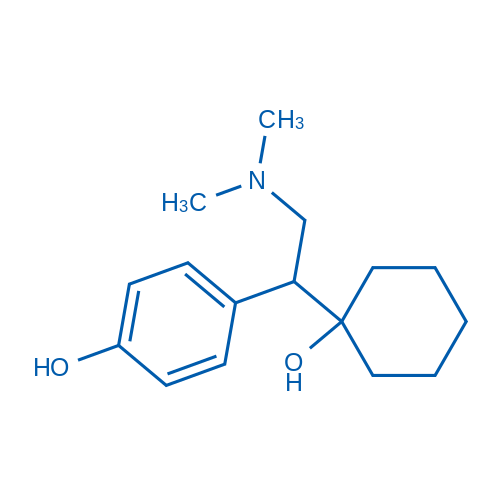
CAS No.: 93413-62-8
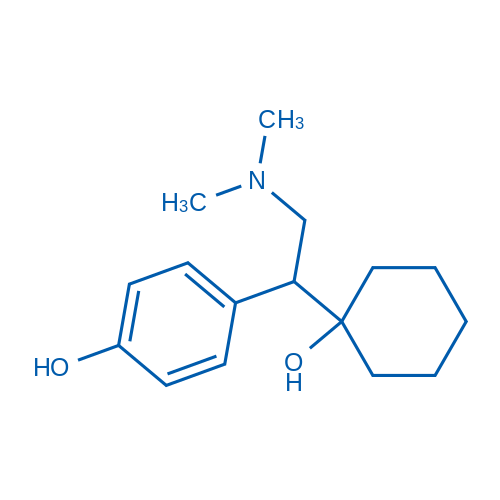
Desvenlafaxine/去甲文拉法辛 Catalog No. CSN10736
Synonyms: WY 45233 Succinate;WY 45233;O-Desmethylvenlafaxine
Desvenlafaxine inhibits serotonin(5-HT) and norepinephrine (NE) reuptake with Ki of 40.2 nM and 558.4 nM, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 5-HT Receptor
Ki:40.2nMnorepinEphrine reuptake
Ki:558.4nM- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00797966 Major Depressive Disorder Phase 2 Completed - - NCT00797966 - - Completed - - NCT01916824 Major Depressive Disorder ... more >> Healthy Controls Collapse << Phase 4 Completed - United States, Georgia ... more >> Emory Mood and Anxiety Disorders Program Atlanta, Georgia, United States, 30322 Collapse << - 更多
- 参考文献
- [1] Sopko MA Jr, Ehret MJ, Grgas M. Desvenlafaxine: another "me too" drug? Ann Pharmacother. 2008 Oct;42(10):1439-46.
- [2] Deecher DC, Beyer CE, et al.Desvenlafaxine succinate: A new serotonin and norepinephrine reuptake inhibitor. J Pharmacol Exp Ther. 2006 Aug;318(2):657-65.
