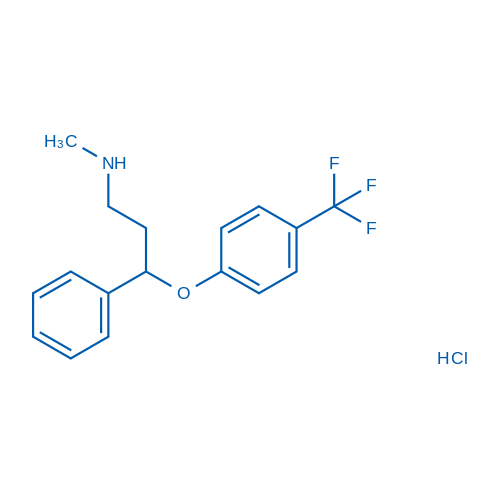
CAS No.: 56296-78-7
盐酸氟西汀 Catalog No. CSN12817
Synonyms: Lilly110140;LY-110140;Fluoxetine (hydrochloride) (CRM)
Fluoxetine HCl, an antidepressant, is an inhibitor of the selective serotonin reuptake with Kd of 0.81 nM against serotonin transporter.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02837224 - - Completed - - NCT02152670 Cocaine Dependence Not Applicable Recruiting May 2021 United States, Connecticut ... more >> Connecticut Mental Health Center Recruiting New Haven, Connecticut, United States, 06519 Contact: Edward Gaiser 203-974-7584 edward.gaiser@yale.edu Principal Investigator: Robert Malison, MD Collapse << NCT02331407 Stroke Not Applicable Completed - United States, New York ... more >> Columbia Univeristy, Neurological Institute New York, New York, United States, 10032 Collapse << - 更多
- 参考文献
- [1] Kodama M, Fujioka T, Duman RS. Chronic olanzapine or fluoxetine administration increases cell proliferation in hippocampus and prefrontal cortex of adult rat. Biol Psychiatry. 2004 Oct 15;56(8):570-80. Erratum in: Biol Psychiatry. 2005 Jan 15;57(2):199.
- [2] Malberg JE, Duman RS. Cell proliferation in adult hippocampus is decreased by inescapable stress: reversal by fluoxetine treatment. Neuropsychopharmacology. 2003 Sep;28(9):1562-71.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 56296-78-7 | 储存条件 |
|
|||||
| 分子式 | C17H19ClF3NO | 运输 | 蓝冰 | |||||
| 分子量 | 345.79 | 别名 | Lilly110140;LY-110140;Fluoxetine (hydrochloride) (CRM);Prozac;Sarafem;Animex-On;Fluoxetin;Pulvules;Eufor;Portal;盐酸氟西汀 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02837224 | - | - | Completed | - | - |
| NCT02152670 | Cocaine Dependence | Not Applicable | Recruiting | May 2021 | United States, Connecticut ... more >> Connecticut Mental Health Center Recruiting New Haven, Connecticut, United States, 06519 Contact: Edward Gaiser 203-974-7584 edward.gaiser@yale.edu Principal Investigator: Robert Malison, MD Collapse << |
| NCT02331407 | Stroke | Not Applicable | Completed | - | United States, New York ... more >> Columbia Univeristy, Neurological Institute New York, New York, United States, 10032 Collapse << |
| NCT02482025 | Drug-Related Side Effects and ... more >>Adverse Reactions Adverse Drug Events Collapse << | Not Applicable | Not yet recruiting | December 31, 2019 | United States, Massachusetts ... more >> VA Boston Healthcare System Jamaica Plain Campus, Jamaica Plain, MA Active, not recruiting Boston, Massachusetts, United States, 02130 Collapse << |
| NCT00456820 | Depression Si... more >>de Effects Sexual Dysfunction Apathy Collapse << | Phase 4 | Completed | - | United States, Indiana ... more >> Indiana University Adult Psychiatry Clinic Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01237275 | Depression Co... more >>ntinuous Antidepressant Abuse Adverse Reaction to Drug Collapse << | Not Applicable | Unknown | December 2016 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of Collapse << |
| NCT00284791 | Adolescent Depression | Not Applicable | Terminated | - | United States, Maine ... more >> Maine Medical Center Outpatient Psychiatry Portland, Maine, United States, 04102 Collapse << |
| NCT01017432 | - | - | Completed | - | United States, Michigan ... more >> William Beaumont Hospital Royal Oak, Michigan, United States, 48073 William Beaumont Hospital Troy, Michigan, United States, 48314 Collapse << |
| NCT00296426 | Medication Reconciliation | Not Applicable | Unknown | August 2010 | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01742832 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60615 Collapse << |
| NCT01742832 | - | - | Completed | - | - |
| NCT00018200 | Back Pain Sci... more >>atica Collapse << | Phase 2 | Completed | - | United States, California ... more >> VA San Diego Healthcare System, San Diego San Diego, California, United States, 92161 Collapse << |
| NCT00018902 | Depression | Phase 2 Phase 3 | Completed | - | United States, California ... more >> University of California at Los Angeles Los Angeles, California, United States, 90095-6967 United States, Oregon Kaiser Permanente Center for Health Research Portland, Oregon, United States, 97227-1098 United States, Pennsylvania Western Psychiatric Institute and Clinic of University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 United States, Rhode Island Brown University Providence, Rhode Island, United States, 02906 United States, Texas University of Texas - Southwestern Medical Center Dallas, Texas, United States, 75390-8589 University of Texas Medical Branch Galveston, Texas, United States, 77555-0188 Collapse << |
| NCT02427334 | Premenstrual Syndrome | Phase 3 | Recruiting | December 2017 | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Cairo university hospitals Recruiting Cairo, Egypt Contact: AbdelGany Hassan, MRCOG, MD 002 01017801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT00676026 | Healthy | Not Applicable | Completed | - | United States, Connecticut ... more >> Yale University School of Medicine; Yale Program for Women's Reproductive Behavioral Health New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01635218 | Moderate Depression ... more >> Menopausal and Postmenopausal Disorders Collapse << | Phase 2 | Completed | - | Mexico ... more >> Hospital Juárez de México Mexico City, Mexico, 07760 Collapse << |
| NCT00676026 | - | - | Completed | - | - |
| NCT01635218 | - | - | Completed | - | - |
| NCT02189213 | Generalized Anxiety Disorder ... more >> Separation Anxiety Disorder Social Phobia Collapse << | Not Applicable | Active, not recruiting | October 2019 | United States, New York ... more >> NYSPI New York, New York, United States, 10032 Collapse << |
| NCT01687478 | Treatment Resistant Depression | Phase 3 | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baoding, China, 071000 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100088 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kunming, China, 650032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210029 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wuhan, China, 430022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710061 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xinxiang, China, 453002 Collapse << |
| NCT01687478 | - | - | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | - |
| NCT00043537 | Social Phobia | Phase 3 | Completed | - | United States, Maryland ... more >> Maryland Center for Anxiety Disorders University of Maryland, College Park, Maryland, United States, 20742 Collapse << |
| NCT00044616 | Bipolar Disorder ... more >> Depression Collapse << | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Depression Research Unit, Univ Penn Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00773942 | Chronic Illness ... more >> Adverse Effects Medication Errors Elderly Patients Collapse << | Not Applicable | Completed | - | United States, Illinois ... more >> University of Illinois at Chicago Chicago, Illinois, United States, 60612 United States, North Carolina Duke University Health System Durham, North Carolina, United States, 27705 United States, Texas Baylor Health Care System Dallas, Texas, United States, 75206 Collapse << |
| NCT00078754 | - | - | Completed | - | - |
| NCT00958568 | Treatment Resistant Depression | Phase 3 | Completed | - | - |
| NCT00958568 | - | - | Completed | - | - |
| NCT00849901 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01740726 | Major Depressive Disorder | Not Applicable | Terminated(Lack of success wit... more >>h recruitment) Collapse << | - | United States, Georgia ... more >> Emory Executive Park Atlanta, Georgia, United States, 30329 Collapse << |
