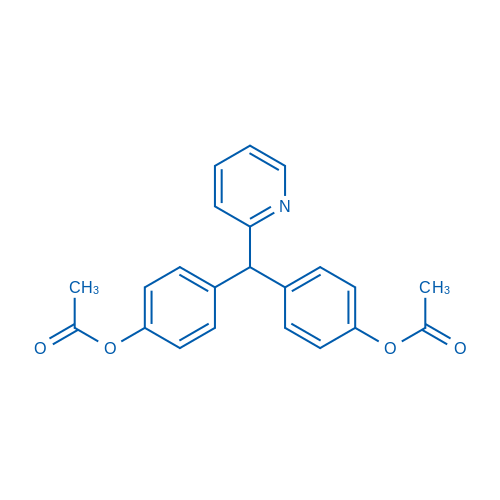
CAS No.: 603-50-9
Bisacodyl/比沙可啶 Catalog No. CSN10462
Synonyms: Durolax;Dulcolax;Fenilaxan
Bisacodyl is a Na+/K+-ATPase inhibitor used as a stimulant laxative drug.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02213172 Irritable Bowel Syndrome-Const... more >>ipation Collapse << Phase 2 Completed - Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << NCT00804167 - - Completed - - NCT01358708 Diarrhea-predominant Irritable... more >> Bowel Syndrome Collapse << Phase 3 Terminated(Administrative reas... more >>ons) Collapse << - France ... more >> Bordeaux, France Colombes, France Marseille, France Nice, France Rouen, France Germany Berlin, Germany Hamburg, Germany Mannheim, Germany Collapse << - 更多
- 参考文献
- [1] Ikarashi N, Baba K, et al. The laxative effect of bisacodyl is attributable to decreased aquaporin-3 expression in the colon induced by increased PGE2 secretion from macrophages. Am J Physiol Gastrointest Liver Physiol. 2011 Nov;301(5):G887-95.
- [2] Saunders DR, Sillery J, Rachmilewitz D, Rubin CE, Tytgat GN. Effect of bisacodyl on the structure and function of rodent and human intestine. Gastroenterology. 1977 May;72(5 Pt 1):849-56.
- [3] Ikarashi N, Baba K, Ushiki T, Kon R, Mimura A, Toda T, Ishii M, Ochiai W, Sugiyama K. The laxative effect of bisacodyl is attributable to decreased aquaporin-3 expression in the colon induced by increased PGE2 secretion from macrophages. Am J Physiol Gastrointest Liver Physiol. 2011 Nov;301(5):G887-95
- [4] Saunders DR, Sillery J, Rachmilewitz D, Rubin CE, Tytgat GN. Effect of bisacodyl on the structure and function of rodent and human intestine. Gastroenterology. 1977 May;72(5 Pt 1):849-56
- [5] Karmeli F, Stalnikowicz R, Rachmilewitz D. Effect of colchicine and bisacodyl on rat intestinal transit and nitric oxide synthase activity. Scand J Gastroenterol. 1997 Aug;32(8):791-6
- [6] Rachmilewitz D, Karmeli F, Okon E. Effects of bisacodyl on cAMP and prostaglandin E2 contents, (Na + K) ATPase, adenyl cyclase, and phosphodiesterase activities of rat intestine. Dig Dis Sci. 1980 Aug;25(8):602-8
- [7] Borrelli F, Mereto E, Capasso F, Orsi P, Sini D, Izzo AA, Massa B, Boggio M, Mascolo N. Effect of bisacodyl and cascara on growth of aberrant crypt foci and malignant tumors in the rat colon. Life Sci. 2001 Sep 7;69(16):1871-7
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 603-50-9 | 储存条件 |
|
|||||
| 分子式 | C22H19NO4 | 运输 | 蓝冰 | |||||
| 分子量 | 361.39 | 别名 | Durolax;Dulcolax;Fenilaxan;Neolax;Ulcolax;比沙可啶 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02213172 | Irritable Bowel Syndrome-Const... more >>ipation Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << |
| NCT00804167 | - | - | Completed | - | - |
| NCT01358708 | Diarrhea-predominant Irritable... more >> Bowel Syndrome Collapse << | Phase 3 | Terminated(Administrative reas... more >>ons) Collapse << | - | France ... more >> Bordeaux, France Colombes, France Marseille, France Nice, France Rouen, France Germany Berlin, Germany Hamburg, Germany Mannheim, Germany Collapse << |
| NCT02047045 | Constipation | Not Applicable | Completed | - | - |
| NCT01211652 | - | - | Completed | - | - |
| NCT01424228 | Constipation | Phase 4 | Completed | - | - |
| NCT01424228 | - | - | Completed | - | - |
| NCT01358708 | - | - | Terminated(Administrative reas... more >>ons) Collapse << | - | - |
| NCT01147926 | - | - | Completed | - | - |
| NCT03610217 | Scleroderma, Systemic ... more >> Sclerosis, Systemic Collapse << | Not Applicable | Not yet recruiting | October 2021 | Canada, Ontario ... more >> Saint Joseph's Health Care London Not yet recruiting London, Ontario, Canada, n6a 4v2 Contact: Janet E Pope, MD, MPH, FRCPSC 15196466332 Janet.Pope@sjhc.london.on.ca Contact: Andreu Fernandez-Codina 151964661023 doccodina@gmail.com Collapse << |
| NCT02547571 | Subjets Requiring a Colonoscop... more >>y Collapse << | Phase 4 | Completed | - | Canada, Alberta ... more >> Forzani & MacPhail Colon Cancer Screening Centre Calgary, Alberta, Canada, T2N 4Z6 University of Alberta Hospital Edmonton, Alberta, Canada, T6G 2X8 Canada, British Columbia St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z 2K5 Canada, Manitoba Health Sciences center and St. Boniface Hospital Winnipeg, Manitoba, Canada, R2H 2A6 Canada, Nova Scotia QEII HSC Halifax, Nova Scotia, Canada, B3H 2Y9 Canada, Ontario Western University London, Ontario, Canada, N6A 5W9 The Ottawa Hospital Ottawa, Ontario, Canada, K1H 1A2 Canada, Quebec CHUM Montreal, Quebec, Canada, H2W 2J3 McGill University Health Centre Montreal, Quebec, Canada, H3G 1A4 Canada Hôpital St-Sacrement, CHU de Québec-Université Laval Québec, Canada, G1S 4L8 Collapse << |
