CAS No.: 59787-61-0
Cyclosporin C Catalog No. CSN16428
Synonyms: Cs-C;环孢素C
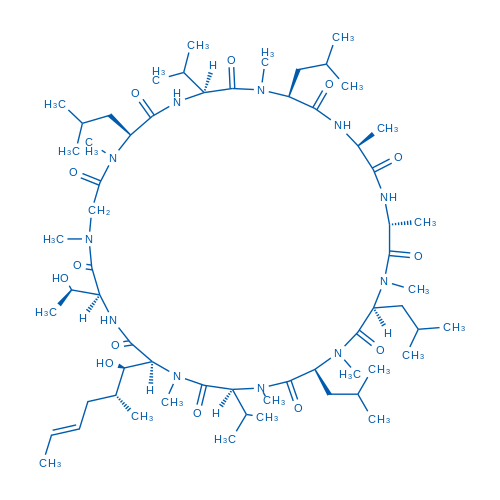
Cyclosporin C, a natural product isolaed and purified from Acremonium luzulae, is an immunosuppressive cyclic polypeptide drug commonly used post-transplant to prevent graft rejection from the cytotoxic effects of T-cells.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00006184 - - Completed - - NCT00001302 Breast Cancer ... more >> Kidney Neoplasm Lymphoma Neoplasm Metastasis Ovarian Cancer Collapse << Phase 1 Completed - United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << NCT00537407 - - Completed - - - 更多
- 参考文献
- [1] Moussaif M, Jacques P, et al. Cyclosporin C is the main antifungal compound produced by Acremonium luzulae. Appl Environ Microbiol. 1997 May;63(5):1739-43.
- [2] Bram RJ, Hung DT, et al. Identification of the immunophilins capable of mediating inhibition of signal transduction by cyclosporin A and FK506: roles of calcineurin binding and cellular location. Mol Cell Biol. 1993 Aug;13(8):4760-9.
- [3] Moussaïf M, Jacques P, Schaarwächter P, Budzikiewicz H, Thonart P. Cyclosporin C is the main antifungal compound produced by Acremonium luzulae. Appl Environ Microbiol. 1997 May;63(5):1739-43
- [4] O'Callaghan CA, Wells AU, Lalvani A, Dhillon PD, Hansell DM, Mitchell DN. Effective use of cyclosporin in sarcoidosis: a treatment strategy based on computed tomography scanning. Eur Respir J. 1994 Dec;7(12):2255-6
- [5] Nicholls S, Domizio P, Williams CB, Dawnay A, Braegger CP, MacDonald TT, Walker-Smith JA. Cyclosporin as initial treatment for Crohn's disease. Arch Dis Child. 1994 Sep;71(3):243-7
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 59787-61-0 | 储存条件 |
|
|
| 分子式 | C62H111N11O13 | 运输 | 蓝冰 | |
| 分子量 | 1218.61 | 别名 | Cs-C;环孢素C |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00006184 | - | - | Completed | - | - |
| NCT00001302 | Breast Cancer ... more >> Kidney Neoplasm Lymphoma Neoplasm Metastasis Ovarian Cancer Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00537407 | - | - | Completed | - | - |
| NCT02461121 | Acute Myeloid Leukemia | Phase 3 | Completed | - | China, Beijing ... more >> Affiliated Hospital of Academy of Military Medical Sciences , Beijing, Beijing, China, 100071 Collapse << |
| NCT00537407 | Chronic Hepatitis C | Phase 2 | Completed | - | United States, California ... more >> Scripps Clinic Liver Disease Research Center la Jolla, California, United States, 92037 United States, Florida University of Florida Gainesville, Florida, United States, 32610 University of Miami Center for Liver Diseases Miami, Florida, United States, 33136 United States, Maryland The Johns Hopkins University School of Medicine Baltimore, Maryland, United States, 21205 United States, Texas Methodist Transplant Physicians Dallas, Texas, United States, 75203 United States, Virginia Metropolitan Research Fairfax, Virginia, United States, 22031 United States, Washington Virginia Mason Medical Center Seattle, Washington, United States, 98101 Collapse << |
| NCT01596062 | - | - | Completed | - | - |
| NCT01596062 | Renal Transplantation | Phase 2 | Completed | - | France ... more >> Novartis Investigative Site Bordeaux Cedex, France, 33076 Novartis Investigative Site Paris cedex 15, France, 75015 Novartis Investigative Site Tours Cedex, France, 37044 Collapse << |
| NCT00854802 | Chronic Hepatitis C | Not Applicable | Completed | - | - |
| NCT00944749 | - | - | Completed | - | - |
| NCT00006184 | Multiple Myeloma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01117142 | - | - | Recruiting | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT02496494 | CYCLOSPORINE/TACROLIMUS | Phase 4 | Unknown | December 2015 | Korea, Republic of ... more >> Kyungpook National University Hospital Recruiting Daegu, Korea, Republic of Contact: Chan-Duck Kim drcdkim@knu.ac.kr Collapse << |
| NCT02143830 | Fanconi Anemia ... more >> Severe Marrow Failure Myelodysplastic Syndrome (MDS) Acute Myelogenous Leukemia (AML) Collapse << | Phase 2 | Recruiting | May 2020 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10174 Contact: Jessica Chaisson 646-888-5718 chaissoj@mskcc.org Principal Investigator: Farid Boulad, MD United States, Ohio Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Principal Investigator: Parinda Mehta, MD United States, Washington Fred Hutchinson Cancer Research Center Recruiting Seattle, Washington, United States, 98109 Contact: Sheri Ballard 206-667-4222 sballard@fredhutch.org Principal Investigator: K. Scott Baker, MD Collapse << |
