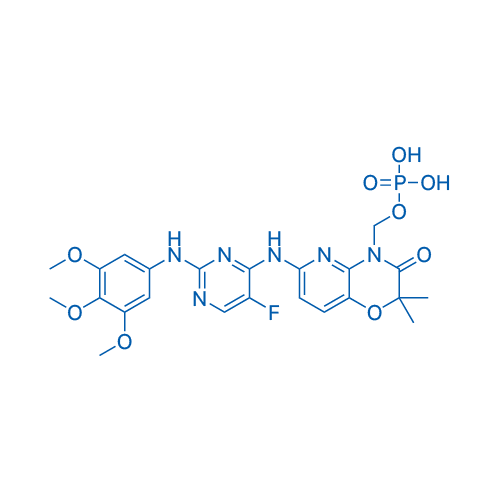
CAS No.: 901119-35-5
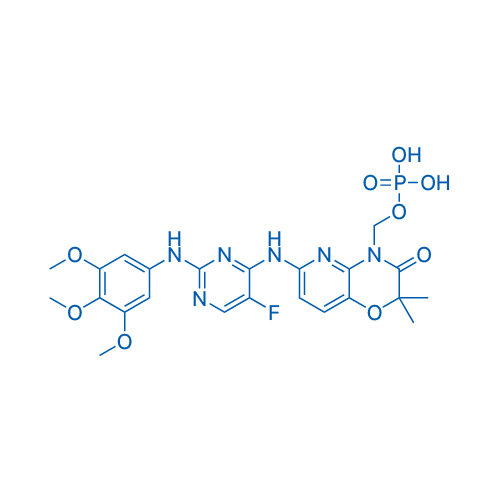
Fostamatinib/福他替尼 Catalog No. CSN15818
Synonyms: R788;福他替尼
Fostamatinib, a prodrug of the active metabolite R406, is a potent Syk inhibitor with IC50 of 41 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00805467 - - Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << - - NCT00665925 Rheumatoid Arthritis Phase 2 Completed - - NCT00752999 Systemic Lupus Erythematosus Phase 2 Withdrawn(Study was withdrawn ... more >>for business reasons before study start.) Collapse << March 2010 - - 更多
- 参考文献
- [1] Chen L, Monti S, et al. SYK-dependent tonic B-cell receptor signaling is a rational treatment target in diffuse large B-cell lymphoma. Blood. 2008 Feb 15;111(4):2230-7.
- [2] Braselmann S, Taylor V, et al. R406, an orally available spleen tyrosine kinase inhibitor blocks fc receptor signaling and reduces immune complex-mediated inflammation. J Pharmacol Exp Ther. 2006 Dec;319(3):998-1008.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 901119-35-5 | 储存条件 |
|
|
| 分子式 | C23H26FN6O9P | 运输 | 蓝冰 | |
| 分子量 | 580.46 | 别名 | R788;福他替尼 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| human Ramos cells | - | Function assay | - | Inhibition of SYK in human Ramos cells, IC50=0.267 μM | 23350847 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00805467 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT00665925 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT00752999 | Systemic Lupus Erythematosus | Phase 2 | Withdrawn(Study was withdrawn ... more >>for business reasons before study start.) Collapse << | March 2010 | - |
| NCT00665626 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT00805467 | Rheumatoid Arthritis | Phase 2 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT01197781 | Drug Drug Interactions ... more >> Healthy Volunteers Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Recruiting Site Overland Park, Kansas, United States Collapse << |
| NCT00665925 | - | - | Completed | - | - |
| NCT01167868 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Research Site Glendale, California, United States Collapse << |
| NCT01682408 | Pharmacokinetics | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT00665626 | - | - | Completed | - | - |
| NCT01645085 | Healthy Volunteers | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT00706342 | Purpura, Thrombocytopenic, Idi... more >>opathic Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Research Site New York, New York, United States, 10065 United States, Ohio Research Site Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02092961 | Rheumatoid Arthritis | Phase 2 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | United States, Arizona ... more >> Research Site Glendale, Arizona, United States Research Site Paradise Valley, Arizona, United States Research Site Phoenix, Arizona, United States United States, New York Research Site Brooklyn, New York, United States United States, Tennessee Research Site Jackson, Tennessee, United States United States, Texas Research Site Austin, Texas, United States Bulgaria Research Site Pleven, Bulgaria Canada, Ontario Research Site Mississauga, Ontario, Canada Czech Republic Research Site Prague, Czech Republic Germany Research Site Hamburg, Germany Research Site Munich, Germany Hungary Research Site Balatonfüred, Hungary Research Site Budapest, Hungary Netherlands Research Site Amsterdam, Netherlands Poland Research Site Warsaw, Poland Russian Federation Research Site Yaroslavl, Russian Federation South Africa Research Site Durban, South Africa Research Site Pretoria, South Africa Research Site Stellenbosch, South Africa United Kingdom Research Site Manchester, United Kingdom Research Site Oxford, United Kingdom Collapse << |