| NCT00083382 | Multiple Myeloma | Phase 2 | Completed | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences/MIRT Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT00083382 | - | - | Completed | - | - |
| NCT01147926 | Male Subjects With Chronic Con... more >>stipation Collapse << | Phase 3 | Completed | - | - |
| NCT02125604 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Research Site Augsburg, Germany Research Site Bamburg, Germany Research Site Bayreuth, Germany Research Site Berlin, Germany Research Site Bochum, Germany Research Site Bonn, Germany Research Site Erbach, Germany Research Site Erlangen, Germany Research Site Freiburg, Germany Research Site Hamburg, Germany Research Site Leipzig, Germany Research Site Marburg, Germany Research Site Minden, Germany Research Site Mittweida, Germany Research Site Munchen, Germany Research Site Munster, Germany Research Site Osnabruck, Germany Research Site Potsdam, Germany Research Site Siegen, Germany Collapse << |
| NCT02125604 | - | - | Completed | - | - |
| NCT00436514 | Colorectal Cancer | Not Applicable | Completed | - | France ... more >> CHU de Nantes Nantes, France, 44093 Collapse << |
| NCT01156129 | Ileus | Phase 1 | Completed | - | United States, Nebraska ... more >> The Nebraska Medical Center Omaha, Nebraska, United States, 68198-3285 Collapse << |
| NCT02028715 | Post-operative Pain | Phase 4 | Completed | - | United States, Ohio ... more >> Aultman Health Foundation Canton, Ohio, United States, 44720 Collapse << |
| NCT01299779 | Hyponatremia | Not Applicable | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT01116206 | Constipation | Phase 3 | Completed | - | Australia ... more >> Adelaide, Australia Box Hill, Australia Kingswood, Australia Kogarah, Australia Newcastle, Australia Parkville, Australia Prahran, Australia Sydney, Australia China Beijing, China Chongqing, China Guangzhou, China Hangzhou, China Hefei, China Jinan, China Nanjing, China Shanghai, China Wuhan, China Xian, China Korea, Republic of Busan, Korea, Republic of Dae-Gu, Korea, Republic of Deajun, Korea, Republic of Gwangju-Si, Korea, Republic of Iksan, Korea, Republic of Seoul, Korea, Republic of Taiwan Taipei, Taiwan Thailand Bamgkok, Thailand Bangkok, Thailand Collapse << |
| NCT00851591 | Lactation | Not Applicable | Terminated(Low enrollment; inv... more >>estigator chose not to continue study) Collapse << | - | United States, Texas ... more >> Texas Tech University Health Sciences Center School of Medicine Amarillo, Texas, United States, 79106 Collapse << |
| NCT00851591 | - | - | Terminated(Low enrollment; inv... more >>estigator chose not to continue study) Collapse << | - | - |
| NCT02887274 | - | - | Recruiting | December 2018 | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Kaohsiung, Taiwan Contact: Cheryl Huang, Bachelor 886 077317123 ext 2639 cheryl60286@gmail.com Collapse << |
| NCT03074448 | Bowel Preparation | Phase 3 | Completed | - | Brazil ... more >> Kaiser Clinica and Day Hospital Sao Jose do Rio Preto, SP, Brazil Collapse << |
| NCT03197922 | Encopresis Au... more >>tism Spectrum Disorder Collapse << | Phase 3 | Recruiting | October 1, 2021 | United States, Georgia ... more >> Marcus Autism Center Recruiting Atlanta, Georgia, United States, 30329 Contact: Nathan Call, PhD 404-785-9428 ncall@emory.edu Principal Investigator: Nathan Call, PhD Collapse << |
| NCT03763266 | Colonoscopy C... more >>olon Adenoma Colorectal Neoplasms Inflammatory Bowel Diseases Colon Disease Collapse << | Not Applicable | Recruiting | December 2019 | Spain ... more >> Hospital Parc Taulí Recruiting Sabadell, Barcelona, Spain, 08208 Contact: Eva Martinez, PhD +34 93 723 10 10 ext 21890 emartinezb@tauli.cat Collapse << |
| NCT02104271 | Radiation Injuries ... more >> Telangiectasis Rectal Bleeding Collapse << | Phase 2 | Completed | - | Brazil ... more >> Barretos Cancer Hospital Barretos, São Paulo, Brazil, 14784400 Collapse << |