| NCT02016625 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1241.61.1 Boehringer Ingelheim Investigational Site Mannheim, Germany Collapse << |
| NCT02016625 | - | - | Completed | - | - |
| NCT00944749 | Anemia, Aplastic ... more >> Anemia, Hypoplastic Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01002859 | Coronary Artery Bypass Surgery... more >> Cardiopulmonary Bypass Collapse << | Phase 2 | Terminated(difficulties to inc... more >>lude patients) Collapse << | - | France ... more >> Department of cardiac surgery - University Hospital of Grenoble Grenoble,, France, 38043 Collapse << |
| NCT00157014 | Heart Diseases ... more >> Heart Transplantation Collapse << | Phase 3 | Completed | - | United States, California ... more >> Los Angeles, California, United States, 90095 Canada, Alberta Calgary, Alberta, Canada, T2N 2T9 Edmonton, Alberta, Canada, T6G 2B7 Edmonton, Alberta, Canada, T6G 2E1 Canada, British Columbia Vancouver, British Columbia, Canada, V6Z 1Y6 Canada, Nova Scotia Halifax, Nova Scotia, Canada, B3H 3A7 Canada, Ontario London, Ontario, Canada, N6A 5A5 Ottawa, Ontario, Canada, K1Y 4W7 Toronto, Ontario, Canada, M5G 1X8 Toronto, Ontario, Canada, M5G 2N2 Canada, Quebec Montreal, Quebec, Canada, H1T 1C8 Montreal, Quebec, Canada, H3A 1A1 Sainte-Foy, Quebec, Canada, G1V 4G5 Collapse << |
| NCT00157014 | - | - | Completed | - | - |
| NCT00176865 | - | - | Completed | - | - |
| NCT00176865 | Hemophagocytic Lymphohistiocyt... more >>osis X-Linked Lymphoproliferative Disorders Chediak-Higashi Syndrome Griscelli Syndrome Immunologic Deficiency Syndromes Langerhans-Cell Histiocytosis Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Masonic Cancer Center University of Minnesota Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT00706680 | Diabetes Graf... more >>t Rejection Collapse << | Phase 4 | Unknown | June 2009 | Canada, British Columbia ... more >> St Paul's Hospital Not yet recruiting Vancouver, British Columbia, Canada, V6Z 2E8 Contact: John Gill 604-681-7191 Principal Investigator: John Gill Sub-Investigator: David Landsberg Canada, Ontario St Michael's Hospital Not yet recruiting Toronto, Ontario, Canada, M5C 2T2 Contact: Ramesh Prasad (416) 867-3722 Ramesh.Prasad@utoronto.ca Principal Investigator: Ramesh Prasad Sub-Investigator: Jeffrey Zaltzman University health Network Recruiting Toronto, Ontario, Canada, M5G 2N2 Contact: Bricio Rodriguez 416-340-4800 ext 5921 bricio.rodriguez@uhn.on.ca Sub-Investigator: Carl Cardella Principal Investigator: Edward Cole Collapse << |
| NCT01028092 | Renal Transplant | Phase 3 | Completed | - | France ... more >> CHU Amiens Amiens, France CHU Pellegrin - Bordeaux Bordeaux, France CHRU de BREST Brest, France, 29609 CHU Cote de Nacre Caen, France CHU Clermont Ferrand Clermont Ferrand, France CHU Dijon Dijon, France CHU Lille Lille, France CHU Limoges Limoges, France CHU Montpellier Montpellier, France CHU Nice Nice, France AP-HP Hopital Necker Paris, France CHU Poitiers Poitiers, France CHU Reims Reims, France CHU Rennes Rennes, France CHU Rouen Rouen, France Hopitaux Universitaires de Strasbourg Strasbourg, France CHU Rangueil - Toulouse Toulouse, France CHU Tours Tours, France Collapse << |
| NCT02397213 | Acute Kidney Injury | Phase 2 | Completed | - | Sweden ... more >> Skane University Hospital Lund, Sweden, 22185 Collapse << |
| NCT03768609 | Healthy | Phase 1 | Not yet recruiting | February 25, 2019 | Belgium ... more >> Clinical Pharmacology Unit Not yet recruiting Merksem, Belgium, 2170 Collapse << |
| NCT01843348 | - | - | Completed | - | - |