- [3] Deecher DC, Beyer CE, Johnston G, et al. Desvenlafaxine succinate: A new serotonin and norepinephrine reuptake inhibitor. J Pharmacol Exp Ther. 2006;318(2):657‐665
- [4] Sopko MA Jr, Ehret MJ, Grgas M. Desvenlafaxine: another "me too" drug?. Ann Pharmacother. 2008;42(10):1439‐1446
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 93413-62-8 | 储存条件 |
|
|||||||||||
| 分子式 | C16H25NO2 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 263.38 | 别名 | WY 45233 Succinate;WY 45233;O-Desmethylvenlafaxine;(±)-O-Desmethyl Venlafaxine;DVS 233;MDD-XR;Pristiq;Desfax;去甲文拉法辛 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00797966 | Major Depressive Disorder | Phase 2 | Completed | - | - |
| NCT00797966 | - | - | Completed | - | - |
| NCT01916824 | Major Depressive Disorder ... more >> Healthy Controls Collapse << | Phase 4 | Completed | - | United States, Georgia ... more >> Emory Mood and Anxiety Disorders Program Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01916824 | - | - | Completed | - | - |
| NCT01350921 | Drug Drug Interaction | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Sweden Research Site Uppsala, Sweden Collapse << |
| NCT01469377 | Major Depressive Disorder | Phase 2 | Completed | - | - |
| NCT01469377 | - | - | Completed | - | - |
| NCT01235338 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Clinical Pharmacology of Miami Miami, Florida, United States, 33014 Collapse << |
| NCT01235338 | - | - | Completed | - | - |
| NCT00834249 | - | - | Completed | - | - |
| NCT02400346 | - | - | Completed | - | - |
| NCT02400346 | Major Depression Disorder | Phase 3 | Completed | - | - |
| NCT00834249 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, H3X 2H9 Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT00834964 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Montreal, Quebec, Canada, H3X 2H9 Anapharm Inc. Sainte-Foy, Quebec, Canada, GIV2K8 Collapse << |
| NCT02054299 | Drug Metabolism ... more >> Membrane Transport Collapse << | Phase 1 | Completed | - | Germany ... more >> Department of Clinical Pharmacology, University Medical Center Goettingen Goettingen, Germany, 37075 Collapse << |
| NCT00834964 | - | - | Completed | - | - |
| NCT02893371 | - | - | Enrolling by invitation | June 2019 | United States, New Mexico ... more >> Christophe G Lambert Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT02058693 | Major Depressive Disorder | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 Collapse << |
| NCT02637193 | Healthy Subjects | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00767624 | Sleep Initiation and Maintenan... more >>ce Disorders Depression Collapse << | Phase 2 Phase 3 | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 United States, North Carolina Duke University Durham, North Carolina, United States United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States Collapse << |
| NCT00767624 | - | - | Completed | - | - |
| NCT00421031 | Vasomotor Symptoms | Phase 3 | Completed | - | - |
| NCT00256685 | Menopause Hot... more >> Flashes Sleep Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT01867255 | Bariatric Surgery ... more >> Gastric Bypass Roux-en-Y Gastric Bypass Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01260896 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, G1V 2K8 Collapse << |
| NCT01260896 | - | - | Completed | - | - |
| NCT01282801 | - | - | Completed | - | - |
| NCT02320240 | - | - | Completed | - | Canada, Quebec ... more >> Lady Davis Institute for Medical Research, Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00366652 | Depression De... more >>pressive Disorder Depressive Disorder, Major Collapse << | Phase 3 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States, 07753 Collapse << |
| NCT02055300 | Major Depressive Disorder | Phase 1 | Completed | - | United States, Ohio ... more >> Medpace Cincinnati, Ohio, United States, 45227 Collapse << |
| NCT01282801 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Anapharm Inc. Sainte-Foy, Quebec, Canada, G1V 2K8 Collapse << |