| NCT01711437 | Colonoscopy | Phase 4 | Unknown | September 2013 | Italy ... more >> Dipartimento di pediatria e neuropsichiatria Policlinico umberto l "università di roma la sapienza" Recruiting Roma, Italy, 00161 Contact: Dipartimento di pediatria e neuropsichiatria Policlinico umberto l "università di roma la sapienza" 0649979326 giovanni.dinardo@uniroma1.it Collapse << |
| NCT02073552 | Colonoscopy Preparation | Phase 4 | Completed | - | Spain ... more >> Hospital Universitario de Canarias Santa Cruz de Tenerife, Spain, 38320 Collapse << |
| NCT00213317 | - | - | Completed | - | - |
| NCT03060512 | Opioid Induced Constipation | Phase 4 | Completed | - | - |
| NCT01575093 | Healthy Volunteers | Phase 1 Phase 2 | Completed | - | United States, Alabama ... more >> Pinnacle Research Group, LLC Anniston, Alabama, United States, 36207 Collapse << |
| NCT03247595 | Colonoscopy | Not Applicable | Recruiting | September 2018 | United States, Maryland ... more >> Woodholme Gastroenterology Recruiting Baltimore, Maryland, United States, 21208 Principal Investigator: Ethan Dubin, MD Sub-Investigator: Lila Tarmin, MD Sinai Hospital Recruiting Baltimore, Maryland, United States, 21215 Sub-Investigator: Sudhir Dutta, MD Sub-Investigator: Rakesh Vinayek, MD Sub-Investigator: Hugh Mai, MD Collapse << |
| NCT03060512 | - | - | Completed | - | - |
| NCT01476735 | Split-dose Bowel Preparation ... more >> Polyethylene Glycol Solution Risk Factors Inadequate Preparation Collapse << | Not Applicable | Completed | - | Poland ... more >> Maria Sklodowska-Curie Memorial cancer Center, Institute of Oncology Warsaw, Poland, 02-781 Collapse << |
| NCT01695863 | Colorectal Cancer | Phase 4 | Unknown | June 2013 | United States, Illinois ... more >> Rush Unversity Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT01093274 | Bowel Preparation | Not Applicable | Unknown | - | Israel ... more >> Rabin Medical Center - Beilinson Hospital Petach Tikva, Israel, 4910 Collapse << |
| NCT00164164 | Colonoscopy | Phase 3 | Completed | - | United States, Massachusetts ... more >> Braintree, Massachusetts, United States, 02184 United States, Texas San Antonio, Texas, United States, 78284 Collapse << |
| NCT02481219 | - | - | Completed | - | - |
| NCT01838408 | Screening Colonoscopy ... more >> Surveillance Colonoscopy Colonoscopy Collapse << | Not Applicable | Completed | - | United States, Florida ... more >> Borland-Groover Clinic Jacksonville, Florida, United States, 32256 Collapse << |
| NCT02977286 | Constipation | Phase 4 | Recruiting | July 2018 | United States, Massachusetts ... more >> Tufts Medical Center Recruiting Boston, Massachusetts, United States, 02111 Contact: John W Devlin, PharmD 617-636-6124 jdevlin@tuftsmedicalcenter.org Contact: Erik Garpestad, MD 6176362362 egarpestad@tuftsmedicalcenter.org Collapse << |
| NCT01044212 | Functional Disorder of Intesti... more >>ne Collapse << | Not Applicable | Completed | - | United States, New York ... more >> University of Rochester Rochester, New York, United States, 14642 Collapse << |
| NCT01627171 | Endoscopy Bow... more >>el Preparation Solutions Collapse << | Phase 4 | Unknown | December 2013 | Canada, Ontario ... more >> St Joseph Hospital Recruiting Hamilton, Ontario, Canada, L8N4A6 Contact: Lisa Balogh-Melanson, BSc 905-522-1155 ext 33673 lbalogh@stjosham.on.ca Principal Investigator: Khurram J Khan, MD,BSc Sub-Investigator: David Morgan, MD, MSc Sub-Investigator: Robert Spaziani, MD Sub-Investigator: Subhas Ganguli, MD, MSc Sub-Investigator: Keith Tsoi, MD Sub-Investigator: Subash Jalali, MBBS Sub-Investigator: Houssein Fergani, MD Sub-Investigator: Abbas Merali, MD Sub-Investigator: Nauzer Forbes, MD Collapse << |
| NCT00164151 | Colonoscopy | Phase 3 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States, 78284 Collapse << |
| NCT01044212 | - | - | Completed | - | - |
| NCT02481219 | Colorectal Cancer Screening | Not Applicable | Completed | - | United States, Alabama ... more >> Pinnacle Research Group, LLC Anniston, Alabama, United States, 36207 United States, Indiana Indiana University Hospital Indianapolis, Indiana, United States, 46202 Indianapolis Gastroenterology and Hepatology Indianapolis, Indiana, United States, 46237 United States, Ohio Dayton Gastroenterology Dayton, Ohio, United States, 45440 United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 United States, Tennessee Franklin Gastroenterology, PLLC Franklin, Tennessee, United States, 37067 Collapse << |
