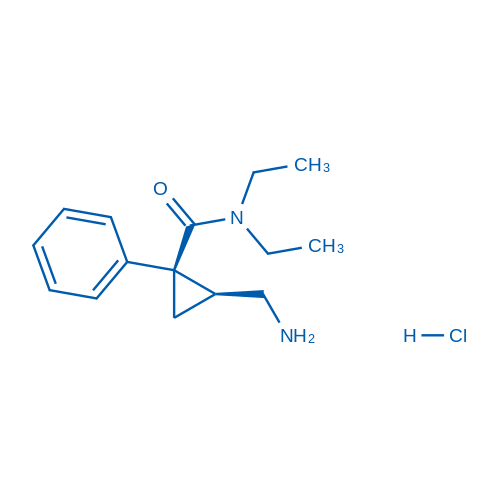
CAS No.: 101152-94-7
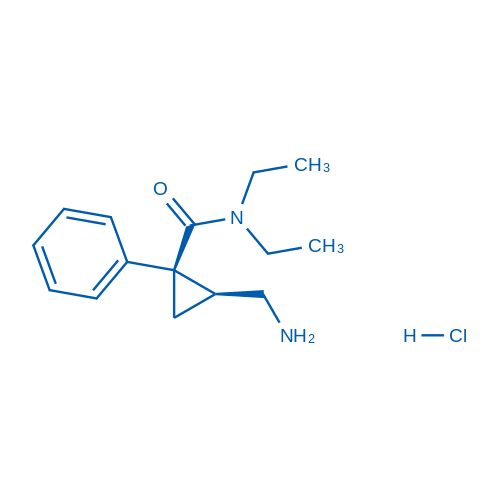
Milnacipran HCl/盐酸米那普仑 Catalog No. CSN11446
Synonyms: Milnacipran (hydrochloride);F 2207;Milnacipran
Milnacipran HCl is an inhibitor of serotonin-norepinephrine reuptake that can be used to treat fibromyalgia.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00605917 - - Completed - - NCT00605917 - - Completed - - NCT00605865 - - Completed - - - 更多
- 参考文献
- [1] Maj J, Rogoz Z, et al. Pharmacological effects of milnacipran, a new antidepressant, given repeatedly on the alpha1-adrenergic and serotonergic 5-HT2A systems. J Neural Transm (Vienna). 2000;107(11):1345-59.
- [2] Moret C, Charveron M, et al. Biochemical profile of midalcipran (F 2207), 1-phenyl-1-diethyl-aminocarbonyl-2-aminomethyl-cyclopropane (Z) hydrochloride, a potential fourth generation antidepressant drug. Neuropharmacology. 1985 Dec;24(12):1211-9.
- [3] Otte C, Gold SM, Penninx BW, et al. Major depressive disorder. Nat Rev Dis Primers. 2016;2:16065
- [4] Wagner G, Schultes MT, Titscher V, Teufer B, Klerings I, Gartlehner G. Efficacy and safety of levomilnacipran, vilazodone and vortioxetine compared with other second-generation antidepressants for major depressive disorder in adults: A systematic review and network meta-analysis. J Affect Disord. 2018;228:1‐12
- [5] Bruno A, Morabito P, Spina E, Muscatello MR. The Role of Levomilnacipran in the Management of Major Depressive Disorder: A Comprehensive Review. Curr Neuropharmacol. 2016;14(2):191‐199
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 101152-94-7 | 储存条件 |
|
|||||||||
| 分子式 | C15H23ClN2O | 运输 | 蓝冰 | |||||||||
| 分子量 | 282.81 | 别名 | Milnacipran (hydrochloride);F 2207;Milnacipran;Levomilnacipran;Ixel;Savella;Dalcipran;Toledomin;盐酸米那普仑 | |||||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00605917 | - | - | Completed | - | - |
| NCT00605917 | - | - | Completed | - | - |
| NCT00605865 | - | - | Completed | - | - |
| NCT00605865 | - | - | Completed | - | - |
| NCT01488266 | Major Depressive Disorder | Not Applicable | Unknown | March 2013 | Korea, Republic of ... more >> Korean Univ Ansan Hospital; Bucheon St.Mary Hospital; DonggukUniv Gyeongju Hospital; Catholic University of Korea St. Paul's Hospital Seoul, Korea, Republic of Taiwan Chang Gung Memorial Hospital; Kaohsiung Medical University Chung-ho Memorial Hospital Taipei, Taiwan Collapse << |
| NCT01352559 | Depression | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT00725101 | - | - | Completed | - | - |
| NCT00817375 | Depression Ad... more >>verse Reaction to Drug Collapse << | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Kangnam, Seoul, Korea, Republic of, 135-710 Contact: Samsung Medical Center Kim, MD.pHD 82-2-3410-3582 dohkwan.kim@samsung.com Collapse << |
| NCT00725101 | - | - | Completed | - | - |
| NCT00225511 | Depression | Phase 3 | Completed | - | - |
| NCT01228357 | Depression De... more >>pressive Symptoms Collapse << | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Kangnam, Seoul, Korea, Republic of Contact: Doh Kwan Kim, MD.PhD 82-2-3410-3582 dohkwan.kim@samsung.com Contact: Woojae Myung, MD. 82-2-3410-6562 smbhealer@gmail.com Collapse << |
| NCT01352572 | Depression | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01254305 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Alabama ... more >> Forest Investigative Site 010 Birmingham, Alabama, United States, 35216 United States, Arkansas Forest Investigative Site 002 Little Rock, Arkansas, United States, 72223 United States, California Forest Investigative Site 001 Cerritos, California, United States, 90703 United States, Florida Forest Investigative Site 014 Fort Myers, Florida, United States, 33912 Forest Investigative Site 006 Jacksonville, Florida, United States, 32216 Forest Investigative Site 017 Orange City, Florida, United States, 32763 Forest Investigative Site 005 Orlando, Florida, United States, 32806 Forest Investigative Site 012 Tampa, Florida, United States, 33613 United States, Georgia Forest Investigative Site 009 Atlanta, Georgia, United States, 30308 United States, Illinois Forest Investigative Site 016 Joliet, Illinois, United States, 60435 United States, Louisiana Forest Investigative Site 004 New Orleans, Louisiana, United States, 70122 United States, Massachusetts Forest Investigative Site 022 Boston, Massachusetts, United States, 02135 United States, New York Forest Investigative Site 011 Bronx, New York, United States, 10467 Forest Investigative Site 015 Cedarhurst, New York, United States, 11516 United States, Ohio Forest Investigative Site 003 Cincinnati, Ohio, United States, 45227 Forest Investigative Site 013 Dayton, Ohio, United States, 45417 United States, Rhode Island Forest Investigative Site 020 Lincoln, Rhode Island, United States, 02865 United States, Tennessee Forest Investigative Site 018 Memphis, Tennessee, United States, 38119 United States, Texas Forest Investigative Site 008 Dallas, Texas, United States, 75235 United States, Wisconsin Forest Investigative Site 007 Middleton, Wisconsin, United States, 53562 Collapse << |
