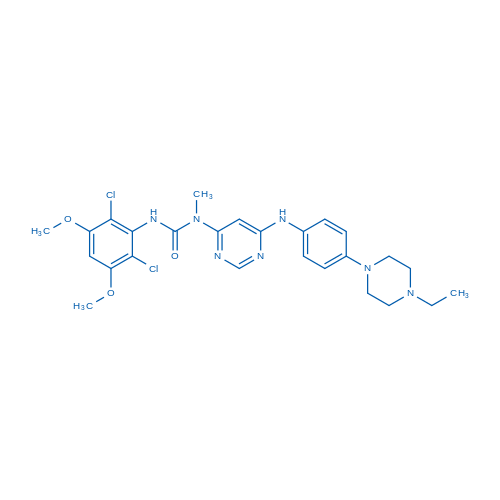
CAS No.: 872511-34-7
Infigratinib Catalog No. CSN16144
Synonyms: BGJ-398;NVP-BGJ398
BGJ-398 is a selective, pan-specific FGFR inhibitor with IC50 of 0.9, 1.4, and 1 nM for FGFR1, FGFR2, and FGFR3, respectively and > 40-fold selective for FGFR versus FGFR4 and VEGFR2, and little activity to Abl, Fyn, Kit, Lck, Lyn and Yes.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 FGFR1
IC50:0.9nMFGFR2
IC50:1.4nMFGFR3
IC50:1nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Rat[1] (p.o.): 4.25 mg/kg, 8.51 mg/kg; Mice: 5 mg/kg[1] (i.v.), 20 mg/kg[1] (p.o.), 50 mg/kg[2] (p.o.)
给药途径 i.v., p.o.
动物药代数据 Animal Nude mice[1] Rats[1] Dose 5 mg/kg (i.v.)
20 mg/kg (p.o.)5 mg/kg (i.v.)
10 mg/kg (p.o.)Administration i.v.
p.o.i.v.
p.o.Cmax 0.86 μM (i.v.)
0.42 μM (p.o.)0.97 μM (i.v.)
0.26 μM (p.o.)T1/2 9.3 h (i.v.)
4.3 h (p.o.)4.5 h (i.v.)
4.8 h (p.o.)F 32% (p.o.) 51% (p.o.) Tmax 1 h (p.o.) 4 h (p.o.) Vss 26 L/kg (i.v.) Vss 9.6 L/kg (i.v.) CL 3.3 L/h/kg (i.v.) 3.6 L/h/kg (i.v.) AUC(normalized to 1 mg/kg) 0.6 μM·h (i.v.)
0.18 μM·h (p.o.)0.49 μM·h (i.v.)
0.25 μM·h (p.o.)- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01820364 Melanoma Phase 2 Terminated(Study was withdrawn... more >> due to scientific and business considerations.) Collapse << - United States, Tennessee ... more >> Sarah Cannon Research Institute Onc Dept Nashville, Tennessee, United States, 37203 Australia, Victoria Novartis Investigative Site East Melbourne, Victoria, Australia, 3002 Canada, Alberta Novartis Investigative Site Edmonton, Alberta, Canada, T6G 1Z2 Germany Novartis Investigative Site Heidelberg, Germany, 69120 Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Switzerland Novartis Investigative Site Zuerich, Switzerland, 8091 Collapse << NCT01820364 - - Terminated(Study was withdrawn... more >> due to scientific and business considerations.) Collapse << - - NCT02159066 Melanoma Phase 2 Active, not recruiting March 30, 2019 United States, Arizona ... more >> Mayo Clinic - Arizona onc Dept Scottsdale, Arizona, United States United States, California University of California at Los Angeles Onc Dept Los Angeles, California, United States, 90095 United States, Massachusetts Massachusetts General Hospital Dept of Onc. Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan Kettering Cancer Center Dept Oncology New York, New York, United States, 90033 United States, Oregon Oregon Health & Science University Onc. Dept Portland, Oregon, United States, 97239 United States, Tennessee Sarah Cannon Research Institute Onc. Dept Nashville, Tennessee, United States, 37203 Australia, Victoria Array BioPharma Investigative Site East Melbourne, Victoria, Australia, 3002 Canada, Ontario Array BioPharma Investigative Site Toronto, Ontario, Canada, M5G 2M9 Canada, Quebec Array BioPharma Investigative Site Montreal, Quebec, Canada, H3T 1E2 Germany Array BioPharma Investigative Site Heidelberg, Germany, 69120 Array BioPharma Investigative Site Köln, Germany, 50937 Array BioPharma Investigative Site Muenchen, Germany, 80336 Array BioPharma Investigative Site Würzburg, Germany, 97080 Italy Array BioPharma Investigative Site Napoli, Italy, 80131 Netherlands Array BioPharma Investigative Site Amsterdam, Netherlands, 1066 CX Spain Array BioPharma Investigative Site Barcelona, Catalunya, Spain, 08035 Switzerland Array BioPharma Investigative Site Zuerich, Switzerland, 8091 United Kingdom Array BioPharma Investigative Site Oxford, United Kingdom, OX3 7LJ Collapse << - 更多
- 参考文献
- [1] Guagnano V, Furet P, et al. Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem. 2011 Oct 27;54(20):7066-83.
- [2] Xiao L, Du E, et al. Inhibition of FGFR Signaling Partially Rescues Hypophosphatemic Rickets in HMWFGF2 Tg Male Mice. Endocrinology. 2017 Oct 1;158(10):3629-3646.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 872511-34-7 | 储存条件 |
|
|||||||||||
| 分子式 | C26H31Cl2N7O3 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 560.48 | 别名 | BGJ-398;NVP-BGJ398 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
