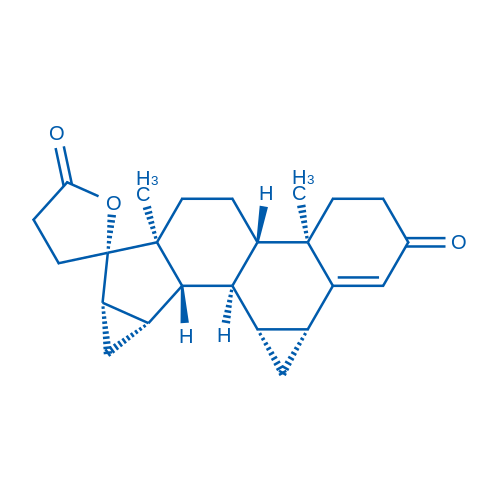
CAS No.: 67392-87-4
Drospirenone/屈螺酮 Catalog No. CSN10873
Synonyms: Dihydrospirorenone;ZK 3059;1,2-dihydro Spirorenone
Drospirenone, analog of spironolactone, is a synthetic progestin.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00185328 Postmenopause Phase 4 Completed - - NCT00442689 Polycystic Ovary Syndrome ... more >> Metabolic Syndrome Collapse << Not Applicable Completed - United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << NCT00442689 - - Completed - - - 更多
- 参考文献
- [1] Fuhrmann U, Krattenmacher R, et al. The novel progestin drospirenone and its natural counterpart progesterone: biochemical profile and antiandrogenic potential. Contraception. 1996 Oct;54(4):243-51.
- [2] Muhn P, Krattenmacher R, et al. Drospirenone: a novel progestogen with antimineralocorticoid and antiandrogenic activity. Pharmacological characterization in animal models. Contraception. 1995 Feb;51(2):99-110.
- [3] Fuhrmann U, Krattenmacher R, Slater EP, Fritzemeier KH. The novel progestin drospirenone and its natural counterpart progesterone: biochemical profile and antiandrogenic potential. Contraception. 1996 Oct;54(4):243-51
- [4] Genazzani AR, Mannella P, Simoncini T. Drospirenone and its antialdosterone properties. Climacteric. 2007 Feb;10 Suppl 1:11-8
- [5] Mallareddy M, Hanes V, White WB. Drospirenone, a new progestogen, for postmenopausal women with hypertension. Drugs Aging. 2007;24(6):453-66
- [6] Christiansen C. Effects of drospirenone/estrogen combinations on bone metabolism. Climacteric. 2005 Oct;8 Suppl 3:35-41
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 67392-87-4 | 储存条件 |
|
|||||
| 分子式 | C24H30O3 | 运输 | 蓝冰 | |||||
| 分子量 | 366.49 | 别名 | Dihydrospirorenone;ZK 3059;1,2-dihydro Spirorenone;drospirenone;67392-87-4;Drospirenona;Drospirenonum;Dehydrospirorenone;屈螺酮 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00185328 | Postmenopause | Phase 4 | Completed | - | - |
| NCT00442689 | Polycystic Ovary Syndrome ... more >> Metabolic Syndrome Collapse << | Not Applicable | Completed | - | United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << |
| NCT00442689 | - | - | Completed | - | - |
| NCT03075956 | Contraception ... more >> Menopause Collapse << | Phase 1 | Completed | - | Bulgaria ... more >> COMAC Sofia, Bulgaria Collapse << |
| NCT01319500 | - | - | Completed | - | - |
| NCT00651846 | Healthy | Phase 4 | Completed | - | Germany ... more >> Berlin, Germany, 10115 Collapse << |
| NCT01319500 | - | - | Completed | - | Germany ... more >> Center for Epidemiology and Health Research Berlin Berlin, Germany, 10115 Collapse << |
| NCT03126773 | - | - | Recruiting | April 30, 2020 | Russian Federation ... more >> Many locations Recruiting Multiple Locations, Russian Federation Collapse << |
| NCT00214903 | - | - | Completed | - | - |
| NCT00948402 | Polycystic Ovarian Syndrome ... more >> Insulin Sensitivity Collapse << | Phase 3 | Completed | - | - |
| NCT01852786 | - | - | Unknown | February 2016 | Lithuania ... more >> Lithuanian University of Health Sciences Kaunas, Lithuania, LT- 50009 Collapse << |
| NCT00651599 | Vasomotor Symptoms | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul, Korea, Republic of, 110-744 Seoul, Korea, Republic of, 135-710 Seoul, Korea, Republic of, 138-736 Seoul, Korea, Republic of Collapse << |
| NCT00593294 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | Italy ... more >> Department of Molecular and Clinical Endocrinology and Oncology, University "Federico II" Naples, Italy, 80131 Collapse << |
