| NCT02371967 | - | - | Active, not recruiting | August 29, 2020 | Sweden
... more >> SU/Sahlgrenska, Hudkliniken
Göteborg, Sweden, 41345
Skånes Universitetssjukhus; Hudkliniken
Lund, Sweden, 222 41
Radiumhemmet
Solna, Sweden, 171 64
Norrlands universitetssjukhus; Onkologkliniken
Umeå, Sweden Collapse << |
| NCT02129101 | Chronic Myelomonocytic Leukemi... more >>a
de Novo Myelodysplastic Syndrome
Essential Thrombocythemia
Myelodysplastic Syndrome
Myelodysplastic/Myeloproliferative Neoplasm
Polycythemia Vera
Previously Treated Myelodysplastic Syndrome
Primary Myelofibrosis
Recurrent Adult Acute Myeloid Leukemia
Recurrent Childhood Acute Myeloid Leukemia
Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Active, not recruiting | October 2018 | United States, Minnesota
... more >>
Mayo Clinic
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03434262 | Anaplastic Astrocytoma
... more >> Anaplastic Ependymoma
Anaplastic Ganglioglioma
Anaplastic Meningioma
Anaplastic Oligodendroglioma
Pleomorphic Xanthoastrocytoma, Anaplastic
Atypical Teratoid/Rhabdoid Tumor
Brain Cancer
Brain Tumor
Central Nervous System Neoplasms
Choroid Plexus Carcinoma
CNS Embryonal Tumor With Rhabdoid Features
Ganglioneuroblastoma of Central Nervous System
CNS Tumor
Embryonal Tumor of CNS
Ependymoma
Glioblastoma
Glioma
Glioma, Malignant
Medulloblastoma
Medulloblastoma; Unspecified Site
Medulloepithelioma
Neuroepithelial Tumor
Neoplasms
Neoplasms, Neuroepithelial
Papillary Tumor of the Pineal Region (High-grade Only)
Pediatric Brain Tumor
Pineal Parenchymal Tumor of Intermediate Differentiation (High-grade Only)
Pineoblastoma
Primitive Neuroectodermal Tumor
Recurrent Medulloblastoma
Refractory Brain Tumor
Neuroblastoma. CNS
Glioblastoma, IDH-mutant
Glioblastoma, IDH-wildtype
Medulloblastoma, Group 3
Medulloblastoma, Group 4
Glioma, High Grade
Neuroepithelial Tumor, High Grade
Medulloblastoma, SHH-activated and TP53 Mutant
Medulloblastoma, SHH-activated and TP53 Wildtype
Medulloblastoma, Chromosome 9q Loss
Medulloblastoma, Non-WNT Non-SHH, NOS
Medulloblastoma, Non-WNT/Non-SHH
Medulloblastoma, PTCH1 Mutation
Medulloblastoma, WNT-activated
Ependymoma, Recurrent
Glioma, Recurrent High Grade
Glioma, Recurrent Malignant
Embryonal Tumor, NOS
Glioma, Diffuse Midline, H3K27M-mutant
Embryonal Tumor With Multilayered Rosettes (ETMR)
Ependymoma, NOS, WHO Grade III
Ependymoma, NOS, WHO Grade II
Medulloblastoma, G3/G4
Ependymoma, RELA Fusion Positive Collapse << | Phase 1 | Recruiting | March 2025 | United States, Tennessee
... more >>
St. Jude Children's Research Hospital
Recruiting
Memphis, Tennessee, United States, 38105
Contact: Tabatha E. Doyle, RN 901-595-2544 tabatha.doyle@stjude.org
Principal Investigator: Giles W. Robinson, MD Collapse << |
| NCT02086552 | Recurrent Plasma Cell Myeloma
... more >>
Refractory Plasma Cell Myeloma Collapse << | Phase 2 | Active, not recruiting | January 30, 2019 | United States, Minnesota
... more >>