| NCT01655095 | Quality of Bowel Preparation f... more >>or the Colon Capsule Colon Capsule Completion Rates Colon Capsule Polyp Detection Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> Hotel Dieu Hospital Kingston, Ontario, Canada Collapse << |
| NCT00817934 | - | - | Unknown | January 2010 | United States, Delaware ... more >> Cedar Tree Surgical Center Millsboro, Delaware, United States, 19966 Collapse << |
| NCT00576810 | Constipation | Phase 3 | Completed | - | Germany ... more >> 1062.7.4926 Boehringer Ingelheim Investigational Site Amberg, Germany 1062.7.4918 Boehringer Ingelheim Investigational Site Bad Essen, Germany 1062.7.4946 Boehringer Ingelheim Investigational Site Bergkamen, Germany 1062.7.4904 Boehringer Ingelheim Investigational Site Berlin, Germany 1062.7.4929 Boehringer Ingelheim Investigational Site Berlin, Germany 1062.7.4902 Boehringer Ingelheim Investigational Site Blankenhain, Germany 1062.7.4927 Boehringer Ingelheim Investigational Site Dortmund, Germany 1062.7.4954 Boehringer Ingelheim Investigational Site Dortmund, Germany 1062.7.4949 Boehringer Ingelheim Investigational Site Dresden, Germany 1062.7.4950 Boehringer Ingelheim Investigational Site Einbeck, Germany 1062.7.4909 Boehringer Ingelheim Investigational Site Freising, Germany 1062.7.4923 Boehringer Ingelheim Investigational Site Fürth, Germany 1062.7.4912 Boehringer Ingelheim Investigational Site Hagen, Germany 1062.7.4953 Boehringer Ingelheim Investigational Site Hamburg, Germany 1062.7.4910 Boehringer Ingelheim Investigational Site Hannover, Germany 1062.7.4920 Boehringer Ingelheim Investigational Site Herne, Germany 1062.7.4944 Boehringer Ingelheim Investigational Site Ingelheim, Germany 1062.7.4936 Boehringer Ingelheim Investigational Site Jena, Germany 1062.7.4925 Boehringer Ingelheim Investigational Site Leipzig, Germany 1062.7.4931 Boehringer Ingelheim Investigational Site Leipzig, Germany 1062.7.4907 Boehringer Ingelheim Investigational Site Leverkusen, Germany 1062.7.4942 Boehringer Ingelheim Investigational Site Ludwigsburg, Germany 1062.7.4906 Boehringer Ingelheim Investigational Site Ludwigsfelde, Germany 1062.7.4941 Boehringer Ingelheim Investigational Site Ludwigshafen, Germany 1062.7.4922 Boehringer Ingelheim Investigational Site Lüdenscheid, Germany 1062.7.4930 Boehringer Ingelheim Investigational Site Lüdenscheid, Germany 1062.7.4911 Boehringer Ingelheim Investigational Site Lützen, Germany 1062.7.4916 Boehringer Ingelheim Investigational Site Marburg, Germany 1062.7.4948 Boehringer Ingelheim Investigational Site Marl, Germany 1062.7.4921 Boehringer Ingelheim Investigational Site Oelde, Germany 1062.7.4935 Boehringer Ingelheim Investigational Site Offenbach, Germany 1062.7.4903 Boehringer Ingelheim Investigational Site Potsdam, Germany 1062.7.4937 Boehringer Ingelheim Investigational Site Recklinghausen, Germany 1062.7.4956 Boehringer Ingelheim Investigational Site Reichenbach, Germany 1062.7.4905 Boehringer Ingelheim Investigational Site Saarbrücken, Germany 1062.7.4933 Boehringer Ingelheim Investigational Site Salzkotten, Germany 1062.7.4943 Boehringer Ingelheim Investigational Site Stendal, Germany 1062.7.4955 Boehringer Ingelheim Investigational Site Stuhr, Germany 1062.7.4957 Boehringer Ingelheim Investigational Site Thedinghausen, Germany 1062.7.4913 Boehringer Ingelheim Investigational Site Wallerfing, Germany 1062.7.4924 Boehringer Ingelheim Investigational Site Wardenburg, Germany 1062.7.4914 Boehringer Ingelheim Investigational Site Wiesbaden, Germany 1062.7.4901 Boehringer Ingelheim Investigational Site Winsen, Germany Collapse << |
| NCT00278967 | Colonoscopy | Phase 4 | Completed | - | United States, California ... more >> Anaheim, California, United States, 92801 United States, Florida Jupiter, Florida, United States, 33458 Miami, Florida, United States, 33173 United States, Louisiana Baton Rouge, Louisiana, United States, 70809 United States, Maryland Laurel, Maryland, United States, 20707 United States, New York Great Neck, New York, United States, 11023 United States, North Carolina Raleigh, North Carolina, United States, 27612 United States, Tennessee Germantown, Tennessee, United States, 38138 United States, Washington Bellevue, Washington, United States, 98004 Spokane, Washington, United States, 99207 Collapse << |