| NCT01805817 | Contraceptive Methods Comparis... more >>on Collapse << | Phase 4 | Unknown | - | Turkey ... more >> Istanbul Training and Research Hospital Recruiting Istanbul, Turkey, 34098 Contact: Cihangir Uzunçakmak, M.D. 0090 505 4505808 cuzuncakmak@gmail.com Principal Investigator: Cihangir Uzuncakmak, M.D. Collapse << |
| NCT00214903 | - | - | Completed | - | Germany ... more >> Center for Epidemiology and Health Research Berlin, Germany, 10115 Collapse << |
| NCT02237131 | Dysmenorrhea ... more >>Pelvic Pain Dyspareunia Endometrioma Collapse << | Phase 4 | Unknown | December 2015 | Greece ... more >> Aretaieion Hospital Recruiting Athens, Greece Contact: Olga B Triantafyllidou, M.D, Msc +306944963506 triantafyllidouolga@yahoo.com Principal Investigator: Nikos Vlahos, AssProfessor Collapse << |
| NCT00785317 | Hormone Replacement Therapy | Phase 4 | Unknown | April 2010 | Sweden ... more >> Karolinska University Hospital Recruiting Stockholm, Sweden Principal Investigator: Eva Lundström Collapse << |
| NCT02710708 | Contraception | Phase 4 | Completed | - | - |
| NCT00089414 | - | - | Terminated(Informed by manufac... more >>turer that CDB-2914 crosses blood-brain barrier invalidating Arm #3 of protocol.) Collapse << | - | - |
| NCT03418363 | Contraceptive Usage ... more >> Coagulation Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, Oregon ... more >> Oregon Health & Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT00089414 | Premenstrual Syndrome ... more >> PMS Premenstrual Dysphoric Disorder PMDD Depression Collapse << | Phase 2 | Terminated(Informed by manufac... more >>turer that CDB-2914 crosses blood-brain barrier invalidating Arm #3 of protocol.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02089620 | Premenstrual Syndrome | Phase 3 | Completed | - | Egypt ... more >> Beni-Suef University Beni-Suef, Egypt Collapse << |
| NCT01291004 | Follicle Development ... more >> Ovarian Follicle Follicle Count Follicle Size Oral Contraceptive Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Teva Investigational Site Philadelphia, Pennsylvania, United States, 19114 United States, Washington Teva Investigational Site Seattle, Washington, United States, 98105 Collapse << |
| NCT02562053 | Premenstrual Syndrome | Phase 3 | Unknown | September 2017 | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Cairo University Hospitals Recruiting Cairo, Egypt Contact: AbdelGany MA Hassan, MRCOG, MD 00217801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT00676065 | - | - | Completed | - | Germany ... more >> Center for Epidemiology and Health Research Berlin, Germany Collapse << |
| NCT00676065 | - | - | Completed | - | - |
| NCT02488538 | Premenstrual Syndrome | Phase 3 | Recruiting | - | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Principal Investigator: Nesreen AA Shehata, MD Cairo University Hospitals Recruiting Cairo, Egypt Contact: AbdelGany MA Hassan, MRCOG, MD 00217801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT01266408 | - | - | Recruiting | December 2025 | Germany ... more >> Center for Epidemiology and Health Research Recruiting Berlin, Germany, 10115 Contact: Suzanne Reed, MSc, PhD +49 30 945 101 63 reed@zeg-berlin.de Contact: Klaas Heinemann, PhD, MD, MBA +49 30 945 101 24 k.heinemann@zeg-berlin.de Principal Investigator: Juergen C Dinger, PhD, MD Collapse << |
| NCT00567164 | Contraception ... more >> Ovulation Inhibition Contraceptives, Oral Collapse << | Phase 3 | Completed | - | - |
| NCT00567164 | - | - | Completed | - | - |