| NCT01843348 | Kidney Transplantation ... more >> Renal Transplantation Collapse << | Phase 3 | Completed | - | France ... more >> Novartis Investigative Site Bordeaux Cedex, France, 33076 Novartis Investigative Site Brest, France, 29200 Novartis Investigative Site Creteil, France, 94010 Novartis Investigative Site Dijon, France, 21079 Novartis Investigative Site Lille Cedex, France, 59037 Novartis Investigative Site Lyon, France, 69437 Novartis Investigative Site Nantes, France, 44035 Novartis Investigative Site Paris, France, 75970 Novartis Investigative Site Poitiers, France, 86000 Novartis Investigative Site St Priest en Jarez Cedex, France, 42277 Novartis Investigative Site Strasbourg, France, 67091 Novartis Investigative Site Toulouse Cedex 4, France, 31054 Germany Novartis Investigative Site Aachen, Germany, 52074 Novartis Investigative Site Berlin, Germany, 13353 Novartis Investigative Site Bochum, Germany, 44892 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Erlangen, Germany, 91052 Novartis Investigative Site Essen, Germany, 45147 Novartis Investigative Site Frankfurt, Germany, 60590 Novartis Investigative Site Freiburg, Germany, 79106 Novartis Investigative Site Hamburg, Germany, 20246 Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Kiel, Germany, 24105 Novartis Investigative Site Mainz, Germany, 55131 Novartis Investigative Site Muenster, Germany, 48149 Novartis Investigative Site Tübingen, Germany, 72076 Collapse << |
| NCT02013791 | Dry Eye Syndromes | Phase 2 | Terminated | - | United States, California ... more >> Sall Research Medical Center Artesia, California, United States, 90701 Lugene Eye Institute Glendale, California, United States, 91204 Lakeside Vision Center Irvine, California, United States, 92604 Steve Yoelin MD Medical Asscociates Newport Beach, California, United States, 92663 Wolstan & Goldberg Eye Associates Torrance, California, United States, 90505 United States, Colorado Vision Institute Colorado Springs, Colorado, United States, 80907 United States, Maryland Johns Hopkins University - Wilmer Eye Institute Baltimore, Maryland, United States, 21287 United States, Wisconsin Eye Centers of Racine and Kenosha Kenosha, Wisconsin, United States, 53142 Collapse << |
| NCT00862979 | Heart Transplantation | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Oeynhausen, Germany, 32545 Novartis Investigative Site Berlin, Germany, 13353 Novartis Investigative Site Hamburg, Germany, 20246 Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Leipzig, Germany, 04289 Novartis Investigative Site Muenster, Germany, 48149 Novartis Investigative Site München, Germany, 81377 Novartis Investigative Site Regensburg, Germany, 93053 Collapse << |
| NCT02013791 | - | - | Terminated | - | - |
| NCT00789308 | Diabetes Mellitus, Type I | Phase 2 | Completed | - | Norway ... more >> University Hospital Rikshospitalet Oslo, Norway Sweden Karolinska University Hospital Stockholm, Sweden Uppsala University Hospital Uppsala, Sweden Collapse << |
| NCT02058875 | Cardiovascular Disease ... more >> Cardiovascular Outcomes Kidney Transplant Recipients Kidney Transplantation Collapse << | Phase 4 | Withdrawn(The study funder ret... more >>racted their grant funding offer before contract signed.) Collapse << | - | Canada, Saskatchewan ... more >> Kidney Health Center Regina, Saskatchewan, Canada, S4R 3C2 St. Paul's Hospital Saskatoon, Saskatchewan, Canada, S7M 0Z9 Collapse << |
| NCT00862979 | - | - | Completed | - | - |
| NCT00514514 | Kidney Transplantation | Phase 3 | Completed | - | Germany ... more >> Novartis Investigative Site Aachen, Germany, 52074 Novartis Investigative Site Berlin, Germany, 10117 Novartis Investigative Site Berlin, Germany, 13353 Novartis Investigative Site Erlangen, Germany, 91054 Novartis Investigative Site Essen, Germany, 45122 Novartis Investigative Site Frankfurt am Main, Germany, 60596 Novartis Investigative Site Freiburg, Germany, 79106 Novartis Investigative Site Hamburg, Germany, 20246 Novartis Investigative Site Hannover Muenden, Germany, 34346 Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Kaiserslautern, Germany, 67655 Novartis Investigative Site Koeln, Germany, 51109 Novartis Investigative Site Lubeck, Germany, 23538 Novartis Investigative Site Munchen, Germany, 81377 Novartis Investigative Site München, Germany, 81675 Novartis Investigative Site Regensburg, Germany, 93053 Switzerland Novartis Investigative Site Bern, Switzerland, 3010 Collapse << |
| NCT02655705 | Psoriasis | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, KS009 Collapse << |
| NCT00226512 | Acute Myeloid Leukemia ... more >> Myelodysplastic Syndrome Collapse << | Phase 3 | Withdrawn(the PI is no longer ... more >>work at Hadassah) Collapse << | - | Israel ... more >> Hadassah Medical Organization Jerusalem, Israel, 91120 Collapse << |
