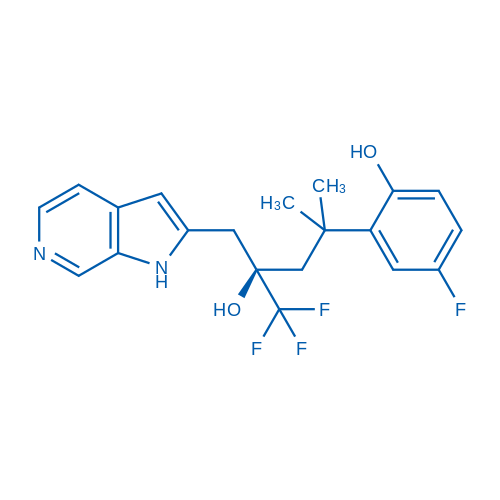
CAS No.: 1245526-82-2
Glucocorticoid receptor agonist Catalog No. CSN17617
Glucocorticoid receptor agonist is a potent Glucocorticoid receptor agonist.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02318043 Asthma Not Applicable Completed - - NCT03478683 Pulmonary Disease, Chronic Obs... more >>tructive Collapse << Phase 4 Active, not recruiting March 14, 2019 - NCT01347060 - - Completed - - - 更多
- 参考文献
- [1] Gauvreau GM, Boulet LP, et al. A nonsteroidal glucocorticoid receptor agonist inhibits allergen-induced late asthmatic responses. Am J Respir Crit Care Med. 2015 Jan 15;191(2):161-7.
- [2] Riether D, Harcken C, et al. Nonsteroidal dissociated glucocorticoid agonists containing azaindoles as steroid A-ring mimetics. J Med Chem. 2010 Sep 23;53(18):6681-98.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1245526-82-2 | 储存条件 |
|
|
| 分子式 | C20H20F4N2O2 | 运输 | 蓝冰 | |
| 分子量 | 396.38 | 别名 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02318043 | Asthma | Not Applicable | Completed | - | - |
| NCT03478683 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Active, not recruiting | March 14, 2019 | - |
| NCT01347060 | - | - | Completed | - | - |
| NCT03261921 | Caudal Block | Phase 1 | Completed | - | - |
| NCT01349257 | Asthma | Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Ltd Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT01332422 | - | - | Completed | - | - |
| NCT01381406 | - | - | Completed | - | - |
| NCT01381406 | - | - | Completed | - | - |
| NCT03478696 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Active, not recruiting | March 14, 2019 | - |
| NCT01381458 | - | - | Completed | - | - |
| NCT01386996 | Asthma | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Phase I Unit Espoo, Finland Collapse << |
| NCT01381471 | - | - | Completed | - | - |
| NCT01347060 | - | - | Completed | - | - |
| NCT01395862 | - | - | Completed | - | - |
| NCT01351792 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Terminated | - | Netherlands ... more >> Department of Pulmonary Diseases - University Medical Center Groningen Groningen, Netherlands, 9713 Collapse << |
| NCT01381471 | - | - | Completed | - | - |
| NCT01397890 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Completed | - | - |
| NCT01395849 | - | - | Completed | - | - |
| NCT01393145 | Chronic Obstructive Pulmonary ... more >>Disease COPD Collapse << | Phase 3 | Withdrawn | August 2012 | - |
| NCT01431924 | - | - | Completed | - | - |
| NCT01423305 | Asthma | Phase 1 | Completed | - | Finland ... more >> Clinical Reseach Services Turku (CRST) Turku, Finland, 20520 Collapse << |
| NCT01457716 | Healthy | Not Applicable | Completed | - | Germany ... more >> Parexel International GmbH Berlin, Germany Collapse << |
| NCT01453881 | Asthma | Phase 3 | Completed | - | Brazil ... more >> Hospital das Clínicas - Universidade Federal de Pernambuco Recife, Pernambuco, Brazil, 50740-520 Collapse << |
| NCT01450774 | Childhood Asthma | Phase 3 | Completed | - | Denmark ... more >> BørneAstmaKlinikken Copenhagen, Denmark, 2100 Collapse << |
| NCT01397890 | - | - | Completed | - | - |
| NCT01466712 | COPD Emphysem... more >>a Small Airway Disease Collapse << | Phase 4 | Unknown | December 2014 | Belgium ... more >> University Hospitals Leuven Leuven, Flanders, Belgium, 3000 Collapse << |
| NCT01462344 | Asthma | Phase 4 | Completed | - | - |
| NCT01462344 | - | - | Completed | - | - |
| NCT01468272 | Asthma in Children | Phase 2 | Completed | - | Denmark ... more >> BørneAstmaKlinikken, Hans Knudsens Plads 1A, Copenhagen,, Denmark, 2100 Collapse << |
| NCT01475721 | Asthma | Phase 4 | Completed | - | - |
| NCT01475721 | - | - | Completed | - | - |
| NCT01494610 | Asthma | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 New Zealand GSK Investigational Site Wellington, New Zealand, 6021 Collapse << |
| NCT01484210 | Asthma | Phase 3 | Completed | - | Czech Republic ... more >> Pneumology surgery Vitezna 201/31 Karlovy Vary, Drahovice, Czech Republic, 56001 Pneumology surgery Vojtesska 237 Kutna Hora, Zizkov, Czech Republic Pneumology and allergology surgery Voldusska 750 Nove Mesto, Czech Republic, 337 01 Pneumology surgery Hostinskeho 1536 Prague, Czech Republic, 1536 Pneumology surgery Generala Janouska 902/17 Prague, Czech Republic, 19800 Collapse << |
| NCT01540708 | Asthma | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01415518 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Completed | - | China, Beijing ... more >> Research Site Beijing, Beijing, China China, Guangdong Research Site Foshan, Guangdong, China Research Site Guangzhou, Guangdong, China Research Site Zhongshan, Guangdong, China China, Hainan Research Site Haikou, Hainan, China China, Hebei Research Site Shijiazhuang, Hebei, China Research Site Tangshan, Hebei, China China, Henan Research Site Zhengzhou, Henan, China China, Hubei Research Site Wuhan, Hubei, China China, Hunan Research Site Changsha, Hunan, China China, Jiangsu Research Site Nanjing, Jiangsu, China China, Jilin Research Site Changchun, Jilin, China China, Liaoning Research Site Shenyang, Liaoning, China China, Shandong Research Site Qingdao, Shandong, China China, Shanghai Research Site Shanghai, Shanghai, China China, Tianjin Research Site Tianjin, Tianjin, China China Research Site Chengdu, China Research Site Chongqin, China Research Site Da Lian, China Research Site Ha'er Bing, China Research Site Huhehaote, China Collapse << |
| NCT01494610 | - | - | Completed | - | - |
| NCT01560689 | Bronchiolitis Obliterans ... more >> Allogeneic Stem Cell Transplantation Collapse << | Phase 2 | Completed | - | France ... more >> Hopital Saint Louis APHP Paris, France, 75010 Collapse << |
| NCT01475032 | Asthma | Phase 3 | Completed | - | Bulgaria ... more >> UMHAT "Alexandrovska Plovdiv, Bulgaria, 4002 France Necker Enfants Malades Hospital Paris, France, 75015 Germany Drez. Gelb & Knecht Bretten, Germany, 75015 Hungary Micro Care Kft Szigetvár, Hungary, 7900 Italy G.Gaslini Institute Genoa, Italy, 16147 Poland Uniwersytetu Medycznego Łódź, Poland, 22 90-153 Romania Spitalul Clinic Universitar de Urgenta Elias Bucuresti, Romania, 011461 Russian Federation Moscow State Medical University Moscow, Russian Federation, 119435 Slovakia NZZ- Detská pneumologická a ftizeologická ambulanci Dolný Smokovec, Slovakia, 059 81 Spain Hospital de Sabadell Barcelona, Spain Ukraine Institute of Pediatrics Kyiv, Ukraine, 04050 Collapse << |
| NCT01415518 | - | - | Completed | - | - |
| NCT01593826 | Asthma | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Clinical Pharmacology Unit Espoo, Finland Collapse << |
| NCT01555099 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | - |
| NCT01602523 | Emphysema Poo... more >>r Sleep Quality Collapse << | Phase 3 | Unknown | August 2014 | United States, Pennsylvania ... more >> Temple Lung Center Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT01570478 | Asthma | Phase 3 | Completed | - | Italy ... more >> Dipartimento Cardio-Polmonare - Azienda Ospedaliero-Universitaria - Padiglione Rasori Parma, Italy, 43100 Collapse << |
| NCT01627158 | Asthma | Phase 1 | Completed | - | Germany ... more >> PAREXEL International GmbH Berlin, Germany, 14050 Collapse << |
| NCT00862264 | Asthma | Phase 3 | Completed | - | - |
| NCT01646424 | - | - | Completed | - | Turkey ... more >> Research Site Adana, Turkey Research Site Afyon, Turkey Research Site Ankara, Turkey Research Site Ayd?n, Turkey Research Site Balikesir, Turkey Research Site Bartin, Turkey Research Site Cankiri, Turkey Research Site Elazig, Turkey Research Site Gaziantep, Turkey Research Site Istanbul, Turkey Research Site Izmir, Turkey Research Site Kayseri, Turkey Research Site Kocaeli, Turkey Research Site Konya, Turkey Research Site Manisa, Turkey Research Site Mersin, Turkey Research Site Rize, Turkey Research Site Sakarya, Turkey Research Site Samsun, Turkey Research Site Yozgat, Turkey Collapse << |
| NCT01657487 | Percentage of Annual Acute Exa... more >>cerbation Quality of Life Collapse << | Phase 4 | Unknown | December 2012 | Taiwan ... more >> Far Eastern Memorial Hospital Recruiting Taipei, Taiwan, 886 Contact: Shih-Lung Cheng, MD, PhD 888-2-89667000 ext 2816 ntuhwyh61@yahoo.com.tw Principal Investigator: Shih-Lung Cheng, MD, PhD Collapse << |
| NCT01668121 | Asthma | Phase 1 | Completed | - | Germany ... more >> PAREXEL International GmbH Berlin, Germany Collapse << |
| NCT01706328 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01709903 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01751113 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | Japan ... more >> GSK Investigational Site Hokkaido, Japan, 070-8644 GSK Investigational Site Ibaraki, Japan, 319-1113 GSK Investigational Site Osaka, Japan, 530-0001 Collapse << |
| NCT01650441 | Asthma | Phase 2 | Completed | - | Belgium ... more >> University Hospital of Antwerp Edegem (Antwerp), Antwerp, Belgium, B-2650 Collapse << |
| NCT01709903 | - | - | Completed | - | - |
| NCT01760304 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Terminated(Sponsor decided to ... more >>stop the study due to expiration of blinded placebo .) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01706328 | - | - | Completed | - | - |
| NCT01751113 | - | - | Completed | - | - |
| NCT01760304 | - | - | Terminated(Sponsor decided to ... more >>stop the study due to expiration of blinded placebo .) Collapse << | - | - |
| NCT01738087 | Asthmatic | Phase 2 | Completed | - | United Kingdom ... more >> The Medicine Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT01766843 | Asthma | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Clinical Pharmacology Unit Espoo, Finland Collapse << |
| NCT01658891 | Asthma in Children | Phase 3 | Withdrawn | February 2016 | Denmark ... more >> BørneAstmaKlinikken Copenhagen, Denmark, 2100 Collapse << |
| NCT01762800 | - | - | Completed | - | - |
| NCT01785901 | - | - | Completed | - | China, Beijing ... more >> Research Site Beijing, Beijing, China China, Chongqing Research Site Chongqing, Chongqing, China China, Guangdong Research Site Guangzhou, Guangdong, China China, Heilongjiang Research Site Harbin, Heilongjiang, China China, Hunan Research Site Changsha, Hunan, China China, Inner Mongolia Research Site Baotou, Inner Mongolia, China China, Jiangsu Research Site Nanjing, Jiangsu, China China, Shandong Research Site Qingdao, Shandong, China China, Sichuan Research Site Chengdu, Sichuan, China China, Zhejiang Research site Hangzhou, Zhejiang, China Research Site Wenzhou, Zhejiang, China Collapse << |
| NCT00424008 | - | - | Completed | - | - |
| NCT01803087 | Asthma | Phase 2 | Completed | - | Poland ... more >> Uniwersytecki Szpital Kliniczny nr 1 Lodz, Poland, 90153 Collapse << |
| NCT01762800 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 814-0180 GSK Investigational Site Fukuoka, Japan, 816-0813 GSK Investigational Site Hiroshima, Japan, 720-0001 GSK Investigational Site Hiroshima, Japan, 734-8530 GSK Investigational Site Hiroshima, Japan, 737-0023 GSK Investigational Site Hiroshima, Japan, 737-0811 GSK Investigational Site Hokkaido, Japan, 070-8644 GSK Investigational Site Hyogo, Japan, 670-0849 GSK Investigational Site Ibaraki, Japan, 300-0053 GSK Investigational Site Ibaraki, Japan, 302-0022 GSK Investigational Site Ibaraki, Japan, 309-1793 GSK Investigational Site Ibaraki, Japan, 311-3193 GSK Investigational Site Ibaraki, Japan, 319-1113 GSK Investigational Site Kagawa, Japan, 760-0018 GSK Investigational Site Kagawa, Japan, 760-8538 GSK Investigational Site Kagawa, Japan, 761-8073 GSK Investigational Site Kanagawa, Japan, 233-0013 GSK Investigational Site Kanagawa, Japan, 253-0083 GSK Investigational Site Kochi, Japan, 780-0901 GSK Investigational Site Kyoto, Japan, 602-8026 GSK Investigational Site Kyoto, Japan, 610-0113 GSK Investigational Site Nara, Japan, 630-0293 GSK Investigational Site Niigata, Japan, 950-1197 GSK Investigational Site Niigata, Japan, 950-2085 GSK Investigational Site Niigata, Japan, 950-8725 GSK Investigational Site Okinawa, Japan, 901-2132 GSK Investigational Site Okinawa, Japan, 904-2143 GSK Investigational Site Okinawa, Japan, 904-2293 GSK Investigational Site Osaka, Japan, 530-0001 GSK Investigational Site Osaka, Japan, 559-0011 GSK Investigational Site Osaka, Japan, 560-0005 GSK Investigational Site Osaka, Japan, 560-8552 GSK Investigational Site Saga, Japan, 840-8571 GSK Investigational Site Shizuoka, Japan, 434-8511 GSK Investigational Site Tokyo, Japan, 103-0027 GSK Investigational Site Tokyo, Japan, 103-0028 GSK Investigational Site Tokyo, Japan, 158-0083 GSK Investigational Site Tokyo, Japan, 187-8510 GSK Investigational Site Yamaguchi, Japan, 755-0241 Collapse << |