| NCT00849901 | - | - | Completed | - | - |
| NCT00961961 | Bipolar Disorder | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 United States, Pennsylvania Depression Research Unit Philadelphia, Pennsylvania, United States, 19104-3309 Collapse << |
| NCT00078754 | Intermittent Explosive Disorde... more >>r Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> The University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00000428 | Fibromyalgia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Office of Raphael Kieval, MD Brockton, Massachusetts, United States, 02402 Office of Ronald J. Rapoport, MD Fall River, Massachusetts, United States, 02720 Office of Eileen Winston, MD Framingham, Massachusetts, United States, 01702 Office of Nicola Mogavero, MD Melrose, Massachusetts, United States, 02176 Office of Jacqueline Feuer, MD Needham, Massachusetts, United States, 02192 Newton-Wellesley Hospital Newton, Massachusetts, United States, 02162 Office of Sharon A. Stotsky, MD North Reading, Massachusetts, United States, 01864 Collapse << |
| NCT00611975 | Healthy | Phase 4 | Completed | - | - |
| NCT00006286 | Major Depressive Disorder ... more >> Depression Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Michigan Wayne State University Detroit, Michigan, United States, 48207 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-5581 United States, New York New York University Medical Center New York, New York, United States, 10016 New York State Psychiatric Institute New York, New York, United States, 10032 United States, North Carolina Behavioral Health Center Charlotte, North Carolina, United States, 28203 United States, Ohio Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Oregon University of Oregon Eugene, Oregon, United States, 97403 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Texas University of Texas Southwestern Medical Center at Dallas Dallas, Texas, United States, 75235 Collapse << |
| NCT01740726 | - | - | Terminated(Lack of success wit... more >>h recruitment) Collapse << | - | - |
| NCT01808612 | Major Depressive Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 471-8513 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 270-0014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 800-0226 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukushima, Japan, 961-0021 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 737-0143 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 065-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 651-0097 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 238-0042 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 616-8421 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 390-0303 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 700-0907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 586-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 339-0057 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tochigi, Japan, 321-0953 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Collapse << |
| NCT01808651 | Major Depressive Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 471-8513 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 270-0014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 800-0226 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukushima, Japan, 963-877 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 737-0143 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 065-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 651-0097 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 238-0042 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 616-8421 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 390-0303 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 700-0907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 586-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 339-0057 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tochigi, Japan, 321-0953 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Collapse << |
| NCT00533117 | Borderline Personality Disorde... more >>r Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT01808651 | - | - | Completed | - | - |
| NCT00612313 | Depression | Not Applicable | Completed | - | United States, Texas ... more >> Children's Medical Center of Dallas, Outpatient Psychiatry Clinic Dallas, Texas, United States, 75235 Collapse << |
| NCT01808612 | - | - | Completed | - | - |
| NCT00533117 | - | - | Completed | - | - |
| NCT00296725 | Major Depression ... more >> Dysthymia Depressive Disorder NOS Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Depression Evaluation Service, New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00612313 | - | - | Completed | - | - |
| NCT00633685 | Posttraumatic Stress Disorder,... more >> Combat-related Collapse << | Phase 4 | Unknown | August 2012 | United States, Texas ... more >> Carl R. Darnall Army Medical Center Recruiting Ft. Hood, Texas, United States, 76544-4752 Contact: Michael Adams, Ph.D. 254-286-7804 michael.adams@amedd.army.mil Principal Investigator: Michael Adams, Ph.D. Collapse << |
| NCT00834834 | - | - | Completed | - | - |
| NCT00285935 | Major Depressive Disorder | Phase 4 | Recruiting | February 2020 | United States, California ... more >> University of California San Francisco Recruiting San Francisco, California, United States, 94143-0984 Contact: Owen Wolkowitz, MD 415-476-7433 Owen.Wolkowitz@ucsf.edu Collapse << |
| NCT01714310 | - | - | Completed | - | - |
| NCT00834834 | Borderline Personality Disorde... more >>r Suicide Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT02357849 | Attenuated Psychosis Syndrome | Phase 4 | Recruiting | July 2019 | United States, New York ... more >> The Zucker Hillside Hospital Recruiting Glen Oaks, New York, United States, 11004 Contact: Christoph U Correll, MD 718-470-4812 ccorrell@nshs.edu Collapse << |
| NCT01714310 | Severe Mood Dysregulation | Phase 2 | Completed | - | United States, California ... more >> UCLA Semel Institute Los Angeles, California, United States, 90095 Collapse << |
| NCT02389712 | Bipolar Disorder | Phase 4 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00909155 | - | - | Completed | - | - |
| NCT00909155 | Major Depressive Disorder | Not Applicable | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison Psychiatry Department Madison, Wisconsin, United States, 53719 Collapse << |
| NCT02017535 | Adolescent Depression | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> University of Minnesota, Department of Psychiatry Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT02238977 | Major Depression ... more >> End Stage Renal Disease Collapse << | Phase 4 | Terminated(Study stopped due t... more >>o difficulty recruiting) Collapse << | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT01824433 | Major Depression | Phase 4 | Completed | - | China, Beijing ... more >> Beijing Anding Hospital Beijing, Beijing, China, 100088 Collapse << |
| NCT02431806 | Major Depressive Disorder | Phase 3 | Recruiting | December 21, 2018 | - |
| NCT00046020 | Depressive Disorder, Major ... more >> Recurrence Collapse << | Phase 4 | Completed | - | - |
| NCT02372799 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00108316 | Depression | Not Applicable | Withdrawn(Study PI resigned) | - | United States, Georgia ... more >> VA Medical Center, Augusta Augusta, Georgia, United States, 30904 Collapse << |
