| NCT01916824 | - | - | Completed | - | - |
| NCT01916824 | Major Depressive Disorder
... more >> Healthy Controls Collapse << | Phase 4 | Completed | - | United States, Georgia
... more >>
Emory Mood and Anxiety Disorders Program
Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01469377 | Major Depressive Disorder | Phase 2 | Completed | - | - |
| NCT01469377 | - | - | Completed | - | - |
| NCT02883660 | - | - | Completed | - | United States, Pennsylvania
... more >>
Thomas Jefferson University
Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT02893371 | - | - | Enrolling by invitation | June 2019 | United States, New Mexico
... more >>
Christophe G Lambert
Albuquerque, New Mexico, United States, 87131 Collapse << |
| NCT00290914 | Major Depressive Disorder | Phase 2 | Completed | - | - |
| NCT01473381 | Major Depressive Disorder | Phase 4 | Completed | - | - |
| NCT00683592 | Major Depressive Disorder | Phase 3 | Completed | - | United States, California
... more >>
Pharmacology Research Institute
Newport Beach, California, United States, 92660
United States, Florida
Florida Clinical Research Center
Bradenton, Florida, United States, 34208
United States, Georgia
Atlanta Institute of Medicine and Research
Atlanta, Georgia, United States, 30328
United States, Oregon
Summit Research Network
Portland, Oregon, United States, 97210
United States, Pennsylvania
University of Pennsylvania Department of Psychiatry Mood and Anxiety Disorders
Philadelphia, Pennsylvania, United States, 19104
United States, Texas
Mood Disorders Research Program and Clinic Exchange Park
Dallas, Texas, United States, 75235
United States, Utah
University of Utah Health Sciences Ctr, Dept of Psychiatry Mood Disorders Clinic
Salt Lake City, Utah, United States, 84132
United States, Washington
Northwest Clinical Research Center
Bellevue, Washington, United States, 98004
Summit Research Network
Seattle, Washington, United States, 98104 Collapse << |
| NCT01473381 | - | - | Completed | - | - |
| NCT01629966 | Generalized Anxiety Disorder | Phase 3 | Completed | - | - |
| NCT00285376 | Depressive Disorder, Major | Phase 3 | Completed | - | United States, California
... more >>
Pharmacology Research Institute
Los Alamitos, California, United States, 90720
Pharmacology Research Institute
Newport Beach, California, United States, 92660
Pharmacology Research Institute
Northridge, California, United States, 91324
Pharmacology Research Institute
Riverside, California, United States, 92506
United States, Georgia
Atlanta Institute of Medicine & Research
Atlanta, Georgia, United States, 30328
Atlanta Institute of Medicine & Research
Marietta, Georgia, United States, 30060
United States, Michigan
Summit Research Network (Michigan), Inc.
Farmington Hills, Michigan, United States, 48336
Summit Research Network (Michigan), Inc.
Flint, Michigan, United States, 48507
United States, New York
Social Psychiatry Research Institute
Brooklyn, New York, United States, 11235
United States, Oregon
Summit Research Network (Oregon) Inc.
Portland, Oregon, United States, 97210
United States, Pennsylvania
University of Pennsylvania Department of Psychiatry Mood and Anxiety Disorders Section
Philadelphia, Pennsylvania, United States, 19104-3309
United States, South Carolina
Southeast Health Consultants, LLC
Charleston, South Carolina, United States, 29407
United States, Utah
University of Utah health Services Center Dept. of Psychiatry Mood and Anxiety Disorders
Salt Lake City, Utah, United States, 84132
United States, Washington
Northwest Clinical Research Center
Bellevue, Washington, United States, 98004
Summit Research Network (Seattle) LLC
Seattle, Washington, United States, 98104 Collapse << |
| NCT02372799 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT02097147 | Healthy | Phase 1 | Completed | - | United States, Florida
... more >>
Forest Investigative Site 001
Miami, Florida, United States, 33014
United States, Kansas
Forest Investigative Site 002
Overland Park, Kansas, United States, 66212
United States, Minnesota
Forest Investigative Site 003
St. Paul, Minnesota, United States, 55114
United States, Virginia
Forest Investigative Site 004
Charlottesville, Virginia, United States, 22903 Collapse << |
| NCT00644358 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00644358 | - | - | Completed | - | - |
| NCT01742832 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Illinois
... more >>
University of Chicago
Chicago, Illinois, United States, 60615 Collapse << |
| NCT01742832 | - | - | Completed | - | - |
| NCT01680900 | - | - | Completed | - | - |
| NCT01608295 | - | - | Completed | - | - |