| NCT00425594 | Colonoscopy | Phase 4 | Completed | - | - |
| NCT01917773 | Colonic Motility Index ... more >> Constipation Collapse << | Phase 4 | Completed | - | United States, Indiana ... more >> Motility Laboratory, Division of Pediatric Gastroenterology, Hepatology and Nutrition at the James Whitcomb Riley Hospital for Children, Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01885104 | Constipation | Phase 2 | Completed | - | - |
| NCT01885104 | - | - | Completed | - | - |
| NCT00831064 | Bowel Preparation | Not Applicable | Completed | - | Canada, Alberta ... more >> University of Alberta Hospital Edmonton, Alberta, Canada Collapse << |
| NCT01398020 | Efficacy of Bowel Preparation ... more >> Ease of Bowel Preparation and Patient Tolerability Collapse << | Not Applicable | Completed | - | Canada, British Columbia ... more >> St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z1Y6 Collapse << |
| NCT02383758 | - | - | Completed | - | - |
| NCT00779649 | Colonoscopy | Phase 4 | Completed | - | - |
| NCT03226145 | Constipation - Functional ... more >> Irritable Bowel Syndrome Characterized by Constipation Collapse << | Not Applicable | Recruiting | June 2019 | United Kingdom ... more >> University of Nottingham Recruiting Nottingham, Notts, United Kingdom, NG7 2RD Contact: Victoria Wilkinson-Smith 7870554040 mszvw@exmail.nottingham.ac.uk Principal Investigator: Robin Spiller Sub-Investigator: Giles Major Sub-Investigator: Victoria Wilkinson-Smith Sub-Investigator: Maura Corsetti Sub-Investigator: Luca Marciani Sub-Investigator: Caroline Hoad Sub-Investigator: Penny Gowland Queen Mary University of London Recruiting London, United Kingdom Contact: Mark Scott m.scott@qmul.ac.uk Contact: Laura Brockway l.brockway@qmul.ac.uk Principal Investigator: Mark Scott Sub-Investigator: Charles Knowles Sub-Investigator: Henriette Heinrich University College London Recruiting London, United Kingdom Contact: Stuart Taylor Contact: Laura Brockway l.brockway@qmul.ac.uk Principal Investigator: Stuart Taylor Sub-Investigator: David Atkinson Sub-Investigator: Anton Emmanuel Collapse << |
| NCT02383758 | Encopresis Au... more >>tistic Disorder Collapse << | Not Applicable | Completed | - | United States, Georgia ... more >> Marcus Autism Center Atlanta, Georgia, United States, 30329 Collapse << |
| NCT01917773 | - | - | Completed | - | - |
| NCT01533688 | Diabetes Mellitus | Not Applicable | Completed | - | United States, Oklahoma ... more >> Oklahoma VA Medical Center Oklahoma City, Oklahoma, United States, 73104 Collapse << |
| NCT02040285 | Gastrointestinal Complication | Phase 4 | Completed | - | Italy ... more >> Policlinico Umberto I Roma, Italy Collapse << |
| NCT02415569 | Bowel Cleansing Quality | Phase 4 | Completed | - | China, Shandong ... more >> Department of Gastroenterology, Qilu Hospital, Shandong University Jinan, Shandong, China, 250012 Collapse << |
| NCT00857272 | Colonoscopy | Phase 4 | Completed | - | United States, California ... more >> Advanced Clinical Research Institute Anaheim, California, United States, 92801 Advanced Clinical Research Institute Orange, California, United States, 92869 Medical Associates Research Group San Diego, California, United States, 92123 United States, Florida United Medical Research New Smyrna Beach, Florida, United States, 32168 United States, Maryland Maryland Digestive Disease Research Laurel, Maryland, United States, 20707 United States, Tennessee Franklin Gastroenterology Franklin, Tennessee, United States, 37067 Collapse << |
| NCT01853709 | Bowel Preparation for Colonosc... more >>opy Collapse << | Phase 4 | Unknown | December 2013 | Spain ... more >> Hospital de Sabadell Not yet recruiting Sabadell, Barcelona, Spain, 08208 Principal Investigator: Felix Junquera Collapse << |
| NCT01044394 | Bowel Preparation | Phase 2 | Withdrawn(A phase III RCT was ... more >>published showing this approach is effective) Collapse << | - | United States, Massachusetts ... more >> Boston Medical Center Boston, Massachusetts, United States, 02118 Collapse << |
