| A10 cells | - | Growth inhibition assay | 48 h | Growth inhibition of rat A10 cells after 48 hrs by MTT assay, IC50=0.001 μM | 22044164 |
| A-549 | - | Growth inhibition assay | - | Cytotoxic concentration required to inhibit 50% cell growth in A-549 lung carcinoma cell lines, ED50=1.20E-06 μM | 1875350 |
| ACHN cell | - | Proliferation assay | - | Antiproliferative activity against drug resistant ACHN cell line expressing MDR1 by Alamar Blue assay, IC50=0.0016 μM | 17286393 |
| BNL 1ME A.7R.1 cells | - | Cytotoxicity assay | - | Cytotoxicity against mouse BNL 1ME A.7R.1 cells, IC50=0.0009 μM | 25785605 |
| Calu6 | - | Proliferation assay | 48 h | Antiproliferative activity against human Calu6 after 48 hrs by spectrophotometry, IC50=0.00094 μM | 21774499 |
| CEM cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human CEM cells after 72 hrs by MTT assay, IC50=0.001 μM | 22676247 |
| DU145 cells | - | Cytotoxicity assay | - | Cytotoxicity against human DU145 cells by SRB assay, GI50=0.00054 μM | 24016002 |
| DU145 cells | - | Growth inhibition assay | - | Growth inhibition of human DU145 cells by sulforhodamine B assay, GI50=0.00054 μM | 23772309 |
| DU145 cells | - | Cytotoxicity assay | 48 h | Cytotoxicity against human DU145 cells after 48 hrs by SRB assay, GI50=0.0008 μM | 18303849 |
| DU145 cells | - | Cytotoxicity assay | - | Cytotoxicity against human DU145 cells by SRB assay, GI50=0.001 μM | 20496923 |
| DU145 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human DU145 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| H460 | - | Cytotoxicity assay | - | Cytotoxicity against human H460 cells by sulforhodamine B test, IC50=0.0012 μM | 18783207 |
| H460 | - | Proliferation assay | - | Antiproliferative activity against human H460 cells, IC50=0.00124 μM | 16722633 |
| HaCaT cells | - | Proliferation assay | - | Antiproliferative activity against human HaCaT cells assessed as cell viability by MTT assay, IC50=0.001 μM | 23063401 |
| HCT 116 | - | Growth inhibition assay | - | Concentration which produces 50% inhibition of growth of human colon tumor HCT 116, IC50=0.0009 μM | 11714613 |
| HCT116 cell | - | Cytotoxicity assay | - | Cytotoxicity against HCT116 cell line, IC50=0.00035 μM | 17249649 |
| HCT116 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HCT116 cells assessed as reduction of [3H]thymidine incorporation after 72 hrs by scintillation counting, IC50=0.00074 μM | 21955454 |
| HCT116 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HCT116 cells after 72 hrs by MTS assay, IC50=0.001 μM | 21112130 |
| HCT116 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HCT116 cells after 72 hrs by MTT assay, IC50=0.001 μM | 22676247 |
| HCT15 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HCT15 cells after 72 hrs by MTS assay, IC50=0.001 μM | 21112130 |
| HeLa cell | - | Cytotoxicity assay | - | Cytotoxicity against HeLa cell line, IC50=0.0003 μM | 17249649 |
| HeLa cells | - | Cytotoxicity assay | - | Cytotoxicity against human HeLa cells by MTT assay, IC50=0.00051 μM | 19879758 |
| HeLa cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HeLa cells after 72 hrs by resazurin based fluorescence assay, IC50=0.0009 μM | 24669888 |
| HepG2 cells | - | Cytotoxicity assay | - | Cytotoxicity against human HepG2 cells, IC50=0.00014 μM | 17127061 |
| HL60 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50=0.0001 μM | 20731355 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50=0.001 μM | 21663319 |