| NCT01573598 | Major Depressive Disorder | Phase 4 | Completed | - | - |
| NCT00683592 | - | - | Completed | - | - |
| NCT01574183 | Marijuana Dependence | Phase 2 | Completed | - | United States, South Carolina
... more >>
Medical University of South Carolina
Charleston, South Carolina, United States, 29425 Collapse << |
| NCT01574183 | - | - | Completed | - | - |
| NCT01766401 | Generalized Anxiety Disorder | Phase 3 | Completed | - | - |
| NCT01473394 | Major Depressive Disorder | Phase 4 | Completed | - | United States, California
... more >>
Forest Investigative Site 009
Beverly Hills, California, United States, 90210
Forest Investigative Site 003
Encino, California, United States, 91316
Forest Investigative Site 010
New port Beach, California, United States, 92660
United States, Florida
Forest Investigative Site 005
Jacksonville, Florida, United States, 32216
Forest Investigative Site 004
Orlando, Florida, United States, 32806
United States, Georgia
Forest Investigative Site 012
Atlanta, Georgia, United States, 30328
United States, Maryland
Forest Investigative Site 001
Baltimore, Maryland, United States, 21285
United States, Ohio
Forest Investigative Site 002
Dayton, Ohio, United States, 45417
United States, Pennsylvania
Forest Investigative Site 011
Philadelphia, Pennsylvania, United States, 19104
United States, Rhode Island
Forest Investigative Site 015
Lincoln, Rhode Island, United States, 02865
United States, Tennessee
Forest Investigative Site 008
Memphis, Tennessee, United States, 38119
United States, Texas
Forest Investigative Site 016
San Antonio, Texas, United States, 78229
United States, Washington
Forest Investigative Site 006
Bellevue, Washington, United States, 98007
Forest Investigative Site 013
Seattle, Washington, United States, 98104 Collapse << |
| NCT01828515 | Memory Impairment | Phase 2 | Completed | - | United States, Texas
... more >>
UT Southwestern Medical Center
Dallas, Texas, United States, 75390 Collapse << |
| NCT01680900 | Hot Flushes | Not Applicable | Completed | - | United States, Pennsylvania
... more >>
Dept OB/GYN, Mudd Professorship Suite
Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01878292 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01608295 | Major Depressive Disorder | Phase 4 | Completed | - | United States, California
... more >>
UCLA Semel Institute
Los Angeles, California, United States, 90095 Collapse << |
| NCT01828515 | - | - | Completed | - | - |
| NCT01473394 | - | - | Completed | - | - |
| NCT02028026 | Major Depressive Disorder
... more >> Anxiety
Comorbidity Collapse << | Phase 4 | Withdrawn(Inability to recruit... more >> eligible subjects) Collapse << | - | United States, Massachusetts
... more >>
Massachusetts General Hospital
Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01999920 | - | - | Completed | - | - |
| NCT01844115 | Generalized Anxiety Disorder | Phase 3 | Completed | - | - |
| NCT01712321 | Social Anxiety Disorder | Not Applicable | Unknown | April 2014 | United States, New York
... more >>
The Medical Research Network, LLC
New York, New York, United States, 10128 Collapse << |
| NCT01715519 | PTSD
Depressi... more >>on Collapse << | Phase 4 | Completed | - | United States, California
... more >>
Veterans Affairs Long Beach Healthcare System
Long Beach, California, United States, 90822
United States, Nebraska
Veterans Affairs Nebraska Western-Iowa Healthcare Systems
Omaha, Nebraska, United States, 68105 Collapse << |
| NCT02015546 | Major Depressive Disorder (MDD... more >>) Collapse << | Phase 3 | Completed | - | United States, North Carolina
... more >>
Duke University Medical Center / Civitan Building
Durham, North Carolina, United States, 27705 Collapse << |
| NCT01856127 | Sexual Dysfunction
... more >> Major Depressive Disorder Collapse << | Phase 4 | Terminated(PI left institution... more >>) Collapse << | - | United States, Pennsylvania
... more >>
University of Pennsylvania
Philadelphia, Pennsylvania, United States, 19104
Thomas Jefferson University
Philadelphia, Pennsylvania, United States, 19107
United States, Virginia
University of Virginia
Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT02436239 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01999920 | Separation Anxiety Disorder | Phase 4 | Completed | - | United States, New York
... more >>
New York State Psychiatric Institute
New York, New York, United States, 10032 Collapse << |
| NCT02015546 | - | - | Completed | - | - |
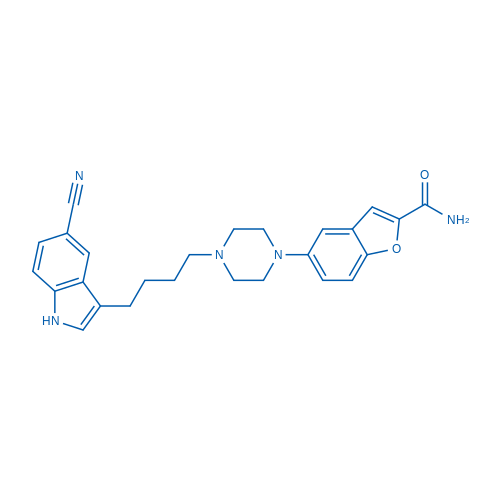
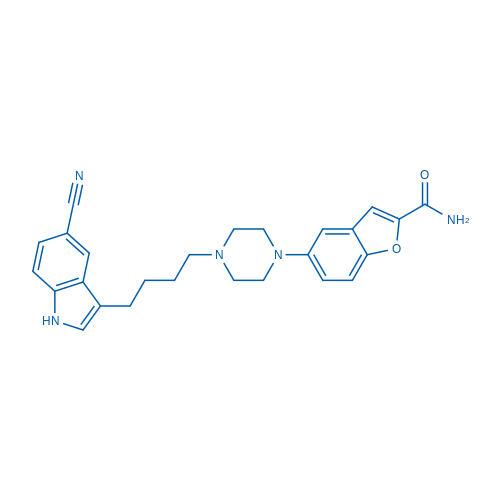

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
