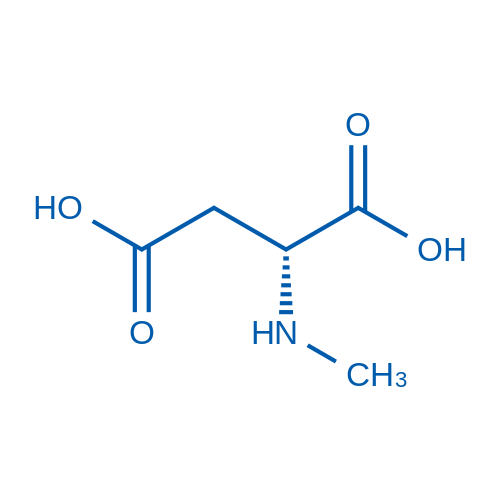
CAS No.: 6384-92-5
N-甲基-D-天冬氨酸 Catalog No. CSN13790
Synonyms: N-Methyl-D-aspartic acid;LC-488A;N-甲基-D-天冬氨酸
NMDA is an amino acid derived specific agonist at the NMDA receptor.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03457961 - - Recruiting July 1, 2019 Hong Kong ... more >> Prince of Wales Hospital Recruiting Hong Kong, Hong Kong, 0000 Contact: Ka Lai YIP +85235053856 Hannahykl@cuhk.edu.hk Principal Investigator: Ho Wan Leung Collapse << NCT00240760 Down's Syndrome ... more >> Dementia Learning Disabilities Collapse << Not Applicable Unknown - United Kingdom ... more >> Northgate Hospital Recruiting Morpeth, Northumberland, United Kingdom, NE61 3BP Contact: Maria Luisa Margallo-Lana, PhD 0044 (0) 1670394000 ext 4079 lana@onetel.com Principal Investigator: Maria Luisa Margallo-Lana, PhD Monyhull Hall Road Recruiting Birmingham, United Kingdom, B30 3QQ Contact: Verinder Prasher, PhD 0044 (0) 121 255 8013 vprasher@compuserver Principal Investigator: Verinder Prasher, PhD Collapse << NCT02532686 Schizophrenia Phase 2 Completed - Taiwan ... more >> Department of Psychiatry, China Medical University Hospital Taichung, Taiwan Collapse << - 更多
- 参考文献
- [1] Cull-Candy S, Brickley S, Farrant M. NMDA receptor subunits: diversity, development and disease. Curr Opin Neurobiol. 2001 Jun;11(3):327-35.
- [2] Yoneda Y, Ogita K. Enhancement of [3H] glutamate binding by N-methyl-D-aspartic acid in rat adrenal. Brain Res. 1987 Mar 17;406(1-2):24-31.
- [3] Yoneda Y, Ogita K. Enhancement of [3H]glutamate binding by N-methyl-D-aspartic acid in rat adrenal. Brain Res. 1987 Mar 17;406(1-2):24-31
- [4] Lapeikaite I, Pupkis V, Neniskis V, Ruksenas O, Kisnieriene V. Glutamate and NMDA affect cell excitability and action potential dynamics of single cell of macrophyte Nitellopsis obtusa. Funct Plant Biol. 2020 Nov;47(12):1032-1040
- [5] Ghislandi AB, Garcez ML, Zambon GM, Constantino LC, Matos DN, Pescador BB, Tasca CI, Boeck CR. Adenosine and NMDA Receptors Modulate Neuroprotection-Induced NMDA Preconditioning in Mice. J Mol Neurosci. 2020 Apr;70(4):590-599
- [6] Kleshchevnikov AM. Dlitel'naia potentsiatsiia NMDA-zavisimogo komponenta VPSP v gippokampe [Long-term potentiation of the NMDA-dependent component of the EPSP in the hippocampus]. Usp Fiziol Nauk. 1998 Oct-Dec;29(4):6-23. Russian
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 6384-92-5 | 储存条件 |
|
|||||
| 分子式 | C5H9NO4 | 运输 | 蓝冰 | |||||
| 分子量 | 147.13 | 别名 | N-Methyl-D-aspartic acid;LC-488A;N-甲基-D-天冬氨酸 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03457961 | - | - | Recruiting | July 1, 2019 | Hong Kong ... more >> Prince of Wales Hospital Recruiting Hong Kong, Hong Kong, 0000 Contact: Ka Lai YIP +85235053856 Hannahykl@cuhk.edu.hk Principal Investigator: Ho Wan Leung Collapse << |
| NCT00240760 | Down's Syndrome ... more >> Dementia Learning Disabilities Collapse << | Not Applicable | Unknown | - | United Kingdom ... more >> Northgate Hospital Recruiting Morpeth, Northumberland, United Kingdom, NE61 3BP Contact: Maria Luisa Margallo-Lana, PhD 0044 (0) 1670394000 ext 4079 lana@onetel.com Principal Investigator: Maria Luisa Margallo-Lana, PhD Monyhull Hall Road Recruiting Birmingham, United Kingdom, B30 3QQ Contact: Verinder Prasher, PhD 0044 (0) 121 255 8013 vprasher@compuserver Principal Investigator: Verinder Prasher, PhD Collapse << |
| NCT02532686 | Schizophrenia | Phase 2 | Completed | - | Taiwan ... more >> Department of Psychiatry, China Medical University Hospital Taichung, Taiwan Collapse << |
| NCT01856556 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Lotus Clinical Research Pasadena, California, United States, 91105 Collapse << |
| NCT00040261 | Depression | Phase 3 | Completed | - | United States, Maryland ... more >> National Institute of Mental Health (NIMH) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00246415 | Alcohol Dependence (Primary Co... more >>ndition) Alcohol Abuse Collapse << | Phase 2 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT01401673 | Cough | Phase 2 | Terminated(Lack of efficacy) | - | United Kingdom ... more >> South Manchester University Hospital Manchester, United Kingdom, M23 9LT Collapse << |
| NCT01507181 | - | - | Completed | - | - |
| NCT00986479 | Treatment Resistant Major Depr... more >>essive Disorder Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Research Site Bethesda, Maryland, United States Collapse << |
| NCT01507181 | Suicidal Ideation | Phase 4 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10027 Collapse << |
| NCT03583554 | Healthy | Phase 1 Phase 2 | Recruiting | March 31, 2019 | United States, Texas ... more >> Michael E. DeBakey VA Medical Center Recruiting Houston, Texas, United States, 77030 Contact: Marijn Lijffijt, PhD 713-798-5642 marijn.lijffijt@bcm.edu Sub-Investigator: Sanjay J. Mathew, MD Sub-Investigator: Alan C. Swann, MD Collapse << |
| NCT00690170 | Healthy Subjects | Phase 1 | Completed | - | United States, Connecticut ... more >> Veterans Administration Hospital West Haven, Connecticut, United States, 06516 Collapse << |
| NCT02589457 | Chronic Hepatitis B | Phase 1 | Completed | - | Korea, Republic of ... more >> Chungnam National University Hospital Daejeon, Jung-gu, Korea, Republic of, 301-721 Collapse << |
| NCT00986479 | - | - | Completed | - | - |
| NCT02649231 | Primary Alcohol Use Disorder | Phase 2 | Recruiting | December 2021 | United Kingdom ... more >> NIHR Exeter Clinical Research Facility Recruiting Exeter, United Kingdom, EX4 5DW Contact: Amy McAndrew, PhD 01392 724070 a.mcandrew@exeter.ac.uk NIHR/Wellcome UCLH Research Facility Recruiting London, United Kingdom, NW1 2BU Contact: Beth Marsh, PhD 02076798275 b.marsh@ucl.ac.uk Collapse << |
| NCT00486902 | Ketamine Adverse Reaction ... more >> Effects of; Anesthesia, Spinal and Epidural, in Pregnancy Complication of Labor and/or Delivery Collapse << | Not Applicable | Completed | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT02579928 | Major Depressive Disorder ... more >> Anxiety Disorder Collapse << | Phase 4 | Active, not recruiting | June 2019 | United States, Connecticut ... more >> Hospital Research Unit at the Yale New Haven Hospital New Haven, Connecticut, United States, 06510 Yale Child Study Center New Haven, Connecticut, United States, 06520 Collapse << |
| NCT00408031 | Major Depressive Disorder | Phase 2 | Completed | - | Israel ... more >> Ezrath Nashim - Herzog Memorial Hospital & Community Clinics Jerusalem, Israel Ezrath Nashim - Herzog Memorial Hospital Jerusalem, Israel Collapse << |
| NCT01541111 | Cancer-related Problem/Conditi... more >>on Collapse << | Not Applicable | Completed | - | Brazil ... more >> Universidade Federal de São Paulo São Paulo, Brazil, 04023062 Collapse << |
| NCT00001365 | Neurodegenerative Disease ... more >> Parkinson's Disease Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Neurological Disorders and Stroke (NINDS) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00630955 | Alcohol Drinking | Phase 2 | Completed | - | United States, Connecticut ... more >> CMHC New Haven, Connecticut, United States, 06519 Collapse << |
| NCT02583841 | Chronic Periodontitis (CP) Sub... more >>jects. Collapse << | Phase 4 | Completed | - | - |
| NCT02587884 | Hepatocellular Carcinoma | Phase 1 | Unknown | December 2017 | China, Shanghai ... more >> Eastern hepatobilliary surgery hospital Not yet recruiting Shanghai, Shanghai, China, 200438 Contact: Feng Shen, MD 0086-021-25070805 shenfengdfgd@yahoo.com.cn Contact: Yong Xia, Doctor 86-021-81875495 Collapse << |
| NCT03212521 | Hepatitis C Virus (HCV) | Phase 4 | Completed | - | - |
| NCT00630955 | - | - | Completed | - | - |
| NCT00055549 | Voice Disorders | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00486902 | - | - | Completed | - | - |
| NCT00588952 | Alcoholism | Not Applicable | Completed | - | United States, Connecticut ... more >> VA Connecticut Healthcare System West Haven, Connecticut, United States, 06516 Collapse << |
| NCT01551329 | Depression Al... more >>cohol Dependence Collapse << | Phase 1 | Completed | - | United States, Connecticut ... more >> VA CT Healthcare Systems West Haven, Connecticut, United States, 06516 Collapse << |
| NCT00854425 | Lymphoma | Phase 2 | Completed | - | China, Shanghai ... more >> Fudan University Cancer Hospital Shanghai, Shanghai, China, 200032 Collapse << |
| NCT00662571 | - | - | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00001929 | Movement Disorders ... more >> Parkinson Disease Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Neurological Disorders and Stroke (NINDS) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00588952 | - | - | Completed | - | - |
| NCT01600885 | - | - | Completed | - | - |
| NCT00001930 | Chorea Huntin... more >>gton's Disease Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Neurological Disorders and Stroke (NINDS) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01585168 | Healthy | Phase 2 | Completed | - | United States, Connecticut ... more >> Olin Neuropsychiatry Research Center at the Institute of Living, Hartford Hospital Hartford, Connecticut, United States, 06106 Collapse << |
| NCT03736538 | Major Depressive Disorder ... more >> Depression Mood Disorders Suicidal Ideation Collapse << | Phase 1 | Enrolling by invitation | December 2021 | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT01585168 | - | - | Completed | - | - |
| NCT03651557 | Cardiac Arrest | Phase 2 | Not yet recruiting | May 2021 | Korea, Republic of ... more >> Kyungpook National University Hospital Not yet recruiting Daegu, Korea, Korea, Republic of, 41944 Contact: Mi Jin Lee, MD Soonchunhyang University Bucheon Hospital Bucheon, Korea, Republic of, 14584 Pusan National University Hospital Not yet recruiting Busan, Korea, Republic of, 49241 Contact: Seok Ran Yeom, MD Chonnam National University Hospital Gwangju, Korea, Republic of, 61469 Gangnam Severance Hospital Seoul, Korea, Republic of, 06273 Samsung Medical Center Not yet recruiting Seoul, Korea, Republic of, 06351 Contact: Jin-Ho Choi, MD Collapse << |
| NCT02437344 | - | - | Completed | - | - |
| NCT03580122 | Classic Galactosemia | Phase 2 | Completed | - | Netherlands ... more >> Academisch Ziekenhuis Maastricht Maastricht, Limburg, Netherlands, 6202 AZ Collapse << |
| NCT01600885 | NMDA Receptor Function | Not Applicable | Completed | - | United States, Connecticut ... more >> Connecticut Mental Health Center New Haven, Connecticut, United States, 06511 Yale Magnetic Resonance Research Center New Haven, Connecticut, United States, 06520 Veterans Affairs Hospital West Haven, Connecticut, United States, 06516 Collapse << |
| NCT02437344 | Opioid Dependence | Phase 2 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT03562585 | - | - | Completed | - | - |
| NCT01014650 | Healthy | Phase 1 | Completed | - | United States, California ... more >> Lotus Clinical Research Pasadena, California, United States, 91105 Collapse << |
| NCT00405886 | Tinnitus | Phase 2 | Completed | - | Austria ... more >> Vienna, Austria Germany Munich, Germany Collapse << |
| NCT00817336 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Nathan Kline Institute Orangeburg, New York, United States, 10962 Collapse << |
| NCT00817336 | - | - | Completed | - | - |
| NCT02051426 | - | - | Completed | - | - |
| NCT03524651 | Iron Deficiency Anemia | Phase 4 | Recruiting | March 1, 2019 | Greece ... more >> General Hospital of Chalkida Not yet recruiting Chalkida, Greece, 34100 Contact: Nikolaos Tsokos, MD 30 22213 50459 ntsokos@hotmail.com Principal Investigator: Nikolaos Tsokos, MD Sub-Investigator: Georgios Haliotis, MD Attikon University Hospital Recruiting Haidari/Athens, Greece, 12462 Contact: Evangelos Giamarellos-Bourboulis, MD, PhD 0030 210 5831994 egiamarel@med.uoa.gr Principal Investigator: Evangelos Giamarellos-Bourboulis, MD, PhD Sub-Investigator: Maria Argiropoulou, MD Sub-Investigator: Evdoxia Kiriazopoulou, MD Collapse << |
| NCT00960219 | Schizophrenias ... more >> Psychoses Psychotic Disorders Schizophrenic Disorders Collapse << | Phase 2 | Completed | - | Taiwan ... more >> Department of Psychiatry, China Medical University Hospital Taichung, Taiwan Collapse << |
| NCT00375778 | Parkinson's Disease | Not Applicable | Completed | - | Denmark ... more >> PET center, Aarhus University Hospital Aarhus, Denmark, 8000 Collapse << |
| NCT02239003 | Mild Cognitive Impairment | Phase 2 | Completed | - | Taiwan ... more >> Kaohsiung Chang Gung Memorial Hospital Kaohsiung, Taiwan, 886 Collapse << |
| NCT00136747 | Tobacco Use Disorder | Phase 1 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT02051426 | Healthy | Not Applicable | Completed | - | - |
| NCT00125515 | - | - | Completed | - | - |
| NCT00131339 | Panic Disorder | Not Applicable | Completed | - | United States, Connecticut ... more >> Hartford Hospital Hartford, Connecticut, United States, 06106 United States, Massachusetts Center for Anxiety and Related Disorders at Boston University Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00088699 | Depression Mo... more >>od Disorders Major Depresssion Collapse << | Phase 1 Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00328276 | Schizophrenias ... more >> Psychoses Psychotic Disorders Schizophrenic Disorders Collapse << | Phase 2 | Completed | - | Taiwan ... more >> Department of Psychiatry, China Medical University Hospital Taichung, Taiwan, 404 Collapse << |
| NCT00980408 | Healthy Individuals | Not Applicable | Completed | - | Germany ... more >> Forschungszentrum Juelich GmbH Juelich, NRW, Germany, 52428 Collapse << |
| NCT00125515 | Opioid Dependence | Phase 2 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT02443350 | - | - | Unknown | - | China, Beijing ... more >> Beijing Tongren Hospital,Capital Medical Universiy Recruiting Beijing, Beijing, China, 100730 Contact: Jia Wei Wang, M.D (86)15910785840 wangjwcq@163.com Collapse << |
| NCT03387046 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 2 | Active, not recruiting | December 25, 2018 | Italy ... more >> Ospedale Binaghi, Università di Cagliari,ASL 8 Cagliari, Italy Ospedale Clinicizzato SS. Annunziata Chieti, Italy Azienda Socio Sanitaria Territoriale della Valle Olona (presidio di Gallarate) Gallarate, Italy Azienda Ospedaliero Universitaria San Martino Genova, Italy Ospedale P.A.Micone Genova, Italy Ospedale San Raffaele Milano, Italy A.O.U. Federico II Napoli, Italy Azienda Ospedaliera di Rilievo Nazionale A. CardarelliAzienda Ospedaliera di Rilievo Nazionale A. Cardarelli Napoli, Italy Seconda Università degli Studi di Napoli Napoli, Italy Azienda Ospedaliera di Padova Padova, Italy I.R.C.C.S. Neuromed-Istituto Neurologico Mediterraneo Pozzilli, Italy Azienda Ospedaliera San Camillo Forlanini Roma, Italy Azienda Ospedaliera Sant'Andrea-Università di Roma La Sapienza Roma, Italy Policlinico Universitario Agostino Gemelli Roma, Italy Azienda Ospedaliera Universitaria Policlinico Tor Vergata Rome, Italy Ospedale S. Paolo Savona, Italy Azienda Ospedaliero Universitaria Ospedali Riuniti Torrette Di Ancona, Italy Collapse << |
| NCT02097706 | Borderline Personality Disorde... more >>r Collapse << | Phase 2 | Recruiting | December 2018 | Australia, Victoria ... more >> Monash Alfred Psychiatry Research Centre Recruiting Melbourne, Victoria, Australia, 3004 Contact: Jayashri Kulkarni, MBBS,MPM,FRANZCP,PhD +61 3 9076 6924 j.kulkarni@alfred.org.au Contact: Anthony deCastella, Dip App Sci, BA, MA +61 3 9076 6554 ext 66554 anthony.decastella@monash.edu Principal Investigator: Jayashri Kulkarni, MBBS,MPM,FRANZCP,PhD Sub-Investigator: Anthony deCastella, Dip AppSci,BA,MA Sub-Investigator: Emorfia Gavrilidis, BAppSci Sub-Investigator: Roisin Worsley, MBBS, FRACP Sub-Investigator: Jasmin Grigg, PhD Collapse << |
| NCT01882829 | Major Depressive Disorder ... more >> Treatment Resistant Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT00142233 | Pancreatitis | Phase 3 | Active, not recruiting | August 2019 | - |
| NCT00088699 | - | - | Completed | - | - |
| NCT00491569 | Schizophrenias ... more >> Psychoses Psychotic Disorders Schizophrenic Disorders Collapse << | Phase 2 | Completed | - | Taiwan ... more >> China Medical University Hospital Taichung, Taiwan, 404 Collapse << |
| NCT01847651 | Hepatic Encephalopathy ... more >> Minimal Hepatic Encephalopathy Cirrhosis Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Liver unit St Mary's Hospital, 10th floor QEQM Wing, South Wharf Road London, United Kingdom, W2 1NY Collapse << |
| NCT01882829 | - | - | Completed | - | - |
| NCT02158182 | Hepatic Encephalopathy | Phase 4 | Completed | - | Mexico ... more >> Hospital General de Mexico Mexico City, Mexico, 06726 Collapse << |
| NCT01140620 | Healthy Volunteers ... more >> Schizophrenia Collapse << | Phase 4 | Completed | - | United Kingdom ... more >> Institute of Psychiatry, King's College London London, Greater London, United Kingdom, SE5 8AF School of Psychology, University of Cardiff Cardiff, United Kingdom, CF10 3AT University of Manchester (Dept of Neuropyschiatry) Manchester, United Kingdom, M13 9PT Collapse << |
| NCT01251055 | Schizophrenia ... more >> Treatment Refractory Collapse << | Phase 2 | Completed | - | Taiwan ... more >> China Medical University Hospital Taichung, Taiwan, 400 Collapse << |
| NCT01041755 | Hepatic Encephalopathy | Phase 4 | Completed | - | Mexico ... more >> Hospital Universitario "José Eleuterio González" Monterrey, Nuevo León, Mexico, 64460 Collapse << |
| NCT00526968 | Human Volunteers | Phase 1 | Completed | - | United Kingdom ... more >> Centre for Neuroimaging Science, Box 089, Institute of Psychiatry London, United Kingdom, SE5 8AF Collapse << |
| NCT00433368 | Hepatic Encephalopathy | Phase 3 | Completed | - | Pakistan ... more >> Aga Khan University Hospital Karachi, Sind, Pakistan, 74800 Collapse << |
| NCT00366886 | - | - | Terminated | - | United States, Georgia ... more >> Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01440829 | Decompensated Cirrhosis ... more >> Portal Hypertension Bleeding Varices Refractory Ascites Collapse << | Phase 2 Phase 3 | Completed | - | China, Shaanxi ... more >> Xijing Hospital of Digestive Diseases, Fourth Military Medical University Xi`an, Shaanxi, China, 710032 Collapse << |
| NCT01785628 | Parkinson's Disease With Demen... more >>tia Collapse << | Not Applicable | Completed | - | Taiwan ... more >> China Medical University Hospital/Neuro Depart. Taichung, Taiwan Collapse << |
| NCT00470314 | Acute Liver Failure | Phase 2 | Unknown | May 2007 | India ... more >> All India Institute of Medical Sciences Delhi, India Collapse << |
| NCT01785628 | - | - | Completed | - | - |
| NCT02082912 | Stroke | Phase 2 | Completed | - | United States, Georgia ... more >> Emory University Atlanta, Georgia, United States, 30322 Collapse << |
| NCT03166501 | Depression Po... more >>st Traumatic Stress Disorder Collapse << | Phase 1 | Not yet recruiting | December 31, 2019 | - |
| NCT02082912 | - | - | Completed | - | - |
| NCT00740142 | Hepatic Encephalopathy | Phase 4 | Unknown | August 2010 | Thailand ... more >> Siriraj Hospital, Mahidol University Recruiting Bangkok, Thailand, 10700 Contact: Siwaporn Chainuvati, MD (662)4197281 siswf@mahidol.ac.th Sub-Investigator: Supot Nimanong, MD Principal Investigator: Siwaporn Chainuvati, MD Collapse << |
| NCT01833897 | Bipolar Disorder | Phase 4 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT01833897 | - | - | Completed | - | - |
| NCT00338598 | Alcoholism Sc... more >>hizophrenia Collapse << | Phase 2 | Completed | - | United States, Connecticut ... more >> VA Connecticut Healthcare System West Haven, Connecticut, United States, 06516 Collapse << |
| NCT00338598 | - | - | Completed | - | - |
| NCT01600469 | Alzheimer's Disease ... more >> Mild Cognitive Impairment Collapse << | Phase 2 | Completed | - | Taiwan ... more >> Kaohsiung Chang Gung Memorial Hospital Kaohsiung, Taiwan, 886 Collapse << |
| NCT01770548 | Autism | Not Applicable | Completed | - | France ... more >> CHRU - Centre de Ressources Autisme (CRA) Brest, France, 29000 CHS du ROUVRAY Rouen, France, 76000 Chru - Cic Tours, France, 37000 CHRU - Pédopsychiatrie Tours, France, F-37000 Collapse << |
| NCT00205712 | Psychoses, Substance-Induced | Phase 4 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine, Psychiatry Dept. St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00205712 | - | - | Completed | - | - |
| NCT00896831 | Hepatic Encephalopathy | Phase 4 | Unknown | September 2010 | Brazil ... more >> Hospital de Clínicas de Porto Alegre Porto Alegre, Rio Grande do Sul, Brazil Collapse << |
| NCT00218465 | Nicotine Dependence | Phase 2 | Completed | - | United States, Massachusetts ... more >> McLean Hospital, Brain Imaging Center Belmont, Massachusetts, United States, 02478 9106 Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00218465 | - | - | Completed | - | - |
| NCT02373124 | Cocaine Use Disorders | Phase 1 Phase 2 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT00364429 | Schizophrenia | Phase 1 | Terminated(terminated) | - | Spain ... more >> GSK Investigational Site Barcelona, Spain Collapse << |
| NCT02559089 | - | - | Unknown | - | - |
| NCT00188383 | Hyperalgesia ... more >>Pain Prostate Cancer Collapse << | Phase 1 Phase 2 | Unknown | - | Canada, Ontario ... more >> University Health Network Recruiting Toronto, Ontario, Canada, M5G 2C4 Contact: Lucia Gagliese, PhD 416-340-4800 ext 4296 lucia.gagliese@uhn.on.ca Principal Investigator: Lucia Gagliese, PhD Sub-Investigator: Vincent Chan, MD Sub-Investigator: Dirk Snijedelaar, MD Sub-Investigator: Joel Katz, PhD Sub-Investigator: Gideon Koren, MD Collapse << |
| NCT03228524 | Brain Injuries | Early Phase 1 | Recruiting | December 1, 2022 | Italy ... more >> IRCCS Neuromed Recruiting Pozzilli, Isernia, Italy, 86077 Contact: Stefania Passarelli +39 0865.915217 direzionescientifica@neuromed.it Collapse << |
| NCT02426983 | Schizophrenia | Phase 1 Phase 2 | Unknown | April 2016 | Canada, Ontario ... more >> University of Ottawa Institute of Mental Health Research Ottawa, Ontario, Canada, K1Z 7K4 Collapse << |
| NCT01722578 | Cirrhosis of Liver ... more >> Hepatic Encephalopathy Collapse << | Phase 4 | Completed | - | India ... more >> Department of Gastroenterology, D.M.C. and Hospital Ludhiana, Punjab, India, 141001 G.B. Pant Hospital New Delhi, India, 110002 Collapse << |
| NCT03336398 | Tinnitus | Phase 2 | Not yet recruiting | December 2022 | United States, New York ... more >> 1051 Riverside Drive New York, New York, United States, 10032 Collapse << |
| NCT00611897 | Cognitive Dysfunction | Phase 1 Phase 2 | Completed | - | United States, Connecticut ... more >> VHA Connecticut West Haven, Connecticut, United States, 06516 Collapse << |
| NCT03062150 | Major Depression | Not Applicable | Recruiting | June 30, 2019 | Germany ... more >> Charité Universitätsmedizin Berlin Recruiting Berlin, Germany, 12203 Contact: Christian Otte, Prof. Dr. +49-30-450-517532 Christian.Otte@charite.de Collapse << |
| NCT01047592 | Schizophrenia | Phase 2 | Completed | - | Taiwan ... more >> Changhua Hospital Changhua, Taiwan, 513 Collapse << |
| NCT00611897 | - | - | Completed | - | - |
| NCT03752463 | Alzheimer Disease | Phase 2 | Recruiting | December 31, 2019 | Taiwan ... more >> Kaohsiung Chang Gung Memorial Hospital Recruiting Kaohsiung, Taiwan, 886 Contact: Chieh-Hsin Lin, MD, PhD 886-7-7317123 ext 8803 cyndi36@gmail.com Principal Investigator: Chieh-Hsin Lin, MD, PhD Collapse << |
| NCT01865578 | Anti-NMDA Receptor Encephaliti... more >>s Collapse << | Not Applicable | Completed | - | Germany ... more >> Charite University Medicine Berlin, Germany, 10098 Collapse << |
| NCT02087202 | Pain | Not Applicable | Unknown | July 2014 | - |
| NCT02675530 | Schizophrenia | Phase 1 | Completed | - | United States, Connecticut ... more >> VHA CT West Haven, Connecticut, United States, 06516 Collapse << |
| NCT00163059 | Depressive Disorder, Major | Phase 2 | Completed | - | United States, Kansas ... more >> Pfizer Investigational Site Wichita, Kansas, United States, 67214-2878 Collapse << |
| NCT00163085 | Parkinson's Disease | Phase 2 | Completed | - | United States, Oregon ... more >> Pfizer Investigational Site Portland, Oregon, United States, 97239 Collapse << |
| NCT01474395 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Phase 2 | Unknown | June 2013 | United States, New York ... more >> Nathan Kline Insitute for Psychiatric Research Recruiting Orangeburg, New York, United States, 10962 Sub-Investigator: Daniel C Javitt, MD Principal Investigator: Joshua T Kantrowitz, MD Collapse << |
| NCT01553435 | Pain Opioid T... more >>olerance Analgesia Tolerance Collapse << | Not Applicable | Unknown | - | Saudi Arabia ... more >> King Abdul Aziz Medical City Riyadh, Central, Saudi Arabia, 11426 Collapse << |
| NCT01602185 | Neuropathic Pain | Phase 2 | Completed | - | France ... more >> CHU Clermont-Ferrand Clermont-Ferrand, France, 63003 Collapse << |
| NCT02994433 | Depressive Disorder, Major ... more >> Depressive Disorder, Treatment-Resistant Collapse << | Phase 1 | Recruiting | June 2019 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Britt Gott, MS 314-362-2463 gottb@wustl.edu Principal Investigator: Charles R Conway, MD Collapse << |
| NCT03414931 | Depressive Disorder, Major | Phase 2 | Recruiting | March 2019 | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Kaohsiung, Taiwan, 886 Contact: Chieh-Hsin Lin, MD, PhD 886-7-7317123 ext 8753 cyndi36@gmail.com Principal Investigator: Chieh-Hsin Lin, MD, PhD China Medical University Hospital Recruiting Taichung, Taiwan, 404 Contact: Hsien-Yuan Lane, MD, PhD 886-921-067260 hylane@gmail.com Collapse << |
| NCT02950233 | Pain, Postoperative | Phase 3 | Recruiting | September 2018 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44106 Contact: Alparslan Turan, MD Principal Investigator: Alparslan Turan, MD Canada, Ontario St. Joseph's Healthcare Recruiting Hamilton, Ontario, Canada, L8N4A6 Contact: Harsha Shanthanna, MD Principal Investigator: Harsha Shanthanna, MD Collapse << |
| NCT01594047 | Post Operative Pain ... more >> Hyperalgesia Collapse << | Not Applicable | Unknown | August 2012 | Italy ... more >> Fondazione IRCCS Istituto Nazionale dei Tumori Recruiting Milano, Italy, 20133 Principal Investigator: Emiliano Tognoli Collapse << |
| NCT01392287 | Major Depressive Disorder | Phase 1 | Completed | - | United States, Massachusetts ... more >> McLean Hospital Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT02343575 | Hyperactive Delirium ... more >> Mixed Delirium Collapse << | Phase 4 | Suspended(The research assista... more >>nt has left the position and we are recruiting a new one.) Collapse << | June 2020 | United States, California ... more >> Stanford Hospital and Clinics Stanford, California, United States, 94305 Collapse << |
| NCT02139540 | Depression Ma... more >>jor Depressive Disorder Collapse << | Phase 2 | Unknown | December 2016 | United States, Missouri ... more >> Barnes-Jewish Hospital St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01656187 | Organic Memory Impairment | Phase 4 | Completed | - | United States, Texas ... more >> Aston Ambulatory Care Center, Allergy and Immunology Dallas, Texas, United States, 75390-8872 Collapse << |
| NCT02659085 | Depressive Disorder, Major | Phase 2 Phase 3 | Recruiting | December 2018 | Sweden ... more >> Department of Psychiatry Recruiting Lund, Sweden, 221 85 Contact: Pouya Movahed, MD, PhD +4646178825 Pouya.movahed_rad@med.lu.se Collapse << |
| NCT03655041 | Sports Nutritional Physiologic... more >>al Phenomena Collapse << | Not Applicable | Completed | - | - |
| NCT00917839 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 2 | Unknown | December 2011 | Switzerland ... more >> Cantonal Hospital St. Gallen Recruiting St. Gallen, Switzerland, 9007 Contact: Norman Putzki, MD +4171494 ext 1663 norman.putzki@kssg.ch Principal Investigator: Norman Putzki, MD Sub-Investigator: Ozgur Yaldizli, MD Collapse << |
| NCT00953615 | Primary Sclerosing Cholangitis | Phase 2 | Terminated(Lack of enrollment) | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03065335 | Healthy Volunteer ... more >> Major Depressive Disorder Depression Collapse << | Phase 1 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT03689790 | Liver Function ... more >> Bariatric Surgery Collapse << | Not Applicable | Active, not recruiting | December 2019 | China, Guangdong ... more >> The frist affiliated hospital of Jinan University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT02656342 | Obsessive-Compulsive Disorder | Phase 4 | Active, not recruiting | December 2023 | Norway ... more >> Haukeland University Hospital Bergen, Norway, 5021 Innlandet Hospital Brumunddal, Norway, 2380 Forde Hospital Forde, Norway, 6812 Sørlandet Hospital Kristiansand, Norway, 4604 Nord Trøndelag Hospital Levanger, Norway, 7601 Akershus University Hospital Lorenskog, Norway, 1478 More and Romsdal Hospital Molde, Norway Østfold Hospital Moss, Norway, 1635 Oslo University Hospital Oslo, Norway, 0424 Vestre Viken Sandvika, Norway, 1300 Stavanger University Hospital Stavanger, Norway Sykehuset i Vestfold Tonsberg, Norway, 3103 Tromso University Hospital Tromso, Norway St. Olavs Hospital Trondheim, Norway, 7006 Collapse << |
| NCT00953615 | - | - | Terminated(Lack of enrollment) | - | - |
| NCT02983734 | Gulf War Illness | Phase 2 | Recruiting | September 2018 | United States, Massachusetts ... more >> Boston University Recruiting Boston, Massachusetts, United States, 02215 Collapse << |
| NCT02719236 | Coxarthrosis | Not Applicable | Recruiting | June 2018 | Romania ... more >> Emergency County Hospital Cluj-Napoca, Orthopedics and Trauma Clinic Recruiting Cluj-Napoca, Cluj, Romania, 400006 Contact: Cozma Elena, Licensed Head Nurse +40 744 517 099 elena_cozma@yahoo.com Sub-Investigator: Adrian Todor, MD, PhD Sub-Investigator: Sergiu Caterev, MD Collapse << |
| NCT03264430 | Perianal Fistula ... more >> Hemorrhoids Collapse << | Early Phase 1 | Active, not recruiting | December 31, 2018 | - |
| NCT02805738 | Chronic Hepatitis B | Phase 3 | Unknown | March 2018 | Korea, Republic of ... more >> Korea University Ansan Hospital Not yet recruiting Ansan, Gyeonggi-do, Korea, Republic of Contact: Hyengjun Lim, MD 82-31-412-6565 gudwns21@medimail.co.kr Bundang Cha Medical Center Not yet recruiting Bundang, Gyeonggi-do, Korea, Republic of Contact: Sunggue Hwang, MD 82-31-780-5213 sghwang@cha.ac.kr Hanyang University Guri Hospital Recruiting Guri, Gyeonggi-do, Korea, Republic of Contact: Joohyun Sohn, MD 82-31-560-2225 sonjh@hanyang.ac.kr Inje University Ilsan Paik Hospital Recruiting Ilsan, Gyeonggi-do, Korea, Republic of Contact: June Sung Lee, MD 82-31-910-7823 jsleemd@paik.ac.kr Gachon University of Medicine and Science Gil Medical Center Active, not recruiting Incheon, Gyeonggi-do, Korea, Republic of Ajou University Hospital Active, not recruiting Suwon, Gyeonggi-do, Korea, Republic of Busan National University Hospital Active, not recruiting Busan, Korea, Republic of Keimyung University Dongsan Medical Center Active, not recruiting Daegu, Korea, Republic of Yeungnam University Medical Center Not yet recruiting Daegu, Korea, Republic of Contact: Heonju Lee, MD 82-53-620-3834 heonjulee@yu.ac.kr Chungnam National University Hospital Not yet recruiting Daejeon, Korea, Republic of Contact: Byengseok Lee, MD 82-2-2019-3314 leeks519@yuhs.ac Jeju National University Hospital Not yet recruiting Jeju, Korea, Republic of Contact: Byungcher Song, MD 82-64-717-1643 drsong@jejunu.ac.kr Chungang University Hospital Active, not recruiting Seoul, Korea, Republic of Hanyang University Hospital Active, not recruiting Seoul, Korea, Republic of Kangnam Severance Hospital Not yet recruiting Seoul, Korea, Republic of Contact: Kwan Sik Lee, MD 82-2-2019-2332 leeks519@yuhs.ac Korea University Guro Hosptial Not yet recruiting Seoul, Korea, Republic of Contact: Jihoon Kim, MD 82-2-2626-3011 kjhhepar@naver.com Samsung Medical Center Active, not recruiting Seoul, Korea, Republic of Seoul Asan Medical Center Active, not recruiting Seoul, Korea, Republic of Seoul National University Hospital Active, not recruiting Seoul, Korea, Republic of Seoul Saint Mary's Hospital Active, not recruiting Seoul, Korea, Republic of Severance Hospital Not yet recruiting Seoul, Korea, Republic of Contact: Do Young Kim, MD 82-2-2228-0525 dyk1025@yuhs.ac Collapse << |
| NCT01317589 | Pain Cancer o... more >>f Head and Neck Collapse << | Phase 4 | Completed | - | Netherlands ... more >> University Hospital Maastricht Maastricht, Netherlands, 6202AZ Collapse << |
| NCT00926900 | Alcohol Drinking | Phase 2 Phase 3 | Unknown | - | Germany ... more >> Central Institute of Mental Health Recruiting Mannheim, Germany, 68159 Contact: Falk Kiefer, Professor +49 621 1703 3522 falk.kiefer@zi-mannheim.de Principal Investigator: Falk Kiefer, Professor Collapse << |
| NCT00210210 | Postoperative Pain ... more >> Breast Cancer Collapse << | Phase 3 | Completed | - | France ... more >> Institut Bergonié - Centre Régional de Luttre Contre le Cancer de Bordeaux et du Sud Ouest Bordeaux, France, 33076 Collapse << |
| NCT03194815 | Psychosis Aut... more >>oimmune Encephalitis Collapse << | Phase 2 | Recruiting | December 2021 | United Kingdom ... more >> Cambridge University Hospitals Recruiting Cambridge, United Kingdom Contact: Alasdair Coles, PhD FRCP +44 (0)1223 762016 ajc1020@medschl.cam.ac.uk University College London Hospitals Nhs Foundation Trust Recruiting London, United Kingdom, NW1 2PG Contact: Michael Zandi, PhD MRCP m.zandi@ucl.ac.uk Oxford Radcliffe Hospitals Recruiting Oxford, United Kingdom Contact: Belinda Lennox, DM MRCPsych : +44(0)1865 613145 belinda.lennox@psych.ox.ac.uk Collapse << |
| NCT01234558 | Major Depressive Disorder | Phase 2 | Completed | - | United States, Illinois ... more >> Mulitple Evanston, Illinois, United States, 60201 Collapse << |
| NCT01203670 | Renal Tolerance ... more >> Digestive Tolerance Collapse << | Not Applicable | Completed | - | France ... more >> Département de Pharmacologie Clinique - Université Victor Segalen Bordeaux 2 - CHU de Bordeaux Bordeaux, France, 33076 Collapse << |
| NCT00155649 | - | - | Completed | - | - |
| NCT02704338 | Autoimmune Diseases | Phase 1 Phase 2 | Unknown | October 2018 | - |
| NCT01634256 | Mild Hepatic Injury | Phase 2 Phase 3 | Completed | - | Korea, Republic of ... more >> Clinical Trial Center for Functional Foods; Chonbuk National University Hospital Jeonju, Jeollabuk-do, Korea, Republic of, 560-822 Collapse << |
| NCT01634256 | - | - | Completed | - | - |
| NCT03775837 | Liver Diseases | Not Applicable | Recruiting | December 31, 2019 | Korea, Republic of ... more >> Integrated Research Institute for Natural Ingredients and Functional Foods Recruiting Yangsan, Korea, Republic of, 50612 Contact: Sang Yeoup Lee, MD 82-55-360-2860 saylee@pnu.edu Collapse << |
| NCT03115489 | Refractory Epilepsy | Phase 2 Phase 3 | Recruiting | April 2020 | United States, Alabama ... more >> UAB Department of Anesthesiology and Perioperative Medicine Recruiting Birmingham, Alabama, United States, 35249 Contact: Adam Sturdivant, MPH 205-934-4042 adamsturdivant@uabmc.edu Contact: Ayesha Bryant, MSPH, MD 205-996-7383 asbryant@uabmc.edu Principal Investigator: Vinodkumar Singh, MD Sub-Investigator: Angela Douglas, MD Sub-Investigator: Casey May, PharmD Collapse << |
| NCT00916864 | - | - | Completed | - | Germany ... more >> Department of Neurology, Goethe-University Frankfurt am Main, Germany, 60590 Collapse << |
| NCT00656292 | - | - | Completed | - | - |
| NCT02684591 | Nonalcoholic Fatty Liver Disea... more >>se HIV Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 Collapse << |
| NCT01341145 | Hypertension | Not Applicable | Completed | - | United States, Texas ... more >> University of Texas at Austin Austin, Texas, United States, 78712 Collapse << |
| NCT01720316 | Schizo-affective Disorder ... more >> Bipolar Disorder Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> McLean Hospital Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT00656292 | Perioperative Inflammatory Res... more >>ponse Collapse << | Phase 4 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00655967 | Alcohol Dependence | Phase 4 | Unknown | December 2008 | United States, New Mexico ... more >> University of New Mexico Recruiting Albuquerque, New Mexico, United States, 87131-0001 Contact: Florian Birkmayer, M.D. 505-272-2223 fbirkmayer@salud.unm.edu Sub-Investigator: Michael Bogenschutz, M.D. Collapse << |
| NCT01720316 | - | - | Completed | - | - |
| NCT01679353 | Inguinal Hernia | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02018276 | Thyroid Cancer | Not Applicable | Completed | - | Korea, Republic of ... more >> Department of Anesthesiology and Pain MedicineSeverance Hospital, Yonsei University Health System Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT01872481 | Phantom Limb Pain | Phase 3 | Completed | - | Colombia ... more >> Fundacion Cardiovascular de Colombia Floridablanca, Santander, Colombia, 0 Collapse << |
| NCT01312129 | - | - | Completed | - | - |
| NCT02304432 | - | - | Completed | - | - |
| NCT01386788 | - | - | Completed | - | Germany ... more >> Prof. Dr. Götz Geldner Ludwigsburg, Germany, 71640 Prof. Th. Zilker München, Germany, 81675 Collapse << |
| NCT01386788 | - | - | Completed | - | - |
| NCT02378740 | Anesthesia Pa... more >>in Surgery Collapse << | Phase 4 | Withdrawn(PI has left the Univ... more >>ersity) Collapse << | - | United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << |
| NCT02108015 | Healthy Volunteers | Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03090776 | Pain, Postoperative ... more >> Post-Mastectomy Chronic Pain Syndrome Collapse << | Early Phase 1 | Not yet recruiting | May 1, 2020 | - |
| NCT03052673 | Postoperative Pain | Phase 4 | Completed | - | India ... more >> Sir Ganga Ram Hospital New Delhi, India, 110060 Collapse << |
| NCT01349231 | Obsessive-compulsive Disorder | Phase 2 | Completed | - | United States, Connecticut ... more >> Connecticut Mental Health Center/ YNHH New Haven, Connecticut, United States, 06520 Collapse << |
| NCT03089905 | Anesthesia Ne... more >>urotoxicity Child Development Collapse << | Phase 3 | Recruiting | April 2022 | United States, Massachusetts ... more >> Boston Children's Hospital Not yet recruiting Boston, Massachusetts, United States, 02115 Contact: Mary Ellen McCann Mary.McCann@childrens.harvard.edu Contact: Charles Berde Charles.Berde@childrens.harvard.edu Principal Investigator: Mary Ellen McCann Sub-Investigator: Charles Berde United States, Ohio Cincinnati Children's Hospital Medical Center Not yet recruiting Cincinnati, Ohio, United States, 45229 Contact: Andreas W Loepke, MD 513-636-4408 Andreas.Loepke@cchmc.org Contact: Chris S Lee, MD 513-636-4408 Christopher.Lee@cchmc.org United States, Oregon Oregon Health and Science University Not yet recruiting Portland, Oregon, United States, 97239 Contact: Ansgar M Brambrink, MD 503-494-8311 brambrin@ohsu.edu United States, Pennsylvania The Children's Hospital of Philadelphia Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Francis X McGowan McGowanF@email.chop.edu Principal Investigator: Francis X McGowan United States, Texas The University of Texas Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390 Contact: Peter Szmuk, MD 214-456-8559 Peter.Szmuk@UTSouthwestern.edu Texas Children's Hospital Not yet recruiting Houston, Texas, United States, 77030 Contact: Dean B Andropoulos, MD 832-826-5831 dra@bcm.edu Australia, New South Wales John Hunter Hospital Not yet recruiting Newcastle, New South Wales, Australia, 2305 Contact: Kim Rackemann kim74r@bigpond.com Principal Investigator: Kim Rackemann Sydney Children's Hospital Not yet recruiting Randwick, New South Wales, Australia, 2031 Contact: Donald Hannah donald.hannah@health.nsw.gov.au Contact: Shona Chung Shona.Chung@health.nsw.gov.au Principal Investigator: Donald Hannah Children's Hospital at Westmead Not yet recruiting Westmead, New South Wales, Australia, 2145 Contact: Justin J Skowno justin.skowno@health.nsw.gov.au Principal Investigator: Justin J Skowno Australia, Queensland Lady Cilento Children's Hospital Not yet recruiting Brisbane, Queensland, Australia, 4101 Contact: Paul Lee-Archer pleearcher@hotmail.com Australia, Victoria Royal Children's Hospital Recruiting Parkville, Victoria, Australia, 3052 Contact: Andrew J Davidson, MD +61393455233 andrew.davidson@rch.org.au Contact: Suzette J Sheppard +61393454901 suzette.sheppard@mcri.edu.au Principal Investigator: Andrew J Davidson, MD Australia, Western Australia Princess Margaret Hospital Not yet recruiting Perth, Western Australia, Australia, 6008 Contact: Britta von ungern Sternberg britta.regli-vonungern@health.wa.gov.au Principal Investigator: Britta von ungern Sternberg Netherlands University Medical Center Groningen Not yet recruiting Groningen, Netherlands Contact: Anthony Absalom a.r.absalom@umcg.nl Principal Investigator: Anthony Absalom Sophia Children's Hospital Not yet recruiting Rotterdam, Netherlands Contact: Jurgen deGraaff j.degraaff@erasmusmc.nl Principal Investigator: Jurgen deGraaff Sweden Uppsala University Hospital Not yet recruiting Uppsala, Sweden Contact: Peter Frykholm peter.frykholm@akademiska.se Principal Investigator: Peter Frykolm Switzerland University Hospital Not yet recruiting Geneva, Switzerland Contact: Laszlo Vutskits Laszlo.Vutskits@unige.ch Principal Investigator: Laszlo Vutskits United Kingdom Great Ormond Street Hospital Not yet recruiting London, United Kingdom Contact: Nicola Disma nicoladisma@gmail.com Principal Investigator: Nicola Disma Sub-Investigator: Suellen Walker Collapse << |
| NCT02304432 | Schizophrenia ... more >> Bipolar Disorder Collapse << | Early Phase 1 | Completed | - | - |
| NCT01312129 | Alcohol Dependence | Not Applicable | Completed | - | United States, New Mexico ... more >> The Mind Research Network Albuquerque, New Mexico, United States, 87106 Collapse << |
| NCT01349231 | - | - | Completed | - | - |
| NCT03597776 | Acute Pain | Phase 4 | Not yet recruiting | July 15, 2021 | - |
| NCT01876823 | Mild Cognitive Impairment ... more >> Major Depressive Disorder Alzheimer's Disease Collapse << | Phase 2 Phase 3 | Completed | - | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Collapse << |
| NCT01808014 | Lumbar Spinal Stenisis or Lumb... more >>ar Herniated Intervertebral Disc Collapse << | Not Applicable | Unknown | June 2014 | Korea, Republic of ... more >> Severance Hospital Recruiting Seoul, Korea, Republic of, 120-752 Contact: Tae Dong Kweon, MD Collapse << |
| NCT01876823 | - | - | Completed | - | - |
| NCT03643666 | Pain, Postoperative | Phase 3 | Recruiting | January 2019 | Egypt ... more >> Kasr Alaini hospital Recruiting Cairo, Egypt Contact: abdelkhalek m Samy 01025854248 dr.abdo86@gmail.com Collapse << |
| NCT02752334 | Pain | Phase 2 | Unknown | May 2016 | Egypt ... more >> Tanta University Hospitals Recruiting Tanta, Egypt, 112 Contact: Nabil M Elshikh, Phd 00201001576606 nelshikh2002@yahoo.com Collapse << |
| NCT01985750 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Early Phase 1 | Recruiting | February 2020 | United Kingdom ... more >> Oxford Centre for Clinical Magnetic Resonance Imaging Recruiting Oxford, Oxfordshire, United Kingdom, OX3 9DU Contact: Kyle Pattinson, BM DPhil FRCA +441865 231509 kyle.pattinson@nda.ox.ac.uk Contact: Sarah Finnegan, DPhil +441865 234544 copd@fmrib.ox.ac.uk Collapse << |
| NCT00595530 | Sickle Cell Disease | Phase 2 | Terminated(Lack of enrollment ... more >>and patient interest in study) Collapse << | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 Connecticut Children's Medical Center Hartford, Connecticut, United States, 06106 Collapse << |
| NCT01969227 | Effects of Ketamine on Respira... more >>tory Stimulation and Transpulmonary Pressure Collapse << | Not Applicable | Recruiting | August 2017 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Matthias Eikermann, MD, PhD 617-643-4408 meikermann@partners.org Contact: Hsin Lin, PharmD hlin13@partners.org Collapse << |
| NCT03316339 | Anesthesia Op... more >>ioid-Related Disorders Collapse << | Phase 3 | Recruiting | June 2020 | France ... more >> Clermont-Ferrand University Hospital Not yet recruiting Clermont-Ferrand, France Contact: Emmanuel FUTIER Beaujon Hospital Not yet recruiting Clichy, France Contact: Catherine PAUGAM-BURTZ Lille University Hospital Not yet recruiting Lille, France Contact: Gilles LEBUFFE Metz-Thionville Hospital Not yet recruiting Metz, France Contact: Julien NADAUD Montpellier University Hospital Not yet recruiting Montpellier, France Contact: Gérald CHANQUES Nantes University Hospital Not yet recruiting Nantes, France Contact: Karim ASEHNOUNE Nimes University Hospital Not yet recruiting Nîmes, France Contact: Philippe CUVILLON Perigueux Hospital Not yet recruiting Périgueux, France Contact: Sebastien OGER Rennes University Hospital Recruiting Rennes, France Contact: Hélène BELOEIL +33 2 99 28 91 76 helene.beloeil@chu-rennes.fr Saint-Brieuc Hospital Not yet recruiting Saint-Brieuc, France Contact: Sylvain LE COEUR Toulouse University Hospital Not yet recruiting Toulouse, France Contact: Vincent MINVILLE Collapse << |
| NCT00595530 | - | - | Terminated(Lack of enrollment ... more >>and patient interest in study) Collapse << | - | - |
| NCT01965093 | - | - | Completed | - | Thailand ... more >> Prince of Songkla University Hat Yai, Songkhla, Thailand, 90112 Collapse << |
| NCT03702803 | Generalized Anxiety Disorder | Phase 2 | Recruiting | April 2019 | Mexico ... more >> Regional General Hospital Number 1 at Cuernavaca, Morelos. Mexican Insitute of Social Security Recruiting Cuernavaca, Morelos, Mexico Contact: Ofelia Romero-Cerecero, Dr. +527773612194 orcerecero@yahoo.com.mx Contact: Jaime Tortoriello, Dr. +527773612155 jtortora2@yahoo.es Sub-Investigator: Alejandro Zamilpa, PhD Sub-Investigator: Enrique Jiménez-Ferrer, PhD Sub-Investigator: Armando Herrera-Arellano, PhD Sub-Investigator: Ofelia Romero-Cerecero, MD, PhD Collapse << |
| NCT01691690 | - | - | Completed | - | - |
| NCT00768430 | Major Depressive Disorder (MDD... more >>) Treatment Resistant Depression (TRD) Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Mount Sinai School of Medicine New York, New York, United States, 10029 United States, Texas Michael E. Dabakey VA Medical Center & Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT01733030 | - | - | Completed | - | United States, Minnesota ... more >> University of MInnesota Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT00768430 | - | - | Completed | - | - |
| NCT00413257 | Hyperalgesia ... more >>Pain, Postoperative Pain, Chronic Disease Collapse << | Phase 2 Phase 3 | Completed | - | France ... more >> Département d'Anesthésie-Réanimation II - Hôpital cardiologique - Groupe Hospitalier Sud, CHU de Bordeaux - 4, Av de Magellan Pessac, France, 33604 Collapse << |
| NCT01691690 | Adenotonsillitis ... more >> Tonsillitis Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Nationwide Children's Hospital Columbus, Ohio, United States, 43205 Collapse << |
| NCT00454259 | Pain, Postoperative | Phase 4 | Completed | - | France ... more >> CHU de Bordeaux Département d'Anesthésie-Réanimation II PESSAC Cedex, Hôpital cardiologique, France, 33604 Collapse << |
| NCT02142712 | - | - | Completed | - | - |
| NCT03485339 | - | - | Recruiting | August 6, 2020 | Hong Kong ... more >> Queen Mary Hospital Recruiting Hong Kong, Hong Kong, 000000 Contact: Albert KK Chung Western Psychiatric Centre Recruiting Hong Kong, Hong Kong, 00000 Contact: Albert Kar Kin Chung, MBBS +852-22556226 chungkka@hku.hk Collapse << |
| NCT02987920 | Pain, Postoperative | Phase 4 | Unknown | January 2018 | - |
| NCT01716858 | Schizophrenia | Phase 2 | Completed | - | Japan ... more >> Chiba University Hospital Chiba, Japan, 260-8670 Collapse << |
| NCT01062932 | Nicotine Addiction | Phase 1 | Completed | - | United States, Texas ... more >> Michael E. DeBakey VA Medical Center Houston, Texas, United States, 77030 Collapse << |
| NCT02142712 | Acute Cerebrovascular Accident... more >> Cerebral Edema Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Shands Hospital at University of Florida Gainesville, Florida, United States, 32610 Collapse << |
| NCT01844427 | ADHD Executiv... more >>e Function Deficits (EFD's) Collapse << | Not Applicable | Withdrawn | - | - |
| NCT03253224 | Arthritis Knee ... more >> Magnesium Sulfate Dexmedetomidine Collapse << | Phase 4 | Terminated(Because the princip... more >>le investigator will participate a visiting scholar program for one year in other country, this study has been terminated.) Collapse << | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam-si, Korea, Republic of, 13620 Collapse << |
| NCT00333242 | Opioid-induced Hyperalgesia | Phase 1 | Unknown | - | United States, California ... more >> UCLA School of Nursing Los Angeles, California, United States, 90095 Collapse << |
| NCT02449746 | Schizophrenia ... more >> Psychosis Auto Immune Disorders Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> University Hospitals Birmingham Birmingham, United Kingdom Cambridge University Hospitals Cambridge, United Kingdom Nottingham University Hospitals Nottingham, United Kingdom Oxford University Hospitals Oxford, United Kingdom University Hospitals Southampton Southampton, United Kingdom Collapse << |
| NCT00618423 | Postoperative Pain | Phase 2 | Completed | - | Switzerland ... more >> University Hospital of Geneva Geneva, Canton of Geneva, Switzerland, 1211 Collapse << |
| NCT01243801 | Persistent Pain ... more >> Postoperative Hyperalgesia Collapse << | Phase 4 | Completed | - | Spain ... more >> Department Anesthesia. Hospital Clinic Barcelona Barcelona, Spain, 08036 Hospital Clinic Barcelona, Spain, 08036 Collapse << |
| NCT00929370 | Schizophrenia | Phase 1 | Terminated(Study has now been ... more >>terminated due to changes in project strategy. Current available data will be analysed and reported in a synoptic study report.) Collapse << | - | United States, California ... more >> GSK Investigational Site Glendale, California, United States, 91206 United States, Texas GSK Investigational Site Bellaire, Texas, United States, 77401 Collapse << |
| NCT03602469 | Arthroscopic Shoulder Surgery | Not Applicable | Not yet recruiting | January 2019 | Egypt ... more >> Mansoura University Not yet recruiting Mansourah, DK, Egypt, 050 Contact: Hanaa M Elbendary, MD 00201005781768 hanaa_elbendary@yahoo.com Contact: Samah El Kenany, MD 00201002262557 sk_20022000@yahoo.com Collapse << |
| NCT01205997 | Post Operative Pain | Phase 1 Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Qazvin university of medical science Qazvin, Iran, Islamic Republic of, 34197/59811 Collapse << |
| NCT03402152 | Bipolar Depression ... more >> Suicidal Ideation Collapse << | Phase 2 Phase 3 | Recruiting | February 28, 2020 | United States, New York ... more >> New York State Psychiatric Institute Recruiting New York, New York, United States, 10032 Contact: Joshua T Kantrowitz, MD 646-774-6738 Joshua.Kantrowitz@nyspi.columbia.edu Collapse << |
| NCT00215904 | Parkinson's Disease | Phase 4 | Completed | - | Israel ... more >> Ezrath Nashim - Herzog Memorial Hospital Jerusalem, Israel, 91351 Collapse << |
| NCT02083926 | Social Anxiety Disorder | Early Phase 1 | Unknown | March 2018 | United States, Connecticut ... more >> Connecticut Mental Health Center New Haven, Connecticut, United States, 06519 Collapse << |
| NCT00181298 | Systemic Lupus Erythematosus | Not Applicable | Completed | - | United States, Maryland ... more >> Johns Hopkins Lupus Center, 1830 East Monument Street, Suite 7500 Baltimore, Maryland, United States, 21205 Collapse << |
| NCT00674219 | OCD Anxiety D... more >>isorder Collapse << | Phase 3 | Completed | - | United States, California ... more >> UCLA Los Angeles, California, United States, 90095 Collapse << |
| NCT03298711 | - | - | Enrolling by invitation | December 31, 2018 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam-si, Gyeonggi-do, Korea, Republic of, 13620 Collapse << |
| NCT01554475 | - | - | Completed | - | Norway ... more >> Kløveråsen Bodø, Norway, N-8076 Collapse << |
| NCT00484484 | Pain | Phase 3 | Completed | - | France ... more >> Hopital Saint Antoine Paris, France, 75012 Collapse << |
| NCT00785863 | Hyperalgesia, Secondary | Phase 4 | Completed | - | Norway ... more >> Ullevaal University Hospital Oslo, Norway, 0407 Collapse << |
| NCT01229514 | - | - | Completed | - | United States, Ohio ... more >> Nationwide Children's Hospital Columbus, Ohio, United States, 43205 Collapse << |
| NCT01326325 | Pain | Phase 3 | Completed | - | France ... more >> Center of Evaluation and Treatment of the pain - Saint-Antoine Hospital Paris, Ile de France, France, 75012 Collapse << |
| NCT00152022 | Attention Deficit Disorder Wit... more >>h Hyperactivity Collapse << | Phase 3 | Completed | - | - |
| NCT00152035 | Attention Deficit Disorder Wit... more >>h Hyperactivity Collapse << | Phase 3 | Completed | - | - |
| NCT02450214 | Pain, Postoperative | Not Applicable | Completed | - | Chile ... more >> Hospital Clinico de la Universidad de Chile Santiago, RM, Chile, 7690306 Collapse << |
| NCT03410446 | Depression Ca... more >>ncer Palliative Care Collapse << | Phase 2 | Recruiting | February 1, 2024 | Canada, Ontario ... more >> Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Madeline Li, M.D. 416-946-4501 ext 7505 madeline.li@uhn.ca Principal Investigator: Madeline Li, M.D. Collapse << |
| NCT00150579 | Attention Deficit Disorder Wit... more >>h Hyperactivity Collapse << | Phase 3 | Completed | - | - |
| NCT02571153 | Postoperative Pain ... more >> Postoperative Nausea and Vomiting Collapse << | Phase 4 | Completed | - | Brazil ... more >> Santa Lucinda Hospital Sorocaba, SP, Brazil, 18030-230 Collapse << |