| NCT00514514 | - | - | Completed | - | - |
| NCT01114529 | - | - | Completed | - | - |
| NCT01598688 | Hematopoietic Stem Cell Transp... more >>lantation Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo Ribeirão Preto, São Paulo, Brazil, 14048-900 Collapse << |
| NCT00051311 | Hematologic Neoplasms | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01369082 | - | - | Completed | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94143 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 University of Illinois Chicago, Illinois, United States, 60612 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02493 United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States, 55455 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01114529 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT00977977 | Nephrotic Syndrome ... more >> Proteinuria Autoimmune Disease Glomerular Disease Membranous Glomerulonephritis Collapse << | Phase 2 | Recruiting | January 1, 2020 | United States, Maryland ... more >> National Naval Medical Center Recruiting Bethesda, Maryland, United States, 20889 National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT00182559 | End Stage Renal Disease | Phase 4 | Completed | - | Austria ... more >> Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT00002833 | Graft Versus Host Disease ... more >> Leukemia Myelodysplastic Syndromes Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas - MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00681213 | Adult Primary Kidney Transplan... more >>tation Collapse << | Phase 4 | Completed | - | United States, Florida ... more >> University of Miami Division of Transplantation Miami, Florida, United States, 33136 Collapse << |
| NCT00135694 | Hepatitis C H... more >>epatitis C, Chronic Nonimmune Nonviral Causes of Liver Failure Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado University of Colorado Denver, Colorado, United States, 80262 United States, Illinois Northwestern University Chicago, Illinois, United States, 60208 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas Baylor University Dallas, Texas, United States, 76798 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01180036 | Idiopathic Membranous Nephropa... more >>thy Collapse << | Phase 2 Phase 3 | Active, not recruiting | October 2019 | - |
| NCT00135694 | - | - | Completed | - | - |
| NCT00154310 | Renal Transplantation | Phase 4 | Completed | - | Germany ... more >> Novartis Investigational Sites Nurnberg, Germany Switzerland Novartis Pharma AG Basel, Switzerland Novartis Investigational Sites Bern, Switzerland Collapse << |
| NCT00154310 | - | - | Completed | - | - |
| NCT02199964 | Dry Eye | Not Applicable | Completed | - | United States, Texas ... more >> Alkek Eye Center, Dept of Ophthalmology, Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT01678937 | - | - | Completed | - | United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << |
| NCT03083782 | Venous Thromboembolism ... more >> Pharmacokinetics Healthy Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00520130 | - | - | Active, not recruiting | - | - |
| NCT00658320 | Kidney Transplantation | Phase 3 | Completed | - | Japan ... more >> Novartis Pharma K.K., Japan Tokyo, Japan, 106-8618 Collapse << |
| NCT00323960 | Juvenile Dermatomyositis | Phase 3 | Unknown | May 2011 | Italy ... more >> Istituto Giannina Gaslini Recruiting Genoa, Italy, 16147 Collapse << |
| NCT00658320 | - | - | Completed | - | - |
| NCT00634920 | Renal Function | Phase 4 | Completed | - | Denmark ... more >> Novartis Investigative Site Aarhus N, Denmark, 8200 Novartis Investigative Site Copenhagen, Denmark, DK-2100 Novartis Investigative Site Herlev, Denmark, 2730 Novartis Investigative Site Odense C, Denmark, DK-5000 Norway Novartis Investigative Site Oslo, Norway, 0424 Sweden Novartis Investigative Site Goteborg, Sweden, 413 45 Novartis Investigative Site Malmo, Sweden, 205 02 Novartis Investigative Site Uppsala, Sweden, 751 85 Collapse << |
| NCT00520130 | Myelodysplastic Syndrome ... more >> Hodgkin's Lymphoma Non-Hodgkin's Disease Acute Leukemia Multiple Myeloma Collapse << | Phase 1 Phase 2 | Active, not recruiting | December 2018 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01598987 | Renal Function ... more >> Liver Transplant Collapse << | Phase 3 | Completed | - | - |