| NCT00824187 | Premenstrual Dysphoric Disorde... more >>r ( PMDD) Collapse << | Phase 3 | Completed | - | China, Guangdong ... more >> Guangzhou, Guangdong, China, 510405 Guangzhou, Guangdong, China, 510630 China, Hubei Wuhan, Hubei, China, 430060 China, Hunan Changsha, Hunan, China, 410011 China, Jiangsu Nanjing, Jiangsu, China, 210029 China, Liaoning Dalian, Liaoning, China, 116011 China, Shandong Jinan, Shandong, China, 250012 China, Shanxi Xi'an, Shanxi, China, 710032 Xi'an, Shanxi, China, 710061 China, Sichuan Chengdu, Sichuan, China, 610041 China, Yunnan Kunming, Yunnan, China, 650032 China, Zhejiang Hangzhou, Zhejiang, China, 310003 China Beijing, China, 100050 Beijing, China, 100191 Beijing, China, 100853 Tianjin, China, 300193 Collapse << |
| NCT03353857 | Biological Availability | Phase 1 | Active, not recruiting | December 18, 2018 | Germany ... more >> CRS Clinical-Research-Services Mannheim GmbH Mannheim, Baden-Württemberg, Germany, 68167 CRS Clinical-Research-Services Mönchengladbach GmbH Mönchengladbach, Nordrhein-Westfalen, Germany, 41061 Collapse << |
| NCT00185419 | Contraception | Phase 3 | Completed | - | China ... more >> ask Contact, China Collapse << |
| NCT01076582 | Oral Contraceptive | Phase 3 | Completed | - | - |
| NCT02855294 | Joint Stabilioty | Phase 4 | Completed | - | Egypt ... more >> Kasr Alainy medical school Cairo, Egypt, 12151 Collapse << |
| NCT00446199 | Vasomotor Symptoms ... more >> Hot Flashes Collapse << | Phase 3 | Completed | - | - |
| NCT00335257 | - | - | Completed | - | Germany ... more >> Center for Epidemiology and Health Research Berlin, Germany, 10115 Collapse << |
| NCT00446199 | - | - | Completed | - | - |
| NCT00335257 | - | - | Completed | - | - |
| NCT01360996 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | United States, Louisiana ... more >> Woman's Hospital Baton Rouge, Louisiana, United States, 70815 Collapse << |
| NCT01360996 | - | - | Completed | - | - |
| NCT02027337 | Polycystic Ovarian Syndrome ... more >> Hyperandrogenism Menstrual Irregularities Collapse << | Phase 4 | Unknown | October 2015 | Russian Federation ... more >> Tyumen State Medical Academy Recruiting Tyumen, Russian Federation, 3452 Contact: Tatyana N Khvoschina hvoschina-t@yandex.ru Principal Investigator: Tatyana N Khvoschina Collapse << |
| NCT01257984 | Contraception | Phase 3 | Completed | - | - |
| NCT00656981 | Acne Vulgaris | Phase 3 | Completed | - | United States, Alabama ... more >> Mobile, Alabama, United States, 36608 United States, Arizona Kingman, Arizona, United States, 86401 United States, California Encino, California, United States, 91436 San Diego, California, United States, 92103 San Diego, California, United States, 92123 Westlake Village, California, United States, 91361 United States, Colorado Castle Rock, Colorado, United States, 80108 Denver, Colorado, United States, 80246 United States, Florida Palm Springs, Florida, United States, 33461 St. Petersburg, Florida, United States, 33702 Venice, Florida, United States, 34285 United States, Idaho Boise, Idaho, United States, 83704 United States, Kansas Shawnee, Kansas, United States, 66203 United States, Kentucky Louisville, Kentucky, United States, 40217 United States, Minnesota Fridley, Minnesota, United States, 55432 United States, New York East Steauket, New York, United States, 11733 New York, New York, United States, 10022 United States, Oregon Medford, Oregon, United States, 97504 Portland, Oregon, United States, 97223-6683 United States, Texas Byran, Texas, United States, 77801 United States, Virginia Arlington, Virginia, United States, 22203 United States, Washington Seattle, Washington, United States, 98105 Collapse << |
