| NCT03345914 | Dermatitis, Atopic | Phase 3 | Recruiting | April 12, 2019 | United States, Alabama
... more >>
Regeneron Investigational Site
Recruiting
Birmingham, Alabama, United States, 35209
United States, California
Regneron Investigational Site
Recruiting
Bakersfield, California, United States, 93309
Regeneron Investigational Site
Recruiting
Long Beach, California, United States, 90808
Regeneron Investigational Site
Recruiting
Mission Viejo, California, United States, 92691
United States, Florida
Regeneron Investigational Site
Recruiting
Coral Gables, Florida, United States, 33146
United States, Georgia
Regeneron Investigational Site
Recruiting
Macon, Georgia, United States, 31217
Regeneron Investigational Site
Recruiting
Sandy Springs, Georgia, United States, 30328
United States, Maryland
Regeneron Investigational Site
Recruiting
Rockville, Maryland, United States, 20850
United States, New York
Regeneron Investigational Site
Recruiting
Forest Hills, New York, United States, 11375
Regeneron Investigational Site
Recruiting
New York, New York, United States, 10029
United States, Texas
Regeneron Investigational Site
Recruiting
Bellaire, Texas, United States, 77401
Regeneron Investigational Site
Recruiting
San Antonio, Texas, United States, 78218
United States, Virginia
Regeneron Investigational Site
Recruiting
Norfolk, Virginia, United States, 23502
Poland
Regeneron Investigational Site
Recruiting
Białystok, Poland, 15-453
Regeneron Investigational Site
Recruiting
Katowice, Poland, 40-611
Regeneron Investigational Site
Recruiting
Katowice, Poland, 40-648
Regeneron Investigational Site
Recruiting
Warsaw, Poland, 01-142
Regeneron Investigational Site
Recruiting
Warsaw, Poland, 02-758
Regeneron Investigational Site
Recruiting
Warszawa, Poland, 01-817
Regeneron Investigational Site
Recruiting
Wrocław, Poland, 50-381
Regeneron Investigational Site
Recruiting
Świętokrzyskie, Poland, 27-400 Collapse << |
| NCT02260986 | Atopic Dermatitis | Phase 3 | Completed | - | - |
| NCT01946386 | Psoriasis Vulgaris | Phase 1 | Completed | - | France
... more >> CPCAD, Centre de Pharmacologie Clinique Appliquée à la Dermatologie
Nice, France, 06202 Collapse << |
| NCT02260986 | - | - | Completed | - | - |
| NCT02404103 | - | - | Completed | - | - |
| NCT00570479 | Uveitis, Posterior
... more >> Glaucoma Collapse << | Phase 1 | Completed | - | United States, Texas
... more >>
Texas Retina Associates
Arlington, Texas, United States, 76012 Collapse << |
| NCT02748512 | Posterior Uveitis
... more >> Intermediate Uveitis
Panuveitis Collapse << | Phase 3 | Completed | - | United States, Colorado
... more >>
Retina Consultants of Southern Colorado PC
Colorado Springs, Colorado, United States, 80909-1183
United States, Massachusetts
Ocular Immunology and Uveitis Foundation
Waltham, Massachusetts, United States, 02451
United States, South Carolina
Charleston Neuroscience Center
Ladson, South Carolina, United States, 29456
United States, Texas
Texas Retina Associates - Fort Worth
Arlington, Texas, United States, 76012 Collapse << |
| NCT02404103 | Childhood Asthma | Not Applicable | Completed | - | United States, Kentucky
... more >>
University of Louisville Pediatric Pulmonology
Louisville, Kentucky, United States, 40202 Collapse << |
| NCT00669071 | - | - | Terminated(Enrollment closed) | - | - |
| NCT01743690 | Oral Lichen Planus
... more >> Candida Infection Collapse << | Not Applicable | Completed | - | Denmark
... more >> University of Copenhagen
Copenhagen, Denmark, 2100 Collapse << |
| NCT01998412 | - | - | Active, not recruiting | January 2020 | United States, Georgia
... more >>
Atlanta, Georgia, United States Collapse << |
| NCT00669071 | Melasma | Phase 4 | Terminated(Enrollment closed) | - | United States, California
... more >>
Dermatology/Cosmetic Laser Associates of La Jolla, Inc.