Mayo Clinic
Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03534947 | Basal Cell Carcinoma
... more >> Basal Cell Carcinoma of Skin, Site Unspecified
Skin Cancer
Invasive Carcinoma Collapse << | Phase 2 | Not yet recruiting | April 2020 | Australia, New South Wales
... more >>
Melanoma Institute Australia
Not yet recruiting
North Sydney, New South Wales, Australia, 2060
Contact: Pascal Guitera, MD PhD +61299117296 pascale.guitera@melanoma.org.au
Principal Investigator: Pascale Guitera Collapse << |
| NCT02303041 | Basal Cell Carcinoma (BCC) of ... more >>the Skin
Nevoid Basal Cell Carcinoma (BCC) Syndrome
Recurrent Skin Cancer Collapse << | Phase 2 | Completed | - | United States, California
... more >>
Stanford University, School of Medicine
Stanford, California, United States, 94305 Collapse << |
| NCT00961896 | Treatment for Basal Cell Carci... more >>nomas (BCCs) in Gorlin Syndrome Patients Collapse << | Phase 2 | Completed | - | Austria
... more >> Novartis Investigative Site
Graz, Austria
Novartis Investigator Site
Vienna, Austria
Switzerland
Novartis Investigative Site
Zurich, Switzerland Collapse << |
| NCT01350115 | Basal Cell Carcinoma
... more >> Gorlin Syndrome
Nevoid Basal Cell Carcinoma Syndrome Collapse << | Phase 2 | Completed | - | Austria
... more >> Novartis Investigative Site
Wien, Austria, A-1090
Belgium
Novartis Investigative Site
Leuven, Belgium, 3000
Canada, Ontario
Novartis Investigative Site
Markham, Ontario, Canada, L3P 1A8
Novartis Investigative Site
Waterloo, Ontario, Canada, N2J 1C4
Canada, Quebec
Novartis Investigative Site
Montreal, Quebec, Canada, H2K 4L5
Germany
Novartis Investigative Site
Kiel, Germany, 24105 Collapse << |
| NCT01350115 | - | - | Completed | - | - |
| NCT01033019 | Sporadic Superficial and Nodul... more >>ar Skin Basal Cell Carcinomas Collapse << | Phase 2 | Terminated(The data from parti... more >>cipants with sBCCs showed insufficient efficacy with current formulation and treatment conditions.) Collapse << | - | Australia, Queensland
... more >>
Novartis Investigative Site
Benowa, Queensland, Australia, 4217
Novartis Investigative Site
Woolloongabba, Queensland, Australia, 4102
Austria
Novartis Investigative Site
Graz, Austria, A-8036
Novartis Investigative Site
Wien, Austria, 1090 Collapse << |
| NCT01033019 | - | - | Terminated(The data from parti... more >>cipants with sBCCs showed insufficient efficacy with current formulation and treatment conditions.) Collapse << | - | - |
| NCT01708174 | Medulloblastoma | Phase 2 | Completed | - | - |
| NCT00961896 | - | - | Completed | - | - |
| NCT01431794 | Resectable Pancreatic Adenocar... more >>cinoma Collapse << | Phase 1
Phase 2 | Active, not recruiting | December 2018 | United States, Maryland
... more >>
Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins
Baltimore, Maryland, United States, 21205 Collapse << |
| NCT01708174 | - | - | Completed | - | - |