| NCT01928823 | Agoraphobia | Phase 2 | Completed | - | Germany ... more >> Department of Psychiatry and Psychotherapy, Charité Campus Mitte - Universitätsmedizin Berlin Berlin, Germany, 10117 Collapse << |
| NCT01707420 | Post Operative Pain Management... more >> in Children With Tonsillectomy/Adenoidectomy Collapse << | Not Applicable | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado Aurora, Colorado, United States, 80045 Collapse << |
| NCT02571153 | - | - | Completed | - | - |
| NCT03296345 | Sickle Cell Disease ... more >> Vaso-Occlusive Crisis Collapse << | Phase 2 | Recruiting | December 2018 | United States, California ... more >> UCSF Benioff Children's Hospital and Research Center Oakland Recruiting Oakland, California, United States, 94609 Contact: Bryan Cooper-Sood, MD 510-428-3888 jocooper@mail.cho.org Contact: Anurag Agrawal, MD 5104283539 AAgrawal@mail.cho.org Principal Investigator: Bryan Cooper-Sood, MD Sub-Investigator: Anurag Agrawal, MD Sub-Investigator: Carolyn Hoppe, MD Sub-Investigator: James Naprawa, MD Sub-Investigator: Anne Marsh, MD Collapse << |
| NCT00202605 | Attention Deficit Disorder Wit... more >>h Hyperactivity Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> Clinical Study Center Little Rock, Arkansas, United States United States, California UCI Child Development Center Irvine, California, United States United States, Nevada Center for Psychiatry and Behavioral Medicine, Inc. Las Vegas, Nevada, United States United States, Texas Bayou City Research, Ltd. Houston, Texas, United States Collapse << |
| NCT02099825 | Anxiety Disorders | Phase 1 | Active, not recruiting | August 2018 | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00073034 | Diabetes Mellitus ... more >> Diabetic Neuropathy, Painful Collapse << | Phase 2 | Terminated | - | - |
| NCT01804920 | Schizophrenia and Schizoaffect... more >>ive Disorder Tardive Dyskinesia Collapse << | Not Applicable | Active, not recruiting | January 2019 | Israel ... more >> Herzog Hospital Jerusalem, Israel Collapse << |
| NCT03666390 | Major Depressive Disorder | Not Applicable | Recruiting | December 31, 2021 | Taiwan ... more >> Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 111 Contact: Mu-Hong Chen, M.D. Collapse << |
| NCT02933684 | Social Anxiety Disorder | Phase 2 | Withdrawn(Unable to access com... more >>ponents necessary to be able to run the study.) Collapse << | December 31, 2016 | United States, Georgia ... more >> Georgia State University Atlanta, Georgia, United States, 30303 Collapse << |
| NCT00567905 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | Taiwan ... more >> Branch of Chinese Medicine, Taipei City Hospital Taipei, Taiwan, 886 Branch of Chinese Medicine Taipei, Taiwan, 886 Collapse << |
| NCT03680183 | - | - | Recruiting | December 31, 2021 | China, Guangdong ... more >> Affiliated Cancer Hospital & Institute of Guangzhou Medical University Recruiting Guangzhou, Guangdong, China, 510182 Contact: Wenying Shu, PhD 86-20-66673666 ext 2006 790840799@qq.com Contact: Tiantian Cheng, MD 86-20-66673677 gyzlgcp@163.com Collapse << |
| NCT03108209 | Phototoxicity | Not Applicable | Recruiting | October 2017 | France ... more >> CHU Nantes Recruiting Nantes, France Contact: Aurélie BOISROBERT Contact 02.40.08.31.22 Collapse << |
| NCT02442700 | - | - | Completed | - | - |
| NCT02442700 | HIV Dyslipide... more >>mia Collapse << | Phase 4 | Completed | - | - |
| NCT02070523 | Acute Lymphoblastic Leukemia | Phase 3 | Unknown | December 2015 | China, Guangdong ... more >> Department of Hematology, Affiliated Hospital of Guangdong Medical College Recruiting Zhanjiang, Guangdong, China, 524000 Contact: Yang Zhi gang +86 13560512702 13560512702@139.com Principal Investigator: Zhi gang Yang Collapse << |
| NCT02046694 | Acute Lymphoblastic Leukemia (... more >>ALL) Collapse << | Early Phase 1 | Recruiting | September 2021 | United States, Maryland ... more >> Johns Hopkins Hospital Recruiting Baltimore, Maryland, United States, 21287 Principal Investigator: Colleen Annesley, MD Sub-Investigator: Patrick A Brown, MD United States, Texas Texas Children's Cancer and Hematology Centers Recruiting Houston, Texas, United States, 77030 Contact: Julie Brackett, MD 832-822-4242 Sub-Investigator: Edward Sisson, MD Principal Investigator: Julie Brackett, MD United States, Washington Seattle Children's Hospital Recruiting Seattle, Washington, United States, 98105 Contact: Colleen Annesley, MD Principal Investigator: Colleen Annesley, MD Collapse << |
| NCT01610440 | Duchenne Muscular Dystrophy | Phase 1 Phase 2 | Unknown | October 2013 | China, Yunnan ... more >> The Second Affiliated Hospital of Kunming Medical College Recruiting Kunming, Yunnan, China, 650031 Contact: Liqing Yao yaoliqing98731@yahoo.com.cn Principal Investigator: Liqing Yao Collapse << |
| NCT03425630 | Hypercholesterolemia | Not Applicable | Completed | - | - |
| NCT02834741 | Healthy Volunteers | Phase 1 | Completed | - | United States, Wisconsin ... more >> Spaulding Clinical West Bend, Wisconsin, United States, 53095 Collapse << |
| NCT00893581 | Mania Bipolar... more >> Disorder Collapse << | Not Applicable | Completed | - | United States, Ohio ... more >> University of Cincinnati Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT02882347 | Liver Failure | Phase 3 | Unknown | - | Korea, Republic of ... more >> Korea university Anam hospital Recruiting Seoul, Korea, Republic of, 90248 Contact: Jae Han, Dr 82-2-920-6407 gelasius1@naver.com Principal Investigator: Jae Han, Dr Collapse << |
| NCT02335632 | Alcoholic Liver Disease | Phase 4 | Unknown | February 2015 | - |
| NCT02387060 | Complications; Cesarean Sectio... more >>n Collapse << | Phase 4 | Unknown | January 2016 | Chile ... more >> Hospital Clinico Pontificia Universidad Catolica Recruiting Santiago, Región Metropolitana, Chile, 8330024 Contact: Hernan Auad, MD 56982933226 hernan_auad@hotmail.com Principal Investigator: Juan Carlos de la Cuadra, MD Collapse << |
| NCT02606019 | - | - | Recruiting | October 2019 | Malaysia ... more >> Ampang Hospital Recruiting Ampang, Selangor, Malaysia Contact: Ridzuan Isa dmiaw2@gmail.com Principal Investigator: Rosaida Mohammad Said Principal Investigator: Ching Soong Khoo Principal Investigator: Melor Mohd Mansor Klinik Kesihatan Ampang (Ampang Health Clinic) Recruiting Ampang, Selangor, Malaysia Contact: Hanif Zailani, MPH hanif@moh.gov.my Collapse << |
| NCT01393665 | Chronic Liver Disease | Phase 2 | Completed | - | Korea, Republic of ... more >> Korea Cancer Center Hospital Seoul, Nowon-gu, Korea, Republic of, 139706 Hanyang University Hospital Seoul, Seongdong-gu, Korea, Republic of, 133792 Collapse << |
| NCT01501162 | - | - | Completed | - | - |
| NCT01501162 | Alcoholic Liver Disease | Phase 4 | Completed | - | Korea, Republic of ... more >> Department of Internal Medicine, Hallym University Chuncheon Sacred Heart Hospital Chuncheon, Korea, Republic of, 200-704 Collapse << |
| NCT01184235 | - | - | Completed | - | United States, North Dakota ... more >> Child Psychopharmacology Institute Fargo, North Dakota, United States, 58103 Collapse << |
| NCT03144167 | Glioblastoma ... more >>Radiotherapy Chemotherapy Collapse << | Not Applicable | Not yet recruiting | January 20, 2021 | France ... more >> CHU Amiens Picardie Not yet recruiting Amiens, Picardie, France, 80054 Contact: Jean-Marc CONSTANS, PhD +33322087511 constans.jean-marc@chu-amiens.fr Collapse << |
| NCT00570336 | Chronic Hepatitis C Virus Infe... more >>ction Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 United States, California Scripps Clinic La Jolla, California, United States, 92037 Kaiser Permanente San Diego, California, United States, 92154 California Pacific Medical Center San Francisco, California, United States, 94115 United States, Colorado University of Colorado Denver Aurora, Colorado, United States, 80045 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Georgia Digestive Healthcare of Georgia Atlanta, Georgia, United States, 30309 United States, Louisiana Tulane University Health Sciences Center New Orleans, Louisiana, United States, 70112 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Michigan Henry Ford Health System West Bloomfield, Michigan, United States, 48322 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New York Bronx VA Medical Center Bronx, New York, United States, 10468 Mt. Sinai School of Medicine New York, New York, United States, 10019 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Consultants of Clinical Research Cincinnati, Ohio, United States, 45219 Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Texas Advanced Liver Therapies - Baylor College of Medicine Houston, Texas, United States, 77030 VAMC - Baylor College of Medicine Houston, Texas, United States, 77030 United States, Virginia McGuire Hospital DVAMC Richmond, Virginia, United States, 23249 Collapse << |
| NCT01939873 | Secons Stage of Labor | Not Applicable | Completed | - | Turkey ... more >> Gökhan Kayseri, Turkey, 38039 Collapse << |
| NCT00600912 | Inflammatory Response ... more >> Hepatic Function Collapse << | Phase 4 | Completed | - | Germany ... more >> Klinikum der Stadt Ludwigshafen, Department of Anesthesiology and Intensive Care Medicine Ludwigshafen, Germany, 67063 Collapse << |
| NCT01499420 | Stable Atherothrombotic Diseas... more >>e Collapse << | Phase 2 | Completed | - | United States, California ... more >> Study Site Chula Vista, California, United States, 91911 United States, Florida Study Site Jacksonville, Florida, United States, 32209 Study Site Miami, Florida, United States, 33126 United States, Kentucky Study Site Lexington, Kentucky, United States, 40536 Study Site Madisonville, Kentucky, United States, 42431 United States, Maine Study Site Auburn, Maine, United States, 04210 United States, Maryland Study Site Baltimore, Maryland, United States, 21215 United States, Michigan Study Site Petoskey, Michigan, United States, 49770 United States, North Carolina Study Site Durham, North Carolina, United States, 27710 United States, Pennsylvania Study Site Philadelphia, Pennsylvania, United States, 19104 United States, South Dakota Study Site Rapid City, South Dakota, United States, 57701 Collapse << |
| NCT02219867 | Depressive Disorder, Major | Not Applicable | Unknown | August 2017 | Israel ... more >> Sheba MC Not yet recruiting Ramat Gan, Israel, 5265601 Contact: Revital Amiaz, Dr 972-35303436 revital.amiaz@sheba.health.gov.il Principal Investigator: Revital Amiaz, Dr Collapse << |
| NCT02489331 | - | - | Recruiting | November 2020 | Korea, Republic of ... more >> Daesung Medical Center Recruiting Bucheon, Korea, Republic of, 14577 Isam Clinic Recruiting Busan, Korea, Republic of, 47354 DongRae BongSeng Hospital Recruiting Busan, Korea, Republic of, 47889 Chung Tae Hoon's Internal Medicine Clinic Withdrawn Daegu, Korea, Republic of, 41579 Hyundai Medical Clinic Recruiting Daegu, Korea, Republic of, 42612 Dr. Koh's Internal Medicine Clinic Withdrawn Daegu, Korea, Republic of, 42765 SANGIN Clinic of Internal Medicine Recruiting Daegu, Korea, Republic of, 42812 New Seoul Internal Medicine, Radiology Clinics, Withdrawn Daejeon, Korea, Republic of, 34944 Kim Jong-hak Internal Medicine Clinic Withdrawn Daejeon, Korea, Republic of, 35023 Lee Jong-hyun Internal Medicine Clinic Withdrawn Daejeon, Korea, Republic of, 35023 Kim Tae Young Internal Medicine Withdrawn Daejeon, Korea, Republic of, 35294 Namyangju Hanyang General Hospital Recruiting Gyeonggi-do, Korea, Republic of, 12048 Gyerim Internal Medicine Clinic Withdrawn Gyeongju, Korea, Republic of, 38146 Kim Young Ho Internal Medicine Clinic Recruiting Gyeongsang, Korea, Republic of, 53042 Jeong Internal Medicine Clinic Withdrawn Incheon, Korea, Republic of, 22140 Hankook Geneal Hospital Withdrawn Jeju, Korea, Republic of, 63183 Gujwa Clinic Withdrawn Jeju, Korea, Republic of, 63361 Jeong Nae in Internal Medicine Clinic Withdrawn Seoul, Korea, Republic of, 8706 Sunchon Jungang Hospital Withdrawn Suncheon, Korea, Republic of, 57956 Collapse << |
| NCT00088140 | Hepatitis C, Chronic | Phase 2 | Completed | - | United States, Arizona ... more >> Mayo Clinic Hospital Phoenix, Arizona, United States, 85054 United States, California Scripps Clinic La Jolla, California, United States, 92067 California Pacific Medical Center San Francisco, California, United States, 94115 University of California, San Francisco San Francisco, California, United States, 94143 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Indiana Indiana University School of Medicine Indianapolis, Indiana, United States, 46202 United States, Massachusetts Beth Israel Deaconness Medical Center Boston, Massachusetts, United States, 02215 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, Minnesota The Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New York Mt. Sinai School of Medicine New York City, New York, United States, 10029 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 Duke University Medical Center Durham, North Carolina, United States, 27715 United States, Ohio University of Cincinnati Cincinnati, Ohio, United States, 45267 Metrohealth Medical Center Cleveland, Ohio, United States, 44109 United States, Virginia Medical College of Virginia Richmond, Virginia, United States, 23298 Collapse << |
| NCT03285867 | - | - | Not yet recruiting | December 2021 | - |
| NCT02369536 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Completed | - | Italy ... more >> IRCCS Neuromed Pozzilli, Isernia, Italy, 86077 Collapse << |
| NCT03121521 | Hepatopathy | Phase 4 | Completed | - | - |
| NCT03645655 | - | - | Not yet recruiting | July 30, 2020 | China, Shanghai ... more >> Shanghai Children's Medical Center, Shanghai Jiaotong University School of Medicine Not yet recruiting Shanghai, Shanghai, China, 200127 Contact: Min Xu, Doctor Collapse << |
| NCT01961583 | Kidney Neoplasm | Phase 2 | Terminated(Termination of [18F... more >>]Fluciclatide production) Collapse << | April 2016 | Korea, Republic of ... more >> Asan Medical Center Seoul, Songpa-gu, Korea, Republic of, 138-736 Collapse << |
| NCT02991651 | Lung Cancer, Nonsmall Cell | Phase 1 | Recruiting | December 2019 | United States, New Hampshire ... more >> Geisel School of Medicine at Dartmouth Recruiting Lebanon, New Hampshire, United States, 03756 Contact: Konstantin H Dragnev, MD Collapse << |
| NCT01884038 | Liver Transplant | Phase 2 Phase 3 | Withdrawn | June 2010 | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center, Starzl Transplantation Institute Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT03531593 | - | - | Completed | - | United States, Missouri ... more >> Children's Mercy Hospital Kansas City, Missouri, United States, 64108 Collapse << |
| NCT03047603 | - | - | Recruiting | May 1, 2018 | China, Shanghai ... more >> Eastern hepatobiliary surgery hospital Recruiting Shanghai, Shanghai, China, 200438 Contact: Tian Yang, Doctor +8618917015805 yangtian6666@hotmail.com Collapse << |
| NCT02543983 | Healthy Volunteers ... more >> Depression Collapse << | Phase 2 | Recruiting | July 21, 2021 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT03384511 | Malignancies ... more >>Stomach Cancer Non-small Cell Lung Cancer Esophageal Cancer Breast Cancer Ovary Cancer Cervical Cancer Collapse << | Phase 4 | Completed | - | China, Shandong ... more >> Jinan, Shandong, China, 250117 Collapse << |
| NCT02241278 | Pain, Postoperative | Phase 4 | Completed | - | Spain ... more >> Hospital Arquitecto Marcide Ferrol, A Coruna, Spain, 15405 Collapse << |
| NCT02811627 | Autism Spectrum Disorder | Early Phase 1 | Recruiting | December 2018 | United States, Missouri ... more >> University of Missouri Recruiting Columbia, Missouri, United States, 65211 Contact: David Q Beversdorf, MD 573-884-1871 beversdorfd@health.missouri.edu Collapse << |
| NCT00993941 | Liver Cirrhosis | Phase 2 | Unknown | April 2011 | China, Guangdong ... more >> Sun Yat-sen University Guangzhou, Guangdong, China Collapse << |
| NCT03319199 | Fatty Liver, Nonalcoholic | Not Applicable | Not yet recruiting | December 2018 | - |
| NCT03613480 | Non-Alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Recruiting | March 1, 2020 | Greece ... more >> Department of Gastroenterology Recruiting Patras, Greece, 26504 Contact: Maria Kalogeropoulou, MSc 00306974190208 mariakalogeropoulou@yahoo.com Collapse << |
| NCT01866644 | Unspecified Complication of Li... more >>ver Transplant Collapse << | Phase 3 | Completed | - | Spain ... more >> Hospital Universitari i Politècnic La Fe Valencia, Spain, 46026 Collapse << |
| NCT02878863 | Hepatitis, Autoimmune | Phase 3 | Withdrawn | July 2017 | China, Sichuan ... more >> West China Hospital Chengdu, Sichuan, China, 610041 Collapse << |
| NCT03729973 | Sore Throat | Phase 3 | Active, not recruiting | April 2019 | Egypt ... more >> Zagazig University Hospitsals Zagazig, Egypt, 055 Collapse << |
| NCT01221454 | Liver Failure | Phase 2 | Unknown | November 2011 | China, Guangdong ... more >> The Third Affiliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT01790490 | - | - | Completed | - | - |
| NCT01942590 | Pompe Disease | Phase 1 Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT01942590 | - | - | Completed | - | - |
| NCT01790490 | Cocaine Dependence | Phase 2 | Completed | - | United States, New York ... more >> NYSPI New York, New York, United States, 10032 Collapse << |
| NCT01223664 | Liver Cirrhosis | Phase 2 | Unknown | November 2011 | China, Guangdong ... more >> Sun Yat-sen University Guangzhou, Guangdong, China, 510630 Collapse << |
| NCT03711500 | Schizophrenia ... more >> Schizo Affective Disorder Collapse << | Phase 1 Phase 2 | Not yet recruiting | December 2020 | United States, New York ... more >> New York State Psychiatric Institute New York, New York, United States, 10032 Nathan Kline Institute Orangeburg, New York, United States, 10962 Collapse << |
| NCT02708667 | - | - | Unknown | May 2016 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510000 Contact: Likun Chen, doctor 13798019964 Collapse << |
| NCT03625284 | Non Alcoholic Fatty Liver | Not Applicable | Not yet recruiting | December 10, 2019 | - |
| NCT01007539 | Methamphetamine Dependence | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul St. Mary's Hospital Seoul, Korea, Republic of, 137-701 Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT02647593 | - | - | Unknown | - | - |
| NCT01547949 | Inflammation ... more >>Pain Collapse << | Phase 4 | Completed | - | United States, Oregon ... more >> Oregon Health & Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT00994981 | Living Donor Liver Transplanta... more >>tion Reperfusion Injury Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> The Catholic University of Korea, Seoul Saint Mary's Hospital Seoul, Korea, Republic of, 137-040 Collapse << |
| NCT02067793 | Major Depressive Disorder | Phase 2 | Completed | - | United States, California ... more >> Pacific Research Partners Oakland, California, United States, 94612 United States, Illinois Psychiatric Medicine Associates Skokie, Illinois, United States, 60076 United States, Kansas University of Kansas School of Medicine Clinical Trial Unit Wichita, Kansas, United States, 67214 United States, Massachusetts Boston Clinical Trials, Inc. Roslindale, Massachusetts, United States, 02131 United States, Missouri Psychiatric Care and Research Center O'Fallon, Missouri, United States, 63368 United States, New York Mount Sinai School of Medicine Elmsford, New York, United States, 10523 New York State Psychiatric Institute New York, New York, United States, 10032 United States, Ohio Lindner Center of HOPE Mason, Ohio, United States, 45040 United States, Tennessee Research Strategies Memphis Memphis, Tennessee, United States, 38119 United States, Texas Research Across America Dallas, Texas, United States, 75234 United States, Utah Lifetree Clinical Research Salt Lake City, Utah, United States, 84106 United States, Virginia Psychiatric Alliance of the Blue Ridge Charlottesville, Virginia, United States, 22903 Collapse << |
| NCT00586066 | Bipolar Disorder | Phase 4 | Completed | - | United States, California ... more >> Cedars Sinai Department of Psychiatry Los Angeles, California, United States, 90048 United States, Illinois Asher Depression Center, Northwestern University Chicago, Illinois, United States, 60611 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00586066 | - | - | Completed | - | - |
| NCT01451723 | Multiple Sclerosis | Phase 2 | Terminated(Unusual high freque... more >>ncy of elevated liver function tests.) Collapse << | - | United States, Louisiana ... more >> LSu Health Sciences Center New Orleans, Louisiana, United States, 70112 Collapse << |
| NCT00858624 | Chronic Cough | Not Applicable | Completed | - | United Kingdom ... more >> University of Manchester, Education and Research Centre, Wythenshawe Hospital Manchester, United Kingdom, M23 9LT Collapse << |
| NCT01451723 | - | - | Terminated(Unusual high freque... more >>ncy of elevated liver function tests.) Collapse << | - | - |
| NCT03377283 | Organ Preservation Solutions | Phase 4 | Completed | - | - |
| NCT02011451 | Multiple Sclerosis | Phase 2 | Withdrawn(unable to enroll) | December 2017 | United States, Louisiana ... more >> LSU Health Sciences Center New Orleans, Louisiana, United States, 70112 Collapse << |
| NCT02366364 | Major Depressive Disorder | Phase 1 | Completed | - | United States, Illinois ... more >> Chicago Research Center, Inc. Chicago, Illinois, United States, 60634 Collapse << |
| NCT01918514 | - | - | - | - | United States, New York ... more >> Upstate Comprehensive Pain Medicine Center East Syracuse, New York, United States, 13057 Contact: Shahryar Mousavi, MD 301-728-7094 mousavis@upstate.edu Contact: Behnam Jafarpour, MD 7033404056 Collapse << |
| NCT01765465 | Postcholecystectomy Syndrome | Phase 4 | Completed | - | Korea, Republic of ... more >> DongGuk University Ilsan Hospital Goyang, Gyeonggi, Korea, Republic of, 410-773 Chung-Ang University Hospital Seoul, Korea, Republic of, 156-755 Collapse << |
| NCT02283710 | Fatty Liver | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Gastroenterology clinic, Sina Hospital Tehran, Iran, Islamic Republic of Collapse << |
| NCT00199303 | Efficacy of Dextromethorphan ... more >> Morphine Consumption Pain Collapse << | Not Applicable | Completed | - | Thailand ... more >> Waraporn Chau-in KhonKaen, Thailand, 40002 Collapse << |
| NCT01765465 | - | - | Completed | - | - |
| NCT01342757 | - | - | Completed | - | - |
| NCT03221894 | - | - | Recruiting | September 2017 | China ... more >> Dongzhimen Hospital Recruiting Beijing, China Contact: Jing Shi, M.D. 86-10-84013132 shijing87@hotmail.com Collapse << |
| NCT01887171 | Early Allograft Dysfunction ... more >> Ischemic Reperfusion Injury Liver Transplantation Hyperfibrinolysis Collapse << | Not Applicable | Completed | - | Belarus ... more >> RSPC for organ and tissue transplantation, Minsk 9th clinic Minsk, Belarus, 220116 Collapse << |
| NCT00322023 | Schizophrenia | Phase 2 | Unknown | June 2008 | United States, Connecticut ... more >> Yale University School of Medicine New Haven, Connecticut, United States, 06512 United States, New York The Zucker Hillside Hospital Glen Oaks, New York, United States, 11004 The Nathan S. Kline Institute for Psychiatric Research Orangeburg, New York, United States, 10962 Collapse << |
| NCT01342757 | Adult Glioblastoma ... more >> Depression Recurrent Adult Brain Tumor Collapse << | Not Applicable | Completed | - | United States, Georgia ... more >> Emory University Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01587768 | - | - | Completed | - | - |
| NCT01887171 | - | - | Completed | - | - |
| NCT03613428 | Acute T Cell Leukemia | Phase 1 Phase 2 | Not yet recruiting | March 30, 2021 | - |
| NCT01503359 | Schizophrenia | Phase 2 | Completed | - | Poland ... more >> Central Clinical Hospital Łódź, Czechosłowacka 8/10, Poland, 92-216 Collapse << |
| NCT02929472 | Physical Activity ... more >> Metabolic Diseases Collapse << | Not Applicable | Completed | - | - |
| NCT00136214 | - | - | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00957619 | Liver Regeneration | Phase 4 | Completed | - | Switzerland ... more >> University Hospital Zurich, Department of Visceral and Transplantation Surgery Zurich, Zürich, Switzerland, 8091 Collapse << |
| NCT00136214 | - | - | Completed | - | - |
| NCT03235531 | Alcohol Dependence ... more >> Alcohol Withdrawal Syndrome Trauma Heavy Drinking Alcohol Use Disorder Collapse << | Phase 4 | Recruiting | January 1, 2020 | United States, West Virginia ... more >> Charleston Area Medical Center, General Hospital, Level 1 Trauma Center Recruiting Charleston, West Virginia, United States, 25301 Contact: Audis Bethea, PharmD, BCPS 304-388-3653 audis.bethea@camc.org Principal Investigator: Audis Bethea, PharmD, BCPS Sub-Investigator: Richard Umstot, MD Sub-Investigator: Chelsea Knotts, MD Sub-Investigator: Brent Stover, MA, LPC, ADC Sub-Investigator: Damayanti Samanta, MS Sub-Investigator: Julton Tomanguillo Chumbe, MD Charleston Area Medical Center Recruiting Charleston, West Virginia, United States, 25304 Contact: Audis Bethea, PharmD, BCPS 304-388-3653 audis.bethea@camc.org Principal Investigator: Audis Bethea, PharmD, BCPS Sub-Investigator: Richard Umstot, MD Sub-Investigator: Chelsea Knotts, MD Sub-Investigator: Brent Stover, MA, LPC, ADC Sub-Investigator: Damayanti Samanta, MS Sub-Investigator: Julton Tomanguillo Chumbe, MD Collapse << |
| NCT00947791 | Bipolar Disorder | Phase 4 | Terminated(Change in available... more >> resources for study procedures) Collapse << | - | United States, New York ... more >> Mount Sinai School of Medicine New York, New York, United States, 10029 Collapse << |
| NCT02557724 | - | - | Completed | - | United States, New Jersey ... more >> University Hospital Newark, New Jersey, United States, 07103 Collapse << |
| NCT01971723 | Supplement | Not Applicable | Completed | - | United States, Florida ... more >> Florida State University Tallahassee, Florida, United States, 32306 Collapse << |
| NCT00947791 | - | - | Terminated(Change in available... more >> resources for study procedures) Collapse << | - | - |
| NCT02992470 | Nonalcoholic Fatty Liver | Not Applicable | Unknown | - | - |
| NCT01117441 | Leukemia | Phase 3 | Active, not recruiting | December 2021 | - |
| NCT02505477 | Schizophrenia ... more >> Cognitive Deficits Schizophrenia; Negative Type Collapse << | Phase 4 | Recruiting | March 2019 | United States, California ... more >> UCLA Semel Institute for Neuroscience and Human Behavior Recruiting Los Angeles, California, United States, 90024 Contact: Yvonne Yang, MD,PHD 310-478-3711 ext 42540 ysyang@mednet.ucla.edu Collapse << |
| NCT02450786 | Parkinson's Disease | Phase 2 | Unknown | March 2018 | Korea, Republic of ... more >> Severance Hospital Recruiting Seoul, Korea, Republic of, 120-752 Contact: Young Ho Sohn, MD, PhD 82-2-2228-1601 yhsohn62@yuhs.ac Collapse << |
| NCT01971723 | - | - | Completed | - | - |
| NCT02724332 | Hepatocellular Carcinoma ... more >> Rapamycin Collapse << | Phase 1 | Completed | - | China, Shanghai ... more >> Eastern hepatobilliary surgery hospital Shanghai, Shanghai, China, 200438 Collapse << |
| NCT03460080 | - | - | Recruiting | December 1, 2018 | Vietnam ... more >> Hanoi Medical University Recruiting Hanoi, Vietnam, 100000 Contact: Bui My Hanh, MD +84983070973 buimyhanh@hmu.edu.vn Contact: Nguyen Hoang Hiep, BSc +84964163292 hiepnhh1993@gmail.com Principal Investigator: Tran Ngoc Anh, MD Sub-Investigator: Duong Mai Chi, MD Collapse << |
| NCT00396695 | - | - | Unknown | - | Israel ... more >> Sieff Government Hospital Recruiting Safed, Israel, 13110 Contact: Igor Krasnov, Ph.D + 972 4 682 8828 Principal Investigator: Alexander Mizruchin, Ph.D Sub-Investigator: Igor Krasnov, Ph.D Collapse << |
| NCT01052090 | Hepatitis C Virus Infection | Phase 1 Phase 2 | Terminated | December 2013 | Taiwan ... more >> Kaohsiung Medical University Chung-Ho Memorial Hospital Kaohsiung, Taiwan PhytoHealth Taipei, Taiwan Collapse << |
| NCT00561782 | - | - | Completed | - | United States, Florida ... more >> VA Medical Center, Miami Miami, Florida, United States, 33125 Collapse << |
| NCT02430818 | Acute Pain Fr... more >>actures Collapse << | Not Applicable | Unknown | March 2017 | United States, Missouri ... more >> Barnes Jewish Hospital Recruiting Saint Louis, Missouri, United States, 63110 Contact: Michelle Jenkerson, RN 314-362-5626 jenkersonm@wusm.wustl.edu Collapse << |
| NCT01563484 | - | - | Unknown | September 2012 | China, Jiangsu ... more >> Zhongda Hospital, Southeast University Nanjing, Jiangsu, China, 210009 Collapse << |
| NCT01946360 | Acute Liver Failure ... more >> Acute on Chronic Liver Failure Collapse << | Not Applicable | Completed | - | India ... more >> Institute of Liver & Biliary Sciences New Delhi, Delhi, India, 110070 Collapse << |
| NCT03334747 | Malaria | Phase 2 | Recruiting | May 14, 2019 | Gabon ... more >> Novartis Investigative Site Recruiting Lambarene, Gabon Ghana Novartis Investigative Site Recruiting Kintampo, Ghana Novartis Investigative Site Recruiting Navrango, Ghana Mali Novartis Investigative Site Recruiting Bamako, Mali Novartis Investigative Site Recruiting Sotuba, Mali Rwanda Novartis Investigative Site Recruiting Kigali, Rwanda Uganda Novartis Investigative Site Recruiting Bushenyi, Uganda Novartis Investigative Site Recruiting Kampala, Uganda Novartis Investigative Site Recruiting Tororo, Uganda Collapse << |
| NCT00516711 | Reperfusion Injury | Not Applicable | Completed | - | Switzerland ... more >> University Hospital of Zurich Zurich, Switzerland Collapse << |
| NCT01913470 | NAFLD | Phase 2 | Completed | - | United States, Georgia ... more >> Emory University / Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 Collapse << |
| NCT00210158 | Chronic Pain | Not Applicable | Completed | - | France ... more >> Institut Bergonié - Centre Régional de Luttre Contre le Cancer DE Bordeaux et du Sud Ouest Bordeaux, France, 33076 Collapse << |
| NCT03599960 | Blastic Plasmacytoid Dendritic... more >> Cell Neoplasm (BPDCN) Collapse << | Phase 2 | Not yet recruiting | May 2022 | France ... more >> Chu Amiens Not yet recruiting Amiens, France, 80054 Contact: GRUSON Bérengère, MD Chu Besancon Not yet recruiting Besancon, France, 25030 Contact: DECONINCK ERIC, MD, PhD Collapse << |
| NCT01118663 | Acetaminophen Overdose | Phase 3 | Terminated | - | United States, Arizona ... more >> Maricopa Medical Center Phoenix, Arizona, United States, 85008 United States, California Loma Linda University Medical Center Loma Linda, California, United States, 92350 University of California Irvine Medical Center Orange, California, United States, 92868 UCSD Medical Center San Diego, California, United States, 92103 United States, Colorado University of Colorado Hospital Aurora, Colorado, United States, 80045 Denver Health and Hospital Authority Denver, Colorado, United States, 80204 United States, Connecticut Hartford Hospital Hartford, Connecticut, United States, 06102 United States, Louisiana LSU Health Sciences Center - Shreveport Shreveport, Louisiana, United States, 71130 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 UMass Memorial Medical Center Worcester, Massachusetts, United States, 01655 United States, Michigan Spectrum Health Butterworth Hospital Grand Rapids, Michigan, United States, 44506 United States, North Carolina East Carolina University Medical Center Greenville, North Carolina, United States, 27834 United States, Ohio Toledo Hospital Toledo, Ohio, United States, 43606 United States, Texas Scott & White Medical Center Temple, Texas, United States, 76508 Collapse << |
| NCT01680003 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Unknown | December 2013 | Malaysia ... more >> Sultamah Bahiyah Hospital Recruiting Alor Setar, Kedah, Malaysia, 05460 Contact: Muhammad Radzi Abu Hassan, Dr +604-7407814 drradzi91@yahoo.co.uk Principal Investigator: Muhammad Radzi Abu Hassan, Dr Kuala Lumpur Hospital Recruiting Wilayah Persekutuan, Kuala Lumpur, Malaysia, 50586 Contact: Shashi Kumar Kumar Bhaskaran, Dr 03-26155555 ext 5691 dr_shashi@hotmail.com Principal Investigator: Shashi Kumar Menon Bhaskaran, Dr Tengku Ampuan Afzan Hospital Recruiting Kuantan, Pahang, Malaysia, 25100 Contact: Tee Hoi Poh, Dr 09-5133334 hptee@hotmail.com Principal Investigator: Tee Hoi Poh, Dr Queen Elizaberth Hospital Recruiting Kota Kinabalu, Sabah, Malaysia, 88586 Contact: Jayaram Menon, Dr +(6)088-517555 ext 7117 drmjayaram@gmail.com Principal Investigator: Jayaram Menon, Dr Ampang Hospital Recruiting Selangor, Malaysia, 68000 Contact: Rosaida Mohd Said, Dr 03-42896155 drrosaida@moh.gov.my Principal Investigator: Rosaida Mohd Said, Dr Collapse << |
| NCT01118663 | - | - | Terminated | - | - |
| NCT01913470 | - | - | Completed | - | - |
| NCT03577938 | Acute on Chronic Liver Failure... more >> Traditional Chinese Medicine Hepatitis B Collapse << | Not Applicable | Not yet recruiting | December 30, 2020 | China, Beijing ... more >> 302 military hospital of China Not yet recruiting Beijing, Beijing, China Contact: Ning Zhang +8615010807936 zhangning198191@sina.com Beijing Ditan hospital Not yet recruiting Beijing, Beijing, China Contact: Xianbo Wang +861301001598 wangxianbo638@163.com Beijing youan hospital Not yet recruiting Beijing, Beijing, China Contact: Xiuhui Li +861351273210 lixiuhui@sohu.com China, Fujian Fuzhou infectious hospital Not yet recruiting Fuzhou, Fujian, China, 350025 Contact: Zhengfang Liu +8618906938388 zhengfang1683399@126.com Xiamen hospital of traditional Chinese medicine Not yet recruiting Xiamen, Fujian, China Contact: Jinmo Tang +8613850085858 13850085858@163.com China, Hubei Hubei hospital of traditional Chinese medicine Not yet recruiting Wuhan, Hubei, China Contact: Hanmin Li +8613018089519 lihanmin69@126.com Tongji hospital, Huazhong university of science and technology Not yet recruiting Wuhan, Hubei, China Contact: Zhengang Zhang +8613071289626 zhangzhg@126.com China, Hunan The first affiliated hospital of Hunan university of traditional Chinese medicine Not yet recruiting Changsha, Hunan, China, 410000 Contact: Kewei Sun +86158731490458 keweisun550@163.com China, Jilin Jilin hepatology hospital Not yet recruiting Changchun, Jilin, China Contact: Shuqin Zhang +8613894892233 zhangshuqin2009@126.com China, Liaoning The sixth people's hospital of Shenyang Not yet recruiting Shenyang, Liaoning, China Contact: Ye Gu +8618502460001 gu3311@126.com China, Shanghai Shanghai public health clinical center Not yet recruiting Shanghai, Shanghai, China Contact: Jiefei Wang +861381901651 wjf_sm@126.com Shuguang hospital, Shanghai university of traditional Chinese medicine Not yet recruiting Shanghai, Shanghai, China Contact: Yunhui Zhuo +861396043598 huihuiz1688@hotmail.com China, Sichuan Chengdu public health clinical center Not yet recruiting Chengdu, Sichuan, China Contact: Lin Wang +8613881731673 wang02736@sina.com China, Tianjin The second people's hospital of Tianjin Not yet recruiting Tianjin, Tianjin, China Contact: Wukui Cao +861382186563 tjcaowk@sina.com China, Zhejiang Hangzhou Xixi hospital Not yet recruiting Hangzhou, Zhejiang, China Contact: Jianchun Guo +861398107528 guojianchun1961@126.com Collapse << |
| NCT01142323 | - | - | Terminated | - | - |
| NCT02343029 | Dementia Cogn... more >>itive Impairment Collapse << | Not Applicable | Completed | - | Germany ... more >> Department of Sports Medicine, Institute of Sports Sciences, Goethe University Frankfurt am Main, Germany, 60487 Institute of General Practice, Goethe University, Frankfurt am Main, Germany, 60590 Collapse << |
| NCT00117286 | - | - | Completed | - | - |
| NCT00117286 | Prostate Cancer | Phase 2 Phase 3 | Completed | - | - |
| NCT00215683 | Prostate Cancer | Phase 2 Phase 3 | Completed | - | Belgium ... more >> UCL Saint Luc Bruxelles, Belgium UZ Gent Gent, Belgium UZ Gasthuisberg, Urology Department Leuven, Belgium Germany Vivantes Klinikum am Urban, Klinik für Urologie Berlin, Germany Urologische Universitätsklinikum, Klinikum Mannheim GmbH, Fakultät für Klinische Medizin Mannheim Mannheim, Germany Hungary Bajcsy-Zsilinszky Hospital, of local Government of Budapest, Dept. of Urology Budapest, Hungary Jahn Ferenc Dél-Pesti Hospital, Dept. of Urology Budapest, Hungary Pez Aladar County Hospital, Dept. of Urology Györ, Hungary Bács-Kiskun County Hospital, Dept. of Urology Kecskemét, Hungary BAZ County Hospital, Dept of Urology Miskolc, Hungary Hospital of Local Government of Szeged, Dept. of Urology Szeged, Hungary MÁV Hospital, Dept. of Urology Szolnok, Hungary Netherlands Academic Medical Center, Dept. of Urology Amsterdam, Netherlands Atrium MC, Dept. of Urology Heerlen, Netherlands Romania "Prof.Dr.Th.Burghele" Hospital - Bucharest Bucharest, Romania "Sf. Ioan" Emergency Hospital - Urology Department Bucharest, Romania CF2 Hospital - Bucharest, Urology Department Bucharest, Romania Fundeni Clinical Institute - Bucharest, Urology Department Bucharest, Romania Russian Federation Moscow State University of Medicine and Dentistry, Department of Urology, Urogynecology and Andrology, City Hospital #50, Urology Department Moscow, Russian Federation Russian Medical Academy of Postgraduate Education, Department of Gerontolology and Geriatrics, Moscow City Hospital #60, Urology Department, 84/1, Entuziastov Shosse (Hospital) Moscow, Russian Federation Russian Medical Academy of Postgraduate Education, Urology Department, Moscow Clinical Hospital n.a. Botkin, Urology Department, 5/16, 2-oy Botkinsky proezd (Hospital) Moscow, Russian Federation Russian State Medical University, Department of Urology and Surgical Nephrology, Moscow City Hospital #1 Moscow, Russian Federation City Hospital #15, Urology Department St Petersburg, Russian Federation City Hospital #26, Urology Department St Petersburg, Russian Federation I.I. Mechnikov St. Petersburg State Medical Academy, Urology Department St Petersburg, Russian Federation Research Institute of Urology of the Ministry of Healthcare of the Russian Federation St Petersburg, Russian Federation St. Petersburg Pavlov Medical School, Urology Department St Petersburg, Russian Federation St.Petersburg Pavlov Medical School, Outpatient Diagnostic Center St Petersburg, Russian Federation "Andros" Urology Clinic, 36A, Lenina St. St. Petersburg, Russian Federation, 197136 South Africa Glenwood Hospital Durban, South Africa WITS Medical School, Level 9 Parktown, South Africa 401 B Medical Centre Pietermaritzburg, South Africa Pretoria Urology Hospital, Suite 2, Hatfield Pretoria, South Africa Sunninghill Clinic, Suite 3 Sunninghill, South Africa Collapse << |
| NCT00268892 | Prostate Cancer | Phase 2 Phase 3 | Completed | - | Belgium ... more >> UZ Gasthuisberg Leuven Leuven, Belgium Finland Helsinki University Hospital, Maria Hospital, Building 11 Helsinki, Finland Central Hospital, North Karelian Joensuu, Finland Oulu University Hospital Oulu, Finland Tampere University Hospital Tampere, Finland France Fédération d'Urologie et Néphrologie, BP69 Hôpital Pasteur Nice, France Germany Gemeinschaftspraxis Dres Effert und Benedic Aachen, Germany Montenegro Clinical Center Novi Sad, Clinic of Urology Novi Sad, Montenegro Netherlands Academic Medical Center, Urology Amsterdam, Netherlands St. Elisabeth Hospital Tilburg, Netherlands Romania "Centrul Medical Privat" Prof. Dr. Ioiart Ioan" Arad, Romania Clinical Hospital "Prof. Dr. Theodor Burghele", Urology Department Bucharest, Romania University CF Hospital No. 2 Bucharest, Romania Russian Federation Andros Clinic St. Petersburg, Russian Federation City Hospital #15 St. Petersburg, Russian Federation City Hospital #26 St. Petersburg, Russian Federation Pavlov State Medical University, Outpatient Diagnostic Center affiliated with the Urology Department St. Petersburg, Russian Federation Pavlov State Medical University, Urology Department St. Petersburg, Russian Federation Serbia Clinical Center of Serbia, Institute of Urology and Nephrology Belgrade, Serbia United Kingdom Mount Vernon Cancer Centre, Marie Cuire Research Wing Northwood, Middlesex, United Kingdom Castle Hill Hospital Hull, North Humberside, United Kingdom Ward 13, NHS Forth Valley Acute Operating Division, Falkirk and District Royal Infirmary, Majors Loans Falkirk, United Kingdom Level 7, Urology Research Unit, Derriford Hospital Plymouth, United Kingdom Collapse << |
| NCT01142323 | Primary Sclerosing Cholangitis | Phase 1 Phase 2 | Terminated | - | United States, Florida ... more >> University of Florida Gainesville, Florida, United States, 32608 University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT02698176 | - | - | Terminated(This study was term... more >>inated due to limited efficacy and not due to safety reasons.) Collapse << | - | - |
| NCT02698176 | NUT Midline Carcinoma (NMC) ... more >> Triple Negative Breast Cancer (TNBC) Non-small Cell Lung Cancer (NSCLC) Castration-resistant Prostate Cancer (CRPC) Collapse << | Phase 1 | Terminated(This study was term... more >>inated due to limited efficacy and not due to safety reasons.) Collapse << | - | - |
| NCT00117312 | Prostate Cancer | Phase 2 | Terminated(After 190 weeks [7 ... more >>patients left] it was terminated for administrative reasons) Collapse << | - | United States, California ... more >> Advanced Urology Medical Center Anaheim, California, United States, 92801 South Orange County Medical Research Center Laguna Woods,, California, United States, 92653 San Bernardino Urological Associates Medical Group San Bernardino, California, United States, 92404 Western Clinical Research Torrance, California, United States, 90505 United States, Colorado Urology Associate PC` Denver, Colorado, United States, 80210 United States, Florida SW Florida Urological Associates Fort Myers, Florida, United States, 33907 Pinellas Urology, Inc. St. Petersburg, Florida, United States, 33710 United States, Maryland Drs. Werner, Murdock & Francis, PA Greenbelt, Maryland, United States, 20770 United States, Nevada Nevada Urology Associates Reno, Nevada, United States, 89511 United States, Oklahoma Urology Specialists of Oklahoma, Inc. Tulsa, Oklahoma, United States, 74104 United States, Texas Urology Clinics of NorthTexas, PA Dallas, Texas, United States, 75231 Urology San Antonio Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT02230683 | Liver Cirrhosis ... more >> Hepatic Cirrhosis Portal Hypertension Collapse << | Phase 2 | Completed | - | United States, Connecticut ... more >> VA Connecticut Healthcare System West Haven, Connecticut, United States, 06516 United States, District of Columbia Johns Hopkins Sibley Memorial Hospital Washington, District of Columbia, United States, 20016 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Mississippi University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 United States, New Jersey Rutgers New Jersey Medical School Newark, New Jersey, United States, 07103 United States, New York North Shore University Hospital Manhasset, New York, United States, 11030 New York University Lagone Medical Center NYC, New York, United States, 10016 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Albert Einstein Medical Center Philadelphia, Pennsylvania, United States, 19141 United States, Texas St. Luke's Health Baylor College of Medicine Houston, Texas, United States, 77030 University of Texas Health Science Center at Houston Houston, Texas, United States, 77030 United States, Utah University of Utah Hospital Salt Lake City, Utah, United States, 84132 United States, Virginia Bon Secours Mary Immaculate Hospital Newport News, Virginia, United States, 23602 Bon Secours St. Mary's Hospital Richmond, Virginia, United States, 23226 McGuire DVAMC Richmond, Virginia, United States, 23249 Collapse << |
| NCT00117312 | - | - | Terminated(After 190 weeks [7 ... more >>patients left] it was terminated for administrative reasons) Collapse << | - | - |
| NCT00215683 | - | - | Completed | - | - |
| NCT00215657 | Prostate Cancer | Phase 2 | Terminated(The doses were not ... more >>effective in maintaining testosterone suppression) Collapse << | - | - |
| NCT00268892 | - | - | Completed | - | - |
| NCT00245466 | Prostate Cancer | Phase 2 | Terminated(The maintenance dos... more >>es were too low to maintain testosterone suppression) Collapse << | - | - |
| NCT02230683 | - | - | Completed | - | - |
| NCT00215657 | - | - | Terminated(The doses were not ... more >>effective in maintaining testosterone suppression) Collapse << | - | - |
| NCT01519921 | Hepatitis B, Chronic | Phase 4 | Completed | - | Korea, Republic of ... more >> Busan, Korea, Republic of, 49241 Daegu, Korea, Republic of, 41944 Seoul, Korea, Republic of, 03722 Seoul, Korea, Republic of, 05505 Seoul, Korea, Republic of, 06351 Seoul, Korea, Republic of, 08308 Seoul, Korea, Republic of, 14647 Collapse << |
| NCT01519921 | - | - | Completed | - | - |
| NCT00200343 | Chronic Hepatitis C | Phase 3 | Completed | - | Japan ... more >> Department of Gastroenterology, University of Tokyo hongo, bunkyo-ku, Tokyo, Japan Collapse << |
| NCT00245466 | - | - | Terminated(The maintenance dos... more >>es were too low to maintain testosterone suppression) Collapse << | - | - |
| NCT00200343 | - | - | Completed | - | - |
| NCT03194893 | Neoplasms | Phase 3 | Recruiting | June 12, 2024 | - |
| NCT01291407 | Gastrointestinal Neoplasms ... more >> Gastric Cancer Collapse << | Phase 1 | Completed | - | China ... more >> Department of Radiation Oncology, Cancer Hospital, Chinese Academy of Medical Sciences (CAMS) and Peking Union Medical College (PUMC) Beijing, China, 100021 Collapse << |
| NCT00982865 | Solid Tumors ... more >>Cancer Collapse << | Phase 1 | Completed | - | Australia ... more >> Westmead Hospital Westmead, Australia Belgium Jules Bordet Institute Brussels, Belgium, B-1000 Ghent University Hospital Ghent, Belgium France Institute Bergonié Bordeaux, France Hôpital Beaujon Paris, France, 92118 Hopital Saint Louis Paris, France Centre Eugène Marquis Rennes, France Institute Claudius Regaud Toulouse, France, 03 31052 Netherlands Netherlands Cancer Institute - Antonie van Leeuwenhoek Hospital Amsterdam, Netherlands Collapse << |
| NCT01281774 | Healthy | Phase 1 | Completed | - | Australia, Queensland ... more >> Q-Pharm Brisbane, Queensland, Australia, 4006 Collapse << |
| NCT01165216 | Non-Small Cell Lung Cancer | Phase 1 | Completed | - | Japan ... more >> Local Institution Chuo-ku, Tokyo, Japan, 1040045 Collapse << |
| NCT00714675 | Metabolism El... more >>derly Collapse << | Not Applicable | Completed | - | France ... more >> Hôpital Emile Roux Limeil-Brevannes, France, 94 Collapse << |
| NCT01877759 | Liver Cirrhosis | Phase 1 Phase 2 | Unknown | December 2014 | India ... more >> Chaitanya Hospital Not yet recruiting Pune,, Maharashtra, India, 411009 Contact: Sachin P Jamadar, D ORTHO +918888788880 sac2751982@gmail.com Contact: Smita S Bhoyar, BAMS.PGCR 9372620569 drsmitabhoyar@rediffmail.com Principal Investigator: Anant E Bagul, MS,Ortho Collapse << |
| NCT00069550 | Rett Syndrome | Phase 3 | Unknown | - | United States, Maryland ... more >> Kennedy Krieger Institute Recruiting Baltimore, Maryland, United States Contact: SakkuBai R. Naidu, MD 443-923-2778 Contact: Genila Bibat, MD 443-923-2778 bibat@kennedykrieger.org Collapse << |
| NCT00982865 | - | - | Completed | - | - |
| NCT01833546 | - | - | Completed | - | - |
| NCT02259114 | NUT Midline Carcinoma ... more >> Triple Negative Breast Cancer Non-small Cell Lung Cancer With Rearranged ALK Gene/Fusion Protein or KRAS Mutation Castrate-resistant Prostate Cancer CRPC Pancreatic Ductal Adenocarcinoma Collapse << | Phase 1 | Completed | - | - |
| NCT01833546 | Solid Tumor P... more >>ancreatic Cancer Collapse << | Phase 1 | Completed | - | Japan ... more >> Research Site Kashiwa, Japan Research Site Tokyo, Japan Collapse << |
| NCT00756080 | Healthy Subjects | Phase 1 | Completed | - | France ... more >> CHU Nantes Nantes, France, 44093 Collapse << |
| NCT02259114 | - | - | Completed | - | - |
| NCT01073202 | Liver Transplantation ... more >> Ischemia-reperfusion Injury Cholestasis Collapse << | Not Applicable | Completed | - | China, Shanghai ... more >> Shanghai First People's Hospital Shanghai, Shanghai, China, 200080 Collapse << |
| NCT03130920 | Liver Metastases | Not Applicable | Completed | - | Croatia ... more >> University Hospital Dubrava Zagreb, Croatia, 10000 Collapse << |
| NCT02937012 | Primary Biliary Cirrhosis | Phase 3 | Recruiting | December 2019 | Mexico ... more >> Instituto Nacional de Ciencias Medicas y Nutricion Salvador Zubiran Recruiting Mexico City, Mexico, 14000 Contact: Edgardo Eric Lopez Mendez, MD (01)(55) 54870900 ext 2710 ericlopezmendez@yahoo.com.mx Contact: Sergio Gabriel Munoz Martinez, MD (01)(55) 54870900 ext 2710 sergio_sg@hotmail.com Principal Investigator: Edgardo Eric Lopez Martinez, MD Sub-Investigator: Sergio Gabriel Munoz Martinez, MD Sub-Investigator: Ignacio Garcia Juarez, MD Sub-Investigator: Ernesto Marquez Guillen, MD Sub-Investigator: Carlos Moctezuma Velazquez, MD Sub-Investigator: Alejandra Tepox Padron, MD Collapse << |
| NCT01165216 | - | - | Completed | - | - |
| NCT03142763 | Cardiovascular Risk Factor | Not Applicable | Active, not recruiting | December 2018 | United States, Connecticut ... more >> University of Connecticut Storrs, Connecticut, United States, 06269 Collapse << |
| NCT03489889 | Ursodeoxycholic Acid ... more >> Primary Biliary Cirrhosis Collapse << | Not Applicable | Completed | - | Brazil ... more >> Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo São Paulo, Brazil, 05403-000 Collapse << |
| NCT02515708 | Liver Transplantation | Not Applicable | Recruiting | September 2020 | United States, Ohio ... more >> Cleveland Clinic Recruiting Cleveland, Ohio, United States, 44195 Contact: Susan Thomas, RN 216-444-5930 thomass4@ccf.org Contact: Cristiano Quintini, MD 216 445-3388 quintic@ccf.org Collapse << |
| NCT03751917 | - | - | Not yet recruiting | December 2023 | Italy ... more >> S.O.C. di Ematologia - A.O. - SS. Antonio e Biagio e Cesare Arrigo Not yet recruiting Alessandria, Italy Contact: Ladetto UO Ematologia con trapianto-Universita' degli Studi di Bari Aldo Moro Not yet recruiting Bari, Italy Contact: Specchia Azienda Ospedaliera - Papa Giovanni XXIII Not yet recruiting Bergamo, Italy Contact: Rambaldi Spedali Civili - Brescia - Azienda Ospedaliera - U.O. Ematologia Not yet recruiting Brescia, Italy Contact: Rossi CTMO - Ematologia - Ospedale "Binaghi" Not yet recruiting Cagliari, Italy Contact: Cabras A.O. Pugliese Ciaccio - Presidio Ospedaliero A.Pugliese - U.O. di Ematologia Not yet recruiting Catanzaro, Italy Contact: Molica U.O.C. di Ematologia 1 e 2 IRCCS AOU San Martino-IST Not yet recruiting Genova, Italy Contact: Angelucci Fondazione IRCCS Ca' Granda Osp. Maggiore Policlinico UOC Oncoematologia Not yet recruiting Milano, Italy Contact: Cortelezzi Ospedale Niguarda " Ca Granda" - SC Ematologia Not yet recruiting Milano, Italy Contact: Cairoli U.O. Ematologia e Trapianto di MIdollo - Ist.Scientifico Ospedale San Raffaele Not yet recruiting Milano, Italy Contact: Ciceri Azienda Ospedaliera "S.Gerardo" Not yet recruiting Monza, Italy Contact: Pioltelli Napoli Azienda Ospedaliera di Rilievo Nazionale "A. Cardarelli" Not yet recruiting Naples, Italy Contact: Ferrara Ospedali Riuniti "Villa Sofia-Cervello" Not yet recruiting Palermo, Italy Contact: Fabbiano U.O. Ematologia Clinica - Azienda USL di Pescara Not yet recruiting Pescara, Italy Contact: Pescara Universita' "Sapienza" - Dip Biotecnologie Cellulari - Divisione di Ematologia Roma, Italy Universita' Cattolica del Sacro Cuore - Policlinico A. Gemelli Not yet recruiting Roma, Italy Contact: De Stefano Universita' degli Studi - Policlinico di Tor Vergata Not yet recruiting Roma, Italy Contact: Arcese Università degli studi di Tor Vergata Not yet recruiting Roma, Italy Contact: Francesco Lo Coco Principal Investigator: Francesco Lo Coco stituto di Ematologia - IRCCS Ospedale Casa Sollievo della Sofferenza Not yet recruiting San Giovanni Rotondo, Italy Contact: Cascavilla Dipartimento di Oncologia Ematologia 2 A.O. Citta' della Salute S. G. Battista Not yet recruiting Torino, Italy Contact: Vitolo ULSS N.6 Osp. S. Bortolo Not yet recruiting Vicenza, Italy Contact: Ruggeri Collapse << |
| NCT01786759 | Preterm Infants | Phase 4 | Unknown | July 2013 | China, Shanghai ... more >> Xinhua hospital Recruiting Shanghai, Shanghai, China, 200092 Contact: Yexuan Tao, PhD 13818334664 taoyexuan@hotmail.cm Principal Investigator: Wei Cai, PhD Principal Investigator: Ying Wang, PhD Sub-Investigator: Kejun Zhou, PhD Sub-Investigator: Qingya Tang, MD Sub-Investigator: Lina Lu, PhD Collapse << |
| NCT02452749 | Hypertension | Not Applicable | Completed | - | United States, Oregon ... more >> Helfgott Research Institute Portland, Oregon, United States, 97201 Collapse << |
| NCT01153269 | - | - | Completed | - | - |
| NCT00313378 | Partial Pneumonectomy Under La... more >>teral or Posterolateral Thoracotomy. Collapse << | Phase 3 | Completed | - | France ... more >> Clermont-Ferrand University Hospital Clermont-Ferrand, Auvergne, France, 63000 Collapse << |
| NCT01443572 | Kasai Operation ... more >> Biliary Atresia Collapse << | Not Applicable | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT01974336 | Short Bowel Syndrome | Phase 4 | Completed | - | China, Jiangsu ... more >> Jinling Hospital Nanjing, Jiangsu, China, 200002 Collapse << |
| NCT03568266 | - | - | Not yet recruiting | November 1, 2024 | United States, California ... more >> USC / Norris Comprehensive Cancer Center Not yet recruiting Los Angeles, California, United States, 90033 Contact: Houda Alachkar 323-442-2696 alachkar@usc.edu Principal Investigator: Houda Alachkar Collapse << |