| NCT01598987 | - | - | Completed | - | - |
| NCT00903188 | Renal Transplantation | Phase 4 | Unknown | April 2015 | Belgium ... more >> Erasme University Hospital Recruiting Brussels, Belgium, 1070 Contact: Daniel Abramowicz, MD/PhD +32/2/555 35 32 dabram@ulb.ac.be Principal Investigator: Daniel Abramowicz, MD/PhD University Hospital Brussels Recruiting Brussels, Belgium, 1090 Contact: Jacques Sennesael, MD/PhD +32/2/477 60 55 Jacques.Sennesael@uzbrussel.be Principal Investigator: Jacques Sennesael, MD University Hospital Antwerp Recruiting Edegem, Belgium, 2650 Contact: Jean-Louis Bosmans, MD/PhD +32/3/821 37 92 jeanlouis.bosmans@ua.ac.be Contact: Angelika Jurgens, Study Coord. +32/3/821 34 21 Angelika.Jurgens@uza.be Principal Investigator: Jean-Louis Bosmans, MD/PhD University Hospital, Ghent Recruiting Gent, Belgium, 9000 Contact: Patrick Peeters, MD/PhD +32/9/332 45 13 patrick.peeters@ugent.be Principal Investigator: Patrick Peeters, MD University Hospital of Liege Recruiting Liège, Belgium, 4000 Contact: Catherine Bonvoisin, MD/PhD +32/4/366 82 58 catherine.bonvoisin@chu.ulg.ac.be Principal Investigator: Catherine Bonvoisin, MD Collapse << |
| NCT02175615 | - | - | Completed | - | Canada, Ontario ... more >> Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT00413920 | - | - | Completed | - | - |
| NCT02547714 | Plaque Psoriasis | Phase 4 | Completed | - | Japan ... more >> Novartis Investigative Site Nagoya city, Aichi, Japan, 467-8602 Novartis Investigative Site Fukuoka-city, Fukuoka, Japan, 814-0180 Novartis Investigative Site Isehara-city, Kanagawa, Japan, 259-1193 Novartis Investigative Site Osaka-city, Osaka, Japan, 550-0012 Novartis Investigative Site Shimotsuke-city, Tochigi, Japan, 329-0498 Novartis Investigative Site Chiyoda-ku, Tokyo, Japan, 102-8798 Novartis Investigative Site Itabashi-ku, Tokyo, Japan, 173-8606 Novartis Investigative Site Itabashi-ku, Tokyo, Japan, 173-8610 Novartis Investigative Site Minato-ku, Tokyo, Japan, 105-8471 Novartis Investigative Site Shinagawa-ku, Tokyo, Japan, 141 8625 Novartis Investigative Site Shinjuku-Ku, Tokyo, Japan, 169-0073 Collapse << |
| NCT00634920 | - | - | Completed | - | - |
| NCT01429844 | Bronchiolitis Obliterans ... more >> Immunosuppression Collapse << | Phase 3 | Completed | - | Australia ... more >> St. Vincent's Hospital Sydney, Australia, NSW 2010 Austria Allgemeines Krankenhaus Wien Wien, Austria, 1090 Belgium Hospital Erasme Bruxelles, Belgium, 1070 Universitaire Ziekenhuizen Leuven, Belgium, 3000 Germany Universitätsklinikum Essen Essen, Germany, 45147 Universitätsklinikum Hamburg-Eppendorf Hamburg, Germany, 20246 Universitätsklinikum Jena Jena, Germany, 07740 Universitätsklinikum Kiel Kiel, Germany, 24105 Spain Hospital Vall d`Hebron Barcelona, Spain, 08035 Hospital Reina Sofia Cordoba, Spain, 14004 Hospital Juan Canalejo La Coruna, Spain, 15006 Clínica Puerta de Hierro Madrid, Spain, 28035 Hospital Marques de Valdecilla Santander, Spain, 39008 Switzerland Centre hospitalier universitaire vaudois Lausanne, Switzerland, 1011 Collapse << |
| NCT00814515 | Moderate to Severe Dry Eye Syn... more >>drome Collapse << | Phase 3 | Completed | - | France ... more >> Hôpital des XV-XX Paris, Île-de-France, France, 75012 Collapse << |
| NCT00413920 | Renal Transplantation | Phase 3 | Completed | - | France ... more >> C.H.U. La Milétrie Poitiers, France Collapse << |
| NCT00461825 | Kidney Transplantation | Phase 3 | Completed | - | France ... more >> Caen university Hospital Caen, France, 14033 Dupuytren University Hospital Limoges, France, 87042 Poitiers University hospital Poitiers, France, 86021 Reims University Hospital Reims, France, 51092 Rouen University Hospital Rouen, France, 76031 Collapse << |
| NCT00775632 | Graft Versus Host Disease ... more >> Bone Marrow Transplantation Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01976390 | Renal Failure | Not Applicable | Active, not recruiting | May 2018 | United States, Ohio ... more >> The Ohio State University Wexner Medical Center Columbus, Ohio, United States, 43212 Collapse << |