| NCT02092961 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT00706342 | - | - | Completed | - | - |
| NCT01264770 | Rheumatoid Arthritis | Phase 2 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT01563978 | - | - | Completed | - | - |
| NCT00923481 | Head and Neck Neoplasms ... more >> Pheochromocytoma Colorectal Neoplasms Carcinoma, Non-Small-Cell Lung Carcinoma, Renal Cell Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00923481 | - | - | Completed | - | - |
| NCT00798096 | - | - | Completed | - | - |
| NCT01725230 | Rheumatoid Arthritis | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT00798096 | T Cell Lymphoma | Phase 2 | Completed | - | United States, California ... more >> Research Site San Francisco, California, United States, 94143 Research Site Stanford, California, United States, 94305 United States, Georgia Research Site Atlanta, Georgia, United States, 30322 United States, Massachusetts Research Site Boston, Massachusetts, United States, 02115 United States, Minnesota Research Site Rochester, Minnesota, United States, 55905 United States, Nebraska Research Site Omaha, Nebraska, United States, 68198 United States, New York Research Site New York, New York, United States, 10021 United States, Texas Research Site Houston, Texas, United States, 77030 Canada, British Columbia Research Site Vancouver, British Columbia, Canada, V5Z 4E6 Canada, Ontario Research Site Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01563978 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT02076399 | Immune Thrombocytopenic Purpur... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT01569074 | Rheumatoid Arthritis | Phase 2 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | Hong Kong ... more >> Research Site New Territories, HK, Hong Kong Research Site Hong Kong, Hong Kong Japan Research Site Matsuyama, Ehime, Japan Research Site Fukuoka-shi, Fukuoka, Japan Research Site Kitakyushu-shi, Fukuoka, Japan Research Site Kurume, Fukuoka, Japan Research Site Sapporo, Hokkaido, Japan Research Site Kato-shi, Hyogo, Japan Research Site Kasama-shi, Ibaraki, Japan Research Site Kumamoto-shi, Kumamoto, Japan Research Site Sendai, Miyagi, Japan Research Site Isahaya, Nagasaki, Japan Research Site Nagasaki-shi, Nagasaki, Japan Research Site Omura-shi, Nagasaki, Japan Research Site Sasebo-shi, Nagasaki, Japan Research Site Shibata, Niigata, Japan Research Site Okayama-shi, Okayama, Japan Research Site Tomigusuku-shi, Okinawa, Japan Research Site Matsue-shi, Shimane, Japan Research Site Hamamatsu-shi, Shizuoka, Japan Research Site Itabashi, Tokyo, Japan Research Site Shinjuku, Tokyo, Japan Research Site Nagasaki, Japan Korea, Republic of Research Site Anyang-si, Gyeonggi-do, Korea, Republic of Research Site Gwangju, Korea, Republic of Research Site Incheon, Korea, Republic of Research Site Seoul, Korea, Republic of Taiwan Research Site Chiayi, Taiwan Research Site Kaohsiung, Taiwan Research Site Taichung, Taiwan Research Site Taipei, Taiwan Thailand Research Site Bangkok, Thailand Research Site Singapore, Thailand Vietnam Research Site Hanoi, Vietnam Research Site Ho Chi Minh, Vietnam Collapse << |
| NCT01245790 | Rheumatoid Arthritis ... more >> Renal Impairment Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Orlando, Florida, United States Collapse << |
| NCT01264770 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT01242514 | Rheumatoid Arthritis | Phase 3 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | January 2015 | - |