| NCT02182622 | Prostate Cancer | Phase 1 | Unknown | - | United States, New Jersey
... more >>
John Theurer Cancer @ HackensackUMC
Not yet recruiting
Hackensack, New Jersey, United States, 07601
Contact: Victoria Sica, RN 551-336-8054 vsica@hackensackumc.org
Principal Investigator: Martin E. Gutierrez, MD
Sub-Investigator: Robert S. Alter, MD
Sub-Investigator: Deena M Atieh Graham, MD
Sub-Investigator: Narjust Perez-Florez, MD Collapse << |
| NCT01757327 | Breast Neoplasms | Phase 2 | Withdrawn(Poor accrual) | December 2019 | - |
| NCT00880308 | Advanced Solid Tumor Cancers
... more >> Medulloblastoma
Basal Cell Carcinoma Collapse << | Phase 1 | Completed | - | United States, Pennsylvania
... more >>
University of Pittsburgh Medical Center SC
Pittsburgh, Pennsylvania, United States, 15213
United States, Texas
Cancer Therapy & Research Center / UT Health Science Center InstituteForDrugDevelopment(5)
San Antonio, Texas, United States, 78229
Spain
Novartis Investigative Site
Barcelona, Catalunya, Spain, 08035
Switzerland
Novartis Investigative Site
Zürich, Switzerland, 8091
United Kingdom
Novartis Investigative Site
Leicester, United Kingdom, LE1 5WW Collapse << |
| NCT01911416 | Pancreatic Ductal Adenocarcino... more >>ma Collapse << | Not Applicable | Withdrawn(No accrual) | - | United States, Utah
... more >>
Huntsman Cancer Institute
Salt Lake City, Utah, United States Collapse << |
| NCT02358161 | Pancreatic Cancer | Phase 1
Phase 2 | Unknown | September 2015 | Netherlands
... more >> Academic Medical Center
Recruiting
Amsterdam, Netherlands, 1105 AZ
Contact: J. W. Wilmink, MD, PhD 31 20 5665955 j.w.wilmink@amc.uva.nl
Contact: E. van Daalen 31 20 5668229 trialmedonc@amc.uva.nl
Principal Investigator: J. W. Wilmink, MD, PhD Collapse << |
| NCT01769768 | Advanced Solid Tumor | Phase 1 | Completed | - | United States, California
... more >>
City of Hope National Medical Center Oncology
Duarte, California, United States, 91010
United States, Kansas
University of Kansas Medical Center CBYM338B2203
Kansas City, Kansas, United States, 66160
United States, Massachusetts
Massachusetts General Hospital Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02114
United States, Michigan
Karmanos Cancer Institute
Detroit, Michigan, United States, 48201
United States, New Hampshire
Dartmouth Hitchcock Medical Center Dartmouth-Hitchcock Med Ctr
Lebanon, New Hampshire, United States, 03756
United States, New Jersey
Hackensack University Medical Center Dept.of HackensackUniv.MedCtr.
Hackensack, New Jersey, United States, 07601
United States, Pennsylvania
University of Pennsylvania--Abramson Cancer Center Abramson Cancer Center
Philadelphia, Pennsylvania, United States, 19104
Fox Chase Cancer Center
Philadelphia, Pennsylvania, United States, 19111-2497
University of Pittsburgh Cancer Institute UPMC Cancer Pavilion
Pittsburgh, Pennsylvania, United States, 15232
United States, South Carolina
Medical University of South Carolina Dept.of Neurosciences/MS Ctr.