| NCT01795664 | Asthma | Phase 3 | Completed | - | Belgium ... more >> Antwerp University Hospital Edegem, Antwerp, Belgium, 2650 Collapse << |
| NCT01817764 | - | - | Completed | - | - |
| NCT00424008 | Asthma | Phase 3 | Completed | - | - |
| NCT01584492 | Asthma | Phase 2 | Completed | - | Poland ... more >> Poradnia Alergologiczna Debica, Debica, Poland, Poland, 39 -200 Ukraine Zaporizhzhia State Medical University Zaporizhzhia, Ukraine, 69063 Collapse << |
| NCT01803555 | Asthma | Phase 3 | Completed | - | - |
| NCT01822899 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01822899 | - | - | Completed | - | - |
| NCT01817764 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01696214 | Asthma | Phase 4 | Completed | - | United States, California ... more >> Airway Research & Clinical Trials Center San Diego, California, United States, 92103 Collapse << |
| NCT01696214 | - | - | Completed | - | - |
| NCT01848769 | Asthma | Phase 2 | Completed | - | Denmark ... more >> BorneAstmaKlinikken Copenhagen, Denmark, 2100 Collapse << |
| NCT01856621 | Asthma | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Clinical Pharmacology Unit Espoo, Finland Collapse << |
| NCT01857479 | Allergic Bronchopulmonary Aspe... more >>rgillosis Collapse << | Phase 2 Phase 3 | Completed | - | India ... more >> Chest Clinic, PGIMER Chandigarh, India, 160012 Collapse << |
| NCT01879410 | - | - | Completed | - | - |
| NCT01834885 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Withdrawn | September 2014 | Austria ... more >> Novartis Investigative Site Feldkirch, Austria, 6800 Novartis Investigative Site Grieskirchen, Austria, 4710 Novartis Investigative Site Hallein, Austria, 5400 Novartis Investigative Site Linz, Austria, 4020 Novartis Investigative Site Thalheim bei Wels, Austria, 4600 Novartis Investigative Site Wels, Austria, 4600 Germany Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Bad Woerishofen, Germany, 86825 Novartis Investigative Site Berlin, Germany, 10117 Novartis Investigative Site Berlin, Germany, 10119 Novartis Investigative Site Berlin, Germany, 10717 Novartis Investigative Site Berlin, Germany, 10789 Novartis Investigative Site Berlin, Germany, 12203 Novartis Investigative Site Berlin, Germany, 12687 Novartis Investigative Site Berlin, Germany, 13156 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Frankfurt, Germany, 60389 Novartis Investigative Site Frankfurt, Germany, 60596 Novartis Investigative Site Grosshansdorf, Germany, 22947 Novartis Investigative Site Hamburg, Germany, 20253 Novartis Investigative Site Hamburg, Germany, 20354 Novartis Investigative Site Hamburg, Germany, 22335 Novartis Investigative Site Hannover, Germany, 30317 Novartis Investigative Site Leipzig, Germany, 04207 Novartis Investigative Site Leipzig, Germany, 04357 Novartis Investigative Site Lübeck, Germany, 23552 Novartis Investigative Site Münster, Germany, 48145 Novartis Investigative Site Potsdam, Germany, 14467 Novartis Investigative Site Rheine, Germany, 48431 Collapse << |
| NCT01860066 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Withdrawn | September 2014 | Argentina ... more >> Novartis Investigative Site San Miguel de Tucuman, Tucuman, Argentina, 4000 Novartis Investigative Site Buenos Aires, Argentina, 1425 Novartis Investigative Site Buenos Aires, Argentina, C1115AAB Novartis Investigative Site Buenos Aires, Argentina, C1122AAK Novartis Investigative Site Buenos Aires, Argentina Novartis Investigative Site Mendoza, Argentina, 5500 Novartis Investigative Site Mendoza, Argentina, M5500CBA Belgium Novartis Investigative Site Antwerpen, Belgium, 2060 Novartis Investigative Site Bruxelles, Belgium, 1000 Novartis Investigative Site Genk, Belgium, 3600 Novartis Investigative Site Gilly, Belgium, 6060 Novartis Investigative Site Hasselt, Belgium, 3500 Novartis Investigative Site Jambes, Belgium, 5100 Colombia Novartis Investigative Site Barranquilla, Colombia Novartis Investigative Site Florida Blanca, Colombia Novartis Investigative Site Medellín, Colombia Czech Republic Novartis Investigative Site Kyjov, CZE, Czech Republic, 697 70 Novartis Investigative Site Cvikov, Czech Republic, 471 54 Novartis Investigative Site Jindrichuv Hradec, Czech Republic, 377 01 Novartis Investigative Site Karlovy Vary, Czech Republic, 360 66 Novartis Investigative Site Kurim, Czech Republic, 664 34 Novartis Investigative Site Pardubice, Czech Republic, 530 09 Novartis Investigative Site Teplice, Czech Republic, 415 01 India Novartis Investigative Site Guntur, Andhra Pradesh, India, 522 001 Novartis Investigative Site Ahmedabad, Gujarat, India, 380 008 Novartis Investigative Site Faridabad, Haryana, India, 121 001 Novartis Investigative Site Bangalore, Karnataka, India, 560 002 Novartis Investigative Site Nagpur, Maharashtra, India, 400 012 Novartis Investigative Site Pune, Maharashtra, India, 411004 Novartis Investigative Site Jaipur, Rajasthan, India, 302004 Novartis Investigative Site Karamsad, India, 388225 Collapse << |
| NCT01879410 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01911364 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Germany ... more >> Institut für klinische Forschung Hessen, Germany Hungary Csongrád Megyei Mellkasi Betegségek Szakkórháza Szeged, Hungary Italy Azienda Ospedaliera Perugia Perugia, Italy Collapse << |
| NCT01912872 | Severe IgE-mediated Asthma | Phase 4 | Unknown | November 2016 | Mexico ... more >> Novartis Investigative Site Completed Tuxtla Gutierrez, Chiapas, Mexico, 29030 Novartis Investigative Site Terminated Mexico City, Distrito Federal, Mexico, 14000 Novartis Investigative Site Completed Mexico, Distrito Federal, Mexico, 03020 Novartis Investigative Site Recruiting Mexico, Distrito Federal, Mexico, 03100 Novartis Investigative Site Recruiting Mexico, Distrito Federal, Mexico, 04700 Novartis Investigative Site Recruiting Mexico, Distrito Federal, Mexico, 04980 Novartis Investigative Site Completed Mexico, Distrito Federal, Mexico, 06090 Novartis Investigative Site Completed Mexico, Distrito Federal, Mexico, 06760 Novartis Investigative Site Withdrawn Mexico, Distrito Federal, Mexico, 10700 Novartis Investigative Site Completed Mexico, Distrito Federal, Mexico, 14050 Novartis Investigative Site Terminated Mexico, Edo. de México, Mexico, 53910 Novartis Investigative Site Completed Nezahualcoyotl, Estado de Mexico, Mexico, 57730 Novartis Investigative Site Active, not recruiting Pachuca de Soto, Hidalgo, Mexico, 42090 Novartis Investigative Site Completed Guadalajara, Jalisco, Mexico, 44600 Novartis Investigative Site Completed Guadalajara, Jalisco, Mexico, 44620 Novartis Investigative Site Completed Guadalajara, Jalisco, Mexico, 44690 Novartis Investigative Site Completed Guadaljara, Jalisco, Mexico, 44500 Novartis Investigative Site Recruiting Tepic, Nayarit, Mexico, 63000 Novartis Investigative Site Recruiting Monterrey, Nuevo León, Mexico, 64020 Novartis Investigative Site Terminated Monterrey, Nuevo León, Mexico, 64718 Novartis Investigative Site Completed Mérida, Yucatán, Mexico, 97070 Collapse << |
| NCT01890863 | Asthma | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01917331 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Hungary ... more >> Dr Beatrix BALINT Szeged, Hungary, 6722 Collapse << |
| NCT01769898 | Bronchiectasis | Phase 4 | Completed | - | China, Guangdong ... more >> State Key Laboratory of Respiratory Research Institute. Guangzhou City, Guangdong, China, 510000 The First Affiliated Hospital of Guangzhou Medical University Guangzhou, Guangdong, China, 510000 Collapse << |
| NCT01836016 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Unknown | May 2015 | China, Henan ... more >> The First Affiliated Hospital of Henan University of Traditional Chinese Medicine Not yet recruiting Zhengzhou, Henan, China, 450000 Principal Investigator: Jiansheng Li, MD Collapse << |
| NCT01933984 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Not Applicable | Unknown | - | China, Taiwan ... more >> Changhua Christian Hospital Recruiting Changhua, Taiwan, China, 50006 Contact: Shin-hwar Wu 886-4-723-8595 ext 3978 126366@cch.org.tw Contact: Kai-huang Lin 886-4-723-8595 ext 3971 54407@cch.org.tw Principal Investigator: Shin-hwar Wu Sub-Investigator: Kai-huang Lin Sub-Investigator: Chin-hsing Li Sub-Investigator: Lih-jen Shyu Collapse << |
| NCT01794780 | COPD | Not Applicable | Completed | - | China ... more >> Novartis Investigative Site Beijing, China, 100730 Collapse << |
| NCT01907334 | - | - | Completed | - | - |
| NCT01978145 | - | - | Completed | - | - |
| NCT01978145 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01907334 | Asthma | Phase 4 | Completed | - | United States, Florida ... more >> University of Florida Asthma Research Lab Gainesville, Florida, United States, 32610-0486 Collapse << |
| NCT02031640 | Asthma | Phase 3 | Completed | - | - |
| NCT01980615 | Healthy Volunteers | Phase 1 | Completed | - | United States, Maryland ... more >> Pearl Therapeutics Study Site Baltimore, Maryland, United States Collapse << |
| NCT02040766 | Asthma | Phase 3 | Completed | - | - |
| NCT02000609 | COPD | Phase 2 | Completed | - | United Kingdom ... more >> Medicine Evaluation Unit Manchester, United Kingdom, M239Qz Collapse << |
| NCT02040779 | - | - | Completed | - | - |
| NCT02045953 | Asthma | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT02040779 | Persistent Asthma | Phase 3 | Completed | - | - |
| NCT02055352 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | Argentina ... more >> Novartis Investigative Site Caba, Buenos Aires, Argentina, C1056ABJ Novartis Investigative Site Florida, Buenos Aires, Argentina, B1602DQD Novartis Investigative Site La Plata, Buenos Aires, Argentina, 1900 Novartis Investigative Site Vicente Lopez, Buenos Aires, Argentina, B1638AAI Novartis Investigative Site Zarate, Buenos Aires, Argentina, 2800 Novartis Investigative Site Santa Rosa, La Pampa, Argentina Novartis Investigative Site Buenos Aires, Argentina, C1125ABE Novartis Investigative Site Ciudad Autonoma de Bs As, Argentina, C1425FVH Novartis Investigative Site Salta, Argentina, 4000 Brazil Novartis Investigative Site Rio de Janeiro, RJ, Brazil, 021941-617 Novartis Investigative Site Porto Alegre, RS, Brazil, 90880-480 Novartis Investigative Site Sao Bernardo do Campo, SP, Brazil, 09750-420 Novartis Investigative Site São Paulo, SP, Brazil, 01244-030 Chile Novartis Investigative Site Santiago, Region Metropolitana, Chile, 8431633 Novartis Investigative Site Rancagua, Chile, 2841959 Dominican Republic Novartis Investigative Site Santo Domingo, Republica Dominicana, Dominican Republic, 10205 Novartis Investigative Site Santo Domingo, Republica Dominicana, Dominican Republic Ecuador Novartis Investigative Site Guayaquil, Guayas, Ecuador, 412 Honduras Novartis Investigative Site San Pedro Sula, Honduras, 21102 Novartis Investigative Site Tegucigalpa, Honduras Mexico Novartis Investigative Site Mexico, Distrito Federal, Mexico, 03310 Novartis Investigative Site Mexico, Distrito Federal, Mexico, 06760 Novartis Investigative Site Mexico, Distrito Federal, Mexico, 14050 Novartis Investigative Site León, Guanajuato, Mexico, 37000 Panama Novartis Investigative Site Panama City, Panamá, Panama Collapse << |