| NCT00569244 | Primary Dysmenorrhea | Phase 3 | Completed | - | Germany ... more >> Heidelberg, Baden-Württemberg, Germany, 69120 Nürnberg, Bayern, Germany, 90491 Frankfurt, Hessen, Germany, 60439 Fulda, Hessen, Germany, 36037 Mühlheim, Hessen, Germany, 63165 Hannover, Niedersachsen, Germany, 30159 Hannover, Niedersachsen, Germany, 30459 Bernburg, Sachsen-Anhalt, Germany, 06406 Blankenburg, Sachsen-Anhalt, Germany, 38889 Burg, Sachsen-Anhalt, Germany, 39288 Jessen, Sachsen-Anhalt, Germany, 06917 Magdeburg, Sachsen-Anhalt, Germany, 39104 Magdeburg, Sachsen-Anhalt, Germany, 39126 Magdeburg, Sachsen-Anhalt, Germany, 39130 Leipzig, Sachsen, Germany, 04207 Leipzig, Sachsen, Germany, 04277 Leipzig, Sachsen, Germany, 04299 Wurzen, Sachsen, Germany, 04808 Gera, Thüringen, Germany, 07545 Kahla, Thüringen, Germany, 07768 Berlin, Germany, 10247 Berlin, Germany, 10409 Berlin, Germany, 12587 Berlin, Germany, 13086 United Kingdom Chesterfield, Derbyshire, United Kingdom, S40 4TF Newcastle Upon Tyne, Tyne and Wear, United Kingdom, NE4 5BE London, United Kingdom, W12 0HS Collapse << |
| NCT00651469 | Acne Vulgaris | Phase 3 | Completed | - | - |
| NCT03037944 | Adenomyosis | Early Phase 1 | Recruiting | December 1, 2018 | Egypt ... more >> Ain Shams University - Maternity Hospital Recruiting Cairo, Egypt Contact: Asmaa MA Mohamdy, Bachelors +201008340829 dr.asmaamamdoh@gmail.com Principal Investigator: Karim Ah Wahba, Asst.Prof Sub-Investigator: Karim Mo Labib, Lecturer Sub-Investigator: Mohamed Es Abbass, Lecturer Collapse << |
| NCT00102141 | Hypertension ... more >>Postmenopause Collapse << | Phase 3 | Completed | - | - |
| NCT02617537 | Dysmenorrhea | Phase 3 | Withdrawn | October 2017 | - |
| NCT01697111 | Endometriosis | Phase 3 | Completed | - | Japan ... more >> Anjo, Aichi, Japan, 446-8510 Ichinomiya, Aichi, Japan, 491-8551 Nagoya, Aichi, Japan, 451-8511 Nagoya, Aichi, Japan, 460-0011 Nagoya, Aichi, Japan, 464-0066 Matsudo, Chiba, Japan, 270-2267 Takasaki, Gunma, Japan, 370-0883 Itami, Hyogo, Japan, 664-8540 Kawanishi, Hyogo, Japan, 666-0125 Kobe, Hyogo, Japan, 654-0047 Kanazawa, Ishikawa, Japan, 920-8530 Kamakura, Kanagawa, Japan, 247-8533 Kawasaki, Kanagawa, Japan, 212-0016 Yokohama, Kanagawa, Japan, 230-0001 Yokohama, Kanagawa, Japan, 231-0023 Toyonaka, Osaka, Japan, 560-0022 Kitamoto, Saitama, Japan, 364-8501 Bunkyo, Tokyo, Japan, 112-0014 Chuo-ku, Tokyo, Japan, 104-0061 Fuchu, Tokyo, Japan, 183-0056 Hachioji, Tokyo, Japan, 192-0046 Itabashi, Tokyo, Japan, 175-0092 Machida, Tokyo, Japan, 194-0022 Minato, Tokyo, Japan, 105-0001 Minato, Tokyo, Japan, 107-0051 Nishitokyo, Tokyo, Japan, 188-0011 Fukui, Japan, 910-0845 Fukui, Japan, 910-8526 Gifu, Japan, 500-8717 Osaka, Japan, 530-0001 Osaka, Japan, 530-0013 Osaka, Japan, 542-0086 Collapse << |
| NCT00185484 | Contraception | Phase 3 | Completed | - | - |
| NCT00266032 | Contraception | Phase 3 | Completed | - | Canada, Quebec ... more >> Drummondville, Quebec, Canada, J2B 1H8 Montreal, Quebec, Canada, H1T 1P6 Pointe-Claire, Quebec, Canada, H9R 4S3 Shawinigan, Quebec, Canada, G9N 2H6 Ste-Foy, Quebec, Canada, G1V 4X7 Canada Quebec, Canada, G1S 2L6 Germany Ettlingen, Baden-Württemberg, Germany, 76275 Karlsruhe, Baden-Württemberg, Germany, 76199 Ansbach, Bayern, Germany, 91522 Krumbach, Bayern, Germany, 86381 Nürnberg, Bayern, Germany, 90491 Dietzenbach, Hessen, Germany, 63128 Frankfurt, Hessen, Germany, 60322 Frankfurt, Hessen, Germany, 60439 Frankfurt, Hessen, Germany, 65929 Frankfurt, Hessen, Germany, 65936 Mühlheim, Hessen, Germany, 63165 Bovenden, Niedersachsen, Germany, 37120 Hannover, Niedersachsen, Germany, 30459 Osnabrück, Niedersachsen, Germany, 49074 Bernburg, Sachsen-Anhalt, Germany, 06406 Jessen, Sachsen-Anhalt, Germany, 06917 Magdeburg, Sachsen-Anhalt, Germany, 39104 Magdeburg, Sachsen-Anhalt, Germany, 39126 Magdeburg, Sachsen-Anhalt, Germany, 39130 Wurzen, Sachsen, Germany, 04808 Gera, Thüringen, Germany, 07545 Jena, Thüringen, Germany, 07747 Kahla, Thüringen, Germany, 07768 Berlin, Germany, 10115 Berlin, Germany, 10247 Berlin, Germany, 12435 Berlin, Germany, 13187 Hamburg, Germany, 21073 Netherlands Groningen, Netherlands, 9713 GZ Collapse << |