Charleston, South Carolina, United States, 29425
Cancer Centers of the Carolinas SC
Greenville, South Carolina, United States, 29605
United States, Texas
Utah Health Science Center at San Antonio
San Antonio, Texas, United States, 78229
United States, Utah
University of Utah / Huntsman Cancer Institute
Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT01125800 | Medulloblastoma
... more >> Rhabdomyosarcoma
Neuroblastoma
Hepatoblastoma
Glioma
Astrocytoma Collapse << | Phase 1
Phase 2 | Completed | - | United States, Georgia
... more >>
Children's Healthcare of Atlanta Childern Hosp - ATL
Atlanta, Georgia, United States, 30342
United States, Maryland
Sidney Kimmel Comprehensive Cancer Center/Johns Hopkins Med. John Hopkins
Baltimore, Maryland, United States, 21231
United States, Massachusetts
Dana Farber Cancer Institute DFCI (3)
Boston, Massachusetts, United States, 02115
United States, Washington
Seattle Children's Hospital CPKC412A2114
Seattle, Washington, United States, 98105
Australia, Victoria
Novartis Investigative Site
Parkville, Victoria, Australia, 3052
Canada, Ontario
Novartis Investigative Site
Toronto, Ontario, Canada, M5G 1X8
Novartis Investigative Site
Toronto, Ontario, Canada, M5G 2M9
France
Novartis Investigative Site
Paris, France, 75231
Novartis Investigative Site
Villejuif Cedex, France, 94805
Italy
Novartis Investigative Site
Bologna, BO, Italy, 40139
Novartis Investigative Site
Milano, MI, Italy, 20133
Novartis Investigative Site
Roma, RM, Italy, 00168
United Kingdom
Novartis Investigative Site
Sutton, Surrey, United Kingdom, SM2 5PT
Novartis Investigative Site
Newcastle Upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT01327053 | Basal Cell Carcinoma | Phase 2 | Completed | - | - |
| NCT01125800 | - | - | Completed | - | - |
| NCT01694589 | Resectable Pancreatic Cancer | Early Phase 1 | Withdrawn(Lack of accrual) | - | United States, New Jersey
... more >>
Rutgers Cancer Institute of New Jersey
New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT01208831 | Advanced Solid Tumor Cancers
... more >> Medulloblastoma
Basal Cell Carcinoma Collapse << | Phase 1 | Completed | - | Hong Kong
... more >> Novartis Investigative Site
Hong Kong, Hong Kong
Japan
Novartis Investigative Site
Nagoya-city, Aichi, Japan, 466-8560
Novartis Investigative Site
Kobe-city, Hyogo, Japan, 650-0017
Taiwan
Novartis Investigative Site
Taipei, Taiwan, 10048 Collapse << |
| NCT01485744 | Pancreatic Cancer | Phase 1 | Active, not recruiting | March 2019 | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02214 Collapse << |
| NCT01576666 | Dose Escalation
... more >> Safety
Preliminary Efficacy
Advanced Solid Tumors
Metastatic Breast Cancer
Advanced Pancreatic Adenocarcinoma
Metastatic Colorectal Cancer
Recurrent Glioblastoma Multiforme
Gastric Cancer
Gastroesophageal Junction Cancer
Triple Negative Metastatic Breast Cancer
Hormone Receptor Positive (ER+/PR+, and Her2-) Metastatic Breast Cancer Collapse << | Phase 1 | Completed | - | - |
| NCT01787552 | Primary Myelofibrosis
... more >> Thrombocythemia, Essential
Thrombocytosis
Myeloproliferative Disorders
Bone Marrow Diseases
Hematologic Diseases
Blood Coagulation Disorders
Blood Platelet Disorders
Hemorrhagic Disorders Collapse << | Phase 1
