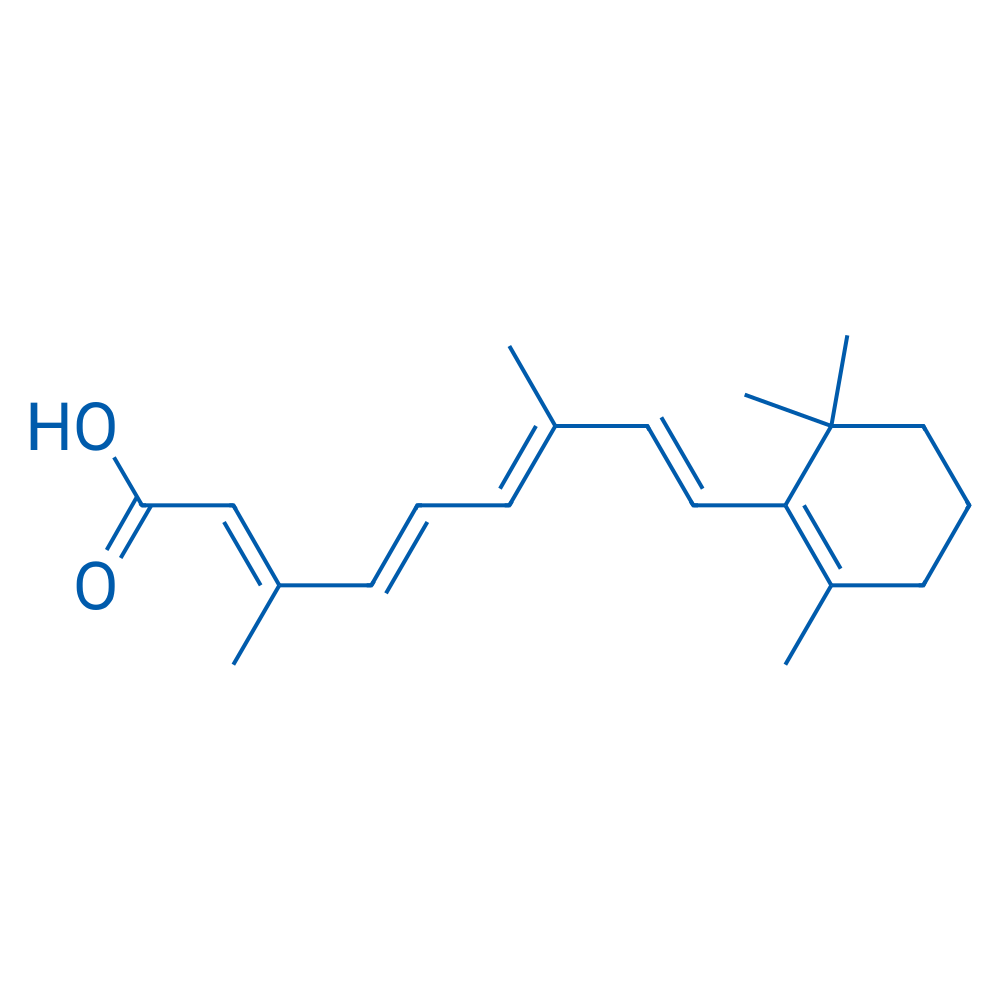
CAS No.: 302-79-4
维甲酸 Catalog No. CSN19418
Synonyms: Vitamin A acid;all-trans-Retinoic acid;ATRA
Retinoic acid is a ligand for retinoic acid receptor (RAR) and retinoid X receptor (RXR) that can prime embryonic stem cells to become neurons and induce granulocytic differentiation.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03291080 Neuroblastoma Phase 1 Phase 2 Recruiting October 28, 2019 United Kingdom ... more >> Bruce Morland Not yet recruiting Birmingham, United Kingdom Contact: Bruce Moreland, MD Dr Antony Ng Recruiting Bristol, United Kingdom Contact: Antony Ng, MD Dr Amos Burke Recruiting Cambridge, United Kingdom Contact: Amos Burke, MD Mark Brougham Not yet recruiting Edinburgh, United Kingdom Contact: Mark Brougham, MD Dr Martin Elliott Recruiting Leeds, United Kingdom Contact: Martin Elliott, MD Dr Lisa Howell Recruiting Liverpool, United Kingdom Contact: Lisa Howell, MD Dr Guiseppe Barone Recruiting London, United Kingdom Contact: Guiseppe Barone, MD Dr Guy Makin Recruiting Manchester, United Kingdom Dr Madhumita Dandapani Recruiting Nottingham, United Kingdom Kate Wheeler Not yet recruiting Oxford, United Kingdom Contact: Kate Wheeler, MD Sucheta Vaidya Recruiting Sutton, United Kingdom Contact: Sucheta Vaidya, MD Collapse << NCT01336387 Healthy, no Evidence of Diseas... more >>e Collapse << Phase 1 Terminated - United States, Wisconsin ... more >> University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << NCT00006220 Leukemia Lymp... more >>homa Multiple Myeloma and Plasma Cell Neoplasm Myelodysplastic Syndromes Collapse << Phase 1 Phase 2 Terminated(Study drug became c... more >>ommercially available.) Collapse << - United States, Missouri ... more >> Washington University Barnard Cancer Center Saint Louis, Missouri, United States, 63110 Collapse << - 更多
- 参考文献
- [1] Uchida G, Yoshimura K, et al. Tretinoin reverses upregulation of matrix metalloproteinase-13 in human keloid-derived fibroblasts. Exp Dermatol. 2003;12 Suppl 2:35-42.
- [2] Manzano VM, Puyol MR, et al. Tretinoin prevents age-related renal changes and stimulates antioxidant defenses in cultured renal mesangial cells. J Pharmacol Exp Ther. 1999 Apr;289(1):123-32.
- [3] Uchida G, Yoshimura K, Kitano Y, Okazaki M, Harii K. Tretinoin reverses upregulation of matrix metalloproteinase-13 in human keloid-derived fibroblasts. Exp Dermatol. 2003;12 Suppl 2:35-42. doi: 10.1034/j.1600-0625.12.s2.6.x. PMID: 14756522.
- [4] Manzano VM, Puyol MR, Puyol DR, Cazaña FJ. Tretinoin prevents age-related renal changes and stimulates antioxidant defenses in cultured renal mesangial cells. J Pharmacol Exp Ther. 1999 Apr;289(1):123-32. PMID: 10086995.
- [5] Guan J, Zhang H, Wen Z, Gu Y, Cheng Y, Sun Y, Zhang T, Jia C, Lu Z, Chen J. Retinoic acid inhibits pancreatic cancer cell migration and EMT through the downregulation of IL-6 in cancer associated fibroblast cells. Cancer Lett. 2014 Apr 1;345(1):132-9. doi: 10.1016/j.canlet.2013.12.006. Epub 2013 Dec 12. PMID: 24334138.
- [6] Park JC, Jeong WJ, Kim MY, Min D, Choi KY. Retinoic-acid-mediated HRas stabilization induces neuronal differentiation of neural stem cells during brain development. J Cell Sci. 2016 Aug 1;129(15):2997-3007. doi: 10.1242/jcs.184366. Epub 2016 May 16. PMID: 27185863.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 302-79-4 | 储存条件 |
|
|||||||
| 分子式 | C20H28O2 | 运输 | 蓝冰 | |||||||
| 分子量 | 300.44 | 别名 | Vitamin A acid;all-trans-Retinoic acid;ATRA;Tretinoin;NSC 122578;NSC 122758;RA;alltrans vitamin A acid;betaretinoic acid;retinoic acid;TRA;trans retinoic acid;trans vitamin A acid;tretinoinum;Vitamin A Acid. US brand names: Aberel;Aknoten;Avita;Renova;RetinA;RetinA MICRO;Vesanoid. Foreign brand names: Airol;Cordes Vas;Dermairol;EpiAberel;Eudyna;RetisolA;StievaA;StievaA Forte;Vitinoin;Abbreviation: ATRA ;Acronyms: ATRA;TRA. Code name: Ro 5488;维生素A酸;维甲酸 | |||||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| COS7 cells | - | Function assay | 16 h | Activity at human RARgamma ligand binding domain expressed in COS7 cells co-transfected with Gal4-DBD assessed as transcriptional activation after 16 hrs by Gal4 response element-driven luciferase reporter gene assay, EC50=0.0002 μM | 19058965 |
| CV-1 cells | - | Function assay | - | Transcriptional activation in CV-1 cells expressing Retinoic acid receptor RAR gamma, EC50=0.0007 μM | 7490725 |
| HL-60 cells | - | Function assay | - | Induction of transglutaminase (TGase) activity in HL-60 cells, EC50=0.005 μM | 8784454 |
| HL-60 cells | - | Function assay | 4 days | Induction of HL-60 cells differentiation over 4 days, ED50=0.12 μM | 9572893 |
| human ACN cell | - | Growth inhibition assay | - | Inhibition of human ACN cell growth in a cell viability assay, IC50=1.64999 μM | SANGER |
| human ATN-1 cell | - | Growth inhibition assay | - | Inhibition of human ATN-1 cell growth in a cell viability assay, IC50=0.0303 μM | SANGER |
| human BPH-1 cell | - | Growth inhibition assay | - | Inhibition of human BPH-1 cell growth in a cell viability assay, IC50=7.43611 μM | SANGER |
| human breast cancer cell SK-BR-3 | - | Proliferation assay | - | Inhibition of ER-negative human breast cancer cell SK-BR-3 proliferation, IC50=0.0017 μM | 12372520 |
| human BV-173 cell | - | Growth inhibition assay | - | Inhibition of human BV-173 cell growth in a cell viability assay, IC50=1.59626 μM | SANGER |
| human COR-L88 cell | - | Growth inhibition assay | - | Inhibition of human COR-L88 cell growth in a cell viability assay, IC50=03.85872 μM | SANGER |
| human CTB-1 cell | - | Growth inhibition assay | - | Inhibition of human CTB-1 cell growth in a cell viability assay, IC50=1.60389 μM | SANGER |
| human D-283MED cell | - | Growth inhibition assay | - | Inhibition of human D-283MED cell growth in a cell viability assay, IC50=0.28262 μM | SANGER |
| human DB cell | - | Growth inhibition assay | - | Inhibition of human DB cell growth in a cell viability assay, IC50=4.76519 μM | SANGER |
| human DEL cell | - | Growth inhibition assay | - | Inhibition of human DEL cell growth in a cell viability assay, IC50=15.3728 μM | SANGER |
| human DU145 cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human DU145 cells assessed as reduction in cell viability after 48 hrs by MTT assay, GI50=11.64 μM | 25701251 |
| human ECC10 cell | - | Growth inhibition assay | - | Inhibition of human ECC10 cell growth in a cell viability assay, IC50=2.38052 μM | SANGER |
| human EW-13 cell | - | Growth inhibition assay | - | Inhibition of human EW-13 cell growth in a cell viability assay, IC50=7.61078 μM | SANGER |
| human EW-3 cell | - | Growth inhibition assay | - | Inhibition of human EW-3 cell growth in a cell viability assay, IC50=0.48028 μM | SANGER |
| human GP5d cell | - | Growth inhibition assay | - | Inhibition of human GP5d cell growth in a cell viability assay, IC50=10.2637 μM | SANGER |
| human HCC1806 cell | - | Growth inhibition assay | - | Inhibition of human HCC1806 cell growth in a cell viability assay, IC50=0.72929 μM | SANGER |
| human HH cell | - | Growth inhibition assay | - | Inhibition of human HH cell growth in a cell viability assay, IC50=0.19554 μM | SANGER |
| human HL-60 cell | - | Growth inhibition assay | - | Inhibition of human HL-60 cell growth in a cell viability assay, IC50=0.28491 μM | SANGER |
| human HT-29 cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human HT-29 cells after 48 hrs by MTT assay, IC50=4.3 μM | 20405849 |
| human HuO-3N1 cell | - | Growth inhibition assay | - | Inhibition of human HuO-3N1 cell growth in a cell viability assay, IC50=0.8316 μM | SANGER |
| human JVM-2 cell | - | Growth inhibition assay | - | Inhibition of human JVM-2 cell growth in a cell viability assay, IC50=1.17316 μM | SANGER |
| human KARPAS-45 cell | - | Growth inhibition assay | - | Inhibition of human KARPAS-45 cell growth in a cell viability assay, IC50=2.59581 μM | SANGER |
| human KU812 cell | - | Growth inhibition assay | - | Inhibition of human KU812 cell growth in a cell viability assay, IC50=0.53609 μM | SANGER |
| human L-363 cell | - | Growth inhibition assay | - | Inhibition of human L-363 cell growth in a cell viability assay, IC50=2.06964 μM | SANGER |
| human LAMA-84 cell | - | Growth inhibition assay | - | Inhibition of human LAMA-84 cell growth in a cell viability assay, IC50=0.99635 μM | SANGER |
| human LNCAP cells | - | Proliferation assay | 48 h | Antiproliferative activity against human LNCAP cells after 48 hrs by MTT assay, IC50=0.5 μM | 19375825 |
| human LNCaP cells | - | Growth inhibition assay | 6 days | Growth inhibition of human LNCaP cells after 6 days by MTT assay, IC50=10 μM | 15615521 |
| human MCF7 cells | - | Growth inhibition assay | 6 days | Growth inhibition of human MCF7 cells after 6 days by MTT assay, IC50=0.58 μM | 15615521 |
| human MC-IXC cell | - | Growth inhibition assay | - | Inhibition of human MC-IXC cell growth in a cell viability assay, IC50=0.15436 μM | SANGER |
| human MDA-MB-231 cells | - | Growth inhibition assay | 5 days | Growth inhibition of human MDA-MB-231 cells after 5 days by MTT assay, GI50=10.85 μM | 18543902 |
| human MDA-MB-453 cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human MDA-MB-453 cells assessed as reduction in cell viability after 48 hrs by MTT assay, GI50=9.51 μM | 25701251 |
| human MEG-01 cell | - | Growth inhibition assay | - | Inhibition of human MEG-01 cell growth in a cell viability assay, IC50=9.61318 μM | SANGER |
| human ML-2 cell | - | Growth inhibition assay | - | Inhibition of human ML-2 cell growth in a cell viability assay, IC50=4.00439 μM | SANGER |
| human MOLT-13 cell | - | Growth inhibition assay | - | Inhibition of human MOLT-13 cell growth in a cell viability assay, IC50=1.145 μM | SANGER |
| human MV-4-11 cell | - | Growth inhibition assay | - | Inhibition of human MV-4-11 cell growth in a cell viability assay, IC50=0.02099 μM | SANGER |
| human NB13 cell | - | Growth inhibition assay | - | Inhibition of human NB13 cell growth in a cell viability assay, IC50=0.25664 μM | SANGER |
| human NB14 cell | - | Growth inhibition assay | - | Inhibition of human NB14 cell growth in a cell viability assay, IC50=7.1271 μM | SANGER |
| human NBsusSR cell | - | Growth inhibition assay | - | Inhibition of human NBsusSR cell growth in a cell viability assay, IC50=1.44987 μM | SANGER |
| human NCI-H1648 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H1648 cell growth in a cell viability assay, IC50=9.73617 μM | SANGER |
| human NCI-H209 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H209 cell growth in a cell viability assay, IC50=2.52132 μM | SANGER |
| human NCI-H526 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H526 cell growth in a cell viability assay, IC50=7.68291 μM | SANGER |
| human NCI-H69 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H69 cell growth in a cell viability assay, IC50=2.26561 μM | SANGER |
| human NCI-H720 cell | - | Growth inhibition assay | - | Inhibition of human NCI-H720 cell growth in a cell viability assay, IC50=1.02621 μM | SANGER |
| human NKM-1 cell | - | Growth inhibition assay | - | Inhibition of human NKM-1 cell growth in a cell viability assay, IC50=8.41009 μM | SANGER |
| human PC3 cells | - | Growth inhibition assay | 6 days | Growth inhibition of human PC3 cells after 6 days by MTT assay, IC50=2 μM | 15615521 |
| human PC3 cells | - | Growth inhibition assay | 5 days | Growth inhibition of human PC3 cells after 5 days by MTT assay, GI50=7.6 μM | 18543902 |
| human RPMI-8226 cell | - | Growth inhibition assay | - | Inhibition of human RPMI-8226 cell growth in a cell viability assay, IC50=0.99334 μM | SANGER |
| human RS4-11 cell | - | Growth inhibition assay | - | Inhibition of human RS4-11 cell growth in a cell viability assay, IC50=4.44737 μM | SANGER |
| human SK-NEP-1 cell | - | Growth inhibition assay | - | Inhibition of human SK-NEP-1 cell growth in a cell viability assay, IC50=0.66231 μM | SANGER |
| human T47D cells | - | Growth inhibition assay | 6 days | Growth inhibition of human T47D cells after 6 days by MTT assay, IC50=0.006 μM | 15615521 |
| human T47D cells | - | Cytotoxic assay | 48 h | Cytotoxicity against human T47D cells assessed as reduction in cell viability after 48 hrs by MTT assay, GI50=0.82 μM | 25701251 |
| human U-266 cell | - | Growth inhibition assay | - | Inhibition of human U-266 cell growth in a cell viability assay, IC50=4.11829 μM | SANGER |
| mouse F9 cells | - | Function assay | 3 days | Induction of terminal differentiation in mouse F9 cells assessed by measuring secreted plasminogen activator activity after 3 days, IC50=0.0001 μM | 17489579 |
| mouse NIH3T3 cells | - | Function assay | - | Activity at RARalpha expressed in mouse NIH3T3 cells by R-SAT assay, EC50=0.00631 μM | 19239230 |
| NB4 cell | - | Function assay | - | Induction of NB4 cell differentiation, ED50=0.61 μM | 9572893 |
| Raji cells | - | Function assay | 48 h | Inhibition of TPA-induced Epstein-Barr virus early antigen activation in Raji cells after 48 hrs, IC50=0.0154 μM | 17503850 |
| T-47D cells | - | Proliferation assay | - | Inhibition of ER positive human breast cancer cell T-47D proliferation, IC50=0.2 μM | 12372520 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03291080 | Neuroblastoma | Phase 1 Phase 2 | Recruiting | October 28, 2019 | United Kingdom ... more >> Bruce Morland Not yet recruiting Birmingham, United Kingdom Contact: Bruce Moreland, MD Dr Antony Ng Recruiting Bristol, United Kingdom Contact: Antony Ng, MD Dr Amos Burke Recruiting Cambridge, United Kingdom Contact: Amos Burke, MD Mark Brougham Not yet recruiting Edinburgh, United Kingdom Contact: Mark Brougham, MD Dr Martin Elliott Recruiting Leeds, United Kingdom Contact: Martin Elliott, MD Dr Lisa Howell Recruiting Liverpool, United Kingdom Contact: Lisa Howell, MD Dr Guiseppe Barone Recruiting London, United Kingdom Contact: Guiseppe Barone, MD Dr Guy Makin Recruiting Manchester, United Kingdom Dr Madhumita Dandapani Recruiting Nottingham, United Kingdom Kate Wheeler Not yet recruiting Oxford, United Kingdom Contact: Kate Wheeler, MD Sucheta Vaidya Recruiting Sutton, United Kingdom Contact: Sucheta Vaidya, MD Collapse << |
| NCT01336387 | Healthy, no Evidence of Diseas... more >>e Collapse << | Phase 1 | Terminated | - | United States, Wisconsin ... more >> University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00006220 | Leukemia Lymp... more >>homa Multiple Myeloma and Plasma Cell Neoplasm Myelodysplastic Syndromes Collapse << | Phase 1 Phase 2 | Terminated(Study drug became c... more >>ommercially available.) Collapse << | - | United States, Missouri ... more >> Washington University Barnard Cancer Center Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT01257906 | MILD TO SEVERE ACNE VULGARIS | Not Applicable | Completed | - | India ... more >> Lotus Labs Pvt. Ltd Bangalore, India, 560 034 Collapse << |
| NCT00156169 | Low HDL Cholesterol | Phase 3 | Completed | - | - |
