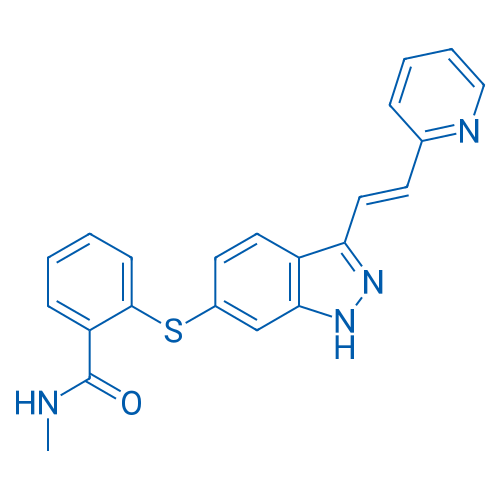
CAS No.: 319460-85-0
阿昔替尼 Catalog No. CSN10361
Synonyms: AG-013736;Brand name: Inlyta;阿昔替尼
Axitinib is a multi-target inhibitor of VEGFR1, VEGFR2, VEGFR3, PDGFRβ and c-Kit with IC50 of 0.1 nM, 0.2 nM, 0.1-0.3 nM, 1.6 nM and 1.7 nM in Porcine aorta endothelial cells, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 c-Kit
IC50:1.7nMPDGFRβ
IC50:1.6nMVEGFR1
IC50:0.1nMVEGFR2
IC50:0.2nMVEGFR3
IC50:0.1nM-0.3nM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00692341 Hepatic Insufficiency Phase 1 Completed - United States, Florida ... more >> Pfizer Investigational Site Miami, Florida, United States, 33169 Pfizer Investigational Site Orlando, Florida, United States, 32809 Collapse << NCT03386929 Non-small Cell Lung Cancer Met... more >>astatic Non-Small Cell Lung Cancer Stage IIIB Collapse << Phase 1 Phase 2 Recruiting December 2022 United States, California ... more >> UCSD Moores Cancer Center Recruiting La Jolla, California, United States, 92093 Contact: Sarah MOORE sam055@ucsd.edu Principal Investigator: Lyudmila BAZHENOVA, MD United States, South Dakota Avera Cancer Center Recruiting Sioux Falls, South Dakota, United States, 57105 Contact: Martha LANG Martha.lang@avera.org Principal Investigator: Benjamin SOLOMON, MD France Centre Léon Bérard Recruiting Lyon, France, 69008 Contact: Séverine LAURENT severine.laurent@lyon.unicancer.fr Principal Investigator: Pierre SAINTIGNY, MD Israel Chiam Sheba Medical Center Recruiting Ramat Gan, Israel, 5265601 Contact: Yona GILADY yona.gilady@sheba.health.gov.il Principal Investigator: Jair BAR, MD Luxembourg Centre Hospitalier Luxembourg Recruiting Luxembourg, Luxembourg, 1210 Contact: Lucile PERNOT lucile.pernot@lih.lu Principal Investigator: Guy BERCHEM, MD Spain Vall Hebron Institute of Oncology Recruiting Barcelona, Spain, 08035 Contact: LLuisa CARBONELL llcarbonell@vhio.net Principal Investigator: Enriqueta Felip, MD Collapse << NCT00389441 Thyroid Neoplasms Phase 2 Completed - - - 更多
- 参考文献
- [1] Hu-Lowe DD, Zou HY, et al. Nonclinical antiangiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2, 3. Clin Cancer Res. 2008;14(22):7272-83.
- [2] Du Four S, Maenhout SK, et al. Axitinib increases the infiltration of immune cells and reduces the suppressive capacity of monocytic MDSCs in an intracranial mouse melanoma model. Oncoimmunology. 2015;4(4):e998107.
- [3] Van der Veken B, De Meyer GRY, Martinet W. Axitinib attenuates intraplaque angiogenesis, haemorrhages and plaque destabilization in mice. Vascul Pharmacol. 2018.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 319460-85-0 | 储存条件 |
|
|||||||||||||
| 分子式 | C22H18N4OS | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 386.47 | 别名 | AG-013736;Brand name: Inlyta;阿昔替尼 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 22RV1 | - | Growth Inhibition Assay | - | IC50=17.5884 μM | SANGER |
| 23132-87 | - | Growth Inhibition Assay | - | IC50=12.0821 μM | SANGER |
| 5637 | - | Growth Inhibition Assay | - | IC50=29.6421 μM | SANGER |
| 697 | - | Growth Inhibition Assay | - | IC50=14.627 μM | SANGER |
| 786-0 | - | Growth Inhibition Assay | - | IC50=37.2794 μM | SANGER |
| 8305C | - | Growth Inhibition Assay | - | IC50=16.7877 μM | SANGER |
| 8505C | - | Growth Inhibition Assay | - | IC50=8.64824 μM | SANGER |
| 8-MG-BA | - | Growth Inhibition Assay | - | IC50=4.26658 μM | SANGER |
| A101D | - | Growth Inhibition Assay | - | IC50=13.5349 μM | SANGER |
| A172 | - | Growth Inhibition Assay | - | IC50=2.13606 μM | SANGER |
| A204 | - | Growth Inhibition Assay | - | IC50=0.00992 μM | SANGER |
| A2058 | - | Growth Inhibition Assay | - | IC50=31.1164 μM | SANGER |
| A2780 | - | Growth Inhibition Assay | - | IC50=1.01146 μM | SANGER |
| A375 | - | Growth Inhibition Assay | - | IC50=31.1696 μM | SANGER |
| A3-KAW | - | Growth Inhibition Assay | - | IC50=4.97352 μM | SANGER |
| A427 | - | Growth Inhibition Assay | - | IC50=1.10381 μM | SANGER |
| A431 | - | Growth Inhibition Assay | - | IC50=47.8338 μM | SANGER |
| A498 | - | Growth Inhibition Assay | - | IC50=41.3015 μM | SANGER |
| A4-Fuk | - | Growth Inhibition Assay | - | IC50=2.47168 μM | SANGER |
| A549 | - | Growth Inhibition Assay | - | IC50=12.3285 μM | SANGER |
| A673 | - | Growth Inhibition Assay | - | IC50=27.2102 μM | SANGER |
| A704 | - | Growth Inhibition Assay | - | IC50=6.10574 μM | SANGER |
| ABC-1 | - | Growth Inhibition Assay | - | IC50=3.93911 μM | SANGER |
| ACN | - | Growth Inhibition Assay | - | IC50=2.23875 μM | SANGER |
| AGS | - | Growth Inhibition Assay | - | IC50=1.11395 μM | SANGER |
| ALL-PO | - | Growth Inhibition Assay | - | IC50=1.80015 μM | SANGER |
| AN3-CA | - | Growth Inhibition Assay | - | IC50=1.08389 μM | SANGER |
| AsPC-1 | - | Growth Inhibition Assay | - | IC50=30.0212 μM | SANGER |
| ATN-1 | - | Growth Inhibition Assay | - | IC50=22.6439 μM | SANGER |
| AU565 | - | Growth Inhibition Assay | - | IC50=17.225 μM | SANGER |
| BB65-RCC | - | Growth Inhibition Assay | - | IC50=20.9285 μM | SANGER |
| BE-13 | - | Growth Inhibition Assay | - | IC50=0.51008 μM | SANGER |
| Becker | - | Growth Inhibition Assay | - | IC50=9.41944 μM | SANGER |
| BFTC-905 | - | Growth Inhibition Assay | - | IC50=33.5136 μM | SANGER |
| BHT-101 | - | Growth Inhibition Assay | - | IC50=29.321 μM | SANGER |
| BPH-1 | - | Growth Inhibition Assay | - | IC50=46.9805 μM | SANGER |
| BT-549 | - | Growth Inhibition Assay | - | IC50=1.56909 μM | SANGER |
| BV-173 | - | Growth Inhibition Assay | - | IC50=0.3932 μM | SANGER |
| C2BBe1 | - | Growth Inhibition Assay | - | IC50=40.9157 μM | SANGER |
| C32 | - | Growth Inhibition Assay | - | IC50=32.3353 μM | SANGER |
| C8166 | - | Growth Inhibition Assay | - | IC50=49.2039 μM | SANGER |
| Ca9-22 | - | Growth Inhibition Assay | - | IC50=10.0665 μM | SANGER |
| CAKI-1 | - | Growth Inhibition Assay | - | IC50=30.7702 μM | SANGER |
| CAL-12T | - | Growth Inhibition Assay | - | IC50=10.6361 μM | SANGER |
| CAL-27 | - | Growth Inhibition Assay | - | IC50=4.24344 μM | SANGER |
| CAL-51 | - | Growth Inhibition Assay | - | IC50=43.5605 μM | SANGER |
| CAL-54 | - | Growth Inhibition Assay | - | IC50=29.5445 μM | SANGER |
| CAL-72 | - | Growth Inhibition Assay | - | IC50=49.6075 μM | SANGER |
| CAMA-1 | - | Growth Inhibition Assay | - | IC50=28.8564 μM | SANGER |
| CaR-1 | - | Growth Inhibition Assay | - | IC50=16.8111 μM | SANGER |
| CAS-1 | - | Growth Inhibition Assay | - | IC50=4.99908 μM | SANGER |
| Ca-Ski | - | Growth Inhibition Assay | - | IC50=29.946 μM | SANGER |
| CFPAC-1 | - | Growth Inhibition Assay | - | IC50=43.4944 μM | SANGER |
| CGTH-W-1 | - | Growth Inhibition Assay | - | IC50=0.00722 μM | SANGER |
| ChaGo-K-1 | - | Growth Inhibition Assay | - | IC50=4.89048 μM | SANGER |
| CHL-1 | - | Growth Inhibition Assay | - | IC50=17.9249 μM | SANGER |
| CHP-212 | - | Growth Inhibition Assay | - | IC50=2.82091 μM | SANGER |
| COLO-320-HSR | - | Growth Inhibition Assay | - | IC50=9.60237 μM | SANGER |
| COLO-684 | - | Growth Inhibition Assay | - | IC50=13.4541 μM | SANGER |
| COLO-800 | - | Growth Inhibition Assay | - | IC50=7.94492 μM | SANGER |
| COLO-829 | - | Growth Inhibition Assay | - | IC50=5.91504 μM | SANGER |
| COR-L105 | - | Growth Inhibition Assay | - | IC50=3.63334 μM | SANGER |
| COR-L23 | - | Growth Inhibition Assay | - | IC50=18.0011 μM | SANGER |
| COR-L88 | - | Growth Inhibition Assay | - | IC50=23.9475 μM | SANGER |
| CP50-MEL-B | - | Growth Inhibition Assay | - | IC50=20.4748 μM | SANGER |
| CTB-1 | - | Growth Inhibition Assay | - | IC50=9.01433 μM | SANGER |
| CTV-1 | - | Growth Inhibition Assay | - | IC50=22.3031 μM | SANGER |
| D-247MG | - | Growth Inhibition Assay | - | IC50=16.8625 μM | SANGER |
| D-263MG | - | Growth Inhibition Assay | - | IC50=9.83384 μM | SANGER |
| D-283MED | - | Growth Inhibition Assay | - | IC50=9.63072 μM | SANGER |
| D-392MG | - | Growth Inhibition Assay | - | IC50=5.57849 μM | SANGER |
| D-423MG | - | Growth Inhibition Assay | - | IC50=8.2172 μM | SANGER |
| D-502MG | - | Growth Inhibition Assay | - | IC50=6.28857 μM | SANGER |
| D-542MG | - | Growth Inhibition Assay | - | IC50=22.5558 μM | SANGER |
| D-566MG | - | Growth Inhibition Assay | - | IC50=1.40683 μM | SANGER |
| Daoy | - | Growth Inhibition Assay | - | IC50=17.7184 μM | SANGER |
| DB | - | Growth Inhibition Assay | - | IC50=13.9928 μM | SANGER |
| DEL | - | Growth Inhibition Assay | - | IC50=1.25457 μM | SANGER |
| Detroit562 | - | Growth Inhibition Assay | - | IC50=40.1268 μM | SANGER |
| DK-MG | - | Growth Inhibition Assay | - | IC50=2.23834 μM | SANGER |
| DMS-114 | - | Growth Inhibition Assay | - | IC50=2.66345 μM | SANGER |
| DMS-273 | - | Growth Inhibition Assay | - | IC50=2.90207 μM | SANGER |
| DOHH-2 | - | Growth Inhibition Assay | - | IC50=20.906 μM | SANGER |
| DOK | - | Growth Inhibition Assay | - | IC50=14.9933 μM | SANGER |
| DU-145 | - | Growth Inhibition Assay | - | IC50=4.12825 μM | SANGER |
| EC-GI-10 | - | Growth Inhibition Assay | - | IC50=43.8347 μM | SANGER |
| EFM-19 | - | Growth Inhibition Assay | - | IC50=5.41737 μM | SANGER |
| EGI-1 | - | Growth Inhibition Assay | - | IC50=21.7454 μM | SANGER |
| EM-2 | - | Growth Inhibition Assay | - | IC50=0.16816 μM | SANGER |
| EoL-1-cell | - | Growth Inhibition Assay | - | IC50=0.0000513 μM | SANGER |
| EPLC-272H | - | Growth Inhibition Assay | - | IC50=19.3708 μM | SANGER |
| ES1 | - | Growth Inhibition Assay | - | IC50=3.83094 μM | SANGER |
| ES3 | - | Growth Inhibition Assay | - | IC50=18.5883 μM | SANGER |
| ES4 | - | Growth Inhibition Assay | - | IC50=29.3172 μM | SANGER |
| ES5 | - | Growth Inhibition Assay | - | IC50=1.17248 μM | SANGER |
| ES6 | - | Growth Inhibition Assay | - | IC50=11.8581 μM | SANGER |