| NCT00456898 | Depressive Disorder, Major ... more >> Diabetic Neuropathies Fibromyalgia Vasomotor Symptoms Collapse << | Phase 1 | Completed | - | - |
| NCT00283842 | Diabetic Neuropathies ... more >> Pain Collapse << | Phase 3 | Terminated(business reasons) | - | - |
| NCT02271412 | Major Depressive Disorder, Rec... more >>urrent, Unspecified Collapse << | Phase 1 | Completed | - | United States, New Jersey ... more >> PRA Health Sciences Marlton, New Jersey, United States, 08053 Collapse << |
| NCT00329147 | Depression | Phase 3 | Completed | - | United States, New Jersey ... more >> Neptune, New Jersey, United States, 07753 Collapse << |
| NCT00401245 | Vasomotor Symptoms | Phase 3 | Completed | - | - |
| NCT00445679 | Depressive Disorder, Major | Phase 3 | Completed | - | - |
| NCT00401245 | - | - | Completed | - | - |
| NCT00283842 | - | - | Terminated(business reasons) | - | - |
| NCT00445679 | - | - | Completed | - | - |
| NCT00452595 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00696787 | - | - | Terminated | - | - |
| NCT00397176 | Vasomotor Symptoms | Phase 1 | Completed | - | - |
| NCT00424892 | Fibromyalgia | Phase 2 Phase 3 | Completed | - | - |
| NCT00696787 | Fibromyalgia | Phase 2 | Terminated | - | - |
| NCT00277823 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01056289 | Major Depressive Disorder | Phase 4 | Completed | - | - |
| NCT01056289 | - | - | Completed | - | - |
| NCT01050218 | - | - | Terminated(Business reasons) | - | - |
| NCT02548949 | - | - | Recruiting | September 2019 | Korea, Republic of ... more >> Chungbuk National University Hospital Recruiting Cheongju-si, Chungcheonbuk-do, Korea, Republic of, 362-711 Hallym University Sacred Heart Hospital Recruiting Anyang-Si, Gyeonggi-do, Korea, Republic of, 431-796 Inje University Ilsan Paik Hospital Recruiting Goyang-si, Gyeonggi-do, Korea, Republic of, 411-706 Bundang Cha Medical Center Recruiting Seongnam-si, Gyeonggi-do, Korea, Republic of, 13496 Bong Seng Memorial Hospital Recruiting Busan, Korea, Republic of, 601-723 Pusan National University Hospital Not yet recruiting Busan, Korea, Republic of, 602-739 Kyungpook National University Hospital Recruiting Daegu, Korea, Republic of, 700-721 Chungnam National University Hospital Recruiting Daejeon, Korea, Republic of, 301-721 Chungnam National University Hospital Not yet recruiting Daejeon, Korea, Republic of, 301-721 Chuncheon Sacred Heart Hospital-Hallym University Recruiting Gangwon-do, Korea, Republic of, 24253 Chosun University Hospital Recruiting Gwangju, Korea, Republic of, 61453 Chonnam National University Hospital Recruiting Gwangju, Korea, Republic of, 61469 Presbyterian Medical Center Recruiting Jeonju, Korea, Republic of, 560-750 Nowon Eulji Medical Center, Eulji University Recruiting Seoul, Korea, Republic of, 01830 Korea University Anam Hospital Recruiting Seoul, Korea, Republic of, 02841 Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Hanyang University Seoul Hospital Recruiting Seoul, Korea, Republic of, 04763 Chung-Ang University Hospital Recruiting Seoul, Korea, Republic of, 06973 Konkuk University Medical Center Recruiting Seoul, Korea, Republic of, 143-729 Collapse << |
| NCT01372150 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01050218 | Diabetic Neuropathy, Painful | Phase 3 | Terminated(Business reasons) | - | - |
| NCT01309542 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01372150 | - | - | Completed | - | - |
| NCT01371708 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01371708 | - | - | Completed | - | - |
| NCT01371721 | - | - | Completed | - | - |
| NCT01371721 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00406640 | Depression De... more >>pressive Disorder Depressive Disorder, Major Collapse << | Phase 3 | Completed | - | - |
| NCT00798707 | - | - | Completed | - | - |
| NCT00406640 | - | - | Completed | - | - |
| NCT01371734 | - | - | Completed | - | - |