| HL60 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50=0.001 μM | 22136312 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50=0.001 μM | 22578111 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells after 72 hrs by MTT assay, IC50=0.001 μM | 23117171 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells after 72 hrs by (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test, IC50=0.001 μM | 25785605 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells after 72 hrs by MTT assay, IC50=0.001 μM | 20420439 |
| HL60 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HL60 cells after 72 hrs by MTT assay, IC50=0.001 μM | 23445496 |
| Hs578T cells | - | Growth inhibition assay | 48 h | Growth inhibition of human Hs578T cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| HT-29 | - | Growth inhibition assay | - | Cytotoxic concentration required to inhibit 50% cell growth in HT-29 colon adenocarcinoma cell lines, ED50=1.20E-06 μM | 1875350 |
| HT-29 | - | Growth inhibition assay | - | Growth inhibition of human HT-29 cells by MTT assay, IC50=0.001 μM | 25689111 |
| Human SH-SY5Y neuroblastoma cells | - | Function assay | - | Inhibitory concentration against Human SH-SY5Y neuroblastoma cells, IC50=0.0015 μM | 15963722 |
| HuTu 80 cells | - | Proliferation assay | 72 h | Antiproliferative activity in human HuTu 80 cells assessed as cell viability after 72 hrs by MTT assay, IC50=0.00137 μM | 22329561 |
| HUVEC | - | Proliferation assay | - | Antiproliferative activity against human HUVEC, IC50=0.0015 μM | 16722633 |
| HUVEC cells | - | Growth inhibition assay | 48 h | Growth inhibition of HUVEC cells after 48 hrs by MTT assay, IC50=0.001 μM | 22044164 |
| K562 cells | - | Cytotoxicity assay | - | Cytotoxicity against human K562 cells, IC50=0.0014 μM | 19837594 |
| KB cell | - | Proliferation assay | - | Antiproliferative activity against human KB cell line by methylene blue dye assay, IC50=0.0015 μM | 17034147 |
| KBV1 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human vinblastine-resistant KBV1 cells after 72 hrs by MTT assay, IC50=0.0004 μM | 20731355 |
| KB-VIN10 cell | - | Proliferation assay | - | Antiproliferative activity against human KB-VIN10 cell line by methylene blue dye assay, IC50=0.0015 μM | 17034147 |
| KB-VIN10 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human KB-VIN10 cells over-expressing P-gp 170/MDR1 after 72 hrs by methylene blue assay, IC50=0.0007 μM | 21641700 |
| M14 | - | Cytotoxicity assay | - | In vitro cytotoxic activity was tested against human melanoma cancer (M14) cell line, GI50=0.0001 μM | 12729640 |
| MCF-7 | - | Growth inhibition assay | - | Cytotoxic concentration required to inhibit 50% cell growth in MCF-7 breast carcinoma cell lines, ED50=3.80E-06 μM | 1875350 |
| MCF7 cells | - | Proliferation assay | 72 h | Antiproliferative activity in human MCF7 cells assessed as cell viability after 72 hrs by MTT assay, IC50=0.00111 μM | 22329561 |
| MDA-MB-231 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human MDA-MB-231 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| MDA-MB-435 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human MDA-MB-435 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| MDA-MB-435 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human MDA-MB-435 cells after 72 hrs by MTT assay, IC50=0.001 μM | 22676247 |
| MDA-MB-468 cells | - | Cytotoxicity assay | 4 days | Cytotoxicity against human MDA-MB-468 cells assessed as cell viability after 4 days by MTS assay, IC50=0.0011 μM | 24210503 |
| melanoma B16 cell | - | Growth inhibition assay | - | Concentration which produces 50% inhibition of growth of murine melanoma B16 cell line, IC50=0.001 μM | 11714613 |
| MLM melanoma cell | - | Growth inhibition assay | - | Cytotoxic concentration required to inhibit 50% cell growth in MLM melanoma cell lines, ED50=1.40E-06 μM | 1875350 |
| Molt4/C8 cells | - | Proliferation assay | - | Antiproliferative activity against human Molt4/C8 cells, IC50=0.0016 μM | 17034150 |