Phase 2 | Completed | - | Australia, New South Wales
... more >>
Novartis Investigative Site
Camperdown, New South Wales, Australia, NSW
Australia, Queensland
Novartis Investigative Site
Woolloongabba, Queensland, Australia, 4102
Belgium
Novartis Investigative Site
Leuven, Belgium, 3000
Canada, Ontario
Novartis Investigative Site
Toronto, Ontario, Canada, M5G 2M9
Canada, Quebec
Novartis Investigative Site
Montreal, Quebec, Canada, H3T 1E2
Denmark
Novartis Investigative Site
Roskilde, Denmark, 4000
France
Novartis Investigative Site
Marseille, France, 13273
Germany
Novartis Investigative Site
Aachen, Germany, 52074
Novartis Investigative Site
Magdeburg, Germany, 39120
Ireland
Novartis Investigative Site
Galway, Ireland
Italy
Novartis Investigative Site
Firenze, FI, Italy, 50134
Novartis Investigative Site
Reggio Calabria, RC, Italy, 89124
Netherlands
Novartis Investigative Site
Amsterdam, Netherlands, 1081 HV
Novartis Investigative Site
Rotterdam, Netherlands, 3015 GD
Spain
Novartis Investigative Site
Barcelona, Catalunya, Spain, 08036
Novartis Investigative Site
Madrid, Spain, 28034
United Kingdom
Novartis Investigative Site
Glasgow, Scotland, United Kingdom, G12 0YN
Novartis Investigative Site
London, United Kingdom, SE1 9RT Collapse << |
| NCT01456676 | Philadelphia Chromosome Positi... more >>ve Chronic Myelogenous Leukemia Collapse << | Phase 1 | Completed | - | Canada, Ontario
... more >>
Novartis Investigative Site
Toronto, Ontario, Canada, M5G 2M9
France
Novartis Investigative Site
Marseille, France, 13273
Germany
Novartis Investigative Site
Frankfurt, Germany, 60590
Novartis Investigative Site
Ulm, Germany, 89081
Italy
Novartis Investigative Site
Roma, RM, Italy, 00161
Spain
Novartis Investigative Site
Pamplona, Navarra, Spain, 31008
Novartis Investigative Site
Madrid, Spain, 28006 Collapse << |
| NCT01487785 | Pancreatic Cancer | Phase 1 | Completed | - | United States, Massachusetts
... more >>
Massachusetts General Hospital Dept. of Mass General Hospital
Boston, Massachusetts, United States, 02114
United States, New York
Memorial Sloan Kettering Cancer Center MSKCC - SC
New York, New York, United States, 10021
United States, Utah
University of Utah / Huntsman Cancer Institute Huntsman UT
Salt Lake City, Utah, United States, 84103
Spain
Novartis Investigative Site
Barcelona, Catalunya, Spain, 08035
United Kingdom
Novartis Investigative Site
Liverpool, United Kingdom, L7 8XP Collapse << |
| NCT01579929 | Lung Cancer | Phase 1 | Completed | - | United States, New Jersey
... more >>
Memoral Sloan Kettering Cancer Center
Basking Ridge, New Jersey, United States
United States, New York
Memorial Sloan Kettering Cancer Center at Commack
Commack, New York, United States, 11725
Memorial Sloan Kettering Cancer Center
New York, New York, United States, 10065
Memorial Sloan Kettering Cancer Center at Mercy Medical Center
Rockville Centre, New York, United States, 11570
Memoral Sloan Kettering Cancer Center@Phelps Memorial Hospital
Sleepy Hollow, New York, United States Collapse << |
| NCT01826214 | Acute Leukemias | Phase 2 | Completed | - | United States, North Carolina
... more >>
Duke University Medical Center SC-5
Durham, North Carolina, United States, 27710
Australia, South Australia