| NCT00276263 | Schizophrenias ... more >> Psychoses Psychotic Disorders Schizophrenic Disorders Collapse << | Phase 2 | Withdrawn(lack of participants... more >>) Collapse << | - | Israel ... more >> Ezrath Nashim - Herzog Memorial Hospital Jerusalem, Israel, 91035 Department of Psychiatry, Hadassah Hospital and Community Clinics Jerusalem, Israel Collapse << |
| NCT03711669 | - | - | Recruiting | April 2021 | Spain ... more >> Hospital Clinic Barcelona Recruiting Barcelona, Spain, 08036 Contact: Maria C Londoño, MD, PhD Hospital del Mar Recruiting Barcelona, Spain Contact: Montserrat Garcia-Retortillo Collapse << |
| NCT00573313 | - | - | Completed | - | - |
| NCT01153269 | - | - | Completed | - | Germany ... more >> Site Reference ID/Investigator# 27575 Berlin, Germany, 10243 Site Reference ID/Investigator# 27592 Berlin, Germany, 10961 Site Reference ID/Investigator# 27588 Dortmund, Germany, 44137 Site Reference ID/Investigator# 27583 Frankfurt, Germany, 60311 Site Reference ID/Investigator# 27587 Frankfurt, Germany, 60596 Site Reference ID/Investigator# 27607 Hamburg, Germany, 20099 Site Reference ID/Investigator# 5355 Krefeld, Germany, 47800 Site Reference ID/Investigator# 27604 Muenster, Germany, 48149 Collapse << |
| NCT03596684 | Atorvastatin | Not Applicable | Not yet recruiting | May 1, 2020 | France ... more >> Endocrinology and diabetology service, Louis Pradel Hospital Not yet recruiting Bron, France, 69500 Contact: Sybil CHARRIERE, MD 4 27 85 66 66 ext +33 sybil.charriere@chu-lyon.fr Sub-Investigator: Philippe MOULIN, MD Principal Investigator: Sybil CHARRIERE, MD Sub-Investigator: Laure Groisne, MD Collapse << |
| NCT00573313 | Liver Disease, Alcoholic | Phase 3 | Completed | - | United States, California ... more >> University of California, Davis Medical Center Sacramento, California, United States, 95817 Collapse << |
| NCT03219320 | Diabetic Peripheral Neuropathy | Phase 2 | Recruiting | December 30, 2018 | - |
| NCT03249103 | Fibromyalgia | Phase 2 | Recruiting | March 30, 2019 | United States, Michigan ... more >> Aptinyx Clinical Site Recruiting Ann Arbor, Michigan, United States, 48109 United States, Ohio Aptinyx Clinical Site Recruiting Cincinnati, Ohio, United States, 45220 Collapse << |
| NCT03299829 | - | - | Recruiting | October 30, 2018 | Taiwan ... more >> Chang Gung Memorial Hospital, Linkou Recruiting Taoyuan City, Taiwan, 333 Contact: Xinyu Huang, Master +886-2-2655-7568 ext 663 tiffany.huang@excelsiorgroup.com.tw Contact: Lijun Chen, Bachelor +886-989-684025 puppy.7151@gmail.com Collapse << |
| NCT03071133 | - | - | Recruiting | November 14, 2019 | Japan ... more >> Local Institution Recruiting Tokyo, Japan, 1620814 Contact: Site 0001 Collapse << |
| NCT00380718 | - | - | Completed | - | - |
| NCT00380718 | Non-small Cell Lung Cancer | Phase 4 | Completed | - | Taiwan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taichung, Taiwan, 407 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taipei, Taiwan, 112 Collapse << |
| NCT02452177 | Liver Disease | Not Applicable | Unknown | February 2016 | China, Jiangsu ... more >> Jinling Hospital Recruiting Nanjing, Jiangsu, China, 210002 Contact: Yousheng Li, M.D., Ph.D. +86 25 80860137 liyousheng6666@126.com Collapse << |
| NCT01973608 | Melanoma | Phase 2 | Terminated(The study is termin... more >>ated prematurely as the sponsor decided to discontinue program of NHS-IL2 [MSB0010445]) Collapse << | - | United States, Massachusetts ... more >> Please Contact U.S. Medical Information Rockland, Massachusetts, United States Collapse << |
| NCT01973608 | - | - | Terminated(The study is termin... more >>ated prematurely as the sponsor decided to discontinue program of NHS-IL2 [MSB0010445]) Collapse << | - | - |
| NCT02478788 | Attention Deficit Hyperactivit... more >>y Disorder Collapse << | Phase 4 | Recruiting | August 2020 | United States, Ohio ... more >> University of Cincinnati, Department of Psychiatry and Behavioral Neuroscience Recruiting Cincinnati, Ohio, United States, 45219 Contact: Leah Fleischer 513-558-3674 fleisclm@ucmail.uc.edu Principal Investigator: Robert K McNamara, PhD Principal Investigator: Melissa P DelBello, MD, MS Collapse << |
| NCT00657618 | - | - | Completed | - | - |
| NCT00657618 | Leishmaniasis | Phase 1 Phase 2 | Completed | - | United States, District of Col... more >>umbia Walter Reed National Military Medical Center Washington, District of Columbia, United States, 20307 Collapse << |
| NCT02984475 | Beta Thalassemia Major Anemia | Phase 4 | Recruiting | April 2019 | Egypt ... more >> Abo El Reesh Hospital Recruiting Cairo., Egypt Contact: Mona Gaber, Bachelor. 0223648368 mona.sobhy@pharma.cu.edu.eg El Demerdash (Ain Shams Teaching Hospital). Completed Cairo., Egypt Collapse << |
| NCT02077374 | - | - | Completed | - | - |
| NCT03453047 | - | - | Recruiting | December 20, 2018 | Turkey ... more >> Akdeniz University Medical Faculty Department of Anesthesiology and Reanimation Recruiting Antalya, Turkey, 07059 Contact: BORA DINC, ASSIST. PROF. +905323865600 drboradinc@gmail.com Contact: ILKER ONGUC AYCAN, ASSOC. PROF +905056702335 ilkeraycan@gmail.com Principal Investigator: BORA DINC, ASSIST. PROF Sub-Investigator: ILKER ONGUC AYCAN, ASSOC. PROF. Collapse << |
| NCT00095147 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT02143323 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Jieon Lee Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT02322879 | Acetaminophen Toxicity | Phase 4 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00095147 | - | - | Completed | - | - |
| NCT02322879 | - | - | Completed | - | - |
| NCT02744105 | Beta Thalassemia Major | Not Applicable | Completed | - | Egypt ... more >> Faculty of Medicine- Tanta University Tanta, Gharbia, Egypt, 0000 Collapse << |
| NCT03769948 | - | - | Not yet recruiting | July 2020 | - |
| NCT01380366 | Short Bowel Syndrome | Phase 4 | Completed | - | United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << |
| NCT01856920 | Medullary Thyroid Cancer (MTC) | Phase 2 | Active, not recruiting | March 1, 2020 | United States, Maryland ... more >> NIH Clinical Center Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03612973 | Chronic Hepatitis c | Not Applicable | Not yet recruiting | December 31, 2021 | - |
| NCT02077374 | Nonalcoholic Steatohepatitis ... more >> Non-alcoholic Fatty Liver Disease Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, Minnesota Mayo Clinic Rochester Rochester, Minnesota, United States, 55905 United States, Missouri Kansas City Research Institute Kansas City, Missouri, United States, 64131 United States, Virginia Mary Immaculate Hospital Newport News, Virginia, United States, 23602 Bon Secours St. Mary's Hospital Richmond, Virginia, United States, 23226 Collapse << |
| NCT02331589 | - | - | Completed | - | - |
| NCT02331589 | Fatigue | Phase 4 | Completed | - | - |
| NCT01380366 | - | - | Completed | - | - |
| NCT03327402 | Attention Deficit Hyperactivit... more >>y Disorder (ADHD) Collapse << | Phase 1 | Recruiting | October 11, 2018 | United States, Arkansas ... more >> Preferred Research Partners Recruiting Little Rock, Arkansas, United States, 72211 United States, Florida Clinical Neuroscience Solutions Inc Recruiting Orlando, Florida, United States, 32801 United States, Ohio Professional Psychiatric Services (PPS) Recruiting Mason, Ohio, United States, 45040 United States, Tennessee Clinical Neuroscience Solutions Inc Recruiting Memphis, Tennessee, United States, 38119 Collapse << |
| NCT02744547 | Beta Thalassemia Major | Not Applicable | Active, not recruiting | December 2017 | Egypt ... more >> Faculty of Medicine- Tanta University Tanta, Gharbia, Egypt, 0000 Collapse << |
| NCT01431521 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 1 | Completed | - | - |
| NCT00060528 | - | - | Completed | - | - |
| NCT01431521 | - | - | Completed | - | - |
| NCT00060528 | Prostatic Neoplasms | Phase 1 Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01462552 | - | - | Completed | - | - |
| NCT01654549 | - | - | Completed | - | - |
| NCT02213094 | - | - | Completed | - | - |
| NCT00665678 | Posttraumatic Stress Disorder ... more >>(PTSD) Collapse << | Phase 4 | Terminated | - | United States, Georgia ... more >> Emory University Atlanta, Georgia, United States, 30306 Collapse << |
| NCT00935012 | TTR-CM | Phase 3 | Active, not recruiting | December 2021 | United States, Illinois ... more >> University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Massachusetts Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 United States, New York Clinical Cardiovascular Research Lab for the Elderly (CCRLE) Columbia Universtiy New York, New York, United States, 10034 Collapse << |
| NCT00136318 | - | - | Completed | - | - |
| NCT03091634 | Qi Stagnation and Blood Stasis... more >> Syndrome Collapse << | Phase 2 | Recruiting | December 1, 2018 | China ... more >> Guang'anmen Hospital, China Academy of Chinese Medical Sciences Recruiting Beijing, China, 100053 Contact: Jie Wang, professor 8610-88001817 wangjie0103@126.com Contact: Haoqiang He, master 8610-88001817 MyStrong@bucm.edu.cn Sub-Investigator: Jie Wang, professor Sub-Investigator: Haoqiang He, master Sub-Investigator: Guang Chen, doctor Sub-Investigator: Jialiang Gao, doctor Sub-Investigator: Qingyong He, professor Huguosi TCM Hospital Affiliated to Beijing University of CM Recruiting Beijing, China Contact: Yu Liu, master 0086-13810113116 Principal Investigator: Yu Liu, master Wangjing Hospital, China Academy of Chinese Medical Sciences Recruiting Beijing, China Contact: Chenhao Zhang, professor 0086-13671393316 Principal Investigator: Chenhao Zhang, professor Collapse << |
| NCT01654549 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Gastroenterology clinic, Sina hospital. Tehran, Iran, Islamic Republic of Collapse << |
| NCT02213094 | Pregnancy Induced Hypertension... more >> Superimposed Preeclampsia Hypertension Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> University of North Carolina Women's Hospital Chapel Hill, North Carolina, United States, 27599-7516 Collapse << |
| NCT01939574 | Glioblastoma | Not Applicable | Unknown | September 2016 | China, Shandong ... more >> Shandong Cancer Hospital and Institute Jinan, Shandong, China, 250117 Collapse << |
| NCT03666832 | Metastatic Pancreatic Cancer | Phase 1 Phase 2 | Not yet recruiting | December 30, 2019 | - |
| NCT01465191 | Postoperative Pain | Phase 4 | Unknown | November 2016 | United States, District of Col... more >>umbia George Washington University Medical Center Not yet recruiting Washington, District of Columbia, United States, 20037 Contact: Jeff Berger, MD 202-715-5296 jberger@mfa.gwu.edu United States, New York Columbia University Medical Center Recruiting New York, New York, United States, 10032 Principal Investigator: Richard M Smiley, MD, PhD Collapse << |
| NCT00136318 | Depression | Phase 3 | Completed | - | Germany ... more >> Department of Gastroenterolgy and Rheumatology, Sektion Hepatology Leipzig, Germany, 04103 Collapse << |
| NCT02684019 | Lack of Drug Action | Phase 1 Phase 2 | Unknown | July 2016 | Egypt ... more >> Theodor Bilharz Research Institute Recruiting Giza, Egypt, 11221 Contact: khalda G Radwan, Professor 00201220411852 khalda.radwan31@gmail.com Principal Investigator: Hend H Kamel, Professor Principal Investigator: Maher F Mahmoud, Professor Principal Investigator: Khalda G Radwan, Professor Sub-Investigator: Nabaweya M Kamal, Professor Sub-Investigator: Mohammed A Maher, Lecturer Sub-Investigator: Eslam A Mohamed, Lecturer Sub-Investigator: Ahmed S Abd El Azeem, Residant Collapse << |
| NCT00986063 | Nevirapine Induced Rash ... more >> Nevirapine Induced Hepatitis HIV Adverse Side Effects AIDS HIV Infections Collapse << | Phase 4 | Completed | - | Thailand ... more >> Division of Infectious Diseases, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University Bangkok, Thailand, 10400 Collapse << |
| NCT02235246 | Gastric Neoplasm | Phase 4 | Completed | - | Korea, Republic of ... more >> Sevrance hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT00483028 | Alzheimer's Disease | Not Applicable | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States United States, New York Pfizer Investigational Site Albany, New York, United States Collapse << |
| NCT03430388 | Systemic Lupus ... more >> Rheumatoid Arthritis Spondyloarthritis Inflammatory Myopathy Systemic Sclerosis Mixed Connective Tissue Disease Takayasu Arteritis Granulomatosis With Polyangiitis Sjogren's Syndrome Juvenile Idiopathic Arthritis Juvenile Dermatomyositis Collapse << | Not Applicable | Recruiting | February 24, 2019 | Brazil ... more >> Rheumatology Division of Hospital das Clínicas da Faculdade de Medicina da Universidade de Sao Paulo Recruiting São Paulo, Sao Paulo, Brazil, 05403-000 Contact: Eloisa Bonfa, MD, PhD 55 11 30617490 eloisa.bonfa@hc.fm.usp.br Collapse << |
| NCT02430285 | Acute Biliary Pancreatitis | Not Applicable | Completed | - | Italy ... more >> Endoscopy Unit, Gastroenterology Department, Humanitas Research Hospital Rozzano, Milan, Italy, 20089 Collapse << |
| NCT02711683 | - | - | Recruiting | March 2018 | China, Shaanxi ... more >> First Affiliated Hospital of Xi'an Jiaotong University Recruiting Xi'an, Shaanxi, China, 710061 Contact: Jin Wang 0086-13572208524 drwangjin@163.com Collapse << |
| NCT01481207 | - | - | Recruiting | December 2019 | Switzerland ... more >> University Children's Hospital Zurich, MRI Center Recruiting Zürich, Switzerland, 8032 Collapse << |
| NCT02399397 | Sepsis System... more >>ic Inflammatory Response Syndrome Septic Shock Collapse << | Phase 4 | Completed | - | Brazil ... more >> Universidade Estadual Paulista Júlio de Mesquita Filho Araraquara, São Paulo, Brazil, 14801902 Collapse << |
| NCT02892916 | Post Operative Cognitive Dysfu... more >>nction Collapse << | Phase 3 | Recruiting | July 2019 | France ... more >> AP-HP - Hôpital Saint-Antoine Recruiting Paris, Île-de-France, France, 75012 Contact: Franck Verdonk, MD franck.verdonk@aphp.fr Collapse << |
| NCT01784484 | - | - | Unknown | - | Israel ... more >> Ziv Medical Center Not yet recruiting Safed, Israel, 13100 Contact: Nimer Assy, MD +972-4-6828442 assy.n@ziv.health.gov.il Collapse << |
| NCT01818830 | - | - | Unknown | - | China, Beijing ... more >> Chinese PLA General Hospital Beijing, Beijing, China, 100853 Collapse << |
| NCT02045563 | - | - | Unknown | - | France ... more >> CHU de DIJON Recruiting Dijon, France, 21079 Contact: Jean-Michel PETIT 3 80 29 34 53 ext +33 jean-michel.petit@chu-dijon.fr Collapse << |
| NCT03213392 | - | - | Completed | - | India ... more >> Postgraduate Institute of Medical Education and Research Chandigarh, India, 160012 Collapse << |
| NCT02537704 | Obesity Overw... more >>eight Collapse << | Not Applicable | Unknown | April 2017 | Mexico ... more >> Centro de Promoción de Salud Nutricional (CPSN) Hermosillo, Sonora, Mexico, 83000 Centro de Salud Urbano Dr. Domingo Olivares Hermosillo, Sonora, Mexico, 83000 Hospital General del Estado de Sonora Hermosillo, Sonora, Mexico, 83000 Hospital Ignacio Chávez Hermosillo, Sonora, Mexico, 83000 Centro Avanzado de Atención a la Salud (CAAPS) Hermosillo, Sonora, Mexico, 83180 Collapse << |
| NCT03247218 | Sickle Cell Disease | Phase 2 | Not yet recruiting | June 30, 2020 | - |
| NCT03649828 | Metabolic Syndrome ... more >> Cardiovascular Diseases Hypertension Collapse << | Not Applicable | Completed | - | - |
| NCT00271596 | Huntington Disease ... more >> Chorea Executive Dysfunction Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Mayo Clinic Arizona Scottsdale, Arizona, United States, 85259 United States, Iowa The University of Iowa Iowa City, Iowa, United States, 52242 United States, New York University of Rochester Rochester, New York, United States, 14618 Collapse << |
| NCT01429610 | Precursor Cell Lymphoblastic L... more >>eukemia-Lymphoma Collapse << | Phase 2 | Active, not recruiting | January 1, 2019 | Korea, Republic of ... more >> Chonnam National University Hwasun Hospital Hwasun, Chollanamdo, Korea, Republic of Hematologic Oncology Clinic, National Cancer Center Ilsan, Kongki, Korea, Republic of Division of Hematology-Oncology, Dong-A University College of Medicine Busan, Korea, Republic of Dong-A University College of Medicine Busan, Korea, Republic of Inje University Busan Paik Hospital Busan, Korea, Republic of Inje University Haeundae-Paik Hospital Busan, Korea, Republic of Kosin University College of Medicine, Kosin University Gospel Hospital Busan, Korea, Republic of Catholic University of Daegu School of Medicine Daegu, Korea, Republic of Keimyung University Dongsan Medical Center Daegu, Korea, Republic of Kyungpook National Unviersity Hospital Daegu, Korea, Republic of Yeungnam University College of Medicine Daegu, Korea, Republic of Chungnam National University Hospital Daejeon, Korea, Republic of Asan Medical Center, University of Ulsan College of Medicine Seoul, Korea, Republic of Chung-Ang University Hospital Seoul, Korea, Republic of Ewha Womans University Mokdong Hospital Seoul, Korea, Republic of Korea University Anam Hospital Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Soonchunhyang University Hospital Seoul, Korea, Republic of Ulsan University Hospital, University of Ulsan College of Medicine Ulsan, Korea, Republic of Collapse << |
| NCT01042496 | Bipolar Depression | Phase 3 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00080236 | Liver Transplantation ... more >> Hepatitis Cholestasis Carcinoma, Hepatocellular Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Mayo Clinic Scottsdale Phoenix, Arizona, United States, 85054 United States, California University of California Los Angeles Los Angeles, California, United States, 90095 University of California San Francisco San Francisco, California, United States, 94143 United States, Indiana Indiana University Medical Center Indianapolis, Indiana, United States, 46202 United States, Louisiana Tulane University Hospital and Clinic New Orleans, Louisiana, United States, 70112 United States, Minnesota Mayo Clinic Rochester Rochester, Minnesota, United States, 55905 United States, New York Mount Sinai School of Medicine New York City, New York, United States, 10029 United States, Ohio University of Cincinnati Cincinnati, Ohio, United States, 45267 United States, Texas Baylor Regional Transplant Institute, Baylor University Medical Center Dallas, Texas, United States, 75246 The University of Texas Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Germany Oberarzt der Klinik für Allgemein-, Viszeral- und Transplantationschirurgie Charité-Virchow Berlin, Germany, D-13353 Klinik für Viszeral- und Transplantationschirurgie Medizinische Hochschule Hannover Hannover, Germany, D-30623 Abteilung für Transplantationschirurgie Johannes Gutenberg-Universität Mainz Mainz, Germany, 55101 Collapse << |
| NCT01042496 | - | - | Completed | - | - |
| NCT02949375 | Chronic Liver Disease | Phase 2 | Recruiting | December 2018 | South Africa ... more >> Steve Biko Academic Hospital Recruiting Pretoria, Gauteng, South Africa, 0001 Contact: Mpho Klass Kgomo, MBBCH +27 (12) 322 7544 kgomomk@worldonline.co.za Collapse << |
| NCT02653651 | Postoperative Pain | Phase 4 | Completed | - | Tunisia ... more >> Ali JENDOUBI Tunis, Tunisia, 1006 Collapse << |
| NCT00271596 | - | - | Completed | - | - |
| NCT01608737 | Hepatitis C, Chronic | Phase 3 | Withdrawn | - | - |
| NCT02613195 | Evidence of Liver Transplantat... more >>ion Collapse << | Phase 3 | Unknown | February 2017 | China, Shanghai ... more >> Ren Ji Hospital Not yet recruiting Shanghai, Shanghai, China, 200127 Contact: Xia Qiang, Investigator +86-21-68383775 xiaqiang@shsmu.edu.cn Contact: Xue Feng, Investigator +8613761267573 fengxue6879@163.com Principal Investigator: Xia Qiang, Investigator Principal Investigator: Xue Feng, Investigator Sub-Investigator: Shi Shaojun China Deparment of Liver Surgery, Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University Shanghai, China, 200127 Collapse << |
| NCT02808013 | Lower Urinary Tract Symptoms | Phase 2 | Unknown | - | - |
| NCT03010176 | Solid Tumors ... more >>Lymphoma Collapse << | Phase 1 | Recruiting | October 22, 2021 | United States, California ... more >> UCLA Medical Center ( Site 0005) Recruiting Santa Monica, California, United States, 90404 Contact: Study Coordinator 310-582-4048 United States, New York Mount Sinai Hospital ( Site 0002) Recruiting New York, New York, United States, 10029 Contact: Study Coordinator 646-543-2859 Columbia University ( Site 0003) Recruiting New York, New York, United States, 10032 Contact: Study Coordinator 646-317-3141 United States, Texas Mary Crowley Cancer Research Center. ( Site 0001) Recruiting Dallas, Texas, United States, 75230 Contact: Study Coordinator 214-658-1945 France Institut Gustave Roussy ( Site 0049) Recruiting Villejuif, France, 94805 Contact: Study Coordinator +33142114211 Israel Sheba Medical Center ( Site 0040) Recruiting Ramat Gan, Israel, 5265601 Contact: Study Coordinator +97235302542 United Kingdom The Royal Marsden Foundation Trust ( Site 0031) Recruiting London, United Kingdom, SW3 6JJ Contact: Study Coordinator +442078118311 Collapse << |
| NCT02802228 | Portal Hypertension ... more >> Liver Cirrhosis Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Tampa General Medical Group Tampa, Florida, United States, 33606 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Ohio The Ohio State University Wexner Medical Center Columbus, Ohio, United States, 43210 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29425 United States, Texas Baylor University Medical Center Dallas, Texas, United States, 75246 United States, Virginia Virginia Commonwealth University Medical Center Richmond, Virginia, United States, 23114 Collapse << |
| NCT00529100 | Non-Small Cell Lung Cancer | Phase 1 Phase 2 | Completed | - | Canada, Alberta ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Edmonton, Alberta, Canada, T6G 1Z2 Canada, Ontario For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamilton, Ontario, Canada, L8V 5C2 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ottawa, Ontario, Canada, K1H 8L6 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00529100 | - | - | Completed | - | - |
| NCT00235716 | Alzheimer's Disease | Phase 3 | Completed | - | United States, Florida ... more >> VA Medical Center, Bay Pines Bay Pines, Florida, United States, 33708 VA Medical Center, Miami Miami, Florida, United States, 33125 United States, Iowa VA Medical Center, Iowa City Iowa City, Iowa, United States, 52246-2208 United States, Maryland VA Maryland Health Care System, Baltimore Baltimore, Maryland, United States, 21201 United States, Massachusetts VA Medical Center, Jamaica Plain Campus Boston, Massachusetts, United States, 02130 United States, Michigan VA Ann Arbor Healthcare System Ann Arbor, Michigan, United States, 48113 United States, Minnesota VA Medical Center, Minneapolis Minneapolis, Minnesota, United States, 55417 United States, North Carolina Salisbury VAMC Salisbury, North Carolina, United States, 28144 United States, Ohio VA Medical Center, Cleveland Cleveland, Ohio, United States, 44106 United States, South Carolina Ralph H Johnson VA Medical Center, Charleston Charleston, South Carolina, United States, 29401-5799 United States, Texas VA North Texas Health Care System, Dallas Dallas, Texas, United States, 75216 United States, Washington VA Puget Sound Health Care System, Seattle Seattle, Washington, United States, 98108 United States, Wisconsin Wlliam S. Middleton Memorial Veterans Hospital, Madison Madison, Wisconsin, United States, 53705 Puerto Rico VA Medical Center, San Juan San Juan, Puerto Rico, 00921 Collapse << |
| NCT01626924 | Perinatal Asphyxia | Phase 2 | Terminated(Recruitment too slo... more >>w and site decided to use hypothermia (exclusion criteria)) Collapse << | - | Turkey ... more >> T.R. Ministry of Health Izmir Tepecik Training and Research Hospital Izmir, Turkey, 35540 Yıl University Medical Faculty Hospital Van, Turkey, 65080 Collapse << |
| NCT02404311 | HIV Infection | Phase 1 Phase 2 | Unknown | January 2017 | South Africa ... more >> CAPRISA eThekwini CRS Durban, KwaZulu-Natal, South Africa, 4001 Isipingo CRS Durban, KwaZulu-Natal, South Africa, 4133 Emavundleni Desmond Tutu HIV Centre CRS Cape Town, Western Cape, South Africa, 7750 Aurum Institute for Health Research Klerksdorp, South Africa, 2570 Perinatal HIV Research Unit Soweto, South Africa, 2013 Collapse << |
| NCT03218657 | Aplastic Anemia | Not Applicable | Recruiting | July 1, 2022 | China, Zhejiang ... more >> Zhejiang Province Traditional Chinese Medical Hospital Recruiting Hangzhou, Zhejiang, China, 310006 Contact: Lin s Yun, master 13588887285 lsyww2003@163.com Collapse << |
| NCT00235716 | - | - | Completed | - | - |
| NCT01330316 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00573300 | - | - | Completed | - | United States, Massachusetts ... more >> Freedom Trail Clinic Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01330316 | - | - | Completed | - | - |
| NCT02036944 | - | - | Terminated(Study stopped d/t l... more >>ack of enrollment and no compelling data.) Collapse << | - | United States, Washington ... more >> Providence Medical Research Center Spokane, Washington, United States, 99204 Collapse << |
| NCT01838759 | - | - | Completed | - | Turkey ... more >> Bulent Ecevit University Hospital Zonguldak, Turkey, 67300 Collapse << |
| NCT01304147 | - | - | Completed | - | - |
| NCT02922751 | - | - | Enrolling by invitation | September 2020 | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 University of California at San Francisco (UCSF) San Francisco, California, United States, 94143 United States, Colorado Children's Hospital Colorado Aurora, Colorado, United States, 80045 United States, Georgia Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 United States, Illinois Ann & Robert H. Lurie Children's Hospital of Chicago Chicago, Illinois, United States, 60611 United States, Indiana Riley Hospital for Children Indianapolis, Indiana, United States, 46202 United States, Ohio Cincinnati Children's Memorial Hospital Cincinnati, Ohio, United States, 60190 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital at Pittsburgh Pittsburgh, Pennsylvania, United States, 15213 United States, Texas Texas Children's Hospital Houston, Texas, United States, 77030 United States, Utah University of Utah Salt Lake City, Utah, United States, 84113 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Canada, Ontario Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT01304147 | Major Depressive Disorder | Not Applicable | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York City, New York, United States, 10029 Collapse << |
| NCT00578786 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 3 | Completed | - | Argentina ... more >> Sanatorio Otamendi Ciudad Autonoma de Buenos Aires, Buenos Aires, Argentina, C1115AAB Hospital Britanico-Buenos Aires Ciudad Autonoma de Buenos Aires, Buenos Aires, Argentina, C1280AEB Instituto del Corazon Denton A. Cooley Ciudad Autonoma de Buenos Aires, Buenos Aires, Argentina, C1416A UAI Hosp. Universitario Ciudad Autonoma de Buenos Aires, Buenos Aires, Argentina, C1437BZL HIGA Hospital Interzonal General de Agudos Oscar Allende Mar del Plata, Buenos Aires, Argentina, 07600 Clinica Independencia Munro Munro, Buenos Aires, Argentina, 01605 Hospital Italiano de Rosario Rosario, Sante Fe, Argentina, 02000 Sanatorio Allende Cordoba, Argentina, X5000JHQ Hospital Italiano de Cordoba Cordoba, Argentina, X5004 FJE Hospital Privado Centro Medico de Cordoba Cordoba, Argentina, X5016KEH Instituto de Cardiologia J.F. Cabral Corrientes, Argentina, W3400AMZ Brazil Hospital Universitario Clementino Fraga Filho Ilha do Fundao, Rio de Janeiro, Brazil, 21941-590 Hospital Madre Teresa Belo Horizonte, Brazil, 30380-090 Hospital San Lucas de Pontificia Universidade Catolica Porto Alegre, Brazil, 90610-000 Complexo Hospitalar Sanata Casa de Porto Alegre Porto Alegre, Brazil, 92020-090 Universidade do Estado de Sao Paulo - UNIFESP Sao Paulo, Brazil, 04023-062 Hospital das Clinicas da FMUSP Sao Paulo, Brazil, 05403-000 Chile Hospital San Juan de Dios Santiago, Santiago de Chile, Chile, CP 8330024 Hospital Clinico Universidad Catolica Santiago, Santiago de Chile, Chile, CP 8350488 Instituto Nacional del Torax Santiago, Santiago de Chile, Chile, CP7500691 Mexico Instituto Nacional de Cardiologia Ignacio Chavez Mexico, DF, DF, Mexico, 14080 Unidad De Investigacion Clinica en Medicina Monterrey, Nuevo Leon, Nuevo Leon, Mexico, 64718 Collapse << |
| NCT01346540 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Italy ... more >> 1199.82.39004 Boehringer Ingelheim Investigational Site Milano, Italy Netherlands 1199.82.3102 Boehringer Ingelheim Investigational Site Maastricht, Netherlands Spain 1199.82.3401 Boehringer Ingelheim Investigational Site Barcelona, Spain 1199.82.3406 Boehringer Ingelheim Investigational Site Madrid, Spain 1199.82.3410 Boehringer Ingelheim Investigational Site Málaga, Spain United Kingdom 1199.82.4401 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.82.4402 Boehringer Ingelheim Investigational Site Manchester, United Kingdom Collapse << |
| NCT02857855 | Infection | Not Applicable | Enrolling by invitation | December 2025 | Poland ... more >> Department of General, Transplant and Liver Surgery, Medical University of Warsaw Warsaw, Mazowieckie, Poland, 02-097 Collapse << |
| NCT01346540 | - | - | Completed | - | - |
| NCT01523782 | Acute Lymphoblastic Leukemia | Phase 2 | Completed | - | - |
| NCT01399619 | - | - | Completed | - | - |
| NCT00578786 | - | - | Completed | - | - |
| NCT03025880 | Advanced Breast Cancer | Phase 2 | Recruiting | July 2020 | Spain ... more >> Institut Català d'Oncologia. Hospital Universitario Germans Trias i Pujol Recruiting Badalona, Barcelona, Spain, 08916 Hospital Universitario de Canarias Recruiting La Laguna, Canarias, Spain, 38320 Hospital Universitario Virgen de la Arrixaca Recruiting El Palmar, Murcia, Spain, 30120 Clínica Universitaria de Navarra Recruiting Pamplona, Navarra, Spain, 31008 Centro Oncológico de Galicia Recruiting A Coruña, Spain, 15009 Complejo Hospitalario Universitario Reina Sofía Recruiting Cordoba, Spain, 14004 Hospital Universitario Ramón y Cajal Recruiting Madrid, Spain, 28034 Hospital Clínico Universitario San Carlos Recruiting Madrid, Spain, 28040 Hospital Universitario Virgen de la Macarena Recruiting Sevilla, Spain, 41009 Hospital Clínico Universitario de Zaragoza "Lozano Blesa" Recruiting Zaragoza, Spain, 50009 Collapse << |
| NCT01523782 | - | - | Completed | - | - |
| NCT01399619 | Hepatitis C, Chronic | Phase 3 | Completed | - | - |
| NCT01322906 | Liver Failure | Phase 2 | Unknown | July 2012 | - |
| NCT00805389 | Hepatitis B | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Bruxelles, Belgium, 1200 GSK Investigational Site Gent, Belgium, 9000 GSK Investigational Site La Louvière, Belgium, 7100 GSK Investigational Site Wilrijk, Belgium, 2610 Germany GSK Investigational Site Tuebingen, Baden-Wuerttemberg, Germany, 72074 GSK Investigational Site Haag, Bayern, Germany, 83527 GSK Investigational Site Muenchen, Bayern, Germany, 80636 GSK Investigational Site Muenchen, Bayern, Germany, 81241 GSK Investigational Site Regensburg, Bayern, Germany, 93053 GSK Investigational Site Wuerzburg, Bayern, Germany, 97070 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Berlin, Germany, 12627 GSK Investigational Site Berlin, Germany, 13125 GSK Investigational Site Hamburg, Germany, 20253 Collapse << |
| NCT02187757 | Hyperlipidemia | Phase 3 | Completed | - | India ... more >> Division of Clinical & Preventive Cardiology, Heart Institute Gurgaon, Delhi, India, 122001 Life Care Institute of Medical Science & Research Ahmedabad, Gujarat, India, 380014 Bhatia Hospital Medical Research Society Mumbai, Maharashtra, India, 400007 Dr. Vikas Govind Pai Clinical Research Foundation Pune, Maharashtra, India, 411005 Fortis Escorts Hospital Jaipur, Rajasthan, India, 302017 Pace Clinical Research Center Bangalore, Tamil Nadu, India, 560043 Singvi Health Centre Chennai, Tamil Nadu, India, 600079 Rangammal Hospital Tiruvannamalai, Tamil Nadu, India, 606003 Collapse << |
| NCT03313713 | Atherosclerosis | Not Applicable | Enrolling by invitation | March 2018 | Italy ... more >> S. Orsola-Malpighi University Hospital Bologna, BO, Italy, 40138 Collapse << |
| NCT00151216 | Batten Disease ... more >> Late Infantile Neuronal Ceroid Lipofuscinosis Collapse << | Phase 1 | Active, not recruiting | June 2019 | United States, New York ... more >> New York Presbyterian Hospital - Weill Medical College of Cornell University New York, New York, United States, 10021 Collapse << |
| NCT00805389 | - | - | Completed | - | - |
| NCT01523808 | Pancreatic Cancer | Phase 1 | Completed | - | - |
| NCT02771249 | Healthy Volunteers | Phase 1 | Recruiting | December 31, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT02200029 | Intrahepatic Cholestasis Assoc... more >>iated With Alcoholic Liver Disease Collapse << | Phase 3 | Completed | - | Russian Federation ... more >> Research facility ORG-000962 Moscow, Russian Federation, 107014 Research facility ORG-000957 Moscow, Russian Federation, 117292 Research facility ORG-000961 Moscow, Russian Federation, 119435 Research facility ID ORG-000960 Moscow, Russian Federation, 119992 Research facility ID ORG-000726 Moscow, Russian Federation, 125284 Research facility ORG-000967 St. Petersburg, Russian Federation, 117630 Research facility ORG-000966 St. Petersburg, Russian Federation, 191015 Research facility ORG-000968 St. Petersburg, Russian Federation, 195257 Research facility ORG-000965 St. Petersburg, Russian Federation, 197022 Research facility ORG-000970 St. Petersburg, Russian Federation, 198216 Research facility ORG-000958 Troitsk, Russian Federation, 142190 Research facility ORG-000969 Yaroslavl, Russian Federation, 150003 Collapse << |
| NCT01903460 | - | - | Completed | - | - |
| NCT01903460 | Alagille Syndrome | Phase 2 | Completed | - | United Kingdom ... more >> Birmingham Children's Hospital Birmingham, West Midlands, United Kingdom, B4 6NH Leeds Teaching Hospitals Leeds, West Yorkshire, United Kingdom, LS9 7TF Kings College Hospital London, United Kingdom, SE5 9RS Collapse << |
| NCT01963845 | - | - | Completed | - | - |
| NCT02150291 | Hepatitis C | Phase 4 | Unknown | November 2015 | Egypt ... more >> Ain Shams University Hospitals Recruiting Cairo, Abbasiya, Egypt, 11566. Contact: Osama A Ahmed 01006154809 dros1977@hotmail.com Principal Investigator: Nermeen N Ashoush Collapse << |
| NCT01963845 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 Collapse << |
| NCT02299739 | - | - | Completed | - | Korea, Republic of ... more >> Jaseng Hospital of Korean Medicine Seoul, Gangnam-Gu, Korea, Republic of, 135-896 Collapse << |
| NCT02650024 | Hepatitis C H... more >>uman Immunodeficiency Virus Collapse << | Phase 4 | Recruiting | July 2020 | United States, California ... more >> Ucsd Hnrp Recruiting San Diego, California, United States, 92103 Contact: Melanie Crescini, B.S. 619-543-5016 mscrescini@ucsd.edu Collapse << |
| NCT01432548 | Ischemic Lesions | Not Applicable | Unknown | October 2012 | Argentina ... more >> Hospital Dr Cosme Argerich Recruiting Buenos Aires, Argentina Contact: Javier Lendoire, MD, PhD 011-4121-0846 • jlendoire@yahoo.com.ar Collapse << |
| NCT01845870 | - | - | Completed | - | China, Tianjin ... more >> General Hospital of Tianjin Medical University Tianjin, Tianjin, China, 300052 Collapse << |
| NCT03056274 | Intrahepatic Cholestases ... more >> Pregnancy Complications Collapse << | Phase 2 Phase 3 | Not yet recruiting | September 2019 | - |
| NCT00941863 | Carcinoma | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Nashville, Tennessee, United States, 37232 United States, Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00795704 | Type 2 Diabetes | Phase 2 Phase 3 | Completed | - | United States, Mississippi ... more >> University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 Collapse << |
| NCT01043523 | - | - | Completed | - | - |
| NCT01043523 | - | - | Completed | - | United States, California ... more >> Palo Alto, California, United States, 94304 United States, New York New York, New York, United States, 10032 United States, North Carolina Durham, North Carolina, United States, 27710 United States, Ohio Cinncinati, Ohio, United States, 45229 United States, Pennsylvania Hershey, Pennsylvania, United States, 17033 Italy Many Locations, Italy Japan Many Locations, Japan Singapore Many Locations, Singapore Taiwan Many Locations, Taiwan Collapse << |
| NCT00085917 | Hepatitis C H... more >>IV Infections Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01465165 | - | - | Terminated(Study procedures we... more >>re not feasible.) Collapse << | - | United States, Utah ... more >> University of Utah Dept of Psychiatry Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02125045 | Health Subjects With Cardiovas... more >>cular Risk Factors Collapse << | Phase 3 | Completed | - | Italy ... more >> Oberdan Parodi, MD Milan, Italy, 20162 Collapse << |
| NCT03669835 | Hepatitis C | Not Applicable | Recruiting | November 1, 2020 | Kazakhstan ... more >> Asfendiyarov Kazakh National Medical University Recruiting Almaty, Kazakhstan, 050000 Contact: Abdugani Mussayev, MD, PhD +77772509406 musaev.dr@mail.ru Principal Investigator: Abdugani Mussayev, MD, PhD Sub-Investigator: Saniya Kozhakhmetova, PhD Sub-Investigator: Baurzhan Zhusupov, MPH Sub-Investigator: Ainur Yeshmanova, PhD Sub-Investigator: Yelena Slavko, PhD Sub-Investigator: Almira Satbayeva, PhD Sub-Investigator: Zarina Zhanen, MD Sub-Investigator: Zhasulan Mergenbayev, MD Sub-Investigator: Akbota Myrzhashova, MD Collapse << |
| NCT01163253 | Psoriasis | Phase 3 | Terminated | - | - |
| NCT00941863 | - | - | Completed | - | - |
| NCT03749070 | Non-Alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Not yet recruiting | June 30, 2021 | - |
| NCT00515463 | Low Bone Mineral Density ... more >> Osteopenia Osteoporosis Collapse << | Phase 3 | Completed | - | - |
| NCT02605616 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Non-alcoholic Steatohepatitis (NASH) Collapse << | Phase 2 | Recruiting | December 2019 | United States, Virginia ... more >> University of Virginia Recruiting Charlottesville, Virginia, United States, 22908 Contact: Pam Morris 434-924-5780 pm2t@hscmail.mcc.virginia.edu Collapse << |
| NCT00515463 | - | - | Completed | - | - |
| NCT00795704 | - | - | Completed | - | - |
| NCT03507699 | Colorectal Neoplasms Malignant... more >> Liver Metastases Collapse << | Phase 1 | Not yet recruiting | May 31, 2021 | Israel ... more >> Sheba Medical Center Ramat Gan, Israel, 52621 Collapse << |
| NCT02564679 | Morbid Obesity ... more >> NAFLD Collapse << | Not Applicable | Completed | - | - |
| NCT02423941 | Liver Transplantation ... more >> Reperfusion Delayed Graft Function Collapse << | Not Applicable | Unknown | May 2017 | Belarus ... more >> RSPC for organ and tissue transplantation, Minsk 9th clinic Recruiting Minsk, Belarus, 220116 Contact: Aliaksei E Shcherba, PhD +375293330689 aleina@tut.by Contact: Denis F Efimov +375(29)6884415 den.efimoff@gmail.com Principal Investigator: Aliaksei E Shcherba, PhD Sub-Investigator: Andrew F Minou Sub-Investigator: Evgeni O Santotski Sub-Investigator: Alexander M Dzyadzko, PhD Sub-Investigator: Denis J Efimov Sub-Investigator: Sergei V Korotkov, PhD Sub-Investigator: Oleg O Rummo, MD PhD Collapse << |
| NCT00085917 | - | - | Completed | - | - |
| NCT00095498 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT01828528 | - | - | Completed | - | Israel ... more >> Liver Disease Center, Sheba Medical Center, Tel-Hashomer Ramat-Gan, Israel, 52621 Collapse << |
| NCT01163253 | - | - | Terminated | - | - |
| NCT02397239 | - | - | Recruiting | - | Taiwan ... more >> National Cheng Kung University Hospital Recruiting Tainan, Taiwan, 704 Contact: Szu-Chun Yang, M.D. +886 972402279 yangszuchun@gmail.com Collapse << |
| NCT02034253 | - | - | Recruiting | November 2018 | United States, Alabama ... more >> Sparks Center Recruiting Birmingham, Alabama, United States, 35294 Contact: David White, MPH, MPA 205-996-9813 dw2777@uab.edu Contact: Ariel Kidwell, BS 205-996-9813 akidwell@uab.edu Principal Investigator: Adrienne C Lahti, MD Collapse << |
| NCT03452540 | Severe Acute Decompensated Alc... more >>oholic Hepatitis Collapse << | Phase 2 | Not yet recruiting | February 2020 | - |
| NCT01855048 | Healthy Volunteers ... more >> Anti-Bacterial Agents Methicillin-Resistant Staphylococcus Aureus Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00344682 | Depressive Disorder | Phase 4 | Completed | - | United States, Massachusetts ... more >> Center for Psychopharmacologic Research and Treatment (University of Massachusetts Medical School) Worcester, Massachusetts, United States, 01605 Collapse << |
| NCT01250743 | Hepatitis C | Not Applicable | Terminated | June 2012 | United States, Kansas ... more >> University of Kansas Medical Center, Department of Integrative Medicine Kansas City, Kansas, United States, 66160 Collapse << |
| NCT00344682 | - | - | Completed | - | - |
| NCT00015873 | Leukemia | Phase 3 | Completed | - | - |
| NCT02491684 | Asthma | Phase 2 | Completed | - | Argentina ... more >> Research Site Buenos Aires, Argentina Research Site C.a.b.a., Argentina Research Site Ciudad Autonomade Buenos Aires, Argentina Research Site Quilmes, Argentina Australia Research Site Bedford Park, Australia Research Site New Lambton, Australia Research Site Westmead, Australia Research Site Woolloongabba, Australia Colombia Research Site Bogota, Colombia Research Site Bogotá, Colombia Research Site Floridablanca, Colombia France Research Site Brest Cedex, France Research Site Dijon, France Research Site Lyon Cedex 04, France Research Site Marseille, France Research Site Montpellier Cedex 5, France Research Site Paris Cedex 18, France Research Site Pessac, France Korea, Republic of Research Site Bucheon-si, Korea, Republic of Research Site Jeonju-si, Korea, Republic of Research Site Seoul, Korea, Republic of Spain Research Site Barcelona, Spain Research Site Marbella (Málaga), Spain Research Site Málaga, Spain Research Site Sevilla, Spain Research Site Valencia, Spain United Kingdom Research Site Blackpool, United Kingdom Research Site Bradford, United Kingdom Research Site Lancaster, United Kingdom Research Site Leeds, United Kingdom Research Site Manchester, United Kingdom Research Site Nottingham, United Kingdom Research Site Southampton, United Kingdom Collapse << |
| NCT01482065 | - | - | Completed | - | - |
| NCT02829268 | Wolfram Syndrome ... more >> Diabetes Mellitus Optic Nerve Atrophy Ataxia Collapse << | Phase 1 Phase 2 | Recruiting | March 2021 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Ashley Simpson, RN, BSN, CPN 314-286-1550 ashley.simpson@wustl.edu Contact: Fumihiko Urano, MD 314-362-8683 urano@wustl.edu Principal Investigator: Fumihiko Urano, MD Collapse << |
| NCT00095498 | - | - | Completed | - | - |
| NCT01482065 | Non Alcoholic Fatty Liver Dise... more >>ase Obstructive Sleep Apnea Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21224 Collapse << |
| NCT01457755 | - | - | Completed | - | - |
| NCT03608748 | - | - | Not yet recruiting | May 1, 2019 | Israel ... more >> Haemek medical center Afula, Israel, 1834111 Collapse << |
| NCT01457768 | - | - | Completed | - | - |
| NCT01137591 | Liver Failure ... more >> Liver Failure, Acute Drug Induced Liver Injury Prevention & Control Fever Collapse << | Not Applicable | Withdrawn(Unable to recruit su... more >>fficient participants due to lack of funding; PI has left the institution.) Collapse << | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT02179918 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> Research Administration Office Los Angeles, California, United States, 90095 Ronald Reagan UCLA Medical Center, Drug Information Center Los Angeles, California, United States, 90095 UCLA Hematology-Oncology Clinic Los Angeles, California, United States, 90095 UCLA Oncology Center Los Angeles, California, United States, 90095 United States, Connecticut Smilow Cancer Center at Yale-New Haven Hospital New Haven, Connecticut, United States, 06510 Smilow Cancer Hospital at Yale-New Haven New Haven, Connecticut, United States, 06510 United States, Texas South Texas Accelerated Research Therapeutics, LLC San Antonio, Texas, United States, 78229 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT01191177 | Liver Disease ... more >> Short Bowel Syndrome Collapse << | Phase 1 Phase 2 | Withdrawn(no subjects recruite... more >>d) Collapse << | January 2012 | United States, Massachusetts ... more >> Children's Hospital Boston Boston, Massachusetts, United States, 02115 Collapse << |
| NCT02179918 | - | - | Completed | - | - |
| NCT01446055 | Critical Limb Ischemia ... more >> Peripheral Arterial Disease Buerger's Disease Collapse << | Phase 1 Phase 2 | Unknown | April 2013 | China, Beijing ... more >> Xuan Wu Hospital Not yet recruiting Beijing, Beijing, China Contact: Yongquan Gu, MD 13910002909 gu-yq@263.net Principal Investigator: Yongquan Gu, MD Collapse << |
| NCT02300298 | - | - | Completed | - | - |
| NCT01846117 | Oxidative Stress ... more >> Inflammation Aging Pain Collapse << | Phase 1 Phase 2 | Completed | - | Canada, Saskatchewan ... more >> Saskatoon Centre for Patient-Oriented Research Saskatoon, Saskatchewan, Canada, S7K 0M7 Collapse << |
| NCT01785212 | Liver Neoplasms ... more >> Liver Metastasis Liver Hemangioma Echinococcosis, Hepatic Collapse << | Not Applicable | Completed | - | Austria ... more >> Dept. of Surgery/Div. of General Surgery Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT02300298 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Japan ... more >> 1199.90.81001 Boehringer Ingelheim Investigational Site Chiba , Kashiwa, Japan 1199.90.81003 Boehringer Ingelheim Investigational Site Kanagawa, Yokohama, Japan 1199.90.81007 Boehringer Ingelheim Investigational Site Osaka, Osakasayama, Japan 1199.90.81006 Boehringer Ingelheim Investigational Site Osaka, Osaka, Japan 1199.90.81004 Boehringer Ingelheim Investigational Site Shizuoka, Sunto-gun, Japan 1199.90.81002 Boehringer Ingelheim Investigational Site Tokyo, Chuo, Japan Collapse << |
| NCT00323414 | Fatty Liver | Phase 2 | Completed | - | United States, Ohio ... more >> MetroHealth Medical Center Cleveland, Ohio, United States, 44109 Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Collapse << |
| NCT03089697 | Staphylococcus Aureus Bacterem... more >>ia Anti-Bacterial Agents Collapse << | Phase 2 | Recruiting | April 2020 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam-si, Gyeonggi-do, Korea, Republic of, 13620 Contact: Hong Bin Kim, M.D, PhD +82-31-787-7021 hbkimmd@snu.ac.kr Seoul National University Hospital Recruiting Seoul, Korea, Republic of, 03080 Contact: Wan Beom Park, M.D, PhD +82-2-2072-2114 wbpark1@snu.ac.kr Collapse << |
| NCT02891577 | - | - | Unknown | September 2017 | France ... more >> Centre Hospitalier Universitaire de Besançon Recruiting Besançon, France, 25030 Contact: Gaël PITON, MD, PhD 0033381668224 gpiton@chu-besancon.fr Collapse << |
| NCT01339078 | Biliary Strictures Post Liver ... more >>Transplantation. Collapse << | Not Applicable | Unknown | December 2015 | Belgium ... more >> University Hospital, Ghent Recruiting Ghent, Belgium Contact: Hans Van Vlierberghe, Ph.D. , M.D. Hans.Vanvlierberghe@ugent.be Principal Investigator: Hans Van Vlierberghe, Ph.D. , M.D. Collapse << |
| NCT00323414 | - | - | Completed | - | - |
| NCT02579265 | Hospitalized Neonates and Infa... more >>nts, Expected to Require Parenteral Nutrition for 28 Days Collapse << | Phase 3 | Recruiting | October 2019 | United States, California ... more >> Lucile Packard Children's Hospital Recruiting Palo Alto, California, United States, 94304 Contact: Katherine McCallie, MD UC San Diego Medical Center Recruiting San Diego, California, United States, 92103 Contact: Jae H Kim, MD, PhD Rady Children's Hospital San Diego Recruiting San Diego, California, United States, 92123 Contact: Brian Lane, MD United States, Connecticut Yale - New Haven Children's Hospital Recruiting New Haven, Connecticut, United States, 06510 Contact: Orly Levit, MD United States, Illinois Ann & Robert H. Lurie Children's Hospital of Chicago Recruiting Chicago, Illinois, United States, 60611-2991 Contact: Daniel T Robinson, MD United States, Massachusetts Boston Children's Hospital Withdrawn Boston, Massachusetts, United States, 02115 United States, Nebraska University of Nebraska Medical Center Recruiting Omaha, Nebraska, United States, 68198-1205 Contact: Ann Anderson Berry, MD, PhD United States, New York Mount Sinai Hospital Recruiting New York, New York, United States, 10029 Contact: Joanne Lai, MD Steven & Alexandra Cohen Children's Medical Center of NY Active, not recruiting New York, New York, United States, 11040 United States, Oklahoma The Children's Hospital at OU Medical Center Recruiting Oklahoma City, Oklahoma, United States, 73104 Contact: Kimberly Ernst, MD United States, Pennsylvania Children's Hospital of Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Maria R Mascarenhas, MBBS Children's Hospital of Pittsburgh of UPMC Recruiting Pittsburgh, Pennsylvania, United States, 15224 Contact: Jeffrey A Rudolph, MD United States, Tennessee Vanderbilt Children's Hospital Recruiting Nashville, Tennessee, United States, 37345 Contact: William Walsh, MD United States, Texas Texas Children's Hospital Recruiting Houston, Texas, United States, 77030 Contact: Amy B Hair, MD Collapse << |
| NCT01525225 | Type 2 Diabetes Mellitus | Phase 1 | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | United States, Arkansas ... more >> Osborne Research Center Little Rock, Arkansas, United States, 72201 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 United States, Kentucky Kosair Charities Pediatric Clinical Research Unit Louisville, Kentucky, United States, 40202 United States, Missouri The Children'S Mercy Hospital And Clinics Kansas City, Missouri, United States, 64108 United States, Ohio Promedica Toledo Children'S Hospital Toledo, Ohio, United States, 43606 Promedica Toledo Childrens Hospital Toledo, Ohio, United States, 43606 United States, Pennsylvania Childrens Hospital Of Pittsburgh Of Upmc Pittsburgh, Pennsylvania, United States, 15224 Collapse << |
| NCT02534961 | Hepatocellular Carcinoma | Phase 4 | Unknown | December 2017 | Taiwan ... more >> Division of Gastroenterology, Department of Medicine, Taipei Veterans General Hospital Recruiting Taipei, Taiwan, 112 Contact: Chien-Wei Su, MD & PhD 886228712121 ext 3352 cwsu2@vghtpe.gov.tw Collapse << |
| NCT01525225 | - | - | Terminated(Release of Post Mar... more >>keting Requirement for this study. Terminated November 2013.) Collapse << | - | - |
| NCT02427087 | Fatty Liver | Not Applicable | Completed | - | Brazil ... more >> University of Sao Paulo School of Medicine Sao Paulo, Brazil, 01246903 Collapse << |
| NCT01115582 | - | - | Completed | - | - |
| NCT02065245 | Aging Frailty | Phase 1 Phase 2 | Active, not recruiting | December 2020 | United States, Florida ... more >> ISCI/University of Miami Miller School of Medicine Miami, Florida, United States, 33136 Collapse << |
| NCT02830841 | Hepatic Ischemic and Reperfusi... more >>on Injury Collapse << | Not Applicable | Unknown | December 2017 | China, Shanghai ... more >> Renji Hospital Recruiting Shanghai, Shanghai, China, 200127 Contact: Weifeng Yu, MD,PhD 862168383702 yuweifeng@renji.com Principal Investigator: Liqun Yang, MD,PhD Principal Investigator: Peiying Li, MD,PhD Collapse << |
| NCT02965911 | Primary Biliary Cirrhosis | Phase 1 Phase 2 | Recruiting | January 2019 | China ... more >> Beijing 302 hospital Recruiting Beijing, China, 100039 Contact: Zhengsheng Zou, Dr. Principal Investigator: Zhengsheng Zou, Dr. Collapse << |
| NCT01115582 | Inborn Errors of Bile Acid Syn... more >>thesis Collapse << | Phase 3 | Completed | - | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 Collapse << |
| NCT02720094 | HIV Infections | Phase 2 Phase 3 | Recruiting | March 30, 2022 | - |
| NCT01845727 | Cutaneous Leishmaniasis | Phase 1 Phase 2 | Completed | - | Colombia ... more >> Programa de Estudios y Control de Enfermedades Tropicales (PECET), Universidad de Antioquia Medellin, Colombia Collapse << |
| NCT03660748 | Obesity | Early Phase 1 | Recruiting | September 1, 2019 | Canada, Ontario ... more >> GI Labs Recruiting Toronto, Ontario, Canada, M5C2N8 Contact: Adish Ezatagha, MSc 416 861-9782 ext 208 aezatagha@gilabs.com Contact: Alexandra L Jenkins, PhD / RN 416 861-9782 ext 200 alexandrajenkins@gilabs.com Collapse << |
| NCT01599676 | Elderly Patients | Not Applicable | Completed | - | France ... more >> HOPITAL BICHAT Claude Bernard Paris, France, 75018 Collapse << |
| NCT01668017 | Advanced Solid Tumors ... more >> Hepatocellular Carcinoma Collapse << | Phase 1 | Terminated(The sponsor decided... more >> not to conduct the expansion part of trial (part 2)) Collapse << | - | Japan ... more >> Please contact Merck Serono Co., Ltd for recruiting locations in, Japan Collapse << |
| NCT01549990 | - | - | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of, 120-752 Collapse << |
| NCT02184858 | Primary Hypertension ... more >> Secondary Hypertension Collapse << | Phase 4 | Completed | - | Belgium ... more >> Department of Pediatrics and Medical Genetics Ghent, Belgium, 9000 Collapse << |
| NCT01668017 | - | - | Terminated(The sponsor decided... more >> not to conduct the expansion part of trial (part 2)) Collapse << | - | - |