| NCT02751255 | Multiple Myeloma | Phase 1 Phase 2 | Active, not recruiting | May 1, 2020 | Netherlands ... more >> VU University Medical Center Amsterdam, NH, Netherlands, 1081HV Collapse << |
| NCT02717884 | Acute Myeloid Leukemia ... more >> Myelodysplastic Syndrome Collapse << | Phase 1 Phase 2 | Recruiting | December 2021 | Germany ... more >> Universitätsklinikum Heidelberg Recruiting Heidelberg, Baden-Wuerttemberg, Germany, 69120 Contact: Alwin Krämer, MD, Prof. +49 6221 ext 5637750 a.kraemer@Dkfz-Heidelberg.de Contact: Anne-Marie Geueke +49 6221 ext 568006 anne-marie.geueke@med.uni-heidelberg.de Principal Investigator: Alwin Krämer, MD,Prof. Sub-Investigator: Tilmann Bochtler, MD,PD Dr Universitätsklinik Düsseldorf, Medical School Duesseldorf Recruiting Düsseldorf, Germany, 40225 Contact: Andrea Kündgen, MD, PD Dr. +49 211 811 ext 6338 kuendgen@med.uni-duesseldorf.de Contact: Ulrike Spiegelberg +49 211 811 ext 7714 spiegelberg@med.uni-duesseldorf.de Principal Investigator: Andrea Kündgen, MD, PD Dr. Sub-Investigator: Ulrich Germing, MD, Prof. Universitätsklinikum Frankfurt Main, Medical School Frankfurt Recruiting Frankfurt Main, Germany, 60590 Contact: Tobias Berg, MD, Dr. +49 69 6301 ext 84004 tobias.berg@kgu.de Contact: Gesine Bug, MD, PD Dr. +49 69 6301 ext 7369 gesine.bug@kgu.de Principal Investigator: Tobias Berg, MD, Dr. Sub-Investigator: Gesine Bug, MD, PD Dr. Universitätsklinikum Freiburg, Medical School Freiburg Recruiting Freiburg, Germany, 79106 Contact: Michael Lübbert, MD, Prof. +49 761 270 ext 35340 michael.luebbert@uniklinik-freiburg.de Contact: Alexandra Schulz, MSc +49 761 270 ext 36710 alexandra.schulz@uniklinik-freiburg.de Sub-Investigator: Ralph Wäsch, MD, Prof. Klinikum München rechts der Isar, Medical School Munich rechts der Isar Recruiting München, Munich, Germany, 81675 Contact: Katharina Götze, MD, Prof. +49 89 4140 ext 5618 katharina.goetze@mri.tum.de Contact: Sandra Eckert +49 89 4140 ext 5637 sandra.eckert@mri.tum.de Principal Investigator: Katharina Götze, MD, Prof. Sub-Investigator: Mareike Verbeek, MD, Dr. Universitätsklinikum Tübingen, Medical School Tuebingen Recruiting Tübingen, Tuebingen, Germany, 72076 Contact: Helmut R Salih, MD, Prof. +49 7071 29 ext 83275 helmut.salih@med.uni-tuebingen.de Contact: Heinz Schwarz, SN +49 7071 29 ext 82883 heinz.schwarz@med.uni-tuebingen.de Principal Investigator: Helmut R Salih, MD, Prof. Sub-Investigator: Marcus M Schittenhelm, MD, Dr. Collapse << |
| NCT00002479 | Lymphoma | Phase 2 | Completed | - | United States, Illinois ... more >> Robert H. Lurie Comprehensive Cancer Center, Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00842907 | Photodamage P... more >>hotoaging Collapse << | Phase 2 | Unknown | August 2010 | Brazil ... more >> Federal University of Sao Paulo Sao Paulo, SP, Brazil, 04022000 Collapse << |
| NCT00006239 | Leukemia Myel... more >>odysplastic Syndromes Myelodysplastic/Myeloproliferative Diseases Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States, 21231-2410 Collapse << |
| NCT00952523 | Acne Vulgaris | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Skin Study Center Broomall, Pennsylvania, United States, 19008 Collapse << |
| NCT00867672 | Acute Myeloid Leukemia | Phase 2 | Completed | - | Germany ... more >> Klinikum der Technischen Universität Aachen Aachen, Germany, 52074 Vivantes Klinikum Neukölln Berlin, Germany, 12351 Augusta-Kranken-Anstalt gGmbH Bochum, Germany, 44791 Klinikum Braunschweig Braunschweig, Germany, 38126 DIAKO Ev. Diakonie-Krankenhaus gGmbH Bremen, Germany, 28239 Universitätsklinikum Düsseldorf Düsseldorf, Germany, 40225 Marien Hospital Düsseldorf Düsseldorf, Germany, 40479 Klinikum Esslingen GmbH Esslingen, Germany, 73730 Universität Frankfurt Frankfurt, Germany Medizinische Universitätsklinik Freiburg Freiburg, Germany, 79106 St. Marien-Hospital Hagen Hagen, Germany, 58095 Universitätsklinikum Halle Halle, Germany, 06120 Evangelisches Krankenhaus Hamm gGmbH Hamm, Germany, 59063 Med. Hochschule Hannover Hannover, Germany, 30625 Universitätsklinikum Jena Jena, Germany, 07747 Ortenau Klinikum Lahr-Ettenheim Lahr, Germany, 77933 Caritas Krankenhaus Lebach Lebach, Germany, 66822 Universitätsklinikum Leipzig AöR Leipzig, Germany, 04103 Klinikum Lüdenscheid Lüdenscheid, Germany, 58515 Philipps-Universität Marburg Marburg, Germany, 35032 TU München München, Germany, 86175 University of Münster Medical Center Münster, Germany, 48149 Ortenau Klinikum Offenburg, Germany, 77654 Studienzentrum Onkologie Ravensburg Ravensburg, Germany, 88212 Eberhard Karls Universität Tübingen Tübingen, Germany, 72076 Universitätsklinikum Ulm Ulm, Germany, 89081 Klinikum Villingen-Schwenningen Villingen-Schwenningen, Germany, 78050 Collapse << |
| NCT00838812 | Acne Vulgaris | Phase 3 | Completed | - | Brazil ... more >> Lal Clinica Valinhos, São Paulo, Brazil, 13276-245 Collapse << |
| NCT00952523 | - | - | Completed | - | - |
| NCT01111994 | Acne | Phase 4 | Unknown | April 2010 | United States, Maryland ... more >> Callender Center for Clinical Research Mitchellville, Maryland, United States, 20721 United States, Pennsylvania Society Hill Dermatology Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00151242 | Acute Myeloid Leukemia | Phase 2 Phase 3 | Completed | - | Austria ... more >> Department of Hematology/Oncology, University Hospital Innsbruck Innsbruck, Austria, 6020 Department of Internal Medicine I, Krankenhaus der Barmherzigen Schwestern Linz, Austria, 4010 Medical Department III, St. Johann-Hospital Salzburg, Austria, 5020 Center of Hematology and Oncology, Hanusch-Hospital Wien, Austria, 1140 Germany Department of General Internal Medicine, University Hospital of Bonn Bonn, Germany, 53127 Medical Department I, Hospital Bremen-Mitte Bremen, Germany, 28177 Clinical Center of Hematology and Oncology, University Hospital of Düsseldorf Düsseldorf, Germany, 40225 Department of Hematology and Oncology, Hospital Essen Süd, Ev. Hospital of Essen-Werden Essen, Germany, 45239 Medical Department III, Hematology/Oncology, University of Frankfurt Frankfurt, Germany, 60590 Department of Internal Medicine III, City Hospital Frankfurt am Main - Höchst Frankfurt, Germany, 65929 Internal Medicine I, University of Freiburg Freiburg, Germany, 79106 Medical Department IV, University Hospital of Giessen Giessen, Germany, 35392 Department of Internal Medicine, Wilhelm-Anton-Hospital gGmbH Goch, Germany, 47574 Centre of Internal Medicine, University Hospital Göttingen Göttingen, Germany, 37075 Department of Oncology and Hematology, University Hospital Eppendorf Hamburg, Germany, 20246 Medical Department II, Hematology and Oncology, General Hospital Altona Hamburg, Germany, 22763 Medical Department III, Clinical Center Hanau Hanau, Germany, 63450 Medical Department III, Hospital Hannover-Siloah Hannover, Germany, 30449 Department of Hematology, Hematology and Oncology, Medizinische Hochschule Hannover Hannover, Germany, 30625 Department of Internal Medicine I, University Hospital of Saarland Homburg, Germany, 66421 Medical Department II, City Hospital Karlsruhe gGmbH Karlsruhe, Germany, 76133 Medical Department II, University Hospital of Kiel Kiel, Germany, 24116 Department of Internal Medicine/Hematology and Oncology, Cartias Hospital Lebach Lebach, Germany, 66822 Department of Hematology and Oncology, Hospital of Lüdenscheid Lüdenscheid, Germany, 58505 Department of Hematology and Internal Oncology, University Hospital of Mainz Mainz, Germany, 55101 Medical Department III, Clinical Center Rechts der Isar München, Germany, 81675 Department of Hematology and Oncology, Clinical Center of Oldenburg gGmbH Oldenburg, Germany, 26133 Department of Hematology and Oncology, Caritas Hospital St. Theresia Saarbrücken, Germany, 66113 Department of Oncology, Clinical Center of Stuttgart Stuttgart, Germany, 70174 Medical Department II, Diakonie Hospital Stuttgart, Germany, 70176 Medical Department I, Hospital of Barmherzige Brüder Trier, Germany, 54292 Department of Internal Medicine II, University Hospital of Tübingen Tübingen, Germany, 72076 Medical Clinic II-Hematology/Oncology, Hospital Villingen-Schwenningen Villingen-Schwenningen, Germany, 78050 Medical Department I, Helios Hospital Wuppertal Wuppertal, Germany, 42283 Collapse << |
| NCT00005969 | Lymphoma | Phase 2 | Withdrawn(Study withdrawn.) | - | - |
| NCT02339740 | Acute Promyelocytic Leukemia W... more >>ith PML-RARA Collapse << | Phase 3 | Recruiting | - | - |
| NCT02620813 | Acne Vulgaris | Phase 4 | Recruiting | May 2019 | United States, California ... more >> Dermatology Research Area Recruiting Davis, California, United States, 95616 Contact: Negar Foolad, MAS 916-734-1509 nfoolad@ucdavis.edu Contact: Lauren A Hassoun, BS 916-734-6550 lahassoun@ucdavis.edu Principal Investigator: Raja K Sivamani, MD UC Davis Department of Dermatology Recruiting Sacramento, California, United States, 95816 Contact: Raja Sivamani, MD rajasivamani@gmail.com Collapse << |
| NCT01183897 | Neuroblastoma | Phase 2 | Active, not recruiting | August 2019 | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01243450 | - | - | Completed | - | - |
| NCT00003598 | Cervical Cancer ... more >> Precancerous/Nonmalignant Condition Collapse << | Not Applicable | Completed | - | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109-0752 Collapse << |
| NCT01243450 | Acne | Phase 3 | Completed | - | United States, New Jersey ... more >> Cu-Tech, LLC Mountain Lakes, New Jersey, United States, 07046 Collapse << |
| NCT01048645 | Non-Small-Cell Lung Carcinoma | Phase 2 | Completed | - | Mexico ... more >> National Institute of Cancerología Mexico City, Mexico, 14080 Collapse << |
| NCT00151255 | Acute Myeloid Leukemia | Phase 3 | Completed | - | Austria ... more >> Department of Hematology/Oncology, University Hospital of Innsbruck Innsbruck, Austria, 6020 Department of Internal Medicine I, Krankenhaus der Barmherzigen Schwestern Linz, Austria, 4010 Medical Department III, St. Johann Hospital Salzburg, Austria, 5020 Center of hematology and oncology, Hanusch Hospital Wien, Austria, 1140 Germany Department of Internal Medicine I, Central Hospital of Augsburg Augsburg, Germany, 86856 Department of General Internal Medicine, University Hospital of Bonn Bonn, Germany, 53127 Medical Department I, Hospital of Bremen-Mitte Bremen, Germany, 28177 Department of Hematology and Oncology, Hospital Essen-Süd, Ev. Hospital of Essen-Werden Essen, Germany, 45239 Department of Internal Medicine III, City Hospital Frankfurt am Main - Höchst Frankfurt, Germany, 65929 Medical Department IV, University Hospital of Giessen Giessen, Germany, 35392 Department of Internal Medicine, Wilhelm-Anton Hospital gGmbH Goch, Germany, 47574 Centre of Internal Medicine, University Hospital of Göttingen Göttingen, Germany, 37075 Department of Oncology and Hematology, University Hospital Eppendorf Hamburg, Germany, 20246 Medical Department II, General Hospital Altona Hamburg, Germany, 22763 Medical Department III, Hospital of Hanau Hanau, Germany, 63450 Medical Department III, Hospital of Hannover-Siloah Hannover, Germany, 30449 Department of Internal Medicine I, University Hospital of Saarland Homburg, Germany, 66421 Medical Department II, City Hospital Karlsruhe gGmbH Karlsruhe, Germany, 76133 Medical Department II, University Hospital of Kiel Kiel, Germany, 24116 Department of Internal Medicine/Hematology and Oncology, Caritas Hospital Lebach Lebach, Germany, 66822 Department of Hematology/Oncology, Clinical Center of Lüdenscheid Lüdenscheid, Germany, 58515 Medical Department III, Clinical Center Rechts der Isar München, Germany, 81675 Department of Hematology and Oncology, Clinical Center of Oldenburg gGmbH Oldenburg, Germany, 26133 Department of Hematology and Oncology/Caritas Hospital St. Theresa Saarbrücken, Germany, 66113 Department of Oncology, Clinical Center of Stuttgart Stuttgart, Germany, 70174 Medical Department II, Diakonie Hospital Stuttgart, Germany, 70176 I. Medical Department, Hospital of Barmerzigen Brüder Trier, Germany, 54292 Medical Center II - Hematology/Oncology, Clinical Center Villingen-Schwenningen Villingen - Schwenningen, Germany, 78050 Medical Department I, Helios Hospital Wuppertal Wuppertal, Germany, 42283 Collapse << |
| NCT00339196 | Acute Myelogenous Leukaemia (A... more >>ML) Myelodysplastic Syndrome (MDS) Collapse << | Phase 2 | Completed | - | France ... more >> Chu Saint Louis Paris, France, 75010 Collapse << |
| NCT01237808 | Acute Myeloid Leukemia (AML) | Phase 3 | Completed | - | Germany ... more >> Ubbo-Emmius Klinik Aurich Aurich, Germany, 26603 Charité Universitätsmedizin Berlin Berlin, Germany, 13353 University Hospital of Bonn Bonn, Germany, 53111 Städtisches Klinikum Braunschweig Braunschweig, Germany, 38114 Klinikum Bremen-Mitte gGmbH Bremen, Germany, 28177 Kliniken Essen Süd, Evangelischs Krankenhaus Essen, Germany, 45239 Klinikum Esslingen Esslingen, Germany, 73730 Medizinische Universitätsklinik Freiburg, Germany, 79106 Medizinisches Versorgungszentrum Osthessen GmbH Fulda, Germany, 36043 Universitätsklinikum Gießen Gießen, Germany, 35392 Wilhelm- Anton- Hospital gGmbH Goch, Germany, 47574 Universitätsmedizin Göttingen Göttingen, Germany, 37075 University Hospital of Hamburg Eppendorf Hamburg, Germany, 20246 Asklepios Klinik Altona Hamburg, Germany, 22763 Medical Department III, Hospital of Hannover-Siloah Hannover, Germany, 30449 Medizinische Hochschule Hannover Hannover, Germany, 30625 SLK-Kliniken Heilbronn GmbH Heilbronn, Germany, 74078 Department of Internal Medicine I, University Hospital of Saarland Homburg, Germany, 66421 Staedtisches Klinikum Karlsruhe Karlsruhe, Germany, 76133 Department of Interial Medicine /Hematology and Oncology, Caritas Hospital Lebach Lebach, Germany, 66822 Klinikum der Johannes Gutenberg Universität Mainz Mainz, Germany, 55131 Klinikum rechts der Isar der TU Muenchen Muenchen, Germany, 81675 Stauferklinikum Schwäbisch Gmünd Mutlangen, Germany, 73557 Pius Hospital Oldenburg Oldenburg, Germany, 26133 Krankenhaus der Barmherzigen Brueder Regensburg, Germany, 93049 Caritas-Klinik St. Theresia Saarbrücken, Germany, 66113 Clinikal Cetner of Stuttgart, Center of Oncology Stuttgart, Germany, 70174 Diakonie-Klinikum Stuttgart Stuttgart, Germany, 70176 Krankenhaus der Barmherzigen Brüder Trier Trier, Germany, 54292 Universitätsklinikum Tübingen Tübingen, Germany, 72076 University hospital of Ulm Ulm, Germany, 89081 Medical Center II - Hematology/Oncology, Clinical Center Villingen-Schwenningen Villingen - Schwenningen, Germany, 78050 Helios Klinikum Wuppertal Wuppertal, Germany, 42283 Collapse << |
| NCT01041833 | Non Small Cell Lung Cancer | Phase 3 | Unknown | September 2013 | Mexico ... more >> Department of Medical Oncology, Instituto Nacional de Cancerología Not yet recruiting Mexico, Mexico, 14080 Contact: Oscar Arrieta, M.D. (+52) (55) 5628-0400 ext 832 ogar@servidor.unam.mx Contact: Oscar Arrieta (+52) (55) 5628-0400 ext 832 ogar@servidor.unam.mx Collapse << |