| NCT01615055 | Cognitive Dysfunction | Early Phase 1 | Withdrawn(Study withdrawn. No ... more >>participants enrolled.) Collapse << | October 2020 | United States, California ... more >> City of Hope Duarte, California, United States, 91010 UCLA Medical Center Los Angeles, California, United States, 90024 Collapse << |
| NCT02238977 | - | - | Terminated(Study stopped due t... more >>o difficulty recruiting) Collapse << | - | - |
| NCT00787111 | Autistic Disorder | Phase 3 | Terminated | - | United States, Arizona ... more >> Southwest Autism Research and Resource Centre Phoenix, Arizona, United States, 85006 United States, California University of California Davis Sacramento, California, United States, 95817 United States, Georgia Institute for Behavioral Medicine Smyrna, Georgia, United States, 30080 United States, Illinois University of Illinois Chicago, Illinois, United States, 60637-1448 AMR-Baber Research Inc. Naperville, Illinois, United States, 60563 United States, Massachusetts Harvard Medical School Medford, Massachusetts, United States, 02155 United States, Michigan Children's Hospital of Michigan Detroit, Michigan, United States, 48201 United States, New Jersey CRCNJ Voorhees, New Jersey, United States, 08043 United States, New York Long Island Jewish Hospital Bethpage, New York, United States, 11714 Mount Sinai School of Medicine New York, New York, United States, 10029 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27514 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Washington Seattle Children's Hosptial University of Washington Seattle, Washington, United States, 98105-0371 Collapse << |
| NCT01699490 | Major Depressive Disorder | Phase 4 | Unknown | July 2015 | Taiwan ... more >> Department of Psychiatry, National Cheng-Kung University Hospital Recruiting Tainan, Taiwan, 701 Contact: Po See Chen, M.D., Ph.D. +886-6-2353535 ext 5213 chenps@mail.ncku.edu.tw Collapse << |
| NCT00061113 | Alcohol-Related Disorders ... more >> Marijuana Abuse Substance-Related Disorders Collapse << | Phase 4 | Completed | - | United States, Colorado ... more >> University of Colorado Denver Aurora, Colorado, United States, 80045 1611 South Federal Blvd. Denver, Colorado, United States, 80219 Collapse << |
| NCT00118404 | Depression | Phase 3 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Western Psychiatric Institute and Clinic Pittsburgh, Pennsylvania, United States, 15213 United States, Texas University of Texas Southwestern Medical Center at Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT00115804 | - | - | Completed | - | - |
| NCT00149643 | Depressive Disorder, Major ... more >> Cannabis Abuse Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Department of Psychiatry Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT03728153 | Acute Stroke ... more >>Stroke, Ischemic Collapse << | Phase 2 | Not yet recruiting | October 31, 2020 | - |
| NCT00149643 | - | - | Completed | - | - |
| NCT00118404 | - | - | Completed | - | - |
| NCT00183339 | - | - | Completed | - | - |
| NCT00245635 | Body Dysmorphic Disorder | Phase 4 | Completed | - | United States, New York ... more >> Montefiore Medical Center, Albert Einstein College of Medicine Bronx, New York, United States, 10467 United States, Rhode Island Rhode Island Hospital Providence, Rhode Island, United States, 02903 Collapse << |
| NCT00183339 | Autistic Disorder | Phase 2 | Completed | - | United States, New York ... more >> Mount Sinai School of Medicine New York, New York, United States, 10029 United States, North Carolina University of North Carolina, Chapel Hill Chapel Hill, North Carolina, United States, 25714 Collapse << |
| NCT00578669 | Major Depressive Disorder ... more >> Nicotine Dependence Depression Collapse << | Phase 3 | Completed | - | United States, Rhode Island ... more >> Butler Hospital Providence, Rhode Island, United States, 02906 Collapse << |
| NCT00265291 | Major Depressive Disorder ... more >> Major Depression Unipolar Depression Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Center for Pharmacogenomics, University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00115804 | Juvenile Primary Fibromyalgia ... more >>Syndrome (JPFS) Fibromyalgia Collapse << | Phase 3 | Completed | - | United States, Ohio ... more >> Women's Health Research Program Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT00733525 | Eating Disorders | Not Applicable | Completed | - | United States, California ... more >> Stanford University Palo Alto, California, United States, 94305 United States, Minnesota University of Minnesota Minneapolis, MN Minneapolis, Minnesota, United States, 55455 United States, New York Cornell University; White Plains, NY White Plains, New York, United States, 10605 United States, North Dakota University of North Dakota / Neuropsychiatric Research Institute Fargo, North Dakota, United States, 58103 Collapse << |
| NCT01146548 | Multiple System Atrophy | Phase 2 | Completed | - | France ... more >> hospital center of Aix enProvence Aix en Provence, France Hospital Gabriel Montpied Clermont-Ferrand, France University Hospital Henri Mondor Creteil, France Hopital Dijon, France Hospital R Salengro Lille, France university hospital Dupuytren Limoges, France university hospital Timone Marseille, France University Hospital Montpellier, France Hospital Nantes, France hospital Pitié Salpêtrière Paris, France University Hospital La Miletrie Poitiers, France Hospital Pontchaillou Rennes, France civil hospital of Strasbourg Strasbourg, France University Hospital Toulouse, France, 31000 Collapse << |
| NCT00965562 | Premenstrual Syndrome | Not Applicable | Completed | - | United States, Connecticut ... more >> Yale School of Medicine New Haven, Connecticut, United States, 06510 Collapse << |
| NCT00245635 | - | - | Completed | - | - |
| NCT01671124 | - | - | Unknown | November 2013 | Taiwan ... more >> Taipei Mecical University Hospital Recruiting Taipei, Taiwan, 100 Contact: Yu-Ru Guo, master 886227361661 ext 6549 dinyty@hotmail.com Sub-Investigator: Chih-Chiang Chiu, Ph.D Sub-Investigator: Hsin-Chien Lee, Ph.D Principal Investigator: Shih-Yi Huang, Ph.D Collapse << |
| NCT00578669 | - | - | Completed | - | - |
| NCT00965562 | - | - | Completed | - | - |
| NCT01545843 | - | - | Completed | - | - |
| NCT01802437 | Adolescent Depression | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> University of Minnesota, Department of Psychiatry Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT00657163 | Ischemic Stroke ... more >> Motor Impairment Collapse << | Phase 2 | Completed | - | France ... more >> University Hospital Besançon, France, 25030 University Hospital René Dubos Cergy-Pontoise, France, 95303 University Hospital Dijon, France, 21000 University Hospital Grenoble, France, 38048 University Hospital Nantes, France, 44093 University Hospital Pitié Salpétrière Paris, France, 75651 University Hospital Sainte Anne Paris, France, 75674 University Hospital Rangueil Toulouse, France, 31052 University Hospital Purpan Toulouse, France, 31059 Collapse << |
| NCT02737930 | Acute Stroke ... more >>Visual Field Loss Collapse << | Phase 2 | Recruiting | June 2020 | United States, New York ... more >> Strong Memorial Hospital Recruiting Rochester, New York, United States, 14642 Contact: Bogachan Sahin, MD, PhD 585-275-2530 bogachan_sahin@urmc.rochester.edu Contact: Ann M Leonhardt, NP 585-275-2530 ann_leonhardt@urmc.rochester.edu Collapse << |
| NCT03390933 | Depression He... more >>modialysis-Induced Symptom Collapse << | Phase 4 | Recruiting | November 30, 2023 | United States, Ohio ... more >> MetroHealth Medical Center Recruiting Cleveland, Ohio, United States, 44109 Contact: Jacqueline Dolata, MBA 216-778-1792 jdolata@metrohealth.org Collapse << |