| NCT03128021 | Major Depressive Disorder | Phase 4 | Recruiting | July 2022 | United States, Pennsylvania ... more >> University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Dana Williams, MS 412-246-5924 barvdm@upmc.edu Contact: Rachel Berta, BA 412-246-5685 goodra@upmc.edu Sub-Investigator: Meryl A Butters, Ph.D. Sub-Investigator: Tae Kim, Ph.D. Sub-Investigator: Dana Tudorascu, Ph.D. Principal Investigator: Howard Aizenstein, M.D., Ph.D. Principal Investigator: Carmen Andreescu, M.D. Collapse << |
| NCT02883660 | - | - | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00969709 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01254305 | - | - | Completed | - | - |
| NCT00606203 | Ischemic Stroke ... more >> Depression Collapse << | Not Applicable | Unknown | September 2011 | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Chiayi, Taiwan, 613 Contact: Jian-An Su, MD +886-5-3621000 ext 2313 jian.7715@gmail.com Principal Investigator: Shih-Young Chou, MD Principal Investigator: Ching-Shu Tsai, MD Collapse << |
| NCT00969709 | - | - | Completed | - | - |
| NCT01034462 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01319825 | Migraine With Aura ... more >> Migraine Without Aura Chronic Migraine Collapse << | Phase 4 | Unknown | July 2012 | United States, California ... more >> California Medical Clinic for Headache Not yet recruiting Santa Monica, California, United States, 90404 Contact: Tatyana Neyman, MD 310-315-1456 tneyman@drkudrow.com Sub-Investigator: Alan Rapoport, MD Collapse << |
| NCT00969150 | - | - | Completed | - | - |
| NCT01914042 | Neuropathic Pain | Phase 2 | Recruiting | December 2019 | Israel ... more >> Rambam Health Care Campus Recruiting Haifa, Israel, 31096 Contact: Elon Eisenberg, MD 972 4 8542880 e_eisenberg@rambam.health.gov.il Principal Investigator: Elon Eisenberg, MD Collapse << |
| NCT01034462 | - | - | Completed | - | - |
| NCT01038323 | Fibromyalgia | Phase 4 | Completed | - | United States, Indiana ... more >> Clinical Research Center for Pain, 250 University Blvd Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT02687165 | Post Treatment Lyme Syndrome (... more >>PTLS) Chronic Pain Collapse << | Not Applicable | Recruiting | February 2018 | United States, New York ... more >> Columbia University Medical Center Recruiting New York, New York, United States, 10032-0000 Contact: Phil Grebowiec, MD 646-774-8100 eb3048@cumc.coumbia.edu Collapse << |
| NCT00793520 | - | - | Terminated | - | - |
| NCT00969150 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00757731 | Fibromyalgia Syndrome | Phase 3 | Completed | - | Czech Republic ... more >> Rheumatology Ambulance Pardubice, Czech Republic, 530 02 Finland Kuopion Oma Laakari Oy Kuopio, Finland, 70100 France Hopital Hotel Dieu Paris, France Germany KKSK KLINIKUM DER UNIVERSITAET ZU KOELN - Anaesthesiology and Intensive Care Koln, Germany, D50931 Italy Ospedale Luigi Sacco Milano, Italy, 20157 Norway Center For Clinical Studies Lillehamer, Norway, 2609 Portugal Hospital Egas Moniz Lisboa, Portugal, 1349-019 Romania Dr I CANTACUZINO CLINICAL HOSPITAL Bucharest, Romania, 020475 Spain Hospital de La Esperanza Barcelona, Spain, 08024 Sweden Gottfriesclinic Ab Molndal, Sweden, 43137 Collapse << |
| NCT01038323 | - | - | Completed | - | - |
| NCT02265367 | Tobacco Use Disorder | Not Applicable | Completed | - | United States, Minnesota ... more >> Clinical and Translational Sciences Institute Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT01747044 | Fibromylagia | Phase 2 | Unknown | November 2014 | France ... more >> CHU de Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 04 73 75 11 95 placarin@chu-clermontferrand.fr Collapse << |
| NCT01377194 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01377194 | - | - | Completed | - | - |
| NCT03569475 | Major Depressive Disorder | Phase 3 | Recruiting | October 21, 2021 | United States, Arkansas ... more >> Woodland International Research Group Recruiting Little Rock, Arkansas, United States, 72211 United States, California Care Access Research, Beverly Hills Recruiting Beverly Hills, California, United States, 90212 Behavioral Research Specialists, LLC Recruiting Glendale, California, United States, 91206 Excell Research, Inc Recruiting Oceanside, California, United States, 92056 Orange County Neuropsychiatric Research Center, LLC Not yet recruiting Orange, California, United States, 92868 Elite Clinical Trials, Inc. Not yet recruiting Wildomar, California, United States, 92595 United States, Georgia Atlanta Behavioral Research, LLC Not yet recruiting Atlanta, Georgia, United States, 30338 United States, Illinois Joliet Center Clinical Research Not yet recruiting Joliet, Illinois, United