| NCT02031640 | - | - | Completed | - | - |
| NCT02062463 | Asthma | Phase 4 | Completed | - | - |
| NCT02040597 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 1 | Completed | - | Poland ... more >> Biovirtus Research Site Nadarzyn, Mokra 7, Poland, 05-830 Medical University in Lodz Lodz, ul. Kopcińskiego 22, Poland, 90-153 Collapse << |
| NCT02091986 | Asthma | Phase 3 | Completed | - | - |
| NCT02055352 | - | - | Completed | - | - |
| NCT02040766 | - | - | Completed | - | - |
| NCT02113436 | Asthma | Phase 4 | Completed | - | - |
| NCT02113436 | - | - | Completed | - | - |
| NCT02091986 | - | - | Completed | - | - |
| NCT02119234 | COPD | Phase 1 | Completed | - | Poland ... more >> Medical University in Lodz Lodz, ul. Kopcińskiego 22, Poland, 90-153 Collapse << |
| NCT02149199 | Asthma | Phase 3 | Completed | - | - |
| NCT02136875 | COPD Chronic ... more >>Obstructive Pulmonary Disease Collapse << | Phase 4 | Completed | - | Canada, Quebec ... more >> Montreal Chest Institute Montreal, Quebec, Canada, H2X2P4 Collapse << |
| NCT02189304 | COPD | Phase 1 | Completed | - | United States, Maryland ... more >> SNBL Clinical Pharmacology Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT02162485 | Asthma | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Pharmacology Unit Espoo, Finland Collapse << |
| NCT02162784 | Asthma | Phase 2 | Completed | - | Taiwan ... more >> Chung Shan Medical University Hospital TaiChung, Taiwan National Taiwan University Hospital Taipei, Taiwan Tri-Service General Hospital Taipei, Taiwan Collapse << |
| NCT02197975 | COPD | Phase 1 | Completed | - | United States, Maryland ... more >> SNBL Clinical Pharmacology Center Baltimore, Maryland, United States, 21201 Collapse << |
| NCT02196077 | - | - | Completed | - | - |
| NCT02218762 | Asthma | Phase 1 | Withdrawn(It was decided that ... more >>data from this study are no longer required and therefore it is not necessary to expose patients to the study medication) Collapse << | June 2015 | - |
| NCT02237508 | Asthma | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Clinical Ltd Ruddington, United Kingdom, NG11 6JS Collapse << |
| NCT02196077 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pearl Investigative Site Rolling Hills Estates, California, United States United States, Florida Pearl Investigative Site Clearwater, Florida, United States Pearl Investigative Site Panama City, Florida, United States Pearl Investigative Site Tampa, Florida, United States Pearl Investigative Site Winter Park, Florida, United States United States, Minnesota Pearl Investigative Site Edina, Minnesota, United States Pearl Investigative Site Minneapolis, Minnesota, United States Pearl Investigative Site Woodbury, Minnesota, United States United States, Missouri Pearl Investigative Site Saint Charles, Missouri, United States United States, North Carolina Pearl Investigative Site Charlotte, North Carolina, United States United States, Ohio Pearl Investigative Site Cincinnati, Ohio, United States Pearl Investigative Site Columbus, Ohio, United States Pearl Investigative Site Dublin, Ohio, United States United States, Oregon Pearl Investigative Site Medford, Oregon, United States United States, South Carolina Pearl Investigative Site Easley, South Carolina, United States Pearl Investigative Site Greenville, South Carolina, United States Pearl Investigative Site Spartanburg, South Carolina, United States Collapse << |
| NCT02224157 | Asthma | Phase 3 | Completed | - | - |
| NCT02260492 | Asthma | Phase 1 | Completed | - | - |
| NCT02260492 | - | - | Completed | - | - |
| NCT02345993 | Asthma Asthma... more >>tic Crisis Collapse << | Phase 4 | Unknown | August 2015 | Mexico ... more >> National Institute of Respiratory Diseases Mexico, Distrito Federal, Mexico, 14080 Collapse << |
| NCT02227394 | Asthma Asthma... more >> Chronic Asthma Bronchial Asthmatic Collapse << | Phase 2 | Completed | - | Belgium ... more >> Antwerp University Hospital Edegem, Antwerp, Belgium, 2650 Collapse << |
| NCT02384577 | - | - | Completed | - | Germany ... more >> Teva Investigational Sites Berlin, Germany Collapse << |
| NCT02415179 | Asthma | Not Applicable | Completed | - | Thailand ... more >> Faculty of Medicine, Ramathibodi Hospital Bangkok, Thailand Collapse << |
| NCT02148120 | Asthma | Phase 2 | Completed | - | United Kingdom ... more >> Hammersmith Medicines Research London, United Kingdom, NW10 7EW Respiratory Clinical Trials, Hearth Lung Centre London, United Kingdom, W1G 8HU Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02270424 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Unknown | December 2016 | - |
| NCT02388373 | Asthma | Phase 4 | Completed | - | United Kingdom ... more >> Research in Real Life Ltd Cambridge, United Kingdom, CB24 3BA Collapse << |
| NCT01387178 | - | - | Completed | - | - |
| NCT00633919 | Allergy | Phase 2 Phase 3 | Completed | - | Spain ... more >> Fernando Rodríguez Santander, Cantabria, Spain Collapse << |
| NCT02245672 | Asthma | Phase 3 | Completed | - | - |
| NCT01387178 | - | - | Completed | - | - |
| NCT02215122 | Asthma COPD | Phase 1 | Completed | - | United States, Texas ... more >> Worldwide Clinical Trials Early Phase Services San Antonio, Texas, United States, 78217 Collapse << |
| NCT02061280 | Asthma | Phase 4 | Completed | - | United States, Delaware ... more >> Alfred I. duPont Hospital for Children Wilmington, Delaware, United States, 19803 United States, Florida Nemours Children's Specialty Care Jacksonville, Florida, United States, 32207 Nemours Children's Hospital Orlando, Florida, United States, 32806 Nemours Children's Specialty Care Pensacola, Florida, United States, 32504 United States, Missouri Washington University in St. Louis Saint Louis, Missouri, United States, 63130 Collapse << |
| NCT00633919 | - | - | Completed | - | - |
| NCT02061280 | - | - | Completed | - | - |
| NCT01524198 | - | - | Completed | - | - |
| NCT02466347 | Asthma Chroni... more >>c Obstructive Pulmonary Disease (COPD) Collapse << | Phase 1 | Completed | - | Taiwan ... more >> Clinical Pharmacology Unit of Mackay Memorial Hospital Tamshui Branch New Taipei City, Taiwan Collapse << |
| NCT01524198 | Status Asthmaticus | Phase 2 Phase 3 | Completed | - | Saudi Arabia ... more >> King Fahad Medical City Riyadh, Saudi Arabia, 11525 Collapse << |
| NCT02466503 | Asthma Chroni... more >>c Obstructive Pulmonary Disease (COPD) Collapse << | Phase 1 | Completed | - | Taiwan ... more >> Clinical Pharmacology Unit of Mackay Memorial Hospital Tamshui Branch New Taipei City, Taiwan Collapse << |
| NCT00102882 | Asthma | Phase 4 | Completed | - | - |
| NCT02233803 | Asthma | Phase 4 | Completed | - | Argentina ... more >> GSK Investigational Site Ciudad autónoma de Buenos Aires, Buenos Aires, Argentina, C1028AAP GSK Investigational Site La Plata, Buenos Aires, Argentina, 1900 GSK Investigational Site Mar del Plata, Buenos Aires, Argentina, 7600 GSK Investigational Site Mar del Plata, Buenos Aires, Argentina, B7600FZN GSK Investigational Site Vicente Lopez, Buenos Aires, Argentina, B1602DOH GSK Investigational Site Rosario, Santa Fe, Argentina, 2000 GSK Investigational Site Berazategui, Buenos Aires, Argentina, B1884AAC GSK Investigational Site Berazategui, Argentina, 1886 GSK Investigational Site Ciudad Autónoma de Buenos Aires, Argentina, C1121ABE GSK Investigational Site Monte Grande, Argentina, 1842 GSK Investigational Site Rosario, Argentina, 2000 Collapse << |
| NCT01687296 | Asthma | Phase 3 | Completed | - | China, Hunan ... more >> GSK Investigational Site Changsha, Hunan, China, 410005 China, Jilin GSK Investigational Site Yanji, Jilin, China, 133000 China, Liaoning GSK Investigational Site Shenyang, Liaoning, China, 110004 China, Zhejiang GSK Investigational Site Wenzhou, Zhejiang, China, 323027 China GSK Investigational Site Beijing, China, 100191 GSK Investigational Site Beijing, China GSK Investigational Site Changchun, China GSK Investigational Site Chongqing, China, 400014 GSK Investigational Site Shanghai, China, 200040 GSK Investigational Site Shanghai, China, 200092 GSK Investigational Site Wuxi, China, 214023 Collapse << |
| NCT02233803 | - | - | Completed | - | - |
| NCT01687296 | - | - | Completed | - | - |
| NCT00449501 | Asthma | Phase 3 | Completed | - | - |
| NCT02850484 | Asthma Chroni... more >>c Obstructive Pulmonary Disease (COPD) Collapse << | Phase 1 | Completed | - | - |
| NCT00319306 | Asthma | Phase 3 | Completed | - | - |
| NCT00461500 | Asthma | Phase 4 | Completed | - | - |
| NCT00461500 | - | - | Completed | - | - |
| NCT02139644 | - | - | Completed | - | - |
| NCT00463866 | Asthma | Phase 4 | Completed | - | - |
| NCT00449527 | Asthma | Phase 3 | Completed | - | - |
| NCT00480649 | Asthma | Phase 4 | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Anyang-Si, Korea, Republic of, 431-070 GSK Investigational Site Busan-si, Korea, Republic of GSK Investigational Site Busan, Korea, Republic of GSK Investigational Site Junjoo-Si, Korea, Republic of GSK Investigational Site Kyungki-Do Anyang-si, Korea, Republic of GSK Investigational Site Kyungki-Do, Korea, Republic of GSK Investigational Site Pusan, Korea, Republic of, 602-739 GSK Investigational Site Pusan, Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 120-752 GSK Investigational Site Seoul, Korea, Republic of, 140-743 GSK Investigational Site Seoul, Korea, Republic of, 152-703 GSK Investigational Site Seoul, Korea, Republic of GSK Investigational Site 서울시, Korea, Republic of GSK Investigational Site 전주시, Korea, Republic of Collapse << |
| NCT00487773 | Asthma | Phase 4 | Completed | - | Poland ... more >> Department of Pediatrics and Allergy, Medical University of Lodz Lodz, Poland, 93-513 Collapse << |
| NCT02139644 | Asthma | Phase 3 | Completed | - | - |
| NCT00489853 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | Germany ... more >> Research Site Berlin, Germany Research Site Erfurt, Germany Research Site Fulda, Germany Research Site Furth, Germany Research Site Geesthacht, Germany Research Site Grobhansdorf, Germany Research Site Leipzig, Germany Research Site Neuruppin, Germany Switzerland Research Site Basel, Basel Stadt, Switzerland Collapse << |
| NCT00463866 | - | - | Completed | - | - |
| NCT00476099 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | France ... more >> Thomas Similowski Paris, France, 75000 Collapse << |
| NCT00489853 | - | - | Completed | - | - |
| NCT00496470 | - | - | Completed | - | - |
