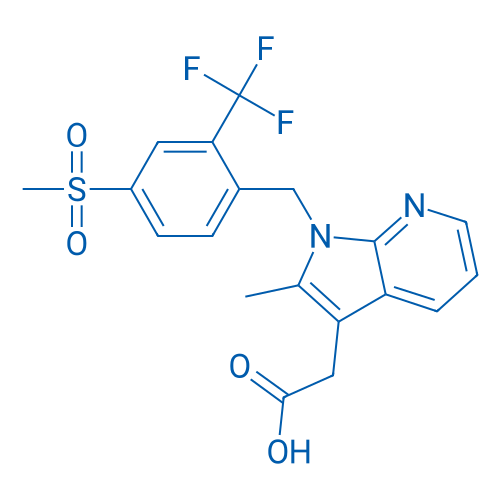
CAS No.: 872365-14-5
Fevipiprant Catalog No. CSN18830
Synonyms: QAW039;NVP-QAW039;NVP-QAW-039 QAW-039
Fevipiprant is a selective, potent, reversible competitive CRTh2 antagonist with an in vitro dissociation constant KD value of 1.1nM at the CRTh2 receptor and an IC50 value of 0.44 nM for inhibition of PGD2-induced eosinophil shape change in human whole blood.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01804400 Allergic Rhinitis Phase 2 Completed - Germany ... more >> Novartis Investigative Site Hannover, Germany, 30625 Collapse << NCT01836471 Asthma Phase 2 Completed - - NCT01253603 Persistent Asthma Phase 2 Completed - - - 更多
- 参考文献
- [1] Sandham DA, Barker L, et al. Discovery of Fevipiprant (NVP-QAW039), a Potent and Selective DP2 Receptor Antagonist for Treatment of Asthma. ACS Med Chem Lett. 2017 Apr 25;8(5):582-586.
- [2] Sykes DA, Bradley ME, et al. Fevipiprant (QAW039), a Slowly Dissociating CRTh2 Antagonist with the Potential for Improved Clinical Efficacy. Mol Pharmacol. 2016 May;89(5):593-605.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 872365-14-5 | 储存条件 |
|
|||
| 分子式 | C19H17F3N2O4S | 运输 | 蓝冰 | |||
| 分子量 | 426.41 | 别名 | QAW039;NVP-QAW039;NVP-QAW-039 QAW-039 | |||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