| ES7 | - | Growth Inhibition Assay | - | IC50=1.11334 μM | SANGER |
| ES8 | - | Growth Inhibition Assay | - | IC50=3.34999 μM | SANGER |
| ESS-1 | - | Growth Inhibition Assay | - | IC50=23.8059 μM | SANGER |
| ETK-1 | - | Growth Inhibition Assay | - | IC50=13.0779 μM | SANGER |
| EW-1 | - | Growth Inhibition Assay | - | IC50=4.53014 μM | SANGER |
| EW-11 | - | Growth Inhibition Assay | - | IC50=3.63022 μM | SANGER |
| EW-13 | - | Growth Inhibition Assay | - | IC50=32.9408 μM | SANGER |
| EW-16 | - | Growth Inhibition Assay | - | IC50=5.75583 μM | SANGER |
| EW-22 | - | Growth Inhibition Assay | - | IC50=36.4072 μM | SANGER |
| G-361 | - | Growth Inhibition Assay | - | IC50=0.32325 μM | SANGER |
| G-402 | - | Growth Inhibition Assay | - | IC50=1.14694 μM | SANGER |
| GCT | - | Growth Inhibition Assay | - | IC50=2.00916 μM | SANGER |
| GI-1 | - | Growth Inhibition Assay | - | IC50=10.3533 μM | SANGER |
| GI-ME-N | - | Growth Inhibition Assay | - | IC50=7.64934 μM | SANGER |
| GOTO | - | Growth Inhibition Assay | - | IC50=2.53013 μM | SANGER |
| GP5d | - | Growth Inhibition Assay | - | IC50=16.342 μM | SANGER |
| GT3TKB | - | Growth Inhibition Assay | - | IC50=16.0676 μM | SANGER |
| H4 | - | Growth Inhibition Assay | - | IC50=1.06422 μM | SANGER |
| H9 | - | Growth Inhibition Assay | - | IC50=12.5577 μM | SANGER |
| HAL-01 | - | Growth Inhibition Assay | - | IC50=7.60644 μM | SANGER |
| HC-1 | - | Growth Inhibition Assay | - | IC50=32.1446 μM | SANGER |
| HCC1187 | - | Growth Inhibition Assay | - | IC50=4.21597 μM | SANGER |
| HCC1395 | - | Growth Inhibition Assay | - | IC50=3.99484 μM | SANGER |
| HCC1419 | - | Growth Inhibition Assay | - | IC50=7.53 μM | SANGER |
| HCC1569 | - | Growth Inhibition Assay | - | IC50=6.53737 μM | SANGER |
| HCC1806 | - | Growth Inhibition Assay | - | IC50=3.27657 μM | SANGER |
| HCC1937 | - | Growth Inhibition Assay | - | IC50=24.1 μM | SANGER |
| HCC2998 | - | Growth Inhibition Assay | - | IC50=7.60743 μM | SANGER |
| HCC38 | - | Growth Inhibition Assay | - | IC50=24.2394 μM | SANGER |
| HCE-T | - | Growth Inhibition Assay | - | IC50=2.20598 μM | SANGER |
| HCT-15 | - | Growth Inhibition Assay | - | IC50=40.5907 μM | SANGER |
| HDLM-2 | - | Growth Inhibition Assay | - | IC50=23.1478 μM | SANGER |
| HD-MY-Z | - | Growth Inhibition Assay | - | IC50=1.86281 μM | SANGER |
| HEC-1 | - | Growth Inhibition Assay | - | IC50=8.78439 μM | SANGER |
| HEL | - | Growth Inhibition Assay | - | IC50=49.4064 μM | SANGER |
| H-EMC-SS | - | Growth Inhibition Assay | - | IC50=0.11005 μM | SANGER |
| HGC-27 | - | Growth Inhibition Assay | - | IC50=4.2966 μM | SANGER |
| HH | - | Growth Inhibition Assay | - | IC50=26.5529 μM | SANGER |
| HL-60 | - | Growth Inhibition Assay | - | IC50=20.2141 μM | SANGER |
| HLE | - | Growth Inhibition Assay | - | IC50=9.90909 μM | SANGER |
| HMV-II | - | Growth Inhibition Assay | - | IC50=1.70488 μM | SANGER |
| HN | - | Growth Inhibition Assay | - | IC50=40.1782 μM | SANGER |
| HO-1-N-1 | - | Growth Inhibition Assay | - | IC50=11.3398 μM | SANGER |
| HOP-62 | - | Growth Inhibition Assay | - | IC50=0.10836 μM | SANGER |
| HOS | - | Growth Inhibition Assay | - | IC50=4.6977 μM | SANGER |
| Hs-578-T | - | Growth Inhibition Assay | - | IC50=4.31468 μM | SANGER |
| HSC-2 | - | Growth Inhibition Assay | - | IC50=44.009 μM | SANGER |
| HSC-3 | - | Growth Inhibition Assay | - | IC50=12.9276 μM | SANGER |
| HSC-4 | - | Growth Inhibition Assay | - | IC50=1.93839 μM | SANGER |
| HT-1080 | - | Growth Inhibition Assay | - | IC50=37.125 μM | SANGER |
| HT-1197 | - | Growth Inhibition Assay | - | IC50=14.0809 μM | SANGER |
| HT-144 | - | Growth Inhibition Assay | - | IC50=17.6598 μM | SANGER |
| HuH-7 | - | Growth Inhibition Assay | - | IC50=2.42165 μM | SANGER |
| HuO9 | - | Growth Inhibition Assay | - | IC50=17.7031 μM | SANGER |
| HuP-T4 | - | Growth Inhibition Assay | - | IC50=11.0328 μM | SANGER |
| HUTU-80 | - | Growth Inhibition Assay | - | IC50=6.41688 μM | SANGER |
| IA-LM | - | Growth Inhibition Assay | - | IC50=7.68613 μM | SANGER |
| IGR-1 | - | Growth Inhibition Assay | - | IC50=36.8184 μM | SANGER |
| IGROV-1 | - | Growth Inhibition Assay | - | IC50=7.32386 μM | SANGER |
| J82 | - | Growth Inhibition Assay | - | IC50=17.9635 μM | SANGER |
| JAR | - | Growth Inhibition Assay | - | IC50=0.23989 μM | SANGER |
| JEG-3 | - | Growth Inhibition Assay | - | IC50=4.15916 μM | SANGER |
| J-RT3-T3-5 | - | Growth Inhibition Assay | - | IC50=6.54572 μM | SANGER |
| JVM-3 | - | Growth Inhibition Assay | - | IC50=5.72325 μM | SANGER |
| K5 | - | Growth Inhibition Assay | - | IC50=0.42127 μM | SANGER |
| K-562 | - | Growth Inhibition Assay | - | IC50=11.7248 μM | SANGER |
| KARPAS-45 | - | Growth Inhibition Assay | - | IC50=5.84325 μM | SANGER |
| KASUMI-1 | - | Growth Inhibition Assay | - | IC50=0.00682 μM | SANGER |
| KE-37 | - | Growth Inhibition Assay | - | IC50=13.5887 μM | SANGER |
| KG-1 | - | Growth Inhibition Assay | - | IC50=0.37642 μM | SANGER |
| KINGS-1 | - | Growth Inhibition Assay | - | IC50=20.7967 μM | SANGER |
| KM12 | - | Growth Inhibition Assay | - | IC50=5.29827 μM | SANGER |
| KM-H2 | - | Growth Inhibition Assay | - | IC50=5.92395 μM | SANGER |
| KMOE-2 | - | Growth Inhibition Assay | - | IC50=3.91808 μM | SANGER |
| KNS-81-FD | - | Growth Inhibition Assay | - | IC50=45.7365 μM | SANGER |
| KOSC-2 | - | Growth Inhibition Assay | - | IC50=42.2699 μM | SANGER |
| KP-4 | - | Growth Inhibition Assay | - | IC50=29.516 μM | SANGER |
| KP-N-YS | - | Growth Inhibition Assay | - | IC50=7.2072 μM | SANGER |
| KS-1 | - | Growth Inhibition Assay | - | IC50=10.6301 μM | SANGER |
| KU812 | - | Growth Inhibition Assay | - | IC50=0.16577 μM | SANGER |
| KY821 | - | Growth Inhibition Assay | - | IC50=21.582 μM | SANGER |
| KYSE-510 | - | Growth Inhibition Assay | - | IC50=35.5042 μM | SANGER |
| L-363 | - | Growth Inhibition Assay | - | IC50=2.28061 μM | SANGER |
| L-428 | - | Growth Inhibition Assay | - | IC50=30.405 μM | SANGER |
| LAMA-84 | - | Growth Inhibition Assay | - | IC50=0.17665 μM | SANGER |
| LAN-6 | - | Growth Inhibition Assay | - | IC50=37.9582 μM | SANGER |
| LB1047-RCC | - | Growth Inhibition Assay | - | IC50=4.81418 μM | SANGER |
| LB2241-RCC | - | Growth Inhibition Assay | - | IC50=39.8205 μM | SANGER |
| LB373-MEL-D | - | Growth Inhibition Assay | - | IC50=32.1971 μM | SANGER |
| LB771-HNC | - | Growth Inhibition Assay | - | IC50=12.765 μM | SANGER |
| LC-2-ad | - | Growth Inhibition Assay | - | IC50=4.22177 μM | SANGER |
| LCLC-103H | - | Growth Inhibition Assay | - | IC50=22.4752 μM | SANGER |
| LK-2 | - | Growth Inhibition Assay | - | IC50=15.6449 μM | SANGER |
| LNCaP-Clone-FGC | - | Growth Inhibition Assay | - | IC50=34.8515 μM | SANGER |
| LoVo | - | Growth Inhibition Assay | - | IC50=16.9488 μM | SANGER |
| LOXIMVI | - | Growth Inhibition Assay | - | IC50=6.11248 μM | SANGER |
| LS-411N | - | Growth Inhibition Assay | - | IC50=26.2038 μM | SANGER |
| LU-135 | - | Growth Inhibition Assay | - | IC50=33.801 μM | SANGER |
| LXF-289 | - | Growth Inhibition Assay | - | IC50=1.43996 μM | SANGER |
| M14 | - | Growth Inhibition Assay | - | IC50=37.1642 μM | SANGER |
| MCF7 | - | Growth Inhibition Assay | - | IC50=46.5533 μM | SANGER |
| MC-IXC | - | Growth Inhibition Assay | - | IC50=29.2366 μM | SANGER |
| MDA-MB-231 | - | Growth Inhibition Assay | - | IC50=18.6502 μM | SANGER |
| MDA-MB-361 | - | Growth Inhibition Assay | - | IC50=10.4432 μM | SANGER |
| MDA-MB-415 | - | Growth Inhibition Assay | - | IC50=7.89186 μM | SANGER |
| ME-180 | - | Growth Inhibition Assay | - | IC50=31.6542 μM | SANGER |
| MEG-01 | - | Growth Inhibition Assay | - | IC50=0.42361 μM | SANGER |
| MEL-JUSO | - | Growth Inhibition Assay | - | IC50=9.90127 μM | SANGER |
| MFM-223 | - | Growth Inhibition Assay | - | IC50=0.44776 μM | SANGER |
| MHH-NB-11 | - | Growth Inhibition Assay | - | IC50=2.70299 μM | SANGER |
| MHH-PREB-1 | - | Growth Inhibition Assay | - | IC50=18.5697 μM | SANGER |
| MIA-PaCa-2 | - | Growth Inhibition Assay | - | IC50=36.0496 μM | SANGER |
| MKN1 | - | Growth Inhibition Assay | - | IC50=47.5137 μM | SANGER |
| MKN45 | - | Growth Inhibition Assay | - | IC50=6.26016 μM | SANGER |
| ML-2 | - | Growth Inhibition Assay | - | IC50=45.2621 μM | SANGER |
| MN-60 | - | Growth Inhibition Assay | - | IC50=19.0592 μM | SANGER |
| MOLT-13 | - | Growth Inhibition Assay | - | IC50=19.4546 μM | SANGER |
| MOLT-16 | - | Growth Inhibition Assay | - | IC50=29.7269 μM | SANGER |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=11.5985 μM | SANGER |
| Mo-T | - | Growth Inhibition Assay | - | IC50=42.8958 μM | SANGER |
| MS-1 | - | Growth Inhibition Assay | - | IC50=35.3038 μM | SANGER |
| MSTO-211H | - | Growth Inhibition Assay | - | IC50=30.15 μM | SANGER |
| MV-4-11 | - | Growth Inhibition Assay | - | IC50=2.59169 μM | SANGER |
| MZ2-MEL | - | Growth Inhibition Assay | - | IC50=37.4501 μM | SANGER |
| NB10 | - | Growth Inhibition Assay | - | IC50=1.32557 μM | SANGER |
| NB12 | - | Growth Inhibition Assay | - | IC50=21.0394 μM | SANGER |
| NB13 | - | Growth Inhibition Assay | - | IC50=25.2611 μM | SANGER |
| NB14 | - | Growth Inhibition Assay | - | IC50=1.01801 μM | SANGER |
| NB17 | - | Growth Inhibition Assay | - | IC50=9.1887 μM | SANGER |
| NB6 | - | Growth Inhibition Assay | - | IC50=46.111 μM | SANGER |
| NB7 | - | Growth Inhibition Assay | - | IC50=16.6971 μM | SANGER |
| NBsusSR | - | Growth Inhibition Assay | - | IC50=5.03514 μM | SANGER |
| NCI-H1048 | - | Growth Inhibition Assay | - | IC50=6.10599 μM | SANGER |
| NCI-H1092 | - | Growth Inhibition Assay | - | IC50=35.2747 μM | SANGER |
| NCI-H1155 | - | Growth Inhibition Assay | - | IC50=5.38185 μM | SANGER |