| NCT00497237 | Asthma | Phase 3 | Completed | - | Bulgaria ... more >> Clinic of Pulmonology, UMHAT "Sveti Georgi" Plovdiv, Bulgaria, 4002 First Department of Pulmonology, Regional Dispensery of Pulmonology and Phtisiatric Diseases with Stationary (RDPPDS) Ruse, Bulgaria, 7002 Clinic of Pulmonology, University Hospital "Lozenetz" Sofia, Bulgaria First Internal Clinic, Endocrinology and Pulmonology Department MHAT Stara Zagora, Bulgaria, 6000 Italy U.O.C. S.Anna e S. Sebastiano - Malattie dell'apparato respiratorio Caserta, Italy Ospedale S. Camillo de Lellis - U.O.C. Pneumologia Chieti, Italy Ospedale Cardarelli - Fisiopatologia Respiratoria Napoli, Italy CNR - Dipartimento di Fisiopatologia Respiratoria Palermo, Italy Ukraine Department of Hospital Pediatrics Crimean State Medical University. Pulmonology Department of Republican Clinical Children's Hospital Crimea, Ukraine, 95004 Pulmonological Department #2 Kharkiv, Ukraine, 61035 Pulmonological Department of the Institute of Therapy, Ukrainian Academy of Medical Sciences Kharkiv, Ukraine, 61035 Pulmonological and Allergological Department of the Kharkov Regional Clinical Hospital Kharkov, Ukraine, 61022 Department of General Practice- Family medicine. Medical Academy of postgraduate education. Kharkov, Ukraine Department of Diagnostic, Therapy and Clinical Pharmacology of Lung Diseases of the Institute of Phthisiology and Pulmonology Academy of Medical Science of the Ukraine Kiev, Ukraine, 03680 Institute of pthysiology and pulmonology Academy of medical science of the Ukraine. Kiev, Ukraine, 03680 Pulmonology Department of the Institute of Phthisiology and Pulmonology AMS of the Ukraine Kiev, Ukraine, 03680 Clinical Hospital 8, Department of pediatrics and clinical laboratories Kriviy Rig, Ukraine Department of Hospital Therapy of Lugansk State Medical Institute. Lugansk Regional Clinical Hospital Lugansk, Ukraine, 91045 Collapse << |
| NCT00479739 | Asthma | Phase 4 | Completed | - | - |
| NCT00497523 | Bronchial Asthma | Phase 3 | Completed | - | Poland ... more >> Zaklad Alergologii Dzieciecej Bialystok, Poland Specjalistyczne Centrum Leczenia Dzieci i Mlodziezy Krakow, Poland Wojskovy Szpital Klinikzny Krakow, Poland Priwatna Pomoc Lekarska Lodz, Poland Alergovita, alergologia Dziecieca Lublin, Poland Priwtny Gabinet Pediatriczno - Alergologiczny Rabka Zdroj, Poland Ukraine City Children's Clinical Hospital n. 2, Dniepropetrovs'k State Medical Academy, Department of preliminary study of childhood disesas Dniepropetrovsk, Ukraine Dniepropetrovsk Regional Children's Clinical Hospital, State Medical Academy, Hospital Paediatric Department N.1 Dniepropetrovsk, Ukraine Department of Hospital Pediatrics, Kharkiv State Medical University Region Clinical Children's Hospital n. 2 Kharkiv, Ukraine Respiratory Diseases Children's Clinic, Institute of Phthisiology and Pulmonology,Academy of Medical Science of the Ukraine, Pulmonology Kiev, Ukraine Children's Hospital "OHMATDYT" Department of Pediatrics N. 1 of Kyiv Medical Academy of Postgraduate Education Kyiv, Ukraine Department of Clinical rehabilitation of children with bodily diseasesof Institute of Pediatrics, Obstetrics and Gynecology Kyiv, Ukraine Institute of Pediatrics, Obstetrics and Gynecology. Department of Children's Pulmonology Diseases and Ecological Problems of Health Kyiv, Ukraine Department of Paediatric and Neonatology, Odessa State Medical University, Odessa Region Clinical Children's Hospital Odessa, Ukraine Poltava regional children's clinical hospital, Ukranian stomatological academy, department of pediatrics Poltava, Ukraine Pediatrics Crimean State Medical University Pulmonology, Department of Republican Clinical Children's Hospital Simferopol, Ukraine Children's clinical hospital N.1, Zaporizzhya Medical Academy of post graduate education, department of pediatrics Zaporizhya, Ukraine City Clinical Hospital N. 1 , Zaporizzhya Medical Academy of post graduate education, department of paediatrics Zaporizhya, Ukraine Regional Children Clinical Hospital. Department of Pulmonology. Zaporizhya, Ukraine Collapse << |
| NCT00496470 | Chronic Obstructive Pulmonary ... more >>Disease, COPD Collapse << | Phase 4 | Completed | - | - |
| NCT00503009 | Asthma | Phase 4 | Terminated(lack of data) | - | United States, Virginia ... more >> GSK Investigational Site Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00517634 | - | - | Completed | - | - |
| NCT00517634 | Asthma | Phase 4 | Completed | - | United States, Wisconsin ... more >> GSK Investigational Site Madison, Wisconsin, United States, 53792 United Kingdom GSK Investigational Site Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT00503009 | - | - | Terminated(lack of data) | - | - |
| NCT00530842 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00528723 | Bronchial Asthma | Phase 3 | Completed | - | Italy ... more >> Servizio di Fisiopatologia Respiratoria Diagnostica Allergologica e Biologia Respiratoria Endoscopica e Toracica U.O.C. di Pneumologia Ospedale Orlandi Bussolengo, (vr), Italy, 37012 Unità Dipartimentale di Fisiopatologia Respiratoria -Dipartimento di Medicina Interna e Specialità Mediche (DI.MI.) - Ospedale S. Martino e Cliniche Universitarie Convenzionate Genova, Italy, 16129 Unità Operativa Complessa di Clinica fisiologica e Pneumologia - Dipartimento di Medicina Interna e Specialità Mediche (DI.MI.) - Ospedale S. Martino e Cliniche Universitarie Convenzionate Genova, Italy, 16129 Dipartimento di Medicina Ambientale e Sanità Pubblica-Sede di Medicina del Lavoro Servizio di Fisiopatologia Respiratoria Padova, Italy, 35127 Malattie dell'Apparato Respiratorio - Università degli Studi di Pisa Ospedale Cisanello Pisa, Italy, 56100 Russian Federation City Clinical Hospital № 61, Pulmonology Department Moscow, Russian Federation, 119048 Central Clinical Hospital №1 OAO"RZD",Pulmonology Department Moscow, Russian Federation, 125315 Clinical Hospital № 2, Pulmonology Department Yaroslavl, Russian Federation, 150010 Clinical Hospital № 8, Pulmonology Department Yaroslavl, Russian Federation, 150030 Regional Clinical Hospital, Department of Therapeutics of Post Graduate, Yaroslavl Medical Academy Yaroslavl, Russian Federation, 150062 Spain Hospital General Universitario Gregorio Marañón Madrid, Spain, 28007 Ukraine Department of Therapy of Kharkov Medical Academy of Postgraduate Education City Multifield Clinical Hospital n° 25 Kharkov, Ukraine, 61008 Pulmonological and Allergological Department of the Kharkov Regional Clinical Hospital Kharkov, Ukraine, 61022, Central Military Hospital of North Region. Pulmonological Department Kharkov, Ukraine, 61022 Department of propaedeutics of internal diseases #1. Kharkov State Medical University. Kharkov, Ukraine, 61022 Pulmonological Department # 2 City Clinical Hospital # 13 Kharkov, Ukraine, 61035 Pulmonological Department of the Institute of Therapy Ukrainian Academy of Medical Science Kharkov, Ukraine, 61035 Department of General Practice - Family Medecine Medical Academy of post-graduate education. City Clinical Hospital N° 17 Kharkov, Ukraine, 61037 Department of Diagnostic, Therapy and Clinical Pharmacology of Lung Disease of the Institute of Phtisiology and Pulmonology Academy of Medical Sciences of the Ukraine Kiev, Ukraine, 03680 Institute of Phthisiology and Pulmonology Acedemy of Medical Science of the Ukraine Kiev, Ukraine, 03680 Pulmonology Department of the Institute of Phthisiology and Pulmonology AMS of the Ukraine Kiev, Ukraine, 03680 Department of Pediatrics and laboratory diagnostics, Dniepropetrovsk State Medical Academy City Clinical Hospital No. 8. Pulmonology Department. 55 Krivoi Rog, Ukraine, 50082 General Therapy Clinic Central Military Clinical Hospital of Ministry of Defenses of Ukraine Kyiv, Ukraine, 01133 Department of Hospital Therapy of Lugansk State Medical Institute Lugansk, Ukraine, 91045 Department of Hospital Therapy, Lviv State Medical University named by Danylo Galytsky, Lviv Regional Clinical Hospital Lviv, Ukraine, 79010 Collapse << |
| NCT00530842 | - | - | Completed | - | - |
| NCT00542880 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Completed | - | - |
| NCT00535366 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Belgium ... more >> 1249.1.32003 Boehringer Ingelheim Investigational Site Genk, Belgium 1249.1.32001 Boehringer Ingelheim Investigational Site Gent, Belgium 1249.1.32002 Boehringer Ingelheim Investigational Site Hasselt, Belgium 1249.1.32004 Boehringer Ingelheim Investigational Site Oostende, Belgium Denmark 1249.1.45001 Boehringer Ingelheim Investigational Site Aarhus C, Denmark Germany 1249.1.49001 Boehringer Ingelheim Investigational Site Großhansdorf, Germany 1249.1.49002 Boehringer Ingelheim Investigational Site Mannheim, Germany 1249.1.49003 Boehringer Ingelheim Investigational Site Weinheim, Germany Netherlands 1249.1.31002 Boehringer Ingelheim Investigational Site Eindhoven, Netherlands 1249.1.31003 Boehringer Ingelheim Investigational Site Harderwijk, Netherlands 1249.1.31001 Boehringer Ingelheim Investigational Site Heerlen, Netherlands 1249.1.31004 Boehringer Ingelheim Investigational Site Veldhoven, Netherlands Collapse << |
| NCT00542880 | - | - | Completed | - | - |
| NCT00549146 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | Netherlands ... more >> GSK Investigational Site Alkmaar, Netherlands, 1815 JD GSK Investigational Site Almelo, Netherlands, 7609 PP GSK Investigational Site Amsterdam, Netherlands, 1034 CS GSK Investigational Site Amsterdam, Netherlands, 1058 NR GSK Investigational Site Amsterdam, Netherlands, 1091 AC GSK Investigational Site Drachten, Netherlands, 9202 NN GSK Investigational Site Haarlem, Netherlands, 2012 CE GSK Investigational Site Harderwijk, Netherlands, 3844 DG GSK Investigational Site Hengelo, Netherlands, 7555 DL GSK Investigational Site Hoorn, Netherlands, 1624 NP GSK Investigational Site Leeuwarden, Netherlands, 8934 AD GSK Investigational Site Meppel, Netherlands, 7943 KA GSK Investigational Site Nijmegen, Netherlands, 6532 SZ GSK Investigational Site Rotterdam, Netherlands, 3045 PM GSK Investigational Site Sneek, Netherlands, 8601 ZK GSK Investigational Site Utrecht, Netherlands, 2584 CX GSK Investigational Site Voerendaal, Netherlands, 6367 ED Collapse << |
| NCT00556673 | Asthma | Phase 2 | Completed | - | France ... more >> Novartis Investigator Site Poitiers, France, 86000 Germany Novartis Investigator Site Berlin, Germany, 14050 Collapse << |
| NCT00557440 | Asthma | Phase 2 | Completed | - | Belgium ... more >> Novartis Investigator Site Aalst, Belgium Novartis Investigator site Ghent, Belgium Germany Novartis Investigator Site Berlin, Germany Novartis Investigator Site Hannover, Germany Novartis Investigator Site Landsberg, Germany Novartis Investigator Site Rostock, Germany Collapse << |
| NCT00536913 | - | - | Completed | - | - |
| NCT00536913 | Asthma | Phase 3 | Completed | - | Hungary ... more >> Research Site Budapest, Hungary Research Site Debrecen, Hungary Research Site Kaposvar, Hungary Poland Research Site Bialystok, Poland Research Site Bydgoszcz, Poland Research Site Bytom, Poland Research Site Karpacz, Poland Research Site Krakow, Poland Research Site Lodz, Poland Research Site Warszawa, Poland Russian Federation Research Site Moscow, Russian Federation Collapse << |
| NCT00565266 | Asthma | Phase 3 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92093 University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado National Jewish Medical and Research Center Denver, Colorado, United States, 80206 United States, Massachusetts Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 United States, Missouri Washington University, St. Louis Saint Louis, Missouri, United States, 63130 United States, New York Columbia University Health Sciences New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555 United States, Wisconsin University of Wisconsin, Madison Madison, Wisconsin, United States, 53706 Collapse << |
| NCT00565266 | - | - | Completed | - | - |
| NCT00568347 | - | - | Completed | - | United States, California ... more >> Arthur F Gelb MD Lakewood, California, United States, 90712 Collapse << |
| NCT00559312 | COPD | Not Applicable | Completed | - | Canada, Ontario ... more >> Respiratory Investigation Unit at Kingston General Hospital Kingston, Ontario, Canada, K7L 2V7 Collapse << |
| NCT00556673 | - | - | Completed | - | - |
| NCT00569712 | Lung Cancer P... more >>recancerous Condition Collapse << | Phase 1 | Completed | - | Canada, British Columbia ... more >> British Columbia Cancer Agency - Vancouver Cancer Centre Vancouver, British Columbia, Canada, V5Z 4E6 Collapse << |
| NCT00557440 | - | - | Completed | - | - |
