| NCT03447249 | Cystic Fibrosis | Phase 3 | Active, not recruiting | April 16, 2019 | - |
| NCT03460990 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT03447262 | Cystic Fibrosis | Phase 3 | Enrolling by invitation | April 24, 2021 | - |
| NCT03227471 | Cystic Fibrosis | Phase 1
Phase 2 | Completed | - | - |
| NCT03525574 | Cystic Fibrosis | Phase 3 | Enrolling by invitation | June 2021 | - |
| NCT03224351 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT03525548 | Cystic Fibrosis | Phase 3 | Active, not recruiting | March 2019 | - |
| NCT03029455 | Cystic Fibrosis | Phase 1 | Completed | - | United Kingdom
... more >>
Birmingham, United Kingdom
Cambridge, United Kingdom
Exeter, United Kingdom
Glasgow, United Kingdom
Leeds, United Kingdom
Liverpool, United Kingdom
London, United Kingdom
Manchester, United Kingdom
Newcastle upon Tyne, United Kingdom
Southampton, United Kingdom Collapse << |
| NCT03537651 | Cystic Fibrosis | Phase 3 | Enrolling by invitation | December 2020 | - |
| NCT03633526 | Cystic Fibrosis | Phase 3 | Recruiting | January 2020 | United States, Colorado
... more >>
Children's Hospital Colorado
Recruiting
Aurora, Colorado, United States, 80045
United States, Illinois
Ann & Robert Lurie Children's Hospital of Chicago
Recruiting
Chicago, Illinois, United States, 60611
United States, Missouri
The Children's Mercy Hospital
Recruiting
Kansas City, Missouri, United States, 64108
United States, North Carolina
Clinical Research of Charlotte
Recruiting
Charlotte, North Carolina, United States, 28277
United States, Oregon
Oregon Health & Science University
Recruiting
Portland, Oregon, United States, 97239
United States, Texas
Texas Children's Hospital
Recruiting
Houston, Texas, United States, 77030 Collapse << |
| NCT03691779 | Cystic Fibrosis | Phase 3 | Recruiting | January 2020 | United States, California
... more >>
Children's Hospital of Orange County
Recruiting
Orange, California, United States, 92868
United States, Massachusetts
Boston Children's Hospital
Recruiting
Boston, Massachusetts, United States, 02115
United States, Minnesota
Children's Respiratory and Critical Care Specialists, P.A., Children's Hospitals and Clinics of Minnesota
Recruiting
Minneapolis, Minnesota, United States, 55404
United States, New York
Northwell Health- Long Island Jewish Medical Center
Recruiting
New Hyde Park, New York, United States, 11040
United States, Ohio
Rainbow Babies and Children's Hospital/University Hospitals Cleveland Medical Center
Recruiting
Cleveland, Ohio, United States, 44106
Nationwide Children's Hospital
Recruiting
Columbus, Ohio, United States, 43205 Collapse << |
| NCT03768089 | Cystic Fibrosis | Phase 1
Phase 2 | Recruiting | March 2019 | Netherlands
... more >> Academic Medical Center
Recruiting
Amsterdam, Netherlands
PRA Health Sciences Onderzoekscentrum UMCG
Recruiting
Groningen, Netherlands
United Kingdom
The Medicines Evaluation Unit
Recruiting
Manchester, United Kingdom Collapse << |
| NCT03525444 | Cystic Fibrosis | Phase 3 | Active, not recruiting | August 2019 | - |
| NCT02951182 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT03486236 | Cystic Fibrosis | Phase 1 | Completed | - | United Kingdom
... more >> The Medicines Evaluation Unit
Manchester, United Kingdom Collapse << |
| NCT02951195 | Cystic Fibrosis | Phase 2 | Completed | - | United States, Alabama
... more >>
Birmingham, Alabama, United States
United States, California
La Jolla, California, United States
Los Angeles, California, United States
United States, Florida
Orlando, Florida, United States
United States, Illinois
Chicago, Illinois, United States
Peoria, Illinois, United States
United States, Missouri
Saint Louis, Missouri, United States
United States, North Carolina
Chapel Hill, North Carolina, United States
United States, Ohio
Akron, Ohio, United States
Cleveland, Ohio, United States
United States, Oregon
Portland, Oregon, United States
United States, Pennsylvania
Philadelphia, Pennsylvania, United States
United States, South Carolina
Charleston, South Carolina, United States
United States, Texas
Fort Worth, Texas, United States
Houston, Texas, United States
United States, Wisconsin
Madison, Wisconsin, United States Collapse << |
| NCT02508207 | Cystic Fibrosis | Phase 2 | Completed | - | United States, Alabama
... more >>
Birmingham, Alabama, United States
United States, Indiana
Indianapolis, Indiana, United States
United States, Maryland
Baltimore, Maryland, United States
United States, North Carolina
Chapel Hill, North Carolina, United States
United States, Ohio
Cincinnati, Ohio, United States
Toledo, Ohio, United States
United States, Pennsylvania
Pittsburgh, Pennsylvania, United States Collapse << |
| NCT02508207 | - | - | Completed | - | - |
| NCT02412111 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT01531673 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT02070744 | - | - | Completed | - | - |
| NCT02516410 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02070744 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT02347657 | - | - | Completed | - | - |
| NCT02730208 | Cystic Fibrosis | Phase 2 | Completed | - | Australia
... more >>
Chermside, Australia
Melbourne, Australia
Nedlands, Australia
New Lambton Heights, Australia
Parkville SIC, Australia
Randwick, Australia
South Brisbane, Australia
Subiaco, Australia
Westmead, Australia Collapse << |
| NCT01531673 | - | - | Completed | - | - |
| NCT02015507 | Cystic Fibrosis | Phase 1 | Completed | - | - |
| NCT02347657 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02516410 | - | - | Completed | - | - |
| NCT02565914 | Cystic Fibrosis | Phase 3 | Enrolling by invitation | September 2019 | - |
| NCT02953314 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02392234 | - | - | Completed | - | - |
| NCT02392234 | Cystic Fibrosis | Phase 3 | Completed | - | - |
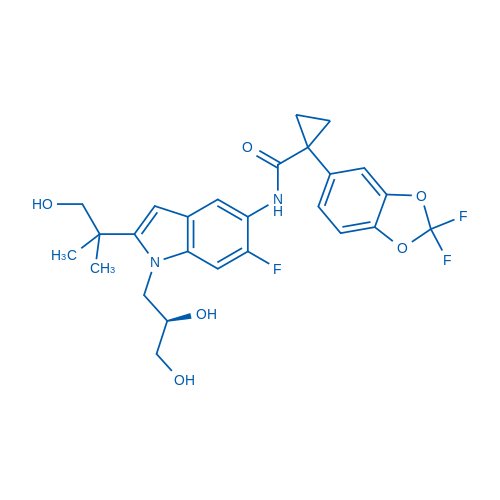

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