| NCI-H1299 | - | Growth Inhibition Assay | - | IC50=33.5621 μM | SANGER |
| NCI-H1563 | - | Growth Inhibition Assay | - | IC50=3.15005 μM | SANGER |
| NCI-H1573 | - | Growth Inhibition Assay | - | IC50=23.4681 μM | SANGER |
| NCI-H1581 | - | Growth Inhibition Assay | - | IC50=1.39042 μM | SANGER |
| NCI-H1648 | - | Growth Inhibition Assay | - | IC50=30.5574 μM | SANGER |
| NCI-H1703 | - | Growth Inhibition Assay | - | IC50=0.000902 μM | SANGER |
| NCI-H1770 | - | Growth Inhibition Assay | - | IC50=18.4615 μM | SANGER |
| NCI-H1792 | - | Growth Inhibition Assay | - | IC50=41.9821 μM | SANGER |
| NCI-H1793 | - | Growth Inhibition Assay | - | IC50=35.6545 μM | SANGER |
| NCI-H2009 | - | Growth Inhibition Assay | - | IC50=15.5095 μM | SANGER |
| NCI-H2029 | - | Growth Inhibition Assay | - | IC50=15.3602 μM | SANGER |
| NCI-H2052 | - | Growth Inhibition Assay | - | IC50=27.086 μM | SANGER |
| NCI-H209 | - | Growth Inhibition Assay | - | IC50=31.8247 μM | SANGER |
| NCI-H2122 | - | Growth Inhibition Assay | - | IC50=33.9966 μM | SANGER |
| NCI-H2228 | - | Growth Inhibition Assay | - | IC50=8.15783 μM | SANGER |
| NCI-H226 | - | Growth Inhibition Assay | - | IC50=13.8808 μM | SANGER |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=12.5026 μM | SANGER |
| NCI-H2405 | - | Growth Inhibition Assay | - | IC50=17.1908 μM | SANGER |
| NCI-H2452 | - | Growth Inhibition Assay | - | IC50=28.0822 μM | SANGER |
| NCI-H28 | - | Growth Inhibition Assay | - | IC50=5.87918 μM | SANGER |
| NCI-H292 | - | Growth Inhibition Assay | - | IC50=1.66383 μM | SANGER |
| NCI-H358 | - | Growth Inhibition Assay | - | IC50=11.2657 μM | SANGER |
| NCI-H446 | - | Growth Inhibition Assay | - | IC50=3.22805 μM | SANGER |
| NCI-H460 | - | Growth Inhibition Assay | - | IC50=16.7326 μM | SANGER |
| NCI-H510A | - | Growth Inhibition Assay | - | IC50=37.9412 μM | SANGER |
| NCI-H520 | - | Growth Inhibition Assay | - | IC50=23.4948 μM | SANGER |
| NCI-H526 | - | Growth Inhibition Assay | - | IC50=3.99126 μM | SANGER |
| NCI-H661 | - | Growth Inhibition Assay | - | IC50=15.9075 μM | SANGER |
| NCI-H720 | - | Growth Inhibition Assay | - | IC50=7.19345 μM | SANGER |
| NCI-H810 | - | Growth Inhibition Assay | - | IC50=26.3112 μM | SANGER |
| NCI-H82 | - | Growth Inhibition Assay | - | IC50=5.92771 μM | SANGER |
| NCI-SNU-1 | - | Growth Inhibition Assay | - | IC50=26.5454 μM | SANGER |
| NEC8 | - | Growth Inhibition Assay | - | IC50=0.72113 μM | SANGER |
| NH-12 | - | Growth Inhibition Assay | - | IC50=11.5378 μM | SANGER |
| NKM-1 | - | Growth Inhibition Assay | - | IC50=1.60556 μM | SANGER |
| NOMO-1 | - | Growth Inhibition Assay | - | IC50=27.8532 μM | SANGER |
| NOS-1 | - | Growth Inhibition Assay | - | IC50=8.51534 μM | SANGER |
| NTERA-S-cl-D1 | - | Growth Inhibition Assay | - | IC50=20.4978 μM | SANGER |
| NUGC-3 | - | Growth Inhibition Assay | - | IC50=10.4402 μM | SANGER |
| OAW-42 | - | Growth Inhibition Assay | - | IC50=5.98821 μM | SANGER |
| OC-314 | - | Growth Inhibition Assay | - | IC50=6.91159 μM | SANGER |
| OCI-AML2 | - | Growth Inhibition Assay | - | IC50=17.5317 μM | SANGER |
| OMC-1 | - | Growth Inhibition Assay | - | IC50=14.7888 μM | SANGER |
| ONS-76 | - | Growth Inhibition Assay | - | IC50=4.24192 μM | SANGER |
| OS-RC-2 | - | Growth Inhibition Assay | - | IC50=10.1045 μM | SANGER |
| OVCAR-4 | - | Growth Inhibition Assay | - | IC50=10.7068 μM | SANGER |
| OVCAR-5 | - | Growth Inhibition Assay | - | IC50=41.7767 μM | SANGER |
| P12-ICHIKAWA | - | Growth Inhibition Assay | - | IC50=24.6258 μM | SANGER |
| P30-OHK | - | Growth Inhibition Assay | - | IC50=46.8117 μM | SANGER |
| PA-1 | - | Growth Inhibition Assay | - | IC50=1.82725 μM | SANGER |
| PC-3 | - | Growth Inhibition Assay | - | IC50=27.8941 μM | SANGER |
| PFSK-1 | - | Growth Inhibition Assay | - | IC50=12.9507 μM | SANGER |
| PSN1 | - | Growth Inhibition Assay | - | IC50=21.6453 μM | SANGER |
| QIMR-WIL | - | Growth Inhibition Assay | - | IC50=12.8258 μM | SANGER |
| Ramos-2G6-4C10 | - | Growth Inhibition Assay | - | IC50=11.932 μM | SANGER |
| RH-18 | - | Growth Inhibition Assay | - | IC50=43.805 μM | SANGER |
| RMG-I | - | Growth Inhibition Assay | - | IC50=49.4444 μM | SANGER |
| RO82-W-1 | - | Growth Inhibition Assay | - | IC50=11.9064 μM | SANGER |
| RPMI-2650 | - | Growth Inhibition Assay | - | IC50=24.6162 μM | SANGER |
| RPMI-7951 | - | Growth Inhibition Assay | - | IC50=13.5136 μM | SANGER |
| RPMI-8226 | - | Growth Inhibition Assay | - | IC50=3.62616 μM | SANGER |
| RS4-11 | - | Growth Inhibition Assay | - | IC50=24.142 μM | SANGER |
| RT-112 | - | Growth Inhibition Assay | - | IC50=2.13424 μM | SANGER |
| S-117 | - | Growth Inhibition Assay | - | IC50=6.50267 μM | SANGER |
| SBC-5 | - | Growth Inhibition Assay | - | IC50=14.1362 μM | SANGER |
| SCC-15 | - | Growth Inhibition Assay | - | IC50=10.4718 μM | SANGER |
| SCC-4 | - | Growth Inhibition Assay | - | IC50=3.50396 μM | SANGER |
| SF126 | - | Growth Inhibition Assay | - | IC50=3.30017 μM | SANGER |
| SF295 | - | Growth Inhibition Assay | - | IC50=3.02597 μM | SANGER |
| SF539 | - | Growth Inhibition Assay | - | IC50=26.8018 μM | SANGER |
| SiHa | - | Growth Inhibition Assay | - | IC50=13.8346 μM | SANGER |
| SJRH30 | - | Growth Inhibition Assay | - | IC50=17.8489 μM | SANGER |
| SKG-IIIa | - | Growth Inhibition Assay | - | IC50=14.8001 μM | SANGER |
| SK-HEP-1 | - | Growth Inhibition Assay | - | IC50=31.4271 μM | SANGER |
| SK-LMS-1 | - | Growth Inhibition Assay | - | IC50=34.4107 μM | SANGER |
| SK-MEL-1 | - | Growth Inhibition Assay | - | IC50=22.8368 μM | SANGER |
| SK-MEL-2 | - | Growth Inhibition Assay | - | IC50=16.4485 μM | SANGER |
| SK-MEL-24 | - | Growth Inhibition Assay | - | IC50=41.4725 μM | SANGER |
| SK-N-AS | - | Growth Inhibition Assay | - | IC50=25.8584 μM | SANGER |
| SK-N-DZ | - | Growth Inhibition Assay | - | IC50=26.049 μM | SANGER |
| SK-NEP-1 | - | Growth Inhibition Assay | - | IC50=7.6961 μM | SANGER |
| SK-N-FI | - | Growth Inhibition Assay | - | IC50=7.63033 μM | SANGER |
| SK-OV-3 | - | Growth Inhibition Assay | - | IC50=1.06633 μM | SANGER |
| SK-PN-DW | - | Growth Inhibition Assay | - | IC50=7.4815 μM | SANGER |
| SK-UT-1 | - | Growth Inhibition Assay | - | IC50=16.565 μM | SANGER |
| SN12C | - | Growth Inhibition Assay | - | IC50=9.45234 μM | SANGER |
| SNB75 | - | Growth Inhibition Assay | - | IC50=31.2435 μM | SANGER |
| SNU-387 | - | Growth Inhibition Assay | - | IC50=32.3191 μM | SANGER |
| SNU-423 | - | Growth Inhibition Assay | - | IC50=4.6079 μM | SANGER |
| SNU-449 | - | Growth Inhibition Assay | - | IC50=7.01072 μM | SANGER |
| SNU-C2B | - | Growth Inhibition Assay | - | IC50=18.2276 μM | SANGER |
| SW1088 | - | Growth Inhibition Assay | - | IC50=7.65826 μM | SANGER |
| SW1463 | - | Growth Inhibition Assay | - | IC50=30.5383 μM | SANGER |
| SW1573 | - | Growth Inhibition Assay | - | IC50=4.55163 μM | SANGER |
| SW1710 | - | Growth Inhibition Assay | - | IC50=13.3641 μM | SANGER |
| SW48 | - | Growth Inhibition Assay | - | IC50=27.8005 μM | SANGER |
| SW620 | - | Growth Inhibition Assay | - | IC50=38.4974 μM | SANGER |
| SW756 | - | Growth Inhibition Assay | - | IC50=0.99846 μM | SANGER |
| SW780 | - | Growth Inhibition Assay | - | IC50=1.65158 μM | SANGER |
| SW948 | - | Growth Inhibition Assay | - | IC50=19.3934 μM | SANGER |
| SW962 | - | Growth Inhibition Assay | - | IC50=49.9332 μM | SANGER |
| SW982 | - | Growth Inhibition Assay | - | IC50=3.33875 μM | SANGER |
| T-24 | - | Growth Inhibition Assay | - | IC50=2.48037 μM | SANGER |
| T47D | - | Growth Inhibition Assay | - | IC50=10.155 μM | SANGER |
| T98G | - | Growth Inhibition Assay | - | IC50=9.90203 μM | SANGER |
| TE-1 | - | Growth Inhibition Assay | - | IC50=8.44316 μM | SANGER |
| TE-11 | - | Growth Inhibition Assay | - | IC50=8.99551 μM | SANGER |
| TGBC11TKB | - | Growth Inhibition Assay | - | IC50=9.02241 μM | SANGER |
| TGBC24TKB | - | Growth Inhibition Assay | - | IC50=24.0312 μM | SANGER |
| TI-73 | - | Growth Inhibition Assay | - | IC50=22.3498 μM | SANGER |
| U-118-MG | - | Growth Inhibition Assay | - | IC50=28.0123 μM | SANGER |
| U251 | - | Growth Inhibition Assay | - | IC50=46.994 μM | SANGER |
| U-2-OS | - | Growth Inhibition Assay | - | IC50=26.7382 μM | SANGER |
| UACC-257 | - | Growth Inhibition Assay | - | IC50=1.82163 μM | SANGER |
| UMC-11 | - | Growth Inhibition Assay | - | IC50=27.9343 μM | SANGER |
| UM-UC-3 | - | Growth Inhibition Assay | - | IC50=23.1944 μM | SANGER |
| VA-ES-BJ | - | Growth Inhibition Assay | - | IC50=2.24957 μM | SANGER |
| VMRC-RCZ | - | Growth Inhibition Assay | - | IC50=14.5774 μM | SANGER |
| WM-115 | - | Growth Inhibition Assay | - | IC50=27.7767 μM | SANGER |
| YAPC | - | Growth Inhibition Assay | - | IC50=24.8214 μM | SANGER |
| YH-13 | - | Growth Inhibition Assay | - | IC50=2.21671 μM | SANGER |
| YKG-1 | - | Growth Inhibition Assay | - | IC50=31.0263 μM | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00692341 | Hepatic Insufficiency | Phase 1 | Completed | - | United States, Florida ... more >> Pfizer Investigational Site Miami, Florida, United States, 33169 Pfizer Investigational Site Orlando, Florida, United States, 32809 Collapse << |