| NCT01899859 | Non-Alcoholic Steatohepatitis ... more >>(NASH) Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Brooke Army Medical Ctr. San Antonio, Texas, United States, 78234 Collapse << |
| NCT03586674 | Chronic Cholestasis | Phase 2 | Completed | - | Egypt ... more >> Dr. Yassin Abdel Ghaffar Charity Center For Liver Diseases & Researches Cairo, Nasr City, Egypt National liver istitute Shibīn Al Kawm, Egypt Collapse << |
| NCT02466516 | Non-Alcoholic Steatohepatitis ... more >>(NASH) Collapse << | Phase 2 | Completed | - | - |
| NCT02106182 | Nocturia Beni... more >>gn Prostatic Hyperplasia Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Bucheon St.Mary's Hospital Bucheon, Gyeonggi-do, Korea, Republic of, 420-818 Ajou University Hospital Suwon, Gyeonggi-do, Korea, Republic of, 443-380 Pusan Natonal University Hospital Busan, Korea, Republic of, 602-739 Eulji University Hospital Daejeon, Korea, Republic of, 302-799 Hanyang University Hospital Seoul, Korea, Republic of, 133-792 Soon Chun Hyang University Hospital Seoul, Korea, Republic of, 140-887 Collapse << |
| NCT01579513 | Congenital Heart Disease ... more >> Disorder of Fetus or Newborn Collapse << | Not Applicable | Active, not recruiting | December 2018 | United States, Georgia ... more >> Children's Healthcare of Atlanta/Emory University Atlanta, Georgia, United States, 30233 United States, South Carolina Medical University of South Carolina, Pediatric Cardiology Charleston, South Carolina, United States, 29425 Collapse << |
| NCT00502593 | Influenza | Phase 2 | Completed | - | Spain ... more >> GSK Investigational Site Blanes (Girona), Spain, 17300 GSK Investigational Site L'Eliana, Valencia, Spain, 46183 GSK Investigational Site Paiporta, Valencia, Spain, 46200 GSK Investigational Site Quart De Poblet, Valencia, Spain, 46930 GSK Investigational Site Valencia, Spain, 46008 GSK Investigational Site Valencia, Spain, 46011 GSK Investigational Site Valencia, Spain, 46024 Collapse << |
| NCT03679013 | Pain, Postoperative | Phase 4 | Not yet recruiting | May 12, 2019 | - |
| NCT01826760 | - | - | Completed | - | China, Zhejiang ... more >> Wenzhou Medical College Wenzhou, Zhejiang, China, 325000 Collapse << |
| NCT02705807 | - | - | Completed | - | - |
| NCT00436826 | Multiple Sclerosis | Phase 2 | Completed | - | - |
| NCT00436826 | - | - | Completed | - | - |
| NCT00514215 | Kidney Cancer ... more >> Lung Cancer Metastatic Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201-1379 Collapse << |
| NCT00502593 | - | - | Completed | - | - |
| NCT00514215 | - | - | Completed | - | - |
| NCT02705807 | Cardiovascular Disease | Phase 4 | Completed | - | Japan ... more >> GSK Investigational Site Okayama, Japan, 701-1192 Collapse << |
| NCT01940393 | Urticaria | Phase 4 | Completed | - | Colombia ... more >> Medellin Medellin, Antioquia, Colombia Collapse << |
| NCT01207648 | - | - | Completed | - | - |
| NCT01207648 | - | - | Completed | - | United States, Alabama ... more >> Research Site Birmingham, Alabama, United States United States, California Research Site San Francisco, California, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, New York Research Site Buffalo, New York, United States Research Site Rochester, New York, United States Research Site Stoney Brook, New York, United States Argentina Research Site Buenos Aires, Argentina Canada Research Site Toronto, Canada France Research Site Le Kremlin Bicêtre Cedex, France Italy Research Site Bari, Italy Research Site Catania, Italy Research Site Gallarate, Italy Research Site Milan, Italy Research Site Rome, Italy Research Site Torino, Italy Russian Federation Research Site Moscow, Russian Federation Tunisia Research Site Tunis, Tunisia Venezuela Research Site Maracaibo, Venezuela Collapse << |
| NCT01712711 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Gastroenterology clinic, Sina Hospital Tehran, Iran, Islamic Republic of Collapse << |
| NCT01712711 | - | - | Completed | - | - |
| NCT01657773 | - | - | Completed | - | China, Zhejiang ... more >> Wenzhou Medical College Wenzhou, Zhejiang, China, 325000 Collapse << |
| NCT01800383 | - | - | Unknown | February 2018 | United States, Massachusetts ... more >> McLean Hospital Recruiting Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT03285217 | Sarcopenia Ci... more >>rrhosis Malnutrition Collapse << | Not Applicable | Recruiting | December 31, 2018 | Spain ... more >> Hospital Universitario Miguel Servet Recruiting Zaragoza, Spain, 50009 Contact: Jose M Arbones-Mainar, PhD +34 976 769 565 Collapse << |
| NCT01691794 | HIV, Pediatric | Phase 4 | Completed | - | United States, Arizona ... more >> Phoenix Children'S Hospital Phoenix, Arizona, United States, 85016 United States, Georgia Grady Health System Ponce Family And Youth Clinic Atlanta, Georgia, United States, 30322 United States, Texas Children'S Medical Center Of Dallas Dallas, Texas, United States, 75235 Argentina Local Institution Buenos Aires, Bs As, Buenos Aires, Argentina, 1141 Local Institution Buenos Aires, Argentina, 1181 Local Institution Buenos Aires, Argentina, 1425 Brazil Local Institution Salvador, Bahia, Brazil, 40110-160 Local Institution Salvador, Bahia, Brazil, 40110 Local Institution Porto Alegre, RIO Grande DO SUL, Brazil, 90035-903 Local Institution Porto Alegre, Rio Grande Do Sul, Brazil, 90035 Local Institution Sao Paulo, SP, Brazil, 01416-901 Local Institution Sao Paulo, Brazil, 01246 Chile Local Institution Santiago, Metropolitana, Chile, 8207257 Local Institution Santiago, Metropolitana, Chile, 8380418 Mexico Local Institution Guadalajara, Jalisco, Mexico, 44280 Local Institution Monterrey, Nuevo LEON, Mexico, 64000 Local Institution Merida, Yucatan, Mexico, 97000 Local Institution Puebla, Mexico, 72000 Peru Local Institution Lima, Peru, 1 Local Institution Lima, Peru, LIMA 10 Russian Federation Local Institution Moscow, Russian Federation, 129110 Local Institution St. Petersburg, Russian Federation, 190103 South Africa Local Institution Port Elizabeth, Eastern CAPE, South Africa, 6001 Local Institution Bloemfontein, FREE State, South Africa, 9301 Local Institution Soweto, Gauteng, South Africa, 2013 Local Institution Cape Town, Western CAPE, South Africa, 7505 Local Institution Cape Town, Western CAPE, South Africa, 7530 Collapse << |
| NCT02057692 | Alagille Syndrome | Phase 2 | Completed | - | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027 University of California at San Francisco San Francisco, California, United States, 94143 United States, Colorado Children's Hospital Colorado Aurora, Colorado, United States, 80045 United States, Georgia Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 United States, Illinois Ann & Robert H. Lurie Children's Hospital of Chicago Chicago, Illinois, United States, 60611 United States, Indiana Riley Hospital for Children Indianapolis, Indiana, United States, 46202 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania The Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children's Hospital of Pittsburgh of UPMC Pittsburgh, Pennsylvania, United States, 15224 United States, Texas Baylor College of Medicine/Texas Children's Hospital Houston, Texas, United States, 77030 United States, Utah University of Utah Salt Lake City, Utah, United States, 84113 United States, Washington Seattle Children's Hospital Seattle, Washington, United States, 98105 Canada, Ontario The Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT02057692 | - | - | Completed | - | - |
| NCT01400451 | Melanoma | Phase 1 | Terminated(More than 2 of 6 pa... more >>tients treated experienced dose limiting toxicities.) Collapse << | - | United States, California ... more >> University Of California Los Angeles Los Angeles, California, United States, 90095 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02215 Dana Farber Cancer Inst Boston, Massachusetts, United States, 02215 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT02978339 | Primary Sclerosing Cholangitis | Phase 1 Phase 2 | Active, not recruiting | November 2018 | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905-0001 Collapse << |
| NCT01370811 | Sialorrhoea | Phase 2 | Completed | - | United States, Michigan ... more >> QUEST Research Institute Bingham Farms, Michigan, United States, 48025 Collapse << |
| NCT01400451 | - | - | Terminated(More than 2 of 6 pa... more >>tients treated experienced dose limiting toxicities.) Collapse << | - | - |
| NCT00442572 | Hepatitis B, Chronic | Phase 2 | Completed | - | Bulgaria ... more >> Mhat St. Ivan Rilski; Clinic of Gastroenterology Sofia, Bulgaria, 1612 Collapse << |
| NCT01759628 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Gastroenterology clinic, Sina Hospital (Tehran) / Beheshti Hospital (Kashan) Tehran / Kashan, Iran, Islamic Republic of Collapse << |
| NCT00442572 | - | - | Completed | - | - |
| NCT02761395 | AL-Hijama in Thalassmia Major | Not Applicable | Unknown | May 2017 | Egypt ... more >> Faculty of Medicine- Tanta University Completed Tanta, Gharbia, Egypt, 0000 Faculty of Medicine- Tanta University Recruiting Tanta, Gharbia, Egypt, 0000 Contact: Mohamed R El-Shanshory, MD +201005680834 elshanshory@gmail.com Contact: Nahed M Hablas, MD +201010560350 nahedhablas79@gmail.com Principal Investigator: Mohamed M El-Shanshory, MD Principal Investigator: Nahed AR Hablas, MD Principal Investigator: Salah M El Sayed, MD Collapse << |
| NCT01691794 | - | - | Completed | - | - |
| NCT03664596 | Non-alcoholic Steatohepatitis | Not Applicable | Recruiting | June 2019 | Kazakhstan ... more >> Medical Centre Hospital of the President's Affairs Administration Recruiting Astana, Kazakhstan, 010000 Contact: Bakytzhan Bimbetov, MD, PhD +7701 383 6072 bimbetov2010@mail.ru Sub-Investigator: Abay Zhangabylov, MD, PhD Sub-Investigator: Marzhan Rakhimzhanova, MD, MPH Collapse << |
| NCT01808313 | Vascular Disease | Phase 3 | Completed | - | China, Heilongjiang ... more >> GSK Investigational Site Harbin, Heilongjiang, China, 150001 China, Hubei GSK Investigational Site Wuhan, Hubei, China, 430022 China, Hunan GSK Investigational Site Hunan, Hunan, China, 410008 China, Jilin GSK Investigational Site Changchun, Jilin, China, 130021 China, Shaanxi GSK Investigational Site Xian, Shaanxi, China, 710032 China, Shandong GSK Investigational Site Jinan, Shandong, China, 250012 China GSK Investigational Site Beijing, China, 100032 GSK Investigational Site Beijing, China, 100034 GSK Investigational Site Beijing, China, 100037 GSK Investigational Site Beijing, China, 100038 GSK Investigational Site Shanghai, China, 200001 GSK Investigational Site Shanghai, China, 200433 Collapse << |
| NCT01871805 | Non-Small Cell Lung Cancer | Phase 1 Phase 2 | Completed | - | - |
| NCT02741245 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01944618 | - | - | Terminated() | - | Australia, Victoria ... more >> Local Institution Clayton, Victoria, Australia, VIC 3168 Collapse << |
| NCT02741245 | - | - | Completed | - | - |
| NCT03099135 | Hepatitis C | Phase 3 | Terminated(Recruitment prematu... more >>rely stopped based on decision to stop HPC program (not due to safety or efficacy results) + On 31 Jan 2018 decision to stop the study) Collapse << | - | Canada, British Columbia ... more >> LAIR Centre Vancouver, British Columbia, Canada, V5Z 1H2 Vancouver Prostate Centre, Gordon and Leslie Diamond Health Care Centre Vancouver, British Columbia, Canada, V5Z 1M9 GI Research Institute (G.I.R.I.) Vancouver, British Columbia, Canada, V6Z 2K5 Vancouver ID Research and Care Centre Society Vancouver, British Columbia, Canada, V6Z2C7 PerCuro Clinical Research Ltd. Victoria, British Columbia, Canada, V8V 3P9 New Zealand Auckland District Health Board Auckland, New Zealand, 1142 Christchurch Clinical Studies Trust Christchurch, New Zealand, 8011 Poland Wojewodzki Szpital Specjalistyczny im. dr Wl. Bieganskiego Lodz, Poland, 91-347 Hepid Diagnostyka I Terapia Tomasiewicz, Kiciak - Lekarze Spółka Partnerska Lublin, Poland, 20-884 ID Clinic Mysłowice, Poland, 41-400 SP ZOZ Wojewodzki Szpital Zakazny Warszawa, Poland, 01-201 EMC Instytut Medyczny SA,Przychodnia przy Lowieckiej Wroclaw, Poland, 50-220 Singapore National University Hospital Singapore, Singapore, 119074 Singapore General Hospital Singapore, Singapore, 169856 Khootech Puat Hospital Singapore, Singapore, 768828 Collapse << |
| NCT01808313 | - | - | Completed | - | - |
| NCT01871805 | - | - | Completed | - | - |
| NCT01155466 | Parkinson Disease | Phase 3 | Completed | - | - |
| NCT00675428 | Multiple Myeloma | Phase 1 Phase 2 | Terminated(Sponsor decision du... more >>e to low enrollment and not safety concerns.) Collapse << | - | United States, Arizona ... more >> Research Center Scottsdale, Arizona, United States United States, Minnesota Research Center Rochester, Minnesota, United States Collapse << |
| NCT01155466 | - | - | Completed | - | - |
| NCT00615017 | Severe Sepsis | Phase 2 | Completed | - | - |
| NCT00675428 | - | - | Terminated(Sponsor decision du... more >>e to low enrollment and not safety concerns.) Collapse << | - | - |
| NCT01294436 | Type2 Diabetes ... more >> High Blood Sugar Collapse << | Phase 3 | Completed | - | Japan ... more >> Research Site Nagoya, Aichi, Japan Research Site Owariasahi, Aichi, Japan Research Site Toyohashi, Aichi, Japan Research Site Hirosaki, Aomori, Japan Research Site Niihama, Ehime, Japan Research Site Itoshima, Fukuoka, Japan Research Site Yukuhashi, Fukuoka, Japan Research Site Annaka, Gunma, Japan Research Site OTA, Gunma, Japan Research Site Aki-gun, Hiroshima, Japan Research Site Sapporo, Hokkaido, Japan Research Site Sanuki, Kagawa, Japan Research Site Takamatsu, Kagawa, Japan Research Site Kamakura, Kanagawa, Japan Research Site Kawasaki, Kanagawa, Japan Research Site Yokohamashi, Kanagawa, Japan Research Site Yokohama, Kanagawa, Japan Research Site Zushi, Kanagawa, Japan Research Site Sendai, Miyagi, Japan Research Site Matsumoto, Nagano, Japan Research Site Suita, Osaka, Japan Research Site Otsu, Shiga, Japan Research Site Atami, Shizuoka, Japan Research Site Komatsushima, Tokushima, Japan Research Site Chiyoda, Tokyo, Japan Research Site Chuo, Tokyo, Japan Research Site Mitaka, Tokyo, Japan Research Site OTA, Tokyo, Japan Research Site Shibuya, Tokyo, Japan Research Site Shinjuku, Tokyo, Japan Research Site Taito, Tokyo, Japan Research Site Takaoka, Toyama, Japan Research Site UBE, Yamaguchi, Japan Research Site Fukuoka, Japan Research Site Hiroshima, Japan Research Site Kochi, Japan Research Site Osaka, Japan Research Site Shizuoka, Japan Research Site Toyama, Japan Collapse << |
| NCT01294436 | - | - | Completed | - | - |
| NCT01524900 | - | - | Completed | - | - |
| NCT00681811 | - | - | Terminated(Terminated prior to... more >> planned completion date due to lack of efficacy.) Collapse << | - | - |
| NCT03416062 | Jaundice; Malignant | Phase 3 | Active, not recruiting | September 2019 | Russian Federation ... more >> Irkutsk Regional Clinical Hospital Irkutsk, Russian Federation, 664079 North-West State Medical University named after I.I. Mechnikov Saint Petersburg, Russian Federation, 191015 Collapse << |
| NCT03058562 | Tourette Syndrome ... more >> Chronic Motor Tic Disorder Collapse << | Phase 1 | Completed | - | Germany ... more >> Medizinische Hochschule Hannover Hannover, Niedersachsen, Germany, 30625 Collapse << |
| NCT03089320 | Unhealthy Alcohol Use | Not Applicable | Recruiting | August 31, 2024 | United States, California ... more >> Greater Los Angeles VA Healthcare Center Infectious Disease Section Not yet recruiting Los Angeles, California, United States, 90073 Contact: Matthew Goetz, MD 310-478-3711 ext 43818 Matthew.goetz@va.gov United States, District of Columbia Washington DC Veterans Affairs Not yet recruiting Washington, District of Columbia, United States, 20422 Contact: Cynthia Gilbert, MD 202-745-7560 Cynthia.gibert@med.va.gov United States, Georgia VA Medical Center Recruiting Atlanta, Georgia, United States, 30033 Contact: Vincent Marconi, MD 404-321-6111 ext 7592 vcmarco@emory.edu United States, New York James J. Peters VA Medical Center Recruiting Bronx, New York, United States, 10468 Contact: Sheldon Brown, MD 718-584-9000 ext 5842 Sheldon.brown@va.gov VA NY Harbor Healthcare System Recruiting New York, New York, United States, 10010 Contact: Michael Simberkoff, MD 212-951-3417 Mike.SimberkoffMD@va.gov United States, Texas VA North Texas Healthcare System Not yet recruiting Dallas, Texas, United States, 75216 Contact: Roger Berdino, MD 214-857-1410 Roger.Bedimo@va.gov VAMC Houston Recruiting Houston, Texas, United States, 77030 Contact: Maria Rodriguez-Barradas, MD 713-794-8856 maria.rodriguez-barradas@va.gov Collapse << |
| NCT03758352 | Liver Transplant; Complication... more >>s Ischemia Reperfusion Injury Collapse << | Not Applicable | Not yet recruiting | November 2020 | - |
| NCT02726542 | NAFLD | Early Phase 1 | Recruiting | September 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Takara L. Stanley, MD 617-726-5312 tstanley@partners.org Collapse << |
| NCT01235689 | Crohn's Disease | Phase 3 | Completed | - | - |
| NCT02655705 | Psoriasis | Phase 4 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of, KS009 Collapse << |
| NCT01524900 | - | - | Completed | - | - |
| NCT00615017 | - | - | Completed | - | - |
| NCT00681811 | Late Infantile Metachromatic L... more >>eukodystrophy Collapse << | Phase 2 | Terminated(Terminated prior to... more >> planned completion date due to lack of efficacy.) Collapse << | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark, 2100 Collapse << |
| NCT03051256 | Depressive Disorder, Major ... more >> Depressive Disorder, Treatment-Resistant Collapse << | Phase 2 | Recruiting | May 2019 | United States, Arkansas ... more >> Woodland International Research Group Recruiting Little Rock, Arkansas, United States, 72211 Contact: Heather Spencer 501-221-8681 hspencer@woodlandintlresearchgrp.com Contact: Valarie Valdez 501-221-8681 valarie@woodlandintlresearchgrp.com Principal Investigator: George Kanis United States, California Collaborative Neuroscience Network, LLC Not yet recruiting Garden Grove, California, United States, 92845-2506 Contact: Hanna Voltattorni 562-304-1742 hannavoltattorni@cnstrial.com Contact: Irene Acacio 562-304-1742 ireneacacio@cnstrial.com Principal Investigator: David Walling Artemis Institute for Clinical Research Not yet recruiting San Diego, California, United States, 92103 Contact: Beth Panella bd@artemis-research.com Contact: Maria Nunez mnunez@artemis-research.com Principal Investigator: Vishaal Mehra United States, Florida Innovative Clinical Research, Inc Recruiting Hialeah, Florida, United States, 33012 Contact: Maryan Negron mnegron@segaltrials.com Principal Investigator: Rishi Kakar United States, Georgia iResearch Atlanta, LLC Not yet recruiting Decatur, Georgia, United States, 30030 Contact: Amanda T Goza 404-537-1281 Amanda.Goza@iResearchAtlanta.com Contact: Casey Chacona 404-537-1281 Casey.Chacona@iResearchAtlanta.com Principal Investigator: David Purselle United States, Missouri St. Louis Clinical Trials, LLC Recruiting Saint Louis, Missouri, United States, 63141 Contact: Emra Okanovic 314-771-6387 eokanovic@slct.co Contact: Jacqueline Doan 314-771-6387 jdoan@slct.co Principal Investigator: Daniel Gruener United States, New Jersey Hassman Research Institute Recruiting Berlin, New Jersey, United States, 08009 Contact: Lisa Speight 856-753-7335 ext 716 lspeight@hritrials.com Contact: Vania DaSilva, BA 856-753-7335 vdasilva@hritrials.com Principal Investigator: Howard Hassman, DO United States, Ohio Midwest Clinical Research Center Recruiting Dayton, Ohio, United States, 45417-3445 Contact: Crystal Jackson 937-424-1050 cjackson@midwestclinical.com Contact: Melissa Ellis 937-424-1050 MEllis@midwestclinical.com Principal Investigator: Otto Dueno United States, Texas Pillar Clinical Research, LLC Recruiting Richardson, Texas, United States, 75080 Contact: Quynh A Doan 214-396-4844 ext 206 qadoan@pillarhc.com Contact: Iliana Maldonado 214-396-4844 ext 208 imaldonado@pillarhc.com Principal Investigator: Scott Bartley Collapse << |
| NCT03261817 | Schizophrenia ... more >> Physical Activity Schizo Affective Disorder Collapse << | Not Applicable | Recruiting | December 31, 2022 | France ... more >> Caen University Hospital Recruiting Caen, France, 14033 Contact: Sonia Dollfus, MD, PhD + 33 2 31 06 5018 dollfus-s@chu-caen.fr Principal Investigator: Sonia Dollfus, MD, PhD Collapse << |
| NCT02302560 | - | - | Withdrawn | November 2019 | Germany ... more >> Department of Anesthesiology and Intensive Care Medicine Berlin Berlin, Germany, 13353 Collapse << |
| NCT01235689 | - | - | Completed | - | - |
| NCT00537030 | Adult Acute Lymphoblastic Leuk... more >>emia Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Unknown | - | - |
| NCT02196831 | Human Immunodeficiency Virus (... more >>HIV) Nonalcoholic Fatty Liver Disease (NAFLD) Nonalcoholic Steatohepatitis (NASH) Collapse << | Not Applicable | Active, not recruiting | July 2019 | United States, Maryland ... more >> National Institutes of Health Bethesda, Maryland, United States, 20892 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00537030 | - | - | Unknown | - | - |
| NCT02394301 | - | - | Suspended(Poor enrollment) | December 2015 | United States, Florida ... more >> The Medimix Specialty Pharmacy, LLC Jacksonville, Florida, United States, 32216 Collapse << |
| NCT03475836 | Cognitive Change ... more >> Affect Collapse << | Not Applicable | Completed | - | - |
| NCT02386553 | Spinal Muscular Atrophy | Phase 2 | Active, not recruiting | January 25, 2022 | United States, Colorado ... more >> Children's Hospital Colorado Aurora, Colorado, United States, 80045 United States, Florida Nemours Children's Clinic Orlando, Florida, United States, 32827 United States, Illinois Ann & Robert H. Lurie Children's Hospital of Chicago Chicago, Illinois, United States, 60611 United States, Maryland The Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, New York Columbia University Medical Center, The SMA Clinical Research Center New York, New York, United States, 10032 United States, Utah Primary Children's Medical Center Salt Lake City, Utah, United States, 84112 Australia, Victoria Royal Children's Hospital- Melbourne Parkville, Victoria, Australia, 3052 Germany Dept. of Neuropediatrics and Muscle Disorders Universitätsklinikum Freiburg Freiburg, Baden-Württemberg, Germany, 79106 Italy Bambino Gesu' Children's Research Hospital Roma, Lazio, Italy, 165 Fondazione Serena Onlus - Centro Clinico Nemo Milano, Italy, 20162 Qatar Hamad General Hospital Doha, Qatar, 3050 Taiwan Kaohsiung Medical University Hospital Kaohsiung City, Taiwan, 806 National Taiwan University Hospital Taipei, Taiwan, 100 Turkey Hacettepe University Medical Faculty Ankara, Sihhiye, Turkey, 6100 Collapse << |
| NCT02130635 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Grosshansdorf, Schleswig-Holstein, Germany, 22927 GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT03471143 | Niemann-Pick Disease, Type C | Phase 1 Phase 2 | Recruiting | February 2022 | United States, Missouri ... more >> St. Louis Children's Hospital Recruiting Saint Louis, Missouri, United States, 63110 Contact: Daniel S Ory, MD 314-362-8737 dory@wustl.edu Contact: Laura A Linneman, RN 314-454-4950 llinneman@wustl.edu Principal Investigator: Daniel S Ory, MD Principal Investigator: F S Cole, MD Collapse << |
| NCT00126087 | Stroke | Phase 4 | Terminated(no further funding ... more >>available for recruiting and testing participants) Collapse << | - | Germany ... more >> University of Münster, Department of Neurology Münster, Nordrhein-Westfalen, Germany, 48129 Collapse << |
| NCT03434470 | - | - | Recruiting | November 30, 2019 | Norway ... more >> University of Bergen, Haukeland University Hospital Recruiting Bergen, Hordaland, Norway, 5057 Contact: Anesa Mulabecirovic, MD anesa.mulabecirovic@uib.no Contact: Roald Havre, MD,PhD roald.flesland.havre@helse-bergen.no Principal Investigator: Anesa Mulabecirovic, MD, PhD-Cand Sub-Investigator: Alexander Rafael Leiva, MD Collapse << |
| NCT02130635 | - | - | Completed | - | - |
| NCT01572792 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02741791 | Treatment Resistant Major Depr... more >>essive Disorder Collapse << | Phase 3 | Recruiting | March 2019 | - |
| NCT03595579 | Major Depressive Disorder | Phase 2 | Active, not recruiting | January 2019 | United States, California ... more >> Axsome Study Site Beverly Hills, California, United States, 90210 United States, Florida Axsome Study Site Jacksonville, Florida, United States, 32256 Axsome Study Site Orlando, Florida, United States, 32801 United States, Washington Axsome Study Site Bellevue, Washington, United States, 98007 Collapse << |
| NCT03510741 | Schizophrenia ... more >> Schizophreniform Disorders Schizoaffective Disorder Collapse << | Phase 2 | Not yet recruiting | July 30, 2020 | Pakistan ... more >> Balochistan Institute of Behavioral Science Not yet recruiting Quetta, Balochistan, Pakistan Contact: Prof. Ghulam Rasool Institute of Psychiatry, Rawalpindi Not yet recruiting Rawalpindi, Islamabad, Pakistan Contact: Prof. Fareed A Minhas Abbasi Shaheed Hsopital Not yet recruiting Karachi, Sindh, Pakistan Contact: Dr. Sohail Ahmed Civil Hospital Karachi Not yet recruiting Karachi, Sindh, Pakistan Contact: Prof. Raza Ur Rehman Karwan e Hayat Not yet recruiting Karachi, Sindh, Pakistan Contact: Dr. Ajmal Kazmi Collapse << |
| NCT01572792 | - | - | Completed | - | - |
| NCT00124982 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT00619489 | Ulcerative Colitis ... more >> Crohn's Disease Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> London Health Sciences Centre London, Ontario, Canada Collapse << |
| NCT00005658 | Psychotic Disorder ... more >> Schizoaffecitve Disorder Schizophrenia Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institute of Mental Health (NIMH) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00631475 | Idiopathic Pulmonary Fibrosis | Phase 3 | Completed | - | - |
| NCT00619489 | - | - | Completed | - | - |
| NCT01914263 | Hepatocellular Carcinoma ... more >> Renal Cell Carcinoma Lung Cancer Collapse << | Phase 1 | Unknown | June 2016 | China, Hainan ... more >> Hannan BOAO Life infinity international anti-aging medical center Recruiting Qionghai, Hainan, China, 571434 China, Liaoning The 210 Hospital of Chinese People's Liberation Army Recruiting Dalian, Liaoning, China, 116021 Contact: Dayue Han, MD 86-13387854002 tc210dl@hotmail.com Principal Investigator: Dayue Han, MD China, Shaanxi The 323 Hospital of Chinese People's Liberation Army Recruiting Xi'an, Shaanxi, China, 710054 Contact: Liming wang, MD 86-29-84756502 wanglm@fmmu.edu.cn Principal Investigator: Liming Wang, MD Collapse << |
| NCT01735071 | Ovarian Epithelial Cancer Recu... more >>rrent Collapse << | Phase 2 | Active, not recruiting | December 2018 | Italy ... more >> Azienda Ospedaliera Spedali Civili di Brescia Brescia, Italy AO Fatebenefratelli e Oftalmico Milano, Italy Istituto Europeo di Oncologia Milan, Italy Azienda Ospedaliera S. Gerardo Monza, Italy Istituto Oncologico Veneto Padova, Italy Policlinico Universitario Agostino Gemelli di Roma Roma, Italy Mauriziano Hospital Torino, Italy Collapse << |
| NCT02562963 | Non-small Cell Lung Cancer ... more >> Gastric Cancer Hepatocellular Carcinoma Colorectal Cancer Collapse << | Phase 1 Phase 2 | Recruiting | December 2018 | China ... more >> Hua Xin Hosptial First Hosptial of Tsinghua University Recruiting Beijing, China, 100016 Contact: Minghui Zhang, PhD 0086-10-62799520 mh-zhang@tsinghua.edu.cn Principal Investigator: Minghui Zhang, PhD Shanghai Public Health Clinical Center Recruiting Shanghai, China, 201508 Contact: Xiaoyan Zhang, MD, PhD 0086-21-37990333 ext 7310 zhangxiaoyan@shaphc.org Principal Investigator: Xiaoyan Zhang, MD, PhD Collapse << |
| NCT00631475 | - | - | Completed | - | - |
| NCT01389973 | Primary Biliary Cirrhosis | Phase 2 | Completed | - | United States, Florida ... more >> Jacksonville, Florida, United States Miami, Florida, United States Naples, Florida, United States United States, Mississippi Tupelo, Mississippi, United States United States, New York New York, New York, United States United States, Texas Houston, Texas, United States Canada, British Columbia Vancouver, British Columbia, Canada Canada, Nova Scotia Halifax, Nova Scotia, Canada Canada, Ontario Toronto, Ontario, Canada Canada, Quebec Montreal, Quebec, Canada Collapse << |
| NCT00124982 | - | - | Completed | - | - |
| NCT01935115 | Treatment Resistant Depression | Phase 4 | Completed | - | Canada, Saskatchewan ... more >> Royal University Hospital Saskatoon, Saskatchewan, Canada, S7N 0W8 Collapse << |
| NCT01389973 | - | - | Completed | - | - |
| NCT03722576 | Primary Sclerosing Cholangitis | Phase 2 | Not yet recruiting | November 2020 | United States, Arizona ... more >> Keith D. Lindor, MD Not yet recruiting Phoenix, Arizona, United States, 85004 Contact: Elizabeth Carey, MD 480-515-6296 carey.elizabeth@mayo.edu Contact: Nicholas F LaRusso 507-284-1006 larusso.nicholas@mayo.edu Collapse << |
| NCT01908192 | Schizophrenia | Phase 2 Phase 3 | Recruiting | June 30, 2019 | - |
| NCT00963924 | Schizophrenia | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT02579642 | Depressive Disorder | Phase 4 | Enrolling by invitation | May 2019 | Canada, Alberta ... more >> Alberta Hospital Edmonton Edmonton, Alberta, Canada, T5J 2J7 University of Alberta Hospital Edmonton, Alberta, Canada, T6G 2B7 Collapse << |
| NCT00352586 | - | - | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01811472 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Novartis Investigative Site Mobile, Alabama, United States, 36608 United States, California Novartis Investigative Site San Diego, California, United States, 92114 United States, Florida Novartis Investigative Site Gainesville, Florida, United States, 32610-0277 Novartis Investigative Site Miami, Florida, United States, 33126 Novartis Investigative Site Tamarac, Florida, United States, 33319 United States, Hawaii Novartis Investigative Site Honolulu, Hawaii, United States, 96814 United States, Kentucky Novartis Investigative Site Louisville, Kentucky, United States, 40213 United States, Mississippi Novartis Investigative Site Tupelo, Mississippi, United States, 38801 United States, Texas Novartis Investigative Site Houston, Texas, United States, 77030 Novartis Investigative Site Plano, Texas, United States, 75093 United States, Virginia Novartis Investigative Site Richmond, Virginia, United States, 23298 Collapse << |
| NCT00963924 | - | - | Completed | - | - |
| NCT01578239 | - | - | Active, not recruiting | - | - |
| NCT02766790 | Healthy | Not Applicable | Unknown | July 2017 | United States, Missouri ... more >> QPS Bio-Kinetic, LLC Recruiting Springfeild, Missouri, United States, 65802 Contact: Brendon Bourg 417-425-3033 brendon.bourg@qps.com Collapse << |
| NCT01811472 | - | - | Completed | - | - |
| NCT00819910 | Hypertriglyceridemia in Type 4... more >> Hyperlipidemia Non Diabetic Subjects With Normoglycemia Collapse << | Phase 4 | Terminated(Slow recruitment an... more >>d increase in deployment overseas limiting follow up) Collapse << | - | United States, Texas ... more >> Brooke Army Medical Center San Antonio, Texas, United States, 78234 Collapse << |
| NCT00819910 | - | - | Terminated(Slow recruitment an... more >>d increase in deployment overseas limiting follow up) Collapse << | - | - |
| NCT01578239 | Carcinoid Tumor of the Small B... more >>owel Neuroendocrine Tumour Collapse << | Phase 3 | Active, not recruiting | February 2021 | - |
| NCT01894243 | Solid Tumours | Phase 1 | Completed | - | Czech Republic ... more >> Research Site Brno, Czech Republic France Research Site Bordeaux Cedex, France Research Site Bordeaux, France Research Site Dijon, France Research Site Grenoble cedex 9, France Research Site Lyon Cedex 8, France Research Site Paris Cedex 13, France Korea, Republic of Research Site Seoul, Korea, Republic of Netherlands Research Site Amsterdam, Netherlands Research Site Maastricht, Netherlands United Kingdom Research Site London, United Kingdom Research Site Sutton, United Kingdom Collapse << |
| NCT01825044 | Traumatic Brain Injury | Phase 2 | Completed | - | Denmark ... more >> Dept. of Neurosurgery, Rigshospitalet, University of Copenhagen Copenhagen, Denmark, 2100 Collapse << |
| NCT01201720 | Liver Cirrhosis ... more >> Liver Failure Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Clínic of Barcelona Barcelona, Spain, 08028 Collapse << |
| NCT02594462 | Sickle Cell Disease | Phase 4 | Active, not recruiting | June 2019 | Brazil ... more >> Bahiana School of Medicne and Public Health Salvador, Bahia, Brazil University of Sao Paulo Ribeirao Preto, Sao Paulo, Brazil, 14049-900 Collapse << |
| NCT01348347 | Neoplasms | Phase 1 | Completed | - | Japan ... more >> 1230.15.001 National Cancer Center Hospital, Chuo-ku, Tokyo, Japan Collapse << |
| NCT02679417 | Non-alcoholic Fatty Liver Dise... more >>ase Collapse << | Not Applicable | Completed | - | Thailand ... more >> Faculty of Medicine Siriraj Hospital Bangkoknoi, Bangkok, Thailand, 10700 Collapse << |
| NCT03114215 | ALS Amyotroph... more >>ic Lateral Sclerosis Motor Neuron Disease Collapse << | Phase 2 | Active, not recruiting | December 2017 | France ... more >> Hopital Gui De Chauliac Montpellier, France, 34000 Collapse << |
| NCT01703260 | Nonalcoholic Steatohapatitis | Phase 2 | Terminated(Company decision; N... more >>o safety or efficacy concerns (see detailed description)) Collapse << | - | United States, California ... more >> Coronado, California, United States, 92118 Coronado, California, United States United States, Maryland Annapolis, Maryland, United States, 21401 Baltimore, Maryland, United States, 21202 Collapse << |
| NCT01348347 | - | - | Completed | - | - |
| NCT02390843 | Retinoblastoma ... more >> Clear Cell Sarcoma Renal Cell Carcinoma Rhabdoid Tumor Wilms Tumor Hepatoblastoma Neuroblastoma Germ Cell Tumors Ewings Sarcoma Non-rhabdomyosarcoma Soft Tissue Sarcoma Osteosarcoma Rhabdomyosarcoma Collapse << | Phase 1 | Recruiting | February 2021 | United States, Georgia ... more >> Children's Healthcare of Atlanta/Emory University Recruiting Atlanta, Georgia, United States, 30322 Contact: Amy Autry-Bush 404-785-6011 amy.autry-bush@choa.org Principal Investigator: Kelly Goldsmith, MD Sub-Investigator: William T Cash, MD Collapse << |
| NCT01456039 | Lymphoma, T-cell, Peripheral | Phase 1 Phase 2 | Active, not recruiting | June 30, 2018 | Japan ... more >> Nagoya Daini Red Cross Hospital Nagoya, Aichi, Japan, 466-8650 Nagoya City University Hospital Nagoya, Aichi, Japan, 467-8602 National Cancer Center Hospital East Kashiwa, Chiba, Japan, 277-8577: Ehime University Hospital To-on, Ehime, Japan, 791-0295 Chugoku Central Hospital Fukuyamashi, Hiroshima, Japan, 720-0001 Sapporo Hokuyu Hospital Sapporo, Hokkaido, Japan, 003-0006 Sapporo Medical University Hospital Sapporo, Hokkaido, Japan, 060-8543 Tokai University School of Medicine Isehara, Kanagawa, Japan, 259-1193 Kochi Medical School Hospital Nankoku, Kochi, Japan, 783-8505 Kinki University Hospital Sayama, Osaka, Japan, 589-8511 National Cancer Center Hospital Chuo, Tokyo, Japan, 104-0045 Japanese Red Cross Medical Center Shibuya, Tokyo, Japan, 150-8935 Kyushu University Hospital Fukuoka, Japan, 812-8582 Kumamoto University Hospital Kumamoto, Japan, 860-8556 University hospital, Kyoto prefectural University of Medicine Kyoto, Japan, 602-8566 Tohoku University Hospital Miyagi, Japan, 980-8574 The Cancer Institute Hospital of JFCR Tokyo, Japan, 135-8550 Collapse << |
| NCT01456039 | - | - | Active, not recruiting | - | - |
| NCT02647671 | Colonic Cancer | Phase 1 | Active, not recruiting | March 2019 | United States, Michigan ... more >> University of Michigan Hospital Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02309723 | Alzheimer's Disease ... more >> Mild Cognitive Impairment Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01703260 | - | - | Terminated(Company decision; N... more >>o safety or efficacy concerns (see detailed description)) Collapse << | - | - |
| NCT02309723 | - | - | Completed | - | - |
| NCT03456284 | Liver Transplantation | Not Applicable | Not yet recruiting | December 2023 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Cristiano Quintini, MD 216-445-3388 quintic@ccf.org Contact: Kevin Smith 2164445868 smithk24@ccf.org Collapse << |
| NCT01449006 | - | - | Completed | - | - |
| NCT03036332 | Metabolic Syndrome | Not Applicable | Completed | - | - |
| NCT01637233 | - | - | Completed | - | Argentina ... more >> Hospital Ramos Mejía Buenos Aires, Argentina, C1221ADC Fundación IDEAA Buenos Aires, Argentina, C1405CKC CAICI Rosario, Argentina Thailand Chulalongkorn University Hospital Bangkok, Thailand, 10330 United Kingdom Brighton & Sussex University NHS Trust Brighton, Sussex, United Kingdom, BN21ES, Imperial Healthcare, St. Mary's Hospital London, United Kingdom, W2 Collapse << |
| NCT01512199 | Metastatic Breast Cancer | Phase 1 Phase 2 | Terminated(Improved, different... more >> standard of care caused business decision to terminate) Collapse << | - | Argentina ... more >> Unidad de Investigación FP Clinical Pharma en Centro Medico Integral Fitz Roy Acevedo, Ciudad Autónoma de Buenos Aires, Argentina Centro Medico San Roque San Miguel, Tucuman, Argentina, T4000LAK Hospital Britanico Buenos Aires, Argentina, 01280 Sanatorio de la Providencia Buenos Aires, Argentina, C1050AAK Instituto Damic - Fundacion Rusculleda Cordoba, Argentina, X5003DCE ISIS Centro Especializado Santa Fe, Argentina, S3000CVK Chile Instituto de Tereplas Oncologicas Providencia INTOP Providencia, Santiago, Chile Hospital Clinico San Borja Arriaran Santiago, Chile Collapse << |
| NCT01393678 | Chronic Liver Disease | Phase 3 | Completed | - | Korea, Republic of ... more >> Hanyang University Hospital Seoul, Seongdong-gu,, Korea, Republic of Collapse << |
| NCT02911961 | Acetaminophen Exposure | Phase 4 | Recruiting | September 2020 | United States, Colorado ... more >> University of Colorado Denver Recruiting Denver, Colorado, United States, 80045 Contact: Clinical Research Coordinator 303-389-1800 Clinical.Trials@rmpdc.org Collapse << |
| NCT01434654 | - | - | Terminated(Slower than expecte... more >>d recruitment) Collapse << | - | Australia, New South Wales ... more >> St Vincent's Hospital Sydney, New South Wales, Australia, 2010 Australia, Victoria The Alfred Hospital Prahran, Victoria, Australia, 3181 Collapse << |
| NCT01449006 | Human Immunodeficiency Virus (... more >>HIV) HIV Associated Neurocognitive Disorders (HAND) Collapse << | Phase 4 | Completed | - | Australia, New South Wales ... more >> St. Vincent's Hospital Sydney, New South Wales, Australia, 2010 Australia, Victoria The Alfred Hospital Melbourne, Victoria, Australia, 3181 Collapse << |
| NCT01076179 | - | - | Completed | - | - |
| NCT02441283 | Hepatitis C | Phase 2 | Active, not recruiting | July 18, 2020 | - |
| NCT02122146 | Neoplasms | Phase 1 | Terminated(The study was prema... more >>turely discontinued due to a business-related decision on 09-FEB-2016. The decision to terminate the trial was based on the overall results.) Collapse << | - | United States, California ... more >> Keck Hospital of USC Los Angeles, California, United States, 90033 LAC&USC Medical Center Los Angeles, California, United States, 90033 USC/Norris Comprehensive Cancer Center / Investigational Drug Services Los Angeles, California, United States, 90033 USC/Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 United States, Colorado Anschutz Cancer Pavilion Aurora, Colorado, United States, 80045 University of Colorado Denver CTO (CTRC) Aurora, Colorado, United States, 80045 University of Colorado Hospital Aurora, Colorado, United States, 80045 United States, Washington Swedish Medical Center Seattle, Washington, United States, 98104 Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT03323216 | - | - | Recruiting | March 31, 2024 | Germany ... more >> University Hospital RWTH Aachen Recruiting Aachen, NRW, Germany, 52074 Contact: Ben A Kappel, MD PhD Collapse << |
| NCT03545672 | - | - | Recruiting | December 1, 2018 | China, Shanghai ... more >> Renji Hospital Recruiting Shanghai, Shanghai, China, 200127 Contact: Zehao Fung, postgraduate fengzehao2016@126.com Contact: Pan Jiang, postgraduate jiangpanmed@163.com Collapse << |
| NCT01434654 | - | - | Terminated(Slower than expecte... more >>d recruitment) Collapse << | - | - |
| NCT01148771 | Healthy | Phase 4 | Completed | - | United States, Connecticut ... more >> Hartford Hospital Hartford, Connecticut, United States, 06102 Collapse << |
| NCT01148771 | - | - | Completed | - | - |
| NCT03249792 | Solid Tumor L... more >>ymphoma Collapse << | Phase 1 | Recruiting | October 25, 2021 | United States, California ... more >> University of California San Diego Moores Cancer Center ( Site 0004) Recruiting La Jolla, California, United States, 92093 Contact: Study Coordinator 858-822-7742 United States, Illinois University of Chicago ( Site 0002) Recruiting Chicago, Illinois, United States, 60637 Contact: Study Coordinator 773-702-1835 United States, New York New York Presbyterian Hospital/Columbia University ( Site 0001) Recruiting New York, New York, United States, 10032 Contact: Study Coordinator 646-317-5381 United States, Texas Mary Crowley Cancer Research Center. ( Site 0005) Recruiting Dallas, Texas, United States, 75230 Contact: Study Coordinator 214-658-1945 MD Anderson Cancer Centr. ( Site 0006) Active, not recruiting Houston, Texas, United States, 77030 Israel Sheba Medical Center ( Site 0301) Recruiting Ramat-Gan, Israel, 5265601 Contact: Study Coordinator +97235302542 Collapse << |
| NCT02122146 | - | - | Terminated(The study was prema... more >>turely discontinued due to a business-related decision on 09-FEB-2016. The decision to terminate the trial was based on the overall results.) Collapse << | - | - |
| NCT00761267 | - | - | Completed | - | - |
| NCT02125877 | - | - | Completed | - | - |
| NCT02125877 | Chronic Iron Overload Due to T... more >>ransfusion-dependant Anemias Collapse << | Phase 2 | Completed | - | - |
| NCT01076179 | - | - | Completed | - | - |
| NCT00761267 | Candidemia | Phase 3 | Completed | - | - |
| NCT03345589 | Primary Biliary Cholangitis | Phase 4 | Recruiting | January 1, 2019 | China, Sichuan ... more >> West China Hospital Recruiting Chengdu, Sichuan, China, 610041 Contact: Li Yang +8618980601276 yangli_hx@scu.edu.cn Collapse << |
| NCT00385580 | Prostate Cancer | Phase 2 | Completed | - | United States, California ... more >> Cedars Sinai Medical Center Los Angeles, California, United States, 90048 United States, Illinois University Of Chicago Chicago, Illinois, United States, 60637 United States, Maryland The Bunting Blaustein Cancer Research Building Baltimore, Maryland, United States, 21231 United States, New York Memorial Sloan-Kettering Cancer Center-Sidney Kimmel Center New York, New York, United States, 10021 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 United States, Wisconsin University Of Wisconsin Paul P Carbone Comprehensive Ca Ctr Madison, Wisconsin, United States, 53792 France Local Institution Montpellier, France, 34298 Local Institution Paris, France, 75015 Local Institution Villejuif Cedex, France, 94800 Italy Local Institution Rome, Italy, 00152 Collapse << |
| NCT00385580 | - | - | Completed | - | - |
| NCT02014467 | - | - | Completed | - | - |
| NCT00207090 | Advanced Solid Tumors ... more >> Neoplasms Collapse << | Phase 1 | Completed | - | United States, Ohio ... more >> The Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02261376 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Hammersmith Medicines Research Ltd London, United Kingdom, NW10 7EW Collapse << |
| NCT02014467 | Osteoporosis, Postmenopausal | Phase 3 | Completed | - | China, Sichuan ... more >> GSK Investigational Site Chengdu, Sichuan, China, 610041 GSK Investigational Site Chengdu, Sichuan, China, 610072 China GSK Investigational Site Beijing, China, 100035 GSK Investigational Site Beijing, China, 100050 GSK Investigational Site Beijing, China, 100730 GSK Investigational Site Chengdu, China, 610083 GSK Investigational Site Shanghai, China, 200040 GSK Investigational Site Shanghai, China, 200233 Collapse << |
| NCT01910402 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Active, not recruiting | December 31, 2020 | - |
| NCT01910402 | - | - | Active, not recruiting | - | - |
| NCT01262898 | Gastroparesis | Phase 2 | Completed | - | - |
| NCT00207090 | - | - | Completed | - | - |
| NCT02299375 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | - |
| NCT02299375 | - | - | Completed | - | - |
| NCT03642405 | - | - | Active, not recruiting | January 1, 2022 | Denmark ... more >> Herlev Hospital Herlev, Denmark, 2730 Collapse << |
| NCT03564691 | Neoplasms | Phase 1 | Recruiting | July 27, 2022 | United States, Michigan ... more >> Henry Ford Health System ( Site 0002) Recruiting Detroit, Michigan, United States, 48202 Contact: Study Coordinator 313-916-7453 United States, Texas South Texas Accelerated Research Therapeutics, LLC (START) ( Site 0001) Recruiting San Antonio, Texas, United States, 78229 Contact: Study Coordinator 210-593-5265 Canada, Ontario The Ottawa Hospital ( Site 0031) Recruiting Ottawa, Ontario, Canada, K1H 8L6 Contact: Study Coordinator 613737770070185 Princess Margaret Cancer Centre ( Site 0033) Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Study Coordinator 4169462911 Israel Rambam Medical Center ( Site 0042) Recruiting Haifa, Israel, 3109601 Contact: Study Coordinator +972535316961 Sourasky Medical Center ( Site 0041) Recruiting Tel Aviv, Israel, 6423906 Contact: Study Coordinator +97236973082 Collapse << |
| NCT03665948 | Nutrition Ket... more >>ogenic Dieting Low Glycemic Index Diet Sport Crossfit Collapse << | Not Applicable | Completed | - | Poland ... more >> Institute of Human Nutrition and Dietetics, Poznan University of Life Sciences Poznań, Poland, 60-624 Collapse << |
| NCT01262898 | - | - | Completed | - | - |
| NCT03385369 | Safety Health... more >>y Subjects Collapse << | Phase 1 | Completed | - | United States, California ... more >> Research Site Anaheim, California, United States, 92801 Collapse << |
| NCT03418935 | Jaundice, Obstructive | Phase 3 | Active, not recruiting | September 2019 | Russian Federation ... more >> Irkutsk Regional Clinical Hospital Irkutsk, Russian Federation, 664079 North-West State Medical University named after I.I. Mechnikov Saint Petersburg, Russian Federation, 191015 State Budgetary Health Care Institution "City Hospital № 26" Saint Petersburg, Russian Federation, 196247 "BioEk" Ltd. Saint Petersburg, Russian Federation, 197342 Leningrad Regional Clinical Hospital Saint Petersburg, Russian Federation Collapse << |
| NCT02294682 | - | - | Completed | - | - |
| NCT01949545 | Solid Tumors ... more >>Hematologic Malignancies Hepatic Impairment Collapse << | Phase 1 | Completed | - | United States, Michigan ... more >> Karmanos Cancer Institute Detroit, Michigan, United States United States, North Carolina Duke Cancer Institute Durham, North Carolina, United States United States, Ohio Gabrail Cancer Center Canton, Ohio, United States United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States United States, Washington Evergreen Hematology and Oncology Spokane, Washington, United States France Institut Gustave Roussy Paris, Villejuif Cedex, France Netherlands University Medical Centre Utrecht Utrecht, Netherlands United Kingdom Northern Ireland Cancer Trials Centre - Queen's University Belfast TriaIs Centre Belfast, Northern Ireland, United Kingdom Beatson West of Scotland Cancer Centre Glasgow, Scotland, United Kingdom Velindre Hospital Cardlff, Wales, United Kingdom Sir Bobby Robson Cancer Trials Research Centre Newcastle, United Kingdom Collapse << |
| NCT02294682 | Gonorrhea | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Fountain Valley, California, United States, 92708 GSK Investigational Site Los Angeles, California, United States, 90028 GSK Investigational Site Los Angeles, California, United States, 90069 GSK Investigational Site Palm Springs, California, United States, 92262 GSK Investigational Site San Francisco, California, United States, 94103 United States, Florida GSK Investigational Site Orlando, Florida, United States, 32806 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30308 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, Louisiana GSK Investigational Site New Orleans, Louisiana, United States, 70119 United States, Nebraska GSK Investigational Site Omaha, Nebraska, United States, 68114 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44109 United States, Pennsylvania GSK Investigational Site Jenkintown, Pennsylvania, United States, 19046 Puerto Rico GSK Investigational Site San Juan, Puerto Rico, 00927 United Kingdom GSK Investigational Site London, United Kingdom, SW10 9NH Collapse << |
| NCT01571960 | HIV Infections | Phase 1 | Completed | - | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, California Bridge HIV CRS San Francisco, California, United States, 94143 United States, Massachusetts Brigham and Women's Hospital Vaccine CRS (BWH VCRS) Boston, Massachusetts, United States, 02115-6110 United States, New York University of Rochester Vaccines to Prevent HIV Infection CRS Rochester, New York, United States, 14642 Collapse << |
| NCT01788358 | Hypertension | Phase 3 | Completed | - | - |
| NCT01788358 | - | - | Completed | - | - |
| NCT02634073 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 4 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT02578693 | - | - | Recruiting | May 2020 | China, Beijing ... more >> Liver Fibrosis Diagnosis and Treatment Centre, 302 Hospital Recruiting Beijing, Beijing, China, 100039 Contact: Guofeng Chen, MD (8610)66933427 guofengchen302@163.com Principal Investigator: George KK Lau, MD Principal Investigator: Guofeng Chen, MD China, Hong Kong Humanity and Health GI and Liver Centre Recruiting Hong Kong, Hong Kong, China, 00852 Contact: George KK Lau, MD (852)28613777 gkklau@netvigator.com Principal Investigator: George KK Lau, MD Collapse << |
| NCT02293486 | Oxidative Stress | Early Phase 1 | Completed | - | - |
| NCT02634073 | - | - | Completed | - | - |
| NCT03425578 | Healthy | Not Applicable | Recruiting | December 2018 | Canada, Ontario ... more >> University of Waterloo Recruiting Waterloo, Ontario, Canada, N2L 3G1 Contact: Kirsten E Bell, PhD Collapse << |
| NCT01949545 | - | - | Completed | - | - |
| NCT02064985 | Stable Coronary Heart Disease ... more >>(CHD) Collapse << | Phase 4 | Completed | - | China ... more >> Research Site Beijing, China Collapse << |
| NCT00350727 | Glioma | Phase 2 | Completed | - | - |
| NCT03436316 | Asthma | Phase 1 | Recruiting | January 16, 2019 | Germany ... more >> Research Site Recruiting Berlin, Germany, 14050 Collapse << |
| NCT01538342 | Psoriasis Ato... more >>pic Dermatitis Collapse << | Not Applicable | Unknown | July 2016 | France ... more >> Assistance Publique - Hôpitaux de Marseille Recruiting Marseille, France, 13 005 Contact: Marie-Aleth RICHARD, PU-PH 04 91 38 79 91 ext +33 mrichard@ap-hm.fr Principal Investigator: Marie-Aleth RICHARD, PU-PH University Hospital of Nice Active, not recruiting Nice, France, 06000 University Hospital of Toulouse Not yet recruiting Toulouse, France, 31059 Contact: Carle PAUL, PU-PH 05 61 77 76 75 ext +33 paul.c@chu-toulouse.fr Principal Investigator: Carle PAUL, PU-PH Collapse << |
| NCT03329599 | Atherosclerosis ... more >> Adherence, Medication Tolerance Collapse << | Phase 2 | Not yet recruiting | August 1, 2019 | Ghana ... more >> Kwame Nkrumah University of Science & Technology Not yet recruiting Kumasi, Ghana Contact: Stephen Sarfo, MD stephensarfo78@gmail.com Collapse << |
| NCT02973295 | Non-Alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 4 | Not yet recruiting | June 30, 2019 | Croatia ... more >> Clinical Hospital Centre Rijeka, Kresimirova 42, Croatia, 51000 Collapse << |
| NCT02064985 | - | - | Completed | - | - |
| NCT02383485 | Overweight and Obesity ... more >> Non-alcoholic Fatty Liver Disease Collapse << | Not Applicable | Completed | - | United States, Georgia ... more >> Georgia Prevention Institute Augusta, Georgia, United States, 30912 Collapse << |
| NCT00350727 | - | - | Completed | - | - |
| NCT02398201 | Mitochondrial Diseases | Phase 2 | Completed | - | United Kingdom ... more >> Clinical Research Facility, Royal Victoria Infirmary Newcastle upon Tyne, Tyne and Wear, United Kingdom, NE1 4LP Collapse << |
| NCT00560404 | Anemia | Phase 3 | Completed | - | Brazil ... more >> Belo Horizonte, Brazil, 30150-320 Brasilia, Brazil, 70390-108 Campinas, Brazil, 13086-970 Campo Grande, Brazil, 79002-073 Cariacica, Brazil, 29152-230 Curitiba, Brazil, 80050-350 Curitiba, Brazil, 80440-020 Fortaleza, Brazil, 60430160 Jaboatão Dos Guararapes, Brazil, 54400-170 Joinville, Brazil, 89227-680 Juiz de Fora, Brazil, 36036900 Londrina, Brazil, 86015-000 Natal, Brazil, 59020-110 Porto Alegre, Brazil, 90035-903 Porto Alegre, Brazil, 90610000 Ribeirão Preto, Brazil, 14025-170 Rio de Janeiro, Brazil Salvador, Brazil, 40110-060 Sao Jose Do Rio Preto, Brazil, 15090-000 Sao Luis, Brazil, 65020-305 Sao Paulo, Brazil, 01246-903 Sao Paulo, Brazil, 01323-900 Sao Paulo, Brazil, 01532-001 Sao Paulo, Brazil, 03065-000 Sao Paulo, Brazil, O4023 Sorocaba, Brazil, 18030-210 Collapse << |
| NCT00560404 | - | - | Completed | - | - |
| NCT01739517 | Short Bowel Syndrome ... more >> Cholestasis Collapse << | Phase 2 | Unknown | December 2013 | United States, South Carolina ... more >> Palmetto Health Children's Hospital Recruiting Columbia, South Carolina, United States, 29203 Contact: Terra R Varner, PharmD 803-434-3034 terra.varner@palmettohealth.org Principal Investigator: Terra R Varner, PharmD Collapse << |