| NCT00798707 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01371734 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00369343 | Depression De... more >>pressive Disorder Depressive Disorder, Major Collapse << | Phase 3 | Completed | - | - |
| NCT00369343 | - | - | Completed | - | - |
| NCT00369434 | Menopause Vas... more >>omotor System Collapse << | Phase 3 | Completed | - | - |
| NCT00727064 | - | - | Completed | - | - |
| NCT01190514 | Depression - Major Depressive ... more >>Disorder Collapse << | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT01190514 | - | - | Completed | - | - |
| NCT00727064 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Clinical Research Institute Wichita, Kansas, United States, 67211 Collapse << |
| NCT00619619 | Depression Ma... more >>jor Depressive Disorder Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> Pfizer Investigational Site Little Rock, Arkansas, United States, 72205 United States, Florida Pfizer Investigational Site North Miami, Florida, United States, 33161 United States, Indiana Pfizer Investigational Site Terre Haute, Indiana, United States, 47802 United States, Kansas Pfizer Investigational Site Wichita, Kansas, United States, 67211 United States, Louisiana Pfizer Investigational Site New Orleans, Louisiana, United States, 70114 United States, New York Pfizer Investigational Site New York, New York, United States, 10032 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44106-5080 United States, Pennsylvania Pfizer Investigational Site Hershey, Pennsylvania, United States, 17033 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77008 Collapse << |
| NCT00619619 | - | - | Completed | - | - |
| NCT00384033 | - | - | Completed | - | - |
| NCT00384033 | Depressive Disorder, Major | Phase 3 | Completed | - | United States, California ... more >> Pfizer Investigational Site Beverly Hills, California, United States, 90210 Pfizer Investigational Site Burbank, California, United States, 91506 Pfizer Investigational Site Encino, California, United States, 91316 Pfizer Investigational Site Los Alamitos, California, United States, 90720 Pfizer Investigational Site Newport Beach, California, United States, 92660 Pfizer Investigational Site Northridge, California, United States, 91324 Pfizer Investigational Site Orange, California, United States, 92868 Pfizer Investigational Site Pasadena, California, United States, 91105 Pfizer Investigational Site Upland, California, United States, 91786 United States, Florida Pfizer Investigational Site South Miami, Florida, United States, 33143 Pfizer Investigational Site St. Petersburg, Florida, United States, 33702 United States, Illinois Pfizer Investigational Site Edwardsville, Illinois, United States, 62025 United States, Michigan Pfizer Investigational Site Farmington Hills, Michigan, United States, 48336 Pfizer Investigational Site Flint, Michigan, United States, 48507 Pfizer Investigational Site Okemos, Michigan, United States, 48864 United States, New Jersey Pfizer Investigational Site Clementon, New Jersey, United States, 08021 United States, Ohio Pfizer Investigational Site Dayton, Ohio, United States, 45408 United States, Oregon Pfizer Investigational Site Portland, Oregon, United States, 97210 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19149 United States, Utah Pfizer Investigational Site Salt Lake City, Utah, United States, 84107 United States, Washington Pfizer Investigational Site Seattle, Washington, United States, 98104 United States, Wisconsin Pfizer Investigational Site Brown Deer, Wisconsin, United States, 53223 Collapse << |