States, 60435 Capstone Clinical Research Recruiting Libertyville, Illinois, United States, 60048 AMR-Baber Research, Inc. Not yet recruiting Naperville, Illinois, United States, 60563 Southern Illinois University School of Medicine Not yet recruiting Springfield, Illinois, United States, 62702 United States, Kansas Psychiatric Associates Not yet recruiting Overland Park, Kansas, United States, 66211 United States, Maryland Kennedy Krieger Institute Not yet recruiting Baltimore, Maryland, United States, 21205 United States, Michigan Rochester Center for Behavioral Medicine Not yet recruiting Rochester Hills, Michigan, United States, 48307 United States, Missouri Millennium Center for Clinical Research Not yet recruiting Creve Coeur, Missouri, United States, 63141 United States, New York Finger Lakes Clinical Research Recruiting Rochester, New York, United States, 14618 United States, Ohio University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45219 The Ohio State University Department of Psychiatry Not yet recruiting Columbus, Ohio, United States, 43210 United States, Texas BioBehavioral Research of Austin Recruiting Austin, Texas, United States, 78759 United States, Utah Ericksen Research and Development Not yet recruiting Clinton, Utah, United States, 84015 Collapse << |
| NCT00436033 | Fibromyalgia Syndrome | Phase 3 | Completed | - | Czech Republic ... more >> Rheumatology Ambulance Pardubice, Czech Republic, 530 02 Denmark FREDERIKSBERG HOSPITAL - Clinic of Rheumatology Frederiksberg, Denmark, F2000 Finland Kuopion Oma Laakari Oy Kuopio, Finland, 70100 France Hopital Hotel Dieu Paris, France Germany KKSK KLINIKUM DER UNIVERSITAET ZU KOELN - Anaesthesiology and Intensive Care Koln, Germany, D50931 Italy Ospedale Luigi Sacco Milano, Italy, 20157 Norway Center For Clinical Studies Lillehamer, Norway, 2609 Poland "Nasz Lekarz" Torun, Poland Portugal Hospital Egas Moniz Lisboa, Portugal, 1349-019 Romania Dr I CANTACUZINO CLINICAL HOSPITAL Bucharest, Romania, 020475 Spain Hospital de La Esperanza Barcelona, Spain, 08024 Sweden Gottfriesclinic Ab Molndal, Sweden, 43137 United Kingdom KINGS COLLEGE HOSPITAL - Clinic Trial Unit Academic Department of Rheumatology London, United Kingdom, SE59RJ Collapse << |
| NCT01288807 | Fibromyalgia | Phase 4 | Completed | - | United States, California ... more >> UCSD Medical Center, La Jolla San Diego, California, United States, 92037 Collapse << |
| NCT00797797 | - | - | Completed | - | - |
| NCT00793520 | Fibromyalgia | Phase 3 | Terminated | - | United States, Michigan ... more >> Forest Investigative Site Ann Arbor, Michigan, United States, 48106 Collapse << |
| NCT00314249 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT00797797 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT00314249 | - | - | Completed | - | - |
| NCT00098124 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT02783430 | Depression | Phase 2 Phase 3 | Recruiting | September 2019 | France ... more >> University Hospital, Not yet recruiting Lille, France Contact: Magali Pierrat, MD Principal Investigator: Magali Pierrat, MD Centre Hospitalier de Valenciennes Recruiting Valenciennes, France Principal Investigator: Antoine Lemaire, MD Collapse << |
| NCT01331109 | - | - | Terminated | - | - |
| NCT01393522 | Chronic Migraine | Not Applicable | Completed | - | United States, Missouri ... more >> Mercy Health Research St. Louis, Missouri, United States, 63141 United States, North Carolina Headache Wellness Center Greensboro, North Carolina, United States, 27405 Collapse << |
| NCT02431806 | Major Depressive Disorder | Phase 3 | Recruiting | December 21, 2018 | - |
| NCT01331109 | Primary Fibromyalgia | Phase 2 | Terminated | - | - |
| NCT01289236 | Chronic Shoulder Pain | Phase 4 | Completed | - | United States, Florida ... more >> Delray Research Associates Delray Beach, Florida, United States, 33484 Collapse << |
| NCT00757679 | Fibromyalgia Syndrome | Phase 2 | Completed | - | Switzerland ... more >> Clinical Pharmacology & Toxicology Multidisciplinary Pain Centre Geneve, Switzerland Collapse << |
| NCT01026077 | - | - | Recruiting | December 2019 | United States, North Carolina ... more >> INC Research Recruiting Wilmington, North Carolina, United States, 28405 Contact 877-643-3010 pregnancyregistries@incresearch.com Collapse << |
| NCT00618956 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT01510457 | - | - | Completed | - | - |
| NCT00618956 | - | - | Completed | - | - |
| NCT01328002 | Primary Fibromyalgia Syndrome | Phase 2 | Terminated | - | - |
| NCT01329406 | Osteoarthritis | Phase 4 | Unknown | September 2012 | United States, Massachusetts ... more >> Analgesic Solutions Recruiting Natick, Massachusetts, United States, 01760 Contact: Karen Cowles, RN, MPH 781-444-9605 ext 121 kcowles@analgesicsolutions.com Contact: Courtney Lincoln 781-444-9605 ext 119 clincoln@analgesicsolutions.com Principal Investigator: Stephen L. Wright, M.D. Sub-Investigator: Nathaniel P. Katz, M.D., M.S. Sub-Investigator: Eric Osgood, M.D. Collapse << |
