| NCT02530437 | Gastroesophageal Junction Aden... more >>ocarcinoma
Stage IB Esophageal Adenocarcinoma AJCC v7
Stage II Esophageal Adenocarcinoma AJCC v7
Stage IIA Esophageal Adenocarcinoma AJCC v7
Stage IIB Esophageal Adenocarcinoma AJCC v7
Stage IIIA Esophageal Adenocarcinoma AJCC v7
Stage IIIB Esophageal Adenocarcinoma AJCC v7 Collapse << | Phase 1
Phase 2 | Active, not recruiting | March 31, 2023 | United States, Texas
... more >>
M D Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT02784795 | Solid Tumor
B... more >>reast Cancer
Colon Cancer
Cholangiocarcinoma
Soft Tissue Sarcoma Collapse << | Phase 1 | Active, not recruiting | January 31, 2019 | United States, Florida
... more >>
Sylvester Comprehensive Cancer Center
Miami, Florida, United States, 33136
United States, Michigan
Karmanos Cancer Institute
Detroit, Michigan, United States, 48201
United States, New York
Memorial Sloan Kettering Cancer Center
New York, New York, United States, 10065
United States, Texas
University of Texas MD Anderson Cancer Center
Houston, Texas, United States, 77030
Denmark
Rigshospitalet
Copenhagen, København Ø, Denmark, 2100
France
Institut Bergonie
Bordeaux, France, 33076
Centre Leon Berard
Lyon Cedex 08, France, 69373
Gustave Roussy
Villejuif Cedex, France, 94805
Spain
Hospital Universitari Vall d'Hebron
Barcelona, Spain, 08035
Fundacion Jimenez Diaz
Madrid, Spain, 28040
Hospital Madrid Norte Sanchinarro
Madrid, Spain, 28050 Collapse << |
| NCT01681186 | Healthy Volunteers | Phase 1 | Completed | - | United States, Texas
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Dallas, Texas, United States Collapse << |
| NCT01746745 | Healthy Volunteers | Phase 1 | Completed | - | United States, Wisconsin
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Madison, Wisconsin, United States, 53704 Collapse << |
| NCT01226485 | Advanced Cancer | Phase 1 | Completed | - | United States, Arizona
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Scottsdale, Arizona, United States, 85260
United States, California
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Redwood City, California, United States, 94063
United States, Colorado
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Aurora, Colorado, United States, 80045
United States, Florida
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Fort Myers, Florida, United States, 33908
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Tampa, Florida, United States, 33612
United States, Nevada
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Las Vegas, Nevada, United States, 89169
United States, New York
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
New York, New York, United States, 10065
United States, Oklahoma
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Oklahoma City, Oklahoma, United States, 73104
United States, Tennessee
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Nashville, Tennessee, United States, 37203
United States, Texas
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Fort Worth, Texas, United States, 76177
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Houston, Texas, United States, 77030
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Tyler, Texas, United States, 75702 Collapse << |
| NCT01722292 | Small Cell Lung Carcinoma | Phase 1
Phase 2 | Terminated(Change in clinical ... more >>strategy.) Collapse << | - | United States, Arkansas
... more >>
Highlands Oncology Group
Fayetteville, Arkansas, United States, 72703
United States, Georgia
Northeast Georgia Cancer Care, LLC
Athens, Georgia, United States, 30607
United States, Nevada
Comprehensive Cancer Centers of Nevada
Las Vegas, Nevada, United States, 89169
United States, New York
New York Oncology Hematology Associate
Albany, New York, United States, 12206
Mount Sinai Medical Center
New York, New York, United States, 10029
United States, South Carolina
Clinical Research Unit (ITOR) Greenville Hospital System
Greenville, South Carolina, United States, 29605
United States, Tennessee
Accelerated Comm. Oncology Research Network (ACORN)
Memphis, Tennessee, United States, 38119
The West Clinic
Memphis, Tennessee, United States, 38120
United States, Texas
US Oncology
The Woodlands, Texas, United States, 77380
Tyler Cancer Center
Tyler, Texas, United States, 75702
United States, Washington
Northwest Cancer Specialists PC
Vancouver, Washington, United States, 98684
Yakima Valley Memorial Hospital
Yakima, Washington, United States, 98902
United Kingdom
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
London, United Kingdom, SE1 9RT
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Manchester, United Kingdom, M20 4BX Collapse << |
| NCT01697514 | Medulloblastoma, Childhood
... more >> Rhabdomyosarcoma Collapse << | Phase 1 | Withdrawn(Trial stopped early ... more >>for poor accrual.) Collapse << | July 2016 | United States, California
... more >>
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
San Diego, California, United States, 92123
United States, Colorado
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Aurora, Colorado, United States, 80045
United States, Ohio
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Cincinnati, Ohio, United States, 45229
United States, Oregon
For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician.
Portland, Oregon, United States, 97239 Collapse << |
| NCT01919398 | Neoplasm Metastasis | Phase 1 | Active, not recruiting | June 2017 | Japan
... more >> For additional information regarding investigative sites for this trial, contact 1-858-255-5959 Mon - Fri from 9 AM to 5 PM Pacific Time (PST), or speak with your personal physician.
Shizuoka, Japan, 411-8777
For additional information regarding investigative sites for this trial, contact 1-858-255-5959 Mon - Fri from 9 AM to 5 PM Pacific Time (PST), or speak with your personal physician.
Tokyo, Japan, 104-0045 Collapse << |
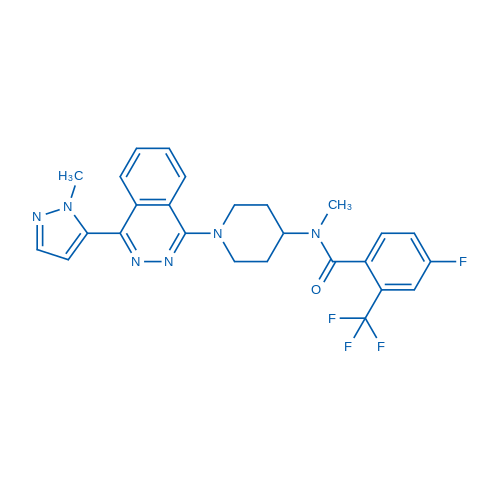
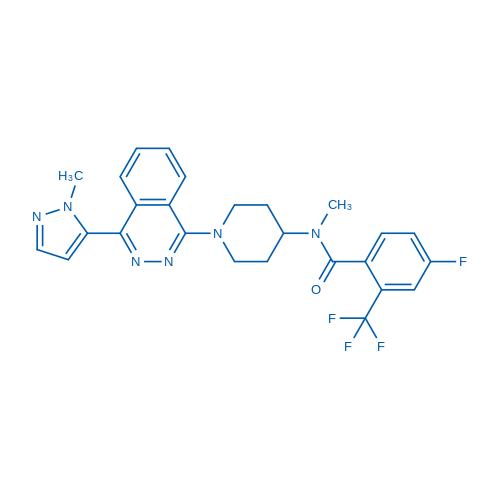

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