| NCT00863798 | Major Depressive Disorder | Phase 3 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmingham, Alabama, United States, 35216 United States, California Pfizer Investigational Site Encino, California, United States, 91316 Pfizer Investigational Site Newport Beach, California, United States, 92660 Pfizer Investigational Site Redlands, California, United States, 92374 Pfizer Investigational Site Upland, California, United States, 91786 United States, Colorado Pfizer Investigational Site Aurora, Colorado, United States, 80045 Pfizer Investigational Site Denver, Colorado, United States, 80204 United States, Connecticut Pfizer Investigational Site Cromwell, Connecticut, United States, 06416 United States, Florida Pfizer Investigational Site Maitland, Florida, United States, 32751 Pfizer Investigational Site North Miami, Florida, United States, 33161 Pfizer Investigational Site South Miami, Florida, United States, 33143 United States, Indiana Pfizer Investigational Site Indianapolis, Indiana, United States, 46260 United States, Missouri Pfizer Investigational Site St. Louis, Missouri, United States, 63139 United States, New York Pfizer Investigational Site New York, New York, United States, 10128 Pfizer Investigational Site Staten Island, New York, United States, 10312 United States, Ohio Pfizer Investigational Site Toledo, Ohio, United States, 43623 United States, Oregon Pfizer Investigational Site Eugene, Oregon, United States, 97401 Pfizer Investigational Site Portland, Oregon, United States, 97210 Pfizer Investigational Site Salem, Oregon, United States, 97301 United States, Pennsylvania Pfizer Investigational Site Media, Pennsylvania, United States, 19063 United States, Virginia Pfizer Investigational Site Herndon, Virginia, United States, 20170 Pfizer Investigational Site Midlothian, Virginia, United States, 23112 United States, Wisconsin Pfizer Investigational Site Middleton, Wisconsin, United States, 53562 Pfizer Investigational Site Waukesha, Wisconsin, United States, 53188 Collapse << |
| NCT00440427 | Depressive Disorder, Major | Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02135 Collapse << |
| NCT00329186 | Healthy | Phase 3 | Completed | - | United States, Kansas ... more >> Wichita, Kansas, United States, 67207 Collapse << |
| NCT00863798 | - | - | Completed | - | - |
| NCT00300378 | Depressive Disorder, Major | Phase 3 | Completed | - | - |
| NCT01468610 | Major Depressive Disorder | Not Applicable | Completed | - | Canada, British Columbia ... more >> University of British Columbia, Department of Psychiatry Vancouver, British Columbia, Canada, V6T 2A1 Collapse << |
| NCT00683800 | - | - | Completed | - | - |
| NCT00683800 | Vasomotor Symptoms | Phase 3 | Completed | - | - |
| NCT00669110 | Depressive Disorder, Major Dep... more >>ressive Disorder Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> Pfizer Investigational Site Little Rock, Arkansas, United States, 72205 United States, Florida Pfizer Investigational Site North Miami, Florida, United States, 33161 United States, Indiana Pfizer Investigational Site Terre Haute, Indiana, United States, 47802 United States, Kansas Pfizer Investigational Site Wichita, Kansas, United States, 67211 United States, Louisiana Pfizer Investigational Site New Orleans, Louisiana, United States, 70114 United States, New York Pfizer Investigational Site New York, New York, United States, 10032 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44106 United States, Pennsylvania Pfizer Investigational Site Hershey, Pennsylvania, United States, 17033 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77008 Collapse << |
| NCT00952653 | - | - | Completed | - | - |
| NCT00669110 | - | - | Completed | - | - |
| NCT00952653 | Major Depressive Disorder | Phase 4 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00887224 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01221935 | - | - | Completed | - | - |
| NCT00887224 | - | - | Completed | - | - |
| NCT02330679 | Major Depressive Disorder | Phase 4 | Unknown | December 2016 | Canada, Alberta ... more >> University of Calgary, TRW Building, Foothills Hospital Campus Recruiting Calgary, Alberta, Canada, T2N4Z6 Contact: Rajamannar Ramasubbu, MD, FRCP(C) 403-210-6890 rramasub@ucalgary.ca Contact: Darren Clark, PhD 403-210-6353 dlclark@ucalgary.ca Principal Investigator: Rajamannar Ramasubbu, MD, FRCP(C) University of Calgary: Foothills Hospital Recruiting Calgary, Alberta, Canada, T2N4Z6 Principal Investigator: Rajamannar Ramasubbu, MD, FRCP(C) Collapse << |