| NCT01545843 | Depression | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02916238 | Major Depression | Phase 4 | Completed | - | Haiti ... more >> GHESKIO Centers Port-au-Prince, Haiti Collapse << |
| NCT03436173 | Depression | Not Applicable | Completed | - | - |
| NCT00006204 | Alcoholism Al... more >>cohol Dependence Depression Collapse << | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Department of Psychiatry, Western Psychiatric Institute and Clinic of the University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT03228732 | Type 1 Diabetes Mellitus | Early Phase 1 | Recruiting | December 10, 2020 | United States, Maryland ... more >> University of Maryland Recruiting Baltimore, Maryland, United States, 21201 Contact: Maka S Hedrington, MD 410-706-5623 mhedrington@som.umaryland.edu Collapse << |
| NCT02473250 | Bipolar Disorder | Phase 4 | Recruiting | September 2019 | United States, New York ... more >> New York State Psychiatric Institute/Columbia University Medical Center Recruiting New York, New York, United States, 10032 Contact: Martin Lan, MD PHD 646-774-7610 martin.lan@NYSPI.COLUMBIA.EDU Collapse << |
| NCT02929667 | Epilepsy SUDE... more >>P Collapse << | Phase 2 | Recruiting | April 2019 | United States, Iowa ... more >> The Univeristy of Iowa Hospitals and Clinics Not yet recruiting Iowa City, Iowa, United States, 52242 Contact: Deidre Dragon 319-356-4337 deidre-nitschke-dragon@uiowa.edu Contact: Rup K Sainju, MBBS 319-353-7767 rup-sainju@uiowa.edu Principal Investigator: Rup K Sainju, MBBS Sub-Investigator: Brian K Gehlbach, MD Sub-Investigator: Mark Granner, MD Sub-Investigator: George B Richerson, MD, PhD Univeristy of Iowa Hospitals and Clinics Recruiting Iowa City, Iowa, United States, 52242 Contact: Deidre Dragon 319-356-4337 deidre-nitschke-dragon@uiowa.edu Contact: Rup K Sainju, MBBS 319-353-7767 rup-sainju@uiowa.edu Principal Investigator: Rup K Sainju, MBBS Collapse << |
| NCT00027378 | Alcoholism De... more >>pression Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Western Psychiatric Institute and Clinic Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT03476525 | - | - | Recruiting | October 2026 | Norway ... more >> St. Olavs University Hospital Recruiting Trondheim, Norway Contact: Magnus Strømmen, MSc Collapse << |
| NCT00592852 | Pediatric Bipolar Disorder ... more >> Pediatric OCD Collapse << | Phase 4 | Terminated(Slow subject recrui... more >>tment.) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Cambridge, Massachusetts, United States, 02138 Collapse << |
| NCT00592852 | - | - | Terminated(Slow subject recrui... more >>tment.) Collapse << | - | - |
| NCT00027378 | - | - | Completed | - | - |
| NCT01569126 | - | - | Completed | - | - |
| NCT01569126 | Healthy Volunteer | Phase 1 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 532-0003 Collapse << |
| NCT00592670 | Type 1 Diabetes | Not Applicable | Completed | - | United States, Tennessee ... more >> Vanderbilt University Nashville, Tennessee, United States, 37232-0475 Collapse << |
| NCT00678574 | Premenstrual Dysphoric Disorde... more >>r Premenstrual Syndrome Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale University New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01361217 | - | - | Completed | - | - |
| NCT00011765 | Domestic Violence | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00603018 | Anorexia Nervosa | Not Applicable | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00678574 | - | - | Completed | - | - |
| NCT00986310 | Uncontrolled Partial Epilepsy ... more >> Ictal Hypoventilation Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California, Davis Sacramento, California, United States, 95817 Collapse << |
| NCT01361217 | Drug-Drug Interaction ... more >> Healthy Volunteers Collapse << | Not Applicable | Completed | - | - |
| NCT02723903 | Syndrome X | Phase 1 Phase 2 | Unknown | December 2017 | China ... more >> Renji Hospital Recruiting Shanghai, China Contact: Wang lingling, Postgraduate 15000053961 Contact: Chen bingxu, Postgraduate 15800539513 Collapse << |
| NCT02965274 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT00603018 | - | - | Completed | - | - |
| NCT02965261 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT00573547 | Depression | Phase 4 | Withdrawn(Principal Investigat... more >>or decided not to initiate the study.) Collapse << | - | United States, Nebraska ... more >> Creighton Department of Psychiatry Omaha, Nebraska, United States, 68131 Creighton University Department of Psychiatry Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00649636 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT01672255 | Type 1 Diabetes ... more >> Hypoglycemia Associated Autonomic Failure Collapse << | Early Phase 1 | Recruiting | December 2018 | United States, Maryland ... more >> University of Maryland, Baltimore Recruiting Baltimore, Maryland, United States, 21201 Contact: Donna Tate 410-706-5643 Principal Investigator: Stephen N. Davis, MBBS Collapse << |
| NCT00913718 | Depression | Phase 1 | Completed | - | - |
| NCT03448159 | Stroke Cerebr... more >>ovascular Accident Cerebral Infarction Brain Infarction Brain Ischemia Cerebrovascular Disorders Brain Diseases Central Nervous System Diseases Nervous System Diseases Vascular Diseases Collapse << | Phase 2 | Not yet recruiting | December 31, 2020 | Canada, Alberta ... more >> University of Calgary Not yet recruiting Calgary, Alberta, Canada, T2N 1N4 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Sean Dukelow, MD Canada, British Columbia University of British Columbia & GF Strong Centre Not yet recruiting Vancouver, British Columbia, Canada, V5Z 2G9 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Janice Eng, PhD Canada, Newfoundland and Labrador Memorial University of Newfoundland Not yet recruiting St. John's, Newfoundland and Labrador, Canada, A1B 3X9 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Michelle Ploughman, PhD Canada, Nova Scotia Dalhousie University Not yet recruiting Halifax, Nova Scotia, Canada, B3H 4R2 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Marilyn Mackay-Lyons, PhD Canada, Ontario Parkwood Institute Not yet recruiting London, Ontario, Canada, N6C 0A7 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Robert Teasell, MD Sunnybrook Health Sciences Centre Not yet recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Sandra Black, MD Toronto Rehabilitation Institute - University Health Network Not yet recruiting Toronto, Ontario, Canada, M5G 2A2 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Mark Bayley, MD University of Waterloo Not yet recruiting Waterloo, Ontario, Canada, N2L 3G1 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: William McIlroy, PhD Sub-Investigator: Robyn Ibey, PhD Collapse << |
| NCT00913588 | Depression | Phase 1 | Completed | - | - |
| NCT00427128 | Major Depression | Phase 4 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, New York New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT01247285 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT00947076 | Depression | Phase 1 | Completed | - | - |
| NCT00778024 | Healthy | Not Applicable | Completed | - | United States, Missouri ... more >> Gateway medical research St. Charles, Missouri, United States, 63301 Collapse << |
| NCT01247272 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT01247285 | - | - | Completed | - | - |