| NCT01108731 | Fibromyalgia | Phase 2 Phase 3 | Completed | - | United States, New York ... more >> Pain and Fatigue Study Center - Beth Israel Medical Center New York, New York, United States, 10003 Collapse << |
| NCT02720198 | Major Depressive Disorder | Phase 3 | Completed | - | United States, Georgia ... more >> Institute for Advanced Medical Research Alpharetta, Georgia, United States, 30005 Collapse << |
| NCT01337700 | Autism Spectrum Disorder ... more >> Asperger Syndrome Aspergers Syndrome Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Montefiore Medical Center, Albert Einstein College of Medicine Bronx, New York, United States, 10467 Collapse << |
| NCT01471379 | Irritable Bowel Syndrome | Phase 2 | Terminated(Due to recruitment ... more >>difficulties the study is terminated.) Collapse << | - | United States, North Carolina ... more >> UNC Center for Functional GI and Motility Disorders Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01510457 | Knee Osteoarthritis ... more >> Degenerative Joint Disease Chronic Pain Collapse << | Phase 4 | Completed | - | - |
| NCT01471379 | - | - | Terminated(Due to recruitment ... more >>difficulties the study is terminated.) Collapse << | - | - |
| NCT01418651 | Fibromyalgia | Phase 3 | Terminated(Lack of subjects) | August 2011 | United States, Arizona ... more >> Banner Sun Health Research Institute Sun City, Arizona, United States, 85351 Collapse << |
| NCT03249311 | None (i.e. Healthy Volunteers) | Phase 4 | Recruiting | January 31, 2020 | Canada, Ontario ... more >> Institute of Mental Health Research, Royal Ottawa Mental Health Centre Recruiting Ottawa, Ontario, Canada, K1Z 7K4 Contact: Holly English, RN 613-722-6521 ext 6405 holly.english@theroyal.ca Contact: Wendy Fusee, RN 613-722-6521 ext 7828 wendy.fusee@theroyal.ca Principal Investigator: Pierre Blier, MD Collapse << |
| NCT01221740 | Back Pain | Phase 3 | Withdrawn(Company decided to d... more >>iscontinue) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Pain Trials Center Chestnut Hill, Massachusetts, United States, 02467 Collapse << |
| NCT01108731 | - | - | Completed | - | - |
| NCT01173055 | Fibromyalgia | Phase 4 | Completed | - | United States, Michigan ... more >> University of Michigan, Chronic Pain and Fatigue Research Center Ann Arbor, Michigan, United States, 48106 Collapse << |
| NCT01173055 | - | - | Completed | - | - |
| NCT02466958 | Major Depressive Disorder | Phase 4 | Active, not recruiting | December 2018 | United States, California ... more >> UCLA Semel Institute - Neuropsychiatric Institute (NPI) Los Angeles, California, United States, 90095 Collapse << |
| NCT01777581 | Radicular Pain Related to Lumb... more >>osacral Disc Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01014585 | Fibromyalgia | Phase 4 | Completed | - | - |
| NCT01014585 | - | - | Completed | - | - |
| NCT01288937 | Idiopathic Peripheral Neuropat... more >>hy Collapse << | Not Applicable | Terminated(Poor recruitment) | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT01225068 | Low Back Pain | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT01304589 | Vestibulodynia ... more >> Vulvodynia Collapse << | Phase 3 | Completed | - | United States, Tennessee ... more >> Women's Health Specialists, PLLC Germantown, Tennessee, United States, 38138 Collapse << |
| NCT01777581 | - | - | Completed | - | - |
| NCT01125423 | Fibromyalgia | Phase 4 | Unknown | - | United States, New York ... more >> Upstate Clinical Research, LLC Recruiting Albany, New York, United States, 12205 Contact: Suzanne Alterman, RN 518-533-1500 salterman@upstateneuro.com Principal Investigator: James Wymer, MD Collapse << |
| NCT01304589 | - | - | Completed | - | - |
| NCT01077375 | Fibromyalgia | Phase 4 | Completed | - | - |
| NCT01077375 | - | - | Completed | - | - |
| NCT01225991 | Rheumatoid Arthritis | Phase 4 | Completed | - | United States, California ... more >> UCLA Semel Institute Los Angeles, California, United States, 90095 Collapse << |
| NCT01225068 | - | - | Completed | - | - |
| NCT01225991 | - | - | Completed | - | - |
| NCT01294059 | Fibromyalgia | Phase 1 | Completed | - | United States, Florida ... more >> Center for Musculoskeletal Pain Research Gainesville, Florida, United States, 32610 Collapse << |
| NCT01829243 | Fibromyalgia ... more >>Neurocognition Collapse << | Phase 3 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center / Civitan Building Durham, North Carolina, United States, 27705 Collapse << |
| NCT01780389 | Knee Pain After Total Knee Art... more >>hroplasty Osteoarthritis Pain Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT01780389 | - | - | Completed | - | - |
| NCT02288325 | Depressive Disorder, Major | Phase 4 | Completed | - | - |
| NCT01829243 | - | - | Completed | - | - |