| NCT01121484 | Major Depressive Disorder | Phase 4 | Completed | - | - |
| NCT01492621 | Major Depression | Phase 4 | Unknown | November 2013 | Canada, Quebec ... more >> Centre de Recherche Fernand-Seguin Recruiting Montreal, Quebec, Canada, H1N3V2 Contact: Souad Lahlafi, Bnurs 5142514000 ext 3416 slahlafi.crfs@ssss.gouv.qc.ca Contact: Louise Normandeau, Nurs 5142514000 ext 3535 lnormandeau.hlhl@ssss.gouv.qc.ca Collapse << |
| NCT01975480 | Generalized Anxiety Disorder | Phase 4 | Recruiting | August 1, 2018 | Canada, Ontario ... more >> START Clinic for Mood and Anxiety Disorders Recruiting Toronto, Ontario, Canada, M4W 2N4 Contact: Martin A Katzman, BSc, MD, FRCPC 416-598-9344 mkatzman@startclinic.ca Principal Investigator: Martin A Katzman, BSc Collapse << |
| NCT01101152 | Healthy | Phase 1 | Unknown | December 2011 | Canada, Ontario ... more >> University of Ottawa Institute of Mental Health Research Recruiting Ottawa, Ontario, Canada, K1Z 7K4 Contact: Olga Chernoloz, B.Sc.Pharm 613-722-6521 ext 6405 olga.chernoloz@rohcg.on.ca Principal Investigator: Pierre Blier, M.D., Ph.D. Collapse << |
| NCT00824291 | Depressive Disorder, Major | Phase 3 | Completed | - | - |
| NCT01121484 | - | - | Completed | - | - |
| NCT01803711 | Major Depression ... more >> Cancer Diabetes Cardiovascular Diseases Collapse << | Phase 2 Phase 3 | Terminated(Lack of recruitment... more >> and no resources) Collapse << | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 Collapse << |
| NCT00824291 | - | - | Completed | - | - |
| NCT01189500 | Pharmacokinetics | Phase 4 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site Miami, Florida, United States, 33126 Pfizer Investigational Site Miami, Florida, United States, 33134 Collapse << |
| NCT01803711 | - | - | Terminated(Lack of recruitment... more >> and no resources) Collapse << | - | - |
| NCT01443208 | Major Depressive Disorder | Phase 1 | Completed | - | Korea, Republic of ... more >> Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01189500 | - | - | Completed | - | - |
| NCT01432457 | Major Depressive Disorder | Phase 4 | Completed | - | - |
| NCT00831415 | - | - | Completed | - | - |
| NCT00831415 | Major Depressive Disorder | Phase 3 | Completed | - | Japan ... more >> Pfizer Investigational Site Aichi, Japan Pfizer Investigational Site Chiba, Japan Pfizer Investigational Site Fukuoka, Japan Pfizer Investigational Site Fukushima, Japan Pfizer Investigational Site Gunma, Japan Pfizer Investigational Site Hiroshima, Japan Pfizer Investigational Site Hokkaido, Japan Pfizer Investigational Site Hyogo, Japan Pfizer Investigational Site Ishikawa, Japan Pfizer Investigational Site Kanagawa, Japan Pfizer Investigational Site Kumamoto, Japan Pfizer Investigational Site Kyoto, Japan Pfizer Investigational Site Osaka, Japan Pfizer Investigational Site Saga, Japan Pfizer Investigational Site Saitama, Japan Pfizer Investigational Site Shiga, Japan Pfizer Investigational Site Tokyo, Japan Collapse << |
| NCT01977378 | Major Depressive Disorder | Phase 2 Phase 3 | Unknown | - | China, Guangdong ... more >> Guangdong General Hospital Recruiting Guangzhou, Guangdong, China China, Hunan The Second Xiangya Hospital of Central South University Recruiting Changsha, Hunan, China China, Sichuan West China Hospital Recruiting Chengdu, Sichuan, China China Beijing An Ding Hospital Recruiting Beijing, China, 100088 Beijing HuiLongGuan Hospital Recruiting Beijing, China, 100096 The Shanghai Mental Health Recruiting Shanghai, China, 200030 Contact: Huafang Li, Professor 86-021-34773128 lhlh_5@163.com Principal Investigator: Huafang Li, Professor Collapse << |
| NCT01432457 | - | - | Completed | - | - |
| NCT01316302 | - | - | Completed | - | - |
| NCT01353963 | - | - | Terminated | - | Philippines ... more >> Private Clinic Las Pinas, Philippines Private Clinic Manila, Philippines Private Clinic Pasay City, Philippines Collapse << |
| NCT01527786 | Depression | Phase 3 | Completed | - | Canada, British Columbia ... more >> BC Women's Hospital Vancouver, British Columbia, Canada, V6H 3N1 Collapse << |