| NCT01802255 | Poisoning by Inhaled Anaesthet... more >>ic Fluoride Poisoning Recovery From Sedation Renal Function Hepatic Function Collapse << | Phase 3 | Recruiting | June 2020 | Spain ... more >> Hospital Clínico Universitario de Valencia Recruiting Valencia, Spain, 46010 Contact: F Javier Belda, MD, PhD Collapse << |
| NCT03579823 | Healthy Volunteers | Phase 1 | Active, not recruiting | August 15, 2018 | Australia, Victoria ... more >> Nucleus Network Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01378767 | Effects of Krill Oil on Human ... more >>Health Collapse << | Not Applicable | Completed | - | United States, Tennessee ... more >> Cardiorespiratory/Metabolic Laboratory Memphis, Tennessee, United States, 38152 Collapse << |
| NCT00502996 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01290887 | - | - | Terminated(Business decision) | - | - |
| NCT01791205 | - | - | Completed | - | Italy ... more >> Reggio Calabria, Calabria, Italy, 89133 Napoli, Campania, Italy, 80131 Telese Terme, Campania, Italy, 82037 Bologna, Emilia-Romagna, Italy, 40138 Roma, Lazio, Italy, 00133 Roma, Lazio, Italy, 00152 Roma, Lazio, Italy, 00161 Roma, Lazio, Italy, 00189 Legnano, Lombardia, Italy, 20025 Milano, Lombardia, Italy, 20157 Milano, Lombardia, Italy, 20162 Monza, Lombardia, Italy, 20052 Pavia, Lombardia, Italy, 27100 Ancona, Marche, Italy, 60020 Jesi, Marche, Italy, 60035 Torino, Piemonte, Italy, 10126 Torino, Piemonte, Italy, 10128 Bari, Puglia, Italy, 70124 Martina Franca, Puglia, Italy, 74015 Sassari, Sardegna, Italy, 07100 Firenze, Toscana, Italy, 50139 Pisa, Toscana, Italy, 56100 Prato, Toscana, Italy, 59100 Siena, Toscana, Italy, 53100 Perugia, Umbria, Italy, 06122 Cona (Ferrara), Veneto, Italy, 44124 Padova, Veneto, Italy, 35128 Verona, Veneto, Italy, 37126 Collapse << |
| NCT01791205 | - | - | Completed | - | - |
| NCT03495362 | Pre-diabetic | Not Applicable | Recruiting | March 2019 | China, Hubei ... more >> Huazhong University of Science and Technology Recruiting Wuhan, Hubei, China, 430030 Contact: Liegang Liu, MD, PhD +86-27-83650522 liegangliu@gmail.com Principal Investigator: Shufang Xu, PhD Principal Investigator: Liangkai Chen, MD Principal Investigator: Xiaoli Hu, MD Principal Investigator: Qiang Wang, MD Collapse << |
| NCT03165877 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | - |
| NCT01290887 | Eosinophilic Asthma | Phase 3 | Terminated(Business decision) | - | - |
| NCT01343888 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02596360 | Experimental Pain ... more >> Hyperalgesia Collapse << | Phase 1 | Completed | - | France ... more >> CHU Clermont-Ferrand Clermont-Ferrand, France, 63003 Collapse << |
| NCT01343888 | - | - | Completed | - | - |
| NCT02828579 | - | - | Completed | - | - |
| NCT00489489 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy Spain Sanofi-Aventis Administrative Office Barcelona, Spain Collapse << |
| NCT03405818 | - | - | Completed | - | - |
| NCT00489489 | - | - | Completed | - | - |
| NCT01736475 | - | - | Completed | - | - |
| NCT00502996 | - | - | Completed | - | - |
| NCT03405818 | Onychomycosis ... more >> Tinea Unguium Collapse << | Phase 4 | Completed | - | United States, California ... more >> Madera Family Medical Group Madera, California, United States, 93637 Stanford University School of Medicine Palo Alto, California, United States, 94304 United States, District of Columbia MedStar Health Research Institute - MedStar Georgetown University Hospital Washington, District of Columbia, United States, 20016 United States, Florida Doctors Research Network South Miami, Florida, United States, 33143 United States, New York University Hospital, SUNY Downstate Medical Center Brooklyn, New York, United States, 11203 Skin Specialty Dermatology New York, New York, United States, 10155 United States, Oregon Cyn3rgy Research Gresham, Oregon, United States, 97030 Oregon Dermatology & Research Center Portland, Oregon, United States, 97210 United States, Texas West Houston Clinical Research Services LLC Houston, Texas, United States, 77055 Texas Dermatology and Laser Specialists San Antonio, Texas, United States, 78218 United States, Utah Jordan Valley Dermatology Center West Jordan, Utah, United States, 84088 United States, Virginia PI Coor Clinical Research, LLC Burke, Virginia, United States, 22015 Collapse << |
| NCT01736475 | Hemophilia A | Phase 2 Phase 3 | Completed | - | - |
| NCT01579474 | Hepatitis C | Phase 3 | Completed | - | Japan ... more >> 1220.54.08104 Boehringer Ingelheim Investigational Site Chuo-ku, Chiba, Japan 1220.54.08118 Boehringer Ingelheim Investigational Site Chuo-ku, Kobe, Hyogo, Japan 1220.54.08108 Boehringer Ingelheim Investigational Site Fukui, Fukui, Japan 1220.54.08110 Boehringer Ingelheim Investigational Site Gifu, Gifu, Japan 1220.54.08105 Boehringer Ingelheim Investigational Site Itabashi-ku, Tokyo, Japan 1220.54.08112 Boehringer Ingelheim Investigational Site Izunokuni, Shizuoka, Japan 1220.54.08107 Boehringer Ingelheim Investigational Site Kanazawa, Ishikawa, Japan 1220.54.08120 Boehringer Ingelheim Investigational Site Kita-gun, Kagawa, Japan 1220.54.08109 Boehringer Ingelheim Investigational Site Kofu, Yamanashi, Japan 1220.54.08123 Boehringer Ingelheim Investigational Site Kurume, Fukuoka, Japan 1220.54.08121 Boehringer Ingelheim Investigational Site Mtsuyama, Ehime, Japan 1220.54.08113 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1220.54.08117 Boehringer Ingelheim Investigational Site Nishinomiya, Hyogo, Japan 1220.54.08111 Boehringer Ingelheim Investigational Site Ogaki, Gifu, Japan 1220.54.08124 Boehringer Ingelheim Investigational Site Oo mura, Nagasaki,, Japan 1220.54.08115 Boehringer Ingelheim Investigational Site Osaka, Osaka, Japan 1220.54.08116 Boehringer Ingelheim Investigational Site Osakasayama, Osaka, Japan 1220.54.08101 Boehringer Ingelheim Investigational Site Sapporo, Hokkaido, Japan 1220.54.08102 Boehringer Ingelheim Investigational Site Sendai, Miyagi, Japan 1220.54.08119 Boehringer Ingelheim Investigational Site Tanabe, Wakayama, Japan 1220.54.08106 Boehringer Ingelheim Investigational Site Toyama,Toyama, Japan 1220.54.08114 Boehringer Ingelheim Investigational Site Tsu, Mie, Japan 1220.54.08122 Boehringer Ingelheim Investigational Site Yahatanishi-ku, Kitakyusyu, Fukuoka, Japan 1220.54.08125 Boehringer Ingelheim Investigational Site Yamagata, Yamagata, Japan Collapse << |
| NCT00475865 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Vienna, Austria Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milan, Italy United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT00475865 | - | - | Completed | - | - |
| NCT00811395 | Multiple Sclerosis | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Austria Sanofi-Aventis Administrative Office Wien, Austria Canada Sanofi-Aventis Administrative Office Laval, Canada Germany Sanofi-Aventis Administrative Office Berlin, Germany Italy Sanofi-Aventis Administrative Office Milano, Italy Spain Sanofi-Aventis Administrative Office Barcelona, Spain United Kingdom Sanofi-Aventis Administrative Office Guildford, United Kingdom Collapse << |
| NCT01579474 | - | - | Completed | - | - |
| NCT00737464 | Anemia | Phase 4 | Completed | - | India ... more >> Indraprastha Apollo Hospitals New Delhi, Delhi, India, 110076 Manipal Institute of Nephrology and Urology; Nephrology Bangalore, India, 560017 Columbia Asia Medical Centre - Hebbal; Nephrology Bangalore, India, 560024 Tanker Foundation Chennai, India, 600 037 MIOT Institute of Nephrology ; Nephrology Chennai, India, 6000089 Madras Medical Mission Hospital Chennai, India, 600037 International Hospital Guwahati, India, 781005 Kamineni Hospitals; Nephrology Hyderabad, India, 500068 Nizam's Institute of Medical Sciences; Nephrology Hyderabad, India, 500082 Regency Hospital Ltd.; Nephrology Kanpur, India, 208005 Wockhardt Hospital and Kidney Institute; Nephrology Kolkata, India, 700029 Meerut Kidney Hospital Meerut, India, 250001 Silver Oaks Hospital; Nephrology Mohali, India, 160 063 Apex Kidney Care Mumbai, India, 400080 Lancelot Dialysis Center Mumbai, India, 400092 Fortis Flt. Lt. Rajan Dhall Hospital; Nephrology New Delhi, India, 110070 Ruby Hall Clinic Pune, India, 411 001 Collapse << |
| NCT03412552 | - | - | Completed | - | Egypt ... more >> Kasr Alainy medical school Cairo, Egypt, 12151 Collapse << |
| NCT00811395 | - | - | Completed | - | - |
| NCT00282243 | Liver Transplantation | Phase 2 | Completed | - | United States, California ... more >> Palo Alto, California, United States United States, Colorado Denver, Colorado, United States, 80262 United States, Maryland Baltimore, Maryland, United States, 21287 United States, Michigan Ann Arbor, Michigan, United States, 48109 United States, Minnesota Minneapolis, Minnesota, United States, 55455 Rochester, Minnesota, United States, 55905 United States, New York New York, New York, United States, 10029 United States, Ohio Cincinnati, Ohio, United States, 45267 United States, Texas Dallas, Texas, United States, 75246 United States, Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00737464 | - | - | Completed | - | - |
| NCT00282243 | - | - | Completed | - | - |
| NCT00116779 | Prostate Cancer | Phase 2 | Completed | - | - |
| NCT03552848 | Acute Respiratory Distress Syn... more >>drome (ARDS) Multiple Organ Failure Collapse << | Not Applicable | Not yet recruiting | September 1, 2018 | China, Fujian ... more >> Department of Cardiovascular Surgery Fuzhou, Fujian, China, 350001 Collapse << |
| NCT00116779 | - | - | Completed | - | - |
| NCT01437540 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01747330 | Pancreatic Exocrine Insufficie... more >>ncy Collapse << | Phase 3 | Completed | - | Russian Federation ... more >> Site Reference ID/Investigator# 80716 Barnaul, Russian Federation, 656019 Site Reference ID/Investigator# 80693 Moscow, Russian Federation, 117997 Site Reference ID/Investigator# 80698 Novosibirsk, Russian Federation, 630091 Site Reference ID/Investigator# 80713 Novosibirsk, Russian Federation Site Reference ID/Investigator# 80715 Orenburg, Russian Federation, 460000 Site Reference ID/Investigator# 80694 St. Petersburg, Russian Federation, 194156 Site Reference ID/Investigator# 80714 Tomsk, Russian Federation, 634050 Site Reference ID/Investigator# 80697 Voronezh, Russian Federation, 394036 Site Reference ID/Investigator# 80696 Yaroslavl, Russian Federation, 150003 Collapse << |
| NCT02617732 | Type 2 Diabetes Mellitus | Not Applicable | Unknown | February 2018 | China, Beijing ... more >> Peking Union Medical College Hospital traditional Chinese medicine department Not yet recruiting Beijing, Beijing, China, 100730 Contact: Qian Zhang, Doctor (86+)13810387388 qianqian0409@163.com Contact: Zhen Tang, Master (86+)18810941269 1225530172@qq.com Collapse << |
| NCT01747330 | - | - | Completed | - | - |
| NCT00494312 | Diabetes Mellitus ... more >> Liver Collapse << | Phase 4 | Completed | - | - |
| NCT03055169 | - | - | Completed | - | France ... more >> CHU Cote de Nâcre Caen, France Hôpital Roger Salengro, CHRU de Lille Lille, France CH Victor Provot Roubaix, France CHU Charles Nicolle Rouen, France CH Tourcoing Tourcoing, France Collapse << |
| NCT03714568 | Pharmacokinetic | Phase 1 | Recruiting | September 2019 | China, Hunan ... more >> The Third Hospital of Changsha Recruiting Changsha, Hunan, China, 410015 Principal Investigator: Li Xin, Doctor Collapse << |
| NCT00817609 | Stroke Acute ... more >>Ischemic Stroke Collapse << | Phase 2 Phase 3 | Unknown | - | China, Shanghai ... more >> Zhongshan Hospital of Fudan University Not yet recruiting Shanghai, Shanghai, China, 200032 Contact: Dingfang Cai, MD doctorcn@hotmail.com Principal Investigator: Zhan Sun, MD Sub-Investigator: Wenwei Li, MD Sub-Investigator: Junpeng Gao, MD, PhD Sub-Investigator: Chenling Yao, MD Sub-Investigator: Zhenju Song, MD Sub-Investigator: Chaoyang Tong, MD Sub-Investigator: Yuping Tang, MD, PhD Sub-Investigator: Chunfei Jiang, MD Collapse << |
| NCT01757184 | Cholesterol Ester Storage Dise... more >>ase (CESD) Lysosomal Acid Lipase Deficiency Collapse << | Phase 3 | Active, not recruiting | January 2019 | - |
| NCT01437540 | - | - | Completed | - | - |
| NCT01083186 | - | - | Completed | - | Greece ... more >> Site Reference ID/Investigator# 47002 Arta, Greece, 47100 Site Reference ID/Investigator# 43772 Athens, Greece, 115 21 Site Reference ID/Investigator# 27492 Athens, Greece, 115 27 Site Reference ID/Investigator# 43769 Athens, Greece, 115 27 Site Reference ID/Investigator# 38257 Athens, Greece, 11526 Site Reference ID/Investigator# 47003 Athens, Greece, 11526 Site Reference ID/Investigator# 27497 Athens, Greece, 11527 Site Reference ID/Investigator# 47004 Athens, Greece, 11527 Site Reference ID/Investigator# 27489 Athens, Greece, 11528 Site Reference ID/Investigator# 27495 Athens, Greece, 16673 Site Reference ID/Investigator# 43773 Athens, Greece, 17237 Site Reference ID/Investigator# 27498 Haidari, Athens, Greece, 12462 Site Reference ID/Investigator# 38259 Ioannina, Greece, 45500 Site Reference ID/Investigator# 22121 Larissa, Greece, 411 10 Site Reference ID/Investigator# 43771 Larissa, Greece, 41223 Site Reference ID/Investigator# 43767 Maroussi Athens, Greece, 15123 Site Reference ID/Investigator# 27496 Nikaia, Greece, 18454 Site Reference ID/Investigator# 43768 North Ionia, Athens, Greece, 142 33 Site Reference ID/Investigator# 27491 Piraeus, Greece, 18536 Site Reference ID/Investigator# 27494 Thessaloniki, Greece, 546 36 Site Reference ID/Investigator# 27493 Thessaloniki, Greece, 54636 Site Reference ID/Investigator# 38255 Thessaloniki, Greece, 56403 Site Reference ID/Investigator# 43770 Thessaloniki, Greece, 570 01 Site Reference ID/Investigator# 39839 Thessaloniki, Greece Collapse << |
| NCT01083186 | - | - | Completed | - | - |
| NCT00665925 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT00665626 | - | - | Completed | - | - |
| NCT00730639 | Metastatic Castration-resistan... more >>t Prostrate Cancer Renal Cell Carcinoma Metastatic Melanoma Non-small Cell Lung Cancer Collapse << | Phase 1 | Active, not recruiting | May 14, 2019 | United States, Arizona ... more >> Pinnacle Oncology Hematology Scottsdale, Arizona, United States, 85258 United States, Connecticut Yale University School Of Medicine New Haven, Connecticut, United States, 06520 United States, Florida H. Lee Moffitt Cancer Center & Research Institute Tampa, Florida, United States, 33612-9497 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21231 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana Farber Cancer Institute Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Michigan University Of Michigan Cancer Center Ann Arbor, Michigan, United States, 48109 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Carolina Biooncology Institute Huntersville, North Carolina, United States, 28078 United States, Ohio Christ Hospital Cincinnati, Ohio, United States, 45219 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 Vanderbilt-Ingram Cancer Ctr Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01757184 | - | - | Active, not recruiting | - | - |
| NCT02636634 | Brain Metastases ... more >> Growing Lesions After Radiosurgical Treatment Collapse << | Phase 3 | Completed | - | France ... more >> CHU Pitié-Salpêtrière - Service de Neurochirurgie du Pr Philippe Cornu - Babinsky Paris, France, 75013 Collapse << |
| NCT01280604 | Hyperlipidemias | Not Applicable | Completed | - | United States, Georgia ... more >> Kaiser Permanente of Georgia Atlanta, Georgia, United States, 30305 Collapse << |
| NCT01280604 | - | - | Completed | - | - |
| NCT00665626 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT00730639 | - | - | Active, not recruiting | - | - |
| NCT02981680 | Acute Kidney Injury ... more >> Coronary Artery Bypass Ischemic Preconditioning Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Shiraz University of Medical Sciences Shiraz, Fars, Iran, Islamic Republic of Collapse << |
| NCT02505334 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | Japan ... more >> Novo Nordisk Investigational Site Annaka-shi, Gunma, Japan, 379 0116 Novo Nordisk Investigational Site Chitose, Hokkaido, Japan, 066-0032 Novo Nordisk Investigational Site Chuo-ku Tokyo, Japan, 103-0027 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0002 Novo Nordisk Investigational Site Chuo-ku, Tokyo, Japan, 103 0027 Novo Nordisk Investigational Site Chuo-ku,, Japan, 104 0061 Novo Nordisk Investigational Site Fukuoka-shi, Fukuoka, Japan Novo Nordisk Investigational Site Higashiosaka-shi, Osaka, Japan Novo Nordisk Investigational Site Hokkaido, Japan, 078-8236 Novo Nordisk Investigational Site Ichikawa-shi, Chiba, Japan Novo Nordisk Investigational Site Iruma-shi, Saitama, Japan, 358 0011 Novo Nordisk Investigational Site Izumisano-shi, Japan, 598 0048 Novo Nordisk Investigational Site Kashiwara-shi, Osaka, Japan, 582 0005 Novo Nordisk Investigational Site Kawagoe-shi, Saitama, Japan, 350 0851 Novo Nordisk Investigational Site Kobe-shi, Hyogo, Japan Novo Nordisk Investigational Site Kumamoto-shi,Kumamoto, Japan, 862 0976 Novo Nordisk Investigational Site Mito-shi, Ibaraki, Japan, 310-0845 Novo Nordisk Investigational Site Mito-shi, Ibaraki, Japan Novo Nordisk Investigational Site Miyazaki-shi, Japan, 880 0034 Novo Nordisk Investigational Site Naka-shi, Ibaraki, Japan, 311 0113 Novo Nordisk Investigational Site Neyagawa-shi, Osaka, Japan Novo Nordisk Investigational Site Nishinomiya-shi, Hygo, Japan, 662 0971 Novo Nordisk Investigational Site Nishinomiya-shi, Hyogo, Japan, 663-8501 Novo Nordisk Investigational Site Oita-shi, Japan, 870 0039 Novo Nordisk Investigational Site Okawa-shi, Fukuoka, Japan, 831 0016 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 553 0003 Novo Nordisk Investigational Site Osaka-shi, Osaka, Japan, 5590012 Novo Nordisk Investigational Site Oyama-shi, Tochigi, Japan, 323 0022 Novo Nordisk Investigational Site Saga-shi,Saga, Japan, 849 0937 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060 0062 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 060-0001 Novo Nordisk Investigational Site Sapporo-shi, Hokkaido, Japan, 062 0007 Novo Nordisk Investigational Site Sappro-shi, Hokkaido, Japan, 060 8648 Novo Nordisk Investigational Site Shimotsuke-shi, Tochigi, Japan, 329 0433 Novo Nordisk Investigational Site Shinjuku-ku, Tokyo, Japan, 160-0008 Novo Nordisk Investigational Site Shizuoka-shi, Japan, 424 0853 Novo Nordisk Investigational Site Suita-shi, Osaka, Japan, 565-0853 Novo Nordisk Investigational Site Takatsuki-shi, Osaka, Japan, 569 1096 Novo Nordisk Investigational Site Tokyo, Japan, 103-0028 Novo Nordisk Investigational Site Tokyo, Japan, 105-8471 Novo Nordisk Investigational Site Tokyo, Japan, 123-0845 Novo Nordisk Investigational Site Tokyo, Japan, 144-0051 Novo Nordisk Investigational Site Tokyo, Japan, 169-0073 Novo Nordisk Investigational Site Tokyo, Japan, 181-0013 Novo Nordisk Investigational Site Yokohama, Kanagawa, Japan, 236-0004 Novo Nordisk Investigational Site Yokohama-shi Kanagawa, Japan, 232-0064 Novo Nordisk Investigational Site Yokohama-shi, Japan, 235 0045 Collapse << |
| NCT00140725 | Chronic Hepatitis B | Phase 3 | Completed | - | Hong Kong ... more >> GSK Investigational Site Shatin, Hong Kong Collapse << |
| NCT00665925 | - | - | Completed | - | - |
| NCT00135707 | Preeclampsia | Phase 3 | Completed | - | United States, Alabama ... more >> University of Alabama - Birmingham Birmingham, Alabama, United States, 35233 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Michigan Wayne State University Detroit, Michigan, United States, 48201 United States, New York Columbia University New York, New York, United States, 10032 United States, North Carolina University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599 Wake Forest University School of Medicine Winston-Salem, North Carolina, United States, 27157 United States, Ohio Case Western University Cleveland, Ohio, United States, 44109 Ohio State University Columbus, Ohio, United States, 43210 United States, Oregon Oregon Health and Sciences University Portland, Oregon, United States, 97239 United States, Pennsylvania Drexel University Philadelphia, Pennsylvania, United States, 19107 University of Pittsburgh Magee Womens Hospital Pittsburgh, Pennsylvania, United States, 15213 United States, Rhode Island Brown University Providence, Rhode Island, United States, 02905 United States, Texas University of Texas - Southwest Dallas, Texas, United States, 75235 University of Texas Medical Branch Galveston, Texas, United States, 77555 University of Texas - Houston Houston, Texas, United States, 77030 United States, Utah University of Utah Medical Center Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT00737711 | Anemia | Phase 4 | Completed | - | India ... more >> Ahmedabad, India, 380007 Bangalore, India, 560034 Chennai, India, 603103 Coimbatore, India, 641004 Gujarat, India, 387 001 Hyderabad, India, 500001 Kerala, India, 682017 Kolkata, India, 700099 Ludhiana, India Mumbai, India, 400026 Mumbai, India New Delhi, India, 110 060 New Delhi, India, 110017 New Delhi, India, 110026 Vellore, India, 632 004 Vishakpatnam, India, 530002 Collapse << |
| NCT00737711 | - | - | Completed | - | - |
| NCT02943707 | Liver Diseases | Phase 2 | Recruiting | October 2020 | China, Zhejiang ... more >> the First Affiliated Hospital of Wenzhou Medical University Recruiting Wenzhou, Zhejiang, China, 325000 Contact: yongping chen 8613505777281 13505777281@163.com Contact: lanman xu 8613587646315 13587646315@163.com Principal Investigator: yongping chen, MD Principal Investigator: lanman xu, PhD Principal Investigator: dazhi chen, MD Principal Investigator: minghua zheng, PhD Principal Investigator: keqing shi, MD Principal Investigator: yu huang, MD Principal Investigator: faling wu, MD Principal Investigator: ruicong chen, MD Principal Investigator: yunlei xiao, MD Collapse << |
| NCT02505334 | - | - | Completed | - | - |
| NCT01316939 | Crohn's Disease | Phase 3 | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT00724867 | - | - | Completed | - | - |
| NCT00724867 | Systemic Lupus Erythematosus | Phase 3 | Completed | - | - |
| NCT00560391 | Multiple Myeloma | Phase 1 | Completed | - | United States, Arizona ... more >> Mayo Clinic Arizona Scottsdale, Arizona, United States, 85259 Australia, Victoria Local Institution Prahran, Victoria, Australia, 3181 France Local Institution Toulouse Cedex 03, France, 31059 Local Institution Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT01316939 | - | - | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT01473524 | Primary Biliary Cirrhosis | Phase 3 | Active, not recruiting | June 2018 | - |
| NCT00135707 | - | - | Completed | - | - |
| NCT00708552 | - | - | Completed | - | - |
| NCT01898650 | Craniosynostosis | Not Applicable | Completed | - | United States, Missouri ... more >> St. Louis Children's Hospital Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT02709681 | - | - | Completed | - | Italy ... more >> Ospedali Civili Riuniti di Sciacca Sciacca, Agrigento, Italy, 92019 Ospedale Vittorio Emanuele III Gela, Caltanisetta, Italy, 93012 Azienda Ospedaliero-Universitaria Bari, Italy U.O. Oncoematologia Pediatrica Ospedali Civili Brescia, Italy, 25100 Ospedale "A. Perrino" Brindisi, Italy, 72100 Azienda Ospedaliera Universitaria Di Cagliari Cagliari, Italy, 09121 Ospedale Vittorio Emanuele Catania, Italy, 95123 ARNAS "Garibaldi" Catania, Italy, 95124 University of Catania Catania, Italy, 95124 A.O. "Pugliese-Ciaccio" Catanzaro, Italy, 88100 Osp.San Giovanni Di Dio Crotone, Italy Azienda Ospedaliero Universitaria Meyer Firenze, Italy, 50139 E.O. Ospedali Galliera Genova, Italy, 16128 Università degli Studi di Milano Milano, Italy, 20122 Azienda Ospedaliero-Universitaria di Modena - Policlinico Modena, Italy, 41124 Clinica pediatrica Monza S. Gerardo Monza, Italy, 20052 Azienda Ospedaliera San Gerardo di Monza Monza, Italy, 20900 Aorn A. Cardarelli Napoli, Italy Clinica di Onco-Ematologia Pediatrica, Università di Padova Padova, Italy, 35128 A.R.N.A.S. "Civico" Palermo, Italy, 90100 Azienda Ospedaliera Ospedali Riuniti Villa Sofia Cervello Palermo, Italy, 90146 Azienda Ospedaliero-Universitaria di Parma Parma, Italy, 43126 Policlinico San Matteo Pavia, Italy, 27100 Centro Emofilia e Medicina Trasfusionale - Pres. Ospedaliero Ravenna, Italy, 48121 A. O. Bianchi Melacrino Morelli Reggio Calabria, Italy, 89100 Ospedale S. Eugenio - FF UOSD DH Talassemici Roma, Italy, 00144 Università Cattolica del Sacro Cuore - Policlinico A.Gemelli Roma, Italy, 00168 U.O.S. Talassemia P.O. Umberto I° Siracusa, Italy, 96100 Università degli Suidi di Torino Torino, Italy, 90146 Policlinico G.B. Rossi Verona, Italy, 37122 Collapse << |
| NCT03462121 | Mild to Moderate Dementia Due ... more >>to Alzheimer's Disease With or Without Coexisting Cerebrovascular Disease Collapse << | Phase 2 | Recruiting | April 30, 2019 | Canada, Ontario ... more >> Kawartha Centre - Redefining Healthy Aging Recruiting Peterborough, Ontario, Canada, K9H 2P4 Contact: Jennifer K Ingram, MD FRCPC 705-749-3906 Contact: Krista Gillis 05-930-2667 (24 Hr.) Principal Investigator: Jennifer K Ingram, MD Toronto Memory Program Recruiting Toronto, Ontario, Canada, M3B 2S7 Principal Investigator: Sharon Cohen, MD Gerontion Research Inc., The Center for Memory & Aging Recruiting Toronto, Ontario, Canada, M4G 3E8 Principal Investigator: Giovanni Marotta, MD Canada, Quebec Recherches Neuro-Hippocampe Inc. d/b/a Clinique de la Memoire de l'Outaouais Recruiting Gatineau, Quebec, Canada, J8T 8J1 Principal Investigator: Richard Bergeron, MD Diex Recherche Sherbrooke Inc. Recruiting Sherbrooke, Quebec, Canada, J1L 0H8 Principal Investigator: Marcel Germain, MD Collapse << |
| NCT01473524 | - | - | Active, not recruiting | - | - |
| NCT00708552 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT00560391 | - | - | Completed | - | - |
| NCT00345605 | Argininosuccinic Aciduria ... more >> Amino Acid Metabolism, Inborn Errors Urea Cycle Disorders Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00345605 | - | - | Completed | - | - |
| NCT02706678 | Renal Transplant Recipients | Phase 4 | Completed | - | China ... more >> Beijing, China Changchun, China Chengdu, China Guangzhou, China Nanjing, China Nanning, China Shanghai, China Shenyang, China Wuhan, China Collapse << |
| NCT00310375 | Epilepsy | Phase 3 | Completed | - | - |
| NCT01718730 | - | - | Active, not recruiting | August 2019 | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03439280 | Relapsed/Refractory ... more >> Multiple Myeloma Collapse << | Phase 1 Phase 2 | Recruiting | October 28, 2020 | United States, California ... more >> City of Hope - Duarte Recruiting Duarte, California, United States, 91010-3012 United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 United States, New York Mount Sinai Hospital-The Donald H. Ruttenberg Cancer Treatment Center Recruiting New York, New York, United States, 10011 Weill Cornell Medical Center, Div. of Hematology Medical Oncology Recruiting New York, New York, United States, 10021-4635 United States, Oregon Oregon Health & Science University Knight Cancer Institute Recruiting Portland, Oregon, United States, 97239 United States, Texas The University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Texas Oncology - San Antonio Northwest Recruiting San Antonio, Texas, United States, 78217 United States, Virginia Virginia Cancer Specialists - Fairfax Office Recruiting Fairfax, Virginia, United States, 22031 United States, Wisconsin Medical College of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00310375 | - | - | Completed | - | - |
| NCT01495000 | Osteoporosis, Postmenopausal | Phase 3 | Completed | - | India ... more >> GSK Investigational Site Ahmedabad, India, 380015 GSK Investigational Site Bangalore, India, 560043 GSK Investigational Site Bangalore, India, 560052 GSK Investigational Site Bangalore, India, 560054 GSK Investigational Site Delhi, India, 110060 GSK Investigational Site Mangalore, India, 575002 GSK Investigational Site Nagpur, India, 440010 GSK Investigational Site Nagpur, India, 440012 GSK Investigational Site Pune, India, 411030 GSK Investigational Site Trivandrum, India, 695011 GSK Investigational Site Vadodra, India, 390007 Collapse << |
| NCT02735694 | Sleep Apnea Syndromes | Phase 1 | Terminated(Analysis of initial... more >> sample shows negative results.) Collapse << | November 2016 | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2N 4N1 Canada, Manitoba Sleep Disorders Centre Winnipeg, Manitoba, Canada, R3C 1A2 Collapse << |
| NCT00546715 | Chronic Hepatitis C | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Advanced Clinical Research Institute Anaheim, California, United States, 92801 United States, Florida Orlando Clinical Research Center Orlando, Florida, United States, 32809 United States, Maryland Parexel International Corporation Baltimore, Maryland, United States, 21225 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New Jersey Bristol-Myers Squibb Clinical Pharmacology Unit Hamilton, New Jersey, United States, 08690 United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 United States, Virginia University Of Virginia Digestive Health Center Of Excellence Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00964041 | Schizophrenia | Phase 4 | Withdrawn | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00920426 | - | - | Completed | - | - |
| NCT01495000 | - | - | Completed | - | - |
| NCT03639701 | Mitochondrial DNA Depletion Sy... more >>ndrome 2 Myopathic Type Thymidine Kinase 2 Deficiency Collapse << | Phase 1 Phase 2 | Enrolling by invitation | April 1, 2024 | United States, New York ... more >> Columbia University New York, New York, United States, 10032 Collapse << |
| NCT00945282 | - | - | Completed | - | - |
| NCT00546715 | - | - | Completed | - | - |
| NCT00920426 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Fort Lauderdale, Florida, United States, 33308 GSK Investigational Site Orlando, Florida, United States, 32803 GSK Investigational Site Vero Beach, Florida, United States, 32960 United States, North Carolina GSK Investigational Site Charlotte, North Carolina, United States, 28209 Collapse << |
| NCT00296504 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Completed | - | United States, California ... more >> GSK Investigational Site Fountain Valley, California, United States, 92708 GSK Investigational Site San Francisco, California, United States, 94115-1931 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80262 United States, Florida GSK Investigational Site Fort Lauderdale, Florida, United States, 33145 GSK Investigational Site Orlando, Florida, United States, 32804 GSK Investigational Site Orlando, Florida, United States, 32806 GSK Investigational Site Sarasota, Florida, United States, 34239 GSK Investigational Site Tampa, Florida, United States, 33614 United States, New York GSK Investigational Site Manhasset, New York, United States, 11030 United States, Texas GSK Investigational Site Galveston, Texas, United States, 77555-1188 Brazil GSK Investigational Site Campinas, São Paulo, Brazil, 13083970 Chile GSK Investigational Site Santiago, Región Metro De Santiago, Chile France GSK Investigational Site Le Kremlin Bicêtre Cedex, France, 94275 GSK Investigational Site Marseille, France, 13005 GSK Investigational Site Paris Cedex 10, France, 75475 GSK Investigational Site Paris Cedex 20, France, 75970 GSK Investigational Site Vandoeuvre Les Nancy Cedex, France, 54511 GSK Investigational Site Villejuif Cedex, France, 94805 Italy GSK Investigational Site Genova, Liguria, Italy, 16128 Portugal GSK Investigational Site Coimbra, Portugal, 3000-075 Spain GSK Investigational Site Badajoz, Spain, 6080 GSK Investigational Site Barcelona, Spain, 08036 United Kingdom GSK Investigational Site London, United Kingdom, NW3 2QG GSK Investigational Site London, United Kingdom, SE1 7EH Collapse << |
| NCT00945282 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 2 | Completed | - | Argentina ... more >> GSK Investigational Site Buenos Aires, Argentina, B1602DBG Collapse << |
| NCT00819013 | - | - | Completed | - | - |
| NCT00296504 | - | - | Completed | - | - |
| NCT02833584 | Dengue Transa... more >>minitis Collapse << | Not Applicable | Completed | - | Thailand ... more >> Phramongkutklao College of Medicine and Hospital Bangkok, Thailand, 10400 Anandamahidol Hospital Lopburi, Thailand, 15000 Fort Adisorn Hospital Saraburi, Thailand, 18000 Collapse << |
| NCT00819013 | Influenza | Phase 1 | Completed | - | United States, Florida ... more >> Miami Research Associates Miami, Florida, United States, 33143 United States, Kansas Johnson County Clin-Trials Lenexa, Kansas, United States, 66219 United States, Washington Northwest Kinetics Tacoma, Washington, United States, 98418 Collapse << |
| NCT02469298 | Virus Diseases | Phase 2 | Completed | - | - |
| NCT02061540 | - | - | Completed | - | - |
| NCT02061540 | Primary Sclerosing Cholangitis... more >> (PSC) Collapse << | Phase 2 | Completed | - | United States, California ... more >> Scripps Clinic : La Jolla, California, United States, 92037 University of California at Davis Sacramento, California, United States, 95817 United States, Colorado University of Colorado Aurora, Colorado, United States, 80045 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, North Carolina Duke University Durham, North Carolina, United States, 27710 Canada, Alberta University of Calgary Liver Unit Calgary, Alberta, Canada, T2N 4Z6 Canada, Ontario University Health Network, Toronto Western Hospital Toronto, Ontario, Canada, M5T 2S8 United Kingdom University of Birmingham Birmingham, England, United Kingdom, B15 2TT Royal Free Hospital London, United Kingdom, NW3 2QG Collapse << |
| NCT00996840 | Lung Injury, Acute | Phase 2 | Completed | - | United States, Kentucky ... more >> GSK Investigational Site Lexington, Kentucky, United States, 40536-0293 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 GSK Investigational Site Winston-Salem, North Carolina, United States, 27157 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37232-7110 Collapse << |
| NCT02756208 | HIV Infection | Phase 1 | Completed | - | United States, Maryland ... more >> University of Maryland, Institute of Human Virology Baltimore, Maryland, United States, 21201 Collapse << |
| NCT02469298 | - | - | Completed | - | - |
| NCT00996840 | - | - | Completed | - | - |
| NCT00664521 | Rheumatoid Arthritis | Phase 2 | Completed | - | France ... more >> Research Site Nice, France Research Site Paris, France Research Site Strasbourg, France Netherlands Research Site Amsterdam, Netherlands Sweden Research Site Malmö, Sweden Research Site Stockholm, Sweden United Kingdom Research Site Newcastle, United Kingdom Research Site Norwich, United Kingdom Collapse << |
| NCT03503708 | Alcoholic Liver Disease | Not Applicable | Not yet recruiting | November 30, 2028 | India ... more >> Mazumdar Shaw Medical Centre Not yet recruiting Bangalore, Karnataka, India, 560099 Contact: Alben Sigamani, MD 8884431444 alben.sigamani.dr@narayanahealth.org Contact: Sanjaya Chauhan, PharmD 9611252350 ext Chauhan drsanjayachauhan49@gmail.com Collapse << |
| NCT00664521 | - | - | Completed | - | - |
| NCT02446925 | Hepatocellular Carcinoma | Not Applicable | Suspended(Insufficient number ... more >>of subjects meeting inclusion criteria) Collapse << | November 2018 | - |
| NCT01513902 | - | - | Completed | - | - |
| NCT00770445 | Diabetes Mellitus | Phase 4 | Completed | - | Germany ... more >> Lichtenfels, Bavaria, Germany Frankfurt, Hessen, Germany Kassel, Hessen, Germany Wiesbaden, Hessen, Germany Bad Oeynhausen, North Rhine-Westphalia, Germany Dinslaken, North Rhine-Westphalia, Germany Duisburg, North Rhine-Westphalia, Germany Essen, North Rhine-Westphalia, Germany Münster, North Rhine-Westphalia, Germany Wuppertal, North Rhine-Westphalia, Germany Mainz, Rheinland-Pfalz, Germany Diez, Rhineland-Palatinate, Germany Landau, Rhineland-Palatinate, Germany Dresden, Saxony, Germany Leipzig, Saxony, Germany Jena, Thüringen, Germany Berlin, Germany Hamburg, Germany Collapse << |
| NCT03308565 | Spinal Cord Injuries ... more >> Paralysis Collapse << | Phase 1 | Recruiting | November 2023 | United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01513902 | Juvenile Idiopathic Arthritis | Phase 1 | Completed | - | United States, Minnesota ... more >> Explorer Clinic, University of Minnesota Children's Hospital Minneapolis, Minnesota, United States, 55454 Clinical and Translational Science Institute Masonic Clinical Research Unit (Administration Only) Minneapolis, Minnesota, United States, 55455 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Oregon Randall Children's Hospital at Legacy Emanuel Portland, Oregon, United States, 97227 Germany PRI - Pediatric Rheumatology Research Institute GmbH Bad Bramstedt, Germany, 24576 Hamburger Zentrum fuer Kinder-und Jugendrheumatologie SchoenKlinik Hamburg Eilbek Hamburg, Germany, 22081 Asklepios Klinik Sankt Augustin Gmbh St. Augustin, Germany, 53757 Poland Wojewodzki Specjalistyczny Szpital Dzieciecy im. Sw. Ludwika w Krakowie Krakow, Poland, 31-503 Klinika Kardiologii i Reumatologii Dzieciecej Lodz, Poland, 91-738 Slovakia Narodny ustav reumatickych chorob Piestany, Slovakia, 921 12 Collapse << |
| NCT02533544 | - | - | Recruiting | October 2018 | Korea, Republic of ... more >> The Catholic University of Korea, Daejeon St.Mary's Hosptial Recruiting Junggu, Daejeon, Korea, Republic of Contact: Myeong Jun Song 8242-220-9291 Collapse << |
| NCT01785615 | Metabolic Syndrome | Phase 1 Phase 2 | Completed | - | United States, New York ... more >> University of Rochester Medical Center Rochester, New York, United States, 14642 Collapse << |
| NCT03385395 | Alpha 1-Antitrypsin Deficiency | Phase 2 | Withdrawn(IND has been closed.... more >>) Collapse << | December 2019 | - |
| NCT01785615 | - | - | Completed | - | - |
| NCT03517254 | Anterior Cruciate Ligament Rec... more >>onstruction Collapse << | Not Applicable | Recruiting | April 2019 | Mexico ... more >> Instituto Nacional de Rehabilitación Recruiting Mexico City, Mexico, 14389 Contact: Sandra Hernández Valencia, MSc +5255 5999-1000 ext 15212 sandrahv710@gmail.com Contact: Saúl León Ballesteros, MD, MSc +5255 5999-1000 ext 15212 saulno1@yahoo.com Principal Investigator: Sandra Hernández Valencia, MSc,PhD (c) Principal Investigator: Saúl León, MD,MSc Sub-Investigator: Laura Moreno Altamirano, MD,MSc,PhD Sub-Investigator: Patricia Clark Peralta, MD,MSc,PhD Sub-Investigator: Guadalupe Gomez Pineda, MD Sub-Investigator: Juan Manuel Mejía Aranguré, MD,MSc,PhD Sub-Investigator: Rebeca Salas Romero, MSc Sub-Investigator: Andrea Pegueros Pérez, MSc Collapse << |
| NCT01156311 | Relapsing-Remitting Multiple S... more >>clerosis Multiple Sclerosis Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Research Site Gilbert, Arizona, United States Research Site Phoenix, Arizona, United States United States, Connecticut Research Site Danbury, Connecticut, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Indiana Research Site Fort Wayne, Indiana, United States United States, Maryland Research Site Baltimore, Maryland, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Minnesota Research Site Golden Valley, Minnesota, United States United States, New Jersey Research Site Teaneck, New Jersey, United States United States, New York Research Site Patchogue, New York, United States United States, Ohio Research Site Cleveland, Ohio, United States Research Site Dayton, Ohio, United States United States, Oregon Research Site Portland, Oregon, United States United States, Tennessee Research Site Cordova, Tennessee, United States Research Site Franklin, Tennessee, United States United States, Wisconsin Research Site Milwaukee, Wisconsin, United States Collapse << |
| NCT02612051 | Idiopathic Pulmonary Fibrosis | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, SE1 1YR Collapse << |
| NCT01948453 | Coronary Artery Disease | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Rajaei Cardiovascular, Medical & Research Center Tehran, Iran, Islamic Republic of Collapse << |
| NCT00846963 | Cholestasis | Phase 2 Phase 3 | Completed | - | Canada, Quebec ... more >> CHU Sainte-Justine Montréal, Quebec, Canada, H3T 1C5 Collapse << |
| NCT01156311 | - | - | Completed | - | - |
| NCT00468286 | Prostate Cancer | Phase 2 | Completed | - | - |
| NCT01318525 | Liver Failure, Acute | Phase 2 | Unknown | September 2012 | France ... more >> CHU de Besançon Not yet recruiting Besançon Cedex, France, 25030 Principal Investigator: Vincent Di Martino CHU Clermont-Ferrand Not yet recruiting Clermont-Ferrand Cedex 1, France, 63003 Principal Investigator: Armand Abergel Hôpital Beaujon Recruiting Clichy, France, 92110 Principal Investigator: François Durand CHU de Grenoble Recruiting Grenoble Cedex 9, France, 38043 Principal Investigator: Vincent Leroy Hôpital Claude Huriez Not yet recruiting Lille cedex, France, 59037 Principal Investigator: Philippe Mathurin Hôpital Croix-Rousse Recruiting Lyon, France, 69004 Principal Investigator: Si Nafa Si Ahmed Hôpital Conception Not yet recruiting Marseille Cedex 5, France, 13385 Principal Investigator: Danielle Botta Fridlund Hôpital Saint-Eloi Recruiting Montpellier Cedex 5, France, 34295 Principal Investigator: Dominique Larrey Hôpital de l'Archet 2 Not yet recruiting Nice, France, 06202 Principal Investigator: Jean Gugenheim Hôpital Saint Antoine Recruiting Paris Cedex 12, France, 75571 Principal Investigator: Nicolas Carbonell Hôpital La Pitié Salpétrière Recruiting Paris Cedex 13, France, 75651 Principal Investigator: Marika Rudler Centre Hépatobiliaire Paul Brousse Recruiting Villejuif Cedex, France, 94804 Principal Investigator: Didier Samuel Collapse << |
| NCT03395262 | Weight Loss | Not Applicable | Active, not recruiting | December 31, 2018 | United States, Texas ... more >> Exercise & Sport Nutrition Lab - Human Clinical Research Facility College Station, Texas, United States, 77845 Collapse << |
| NCT00468286 | - | - | Completed | - | - |
| NCT01438606 | HIV Infections | Phase 1 | Completed | - | United States, California ... more >> Bridge HIV CRS San Francisco, California, United States, 94143 United States, Georgia The Hope Clinic of the Emory Vaccine Center CRS Decatur, Georgia, United States, 30030 United States, Pennsylvania Penn Prevention CRS Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01578889 | HIV Infections | Phase 1 | Completed | - | United States, New York ... more >> Columbia P&S CRS New York, New York, United States, 10032-3732 New York Blood Center CRS New York, New York, United States, 10065 University of Rochester Vaccines to Prevent HIV Infection CRS Rochester, New York, United States, 14642 United States, Pennsylvania Penn Prevention CRS Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232-2582 Collapse << |
| NCT03613740 | Metabolic Syndrome | Phase 2 | Not yet recruiting | September 2020 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00116753 | Prostate Cancer | Phase 2 | Completed | - | - |
| NCT00116753 | - | - | Completed | - | - |
| NCT03687476 | Niemann-Pick Disease, Type C | Phase 2 | Not yet recruiting | October 30, 2022 | - |
| NCT01970449 | HIV Infections | Phase 1 | Withdrawn | - | - |
| NCT01041677 | Obesity Nutri... more >>tional and Metabolic Diseases Metabolic Diseases Nutrition Disorders Overweight Collapse << | Phase 2 | Completed | - | Finland ... more >> Helsinki, Finland Netherlands Zuidlaren, Netherlands Sweden Göteborg, Sweden Collapse << |
| NCT00261443 | Bipolar Disorder | Phase 4 | Completed | - | - |
| NCT02759315 | - | - | Terminated(The study was termi... more >>nated based on review of Phase 2 efficacy data) Collapse << | - | - |
| NCT03372083 | Iron Over Load | Phase 4 | Recruiting | November 14, 2019 | Italy ... more >> Novartis Investigative Site Recruiting Cona, FE, Italy, 44100 Novartis Investigative Site Recruiting Cagliari, ITA, Italy, 09121 Novartis Investigative Site Recruiting Napoli, Italy, 80138 Lebanon Novartis Investigative Site Recruiting Hazmiyeh, Beirut, Lebanon, PO BOX 213 Oman Novartis Investigative Site Recruiting Muscat, Oman, 123 Russian Federation Novartis Investigative Site Recruiting Moscow, Russian Federation, 117198 Thailand Novartis Investigative Site Recruiting Bangkok noi, Bangkok, Thailand, 10700 Novartis Investigative Site Completed Muang, Chiangmai, Thailand, 50200 United Arab Emirates Novartis Investigative Site Recruiting Dubai, United Arab Emirates, 9115 Collapse << |
| NCT03653091 | Type 2 Diabetes | Not Applicable | Recruiting | February 2020 | United States, Florida ... more >> Florida Hospital / TRANSLATIONAL RESEARCH INSTITUTE FOR METABOLISM AND DIABETES (TRI) Recruiting Orlando, Florida, United States, 32804 Contact: Quianna Stokes-Washington 407-303-7100 TRI@flhosp.org Principal Investigator: Steven Smith, MD Sub-Investigator: Robert Hawes, MD United States, Pennsylvania University of Pennsylvania - Penn Metabolic Medicine Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Anastassia Amaro, MD 215-294-9525 Anastassia.Amaro@uphs.upenn.edu Contact: Gregory G. Ginsberg, MD 215-349-8222 Gregory.Ginsberg@uphs.upenn.edu Principal Investigator: Anastassia Amaro, MD Sub-Investigator: Gregory G. Ginsberg, MD Collapse << |
| NCT01925287 | Pharmacokinetics of Novel Curc... more >>umin Formulations Safety of Novel Curcumin Formulations Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Germany, 70599 Collapse << |
| NCT02759315 | Hepatitis C, Chronic | Phase 2 | Terminated(The study was termi... more >>nated based on review of Phase 2 efficacy data) Collapse << | - | - |
| NCT01910428 | Acute Lymphoblastic Leukemia ... more >> Lymphoblastic Lymphoma Collapse << | Phase 1 | Terminated(Sponsor decision ba... more >>sed off site/PI feedback about) Collapse << | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 United States, Maryland University of Maryland, Greenebaum Comprehensive Cancer Center Baltimore, Maryland, United States, 21201 United States, New York Monter Cancer Center Lake Success, New York, United States, 11042 United States, North Carolina Duke University Durham, North Carolina, United States, 27705 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 Collapse << |
| NCT00548964 | Major Depressive Disorder | Phase 1 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT03771729 | CKD | Early Phase 1 | Not yet recruiting | June 15, 2019 | - |
| NCT03636035 | Mild Knee Osteoarthritis | Not Applicable | Not yet recruiting | December 31, 2019 | - |
| NCT02217943 | Diabetes Obes... more >>ity Collapse << | Not Applicable | Active, not recruiting | July 1, 2021 | China ... more >> Beijing Shijitan Hospital Beijing, China China-Japan Union Hospital of Jilin University Changchun, China Guangzhou Overseas Chinese Hospital Guangzhou, China Jiangsu Province Hospital Jiangsu, China Shanghai East Hospital Shanghai, China Shanghai No. 6 People's Hospital Shanghai, China Collapse << |
| NCT03504033 | Liver Function After Partial L... more >>iver Resection Collapse << | Phase 4 | Not yet recruiting | April 2020 | Germany ... more >> University Hospital RWTH Aachen University, Department of Anesthesiology Not yet recruiting Aachen, Germany, 52074 Contact: Sebastian Ziemann, MD +49 241 80 88179 sziemann@ukaachen.de Contact: Ana Kowark, MD +49 241 80 88179 akowark@ukaachen.de Principal Investigator: Ana Kowark, MD Sub-Investigator: Rolf Rossaint, MD Sub-Investigator: Mark Coburn, MD Sub-Investigator: Julia Van Waesberghe, MD Sub-Investigator: Sebastian Ziemann, MD Collapse << |
| NCT00261443 | - | - | Completed | - | - |
| NCT03052062 | Obese | Phase 1 Phase 2 | Completed | - | France ... more >> Centre d'Investigation Clinique Clermont-Ferrand, France, 63000 Collapse << |
| NCT00793793 | Hepatitis C, Chronic | Phase 1 | Completed | - | United States, California ... more >> 1220.2.10 Boehringer Ingelheim Investigational Site San Francisco, California, United States 1220.2.15 Boehringer Ingelheim Investigational Site San Francisco, California, United States United States, Maryland 1220.2.17 Boehringer Ingelheim Investigational Site Baltimore, Maryland, United States United States, New York 1220.2.11 Boehringer Ingelheim Investigational Site New York, New York, United States 1220.2.12 Boehringer Ingelheim Investigational Site New York, New York, United States United States, Texas 1220.2.14 Boehringer Ingelheim Investigational Site Austin, Texas, United States France 1220.2.3304A Boehringer Ingelheim Investigational Site Lyon, France 1220.2.3303A Boehringer Ingelheim Investigational Site Marseille, France 1220.2.3301A Boehringer Ingelheim Investigational Site Paris, France 1220.2.3302A Boehringer Ingelheim Investigational Site Paris, France Germany 1220.2.49002 Boehringer Ingelheim Investigational Site Berlin, Germany 1220.2.49005 Boehringer Ingelheim Investigational Site Düsseldorf, Germany 1220.2.49006 Boehringer Ingelheim Investigational Site Hannover, Germany 1220.2.49004 Boehringer Ingelheim Investigational Site Kiel, Germany 1220.2.49003 Boehringer Ingelheim Investigational Site Mainz, Germany Spain 1220.2.34001 Boehringer Ingelheim Investigational Site Madrid, Spain Collapse << |
| NCT00793793 | - | - | Completed | - | - |
| NCT00703391 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT02118766 | - | - | Completed | - | - |
| NCT00703391 | - | - | Completed | - | - |
| NCT03569852 | Fasting Inter... more >>mittent Fasting Athletic Performance Cardiovascular Risk Factor Resting Energy Expenditure Collapse << | Not Applicable | Recruiting | July 15, 2020 | United States, California ... more >> UC Davis, Western Human Nutrition Research Center Recruiting Davis, California, United States, 95616 Collapse << |
| NCT02118766 | Dermatitis, Atopic | Phase 3 | Completed | - | United States, Georgia ... more >> Anacor Investigational Site Stockbridge, Georgia, United States Collapse << |
| NCT03545581 | Hereditary Fructose Intoleranc... more >>e Collapse << | Not Applicable | Recruiting | April 1, 2019 | Belgium ... more >> Guillaume Debray Recruiting Liège, Belgium Contact: Guillaume F Debray, MD +3242249057 fg.debray@chu.ulg.ac.be Collapse << |
| NCT03112083 | Adverse Drug Event ... more >> Side Effects of Drugs Collapse << | Not Applicable | Completed | - | Finland ... more >> Oy Medfiles Ltd Kuopio, Finland, 70701 Collapse << |
| NCT02308111 | Liver Cirrhosis, Biliary | Phase 4 | Recruiting | April 2025 | - |
| NCT01231321 | Rheumatoid Arthritis | Phase 3 | Completed | - | Russian Federation ... more >> Site Ref # / Investigator 17682 Kazan, Russian Federation, 420095 Site Ref # / Investigator 7417 Moscow, Russian Federation, 115522 Site Ref # / Investigator 17681 Moscow, Russian Federation, 117513 Site Ref # / Investigator 7401 Moscow, Russian Federation, 119049 Site Ref # / Investigator 18081 Saint Petersburg, Russian Federation, 191015 Collapse << |
| NCT00585546 | Heart Failure ... more >> Dilated Cardiomyopathy Collapse << | Phase 1 | Terminated(No longer could obt... more >>ain clenbuterol) Collapse << | - | United States, District of Col... more >>umbia Georgetown Hospital Washington, D.C., District of Columbia, United States, 20010 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Michigan University of Michigan Health System Ann Arbor, Michigan, United States, 48109 United States, New York Montefiore Medical Center The Bronx, New York, United States, 10467 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19014 United States, Texas Texas Heart Institute Houston, Texas, United States, 77030 Collapse << |
| NCT03061721 | Non-Alcoholic Fatty Liver Dise... more >>ase Nonalcoholic Steatohepatitis Collapse << | Phase 2 | Recruiting | April 2019 | United States, California ... more >> Precision Research Recruiting Chula Vista, California, United States, 91910 Contact: Research Site Cedars-Sinai Medical Center Recruiting Los Angeles, California, United States, 90048 Contact: Research Site Catalina Research Institute Recruiting Montclair, California, United States, 91763 Contact: Joe Calleros California liver research institute Recruiting Pasadena, California, United States, 91105 Contact: Stephanie Tang Precision Research Recruiting San Diego, California, United States, 92114 Contact: Research Site Medical Associates Research Group Recruiting San Diego, California, United States, 92123 Contact: Tina Delrosa United States, Florida University of Florida Recruiting Gainesville, Florida, United States, 32601 Contact: Research Site Schiff Center for Liver Diseases/University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Diane Sabogal Avail Clinical Research Recruiting Orange City, Florida, United States, 32763 Contact: Samantha Watts United States, Indiana Indiana University Recruiting Indianapolis, Indiana, United States, 46290 Contact: Research Site United States, Maryland Mercy Medical Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Research Site United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109-5362 Contact: Elizabeth Wu Henry Ford Hospital Recruiting Detroit, Michigan, United States, 48202 Contact: Research Site United States, North Carolina Wake Internal Medicine Withdrawn Raleigh, North Carolina, United States, 27612 United States, Ohio Awasty Research Network, LLC Recruiting Marion, Ohio, United States, 43302 Contact: Elisia Baldwin United States, Pennsylvania Einstein Medical Center Recruiting Philadelphia, Pennsylvania, United States, 19141 Contact: Stacey J Carmody United States, Tennessee Gastro One Recruiting Germantown, Tennessee, United States, 38138 Contact: Laurie Martin AIG Research Recruiting Hermitage, Tennessee, United States, 37076 Contact: Research Site United States, Texas Liver Consultants Recruiting Dallas, Texas, United States, 75246 Contact: Research Site The Liver Institute Recruiting San Antonio, Texas, United States, 78215 Contact: Research Site United States, Washington Swedish Medical Center Recruiting Seattle, Washington, United States, 89104 Contact: Research Site Collapse << |
