| NCT02456428 | - | - | Completed | - | Canada, Quebec
... more >> Lady Davis Institute for Medical Research, Jewish General Hospital
Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02476760 | - | - | Completed | - | Canada, Quebec
... more >> Lady Davis Institute for Medical Research, Jewish General Hospital
Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT02685410 | HIV Prevention
... more >> STI Prevention Collapse << | Not Applicable | Completed | - | United States, Connecticut
... more >>
Yale New Haven Hospital
New haven, Connecticut, United States, 06510 Collapse << |
| NCT03157895 | Relationship, Family (Foster)
... more >>
Substance Use Prevention Collapse << | Not Applicable | Recruiting | January 31, 2020 | United States, Washington
... more >>
Social Development Research Group
Recruiting
Seattle, Washington, United States, 98115
Contact: Jessica Colito, MSW 206-221-5250 jcolito@uw.edu
Contact: Patricia Gomez 206-616-7931 pagomez@uw.edu
Principal Investigator: Kevin Haggerty, PhD
Sub-Investigator: Susan Barkan, PhD Collapse << |
| NCT02475499 | - | - | Completed | - | Canada, Quebec
... more >> Lady Davis Institute for Medical Research, Jewish General Hospital
Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00985400 | Colorectal Cancer | Not Applicable | Active, not recruiting | November 2020 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030-4009 Collapse << |
| NCT00454766 | - | - | Active, not recruiting | February 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT00853203 | - | - | Active, not recruiting | January 2019 | United States, Texas
... more >>
Kelsey-Seybold Clinics
Houston, Texas, United States, 77030
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01504919 | Psychosocial Problem | Not Applicable | Active, not recruiting | January 2019 | United States, Texas
... more >>
William Marsh Rice University
Houston, Texas, United States, 77005
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01311856 | - | - | Active, not recruiting | March 2020 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT00473798 | - | - | Active, not recruiting | July 2020 | United States, Texas
... more >>
Hermann Professional Building Clinic
Houston, Texas, United States, 77030
LBJ Hospital
Houston, Texas, United States, 77030
Texas Medical Center Clinic
Houston, Texas, United States, 77030
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030
Canada, British Columbia
Vancouver General Hospital/British Columbia Cancer Agency
Vancouver, British Columbia, Canada, V5Z 1M9 Collapse << |
| NCT00533338 | Breast Cancer | Not Applicable | Active, not recruiting | July 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01504789 | Breast Cancer | Not Applicable | Active, not recruiting | February 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030
Puerto Rico
University of Puerto Rico
San Juan, Puerto Rico, 00936 Collapse << |
| NCT02774759 | Malignant Neoplasms of Female ... more >>Genital Organs Collapse << | Not Applicable | Active, not recruiting | October 2020 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT00633633 | Heart Failure | Not Applicable | Active, not recruiting | February 2021 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030
The University of Texas Health Science Center at San Antonio
San Antonio, Texas, United States, 78249 Collapse << |
| NCT01697592 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00501761 | - | - | Active, not recruiting | May 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01646788 | Obstructive Lesions of Arterio... more >>venous Dialysis Fistulae Collapse << | Not Applicable | Unknown | January 2015 | Israel
... more >> The Rabin Medical Center, Hasharon Hospital
Not yet recruiting
Petah Tikva, Israel Collapse << |
| NCT00542438 | Cardiovascular Disease | Not Applicable | Active, not recruiting | October 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT02593422 | Ovarian Cancer | Not Applicable | Completed | - | United States, Maryland
... more >>
University of Maryland, College Park
College Park, Maryland, United States, 20742 Collapse << |
| NCT02194387 | Breast Cancer
... more >> Ovarian Cancer Collapse << | Not Applicable | Recruiting | September 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Recruiting