| NCT01247272 | - | - | Completed | - | - |
| NCT01166087 | Healthy | Phase 1 | Completed | - | India ... more >> Lotus Labs Private Limited Mylapore, Chennai, India, 600 004 Collapse << |
| NCT01166100 | Healthy | Phase 1 | Completed | - | India ... more >> Lotus Labs Private Limited Mylapore, Chennai, India, 600 004 Collapse << |
| NCT02482025 | Drug-Related Side Effects and ... more >>Adverse Reactions Adverse Drug Events Collapse << | Not Applicable | Not yet recruiting | December 31, 2019 | United States, Massachusetts ... more >> VA Boston Healthcare System Jamaica Plain Campus, Jamaica Plain, MA Active, not recruiting Boston, Massachusetts, United States, 02130 Collapse << |
| NCT02837224 | - | - | Completed | - | - |
| NCT00456820 | Depression Si... more >>de Effects Sexual Dysfunction Apathy Collapse << | Phase 4 | Completed | - | United States, Indiana ... more >> Indiana University Adult Psychiatry Clinic Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00296426 | Medication Reconciliation | Not Applicable | Unknown | August 2010 | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT02331407 | Stroke | Not Applicable | Completed | - | United States, New York ... more >> Columbia Univeristy, Neurological Institute New York, New York, United States, 10032 Collapse << |
| NCT01742832 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60615 Collapse << |
| NCT00284791 | Adolescent Depression | Not Applicable | Terminated | - | United States, Maine ... more >> Maine Medical Center Outpatient Psychiatry Portland, Maine, United States, 04102 Collapse << |
| NCT02152670 | Cocaine Dependence | Not Applicable | Recruiting | May 2021 | United States, Connecticut ... more >> Connecticut Mental Health Center Recruiting New Haven, Connecticut, United States, 06519 Contact: Edward Gaiser 203-974-7584 edward.gaiser@yale.edu Principal Investigator: Robert Malison, MD Collapse << |
| NCT00018200 | Back Pain Sci... more >>atica Collapse << | Phase 2 | Completed | - | United States, California ... more >> VA San Diego Healthcare System, San Diego San Diego, California, United States, 92161 Collapse << |
| NCT00018902 | Depression | Phase 2 Phase 3 | Completed | - | United States, California ... more >> University of California at Los Angeles Los Angeles, California, United States, 90095-6967 United States, Oregon Kaiser Permanente Center for Health Research Portland, Oregon, United States, 97227-1098 United States, Pennsylvania Western Psychiatric Institute and Clinic of University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 United States, Rhode Island Brown University Providence, Rhode Island, United States, 02906 United States, Texas University of Texas - Southwestern Medical Center Dallas, Texas, United States, 75390-8589 University of Texas Medical Branch Galveston, Texas, United States, 77555-0188 Collapse << |
| NCT02427334 | Premenstrual Syndrome | Phase 3 | Recruiting | December 2017 | Egypt ... more >> BeniSuef University hospitals Recruiting BeniSuef, Egypt Contact: Nesreen AA Shehata, MD +2001227866337 nesoomar@yahoo.com Cairo university hospitals Recruiting Cairo, Egypt Contact: AbdelGany Hassan, MRCOG, MD 002 01017801604 abdelgany2@gmail.com Principal Investigator: AbdelGany MA Hassan, MRCOG, MD Collapse << |
| NCT01742832 | - | - | Completed | - | - |
| NCT01017432 | - | - | Completed | - | United States, Michigan ... more >> William Beaumont Hospital Royal Oak, Michigan, United States, 48073 William Beaumont Hospital Troy, Michigan, United States, 48314 Collapse << |
| NCT01237275 | Depression Co... more >>ntinuous Antidepressant Abuse Adverse Reaction to Drug Collapse << | Not Applicable | Unknown | December 2016 | Korea, Republic of ... more >> Samsung Medical Center Kangnam, Seoul, Korea, Republic of Collapse << |
| NCT00676026 | - | - | Completed | - | - |
| NCT01635218 | - | - | Completed | - | - |
| NCT01635218 | Moderate Depression ... more >> Menopausal and Postmenopausal Disorders Collapse << | Phase 2 | Completed | - | Mexico ... more >> Hospital Juárez de México Mexico City, Mexico, 07760 Collapse << |
| NCT02189213 | Generalized Anxiety Disorder ... more >> Separation Anxiety Disorder Social Phobia Collapse << | Not Applicable | Active, not recruiting | October 2019 | United States, New York ... more >> NYSPI New York, New York, United States, 10032 Collapse << |
| NCT00676026 | Healthy | Not Applicable | Completed | - | United States, Connecticut ... more >> Yale University School of Medicine; Yale Program for Women's Reproductive Behavioral Health New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01808651 | Major Depressive Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 471-8513 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 270-0014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 800-0226 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukushima, Japan, 963-877 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 737-0143 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 065-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 651-0097 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 238-0042 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 616-8421 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 390-0303 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 700-0907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 586-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 339-0057 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tochigi, Japan, 321-0953 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Collapse << |
| NCT01808612 | Major Depressive Disorder | Phase 3 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aichi, Japan, 471-8513 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 270-0014 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukuoka, Japan, 800-0226 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Fukushima, Japan, 961-0021 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 737-0143 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hokkaido, Japan, 065-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hyogo, Japan, 651-0097 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 238-0042 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kyoto, Japan, 616-8421 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nagano, Japan, 390-0303 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Okayama, Japan, 700-0907 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 586-0012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Saitama, Japan, 339-0057 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tochigi, Japan, 321-0953 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Collapse << |
| NCT01808612 | - | - | Completed | - | - |
| NCT00849901 | - | - | Completed | - | - |
| NCT01808651 | - | - | Completed | - | - |
| NCT01687478 | Treatment Resistant Depression | Phase 3 | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baoding, China, 071000 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100088 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410011 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510080 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310009 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kunming, China, 650032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210029 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wuhan, China, 430022 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710061 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xinxiang, China, 453002 Collapse << |