| Molt4/C8 cells | - | Proliferation assay | 3 days | Antiproliferative effect against human Molt4/C8 cells after 3 days, IC50=0.0016 μM | 17419607 |
| NCI/ADR (MDR) cells | - | Function assay | - | Cell cycle arrest in NCI/ADR (MDR) cells by accumulation at G2/M phase, IC50=0.0015 μM | 16377187 |
| NCI/ADR-RES cells | - | Growth inhibition assay | 48 h | Growth inhibition of human NCI/ADR-RES cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| NCI/ADR-RES cells | - | Cytotoxicity assay | - | Cytotoxicity against human NCI/ADR-RES cells assessed as growth inhibition by trypan blue exclusion assay, IC50=0.0013 μM | 23214452 |
| NCI/ADR-RES cells | - | Cytotoxicity assay | 96 h | Cytotoxicity against human NCI/ADR-RES cells assessed as growth inhibition after 96 hrs by Giemsa staining-based light microscopy, IC50=0.0013 μM | 25025991 |
| NCIH460 cells | - | Cytotoxicity assay | 48 h | Cytotoxicity against human NCIH460 cells after 48 hrs by SRB assay, GI50=0.0006 μM | 18303849 |
| NCI-H460 cells | - | Cytotoxicity assay | 72 h | Cytotoxic activity against human NCI-H460 cells after 72 hrs by sulforhodamine B test, IC50=0.0013 μM | 18396857 |
| NCI-H522 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human NCI-H522 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| OVCAR3 | - | Growth inhibition assay | 48 h | Growth inhibition of human OVCAR3 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| OVCAR8 cells | - | Growth inhibition assay | 96 h | Growth inhibition of human OVCAR8 cells overexpressing P-glycoprotein after 96 hrs, IC50=0.0013 μM | 22044164 |
| OVCAR8 cells | - | Cytotoxicity assay | 96 h | Cytotoxicity against human OVCAR8 cells assessed as growth inhibition after 96 hrs by Giemsa staining-based light microscopy, IC50=0.0013 μM | 25025991 |
| OVCAR8 cells | - | Cytotoxicity assay | - | Cytotoxicity against human OVCAR8 cells assessed as growth inhibition by trypan blue exclusion assay, IC50=0.0013 μM | 23214452 |
| PC3 cells | - | Growth inhibition assay | 48 h | Growth inhibition of human PC3 cells after 48 hrs by SRB assay, GI50=0.001 μM | 22982122 |
| RS4:11 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human RS4:11 cells after 72 hrs by (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test, IC50=0.0008 μM | 25785605 |
| SKMEL-5 | - | Growth inhibition assay | - | Cytotoxic concentration required to inhibit 50% cell growth in SKMEL-5 melanoma cell lines, ED50=3.00E-08 μM | 1875350 |
| SK-N-BE | - | Proliferation assay | 72 h | Antiproliferative activity in human SK-N-BE cells assessed as cell viability after 72 hrs by MTT assay, IC50=0.00058 μM | 22329561 |
| SKOV3 | - | Cytotoxicity assay | - | Cytotoxicity against human SKOV3 cells by SRB assay, GI50=0.00042 μM | 24016002 |
| SK-OV3 | - | Cytotoxicity assay | - | In vitro cytotoxic activity tested against human ovarian cancer (SK-OV3) cell line, GI50=0.0001 μM | 12729640 |
| SK-OV-3 | - | Growth inhibition assay | 48 h | Growth inhibition of human SK-OV-3 after 48 hrs by SRB assay, GI50=0.00013 μM | 18722127 |
| SKOV3 cells | - | Growth inhibition assay | - | Growth inhibition of human SKOV3 cells by sulforhodamine B assay, GI50=0.00042 μM | 23772309 |
| SKOV3 cells | - | Growth inhibition assay | - | Growth inhibition of human SKOV3 cells by MTT assay, IC50=0.001 μM | 25689111 |
| SW620 cells | - | Proliferation assay | 72 h | Antiproliferative activity in human SW620 cells assessed as cell viability after 72 hrs by MTT assay, IC50=0.00152 μM | 22329561 |
| ZR-75-1 | - | Cytotoxicity assay | - | Cytotoxicity against ZR-75-1 cell line, IC50=0.00024 μM | 17249649 |
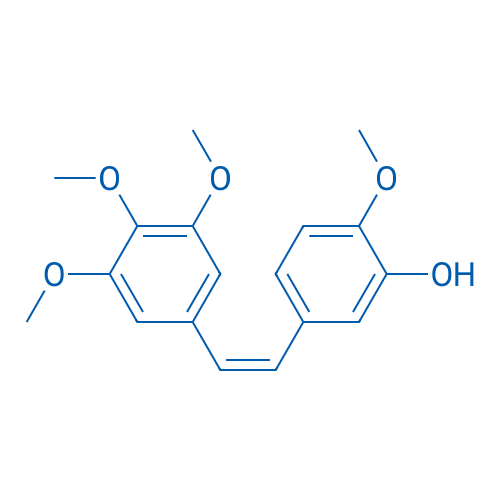

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