| NCT01197755 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01640054 | Rheumatoid Arthritis | Phase 2 | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | Hong Kong ... more >> Research Site Hongkong, Shatin, Hong Kong Research Site Hong Kong, Hong Kong Japan Research Site Fukuoka-shi, Fukuoka, Japan Research Site Kitakyushu-shi, Fukuoka, Japan Research Site Sapporo-shi, Hokkaido, Japan Research Site Kato-shi, Hyogo, Japan Research Site Sendai-shi, Miyagi, Japan Research Site Nagasaki-shi, Nagasaki, Japan Research Site Omura-shi, Nagasaki, Japan Research Site Sasebo-shi, Nagasaki, Japan Research Site Okayama-shi, Okayama, Japan Research Site Tomigusuku-shi, Okinawa, Japan Research Site Hamamatsu-shi, Shizuoka, Japan Research Site Itabashi-ku, Tokyo, Japan Research Site Shinjuku-ku, Tokyo, Japan Korea, Republic of Research Site Anyang-si, Gyeonggi-do, Korea, Republic of Research Site Gwangju, Korea, Republic of Research Site Incheon, Korea, Republic of Research Site Seoul, Korea, Republic of Taiwan Research Site Kaohsiung, Taiwan Research Site Taichung, Taiwan Research Site Taipei, Taiwan Thailand Research Site Bangkok, Thailand Research Site Singapore, Thailand Vietnam Research Site Hanoi, Vietnam Research Site Ho Chi Minh, Vietnam Collapse << |
| NCT01242514 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT01197755 | - | - | Completed | - | - |
| NCT01569074 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT01640054 | - | - | Terminated(AZ decision to disc... more >>ontinue fostamatinib development in RA; rights to fostamatinib returned to Rigel Pharmaceuticals.) Collapse << | - | - |
| NCT00446095 | - | - | Completed | - | - |
| NCT01309854 | Rheumatoid Arthritis ... more >> Healthy Volunteers Pharmacokinetics Pioglitazone Drug-drug Interaction Amount of Pioglitazone in Blood Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01197534 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT00446095 | Lymphoma | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States, 90095 Research Site Stanford, California, United States, 94305 United States, Georgia Research Site Atlanta, Georgia, United States, 30322 United States, Illinois Research Site Chicago, Illinois, United States, 60612 United States, Indiana Research Site Indianapolis, Indiana, United States, 46202 United States, Massachusetts Research Site Boston, Massachusetts, United States, 02115 United States, Minnesota Research Site Rochester, Minnesota, United States, 59905 United States, Nebraska Research Site Omaha, Nebraska, United States, 68198 United States, New York Research Site New York, New York, United States, 10065 Research Site Rochester, New York, United States, 14642 United States, Ohio Research Site Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01197521 | - | - | Completed | - | - |
| NCT01222455 | Hepatic Impairment ... more >> Healthy Volunteers Pharmacokinetics Amount of R406 in Blood Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Orlando, Florida, United States Collapse << |
| NCT01197534 | - | - | Completed | - | - |
| NCT03363334 | - | - | - | - | - |
| NCT01197521 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01208155 | Bioavailability ... more >> Pharmacokinetics Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01387308 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT02112838 | IGA Nephropathy | Phase 2 | Active, not recruiting | December 2018 | United States, California ... more >> Stanford University Medical Palo Alto, California, United States, 94304 United States, Georgia Nephrology Associates PC, University Hospital, Professional Center 1 Augusta, Georgia, United States, 30901 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Tennessee Southeast Renal Research Institute Chattanooga, Tennessee, United States, 37408 Austria Medical University of