| NCT01231321 | - | - | Completed | - | - |
| NCT00413699 | Arthritis, Rheumatoid | Phase 3 | Completed | - | - |
| NCT02141659 | Hormone Treatment-naïve Partic... more >>ipants With Prostate Cancer Collapse << | Phase 1 | Completed | - | Japan ... more >> Sakura-shi, Chiba, Japan Maebashi-shi, Gunma, Japan Sapporo-shi, Hokkaido, Japan Kanazawa-shi, Ishikawa, Japan Yokohama-shi, Kanagawa, Japan Chiyoda-ku, Tokyo, Japan Collapse << |
| NCT00413699 | - | - | Completed | - | - |
| NCT03764670 | - | - | Not yet recruiting | December 2020 | Taiwan ... more >> National Taiwan University Hospital Not yet recruiting Taipei, Taiwan Contact: Meng-Kun Tsai, Professor +886-972651461 mengkuntsai@ntu.edu.tw Collapse << |
| NCT03706690 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Recruiting | January 31, 2025 | - |
| NCT02141659 | - | - | Completed | - | - |
| NCT03318809 | Heart Failure | Phase 1 | Completed | - | United States, California ... more >> Research Site Tustin, California, United States, 92780 United States, Florida Research Site Orlando, Florida, United States, 32809 United States, Minnesota Research Site Minneapolis, Minnesota, United States, 55404 United States, Texas Research Site San Antonio, Texas, United States, 78215 Collapse << |
| NCT03771170 | Peritonitis I... more >>ntoxication Collapse << | Phase 4 | Recruiting | January 31, 2020 | Georgia ... more >> "Unimedi Adjara" LLC Recruiting Batumi, Georgia Contact: Kahaber Kishibadje, MD Principal Investigator: Kahaber Kishibadje, MD Kutaisi Referral Hospital Recruiting Kutaisi, Georgia Contact: David Tevdoradze Principal Investigator: David Tevdoradze JSC EVEX/LTD Traumatology Recruiting Tbilisi, Georgia Contact: Jano Vashadze, MD Principal Investigator: Jano Vashadze, MD Moldova, Republic of Institute of Ambulance Recruiting Chisinau, Moldova, Republic of Contact: Adrian Beliy, MD Principal Investigator: Adrian Beliy, MD Municipal Clinical Hospital Recruiting Chisinau, Moldova, Republic of Contact: Silvia Agop, MD Principal Investigator: Silvia Agop, MD Republican Clinical Hospital Recruiting Chisinau, Moldova, Republic of Contact: Victor Cojocaru, MD Principal Investigator: Viktor Kogokaru Ukraine Institute of General and Urgent Surgery of the National Academy of Medical Sciences of Ukraine Recruiting Kharkov, Ukraine Contact: Peev Stanislav, MD Principal Investigator: Peev Stanislav, MD Kyiv City Clinical Hospital No.4; National Medical University named after O.O. Bogomolets Recruiting Kyiv, Ukraine Contact: Igor Kolosovich, MD Principal Investigator: Igor Kolosovich, MD HSEE of Ukraine "Ukrainian Medical Stomatological Academy"; Poltava Central District Hospital Recruiting Poltava, Ukraine Contact: Alexey Ligonenko, MD Principal Investigator: Alexey Ligonenko, MD Vinnytsa Regional Clinical Hospital Active, not recruiting Vinnitsa, Ukraine Uzbekistan Republic Centre of Ambulance Recruiting Tashkent, Uzbekistan Contact: Visolat Sharipova, MD Principal Investigator: Visolat Sharipova, MD Collapse << |
| NCT02909400 | Mitochondrial Diseases ... more >> Mitochondrial Myopathies Mitochondrial Encephalomyopathies MELAS MIDD Collapse << | Phase 2 | Completed | - | Netherlands ... more >> Radboud University Medical Center Nijmegen, Netherlands Collapse << |
| NCT03732820 | Metastatic Castration-resistan... more >>t Prostate Cancer Collapse << | Phase 3 | Recruiting | August 17, 2022 | - |
| NCT00585546 | - | - | Terminated(No longer could obt... more >>ain clenbuterol) Collapse << | - | - |
| NCT01363297 | Acute Lymphocytic Leukemia | Phase 2 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States, 91010-3000 Stanford Unversity Cancer Clinical Trials Office Palo Alto, California, United States, 94304 Stanford Unversity Hospital and Clinics, CTRU Palo Alto, California, United States, 94304 Stanford Cancer Institute Stanford, California, United States, 94305 Stanford University Hospital and Clinics Stanford, California, United States, 94305 United States, Illinois The University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Massachusetts Massachusetts General Hospital (MGH) Boston, Massachusetts, United States, 02114 Brigham and Women's Hospital (BWH) Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 Karmanos Cancer Institute at Farmington Hills Farmington Hills, Michigan, United States, 48334 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Texas The University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 Collapse << |
| NCT03764085 | Sepsis Intoxi... more >>cation; Septic Collapse << | Phase 4 | Recruiting | October 31, 2019 | Georgia ... more >> "Unimedi Adjara" LLC Recruiting Batumi, Georgia Contact: Sophio Beridze Kutaisi Referral Hospital Recruiting Kutaisi, Georgia Contact: Nino Babunashvili "Unimedi Kakheti" Ltd Recruiting Telavi, Georgia Contact: Maya Makkhetelashvili Kazakhstan Hospital of the Medical Center of the Administration of the President of the Republic of Kazakhstan Recruiting Astana, Kazakhstan Contact: Nasrulla Shanazarov Moldova, Republic of Institute of Ambulance Recruiting Chisinau, Moldova, Republic of Contact: Adrian Belyy State Clinical Hospital of Traumatology and Orthopedy Recruiting Chisinau, Moldova, Republic of Contact: Octavian Cherempey State Clinical Hospital Recruiting Chisinau, Moldova, Republic of Contact: Victor Cojocaru Ukraine Dnipro`s Medical Academy of Ministry of Health of Ukraine Recruiting Dnipro, Ukraine, 49005 Contact: Yurii Kobelyatsky Zaicev's Institute of general and urgent surgery NMA Ukraine Recruiting Kharkiv, Ukraine, 61018 Contact: Stanislav Peev HSEE of Ukraine "Ukrainian Medical Stomatological Academy"; Poltava Central District Hospital Recruiting Poltava, Ukraine Contact: Alexey Lyhonenko Zaporizka Medical Academy of postgraduate education, Zaporizkyi Clinical Hospital N9 Recruiting Zaporizhzhya, Ukraine, 69065 Contact: Volodymyr Koshlya Uzbekistan Republic Centre of Ambulance Recruiting Tashkent, Uzbekistan Contact: Vіsolat Sharipova Collapse << |
| NCT02457793 | Non-Small Cell Lung Cancer, Me... more >>tastatic Colorectal Cancer, Metastatic Non Small Cell Lung Cancer, Metastatic Cancers, Melanoma Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado Aurora, Colorado, United States, 80045-2517 United States, Connecticut Yale Cancer Center New Haven, Connecticut, United States, 06520 United States, Massachusetts Dana Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 Collapse << |
| NCT03669692 | Prostatic Hyperplasia, Benign ... more >> Metabolic Syndrome Collapse << | Not Applicable | Recruiting | January 1, 2023 | Spain ... more >> Jose Luis Ponce Diaz-Reixa Recruiting A Coruña, Spain, 15006 Contact: Jose L Ponce, MD +34981178057 Jose.Luis.Ponce.DIaz-Reixa@sergas.es Principal Investigator: Jose L Ponce, MD Collapse << |
| NCT01363297 | - | - | Completed | - | - |
| NCT03686969 | Dermatomyositis | Phase 3 | Recruiting | June 2020 | Russian Federation ... more >> I.M. SECHENOV FIRST MOSCOW STATE MEDICAL UNIVERSITY Rheumatology Department Of, Clinici Of Nephrology Recruiting Moscow, Russian Federation, 119992 Collapse << |
| NCT01816958 | - | - | Completed | - | United States, Illinois ... more >> The Neuroscience Center Deerfield, Illinois, United States, 60015 Collapse << |
| NCT03658213 | Breast Cancer | Phase 3 | Not yet recruiting | December 31, 2020 | China, Beijing ... more >> The 307th hospital of Chinese People's Liberation Army Not yet recruiting Beijing, Beijing, China Contact: Zefei JIANG Collapse << |
| NCT03735420 | Healthy | Phase 1 | Not yet recruiting | August 2021 | - |
| NCT02205476 | Hypertrophic Scars Resulting F... more >>rom Prior Breast Scar Revision Surgery Collapse << | Phase 2 | Terminated(B5301001 study did ... more >>not meet its predefined efficacy endpoints, the long term follow up study B5301012 was terminated in 1/6/2015. No safety concerns identified.) Collapse << | - | United States, Florida ... more >> Stephan Baker MD PA Coral Gables, Florida, United States, 33146 Bayside Ambulatory Center Miami, Florida, United States, 33133 United States, Georgia Kavali Plastic Surgery and Skin Renewal Center Atlanta, Georgia, United States, 30328 Primeter outpatient surgery center Atlanta, Georgia, United States, 30328 United States, Missouri Body Aesthetic Research Center Saint Louis, Missouri, United States, 63141 Collapse << |
| NCT02205476 | - | - | Terminated(B5301001 study did ... more >>not meet its predefined efficacy endpoints, the long term follow up study B5301012 was terminated in 1/6/2015. No safety concerns identified.) Collapse << | - | - |
| NCT02457793 | - | - | Completed | - | - |
| NCT02129231 | Oxidative Stress ... more >> Diabetic Polyneuropathy Collapse << | Phase 2 | Completed | - | Mexico ... more >> Cardiovascular Research Unit Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00325299 | Alcohol-Induced Disorders | Phase 4 | Completed | - | Finland ... more >> Finnish Foundation for Alcohol Studies Helsinki, Finland, 00531 Collapse << |
| NCT02354339 | Metabolic Syndrome X | Phase 2 | Completed | - | Mexico ... more >> Instituto de Terapéutica Experimental y Clínica Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT01524159 | Cardiovascular Disease. | Not Applicable | Completed | - | China, Beijing ... more >> Hebei Yiling Hospital Shijiazhuang, Beijing, China, 050091 Collapse << |
| NCT03665519 | Primary Biliary Cirrhosis | Not Applicable | Recruiting | December 2020 | Kazakhstan ... more >> National Research Medical Center Recruiting Astana, Kazakhstan, 010000 Contact: Galiya Shaimardanova, PhD +77017299148 galiya_masugut@mail.ru Contact: Gulmira Dossatayeva, MD, MBA +77015127500 g_dossatayeva@inbox.ru Principal Investigator: Abay Baigenzhin, MD, PhD Sub-Investigator: Gulmira Dossatayeva, MD, MBA Sub-Investigator: Maiya Zhumabayeva, MD Sub-Investigator: Galiya Shaimardanova, PhD Collapse << |
| NCT00896064 | Infections, Streptococcal | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Collapse << |
| NCT01098500 | - | - | Completed | - | - |
| NCT02929901 | Type 2 Diabetes Nonalcoholic F... more >>atty Liver Collapse << | Phase 2 Phase 3 | Unknown | December 2017 | Iran, Islamic Republic of ... more >> NNFTRI clinic Tehran, Iran, Islamic Republic of, 19435 Collapse << |
| NCT02706834 | Dose Finding Study | Phase 1 | Completed | - | United States, Maryland ... more >> Baltimore, Maryland, United States United States, Texas Austin, Texas, United States Collapse << |
| NCT02319577 | Non Small Cell Lung Cancer | Phase 2 | Unknown | December 2015 | Italy ... more >> IRCCS Azienda Ospedaliera Universitaria San Martino - IST Istituto Nazionale per la Ricerca sul Cancro Recruiting Genova, Italy, 16132 Contact: Francesco Grossi, MD +39 010 5600385 francesco.grossi@hsanmartino.it Collapse << |
| NCT01098500 | - | - | Completed | - | - |
| NCT01215227 | Parkinson Disease ... more >> Idiopathic Parkinson Disease Collapse << | Phase 3 | Terminated | - | - |
| NCT03159507 | Healthy | Phase 4 | Recruiting | February 2019 | Canada, Quebec ... more >> SCF Pharma Recruiting Rimouski, Quebec, Canada, G5M 1J5 Contact: Samuel Fortin, PhD 514 317-9347 sfortin@scfpharma.com Collapse << |
| NCT03084783 | HSV Encephalitis | Phase 3 | Not yet recruiting | December 1, 2020 | France ... more >> Hôpital Gui de Chauliac Montpellier, France, 34295 CHU Hôtel-Dieu Nantes, France, 44093 Hôpital Bichat-Claude Bernard, APHP Paris, France, 75877 CHU Rennes, Hôpital Pontchaillou Rennes, France, 35000 Hôpital Charles Nicolle Rouen, France, 76031 Hôpital Delafontaine Saint-Denis, France, 93205 CHU Strasbourg Strasbourg, France, 67091 CHRU de Nancy, Hopitaux de Brabois Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT03174041 | Healthy Participants | Phase 1 | Completed | - | United States, Florida ... more >> Covance Research Unit - Daytona Daytona Beach, Florida, United States, 32117 United States, Indiana Covance Clinical Research Unit Inc.; Covance Gfi Research Evansville, Indiana, United States, 47710 United States, Wisconsin Covance Clinical Research Unit, Inc Madison, Wisconsin, United States, 53704 Collapse << |
| NCT00896064 | - | - | Completed | - | - |
| NCT02706834 | - | - | Completed | - | - |
| NCT01777256 | Systemic Lupus Erythematosus | Phase 2 | Terminated(This study met a pr... more >>otocol defined futility criterion and will be terminated following completion of the Follow Up Visit on the last subject randomised.) Collapse << | - | - |
| NCT03151837 | Diabetes Mellitus, Type 2 | Not Applicable | Completed | - | Taiwan ... more >> Chung Shan Medical University Hospital Taichung, Taiwan, 40201 Collapse << |
| NCT03301506 | Primary Biliary Cirrhosis | Phase 2 Phase 3 | Recruiting | March 2022 | - |
| NCT02290106 | Obesity Fatty... more >> Liver, Nonalcoholic Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01215227 | - | - | Terminated | - | - |
| NCT02028676 | Human Immunodeficiency Virus | Phase 4 | Completed | - | Uganda ... more >> MRC /UVRI Uganda Research Unit on AIDS Entebbe, Uganda Joint Clinical Research Centre Kampala, Uganda Baylor College of Medicine Children's Foundation Mulago, Uganda Zimbabwe University of Zimbabwe Medical School Harare, Zimbabwe Collapse << |
| NCT01942915 | Liver Cirrhosis | Phase 1 | Unknown | December 2014 | China, Hebei ... more >> the First Hospital of Hebei Medical University Shijiazhuang, Hebei, China, 050000 Collapse << |
| NCT01098487 | - | - | Completed | - | - |
| NCT02363946 | - | - | Terminated(Company decision to... more >> terminate the trial) Collapse << | - | - |
| NCT00628108 | Allergic Rhinitis ... more >> Chronic Urticaria Collapse << | Phase 3 | Completed | - | United States, Alabama ... more >> Birmingham, Alabama, United States United States, Arkansas Hot Springs, Arkansas, United States Jonesboro, Arkansas, United States Little Rock, Arkansas, United States United States, California Bakersfield, California, United States Crescent City, California, United States Huntington Beach, California, United States Los Angeles, California, United States Orange, California, United States Roseville, California, United States Stockton, California, United States United States, Georgia Albany, Georgia, United States Gainesville, Georgia, United States United States, Illinois Normal, Illinois, United States United States, Missouri Bridgeton, Missouri, United States United States, Nebraska Omaha, Nebraska, United States United States, Oklahoma Oklahoma City, Oklahoma, United States United States, South Carolina Barnwell, South Carolina, United States Spartanburg, South Carolina, United States United States, Tennessee Kingsport, Tennessee, United States United States, Texas Austin, Texas, United States Dallas, Texas, United States El Paso, Texas, United States San Antonio, Texas, United States Sugarland, Texas, United States United States, Virginia Richmond, Virginia, United States Collapse << |
| NCT01098487 | Purpura, Thrombocytopaenic, Id... more >>iopathic Collapse << | Phase 4 | Completed | - | - |
| NCT02028676 | - | - | Completed | - | - |
| NCT00628108 | - | - | Completed | - | - |
| NCT00619801 | Allergic Rhinitis ... more >> Chronic Urticaria Collapse << | Phase 3 | Completed | - | - |
| NCT03286634 | Down Syndrome ... more >> Acute Lymphoblastic Leukemia Childhood Cancer Collapse << | Phase 2 | Recruiting | March 31, 2033 | Hong Kong ... more >> Prince of Wales Hospital Recruiting Shatin, New Territories, Hong Kong Contact: Chi Kong Li, MBBS ckli@cuhk.edu.hk Japan Kagoshima University Hospital Recruiting Kagoshima, Japan, 890-8544 Contact: Yasuhiro Okamoto okamoto@m2.kufm.kagoshima-u.ac.jp Malaysia University of Malaya Medical Centre Not yet recruiting Kuala Lumpur, Malaysia, 59100 Contact: Hany Arrifin, MBBS hany@ummc.edu.my Subang Jaya Medical Centre Not yet recruiting Subang Jaya, Malaysia, 47500 Contact: Hai Peng Lin, MBBS flslhp@gmail.com Sub-Investigator: Lee Lee Chan, MBBS Singapore National University Hospital Recruiting Singapore, Singapore, 119074 Contact: Allen Yeoh, MBBS allen_yeoh@nuhs.edu.sg KK Women's and Children's Hospital Not yet recruiting Singapore, Singapore, 229899 Contact: Ah Moy Tan, MBBS tan.ah.moy01@singhealth.com.sg Taiwan National Taiwan University Children's Hospital Not yet recruiting Taipei, Taiwan, 100 Contact: Tsamn Lin Dong, MD dtlin@ntuh.gov.tw Mackay Memorial Hospital Not yet recruiting Taipei, Taiwan, 10449 Contact: Hsi-Che Liu, MD hsiche@mmh.org.tw Chang Gung Memorial Hopsital, Linkou Not yet recruiting Taoyuan, Taiwan, 333 Contact: Shih-Hsiang Chen, MD samechen@cgmh.org.tw Collapse << |
| NCT02363946 | Alpha-1 Antitrypsin Deficiency | Phase 1 | Terminated(Company decision to... more >> terminate the trial) Collapse << | - | Australia, Victoria ... more >> Nucleus Network Ltd Melbourne, Victoria, Australia, 3004 Germany Universitatsklinikum des Saarlandes Homburg, Germany Netherlands Leiden University Medical Center Leiden, Netherlands, 2333ZA United Kingdom Queen Elizabeth Hospital Edgbaston, Birmingham, United Kingdom, B15 2WB Collapse << |
| NCT00619801 | - | - | Completed | - | - |
| NCT02358044 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00767520 | Breast Cancer | Phase 2 | Completed | - | United States, California ... more >> Compassionate Cancer Care Medical Group, Inc Fountain Valley, California, United States, 92708 Compassionate Cancer Care Medical Group Inc Riverside, California, United States, 92501 United States, Pennsylvania Pennsylvania Oncology/Hematology Associates Philadelphia, Pennsylvania, United States, 19106 United States, Tennessee The West Clinic Memphis, Tennessee, United States, 38120 Czech Republic Local Institution Hradec Kralove, Czech Republic, 500 05 Local Institution Prague 5, Czech Republic, 150 06 France Local Institution Lille, France, 59000 Local Institution Paris Cedex 13, France, 75651 Local Institution Saint-Cloud Cedex, France, 92211 Ireland Local Institution Dublin, Ireland, 24 Poland Local Institution Gdansk, Poland, 80-462 Local Institution Gdansk, Poland, 80-952 Local Institution Lodz, Poland, 93-509 Local Institution Opole, Poland, 45-060 Spain Local Institution Madrid, Spain, 28033 Local Institution Madrid, Spain, 28041 Local Institution Torrevieja, Spain, 03186 Sweden Local Institution Vasteras, Sweden, 72189 United Kingdom Local Institution Chelmsford, Essex, United Kingdom, CM1 7ET Local Institution London, Greater London, United Kingdom, NW1 2BU Local Institution Coventry, Warwickshire, United Kingdom, CV22DX Collapse << |
| NCT00767520 | - | - | Completed | - | - |
| NCT00753688 | - | - | Completed | - | - |
| NCT00547521 | Rheumatoid Arthritis (RA) | Phase 3 | Completed | - | - |
| NCT00753688 | Sarcoma, Soft Tissue | Phase 3 | Completed | - | - |
| NCT00547521 | - | - | Completed | - | - |
| NCT01222962 | Malaria, Falciparum | Phase 1 | Completed | - | Australia ... more >> CMAX, a division of IDT Australia Limited Adelaide, Australia, SA 5000 Collapse << |
| NCT01222949 | Malaria, Falciparum | Phase 1 | Completed | - | Australia ... more >> CMAX, a division of IDT Australia Limited Adelaide, Australia, SA 5000 Nucleus Network Limited Melbourne, Australia, VIC 3004 Collapse << |
| NCT02358044 | - | - | Completed | - | - |
| NCT01860716 | Evidence of Liver Transplantat... more >>ion Collapse << | Phase 3 | Unknown | December 2013 | Spain ... more >> Hospital Clínico Universitario Lozano Blesa Not yet recruiting Zaragoza, Spain, 50009 Contact: Francisco A. García-Gil, Physician 0034976765700 agarciagil@telefonica.net Principal Investigator: Francisco A. García-Gil, Physician Collapse << |
| NCT01262976 | Tuberculosis | Phase 2 | Completed | - | India ... more >> GSK Investigational Site Tharamani Chennai, India, 600113 Collapse << |
| NCT01905215 | Infections, Respiratory Syncyt... more >>ial Virus Collapse << | Phase 1 | Completed | - | Canada, Nova Scotia ... more >> GSK Investigational Site Halifax, Nova Scotia, Canada, B3K 6R8 Canada, Ontario GSK Investigational Site Brampton, Ontario, Canada, L6T 0G1 GSK Investigational Site Toronto, Ontario, Canada, M9W 4L6 Collapse << |
| NCT00787137 | Arthritis, Psoriatic | Phase 1 | Terminated(poor recruitment) | - | Serbia ... more >> Professor Nemanja Damjanov Belgrade, Serbia Collapse << |
| NCT03202901 | Knee Injuries ... more >> Inflammation Knees Collapse << | Not Applicable | Not yet recruiting | March 21, 2018 | Spain ... more >> Technological Center of Nutrition and Health (CTNS) Not yet recruiting Reus, Spain, 43204 Contact: Francesc Puiggròs, PhD 977751382 francesc.puiggros@eurecat.org Collapse << |
| NCT02193867 | Lysosomal Acid Lipase Deficien... more >>cy Collapse << | Phase 2 | Active, not recruiting | April 2018 | United States, California ... more >> Irvine, California, United States Italy Naples, Italy United Kingdom Birmingham, United Kingdom Manchester, United Kingdom Collapse << |
| NCT01262976 | - | - | Completed | - | - |
| NCT00787137 | - | - | Terminated(poor recruitment) | - | - |
| NCT00023075 | - | - | Completed | - | United States, Maryland ... more >> National Institute of Neurological Disorders and Stroke (NINDS) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02680847 | Moderate-severe Pain | Phase 4 | Terminated(The trial was termi... more >>nated on 08FEB2018. Pfizer has decided to withdraw the New Drug Application and has notified FDA. There are no efficacy or safety concerns.) Collapse << | - | United States, California ... more >> Research Center For Clinical Studies-West, Inc. Lancaster, California, United States, 93534 Children's Hopsital Los Angeles Los Angeles, California, United States, 90027 Children's Hospital Of Los Angeles - University Of Southern California School Of Medicine Los Angeles, California, United States, 90027 Shriners Hospitals For Children Northern California Sacramento, California, United States, 95817 UC Davis Health Attn: Peter Trovitch, PharmD Sacramento, California, United States, 95817 University of California Davis Sacramento, California, United States, 95817 United States, Illinois University of Illinois at Chicago Clinical Research Center Chicago, Illinois, United States, 60612 University of Illinois Hospital and Health Sciences Systems Chicago, Illinois, United States, 60612 University of Illinois Hospital at the Medical Center Chicago, Illinois, United States, 60612 United States, North Carolina East Carolina University Brody School of Medicine(ECU) Greenville, North Carolina, United States, 27834 Leo Jenkins Cancer Center Pharmacy Greenville, North Carolina, United States, 27834 United States, South Carolina Medical University of South Carolina Children's Hospital Charleston, South Carolina, United States, 29425 Medical University of South Carolina, Investigational Drugs Services Charleston, South Carolina, United States, 29425 Medical University of South Carolina, Rutledge Tower, Pediatric Clinic Charleston, South Carolina, United States, 29425 Medical University of South Carolina, SCTR Research Nexus Charleston, South Carolina, United States, 29425 United States, Texas Road Runner Research, Ltd San Antonio, Texas, United States, 78249 Collapse << |
| NCT00457197 | - | - | Completed | - | - |
| NCT02680847 | - | - | Terminated(The trial was termi... more >>nated on 08FEB2018. Pfizer has decided to withdraw the New Drug Application and has notified FDA. There are no efficacy or safety concerns.) Collapse << | - | - |
| NCT00457197 | Bipolar Disorder ... more >> Alcohol Dependence Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> Psychoneuroendocrine Research Program Dallas, Texas, United States, 75235 Collapse << |
| NCT03473925 | Solid Tumors ... more >>Non-small Cell Lung Cancer Castration Resistant Prostate Cancer Microsatellite Stable Colorectal Cancer Collapse << | Phase 2 | Recruiting | August 9, 2019 | United States, Arizona ... more >> Honor Health ( Site 0005) Recruiting Scottsdale, Arizona, United States, 85258 Contact: Study Coordinator 623-238-7685 United States, Florida Florida Cancer Specialists ( Site 0003) Recruiting Sarasota, Florida, United States, 34232 Contact: Study Coordinator 941-377-9993 United States, Michigan Henry Ford Health System ( Site 0006) Recruiting Detroit, Michigan, United States, 48202 Contact: Study Coordinator 313-916-1784 United States, North Carolina Duke University Medical Center ( Site 0004) Recruiting Durham, North Carolina, United States, 27710 Contact: Study Coordinator 919-668-8797 Australia, New South Wales Scientia Clinical Research ( Site 0021) Recruiting Sydney, New South Wales, Australia, 2031 Contact: Study Coordinator +61293825807 Australia, Victoria Peter MacCallum Cancer Centre ( Site 0023) Recruiting Melbourne, Victoria, Australia, 3000 Contact: Study Coordinator +61385595000 Canada, Ontario Princess Margaret Cancer Centre ( Site 0031) Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Study Coordinator 41694645015634 Canada, Quebec Jewish General Hospital ( Site 0032) Recruiting Montreal, Quebec, Canada, H3T 1E2 Contact: Study Coordinator 5143408222 Israel Sourasky Medical Center ( Site 0012) Recruiting Tel Aviv, Israel, 6423906 Contact: Study Coordinator +97236973082 Korea, Republic of Severance Hospital Yonsei University Health System ( Site 0041) Recruiting Seoul, Korea, Republic of, 03722 Contact: Study Coordinator +82222288132 Collapse << |
| NCT03226067 | Primary Biliary Cirrhosis | Phase 2 | Active, not recruiting | September 26, 2019 | - |
| NCT01880593 | Depression | Phase 2 | Completed | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT01880593 | - | - | Completed | - | - |
| NCT01376726 | HIV Infections | Phase 1 | Completed | - | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232-2582 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 Collapse << |
| NCT00119678 | Systemic Lupus Erythematosus | Phase 2 | Completed | - | - |
| NCT03593707 | Healthy Volunteers | Phase 1 | Suspended(Sponsor elected to v... more >>oluntarily pause study.) Collapse << | February 12, 2019 | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT00119678 | - | - | Completed | - | - |
| NCT00519376 | - | - | Completed | - | - |
| NCT00519376 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Wiesbaden, Hessen, Germany, 65187 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Berlin, Germany, 14050 GSK Investigational Site Berlin, Germany, 14057 Collapse << |
| NCT00515502 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Berlin, Germany, 14050 GSK Investigational Site Hamburg, Germany, 22291 Collapse << |
| NCT00046267 | HIV Infections ... more >> Hypercholesterolemia Hypertriglyceridemia Diabetes Mellitus Collapse << | Not Applicable | Completed | - | United States, California ... more >> UCLA School of Medicine Los Angeles, California, United States, 90095 Univ of California, San Diego Antirviral Research San Diego, California, United States, 92103 San Mateo County AIDS Program Stanford, California, United States, 94305 Stanford Univ Stanford, California, United States, 94305 Willow Clinic Stanford, California, United States, 94305 United States, Colorado Univ of Colorado Health Sciences Ctr, Denver Denver, Colorado, United States, 80262-3706 United States, Florida Univ of Miami School of Medicine Miami, Florida, United States, 33136-1013 United States, Indiana Indiana University Hospital Indianapolis, Indiana, United States, 46202 Methodist Hosp of Indiana Indianapolis, Indiana, United States, 46202 Wishard Hospital Indianapolis, Indiana, United States, 46202 United States, Minnesota Univ of Minnesota Minneapolis, Minnesota, United States, 55455-0392 United States, Missouri St. Louis Connect Care St. Louis, Missouri, United States, 63108 Washington Univ (St. Louis) St. Louis, Missouri, United States, 63108 United States, Ohio Univ of Cincinnati Cincinnati, Ohio, United States, 45267-0405 Case Western Reserve Univ Cleveland, Ohio, United States, 44106 United States, Pennsylvania Univ of Pittsburgh Pittsburgh, Pennsylvania, United States, 15213-2582 Collapse << |
| NCT01949090 | Influenza | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Hollywood, Florida, United States, 33024 United States, Idaho GSK Investigational Site Meridian, Idaho, United States, 83642 United States, Kansas GSK Investigational Site Lenexa, Kansas, United States, 66219 GSK Investigational Site Wichita, Kansas, United States, 67207 United States, Kentucky GSK Investigational Site Lexington, Kentucky, United States, 40509 Canada, Nova Scotia GSK Investigational Site Truro, Nova Scotia, Canada, B2N 1L2 Canada, Ontario GSK Investigational Site Sudbury, Ontario, Canada, P3E 1H5 GSK Investigational Site Toronto, Ontario, Canada, M9W 4L6 Collapse << |
| NCT01949090 | - | - | Completed | - | - |
| NCT01556633 | Healthy Volunteer | Phase 1 | Completed | - | New Zealand ... more >> Christchurch, New Zealand, 8011 Grafton, New Zealand, 1010 Collapse << |
| NCT00640328 | Multiple Sclerosis | Phase 2 | Completed | - | - |
| NCT00410410 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT01556633 | - | - | Completed | - | - |
| NCT02583685 | Chronic Hepatitis C Infection | Phase 2 | Recruiting | March 2019 | China, Beijing ... more >> Liver Fibrosis Diagnosis and Treatment Centre, 302 Hospital Recruiting Beijing, Beijing, China, 100039 Contact: Guofeng Chen, MD (8610)66933427 guofengchen302@163.com Principal Investigator: George KK Lau, MD Principal Investigator: Guofeng Chen, MD China, Hong Kong Humanity and Health GI and Liver Centre Recruiting Hong Kong, Hong Kong, China, 00852 Contact: George KK Lau, MD (852)28613777 gkklau@netvigator.com Principal Investigator: George KK Lau, MD Collapse << |
| NCT00625404 | HIV Infections | Phase 3 | Completed | - | Kenya ... more >> Bondo Clinic, Bondo District Hospital Bondo, Nyanza, Kenya South Africa Setshaba Research Centre Pretoria, Gauteng, South Africa Josha Research Center Bloemfontein, South Africa Tanzania Arusha Clinic, Levolosi Health Center Arusha, Tanzania Collapse << |
| NCT03435627 | - | - | Enrolling by invitation | November 30, 2021 | Japan ... more >> Novo Nordisk Investigational Site Maebashi-shi, Gunma, Japan, 371-8511 Novo Nordisk Investigational Site Osaka, Japan, 594-1101 Novo Nordisk Investigational Site Tokyo, Japan, 157 8535 Collapse << |
| NCT00625404 | - | - | Completed | - | - |
| NCT00515502 | - | - | Completed | - | - |
| NCT00640328 | - | - | Completed | - | - |
| NCT00583362 | Systemic Lupus Erythematosus | Phase 2 | Completed | - | - |
| NCT00410410 | - | - | Completed | - | - |
| NCT00583362 | - | - | Completed | - | - |
| NCT00712933 | Systemic Lupus Erythematosus | Phase 3 | Completed | - | - |
| NCT02654080 | HIV Infections | Phase 1 | Active, not recruiting | - | United States, Ohio ... more >> Case Clinical Research Site Cleveland, Ohio, United States, 44106 United States, Pennsylvania Penn Prevention CRS Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00937326 | - | - | Completed | - | - |
| NCT01222078 | Diabetes Mellitus, Type 1 | Phase 2 | Terminated(A Phase 3 study rec... more >>ently reported and demonstrated that the dose of otelixizumab in OTX113390 is not effective.) Collapse << | - | France ... more >> GSK Investigational Site Paris Cedex 18, France, 75877 Germany GSK Investigational Site Ulm, Baden-Wuerttemberg, Germany, 89081 GSK Investigational Site Leipzig, Sachsen, Germany, 04103 Collapse << |
| NCT01641692 | Asthma | Phase 2 | Completed | - | - |
| NCT00712933 | - | - | Completed | - | - |
| NCT01222078 | - | - | Terminated(A Phase 3 study rec... more >>ently reported and demonstrated that the dose of otelixizumab in OTX113390 is not effective.) Collapse << | - | - |
| NCT00941876 | HIV Infection ... more >> HIV Infections Collapse << | Not Applicable | Completed | - | Tanzania ... more >> Muhimbili University of Health and Allied Services Dar es Salaam, Tanzania Collapse << |
| NCT00937326 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00071760 | - | - | Active, not recruiting | - | - |
| NCT00071760 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 2 | Active, not recruiting | June 2021 | United States, Florida ... more >> GSK Investigational Site Jacksonville, Florida, United States, 32209 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27705 Argentina GSK Investigational Site Buenos Aires, Argentina, 1405 Mexico GSK Investigational Site Mexico, D.F., Mexico, 06720 GSK Investigational Site Mexico, Mexico, 6720 Portugal GSK Investigational Site Almada, Portugal, 2805-267 GSK Investigational Site Amadora, Portugal, 2700 GSK Investigational Site Lisboa, Portugal, 1649-035 Puerto Rico GSK Investigational Site San Juan, Puerto Rico, 00935 Russian Federation GSK Investigational Site Moscow, Russian Federation, 105275 GSK Investigational Site Moscow, Russian Federation, 129110 GSK Investigational Site St. Petersburg, Russian Federation, 196645 South Africa GSK Investigational Site Durban, KwaZulu- Natal, South Africa, 4013 GSK Investigational Site Parow Valley, Western Province, South Africa, 7505 GSK Investigational Site Soweto, South Africa, 2013 Collapse << |
| NCT01641692 | - | - | Completed | - | - |
| NCT01648140 | Hepatitis C, Chronic | Phase 2 | Completed | - | - |
| NCT01113346 | Viral Gastroenteritis | Phase 2 Phase 3 | Unknown | February 2011 | Russian Federation ... more >> Arhangelsk Regional Children's Clinical Hospital named after P.G. Vizshletsov Recruiting Arhangelsk, Arhangelskaya oblast, Russian Federation, 163062 Contact: Larisa Titova, MD, PhD +7-8182-280433 irina-bul@yandex.ru Contact: Olga Samodova, MD, DrSc +7-8182-22-95-42 samodova@atknet.ru Principal Investigator: Larisa Titova, MD, PhD Sub-Investigator: Olga Samodova, MD, DrSc St. Vladimir Children's Moscow Clinical Hospital Recruiting Moscow, Russian Federation, 107014 Contact: Elena Meskina, MD, DrSc +7-916-1216172 meskinaelena@mail.ru Contact: Lyudmila Feklisova, MD, DrSc +7-495-2682226 vespamen@yandex.ru Sub-Investigator: Lyudmila Feklisova, MD, DrSc Principal Investigator: Elena Meskina, MD, DrSc Collapse << |
| NCT01648140 | - | - | Completed | - | - |
| NCT00604825 | Menopausal and Female Climacte... more >>ric States Collapse << | Phase 2 | Completed | - | - |
| NCT00146328 | HIV Infections | Phase 2 Phase 3 | Completed | - | - |
| NCT03694197 | Heterozygous Familial Hypercho... more >>lesterolemia Non-familial Hypercholesterolemia Collapse << | Phase 4 | Enrolling by invitation | November 24, 2023 | United States, Texas ... more >> Regeneron Research Site Schertz, Texas, United States, 78154 Collapse << |
| NCT00732472 | - | - | Completed | - | - |
| NCT00146328 | - | - | Completed | - | - |
| NCT01332292 | Asthma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Cypress, California, United States, 90630 GSK Investigational Site Huntington Beach, California, United States, 92647 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80206 United States, Illinois GSK Investigational Site Normal, Illinois, United States, 61761 United States, Oregon GSK Investigational Site Medford, Oregon, United States, 97504 Collapse << |
| NCT01332292 | - | - | Completed | - | - |
| NCT01453023 | Asthma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Huntington Beach, California, United States, 92647 Collapse << |
| NCT01453296 | Asthma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Cypress, California, United States, 90630 GSK Investigational Site Huntington Beach, California, United States, 92647 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80206 United States, Illinois GSK Investigational Site Normal, Illinois, United States, 61761 United States, Oregon GSK Investigational Site Medford, Oregon, United States, 97504 Collapse << |
| NCT00604825 | - | - | Completed | - | - |
| NCT00732472 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Reading, Berkshire, United Kingdom, RG2 0TG GSK Investigational Site Buckshaw Village, Chorley, Lancashire, United Kingdom, PR7 7NA GSK Investigational Site Waterloo, Liverpool, Merseyside, United Kingdom, L22 0LG GSK Investigational Site Clydebank, Glasgow, United Kingdom, G81 2DR GSK Investigational Site Edgbaston, Birmingham, United Kingdom, B15 2SQ GSK Investigational Site Llanishen, United Kingdom, CF14 5GJ GSK Investigational Site Manchester, United Kingdom, M15 6SX Collapse << |
| NCT01453023 | - | - | Completed | - | - |
| NCT00128492 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT00000372 | Schizophrenia | Phase 3 | Withdrawn(Pairing D-Cycloserin... more >>e with Clozapine was found to worsen negative side effects in patients with Schizophrenia, so the study was suspended.) Collapse << | - | United States, Massachusetts ... more >> Freedom Trail Clinic Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00128492 | - | - | Completed | - | - |
| NCT03563846 | Lactose Intolerance | Phase 1 | Completed | - | United States, Florida ... more >> Syneos Health Miami, Florida, United States, 33136 Collapse << |
| NCT01453296 | - | - | Completed | - | - |
| NCT01381549 | Infections, Urinary Tract | Phase 2 | Terminated(Microbiological fin... more >>dings of resistance on therapy in patients with complicated urinary tract infection) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90033 United States, Iowa GSK Investigational Site Council Bluffs, Iowa, United States, 51503 United States, Kansas GSK Investigational Site Topeka, Kansas, United States, 66604 United States, Texas GSK Investigational Site Corsicana, Texas, United States, 75110 Canada, Quebec GSK Investigational Site Chicoutimi, Quebec, Canada, G7H 5H6 GSK Investigational Site Sherbrooke, Quebec, Canada, J1H 5N4 France GSK Investigational Site Suresnes, France, 92151 GSK Investigational Site Toulouse cedex 9, France, 31059 Greece GSK Investigational Site Athens, Greece, 11527 GSK Investigational Site Chaidari, Greece, 12462 GSK Investigational Site Goudi, Athens, Greece, 11527 GSK Investigational Site Thessaloniki, Greece, 56429 Russian Federation GSK Investigational Site Irkutsk, Russian Federation, 664079 GSK Investigational Site Moscow, Russian Federation, 125367 GSK Investigational Site Rostov-on-Don, Russian Federation, 344022 GSK Investigational Site Smolensk, Russian Federation, 214018 GSK Investigational Site St'Petersburg, Russian Federation, 194354 Spain GSK Investigational Site Alicante, Spain, 03010 GSK Investigational Site Elche (Alicante), Spain, 03203 GSK Investigational Site Getafe/Madrid, Spain, 28905 GSK Investigational Site Granada, Spain, 18003 GSK Investigational Site Madrid, Spain, 28040 GSK Investigational Site Murcia, Spain GSK Investigational Site Pama de Mallorca, Spain, 07010 Collapse << |
| NCT01018186 | Asthma | Phase 3 | Completed | - | - |
| NCT00088465 | - | - | Completed | - | - |
| NCT01018186 | - | - | Completed | - | - |
| NCT00088465 | Schizophrenic Disorders ... more >> Schizoaffective Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT01908660 | - | - | Completed | - | Spain ... more >> Hospital Universitario de Valme Seville, Spain, 41014 Collapse << |
| NCT01218477 | Leukemia | Phase 1 Phase 2 | Completed | - | United States, California ... more >> University Of California Medical Center San Francisco, California, United States, 94143 United States, Maryland Sidney Kimmel Cancer Center At Johns Hopkins Baltimore, Maryland, United States, 21287 United States, Texas Ut M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Canada, Ontario Local Institution Hamilton, Ontario, Canada, L8N 3Z5 Local Institution Toronto, Ontario, Canada, M5G 2M9 Finland Local Institution Helsinki, Finland, 00290 France Local Institution Bordeaux, France, 33076 Local Institution Poitiers Cedex, France, 86021 Germany Local Institution Frankfurt/main, Germany, 60590 Italy Local Institution Bologna, Italy, 40138 Local Institution Orbassano(To), Italy, 10043 United Kingdom Local Institution Glasgow, United Kingdom, G12 0YN Collapse << |
| NCT01218477 | - | - | Completed | - | - |
| NCT02999152 | Bone Metastasis ... more >> Malignant Hemopathy Collapse << | Not Applicable | Recruiting | December 31, 2019 | France ... more >> Institut de Cancérologie Lucien Neuwirth Recruiting Saint-Priest en Jarez, France, 42270 Principal Investigator: Nicolas Magné, PhD Sub-Investigator: Guy De Laroche, MD Sub-Investigator: Grégoire Pigne, MD Sub-Investigator: Denis Guyotat, PhD Sub-Investigator: Jérôme Cornillon, MD Institut Gustave Roussy Not yet recruiting Villejuif, France, 94800 Contact: Valentine Martin, MD Principal Investigator: Valentine Martin, MD Sub-Investigator: Angela Boros, MD Collapse << |
| NCT01725126 | Obesity | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Kansas GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT01381549 | - | - | Terminated(Microbiological fin... more >>dings of resistance on therapy in patients with complicated urinary tract infection) Collapse << | - | - |
| NCT01883674 | Sarcopenia | Not Applicable | Completed | - | Canada, Quebec ... more >> Centre de recherche sur le vieillissement Sherbrooke, Quebec, Canada, J1H 4C4 Collapse << |
| NCT03093688 | Non-small Cell Lung Cancer ... more >> Small Cell Lung Cancer Pancreas Cancer Hepatocellular Carcinoma Gastric Cancer Renal Cell Carcinoma Collapse << | Phase 1 Phase 2 | Recruiting | December 31, 2019 | China, Shanghai ... more >> Shanghai Public Health Clinical Center Recruiting Shanghai, Shanghai, China, 201508 Contact: Yan X Zhang, M.D. 0086-021-37990333 ext 7310 zhangxiaoyan@shaphc.org Collapse << |
| NCT01725126 | - | - | Completed | - | - |
| NCT01209078 | Skin Infections, Bacterial | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Anniston, Alabama, United States, 36207 United States, California GSK Investigational Site Chula Vista, California, United States, 91911 GSK Investigational Site La Mesa, California, United States, 91942 GSK Investigational Site Long Beach, California, United States, 90813 GSK Investigational Site Oceanside, California, United States, 92056 United States, Hawaii GSK Investigational Site Honolulu, Hawaii, United States, 96813 United States, Pennsylvania GSK Investigational Site West Reading, Pennsylvania, United States, 19611 Collapse << |
| NCT00089583 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 2 | Completed | - | - |
| NCT01736033 | Benign Prostate Hyperplasia | Phase 4 | Unknown | June 2017 | Korea, Republic of ... more >> Chungbuk National University Hospital Cheongju, Chungbuk, Korea, Republic of Soon Chun Hyang University Hospital Cheonan Cheonan, Chungnam, Korea, Republic of Hallym University Sacred Heart Hospital Anyang, Gyeoggi, Korea, Republic of Bucheon St. Mary's Hospital Bucheon, Gyeonggi, Korea, Republic of Soon Chun Hyang University Hospital Bucheon Bucheon, Gyeonggi, Korea, Republic of Seoul National University Bundang Hospital Seongnam, Gyeonggi, Korea, Republic of Ajou University Hospital Suwon, Gyeonggi, Korea, Republic of Chonnam National University Hwasun Hospital Hwasun, Jeonnam, Korea, Republic of Pusan National University Hospital Busan, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Eulji General Hospital Seoul, Korea, Republic of Collapse << |
| NCT01831791 | Alopecia | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 812-0025 GSK Investigational Site Osaka, Japan, 530-0057 GSK Investigational Site Osaka, Japan, 532-0003 GSK Investigational Site Tokyo, Japan, 103-0028 GSK Investigational Site Tokyo, Japan, 160-0022 Collapse << |
| NCT03499249 | Biliary Atresia | Phase 2 | Recruiting | December 31, 2021 | United States, Texas ... more >> Texas Children's Hospital and Baylor College of Medicine Recruiting Houston, Texas, United States, 77030 Contact: Sanjiv Harpavat, MD PhD 832-824-2099 ext 2144 harpavat@bcm.edu Principal Investigator: Sanjiv Harpavat, MD PhD Sub-Investigator: Benjamin Shneider, MD Sub-Investigator: Mary Brandt, MD Sub-Investigator: Dana Cerminara, PharmD Collapse << |
| NCT00089583 | - | - | Completed | - | - |
| NCT00566020 | Bipolar Disorder | Phase 3 | Completed | - | - |
| NCT01586364 | Atrophy Vagin... more >>al Diseases Collapse << | Phase 3 | Completed | - | - |
| NCT01209078 | - | - | Completed | - | - |
| NCT02460159 | Hypercholesterolemia ... more >> Heterozygous Familial Hypercholesterolemia Collapse << | Phase 3 | Completed | - | - |
| NCT00742508 | Heart Failure, Congestive | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 296-8602 GSK Investigational Site Ehime, Japan, 794-0006 GSK Investigational Site Hiroshima, Japan, 737-0023 GSK Investigational Site Hokkaido, Japan, 060-0033 GSK Investigational Site Hokkaido, Japan, 063-0005 GSK Investigational Site Kanagawa, Japan, 210-0852 GSK Investigational Site Kanagawa, Japan, 238-8558 GSK Investigational Site Mie, Japan, 511-0068 GSK Investigational Site Nagano, Japan, 397-8555 GSK Investigational Site Nagasaki, Japan, 859-3615 GSK Investigational Site Oita, Japan, 879-5593 GSK Investigational Site Osaka, Japan, 565-8565 GSK Investigational Site Saga, Japan, 843-0393 GSK Investigational Site Saitama, Japan, 364-8501 GSK Investigational Site Shizuoka, Japan, 410-2295 GSK Investigational Site Shizuoka, Japan, 411-8611 GSK Investigational Site Shizuoka, Japan, 427-8502 GSK Investigational Site Shizuoka, Japan, 430-8502 GSK Investigational Site Tokyo, Japan, 141-0001 GSK Investigational Site Tokyo, Japan, 142-8666 GSK Investigational Site Tokyo, Japan, 153-8515 GSK Investigational Site Tokyo, Japan, 196-0003 GSK Investigational Site Wakayama, Japan, 640-8158 Collapse << |
| NCT00566020 | - | - | Completed | - | - |
| NCT01831791 | - | - | Completed | - | - |
| NCT01586364 | - | - | Completed | - | - |
| NCT02613910 | Pemphigus | Phase 3 | Terminated(Novartis has acquir... more >>ed the rights to ofatumumab and terminated the OPV116910 and OPV117059 studies. This decision is not linked to any safety consideration.) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90045 United States, Michigan GSK Investigational Site Ann Arbor, Michigan, United States, 48103 Collapse << |
| NCT01876108 | Fatty Liver L... more >>iver Dysfunction Insulin Resistance Collapse << | Phase 2 | Completed | - | Iran, Islamic Republic of ... more >> Gastroenterology clinic, Sina Hospital Tehran, Iran, Islamic Republic of Collapse << |
| NCT00742508 | - | - | Completed | - | - |
| NCT00733304 | Macular Degeneration | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Beverly Hills, California, United States, 90211 GSK Investigational Site Sacramento, California, United States, 95841 United States, Florida GSK Investigational Site Winter Haven, Florida, United States, 33880 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46280 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02111 United States, Michigan GSK Investigational Site Ann Arbor, Michigan, United States, 48105 GSK Investigational Site Grand Rapids, Michigan, United States, 49525 United States, Utah GSK Investigational Site Salt Lake City, Utah, United States, 84132 Australia, New South Wales GSK Investigational Site Sydney, New South Wales, Australia, 2145 GSK Investigational Site Sydney, New South Wales, Australia, 2150 Australia, Victoria GSK Investigational Site Melbourne, Victoria, Australia Australia, Western Australia GSK Investigational Site Perth, Western Australia, Australia, 6009 Italy GSK Investigational Site Milano, Lombardia, Italy, 20132 GSK Investigational Site Milano, Lombardia, Italy, 20157 GSK Investigational Site Torino, Piemonte, Italy, 10122 GSK Investigational Site Firenze, Toscana, Italy, 50134 GSK Investigational Site Padova, Veneto, Italy, 35128 Collapse << |
| NCT00255190 | Gastroesophageal Reflux Diseas... more >>e Collapse << | Phase 3 | Completed | - | - |
| NCT02460159 | - | - | Completed | - | - |
| NCT00736099 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00255190 | - | - | Completed | - | - |
| NCT02793154 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(Early termination d... more >>ue to insufficient enrollment.) Collapse << | - | United States, Kentucky ... more >> GSK Investigational Site Louisville, Kentucky, United States, 40213 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75230 Collapse << |
| NCT02613910 | - | - | Terminated(Novartis has acquir... more >>ed the rights to ofatumumab and terminated the OPV116910 and OPV117059 studies. This decision is not linked to any safety consideration.) Collapse << | - | - |
| NCT00736099 | - | - | Completed | - | - |
| NCT02721277 | Cholestasis J... more >>aundice, Obstructive Collapse << | Phase 1 Phase 2 | Terminated | - | United States, Florida ... more >> University of Florida Gainesville, Florida, United States, 32610 Collapse << |
| NCT02721277 | - | - | Terminated | - | - |
| NCT03088345 | Circulatory Perfusion Disorder... more >> Congenital Heart Disease Single-ventricle Collapse << | Phase 2 Phase 3 | Recruiting | December 2020 | United States, Wisconsin ... more >> Children's Hospital of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Amee Bigelow, MD 414-266-4730 abigelow@mcw.edu Collapse << |
| NCT01841684 | Hyperlipoproteinemia Type II ... more >> Homozygous Familial Hypercholesterolemia Collapse << | Phase 3 | Terminated | - | - |
| NCT00733304 | - | - | Completed | - | - |
| NCT01527110 | Influenza, Human | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 816-0864 GSK Investigational Site Kanagawa, Japan, 247-8533 GSK Investigational Site Miyagi, Japan, 983-0824 GSK Investigational Site Miyagi, Japan, 985-8506 GSK Investigational Site Okayama, Japan, 700-8557 GSK Investigational Site Osaka, Japan, 581-0011 GSK Investigational Site Shizuoka, Japan, 432-8002 Collapse << |
| NCT00811018 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 3 | Terminated(This trial was prem... more >>aturely terminated on Dec 9 2010 due to safety concerns, specifically emerging evidence of hepatic injury.) Collapse << | - | - |
| NCT02793154 | - | - | Terminated(Early termination d... more >>ue to insufficient enrollment.) Collapse << | - | - |
| NCT01527110 | - | - | Completed | - | - |
| NCT00811018 | - | - | Terminated(This trial was prem... more >>aturely terminated on Dec 9 2010 due to safety concerns, specifically emerging evidence of hepatic injury.) Collapse << | - | - |
| NCT01220128 | Neoplasms, Breast | Phase 2 | Terminated(Study termination d... more >>ue to negative Ph III of another study product from same technology platform.) Collapse << | - | - |
| NCT02550288 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00048581 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT03586947 | Gynaecological Disease ... more >> Vitamin D Deficiency Uterine Fibroids Collapse << | Not Applicable | Not yet recruiting | July 1, 2021 | China, Zhejiang ... more >> The Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University Not yet recruiting Wenzhou, Zhejiang, China, 325000 Contact: TING LI feclinicalresearch@163.com Collapse << |
| NCT01220128 | - | - | Terminated(Study termination d... more >>ue to negative Ph III of another study product from same technology platform.) Collapse << | - | - |
| NCT02434497 | Homozygous Familial Hyperchole... more >>sterolemia (HoFH) Collapse << | Phase 3 | Completed | - | Belgium ... more >> Research Site Brussels (Woluwé-St-Lambert), Belgium, 1200 Canada, Quebec Research Site Chicoutimi, Quebec, Canada, G7H 7K9 Denmark Research Site Copenhagen, Denmark, DK-2100 Israel Research Site Halfa, Israel, 31096 Malaysia Research Site Kubang Kerian, Malaysia, 16150 Taiwan Research Site Taipei City, Taiwan, 11217 Collapse << |
| NCT02550288 | - | - | Completed | - | - |
| NCT01335698 | - | - | Completed | - | - |
| NCT01335698 | HIV | Phase 3 | Completed | - | - |
| NCT02434497 | - | - | Completed | - | - |
| NCT01070784 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Japan ... more >> Research Site Nagoya, Aichi, Japan Research Site Toyota, Aichi, Japan Research Site Yanagawa, Fukuoka, Japan Research Site Asahikawa, Hokkaido, Japan Research Site Sapporo, Hokkaido, Japan Research Site Itami, Hyogo, Japan Research Site Hitachi, Ibaraki, Japan Research Site Tsukuba, Ibaraki, Japan Research Site Sakaide, Kagawa, Japan Research Site Fujisawa, Kanagawa, Japan Research Site Yokohama, Kanagawa, Japan Research Site Koshi, Kumamoto, Japan Research Site Shibata, Miyagi, Japan Research Site Chuo, Tokyo, Japan Research Site Setagaya, Tokyo, Japan Research Site Hiroshima, Japan Research Site Kyoto, Japan Collapse << |
| NCT01070784 | - | - | Completed | - | - |
| NCT03081117 | HIV Dementia ... more >>HIV-Associated Cognitive Motor Complex Collapse << | Phase 1 Phase 2 | Recruiting | July 2021 | United States, Maryland ... more >> The Johns Hopkins Institute for Clinical and Translational Research, Adult Outpatient Clinical Research Unit Recruiting Baltimore, Maryland, United States, 21287 Contact: Mary De'Jarnette, RN, MSN 410-955-2760 crus@jhmi.edu Collapse << |
| NCT01047553 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | Japan ... more >> Research Site Nagoya, Aichi, Japan Research Site Akita-shi, Akita, Japan Research Site Chitose, Hokkaido, Japan Research Site Obihiro, Hokkaido, Japan Research Site Sapporo, Hokkaido, Japan Research Site AKO, Hyogo, Japan Research Site Kobe-shi, Hyogo, Japan Research Site Hitachi, Ibaraki, Japan Research Site Tsukuba, Ibaraki, Japan Research Site Kanazawa, Ishikawa, Japan Research Site Sakaide, Kagawa, Japan Research Site Fujisawa, Kanagawa, Japan Research Site Kawasaki-shi, Kanagawa, Japan Research Site Yokohama, Kanagawa, Japan Research Site Koshi, Kumamoto, Japan Research Site Nagaoka, Niigata, Japan Research Site Saiki-shi, Oita, Japan Research Site Moriguchi, Osaka, Japan Research Site Matsue, Shimane, Japan Research Site Bunkyo, Tokyo, Japan Research Site Chuo, Tokyo, Japan Research Site Katsushika-ku, Tokyo, Japan Research Site Kodaira, Tokyo, Japan Research Site Setagaya, Tokyo, Japan Research Site Tosima-ku, Tokyo, Japan Research Site Fukuoka, Japan Research Site Kyoto, Japan Collapse << |