| NCT00633217 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Jasper, Alabama, United States, 35501 GSK Investigational Site Mobile, Alabama, United States, 36608 United States, Louisiana GSK Investigational Site Lafayette, Louisiana, United States, 70503 GSK Investigational Site New Orleans, Louisiana, United States, 70115 GSK Investigational Site Sunset, Louisiana, United States, 70584 United States, Missouri GSK Investigational Site St. Charles, Missouri, United States, 63301 United States, North Carolina GSK Investigational Site Elizabeth City, North Carolina, United States, 27909 United States, Pennsylvania GSK Investigational Site Erie, Pennsylvania, United States, 16508 United States, South Carolina GSK Investigational Site Charleston, South Carolina, United States, 29406-7108 GSK Investigational Site Gaffney, South Carolina, United States, 29340 GSK Investigational Site Greenville, South Carolina, United States, 29615 GSK Investigational Site Spartanburg, South Carolina, United States, 29303 GSK Investigational Site Union, South Carolina, United States, 29309 United States, Texas GSK Investigational Site Corsicana, Texas, United States, 75110 United States, Virginia GSK Investigational Site Richmond, Virginia, United States, 23229 United States, West Virginia GSK Investigational Site Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT00646009 | Asthma | Phase 3 | Completed | - | - |
| NCT00633217 | - | - | Completed | - | - |
| NCT00646321 | Asthma | Phase 3 | Completed | - | - |
| NCT00646529 | Asthma | Phase 3 | Completed | - | - |
| NCT00646516 | Mild or Moderate Asthma | Phase 3 | Completed | - | - |
| NCT00646620 | Asthma | Phase 3 | Completed | - | - |
| NCT00651651 | Asthma | Phase 3 | Completed | - | - |
| NCT00646594 | Asthma | Phase 3 | Completed | - | - |
| NCT00652392 | Asthma | Phase 3 | Completed | - | - |
| NCT00652002 | Asthma | Phase 3 | Completed | - | - |
| NCT00658255 | Stable Asthma | Phase 2 | Completed | - | - |
| NCT00624754 | Obstructive Airway Disease | Phase 2 | Completed | - | France ... more >> Hôpital Saint Louis Paris, France, 75010 Collapse << |
| NCT00662805 | - | - | Completed | - | - |
| NCT00490243 | Asthma | Phase 4 | Completed | - | Poland ... more >> Department of Pediatrics and Allergy, Medical University of Lodz, Poland Lodz, Poland, 93-513 Collapse << |
| NCT00741767 | Chronic Obstructive Pulmonary ... more >>Disease Sleep Disorders Collapse << | Not Applicable | Withdrawn(Study subject enroll... more >>ment difficulties) Collapse << | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00702325 | Asthma | Phase 4 | Completed | - | - |
| NCT00651768 | Asthma | Phase 3 | Completed | - | - |
| NCT00784550 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00782314 | - | - | Completed | - | Slovenia ... more >> Research Site Celje, Slovenia Research Site Domzale, Slovenia Research Site Dvorec Sela, Slovenia Research Site Grosuplje, Slovenia Research Site Idrija, Slovenia Research Site Izola, Slovenia Research Site Jesenice, Slovenia Research Site Kamnik-Duplica, Slovenia Research Site Kranj, Slovenia Research Site Litija, Slovenia Research Site Ljubljana, Slovenia Research Site Lucija, Slovenia Research Site Maribor, Slovenia Research Site Murska Sobota, Slovenia Research Site Novo mesto, Slovenia Research Site Ptuja, Slovenia Research Site Ravne na Koroskem, Slovenia Research Site Sezana, Slovenia Research Site Slovenj Gradec, Slovenia Research Site Slovenska Bistrica, Slovenia Research Site Topolsica, Slovenia Research Site Velenje, Slovenia Research Site Vrhnika, Slovenia Research Site Zagorje, Slovenia Collapse << |
| NCT00651547 | Asthma | Phase 3 | Completed | - | - |
| NCT00800241 | - | - | Completed | - | Thailand ... more >> Research Site Buriram, Thailand Research Site Chanthaburi, Thailand Research Site Chiang Mai, Thailand Research Site Mahasarakham, Thailand Research Site Nakhon Phanom, Thailand Research Site Nakhon Srithammarat, Thailand Research Site Nakhonpathom, Thailand Research Site Phattalung, Thailand Research Site Phitsanulok, Thailand Research Site Rayong, Thailand Research Site Roi Et, Thailand Research Site Samutsakhon, Thailand Research Site Surat Thani, Thailand Research Site Trang, Thailand Collapse << |
| NCT00702325 | - | - | Completed | - | - |
| NCT00830505 | Asthma | Phase 4 | Unknown | December 2009 | United Kingdom ... more >> Asthma and Allergy Research Group Dundee, Angus, United Kingdom, DD1 9SY Collapse << |
| NCT00837629 | - | - | Completed | - | - |
| NCT00850265 | Asthma | Not Applicable | Completed | - | Spain ... more >> Hospital General de Requena Requena-Valencia, Valencia, Spain, 46340 Collapse << |
| NCT00862394 | Asthma | Phase 3 | Completed | - | Germany ... more >> Oliver Kornmann - National Coordinator in Germany Mainz, Germany, 55131 Collapse << |
| NCT00861926 | Asthma | Phase 3 | Completed | - | Italy ... more >> Pr Papi Ferrara, Italy Collapse << |
| NCT00784550 | - | - | Completed | - | - |
| NCT00868426 | Healthy | Phase 1 | Completed | - | Finland ... more >> Orion Pharma Phase I Unit Espoo, Finland, 02101 Collapse << |
| NCT00864812 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Soonchunhyang University Bucheon Hospital Bucheon, Korea, Republic of Inje university Pusan Paik hospital Busan, Korea, Republic of Chonbuk national university hospital Chunbuk, Korea, Republic of Chungbuk national university hospital Chungbuk, Korea, Republic of Chungnam National University Hospital Chungnam, Korea, Republic of Keimyung university dongsan medical center Daegu, Korea, Republic of Kyungpook national university hospital Daegu, Korea, Republic of Yeungnam University Hospital Daegu, Korea, Republic of Hallym University scared heart hospital Gyeonggi-do, Korea, Republic of Gachon University Gil Hospital Inchon, Korea, Republic of Incheon St. Mary's Hospital Inchon, Korea, Republic of Inha university Hospital Inchon, Korea, Republic of Gyeongsang national university hospital Jinju, Korea, Republic of Kyunghee university east-west neo medical center Seoul, Korea, Republic of, 134-727 Asan Medical Center Seoul, Korea, Republic of Boramae Medical Center Seoul, Korea, Republic of Ewha womans university mokdong hospital Seoul, Korea, Republic of Hanyang University Hospital Seoul, Korea, Republic of Inje university Seoul Paik Hospital Seoul, Korea, Republic of Kangdong Scared heart Hospital Seoul, Korea, Republic of Kangnam St. Mary's Hospital Seoul, Korea, Republic of Konkuk university hospital Seoul, Korea, Republic of Korea University Guro Hospital Seoul, Korea, Republic of Kyunghee university medical center Seoul, Korea, Republic of Samsung medical center Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Severance Hospital Seoul, Korea, Republic of Soonchunhyang University hospital Seoul, Korea, Republic of St. Paul's Hospital Seoul, Korea, Republic of Ajou university hospital Suwon, Korea, Republic of Uijeongbu St. Mary's Hospital Uijeongbu, Korea, Republic of Wonju Christian Hospital Wonju, Korea, Republic of Collapse << |
| NCT00908362 | Smoke-related Lung Diseases ... more >> Chronic Obstructive Pulmonary Disease Collapse << | Phase 1 | Completed | - | Germany ... more >> Dep. of Pneumology, University of Rostock Rostock, Mecklenburg-Vorpommern, Germany, 18057 Collapse << |
| NCT00868023 | Asthma | Phase 2 | Completed | - | United Kingdom ... more >> Pr DS SINGH Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT00829257 | Asthma | Phase 4 | Completed | - | United Kingdom ... more >> Asthma and Allergy Research Group, Ninewells Hospital and Medical School Dundee, Scotland, United Kingdom, DD1 9SY Collapse << |
| NCT00920959 | Asthma | Phase 3 | Completed | - | - |
| NCT00927758 | Asthma | Phase 2 | Completed | - | United States, Florida ... more >> University of Florida Gainesville, Florida, United States, 32610-0486 United States, Iowa CompleWare Iowa City, Iowa, United States, 52244 United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00927758 | - | - | Completed | - | - |
| NCT00901368 | Asthmatic Patients | Phase 4 | Completed | - | France ... more >> Hôpital Nord Marseille, France, 13015 Germany Allergologie imUmkreis der Praxis Pneumologie Gelsenkirchen, Nordrhein-Westfalen, Germany, 45879 Netherlands Atrium Medisch Centrum Heerlen, Heerlen, Netherlands, 6419 PC Spain Hospital Universitario La Fe Valencia, Spain, 46009 Collapse << |
| NCT00419744 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00939341 | Asthma | Phase 4 | Completed | - | - |
| NCT00920543 | Asthma | Phase 4 | Completed | - | - |
| NCT00910793 | Asthma | Phase 4 | Completed | - | Belgium ... more >> University Hospital Antwerp Antwerp, Belgium, 2650 Collapse << |
| NCT00419744 | - | - | Completed | - | - |
| NCT00929851 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | United Kingdom ... more >> UCL Medical School London, United Kingdom Collapse << |
| NCT00984659 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00964535 | Asthma | Phase 1 Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT00939341 | - | - | Completed | - | - |
| NCT00996697 | COPD | Phase 4 | Completed | - | United Kingdom ... more >> Asthma and Allergy Research Group, Ninewells Hospital and Medical School Dundee, Tayside, United Kingdom, DD1 9SY Collapse << |
| NCT00989833 | - | - | Completed | - | - |
| NCT00984659 | - | - | Completed | - | - |
| NCT00441441 | Asthma | Phase 3 | Completed | - | - |
| NCT00989833 | Exercise Induced Asthma | Phase 2 | Completed | - | Norway ... more >> Research Site Oslo, Norway Research Site Trondheim, Norway Sweden Research Site Goteborg, Sweden Research Site Huddinge, Sweden Research Site Linkoping, Sweden Research Site Lulea, Sweden Research Site Lund, Sweden Research Site Skene, Sweden Research Site Stockholm, Sweden Research Site Uppsala, Sweden Collapse << |
| NCT00997477 | Asthma | Phase 4 | Withdrawn | - | Turkey ... more >> Novartis Investigator Site Ankara, Turkey Novarits Investigator Site Istanbul, Turkey Novartis Investigator Site Istanbul, Turkey Novartis Investigator Site Izmir, Turkey Collapse << |
| NCT01023516 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | - |
| NCT00441441 | - | - | Completed | - | - |
| NCT01001364 | ASTHMA | Phase 3 | Completed | - | Brazil ... more >> Centro de Pesquisa Clínica Stelmach São Paulo, SP, Brazil, 05437 010 Collapse << |
| NCT01065272 | Bronchiolitis | Phase 1 | Completed | - | Qatar ... more >> Hamad Medical Corporation, Pediatric Emergency Center,Alsaad. Doha, Qatar, 3050 Collapse << |
| NCT01023516 | - | - | Completed | - | - |
| NCT01069289 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01069289 | - | - | Completed | - | - |
| NCT01070784 | - | - | Completed | - | - |
| NCT00476268 | Asthma | Phase 3 | Completed | - | - |
| NCT01070888 | Exercise Induced Asthma | Phase 4 | Terminated(Unable to reach tar... more >>get goal) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Children's Hospital Boston Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00314509 | Asthma | Phase 2 | Completed | - | France ... more >> Altana Pharma/Nycomed Grenoble, France, 38100 Altana Pharma/Nycomed Montpellier, France, 34295 Germany Altana Pharma/Nycomed Berlin, Germany, 14050 Altana Pharma/Nycomed Großhansdorf, Germany, 22927 Altana Pharma/Nycomed Mainz, Germany, 55131 Altana Pharma/Nycomed Rüdersdorf, Germany, 15562 Altana Pharma/Nycomed Wiesbaden, Germany, 65187 Hungary Altana Pharma/Nycomed Balassagyarmat, Hungary, 2660 Altana Pharma/Nycomed Balatonfüred, Hungary, 8230 Altana Pharma/Nycomed Budapest, Hungary, 1125 Altana Pharma/Nycomed Budapest, Hungary, 1529 Altana Pharma/Nycomed Csorna, Hungary, 9300 Altana Pharma/Nycomed Debrecen, Hungary, 4043 Altana Pharma/Nycomed Györ, Hungary, 9024 Altana Pharma/Nycomed Matrahaza, Hungary, 3233 Altana Pharma/Nycomed Mosonmagyaróvár, Hungary, 9200 Altana Pharma/Nycomed Nyíregyháza, Hungary, 4400 Altana Pharma/Nycomed Szolnok, Hungary, 5006 Altana Pharma/Nycomed Szombathely, Hungary, 9700 South Africa Altana Pharma/Nycomed Arcadia, Pretoria, South Africa, 132 Altana Pharma/Nycomed Bellville, Cape Town, South Africa, 7530 Altana Pharma/Nycomed Bloemfontein, South Africa, 9300 Altana Pharma/Nycomed Cape Town, Tygerberg, South Africa, 7505 Altana Pharma/Nycomed Mowbray, Cape Town, South Africa, 7925 Altana Pharma/Nycomed Panorama / RSA-Cape Town, South Africa, 7500 Altana Pharma/Nycomed Pretoria, South Africa Collapse << |