| NCT01773096 | Constipation | Phase 4 | Completed | - | United States, Washington ... more >> Shriners Hospitals for Children- Spokane Spokane, Washington, United States, 99203 Collapse << |
| NCT02976805 | Colonoscopy B... more >>owel Preparation Collapse << | Phase 3 | Recruiting | December 2019 | Canada, Alberta ... more >> University of Alberta Hospital Not yet recruiting Edmonton, Alberta, Canada Contact: Richard Sultanian, MD Canada, Ontario London Health Sciences Centre Recruiting London, Ontario, Canada Contact: Michael Sey, MD 519-667-6582 Canada, Quebec CHUM Not yet recruiting Montreal, Quebec, Canada Contact: Daniel Von Renteln, MD McGill University Health Centre Not yet recruiting Montreal, Quebec, Canada Contact: Alan Barkun, MD Collapse << |
| NCT01533688 | - | - | Completed | - | - |
| NCT01879605 | Constipation ... more >>Ileus Surgery Collapse << | Phase 4 | Completed | - | Norway ... more >> Oslo Universitetssykehus Oslo, Norway, 0424 Collapse << |
| NCT00857272 | - | - | Completed | - | - |
| NCT00410215 | Colonoscopy | Phase 4 | Completed | - | Canada, Ontario ... more >> Hotel Dieu Hospital Kingston, Ontario, Canada, K7L 5G2 Collapse << |
| NCT01073943 | Bowel Preparation | Phase 3 | Completed | - | United States, Florida ... more >> Florida Clinical Research Group Clearwater, Florida, United States Investigational site Hollywood, Florida, United States Nature Coast Clinical Research Inverness, Florida, United States United States, Louisiana Gastroenterology Associates, LLC Baton Rouge, Louisiana, United States Louisiana Research Center Shreveport, Louisiana, United States United States, Maryland Investigative Clinical Research Annapolis, Maryland, United States United States, New Jersey NJ Physicians, LLC Passaic, New Jersey, United States United States, North Carolina Vital re:Search, Inc Greensboro, North Carolina, United States United States, Ohio Gastroenterology Research Consultants of Greater Cincinnati Cincinnati, Ohio, United States United States, Pennsylvania Albert Einstein Medical Center Philadelphia, Pennsylvania, United States United States, Tennessee Gastroenterology Associates Kingsport, Tennessee, United States Collapse << |
| NCT01984008 | Bowel Preparation Before Colon... more >>oscopy Collapse << | Phase 3 | Unknown | July 2015 | Taiwan ... more >> China Medical University Hospital Taichung, Taiwan, 40447 Collapse << |
| NCT01073930 | Bowel Preparation | Phase 3 | Completed | - | United States, Alabama ... more >> Clinical Research Associates Huntsville, Alabama, United States United States, Arkansas Arkansas Gastroenterology Sherwood, Arkansas, United States United States, Connecticut Connecticut Gastroenterology Institute Bristol, Connecticut, United States United States, Indiana Indiana University Hospital Indianapolis, Indiana, United States United States, Louisiana Louisiana Research Center LLC Shreveport, Louisiana, United States United States, North Carolina Piedmont Medical Research Associates Winston-Salem, North Carolina, United States United States, Pennsylvania Hillmont GI, P.C. Flourtown, Pennsylvania, United States United States, Tennessee ClinSearch Chattanooga, Tennessee, United States United States, Virginia Blue Ridge Medical Research Lynchburg, Virginia, United States Collapse << |
| NCT01073943 | - | - | Completed | - | - |
| NCT01865916 | Colonoscopy | Phase 4 | Completed | - | Canada, British Columbia ... more >> GI Clinic, St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z 2K5 Collapse << |
| NCT01510977 | Colonoscopy Failure ... more >> Poor Bowel Preparation Collapse << | Phase 4 | Terminated | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT02761317 | Bowel Preparation | Phase 4 | Unknown | November 2016 | China, Shandong ... more >> Department of Gastroenterology, Qilu Hospital, Shandong University Recruiting Jinan, Shandong, China, 250012 Contact: Yanqing Li, PhD. MD. 86-531-82169236 ext 82169508 qiluliyanqing@gmail.com Principal Investigator: Yanqing li, PhD. MD. Collapse << |
| NCT01533090 | Colonic Polyps ... more >> Cancer Colon Inflammatory Bowel Disease Collapse << | Not Applicable | Completed | - | Italy ... more >> Digestive Endoscopy Unit of Catholic University Rome, Italy, 00168 Collapse << |