| NCT00537862 | Kidney Transplant | Phase 4 | Completed | - | Korea, Republic of ... more >> Novartis Investigative Site Busan, Korea, Republic of, (614-735) Novartis Investigative Site Daegu, Korea, Republic of, (700-712) Novartis Investigative Site Daegu, Korea, Republic of, (700-721) Novartis Investigative Site Jeollanam-do, Korea, Republic of, (501-757) Novartis Investigative Site Seoul, Korea, Republic of, (110-744) Novartis Investigative Site Seoul, Korea, Republic of, (120-752) Novartis Investigative Site Seoul, Korea, Republic of, (137-701) Novartis Investigative Site Seoul, Korea, Republic of, (138-736) Collapse << |
| NCT00332462 | Liver Transplantation | Phase 4 | Completed | - | Germany ... more >> Novartis Investigational Site Various Cities, Germany Collapse << |
| NCT02547714 | - | - | Completed | - | - |
| NCT00332462 | - | - | Completed | - | - |
| NCT00363779 | - | - | Terminated(Study terminated du... more >>e to low accrual and the investigator left the NIH.) Collapse << | - | - |
| NCT00663455 | Kidney Transplant | Phase 4 | Terminated(Recruitment goals w... more >>ere not achieved. Extension of the trial concept had to be abjected due to methodological reasons.) Collapse << | - | Germany ... more >> Dept. of Pediatric Nephrology, University Hospital Erlangen Erlangen, Germany Dept. of Pediatric Nephrology, University Hospital Freiburg Freiburg, Germany Dept. of Pediatric Nephrology, University Hospital Hamburg Hamburg, Germany Dept. of Pediatric Nephrology, University Hospital Hannover Hannover, Germany Dept. of Pediatric Nephrology, University Hospital Heidelberg Heidelberg, Germany Dept. of Pediatric Nephrology, University Hospital Jena Jena, Germany Dept. of Pediatric Nephrology, Community Hospital Memmingen Memmingen, Germany Dept. of Pediatric Nephrology, University Hospital Muenster Muenster, Germany Dept. of Pediatric Nephrology, University Hospital München Munich, Germany Dept. of Pediatric Nephrology, University Hospital Rostock Rostock, Germany Collapse << |
| NCT01762657 | Type 1 Diabetes | Phase 3 | Withdrawn(Study stopped prior ... more >>to Patient Enrollment) Collapse << | September 2015 | - |
| NCT00363779 | Large Granular Lymphocytic Leu... more >>kemia LGL Leukemia Collapse << | Phase 2 | Terminated(Study terminated du... more >>e to low accrual and the investigator left the NIH.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01825044 | Traumatic Brain Injury | Phase 2 | Completed | - | Denmark ... more >> Dept. of Neurosurgery, Rigshospitalet, University of Copenhagen Copenhagen, Denmark, 2100 Collapse << |
| NCT00668369 | Hepatitis C Virus Infection ... more >> Liver Transplantation Collapse << | Phase 2 | Completed | - | Spain ... more >> Hospital Clinic Barcelona, University of Barcelona Barcelona, Spain, 08036 Collapse << |
| NCT00524953 | Allogeneic Transplantation (No... more >>n T-cell Depleted) Collapse << | Phase 1 | Withdrawn | - | Israel ... more >> Hadassah Medical Organization Jerusalem, Israel Collapse << |
| NCT00692250 | Healthy | Phase 1 | Completed | - | Netherlands ... more >> University Medical Center Groningen- Biotech Center Groningen, Netherlands Collapse << |
| NCT00154284 | Organ Transplantation, Renal T... more >>ransplantation Collapse << | Phase 3 | Completed | - | - |
| NCT00154284 | - | - | Completed | - | - |
| NCT00264355 | Heart Transplantation ... more >> Acute Renal Failure Collapse << | Phase 4 | Completed | - | Norway ... more >> Rikshospitalet, Department of Thoracic surgery Oslo, Norway, Oslo Collapse << |
| NCT01592006 | Hepatitis C | Phase 4 | Terminated(Low accrual) | - | United States, Illinois ... more >> University of Chicago Medical Center Chicago, Illinois, United States, 60637 Collapse << |
| NCT00238901 | Liver Transplant | Phase 3 | Completed | - | - |
| NCT00251004 | - | - | Completed | - | - |
| NCT00425308 | Renal Transplantation | Phase 3 | Completed | - | France ... more >> Novartis Investigative Site Paris, France Collapse << |
| NCT02472054 | Hemophagocytic Lymphohistiocyt... more >>osis (HLH) Collapse << | Phase 1 Phase 2 | Recruiting | April 2020 | France ... more >> Hôpital Necker-Enfants Malades Recruiting Paris, France, 75015 Contact: Despina MOSHOUS, MD, PhD +33 (0) 1 44 49 48 23 despina.moshous@inserm.fr Contact: Valérie JOLAINE, Master +33 (0) 1 42 19 28 79 valerie.jolaine@nck.aphp.fr Collapse << |