| NCT01117116 | Asthma | Not Applicable | Completed | - | United States, Colorado ... more >> National Jewish Health Denver, Colorado, United States, 80206 Collapse << |
| NCT01136655 | Asthma | Phase 2 | Completed | - | - |
| NCT01070784 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Japan ... more >> Research Site Nagoya, Aichi, Japan Research Site Toyota, Aichi, Japan Research Site Yanagawa, Fukuoka, Japan Research Site Asahikawa, Hokkaido, Japan Research Site Sapporo, Hokkaido, Japan Research Site Itami, Hyogo, Japan Research Site Hitachi, Ibaraki, Japan Research Site Tsukuba, Ibaraki, Japan Research Site Sakaide, Kagawa, Japan Research Site Fujisawa, Kanagawa, Japan Research Site Yokohama, Kanagawa, Japan Research Site Koshi, Kumamoto, Japan Research Site Shibata, Miyagi, Japan Research Site Chuo, Tokyo, Japan Research Site Setagaya, Tokyo, Japan Research Site Hiroshima, Japan Research Site Kyoto, Japan Collapse << |
| NCT01167010 | Asthma | Phase 3 | Completed | - | Brazil ... more >> Centro de Referencia em Enfermidades Respiratoria e Alergica Salvador, BA, Brazil Hospital de Clinicas UFPR Curitiba, PR, Brazil Clinica de Alergia Martti Antila Sorocaba, SP, Brazil IMA Brasil - Instituto de Pesquisa Clínica e Medicina Avançada São Paulo, SP, Brazil Stelmach Pesquisa Clinica São Paulo, SP, Brazil Collapse << |
| NCT01136655 | - | - | Completed | - | - |
| NCT01070888 | - | - | Terminated(Unable to reach tar... more >>get goal) Collapse << | - | - |
| NCT01147848 | Asthma | Phase 3 | Completed | - | - |
| NCT01179152 | Asthma Exacerbation | Not Applicable | Unknown | October 2012 | Israel ... more >> Meir Medical Center Recruiting Kfar Saba, Israel Contact: Avner He Cohen, MD Collapse << |
| NCT01186653 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Respiratory Medicine, Castgle Hill Hospital Cottingham, East Yorkshire, United Kingdom, HU16 5JQ Collapse << |
| NCT01181063 | Asthma | Phase 1 | Completed | - | Finland ... more >> Phase I Unit, Orion Pharma Espoo, Finland Collapse << |
| NCT01202097 | Asthma | Phase 3 | Completed | - | Brazil ... more >> Hospital de Clinicas UFPR Curitiba, PR, Brazil Centro de Referencia em Enfermidades Respiratoria e Alergica Salvador, BA, Brazil Clinica de Alergia Martti Antila Sorocaba, SP, Brazil Centro de Estudos de Pneumologia FMABC São Paulo, Brazil IMA Brasil - Instituto de Pesquisa Clínica e Medicina Avançada São Paulo, Brazil Stelmach Pesquisa Clinica São Paulo, Brazil Collapse << |
| NCT01204034 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Belgium ... more >> University Hospital of Antwerp Edegem (Antwerp), Antwerp, Belgium, B-2650 Collapse << |
| NCT01147848 | - | - | Completed | - | - |
| NCT01245569 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Italy ... more >> Policlinico Umberto I - VIII Padiglione Rome, Italy, 00161 Collapse << |
| NCT01257048 | COPD Method Evaluation ... more >> Chronic Obstructive Pulmonary Disease Method Evaluation Collapse << | Early Phase 1 | Completed | - | Sweden ... more >> Research Site Lund, Sweden United Kingdom Research Site Manchester, United Kingdom Collapse << |
| NCT01006655 | Asthma Bronch... more >>ial Hyperresponsiveness Collapse << | Phase 2 | Completed | - | Israel ... more >> Rambam Medical Center Haifa, Israel Collapse << |
| NCT01191424 | Asthma | Phase 2 | Completed | - | Denmark ... more >> Copenhagen University Hospital Copenhagen, Denmark, 2800 United Kingdom Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT01253473 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham Medical Center Birmingham, Alabama, United States, 35294 United States, California Harbor UCLA Medical Center Torrance, California, United States, 90502 United States, Colorado National Jewish Health Denver, Colorado, United States, 80206 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 United States, Massachusetts Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Reliant Medical Group Worcester, Massachusetts, United States, 01608 United States, Pennsylvania Temple University Medical Center Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT01327001 | - | - | Completed | - | - |
| NCT01332344 | - | - | Completed | - | - |
| NCT01315249 | - | - | Completed | - | - |
| NCT01006655 | - | - | Completed | - | - |
| NCT01329276 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | Belgium ... more >> University Hospital Antwerp Edegem (Antwerp), Antwerp, Belgium, 2650 Collapse << |
| NCT01332357 | - | - | Completed | - | - |
| NCT01315249 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT01332357 | - | - | Completed | - | - |
| NCT01332409 | - | - | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 296-8602 Collapse << |
| NCT01772368 | Asthma | Phase 2 | Completed | - | United States, Colorado ... more >> Teva Investigational Site 10453 Denver, Colorado, United States United States, Massachusetts Teva Investigational Site 10452 North Dartmouth, Massachusetts, United States United States, Missouri Teva Investigational Site 10455 Saint Louis, Missouri, United States United States, New Jersey Teva Investigational Site 10454 Skillman, New Jersey, United States United States, North Carolina Teva Investigational Site 10448 Raleigh, North Carolina, United States United States, Oregon Teva Investigational Site 10451 Medford, Oregon, United States Teva Investigational Site 10449 Portland, Oregon, United States United States, Texas Teva Investigational Site 10457 El Paso, Texas, United States Teva Investigational Site 10450 New Braunfels, Texas, United States Teva Investigational Site 10456 San Antonio, Texas, United States Collapse << |
| NCT02526758 | Pulmonary Disease,Chronic Obst... more >>ructive; Collapse << | Phase 4 | Unknown | May 2016 | China, Guangdong ... more >> Zhujiang Hospital Recruiting Guangzhou, Guangdong, China, 510280 Contact: Shuhan Wu, Master 13268268627 664397041@qq.com Collapse << |
| NCT01772368 | - | - | Completed | - | - |
| NCT02924688 | Asthma | Phase 3 | Active, not recruiting | February 21, 2019 | - |
| NCT02499887 | Asthma COPD | Not Applicable | Unknown | September 2016 | United States, New York ... more >> Erie County Medical Center Recruiting Buffalo, New York, United States, 14215 Contact: Heather Lindstrom, PhD Collapse << |
| NCT02556073 | Asthma | Not Applicable | Unknown | December 2016 | Taiwan ... more >> Taipei Veterans General Hospital Taipei City, Taiwan, 886 Collapse << |
| NCT02373137 | Keratoplasty ... more >>Grafting, Corneal Transplantation, Corneal Transplantation, Cornea Keratoplasty, Lamellar Collapse << | Phase 4 | Active, not recruiting | December 2019 | United States, California ... more >> Byers Eye Institute, Stanford University Palo Alto, California, United States, 94303 F. I. Proctor Foundation, University of California, San Francisco San Francisco, California, United States, 94143 United States, Oregon Casey Eye Institute, Oregon Health & Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT02175771 | - | - | Completed | - | - |
| NCT00252785 | Asthma | Phase 3 | Completed | - | Japan ... more >> Research Site Komaki, Aichi, Japan Research Site Seto, Aichi, Japan Research Site Asahi, Chiba, Japan Research Site Noda, Chiba, Japan Research Site Touon, Ehime, Japan Research Site Mizumaki, Fukuoka, Japan Research Site Isesaki, Gunma, Japan Research Site Maebashi, Gunma, Japan Research Site Ora, Gunma, Japan Research Site Ota, Gunma, Japan Research Site Chitose, Hokkaido, Japan Research Site Kitahiroshima, Hokkaido, Japan Research Site Obihiro, Hokkaido, Japan Research Site Sapporo, Hokkaido, Japan Research Site Tomakomai, Hokkaido, Japan Research Site Morioka, Iwate, Japan Research Site Takamatsu, Kagawa, Japan Research Site Sendai, Miyagi, Japan Research Site Beppu, Ohita, Japan Research Site Tsukubo, Okayama, Japan Research Site Kishiwada, Osaka, Japan Research Site Oskasayama, Osaka, Japan Research Site Takatsuiki, Osaka, Japan Research Site Koshigaya, Saitama, Japan Research Site Minamisaitama, Saitama, Japan Research Site Arakawa, Tokyo, Japan Research Site Chiyoda, Tokyo, Japan Research Site Itabashi, Tokyo, Japan Research Site Kodaira, Tokyo, Japan Research Site Nakano-ku, Tokyo, Japan Research Site Ota-ku, Tokyo, Japan Research Site Shinagawa-ku, Tokyo, Japan Research Site Sumida, Tokyo, Japan Research Site Ube, Yamaguchi, Japan Research Site Gifu, Japan Research Site Hiroshima, Japan Research Site Kagoshima, Japan Research Site Kyoto, Japan Research Site Oita, Japan Research Site Okayama, Japan Research Site Osaka, Japan Research Site Tochigi, Japan Research Site Toyama, Japan Collapse << |
| NCT00252863 | Asthma | Phase 3 | Completed | - | - |
| NCT03219866 | COPD COPD Exa... more >>cerbation Collapse << | Phase 4 | Recruiting | September 2019 | United States, North Carolina ... more >> Wake Forest Baptist Health Recruiting Winston-Salem, North Carolina, United States, 27104 Contact: Jill A Ohar, MD, FCCP 336-716-1210 johar@wakehealth.edu Collapse << |
| NCT00403286 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | - |
| NCT00411372 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Withdrawn(No subjects enrolled... more >>. Study was canceled before active) Collapse << | - | - |
| NCT00413387 | Bronchial Asthma | Phase 3 | Completed | - | Austria ... more >> Ambulance For Paediatric and Pulmonology Wien, Austria Poland Nzoz "Medex"Poradnia Alergologiczna Bielsko-Biala, Poland Centrum Uslug Medycznych Krakow, Poland Centrum Alergologii Lodz, Poland Prywatny Gabinet Lekarski Lodz, Poland Uniwersytet Medyczny Lodz, Poland Nzoz Lekarze Specjalisci Wroclaw, Poland Ukraine Internal Medicine Department, Dniepropetrovsk State Medical Academy. City Clinical Hospital no. 4 Dniepropetrovsk, Ukraine Institute of Therapy, Ukranian Academy of Medical Science. Pulmonological Departement Kharkiv, Ukraine Kharkov Regional Clinical Hospital. Pulmonological and Allergological Department Kharkov, Ukraine Institute of Phthisiology and Pulmonology Academy of Medical Science of the Ukraine, Pulmonology Departement Kiev, Ukraine Institute of Phthisiology and Pulmonology Academy of Medical Science of the Ukraine. Department of Diagnostic, Therapy and Clinical Pharmacology of Lung Diseases Kiev, Ukraine Kiev Medical Academy of Postdiploma Education. Department of Medical Genetics, Clinical Immunology and Allergology Kiev, Ukraine Collapse << |
| NCT00419757 | Asthma | Phase 3 | Completed | - | - |
| NCT02175771 | Persistent Asthma | Phase 3 | Completed | - | - |
| NCT00448435 | Bronchial Asthma | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 260-0001 GSK Investigational Site Kanagawa, Japan, 245-0018 GSK Investigational Site Saitama, Japan, 360-0018 GSK Investigational Site Saitama, Japan, 360-0812 GSK Investigational Site Tokyo, Japan, 154-0002 GSK Investigational Site Tokyo, Japan, 154-0017 GSK Investigational Site Tokyo, Japan, 158-0083 GSK Investigational Site Tokyo, Japan, 158-0097 Collapse << |
| NCT00421122 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | China, Beijing ... more >> Research Site Beijing, Beijing, China China, Guangdong Research Site Guangzhou, Guangdong, China China, Jiangsu Research Site Nanjing, Jiangsu, China China, Liaoling Research Site Shenyang, Liaoling, China China, Shanghai Research Site Shanghai, Shanghai, China China, Shanxi Research Site Xi'an, Shanxi, China China, Sichuan Research Site Chengdu, Sichuan, China China, Zhejiang Research Site Hangzhou, Zhejiang, China Collapse << |