| NCT00819312 | Contraception | Phase 3 | Completed | - | China, Guangdong ... more >> Guangzhou, Guangdong, China, 510120 Guangzhou, Guangdong, China China, Hubei Wuhan, Hubei, China, 430022 China, Jiangsu Nanjing, Jiangsu, China, 210006 Nanjing, Jiangsu, China, 210009 Yangzhou, Jiangsu, China, 225001 China, Liaoning Shenyang, Liaoning, China, 110004 China, Shandong Jinan, Shandong, China China, Shanxi Xi An, Shanxi, China, 710032 China, Sichuan Chengdu, Sichuan, China China Beijing, China, 100034 Beijing, China, 100044 Chongqing, China, 400010 Chongqing, China Shanghai, China, 200011 Shanghai, China, 200127 Hong Kong Hongkong, Hong Kong Collapse << |
| NCT00522873 | Postmenopause | Phase 3 | Completed | - | - |
| NCT00522873 | - | - | Completed | - | - |
| NCT00266032 | - | - | Completed | - | - |
| NCT01254292 | Contraception | Phase 3 | Completed | - | - |
| NCT00511199 | Contraception | Phase 3 | Completed | - | - |
| NCT01254292 | - | - | Completed | - | - |
| NCT01432028 | Postmenopause | Not Applicable | Completed | - | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre, Rio Grande do Sul, Brazil Collapse << |
| NCT00511199 | - | - | Completed | - | - |
| NCT00413062 | Contraception | Phase 3 | Completed | - | - |
| NCT01603745 | Polycystic Ovary Syndrome | Phase 1 | Unknown | October 2012 | Italy ... more >> Catholic university of Sacred Heart Recruiting Rome, Italy, 00168 Contact: Rosanna Apa, MD sandrina84@hotmail.com Principal Investigator: Rosanna Apa, MD Collapse << |
| NCT00511433 | - | - | Completed | - | - |
| NCT00511433 | Contraception | Phase 3 | Completed | - | - |
| NCT01195974 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Terminated(In a Phase II study... more >> in HIV-infected patients there were a number of seizures, although exact causality could not be assessed phase 1 activity was terminated.) Collapse << | - | United States, North Dakota ... more >> GSK Investigational Site Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00988910 | - | - | Completed | - | Italy ... more >> Many Locations, Italy Collapse << |
| NCT00633360 | Premenstrual Syndrome ... more >> Depression Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00633360 | - | - | Completed | - | - |
| NCT00413062 | - | - | Completed | - | - |
| NCT00640224 | - | - | Completed | - | - |
| NCT00631124 | Contraception | Phase 2 | Completed | - | Germany ... more >> Berlin, Germany, 10115 Berlin, Germany, 13353 Collapse << |
| NCT00653614 | Contraceptive, Oral, Hormonal | Phase 2 | Completed | - | Germany ... more >> Krumbach, Bayern, Germany, 86381 Nürnberg, Bayern, Germany, 90491 Dietzenbach, Hessen, Germany, 63128 Frankfurt, Hessen, Germany, 60322 Frankfurt, Hessen, Germany, 65936 Fulda, Hessen, Germany, 36037 Mühlheim, Hessen, Germany, 63165 Blankenburg, Sachsen-Anhalt, Germany, 38889 Burg, Sachsen-Anhalt, Germany, 39288 Jessen, Sachsen-Anhalt, Germany, 06917 Magdeburg, Sachsen-Anhalt, Germany, 39104 Magdeburg, Sachsen-Anhalt, Germany, 39126 Dippoldiswalde, Sachsen, Germany, 01744 Dresden, Sachsen, Germany, 01099 Dresden, Sachsen, Germany, 01169 Leipzig, Sachsen, Germany, 04207 Leipzig, Sachsen, Germany, 04299 Wurzen, Sachsen, Germany, 04808 Gera, Thüringen, Germany, 07545 Berlin, Germany, 10247 Berlin, Germany, 12587 Berlin, Germany, 13086 Berlin, Germany, 13507 Hamburg, Germany, 21073 Hamburg, Germany, 22159 Collapse << |
| NCT02874248 | Contraception | Phase 1 | Completed | - | - |