| NCT01047189 | Acne Vulgaris | Phase 4 | Completed | - | United States, North Carolina ... more >> Wake Forest University Health Sciences, Dermatology Winston-Salem, North Carolina, United States, 27157 Collapse << |
| NCT01047189 | - | - | Completed | - | - |
| NCT02704507 | Acne | Not Applicable | Unknown | June 2016 | - |
| NCT00823901 | Rosacea | Phase 2 Phase 3 | Completed | - | United States, California ... more >> Department of Dermatology - Stanford School of Medicine Stanford, California, United States, 94305 United States, Massachusetts CURTIS - Massachussetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00823901 | - | - | Completed | - | - |
| NCT00528450 | - | - | Terminated(Lack of accrual) | - | - |
| NCT00528450 | Leukemia | Phase 2 | Terminated(Lack of accrual) | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00919555 | Amyotrophic Lateral Sclerosis | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> Phoenix Neurological Associates, LTD Phoenix, Arizona, United States, 85018 Collapse << |
| NCT00200161 | - | - | Completed | - | - |
| NCT00200161 | Glioblastoma ... more >>Gliomas Collapse << | Phase 2 | Completed | - | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00326170 | Myelodysplastic Syndrome ... more >> Acute Myelogenous Leukemia Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> The University of Texas M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00969722 | Primary Refractory Neuroblasto... more >>ma Collapse << | Phase 2 | Terminated(Lack of enrollment.... more >>) Collapse << | - | United States, Arizona ... more >> Phoenix Children's Hospital Phoenix, Arizona, United States, 85016 United States, California Rady Children's Hospital of San Diego San Diego, California, United States, 92123 United States, District of Columbia Georgetown Medical Center Washington, District of Columbia, United States, 20057 United States, Florida All Children's Hospital in Florida St. Petersburg, Florida, United States, 33701 United States, Louisiana LSU Health Sciences Center; Children's Hospital New Orleans, Louisiana, United States, 70118 United States, Minnesota Children's Hospitals and Clinics of Minnesota Minneapolis, Minnesota, United States, 55404 United States, New York Children's Hospital at Montefiore Bronx, New York, United States, 10467 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27705 United States, Ohio Nationwide Childrens Hospital Columbus, Ohio, United States, 43205 United States, Oklahoma University of Oklahoma Cancer Center Oklahoma City, Oklahoma, United States, 73104 United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 United States, Texas US Oncology Dallas, Texas, United States, 75230 The University of Texas M.D. Anderson Cancer Center Houston, Texas, United States, 77030 United States, Utah University of Utah Medical Center Salt Lake City, Utah, United States, 84113 United States, Vermont Vermont Cancer Center Burlington, Vermont, United States, 05405 Collapse << |
| NCT03392623 | Melasma | Early Phase 1 | Completed | - | - |
| NCT00617409 | - | - | Active, not recruiting | - | - |
| NCT00617409 | Small Cell Lung Cancer | Phase 2 | Active, not recruiting | December 2019 | United States, Florida ... more >> H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01915758 | Acne Vulgaris | Phase 1 | Completed | - | - |
| NCT00326170 | - | - | Completed | - | - |
| NCT00413166 | Acute Promyelocytic Leukemia | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01667263 | Autoimmune Thrombocytopenia | Phase 2 | Completed | - | China, Beijing ... more >> Beijing Hospital, Ministry of Health Beijing, Beijing, China, 100044 Peking University People's Hospital, Peking University Insititute of Hematology Beijing, Beijing, China, 100044 Beijing Tongren Hospital Beijing, Beijing, China PLA Navy General Hospital Beijing, Beijing, China China, Shandong Qilu Hospital, Shandong University Jinan, Shandong, China Collapse << |
| NCT02842827 | Acute Myeloid Leukemia ... more >> Myelodysplastic Syndrome Collapse << | Phase 1 | Active, not recruiting | December 2018 | Australia, South Australia ... more >> Royal Adelaide Hospital Adelaide, South Australia, Australia Collapse << |
| NCT01276730 | Cervical Cancer | Phase 2 | Completed | - | India ... more >> Chittaranjan National Cancer Institute Kolkata, India, 700026 Collapse << |
| NCT01135069 | Acne | Phase 3 | Completed | - | United States, Florida ... more >> Moore Clinical Research Land O Lakes, Florida, United States, 34628 Collapse << |
| NCT00603772 | Healthy | Phase 1 | Completed | - | United States, North Carolina ... more >> Suncare Research Laboratories Winston-Salem, North Carolina, United States, 27106 Collapse << |
| NCT01135069 | - | - | Completed | - | - |
| NCT00440024 | Sun-Damaged Skin ... more >> Retinoid Intolerance Collapse << | Not Applicable | Completed | - | United States, Michigan ... more >> University of Michigan Department of Dermatology Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00382200 | Myelodysplastic Syndromes | Phase 1 Phase 2 | Suspended | July 2019 | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01313728 | - | - | Completed | - | - |
| NCT01313728 | Acne Vulgaris | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Skin Study Center Broomall, Pennsylvania, United States, 19008 Collapse << |
| NCT01522456 | - | - | Completed | - | - |
| NCT01916187 | Neuroblastoma | Phase 1 | Withdrawn(drug company withdrw... more >>w support following a re-examination of the benefit-risk assessment for the investigational use of imetelstat in this population) Collapse << | - | Canada, Quebec ... more >> CHU Sainte-Justine Montreal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT00756288 | Actinic Keratoses | Not Applicable | Withdrawn | December 2017 | - |
| NCT00007631 | Carcinoma, Basal Cell ... more >> Carcinoma, Squamous Cell Skin Neoplasms Collapse << | Phase 3 | Completed | - | United States, Arizona ... more >> Carl T. Hayden VA Medical Center Phoenix, Arizona, United States, 85012 United States, California VA Medical Center, Long Beach Long Beach, California, United States, 90822 United States, Florida VA Medical Center, Miami Miami, Florida, United States, 33125 United States, Illinois Edward Hines, Jr. VA Hospital Hines, Illinois, United States, 60141-5000 United States, North Carolina VA Medical Center, Durham Durham, North Carolina, United States, 27705 United States, Oklahoma VA Medical Center, Oklahoma City Oklahoma City, Oklahoma, United States, 73104 United States, Rhode Island VA Medical Center, Providence Providence, Rhode Island, United States, 02908 Collapse << |
| NCT00717652 | Melasma | Phase 2 Phase 3 | Suspended | - | Brazil ... more >> Lal Clinica Pesquisa E Desenvolvimento Ltda Valinhos, S, Brazil, 13270000 Collapse << |
| NCT00835198 | Acne Vulgaris | Phase 4 | Completed | - | United States, Ohio ... more >> Strongsville, Ohio, United States Collapse << |
| NCT01522456 | Acne Vulgaris | Phase 4 | Completed | - | United States, Texas ... more >> Stephens and Associates Dallas, Texas, United States, 75006 Collapse << |
| NCT00621218 | Rosacea | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Product Investigations, Inc. Conshohocken, Pennsylvania, United States, 19428 Collapse << |
| NCT00835198 | - | - | Completed | - | - |
| NCT00939133 | Acne | Phase 4 | Completed | - | Canada, Ontario ... more >> Dermatrials Research Hamilton, Ontario, Canada, L8N 1V6 Collapse << |
| NCT00647556 | Photoaging | Phase 3 | Completed | - | United States, Michigan ... more >> University of Michigan Department of Dermatology Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00240513 | Acne Vulgaris | Phase 4 | Terminated(Too much difficulty... more >> recruiting and retaining patients, PI decided to stop.) Collapse << | - | Canada, British Columbia ... more >> Derm Research @ 888 Inc Vancouver, British Columbia, Canada, V5Z 3Y1 Collapse << |
| NCT00001438 | HIV Infections ... more >> Immunologic Deficiency Syndromes Lymphoproliferative Disorders Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01261923 | Hepatic Insufficiency | Phase 1 Phase 2 | Completed | - | Denmark ... more >> Gentofte Hospital Hellerup, Denmark, 2900 Collapse << |
| NCT00892190 | Acute Myelogenous Leukemia ... more >> Myelodysplastic Syndrome Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Cancer Institute - Hillman Cancer Center Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT01422785 | Acne Vulgaris | Phase 4 | Unknown | - | United States, New York ... more >> Mt Sinai Hospital - Dept of Dermatology Recruiting New York, New York, United States, 10029 Contact: Vicky Wong 212-241-3288 Principal Investigator: Joshua Zeichner, MD Collapse << |
| NCT00647556 | - | - | Completed | - | - |
| NCT02249767 | Acne | Phase 3 | Completed | - | - |
| NCT01027793 | Stretch Marks ... more >> Striae Treatment Collapse << | Phase 4 | Completed | - | Brazil ... more >> Brazilian Center for Studies in Dermatology Porto Alegre, Rio Grande do Sul, Brazil, 90570 040 Collapse << |
| NCT02278861 | Actinic Keratosis | Phase 2 | Completed | - | Brazil ... more >> Dermatology Outpatient Clinic - Hospital das Clínicas - Federal University of Goias (UFG) Goiânia, Goiás, Brazil, 74605-020 Collapse << |
| NCT02261779 | AML | Phase 1 Phase 2 | Unknown | September 2017 | Germany ... more >> Universitaetsklinikum Halle Recruiting Halle, Germany, D-06120 Contact: Contact Person Collapse << |
| NCT02410980 | Collagen Fibril Alteration | Early Phase 1 | Completed | - | United States, California ... more >> Beckman Laser Institute Medical Clinic Irvine, California, United States, 92612 Collapse << |
| NCT01283464 | Photodamaged Skin ... more >> Wrinkles Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Johns Hopkins Dept. of Dermatology Baltimore, Maryland, United States, 21231 Collapse << |
| NCT01935960 | Healthy Subject | Phase 1 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Cancer Center Birmingham, Alabama, United States, 35233 United States, Iowa University of Iowa/Holden Comprehensive Cancer Center Iowa City, Iowa, United States, 52242 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00001504 | Breast Cancer ... more >> Breast Neoplasm Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01283464 | - | - | Completed | - | - |
| NCT01668615 | - | - | Completed | - | China, Jiangsu ... more >> Jiangsu Institute of Hematology Suzhou, Jiangsu, China, 215006 Collapse << |
| NCT01125930 | Rosacea | Phase 3 | Terminated(Due to slow recruit... more >>ment and sponsor request study ended early) Collapse << | - | United States, Michigan ... more >> University of Michigan Department of Dermatology Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00002586 | Drug Toxicity ... more >> Lung Cancer Collapse << | Phase 2 | Completed | - | United States, Colorado ... more >> University of Colorado Cancer Center at University of Colorado Health Sciences Center Denver, Colorado, United States, 80010 National Jewish Center for Immunology and Respiratory Medicine Denver, Colorado, United States, 80206 Lung Cancer Institute Denver, Colorado, United States, 80218 Veterans Affairs Medical Center - Denver Denver, Colorado, United States, 80220 Collapse << |
| NCT00001509 | Nephroblastoma ... more >> Neuroblastoma Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03433378 | Acne Vulgaris | Early Phase 1 | Completed | - | United States, North Carolina ... more >> Catawba Research, LLC Charlotte, North Carolina, United States, 28217 Collapse << |
| NCT03427554 | Acne Vulgaris | Early Phase 1 | Completed | - | United States, North Carolina ... more >> Catawba Research, LLC Charlotte, North Carolina, United States, 28217 Collapse << |
| NCT01820624 | Adult Acute Megakaryoblastic L... more >>eukemia (M7) Adult Acute Monoblastic Leukemia (M5a) Adult Acute Monocytic Leukemia (M5b) Adult Acute Myeloblastic Leukemia With Maturation (M2) Adult Acute Myeloblastic Leukemia Without Maturation (M1) Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Myelomonocytic Leukemia (M4) Adult Erythroleukemia (M6a) Adult Pure Erythroid Leukemia (M6b) Recurrent Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Completed | - | United States, Ohio ... more >> University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Collapse << |
| NCT01125930 | - | - | Terminated(Due to slow recruit... more >>ment and sponsor request study ended early) Collapse << | - | - |
| NCT00136461 | Acute Myelogenous Leukemia ... more >> Myelodysplastic Syndrome Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01406080 | Photoaging Ph... more >>otodamage Collapse << | Phase 3 | Completed | - | Brazil ... more >> Centro de Dermatologia Dona Libania Fortaleza, Ceará, Brazil, 60035-101 Santa Casa de Belo Horizonte Belo Horizonte, Minas Gerais, Brazil, 30150-221 Hospital de Clínicas da Universidade Federal do Paraná Curitiba, Paraná, Brazil, 80060-900 Universidade Federal de São Paulo - UNIFESP - UNICCO São Paulo, Brazil, 04022-000 Collapse << |
| NCT01183884 | Neuroblastoma | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01404949 | Acute Promyelocytic Leukemia | Phase 2 | Active, not recruiting | July 2019 | United States, California ... more >> University of Southern California Los Angeles, California, United States, 90033 United States, Illinois Northwestern University Evanston, Illinois, United States, 60208 United States, Maryland National Heart, Lung, and Blood Institute (NIH) Bethesda, Maryland, United States, 20824 United States, New Jersey Memorial Sloan Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 Memorial Sloan Kettering Monmouth Middletown, New Jersey, United States, 07748 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 New York Presbyterian Hospital-Weill Medical College of Cornell University New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 Canada, Ontario Princess Margaret Hospital/Ontario Cancer Institute Toronto, Ontario, Canada Collapse << |
| NCT03359174 | Cholangitis, Sclerosing | Phase 2 | Recruiting | January 1, 2021 | United States, Connecticut ... more >> Yale University Recruiting New Haven, Connecticut, United States, 06510 Collapse << |
| NCT01987297 | Acute Promyelocytic Leukemia | Phase 4 | Unknown | October 2017 | China ... more >> Department of Hematology Recruiting Shanghai, China, 200025 Contact: Jiong HU hujiong@medmail.com.cn Principal Investigator: Jun-min Li, M.D Collapse << |
| NCT01183416 | Neuroblastoma | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01349556 | Medication Reaction | Not Applicable | Withdrawn(Study withdrawn due ... more >>to volunteer disinterest in participating.) Collapse << | - | - |