| NCT00425308 | - | - | Completed | - | - |
| NCT00087581 | Kidney Transplantation | Phase 4 | Completed | - | - |
| NCT00110110 | Retinoblastoma | Phase 2 | Active, not recruiting | December 2019 | Canada, British Columbia ... more >> Children's and Women's Hospital of British Columbia Vancouver, British Columbia, Canada, V6H 3V4 Canada, Ontario Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Canada, Quebec Montreal Children's Hospital at McGill University Health Center Montreal, Quebec, Canada, H3H 1P3 Chile Hospital San Juan de Dios Santiago, Chile, 8500000 India Sankara Nethralaya Super Specialty Clinic Chennai, India, 600 006 Singapore Kandang Kerbau Women's and Children's Hospital Singapore, Singapore, 229899 Collapse << |
| NCT00371826 | - | - | Completed | - | - |
| NCT01592006 | - | - | Terminated(Low accrual) | - | - |
| NCT00286871 | Chronic Hepatitis C ... more >> Organ Transplantation Immunosuppression Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00150085 | Tacrolimus-associated Abnormal... more >> Glucose Metabolism in Kidney and Liver Transplant Recipients Collapse << | Phase 4 | Terminated | - | - |
| NCT01467505 | Hepatitis C | Phase 2 | Terminated(The study was termi... more >>nated early by the sponsor on 13 January 2014 due to a decision to modify the drug development plan.) Collapse << | - | - |
| NCT00371826 | Renal Transplanted Recipients | Phase 4 | Completed | - | Australia ... more >> Royal Prince Alfred Hospital NSW, Australia Westmead Hospital NSW, Australia Princess Alexandra Hospital QLD, Australia Monash Medical Centre Sale, Australia Queen Elizabeth Hospital Sale, Australia Royal Melbourne Hospital VIC, Australia Sir Charles Gairdner Hospital WA, Australia Collapse << |
| NCT01467505 | - | - | Terminated(The study was termi... more >>nated early by the sponsor on 13 January 2014 due to a decision to modify the drug development plan.) Collapse << | - | - |
| NCT00171496 | Kidney Transplant | Phase 4 | Completed | - | - |
| NCT01504906 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Research site Overland Park, Kansas, United States Collapse << |
| NCT01023815 | de Novo Kidney Transplant Reci... more >>pients Renal Transplantation Collapse << | Phase 3 | Completed | - | Italy ... more >> Novartis Investigative Site Ancona, Italy, 60100 Novartis Investigative Site Bologna, Italy Novartis Investigative Site Brescia, Italy Novartis Investigative Site Cagliari, Italy Novartis Investigative Site Catania, Italy Novartis Investigative Site Coppito, Italy Novartis Investigative Site Firenze, Italy Novartis Investigative Site Genova, Italy, 16132 Novartis Investigative Site Milano, Italy, 20122 Novartis Investigative Site Modena, Italy, 41100 Novartis Investigative Site Napoli, Italy Novartis Investigative Site Novara, Italy, 28100 Novartis Investigative Site Padova, Italy Novartis Investigative Site Palermo, Italy Novartis Investigative Site Parma, Italy Novartis Investigative Site Perugia, Italy, 06070 Novarits Investigative Site Pisa, Italy Novartis Investigative Site Roma, Italy Novartis Investigative Site Rome, Italy Novartis Investigative Site Salerno, Italy Novartis Investigative Site Sassari, Italy, 07100 Novartis Investigative Site Siena, Italy, 53100 Novartis Investigative Site Torino, Italy, 10126 Novartis Investigative Site Treviso, Italy Novartis InvestigativeSite Udine, Italy Novartis Investigative Site Varese, Italy Novartis Investigative Site Verona, Italy Novartis Investigative Site Vicenza, Italy Collapse << |
| NCT01762644 | Type 1 Diabetes | Phase 3 | Withdrawn(This study was withd... more >>rawn prior to enrollment.) Collapse << | August 2016 | - |
| NCT01718652 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Antwerpen, Belgium Collapse << |
| NCT03665766 | - | - | Completed | - | - |
| NCT00171509 | Liver Transplant | Phase 4 | Completed | - | - |
| NCT02042027 | Corneal Neovascularization ... more >> Corneal Graft Failure Anterior Segment Inflammation Collapse << | Phase 1 | Withdrawn(No participants enro... more >>lled) Collapse << | - | United States, Utah ... more >> John A. Moran Eye Center Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT00710801 | - | - | Completed | - | Canada, Alberta ... more >> University of Alberta Hospital Edmonton, Alberta, Canada, T6G 2B7 Collapse << |