Novartis Investigative Site
Adelaide, South Australia, Australia, 5000
Australia, Victoria
Novartis Investigative Site
Prahran, Victoria, Australia, 3181
Austria
Novartis Investigative Site
Salzburg, Austria, 5020
Novartis Investigative Site
Wien, Austria, 1090
Belgium
Novartis Investigative Site
Leuven, Belgium, 3000
Novartis Investigative Site
Yvoir, Belgium, 5530
Canada, Quebec
Novartis Investigative Site
Montreal, Quebec, Canada, H1T 2M4
Novartis Investigative Site
Montreal, Quebec, Canada, H3T 1E2
Germany
Novartis Investigative Site
Dresden, Germany, 01307
Novartis Investigative Site
Frankfurt, Germany, 60590
Novartis Investigative Site
Magdeburg, Germany, 39120
Novartis Investigative Site
Ulm, Germany, 89081
Hungary
Novartis Investigative Site
Debrecen, Hungary, 4032
Netherlands
Novartis Investigative Site
Amsterdam, Netherlands, 1081 HV
Novartis Investigative Site
Nijmegen, Netherlands, 6525 GA
Novartis Investigative Site
Rotterdam, Netherlands, 3015 CE
Novartis Investigative Site
Rotterdam, Netherlands, 3075 EA
Norway
Novartis Investigative Site
Bergen, Norway, NO-5021
Novartis Investigative Site
Trondheim, Norway, 7006
Spain
Novartis Investigative Site
Salamanca, Castilla y Leon, Spain, 37007
Novartis Investigative Site
Valencia, Comunidad Valenciana, Spain, 46026
United Kingdom
Novartis Investigative Site
London, United Kingdom, WC1E 6HX Collapse << |
| NCT01529450 | Basal Cell Carcinoma | Not Applicable | Completed | - | United States, California
... more >>
Stanford University, School of Medicine
Stanford, California, United States, 94305 Collapse << |
| NCT01529450 | - | - | Completed | - | - |
| NCT01327053 | - | - | Completed | - | - |
| NCT03070691 | Basal Cell Carcinoma | Phase 2
Phase 3 | Withdrawn | - | - |
| NCT01954355 | Solid Tumor
O... more >>varian Cancer Collapse << | Phase 1 | Completed | - | Switzerland
... more >> Istituto Oncologico della Svizzera Italiana
Bellinzona, Switzerland, 6500
Kantonsspital Graubünden
Chur, Switzerland, 7000
Centre Pluridisciplinaire d`Oncologie CHUV
Lausanne, Switzerland, 1011
Kantonsspital St. Gallen
St. Gallen, Switzerland, 9007 Collapse << |
| NCT02002689 | - | - | Terminated(Low enrollment) | - | - |
| NCT02002689 | PTCH1 or SMO Activated Solid a... more >>nd Hematologic Tumors Collapse << | Phase 2 | Terminated(Low enrollment) | - | United States, California
... more >>
University of California Davis Cancer Center UC Davis Cancer (3)
Sacramento, California, United States, 95817
United States, Colorado
Rocky Mountain Cancer Centers RMCC - Aurora
Greenwood Village, Colorado, United States
United States, Illinois
Lurie Children's Hospital of Chicago Developmental Therapeutics
Chicago, Illinois, United States, 60611
United States, Minnesota
Minnesota Oncology Hematology, P.A. Southdate Medical Center
Minneapolis, Minnesota, United States, 55404
United States, Ohio
Cleveland Clinic Foundation Cleveland Clinic (19)
Cleveland, Ohio, United States, 44195
United States, South Dakota
Sanford Research Sanford Health
Sioux Falls, South Dakota, United States, 57104
United States, Texas
Oncology Consultants Oncology Group
Houston, Texas, United States, 77024
MD Anderson Cancer Center/University of Texas MD Anderson Cancer Center (3)
Houston, Texas, United States, 77030