| NCT00953017 | Colonoscopy | Phase 4 | Completed | - | United States, Texas ... more >> Brooke Army Medical Center San Antonio, Texas, United States, 78248 Collapse << |
| NCT02119000 | Hospitalized Patients | Phase 4 | Unknown | September 2016 | Canada, Quebec ... more >> McGill University Health Centre Recruiting Montreal, Quebec, Canada, H3G1A4 Contact: Alan Barkun, MD 514-934-8233 Principal Investigator: Alan Barkun, MD Centre hospitalier universitaire de Sherbrooke Recruiting Sherbrooke, Quebec, Canada, J1G 2E8 Contact: Charles Ménard 819-346-1110 ext 23553 Principal Investigator: Charles Ménard, MD Collapse << |
| NCT00953017 | - | - | Completed | - | - |
| NCT03146052 | Polyps Colon ... more >>Preparation Collapse << | Not Applicable | Completed | - | Italy ... more >> ASST Rhodense Garbagnate Milanese, Lombardia, Italy, 20020 Collapse << |
| NCT01528098 | - | - | Unknown | October 2012 | - |
| NCT01685853 | Colonoscopy | Phase 3 | Completed | - | Italy ... more >> European Institute of Oncology Milan, Italy, 20141 Collapse << |
| NCT02609607 | Constipation ... more >>Fecal Incontinence Multiple Sclerosis Collapse << | Phase 2 | Terminated(Poor enrollment; fu... more >>nding ending Dec 31, 2018.) Collapse << | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT01073930 | - | - | Completed | - | - |
| NCT01427296 | Colon Cleansing | Phase 4 | Completed | - | United States, California ... more >> San Carlos, California, United States United States, Connecticut Bristol, Connecticut, United States United States, Georgia Macon, Georgia, United States Suwanee, Georgia, United States United States, Maryland Hagerstown, Maryland, United States United States, Michigan Wyoming, Michigan, United States United States, Missouri Mexico, Missouri, United States United States, New Jersey Vineland, New Jersey, United States United States, New York Great Neck, New York, United States New York, New York, United States United States, North Carolina Fayetteville, North Carolina, United States High Point, North Carolina, United States Raleigh, North Carolina, United States United States, Ohio Cincinnati, Ohio, United States Mentor, Ohio, United States United States, Texas Plano, Texas, United States United States, Virginia Chesapeake, Virginia, United States United States, Washington Bellevue, Washington, United States Collapse << |
| NCT01531140 | Colonoscopy Preparation | Phase 4 | Completed | - | Poland ... more >> Warsaw Medical University Warsaw, Poland, 01-184 Child Health Center Warsaw, Poland, 04-730 Collapse << |
| NCT03410524 | Colonoscopy | Phase 3 | Completed | - | United States, Nevada ... more >> Gastroenterology Consultants Reno - North Office and Endoscopy Center Reno, Nevada, United States, 89502 Gastroenterology Consultants Reno - South Meadows Office and Endoscopy Center Reno, Nevada, United States, 89521 Collapse << |
| NCT01916564 | Bowel Preparation for Morning ... more >>Colonoscopy Collapse << | Not Applicable | Completed | - | Malaysia ... more >> Endoscocpy Unit, University of Malaya Medical Centre Kuala Lumpur, Wilayah Persekutuan Kuala Lumpur, Malaysia, 59100 Collapse << |
| NCT01063049 | Preparation for Colonoscopy | Phase 4 | Completed | - | United States, Illinois ... more >> Gastroenterology Services Downers Grove, Illinois, United States, 60515 Collapse << |
| NCT01063049 | - | - | Completed | - | - |
| NCT02386449 | Bowel Preparation | Phase 4 | Completed | - | Brazil ... more >> Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (there may be other sites in this country) Sao Paulo, Brazil Collapse << |
| NCT01427296 | - | - | Completed | - | - |
| NCT00509327 | Postoperative Ileus | Phase 4 | Completed | - | Switzerland ... more >> Department of Surgery, Triemli Hospital, Zurich, Switzerland Zurich, Switzerland Collapse << |
| NCT01887158 | Intestinal Cancer | Phase 4 | Completed | - | Italy ... more >> Alessandro Mussetto Ravenna, RA, Italy, 48100 Ospedale S.Maria delle Croci Ravenna, RA, Italy, 48100 Collapse << |
