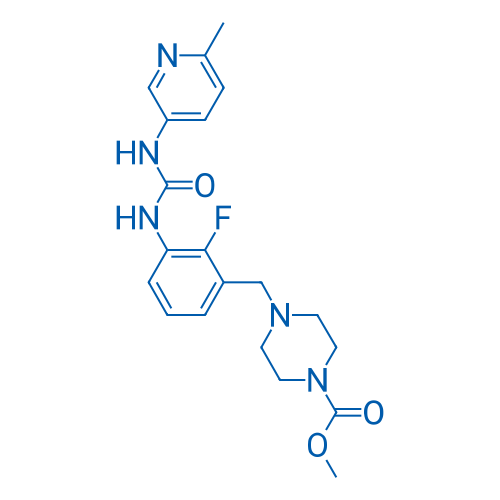
CAS No.: 873697-71-3
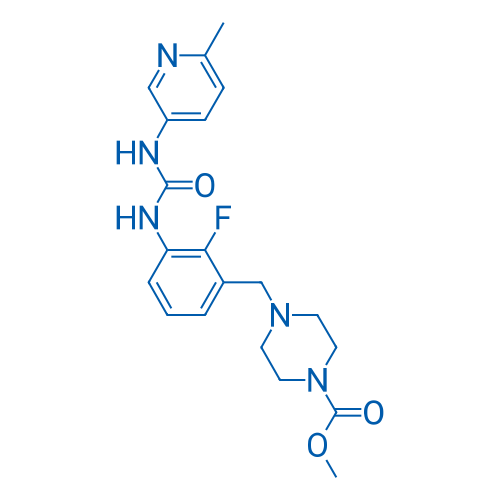
Omecamtiv mecarbil Catalog No. CSN16232
Synonyms: CK-1827452;Omecamtiv Mecarbil
Omecamtiv mecarbil is a selective sarcomere-directed cardiac myosin activator with an EC50 of 0.6 μM.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
动物研究 剂量 Rat: 1.2 mg/kg[3] (femoral vein perfusion)
给药途径 i.v.
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00682565 Heart Failure ... more >> Myocardial Ischemia Angina Pectoris Collapse << Phase 2 Completed - Georgia ... more >> Tbilisi State Medical University Clinic #1 Tbilisi, Georgia, 0102 Cardio-Reanimation Centre Tbilisi, Georgia, 0141 Cardiology Clinic Tbilisi, Georgia, 0144 National Center of Therapy Tbilisi, Georgia, 0159 Multiprofile Clinical Hospital of Tbilisi #2 Tbilisi, Georgia, 0164 Diagnostic Services Clinic Tbilisi, Georgia, 0179 Russian Federation Altay Territory Cardiology Dispensary Barnaul, Russian Federation City Hospital #1 Barnaul, Russian Federation City Clinical Hospital #59 Moscow, Russian Federation City Clinical Hospital #64 Moscow, Russian Federation Moscow Municipal Clinical Hospital #4 Moscow, Russian Federation Federal Center of Heart, Blood and Endocrinology n.a. Almazov St. Petersburg, Russian Federation Research Centre for Cardiology n.a. Almazov under Roszdrav St. Petersburg, Russian Federation Volgograd Regional Cardiology Center Volgograd, Russian Federation Collapse << NCT00682565 - - Completed - - NCT00624442 Heart Failure Phase 2 Completed - United States, California ... more >> University of California, San Diego Medical Center San Diego, California, United States, 92103 United States, Delaware Christiana Care Health Services, Inc. Newark, Delaware, United States, 19713 Georgia Diagnostic Services Clinic Tbilisi, Georgia Russian Federation Russian Cardiological Research and Production Complex Moscow, Russia, Russian Federation, 121552 Dzhanelidze Research Institute for Emergency Medical Care St. Petersburg, Russia, Russian Federation, 192242 Almazov Federal Heart, Blood and Endocrinology Center St. Petersburg, Russia, Russian Federation, 194156 St. Petersburg State Medical University St. Petersburg, Russia, Russian Federation, 197089 United Kingdom Castle Hill Hospital, University of Hull Hull, England, United Kingdom, HU16 5JQ King's College Hospital London, England, United Kingdom, SE5 9RS St. George's Hospital London, England, United Kingdom, SW17 ORE St. Mary's Hospital & Imperial College London, England, United Kingdom, W2 1LA Manchester Heart Centre, Manchester Royal Infirmary Manchester, England, United Kingdom, M13 9WL ICON Development Solutions Manchester, England, United Kingdom, M15 6SH Wythenshawe Hospital Manchester, England, United Kingdom, M23 9LT Northwick Park Hospital Middlesex, England, United Kingdom, HA1 3UJ Ninewells Hospital and Medical School Dundee, Scotland, United Kingdom, DD1 9SY BHF Cardiovascular Centre Glasgow, Scotland, United Kingdom, G12 8TA Collapse << - 更多
- 参考文献
- [1] Kaplinsky E, Mallarkey G, et al. Cardiac myosin activators for heart failure therapy: focus on omecamtiv mecarbil. Drugs Context. 2018 Apr 23;7:212518.
- [2] Morgan BP, Muci A, et al. Discovery of omecamtiv mecarbil the first, selective, small molecule activator of cardiac Myosin. ACS Med Chem Lett. 2010 Aug 20;1(9):472-7.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 873697-71-3 | 储存条件 |
|
|||||
| 分子式 | C20H24FN5O3 | 运输 | 蓝冰 | |||||
| 分子量 | 401.43 | 别名 | CK-1827452;Omecamtiv Mecarbil | |||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