United States, Utah
Intermountain Medical Center Intermountain Healthcare
Murray, Utah, United States, 84157
United States, Washington
Seattle Cancer Care Alliance Skagit Valley Hospital
Seattle, Washington, United States, 98109-1023 Collapse << |
| NCT01826214 | - | - | Completed | - | - |
| NCT02027376 | Advanced Breast Cancer | Phase 1 | Completed | - | Spain
... more >> Complejo Hospitalario Universitario A Coruña
A Coruña, Spain, 15006
Hospital General Universitario Gregorio Marañón
Madrid, Spain, 28007
Hospital Clínico Universitario San Carlos
Madrid, Spain, 28040
Hospital Clínico Universitario Virgen de la Victoria
Málaga, Spain, 29010
Hospital Universitario Virgen del Rocío
Sevilla, Spain, 41071 Collapse << |
| NCT01764776 | Normal Hepatic Function
... more >> Impaired Hepatic Function Collapse << | Phase 1 | Completed | - | United States, Florida
... more >>
University of Miami Div. of Clinical Pharmacology
Miami, Florida, United States, 33136
Belgium
Novartis Investigative Site
Bruxelles, Belgium, 1200
Bulgaria
Novartis Investigative Site
Sofia, Bulgaria, 1612
Germany
Novartis Investigative Site
Berlin, Germany, 14050
Israel
Novartis Investigative Site
Tel-Aviv, Israel, 64239 Collapse << |
| NCT02151864 | Hepatocellular Carcinoma
... more >> Cirrhosis Collapse << | Phase 1 | Active, not recruiting | January 2019 | United States, California
... more >>
UCSD Moores Cancer Center
La Jolla, California, United States, 92093 Collapse << |
| NCT02027376 | - | - | Completed | - | - |
| NCT02254551 | - | - | Terminated(Safety lead-in data... more >> did not support continuation of study.) Collapse << | - | - |
| NCT02138929 | Esophageal Cancer | Phase 1 | Active, not recruiting | November 2020 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT02254551 | Multiple Myeloma | Phase 2 | Terminated(Safety lead-in data... more >> did not support continuation of study.) Collapse << | - | United States, Colorado
... more >>
Colorado Blood Cancer Institute
Denver, Colorado, United States, 80218
United States, Ohio
Oncology Hematology Care
Cincinnati, Ohio, United States, 45236
United States, Tennessee
Tennessee Oncology
Chattanooga, Tennessee, United States, 37404
Tennessee Oncology PLLC
Nashville, Tennessee, United States, 37203
United States, Texas
Texas Transplant Institute/Methodist Healthcare
San Antonio, Texas, United States, 78229 Collapse << |
| NCT02086513 | Graft Versus Host Disease | Phase 1 | Terminated | - | United States, Massachusetts
... more >>
Beth Israel Deaconess Medical Center
Boston, Massachusetts, United States, 02215
Dana Farber Cancer Institute
Boston, Massachusetts, United States, 02215
Massachusetts General Hospital
Boston, Massachusetts, United States, 02215 Collapse << |
| NCT02111187 | Prostate Cancer | Phase 1 | Completed | - | United States, Maryland
... more >>
Johns Hopkins Hospital
Baltimore, Maryland, United States, 21205 Collapse << |
| NCT02195973 | Recurrent Ovarian Cancer | Phase 1 | Completed | - | United States, Alabama
... more >>
University of Alabama at Birmingham
Birmingham, Alabama, United States, 35294 Collapse << |
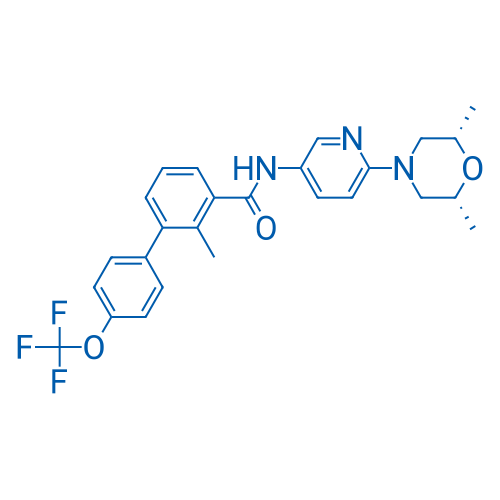
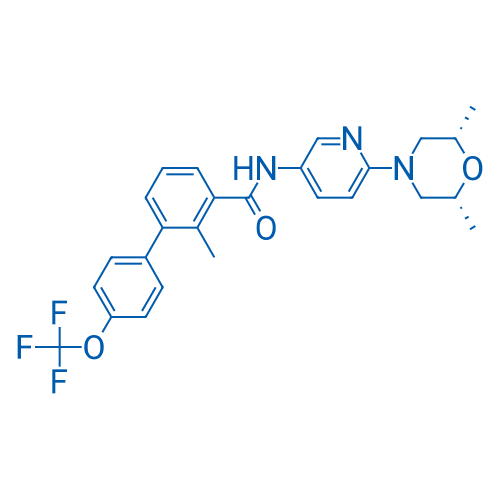

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