| NCT00044616 | Bipolar Disorder ... more >> Depression Collapse << | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Depression Research Unit, Univ Penn Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01687478 | - | - | Terminated(Interim assessment ... more >>Lack of efficacy) Collapse << | - | - |
| NCT00043537 | Social Phobia | Phase 3 | Completed | - | United States, Maryland ... more >> Maryland Center for Anxiety Disorders University of Maryland, College Park, Maryland, United States, 20742 Collapse << |
| NCT00849901 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00773942 | Chronic Illness ... more >> Adverse Effects Medication Errors Elderly Patients Collapse << | Not Applicable | Completed | - | United States, Illinois ... more >> University of Illinois at Chicago Chicago, Illinois, United States, 60612 United States, North Carolina Duke University Health System Durham, North Carolina, United States, 27705 United States, Texas Baylor Health Care System Dallas, Texas, United States, 75206 Collapse << |
| NCT00958568 | Treatment Resistant Depression | Phase 3 | Completed | - | - |
| NCT00078754 | Intermittent Explosive Disorde... more >>r Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> The University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT01740726 | Major Depressive Disorder | Not Applicable | Terminated(Lack of success wit... more >>h recruitment) Collapse << | - | United States, Georgia ... more >> Emory Executive Park Atlanta, Georgia, United States, 30329 Collapse << |
| NCT00078754 | - | - | Completed | - | - |
| NCT01740726 | - | - | Terminated(Lack of success wit... more >>h recruitment) Collapse << | - | - |
| NCT00006286 | Major Depressive Disorder ... more >> Depression Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Michigan Wayne State University Detroit, Michigan, United States, 48207 United States, Nebraska University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-5581 United States, New York New York University Medical Center New York, New York, United States, 10016 New York State Psychiatric Institute New York, New York, United States, 10032 United States, North Carolina Behavioral Health Center Charlotte, North Carolina, United States, 28203 United States, Ohio Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 Case Western Reserve University Cleveland, Ohio, United States, 44106 United States, Oregon University of Oregon Eugene, Oregon, United States, 97403 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Texas University of Texas Southwestern Medical Center at Dallas Dallas, Texas, United States, 75235 Collapse << |
| NCT00961961 | Bipolar Disorder | Phase 4 | Completed | - | United States, Illinois ... more >> Rush University Medical Center Chicago, Illinois, United States, 60612 United States, Pennsylvania Depression Research Unit Philadelphia, Pennsylvania, United States, 19104-3309 Collapse << |
| NCT00611975 | Healthy | Phase 4 | Completed | - | - |
| NCT00285935 | Major Depressive Disorder | Phase 4 | Recruiting | February 2020 | United States, California ... more >> University of California San Francisco Recruiting San Francisco, California, United States, 94143-0984 Contact: Owen Wolkowitz, MD 415-476-7433 Owen.Wolkowitz@ucsf.edu Collapse << |
| NCT00909155 | Major Depressive Disorder | Not Applicable | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison Psychiatry Department Madison, Wisconsin, United States, 53719 Collapse << |
| NCT00296725 | Major Depression ... more >> Dysthymia Depressive Disorder NOS Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Depression Evaluation Service, New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00909155 | - | - | Completed | - | - |
| NCT00533117 | - | - | Completed | - | - |
| NCT00533117 | Borderline Personality Disorde... more >>r Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00612313 | Depression | Not Applicable | Completed | - | United States, Texas ... more >> Children's Medical Center of Dallas, Outpatient Psychiatry Clinic Dallas, Texas, United States, 75235 Collapse << |
| NCT00612313 | - | - | Completed | - | - |
| NCT01714310 | Severe Mood Dysregulation | Phase 2 | Completed | - | United States, California ... more >> UCLA Semel Institute Los Angeles, California, United States, 90095 Collapse << |
| NCT00834834 | Borderline Personality Disorde... more >>r Suicide Collapse << | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00000428 | Fibromyalgia | Phase 4 | Completed | - | United States, Massachusetts ... more >> Office of Raphael Kieval, MD Brockton, Massachusetts, United States, 02402 Office of Ronald J. Rapoport, MD Fall River, Massachusetts, United States, 02720 Office of Eileen Winston, MD Framingham, Massachusetts, United States, 01702 Office of Nicola Mogavero, MD Melrose, Massachusetts, United States, 02176 Office of Jacqueline Feuer, MD Needham, Massachusetts, United States, 02192 Newton-Wellesley Hospital Newton, Massachusetts, United States, 02162 Office of Sharon A. Stotsky, MD North Reading, Massachusetts, United States, 01864 Collapse << |
| NCT01714310 | - | - | Completed | - | - |
| NCT00834834 | - | - | Completed | - | - |
| NCT02357849 | Attenuated Psychosis Syndrome | Phase 4 | Recruiting | July 2019 | United States, New York ... more >> The Zucker Hillside Hospital Recruiting Glen Oaks, New York, United States, 11004 Contact: Christoph U Correll, MD 718-470-4812 ccorrell@nshs.edu Collapse << |
| NCT00633685 | Posttraumatic Stress Disorder,... more >> Combat-related Collapse << | Phase 4 | Unknown | August 2012 | United States, Texas ... more >> Carl R. Darnall Army Medical Center Recruiting Ft. Hood, Texas, United States, 76544-4752 Contact: Michael Adams, Ph.D. 254-286-7804 michael.adams@amedd.army.mil Principal Investigator: Michael Adams, Ph.D. Collapse << |
| NCT01824433 | Major Depression | Phase 4 | Completed | - | China, Beijing ... more >> Beijing Anding Hospital Beijing, Beijing, China, 100088 Collapse << |
| NCT02372799 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT02431806 | Major Depressive Disorder | Phase 3 | Recruiting | December 21, 2018 | - |
| NCT02389712 | Bipolar Disorder | Phase 4 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT02238977 | Major Depression ... more >> End Stage Renal Disease Collapse << | Phase 4 | Terminated(Study stopped due t... more >>o difficulty recruiting) Collapse << | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 Collapse << |
| NCT02238977 | - | - | Terminated(Study stopped due t... more >>o difficulty recruiting) Collapse << | - | - |
| NCT03728153 | Acute Stroke ... more >>Stroke, Ischemic Collapse << | Phase 2 | Not yet recruiting | October 31, 2020 | - |
| NCT00046020 | Depressive Disorder, Major ... more >> Recurrence Collapse << | Phase 4 | Completed | - | - |
| NCT00108316 | Depression | Not Applicable | Withdrawn(Study PI resigned) | - | United States, Georgia ... more >> VA Medical Center, Augusta Augusta, Georgia, United States, 30904 Collapse << |
| NCT01699490 | Major Depressive Disorder | Phase 4 | Unknown | July 2015 | Taiwan ... more >> Department of Psychiatry, National Cheng-Kung University Hospital Recruiting Tainan, Taiwan, 701 Contact: Po See Chen, M.D., Ph.D. +886-6-2353535 ext 5213 chenps@mail.ncku.edu.tw Collapse << |
