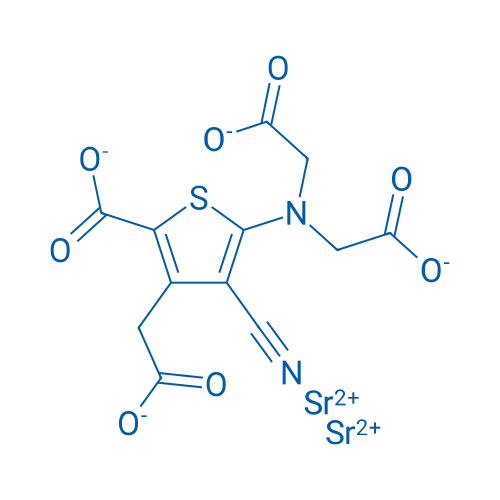
CAS No.: 135459-87-9
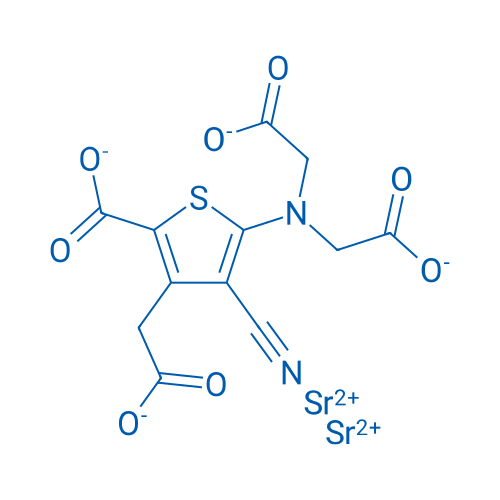
Strontium Ranelate/雷尼酸锶 Catalog No. CSN11988
Synonyms: Distrontium renelate;S12911;S12911-2
Strontium ranelate stimulates the calcium sensing receptors (CaSR) and leads to the differentiation of pre-osteoblast to osteoblast which increases the bone formation.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02637180 - - Completed - Greece ... more >> NATIONAL AND KAPODISTRIAN UNIVERSITY OF ATHENS, MEDICAL SCHOOL, 2nd DEPARTMENT OF ORTHOPAEDIC SURGERY Athens, Attiki, Greece, 14233 Democritus University of Thrace, School of Medicine, Department of Orthopaedic Surgery Alexandroupolis, Evros, Greece, 68100 Orthopaedic Department, Faculty of Medicine, School of Health Sciences, University of Thessalia Larissa, Greece, 41110 Aristotle University of Thessaloniki, 3rd University Orthopaedic Department, Papageorgiou General Hospital Thessaloniki, Greece, 56403 Collapse << NCT00409032 Osteoporosis Phase 2 Completed - Denmark ... more >> PhaseOneTrials Hvidovre, Denmark, 2650 Odense University Hospital Odense, Denmark, 5000 United Kingdom Medinova Clinic Northwood, Middlesex, United Kingdom Synexus Wales Clinical Research Centre Cardiff, United Kingdom, CF14 5GJ Synexus Scotland Clinical Research Centre Glasgow, United Kingdom, G81 2DR Synexus Limited Reading Clinical Research Centre Reading, United Kingdom, RG2 7AG University of Sheffield Sheffield, United Kingdom, S5 7AU Synexus Crosby Clinical Research Centre Waterloo, United Kingdom, L22 0LG Synexus Wigan Clinical Research Centre Wigan, United Kingdom, WN1 1XX Collapse << NCT00239629 Postmenopausal Osteoporosis Phase 4 Completed - Czech Republic ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Prague, Czech Republic, 128 21 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 20354 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Heidelberg, Germany, 69120 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Magdeburg, Germany, D-39110 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Vogelsang, Germany, 39245 Greece For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kifissia, Greece, 145 61 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Thessaloniki, Greece, 56429 Israel For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Haifa, Israel, 31096 Mexico For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guadalajara, Mexico, 44670 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08025 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Madrid, Spain, 28041 Collapse << - 更多
- 参考文献
- [1] Guo X, Wei S, et al. Dose-dependent Effects of Strontium Ranelate on Ovariectomy Rat Bone Marrow Mesenchymal Stem Cells and Human Umbilical Vein Endothelial Cells. Int J Biol Sci. 2016 Nov 25;12(12):1511-1522.
- [2] Fernandez JM, Molinuevo MS, et al. Strontium ranelate prevents the deleterious action of advanced glycation endproducts on osteoblastic cells via calcium channel activation. Eur J Pharmacol. 2013 Apr 15;706(1-3):41-7.
- [3] Bonnelye E, Chabadel A, Saltel F, Jurdic P. Dual effect of strontium ranelate: stimulation of osteoblast differentiation and inhibition of osteoclast formation and resorption in vitro. Bone. 2008 Jan;42(1):129-38.
- [4] Delannoy P, Bazot D, Marie PJ. Long-term treatment with strontium ranelate increases vertebral bone mass without deleterious effect in mice. Metabolism. 2002 Jul;51(7):906-11.
- [5] Bain SD, Jerome C, Shen V, Dupin-Roger I, Ammann P. Strontium ranelate improves bone strength in ovariectomized rat by positively influencing bone resistance determinants. Osteoporos Int. 2009 Aug;20(8):1417-28.
- [6] Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel EM, Pors-Nielsen S, Rizzoli R, Genant HK, Reginster JY. The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med. 2004 Jan 29;350(5):459-68.
- [7] Marie PJ. Strontium ranelate in osteoporosis and beyond: identifying molecular targets in bone cell biology. Mol Interv. 2010 Oct;10(5):305-12.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 135459-87-9 | 储存条件 |
|
|
| 分子式 | C12H6N2O8SSr2 | 运输 | 蓝冰 | |
| 分子量 | 513.49 | 别名 | Distrontium renelate;S12911;S12911-2;trade mane: Protelos or Proto;雷尼酸锶 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