| NCT00528437 | Brain Tumors | Phase 2 | Active, not recruiting | July 2019 | United States, Arizona ... more >> Phoenix Children's Hospital Phoenix, Arizona, United States, 85016 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Hawaii Hawaii Pacific Health Lihue, Hawaii, United States, 96766 United States, Illinois Children's Memorial Hospital Chicago, Illinois, United States, 60614 United States, Indiana Riley Hospital for Children Indianapolis, Indiana, United States, 46202 United States, Minnesota Children's Hospital and Clinics of Minnesota Minneapolis, Minnesota, United States, 55404 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Steven and Alexandra Cohen Children's Medical Center of New York- North Shore LIJ New Hyde Park, New York, United States, 11040 NYU Hassenfeld Center New York, New York, United States, 10016 United States, Ohio Nationwide Children's Hospital Columbus, Ohio, United States, 43205 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, South Carolina Medical Univ. of South Carolina Charleston, South Carolina, United States, 29425 United States, Tennessee Vanderbilt Univ. Nashville, Tennessee, United States, 37240 United States, Texas Children's Medical Center of Dallas Dallas, Texas, United States, 75235 MD Anderson Cancer Center (MDACC) Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth Univ. Richmond, Virginia, United States, 23284 Canada, Ontario Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01183429 | Neuroblastoma | Phase 2 | Active, not recruiting | August 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01275274 | Anti-Neutrophil Cytoplasmic An... more >>tibody-Associated Vasculitis Collapse << | Phase 2 | Withdrawn(no subject enrolled ... more >>in nearly 2 years) Collapse << | - | United States, North Carolina ... more >> UNC Kidney Center Chapel Hill, North Carolina, United States, 27599-7155 Collapse << |
| NCT01882127 | Purpura Idiop... more >>athic Thrombocytopenic Purpura Collapse << | Phase 3 | Withdrawn(No eligible patient ... more >>was enrolled.) Collapse << | - | China, Shandong ... more >> Qilu Hospital, Shandong University Jinan, Shandong, China, 250000 Collapse << |
| NCT02439099 | - | - | Recruiting | December 2018 | Germany ... more >> Department of Psychiatry, Charité - Campus Benjamin Franklin Recruiting Berlin, Germany, 12203 Collapse << |
| NCT02497833 | Hypercholesterolemia | Early Phase 1 | Completed | - | China, Guangdong ... more >> Guangdong Provincial Key Laboratory of Food, Nutrition and Health, Department of Nutrition, School of Public Health, Sun Yat-Sen University (Northern Campus) Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT01448733 | Acne Vulgaris | Phase 4 | Unknown | October 2011 | United States, New York ... more >> Mt Sinai Hospital, Department of Dermatology New York, New York, United States, 10029 Collapse << |
| NCT01985477 | Myeloma | Phase 1 | Terminated(Terminated Phase I ... more >>due to slow accrual without progression to Phase II.) Collapse << | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02403778 | Melanoma | Phase 2 | Active, not recruiting | March 2020 | United States, Colorado ... more >> University of Colorado Hospital Aurora, Colorado, United States, 80045 Collapse << |
| NCT00533169 | Neuroblastoma | Phase 1 | Terminated(Lack of enrollment.... more >>) Collapse << | - | United States, Texas ... more >> U.T.M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02061384 | Male Infertility ... more >> Klinefelter's Syndrome Y-chromosome Microdeletions Collapse << | Phase 2 | Active, not recruiting | December 2018 | United States, Washington ... more >> University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT01048086 | Neuroblastoma ... more >> Neuroendocrine Tumor Collapse << | Phase 2 | Withdrawn(No patients were enr... more >>olled because the company that pledged the drug does not yet have it available.) Collapse << | - | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52242 Collapse << |
| NCT03611595 | Solid Tumor | Phase 1 | Not yet recruiting | September 2020 | United States, California ... more >> UC San Diego Moores Cancer Center La Jolla, California, United States, 92093 Collapse << |
| NCT03323801 | Male Infertility, Azoospermia | Phase 2 | Recruiting | December 31, 2019 | United States, Washington ... more >> University of Washington Medical Center Recruiting Seattle, Washington, United States, 98195 Contact: Iris Nielsen 206-221-5473 nielseni@uw.edu Contact: Kathy Winter 206-616-0484 klwinter@uw.edu Principal Investigator: John K Amory, MD, MPH Collapse << |
| NCT01456468 | Cholangitis, Sclerosing | Phase 1 | Completed | - | United States, Connecticut ... more >> Yale University School of Medicine - 333 Cedar St - 1080 LMP New Haven, Connecticut, United States, 06520 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01891526 | - | - | Completed | - | Denmark ... more >> Gentofte Hospital Gentofte, Denmark, 2900 Collapse << |
| NCT03200847 | Stage IV Melanoma ... more >> Stage III Melanoma Advanced Melanoma Collapse << | Phase 1 Phase 2 | Recruiting | May 2019 | United States, Colorado ... more >> University of Colorado Denver Recruiting Aurora, Colorado, United States, 80045 Contact: Christine Christensen 720-848-0592 christine.christensen@ucdenver.edu Principal Investigator: Martin McCarter, MD Collapse << |
| NCT00002143 | HIV Infections ... more >> Psoriasis Collapse << | Phase 3 | Completed | - | United States, New York ... more >> Beth Israel Med Ctr New York, New York, United States, 10003 Collapse << |
| NCT01472900 | Acne Vulgaris | Not Applicable | Completed | - | Thailand ... more >> Faculty of Medicine, Chulalongkorn University Bangkok, Thailand, 10330 Collapse << |
| NCT01184274 | Leukemia Soli... more >>d Tumours Collapse << | Phase 1 | Completed | - | Canada, Alberta ... more >> Alberta Children's Hospital Calgary, Alberta, Canada, T3B 6A8 Stollery Children's Hospital Edmonton, Alberta, Canada, T6G 2B7 Canada, British Columbia Children's and Women's Health Centre of BC Branch Vancouver, British Columbia, Canada, V6H 3V4 Canada, Manitoba CancerCare Manitoba Winnipeg, Manitoba, Canada, R3E 0V9 Canada, Nova Scotia Izaak Walton Killam (IWK) Health Centre Halifax, Nova Scotia, Canada, B3K 6R8 Canada, Ontario Children's Hospital of Eastern Ontario Ottawa, Ontario, Canada, K1H 8L1 Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Canada, Quebec CHU Sainte-Justine Montreal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT00520208 | Acute Promyelocytic Leukemia | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 United States, Texas UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00624676 | Acne Vulgaris | Not Applicable | Completed | - | Canada, Quebec ... more >> Innovaderm Research Laval, Quebec, Canada Innovaderm Research Montreal, Quebec, Canada Collapse << |
| NCT00723437 | Psoriasis | Phase 4 | Completed | - | Italy ... more >> Department of Dermatology University of Bologna Bologna, Italy, 40138 Collapse << |
| NCT01982578 | Alzheimer's Disease | Not Applicable | Unknown | September 2018 | Spain ... more >> Hospital de La Ribera Recruiting Alzira, Valencia, Spain, 46600 Contact: Francisco Tarazona, MD +34 962 458 100 FJTarazona@Hospital-Ribera.com Hospital Clínico Universitario de Valencia Recruiting Valencia, Spain, 46010 Contact: José Miguel Lainez, M.D. 963 86 26 00 jlaineza@medynet.com Universitat de València Recruiting Valencia, Spain, 46010 Contact: Jose Viña, MD PhD (hon) 0034 963864646 ext 64650 jose.vina@uv.es Principal Investigator: Jose Viña, MD PhD (hon) Collapse << |
| NCT01743053 | Venous Leg Ulcers | Phase 4 | Completed | - | France ... more >> Hôpital Lapeyronie Montpellier, France United Kingdom Bradford Teaching Hospitals Bradford, United Kingdom, BD9 6RJ Cambridge University Hospitals - Addenbrooke's Hospital Cambridge, United Kingdom, CB2 0QQ Cardiff University Cardiff, United Kingdom, CF24 0DE Doncaster and Bassetlaw Hospitals Doncaster, United Kingdom, DN2 5LT The Leeds Teaching Hospitals - James's University Hospital Leeds, United Kingdom, LS9 7TF University Hospital of South Manchester - Wythenshawe Hospital Manchester, United Kingdom, M23 9LT Collapse << |
| NCT02992249 | - | - | Suspended(Program under evalua... more >>tion following FDA approval of the RECELL device.) Collapse << | - | - |
| NCT02799121 | Diabetic Foot Ulcer | Phase 4 | Recruiting | - | United Kingdom ... more >> King's College Hospital, London Recruiting London, United Kingdom, SE5 9RS Contact: Naveen Cavale naveen.cavale@nhs.net Principal Investigator: Naveen Cavale Northwick Park Hospital Recruiting London, United Kingdom Contact: Cheryl Pavel cpavel@nhs.net Principal Investigator: Sophie Renton, Miss Manchester Royal Infirmary Recruiting Manchester, United Kingdom, M13 9WL Contact: Tawqeer Rashid Tawqeer.rashid@cmft.nhs.uk Contact: Frank Bowling Frank.Bowling@manchester.ac.uk Principal Investigator: Tawqeer Rashid Collapse << |
| NCT02994654 | Burns | Not Applicable | Active, not recruiting | December 2018 | United States, Arizona ... more >> Arizona Burn Center at Maricopa Integrated Health Systems Phoenix, Arizona, United States, 85008 United States, District of Columbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 United States, Florida Tampa General Hospital Tampa, Florida, United States, 33606 United States, North Carolina Wake Forest Baptist Medical Center Winston-Salem, North Carolina, United States, 27157 United States, Tennessee University of Tennessee Health Science Center Memphis, Tennessee, United States, 38103-3409 United States, Texas U.S.Army Institute of Surgical Research Fort Sam Houston, Texas, United States, 78234-7767 Collapse << |
| NCT00055991 | Breast Cancer | Phase 1 | Completed | - | United States, District of Col... more >>umbia Lombardi Comprehensive Cancer Center at Georgetown University Medical Center Washington, District of Columbia, United States, 20007 United States, Texas M.D. Anderson Cancer Center at University of Texas Houston, Texas, United States, 77030-4009 Dan L. Duncan Cancer Center at Baylor College of Medicine Houston, Texas, United States, 77030 Cancer Therapy and Research Center San Antonio, Texas, United States, 78229 Collapse << |
| NCT03626701 | Burns | Not Applicable | Not yet recruiting | August 31, 2021 | - |
| NCT02380612 | Burns | Not Applicable | Completed | - | United States, Arizona ... more >> Arizona Burn Center at Maricopa Medical Center Phoenix, Arizona, United States, 85008 United States, District of Columbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 United States, Florida Tampa General Hospital Tampa, Florida, United States, 33606 United States, North Carolina North Carolina Jaycee Burn Center Chapel Hill, North Carolina, United States, 27599 Wake Forest Baptist Medical Center Winston-Salem, North Carolina, United States, 27157 United States, Tennessee University of Tennessee Health Science Center Memphis, Tennessee, United States, 38103-3409 United States, Texas USAISR - US Army Institute of Surgical Research Fort Sam Houston, Texas, United States, 78234 Collapse << |
| NCT03233165 | Leukoplakic Lesions | Not Applicable | Completed | - | - |
| NCT03333941 | - | - | Active, not recruiting | June 2019 | United States, Arizona ... more >> Arizona Burn Center at Maricopa Integrated Health Systems Phoenix, Arizona, United States, 85008 United States, District of Columbia MedStar Washington Hospital Center Washington, District of Columbia, United States, 20010 United States, Florida Tampa General Hospital Tampa, Florida, United States, 33606 United States, Louisiana University Medical Center New Orleans, Louisiana, United States, 70112 United States, North Carolina Wake Forest Baptist Medical Center Winston-Salem, North Carolina, United States, 27157 United States, Tennessee University of Tennessee Health Science Center Memphis, Tennessee, United States, 38103-3409 Collapse << |
| NCT03049319 | Healthy Participants and/or Pa... more >>tients Collapse << | Not Applicable | Active, not recruiting | December 2018 | United States, North Carolina ... more >> Elias Oussedik Winston-Salem, North Carolina, United States, 27106 Collapse << |
| NCT00793845 | Neuroblastoma | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT03624192 | Skin; Deformity | Not Applicable | Recruiting | December 31, 2020 | United States, Arizona ... more >> Arizona Burn Center at Maricopa Intergrated Health Systems Recruiting Phoenix, Arizona, United States, 85008 Contact: Morgan Baker, MSN, RN 602-344-5977 Morgan.Baker@mihs.org Principal Investigator: Kevin Foster, MD, MBA, FACS Collapse << |
| NCT01479855 | - | - | Unknown | December 2013 | Norway ... more >> OsloUH, Ullevål Recruiting Oslo, Norway, 0424 Principal Investigator: Ingun D Ulstein, MD PhD Collapse << |
| NCT03061656 | High Risk Neuroblastoma | Phase 2 | Active, not recruiting | December 31, 2018 | - |
| NCT01306955 | Behcet's Disease | Phase 4 | Unknown | August 2011 | Iran, Islamic Republic of ... more >> Rheumatology Research Center, Tehran University of Medical Science Recruiting Tehran, Iran, Islamic Republic of, 1411713135 Contact: Shahram Farhad, Professor 0098-21-8802-6956 Shahramf@tums.ac.ir Contact: Mastaneh Mohammadi, MD 0098-21-8802-6956 Mastanehmohammadi88@gmail.com Collapse << |
| NCT01782742 | - | - | Completed | - | - |
| NCT01569724 | Hypertriglyceridemia ... more >> Cutaneous T Cell Lymphoma Collapse << | Phase 4 | Completed | - | France ... more >> Service de dermatologie - Hôpital de Pontchaillou Rennes, France, 35033 Collapse << |
| NCT02693171 | - | - | Terminated(Significant short a... more >>nd intermediate-term drug supply shortages.) Collapse << | December 2018 | United States, Alabama ... more >> University Of Alabama At Birmingham Birmingham, Alabama, United States, 35233 United States, Arkansas Arkansas Children's Hospital Little Rock, Arkansas, United States, 72202 United States, California Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Children's Hospital of Orange County Orange, California, United States, 92868 Rady Children's Hospital- San Diego San Diego, California, United States, 92123 United States, Illinois University of Chicago Chicago, Illinois, United States, 60637 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Michigan University of Michigan - C S Mott Children's Hospital Ann Arbor, Michigan, United States, 48109 United States, Minnesota Children's Hospitals and Clinics of Minnesota - Minneapolis Minneapolis, Minnesota, United States, 55404 United States, Missouri Children's Mercy Hospital Kansas City, Missouri, United States, 64108 United States, New York Columbia University Medical Center New York, New York, United States, 10032 United States, North Carolina Carolinas Medical Center / Levine Children's Hospital Charlotte, North Carolina, United States, 28203 Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Nationwide Children's Hospital Columbus, Ohio, United States, 43205 United States, Pennsylvania Penn State Hershey Children's Hospital Hershey, Pennsylvania, United States, 17033 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 United States, Texas Cook Children's Health Care System Fort Worth, Texas, United States, 76104 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Collapse << |
| NCT01782742 | Alzheimer's Disease | Phase 2 | Completed | - | United States, Nevada ... more >> Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada, United States, 89106 Collapse << |