| NCT01359826 | Systemic Lupus Erythematosus ... more >> Widespread Pain Fibromyalgia Collapse << | Phase 4 | Withdrawn(PI has let the insti... more >>tution. We are unable to locate any study documents that would indicate study enrollment) Collapse << | - | United States, California ... more >> Loma Linda University Medical Center Loma Linda, California, United States, 92354 Collapse << |
| NCT01207453 | Arthritis, Rheumatoid | Phase 4 | Completed | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT02288325 | - | - | Completed | - | - |
| NCT01207453 | - | - | Completed | - | - |
| NCT01234675 | Sleep Disorders ... more >> Fibromyalgia Sleep Collapse << | Phase 4 | Completed | - | United States, Ohio ... more >> Cleveland Sleep Research Center Middleburg Heights, Ohio, United States, 44130 Collapse << |
| NCT01234675 | - | - | Completed | - | - |
| NCT00793520 | Fibromyalgia | Phase 3 | Terminated | - | United States, Michigan ... more >> Forest Investigative Site Ann Arbor, Michigan, United States, 48106 Collapse << |
| NCT00605917 | - | - | Completed | - | - |
| NCT00605865 | - | - | Completed | - | - |
| NCT00605865 | - | - | Completed | - | - |
| NCT00725101 | - | - | Completed | - | - |
| NCT01488266 | Major Depressive Disorder | Not Applicable | Unknown | March 2013 | Korea, Republic of ... more >> Korean Univ Ansan Hospital; Bucheon St.Mary Hospital; DonggukUniv Gyeongju Hospital; Catholic University of Korea St. Paul's Hospital Seoul, Korea, Republic of Taiwan Chang Gung Memorial Hospital; Kaohsiung Medical University Chung-ho Memorial Hospital Taipei, Taiwan Collapse << |
| NCT01352559 | Depression | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT00725101 | - | - | Completed | - | - |
| NCT00605917 | - | - | Completed | - | - |
| NCT02883660 | - | - | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00817375 | Depression Ad... more >>verse Reaction to Drug Collapse << | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Kangnam, Seoul, Korea, Republic of, 135-710 Contact: Samsung Medical Center Kim, MD.pHD 82-2-3410-3582 dohkwan.kim@samsung.com Collapse << |
| NCT01228357 | Depression De... more >>pressive Symptoms Collapse << | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Kangnam, Seoul, Korea, Republic of Contact: Doh Kwan Kim, MD.PhD 82-2-3410-3582 dohkwan.kim@samsung.com Contact: Woojae Myung, MD. 82-2-3410-6562 smbhealer@gmail.com Collapse << |
| NCT00225511 | Depression | Phase 3 | Completed | - | - |
| NCT01254305 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Alabama ... more >> Forest Investigative Site 010 Birmingham, Alabama, United States, 35216 United States, Arkansas Forest Investigative Site 002 Little Rock, Arkansas, United States, 72223 United States, California Forest Investigative Site 001 Cerritos, California, United States, 90703 United States, Florida Forest Investigative Site 014 Fort Myers, Florida, United States, 33912 Forest Investigative Site 006 Jacksonville, Florida, United States, 32216 Forest Investigative Site 017 Orange City, Florida, United States, 32763 Forest Investigative Site 005 Orlando, Florida, United States, 32806 Forest Investigative Site 012 Tampa, Florida, United States, 33613 United States, Georgia Forest Investigative Site 009 Atlanta, Georgia, United States, 30308 United States, Illinois Forest Investigative Site 016 Joliet, Illinois, United States, 60435 United States, Louisiana Forest Investigative Site 004 New Orleans, Louisiana, United States, 70122 United States, Massachusetts Forest Investigative Site 022 Boston, Massachusetts, United States, 02135 United States, New York Forest Investigative Site 011 Bronx, New York, United States, 10467 Forest Investigative Site 015 Cedarhurst, New York, United States, 11516 United States, Ohio Forest Investigative Site 003 Cincinnati, Ohio, United States, 45227 Forest Investigative Site 013 Dayton, Ohio, United States, 45417 United States, Rhode Island Forest Investigative Site 020 Lincoln, Rhode Island, United States, 02865 United States, Tennessee Forest Investigative Site 018 Memphis, Tennessee, United States, 38119 United States, Texas Forest Investigative Site 008 Dallas, Texas, United States, 75235 United States, Wisconsin Forest Investigative Site 007 Middleton, Wisconsin, United States, 53562 Collapse << |
| NCT01254305 | - | - | Completed | - | - |
| NCT03128021 | Major Depressive Disorder | Phase 4 | Recruiting | July 2022 | United States, Pennsylvania ... more >> University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Dana Williams, MS 412-246-5924 barvdm@upmc.edu Contact: Rachel Berta, BA 412-246-5685 goodra@upmc.edu Sub-Investigator: Meryl A Butters, Ph.D. Sub-Investigator: Tae Kim, Ph.D. Sub-Investigator: Dana Tudorascu, Ph.D. Principal Investigator: Howard Aizenstein, M.D., Ph.D. Principal Investigator: Carmen Andreescu, M.D. Collapse << |