| NCT00787111 | Autistic Disorder | Phase 3 | Terminated | - | United States, Arizona ... more >> Southwest Autism Research and Resource Centre Phoenix, Arizona, United States, 85006 United States, California University of California Davis Sacramento, California, United States, 95817 United States, Georgia Institute for Behavioral Medicine Smyrna, Georgia, United States, 30080 United States, Illinois University of Illinois Chicago, Illinois, United States, 60637-1448 AMR-Baber Research Inc. Naperville, Illinois, United States, 60563 United States, Massachusetts Harvard Medical School Medford, Massachusetts, United States, 02155 United States, Michigan Children's Hospital of Michigan Detroit, Michigan, United States, 48201 United States, New Jersey CRCNJ Voorhees, New Jersey, United States, 08043 United States, New York Long Island Jewish Hospital Bethpage, New York, United States, 11714 Mount Sinai School of Medicine New York, New York, United States, 10029 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27514 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Washington Seattle Children's Hosptial University of Washington Seattle, Washington, United States, 98105-0371 Collapse << |
| NCT00061113 | Alcohol-Related Disorders ... more >> Marijuana Abuse Substance-Related Disorders Collapse << | Phase 4 | Completed | - | United States, Colorado ... more >> University of Colorado Denver Aurora, Colorado, United States, 80045 1611 South Federal Blvd. Denver, Colorado, United States, 80219 Collapse << |
| NCT01615055 | Cognitive Dysfunction | Early Phase 1 | Withdrawn(Study withdrawn. No ... more >>participants enrolled.) Collapse << | October 2020 | United States, California ... more >> City of Hope Duarte, California, United States, 91010 UCLA Medical Center Los Angeles, California, United States, 90024 Collapse << |
| NCT00115804 | Juvenile Primary Fibromyalgia ... more >>Syndrome (JPFS) Fibromyalgia Collapse << | Phase 3 | Completed | - | United States, Ohio ... more >> Women's Health Research Program Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT01569126 | Healthy Volunteer | Phase 1 | Completed | - | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Osaka, Japan, 532-0003 Collapse << |
| NCT02017535 | Adolescent Depression | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> University of Minnesota, Department of Psychiatry Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT01569126 | - | - | Completed | - | - |
| NCT00149643 | - | - | Completed | - | - |
| NCT00118404 | Depression | Phase 3 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Western Psychiatric Institute and Clinic Pittsburgh, Pennsylvania, United States, 15213 United States, Texas University of Texas Southwestern Medical Center at Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT00958568 | - | - | Completed | - | - |
| NCT00118404 | - | - | Completed | - | - |
| NCT00183339 | Autistic Disorder | Phase 2 | Completed | - | United States, New York ... more >> Mount Sinai School of Medicine New York, New York, United States, 10029 United States, North Carolina University of North Carolina, Chapel Hill Chapel Hill, North Carolina, United States, 25714 Collapse << |
| NCT00149643 | Depressive Disorder, Major ... more >> Cannabis Abuse Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Department of Psychiatry Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00115804 | - | - | Completed | - | - |
| NCT00245635 | Body Dysmorphic Disorder | Phase 4 | Completed | - | United States, New York ... more >> Montefiore Medical Center, Albert Einstein College of Medicine Bronx, New York, United States, 10467 United States, Rhode Island Rhode Island Hospital Providence, Rhode Island, United States, 02903 Collapse << |
| NCT00183339 | - | - | Completed | - | - |
| NCT00245635 | - | - | Completed | - | - |
| NCT00265291 | Major Depressive Disorder ... more >> Major Depression Unipolar Depression Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Center for Pharmacogenomics, University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00578669 | Major Depressive Disorder ... more >> Nicotine Dependence Depression Collapse << | Phase 3 | Completed | - | United States, Rhode Island ... more >> Butler Hospital Providence, Rhode Island, United States, 02906 Collapse << |
| NCT00578669 | - | - | Completed | - | - |
| NCT00733525 | Eating Disorders | Not Applicable | Completed | - | United States, California ... more >> Stanford University Palo Alto, California, United States, 94305 United States, Minnesota University of Minnesota Minneapolis, MN Minneapolis, Minnesota, United States, 55455 United States, New York Cornell University; White Plains, NY White Plains, New York, United States, 10605 United States, North Dakota University of North Dakota / Neuropsychiatric Research Institute Fargo, North Dakota, United States, 58103 Collapse << |
| NCT01146548 | Multiple System Atrophy | Phase 2 | Completed | - | France ... more >> hospital center of Aix enProvence Aix en Provence, France Hospital Gabriel Montpied Clermont-Ferrand, France University Hospital Henri Mondor Creteil, France Hopital Dijon, France Hospital R Salengro Lille, France university hospital Dupuytren Limoges, France university hospital Timone Marseille, France University Hospital Montpellier, France Hospital Nantes, France hospital Pitié Salpêtrière Paris, France University Hospital La Miletrie Poitiers, France Hospital Pontchaillou Rennes, France civil hospital of Strasbourg Strasbourg, France University Hospital Toulouse, France, 31000 Collapse << |
| NCT00657163 | Ischemic Stroke ... more >> Motor Impairment Collapse << | Phase 2 | Completed | - | France ... more >> University Hospital Besançon, France, 25030 University Hospital René Dubos Cergy-Pontoise, France, 95303 University Hospital Dijon, France, 21000 University Hospital Grenoble, France, 38048 University Hospital Nantes, France, 44093 University Hospital Pitié Salpétrière Paris, France, 75651 University Hospital Sainte Anne Paris, France, 75674 University Hospital Rangueil Toulouse, France, 31052 University Hospital Purpan Toulouse, France, 31059 Collapse << |
| NCT01545843 | Depression | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01671124 | - | - | Unknown | November 2013 | Taiwan ... more >> Taipei Mecical University Hospital Recruiting Taipei, Taiwan, 100 Contact: Yu-Ru Guo, master 886227361661 ext 6549 dinyty@hotmail.com Sub-Investigator: Chih-Chiang Chiu, Ph.D Sub-Investigator: Hsin-Chien Lee, Ph.D Principal Investigator: Shih-Yi Huang, Ph.D Collapse << |
| NCT01802437 | Adolescent Depression | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> University of Minnesota, Department of Psychiatry Minneapolis, Minnesota, United States, 55454 Collapse << |
| NCT02737930 | Acute Stroke ... more >>Visual Field Loss Collapse << | Phase 2 | Recruiting | June 2020 | United States, New York ... more >> Strong Memorial Hospital Recruiting Rochester, New York, United States, 14642 Contact: Bogachan Sahin, MD, PhD 585-275-2530 bogachan_sahin@urmc.rochester.edu Contact: Ann M Leonhardt, NP 585-275-2530 ann_leonhardt@urmc.rochester.edu Collapse << |
| NCT02929667 | Epilepsy SUDE... more >>P Collapse << | Phase 2 | Recruiting | April 2019 | United States, Iowa ... more >> The Univeristy of Iowa Hospitals and Clinics Not yet recruiting Iowa City, Iowa, United States, 52242 Contact: Deidre Dragon 319-356-4337 deidre-nitschke-dragon@uiowa.edu Contact: Rup K Sainju, MBBS 319-353-7767 rup-sainju@uiowa.edu Principal Investigator: Rup K Sainju, MBBS Sub-Investigator: Brian K Gehlbach, MD Sub-Investigator: Mark Granner, MD Sub-Investigator: George B Richerson, MD, PhD Univeristy of Iowa Hospitals and Clinics Recruiting Iowa City, Iowa, United States, 52242 Contact: Deidre Dragon 319-356-4337 deidre-nitschke-dragon@uiowa.edu Contact: Rup K Sainju, MBBS 319-353-7767 rup-sainju@uiowa.edu Principal Investigator: Rup K Sainju, MBBS Collapse << |