Houston, Texas, United States, 77030 Collapse << |
| NCT01682824 | - | - | Active, not recruiting | May 31, 2019 | United States, Texas
... more >>
M D Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01242202 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | Japan
... more >>
Chubu, Japan
Hokkaido, Japan
Kansai, Japan
Kantou, Japan
Kyushu, Japan
Touhoku, Japan Collapse << |
| NCT01263470 | - | - | Completed | - | - |
| NCT01697592 | - | - | Completed | - | - |
| NCT02287402 | Impaired Glucose Tolerance (IG... more >>T) Collapse << | Phase 4 | Completed | - | - |
| NCT01263470 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Japan
... more >>
Okayama, Japan, 701-0192 Collapse << |
| NCT02287402 | - | - | Completed | - | - |
| NCT00724542 | Coronary Heart Disease
... more >> Impaired Glucose Tolerance Collapse << | Not Applicable | Unknown | December 2010 | China, Beijing
... more >> Peking University First Hospital
Recruiting
Beijing, Beijing, China, 100034
Contact: Wenhui Ding, Professor 86-10-66551122 ext 5001 dwh_rd@126.com Collapse << |
| NCT01263496 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT02097342 | Type 2 Diabetes Mellitus
... more >> Insulin Sensitivity/Resistance Collapse << | Phase 4 | Unknown | January 2015 | India
... more >> Postgraduate Institute of Medical Education & Research
Recruiting
Chandigarh, India, 160012
Contact: Anil Bhansali, MD DM anilbhansaliendocrine@gmail.com Collapse << |
| NCT00970528 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of
... more >>
Wonju-si, Gang''weondo, Korea, Republic of, 220-701
Busan, Korea, Korea, Republic of, 602-714
Gyeonggi-do, Korea, Korea, Republic of, 463-500
Gyeonggi-do, Korea, Korea, Republic of, 471-701
Pusan, Korea, Korea, Republic of, 602-739
Seoul, Korea, Korea, Republic of, 150-713
Daegu, Korea, Republic of, 705-717
Jeonju-si, Korea, Republic of, 456-712
Seongbuk-gu Seoul, Korea, Republic of, 136-705
Seoul, Korea, Republic of, 134-701
Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01263496 | - | - | Completed | - | - |
| NCT00212017 | Impaired Glucose Tolerance
... more >> Myocardial Infarction Collapse << | Not Applicable | Terminated(We obtained the fin... more >>al results due to the interim analysis.) Collapse << | - | Japan
... more >> National Cardiovascular Center
Suita, Osaka, Japan, 565-8565 Collapse << |
| NCT03546972 | Obesity and Other Hyperaliment... more >>ation Collapse << | Not Applicable | Recruiting | November 2019 | United States, Texas
... more >>
University of Texas MD Anderson Cancer Center
Recruiting
Houston, Texas, United States, 77030
Contact kbasenen@mdanderson.org Collapse << |
| NCT01263483 | Type 2 Diabetes Mellitus | Phase 2
Phase 3 | Completed | - | - |
| NCT01263509 | Type 2 Diabetes Mellitus | Phase 2
Phase 3 | Completed | - | - |
| NCT01263483 | - | - | Completed | - | - |
| NCT00411554 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
| NCT01263509 | - | - | Completed | - | - |
| NCT00411554 | - | - | Completed | - | - |
| NCT00654381 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan
... more >> 1218.23.05 Boehringer Ingelheim Investigational Site
Asahi, Chiba, Japan
1218.23.06 Boehringer Ingelheim Investigational Site
Funabashi, Chiba, Japan
1218.23.21 Boehringer Ingelheim Investigational Site
Hitachinaka, Ibaraki, Japan
1218.23.44 Boehringer Ingelheim Investigational Site
Hitachiota, Ibaraki, Japan
1218.23.09 Boehringer Ingelheim Investigational Site
Imizu, Toyama, Japan
1218.23.45 Boehringer Ingelheim Investigational Site
Inashiki-gun, Ibaraki, Japan
1218.23.13 Boehringer Ingelheim Investigational Site
Izumisano, Osaka, Japan
1218.23.27 Boehringer Ingelheim Investigational Site
Kariya, Aichi, Japan
1218.23.47 Boehringer Ingelheim Investigational Site
Kitakatsushika-gun, Saitama, Japan
1218.23.39 Boehringer Ingelheim Investigational Site
Kitakyuushuu, Fukuoka, Japan
1218.23.02 Boehringer Ingelheim Investigational Site
Koriyama, Fukushima, Japan
1218.23.03 Boehringer Ingelheim Investigational Site
Koriyama, Fukushima, Japan
1218.23.10 Boehringer Ingelheim Investigational Site
Kyoto, Kyoto, Japan
1218.23.37 Boehringer Ingelheim Investigational Site