LaJolla, California, United States, 92037
United States, Tennessee
Tennessee Clinical Research Center
Nashville, Tennessee, United States, 37215 Collapse << |
| NCT00032396 | Macular Degeneration | Phase 2 | Unknown | - | United States, Massachusetts
... more >>
Control Delivery Systems, Inc.
Watertown, Massachusetts, United States, 02472 Collapse << |
| NCT00469183 | Melasma | Phase 4 | Completed | - | United States, California
... more >>
Vitiligo and Pigmentation Institute of Southern California
Los Angeles, California, United States, 90036
United States, Texas
University of Texas Southwestern Medical Center of Dallas
Dallas, Texas, United States, 75390 Collapse << |
| NCT02472366 | - | - | Completed | - | - |
| NCT02472366 | Chronic Diabetic Macular Edema | Phase 4 | Completed | - | - |
| NCT00720928 | Ocular Behcet's Disease,
... more >> Non-Infectious Uveitis
Refractory Uveitis Collapse << | Phase 4 | Unknown | May 2010 | Korea, Republic of
... more >>
Asan Medical Center
Recruiting
Seoul, Korea, Republic of, 138-736
Contact: Young Hee Yoon, MD 822-3010-3680 yhyoon@amc.seoul.kr
Contact: Don-Il Ham, MD 822-3410-3567 diham@smc.samsung.co.kr Collapse << |
| NCT02080091 | - | - | Terminated | - | - |
| NCT00132691 | - | - | Completed | - | - |
| NCT00975312 | Lentigo | Phase 2 | Completed | - | Chile
... more >> Pontificia Universidad Católica de Chile
Santiago, Chile, 6904413 Collapse << |
| NCT03238157 | Radiation Retinopathy | Phase 2
Phase 3 | Not yet recruiting | September 1, 2019 | United States, Ohio
... more >>
Cole Eye Instiute
Not yet recruiting
Cleveland, Ohio, United States, 44195
Contact: Arun Singh, MD 216-445-9479 singha@ccf.org
Contact: Rachel Rusnak 2164451256 rusnakr@ccf.org Collapse << |
| NCT02080091 | - | - | Terminated | - | Germany
... more >>
Neubrandenburg, Germany Collapse << |
| NCT00472966 | Melasma | Phase 4 | Completed | - | United States, Florida
... more >>
Skin Care Research, Inc.
Boca Raton, Florida, United States, 33486 Collapse << |
| NCT00132691 | Uveitis | Phase 4 | Completed | - | - |
| NCT00008515 | Choroidal Neovascularization
... more >> Macular Degeneration Collapse << | Phase 1 | Completed | - | United States, Maryland
... more >>
National Eye Institute (NEI)
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02168478 | - | - | Completed | - | - |
| NCT01867164 | Vaginitis
Inf... more >>ectious Vaginosis Collapse << | Phase 3 | Completed | - | Mexico
... more >>
DF, Mexico
Mexico D.F., Mexico Collapse << |
| NCT01694186 | Posterior Uveitis
... more >> Intermediate Uveitis
Panuveitis Collapse << | Phase 3 | Active, not recruiting | December 2018 | - |
| NCT01867164 | - | - | Completed | - | - |
| NCT00695318 | Age-Related Macular Degenerati... more >>on Collapse << | Phase 2 | Terminated | - | United States, Michigan
... more >>
Kresge Eye Institute
Detroit, Michigan, United States Collapse << |
| NCT02746991 | Posterior Uveitis
... more >> Intermediate Uveitis
Panuveitis Collapse << | Phase 3 | Active, not recruiting | June 2020 | India