| NCT01545843 | - | - | Completed | - | - |
| NCT03390933 | Depression He... more >>modialysis-Induced Symptom Collapse << | Phase 4 | Recruiting | November 30, 2023 | United States, Ohio ... more >> MetroHealth Medical Center Recruiting Cleveland, Ohio, United States, 44109 Contact: Jacqueline Dolata, MBA 216-778-1792 jdolata@metrohealth.org Collapse << |
| NCT03436173 | Depression | Not Applicable | Completed | - | - |
| NCT03476525 | - | - | Recruiting | October 2026 | Norway ... more >> St. Olavs University Hospital Recruiting Trondheim, Norway Contact: Magnus Strømmen, MSc Collapse << |
| NCT00006204 | Alcoholism Al... more >>cohol Dependence Depression Collapse << | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Department of Psychiatry, Western Psychiatric Institute and Clinic of the University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00027378 | Alcoholism De... more >>pression Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Western Psychiatric Institute and Clinic Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02916238 | Major Depression | Phase 4 | Completed | - | Haiti ... more >> GHESKIO Centers Port-au-Prince, Haiti Collapse << |
| NCT00027378 | - | - | Completed | - | - |
| NCT02473250 | Bipolar Disorder | Phase 4 | Recruiting | September 2019 | United States, New York ... more >> New York State Psychiatric Institute/Columbia University Medical Center Recruiting New York, New York, United States, 10032 Contact: Martin Lan, MD PHD 646-774-7610 martin.lan@NYSPI.COLUMBIA.EDU Collapse << |
| NCT00592670 | Type 1 Diabetes | Not Applicable | Completed | - | United States, Tennessee ... more >> Vanderbilt University Nashville, Tennessee, United States, 37232-0475 Collapse << |
| NCT00592852 | Pediatric Bipolar Disorder ... more >> Pediatric OCD Collapse << | Phase 4 | Terminated(Slow subject recrui... more >>tment.) Collapse << | - | United States, Massachusetts ... more >> Massachusetts General Hospital Cambridge, Massachusetts, United States, 02138 Collapse << |
| NCT03228732 | Type 1 Diabetes Mellitus | Early Phase 1 | Recruiting | December 10, 2020 | United States, Maryland ... more >> University of Maryland Recruiting Baltimore, Maryland, United States, 21201 Contact: Maka S Hedrington, MD 410-706-5623 mhedrington@som.umaryland.edu Collapse << |
| NCT01361217 | Drug-Drug Interaction ... more >> Healthy Volunteers Collapse << | Not Applicable | Completed | - | - |
| NCT00592852 | - | - | Terminated(Slow subject recrui... more >>tment.) Collapse << | - | - |
| NCT00678574 | Premenstrual Dysphoric Disorde... more >>r Premenstrual Syndrome Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> Yale University New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01361217 | - | - | Completed | - | - |
| NCT00603018 | Anorexia Nervosa | Not Applicable | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00965562 | - | - | Completed | - | - |
| NCT00965562 | Premenstrual Syndrome | Not Applicable | Completed | - | United States, Connecticut ... more >> Yale School of Medicine New Haven, Connecticut, United States, 06510 Collapse << |
| NCT00603018 | - | - | Completed | - | - |
| NCT00573547 | Depression | Phase 4 | Withdrawn(Principal Investigat... more >>or decided not to initiate the study.) Collapse << | - | United States, Nebraska ... more >> Creighton Department of Psychiatry Omaha, Nebraska, United States, 68131 Creighton University Department of Psychiatry Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00986310 | Uncontrolled Partial Epilepsy ... more >> Ictal Hypoventilation Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California, Davis Sacramento, California, United States, 95817 Collapse << |
| NCT01672255 | Type 1 Diabetes ... more >> Hypoglycemia Associated Autonomic Failure Collapse << | Early Phase 1 | Recruiting | December 2018 | United States, Maryland ... more >> University of Maryland, Baltimore Recruiting Baltimore, Maryland, United States, 21201 Contact: Donna Tate 410-706-5643 Principal Investigator: Stephen N. Davis, MBBS Collapse << |
| NCT00678574 | - | - | Completed | - | - |
| NCT02965274 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT02965261 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT03448159 | Stroke Cerebr... more >>ovascular Accident Cerebral Infarction Brain Infarction Brain Ischemia Cerebrovascular Disorders Brain Diseases Central Nervous System Diseases Nervous System Diseases Vascular Diseases Collapse << | Phase 2 | Not yet recruiting | December 31, 2020 | Canada, Alberta ... more >> University of Calgary Not yet recruiting Calgary, Alberta, Canada, T2N 1N4 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Sean Dukelow, MD Canada, British Columbia University of British Columbia & GF Strong Centre Not yet recruiting Vancouver, British Columbia, Canada, V5Z 2G9 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Janice Eng, PhD Canada, Newfoundland and Labrador Memorial University of Newfoundland Not yet recruiting St. John's, Newfoundland and Labrador, Canada, A1B 3X9 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Michelle Ploughman, PhD Canada, Nova Scotia Dalhousie University Not yet recruiting Halifax, Nova Scotia, Canada, B3H 4R2 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Marilyn Mackay-Lyons, PhD Canada, Ontario Parkwood Institute Not yet recruiting London, Ontario, Canada, N6C 0A7 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Robert Teasell, MD Sunnybrook Health Sciences Centre Not yet recruiting Toronto, Ontario, Canada, M4N 3M5 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Sandra Black, MD Toronto Rehabilitation Institute - University Health Network Not yet recruiting Toronto, Ontario, Canada, M5G 2A2 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: Mark Bayley, MD University of Waterloo Not yet recruiting Waterloo, Ontario, Canada, N2L 3G1 Contact: Farrell Leibovitch 416-480-6100 ext 7548 farrell@canadianstroke.ca Principal Investigator: William McIlroy, PhD Sub-Investigator: Robyn Ibey, PhD Collapse << |
| NCT00649636 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00947076 | Depression | Phase 1 | Completed | - | - |
| NCT00011765 | Domestic Violence | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00913588 | Depression | Phase 1 | Completed | - | - |
| NCT01247285 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT00427128 | Major Depression | Phase 4 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, New York New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00913718 | Depression | Phase 1 | Completed | - | - |
| NCT01247285 | - | - | Completed | - | - |
| NCT01166100 | Healthy | Phase 1 | Completed | - | India ... more >> Lotus Labs Private Limited Mylapore, Chennai, India, 600 004 Collapse << |
| NCT01166087 | Healthy | Phase 1 | Completed | - | India ... more >> Lotus Labs Private Limited Mylapore, Chennai, India, 600 004 Collapse << |
| NCT01247272 | - | - | Completed | - | - |
| NCT00778024 | Healthy | Not Applicable | Completed | - | United States, Missouri ... more >> Gateway medical research St. Charles, Missouri, United States, 63301 Collapse << |
| NCT01247272 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58102 Collapse << |
| NCT02723903 | Syndrome X | Phase 1 Phase 2 | Unknown | December 2017 | China ... more >> Renji Hospital Recruiting Shanghai, China Contact: Wang lingling, Postgraduate 15000053961 Contact: Chen bingxu, Postgraduate 15800539513 Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