Marugame, Kagawa, Japan
1218.23.38 Boehringer Ingelheim Investigational Site
Marugame, Kagawa, Japan
1218.23.23 Boehringer Ingelheim Investigational Site
Matsumoto, Nagano, Japan
1218.23.07 Boehringer Ingelheim Investigational Site
Meguro-ku, Tokyo, Japan
1218.23.25 Boehringer Ingelheim Investigational Site
Nagoya, Aichi, Japan
1218.23.26 Boehringer Ingelheim Investigational Site
Nagoya, Aichi, Japan
1218.23.28 Boehringer Ingelheim Investigational Site
Nagoya, Aichi, Japan
1218.23.29 Boehringer Ingelheim Investigational Site
Nagoya, Aichi, Japan
1218.23.30 Boehringer Ingelheim Investigational Site
Nagoya, Aichi, Japan
1218.23.04 Boehringer Ingelheim Investigational Site
Naka, Ibaraki, Japan
1218.23.15 Boehringer Ingelheim Investigational Site
Nishi-ku, Hiroshima, Hiroshima, Japan
1218.23.34 Boehringer Ingelheim Investigational Site
Nishi-ku, Sakai, Osaka, Japan
1218.23.22 Boehringer Ingelheim Investigational Site
Nishishinjyuku, Shinjyuku-ku, Tokyo, Japan
1218.23.16 Boehringer Ingelheim Investigational Site
Oita, Oita, Japan
1218.23.40 Boehringer Ingelheim Investigational Site
Oita, Oita, Japan
1218.23.36 Boehringer Ingelheim Investigational Site
Okayama, Okayama, Japan
1218.23.11 Boehringer Ingelheim Investigational Site
Osaka, Osaka, Japan
1218.23.12 Boehringer Ingelheim Investigational Site
Osaka, Osaka, Japan
1218.23.32 Boehringer Ingelheim Investigational Site
Osaka, Osaka, Japan
1218.23.33 Boehringer Ingelheim Investigational Site
Osaka, Osaka, Japan
1218.23.35 Boehringer Ingelheim Investigational Site
Osaka, Osaka, Japan
1218.23.17 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.18 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.19 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.41 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.42 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.43 Boehringer Ingelheim Investigational Site
Sapporo, Hokkaido, Japan
1218.23.01 Boehringer Ingelheim Investigational Site
Sendai, Miyagi, Japan
1218.23.20 Boehringer Ingelheim Investigational Site
Sendai, Miyagi, Japan
1218.23.46 Boehringer Ingelheim Investigational Site
Shinjuku-ku, Tokyo, Japan
1218.23.08 Boehringer Ingelheim Investigational Site
Shinjyuku-ku,Tokyo, Japan
1218.23.14 Boehringer Ingelheim Investigational Site
Suita, Osaka, Japan
1218.23.31 Boehringer Ingelheim Investigational Site
Takatsuki, Osaka, Japan
1218.23.48 Boehringer Ingelheim Investigational Site
Yokohama, Kanagawa, Japan Collapse << |
| NCT00837577 | - | - | Completed | - | - |
| NCT01309698 | Type 2 Diabetes | Phase 4 | Completed | - | Japan
... more >> Novartis Investigative Site
Tokyo, Japan Collapse << |
| NCT00654381 | - | - | Completed | - | - |
| NCT00368134 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | Japan
... more >> Novartis Pharmaceuticals
Tokyo, Japan Collapse << |
| NCT01055652 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | Japan
... more >> Research Site
Moriya, Japan
Research Site
Osaka, Japan Collapse << |
| NCT02049814 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Anhui
... more >>
Hefei, Anhui, China
Maanshan, Anhui, China
China, Beijing
Beijing, Beijing, China
China, Guangdong
Guangzhou, Guangdong, China
Shenzhen, Guangdong, China
Taishan, Guangdong, China
China, Hunan
Zhuzhou, Hunan, China
China, Jiangsu
Nanjing, Jiangsu, China
Xuzhou, Jiangsu, China
China, Jilin
Changchun, Jilin, China
Jilin, Jilin, China
China, Liaoning
Shenyang, Liaoning, China
China, Shandong
Qingdao, Shandong, China
China, Shanghai
Shanghai, Shanghai, China
China, Shanxi
Yan An, Shanxi, China
Yanan, Shanxi, China
China, Tianjin
Tianjin, Tianjin, China
China, Zhejiang
Wenzhou, Zhejiang, China Collapse << |
| NCT01993927 | - | - | Completed | - | Japan
... more >>
Osaka, Japan Collapse << |
| NCT01993927 | - | - | Completed | - | - |
| NCT00663884 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | Korea, Republic of
... more >>
The Catholic Univ. of Korea, Kangnam ST. Mary's Hospital
Seoul, Korea, Republic of, 137-701 Collapse << |
| NCT00837577 | Diabetes Mellitus, Non-Insulin... more >>-Dependent Collapse << | Phase 3 | Completed | - | - |
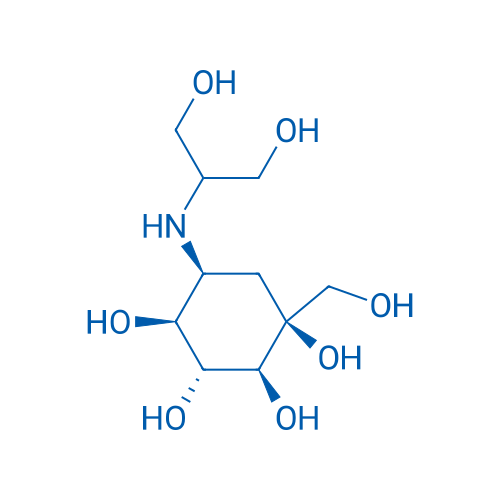

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