| NCT01237821 | Acne Vulgaris | Not Applicable | Completed | - | United States, Pennsylvania ... more >> Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT01226303 | Acute Promyelocytic Leukemia | Phase 3 | Unknown | December 2018 | Italy ... more >> Dipartimento di Biotecnologie Cellulari ed Ematologia Recruiting Roma, Italy, 00161 Contact: Testi, Dr 06.857951 ext +39 testi@bce.uniroma1.it Principal Investigator: Annamaria Testi, Dr Collapse << |
| NCT01701479 | Neuroblastoma | Phase 1 Phase 2 | Unknown | December 2013 | Austria ... more >> St. Anna Kinderspital Recruiting Vienna, Austria, 1090 Contact: Ruth Ladenstein, MD +43(1)40470-4750 ruth.ladenstein@ccri.at Principal Investigator: Ruth Ladenstein, MD France Institut Curie Recruiting Paris, France, 75248 Contact: Jean Michon, MD +33-1-44-35-4550 jean.michon@curie.net Principal Investigator: Jean Michon, MD Sub-Investigator: Daniel Orbach, MD Institut Gustave Roussy Recruiting Villejuif, France, 94805 Contact: Dominique Valteau-Couanet, MD +33-1-42114872 valteau@igr.fr Principal Investigator: Dominique Valteau-Couanet, MD Sub-Investigator: Angela Digiannatale, MD Germany University Children's Hospital Recruiting Greifswald, Germany, 17475 Contact: Holger Lode, MD, PhD +49 3834 866301 lode@uni-greifswald.de Principal Investigator: Holger Lode, Md, Phd Israel Schneider Children's Medical Centre of Israel Recruiting Petach Tikvah, Israel, 49202 Contact: Isaac Yaniv, MD +972-3-9253669 iyaniv@clalit.org.it Principal Investigator: Isaac Yaniv, MD Sub-Investigator: Shifra Ash, MD Italy Gaslini Children's Hospital Recruiting Genova, Italy, 16147 Contact: Alberto Garaventa, MD +39-010-5636694-714 albertogaraventa@ospedale-gaslini.ge.it Principal Investigator: Alberto Garaventa, MD Sub-Investigator: Carla Manzitti, MD Spain Hospital Universitario La Fe Recruiting Valencia, Spain, 46009 Contact: Victoria Castel, MD +34-96-197-3304 castel_vic@gva.es Principal Investigator: Victoria Castel, MD Sub-Investigator: Adela Canete, MD United Kingdom Birmingham Children's Hospital NHS Foundation Trust Not yet recruiting Birmingham, United Kingdom, B4 6NH Contact: Pamela Kearns, MBChB, MRCP (paeds), PhD +44121 3338238 p.r.kearns@bham.ac.uk Principal Investigator: Pamela Kearns, MBChB, MRCP (paeds), PhD University Hospitals Bristol NHS Foundation Trust Not yet recruiting Bristol, United Kingdom, BS2 8BJ Contact: Helen Rees, MBBS, MRCP(Paeds), DCH +44117 342 28815 Helen.rees@uhbristol.nhs.uk Principal Investigator: Helen Rees, MBBS, MRCP(Paeds), DCH Leeds Teaching Hospitals NHS Trust Not yet recruiting Leeds, United Kingdom, LS1 3EX Contact: Martin Elliott, MBChB, MRCP, PhD +44113 3928039 Martin.elliott@leedsth.nhs.uk Principal Investigator: Martin Elliott, MBChB, MRCP, PhD Alder Hey Children's NHS Foundation Trust Not yet recruiting Liverpool, United Kingdom, L12 2AP Contact: Lisa Howell, B.Med.Sci, BmBs, MRCPCH +441512525976 Lisa.howell@alderhey.nhs.uk Principal Investigator: Lisa Howell, B.Med.Sci, BmBs, MRCPCH University College Hospitals NHS Foundation Trust Not yet recruiting London, United Kingdom, NW1 2BU Contact: Ananth Shankar, DCH, MD, MRCP +44845 1555000 ext 79490 ananth.shankar@uclh.nhs.uk Principal Investigator: Ananth Shankar, DCH, MD, MRCP Great Ormond Street Hospital for Children NHS Foundation Trust Not yet recruiting London, United Kingdom, WC1N 3JH Contact: John Anderson, MBBS, MRCP (Paeds), PhD +442074059200 ext 8832 j.anderson@ich.ucl.ac.uk Principal Investigator: John Anderson, MBBS, MRCP (Paeds), PhD The Newcastle upon Tyne Hospitals NHS Foundation Trust Not yet recruiting Newcastle, United Kingdom, NE1 4LP Contact: Deborah Tweddle, MBChB (Hons), PhD, FRCPCH +44191 2824068 deborah.tweddle@nuth.nhs.uk Principal Investigator: Deborah Tweddle, MBChB (Hons), PhD, FRCPCH Collapse << |
| NCT00648986 | Brittle Nails | Phase 4 | Completed | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00482833 | Leukemia | Phase 3 | Active, not recruiting | December 2018 | - |
| NCT03751917 | - | - | Not yet recruiting | December 2023 | Italy ... more >> S.O.C. di Ematologia - A.O. - SS. Antonio e Biagio e Cesare Arrigo Not yet recruiting Alessandria, Italy Contact: Ladetto UO Ematologia con trapianto-Universita' degli Studi di Bari Aldo Moro Not yet recruiting Bari, Italy Contact: Specchia Azienda Ospedaliera - Papa Giovanni XXIII Not yet recruiting Bergamo, Italy Contact: Rambaldi Spedali Civili - Brescia - Azienda Ospedaliera - U.O. Ematologia Not yet recruiting Brescia, Italy Contact: Rossi CTMO - Ematologia - Ospedale "Binaghi" Not yet recruiting Cagliari, Italy Contact: Cabras A.O. Pugliese Ciaccio - Presidio Ospedaliero A.Pugliese - U.O. di Ematologia Not yet recruiting Catanzaro, Italy Contact: Molica U.O.C. di Ematologia 1 e 2 IRCCS AOU San Martino-IST Not yet recruiting Genova, Italy Contact: Angelucci Fondazione IRCCS Ca' Granda Osp. Maggiore Policlinico UOC Oncoematologia Not yet recruiting Milano, Italy Contact: Cortelezzi Ospedale Niguarda " Ca Granda" - SC Ematologia Not yet recruiting Milano, Italy Contact: Cairoli U.O. Ematologia e Trapianto di MIdollo - Ist.Scientifico Ospedale San Raffaele Not yet recruiting Milano, Italy Contact: Ciceri Azienda Ospedaliera "S.Gerardo" Not yet recruiting Monza, Italy Contact: Pioltelli Napoli Azienda Ospedaliera di Rilievo Nazionale "A. Cardarelli" Not yet recruiting Naples, Italy Contact: Ferrara Ospedali Riuniti "Villa Sofia-Cervello" Not yet recruiting Palermo, Italy Contact: Fabbiano U.O. Ematologia Clinica - Azienda USL di Pescara Not yet recruiting Pescara, Italy Contact: Pescara Universita' "Sapienza" - Dip Biotecnologie Cellulari - Divisione di Ematologia Roma, Italy Universita' Cattolica del Sacro Cuore - Policlinico A. Gemelli Not yet recruiting Roma, Italy Contact: De Stefano Universita' degli Studi - Policlinico di Tor Vergata Not yet recruiting Roma, Italy Contact: Arcese Università degli studi di Tor Vergata Not yet recruiting Roma, Italy Contact: Francesco Lo Coco Principal Investigator: Francesco Lo Coco stituto di Ematologia - IRCCS Ospedale Casa Sollievo della Sofferenza Not yet recruiting San Giovanni Rotondo, Italy Contact: Cascavilla Dipartimento di Oncologia Ematologia 2 A.O. Citta' della Salute S. G. Battista Not yet recruiting Torino, Italy Contact: Vitolo ULSS N.6 Osp. S. Bortolo Not yet recruiting Vicenza, Italy Contact: Ruggeri Collapse << |
| NCT03112863 | Wrinkle Photo... more >>aging Collapse << | Phase 1 Phase 2 | Recruiting | March 2027 | United States, California ... more >> UC Davis Department of Dermatology Recruiting Sacramento, California, United States, 95816 Contact: Manisha Notay, MBBS 916-734-6550 mnotay@ucdavis.edu Collapse << |
| NCT00167219 | Juvenile Myelomonocytic Leukem... more >>ia Collapse << | Phase 1 Phase 2 | Recruiting | December 2020 | United States, Minnesota ... more >> Masonic Cancer Center, University of Minnesota Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Kim Nelson 612-273-2925 knelso62@fairview.org Collapse << |
| NCT02849860 | Acne | Phase 1 | Completed | - | United States, Texas ... more >> Valeant Site 02 Austin, Texas, United States, 78759 Valeant Site 01 College Station, Texas, United States, 77845 Collapse << |
| NCT00196768 | Relapsed Acute Promyelocytic L... more >>eukemia Refractory Acute Promyelocytic Leukemia Collapse << | Phase 4 | Unknown | - | Germany ... more >> Eva Lengfelder, MD, PhD Recruiting Mannheim, Germany, 68305 Principal Investigator: Eva Lengfelder, MD, PhD Collapse << |
| NCT03624270 | Acute Promyelocytic Leukemia | Phase 2 | Recruiting | December 31, 2023 | Hong Kong ... more >> Department of Medicine, the University of Hong Kong, Queen Mary Hospital Recruiting Hong Kong, N/A = Not Applicable, Hong Kong Contact: Harinder Singh Harry Gill +852 22554542 gillhsh@hku.hk Collapse << |
| NCT02721173 | Acne Vulgaris | Phase 4 | Completed | - | India ... more >> AIIMS, Bhubaneswar Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT00145106 | Acne Vulgaris | Phase 4 | Completed | - | United States, Pennsylvania ... more >> KGL, Inc Broomall, Pennsylvania, United States, 19008 Collapse << |
| NCT00504764 | Acute Promyelocytic Leukemia | Phase 4 | Completed | - | - |
| NCT03042429 | Neuroblastoma | Phase 3 | Completed | - | Germany ... more >> University of Cologne Koln, Germany, 50924 Collapse << |
| NCT02850003 | Acne | Phase 1 | Completed | - | United States, Texas ... more >> Valeant Site 01 Austin, Texas, United States, 78759 Collapse << |
| NCT03259516 | Myelodysplastic Syndromes | Phase 1 Phase 2 | Recruiting | December 25, 2021 | Russian Federation ... more >> First Pavlov State Medical University of St. Petersburg Recruiting Saint-Petersburg, Russian Federation, 197089 Contact: Ivan S. Moiseev, MD, PhD +79217961951 moisiv@mail.ru Contact: Elena V Morozova, MD, PhD +79119278229 dr_morozova@mail.ru Principal Investigator: Boris V Afanasyev, MD, Prof Sub-Investigator: Elena V Morozova, MD, PhD Sub-Investigator: Yulia V Rudnizkaya, MD, PhD Sub-Investigator: Ivan S Moiseev, MD, PhD Sub-Investigator: Nikolai Yu Tcvetkov, MD Collapse << |
| NCT03325738 | - | - | Recruiting | June 30, 2018 | France ... more >> PEYRADE Frédéric Recruiting Nice, France, 06189 Contact: Frédéric PEYRADE +33492031022 frederic.peyrade@nice.unicancer.fr Collapse << |
| NCT00534898 | Schizophrenia ... more >> Schizoaffective Disorder Cognition Collapse << | Phase 3 | Withdrawn(The study withdrawn ... more >>due to lack financial support) Collapse << | - | Israel ... more >> Be'er Sheva Mental Health Center, Sha'ar Menashe Mental Health Center Be'er Sheva, Hadera, Israel, 84170 Collapse << |
| NCT00555399 | Glioblastoma Multiforme ... more >> Anaplastic Glioma Collapse << | Phase 1 Phase 2 | Active, not recruiting | November 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01680510 | Retinitis Pigmentosa | Phase 2 Phase 3 | Recruiting | December 2018 | Israel ... more >> Sheba Medical Center Recruiting Tel Hashomer, Israel, 52621 Contact: Ygal Rotenstreich, Dr. 972-3-5302880 Principal Investigator: Ygal Rotenstreich, Dr. Collapse << |
| NCT01256697 | Retinitis Pigmentosa | Not Applicable | Completed | - | Israel ... more >> Sheba Medical Center Tel Hashomer, Israel Collapse << |
| NCT01085279 | Pigmentation Disorder | Phase 2 | Completed | - | Netherlands ... more >> Netherlands Institute for Pigment disorders Amsterdam, Netherlands, 1105 AZ Collapse << |
| NCT00993538 | Acute Leukaemia | Not Applicable | Unknown | - | France ... more >> Service d'Hématologie et d'Oncologie Recruiting Strasbourg, France, 67098 Contact: Raoul Herbrecht, MD 3.33.88.12.83.79 Raoul.herbrecht@chru-strasbourg.fr Principal Investigator: Raoul Herbrecht, MD Sub-Investigator: Cécile Fohrer, MD Sub-Investigator: Luc Fornecker, MD Sub-Investigator: Bruno Lioure, MD Sub-Investigator: Karin Bilger, MD Service de Pédiaterie - Hôpital de Hautepierre Recruiting Strasbourg, France, 67098 Contact: Patrick Lutz, MD 3.33.88.12.80.90 Patrick.lutz@chru-strasbourg.fr Principal Investigator: Patrick Lutz, MD Sub-Investigator: Natacha Entz-Werle, MD Collapse << |
| NCT00985530 | Acute Promyelocytic Leukemia | Phase 1 | Terminated(Supporting agency w... more >>ithdrew funding for study.) Collapse << | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00903422 | Myelodysplastic Syndrome | Phase 1 | Completed | - | - |
| NCT02173054 | Acne | Phase 3 | Completed | - | Thailand ... more >> Siriraj Hospital Mahidol Univeristy Bangkok, Thailand, 10700 Collapse << |
| NCT02173054 | - | - | Completed | - | - |
| NCT02752763 | Dry Eye | Phase 4 | Completed | - | Turkey ... more >> Niğde State Hospital Niğde, Turkey, 51100 Collapse << |
| NCT02381483 | Brown Adipose Tissue | Not Applicable | Completed | - | Austria ... more >> Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT02979769 | Fibrodysplasia Ossificans Prog... more >>ressiva Collapse << | Phase 2 | Active, not recruiting | December 2018 | France ... more >> Hôpital Necker-Enfants Malades, Department of Genetics Paris, France Collapse << |
| NCT00747851 | - | - | Recruiting | December 2019 | United States, Utah ... more >> University of Utah Recruiting Salt Lake City, Utah, United States, 84112 Contact: Christian C Yost, M.D. 801-581-7052 christian.yost@hmbg.utah.edu Principal Investigator: Christian C Yost, M.D. Collapse << |
| NCT01404702 | Neuroblastoma | Phase 1 | Terminated(Accrual slower than... more >> anticipated) Collapse << | - | United States, Alabama ... more >> University of Alabama at Birmingham-Children's of Alabama Birmingham, Alabama, United States, 35233 Collapse << |
| NCT03026946 | Hand Eczema | Phase 3 | Recruiting | March 2020 | Netherlands ... more >> University Medical Center Groningen Recruiting Groningen, Netherlands, 9700RB Contact: MLA Schuttelaar, MD, PhD +31503612520 m.l.a.schuttelaar@umcg.nl Contact: JAF Oosterhaven, MD +31503610795 j.a.f.oosterhaven@umcg.nl Collapse << |
| NCT03022019 | Non-segmental Vitiligo ... more >> Vitiligo Collapse << | Not Applicable | Unknown | December 2018 | Netherlands ... more >> Nipd / Amc Amsterdam, Netherlands, 1105AZ Collapse << |
| NCT01079988 | Psoriasis | Phase 4 | Completed | - | - |
| NCT03004846 | Psoriasis | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Hamburg, Germany, 20095 Collapse << |
| NCT02942758 | Acute Myeloid Leukemia | Phase 2 | Recruiting | February 2020 | Germany ... more >> University Hospital Regensburg Recruiting Regensburg, Germany, 93053 Collapse << |
| NCT00907582 | Acute Promyelocytic Leukemia ... more >> Relapse Collapse << | Phase 2 | Terminated(slow recruitment) | - | China ... more >> Rui Jin Hospital, Shanghai JiaoTong University School of Medicine Shanghai, China, 200025 Collapse << |
| NCT01079988 | - | - | Completed | - | - |
| NCT01295762 | Neuroblastoma | Not Applicable | Active, not recruiting | July 2019 | France ... more >> Hopital D'Estaing Clermont Ferrand, France, 63100 Chu Grenoble - Hopital Nord La Tronche, France, 38700 IHOP Lyon, France, 69008 Chu - Hopital Nord Saint Priest En Jarez, France, 42270 Collapse << |
| NCT00282724 | Ichthyosis, Lamellar | Phase 2 Phase 3 | Completed | - | Belgium ... more >> Academisch Ziekenhuis Vrije Universiteit Brussel Brussels, Belgium Geel Geel, Belgium Canada Hôpital Saint-Justine Montreal, Canada Newlab Clinical Research Inc. St John, Canada Dominican Republic Instituto Dermatologico Santo Domingo, Dominican Republic France Hôtel Dieu CHU Nantes, France Germany Tomesa Fachklinik Bad Salzschlirf, Germany Dueren Dueren, Germany Otto-von-Guericke-Universität Magdeburg, Germany University Hospital Muenster Muenster, Germany Italy Fondazione Policlinico Mangiagalli e Regina Elena Milano, Italy Istituto Dermopatico dell'Immacolata Rome, Italy Netherlands Academisch Ziekenhuis Maastricht Maastricht, Netherlands University Hospital Rotterdam Rotterdam, Netherlands Norway Rikshospitalet Universitetsklinikk Oslo, Norway Sweden Uppsala University Hospital Uppsala, Sweden Collapse << |
| NCT01538732 | Lichen Planus (LP) | Phase 2 | Unknown | March 2014 | Switzerland ... more >> University Hospital Zurich, Division of Dermatology Recruiting Zurich, ZH, Switzerland, 8091 Collapse << |