| NCT00640224 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Children's Hospital of Pittsburgh Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT00420342 | Postmenopause ... more >> Hypertension Pre-Hypertension Collapse << | Phase 2 | Completed | - | United States, California ... more >> Greenbrae, California, United States, 94904 San Diego, California, United States, 92108 United States, Florida Daytona Beach, Florida, United States, 32114 Miami, Florida, United States, 33136 United States, Michigan Paw Paw, Michigan, United States, 49079 United States, Nevada Las Vegas, Nevada, United States, 89122 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19114 United States, South Carolina Columbia, South Carolina, United States, 29201 United States, Virginia Arlington, Virginia, United States, 22203 Collapse << |
| NCT00420342 | - | - | Completed | - | - |
| NCT00302848 | - | - | Completed | - | Germany ... more >> Center for Epidemiology and Health Research Berlin, Germany, 10115 Collapse << |
| NCT03074045 | Contraception | Phase 3 | Active, not recruiting | September 1, 2022 | Russian Federation ... more >> Altai State Medical University Barnaul, Russian Federation, 656038 Scientific Center of family health & human reprod. problems Irkutsk, Russian Federation, 664003 Krasnoyarsk State Medical University Krasnoyarsk, Russian Federation, 660022 City Clinical Hospital #13 Moscow, Russian Federation, 115280 LLC Reafan Novosibirsk, Russian Federation, 630099 Medical Center "Avicenna" Novosibirsk, Russian Federation, 630099 Smolensk State Medical University Smolensk, Russian Federation, 214019 LLC Medical center PRIME ROSE St. Petersburg, Russian Federation, 197374 Regional perinatal center Yaroslavl, Russian Federation, 150042 Collapse << |
| NCT00302848 | - | - | Completed | - | - |
| NCT00468481 | Neural Tube Defects ... more >> Contraception Oral Contraceptives (OC) Collapse << | Phase 3 | Completed | - | United States, California ... more >> Orange County Clinical Trials Anaheim, California, United States, 92801 Medical Center for Clinical Research San Diego, California, United States, 92108 United States, Maryland SNBL Clinical Pharmacology Center, Inc. Baltimore, Maryland, United States, 21201 United States, New York Columbia University Medical Center New York, New York, United States, 10032 United States, North Carolina AAIPharma, Inc. Morrisville, North Carolina, United States, 27560 Lyndhurst Gynecologic Associates Winston-Salem, North Carolina, United States, 27103 United States, South Carolina Coastal Carolina Research Center Mt. Pleasant, South Carolina, United States, 29464 United States, Tennessee New Orleans Center for Clinical Research Knoxville, Tennessee, United States, 37920 United States, Washington NorthWest Kinetics Tacoma, Washington, United States, 98418 Collapse << |
| NCT03091595 | Prevention of Pregnancy | Phase 2 | Completed | - | Netherlands ... more >> Dinox BV Groningen, Netherlands, 9713 CZ Collapse << |
| NCT01258660 | Contraception | Phase 1 | Completed | - | Germany ... more >> Nuvisan GmbH Neu-Ulm, Bayern, Germany, 89231 Collapse << |
| NCT00468481 | - | - | Completed | - | - |
| NCT01892904 | Dysmenorrhea | Phase 3 | Completed | - | Japan ... more >> Matsudo, Chiba, Japan, 270-2267 Hachioji, Tokyo, Japan, 192-0046 Minato, Tokyo, Japan, 108-0071 Setagaya-ku, Tokyo, Japan, 157-0066 Suginami-ku, Tokyo, Japan, 167-0051 Toshima-ku, Tokyo, Japan, 171-0021 Osaka, Japan, 530-0013 Osaka, Japan, 542-0086 Collapse << |
| NCT01258660 | - | - | Completed | - | - |
| NCT00367276 | Healthy | Phase 3 | Completed | - | - |
| NCT01902264 | Contraception | Phase 3 | Completed | - | Mexico ... more >> Mexico, D.F., Distrito Federal, Mexico, CP 14050 Collapse << |