| NCT01352572 | Depression | Not Applicable | Unknown | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01319825 | Migraine With Aura ... more >> Migraine Without Aura Chronic Migraine Collapse << | Phase 4 | Unknown | July 2012 | United States, California ... more >> California Medical Clinic for Headache Not yet recruiting Santa Monica, California, United States, 90404 Contact: Tatyana Neyman, MD 310-315-1456 tneyman@drkudrow.com Sub-Investigator: Alan Rapoport, MD Collapse << |
| NCT01914042 | Neuropathic Pain | Phase 2 | Recruiting | December 2019 | Israel ... more >> Rambam Health Care Campus Recruiting Haifa, Israel, 31096 Contact: Elon Eisenberg, MD 972 4 8542880 e_eisenberg@rambam.health.gov.il Principal Investigator: Elon Eisenberg, MD Collapse << |
| NCT00969709 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00606203 | Ischemic Stroke ... more >> Depression Collapse << | Not Applicable | Unknown | September 2011 | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Chiayi, Taiwan, 613 Contact: Jian-An Su, MD +886-5-3621000 ext 2313 jian.7715@gmail.com Principal Investigator: Shih-Young Chou, MD Principal Investigator: Ching-Shu Tsai, MD Collapse << |
| NCT01038323 | Fibromyalgia | Phase 4 | Completed | - | United States, Indiana ... more >> Clinical Research Center for Pain, 250 University Blvd Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01034462 | - | - | Completed | - | - |
| NCT00969709 | - | - | Completed | - | - |
| NCT01038323 | - | - | Completed | - | - |
| NCT00969150 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00793520 | - | - | Terminated | - | - |
| NCT00969150 | - | - | Completed | - | - |
| NCT02687165 | Post Treatment Lyme Syndrome (... more >>PTLS) Chronic Pain Collapse << | Not Applicable | Recruiting | February 2018 | United States, New York ... more >> Columbia University Medical Center Recruiting New York, New York, United States, 10032-0000 Contact: Phil Grebowiec, MD 646-774-8100 eb3048@cumc.coumbia.edu Collapse << |
| NCT01034462 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT02265367 | Tobacco Use Disorder | Not Applicable | Completed | - | United States, Minnesota ... more >> Clinical and Translational Sciences Institute Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT00098124 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT00757731 | Fibromyalgia Syndrome | Phase 3 | Completed | - | Czech Republic ... more >> Rheumatology Ambulance Pardubice, Czech Republic, 530 02 Finland Kuopion Oma Laakari Oy Kuopio, Finland, 70100 France Hopital Hotel Dieu Paris, France Germany KKSK KLINIKUM DER UNIVERSITAET ZU KOELN - Anaesthesiology and Intensive Care Koln, Germany, D50931 Italy Ospedale Luigi Sacco Milano, Italy, 20157 Norway Center For Clinical Studies Lillehamer, Norway, 2609 Portugal Hospital Egas Moniz Lisboa, Portugal, 1349-019 Romania Dr I CANTACUZINO CLINICAL HOSPITAL Bucharest, Romania, 020475 Spain Hospital de La Esperanza Barcelona, Spain, 08024 Sweden Gottfriesclinic Ab Molndal, Sweden, 43137 Collapse << |
| NCT01288807 | Fibromyalgia | Phase 4 | Completed | - | United States, California ... more >> UCSD Medical Center, La Jolla San Diego, California, United States, 92037 Collapse << |
| NCT01747044 | Fibromylagia | Phase 2 | Unknown | November 2014 | France ... more >> CHU de Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 04 73 75 11 95 placarin@chu-clermontferrand.fr Collapse << |
| NCT01377194 | - | - | Completed | - | - |
| NCT00797797 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT00436033 | Fibromyalgia Syndrome | Phase 3 | Completed | - | Czech Republic ... more >> Rheumatology Ambulance Pardubice, Czech Republic, 530 02 Denmark FREDERIKSBERG HOSPITAL - Clinic of Rheumatology Frederiksberg, Denmark, F2000 Finland Kuopion Oma Laakari Oy Kuopio, Finland, 70100 France Hopital Hotel Dieu Paris, France Germany KKSK KLINIKUM DER UNIVERSITAET ZU KOELN - Anaesthesiology and Intensive Care Koln, Germany, D50931 Italy Ospedale Luigi Sacco Milano, Italy, 20157 Norway Center For Clinical Studies Lillehamer, Norway, 2609 Poland "Nasz Lekarz" Torun, Poland Portugal Hospital Egas Moniz Lisboa, Portugal, 1349-019 Romania Dr I CANTACUZINO CLINICAL HOSPITAL Bucharest, Romania, 020475 Spain Hospital de La Esperanza Barcelona, Spain, 08024 Sweden Gottfriesclinic Ab Molndal, Sweden, 43137 United Kingdom KINGS COLLEGE HOSPITAL - Clinic Trial Unit Academic Department of Rheumatology London, United Kingdom, SE59RJ Collapse << |
| NCT00797797 | - | - | Completed | - | - |
| NCT00314249 | - | - | Completed | - | - |
| NCT02783430 | Depression | Phase 2 Phase 3 | Recruiting | September 2019 | France ... more >> University Hospital, Not yet recruiting Lille, France Contact: Magali Pierrat, MD Principal Investigator: Magali Pierrat, MD Centre Hospitalier de Valenciennes Recruiting Valenciennes, France Principal Investigator: Antoine Lemaire, MD Collapse << |