| NCT03386929 | Non-small Cell Lung Cancer Met... more >>astatic Non-Small Cell Lung Cancer Stage IIIB Collapse << | Phase 1 Phase 2 | Recruiting | December 2022 | United States, California ... more >> UCSD Moores Cancer Center Recruiting La Jolla, California, United States, 92093 Contact: Sarah MOORE sam055@ucsd.edu Principal Investigator: Lyudmila BAZHENOVA, MD United States, South Dakota Avera Cancer Center Recruiting Sioux Falls, South Dakota, United States, 57105 Contact: Martha LANG Martha.lang@avera.org Principal Investigator: Benjamin SOLOMON, MD France Centre Léon Bérard Recruiting Lyon, France, 69008 Contact: Séverine LAURENT severine.laurent@lyon.unicancer.fr Principal Investigator: Pierre SAINTIGNY, MD Israel Chiam Sheba Medical Center Recruiting Ramat Gan, Israel, 5265601 Contact: Yona GILADY yona.gilady@sheba.health.gov.il Principal Investigator: Jair BAR, MD Luxembourg Centre Hospitalier Luxembourg Recruiting Luxembourg, Luxembourg, 1210 Contact: Lucile PERNOT lucile.pernot@lih.lu Principal Investigator: Guy BERCHEM, MD Spain Vall Hebron Institute of Oncology Recruiting Barcelona, Spain, 08035 Contact: LLuisa CARBONELL llcarbonell@vhio.net Principal Investigator: Enriqueta Felip, MD Collapse << |
| NCT00389441 | Thyroid Neoplasms | Phase 2 | Completed | - | - |
| NCT00460603 | Colorectal Neoplasms | Phase 1 Phase 2 | Completed | - | - |
| NCT01469052 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site San Francisco, California, United States, 94115 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77030 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00692341 | - | - | Completed | - | - |
| NCT01469052 | - | - | Completed | - | - |
| NCT02184416 | - | - | Recruiting | July 20, 2019 | - |
| NCT00389441 | - | - | Completed | - | - |
| NCT00460603 | - | - | Completed | - | - |
| NCT00600821 | Non-Small-Cell Lung Carcinoma ... more >> Adenocarcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT00094055 | Thyroid Neoplasms | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Santa Monica, California, United States, 90404 United States, Colorado Pfizer Investigational Site Aurora, Colorado, United States, 80010 Pfizer Investigational Site Denver, Colorado, United States, 80220 United States, Illinois Pfizer Investigational Site Chicago, Illinois, United States, 60637 United States, Maryland Pfizer Investigational Site Baltimore, Maryland, United States, 21231-1000 Pfizer Investigational Site Baltimore, Maryland, United States, 51231 United States, Michigan Pfizer Investigational Site Ann Arbor, Michigan, United States, 48109 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77030-4009 Collapse << |
| NCT00094055 | - | - | Completed | - | - |
| NCT00726752 | Neoplasms | Phase 1 | Completed | - | Japan ... more >> Pfizer Investigational Site Kobe-shi, Hyogo-ken, Japan Collapse << |
| NCT01020136 | Healthy Volunteer | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT00726752 | - | - | Completed | - | - |
| NCT03086174 | Kidney Cancer Stage Iv ... more >> Advanced Melanoma Collapse << | Phase 1 | Recruiting | February 2021 | China, Beijing ... more >> Beijing Cancer Hospital Recruiting Beijing, Beijing, China, 100142 Contact: Jun Guo, Phd; Md guoj307@126.com Principal Investigator: Jun Guo, PhD; MD Sub-Investigator: xinan Sheng, MD Collapse << |
| NCT00094107 | Melanoma Skin... more >> Neoplasms Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Orange, California, United States, 92868 United States, Florida Pfizer Investigational Site Miami Beach, Florida, United States, 33140 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02115 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 United States, Pennsylvania Pfizer Investigational Site Clairton, Pennsylvania, United States, 15025 Pfizer Investigational Site Greensburg, Pennsylvania, United States, 15601 Pfizer Investigational Site Johnstown, Pennsylvania, United States, 15901 Pfizer Investigational Site Pittsburgh, Pennsylvania, United States, 15232-1305 Pfizer Investigational Site Pittsburgh, Pennsylvania, United States, 15232 Pfizer Investigational Site Wexford, Pennsylvania, United States, 15090 France Pfizer Investigational Site Paris Cedex 13, France, 75651 Collapse << |
| NCT00600821 | - | - | Completed | - | - |
| NCT00094107 | - | - | Completed | - | - |
| NCT00219557 | Pancreatic Neoplasms | Phase 2 | Completed | - | - |
| NCT00918632 | Healthy Volunteers | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, 1070 Collapse << |
| NCT00071006 | Acute Myeloid Leukemia (AML) ... more >> Myelodysplastic Syndrome Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Pfizer Investigational Site Houston, Texas, United States, 77030 Collapse << |
| NCT03538717 | - | - | Recruiting | June 30, 2019 | Spain ... more >> Hospital Duran i Reynals Not yet recruiting Hospitalet de Llobregat, Barcelona, Spain, 08908 Hospital Universitario Parc Taulí Not yet recruiting Sabadell, Barcelona, Spain, 08208 Hospital Sant Pau i Santa Tecla Not yet recruiting Tarragona, Barcelona, Spain, 43003 Hospital Universitario Mutua Terrassa Not yet recruiting Terrassa, Barcelona, Spain, 08221 Complejo Hospitalario la Mancha Centro Not yet recruiting Alcazar de San Juan, Ciudad REAL, Spain, 13600 Hospital Universitario Principe de Asturias Not yet recruiting Alcalá de Henares, Madrid, Spain, 28805 Hospital Universitario Fundacion Alcorcón Recruiting Alcorcón, Madrid, Spain, 28922 Hospital Universitario Severo Ochoa Recruiting Leganés, Madrid, Spain, 28911 Hospital Universitario Rey Juan carlos Not yet recruiting Mostoles, Madrid, Spain, 28933 Hospital Infanta Cristina Not yet recruiting Parla, Madrid, Spain, 28981 Hospital Universitario Infanta Sofía Not yet recruiting San Sebatián De Los Reyes, Madrid, Spain, 28703 Hospital Universitario Infanta Elena Not yet recruiting Valdemoro, Madrid, Spain, 28342 Hospital Universitario Son Espases Not yet recruiting Palma de Mallorca, Mallorca, Spain, 07120 HU de Navarra, Pamplona Not yet recruiting Pamplona, Navarra, Spain, 31008 Hospital Universitario de Canarias Not yet recruiting La Cuesta, Santa Cruz De Tenerife, Tenerife, Spain, 38320 Hospital Universitario Nuestra Señora de la Candelaria, Tenerife Not yet recruiting Santa Cruz de Tenerife, Tenerife, Spain, 38010 Hospita General Nuestra Señora del Prado Not yet recruiting Talavera de la Reina, Toledo, Spain, 45600 Hospital del Mar Not yet recruiting Barcelona, Spain, 08003 Hospital Universitario Vall d'Hebrón Not yet recruiting Barcelona, Spain, 08036 HU Clinic i Provincial Not yet recruiting Barcelona, Spain, 08036 Hospital Universitario Sant Pau i Santa Creu Recruiting Barcelona, Spain, 08041 Hospital General Universitario de Ciudad Real Not yet recruiting Ciudad Real, Spain, 13005 Hospital Reina Sofía Not yet recruiting Córdoba, Spain, 14004 Hospital Universitario de Cabueñes Not yet recruiting Gijón, Spain, 33394 Complejo Hospitalario de Jaén Not yet recruiting Jaén, Spain, 23007 HU De León Not yet recruiting León, Spain, 24080 Hospital Universitario Lucus Augusti (HULA_ Lugo) Not yet recruiting Lugo, Spain, 27003 Hospital Universitario Ramón y Cajal Recruiting Madrid, Spain, 28034 Hospital Universitario Fundación Jimenez Diaz Not yet recruiting Madrid, Spain, 28040 Hospital Universitario 12 de Octubre Not yet recruiting Madrid, Spain, 28041 Hospital Universitario La Paz Not yet recruiting Madrid, Spain, 28046 Centro Integral Oncológico Clara Campal Not yet recruiting Madrid, Spain, 28050 Complejo Hospitalario Universitario de Ourense CHUOU Not yet recruiting Orense, Spain, 32005 Hospital Universitario Central de Asturias Not yet recruiting Oviedo, Spain, 33011 Hospital Universitario Santiago de Compostela Not yet recruiting Santiago de Compostela, Spain, 15076 Complejo Asistencial de Segovia Not yet recruiting Segovia, Spain, 40002 Hospita Virgen de la Salud de Toledo Not yet recruiting Toledo, Spain, 45004 Instituto Valenciano de Oncología Not yet recruiting Valencia, Spain, 46009 Hospital Clínico Universitario de Valencia Recruiting Valencia, Spain, 46010 Hospital Universitario y Politécnico La Fe Not yet recruiting Valencia, Spain, 46026 Hospital Universitario de Vigo- Hospital Álvaro Cunqueiro Recruiting Vigo, Spain, 36312 H. Universitario Miguel Servet, Zaragoza Not yet recruiting Zaragoza, Spain, 50009 Hospital Clínico Uiversitario Lozano Blesa Not yet recruiting Zaragoza, Spain, 50009 Collapse << |
| NCT01999972 | Advanced Solid Tumors | Phase 1 | Active, not recruiting | July 31, 2018 | United States, Illinois ... more >> University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Indiana Investigational Drug Services IUHSCC Indianapolis, Indiana, United States, 46202 IU Health University Hospital Indianapolis, Indiana, United States, 46202 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Michigan Wayne State University, Dept. of Oncology Detroit, Michigan, United States, 48201 United States, Minnesota University of Minnesota Health Clinics and Surgery Center Minneapolis, Minnesota, United States, 55455 University of Minnesota Medical Center, Fairview IDS Pharmacy Minneapolis, Minnesota, United States, 55455 University of Minnesota Medical Center, Fairview Minneapolis, Minnesota, United States, 55455 University of Minnesota Physicians Masonic Cancer Center Minneapolis, Minnesota, United States, 55455 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Utah Huntsman Cancer Hospital Salt Lake City, Utah, United States, 84112 Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 John A Moran Eye Center Salt Lake City, Utah, United States, 84132 United States, Wisconsin University Station Ophthalmology Clinic Madison, Wisconsin, United States, 53705 University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 United Kingdom St Bartholomew's Hospital - Barts Health NHS Trust London, United Kingdom, EC1A 7BE The Royal Marsden NHS Foundation Trust, Royal Marsden Hospital London, United Kingdom, SW3 6JJ Collapse << |
| NCT00768755 | Carcinoma, Non-Small Cell Lung | Phase 1 Phase 2 | Completed | - | - |
| NCT00219557 | - | - | Completed | - | - |
| NCT00447005 | Carcinoma | Phase 1 | Completed | - | Japan ... more >> Pfizer Investigational Site Kashiwa, Chiba, Japan Collapse << |
| NCT00768755 | - | - | Completed | - | - |