| NCT02957630 | Contraception ... more >> Liver Metabolism Hemostasis Parameter Collapse << | Phase 1 Phase 2 | Completed | - | Netherlands ... more >> Dinox BV Groningen, Netherlands, 9713 GZ Collapse << |
| NCT02757469 | Primary Ovarian Insufficiency ... more >> Menopause, Premature Collapse << | Not Applicable | Not yet recruiting | May 2021 | China, Beijing ... more >> Navy General Hospital Not yet recruiting Beijing, Beijing, China, 100048 Contact: Yunhai Chuai, Dr +86-18810892004 wangyh85@foxmail.com Principal Investigator: Wei Shang, Dr Principal Investigator: Lei Chen, Dr Principal Investigator: Yunhai Chuai, Dr Sub-Investigator: Mingming Shu, Dr Sub-Investigator: Ming Zhou, Dr Sub-Investigator: Mengnan Chen, Dr Sub-Investigator: Huiming Han, Dr Collapse << |
| NCT03512860 | Contraception | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Sciences Ruddington, Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT03124524 | Primary Dysmenorrhea | Phase 4 | Completed | - | - |
| NCT02384408 | Menopause | Phase 4 | Completed | - | Brazil ... more >> Vale Do Sapucai University (Univas) Pouso Alegre, MG, Brazil, 37550-000 Collapse << |
| NCT00915174 | Healthy | Phase 1 | Completed | - | Germany ... more >> AAIharma Deutschland GmbH & Co. KG Neu Ulm, Bavaria, Germany, 89231 Collapse << |
| NCT00722761 | Acne Vulgaris | Phase 3 | Completed | - | United States, Massachusetts ... more >> Clinical Unit for Research Trials in Skin Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00722800 | - | - | Terminated(Poor recruitment) | - | - |
| NCT01581814 | Polycystic Ovary Syndrome | Phase 3 | Completed | - | Italy ... more >> Policlinico A. Gemelli Rome, Italy, 00168 Collapse << |
| NCT00722761 | - | - | Completed | - | - |
| NCT01511822 | PCOS Polycyst... more >>ic Ovary Syndrome Collapse << | Phase 4 | Completed | - | Italy ... more >> Istituto di Patologia Ostetrica e Ginecologica Catania, Italy Collapse << |
| NCT01253174 | Contraception | Phase 1 | Completed | - | Germany ... more >> Scope International Hamburg, Germany, 22525 Collapse << |
| NCT00461305 | Dysmenorrhea | Phase 2 Phase 3 | Completed | - | Japan ... more >> Nagoya, Aichi, Japan, 460-0007 Nagoya, Aichi, Japan, 460-0011 Nagoya, Aichi, Japan, 464-0066 Maebashi, Gunma, Japan, 371-0024 Takasaki, Gunma, Japan, 370-0883 Kobe, Hyogo, Japan, 650-0021 Nishinomiya, Hyogo, Japan, 663-8204 Yamato, Kanagawa, Japan, 242-0007 Yokohama, Kanagawa, Japan, 231-0861 Sendai, Miyagi, Japan, 980-0021 Sendai, Miyagi, Japan, 981-0933 Sendai, Miyagi, Japan, 984-0042 Toyonaka, Osaka, Japan, 560-0022 Chuo-ku, Tokyo, Japan, 104-0061 Hachioji, Tokyo, Japan, 192-0046 Machida, Tokyo, Japan, 194-0022 Musashino, Tokyo, Japan, 180-0003 Ota-ku, Tokyo, Japan, 144-0052 Setagaya-ku, Tokyo, Japan, 156-0042 Setagaya-ku, Tokyo, Japan, 157-0066 Shibuya-ku, Tokyo, Japan, 150-0013 Shinagawa-ku, Tokyo, Japan, 140-0013 Suginami-ku, Tokyo, Japan, 167-0051 Toshima-ku, Tokyo, Japan, 171-0021 Osaka, Japan, 530-0013 Osaka, Japan, 534-0014 Collapse << |
| NCT00356447 | Vasomotor Symptoms | Phase 3 | Completed | - | China, Hubei ... more >> Wuhan, Hubei, China, 430032 China, Jiangsu Nanjing, Jiangsu, China, 210029 China, Shandong Jinan, Shandong, China, 250012 China Beijing, China, 100020 Beijing, China, 100034 Beijing, China, 100083 Beijing, China, 100730 Chongqing, China, 400010 Shanghai, China, 200011 Collapse << |
| NCT00722800 | Hidradenitis Suppurativa | Phase 2 | Terminated(Poor recruitment) | - | United States, Massachusetts ... more >> Clinical Unit for Research Trials in Skin Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01253187 | - | - | Completed | - | - |
| NCT00461305 | - | - | Completed | - | - |
| NCT01253174 | - | - | Completed | - | - |