| NCT00419757 | - | - | Completed | - | - |
| NCT01110200 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT02584738 | Asthma | Phase 4 | Unknown | January 2018 | Mexico ... more >> Hospital General Naval de Alta Especialidad Recruiting México, Distrito Federal, Mexico, 04480 Contact: Jesús Abisai Uicab Saucedo, Pediatrician (52)5550371200 ext 5423 abisaipec@msn.com Collapse << |
| NCT01110200 | - | - | Completed | - | - |
| NCT02972476 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Recruiting | April 2020 | United Kingdom ... more >> NHS Tayside Recruiting Dundee, Tayside, United Kingdom, DD1 9SY Contact: Philip Short, MBChB MRCP 01382660111 p.short@dundee.ac.uk Contact: Gayle Scott, BSc 01382383533 g.x.scott@dundee.ac.uk Blackpool Teaching Hospital NHS Foundation Trust Recruiting Blackpool, United Kingdom, FY3 8NR Contact: Mohammad Paracha, MBBS, MRCP, FRCP 01253 951444 Dr.Paracha@bfwhospitals.nhs.uk NHS Lothian Recruiting Edinburgh, United Kingdom, EH16 4TJ Contact: Gourab Choudhury, MBBS, MRCP, SCE 01312421869 gourab.choudhury@nhslothian.scot.nhs.uk NHS Greater Glasgow and Clyde Recruiting Glasgow, United Kingdom Contact: Rekha Chaudhuri, MD, MBBS 0141 211 0095 Rekha.chaudhuri@ggc.scot.nhs.uk NHS Fife Recruiting Kirkcaldy, United Kingdom, KY2 5AH Contact: Devesh Dhasmana, MBBS, MRCP, PhD 01592 643355 devesh.dhasmana@nhs.net NHS Lanarkshire Recruiting Wishaw, United Kingdom, ML2 0DP Contact: Manish Patel, MBChB, FRCP, PhD 07876786475 manishpatel2@nhs.net Collapse << |
| NCT02528214 | - | - | Completed | - | - |
| NCT00448435 | - | - | Completed | - | - |
| NCT00553059 | Chemotherapy-induced Nausea an... more >>d Vomiting Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 3 | Completed | - | United States, Missouri ... more >> Cancer Research for the Ozarks Springfield, Missouri, United States, 65807 United States, South Carolina CCOP - Greenville Greenville, South Carolina, United States, 29615 United States, Texas University of Texas M.D. Anderson CCOP Research Base Houston, Texas, United States, 77030-4009 United States, Vermont Vermont Cancer Center at University of Vermont Burlington, Vermont, United States, 05405 Collapse << |
| NCT01615874 | Asthma | Phase 2 | Withdrawn | September 2013 | - |
| NCT02528214 | Asthma | Phase 3 | Completed | - | - |
| NCT00404261 | Asthma | Phase 4 | Completed | - | Australia, Queensland ... more >> GSK Investigational Site Auchenflower, Queensland, Australia, 4066 GSK Investigational Site Caboolture, Queensland, Australia, 4510 GSK Investigational Site Kippa Ring, Queensland, Australia, 4021 Australia, Western Australia GSK Investigational Site Perth, Western Australia, Australia, 6000 Collapse << |
| NCT03270033 | Pain, Postoperative ... more >> Shoulder Joint Disorder Ambulatory Surgical Procedures Brachial Plexus Block Collapse << | Phase 4 | Active, not recruiting | October 2018 | Canada, Manitoba ... more >> Pan Am Surgical Centre Winnipeg, Manitoba, Canada, R3M 3E4 Collapse << |
| NCT01783821 | - | - | Completed | - | - |
| NCT01787097 | Chronic Obstructive Lung Disea... more >>se Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Royal Brompton and Harefield NHS trust London, United Kingdom, SW3 6NP Collapse << |
| NCT01656005 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Asthma and Allergy Research Group, Ninewells Hospital and Medical School, University of Dundee Dundee, United Kingdom, DD1 9SY Collapse << |
| NCT02757209 | Asthma COPD | Not Applicable | Completed | - | Italy ... more >> 3) Fondazione S. Maugeri - IRCCS - Dipartimento di Pneumologia Riabilitativa Cassano delle Murge, Bari, Italy, 70020 University Hospital S Anna Ferrara, Fe, Italy, 44124 4) Clinica di Malattie dall'Apparato Respiratorio Fondazione Salvatore Maugeri Tradate, Varese, Italy, 21049 Collapse << |
| NCT02676089 | Asthma | Phase 3 | Completed | - | - |
| NCT02676076 | Asthma | Phase 3 | Completed | - | Germany ... more >> Chiesi Clinical Trial Site 276814 Rostock, Germany Collapse << |
| NCT01712854 | Chronic Obstructive Lung Disea... more >>se Collapse << | Phase 4 | Terminated(Closed by study spo... more >>nsor; PI left the institution; responsible party changed to Columbia) Collapse << | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT01312961 | Asthma | Phase 2 | Completed | - | - |
| NCT01712854 | - | - | Terminated(Closed by study spo... more >>nsor; PI left the institution; responsible party changed to Columbia) Collapse << | - | - |
| NCT01471340 | Asthma | Phase 4 | Completed | - | - |
| NCT03387852 | Asthma | Phase 2 | Recruiting | August 27, 2019 | - |
| NCT01783821 | Acute Respiratory Distress Syn... more >>drome (ARDS) Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> University of Arizona Tucson, Arizona, United States, 85721 United States, California Stanford University Stanford, California, United States, 94305 United States, Florida Mayo Clinic in Florida Jacksonville, Florida, United States, 32224 United States, Massachusetts Beth Israel Medical Center Boston, Massachusetts, United States, 02215 United States, Minnesota Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01471340 | - | - | Completed | - | - |
| NCT01782326 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT03627858 | - | - | Recruiting | June 2020 | Belgium ... more >> ZNA Middelheim Recruiting Antwerp, Belgium, 2020 Contact: Lieven Bedert, MD lieven.bedert@zna.be Universitair Ziekenhuis Antwerpen Not yet recruiting Antwerp, Belgium, 2650 Contact: Veronique Verplancke, MD GZA Campus Sint-Vincentius Recruiting Antwerp, Belgium Contact: Inge Hantson Hôpital Erasme Recruiting Brussels, Belgium Contact: Dimitri Leduc UZ Brussel Recruiting Brussel, Belgium Contact: Daniel Schuermans Grand Hôpital de Charleroi Recruiting Charleroi, Belgium, 6000 Contact: Pierre Fievez, MD Clinique Notre-Dame de Grâce ASBL Gosselies Recruiting Charleroi, Belgium Contact: Véronique Goblet UZ Gent Recruiting Gent, Belgium Contact: Stefanie Vermeersch AZ Groeninge Recruiting Kortrijk, Belgium Contact: Fem De Plus CHR de la Citadelle Recruiting Liège, Belgium Contact: Geoffroy De Fooz AZ Sint-Maarten Recruiting Mechelen, Belgium Contact: Muriel Lins Hôpital André Vésale Recruiting Montigny-le-Tilleul, Belgium Contact: Rudi Peché, MD Clinique Saint-Luc Bouge Recruiting Namur, Belgium Contact: Richard Frognier AZ Delta Recruiting Roeselare, Belgium Contact: Lore Carton Hospital Center De Wallonie Picarde Recruiting Tournai, Belgium Contact: Alain Palem, MD Collapse << |
| NCT01782326 | - | - | Completed | - | - |
| NCT02546349 | COPD | Phase 4 | Unknown | January 2016 | Taiwan ... more >> Taipei Veterans General Hospital Taipei City, Taiwan, 886 Collapse << |
| NCT01242098 | - | - | Completed | - | United Kingdom ... more >> Optimum Patient Care Cawston, Norfolk, United Kingdom, NR10 4FE Collapse << |
| NCT01312961 | - | - | Completed | - | - |
| NCT02301975 | Asthma | Phase 3 | Completed | - | - |
| NCT02446418 | Asthma | Phase 3 | Completed | - | - |
| NCT02301975 | - | - | Completed | - | - |
| NCT02127866 | Asthma | Phase 2 | Completed | - | - |
| NCT02741271 | Asthma | Phase 3 | Completed | - | - |
| NCT02094274 | Asthma | Phase 1 | Completed | - | Germany ... more >> Almirall Investigational Site #1 Berlin, Germany, 14050 United Kingdom Almirall Investigational Site #2 Harrow, United Kingdom, HA1 3UJ Collapse << |
| NCT00521222 | Asthma | Not Applicable | Unknown | November 2013 | United States, New Jersey ... more >> Hackensack University Medical Center Hackensack, New Jersey, United States, 07601 United States, New York Columbia University Medical Center Eastside New York, New York, United States, 10022 Columbia Presbyterian Medical Center New York, New York, United States, 10032 Collapse << |
| NCT01437995 | Asthma | Phase 4 | Completed | - | United States, Arizona ... more >> University of Arizona, Arizona Respiratory Center Tucson, Arizona, United States, 85724 United States, California University of California, San Diego San Diego, California, United States, 92103 United States, Colorado National Jewish Health Denver, Colorado, United States, 80206 United States, Florida Nemours Children's Clinic Jacksonville, Florida, United States, 32207 University of Miami/University of South Florida Tampa, Florida, United States, 33613 United States, Illinois The Illinois Consortium Chicago, Illinois, United States, 60611 United States, Indiana St. Vincent Healthcare Indianapolis, Indiana, United States, 46260 St. Vincent Hospital and Health Care Center, Inc Indianapolis, Indiana, United States, 46290 United States, Louisiana Louisiana State University Health Sciences Center, The Ernest N. Morial Asthma, Allergy and Respiratory Disease Center New Orleans, Louisiana, United States, 70112 United States, Missouri University of Missouri, Kansas City School of Medicine Kansas City, Missouri, United States, 64108 Washington University/St. Louis University St. Louis, Missouri, United States, 63110 United States, New York Hofstra North Shore-LIJ School of Medicine New Hyde Park, New York, United States, 11040 New York University School of Medicine New York, New York, United States, 10016 Maria Fareri Children's Hospital at Westchester Medical Center and New York Medical College Valhalla, New York, United States, 10595 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio The Ohio State University Medical Center Columbus, Ohio, United States, 43221 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 United States, Vermont Northern New England Consortium Colchester, Vermont, United States, 05446 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT03691961 | Asthma | Not Applicable | Recruiting | January 24, 2020 | France ... more >> Foch Hospital Recruiting Suresnes, France, 92150 Contact: Hélène Salvator, Dr 33 (0)1 46 25 29 55 h.salvator@hopital-foch.com Principal Investigator: Hélène Salvator, Dr Collapse << |
| NCT01736917 | Chemotherapy-Induced Nausea an... more >>d Vomiting Collapse << | Phase 2 | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, Missouri Siteman Cancer Center St. Louis, Missouri, United States, 63110 United States, Nebraska Nebraska Cancer Specialists Omaha, Nebraska, United States, 68114 United States, South Carolina MUSC Hollings Cancer Center Charleston, South Carolina, United States, 29425 Collapse << |
| NCT01437995 | - | - | Completed | - | - |
| NCT01599871 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Spain ... more >> Hospital Universitario Son Espases Palma de Mallorca, Baleares, Spain, 07010 Hospital Universitario Nuestra Señora de la Candelaria Santa Cruz de Tenerife, Islas Canarias, Spain, 38010 Clínica Universitaria de Navarra Pamplona, Navarra, Spain, 31008 Hospital del Mar Barcelona, Spain, 08003 Hospital de Sant Pau Barcelona, Spain, 08025 Hospital Clínic de Barcelona Barcelona, Spain, 08036 Hospital Gregorio Marañón Madrid, Spain, 28009 Fundación Jiménez Díaz Madrid, Spain, 28040 Hospital 12 de Octubre Madrid, Spain, 28044 Hospital Miguel Servet Zaragoza, Spain, 50004 Collapse << |
| NCT01736917 | - | - | Completed | - | - |
| NCT01313585 | - | - | Completed | - | United Kingdom ... more >> General Practice Research Database London, United Kingdom Collapse << |
| NCT02345161 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT02934945 | Bronchial Asthma | Phase 4 | Unknown | March 2017 | China, Guangdong ... more >> Zhujiang Hospital of Southern Medical University Recruiting Guangzhou, Guangdong, China, 510282 Contact: Shuyu Chen, M.D 13580421202 1137968694@qq.com Contact: Jielu Liu, M.D Collapse << |