| NCT01280110 | - | - | Completed | - | - |
| NCT00841776 | Acne | Phase 4 | Completed | - | United States, Kentucky ... more >> Dermatology Specialists, PSC Louisville, Kentucky, United States, 40202 United States, Ohio Skin We Care Dermatology Mason, Ohio, United States, 45040 Collapse << |
| NCT00841776 | - | - | Completed | - | - |
| NCT03026907 | Hand Eczema | Phase 3 | Recruiting | December 2019 | Netherlands ... more >> University Medical Center Groningen Recruiting Groningen, Netherlands, 9700RB Contact: MLA Schuttelaar, MD, PhD +31503612520 m.l.a.schuttelaar@umcg.nl Contact: JAF Oosterhaven, MD +31503610795 j.a.f.oosterhaven@umcg.nl Collapse << |
| NCT01020539 | Acute Myelogenous Leukemia ... more >> Myelodysplastic Syndrome Juvenile Myelomonocytic Leukemia Collapse << | Phase 1 | Active, not recruiting | March 2018 | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT02401295 | Relapsed Multiple Myeloma | Phase 1 | Completed | - | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52242 Collapse << |
| NCT01280110 | Dry Eye Syndromes | Phase 4 | Completed | - | Brazil ... more >> Department of Ophthalmology, University of Campinas Campinas, SP, Brazil Collapse << |
| NCT00535574 | Schizophrenia | Phase 3 | Completed | - | Israel ... more >> Be'er Sheva Mental Health Center Be'er Sheva,, Be'er Sheva, Israel, 84170 Abarbanel Mental Health Center Bat-Yam, Israel Sha'ar Menashe Mental Health Center, Hadera,, Israel Collapse << |
| NCT00201279 | Oral Cavity S... more >>quamous Cell Carcinoma Collapse << | Phase 3 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT00697593 | Chronic Plaque Psoriasis | Phase 4 | Terminated(The study was termi... more >>nated after the European Medicines Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | Canada, Ontario ... more >> Probity Medical Research City Waterloo, Ontario, Canada, N2J 1C4 Collapse << |
| NCT01728155 | LOW AND INTERMEDIATE PAEDIATRI... more >>C NEUROBLASTOMA AND NEONATAL SUPRARENAL MASSES Collapse << | Phase 3 | Recruiting | December 2031 | - |
| NCT03076021 | CYP2D6 Polymorphism | Phase 4 | Recruiting | July 26, 2026 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Mary F Hebert, PharmD 206-616-5016 mhebert@uw.edu Collapse << |
| NCT00697593 | - | - | Terminated(The study was termi... more >>nated after the European Medicines Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT00615784 | Acute Myeloid Leukemia | Phase 2 | Unknown | - | United States, Pennsylvania ... more >> Abramson Cancer Center of University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01504490 | Solid Tumors ... more >>Lymphoma Multiple Myeloma Collapse << | Phase 1 | Terminated(Drug no longer avai... more >>lable) Collapse << | - | United States, District of Col... more >>umbia Georgetown Lombardi Comprehensive Cancer Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT03287011 | Chronic Gingivitis, Plaque Ind... more >>uced Collapse << | Not Applicable | Recruiting | April 30, 2018 | Malaysia ... more >> Avita Rath Recruiting Petaling Jaya, Selangor, Malaysia, 47810 Contact: Avita Rath, MDS 60109677452 avitashanti@segi.edu.my Collapse << |
| NCT02715960 | Psoriasis Vulgaris | Phase 2 | Unknown | December 2017 | China, Hunan ... more >> Xiangya Hospital Central South University Recruiting Changsha, Hunan, China, 410008 Contact: Yijing He, MD, PhD +86-1587481262 yijinghe@gmail.com Collapse << |
| NCT02190747 | Fibrodysplasia Ossificans Prog... more >>ressiva Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California San Francisco, Division of Endocrinology and Metabolism San Francisco, California, United States, 94143 United States, Pennsylvania University of Pennsylvania, Center for Research in FOP & Related Disorders Philadelphia, Pennsylvania, United States, 19104 France Hôpital Necker-Enfants Malades, Department of Genetics Paris, France United Kingdom The Royal National Orthopaedic Hospital, Brockley Hill Stanmore, Middlesex, United Kingdom, HA7 4LP Collapse << |
| NCT01138917 | Burns | Not Applicable | Completed | - | United States, Arizona ... more >> Arizona Burn Center at Maricopa Integrated Health Systems Phoenix, Arizona, United States, 85008 United States, California University of California Davis Regional Burn Center Sacramento, California, United States, 95817 United States, District of Columbia The Burn Center at Washington Hospital Center Washington, District of Columbia, United States, 20010 United States, Florida Shands Burn Center at University of Florida Gainesville, Florida, United States, 32610 Tampa General Hospital Tampa, Florida, United States, 33606 United States, Indiana Indiana University - Richard M. Fairbanks Burn Center at Wishard Memorial Hospital Indianapolis, Indiana, United States, 46202 United States, North Carolina NC Jaycee Burn Center at University of NC at Chapel Hill Chapel Hill, North Carolina, United States, 27599 Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 United States, Tennessee University of Tennessee Medical Center Memphis, Tennessee, United States, 38163 United States, Texas USAISR Fort Sam Houston, Texas, United States, 78234 Texas Tech University Health Sciences Center Lubbock, Texas, United States, 79430 United States, Virginia Virginia Commonwealth University Health System- Evans Haynes Burn Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT02548052 | Psoriasis | Phase 1 | Completed | - | Germany ... more >> GSK Investigational Site Hamburg, Germany, 20095 Collapse << |
| NCT00628329 | - | - | Withdrawn | December 2012 | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Collapse << |
| NCT02688140 | Acute Promyelocytic Leukemia | Phase 3 | Recruiting | January 2022 | France ... more >> French-Belgian-Swiss APL study group Not yet recruiting Multiple Locations, France Contact: Pierre Fenaux, Prof. Dr. pierre.fenaux@avc.ap-hop-paris.fr Contact: Lionel Ades, Dr. lionel.ades@sls.aphp.fr Germany AML-CG study group Recruiting Multiple Locations, Germany Contact: Eva Lengfelder, Prof. Dr. Eva.Lengfelder@umm.de AML-SG study group Recruiting Multiple Locations, Germany Contact: Richard F. Schlenk, Prof. Dr. Richard.Schlenk@uniklinik-ulm.de OSHO study group Recruiting Multiple Locations, Germany Contact: Dietger Niederwieser, Prof. Dr. Dietger.Niederwieser@medizin.uni-leipzig.de SAL study group Recruiting Multiple Locations, Germany Contact: Uwe Platzbecker, Prof. Dr. +49 351 458 ext 3192 uwe.platzbecker@uniklinikum-dresden.de Contact: Michaela Sauer +49 351 458 ext 3192 michaela.sauer@uniklinikum-dresden.de Italy GIMEMA study group Not yet recruiting Multiple Locations, Italy Contact: Francesco Lo-Coco, Prof. Dr. Francesco.lo.coco@uniroma2.it Contact: Fabio Efficace, Dr. f.efficace@gimema.it Netherlands HOVON study group Not yet recruiting Multiple Locations, Netherlands Contact: Edo Vellenga, Prof. Dr. e.vellenga@umcg.nl Spain PETHEMA study group Not yet recruiting Multiple Locations, Spain Contact: Miguel A. Sanz, Prof. Dr. sanz_mig@gva.es Contact: Pau Montesinos, Prof. Dr. montesinos_pau@gva.es Collapse << |
| NCT00146120 | Acute Myeloid Leukemia | Phase 3 | Completed | - | Austria ... more >> Department of Hematology / Oncology, University Hospital of Innsbruck Innsbruck, Austria, 6020 III Medical Department, Hematology and Oncology Center, Hanuschhospital Wien Wien, Austria, 1140 Germany Department of General Internal Medicine, University Hospital of Bonn Bonn, Germany, 53127 Department of Internal Medicine Hematology, Heinrich-Heine University Düsseldorf, Germany, 40225 Department of Interial Medicine III, City Hospital Frankfurt Am Main - Höchst Frankfurt, Germany, 65927 Medical Department IV, University of Gießen Gießen, Germany, 35392 Department of Interial Medicine, Wilhelm-Anton-Hospital Goch Goch, Germany, 47574 Centre of Interial Medicine, University of Göttingen Göttingen, Germany, 37075 Medical Department III of Hematology and Oncology, General Hospital Altona Hamburg, Germany, 22763 Department of Interial Medicine V, University of Heidelberg Heidelberg, Germany, 69120 Department of Interial Medicine I, University Hospital of Saarland Homburg, Germany, 66421 Medical Department II, City Hospital Karlsruhe gGmbH Karlsruhe, Germany, 76133 Medical Department II, University Hospital of Kiel Kiel, Germany, 24116 Department of Interial Medicine /Hematology and Oncology, Caritas Hospital Lebach Lebach, Germany, 66822 I. Medical Department, City Hospital München-Schwabing München, Germany, 80804 Medical Department III, Clinical Center rechts der Isar München, Germany, 81675 Department of Hematology and Oncology, City Hospital Neunkirchen gGmbH Neunkirchen, Germany, 66538 Department of Hematology and Oncology, Clinical Center of Oldenburg gGmbH Oldenburg, Germany, 26133 Department of Hematologie and Oncology, Caritas Hospital St. Theresa Saarbrücken Saarbrücken, Germany, 66113 Clinikal Cetner of Stuttgart, Center of Oncology Stuttgart, Germany, 70174 Medical Department I, Clinical Center of Stuttgart Stuttgart, Germany, 70191 Hospital of Barmherzige Brüder, I Medical Department Trier, Germany, 54292 Collapse << |
| NCT01245946 | Acne | Phase 2 | Completed | - | Chile ... more >> Departamento de dermatología, Centro Médico San Joaquín, Pontificia Universidad Católica de Chile Santiago, San Joaquín, Chile, 1234 Collapse << |
| NCT00804050 | Myelodysplastic Syndromes | Phase 3 | Terminated(Difficulties of enr... more >>ollment) Collapse << | - | Italy ... more >> Ospedale SS. Antonio, Biagio e Cesare Arrigo Alessandria, Italy Ospedale degli Infermi Biella, Italy Spedali civili Brescia, Italy Ospedale Santo Spirito Casale (AL), Italy Ospedale Maggiore Chieri (TO), Italy Policlinico dell'Annunziata Cosenza, Italy Ospedale Santa Croce e Carle Cuneo, Italy Ospedale Santa Croce Fano (PU), Italy Ospedale San Martino Genova, Italy Ospedale civile Ivrea (TO), Italy Ospedale San Gerardo Monza (MI), Italy Ospedale Maggiore della Carità Novara, Italy Ospedale Civile Ovada (AL), Italy Istituto clinico Humanitas Rozzano (MI), Italy Ospedale San Giovanni Battista Molinette Torino, Italy Ospedale Cardinale Panico Tricase (LE), Italy Ospedale Sant'Andrea Vercelli, Italy Ospedale San Bortolo Vicenza, Italy Collapse << |
| NCT00469183 | Melasma | Phase 4 | Completed | - | United States, California ... more >> Vitiligo and Pigmentation Institute of Southern California Los Angeles, California, United States, 90036 United States, Texas University of Texas Southwestern Medical Center of Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT00038376 | Lymphoma, T-Cell ... more >> Mycosis Fungoides Hematologic Neoplasms Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> M. D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02177812 | Leukaemia, Myelocytic, Acute | Phase 1 | Terminated(The risk benefit in... more >> relapsed refractory AML does not favor continuation of the study.) Collapse << | - | United States, Georgia ... more >> GSK Investigational Site Augusta, Georgia, United States, 30912 United States, New York GSK Investigational Site Bronx, New York, United States, 10461 GSK Investigational Site Bronx, New York, United States, 10467 GSK Investigational Site New York, New York, United States, 10065 United States, Texas GSK Investigational Site Houston, Texas, United States, 77030 Australia, Victoria GSK Investigational Site Melbourne, Victoria, Australia, 3004 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M5G 2M9 GSK Investigational Site Toronto, Ontario, Canada, M5G 2M Collapse << |
| NCT00808899 | Neuroblastoma | Phase 2 | Terminated(Voluntarily closed ... more >>and terminated by the PI due to lack of feasibility) Collapse << | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT01206348 | Acne | Phase 4 | Completed | - | United States, Alabama ... more >> Birmingham, Alabama, United States United States, California Santa Monica, California, United States United States, Delaware Hockessin, Delaware, United States United States, Massachusetts Chestnut Hill, Massachusetts, United States United States, Michigan Detroit, Michigan, United States United States, Nevada Henderson, Nevada, United States United States, North Carolina High Point, North Carolina, United States United States, Pennsylvania Hershey, Pennsylvania, United States United States, Tennessee Nashville, Tennessee, United States United States, Utah Salt Lake City, Utah, United States Collapse << |
| NCT00808899 | - | - | Terminated(Voluntarily closed ... more >>and terminated by the PI due to lack of feasibility) Collapse << | - | - |
| NCT00863278 | Melanosis | Phase 4 | Terminated | - | France ... more >> CHU de Nice - 4 avenue Reine Victoria - Hôpital de Cimiez Nice, Alpes-Maritimes, France, 06001 Collapse << |
| NCT01404130 | Acne Vulgaris | Phase 4 | Completed | - | United Kingdom ... more >> Anthony Ormerod Aberdeen, Aberdeenshire, United Kingdom, AB25 2AN Collapse << |
| NCT01461655 | - | - | Completed | - | - |
| NCT00309621 | Hand Dermatoses | Phase 3 | Completed | - | Canada, Alberta ... more >> Edmonton, Alberta, Canada, T6G2G3 Canada, British Columbia Vancouver, British Columbia, Canada, V5O3Y1 Canada, Manitoba Winnipeg, Manitoba, Canada, R3C1R4 Canada, Ontario Hawkesbury, Ontario, Canada, K6A1A1 London, Ontario, Canada, N6H1S9 Markham, Ontario, Canada, L3P1A8 Oakville, Ontario, Canada, L6J7W5 Canada, Quebec Montreal, Quebec, Canada, H3X1V6 Sheerbroke, Quebec, Canada, J1H1Z1 Canada Quebec, Canada, G1J1X7 Germany Berlin, Germany, 12353 Berlin, Germany, 13439 Frankfurt, Germany, 60326 Hamburg, Germany, 20246 Hamburg, Germany, 20354 Hamburg, Germany, 22143 Abt. Klinische Sozialmedizin Heidelberg, Germany, 69115 Ibbenbüren, Germany, 49477 Köln, Germany, 51143 Lübeck, Germany, 23558 Mönchengladbach, Germany, 41061 München, Germany, 81375 Münster, Germany, 48143 Osnabrück, Germany, 49078 Potsdam, Germany, 14482 Viersen, Germany, 41747 Wuppertal, Germany, 42275 Poland Gdansk, Poland Krakow, Poland, 31-462 Lublin, Poland Opole, Poland, 45-372 Szcecin, Poland Wroclaw, Poland Collapse << |
| NCT00002737 | Kidney Cancer | Phase 3 | Completed | - | Belgium ... more >> Onze Lieve Vrouw Ziekenhuis Aalst Aalst, Belgium, B-9300 Algemeen Ziekenhuis Middelheim Antwerp, Belgium, 2020 Institut Jules Bordet Brussels, Belgium, 1000 Universitair Ziekenhuis Antwerpen Edegem, Belgium, B-2650 U.Z. Gasthuisberg Leuven, Belgium, B-3000 Hungary National Institute of Oncology Budapest, Hungary, 1125 Italy Ospedale di Circolo e Fondazione Macchi Varese, Italy, 21100 Netherlands Antoni van Leeuwenhoekhuis Amsterdam, Netherlands, 1066 CX Academisch Medisch Centrum Amsterdam, Netherlands, 1105 AZ Groot Ziekengasthuis 's-Hertogenbosch Hertogenbosch, Netherlands, 5211 NL Leiden University Medical Center Leiden, Netherlands, 2300 CA University Medical Center Nijmegen Nijmegen, Netherlands, NL-6252 HB University Hospital - Rotterdam Dijkzigt Rotterdam, Netherlands, 3000 CA Rotterdam Cancer Institute Rotterdam, Netherlands, 3075 EA Academisch Ziekenhuis Utrecht Utrecht, Netherlands, 3508 GA Norway Norwegian Radium Hospital Oslo, Norway, N-0310 Russian Federation Russian Academy of Medical Sciences Cancer Research Center Moscow, Russian Federation, 115478 Switzerland Kantonspital Aarau Aarau, Switzerland, 5001 Ospedale San Giovanni Bellinzona, Switzerland, CH-6500 Inselspital, Bern Bern, Switzerland, CH-3010 Ratisches Kantons und Regionalspital Chur, Switzerland, CH-7000 Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland, CH-1011 Kantonsspital - Saint Gallen Saint Gallen, Switzerland, CH-9007 Universitaetsspital Zurich, Switzerland, CH-8091 Turkey Marmara University Hospital Istanbul, Turkey, 81190 United Kingdom Bristol Royal Infirmary Bristol, England, United Kingdom, BS2 8HW Beatson Oncology Centre Glasgow, Scotland, United Kingdom, G11 6NT Collapse << |