| NCT01253187 | Contraception | Phase 1 | Completed | - | Netherlands ... more >> Dinox B.V. Groningen, Netherlands, 9713GZ Collapse << |
| NCT00511797 | Dysmenorrhea | Phase 2 Phase 3 | Completed | - | Japan ... more >> Maebashi, Gunma, Japan, 371-0024 Kobe, Hyogo, Japan, 650-0021 Nishinomiya, Hyogo, Japan, 663-8204 Yokohama, Kanagawa, Japan, 231-0861 Sendai, Miyagi, Japan, 981-0933 Chuo-ku, Tokyo, Japan, 104-0061 Hachioji, Tokyo, Japan, 192-0046 Setagaya-ku, Tokyo, Japan, 156-0042 Setagaya-ku, Tokyo, Japan, 157-0066 Toshima-ku, Tokyo, Japan, 171-0021 Kyoto, Japan, 612-0064 Osaka, Japan, 530-0013 Collapse << |
| NCT00511797 | - | - | Completed | - | - |
| NCT00745901 | Contraception | Phase 4 | Completed | - | - |
| NCT00818519 | - | - | Completed | - | - |
| NCT01608698 | Body Weight Changes | Phase 4 | Unknown | February 2016 | Thailand ... more >> Family Planning Unit, Department of Obstetric and Gynecology, Faculty of Medicine Siriraj Hospital, Mahidol University Bangkok, Thailand, 10700 Collapse << |
| NCT02852681 | Contraception | Phase 1 | Completed | - | - |
| NCT00818519 | Acne Vulgaris | Phase 3 | Completed | - | China, Guangdong ... more >> Guangzhou, Guangdong, China, 510630 China, Hunan Changsha, Hunan, China, 410011 China, Jiangsu Nanjing, Jiangsu, China, 210042 China, Sichuan Chengdu, Sichuan, China, 610041 China Beijing, China, 100032 Beijing, China, 100853 Shanghai, China, 200043 Collapse << |
| NCT01109979 | - | - | Completed | - | - |
| NCT00745901 | - | - | Completed | - | - |
| NCT02269241 | Contraception | Phase 3 | Completed | - | - |
| NCT01590849 | Contraceptive Affecting the Au... more >>tonomic Nervous System Contraceptive Affecting Blood Pressure Collapse << | Phase 3 | Unknown | - | Brazil ... more >> Marcelo Gil Nisenbaum Sao Paulo, São Paulo, Brazil, 05403-000 Collapse << |
| NCT02342093 | Blood Pressure ... more >> Contraception Collapse << | Phase 4 | Completed | - | Brazil ... more >> University of Sao Paulo Ribeirão Preto, SP, Brazil, 14049-900 Collapse << |
| NCT01109979 | Cardiovascular Diseases | Phase 4 | Completed | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01519401 | Polycystic Ovary Syndrome ... more >> Estro-progestin Drugs Collapse << | Not Applicable | Completed | - | Italy ... more >> Catholic University of Sacred Heart Rome, Italy, 00168 Collapse << |
| NCT01182207 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01482338 | Premenstrual Syndrome | Phase 4 | Completed | - | Thailand ... more >> Family Unit, King Chulalongkorn Memorial Hospital Bangkok, Thailand, 10330 Collapse << |
| NCT02885454 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT01182194 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01182207 | - | - | Completed | - | - |
| NCT01459445 | Polycystic Ovary Syndrome ... more >> Endothelial Dysfunction Collapse << | Not Applicable | Unknown | January 2012 | Romania ... more >> Clinic of Endocrinology Cluj-Napoca, Cluj, Romania, 400349 Collapse << |
| NCT03126097 | Healthy | Phase 1 | Terminated(Study stopping rule... more >>s were met.) Collapse << | - | United States, Arizona ... more >> Celerion Tempe, Arizona, United States, 85283 Collapse << |
| NCT03111511 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Clinical Pharmacology Unit Merksem, Belgium, 2170 Collapse << |
| NCT02817828 | Contraception | Phase 3 | Completed | - | Belgium ... more >> Hopital Saint-Pierre Bruxelles, Belgium, 1000 Collapse << |
| NCT02817841 | Contraception | Phase 3 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Obstetrics and Gynecology Philadelphia, Pennsylvania, United States, 19107 Canada Clinique de Santé des Femmes Quebec, Canada, G1S 2L6 Collapse << |
| NCT01182194 | - | - | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