| NCT00526097 | Constipation | Phase 3 | Completed | - | United Kingdom ... more >> 122.56.44032 Boehringer Ingelheim Investigational Site Addlestone, United Kingdom 122.56.44018 Boehringer Ingelheim Investigational Site Ash Vale, Aldershot, United Kingdom 122.56.44029 Boehringer Ingelheim Investigational Site Ashford, United Kingdom 122.56.44023 Boehringer Ingelheim Investigational Site Atherstone, United Kingdom 122.56.44011 Boehringer Ingelheim Investigational Site Bedworth, United Kingdom 122.56.44009 Boehringer Ingelheim Investigational Site Bennetthorpe, Doncaster, United Kingdom 122.56.44025 Boehringer Ingelheim Investigational Site Bexhill on Sea, United Kingdom 122.56.44012 Boehringer Ingelheim Investigational Site Blackpool, United Kingdom 122.56.44024 Boehringer Ingelheim Investigational Site Burbage, United Kingdom 122.56.44003 Boehringer Ingelheim Investigational Site Cardiff, United Kingdom 122.56.44017 Boehringer Ingelheim Investigational Site Chesterfield, United Kingdom 122.56.44014 Boehringer Ingelheim Investigational Site Chippenham, United Kingdom 122.56.44006 Boehringer Ingelheim Investigational Site Chorley, United Kingdom 122.56.44033 Boehringer Ingelheim Investigational Site Dundee, United Kingdom 122.56.44002 Boehringer Ingelheim Investigational Site Edgbaston, Birmingham, United Kingdom 122.56.44004 Boehringer Ingelheim Investigational Site Glasgow, United Kingdom 122.56.44020 Boehringer Ingelheim Investigational Site Leamington Spa, United Kingdom 122.56.44007 Boehringer Ingelheim Investigational Site Liverpool, United Kingdom 122.56.44005 Boehringer Ingelheim Investigational Site Manchester, United Kingdom 122.56.44010 Boehringer Ingelheim Investigational Site Midsomer Norton, United Kingdom 122.56.44021 Boehringer Ingelheim Investigational Site Newtonabbey, United Kingdom 122.56.44022 Boehringer Ingelheim Investigational Site Paignton, United Kingdom 122.56.44001 Boehringer Ingelheim Investigational Site Slough, United Kingdom 122.56.44026 Boehringer Ingelheim Investigational Site Sunbury-on-Thames, United Kingdom 122.56.44019 Boehringer Ingelheim Investigational Site Swindon, United Kingdom 122.56.44015 Boehringer Ingelheim Investigational Site Warminster, United Kingdom 122.56.44008 Boehringer Ingelheim Investigational Site Wolverhampton, United Kingdom Collapse << |
| NCT01509131 | Colonoscopy | Phase 4 | Completed | - | Italy ... more >> IRCCS Ospedale Casa Sollievo della Sofferenza San Giovanni Rotondo, Foggia, Italy, 71013 IRCCS Humanitas Rozzano, Milano, Italy, 20089 Spedali Civili Brescia, Italy, 25123 Azienda Ospedaliero Universitaria Careggi Firenze, Italy, 50134 Nuovo Regina Margherita Roma, Italy, 00185 Collapse << |
| NCT02211911 | Healthy | Phase 1 | Completed | - | - |
| NCT01415687 | Bowel Preparation for Colonosc... more >>opy Collapse << | Phase 3 | Completed | - | Canada, Alberta ... more >> Foranzi & MacPhail Colon Cancer Screening Centre, Alberta Health Services Calgary, Alberta, Canada, T2N 4Z6 Foranzi & MacPhail Colon Cancer Screening Centre Calgary, Alberta, Canada, T2N 4Z6 Collapse << |
| NCT01745835 | Healthy Person ... more >> Bowel Preparation Before Colonoscopy Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, City, Korea, Republic of Collapse << |
| NCT02211976 | Constipation | Phase 3 | Completed | - | - |
| NCT00526097 | - | - | Completed | - | - |
| NCT02123017 | Colonoscopy Preparation | Phase 2 | Completed | - | United States, Tennessee ... more >> ClinSearch, LLC Chattanooga, Tennessee, United States, 37421 Collapse << |
| NCT02123017 | - | - | Completed | - | - |
| NCT01452737 | Colonoscopy | Not Applicable | Withdrawn | - | Canada, British Columbia ... more >> St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z 1Y6 Collapse << |
| NCT01011426 | Postoperative Nausea and Vomit... more >>ing Abdominal Pain Collapse << | Phase 2 | Completed | - | United States, New Jersey ... more >> Cooper University Hospital Camden, New Jersey, United States, 08103 Collapse << |
| NCT03248726 | Bowel Preparation | Not Applicable | Recruiting | July 13, 2019 | Taiwan ... more >> Wanfang Hospital Recruiting Taipei, Taiwan, 116 Contact: Wei-Cheng Huang, Bachelor +886229307930 egghouses@gmail.com Collapse << |
| NCT03279341 | Chronic Constipation | Phase 4 | Completed | - | Belgium ... more >> TARGID Leuven, Belgium, 3000 Collapse << |
| NCT02980562 | Bowel Disease ... more >> Colon Adenoma Collapse << | Phase 3 | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