| NCT03569475 | Major Depressive Disorder | Phase 3 | Recruiting | October 21, 2021 | United States, Arkansas ... more >> Woodland International Research Group Recruiting Little Rock, Arkansas, United States, 72211 United States, California Care Access Research, Beverly Hills Recruiting Beverly Hills, California, United States, 90212 Behavioral Research Specialists, LLC Recruiting Glendale, California, United States, 91206 Excell Research, Inc Recruiting Oceanside, California, United States, 92056 Orange County Neuropsychiatric Research Center, LLC Not yet recruiting Orange, California, United States, 92868 Elite Clinical Trials, Inc. Not yet recruiting Wildomar, California, United States, 92595 United States, Georgia Atlanta Behavioral Research, LLC Not yet recruiting Atlanta, Georgia, United States, 30338 United States, Illinois Joliet Center Clinical Research Not yet recruiting Joliet, Illinois, United States, 60435 Capstone Clinical Research Recruiting Libertyville, Illinois, United States, 60048 AMR-Baber Research, Inc. Not yet recruiting Naperville, Illinois, United States, 60563 Southern Illinois University School of Medicine Not yet recruiting Springfield, Illinois, United States, 62702 United States, Kansas Psychiatric Associates Not yet recruiting Overland Park, Kansas, United States, 66211 United States, Maryland Kennedy Krieger Institute Not yet recruiting Baltimore, Maryland, United States, 21205 United States, Michigan Rochester Center for Behavioral Medicine Not yet recruiting Rochester Hills, Michigan, United States, 48307 United States, Missouri Millennium Center for Clinical Research Not yet recruiting Creve Coeur, Missouri, United States, 63141 United States, New York Finger Lakes Clinical Research Recruiting Rochester, New York, United States, 14618 United States, Ohio University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45219 The Ohio State University Department of Psychiatry Not yet recruiting Columbus, Ohio, United States, 43210 United States, Texas BioBehavioral Research of Austin Recruiting Austin, Texas, United States, 78759 United States, Utah Ericksen Research and Development Not yet recruiting Clinton, Utah, United States, 84015 Collapse << |
| NCT01026077 | - | - | Recruiting | December 2019 | United States, North Carolina ... more >> INC Research Recruiting Wilmington, North Carolina, United States, 28405 Contact 877-643-3010 pregnancyregistries@incresearch.com Collapse << |
| NCT01331109 | Primary Fibromyalgia | Phase 2 | Terminated | - | - |
| NCT01331109 | - | - | Terminated | - | - |
| NCT00757679 | Fibromyalgia Syndrome | Phase 2 | Completed | - | Switzerland ... more >> Clinical Pharmacology & Toxicology Multidisciplinary Pain Centre Geneve, Switzerland Collapse << |
| NCT01377194 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00618956 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT01393522 | Chronic Migraine | Not Applicable | Completed | - | United States, Missouri ... more >> Mercy Health Research St. Louis, Missouri, United States, 63141 United States, North Carolina Headache Wellness Center Greensboro, North Carolina, United States, 27405 Collapse << |
| NCT00314249 | Fibromyalgia | Phase 3 | Completed | - | - |
| NCT01289236 | Chronic Shoulder Pain | Phase 4 | Completed | - | United States, Florida ... more >> Delray Research Associates Delray Beach, Florida, United States, 33484 Collapse << |
| NCT01329406 | Osteoarthritis | Phase 4 | Unknown | September 2012 | United States, Massachusetts ... more >> Analgesic Solutions Recruiting Natick, Massachusetts, United States, 01760 Contact: Karen Cowles, RN, MPH 781-444-9605 ext 121 kcowles@analgesicsolutions.com Contact: Courtney Lincoln 781-444-9605 ext 119 clincoln@analgesicsolutions.com Principal Investigator: Stephen L. Wright, M.D. Sub-Investigator: Nathaniel P. Katz, M.D., M.S. Sub-Investigator: Eric Osgood, M.D. Collapse << |
| NCT01328002 | Primary Fibromyalgia Syndrome | Phase 2 | Terminated | - | - |
| NCT02431806 | Major Depressive Disorder | Phase 3 | Recruiting | December 21, 2018 | - |
| NCT01108731 | - | - | Completed | - | - |
| NCT02720198 | Major Depressive Disorder | Phase 3 | Completed | - | United States, Georgia ... more >> Institute for Advanced Medical Research Alpharetta, Georgia, United States, 30005 Collapse << |
| NCT00618956 | - | - | Completed | - | - |
| NCT01471379 | Irritable Bowel Syndrome | Phase 2 | Terminated(Due to recruitment ... more >>difficulties the study is terminated.) Collapse << | - | United States, North Carolina ... more >> UNC Center for Functional GI and Motility Disorders Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT01108731 | Fibromyalgia | Phase 2 Phase 3 | Completed | - | United States, New York ... more >> Pain and Fatigue Study Center - Beth Israel Medical Center New York, New York, United States, 10003 Collapse << |