| NCT00879814 | Meningitis, Meningococcal | Phase 1 | Completed | - | United States, Florida ... more >> Miami Research Associates, Inc. South Miami, Florida, United States, 33143 Collapse << |
| NCT00048581 | - | - | Completed | - | - |
| NCT00879814 | - | - | Completed | - | - |
| NCT01047553 | - | - | Completed | - | - |
| NCT00710164 | Acute Ischemic Stroke | Phase 2 Phase 3 | Unknown | - | China, Chengdu ... more >> Department of Nuerology, Kunming General Hospital of Chengdu Military Command Recruiting Kunming, Chengdu, China, 650032 Contact: Shixiang Liu, MD China, Guangdong Department of Nuerology, The Second People's Hospital of Foshan city Recruiting Foshan, Guangdong, China, 528000 Contact: Qingkun Zhou, MD Department of Nuerology, The Second People's Hospital of Jiangmen city Recruiting Jiangmen, Guangdong, China, 529030 Contact: Qiong Zheng, MD China, Guangxi Department of Nuerology, Liuzhou Hospital of Traditional Chinese Medicine Recruiting Liuzhou, Guangxi, China, 545001 Contact: Changjun Lu, MD China, Hebei Department of Nuerology, Cangzhou Central Hospistal Recruiting Cangzhou, Hebei, China, 060001 Contact: Junling Zhang, MD China, Hubei Department of Nuerology, Hubei Hospital of Traditional Chinese Medicine Recruiting Wuhan, Hubei, China, 430061 Contact: Mengjiu Dong, MD China, Hunan Department of Nuerology, The First People's Hospital of Changsha city Recruiting Changsha, Hunan, China, 410005 Contact: Shenhui Zhu, MD China, Shanghai Department of Neurology, Renji Hospital Affiliated to Shanghai Jiao Tong University School of Medicine Recruiting Shanghai, Shanghai, China, 200001 Contact: Yansheng Li Department of Nuerology, Changzheng Hospital Affiliated to the Second Military Medical University Recruiting Shanghai, Shanghai, China, 200003 Contact: Zhongxin Zhao, MD Department of Integrative Medicine, Zhongshan hospital, Fudan University Recruiting Shanghai, Shanghai, China, 200021 Contact: Dingfang Cai Department of Nuerology, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Not yet recruiting Shanghai, Shanghai, China, 200021 Contact: Zhenglong Fang, MD Department of Neurology, Ruijin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine Recruiting Shanghai, Shanghai, China, 200025 Contact: Shengdi Chen, MD Department of Nuerology, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Recruiting Shanghai, Shanghai, China, 200032 Contact: Canxing Yuan, MD Department of Nuerology, Zhongshan hospital, Fudan University Not yet recruiting Shanghai, Shanghai, China, 200032 Contact: Wei Fan, MD Department of Neurology, Huashan hospital, Fudan University Not yet recruiting Shanghai, Shanghai, China, 200040 Contact: Qiang Dong, MD Department of Nuerology, Tongji Hospital of Tongji University Recruiting Shanghai, Shanghai, China, 200065 Contact: Qing Zhan, MD Department of Neurology, Shanghai First People's Hospital Affiliated to Shanghai Jiao Tong University School of Medicine Recruiting Shanghai, Shanghai, China, 200081 Contact: Shaoshi Wang, MD Department of Nuerology, Xinhua Hospital Affiliated to Shanghai Jiaotong University School of Medicine Recruiting Shanghai, Shanghai, China, 200092 Contact: Zhenguo Liu, MD Department of Nuerology, Shanghai Sixth People's Hospital Affiliated to Shanghai Jiaotong University School of Medicine Active, not recruiting Shanghai, Shanghai, China, 200233 Department of Neurology, Changhai Hospital Affiliated to the Second Military Medical University Recruiting Shanghai, Shanghai, China, 200433 Contact: Yangtai Guan, MD Collapse << |
| NCT00932373 | Metastatic Breast Cancer | Phase 1 | Completed | - | - |
| NCT00932373 | - | - | Completed | - | - |
| NCT02462759 | Spinal Muscular Atrophy | Phase 2 | Active, not recruiting | April 9, 2019 | United States, California ... more >> David Geffen School of Medicine at UCLA Los Angeles, California, United States, 90095-8344 United States, Connecticut Connecticut Childrens Medical Hartford, Connecticut, United States, 06106 United States, Maryland The Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Minnesota Gillette Children's Specialty Healthcare Saint Paul, Minnesota, United States, 55101 United States, Texas The University of Texas Southwestern Medical Center Dallas, Texas, United States, 75235 United States, Washington Seattle Children's Research Institute Seattle, Washington, United States, 98105 Germany LMU-Campus Innenstadt Muenchen, Germany, 80337 Collapse << |
| NCT00302952 | - | - | Completed | - | - |
| NCT03685591 | Hormone Receptor Positive Adva... more >>nced Breast Cancer Metastatic Breast Cancer Human Epidermal Receptor 2 Negative Advanced Breast Cancer Castration Resistant Prostate Cancer Collapse << | Phase 1 | Recruiting | August 24, 2023 | United States, Texas ... more >> The University of Texas MD Anderson Cancer Center Not yet recruiting Houston, Texas, United States, 77030-4009 The University of Texas - MD Anderson Cancer Center Not yet recruiting Houston, Texas, United States, 77030 NEXT Oncology Recruiting San Antonio, Texas, United States, 78240 Collapse << |
| NCT02532673 | - | - | Completed | - | - |
| NCT02114658 | Thyroid Carcinoma | Phase 2 | Completed | - | Japan ... more >> Kobe, Hyogo, Japan, 650-0011 Matsumoto, Nagano, Japan, 390-8621 Bunkyo-ku, Tokyo, Japan, 113-8603 Koto-ku, Tokyo, Japan, 135-8550 Osaka, Japan, 545-8586 Collapse << |
| NCT00302952 | Rheumatoid Arthritis | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham, Alabama, United States, 35294 United States, California University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado University of Colorado Aurora, Colorado, United States, 80095 United States, Illinois University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Michigan Justus J. Fiechtner, MD, PLLC Lansing, Michigan, United States, 48910 United States, New York Feinstein Institute for Medical Research NS-LIJ Health System Manhasset, New York, United States, 11030 University of Rochester Rochester, New York, United States, 14642 United States, North Carolina Carolina Bone and Joint Charlotte, North Carolina, United States, 29425 Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Oklahoma Oklahoma Medical Research Foundation Oklahoma City, Oklahoma, United States, 73104 United States, Pennsylvania Altoona Center for Clinical Research Duncansville, Pennsylvania, United States, 16635 University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15261 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29425 United States, Texas Baylor Research Institute Dallas, Texas, United States, 75231 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT03613649 | Electrocardiogram Repolarisati... more >>on Abnormality Gonococcal Infection Collapse << | Phase 1 | Active, not recruiting | January 6, 2019 | United States, Kansas ... more >> Vince and Associates Clinical Research Overland Park, Kansas, United States, 66212 Collapse << |
| NCT02147041 | Diabetes Mellitus ... more >> Hyperlipidemia Collapse << | Phase 2 Phase 3 | Unknown | August 2014 | Taiwan ... more >> Taipei City Hospital Taipei, Taiwan Collapse << |
| NCT02841332 | Glioblastoma | Not Applicable | Terminated(lack of recruitment... more >>) Collapse << | - | France ... more >> UHToulouse Toulouse, France, 31000 Collapse << |
| NCT00720473 | Bipolar Depression | Not Applicable | Completed | - | United States, Massachusetts ... more >> McLean Hospital Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT00720473 | - | - | Completed | - | - |
| NCT02289027 | Ebola Vaccines | Phase 1 Phase 2 | Completed | - | Switzerland ... more >> Clinical Trial Unit Lausanne Lausanne, Switzerland, 1011 Collapse << |
| NCT01861054 | Breast Cancer | Phase 2 | Terminated(Enrollment target n... more >>ot reached) Collapse << | - | United States, Indiana ... more >> Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202-5289 United States, Kansas University of Kansas Cancer Center, 4350 Shawnee Mission Pkwy, Suite 1500, Mailstop 6004 Fairway, Kansas, United States, 66205 United States, New Jersey The Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 United States, New York Weill Cornell Medical College New York, New York, United States, 10065 Montefiore Medical Center, MMC Medical Park at Eastchester The Bronx, New York, United States, 10461 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Magee-Womens Hospital Pittsburgh, Pennsylvania, United States, 15213 United States, Tennessee Sara Cannon Research Institute Nashville, Tennessee, United States, 37203 United States, Texas The Methodist Hospital Research Institute Houston, Texas, United States, 77030 Collapse << |
| NCT01068717 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development, Lp Austin, Texas, United States, 78744 Collapse << |
| NCT01068717 | - | - | Completed | - | - |
| NCT01270958 | Rhinitis, Allergic, Perennial | Phase 4 | Completed | - | Vietnam ... more >> GSK Investigational Site Ha Noi, Vietnam, 084 Collapse << |
| NCT00697593 | Chronic Plaque Psoriasis | Phase 4 | Terminated(The study was termi... more >>nated after the European Medicines Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | Canada, Ontario ... more >> Probity Medical Research City Waterloo, Ontario, Canada, N2J 1C4 Collapse << |
| NCT01270958 | - | - | Completed | - | - |
| NCT00555321 | Immunosuppression in Solid Org... more >>an Transplant Collapse << | Phase 2 | Terminated | - | - |
| NCT01243320 | - | - | Completed | - | - |
| NCT01405794 | Healthy | Phase 1 Phase 2 | Completed | - | United States, Utah ... more >> University of Utah Salt Lake City, Utah, United States, 84112-5820 Collapse << |
| NCT01099579 | HIV Infections | Phase 3 | Completed | - | Brazil ... more >> Local Institution Sao Paolo, SAO Paulo, Brazil, 01246-900 Chile Local Institution Santiago, Metropolitana, Chile, 8380418 Local Institution Santiago, Metropolitana, Chile Mexico Local Institution Df, Distrito Federal, Mexico, 06720 Local Institution Guadalajara, Jalisco, Mexico, 44160 Local Institution Guadalajara, Jalisco, Mexico, 44280 Local Institution Merida, Yucatan, Mexico, 97000 Local Institution Oaxaca, Mexico, 71256 Local Institution Puebla, Mexico, 72000 Peru Local Institution Lima, Peru, 1 Local Institution Lima, Peru South Africa Local Institution Bloemfontein, FREE State, South Africa, 9301 Local Institution Coronationville, Gauteng, South Africa, 2092 Local Institution Soweto, Gauteng, South Africa, 2001 Local Institution Congella, KWA ZULU Natal, South Africa, 4013 Local Institution Cape Town, Western CAPE, South Africa, 7505 Thailand Local Institution Bangkok, Thailand, 10330 Local Institution Bangkok, Thailand, 10700 Collapse << |
| NCT00697593 | - | - | Terminated(The study was termi... more >>nated after the European Medicines Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT01405794 | - | - | Completed | - | - |
| NCT00774163 | Healthy | Phase 1 | Completed | - | Peru ... more >> Community of Santa Clara Iquitos, Loreto, Peru Collapse << |
| NCT01243320 | Healthy | Phase 1 Phase 2 | Completed | - | United States, Utah ... more >> University of Utah Hospital and Clinics Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT03431805 | Postpartum Hemorrhage | Phase 3 | Recruiting | June 30, 2020 | France ... more >> CHU Angers Recruiting Angers, France, 49033 Contact: Guillaume Legendre, MD guillaume.legendre@chu-angers.fr CHU Jean Minjoz Recruiting Besançon, France, 25000 Contact: Didier Riethmuller, MD,PhD didier.riethmuller@univ-fcomte.fr CHU Bordeaux Recruiting Bordeaux, France, 33076 Contact: Loic Sentilhes, MD,PhD loic.sentilhes@chu-bordeaux.fr Contact: Olivier delorme olivier.delorme@chu-bordeaux.fr CHU Hôpital Femme Mère Enfant Not yet recruiting Bron, France, 69677 Contact: Muriel Doret, MD,PhD muriel.doret-dion@chu-lyon.fr CHRU Côte de Nacre Recruiting Caen, France, 14033 Contact: Delphine Vardon, MD +336 66 93 52 86 vardon-d@chu-caen.fr CHU Estain Recruiting Clermont Ferrand, France, 63001 Contact: Denis Gallot, MD,PhD dgallot@chu-clermontferrand.fr Centre Hospitalier Intercommunal de Créteil Recruiting Créteil, France Contact: Bassam Haddad, MD bassam.haddad@chicreteil.fr Hôpital Saint Joseph Marseille Recruiting Marseille, France, 13008 Contact: Raoul Desbrières, MD raoul.desbriere@orange.fr Hopital Nord Recruiting Marseille, France, 13915 Contact: Florence Bretelle, MD,PhD florence.bretelle@ap-hm.fr CHU de Montpellier Recruiting Montpellier, France, 34295 Contact: Florent Fuchs, MD f-fuchs@chu-montpellier.fr CHRU de Nancy Recruiting Nancy, France, 54000 Contact: Olivier Morel, MD,PhD olivier.morel@chru-nancy.fr CHU Nantes Recruiting Nantes, France, 44093 Contact: Norbert Winer, MD,PhD norbert.winer@chu-nantes.fr CHU Nice Not yet recruiting Nice, France, 06202 Contact: André Bongain, MD,PhD bongain.a@chu-nice.fr CHU Nîmes Recruiting Nîmes, France, 30900 Contact: Vincent Letouzey, MD,PhD vincent.letouzey@chu-nimes.fr Hôpital Trousseau Recruiting Paris, France, 75012 Contact: Gilles Kayem, MD,PhD gkayem@gmail.com Hôpital Saint Joseph Paris Recruiting Paris, France, 75014 Contact: Elie Azria, MD eazria@hpsj.fr Maternité de Port-Royal Paris Recruiting Paris, France, 75014 Contact: François Goffinet, MD,PhD francois.goffinet@aphp.fr Hôpital universitaire Necker-Enfants malades Recruiting Paris, France, 75015 Contact: Laurent Salomon, MD,PhD laurentsalomon@gmail.com Hôpital universitaire Robert Debré Recruiting Paris, France, 75019 Contact: Thomas Schmitz, MD,PhD thomas.schmitz@aphp.fr Hôpital Lariboisière Paris Recruiting Paris, France, 75475 Contact: Jeremy Sroussi, MD jeremy.sroussi@aphp.fr Hôpital universitaire Kremlin-Bicètre Recruiting Paris, France, 94270 Contact: Marie Victoire Senat, MD,PhD marie-victoire.senat@aphp.fr CH de Pau Recruiting Pau, France Contact: Caroline BOHEC, MD caroline.bohec@ch-pau.fr Centre Hospitalier Intercommunal Poissy-Saint Germain Recruiting Poissy, France, 78303 Contact: Patrick Rozenberg, MD,PhD prozenberg@chi-poissy-st-germain.fr CHU Rennes Recruiting Rennes, France, 35033 Contact: Hélène Isly, MD,PhD helene.isly@chu-rennes.fr Principal Investigator: Maela Le Lous, MD CHU Charles Nicolle Recruiting Rouen, France, 76000 Contact: Eric Verspyck, MD,PhD Eric.Verspyck@chu-rouen.fr CHU Saint Etienne Recruiting Saint Etienne, France, 42270 Contact: Céline Chauleur, MD,PhD celine.chauleur@chu-st-etienne.fr CHU Strasbourg Recruiting Strasbourg, France, 67098 Contact: Nicolas Sananes, MD Nicolas.SANANES@chru-strasbourg.fr Contact: Bruno Langer, MD, PhD Bruno.Langer@chru-strasbourg.fr Hôpital Paule de Viguier CHU Toulouse Recruiting Toulouse, France, 31059 Contact: Paul Guerby, MD guerby.p@chu-toulouse.fr CHU Tours Recruiting Tours, France, 37044 Contact: Franck Perrotin, MD,PhD franck.perrotin@med.univ-tours.fr Collapse << |
| NCT01099579 | - | - | Completed | - | - |
| NCT00774163 | - | - | Completed | - | - |
| NCT00555321 | - | - | Terminated | - | - |
| NCT00942162 | Melanoma | Phase 2 | Completed | - | - |
| NCT00611884 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT03603665 | Botulism | Phase 1 | Recruiting | September 7, 2019 | United States, North Carolina ... more >> Duke University School of Medicine - Duke Clinical Research Institute - Duke Clinical Research Unit Recruiting Durham, North Carolina, United States, 27710-4000 Collapse << |
| NCT00611884 | - | - | Completed | - | - |
| NCT01602562 | Herpes Simplex | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Aichi, Japan, 446-8602 GSK Investigational Site Aichi, Japan, 453-8511 GSK Investigational Site Aichi, Japan, 466-8560 GSK Investigational Site Hiroshima, Japan, 734-8551 GSK Investigational Site Hyogo, Japan, 650-0047 GSK Investigational Site Hyogo, Japan, 654-0081 GSK Investigational Site Kagoshima, Japan, 890-8520 GSK Investigational Site Kanagawa, Japan, 259-1143 GSK Investigational Site Osaka, Japan, 594-1101 GSK Investigational Site Saitama, Japan, 339-8551 GSK Investigational Site Tokyo, Japan, 162-8655 Collapse << |
| NCT00942162 | - | - | Completed | - | - |
| NCT00613951 | - | - | Completed | - | - |
| NCT00613951 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | Finland ... more >> Novo Nordisk Investigational Site Helsinki, Finland, 00260 Novo Nordisk Investigational Site Kuopio, Finland, 70210 Novo Nordisk Investigational Site Lahti, Finland, 15110 Novo Nordisk Investigational Site Pori, Finland, FI-28100 France Novo Nordisk Investigational Site Bar-Le-Duc, France, 55000 Novo Nordisk Investigational Site GRENOBLE cedex, France, 38043 Novo Nordisk Investigational Site Hayange, France, 57700 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Nanterre, France, 92014 Novo Nordisk Investigational Site NEVERS cedex, France, 58033 Novo Nordisk Investigational Site Pointe à Pitre, France, 97159 Germany Novo Nordisk Investigational Site Berlin, Germany, 12163 Novo Nordisk Investigational Site Pirna, Germany, 01796 Novo Nordisk Investigational Site Riesa, Germany, 01587 Novo Nordisk Investigational Site Saarbrücken, Germany, 66121 Novo Nordisk Investigational Site St. Ingbert, Germany, 66386 Novo Nordisk Investigational Site Völklingen, Germany, 66333 Novo Nordisk Investigational Site Wangen, Germany, 88239 Poland Novo Nordisk Investigational Site Bydgoszcz, Poland, 85-822 Novo Nordisk Investigational Site Gniewkowo, Poland, 88-140 Novo Nordisk Investigational Site Nysa, Poland, 48-300 Novo Nordisk Investigational Site Plock, Poland, 09-400 Novo Nordisk Investigational Site Szczecin, Poland, 70-483 Novo Nordisk Investigational Site Tychy, Poland, 43-100 Novo Nordisk Investigational Site Warszawa, Poland, 02-507 Novo Nordisk Investigational Site Wroclaw, Poland, 50-127 Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Barcelona, Spain, 08035 Novo Nordisk Investigational Site Granada, Spain, 18012 Novo Nordisk Investigational Site Madrid, Spain, 28034 Novo Nordisk Investigational Site San Juan, Spain, 03550 Collapse << |
| NCT01602562 | - | - | Completed | - | - |
| NCT03481894 | Malnutrition | Phase 4 | Recruiting | March 2021 | United States, Illinois ... more >> The University of Chicago Medical Center Not yet recruiting Chicago, Illinois, United States, 60637 Contact: Timothy Sentongo, MD United States, Missouri Children's Mercy Hospital Recruiting Kansas City, Missouri, United States, 64108 Contact: Joel D Lim, MD Collapse << |
| NCT02853435 | Infections, Bacterial | Phase 1 | Completed | - | United States, California ... more >> GSK Investigational Site Anaheim, California, United States, 92801 United States, Texas GSK Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00479882 | - | - | Completed | - | - |
| NCT03031496 | Hypertension | Phase 1 | Completed | - | South Africa ... more >> GSK Investigational Site Bloemfontein, South Africa, 9301 Collapse << |
| NCT02853435 | - | - | Completed | - | - |
| NCT01133665 | - | - | Completed | - | - |
| NCT03031496 | - | - | Completed | - | - |
| NCT00767806 | Chronic Low Back Pain | Phase 3 | Completed | - | United States, Arizona ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chandler, Arizona, United States, 85225 United States, Connecticut For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cromwell, Connecticut, United States, 06416 United States, Florida For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Deland, Florida, United States, 32720 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jacksonville, Florida, United States, 32216 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Brighton, Massachusetts, United States, 02135 United States, Oklahoma For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Oklahoma City, Oklahoma, United States, 73109 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97210 Brazil For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Sa Coma, Brazil, 04230000 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. São Paulo, Brazil, 04026-000 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Aalen, Germany, 73430 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Alzenau, Germany, 63755 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Berlin, Germany, 10629 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ellwangen, Germany, 73479 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 20255 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wiesbaden, Germany, 65189 Netherlands For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rotterdam, Netherlands, 3039 BD Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Krakow, Poland, 30-349 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lublin, Poland, 20-093 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Poznan, Poland, 61-289 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Szczecin, Poland, 70-376 Russian Federation For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Moscow, Russian Federation, 119992 Spain For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Barcelona, Spain, 08025 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bilboa, Spain, 48013 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Getafe, Spain, 28905 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. La Coruña, Spain, 15006 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Madrid, Spain, 28009 Collapse << |
| NCT01133665 | Squamous Cell Carcinoma | Phase 2 | Completed | - | United States, Illinois ... more >> The University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT02927431 | Virus Diseases | Phase 2 | Terminated(The study was stopp... more >>ed early due to lack of enrollment.) Collapse << | - | - |
| NCT00479882 | Primary Hypercholesterolemia ... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT02927431 | - | - | Terminated(The study was stopp... more >>ed early due to lack of enrollment.) Collapse << | - | - |
| NCT00767806 | - | - | Completed | - | - |
| NCT00711009 | - | - | Completed | - | - |
| NCT01729728 | Postoperative Pain ... more >> Acute Pain Collapse << | Phase 2 | Completed | - | United States, Utah ... more >> Jean Brown Research Salt Lake City, Utah, United States, 84124 Collapse << |
| NCT00730236 | Homozygous Familial Hyperchole... more >>sterolemia Collapse << | Phase 3 | Completed | - | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States, 90048 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Canada, Ontario Robarts Research Institute London, Ontario, Canada, N6A 5K8 Canada, Quebec Lipid Clinic and University of Montreal Community Genomic Medicine Center Chicoutimi, Quebec, Canada, G7H 5H6 Italy Dipartimento di Medicina Clinica e Della Patalogie Emergenti Palermo, Sicily, Italy Medicina Interna Universitaria Ferrara, Italy Centro Universitario Dislipidemie Milano, Italy Dipartimento di Clinica e Terapia Medica Roma, Italy South Africa Cardiology Research Bloemfontein, South Africa, 9300 University of Capetown Cape town, South Africa, 7925 Collapse << |
| NCT00576420 | Adjunct to Hemostasis in Vascu... more >>lar Surgery (Synthetic Vascular Grafts) Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Birmingham, Alabama, United States United States, Arizona Tucson, Arizona, United States United States, California Long Beach, California, United States United States, Florida Jacksonville, Florida, United States United States, Kentucky Lexington, Kentucky, United States United States, Louisiana Baton Rouge, Louisiana, United States United States, Pennsylvania Pittsburgh, Pennsylvania, United States United States, Utah Salt Lake City, Utah, United States United States, Virginia Charlottesville, Virginia, United States Virginia Beach, Virginia, United States United States, Washington Seattle, Washington, United States Collapse << |
| NCT03707652 | Increase in Blood Levels of Ni... more >>cotinamide Adenine Dinucleotide (NAD+) Collapse << | Not Applicable | Completed | - | United States, Florida ... more >> Life Extension Clinical Research, Inc. Fort Lauderdale, Florida, United States, 33308 Collapse << |
| NCT00711009 | Human Immunodeficiency Virus I... more >>nfection Collapse << | Phase 3 | Completed | - | - |
| NCT00730236 | - | - | Completed | - | - |
| NCT00576420 | - | - | Completed | - | - |
| NCT00972543 | Chronic Plaque Psoriasis | Phase 4 | Terminated(The study was termi... more >>nated after the European Medicines Evaluation Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT01729728 | - | - | Completed | - | - |
| NCT01294683 | - | - | Terminated | - | - |
| NCT00972543 | - | - | Terminated(The study was termi... more >>nated after the European Medicines Evaluation Agency recommended to suspend the marketing authorisation of Raptiva in the European Union) Collapse << | - | - |
| NCT02262728 | Hepatitis C, Chronic | Phase 2 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States Collapse << |
| NCT01253811 | Congenital Bleeding Disorder ... more >> Congenital FXIII Deficiency Collapse << | Phase 3 | Completed | - | United States, Massachusetts ... more >> Novo Nordisk Clinical Trial Call Center Boston, Massachusetts, United States, 02115 United States, Ohio Novo Nordisk Clinical Trial Call Center Columbus, Ohio, United States, 43205 Israel Petach Tikva, Israel, 49100 United Kingdom Leicester, United Kingdom, LE1 5WW Collapse << |
| NCT02262728 | - | - | Completed | - | - |
| NCT01294683 | Primary Hypercholesterolemia ... more >> Dyslipidemia Collapse << | Phase 3 | Terminated | - | - |
| NCT01253811 | - | - | Completed | - | - |
| NCT00892957 | Hemostasis in Participants Rec... more >>eiving Peripheral Vascular Expanded Polytetrafluoroethylene (ePTFE) Graft Prostheses Collapse << | Phase 3 | Completed | - | - |
| NCT02041520 | HIV/AIDS | Phase 3 | Completed | - | Mexico ... more >> Instituto Mexicano del Seguro Social Leon, Guanajuato, Mexico, 37320 Collapse << |
| NCT03626662 | Cardiovascular Disease | Phase 1 | Recruiting | December 1, 2019 | United States, Florida ... more >> Research Site Recruiting South Miami, Florida, United States, 33143 Australia, Western Australia Research Site Recruiting Nedlands, Western Australia, Australia, 6009 Collapse << |
| NCT02041520 | - | - | Completed | - | - |
| NCT00707967 | Tuberculosis | Phase 2 | Completed | - | Switzerland ... more >> GSK Investigational Site Lausanne, Switzerland, 1011 Collapse << |
| NCT02664727 | Heavy Drinking ... more >> Alcohol Abuse Alcohol Dependence Alcohol Use Disorder Collapse << | Not Applicable | Completed | - | - |
| NCT00723073 | - | - | Completed | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00892957 | - | - | Completed | - | - |
| NCT00707967 | - | - | Completed | - | - |
| NCT03563222 | Malnutrition, Child | Phase 4 | Not yet recruiting | November 2020 | - |
| NCT00445146 | HIV Infections | Phase 2 | Completed | - | - |
| NCT02431767 | HIV Infections | Phase 1 | Completed | - | United States, Georgia ... more >> The Hope Clinic of the Emory Vaccine Center CRS Decatur, Georgia, United States, 30030 United States, New York University of Rochester Vaccines to Prevent HIV Infection CRS Rochester, New York, United States, 14642 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232-2582 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 Collapse << |
| NCT00445146 | - | - | Completed | - | - |
| NCT00723073 | - | - | Completed | - | - |
| NCT00120770 | HIV Infections ... more >> Gonorrhea Chlamydia Infections Collapse << | Phase 3 | Terminated(The study was halte... more >>d in January 2007 due to safety concerns.) Collapse << | - | Nigeria ... more >> Lagos University, College of Medicine, Center 10151 Lagos, Nigeria University of Port Harcourt Teaching Hospital, Center 10152 Port Harcourt, Nigeria Collapse << |
| NCT00596934 | Fatty Liver Disease, Nonalcoho... more >>lic Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT00596934 | - | - | Completed | - | - |
| NCT00950612 | Tuberculosis | Phase 2 | Completed | - | South Africa ... more >> GSK Investigational Site Worcester, Western Province, South Africa, 6850 Collapse << |
| NCT03492216 | HIV/AIDS Tube... more >>rculosis Collapse << | Not Applicable | Recruiting | February 2022 | Uganda ... more >> Infectious Disease Research Collaboration (IDRC) Recruiting Mbarara, Uganda Contact: Dalsone Kwarisiima, MBChB, MPH Mbarara Regional Referral Hospital (MRRH): Immune Suppression Syndrome HIV Recruiting Mbarara, Uganda Contact: Winnie Muyindike, MMED, MBChB Collapse << |
| NCT00122486 | HIV Infections | Phase 2 | Completed | - | Cameroon ... more >> Care and Health Program Douala, Cameroon Ghana Virtual Access Tema, Ghana Nigeria University of College Hospital Ibadan, Nigeria Collapse << |
| NCT00532948 | Glioma | Phase 1 | Completed | - | United States, California ... more >> San Francisco, California, United States, 94143-0780 United States, District of Columbia Washington, District of Columbia, United States, 20010 United States, Illinois Chicago, Illinois, United States, 60614 United States, Massachusetts Boston, Massachusetts, United States, 02115-6084 United States, North Carolina Durham, North Carolina, United States, 27710 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Pittsburgh, Pennsylvania, United States, 15261 United States, Texas Houston, Texas, United States, 77030 United States, Washington Seattle, Washington, United States, 98105 Collapse << |
| NCT00122512 | HIV Infections | Phase 2 | Terminated | - | Malawi ... more >> UNC Project, Kamuzu Central Hospital Lilongwe, Malawi Collapse << |
| NCT00950612 | - | - | Completed | - | - |
| NCT00940498 | Neoplasms | Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Insitute Boston, Massachusetts, United States, 02215 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10022 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 United States, Tennessee Sarah Cannon Research Institute, Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37203-1632 Spain Hospital General Vall D'Hebron Barcelona, Spain, 08035 United Kingdom King's College London London, England, United Kingdom, SE1 9RT Collapse << |
| NCT02500706 | Diabetes Diab... more >>etes Mellitus, Type 1 Collapse << | Phase 3 | Completed | - | - |
| NCT02825251 | Diabetes Diab... more >>etes Mellitus, Type 1 Collapse << | Phase 3 | Completed | - | - |
| NCT00413153 | HIV Infections | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00532948 | - | - | Completed | - | - |
| NCT01900184 | Essential Hypertension | Phase 1 | Completed | - | France ... more >> Biotrial PARIS Rueil-Malmaison, France, 92502 Collapse << |
| NCT02825251 | - | - | Completed | - | - |
| NCT00413153 | - | - | Completed | - | - |
| NCT01900171 | Essential Hypertension | Phase 1 | Completed | - | France ... more >> Biotrial PARIS Rueil-Malmaison, France, 92502 Collapse << |
| NCT00570440 | Pregnancy Prevention | Phase 4 | Completed | - | Dominican Republic ... more >> PROFAMILIA - Santo Domingo, Dominican Republic Ens. Luperon, Santo Domingo, Dominican Republic Nicaragua PROFAMILIA - Managua, Nicaragua Managua, Nicaragua Collapse << |
| NCT00940498 | - | - | Completed | - | - |
| NCT00795600 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | Spain ... more >> Málaga, Spain, 29009 Collapse << |
| NCT01675427 | Hepatitis C, Chronic | Phase 4 | Completed | - | - |
| NCT02500706 | - | - | Completed | - | - |
| NCT02635945 | Asthma | Phase 2 | Recruiting | November 2019 | Spain ... more >> Unitat de Pneumologia Experimental Recruiting Barcelona, Spain, 08025 Principal Investigator: David Ramos Barbón, MD, PhD Collapse << |
| NCT00795600 | - | - | Completed | - | - |
| NCT02513121 | Nonalcoholic Fatty Liver Disea... more >>se Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92093 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01463254 | - | - | Completed | - | Pakistan ... more >> Marie Stopes Society clinics Pakistan, Pakistan Collapse << |
| NCT00972725 | AIDS | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Collapse << |
| NCT01715051 | Cocaine Dependence | Phase 2 | Withdrawn(Funding was not rece... more >>ived) Collapse << | September 2019 | United States, Ohio ... more >> Maryhaven, Inc Columbus, Ohio, United States, 43207 Collapse << |
| NCT01610791 | Rheumatoid Arthritis | Phase 3 | Completed | - | Morocco ... more >> Casablanca, Morocco, 20000 Fés, Morocco, 30000 Khouribga, Morocco, 14000 Kénitra, Morocco, 25000 Marrakech, Morocco, 40000 Meknés, Morocco, 50000 Rabat, Morocco, 10000 Salé, Morocco, 15045 Collapse << |
| NCT01610791 | - | - | Completed | - | - |
| NCT02720081 | Asthma | Phase 2 | Completed | - | - |
| NCT01226433 | - | - | Completed | - | Kenya ... more >> 5 Community Outreach Events Various, Rift Valley & Central, Kenya Collapse << |
| NCT01334190 | - | - | Completed | - | Kenya ... more >> FHI Nairobi, Kenya Collapse << |
| NCT02720081 | - | - | Completed | - | - |
| NCT01218113 | AIDS | Phase 2 | Completed | - | - |
| NCT00972725 | - | - | Completed | - | - |
| NCT01452503 | Contraception | Not Applicable | Completed | - | South Africa ... more >> Commercial City Clinic Department of Health Durban, South Africa, 4001 Collapse << |
| NCT01340599 | Stage I Prostate Cancer ... more >> Stage IIA Prostate Cancer Stage IIB Prostate Cancer Collapse << | Phase 2 | Terminated(Slow accrual) | - | United States, Ohio ... more >> Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Collapse << |
| NCT01675427 | - | - | Completed | - | - |
| NCT01560845 | Liver Cirrhosis ... more >> Portal Hypertension Hepatic Decompensation Surgery Collapse << | Phase 2 Phase 3 | Unknown | June 2014 | China, Zhejiang ... more >> the First Affiliated Hospital of Wenzhou Medical College Recruiting Wenzhou, Zhejiang, China, 325000 Contact: qiqiang zeng, MD 86-0577-88069307 zengqiqiangwz@163.com Principal Investigator: qiqiang zeng, MD Principal Investigator: yi liao, MD Principal Investigator: minghua zheng, MD Principal Investigator: bing liang, MD Principal Investigator: zhiming li, MD Principal Investigator: qigang xu, MD Principal Investigator: yi wang, MD Principal Investigator: zhengping yu, MD Principal Investigator: hongqi shi, MD Collapse << |
| NCT02929498 | Myelodysplastic Syndrome | Phase 2 | Recruiting | May 15, 2019 | United States, Pennsylvania ... more >> GSK Investigational Site Recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01594632 | Contraception | Not Applicable | Completed | - | Dominican Republic ... more >> Profamilia Santo Domingo, Dominican Republic Collapse << |
| NCT00775437 | - | - | Completed | - | - |
| NCT01487876 | Chronic Hepatitis B | Phase 2 | Unknown | December 2012 | China, Bejing ... more >> Guiqiang Wang Recruiting Beijing, Bejing, China, 10000 Contact: Guiqiang Wang, PhD 00861066551122 ext 2597 wanggq@hotmail.com Principal Investigator: Guiqiang Wang, PhD Collapse << |
| NCT01667276 | - | - | Completed | - | Senegal ... more >> Ministry of Health Clinics All Cities Where There Is A Clinic, Senegal Uganda Ministry of Health Clinics, Uganda All Cities Where There Is A Clinic, Uganda Collapse << |
| NCT01721798 | HIV | Not Applicable | Completed | - | South Africa ... more >> University of Cape Town Cape Town, Western Cape Province, South Africa, 7701 Collapse << |
| NCT02547974 | Respiratory Disorders | Phase 1 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 GSK Investigational Site Leuven, Belgium, 3000 GSK Investigational Site Wilrijk, Belgium, 2610 Collapse << |
| NCT01808352 | HIV | Phase 4 | Completed | - | United States, California ... more >> UCLA Vine Street Clinic Los Angeles, California, United States, 90095 United States, District of Columbia George Washington University CRS Washington, District of Columbia, United States, 20007 United States, North Carolina UNC CRS Chapel Hill, North Carolina, United States, 27514 Collapse << |
| NCT01903889 | HIV | Not Applicable | Completed | - | Uganda ... more >> Bukinda Health Center Kabale, Uganda Kamkira Health Center Kabale, Uganda Kamwezi Health Center Kabale, Uganda Maziba Health Center Kabale, Uganda Mparo Health Center Kabale, Uganda Muko Health Center Kabale, Uganda Rubaya Health Center Kabale, Uganda Collapse << |
| NCT02547974 | - | - | Completed | - | - |
| NCT01218113 | - | - | Completed | - | - |
| NCT00397943 | Tuberculosis | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Collapse << |
| NCT00775437 | Juvenile Idiopathic Arthritis | Phase 3 | Completed | - | - |
| NCT02467413 | Alzheimer's Disease ... more >> Vascular Dementia Collapse << | Phase 2 | Not yet recruiting | December 2021 | Taiwan ... more >> National Cheng Kung University Hospital Not yet recruiting Tainan, Taiwan, 704 Contact: Ming-Chyi Pai, PhD +886-6-2353535 ext 5534 pair@mail.ncku.edu.tw Principal Investigator: Ming-Chyi Pai, MD, PhD Collapse << |
| NCT00621322 | Tuberculosis | Phase 2 | Completed | - | Philippines ... more >> GSK Investigational Site Manila, Philippines, 1000 Collapse << |
| NCT00791648 | - | - | Completed | - | - |
| NCT00791648 | Acute Kidney Injury ... more >> Post-Operative Delirium Icu Delirium Acute Renal Failure Delirium Collapse << | Not Applicable | Completed | - | United States, Tennessee ... more >> Vanderbilt University Medical Center Nashville, Tennessee, United States, 37212 Collapse << |
| NCT00397943 | - | - | Completed | - | - |
| NCT01432587 | - | - | Completed | - | Sweden ... more >> Dept of Clinical Medicin, Ptieå Hospital Piteå, Sweden, SE-941 28 Dept of clinical medicin, Skellefteå Hospital Skellefteå, Sweden, SE-931 86 Dept of Clinical Medicine, Umeå University Hospital Umeå, Sweden, SE-90185 Collapse << |
| NCT00621322 | - | - | Completed | - | - |
| NCT02177812 | Leukaemia, Myelocytic, Acute | Phase 1 | Terminated(The risk benefit in... more >> relapsed refractory AML does not favor continuation of the study.) Collapse << | - | United States, Georgia ... more >> GSK Investigational Site Augusta, Georgia, United States, 30912 United States, New York GSK Investigational Site Bronx, New York, United States, 10461 GSK Investigational Site Bronx, New York, United States, 10467 GSK Investigational Site New York, New York, United States, 10065 United States, Texas GSK Investigational Site Houston, Texas, United States, 77030 Australia, Victoria GSK Investigational Site Melbourne, Victoria, Australia, 3004 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M5G 2M9 GSK Investigational Site Toronto, Ontario, Canada, M5G 2M Collapse << |
| NCT01556906 | Homozygous Familial Hyperchole... more >>sterolemia Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT02075541 | Respiratory Disorders | Phase 2 | Completed | - | Sweden ... more >> GSK Investigational Site Eskilstuna, Sweden, SE-631 88 GSK Investigational Site Göteborg, Sweden, SE-413 45 GSK Investigational Site Örebro, Sweden, SE-703 62 United Kingdom GSK Investigational Site Llanelli, Carmarthenshire, United Kingdom, SA14 8QF GSK Investigational Site Leicester, Leicestershire, United Kingdom, LE3 9QP GSK Investigational Site Stoke on Trent, Staffordshire, United Kingdom, ST4 6QG GSK Investigational Site Bradford, United Kingdom, BD9 6RJ GSK Investigational Site Dundee, United Kingdom, DD1 9SY GSK Investigational Site Edinburgh, United Kingdom, EH16 4SA GSK Investigational Site Liverpool, United Kingdom, L9 7AL GSK Investigational Site Poole, Dorset, United Kingdom, BH15 2JB GSK Investigational Site Salford, United Kingdom, M6 8HD GSK Investigational Site Southampton, United Kingdom, SO16 6YD GSK Investigational Site Wolverhampton, United Kingdom, WV10 0QP Collapse << |
| NCT01556906 | - | - | Completed | - | - |
| NCT01990794 | - | - | Completed | - | - |
| NCT00530790 | - | - | Completed | - | - |
| NCT03598283 | - | - | Recruiting | June 30, 2020 | United States, Texas ... more >> University of Texas Medical Branch Recruiting Galveston, Texas, United States, 77550 Contact: Christian Tapking, MD 409-770-6964 chtapkin@utmb.edu Collapse << |
| NCT01462877 | Dyslipidemias ... more >> Cardiovascular Diseases Hypertriglyceridemia Collapse << | Phase 4 | Completed | - | China ... more >> Site Reference ID/Investigator# 64695 Xiamen, China, 36100 Collapse << |
| NCT01990794 | - | - | Completed | - | - |
| NCT01462877 | - | - | Completed | - | - |
| NCT01165203 | Herpes Zoster | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Long Beach, California, United States, 90813 GSK Investigational Site San Francisco, California, United States, 94115 United States, Florida GSK Investigational Site Orlando, Florida, United States, 32803 United States, New Mexico GSK Investigational Site Santa Fe, New Mexico, United States, 87505 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75246 Germany GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Bochum, Nordrhein-Westfalen, Germany, 44791 GSK Investigational Site Duesseldorf, Nordrhein-Westfalen, Germany, 40237 GSK Investigational Site Essen, Nordrhein-Westfalen, Germany, 45122 GSK Investigational Site Berlin, Germany, 13353 GSK Investigational Site Hamburg, Germany, 20146 United Kingdom GSK Investigational Site Woolwich, London, London, United Kingdom, SE18 4QH GSK Investigational Site London, United Kingdom, E1 1BB GSK Investigational Site London, United Kingdom, NW3 2QG GSK Investigational Site London, United Kingdom, SW10 9TH Collapse << |
| NCT01165203 | - | - | Completed | - | - |
| NCT02075541 | - | - | Completed | - | - |
| NCT01202253 | - | - | Completed | - | United Kingdom ... more >> Pfizer Investigational Site London, United Kingdom, SE5 9RS Collapse << |
| NCT00531206 | - | - | Completed | - | - |
| NCT00530790 | Restless Legs Syndrome | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 802-0084 GSK Investigational Site Fukuoka, Japan, 810-0044 GSK Investigational Site Fukuoka, Japan, 830-0011 GSK Investigational Site Hiroshima, Japan, 733-0031 GSK Investigational Site Kanagawa, Japan, 210-0024 GSK Investigational Site Osaka, Japan, 550-0004 GSK Investigational Site Osaka, Japan, 589-0022 GSK Investigational Site Osaka, Japan, 599-8263 GSK Investigational Site Tochigi, Japan, 321-0293 GSK Investigational Site Tokyo, Japan, 151-0053 GSK Investigational Site Tokyo, Japan, 187-0041 Collapse << |
| NCT02663453 | Cholestasis | Phase 3 | Completed | - | - |
| NCT01202253 | - | - | Completed | - | - |
| NCT02663453 | - | - | Completed | - | - |
| NCT00531206 | - | - | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Aachen, Germany Boehringer Ingelheim Investigational Site Berlin, Germany Boehringer Ingelheim Investigational Site Bremen, Germany Boehringer Ingelheim Investigational Site Dortmund, Germany Boehringer Ingelheim Investigational Site Düsseldorf, Germany Boehringer Ingelheim Investigational Site Erlangen, Germany Boehringer Ingelheim Investigational Site Frankfurt/Main, Germany Boehringer Ingelheim Investigational Site Freiburg, Germany Boehringer Ingelheim Investigational Site Gießen, Germany Boehringer Ingelheim Investigational Site Halle/Saale, Germany Boehringer Ingelheim Investigational Site Hamburg, Germany Boehringer Ingelheim Investigational Site Hannover, Germany Boehringer Ingelheim Investigational Site Homburg/Saar, Germany Boehringer Ingelheim Investigational Site Karlsruhe, Germany Boehringer Ingelheim Investigational Site Krefeld, Germany Boehringer Ingelheim Investigational Site Köln, Germany Boehringer Ingelheim Investigational Site Leipzig, Germany Boehringer Ingelheim Investigational Site Magdeburg, Germany Boehringer Ingelheim Investigational Site Mainz, Germany Boehringer Ingelheim Investigational Site München, Germany Boehringer Ingelheim Investigational Site Münster, Germany Boehringer Ingelheim Investigational Site Nürnberg, Germany Boehringer Ingelheim Investigational Site Oldenburg, Germany Boehringer Ingelheim Investigational Site Osnabrück, Germany Boehringer Ingelheim Investigational Site Saarbrücken, Germany Boehringer Ingelheim Investigational Site Stuttgart, Germany Boehringer Ingelheim Investigational Site Wuppertal, Germany Collapse << |
| NCT02372175 | Dengue | Phase 1 | Recruiting | October 2019 | United States, New York ... more >> State University of New York, Upstate Medical University (SUNY-UMU) Recruiting Syracuse, New York, United States, 13210 Contact: Timothy P Endy, MD, MPH 315-464-8668 endyt@upstate.edu Principal Investigator: Timothy P Endy, MD, MPH Sub-Investigator: Mark Polhemus, MD Sub-Investigator: Ambika Eranki, MD Sub-Investigator: Kimberly Amodio, PA Sub-Investigator: Jennifer Knutsen, PA Sub-Investigator: Donald Blair, MD Collapse << |
| NCT02177136 | Primary Sclerosing Cholangitis... more >> (PSC) Collapse << | Phase 2 | Completed | - | - |
| NCT02177136 | - | - | Completed | - | - |
| NCT02817620 | Metabolic Syndrome | Not Applicable | Completed | - | France ... more >> Biofortis Mérieux NutriSciences Clinical Investigation Center Saint Herblain, France, 44800 Collapse << |
| NCT00310388 | Epilepsy | Phase 3 | Terminated(No longer any benef... more >>it in collecting data since retigabine has been withdrawn from market.) Collapse << | - | - |
| NCT00063635 | - | - | Completed | - | - |
| NCT01822821 | Pain | Not Applicable | Completed | - | United States, Ohio ... more >> Cleveland Clinic Cleveland, Ohio, United States, 44195 Collapse << |
| NCT00063635 | Fatty Liver | Phase 3 | Completed | - | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94143 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 United States, Missouri St. Louis University St. Louis, Missouri, United States, 63110 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44109 United States, Texas Texas Children's Hospital Houston, Texas, United States, 77030 United States, Virginia Virginia Commonwealth University Richmond, Virginia, United States, 23298 United States, Washington University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01822821 | - | - | Completed | - | - |
| NCT01610011 | - | - | Completed | - | - |
| NCT01074359 | Neuromuscular Disease | Phase 2 | Terminated(Terminated for Comm... more >>ercial Reasons. There were no safety issues involved in the decision to terminate the study.) Collapse << | - | United Kingdom ... more >> University of Newcastle upon Tyne Newcastle, Framlington Place, United Kingdom, NE2 4HH Collapse << |
| NCT01923480 | Parenteral Nutrition (No Prima... more >>ry Condition Studied) Collapse << | Phase 4 | Terminated | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 United States, Texas Texas A&M University, Center for Translational Research in Aging & Longevity College Station, Texas, United States, 77843-4253 Collapse << |
| NCT00920218 | Herpes Zoster | Phase 2 | Completed | - | United States, Arkansas ... more >> GSK Investigational Site Little Rock, Arkansas, United States, 72205 United States, California GSK Investigational Site Duarte, California, United States, 91010 GSK Investigational Site Frisco, California, United States, 94143 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02115 United States, Minnesota GSK Investigational Site Minnesota, Minnesota, United States, 55455 GSK Investigational Site Rochester, Minnesota, United States, 55905 United States, New York GSK Investigational Site New York, New York, United States, 10065 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599 GSK Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44195 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT02874586 | Hepatitis, Autoimmune | Not Applicable | Unknown | September 2017 | China, Sichuan ... more >> West China Hospital Recruiting Chengdu, Sichuan, China, 610041 Contact: Li Yang, MD +8618980601276 yangli_hx@scu.edu.cn Collapse << |
| NCT02472509 | Hemolytic Disorders ... more >> Gallstones Collapse << | Phase 4 | Terminated(Study not feasible ... more >>- very low recruitment rate) Collapse << | - | Israel ... more >> Shaare Zedek Medical Center Jerusalem, Israel, 91031 Collapse << |
| NCT01610011 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> McLean Imaging Center, McLean Hospital Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT02760901 | Cisplatin Nephrotoxicity | Phase 2 | Completed | - | - |
| NCT00310388 | - | - | Terminated(No longer any benef... more >>it in collecting data since retigabine has been withdrawn from market.) Collapse << | - | - |
| NCT00836719 | Multiple Sclerosis | Phase 1 | Completed | - | United States, Louisiana ... more >> Louisiana Health Sciences Center New Orleans New Orleans, Louisiana, United States, 70112 Collapse << |
| NCT01923480 | - | - | Terminated | - | - |
| NCT00836719 | - | - | Completed | - | - |
| NCT01397227 | Malaria | Phase 1 Phase 2 | Completed | - | United States, Washington ... more >> Malaria Clinical Trials Center at Seattle Biomedical Research Institute Seattle, Washington, United States, 98109 Collapse << |
| NCT00920218 | - | - | Completed | - | - |
| NCT02491463 | Respiratory Synctial Virus Inf... more >>ections Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Oxford, Oxfordshire, United Kingdom, OX3 7LJ Collapse << |
| NCT00244725 | Deep Vein Thrombosis ... more >> Fibrillation, Atrial Venous Thromboembolism Pulmonary Embolism Collapse << | Phase 2 | Completed | - | - |
| NCT03612856 | End Stage Renal Disease ... more >> Thrombosis Collapse << | Phase 2 | Recruiting | May 2020 | United States, Florida ... more >> Orlando Clinical Research Center Recruiting Orlando, Florida, United States, 32809 Contact: Charles Johnson 407-240-7876 Collapse << |
| NCT02491463 | - | - | Completed | - | - |
| NCT00244725 | - | - | Completed | - | - |
| NCT00488514 | Migraine Disorders | Phase 3 | Completed | - | - |
| NCT00646776 | Antivirals/HIV | Phase 1 | Completed | - | United States, New Jersey ... more >> Bristol-Myers Squibb Clinical Pharmacology Unit Hamilton, New Jersey, United States, 08690 Collapse << |
| NCT03122223 | HIV Infections | Phase 1 Phase 2 | Active, not recruiting | September 30, 2019 | United States, California ... more >> Bridge HIV CRS San Francisco, California, United States, 94143 United States, Georgia The Hope Clinic of the Emory Vaccine Center CRS Decatur, Georgia, United States, 30030 United States, Massachusetts Brigham and Women's Hospital Vaccine CRS (BWH VCRS) Boston, Massachusetts, United States, 02115-6110 Fenway Health (FH) CRS Boston, Massachusetts, United States, 02215-4302 United States, New York University of Rochester Vaccines to Prevent HIV Infection CRS Rochester, New York, United States, 14642 United States, Ohio Case Clinical Research Site Cleveland, Ohio, United States, 44106 United States, Pennsylvania Penn Prevention CRS Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232-2582 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 Tanzania National Institute for Medical Research (NIMR) - Mbeya Medical Research Center (MMRC) Network CRS Mbeya, Tanzania Zambia Matero Reference Clinic CRS Lusaka, Zambia, 10101 Zimbabwe Seke South CRS Chitungwiza, Zimbabwe Collapse << |
| NCT02550912 | Autism | Phase 2 | Completed | - | Egypt ... more >> Faculty of Pharmacy AinShams University Cairo, Egypt, 02 Collapse << |
| NCT03693612 | Neoplasms | Phase 1 Phase 2 | Recruiting | June 16, 2022 | United States, Massachusetts ... more >> GSK Investigational Site Not yet recruiting Boston, Massachusetts, United States, 02215 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com United States, New York GSK Investigational Site Not yet recruiting New York, New York, United States, 10016- Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting New York, New York, United States, 10032 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com United States, Oklahoma GSK Investigational Site Not yet recruiting Oklahoma City, Oklahoma, United States, 73104 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com United States, Pennsylvania GSK Investigational Site Not yet recruiting Pittsburgh, Pennsylvania, United States, 15232 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com United States, Texas GSK Investigational Site Recruiting San Antonio, Texas, United States, 78240 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Principal Investigator: Anthony Tolcher Australia, New South Wales GSK Investigational Site Not yet recruiting Darlinghurst, New South Wales, Australia, 2010 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Australia, Victoria GSK Investigational Site Not yet recruiting Melbourne, Victoria, Australia, 3000 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Canada, Ontario GSK Investigational Site Not yet recruiting Ottawa, Ontario, Canada, K1H 8L Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Toronto, Ontario, Canada, M5G 2M Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00393484 | Chronic Hepatitis B | Phase 4 | Completed | - | Korea, Republic of ... more >> Local Institution Bucheon, Korea, Republic of, 420-717 Local Institution Busan, Korea, Republic of, 602-739 Local Institution Chuncheon, Korea, Republic of, 200-704 Local Institution Daegu, Korea, Republic of, 700-721 Local Institution Daegu, Korea, Republic of, 705-030 Local Institution Daejeon, Korea, Republic of, 301-721 Local Institution Guri, Korea, Republic of, 471-020 Local Institution Jeonju, Korea, Republic of, 561-180 Local Institution Seoul, Korea, Republic of, 130-702 Local Institution Seoul, Korea, Republic of, 135-270 Local Institution Seoul, Korea, Republic of, 135-710 Local Institution Seoul, Korea, Republic of, 136-705 Local Institution Seoul, Korea, Republic of, 138-040 Local Institution Seoul, Korea, Republic of, 150-950 Local Institution Seoul, Korea, Republic of, 152-050 Collapse << |
| NCT00488514 | - | - | Completed | - | - |
| NCT00646776 | - | - | Completed | - | - |