| NCT03172754 | Renal Cell Carcinoma | Phase 1 Phase 2 | Recruiting | April 2019 | United States, Pennsylvania ... more >> Fox Chase Cancer Center Recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: Matthew Zibelman, MD 215-214-1515 matthew.zibelman@fccc.edu Collapse << |
| NCT00735904 | Carcinoma, Non-Small-Cell Lung... more >> (NSCLC) Collapse << | Phase 2 | Completed | - | Poland ... more >> Pfizer Investigational Site Torun, Poland, 87-100 Pfizer Investigational Site Wodzislaw Sl., Poland, 44-300 Romania Pfizer Investigational Site Cluj-Napoca, Cluj, Romania, 400015 Pfizer Investigational Site Bucuresti, Romania, 022328 Pfizer Investigational Site Oradea, Romania, 410032 South Africa Pfizer Investigational Site Parktown, South Africa, 2193 Ukraine Pfizer Investigational Site Dnipropetrovsk, Ukraine, 49102 Pfizer Investigational Site Donetsk, Ukraine, 83092 Pfizer Investigational Site Kyiv, Ukraine, 04107 Pfizer Investigational Site Lviv, Ukraine, 79031 Collapse << |
| NCT00735904 | - | - | Completed | - | - |
| NCT02667886 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT00071006 | - | - | Completed | - | - |
| NCT01210495 | Hepatocellular Carcinoma | Phase 2 | Completed | - | - |
| NCT00282048 | Kidney Neoplasms ... more >> Carcinoma, Renal Cell Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> Pfizer Investigational Site Chicago, Illinois, United States, 60637 United States, New York Pfizer Investigational Site Bronx, New York, United States, 10466 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111-2497 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00569946 | Carcinoma, Renal Cell | Phase 2 | Completed | - | Japan ... more >> Pfizer Investigational Site Kashiwa, Chiba, Japan Pfizer Investigational Site Sapporo, Hokkaido, Japan Pfizer Investigational Site Tsukuba, Ibaraki, Japan Pfizer Investigational Site Morioka, Iwate, Japan Pfizer Investigational Site Nankoku-shi, Kochi-ken, Japan Pfizer Investigational Site Osakasayama, Osaka, Japan Pfizer Investigational Site Hamamatsu-City, Shizuoka, Japan Pfizer Investigational Site Sunto-gun, Shizuoka, Japan Pfizer Investigational Site Arakawa-ku, Tokyo, Japan Pfizer Investigational Site Chuo-ku, Tokyo, Japan Pfizer Investigational Site Itabashi-ku, Tokyo, Japan Pfizer Investigational Site Akita, Japan Pfizer Investigational Site Fukuoka, Japan Pfizer Investigational Site Kyoto, Japan Pfizer Investigational Site Osaka, Japan Pfizer Investigational Site Tokushima, Japan Pfizer Investigational Site Yamagata, Japan Collapse << |
| NCT00454649 | Neoplasms | Phase 1 | Completed | - | United States, Georgia ... more >> Pfizer Investigational Site Augusta, Georgia, United States, 30909 Pfizer Investigational Site Augusta, Georgia, United States, 30912 United States, Illinois Pfizer Investigational Site Harvey, Illinois, United States, 60426-4265 Pfizer Investigational Site Harvey, Illinois, United States, 60426 Pfizer Investigational Site Tinley Park, Illinois, United States, 60477 United States, Indiana Pfizer Investigational Site Hobart, Indiana, United States, 46342 Pfizer Investigational Site Munster, Indiana, United States, 46321 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19111 United States, Texas Pfizer Investigational Site Houston, Texas, United States, 77030-4009 United States, Washington Pfizer Investigational Site Kennewick, Washington, United States, 99336 Poland Pfizer Investigational Site Warszawa, Poland, 02-781 Spain Pfizer Investigational Site Barcelona, Spain, 08003 Pfizer Investigational Site Madrid, Spain, 28046 Collapse << |
| NCT00447005 | - | - | Completed | - | - |
| NCT00094094 | - | - | Completed | - | - |
| NCT00094094 | Lung Neoplasms ... more >> Carcinoma, Non-small Cell Lung Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Irvine, California, United States, 92612 Pfizer Investigational Site Orange, California, United States, 92868 United States, Illinois Pfizer Investigational Site Chicago, Illinois, United States, 60637 Pfizer Investigational Site Park Ridge, Illinois, United States, 60068 United States, Minnesota Pfizer Investigational Site Coon Rapids, Minnesota, United States, 55433 Pfizer Investigational Site Fridley, Minnesota, United States, 55432 Pfizer Investigational Site Robbinsdale, Minnesota, United States, 55422 United States, North Carolina Pfizer Investigational Site Charlotte, North Carolina, United States, 28203 United States, Tennessee Pfizer Investigational Site Nashville, Tennessee, United States, 37232 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 Germany Pfizer Investigational Site Gauting, Germany, 82131 Collapse << |
| NCT01210495 | - | - | Completed | - | - |
| NCT00282048 | - | - | Completed | - | - |
| NCT00842244 | Stomach Neoplasms ... more >> Advanced Gastric Cancer Collapse << | Phase 1 | Completed | - | Japan ... more >> Pfizer Investigational Site Kashiwa, Chiba, Japan Pfizer Investigational Site Yufu-city, Oita, Japan Korea, Republic of Pfizer Investigational Site Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Pfizer Investigational Site Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-707 Pfizer Investigational Site Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00569946 | - | - | Completed | - | - |
| NCT00842244 | - | - | Completed | - | - |
| NCT01649180 | - | - | Terminated(Closed due to prolo... more >>nged enrollment timelines) Collapse << | - | - |
| NCT02535533 | Advanced Metastatic Clear Cell... more >> Renal Cell Carcinoma (CCRCC) Collapse << | Phase 1 | Suspended(Suspended for interi... more >>m analysis) Collapse << | September 2020 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52242 Collapse << |
| NCT01441414 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | United States, Arizona ... more >> Pinnacle Oncology Hematology Scottsdale, Arizona, United States, 85258 Arizona Oncology Associates, PC - HOPE Tucson, Arizona, United States, 85704 Arizona Oncology Associates, PC-HOPE Tucson, Arizona, United States, 85710 United States, Colorado Rocky Mountain Cancer Centers Aurora, Colorado, United States, 80012 Rocky Mountain Cancer Centers Boulder, Colorado, United States, 80303 Rocky Mountain Cancer Centers Centennial, Colorado, United States, 80112 Rocky Mountain Cancer Centers Colorado Springs, Colorado, United States, 80907 Rocky Mountain Cancer Centers Colorado Springs, Colorado, United States, 80909 Rocky Mountain Cancer Centers Denver, Colorado, United States, 80218 Rocky Mountain Cancer Centers Denver, Colorado, United States, 80220 Rocky Mountain Cancer Centers Lakewood, Colorado, United States, 80228 Rocky Mountain Cancer Centers Littleton, Colorado, United States, 80120-4413 Rocky Mountain Cancer Centers Lone Tree, Colorado, United States, 80124 Rocky Mountain Cancer Centers Longmont, Colorado, United States, 80501 Rocky Mountain Cancer Centers Parker, Colorado, United States, 80138 Rocky Mountain Cancer Centers Pueblo, Colorado, United States, 81008 Rocky Mountain Cancer Centers Thornton, Colorado, United States, 80260 United States, Nebraska Nebraska Methodist Hospital Omaha, Nebraska, United States, 68114 United States, Nevada Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89014 Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89052 Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89074 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89128 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89148 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89169 United States, North Carolina Regional Cancer Care-Durham Durham, North Carolina, United States, 27704 United States, Texas Investigational Products Center (IPC) (drug shipment only) Fort Worth, Texas, United States, 76177 Texas Oncology-Tyler Tyler, Texas, United States, 75702 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 University of Washington Medical Center Seattle, Washington, United States, 98195 Czech Republic Masarykuv onkologicky ustav Brno, Czech Republic, 65653 Collapse << |
| NCT01441414 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01056263 | - | - | Completed | - | United States, California ... more >> Pfizer Investigational Site San Francisco, California, United States, 94115 United States, Massachusetts Pfizer Investigational Site Boston, Massachusetts, United States, 02114 Pfizer Investigational Site Boston, Massachusetts, United States, 02215 United States, New York Pfizer Investigational Site New York, New York, United States, 10065 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 United States, Wisconsin Pfizer Investigational Site Madison, Wisconsin, United States, 53792 France Pfizer Investigational Site Paris Cedex 13, France, 75651 Germany Pfizer Investigational Site Hannover, Germany, 30625 Collapse << |
| NCT01174238 | Melanoma | Phase 2 | Completed | - | United States, California ... more >> Cutaneous Onocology Group at the Helen Dillar Family Comprehensive Cancer Center UCSF San Francisco, California, United States, 94115 Collapse << |
| NCT01056263 | - | - | Completed | - | - |
| NCT01649180 | Renal Cell Carcinoma | Phase 2 | Terminated(Closed due to prolo... more >>nged enrollment timelines) Collapse << | - | United States, Nebraska ... more >> Missouri Valley Cancer Consortium Omaha, Nebraska, United States, 68106 United States, Ohio Cleveland Clinic, Taussig Cancer Institute Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania, Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00454649 | - | - | Completed | - | - |
| NCT00905814 | Healthy Volunteers | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT01540526 | Solid Malignancies ... more >> Metastatic Castrate-resistant Prostate Cancer Collapse << | Phase 1 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Carbone Cancer Center Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01174238 | - | - | Completed | - | - |