| NCT01938625 | - | - | Completed | - | - |
| NCT00163657 | End Stage Liver Disease ... more >> Hepatitis C Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> Baylor Regional Transplant Institute - Baylor University Medical Center Dallas, Texas, United States, 75246 Collapse << |
| NCT00163657 | - | - | Completed | - | - |
| NCT01023815 | - | - | Completed | - | - |
| NCT00284921 | De Novo Kidney Transplant | Phase 3 | Terminated | - | - |
| NCT00154232 | Renal Transplantation | Phase 4 | Completed | - | - |
| NCT01038167 | Hepatitis C | Phase 1 | Completed | - | United States, Texas ... more >> Dallas, Texas, United States Collapse << |
| NCT01469884 | Renal Allograft ... more >> Hepatitis C Collapse << | Phase 4 | Completed | - | Brazil ... more >> Irmandade Da Santa Casa de Misericórdia de Porto Alegre Porto Alegre, Rio Grande Do Sul, Brazil, 90020090 Collapse << |
| NCT01938625 | Hepatitis C, Chronic | Phase 2 | Completed | - | Germany ... more >> Essen, Germany Hamburg, Germany Poland Warszawa, Poland Spain Barcelona, Spain Madrid, Spain Valencia, Spain Collapse << |
| NCT02144792 | Epilepsy | Phase 2 | Unknown | December 2014 | Korea, Republic of ... more >> Seoul National University Hospital Recruiting Seoul, Korea, Republic of Contact: Sang Kun Lee, MD, PhD sangkun2923@gmail.com Principal Investigator: Sang Kun Lee, MD, PhD Collapse << |
| NCT00149994 | - | - | Completed | - | - |
| NCT00938860 | Hepatitis C L... more >>iver Transplantation Collapse << | Phase 4 | Completed | - | - |
| NCT00251004 | Kidney Transplantation ... more >> Graft Rejection Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Novartis East Hanover, New Jersey, United States, 07936 Collapse << |
| NCT00149994 | Liver Transplant | Phase 4 | Completed | - | Switzerland ... more >> Novartis Basel, Switzerland Collapse << |
| NCT00375895 | Chronic Hepatitis C ... more >> Evidence of Liver Transplantation Collapse << | Phase 3 | Terminated(Insufficient enroll... more >>ment) Collapse << | - | France ... more >> Service d'Hépatologie - Hôpital Jean Minjoz Besançon, France, 25030 Service d'Hépatogastroentérologie - Hôpital Beaujon Clichy, France, 92118 Service d'Hépatologie et Gastroentérologie - CH Henri Mondor Créteil, France, 94010 Service des Maladies de l'Appareil Digestif - CHRU Claude Huriez Lille, France, 59037 Service de Chirurgie Générale - Hôpital Edouard Herriot Lyon, France, 69437 Chirurgie Générale - Hôpital de la Conception Marseille, France, 13385 Service d'Hépato-gastro-entérologie - Hôpital Saint Eloi Montpellier, France, 34295 Chirurgie Viscérale et Digestive - Hôpital de l'Archet Nice, France, 06200 Service de Chirurgie Générale - Hôpital Cochin Paris, France, 75679 Service des Maladies du Foie - Hôpital Pontchaillou Rennes, France, 35033 Service de Chirurgie Générale et Transplantation Multi-organe - Hôpital de la Hautepierre Strasbourg, France, 67098 Service d'Hépato-gastro-entérologie - Hôpital de Rangueil Toulouse, France, 31403 Centre Hépato-Biliaire - Hôpital Paul Brousse Villejuif, France, 94804 Collapse << |
| NCT00938860 | - | - | Completed | - | - |
| NCT00260208 | Liver Transplant ... more >> Hepatitis C Collapse << | Phase 4 | Terminated(Study was premature... more >>ly terminated due to poor recruitment.) Collapse << | - | United States, New Jersey ... more >> Novartis Investigative Site East Hanover, New Jersey, United States Switzerland Novartis Investigational Site Zurich, Switzerland, 8091 Collapse << |
| NCT02108301 | Chronic Hepatitis C-virus Infe... more >>ction Renal Transplantation Collapse << | Phase 4 | Unknown | - | Austria ... more >> Medical University of Vienna, Department of Internal Medicine III, Division of Nephrology and Dialysis Vienna, Austria, 1090 Collapse << |
| NCT01479881 | Hepatitis C Virus | Phase 1 | Completed | - | Belgium ... more >> Merksem, Belgium Collapse << |
| NCT00154193 | Heart and Lung Transplant | Phase 4 | Completed | - | Switzerland ... more >> Novartis Basel, Switzerland Collapse << |
| NCT00821587 | - | - | Completed | - | - |
| NCT00821587 | Hepatitis C | Phase 4 | Completed | - | - |
| NCT00260208 | - | - | Terminated(Study was premature... more >>ly terminated due to poor recruitment.) Collapse << | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