... more >> LV. Prasad Eye Institute
Hyderabad, Andhra Pradesh, India, 500034
Sri Sankaradeva Nethralaya, Guwahati
Guwahati, Assam, India, 781028
Regional Institute of Opthalmology
Patna, Bihar, India, 800014
C.H. Nagri Municipal Eye Hospital
Ahmedabad, Gujarat, India, 380006
Seth G.S. Medical College & K.E.M Hospital
Mumbai, Maharashtra, India, 400012
Deenanath Mangeshkar Hospital, Pune
Pune, Maharashtra, India, 411004
PBMA'S, H. V. Desai Eye Hospital
Pune, Maharashtra, India, 411060
Dr. Shroff's Charity Eye Hospital
Daryaganj, New Delhi, India, 110002
Sankara Nethralaya hospital
Chennai, Tamil Nadu, India, 600006
Vasan Eye Care Hospital
Chennai, Tamil Nadu, India, 600015
Sri Ramachandra Hospital
Chennai, Tamil Nadu, India, 600116
J L Rohatgi Memorial Eye Hospital
Kanpur, Uttar Pradesh, India, 208005
King George's Medical University
Lucknow, Uttar Pradesh, India, 226003
ICARE Eye Hospital and Research centre
Noida, Uttar Pradesh, India, 201301
Regional Institute of Ophthalmology
Kolkata, West Bengal, India, 700073 Collapse << |
| NCT00695318 | - | - | Terminated | - | - |
| NCT00605423 | - | - | Completed | - | - |
| NCT01304706 | - | - | Completed | - | - |
| NCT01304706 | Diabetic Macular Edema | Phase 3 | Completed | - | United States, Georgia
... more >>
Alpharetta, Georgia, United States Collapse << |
| NCT00770770 | Macular Edema
... more >> Retinal Vein Occlusion Collapse << | Phase 2 | Terminated | - | United States, Kentucky
... more >>
University of Kentucky Department of Ophthalmology
Lexington, Kentucky, United States
United States, Massachusetts
Ophthalmic Consultants of Boston
Boston, Massachusetts, United States
United States, Ohio
Cole Eye Institute
Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02168478 | Allergy | Phase 4 | Completed | - | United States, New Jersey
... more >>
Michael Caswell PhD., CCRC,CCRA
Fairfield, New Jersey, United States, 07004 Collapse << |
| NCT00344968 | - | - | Completed | - | - |
| NCT00570830 | Intermediate Uveitis
... more >> Posterior Uveitis Collapse << | Not Applicable | Completed | - | United States, North Carolina
... more >>
Duke University Medical Center
Durham, North Carolina, United States, 07710 Collapse << |
| NCT03145025 | - | - | Not yet recruiting | August 2020 | - |
| NCT00770770 | - | - | Terminated | - | - |
| NCT00468871 | Non-infectious Uveitis | Phase 2
Phase 3 | Completed | - | - |
| NCT00502541 | Diabetic Macular Edema | Phase 2
Phase 3 | Completed | - | - |
| NCT00456482 | Non-infectious Uveitis | Phase 2
Phase 3 | Terminated(SARS epidemic in As... more >>ia and Canada) Collapse << | - | - |
| NCT00605423 | Age Related Macular Degenerati... more >>on Collapse << | Phase 2 | Completed | - | United States, Maryland
... more >>
Wilmer Eye Institute, Johns Hopkins University
Baltimore, Maryland, United States, 21287 Collapse << |
| NCT00407082 | Noninfectious Posterior Uveiti... more >>s Collapse << | Phase 2
Phase 3 | Completed | - | United States, North Carolina
... more >>
Duke Eye Center