| NCT01461655 | Acne Vulgaris | Phase 2 | Completed | - | France ... more >> CPCAD - Centre de Pharmacologie Clinique Appliquée à la Dermatologie Nice, France, 06202 Collapse << |
| NCT02200978 | Childhood Acute Promyelocytic ... more >>Leukemia Collapse << | Phase 4 | Recruiting | September 2022 | China, Guangdong ... more >> The First Affiliated Hospital of Sun Yat-Sen University Recruiting Guangzhou, Guangdong, China, 510080 Contact: Xue-Qun Luo, professor L-xuequn@126.com Principal Investigator: Xue-Qun Luo, professor Collapse << |
| NCT03615729 | - | - | Completed | - | - |
| NCT00499616 | Neuroblastoma | Phase 3 | Completed | - | - |
| NCT00004188 | Neuroblastoma | Phase 3 | Completed | - | - |
| NCT00499616 | - | - | Completed | - | - |
| NCT02551718 | Acute Leukemia of Ambiguous Li... more >>neage Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Refractory Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Pamela S. Becker 206-616-1589 pbecker@u.washington.edu Principal Investigator: Pamela S. Becker Collapse << |
| NCT01917032 | Malnutrition | Phase 3 | Unknown | December 2013 | Colombia ... more >> Fundación de Atención a la Niñez (FAN) Recruiting Medellín, Ant, Colombia, 0574 Contact: Viviana Ramírez, Nutricionist 5744480288 nutricion@fan.org.co Collapse << |
| NCT00400556 | Multiple Myeloma ... more >> Cutaneous Lymphoma Collapse << | Phase 1 | Completed | - | Australia, Victoria ... more >> Peter MacCallum Cancer Center East Melbourne, Victoria, Australia, 3002 Collapse << |
| NCT00078988 | Brain Tumor C... more >>entral Nervous System Tumor Collapse << | Phase 3 | Completed | - | - |
| NCT00567567 | - | - | Completed | - | - |
| NCT00567567 | Localized Resectable Neuroblas... more >>toma Localized Unresectable Neuroblastoma Recurrent Neuroblastoma Regional Neuroblastoma Stage 4 Neuroblastoma Stage 4S Neuroblastoma Collapse << | Phase 3 | Completed | - | - |
| NCT00842543 | Obesity | Not Applicable | Completed | - | United States, Florida ... more >> Nemours Children's Clinic Jacksonville, Florida, United States, 32207 Collapse << |
| NCT00392327 | Anaplastic Medulloblastoma ... more >> Medulloblastoma Untreated Childhood Medulloblastoma Collapse << | Phase 3 | Suspended(Temporarily stopped ... more >>for assessment. Contact ctsucontact@westat.com for specifics) Collapse << | - | - |
| NCT00842543 | - | - | Completed | - | - |
| NCT00867178 | Medulloblastoma ... more >> Pineoblastoma Supratentorial Embryonal Tumor, Not Otherwise Specified Untreated Childhood Medulloblastoma Untreated Childhood Pineoblastoma Untreated Childhood Supratentorial Primitive Neuroectodermal Tumor Collapse << | Not Applicable | Active, not recruiting | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Lucile Packard Children's Hospital Stanford University Palo Alto, California, United States, 94304 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Illinois Lurie Children's Hospital-Chicago Chicago, Illinois, United States, 60611 United States, Maryland National Cancer Institute Pediatric Oncology Branch Bethesda, Maryland, United States, 20892 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 United States, Tennessee Pediatric Brain Tumor Consortium Memphis, Tennessee, United States, 38105 St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 Texas Children's Hospital Houston, Texas, United States, 77030 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Collapse << |
| NCT01231854 | Atopic Dermatitis | Phase 4 | Terminated(delayed start of th... more >>e trial, both treatment options already have market authorizations, study medication expired) Collapse << | - | Germany ... more >> Universitätsklinikum Carl Gustav Carus an der TU Dresden Dresden, Sachsen, Germany, 01307 Collapse << |
| NCT03126916 | Childhood Ganglioneuroblastoma... more >> Childhood Neuroblastoma INRG Stage L2 INRG Stage M INRG Stage MS NMYC Gene Amplification Recurrent Neuroblastoma Collapse << | Phase 3 | Recruiting | June 30, 2026 | - |
| NCT00852709 | Myelodysplastic Syndrome ... more >> Acute Myeloid Leukemia Myeloproliferative Disorders Acute Lymphocytic Leukemia Acute Promyelocytic Leukemia Acute Leukemia Chronic Myelogenous Leukemia Myelofibrosis Chronic Myelomonocytic Leukemia Juvenile Myelomonocytic Leukemia Collapse << | Phase 1 | Terminated(Lack of accrual) | - | United States, Arizona ... more >> Phoenix Children's Hospital Phoenix, Arizona, United States, 85016-7710 United States, Colorado University of Colorado Health Sciences Center and The Children's Hospital Aurora, Colorado, United States, 80045 United States, Florida Pediatrix Hematology/Oncology University of Florida College of Medicine Gainesville, Florida, United States, 32610-0296 United States, Georgia Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 United States, Tennessee Vanderbilt Children's Hospital Nashville, Tennessee, United States, 37232-6310 United States, Texas M.D. Anderson Comprehensive Cancer Center Houston, Texas, United States, 77030 Canada, Alberta Southern Alberta Children's Cancer Program Calgary, Alberta, Canada, T3B6A8 Collapse << |
| NCT00075491 | Adult Giant Cell Glioblastoma ... more >> Adult Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 2 | Terminated(Administratively co... more >>mplete.) Collapse << | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00026312 | Localized Resectable Neuroblas... more >>toma Localized Unresectable Neuroblastoma Recurrent Neuroblastoma Regional Neuroblastoma Stage 4 Neuroblastoma Stage 4S Neuroblastoma Collapse << | Phase 3 | Completed | - | - |
| NCT00028106 | Pheochromocytoma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02847533 | Depression | Not Applicable | Unknown | December 2018 | France ... more >> CHU Besançon Not yet recruiting Besançon, France, 25000 Contact: Sophie Depierre 03841218745 sdepierre@chu-besancon.fr Principal Investigator: Emmanuel Haffen, MD PhD Sub-Investigator: Pierre Vandel, MD PhD Sub-Investigator: Djamila Bennabi, MD PhD Sub-Investigator: Nazim Nekrouf, MD PhD Sub-Investigator: Julie Giustiniani, MD Sub-Investigator: Caroline Masse-Sibille, MD Collapse << |
| NCT02758990 | Obesity Rhini... more >>tis Rhinitis, Allergic Respiratory Sounds Dyspnea Headache Migraine Disorders Arthritis Arthralgia Anxiety Sleep Initiation and Maintenance Disorders Sleep Deprivation Collapse << | Not Applicable | Terminated(Recruiting and fina... more >>ncial constraints) Collapse << | - | United States, Texas ... more >> Verifomics, LLC Dallas, Texas, United States, 75219 Collapse << |
| NCT01848028 | - | - | Recruiting | July 2026 | Germany ... more >> Nationwide group of dermatological centers, hospitals and medical offices Recruiting Hamburg, Germany Contact: Matthias Augustin, Prof. Dr. m.augustin@uke.de Principal Investigator: Matthias Augustin, Prof. Dr. Collapse << |
| NCT01704716 | Neuroblastoma | Phase 3 | Recruiting | September 2024 | - |
| NCT00599937 | Leukemia, Promyelocytic, Acute | Phase 3 | Completed | - | - |
| NCT03057626 | - | - | Recruiting | August 1, 2020 | - |
| NCT01763853 | Acute Respiratory Distress Syn... more >>drome (ARDS) Hypovolemia Pulmonary Edema Collapse << | Phase 4 | Unknown | May 2017 | France ... more >> CHU Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick Lacarin 04 73 75 11 95 placarin@chu-clermontferrand.fr Collapse << |
| NCT00026312 | - | - | Completed | - | - |
| NCT01857934 | Neuroblastoma | Phase 2 | Recruiting | July 2021 | United States, Tennessee ... more >> St. Jude Children's Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Wayne L. Furman, MD 866-278-5833 referralinfo@stjude.org Principal Investigator: Wayne L. Furman, MD Collapse << |
| NCT02181790 | Psoriasis, Skin or Nails | Phase 4 | Terminated(Lack of subjects) | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT00322296 | Parapsoriasis | Phase 2 | Terminated(The response rate (... more >>5/8) had reached a statistical significant endpoint.) Collapse << | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT03151473 | - | - | Recruiting | September 30, 2027 | Hong Kong ... more >> Humanity and Health Medical Centre Recruiting Hong Kong, Hong Kong, 00852 Contact: George Lau, MD (852)28613777 gkklau@netvigator.com Principal Investigator: George Lau, MD Collapse << |
| NCT02279095 | Fibrodysplasia Ossificans Prog... more >>ressiva Collapse << | Phase 2 | Active, not recruiting | March 2021 | United States, California ... more >> University of California San Francisco, Division of Endocrinology and Metabolism San Francisco, California, United States, 94143 United States, Minnesota Mayo Clinic, Department of Medicine Rochester, Minnesota, United States, 55905 United States, Pennsylvania University of Pennsylvania, Center for FOP & Related Bone Disorders Philadelphia, Pennsylvania, United States, 19104 Argentina Hospital Italiano de Buenos Aires, Department of Pediatrics Buenos Aires, Argentina Australia, Queensland Queensland University of Technology (QUT) Institute of Health and Biomedical Innovation (IHBI) Woolloongabba, Queensland, Australia, 4102 France Hôpital Necker-Enfants Malades, Department of Genetics Paris, France United Kingdom The Royal National Orthopaedic Hospital, Brockley Hill Stanmore, Middlesex, United Kingdom, HA7 4LP Collapse << |
| NCT02181790 | - | - | Terminated(Lack of subjects) | - | - |
| NCT01041638 | - | - | Completed | - | - |
| NCT03312634 | Fibrodysplasia Ossificans Prog... more >>ressiva Collapse << | Phase 3 | Active, not recruiting | November 2020 | United States, California ... more >> University of California San Francisco, Division of Endocrinology and Metabolism San Francisco, California, United States, 94143 United States, Minnesota Mayo Clinic - 200 1st Street Southwest Rochester, Minnesota, United States, 55905 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Argentina Hospital Italiano de Buenos Aires, Tte General Juan Domingo Peron 4190 Buenos Aires, Argentina, C1199ABB Australia, Queensland Queensland University of Technology, Institute of Health and Biomedical Innovation, TRI, Level 3 Woolloongabba, Queensland, Australia, 4102 Brazil Hospital Israelita Albert Einstein Sao Paulo, SP, Brazil, 05652-900 Canada, Ontario Hospital for Sick Children, 555 University Avenue Toronto, Ontario, Canada, M5G 1X8 Toronto General Hospital Toronto, Ontario, Canada, M5G 2C4 France Groupe Hospitalier Necker Enfants Malades Paris, France, 75015 Italy Istituto Giannina Gaslini, Department of Pediatrics, Unit of Rare Diseases Genova, Liguria, Italy, 16148 Japan The University of Tokyo Hospital Tokyo, Bunkyo-ku, Japan, 113-8655 Spain Hospital Universitari i Politècnic La Fe, Unidad de Reumatología Pediatrica Valencia, Avinguda De Fernando Abril Martorell, Nº 106, Spain, 46026 Sweden Norrlands Universitetssjukhus Umeå, Sweden, SE-90185 United Kingdom Royal National Orthopaedic Hospital, Brockely Hill Stanmore, United Kingdom, HA7 4LP Collapse << |
| NCT03022162 | - | - | Not yet recruiting | August 31, 2017 | - |
| NCT01041638 | Localized Resectable Neuroblas... more >>toma Localized Unresectable Neuroblastoma Regional Neuroblastoma Stage 4 Neuroblastoma Stage 4S Neuroblastoma Collapse << | Phase 3 | Completed | - | - |
| NCT00017225 | Neuroblastoma | Phase 2 | Completed | - | - |
| NCT03442985 | Exostoses, Multiple Hereditary | Phase 2 | Recruiting | February 2021 | United States, California ... more >> Children's Orthopaedic Center Recruiting Los Angeles, California, United States, 90027 Contact: Regina Woon 323-361-2843 rwoon@chla.usc.edu Principal Investigator: Vernon T Tolo, MD Sub-Investigator: Rachel Y Goldstein, MD, MPH University of California-San Francisco Recruiting San Francisco, California, United States, 94158 Contact: Emma Canepa 415-502-2425 Emma.Canepa@ucsf.edu Principal Investigator: Jessica Tenney, MD Sub-Investigator: Edward Hsiao, MD Sub-Investigator: Janet Lee, MD United States, Florida Paley Orthopedic and Spine Institute Recruiting West Palm Beach, Florida, United States, 330407 Contact: Troy Rand 561-531-1553 trand@paleyinstitute.org Principal Investigator: David Feldman, MD United States, Illinois Shriners Hospital for Children - Chicago Recruiting Chicago, Illinois, United States, 60707 Contact: Sahar Hassani, MS 773-622-5400 shassani@shrinenet.org Principal Investigator: Jeffrey Ackman, MD Sub-Investigator: Haluk Altiok, MD Sub-Investigator: Purnendu Gupta, MD Sub-Investigator: Peter Smith, MD United States, Massachusetts Boston Children's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Andrea Hale 167-135-5600 Andrea.Hale@childrens.harvard.edu Principal Investigator: Christina Jacobsen, MD Sub-Investigator: Ingrid Holm, MD, MPH Sub-Investigator: Samantha Spencer, MD United States, Pennsylvania The Children's Hospital of Philadelphia (CHOP) Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Vashisht Arshanapally, BS 267-426-7482 arshanapav@email.chop.edu Principal Investigator: Michael A Levine, MD Sub-Investigator: Edna Mancilla, MD Shriners Hospital for Children - Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19410-4160 Contact: Jamie Landgarten, MS 215-430-4239 jlandgarten@shrinenet.org Principal Investigator: Bethany Lipa, MD United States, Texas Memorial Hermann Hospital Recruiting Houston, Texas, United States, 77030 Contact: Mayank Rao 713-704-2639 Mayank.Rao@uth.tmc.edu Principal Investigator: Ernest Conrad, MD Sub-Investigator: Shiraz Younas, MD Canada, Ontario Hospital for Sick Children Recruiting Toronto, Ontario, Canada, M5G 1X8 Contact: Tony (Yongyao) Tan (416) 813-7654 ext 203575 tony.tan@sickkids.ca Principal Investigator: Roberto Mendoza-Londono, MD Sub-Investigator: Lucie Dupuis, MD Sub-Investigator: Andrew Howard, MD Sub-Investigator: Jennifer Stimec, MD Sub-Investigator: Irene Lara-Corrales, MD Sub-Investigator: Peter Kannu, MD Japan Nagoya University Hospital Recruiting Nagoya, Aichi, Japan Contact: Kumiko Tsukamoto +81-52-561-0166 kumiko-tsukamoto@irom.co.jp Principal Investigator: Hiroshi Kitoh, MD Sub-Investigator: Kenichi Mishima, MD Sub-Investigator: Tadashi Nagata, MD Sub-Investigator: Masaki Matsushita, MD Osaka University Hospital Recruiting Suita, Osaka, Japan, 565-0871 Contact: Hiromi Tanaka (SC) +81662108290 h-tanaka@dmi.med.osaka-u.ac.jp Principal Investigator: Keiichi Ozono, MD, PhD Sub-Investigator: Taichi Kitaoka, MD Sub-Investigator: Hiroyuki Saitou, MD Sub-Investigator: Dr. Kiyoshi Yoshida Sub-Investigator: Dr. Hidetatsu Otani Sub-Investigator: Dr. Kenichiro Hamada Sub-Investigator: Dr. Satoshi Takenaka Sub-Investigator: Hiroto Takahashi, MD Collapse << |