| NCT00576069 | - | - | Unknown | June 2018 | United States, California ... more >> Arthur F Gelb Medical Corporation Recruiting Lakewood, California, United States, 90712 Principal Investigator: Arthur F Gelb, MD Collapse << |
| NCT00471809 | Asthma | Phase 4 | Terminated(The CARE Network DS... more >>MB recommended to the NHLBI that the MARS trial be terminated, based on a futility analysis with 55 randomized children.) Collapse << | - | United States, Arizona ... more >> University of Arizona College of Medicine Tucson, Arizona, United States, 85724 United States, California Los Angeles, Kaiser Permanente Allergy Department Los Angeles, California, United States, 90027 Kaiser Permanente Medical Center San Diego, California, United States, 92111 United States, Colorado National Jewish Medical and Research Center Denver, Colorado, United States, 80206 United States, Missouri Washington University School of Medicine St. Louis, Missouri, United States, 63110 United States, Wisconsin University of Wisconsin - Madison Madison, Wisconsin, United States, 53792-3244 Collapse << |
| NCT02574975 | Asthma | Phase 4 | Unknown | May 2016 | China, Guangdong ... more >> Zhujiang Hospital Recruiting Guangzhou, Guangdong, China, 510280 Contact: Shuhan Wu, Master 13268268627 664397041@qq.com Collapse << |
| NCT00395304 | - | - | Completed | - | - |
| NCT01141465 | - | - | Completed | - | United Kingdom ... more >> General Practice Research Database London, United Kingdom, SW8 5NQ Collapse << |
| NCT02165033 | Asthma | Phase 1 | Completed | - | Taiwan ... more >> Tri-Service General Hospital Taipei, Neihu, Taiwan, 114 Collapse << |
| NCT03207243 | Asthma | Phase 1 Phase 2 | Active, not recruiting | May 23, 2019 | - |
| NCT03066544 | Migraine | Phase 1 Phase 2 | Recruiting | June 30, 2020 | United States, Connecticut ... more >> Hartford HealthCare Headache Center Recruiting Wethersfield, Connecticut, United States, 06109 Contact: Brooke Pellegrino, PhD 860-696-2925 Brooke.Pellegrino@hhchealth.org Principal Investigator: Brian Grosberg, MD Collapse << |
| NCT00395304 | Asthma | Phase 3 | Completed | - | United States, Arizona ... more >> University of Arizona College of Medicine Tucson, Arizona, United States, 85724 United States, California Kaiser Permanente Medical Center San Diego, California, United States, 92111 United States, Colorado National Jewish Medical and Research Center Denver, Colorado, United States, 80206 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, Wisconsin University of Wisconsin - Madison Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00888901 | Idiopathic Thrombocytopenic Pu... more >>rpura Collapse << | Phase 4 | Completed | - | - |
| NCT00200967 | Asthma | Phase 3 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94143-0130 United States, Colorado National Jewish Medical & Research Center Denver, Colorado, United States, 80206 United States, Massachusetts Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 United States, Missouri Washington University Saint Louis, Missouri, United States, 63110 United States, North Carolina Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27103 United States, Wisconsin University of Wisconsin Madison Madison, Wisconsin, United States, 53792-3244 Collapse << |
| NCT00673309 | Burns | Phase 2 Phase 3 | Completed | - | United States, Texas ... more >> University of Texas Medical Branch Galveston, Texas, United States, 77550 Collapse << |
| NCT02165046 | Asthma Chroni... more >>c Obstructive Pulmonary Disease (COPD) Collapse << | Phase 1 | Completed | - | Taiwan ... more >> Tri-Service General Hospital Taipei, Taiwan Collapse << |
| NCT00319488 | Asthma Lung D... more >>iseases Collapse << | Not Applicable | Completed | - | United States, Arizona ... more >> University of Arizona, College of Medicine Tucson, Arizona, United States, 85724 United States, California UCSD School of Medicine LaJolla, California, United States, 92093 United States, Colorado National Jewish Medical and Research Center Denver, Colorado, United States, 80206 United States, Missouri Washington University School of Medicine Patient Oriented Research Unit St. Louis, Missouri, United States, 63110 United States, Pennsylvania Dept. of Health Evaluation Sciences, Penn State College of Medicine Hershey, Pennsylvania, United States, 17033 United States, Wisconsin University of Wisconsin - Madison Madison, Wisconsin, United States, 53792 Collapse << |
| NCT02345161 | - | - | Completed | - | - |
| NCT00706446 | Asthma | Not Applicable | Terminated(Funding was termina... more >>ted) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00706446 | - | - | Terminated(Funding was termina... more >>ted) Collapse << | - | - |
| NCT00200967 | - | - | Completed | - | - |
| NCT03016741 | Castration-Resistant Prostatic... more >> Cancer Metastatic Prostate Carcinoma Recurrent Prostate Carcinoma Stage IV Prostate Cancer Hormone-Refractory Prostate Cancer Collapse << | Phase 4 | Recruiting | August 2021 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Alicia K. Morgans, MD, MPH Principal Investigator: Alicia K. Morgans, MD, MPH United States, Tennessee Vanderbilt-Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Contact: ICC Clinical Trials Information Program 800-811-8480 cip@vanderbilt.edu Collapse << |
| NCT00419952 | Asthma | Phase 3 | Completed | - | - |
| NCT01231230 | Asthma | Not Applicable | Completed | - | - |
| NCT00419952 | - | - | Completed | - | - |
| NCT02048644 | Idiopathic Pulmonary Fibrosis | Phase 2 | Completed | - | United Kingdom ... more >> Respiratory Medicine Clinical trials Unit Cottingham, East Yorkshire, United Kingdom, HU16 5JQ Collapse << |
| NCT01676987 | Asthma | Phase 3 | Completed | - | - |
| NCT01231230 | - | - | Completed | - | - |
| NCT01468532 | Adenocarcinoma of the Prostate... more >> Hormone-resistant Prostate Cancer Recurrent Prostate Cancer Stage IV Prostate Cancer Collapse << | Phase 1 Phase 2 | Unknown | March 2017 | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201 Collapse << |
| NCT00557180 | - | - | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92093 University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado National Jewish Medical & Research Center Denver, Colorado, United States, 80206 United States, Massachusetts Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 United States, Missouri Washington University St Louis, Missouri, United States, 63130 United States, New York Columbia University Medical Center New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555 United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States, 53706 Collapse << |
| NCT03622112 | Asthma | Phase 2 | Not yet recruiting | September 6, 2019 | - |
| NCT02949284 | Stage II Prostate Adenocarcino... more >>ma Stage III Prostate Adenocarcinoma Collapse << | Phase 2 | Recruiting | December 2018 | United States, California ... more >> City of Hope Comprehensive Cancer Center Not yet recruiting Duarte, California, United States, 91010 Contact: Bertram Yuh 626-394-2383 byuh@coh.org Principal Investigator: Bertram Yuh United States, New Jersey Rutgers Cancer Institute of New Jersey Recruiting New Brunswick, New Jersey, United States, 08903 Contact: Isaac Y. Kim 732-235-2043 kimiy@cinj.rutgers.edu Principal Investigator: Isaac Y. Kim Collapse << |
| NCT02319564 | Wheeze | Phase 4 | Withdrawn(Study population too... more >> narrow - unable to recruit any patients.) Collapse << | - | Canada, Alberta ... more >> Alberta Children's Hospital Calgary, Alberta, Canada, T3B 6A8 Collapse << |
| NCT03136562 | - | - | Recruiting | June 1, 2019 | Denmark ... more >> Rigshospitalet Recruiting Copenhagen, Denmark, 2100 Contact: Ulla Feldt-Rasmussen, Professor +4535451023 ufeldt@rh.dk Principal Investigator: Ulla Feldt-Rasmussen, Professor Collapse << |
| NCT03064633 | Postoperative Pain | Phase 4 | Completed | - | Egypt ... more >> Fayoum University hospital Madīnat al Fayyūm, Faiyum Governorate, Egypt, 63514 Collapse << |
| NCT03491696 | Depressive Disorder ... more >> Hormone Disturbance Collapse << | Phase 4 | Not yet recruiting | September 2019 | France ... more >> Hopitaux Universitares de Strasbourg Not yet recruiting Strasbourg, Bas Rhin, France, 67000 Contact: Jack Foucher, MD +33 3 88 11 69 21 CEMNIS@chru-strasbourg.fr Contact: Ludovic C. Jeanjean, Intern +33 6 30 11 09 13 ludovic.jeanjean@icloud.com Sub-Investigator: Jack Foucher, MD Principal Investigator: Ludovic C. Jeanjean, Intern Centre Hospitalier de Rouffach Not yet recruiting Rouffach, Haut Rhin, France, 68250 Contact: Fabrice Duval, MD +33 389787018 f.duval@ch-rouffach.fr Contact: Ludovic C. Jeanjean, Intern +33 6 30 11 09 13 ludovic.jeanjean@icloud.com Sub-Investigator: Alexis Erb, MD Sub-Investigator: Fabrice Duval, MD Principal Investigator: Ludovic C. Jeanjean, Intern Collapse << |
| NCT01321827 | Allergic Bronchopulmonary Aspe... more >>rgillosis Collapse << | Phase 2 Phase 3 | Completed | - | India ... more >> Postgraduate Institute of Medical Education and Research Chandigarh, UT, India, 160012 Collapse << |
| NCT03410160 | - | - | Active, not recruiting | December 2018 | Germany ... more >> University Hospital Wuerzburg, Dept. of medicine I Wuerzburg, Germany, 97080 Collapse << |
| NCT01411046 | - | - | Recruiting | December 2017 | Denmark ... more >> Rigshospitalet Recruiting Copenhagen, Denmark, 2100 Contact: Ulla Feldt-Rasmussen, Professor +4535451023 ulla.feldt-rasmussen@dadlnet.dk Principal Investigator: Ulla Feldt-Rasmussen, Professor Collapse << |
| NCT00159263 | Asthma | Not Applicable | Completed | - | United Kingdom ... more >> Section of Airway Disease, Asthma Lab, Imperial College London, Royal Brompton Hospital London, United Kingdom Collapse << |
| NCT01170429 | Cough Variant Asthma | Phase 4 | Unknown | February 2011 | China, Shanghai ... more >> Xin Zhou Shanghai, Shanghai, China, 200080 Collapse << |
| NCT03062150 | Major Depression | Not Applicable | Recruiting | June 30, 2019 | Germany ... more >> Charité Universitätsmedizin Berlin Recruiting Berlin, Germany, 12203 Contact: Christian Otte, Prof. Dr. +49-30-450-517532 Christian.Otte@charite.de Collapse << |
| NCT03511625 | Rheumatoid Arthritis | Phase 3 | Not yet recruiting | September 2018 | - |
| NCT00744224 | Glucocorticoid-induced Glucome... more >>tabolic Abnormalities Glucocorticoid-induced Beta-cell Dysfunction Collapse << | Not Applicable | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Noord-Holland, Netherlands, 1081HV Collapse << |
| NCT01304901 | Asthma Acute ... more >>Asthma Collapse << | Phase 4 | Completed | - | Turkey ... more >> Kecioren Education and Training Hospital Ankara, Keçiören, Turkey, 06290 Collapse << |
| NCT02767089 | Healthy Adults | Phase 1 | Completed | - | United States, Michigan ... more >> Jasper Clinic, Inc. Kalamazoo, Michigan, United States, 49007 Collapse << |
| NCT01209715 | COPD | Phase 2 | Withdrawn(Due to the ubiquitou... more >>s use of ICS in the treatment of COPD in 2012, it was hard to find the study population.) Collapse << | - | United States, Florida ... more >> University of Miami School of Medicine Miami, Florida, United States, 33136 Collapse << |
| NCT00522678 | Asthma | Phase 1 | Completed | - | Australia, Victoria ... more >> GSK Investigational Site Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01225549 | - | - | Completed | - | - |
| NCT02967159 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Asthma Collapse << | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany, 14050 Collapse << |
| NCT01225549 | Asthma | Phase 2 | Completed | - | Canada, Alberta ... more >> Research Site Calgary, Alberta, Canada Canada, Ontario Research Site Hamilton, Ontario, Canada Canada, Saskatchewan Research Site Saskatoon, Saskatchewan, Canada Canada Research Site Quebec, Canada Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