| NCT01353963 | - | - | Terminated | - | - |
| NCT01316302 | Social Anxiety Disorder | Phase 4 | Completed | - | United States, New York ... more >> The Medical Research Network, LLC New York, New York, United States, 10128 Collapse << |
| NCT00818155 | Healthy | Phase 1 | Completed | - | - |
| NCT01537068 | Dysthymic Disorder ... more >> Dysthymia Chronic Depressive Disorder Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute/3 Columbus Circle Midtown New York, New York, United States, 10019 Depression Evaluation Service (DES), New York State Psychiatric Institute, Columbia University Department of Psychiatry New York, New York, United States, 10032 Collapse << |
| NCT01188668 | - | - | Completed | - | - |
| NCT02988024 | Major Depression | Phase 1 | Completed | - | United States, New Jersey ... more >> Clinilabs, Inc. Eatontown, New Jersey, United States, 07724 Collapse << |
| NCT01537068 | - | - | Completed | - | - |
| NCT01188668 | Pharmacokinetics ... more >> Cytochrome P-450 CYP2D6 CYP3A4 Protein, Human Collapse << | Phase 4 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site Miami, Florida, United States, 33126 Pfizer Investigational Site Miami, Florida, United States, 33134 Collapse << |
| NCT03432221 | - | - | Not yet recruiting | April 3, 2019 | - |
| NCT00888862 | Major Depressive Disorder ... more >> Menopausal Staging and Vasomotor Symptoms (for Females) Collapse << | Phase 3 | Unknown | October 2012 | Canada, Ontario ... more >> Women's Health Concerns Clinic Recruiting Hamilton, Ontario, Canada, L8P 3B6 Contact: Stefanie M Attard 905-522-1155 ext 32048 sattard@stjoes.ca Principal Investigator: Claudio N Soares, MD, PhD Sub-Investigator: Benicio N Frey, MD, PhD Sub-Investigator: Geoff Hall, PhD Sub-Investigator: Meir Steiner, MD, PhD Collapse << |
| NCT02859103 | Depression An... more >>hedonia Collapse << | Phase 4 | Recruiting | January 2019 | Canada, Ontario ... more >> St. Michael's Hospital Recruiting Toronto, Ontario, Canada, M5B 1M8 Contact: Troy Chow, HBSc 416-864-6060 ext 7844 chowt@smh.ca Principal Investigator: Sakina Rizvi, PhD Collapse << |
| NCT02819921 | Hot Flashes B... more >>reast Neoplasms Collapse << | Phase 4 | Recruiting | December 2020 | Korea, Republic of ... more >> National Cancer Center Recruiting Goyang, Gyeonggi-do, Korea, Republic of, 10408 Contact: Hyun-Jung Lee, M.D. Principal Investigator: Hyun-Jung Lee, M.D. Seoul National University Bundang Hospital Not yet recruiting Seongnam, Gyeonggi-do, Korea, Republic of, 13620 Contact: Jeong-Hyun Kim, M.D., Ph.D. Principal Investigator: Jeong-Hyun Kim, M.D., Ph.D. Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Contact: Kyung-Lak Son, M.D. 82-02-2072-3767 klson01@gmail.com Collapse << |
| NCT03733574 | Major Depressive Disorder | Phase 1 | Completed | - | United States, Maryland ... more >> Pharmaron CPC, Inc. Baltimore, Maryland, United States, 21201 Collapse << |
| NCT01974934 | Vascular Depression | Phase 4 | Unknown | - | Canada, Ontario ... more >> Hotel-Dieu Grace Healthcare Recruiting Windsor, Ontario, Canada, N9C 3Z4 Contact: Jeff Geml jeff.geml@hdgh.org Principal Investigator: Corina Velehorschi, MD Collapse << |
| NCT01948895 | Dysthymic Disorder | Not Applicable | Completed | - | Canada, Ontario ... more >> Medical Research Associates Mississauga, Ontario, Canada, L5M 4N4 Centre for Addiction and Mental Health Toronto, Ontario, Canada, M5T 1R8 Collapse << |
| NCT03357796 | Major Depressive Disorder | Phase 1 | Completed | - | United States, New Jersey ... more >> Clinilabs, Inc. Eatontown, New Jersey, United States, 07724 Collapse << |
| NCT02200406 | Depression Op... more >>ioid Dependence Methadone Treatment Collapse << | Phase 4 | Completed | - | Canada, Quebec ... more >> Centre de recherche du Centre Hospitalier de l'Université de Montréal Montréal, Quebec, Canada, H2X0A9 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| 5-HT Receptor | Ki:40.2nM | |
| norepinEphrine reuptake | Ki:558.4nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