| NCT00920816 | Kidney Neoplasms | Phase 3 | Active, not recruiting | May 30, 2021 | - |
| NCT01490866 | - | - | Completed | - | - |
| NCT03736330 | Renal Cancer Metastatic | Phase 2 | Recruiting | November 8, 2021 | China, Guangdong ... more >> Cancer Center, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Fang-Jian Zhou, M.D Ph.D zhoufj@sysucc.org.cn Contact: Zhi-Ling Zhang, M.D Ph.D zhangzhl@sysucc.org.cn Principal Investigator: Fang-Jian Zhou, M.D Ph.D Principal Investigator: Jian-Chuan Xia, Ph.D Sub-Investigator: Zhi-Ling Zhang, M.D Ph.D Collapse << |
| NCT00828919 | Solid Tumors | Not Applicable | Active, not recruiting | May 31, 2021 | - |
| NCT00920816 | - | - | Active, not recruiting | - | - |
| NCT00835978 | Carcinoma, Renal Cell | Phase 2 | Completed | - | - |
| NCT01967576 | Pheochromocytoma ... more >> Paraganglioma Collapse << | Phase 2 | Active, not recruiting | December 1, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02853331 | Renal Cell Carcinoma | Phase 3 | Active, not recruiting | January 27, 2020 | - |
| NCT01490866 | Colorectal Cancer | Phase 2 | Completed | - | United States, Arkansas ... more >> NEA Baptist Clinic Jonesboro, Arkansas, United States, 72401 United States, Florida Florida Cancer Specialists-South Fort Myers, Florida, United States, 33916 Woodlands Medical Specialists Pensacola, Florida, United States, 32503 Florida Cancer Specialists-North St. Petersburg, Florida, United States, 33705 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Indiana Oncology Hematology of SW Indiana Newburgh, Indiana, United States, 47630 Hope Cancer Center Terre Haute, Indiana, United States, 47802 United States, Michigan Grand Rapids Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Nebraska Nebraska Methodist Hospital Omaha, Nebraska, United States, 68114 United States, New Jersey Atlantic Health System Summit, New Jersey, United States, 07901 United States, Tennessee Tennessee Oncology Nashville, Tennessee, United States, 37203 United States, Texas Texas Health Physician Group Dallas, Texas, United States, 75243 Collapse << |
| NCT01334073 | Malignant Advanced Solid Tumor... more >>s Carcinoma, Renal Cell Collapse << | Phase 1 | Completed | - | France ... more >> Professeur Alain RAVAUD Bordeaux, France, 33000 Professeur Jean-Pierre DELORD Toulouse, France, 31000 Collapse << |
| NCT00835978 | - | - | Completed | - | - |
| NCT00678392 | Kidney Neoplasms | Phase 3 | Completed | - | - |
| NCT01385059 | Stage III Prostate Cancer ... more >> Stage IV Prostate Cancer Collapse << | Phase 2 | Active, not recruiting | November 2019 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << |
| NCT00678392 | - | - | Completed | - | - |
| NCT01352728 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Hong Kong ... more >> Department of Clinical Oncology, Prince of Wales Hospital Hong Kong, Hong Kong Collapse << |
| NCT02133742 | Renal Cell Carcinoma | Phase 1 | Active, not recruiting | February 2019 | United States, District of Col... more >>umbia Georgetown University Medical Center Washington, District of Columbia, United States, 20007 United States, Florida H. Lee Moffitt Cancer Center & Research Institute, Inc. Tampa, Florida, United States, 33612 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Drug Shipment Address: Attn. Alicia Sammarco, RPh, NYU Investigational Pharmacy New York, New York, United States, 10016 NYU Langone Medical Center New York, New York, United States, 10016 NYU Langone New York, New York, United States, 10016 United States, Ohio Investigational Drug Services (Drug Management Only) Columbus, Ohio, United States, 43210 James Cancer Hospital Columbus, Ohio, United States, 43210 The James Cancer Hospital Columbus, Ohio, United States, 43210 Martha Morehouse Medical Plaza Columbus, Ohio, United States, 43221 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, Tennessee Henry-Joyce Cancer Clinic Nashville, Tennessee, United States, 37232 United States, Texas Texas Oncology - Baylor Charles A. Sammons Cancer Center Dallas, Texas, United States, 75246 Collapse << |
| NCT00919204 | Healthy Volunteers | Phase 1 | Completed | - | China ... more >> Pfizer Investigational Site Beijng, China, 100730 Collapse << |
| NCT02636725 | Alveolar Soft Part Sarcoma ... more >> Soft Tissue Sarcomas Collapse << | Phase 2 | Recruiting | March 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Breelyn Wilky, MD 305-243-1287 b.wilky@med.miami.edu Principal Investigator: Breelyn Wilky, MD Sub-Investigator: Jonathan Trent, MD Collapse << |
| NCT01486251 | Colorectal Carcinoma | Not Applicable | Completed | - | France ... more >> Institut gustave roussy Villejuif, France, 94805 Collapse << |
| NCT03092856 | Clear Cell Renal Cell Carcinom... more >>a Metastatic Renal Cell Cancer Recurrent Renal Cell Carcinoma Stage IV Renal Cell Cancer Collapse << | Phase 2 | Recruiting | July 19, 2021 | United States, California ... more >> USC / Norris Comprehensive Cancer Center Recruiting Los Angeles, California, United States, 90033 Contact: Sarmad Sadeghi, MD 323-865-0553 sarmad.sadeghi@usc.edu Principal Investigator: Sarmad Sadeghi University of California Davis Comprehensive Cancer Center Not yet recruiting Sacramento, California, United States, 95817 Contact: Primo N. Lara 916-734-3089 Principal Investigator: Primo N. Lara United States, Ohio Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Brian I. Rini 866-223-8100 Principal Investigator: Brian I. Rini Ohio State University Comprehensive Cancer Center Not yet recruiting Columbus, Ohio, United States, 43210 Contact: Amir Mortazavi 614-293-6196 Principal Investigator: Amir Mortazavi United States, Pennsylvania University of Pittsburgh Cancer Institute (UPCI) Not yet recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: Rahul A. Parikh 412-647-8073 parikhr@upmc.edu Principal Investigator: Rahul A. Parikh Collapse << |
| NCT02156895 | - | - | Recruiting | August 21, 2021 | Korea, Republic of ... more >> Pfizer Tower Recruiting Seoul, Korea, Republic of, 04631 Collapse << |
| NCT01529138 | Cancer | Phase 1 | Completed | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01473043 | - | - | - | - | Australia, Queensland ... more >> Pfizer Investigational Site Douglas, Queensland, Australia, 4814 Australia, Tasmania Pfizer Investigational Site Hobart, Tasmania, Australia, 7000 Australia, Victoria Pfizer Investigational Site Wendouree, Victoria, Australia, 3355 Canada, Quebec Pfizer Investigational Site Montreal, Quebec, Canada, H2X 1N8 Collapse << |
| NCT01249547 | Recurrent or Metastatic NPC | Phase 2 | Completed | - | Hong Kong ... more >> Department of Clinical Oncology, Prince of Wales Hospital Hong Kong, Hong Kong Collapse << |
| NCT01483638 | Colorectal Carcinoma | Phase 2 | Terminated(The trial was prema... more >>turely closed due to lack of accrual) Collapse << | - | Spain ... more >> Spanish Cooperative Group for Digestive Tumour Therapy Madrid, Spain, 28046 Collapse << |
| NCT01255137 | Adrenal Cortex Neoplasms | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03289533 | Carcinoma, Hepatocellular | Phase 1 | Recruiting | October 7, 2019 | Japan ... more >> Aichi Cancer Center Hospital Recruiting Nagoya, Aichi, Japan, 464-8681 Iizuka Hospital Active, not recruiting Iizuka, Fukuoka, Japan, 820-8505 Kindai University Hospital, Department of Gastroenterology and Hepatology Active, not recruiting Osaka-Sayama, Osaka, Japan, 589-8511 National Cancer Center Hospital Recruiting Chuo-ku, Tokyo, Japan, 104-0045 Kyorin University Hospital, Department of Medical Oncology Active, not recruiting Mitaka-shi, Tokyo, Japan, 181-8611 Japanese Red Cross Musashino Hospital Active, not recruiting Musashino, Tokyo, Japan, 180-8610 National Hospital Organization Kyushu Medical Center Recruiting Fukuoka, Japan, 810-8563 Collapse << |
| NCT01255137 | - | - | Completed | - | - |
| NCT01321437 | Melanoma Mali... more >>gnant Melanoma Stage IIIA Melanoma Stage IIIB Melanoma Stage IIIC Melanoma Collapse << | Phase 2 | Active, not recruiting | December 2019 | United States, California ... more >> Chao Family Comprehensive Cancer Center Orange, California, United States, 92868 Collapse << |
| NCT03595124 | Metastatic Renal Cell Carcinom... more >>a Renal Cell Carcinoma Associated With Xp11.2 Translocations/TFE3 Gene Fusions Stage III Renal Cell Cancer AJCC v8 Stage IV Renal Cell Cancer AJCC v8 TFE3 Gene Translocation TFEB Gene Translocation Collapse << | Phase 2 | Recruiting | June 18, 2023 | - |
| NCT01409200 | Prostate Cancer | Phase 2 | Active, not recruiting | February 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01508117 | - | - | Terminated(transferred study s... more >>ponsor) Collapse << | - | - |
| NCT01508117 | Glioblastoma Multiforme | Phase 2 | Terminated(transferred study s... more >>ponsor) Collapse << | - | United States, Ohio ... more >> University of Cincinnati Cincinnati, Ohio, United States, 45267-0502 Collapse << |
| NCT01727336 | Advanced Renal Cell Carcinoma | Phase 2 | Active, not recruiting | December 2018 | - |