| NCT00675870 | Acute Promyelocytic Leukemia | Phase 2 | Unknown | - | United States, California ... more >> Sarcoma Oncology Center Recruiting Santa Monica,, California, United States, 90403 Contact: Victoria Chua 310-552-9999 vikychua@aol.com Collapse << |
| NCT00002658 | Leukemia Neut... more >>ropenia Collapse << | Phase 3 | Unknown | - | United Kingdom ... more >> University of Wales College of Medicine Cardiff, Wales, United Kingdom, CF14 4XN Collapse << |
| NCT01903317 | - | - | Withdrawn(No subjects enrolled... more >>) Collapse << | - | United States, California ... more >> University of California Irvine, Dermatology Irvine, California, United States, 92697 Collapse << |
| NCT00180128 | Leukemia, Promyelocytic, Acute | Phase 4 | Unknown | November 2012 | Germany ... more >> Department of Medicine I, University Hospital Carl Gustav Carus Recruiting Dresden, Germany, 01307 Contact: Markus Schaich, MD +49-351-458 ext -4251 markus.schaich@uniklinikum-dresden.de Contact: Silke Soucek +49-351-458 ext -4251 silke.soucek@uniklinikum-dresden.de Collapse << |
| NCT02348814 | - | - | Recruiting | March 2018 | United States, California ... more >> UCLA Clinical and Translational Research Center (CTRC) Recruiting Los Angeles, California, United States, 90095 Contact: Stephanie Okimoto, BA, CCRC 310-267-1040 sokimoto@mednet.ucla.edu Collapse << |
| NCT00124436 | Hand Dermatoses | Phase 3 | Completed | - | Germany ... more >> Thomas Ruzicka Düsseldorf, Germany, 40225 Collapse << |
| NCT00005823 | Leukemia Myel... more >>odysplastic Syndromes Myelodysplastic/Myeloproliferative Neoplasms Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> Queen Elizabeth Hospital at University of Birmingham Birmingham, England, United Kingdom, B15 2RR University College Hospital London, England, United Kingdom, WC1E 6AU University Hospital of Wales Cardiff, Wales, United Kingdom, CF14 4XN Collapse << |
| NCT00519675 | Eczema | Phase 3 | Completed | - | France ... more >> Paris, France Saint Etienne, France Germany Berlin, Germany Dresden, Germany Collapse << |
| NCT01245140 | Psoriasis | Phase 2 | Completed | - | France ... more >> GSK Investigational Site Paris Cedex 10, France, 75475 Germany GSK Investigational Site Muenchen, Bayern, Germany, 80337 GSK Investigational Site Witten, Nordrhein-Westfalen, Germany, 58453 GSK Investigational Site Berlin, Germany, 10117 GSK Investigational Site Berlin, Germany, 10827 GSK Investigational Site Hamburg, Germany, 20354 Netherlands GSK Investigational Site Nijmegen, Netherlands, 6525 GA United Kingdom GSK Investigational Site London, United Kingdom, SE1 7EH Collapse << |
| NCT01407679 | Lupus Erythematosus, Cutaneous | Phase 2 | Terminated | - | Germany ... more >> Department of Dematology, University Hospital Mannheim, Baden-Wuerttemberg, Germany, 68167 Department of Dermatology, Ludwig-Maximilians University Muenchen, Bayern, Germany, 80337 Department of Dermatology, University Hospital Muenster, Westfalen, Germany, 48149 Collapse << |
| NCT00276601 | Leukemia | Phase 2 | Completed | - | United States, Alabama ... more >> Comprehensive Cancer Center at University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Florida University of Florida Shands Cancer Center Gainesville, Florida, United States, 32610-0232 United States, Georgia Blood and Marrow Transplant Group of Georgia Atlanta, Georgia, United States, 30342 United States, Maryland Greenebaum Cancer Center at University of Maryland Medical Center Baltimore, Maryland, United States, 21201 Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States, 21231-2410 United States, Nebraska UNMC Eppley Cancer Center at the University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-3330 United States, Pennsylvania Hillman Cancer Center at University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00707174 | Cancer Lentig... more >>o Maligna Collapse << | Not Applicable | Completed | - | United States, Utah ... more >> Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT01245140 | - | - | Completed | - | - |
| NCT00707174 | - | - | Completed | - | - |
| NCT00050960 | Non-small Cell Lung Cancer | Phase 3 | Completed | - | - |
| NCT00050960 | - | - | Completed | - | - |
| NCT01711554 | Recurrent Neuroblastoma ... more >> Refractory Neuroblastoma Collapse << | Phase 1 | Suspended(Scheduled interim mo... more >>nitoring) Collapse << | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 Lucile Packard Children's Hospital Stanford University Palo Alto, California, United States, 94304 UCSF Medical Center-Parnassus San Francisco, California, United States, 94143 United States, Colorado Children's Hospital Colorado Aurora, Colorado, United States, 80045 United States, Georgia Children's Healthcare of Atlanta - Egleston Atlanta, Georgia, United States, 30322 United States, Illinois University of Chicago Comer Children's Hospital Chicago, Illinois, United States, 60637 United States, Massachusetts Boston Children's Hospital Boston, Massachusetts, United States, 02115 United States, Michigan C S Mott Children's Hospital Ann Arbor, Michigan, United States, 48109 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Texas Cook Children's Medical Center Fort Worth, Texas, United States, 76104 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Canada, Ontario Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT00050973 | Non-small Cell Lung Cancer | Phase 3 | Completed | - | - |
| NCT01560585 | Alzheimer's Disease | Phase 1 Phase 2 | Terminated | - | United States, Ohio ... more >> Parkway Medical Building Beachwood, Ohio, United States, 44122 Collapse << |
| NCT01948570 | - | - | Completed | - | Italy ... more >> DermIng S.r.l Monza, MB, Italy, 20900 Collapse << |
| NCT03626298 | Acne Vulgaris | Phase 4 | Completed | - | - |
| NCT02772874 | - | - | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania, Division of Urogynecology Philadelphia, Pennsylvania, United States, 19106 Collapse << |
| NCT02845232 | - | - | Completed | - | France ... more >> Hospices Civils de Lyon - Centre Hospitalier Lyon Sud, 165 Chemin du Grand Revoyet Pierre-benite, France, 69310 Collapse << |
| NCT00866918 | Childhood Acute Promyelocytic ... more >>Leukemia (M3) Untreated Childhood Acute Myeloid Leukemia and Other Myeloid Malignancies Collapse << | Phase 3 | Completed | - | - |
| NCT01226147 | Lupus Nephritis | Phase 2 | Unknown | February 2013 | Japan ... more >> Kinki University Hospital Recruiting Osaka, Japan, 5898511 Contact: Masanori Funauchi, M.D. mn-funa@med.kindai.ac.jp Principal Investigator: Masanori Funauchi, M.D. Collapse << |
| NCT00898755 | - | - | Completed | - | - |
| NCT00866918 | - | - | Completed | - | - |
| NCT01232179 | Proiriftrative Diabetic Retino... more >>pathy Collapse << | Phase 2 | Unknown | May 2011 | Iran, Islamic Republic of ... more >> Labbafinejad medical center Recruiting Tehran, Iran, Islamic Republic of Contact: Masoud Soheilian, proffesor 00982122585952 masoud_soheilian@yahoo.com Principal Investigator: masoud soheilian, proffesor Collapse << |
| NCT01232855 | - | - | Completed | - | - |
| NCT01526603 | Neuroblastoma | Not Applicable | Recruiting | February 2020 | United States, Minnesota ... more >> Masonic Cancer Center, University of Minnesota Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Kim Nelson 612-273-2925 knelso62@fairview.org Collapse << |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00609739 | - | - | Terminated(Low accrual) | - | - |
| NCT00551460 | Leukemia | Phase 2 | Completed | - | - |
| NCT00112502 | Brain and Central Nervous Syst... more >>em Tumors Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> Hembree Mercy Cancer Center at St. Edward Mercy Medical Center Fort Smith, Arkansas, United States, 72913 United States, Florida University of Texas MD Anderson Cancer Center at Orlando Orlando, Florida, United States, 32806-2134 United States, Georgia CCOP - Atlanta Regional Atlanta, Georgia, United States, 30342-1701 United States, Illinois CCOP - Central Illinois Decatur, Illinois, United States, 62526 United States, Kansas CCOP - Wichita Wichita, Kansas, United States, 67214-3882 United States, Michigan CCOP - Grand Rapids Grand Rapids, Michigan, United States, 49503 CCOP - Kalamazoo Kalamazoo, Michigan, United States, 49007-3731 United States, Missouri CCOP - Kansas City Kansas City, Missouri, United States, 64131 Cancer Research for the Ozarks Springfield, Missouri, United States, 65804 United States, Ohio Arthur G. James Cancer Hospital and Richard J. Solove Research Institute at Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210-1240 United States, South Carolina CCOP - Upstate Carolina Spartanburg, South Carolina, United States, 29303 United States, Texas University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030-4009 Collapse << |
| NCT01083498 | Pigmentation Disorder | Phase 2 | Completed | - | - |
| NCT00551460 | - | - | Completed | - | - |
| NCT02730819 | - | - | Completed | - | - |
| NCT00609739 | Leukemia | Phase 1 Phase 2 | Terminated(Low accrual) | - | United States, Minnesota ... more >> Masonic Cancer Center at University of Minnesota Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT02730819 | Melasma | Phase 2 | Completed | - | United States, Florida ... more >> Mayo Clinic in Florida Jacksonville, Florida, United States, 32224 Collapse << |
| NCT00866073 | Acute Myeloid Leukemia | Phase 2 | Completed | - | Germany ... more >> Medizinische Hochschule Hannover Hannover, Niedersachsen, Germany, 30625 Klinikum der Technischen Universität Aachen Aachen, Germany Universitätsklinikum Düsseldorf Düsseldorf, Germany Katholisches Krankenhaus Hagen Hagen, Germany Klinikum Lüdenscheid Luedenscheid, Germany Universitätsklinikum Ulm Ulm, Germany Klinikum Villingen-Schwenningen Villingen-Schwenningen, Germany Netherlands Leyenburg Hospital The Hague, Netherlands Collapse << |
| NCT02704338 | Autoimmune Diseases | Phase 1 Phase 2 | Unknown | October 2018 | - |
| NCT00254982 | - | - | Completed | - | - |
| NCT00975169 | - | - | Unknown | - | Taiwan ... more >> Department of Internal Medicine, National Taiwan University Hospital Recruiting Taipei, Taiwan Contact: Linda Huang 886 9 67670020 linda590203@gmail.com Principal Investigator: Tien-Jyun Chang, Ph D Collapse << |
| NCT03076320 | Acne Vulgaris Superficial Mixe... more >>d Comedonal and Inflammatory Collapse << | Phase 1 Phase 2 | Recruiting | March 31, 2019 | Mexico ... more >> Hospital Civil de Guadalajara Fray Antonio Alcalde Recruiting Guadalajara, Jalisco, Mexico, 44280 Contact: Yocasta Martínez Alvarado, MD +52 (33) 14794863 yocastadermatologia@gmail.com Contact: Marisol Ramirez Padilla, MD +52 (33) 31563006 yeuxenor@hotmail.com Principal Investigator: Yocasta Martínez-Alvarado, MD Sub-Investigator: Marisol Ramirez-Padilla, MD Collapse << |
| NCT00254982 | Psoriasis | Phase 3 | Completed | - | - |
| NCT02458417 | Segmental Vitiligo ... more >> Piebaldism Collapse << | Phase 4 | Completed | - | Netherlands ... more >> Netherlands Institute for Pigment disorders Amsterdam, Netherlands, 1105 AZ Collapse << |
| NCT00636038 | Vitamin A Status | Phase 1 | Completed | - | Zimbabwe ... more >> National University of Science and Technology Bulawayo, Zimbabwe Collapse << |
| NCT00000764 | HIV Infections ... more >> Anus Neoplasms Collapse << | Phase 1 | Completed | - | United States, Washington ... more >> University of Washington AIDS CRS Seattle, Washington, United States, 98122 Collapse << |
| NCT00972543 | Chronic Plaque Psoriasis | Phase 4 | Terminated(The study was termi... more >>nated after the European Medicines Evaluation Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT00489086 | Neoplastic Syndrome | Phase 2 | Completed | - | United States, California ... more >> Children's Hospital and Research Center Oakland Oakland, California, United States, 94609 United States, New York Herbert Irving Comprehensive Cancer Center at Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00489086 | - | - | Completed | - | - |
| NCT00470912 | Cutaneous Lupus Erythematosus ... more >> Skin Lesion Experimental Photoinduction Protective Sunscreen Collapse << | Phase 3 | Completed | - | Germany ... more >> Heinrich Heine University of Duesseldorf, Depatment of Dermatoly Duesseldorf, NRW, Germany, 40225 Collapse << |
| NCT01640678 | Piebaldism Se... more >>gmental Vitiligo Collapse << | Phase 4 | Unknown | December 2013 | Netherlands ... more >> The Netherlands Institute for pigment disorders, AMC Amsterdam, Netherlands, 1105 AZ Collapse << |
| NCT00141947 | Schizophrenia | Phase 3 | Unknown | - | Israel ... more >> Beersheva Mental Health Center Recruiting Beersheva, Israel Contact: Vladimir Lerner, MD, PhD 972-8-6401408 lernervld@yahoo.com Principal Investigator: Vladimir Lerner, MD, PhD Shaar Manashe Mental Health Center Recruiting Hadera, Israel Contact: Michael Ritsner, MD 9728-627-8750 ritsner@shaar-menashe.org.il Principal Investigator: Michael Ritsner, MD Collapse << |
| NCT00972543 | - | - | Terminated(The study was termi... more >>nated after the European Medicines Evaluation Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT00716014 | Pachyonychia Congenita | Phase 1 | Completed | - | United States, Utah ... more >> Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02421770 | Oral Lichen Planus | Phase 3 | Completed | - | Spain ... more >> Pia Lopez Jornet Murcia, Spain, 30008 Collapse << |
| NCT00321750 | Oral Erosive Lichen Planus | Phase 2 | Terminated | - | - |
| NCT00016159 | Leukemia | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT03237533 | Oral Lichen Planus | Early Phase 1 | Recruiting | December 31, 2017 | Pakistan ... more >> The university of Faisalabad Recruiting Faisalābād, Punjab, Pakistan, 38000 Contact: Sadaf Rashid, BDS +923237791689 durr_e_sadaf@hotmail.com Contact: Arsalan Wahid, M. Phil +923216685228 dr.arsalanmalik@gmail.com Collapse << |
| NCT01553071 | Solid Tumor | Phase 1 | Recruiting | - | United States, Texas ... more >> University of Texas Southwestern Not yet recruiting Dallas, Texas, United States, 75390 Contact: Dong Ying 214-648-5107 ying.dong@utsouthwestern.edu Contact: Marcella Aguilar 214-648-1479 marcella.aguilar@utsouthwestern.edu Principal Investigator: David Gerber, MD Joe Arrington Cancer Center/Covenant Children's Hospital Recruiting Lubbock, Texas, United States, 79410 Contact: Christy Ratheal 806-725-8131 cratheal@covhs.org Principal Investigator: Isaac Tafur, MD University Medical Center Recruiting Lubbock, Texas, United States, 79415 Contact: Melanie Bisbee 806-775-8590 melanie.bisbee@umchealthsystem.com Principal Investigator: Sanjay Awasthi, MD Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