| NCT02466958 | Major Depressive Disorder | Phase 4 | Active, not recruiting | December 2018 | United States, California ... more >> UCLA Semel Institute - Neuropsychiatric Institute (NPI) Los Angeles, California, United States, 90095 Collapse << |
| NCT03249311 | None (i.e. Healthy Volunteers) | Phase 4 | Recruiting | January 31, 2020 | Canada, Ontario ... more >> Institute of Mental Health Research, Royal Ottawa Mental Health Centre Recruiting Ottawa, Ontario, Canada, K1Z 7K4 Contact: Holly English, RN 613-722-6521 ext 6405 holly.english@theroyal.ca Contact: Wendy Fusee, RN 613-722-6521 ext 7828 wendy.fusee@theroyal.ca Principal Investigator: Pierre Blier, MD Collapse << |
| NCT01471379 | - | - | Terminated(Due to recruitment ... more >>difficulties the study is terminated.) Collapse << | - | - |
| NCT01337700 | Autism Spectrum Disorder ... more >> Asperger Syndrome Aspergers Syndrome Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Montefiore Medical Center, Albert Einstein College of Medicine Bronx, New York, United States, 10467 Collapse << |
| NCT01173055 | - | - | Completed | - | - |
| NCT01173055 | Fibromyalgia | Phase 4 | Completed | - | United States, Michigan ... more >> University of Michigan, Chronic Pain and Fatigue Research Center Ann Arbor, Michigan, United States, 48106 Collapse << |
| NCT01221740 | Back Pain | Phase 3 | Withdrawn(Company decided to d... more >>iscontinue) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Pain Trials Center Chestnut Hill, Massachusetts, United States, 02467 Collapse << |
| NCT01777581 | Radicular Pain Related to Lumb... more >>osacral Disc Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01418651 | Fibromyalgia | Phase 3 | Terminated(Lack of subjects) | August 2011 | United States, Arizona ... more >> Banner Sun Health Research Institute Sun City, Arizona, United States, 85351 Collapse << |
| NCT01777581 | - | - | Completed | - | - |
| NCT01294059 | Fibromyalgia | Phase 1 | Completed | - | United States, Florida ... more >> Center for Musculoskeletal Pain Research Gainesville, Florida, United States, 32610 Collapse << |
| NCT01288937 | Idiopathic Peripheral Neuropat... more >>hy Collapse << | Not Applicable | Terminated(Poor recruitment) | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT01014585 | - | - | Completed | - | - |
| NCT01225068 | - | - | Completed | - | - |
| NCT01014585 | Fibromyalgia | Phase 4 | Completed | - | - |
| NCT01304589 | Vestibulodynia ... more >> Vulvodynia Collapse << | Phase 3 | Completed | - | United States, Tennessee ... more >> Women's Health Specialists, PLLC Germantown, Tennessee, United States, 38138 Collapse << |
| NCT01125423 | Fibromyalgia | Phase 4 | Unknown | - | United States, New York ... more >> Upstate Clinical Research, LLC Recruiting Albany, New York, United States, 12205 Contact: Suzanne Alterman, RN 518-533-1500 salterman@upstateneuro.com Principal Investigator: James Wymer, MD Collapse << |
| NCT01077375 | - | - | Completed | - | - |
| NCT01225068 | Low Back Pain | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT01510457 | Knee Osteoarthritis ... more >> Degenerative Joint Disease Chronic Pain Collapse << | Phase 4 | Completed | - | - |
| NCT01780389 | Knee Pain After Total Knee Art... more >>hroplasty Osteoarthritis Pain Collapse << | Phase 4 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT01077375 | Fibromyalgia | Phase 4 | Completed | - | - |
| NCT01780389 | - | - | Completed | - | - |
| NCT01225991 | - | - | Completed | - | - |
| NCT01829243 | Fibromyalgia ... more >>Neurocognition Collapse << | Phase 3 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center / Civitan Building Durham, North Carolina, United States, 27705 Collapse << |
| NCT01510457 | - | - | Completed | - | - |
| NCT01829243 | - | - | Completed | - | - |
| NCT01225991 | Rheumatoid Arthritis | Phase 4 | Completed | - | United States, California ... more >> UCLA Semel Institute Los Angeles, California, United States, 90095 Collapse << |
| NCT02288325 | Depressive Disorder, Major | Phase 4 | Completed | - | - |
| NCT02288325 | - | - | Completed | - | - |
| NCT01304589 | - | - | Completed | - | - |
| NCT01234675 | Sleep Disorders ... more >> Fibromyalgia Sleep Collapse << | Phase 4 | Completed | - | United States, Ohio ... more >> Cleveland Sleep Research Center Middleburg Heights, Ohio, United States, 44130 Collapse << |
| NCT01359826 | Systemic Lupus Erythematosus ... more >> Widespread Pain Fibromyalgia Collapse << | Phase 4 | Withdrawn(PI has let the insti... more >>tution. We are unable to locate any study documents that would indicate study enrollment) Collapse << | - | United States, California ... more >> Loma Linda University Medical Center Loma Linda, California, United States, 92354 Collapse << |
| NCT01207453 | - | - | Completed | - | - |
| NCT01207453 | Arthritis, Rheumatoid | Phase 4 | Completed | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01234675 | - | - | Completed | - | - |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