| NCT01744249 | Neuroendocrine Tumors ... more >> Advanced Cancer Collapse << | Phase 2 Phase 3 | Recruiting | October 2020 | Germany ... more >> Berlin Charité Universitätsmedizin Recruiting Berlin, Germany Principal Investigator: Frank-Ulrich Pape, Dr. Marburg Universitätsklinikum Giessen und Marburg GmbH Recruiting Marburg, Germany, 35043 Principal Investigator: Anja Rinke, Dr. Klinikum rechts der Isar Recruiting München, Germany, 81675 Principal Investigator: Alexander Von Werder, Dr. Italy Azienda Ospedaliera Universitaria di Perugia Recruiting Perugia, Italy, 06129 Principal Investigator: Vicenzo Minotti, Dr. Sapienza, Universitá di Roma, Ospedale sant'Andrea Recruiting Rome, Italy, 00189 Principal Investigator: Francesco Panzuto, Dr. Spain Hospital Central de Asturias Recruiting Oviedo, Asturias, Spain Principal Investigator: Paula Jiménez Fonseca, MD Institut Català d'Oncologia L'Hospitalet Recruiting L'Hospitalet de Llobregat, Barcelona, Spain Principal Investigator: Alexandre Teulé, MD Hospital Universitario Virgen de la Victoria Recruiting Málaga, Malaga, Spain, 29010 Principal Investigator: Isabel Sevilla, MD Hospital Alvaro Cunqueiro Recruiting Vigo, Pontevedra, Spain, 36312 Principal Investigator: M. José Villanueva Silva, MD Complejo Hospitalario Univ A Coruña Recruiting A Coruña, Spain Principal Investigator: Luis antón Aparicio, MD Sub-Investigator: María Quindós, MD Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain Principal Investigator: Jaume Capdevila, MD Hospital Universitario de Burgos Recruiting Burgos, Spain Contact: Guillermo Crespo Herrero, MD Principal Investigator: Guillermo Crespo Herrero, MD Hospital Virgen de las Nieves Recruiting Granada, Spain Principal Investigator: Encarnación González Flores, MD Hospital universitario de Leon Suspended Leon, Spain MD Anderson Cancer Center Recruiting Madrid, Spain, 28033 Principal Investigator: Enrique Grande Pulido, MD Hospital Clara Campal Suspended Madrid, Spain Hospital Clínico San Carlos Recruiting Madrid, Spain Principal Investigator: Javier Sastre Hospital Gregorio Marañón Recruiting Madrid, Spain Principal Investigator: Pilar García Alfonso, MD Hospital Univ La Paz Recruiting Madrid, Spain Principal Investigator: Ana Custodio, MD Hospital Universitario 12 de Octubre Recruiting Madrid, Spain Principal Investigator: Daniel Castellano, MD Hospital Universitario Ramón y Cajal Recruiting Madrid, Spain Principal Investigator: Teresa Alonso Gordoa, MD Hospital Univ de Salamanca Recruiting Salamanca, Spain Principal Investigator: Miguel Navarro, MD Hospital de Donostia Recruiting San Sebastian, Spain Principal Investigator: Adelaida La Casta, MD Sub-Investigator: Sara Arévalo, MD Hospital Marqués de Valdecilla Recruiting Santander, Spain Principal Investigator: Carlos López, MD Hospital Universitario Virgen del Rocío Recruiting Sevilla, Spain Principal Investigator: Marta Benavent, MD Hospital General Universitario de Valencia Recruiting Valencia, Spain, 46014 Principal Investigator: Vega Iranzo, MD Hospital La Fe Withdrawn Valencia, Spain Hospital Universitario Miguel Servet Recruiting Zaragoza, Spain Contact: Vicente Alonso, M.D. Principal Investigator: Vicente Alonso, M.D. United Kingdom Clatterbridge Cancer Centre Suspended Bebington, Wirral, United Kingdom, CH63 4JY Queen Elisabeth Hospital Birmingham Not yet recruiting Birmingham, United Kingdom, B15 2TH Principal Investigator: Tahir Shah, Dr. Royal Free Hospital Recruiting London, United Kingdom, NW3 2QG Principal Investigator: Christine Thirlwell, Prof. Christie NHS Foundation Trust Not yet recruiting Manchester, United Kingdom, MB204BX Principal Investigator: Juan Valle, Prof. Collapse << |
| NCT02684006 | Renal Cell Cancer | Phase 3 | Active, not recruiting | April 30, 2021 | - |
| NCT03291314 | Recurrent Glioblastoma (WHO-Gr... more >>ade IV Glioma) Collapse << | Phase 2 | Recruiting | September 30, 2018 | Belgium ... more >> UZ Brussel Recruiting Brussel, Belgium, 1090 Contact: Bart Neyns, Phd,Md +32 2 477 ext 54 47 bart.neyns@uzbrussel.be Principal Investigator: Bart Neyns, Phd,Md Collapse << |
| NCT02762513 | Squamous Cell Carcinoma of the... more >> Head and Neck Collapse << | Phase 2 | Recruiting | November 2020 | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Cancer Answer Line 800-865-1125 Principal Investigator: Francis Worden, M.D. Collapse << |
| NCT01273662 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei City, Taiwan, 100 Taipei Veterans General Hospital Taipei City, Taiwan, 11217 Tri-Service General Hospital Taipei City, Taiwan, 114 Collapse << |
| NCT01469546 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01140737 | Soft Tissue Sarcoma | Phase 2 | Active, not recruiting | December 2018 | United Kingdom ... more >> Aberdeen Royal Infirmary Aberdeen, Scotland, United Kingdom Western General Hospital Edinburgh, Scotland, United Kingdom Bristol Haematology & Oncology Centre Bristol, United Kingdom St. James's Hospital Leeds, United Kingdom Royal Marsden Hospital London, United Kingdom Royal Marsden London, United Kingdom University College London Hospitals London, United Kingdom Christies Manchester, United Kingdom Nottingham City Hospital Nottingham, United Kingdom Churchill Hospital Oxford, United Kingdom Penella Woll Sheffield, United Kingdom Southampton General Hospital Southampton, United Kingdom Clatterbridge Centre for Oncology Wirral, United Kingdom Collapse << |
| NCT01469546 | - | - | Completed | - | - |
| NCT00859118 | Neoplasms | Phase 1 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Paul P. Carbone Comprehensive Cancer Center Madison, Wisconsin, United States, 53575 Collapse << |
| NCT01798446 | Renal Cell Carcinoma ... more >> Nonclear Cell Temsirolimus Resistance Collapse << | Phase 2 | Unknown | December 2016 | Korea, Republic of ... more >> Seou National University Hospital Recruiting Seoul, Korea, Republic of, 110744 Contact: Se-Hoon Lee, MD, PhD 82220722199 shlee119@snu.ac.kr Principal Investigator: Se-Hoon Lee, MD, PhD Sub-Investigator: Dae Seog Heo, MD, PhD Sub-Investigator: Dong-Wan Kim, MD, PhD Sub-Investigator: Tae Min Kim, MD, PhD Sub-Investigator: Bhumsuk Keam, MD Collapse << |
| NCT01211275 | Malignant Pleural Mesothelioma | Phase 1 Phase 2 | Completed | - | Netherlands ... more >> Antoni van Leeuwenhoekziekenhuis (NKI-AVL) Amsterdam, Noord-Holland, Netherlands, 1066 CX Collapse << |
| NCT01558661 | - | - | Completed | - | - |
| NCT01263769 | Kidney Cancer | Phase 2 | Active, not recruiting | February 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01599754 | Clear Cell Renal Carcinoma | Phase 3 | Terminated(Primary endpoint di... more >>d not reach statistical significance) Collapse << | - | - |
| NCT02493751 | Renal Cell Cancer | Phase 1 | Active, not recruiting | January 22, 2020 | United States, Arizona ... more >> Oncology Research Associates, PLLC d/b/a Pinnacle Oncology Hematology Scottsdale, Arizona, United States, 85258 United States, District of Columbia Georgetown University Medical Center Department of Pharmacy, Research Washington, District of Columbia, United States, 20007 Georgetown University Medical Center Washington, District of Columbia, United States, 20007 United States, Massachusetts Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, New York Drug Shipment address: Attn. Alicia Sammarco, RPh,NYU Investigational Pharmacy New York, New York, United States, 10016 Laura & Isaac Perlmutter Cancer Center At NYU Langone New York, New York, United States, 10016 NYU Langone Medical Center New York, New York, United States, 10016 United States, Ohio Cleveland Clinic Taussig Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Tennessee Henry Joyce Cancer Clinic Nashville, Tennessee, United States, 37232-5310 Vanderbilt University Oncology Pharmacy (ship drug only) Nashville, Tennessee, United States, 37232 United States, Utah University of Utah, Huntsman Cancer Hospital Salt Lake City, Utah, United States, 84112 University of Utah, Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Japan Niigata University Medical & Dental Hospital Chuo-ku, Niigata, Niigata, Japan, 951-8520 Kindai University Hospital Osakasayama, Osaka, Japan, 589-8511 Keio University Hospital Shinjuku-ku, Tokyo, Japan, 160-8582 United Kingdom Mount Vernon Cancer Center, East and North Herts. NHS Trust London, Middlesex, United Kingdom, HA6 2RN St Helier Hospital Carshalton, Surrey, United Kingdom, SM5 1AA St. Bartholomew's Hospital London, United Kingdom, EC1A 7BE The Royal Marsden NHS Foundation Trust London, United Kingdom, SW3 6JJ The Christie NHS Foundation Trust Manchester, United Kingdom, M20 4BX The Royal Marsden NHS Foundation Trust Sutton, United Kingdom, SM2 5PT Collapse << |
| NCT03341845 | Renal Cell Carcinoma | Phase 2 | Recruiting | January 31, 2025 | Netherlands ... more >> Antoni van Leeuwenhoek Recruiting Amsterdam, Netherlands, 1066CX Contact: Axel Bex, MD, PhD 0205129111 a.bex@nki.nl Collapse << |
| NCT01533948 | Extraocular Extension Melanoma... more >> Metastatic Intraocular Melanoma Recurrent Intraocular Melanoma Recurrent Melanoma Stage IIIA Intraocular Melanoma Stage IIIA Melanoma Stage IIIB Intraocular Melanoma Stage IIIB Melanoma Stage IIIC Intraocular Melanoma Stage IIIC Melanoma Stage IV Intraocular Melanoma Stage IV Melanoma Collapse << | Phase 2 | Terminated(low accrual) | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT01558661 | Adenoid Cystic Carcinoma | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01533948 | - | - | Terminated(low accrual) | - | - |
| NCT01806064 | Renal Cell Carcinoma | Phase 1 Phase 2 | Active, not recruiting | June 2019 | - |
| NCT01435122 | Carcinoid Tumor | Phase 2 | Completed | - | United States, California ... more >> University of California San Francisco (UCSF) San Francisco, California, United States, 94115 United States, Florida H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT02639182 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Active, not recruiting | January 2019 | - |
| NCT01435122 | - | - | Completed | - | - |
