| NCT00901979 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01594983 | Non Familial Chylocmicronemia ... more >>Syndrome (Non-FCS) Collapse << | Phase 2 | Completed | - | - |
| NCT01387958 | Hepatitis C | Phase 2 | Terminated(Terminated early du... more >>e to futility) Collapse << | - | New Zealand
... more >> Novartis Investigative Site
Auckland, New Zealand
Novartis Investigative Site
Christchurch, New Zealand Collapse << |
| NCT01146522 | Hyperlipoproteinemia | Phase 1
Phase 2 | Completed | - | Canada
... more >> Novartis Investigative Site
Quebec, Canada Collapse << |
| NCT01811472 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Collapse << | Phase 2 | Completed | - | United States, Alabama
... more >>
Novartis Investigative Site
Mobile, Alabama, United States, 36608
United States, California
Novartis Investigative Site
San Diego, California, United States, 92114
United States, Florida
Novartis Investigative Site
Gainesville, Florida, United States, 32610-0277
Novartis Investigative Site
Miami, Florida, United States, 33126
Novartis Investigative Site
Tamarac, Florida, United States, 33319
United States, Hawaii
Novartis Investigative Site
Honolulu, Hawaii, United States, 96814
United States, Kentucky
Novartis Investigative Site
Louisville, Kentucky, United States, 40213
United States, Mississippi
Novartis Investigative Site
Tupelo, Mississippi, United States, 38801
United States, Texas
Novartis Investigative Site
Houston, Texas, United States, 77030
Novartis Investigative Site
Plano, Texas, United States, 75093
United States, Virginia
Novartis Investigative Site
Richmond, Virginia, United States, 23298 Collapse << |
| NCT01589237 | Familial Chylomicronemia Syndr... more >>ome (FCS) (HLP Type I) Collapse << | Phase 3 | Terminated(Interim analysis su... more >>ggested that size of benefit anticipated from continued participation of patients in Part B no longer supported trial extension beyond Part A) Collapse << | - | United States, Washington
... more >>
Novartis Investigative Site
Seatlle, Washington, United States, 98104
Canada, Quebec
Novartis Investigative Site
Chicoutimi, Quebec, Canada, G7H 7P2
Novartis Investigative Site
Ste-Foy, Quebec, Canada, G1V4M6
Canada
Novartis Investigative Site
Ouest-Montreal, Canada, H2W1R7
France
Novartis Investigative Site
Nantes, France, 44093
Novartis Investigative Site
Paris Cedex 13, France, 75651
Germany
Novartis Investigative Site
Hamburg, Germany, 20246
Netherlands
Novartis Investigative Site
Meibergdreef 9, Netherlands, 1105 AZ
South Africa
Novartis Investigative Site
Cape Town, South Africa, 7925
United Kingdom
Novartis Investigative Site
Manchester, United Kingdom, M13 9NT Collapse << |
| NCT01811472 | - | - | Completed | - | - |
| NCT01474434 | Coronary Artery Disease
... more >> Hypertriglyceridemia Collapse << | Phase 2 | Terminated(The study was termi... more >>nated based on interim analysis. See detailed description.) Collapse << | - | United States, California
... more >>
Novartis Investigative Site
Pasadena, California, United States, 91105 Collapse << |
| NCT01594957 | Hepatic Impairment | Phase 1 | Completed | - | United States, Florida
... more >>
Novartis Investigative Site
Miami, Florida, United States, 33136
Novartis Investigative Site
Orlando, Florida, United States, 32809 Collapse << |
| NCT01589237 | - | - | Terminated(Interim analysis su... more >>ggested that size of benefit anticipated from continued participation of patients in Part B no longer supported trial extension beyond Part A) Collapse << | - | - |
| NCT01514461 | Familial Chylomicronemia Syndr... more >>ome (FCS) Collapse << | Phase 3 | Completed | - | United States, Washington
... more >>
Novartis Investigative Site
Seatlle, Washington, United States, 98104
Canada, Quebec
Novartis Investigative Site
Chicoutimi, Quebec, Canada, G7H 7P2
Novartis Investigative Site
Ste-Foy, Quebec, Canada, G1V4M6
Canada
Novartis Investigative Site
Ouest-Montreal, Canada, H2W1R7
France
Novartis Investigative Site
Bron, France, 69677
Novartis Investigative Site
Nantes, France, 44093
Novartis Investigative Site
Paris Cedex 13, France, 75651
Germany
Novartis Investigative Site
Hamburg, Germany, 20246
Novartis Investigative Site
Köln, Germany, 50937
Netherlands
Novartis Investigative Site
Meibergdreef 9, Netherlands, 1105 AZ
South Africa
Novartis Investigative Site
Cape Town, South Africa, 7925
Spain
Novartis Investigative Site
Malaga, Andalucia, Spain, 29010
Novartis Investigative Site
Sevilla, Andalucia, Spain, 41013
United Kingdom
Novartis Investigative Site
Manchester, United Kingdom, M13 9NT Collapse << |
| NCT01474434 | - | - | Terminated(The study was termi... more >>nated based on interim analysis. See detailed description.) Collapse << | - | - |
| NCT01558323 | Renal Impairment | Phase 1 | Completed | - | United States, Florida
... more >>
Novartis Investigative Site
Orlando, Florida, United States, 32809
United States, Tennessee
Novartis Investigative Site
Knoxville, Tennessee, United States, 37920 Collapse << |
| NCT01514461 | - | - | Completed | - | - |
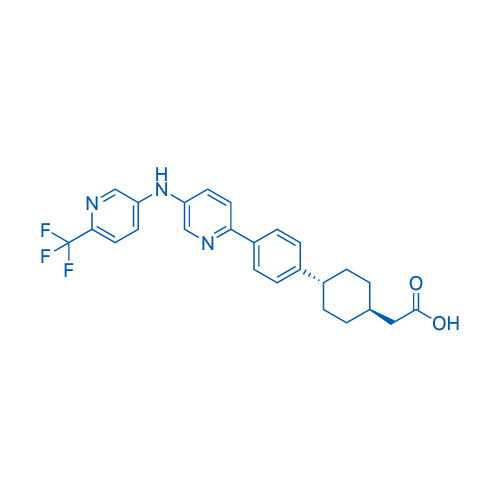

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