Graz Graz, Steiermark, Austria, 8036 Medical University Vienna, Nephrology Vienna, Austria, A-1090 Germany Medical University of Heidelberg Heidelberg, Baden-Wurtemberberg, Germany, 69120 Klinikum der Universität München Munich, Bayern, Germany, 80336 Universitätsklinikum Carl Gustav Carus an der Technischen Universität Dresden Dresden, Sachsen, Germany, 1307 Medical University of Jena Jena, Thueringen, Germany, 07747 Hong Kong Prince of Wales Hospital Hong Kong, Sha Tin, Hong Kong Queen Mary Hospital Hong Kong, Hong Kong Taiwan China Medical University Hospital Taichung, Taiwan, 40447 School of Medicine, Chang Gung University, Chang Gung Memorial Hospital Taoyuan, Taiwan, 333 United Kingdom Addenbrookes Hospital Cambridge, United Kingdom, CB2 0QQ Cardiff University Cardiff, United Kingdom, CF14 4XN Leicester General Hospital Leicester, United Kingdom, LE5 4PW Royal Free Hospital London, United Kingdom, NW3 2PF Hammersmith Hospital London, United Kingdom, W12 0NN Freeman Hospital Newcastle upon Tyne, United Kingdom, NE7 7DN Collapse << |
| NCT01355354 | Healthy Volunteers ... more >> Rheumatoid Arthritis Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT02077192 | Immune Thrombocytopenic Purpur... more >>a Collapse << | Phase 3 | Active, not recruiting | March 2020 | - |
| NCT01336218 | Rheumatoid Arthritis ... more >> Healthy Volunteers Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> Research Site Overland Park, Kansas, United States Collapse << |
| NCT01311622 | Rheumatoid Arthritis ... more >> Healthy Subjects Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT01276262 | Scientific Terminology Rheumat... more >>oid Arthritis, Healthy Female Volunteers, Pharmacokinetics, Oral Contraceptive, Drug-drug Interaction Laymen Terminology Level of Oral Contraceptive in Blood, Oral Contraceptive, Rheumatoid Arthritis, Drug -Drug Interaction Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT02076412 | Immune Thrombocytopenic Purpur... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT01499303 | - | - | Completed | - | - |
| NCT01598571 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Ruddington, Nottingham, United Kingdom Collapse << |
| NCT01608542 | Healthy Japanese Volunteers | Phase 1 | Completed | - | United States, California ... more >> Glendale, California, United States United States, Maryland Baltimore, Maryland, United States Collapse << |
| NCT02612558 | Warm Antibody Autoimmune Hemol... more >>ytic Anemia Collapse << | Phase 2 | Active, not recruiting | June 2019 | - |
| NCT03764618 | Warm Antibody Autoimmune Hemol... more >>ytic Anemia Collapse << | Phase 3 | Not yet recruiting | September 2021 | - |
| NCT03246074 | Ovarian Cancer | Phase 1 | Recruiting | October 1, 2021 | United States, Maryland ... more >> Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Recruiting Baltimore, Maryland, United States, 21287 Contact: Hopkins Breast Trials HopkinsBreastTrials@jhmi.edu Contact: Kimmel Cancer Center Clinical Research Office 410-955-8866 jhcccro@jhmi.edu Principal Investigator: Stephanie Gaillard, MD Collapse << |
| NCT01499303 | Diffuse Large B-Cell Lymphoma | Phase 2 | Completed | - | - |
| NCT02611063 | Hematological Malignancies | Phase 1 | Recruiting | January 2021 | United States, North Carolina ... more >> Duke University of Medicine Recruiting Durham, North Carolina, United States, 27710 Contact: Stefanie Sarantopoulos, MD, PhD 919-668-4383 stefanie.sarantopoulso@duke.edu Contact: Krista Rowe, RN 919-684-7115 krista.rowe@duke.edu Collapse << |
| NCT02433236 | IGA Nephropathy | Phase 2 | Withdrawn(Study withdrawn prio... more >>r to enrollment of first subject) Collapse << | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