| NCT01334112 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Canada, Alberta ... more >> Foothills Hospital, Alberta Health Services Calgary, Alberta, Canada Canada, Ontario University Health Network, Princess Margaret Hospital Toronto, Ontario, Canada Collapse << |
| NCT02859012 | Recurrent ACC, metastaticACC, ... more >>Unreaectable ACC Collapse << | Phase 2 | Active, not recruiting | August 2020 | Korea, Republic of ... more >> Department of Internal Medicine, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT03472560 | Non-Small Cell Lung Cancer ... more >> Urothelial Cancer Collapse << | Phase 2 | Recruiting | September 18, 2020 | United States, Arizona ... more >> Arizona Oncology Associates- Saguaro Cancer Center Recruiting Glendale, Arizona, United States, 85308 Arizona Oncology Associates- Biltmore Cancer Center Recruiting Phoenix, Arizona, United States, 85016 Arizona Oncology Associates- Deer Valley Cancer Center Recruiting Phoenix, Arizona, United States, 85027 Arizona Oncology Associates- East Valley Cancer Center Recruiting Tempe, Arizona, United States, 85284 United States, Michigan Karmanos Cancer Institute Not yet recruiting Detroit, Michigan, United States, 48201 Karmanos Cancer Institute Not yet recruiting Farmington Hills, Michigan, United States, 48334 United States, Missouri Oncology Hematology Associates Recruiting Springfield, Missouri, United States, 65807 United States, Nebraska Oncology Hematology West, PC dba Nebraska Cancer Specialists Recruiting Omaha, Nebraska, United States, 68114 Oncology Hematology West, PC dba Nebraska Cancer Specialists Recruiting Omaha, Nebraska, United States, 68130 United States, Ohio Toledo Clinic Cancer Center- Toledo Not yet recruiting Toledo, Ohio, United States, 43623 United States, South Carolina Saint Francis Hospital Recruiting Greenville, South Carolina, United States, 29601 Saint Francis Hospital Cancer Center Recruiting Greenville, South Carolina, United States, 29607 Hungary Orszagos Onkologiai Intezet Not yet recruiting Budapest, Hungary, H-1122 Pecsi Tudomanyegyetem Klinikai Kozpont Not yet recruiting Pecs, Hungary, H-7624 Tudogyogyintezet Torokbalint Recruiting Torokbalint, Hungary, H-2045 Korea, Republic of National Cancer Center Recruiting Goyang-si, Gyeonggi-do, Korea, Republic of, 10408 Samsung Medical Center Recruiting Gangnam-gu, Seoul, Korea, Republic of, 06351 Asan Medical Center Not yet recruiting Songpa-gu, Seoul, Korea, Republic of, 05505 Severance Hospital, Yonsei University Health System Recruiting Seoul, Korea, Republic of, 03722 Taiwan Chi Mei Hospital, Liouying Recruiting Tainan City, Liouying District, Taiwan, 73657 National Cheng Kung University Hospital Not yet recruiting Tainan, Taiwan, 704 Collapse << |
| NCT02579811 | Metastatic Renal Cell Cancer | Phase 2 | Active, not recruiting | September 2019 | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 United States, Ohio Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 The Ohio State University Comprehensive Cancer Center - Arthur G. James Cancer Hospital and Richard J. Solove Research Institute Columbus, Ohio, United States, 43210 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT02782403 | Leukemia | Phase 1 Phase 2 | Recruiting | March 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact pbose@mdanderson.org Collapse << |
| NCT02597322 | cT2a N0NxM0 Renal Tumor | Phase 2 | Completed | - | France ... more >> Bicêtre Hospital Le Kremlin Bicêtre, France, 94275 Collapse << |
| NCT01693822 | Clear-cell Metastatic Renal Ce... more >>ll Carcinoma Collapse << | Phase 2 | Unknown | September 2016 | United Kingdom ... more >> Royal Marsden Hospital - sutton Recruiting London, Sutton, United Kingdom, SM2 5PT Principal Investigator: James Larkin, Dr Addenbrooke's Hospital Recruiting Cambridge, United Kingdom, CB2 0QQ Principal Investigator: Timothy Eisen, Prof Royal Marsden Hospital Recruiting London, United Kingdom, SW3 6JJ Principal Investigator: James Larkin, Dr Christie Hospital Recruiting Manchester, United Kingdom, M20 4BX Principal Investigator: Robert Hawkins, Prof Derriford Hospital Recruiting Plymouth, United Kingdom, PL6 8DH Principal Investigator: Martin Highley, Dr Royal Surrey County Hospital Recruiting Surrey, United Kingdom, GU2 7XX Principal Investigator: Agnieszka Michael, Dr Collapse << |
| NCT01562197 | Glioblastoma Multiforme | Phase 2 | Active, not recruiting | December 2017 | Belgium ... more >> UZ Brussel Brussel, Belgium, 1090 Collapse << |
| NCT02489695 | Papillary Renal Cell Carcinoma | Phase 2 | Active, not recruiting | January 2019 | France ... more >> ICO Paul Papin Angers, France, 49933 Chu Bordeaux Bordeaux, France, 33075 Centre Francois Baclesse Caen, France, 14076 Centre Geogres François Leclerc Dijon, France, 21079 Centre Leon Berard Lyon, France, 69373 Institut Paoli Calmettes Marseille, France, 13273 ICO - René Gauducheau Saint-herblain, France, 44805 Institut Claudius Regaud Toulouse, France, 31059 ICL Vandoeuvre-les-nancy, France, 54519 Gustave Roussy Villejuif, France, 94805 Collapse << |
| NCT02261207 | Solitary Fibrous Tumor | Phase 2 | Unknown | March 2018 | Italy ... more >> Fondazione IRCCS Istituto Naazionale dei Tumori Not yet recruiting Milan, MI, Italy, 20133 Contact: Silvia Stacchiotti, MD +39 022390 ext 2807 silvia.stacchiotti@istitutotumori.mi.it Principal Investigator: Silvia Stacchiotti, MD Collapse << |
| NCT02164838 | Refractory or Recurrent Solid ... more >>Tumors, Excluding CNS Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT02700568 | Carcinoma, Renal Cell ... more >> Clear Cell Renal Cell Carcinoma Collapse << | Phase 2 | Recruiting | August 2018 | Russian Federation ... more >> Demchenkova Marina Viktorovna Recruiting Irkutsk, Russian Federation Semenov Andrey Vladimirovich Recruiting Ivanovo, Russian Federation Mikhailova Nadezhda Vasilievna Recruiting Kazan, Russian Federation Eskerov Kurban Abdulmutalibovich Active, not recruiting Kirov, Russian Federation Zukov Ruslan Aleksandrovich Recruiting Krasnoyarsk, Russian Federation Ovchinnikova Elena Georgievna Recruiting Nizhny Novgorod, Russian Federation Guseva Irina Vasilievna Recruiting Penza, Russian Federation Vladimirova Lyubov Yur'evna Active, not recruiting Rostov-on-Don, Russian Federation Zolotoreva Tatiana Gennadievna Recruiting Samara, Russian Federation Katkov Alexey Aleksandrovich Active, not recruiting Saratov, Russian Federation Ivannikov Andrey Andreyevich Recruiting Tambov, Russian Federation Usynin Evgeny Anatolievich Recruiting Tomsk, Russian Federation Evstegneyeva Irina Vladimirovna Recruiting Tver, Russian Federation Khmelevsky Andrey Anatolievich Recruiting Ufa, Russian Federation Gurina Ludmila Ivanovna Active, not recruiting Vladivostok, Russian Federation Collapse << |
| NCT03592199 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | December 11, 2022 | Brazil ... more >> Instituto do Cancer do Estado de São Paulo Recruiting São Paulo, Brazil, 01246000 Contact: David QB Muniz, MD +551138932691 david.muniz@hc.fm.usp.br Contact: Diogo A Bastos, MD +551138932691 diogo.bastos@hc.fm.usp.br Principal Investigator: David QB Muniz, MD Sub-Investigator: Carlos Dzik, MD, PHD Sub-Investigator: Diogo A Bastos, MD Sub-Investigator: Jamile A Silva, MD Sub-Investigator: Pedro HI Velho, MD Collapse << |
| NCT02857712 | Salivary Gland Cancers | Phase 2 | Active, not recruiting | June 2019 | Italy ... more >> Fondazione IRCCS Istituto Nazionale dei Tumori Milan, Italy, 20133 Collapse << |
| NCT03494816 | Renal Cell Carcinoma ... more >> Metastatic Renal Cell Carcinoma Non-metastatic Renal Cell Carcinoma Collapse << | Phase 2 | Recruiting | June 1, 2020 | United Kingdom ... more >> Addenbrookes Hospital Recruiting Cambridge, United Kingdom, CB2 0QQ Contact: Ellie Couch Principal Investigator: Kate Fife Western General Hospital Recruiting Edinburgh, United Kingdom, EH4 2XU Contact: Susan Forman Principal Investigator: Steve Leung Beatson West of Scotland Cancer Centre Recruiting Glasgow, United Kingdom, G12 0YN Contact: Maria Nicol Principal Investigator: Rob Jones St George's Hospital Not yet recruiting London, United Kingdom, SW17 0QT Contact: Sam Hollingworth Principal Investigator: Pieter Le Roux Royal Marsden Recruiting London, United Kingdom, SW3 6JJ Contact: Darshana Shah Principal Investigator: David Nicol The Christie Not yet recruiting Manchester, United Kingdom, M20 4BX Contact: Kate Armitage Principal Investigator: Tom Waddell Collapse << |
| NCT02129647 | Neurofibromatosis Type 2 ... more >> Vestibular Schwannomas Collapse << | Phase 2 | Recruiting | January 2019 | United States, New York ... more >> NYU Langone Medical Center Recruiting New York, New York, United States, 10016 Contact: Srivandana Akshintala, MD 212-263-8400 Srivandana.Akshintala@nyumc.org Principal Investigator: Srivandana A Akshintala, MD Sub-Investigator: Jeffrey Allen, MD Sub-Investigator: Katharine McNeill, MD Collapse << |
| NCT02814461 | Advanced Hepatocellular Carcin... more >>oma (HCC) Collapse << | Phase 1 | Recruiting | November 2018 | Taiwan ... more >> Shin Kong Wu Ho-Su Memorial Hospital Recruiting Taipei, Taiwan, 11101 Contact: Kai-Lin Yang, M.D. +886-28332211 ext 2275 M011360@ms.skh.org.tw Collapse << |
| NCT03438708 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | February 1, 2020 | United States, California ... more >> UC San Diego Moores Cancer Center Recruiting La Jolla, California, United States, 92093 Contact: Candace M Winkler, MS 858-822-5398 cmwinkler@ucsd.edu Contact: Arlene Araneta 8588226187 aaraneta@ucsd.edu Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| c-Kit | IC50:1.7nM | |
| PDGFRβ | IC50:1.6nM | |
| VEGFR1 | IC50:0.1nM | |
| VEGFR2 | IC50:0.2nM | |
| VEGFR3 | IC50:0.1nM-0.3nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