Durham, North Carolina, United States, 27710 Collapse << |
| NCT00636493 | Retinal Vein Occlusion | Early Phase 1 | Completed | - | United States, North Carolina
... more >>
Duke University Eye Center
Durham,, North Carolina, United States, 27710 Collapse << |
| NCT00576459 | Diabetic Macular Edema | Phase 2 | Completed | - | China
... more >> The Chinese University of Hong Kong
Hong Kong, China
India
LV Prasad Eye Institute
Banjara Hills, Hyderabad, India, 500 034 Collapse << |
| NCT00952614 | - | - | Completed | - | - |
| NCT00344968 | Diabetic Macular Edema | Phase 3 | Completed | - | United States, Georgia
... more >>
Atlanta, Georgia, United States Collapse << |
| NCT03590587 | - | - | Recruiting | September 16, 2018 | United Arab Emirates
... more >>
Shiekh Khalifa medical city
Recruiting
Abu Dhabi, United Arab Emirates, 51900
Contact: Ahmed Elbarky 00971557443731 ext 00971557443731 AHMED.ALBARQY@fmed.bu.edu.eg
Sub-Investigator: tarek elhamaky, MD Collapse << |
| NCT00490815 | - | - | Completed | - | - |
| NCT00543296 | Uveitis | Phase 4 | Completed | - | United States, North Carolina
... more >>
Duke University Eye Center
Durham, North Carolina, United States, 27710 Collapse << |
| NCT00952614 | Central Retinal Vein Occlusion | Not Applicable | Completed | - | United States, North Carolina
... more >>
Duke Eye Center
Durham, North Carolina, United States, 27710 Collapse << |
| NCT00543296 | - | - | Completed | - | - |
| NCT02359526 | Diabetic Macular Edema | Phase 4 | Completed | - | Portugal
... more >> Center for Clinical Trials - Association for Innovation and Biomedical Research on Light and Image
Coimbra, Portugal, 3000-548
Instituto de Retina de Lisboa
Lisboa, Portugal, 1050-085
Hospital de São João
Porto, Portugal, 4200-319
Hospital Vila Franca Xira
Vila Franca de Xira, Portugal, 2600-009 Collapse << |
| NCT02902744 | Diabetic Macular Edema | Phase 4 | Withdrawn(The study was withdr... more >>awn due to difficult recruitment of patients for the study; No participants were enrolled in the study.) Collapse << | - | United States, Maryland
... more >>
Wilmer Eye Institute, Johns Hopkins University School of Medicine
Baltimore, Maryland, United States, 21218 Collapse << |
| NCT00490815 | Diabetic Macular Edema | Phase 3 | Completed | - | United States, Maryland
... more >>
John Hopkins University
Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01060787 | - | - | Recruiting | May 5, 2018 | United States, Massachusetts
... more >>
Ophthalmic Partners of Boston
Recruiting
Boston, Massachusetts, United States, 02114
Contact: Michael Raziman, MD Collapse << |
| NCT02424019 | Diabetic Macular Edema (DME) | Phase 4 | Active, not recruiting | January 2020 | - |
| NCT01781936 | Uveitis Affecting the Posterio... more >>r Segment Collapse << | Phase 1 | Active, not recruiting | December 2018 | United States, North Carolina
... more >>
Duke Eye Center
Durham, North Carolina, United States, 27710 Collapse << |
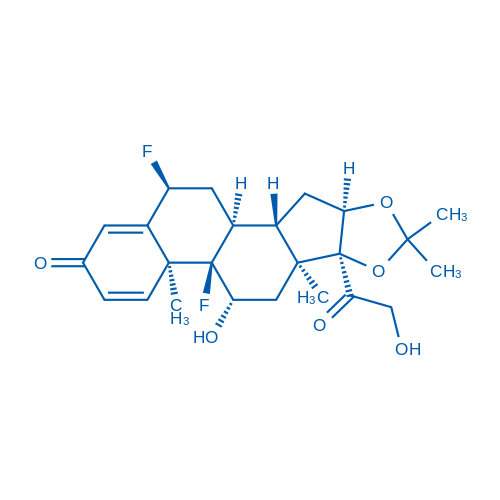

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