| NCT02915016 | HIV Infections | Phase 1 Phase 2 | Active, not recruiting | - | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, Massachusetts Brigham and Women's Hospital Vaccine CRS (BWH VCRS) Boston, Massachusetts, United States, 02115-6110 Fenway Health (FH) CRS Boston, Massachusetts, United States, 02215-4302 United States, New York Columbia P&S CRS New York, New York, United States, 10032-3732 New York Blood Center CRS New York, New York, United States, 10065 University of Rochester Vaccines to Prevent HIV Infection CRS Rochester, New York, United States, 14642 United States, Tennessee Vanderbilt Vaccine (VV) CRS Nashville, Tennessee, United States, 37232-2582 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 South Africa Kliptown Soweto CRS Johannesburg, Gauteng, South Africa, 1409 Aurum Tembisa CRS Johannesburg, Gauteng, South Africa, 1632 Soweto HVTN CRS Johannesburg, Gauteng, South Africa, 1862 Setshaba Research Centre CRS Soshanguve, Gauteng, South Africa, 0152 CAPRISA eThekwini CRS Durban, Kwa Zulu Natal, South Africa, 4013 Qhakaza Mbokodo Research Clinic CRS Ladysmith, Kwa Zulu Natal, South Africa, 3370 Verulam CRS Verulam, Kwa Zulu Natal, South Africa, 4340 Aurum Institute Klerksdorp CRS Klerksdorp, North West Province, South Africa, 2571 Emavundleni CRS Cape Town, Western Cape, South Africa, 7750 Collapse << |
| NCT00393484 | - | - | Completed | - | - |
| NCT00972517 | Influenza | Phase 3 | Completed | - | Germany ... more >> GSK Investigational Site Kehl, Baden-Wuerttemberg, Germany, 77694 GSK Investigational Site Pforzheim, Baden-Wuerttemberg, Germany, 75172 GSK Investigational Site Schwaebisch-Hall, Baden-Wuerttemberg, Germany, 74523 GSK Investigational Site Stuttgart, Baden-Wuerttemberg, Germany, 70469 GSK Investigational Site Bindlach, Bayern, Germany, 95463 GSK Investigational Site Cham, Bayern, Germany, 93413 GSK Investigational Site Muenchen, Bayern, Germany, 81241 GSK Investigational Site Noerdlingen, Bayern, Germany, 86720 GSK Investigational Site Frankenthal, Rheinland-Pfalz, Germany, 67227 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Worms, Rheinland-Pfalz, Germany, 67547 Collapse << |
| NCT02277691 | Essential Hypertension | Phase 3 | Completed | - | Japan ... more >> Nagoya-shi, Aichi, Japan Chiba-shi, Chiba, Japan Itojima-shi, Fukuoka, Japan Kouriyama-shi, Fukushima, Japan Sapporo-shi, Hokkaido, Japan Amagasaki-shi, Hyougo, Japan Tsukuba-shi, Ibaragi, Japan Morioka-shi, Iwate, Japan Sakaide-shi, Kagawa, Japan Takamatsu-shi, Kagawa, Japan Kawasaki-shi, Kanagawa, Japan Kyoto-shi, Kyoto, Japan Uji-shi, Kyoto, Japan Sendai-shi, Miyagi, Japan Hirakata-shi, Osaka, Japan Osaka-shi, Osaka, Japan Takatsuki-shi, Osaka, Japan Saitama-shi, Saitama, Japan Tokorozawa-shi, Saitama, Japan Yaizu-shi, Shizuoka, Japan Chiyoda-ku, Tokyo, Japan Choufu-shi, Tokyo, Japan Kodaira-shi, Tokyo, Japan Koutou-ku, Tokyo, Japan Setagaya-ku, Tokyo, Japan Shinagawa-ku, Tokyo, Japan Shinjuku-ku, Tokyo, Japan Choufu-shi, Japan Kawasaki-shi, Japan Koutou-ku, Japan Morioka-shi, Japan Sakaide-shi, Japan Setagaya-ku, Japan Shinagawa-ku, Japan Shinjuku-ku, Japan Tsukuba-shi, Japan Uji-shi, Japan Yaizu-shi, Japan Collapse << |
| NCT00972517 | - | - | Completed | - | - |
| NCT02997969 | HIV Infections | Phase 1 | Unknown | September 2017 | South Africa ... more >> Aurum Tembisa CRS Recruiting Johannesburg, Gauteng, South Africa, 1632 Contact: Modulakgotla Sebe 27-87-1351645 msebe@auruminstitute.org Contact: Gladys Kobane 27-87-1351533 gkobane@auruminstitute.org Isipingo CRS Recruiting Westville, Kwa Zulu Natal, South Africa, 3630 Contact: Arendevi Pather 27-31-9027494 Arendevi.Pather@mrc.ac.za Contact: Girisha Kistnasami, BSc, DPM 27-31-9027494 girisha.kistnasami@mrc.ac.za Aurum Institute Klerksdorp CRS Recruiting Klerksdorp, North West Province, South Africa, 2571 Contact: Craig Innes 27-87-1351587 cinnes@auruminstitute.org Contact: Tania Adonis 27-87-1351587 tadonis@auruminstitute.org Tanzania National Institute for Medical Research (NIMR) - Mbeya Medical Research Center (MMRC) Network CRS Recruiting Mbeya, Tanzania Contact: Nyanda Elias nelias@nimr-mmrc.org Contact: Lucas Maganga 255-25-2503364 lmaganga@nimr-mmrc.org Zambia Matero Reference Clinic CRS Recruiting Lusaka, Zambia Contact: Stewart Reid 260-966-747677 stewart.reid@cidrz.org Contact: Bupe N Sichalwe 260-966848889 bupe.sichalwe@cidrz.org Collapse << |
| NCT00510146 | - | - | Completed | - | - |
| NCT03234036 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00510146 | Depression, Bipolar | Phase 3 | Completed | - | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100088 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changsha, China, 410008 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chengdu, China, 610041 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Guang Zhou, China, 510370 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310003 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Harbin, China, 150001 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kunming, China, 650032 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Nanjing, China, 210029 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shanghai, China, 200030 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Wu Han, China, 430060 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Xi'An, China, 710032 Japan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hiroshima, Japan, 731-0501 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Shiga, Japan, 525-0037 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tokyo, Japan, 170-0002 Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seongnam-Si, Korea, Republic of, 463-707 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT02277691 | - | - | Completed | - | - |
| NCT00603746 | Asthma | Phase 2 | Completed | - | - |
| NCT02689206 | Anaemia | Phase 2 | Completed | - | - |
| NCT00603746 | - | - | Completed | - | - |
| NCT00603278 | Asthma | Phase 2 | Completed | - | - |
| NCT02689206 | - | - | Completed | - | - |
| NCT02927210 | Healthy Men M... more >>ale Contraception Collapse << | Phase 1 | Recruiting | October 2019 | United States, California ... more >> Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center Recruiting Torrance, California, United States, 90509 Contact: Xiaodan Han 310-222-1865 xhan@labiomed.org Principal Investigator: Christina Wang, MD United States, Washington University of Washington Medical Center & Health Sciences Recruiting Seattle, Washington, United States, 98195 Contact: Kathy Winter 206-616-0484 klwinter@uw.edu Principal Investigator: Stephanie Page, MD, PhD Collapse << |
| NCT00603278 | - | - | Completed | - | - |
| NCT00603382 | - | - | Completed | - | - |
| NCT00603382 | Asthma | Phase 2 | Completed | - | - |
| NCT00141102 | Osteoarthritis ... more >> Arthritis, Rheumatoid Collapse << | Phase 4 | Completed | - | - |
| NCT01244984 | Asthma | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 277-0863 GSK Investigational Site Fukuoka, Japan, 802-0083 GSK Investigational Site Fukuoka, Japan, 816-0813 GSK Investigational Site Gifu, Japan, 501-6062 GSK Investigational Site Gunma, Japan, 373-0021 GSK Investigational Site Hiroshima, Japan, 730-0844 GSK Investigational Site Hiroshima, Japan, 732-0062 GSK Investigational Site Hiroshima, Japan, 739-0402 GSK Investigational Site Hyogo, Japan, 665-0827 GSK Investigational Site Hyogo, Japan, 670-0046 GSK Investigational Site Hyogo, Japan, 672-8064 GSK Investigational Site Ibaraki, Japan, 302-0022 GSK Investigational Site Kanagawa, Japan, 231-8682 GSK Investigational Site Kanagawa, Japan, 253-0041 GSK Investigational Site Kyoto, Japan, 601-1495 GSK Investigational Site Kyoto, Japan, 615-8087 GSK Investigational Site Miyagi, Japan, 983-0824 GSK Investigational Site Miyagi, Japan, 983-8520 GSK Investigational Site Nagano, Japan, 390-0303 GSK Investigational Site Nagano, Japan, 390-8510 GSK Investigational Site Okayama, Japan, 700-0862 GSK Investigational Site Okayama, Japan, 701-0304 GSK Investigational Site Okayama, Japan, 714-0081 GSK Investigational Site Osaka, Japan, 545-8586 GSK Investigational Site Osaka, Japan, 569-1192 GSK Investigational Site Osaka, Japan, 589-0022 GSK Investigational Site Tokyo, Japan, 105-0004 GSK Investigational Site Tokyo, Japan, 134-0083 GSK Investigational Site Tokyo, Japan, 185-0014 GSK Investigational Site Toyama, Japan, 937-0066 Collapse << |
| NCT00141102 | - | - | Completed | - | - |
| NCT01244984 | - | - | Completed | - | - |
| NCT01301456 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Metabolic Diseases Endocrine System Diseases Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 Comprehensive Phase One (A Division of Comprehensive NeuroScience, Inc.) Miramar, Florida, United States, 33025 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Nebraska ICON Clinical Pharmacology, LLC Omaha, Nebraska, United States, 68154 United States, Pennsylvania CRI Worldwide, LLC Philadelphia, Pennsylvania, United States, 19139 United States, Texas Healthcare Discoveries LLC d/b/a ICON Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT00434304 | Parkinson Disease | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Aichi, Japan, 455-8530 GSK Investigational Site Aichi, Japan, 460-0008 GSK Investigational Site Chiba, Japan, 279-0021 GSK Investigational Site Ehime, Japan, 791-0295 GSK Investigational Site Hokkaido, Japan, 070-0901 GSK Investigational Site Iwate, Japan, 020-0878 GSK Investigational Site Kanagawa, Japan, 251-0038 GSK Investigational Site Kyoto, Japan, 600-8811 GSK Investigational Site Osaka, Japan, 570-8507 GSK Investigational Site Saitama, Japan, 343-0032 GSK Investigational Site Tokyo, Japan, 113-8431 GSK Investigational Site Tokyo, Japan, 136-0075 Collapse << |
| NCT03628079 | Cancer of the Gastrointestinal... more >> Tract Cancer of Unknown Origin Collapse << | Phase 1 Phase 2 | Recruiting | June 2020 | Sweden ... more >> Dept of oncology, University Hospital Recruiting Uppsala, Sweden, 75185 Collapse << |
| NCT00289900 | Mixed Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT00434304 | - | - | Completed | - | - |
| NCT02187029 | Gout | Phase 1 | Terminated(B7911002 was premat... more >>urely discontinued due to a safety concern that led to decision to terminate the study on 10 Nov 2014) Collapse << | - | United States, Florida ... more >> MRA Clinical Research, LLC Miami, Florida, United States, 33143 Miami Research Associates, Inc. South Miami, Florida, United States, 33143 United States, Kansas Vince and Associates Clinical Research Inc. Overland Park, Kansas, United States, 66211 Vince and Associates Clinical Research, Inc. Overland Park, Kansas, United States, 66212 Collapse << |
| NCT01301456 | - | - | Completed | - | - |
| NCT00289900 | - | - | Completed | - | - |
| NCT01362244 | - | - | Completed | - | - |
| NCT00371254 | - | - | Completed | - | - |
| NCT01702740 | Lupus Erythematosus, Cutaneous... more >> Lupus Erythematosus, Systemic Collapse << | Phase 1 | Completed | - | - |
| NCT00516139 | - | - | Completed | - | - |
| NCT01362244 | Nasal Polyps | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Netherlands GSK Investigational Site Amsterdam, Netherlands, 1105 AZ United Kingdom GSK Investigational Site Cambridge, United Kingdom GSK Investigational Site London, United Kingdom, EC1M 6BQ GSK Investigational Site London, United Kingdom, SE1 1YR GSK Investigational Site London, United Kingdom, SW3 6NP GSK Investigational Site London, United Kingdom, WC1X 8DA Collapse << |
| NCT00371254 | Breast Cancer ... more >> Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Ucsf-Comprehensive Cancer Center San Francisco, California, United States, 94143 United States, Florida Mayo Clinic Jacksonville Jacksonville, Florida, United States, 32224 United States, Massachusetts Dana-Farber Cancer Inst Boston, Massachusetts, United States, 02115 United States, New York Montefiore Medical Center Bronx, New York, United States, 10461 United States, Texas University Of Texas Md Anderson Cancer Ctr Houston, Texas, United States, 77030 France Local Institution Paris, France, 75231 Local Institution Toulouse Cedex 3, France, 31052 Italy Local Institution Modena, Italy, 41100 Spain Local Institution Barcelona, Spain, 08035 Local Institution Lleida, Spain, 25198 Collapse << |
| NCT02187029 | - | - | Terminated(B7911002 was premat... more >>urely discontinued due to a safety concern that led to decision to terminate the study on 10 Nov 2014) Collapse << | - | - |
| NCT01376388 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 802-0083 GSK Investigational Site Gunma, Japan, 371-0048 GSK Investigational Site Hiroshima, Japan, 732-0052 GSK Investigational Site Hiroshima, Japan, 732-0057 GSK Investigational Site Hokkaido, Japan, 064-0915 GSK Investigational Site Hokkaido, Japan, 080-0805 GSK Investigational Site Hyogo, Japan, 665-0827 GSK Investigational Site Hyogo, Japan, 670-0849 GSK Investigational Site Ibaraki, Japan, 300-0053 GSK Investigational Site Kagawa, Japan, 763-8502 GSK Investigational Site Kanagawa, Japan, 239-0821 GSK Investigational Site Kyoto, Japan, 615-8087 GSK Investigational Site Oita, Japan, 870-0921 GSK Investigational Site Oita, Japan, 876-0047 GSK Investigational Site Osaka, Japan, 576-0016 GSK Investigational Site Osaka, Japan, 589-0022 GSK Investigational Site Osaka, Japan, 590-0064 GSK Investigational Site Shizuoka, Japan, 436-0022 GSK Investigational Site Tokyo, Japan, 105-0004 GSK Investigational Site Tokyo, Japan, 185-0014 GSK Investigational Site Tokyo, Japan, 187-0024 GSK Investigational Site Wakayama, Japan, 641-8510 GSK Investigational Site Yamanashi, Japan, 400-0031 Collapse << |
| NCT00516139 | Epilepsy | Phase 3 | Completed | - | - |
| NCT00526669 | Neoplasms, Gastrointestinal Tr... more >>act Collapse << | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Loma Linda, California, United States, 92354 GSK Investigational Site Los Angeles, California, United States, 90033 United States, District of Columbia GSK Investigational Site Washington, District of Columbia, United States, 20007 United States, Louisiana GSK Investigational Site Shreveport, Louisiana, United States, 71103 United States, Michigan GSK Investigational Site Ann Arbor, Michigan, United States, 48109 GSK Investigational Site Southgate, Michigan, United States, 48195 United States, Missouri GSK Investigational Site Jefferson City, Missouri, United States, 65109 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75137 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H3T 1E2 Korea, Republic of GSK Investigational Site Hwasun, Korea, Republic of, 519-809 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 120-752 GSK Investigational Site Suwon, Gyeonggi-do, Korea, Republic of, 442-723 Mexico GSK Investigational Site Mexico City, Mexico, CP 14080 Russian Federation GSK Investigational Site Astrakhan, Russian Federation, 414044 GSK Investigational Site Chelyabinsk, Russian Federation, 454087 GSK Investigational Site St. Petersburg, Russian Federation, 197022 GSK Investigational Site Ufa,, Russian Federation, 450054 Taiwan GSK Investigational Site Changhua, Taiwan, 500 GSK Investigational Site Taipei, Taiwan, 100 GSK Investigational Site Taipei, Taiwan, 104 GSK Investigational Site Taipei, Taiwan, 112 Collapse << |
| NCT02020889 | Churg-Strauss Syndrome | Phase 3 | Completed | - | - |
| NCT01086410 | Asthma | Phase 3 | Completed | - | United States, California ... more >> GSK Investigational Site Cypress, California, United States, 90630 GSK Investigational Site Huntington Beach, California, United States, 92647 United States, Oklahoma GSK Investigational Site Oklahoma City, Oklahoma, United States, 73103 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78229 Germany GSK Investigational Site Frankfurt, Hessen, Germany, 60596 GSK Investigational Site Magdeburg, Sachsen-Anhalt, Germany, 39112 GSK Investigational Site Grosshansdorf, Schleswig-Holstein, Germany, 22927 GSK Investigational Site Berlin, Germany, 10117 GSK Investigational Site Berlin, Germany, 10787 GSK Investigational Site Hamburg, Germany, 20253 Poland GSK Investigational Site Bialystok, Poland, 15-010 GSK Investigational Site Gdansk, Poland, 80-405 GSK Investigational Site Gidle, Poland, 97-540 GSK Investigational Site Krakow, Poland, 31-023 GSK Investigational Site Lodz, Poland, 93-513 GSK Investigational Site Olsztyn, Poland, 10-357 GSK Investigational Site Warszawa, Poland, 01-138 Collapse << |
| NCT00526669 | - | - | Completed | - | - |
| NCT01453049 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(US FDA/EMA/SFDA dec... more >>isions to rosiglitazone-containing medicines, ethic) Collapse << | - | China, Hubei ... more >> GSK Investigational Site Wuhan, Hubei, China, 430030 China, Jiangsu GSK Investigational Site Suzhou, Jiangsu, China, 215004 China, Liaoning GSK Investigational Site Dalian, Liaoning, China, 116027 China, Zhejiang GSK Investigational Site Hangzhou, Zhejiang, China, 310003 GSK Investigational Site Hangzhou, Zhejiang, China, 310009 China GSK Investigational Site Beijing, China, 100029 GSK Investigational Site Beijing, China, 100730 GSK Investigational Site Chongqing, China, 400016 GSK Investigational Site Shanghai, China, 200080 GSK Investigational Site Shenyang, China, 110003 GSK Investigational Site Tianjin, China, 300052 GSK Investigational Site Wuhan, China, 430022 Collapse << |
| NCT02020889 | - | - | Completed | - | - |
| NCT01435616 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00533897 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01376388 | - | - | Completed | - | - |
| NCT01453049 | - | - | Terminated(US FDA/EMA/SFDA dec... more >>isions to rosiglitazone-containing medicines, ethic) Collapse << | - | - |
| NCT01086410 | - | - | Completed | - | - |
| NCT00533897 | - | - | Completed | - | - |
| NCT00685776 | Coronary Heart Disease (CHD) ... more >> CHD Risk-Equivalent Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02555540 | Prevention of Infections With ... more >>Bordetella Pertussis Collapse << | Phase 4 | Active, not recruiting | December 2017 | Belgium ... more >> Center for Vaccinology Ghent, East-Flanders, Belgium, 9000 Collapse << |
| NCT01435616 | - | - | Completed | - | - |
| NCT01506271 | Intra-abdominal Infections | Phase 2 | Completed | - | - |
| NCT01205451 | Spasticity, Post-Stroke | Phase 3 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China China, Heilongjiang GSK Investigational Site Haerbin, Heilongjiang, China, 150001 China, Hubei GSK Investigational Site Wuhan, Hubei, China, 430060 China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210029 GSK Investigational Site Suzhou, Jiangsu, China, 215004 China, Liaoning GSK Investigational Site Shenyang, Liaoning, China, 110001 China, Sichuan GSK Investigational Site Chengdu, Sichuan, China, 610041 China GSK Investigational Site Beijing, China, 100050 GSK Investigational Site Beijing, China, 100068 GSK Investigational Site Beijing, China, 100730 GSK Investigational Site Hangzhou, China, 310016 GSK Investigational Site Shanghai, China, 200025 GSK Investigational Site Shanghai, China, 200040 Collapse << |
| NCT00281632 | - | - | Completed | - | - |
| NCT00812929 | - | - | Completed | - | - |
| NCT01205451 | - | - | Completed | - | - |
| NCT01505634 | Urinary Tract Infections ... more >> Pyelonephritis Collapse << | Phase 2 | Completed | - | - |
| NCT00281632 | Peritoneal Cancer ... more >> Ovarian Cancer Neoplasms, Ovarian Fallopian Tube Cancer Collapse << | Phase 2 | Completed | - | United States, Georgia ... more >> GSK Investigational Site Atlanta, Georgia, United States, 30342 United States, Texas GSK Investigational Site Austin, Texas, United States, 78731 GSK Investigational Site Bedford, Texas, United States, 76022 GSK Investigational Site Dallas, Texas, United States, 75246 GSK Investigational Site Fort Worth, Texas, United States, 76104 Australia, New South Wales GSK Investigational Site Randwick, New South Wales, Australia, 2031 Australia, Queensland GSK Investigational Site Herston, Queensland, Australia, 4029 Australia GSK Investigational Site Melbourne, Australia, 3084 Singapore GSK Investigational Site Singapore, Singapore, 119074 GSK Investigational Site Singapore, Singapore, 229899 Collapse << |
| NCT00812929 | Asthma | Phase 2 | Completed | - | United States, Colorado ... more >> GSK Investigational Site Denver, Colorado, United States, 80230 United States, Massachusetts GSK Investigational Site North Dartmouth, Massachusetts, United States, 02747 United States, Oklahoma GSK Investigational Site Oklahoma City, Oklahoma, United States, 73120 United States, Pennsylvania GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 United States, South Carolina GSK Investigational Site Orangeburg, South Carolina, United States, 29118 United States, Texas GSK Investigational Site El Paso, Texas, United States, 79925 Collapse << |
| NCT01717300 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01702363 | - | - | Completed | - | - |
| NCT01702363 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 811-1347 GSK Investigational Site Gunma, Japan, 371-0048 GSK Investigational Site Hokkaido, Japan, 080-0805 GSK Investigational Site Hyogo, Japan, 670-0849 GSK Investigational Site Ibaraki, Japan, 300-0053 GSK Investigational Site Ibaraki, Japan, 302-0022 GSK Investigational Site Ishikawa, Japan, 920-8610 GSK Investigational Site Kanagawa, Japan, 239-0821 GSK Investigational Site Kyoto, Japan, 601-1495 GSK Investigational Site Miyagi, Japan, 983-0824 GSK Investigational Site Nagano, Japan, 391-0011 GSK Investigational Site Oita, Japan, 870-0921 GSK Investigational Site Oita, Japan, 876-0047 GSK Investigational Site Osaka, Japan, 530-0001 GSK Investigational Site Osaka, Japan, 589-0022 GSK Investigational Site Shizuoka, Japan, 436-0022 GSK Investigational Site Tokyo, Japan, 103-0027 GSK Investigational Site Tokyo, Japan, 153-8934 GSK Investigational Site Tokyo, Japan, 192-0903 GSK Investigational Site Yamanashi, Japan, 400-0031 Collapse << |
| NCT00627926 | - | - | Completed | - | - |
| NCT01297270 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02045797 | Infections, Bacterial | Phase 2 | Completed | - | - |
| NCT01860729 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00627926 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT01297270 | - | - | Completed | - | - |
| NCT00810277 | Rheumatoid Arthritis | Phase 3 | Completed | - | Finland ... more >> Kiljavan Lääketutkimus Oy Hyvinkää, Finland, 05800 Kanta-Hämeen Keskussairaala ; Reumatologia Hämeenlinna, Finland, 13530 Kanta-Hämeen keskussairaala; Riihimäen aluesairaala Reumapoliklinikka Riihimäki, Finland, 11101 Collapse << |
| NCT00857857 | Asthma | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Wiesbaden, Hessen, Germany, 65187 GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Grosshansdorf, Schleswig-Holstein, Germany, 22927 GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT00810277 | - | - | Completed | - | - |
| NCT02045797 | - | - | Completed | - | - |
| NCT00857857 | - | - | Completed | - | - |
| NCT00573794 | - | - | Completed | - | - |
| NCT00573794 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT01382212 | - | - | Completed | - | - |
| NCT01382212 | End-Stage Renal Disease ... more >> Secondary Hyperparathyroidism Collapse << | Phase 3 | Completed | - | - |
| NCT01622231 | Rhinitis | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 272-0143 GSK Investigational Site Chiba, Japan, 277-0882 GSK Investigational Site Gifu, Japan, 501-3247 GSK Investigational Site Saitama, Japan, 355-0062 GSK Investigational Site Tokyo, Japan, 160-0017 GSK Investigational Site Tokyo, Japan, 170-0005 Collapse << |
| NCT03298373 | Healthy Men M... more >>ale Contraception Collapse << | Phase 1 | Recruiting | December 2018 | United States, California ... more >> Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center Recruiting Torrance, California, United States, 90509 Contact: Christina Wang, MD 310-222-2503 wang@labiomed.org United States, Washington University of Washington Medical Center & Health Sciences Recruiting Seattle, Washington, United States, 98195 Contact: Stephanie Page, MD, PhD 206-616-1818 page@uw.edu Collapse << |
| NCT01622231 | - | - | Completed | - | - |
| NCT00655824 | Arthritis, Rheumatoid | Phase 2 | Terminated(The clinical develo... more >>pment of intravenously administered ofatumumab in RA will no longer be pursued, so this study was prematurely terminated by the Sponsor.) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90095 United States, Florida GSK Investigational Site Fort Lauderdale, Florida, United States, 33334 United States, New York GSK Investigational Site New York, New York, United States, 10003 United States, Pennsylvania GSK Investigational Site Duncansville, Pennsylvania, United States, 16635 Denmark GSK Investigational Site Glostrup, Denmark, 2600 GSK Investigational Site Hellerup, Denmark, 2900 GSK Investigational Site Koebenhavn, Denmark, 2100 Hungary GSK Investigational Site Szombathely, Hungary, 9700 Poland GSK Investigational Site Warszawa, Poland, 02-256 United Kingdom GSK Investigational Site Ipswich, United Kingdom, IP4 5PD Collapse << |
| NCT00168831 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00168831 | - | - | Completed | - | - |
| NCT00796991 | - | - | Completed | - | - |
| NCT01721317 | - | - | Terminated(After reevaluation ... more >>of the benefit risk profile of ezogabine/retigabine, GSK does not believe the early adjunctive treatment study population is appropriate.) Collapse << | - | - |
| NCT02292771 | Obstetric Labour, Premature | Phase 3 | Terminated(The study was stopp... more >>ed due to the feasibility of recruiting the study in a timely manner) Collapse << | - | - |
| NCT00655824 | - | - | Terminated(The clinical develo... more >>pment of intravenously administered ofatumumab in RA will no longer be pursued, so this study was prematurely terminated by the Sponsor.) Collapse << | - | - |
| NCT00796991 | Advanced Melanoma | Phase 1 | Completed | - | United States, California ... more >> The Angeles Clinic & Research Inst. Los Angeles, California, United States, 90025 United States, Florida H Lee Moffit Cancer Cnt And Res Inst Tampa, Florida, United States, 33612 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10021 United States, North Carolina Blumenthal Cancer Center, Carolinas Medical Center Charlotte, North Carolina, United States, 28204 Collapse << |
| NCT02000440 | Glomerulosclerosis, Focal Segm... more >>ental Collapse << | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90025 GSK Investigational Site Stanford, California, United States, 94304 United States, Louisiana GSK Investigational Site New Orleans, Louisiana, United States, 70112 United States, Michigan GSK Investigational Site Ann Arbor, Michigan, United States, 48109 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599-7155 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44109 GSK Investigational Site Columbus, Ohio, United States, 43210 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19140 Canada, Alberta GSK Investigational Site Edmonton, Alberta, Canada, T6G 2B7 Canada, British Columbia GSK Investigational Site Vancouver, British Columbia, Canada, V6Z 1Y6 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M4N 3M5 GSK Investigational Site Toronto, Ontario, Canada, M5G 2C4 Collapse << |
| NCT01358864 | Hepatitis C, Chronic | Phase 3 | Completed | - | - |
| NCT01721317 | Seizures | Phase 4 | Terminated(After reevaluation ... more >>of the benefit risk profile of ezogabine/retigabine, GSK does not believe the early adjunctive treatment study population is appropriate.) Collapse << | - | United States, California ... more >> GSK Investigational Site Fresno, California, United States, 93710 GSK Investigational Site Newport Beach, California, United States, 92658 GSK Investigational Site Newport Beach, California, United States, 92663 GSK Investigational Site Santa Monica, California, United States, 90404 United States, Colorado GSK Investigational Site Colorado Springs, Colorado, United States, 80907 United States, Florida GSK Investigational Site Pensacola, Florida, United States, 32514 GSK Investigational Site Port Charlotte, Florida, United States, 33952 United States, Illinois GSK Investigational Site Peoria, Illinois, United States, 61637 United States, Maryland GSK Investigational Site Bethesda, Maryland, United States, 20817 United States, Minnesota GSK Investigational Site Golden Valley, Minnesota, United States, 55422 United States, New Jersey GSK Investigational Site Hackensack, New Jersey, United States, 07601 GSK Investigational Site Livingston, New Jersey, United States, 07039 United States, Oregon GSK Investigational Site Medford, Oregon, United States, 97504 United States, Texas GSK Investigational Site Arlington, Texas, United States, 76017 GSK Investigational Site Dallas, Texas, United States, 75251 GSK Investigational Site Kingwood, Texas, United States, 77339 GSK Investigational Site San Antonio, Texas, United States, 78229 GSK Investigational Site Temple, Texas, United States, 76508 United States, Washington GSK Investigational Site Spokane, Washington, United States, 99204 Greece GSK Investigational Site Athens, Greece, 10676 GSK Investigational Site Thessaloniki, Greece, 57010 Collapse << |
| NCT02292771 | - | - | Terminated(The study was stopp... more >>ed due to the feasibility of recruiting the study in a timely manner) Collapse << | - | - |
| NCT01358864 | - | - | Completed | - | - |
| NCT02000440 | - | - | Completed | - | - |
| NCT00320489 | Schizophrenia | Phase 3 | Completed | - | - |
| NCT02296541 | HIV Infections | Phase 1 | Active, not recruiting | September 8, 2020 | United States, Alabama ... more >> Alabama CRS Birmingham, Alabama, United States, 35294 United States, California Bridge HIV CRS San Francisco, California, United States, 94143 United States, Georgia The Hope Clinic of the Emory Vaccine Center CRS Decatur, Georgia, United States, 30030 United States, Massachusetts Brigham and Women's Hospital Vaccine CRS (BWH VCRS) Boston, Massachusetts, United States, 02115-6110 Fenway Health (FH) CRS Boston, Massachusetts, United States, 02215-4302 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 Switzerland Lausanne Vaccine and Immunotherapy Center CRS Lausanne, Vaud, Switzerland, 1011 Collapse << |
| NCT01310413 | Influenza | Phase 3 | Completed | - | United States, California ... more >> GSK Investigational Site Paramount, California, United States, 90723 GSK Investigational Site Sacramento, California, United States, 95816 United States, Kansas GSK Investigational Site Newton, Kansas, United States, 67114 United States, Kentucky GSK Investigational Site Bardstown, Kentucky, United States, 40004 United States, Louisiana GSK Investigational Site Metairie, Louisiana, United States, 70006 United States, Missouri GSK Investigational Site Saint Louis, Missouri, United States, 63141 United States, Nebraska GSK Investigational Site Omaha, Nebraska, United States, 68134 United States, Nevada GSK Investigational Site Henderson, Nevada, United States, 89014 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44121 United States, Texas GSK Investigational Site Fort Worth, Texas, United States, 76135 GSK Investigational Site San Angelo, Texas, United States, 76904 Canada, British Columbia GSK Investigational Site Coquitlam, British Columbia, Canada, V3K 3P4 Canada, Ontario GSK Investigational Site Hamilton, Ontario, Canada, L8L 5G8 GSK Investigational Site Sudbury, Ontario, Canada, P3E 1H5 Canada, Quebec GSK Investigational Site Sherbrooke, Quebec, Canada, J1H 1Z1 GSK Investigational Site Sherbrooke, Quebec, Canada, J1H 2G2 Thailand GSK Investigational Site Khon Kaen, Thailand, 40002 Collapse << |
| NCT01602549 | Gastroparesis | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Germany GSK Investigational Site Ulm, Baden-Wuerttemberg, Germany, 89081 GSK Investigational Site Bochum, Nordrhein-Westfalen, Germany, 44791 GSK Investigational Site Bonn, Nordrhein-Westfalen, Germany, 53105 GSK Investigational Site Koeln, Nordrhein-Westfalen, Germany, 50937 GSK Investigational Site Jena, Thueringen, Germany, 07747 GSK Investigational Site Berlin, Germany, 13088 Sweden GSK Investigational Site Uppsala, Sweden, SE-751 85 United Kingdom GSK Investigational Site Cambridge, United Kingdom, C2B 2PY GSK Investigational Site Newcastle Upon Tyne, United Kingdom, NE4 5PL GSK Investigational Site Norwich, United Kingdom, NR4 7UY Collapse << |
| NCT00320489 | - | - | Completed | - | - |
| NCT01466062 | Respiratory Syncytial Virus In... more >>fection Collapse << | Phase 3 | Completed | - | Japan ... more >> Site Reference ID/Investigator# 56847 Hyogo, Japan Site Reference ID/Investigator# 56845 Shimotsuke, Japan Site Reference ID/Investigator# 56842 Tokyo, Japan Site Reference ID/Investigator# 56844 Tokyo, Japan Site Reference ID/Investigator# 56846 Tokyo, Japan Site Reference ID/Investigator# 56843 Yokohama, Japan Collapse << |
| NCT01587898 | Anaemia | Phase 2 | Completed | - | - |
| NCT03726996 | Infantile Neuroaxonal Dystroph... more >>y Collapse << | Phase 4 | Not yet recruiting | November 30, 2020 | United States, North Carolina ... more >> Duke University Health Center Not yet recruiting Durham, North Carolina, United States, 27710 Contact: Janet Wootton, RN 919-668-4809 janet.wootton@duke.edu Collapse << |
| NCT02249468 | - | - | Terminated(Stop the study for ... more >>futility based on recommendation from Independent Data Committee.) Collapse << | - | Singapore ... more >> National University Hospital Singapore, Singapore, 119074 Collapse << |
| NCT00536809 | Neoplasms, Colorectal | Phase 1 | Completed | - | United States, Wisconsin ... more >> GSK Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01310413 | - | - | Completed | - | - |
| NCT01899768 | Cough | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Manchester, Lancashire, United Kingdom, M23 9LT GSK Investigational Site Belfast, United Kingdom, BT9 7AB Collapse << |
| NCT01602549 | - | - | Completed | - | - |
| NCT00536809 | - | - | Completed | - | - |
| NCT01466062 | - | - | Completed | - | - |
| NCT01899768 | - | - | Completed | - | - |
| NCT01587898 | - | - | Completed | - | - |
| NCT02377466 | Obstetric Labour, Premature | Phase 3 | Terminated(Asset terminated by... more >> PIB) Collapse << | - | - |
| NCT03526588 | Congenital Diaphragmatic Herni... more >>a Collapse << | Phase 1 | Recruiting | November 1, 2027 | United States, Texas ... more >> The University of Texas Health Science Center at Houston Recruiting Houston, Texas, United States, 77030 Contact: Matthew T. Harting, MD, MS 855-566-6273 Collapse << |
| NCT02377466 | - | - | Terminated(Asset terminated by... more >> PIB) Collapse << | - | - |
| NCT00168844 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00979420 | - | - | Completed | - | - |
| NCT00168844 | - | - | Completed | - | - |
| NCT03400488 | Coronary Artery Disease | Phase 1 | Completed | - | United States, California ... more >> Research Site Glendale, California, United States, 91206 Collapse << |
| NCT03710356 | Telomere Shortening ... more >> Telomere Length, Mean Leukocyte Collapse << | Phase 1 Phase 2 | Not yet recruiting | October 20, 2022 | - |
| NCT02345161 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00979420 | - | - | Completed | - | - |
| NCT02880852 | Systemic Lupus Erythematosus | Phase 1 | Completed | - | China ... more >> GSK Investigational Site Shanghai, China GSK Investigational Site Suzhou, China, 215004 Collapse << |
| NCT02880852 | - | - | Completed | - | - |
| NCT02497937 | - | - | Completed | - | - |
| NCT01568892 | HIV Infections | Phase 3 | Completed | - | - |
| NCT02345161 | - | - | Completed | - | - |
| NCT00848510 | Colorectal and Ovarian Cancer ... more >>Patients With Liver Metastases Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Christie Hospital Manchester, United Kingdom Collapse << |
| NCT00848510 | - | - | Completed | - | - |
| NCT01494610 | - | - | Completed | - | - |
| NCT02497937 | Heart Failure | Phase 2 | Completed | - | United States, Minnesota ... more >> GSK Investigational Site Minneapolis, Minnesota, United States, 55415 GSK Investigational Site Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01494610 | Asthma | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 New Zealand GSK Investigational Site Wellington, New Zealand, 6021 Collapse << |
| NCT01328054 | Cancer | Phase 4 | Completed | - | United States, Michigan ... more >> GSK Investigational Site Detroit, Michigan, United States, 48202 United States, New Hampshire GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Utah GSK Investigational Site Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02564042 | Psoriasis | Phase 2 | Completed | - | - |
| NCT01328054 | - | - | Completed | - | - |
| NCT01568892 | - | - | Completed | - | - |
| NCT01316887 | - | - | Completed | - | - |
| NCT02564055 | Dermatitis, Atopic | Phase 2 | Completed | - | - |
| NCT00804570 | Alcohol Dependence | Phase 2 | Completed | - | United States, Indiana ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Indianapolis, Indiana, United States, 46260 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lafayette, Indiana, United States, 47905 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21205 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Belmont, Massachusetts, United States, 02478 United States, Minnesota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rochester, Minnesota, United States, 55905 United States, Nebraska For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Omaha, Nebraska, United States, 68133 United States, New Hampshire For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lebanon, New Hampshire, United States, 03756 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New York, New York, United States, 10016 United States, North Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chapel Hill, North Carolina, United States, 27599 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Charlotte, North Carolina, United States, 28211 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45237 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97210 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Philadelphia, Pennsylvania, United States, 19104 United States, Rhode Island For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Providence, Rhode Island, United States, 02908 United States, South Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Charleston, South Carolina, United States, 29425 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 United States, Washington For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seattle, Washington, United States, 98104 Collapse << |
| NCT01667679 | Migraine Head... more >>aches Collapse << | Phase 3 | Completed | - | United States, California ... more >> San Francisco Clinical Research Center San Francisco, California, United States, 94109 California Medical Clinic for Headache Santa Monica, California, United States, 90404 United States, Connecticut Associated Neurologists of Southern CT, P.C. Fairfied, Connecticut, United States, 06824 United States, Florida Premiere Research Institute West Palm Beach, Florida, United States, 33407 United States, Massachusetts MedVadis Watertown, Massachusetts, United States, 02472 United States, Michigan Michigan Head and Pain Institute Ann Arbor, Michigan, United States, 48104-5199 United States, Missouri ClinVest Springfield, Missouri, United States, 65807 Mercy Health Research St. Louis, Missouri, United States, 63141 United States, New York DENT Neurologic Institute Amherst, New York, United States, 14226 United States, North Carolina Headache Welness Center Greensboro, North Carolina, United States, 27405 PMG Research of Winston Salem, LLC Winston Salem, North Carolina, United States, 27103 United States, Pennsylvania Jefferson Headache Center Philadelphia, Pennsylvania, United States, 19107 United States, South Carolina Coastal Carolina Research Center Mt. Pleasant, South Carolina, United States, 29464 Collapse << |
| NCT01316887 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT02564042 | - | - | Completed | - | - |
| NCT00804570 | - | - | Completed | - | - |
| NCT02753413 | Respiratory Syncytial Virus In... more >>fections Collapse << | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Wilrijk, Belgium, 2610 Collapse << |
| NCT03569826 | - | - | Recruiting | December 2025 | Canada, Alberta ... more >> University of Alberta Hospital Recruiting Edmonton, Alberta, Canada, T6G 2B7 Contact: Andrew Mason, MD (780) 492-8176 am16@ualberta.ca Canada, Ontario Toronto General Hospital Recruiting Toronto, Ontario, Canada, M5G 2C4 Contact: Bettina Hansen, PhD 416-340-5157 bettina.hansen@utoronto.ca Principal Investigator: Bettina Hansen, PhD Collapse << |
| NCT00570492 | Rhinitis, Allergic, Perennial | Phase 4 | Completed | - | - |
| NCT00570492 | - | - | Completed | - | - |
| NCT01667679 | - | - | Completed | - | - |
| NCT03024827 | Epileptic Encephalopathy | Phase 1 | Recruiting | December 2019 | Canada, British Columbia ... more >> University of British Columbia Recruiting Vancouver, British Columbia, Canada, V6T1Z4 Contact: Vesna Popovska vpopovska@cw.bc.ca Principal Investigator: Linda Huh Canada, Manitoba University of Manitoba Recruiting Winnipeg, Manitoba, Canada, R3T 2N2 Contact: Lise Bourrier LBourrier@chrim.ca Principal Investigator: Edward Leung Canada, Quebec Universite de Montreal Recruiting Montreal, Quebec, Canada, H3T1J4 Contact: Jonathan Bitton jonathan.bitton@umontreal.ca Principal Investigator: Philippe Major Canada, Saskatchewan University of Saskatchewan Recruiting Saskatoon, Saskatchewan, Canada, S7N5C5 Contact: Doris Newmeyer 306-844-1208 doris.newmeyer@usask.ca Principal Investigator: Richard Huntsman Sub-Investigator: Blair Seifert Sub-Investigator: Jose Tellez-Zentino Sub-Investigator: Jane Alcorn Sub-Investigator: Bryan Acton Sub-Investigator: Andrew Lyon Sub-Investigator: June Lim Sub-Investigator: Simona Hasal Collapse << |
| NCT01808261 | Cerebrovascular Accident | Phase 2 | Terminated(This study was term... more >>inated for futility.) Collapse << | - | - |
| NCT02753413 | - | - | Completed | - | - |
| NCT00849875 | Melanoma | Phase 2 | Terminated(Early end of trial ... more >>notification after termination of long term follow up due to lack of scientific justification to continue collect information.) Collapse << | - | Belgium ... more >> GSK Investigational Site Brussels, Belgium, 1200 GSK Investigational Site Brussel, Belgium, 1090 GSK Investigational Site Bruxelles, Belgium, 1180 GSK Investigational Site Liège, Belgium, 4000 GSK Investigational Site Roeselare, Belgium, 8800 GSK Investigational Site Yvoir, Belgium, 5530 France GSK Investigational Site Caen, France, 14033 GSK Investigational Site Lille, France, 59037 GSK Investigational Site Marseille Cedex 5, France, 13385 GSK Investigational Site Nantes, France, 44093 GSK Investigational Site Paris Cedex 10, France, 75475 GSK Investigational Site Paris, France, 75018 GSK Investigational Site Reims, France, 51092 GSK Investigational Site Villejuif, France, 94805 Collapse << |
| NCT00849875 | - | - | Terminated(Early end of trial ... more >>notification after termination of long term follow up due to lack of scientific justification to continue collect information.) Collapse << | - | - |
| NCT00612040 | Diabetes Diab... more >>etes Mellitus, Type 1 Collapse << | Phase 2 | Completed | - | - |
| NCT01808261 | - | - | Terminated(This study was term... more >>inated for futility.) Collapse << | - | - |
| NCT00614055 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | France ... more >> Novo Nordisk Investigational Site Alès, France, 30100 Novo Nordisk Investigational Site Brest, France, 29609 Novo Nordisk Investigational Site LA ROCHELLE cedex, France, 17019 Novo Nordisk Investigational Site Le Creusot, France, 71200 Novo Nordisk Investigational Site Mont de Marsan, France, 40024 Novo Nordisk Investigational Site Venissieux, France, 69200 Germany Novo Nordisk Investigational Site Bad Kreuznach, Germany, 55545 Novo Nordisk Investigational Site Bad Mergentheim, Germany, 97980 Novo Nordisk Investigational Site Dormagen, Germany, 41539 Novo Nordisk Investigational Site Hohenmölsen, Germany, 06679 Novo Nordisk Investigational Site Neuss, Germany, 41460 Novo Nordisk Investigational Site Neuwied, Germany, 56564 Norway Novo Nordisk Investigational Site Elverum, Norway, 2408 Novo Nordisk Investigational Site Hamar, Norway, 2317 Novo Nordisk Investigational Site Kongsvinger, Norway, 2212 Novo Nordisk Investigational Site Oslo, Norway, 0586 Novo Nordisk Investigational Site Stavanger, Norway, 4011 Novo Nordisk Investigational Site Tromsø, Norway, 9038 Romania Novo Nordisk Investigational Site Bucharest, Romania, 011234 Novo Nordisk Investigational Site Bucharest, Romania, 020042 Novo Nordisk Investigational Site Bucharest, Romania, 020475 Novo Nordisk Investigational Site Bucharest, Romania, 020992 Novo Nordisk Investigational Site Bucharest, Romania Spain Novo Nordisk Investigational Site Almería, Spain, 04001 Novo Nordisk Investigational Site Partida de Bacarot, Spain, 03114 Novo Nordisk Investigational Site Sevilla, Spain, 41010 Novo Nordisk Investigational Site Valencia, Spain, 46014 Novo Nordisk Investigational Site Valladolid, Spain, 47011 Collapse << |
| NCT00612040 | - | - | Completed | - | - |
| NCT01494584 | Epilepsy | Phase 2 | Terminated(Terminated after pl... more >>acing the study on hold at the request of the FDA) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90027 United States, Florida GSK Investigational Site Gulf Breeze, Florida, United States, 32561 GSK Investigational Site Port Charlotte, Florida, United States, 33952 GSK Investigational Site Wellington, Florida, United States, 33414 United States, Minnesota GSK Investigational Site Saint Paul, Minnesota, United States, 55102-2534 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Tennessee GSK Investigational Site Memphis, Tennessee, United States, 38105 United States, Texas GSK Investigational Site Austin, Texas, United States, 78723 GSK Investigational Site Dallas, Texas, United States, 75230-2507 Collapse << |
| NCT01494584 | - | - | Terminated(Terminated after pl... more >>acing the study on hold at the request of the FDA) Collapse << | - | - |
| NCT02564055 | - | - | Completed | - | - |
| NCT00614055 | - | - | Completed | - | - |
| NCT01819636 | Hypertriglyceridemia | Not Applicable | Completed | - | France ... more >> OPTIMED Gières, France, 38610 Naturalpha Lille, France, 59020 Biofortis Saint Herblain, France, 44800 Collapse << |
| NCT02195817 | Alcohol Dependence | Phase 4 | Terminated(The study was termi... more >>nated due to enrolment challenges) Collapse << | - | - |
| NCT02885207 | Epilepsy | Not Applicable | Unknown | July 2017 | France ... more >> CHU de Besançon Not yet recruiting Besançon, France, 25000 Contact: BERGER Eric, MD 03 81 66 81 66 neurologie-secret@chu-besancon.fr CHU de Dijon Not yet recruiting Dijon, France, 21079 Contact: LEMESLE Matine, MD 03 80 29 30 31 martine.lemesle@chu-dijon.fr CHU de Nancy Not yet recruiting Nancy, France, 54000 Contact: MAILLIARD Louis, MD 03 83 85 16 09 Les Hôpitaux Universitaires de Strasbourg Recruiting Strasbourg, France, 67091 Principal Investigator: BENOILID Aurélien, MD Collapse << |
| NCT02539576 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, California ... more >> GSK Investigational Site Glendale, California, United States, 91206 Collapse << |
| NCT00009737 | Colorectal Cancer | Phase 3 | Completed | - | - |
| NCT02198703 | Moderate Hypercholesterolemia | Not Applicable | Completed | - | France ... more >> Naturalpha Lille, France, 59020 Biofortis Saint-Herblain, France, 44800 Collapse << |
| NCT02838121 | Prevention of BMD Loss After T... more >>HR Serum Markers of Bone Turnover Functional Outcomes Collapse << | Phase 3 | Completed | - | Taiwan ... more >> Chang Gung Memorial Hospital Kweishan, Taoyuan, Taiwan, 333 Collapse << |
| NCT03089840 | End Stage Liver Disease ... more >> Liver Diseases Collapse << | Phase 1 Phase 2 | Recruiting | January 2020 | Canada, Alberta ... more >> University of Alberta Recruiting Edmonton, Alberta, Canada, T6G2C8 Contact: Andrew Malcolm, PhD 780-407-6952 andrew.malcolm@ualberta.ca Principal Investigator: James Shapiro, MD, PhD Collapse << |
| NCT03251014 | Thrombosis | Not Applicable | Completed | - | - |
| NCT02368249 | Kidney Failure | Phase 2 | Withdrawn(no focus on this top... more >>ic) Collapse << | February 2025 | Switzerland ... more >> Department of Visceral and Transplantation Surgery of the University Hospital of Zurich Zurich, Switzerland, 8091 Collapse << |
| NCT02664766 | Heavy Drinking ... more >> Alcohol Abuse Alcohol Use Disorder Collapse << | Not Applicable | Completed | - | - |
| NCT01478126 | Coronary Artery Disease | Phase 3 | Completed | - | Russian Federation ... more >> State Research Institute of Circulation Pathology Novosibirsk, Russian Federation Collapse << |
| NCT02856997 | Peripheral T Cell Lymphoma | Phase 2 | Not yet recruiting | - | - |
| NCT02970838 | Obesity Diabe... more >>tes Type 2 Weight Loss Collapse << | Not Applicable | Completed | - | Germany ... more >> University Medicine Greifswald Greifswald, Germany, 17475 Collapse << |
| NCT02944812 | Peripheral T Cell Lymphoma | Phase 2 | Recruiting | January 2019 | China, Guangdong ... more >> Guangdong general hospital Recruiting Guangzhou, Guangdong, China, 510080 Contact: Wenyu Li, MD PhD +86 20 81884713-80412 Collapse << |
| NCT02815007 | Advanced EGFR-TKI-resistant No... more >>n-Small Cell Lung Cancer Collapse << | Phase 2 | Unknown | June 2018 | - |
| NCT00009737 | - | - | Completed | - | - |
| NCT02109445 | - | - | Terminated(The study was termi... more >>nated on 24JUN15 due to change in strategy of PF-03084014 development.No safety/efficacy concerns were behind the reason of trial termination) Collapse << | - | - |
| NCT02878278 | Lymphoma, Extranodal NK-T-Cell... more >> EBV Collapse << | Phase 2 | Not yet recruiting | September 2019 | - |
| NCT03678480 | Primary Sclerosing Cholangitis... more >> Cholangitis Cholangitis, Sclerosing Bile Duct Diseases Biliary Tract Diseases Digestive System Diseases Adolescent Collapse << | Phase 2 | Not yet recruiting | November 4, 2019 | - |
| NCT00385671 | Diabetic Neuropathy, Painful | Phase 4 | Completed | - | United States, California ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Torrance, California, United States, 90503 United States, Connecticut For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cromwell, Connecticut, United States, 06416 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Staten Island, New York, United States, 10312 United States, North Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Greenville, North Carolina, United States, 27834 United States, Utah For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Midvale, Utah, United States, 84047 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bochum, Germany, D-44805 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dresden, Germany, 01307 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 20354 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Siegen, Germany, 57072 Collapse << |
| NCT02109445 | Metastatic Cancer Pancreas | Phase 2 | Terminated(The study was termi... more >>nated on 24JUN15 due to change in strategy of PF-03084014 development.No safety/efficacy concerns were behind the reason of trial termination) Collapse << | - | United States, Colorado ... more >> Anschutz Inpatient Pavilion Aurora, Colorado, United States, 80045 Drug Shipment: Anschutz Cancer Pavilion Aurora, Colorado, United States, 80045 University of Colorado Cancer Center Aurora, Colorado, United States, 80045 University of Colorado Denver, CTO (CTRC) Aurora, Colorado, United States, 80045 United States, New York University of Rochester Investigational Drug Pharmacy Rochester, New York, United States, 14642 University of Rochester Rochester, New York, United States, 14642 Collapse << |
| NCT02644603 | Carcinoma, Hepatocellular | Not Applicable | Active, not recruiting | June 2017 | China, Shanghai ... more >> Zhongshan Hospital Shanghai, Shanghai, China, 200032 Collapse << |
| NCT02771756 | Breast Cancer ... more >> Lung Cancer Collapse << | Not Applicable | Unknown | May 2016 | China, Beijing ... more >> National Cancer Institute, Cancer Hospital Chinese Academy of Medical Sciences Recruiting Beijing, Beijing, China, 100021 Contact: Peng Yuan, MD 86-10-87788528 Yuanpeng01@hotmail.com Collapse << |
| NCT00725712 | Neoplasms, Gastrointestinal Tr... more >>act Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Birmingham, Alabama, United States, 35294 United States, Arizona GSK Investigational Site Scottsdale, Arizona, United States, 85258 United States, California GSK Investigational Site Los Angeles, California, United States, 90024 GSK Investigational Site Stanford, California, United States, 94305 United States, District of Columbia GSK Investigational Site Washington, D.C., District of Columbia, United States, 20007 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30309 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60637 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02114 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48201 United States, Montana GSK Investigational Site Billings, Montana, United States, 59101 United States, New Mexico GSK Investigational Site Albuquerque, New Mexico, United States, 87131 United States, New York GSK Investigational Site New York, New York, United States, 10016 GSK Investigational Site New York, New York, United States, 10021 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Oregon GSK Investigational Site Portland, Oregon, United States, 97239 United States, Texas GSK Investigational Site Austin, Texas, United States, 78705 United States, Wisconsin GSK Investigational Site Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00725712 | - | - | Completed | - | - |
| NCT02119260 | Oedema, Pulmonary | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 2GG GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT00385671 | - | - | Completed | - | - |
| NCT00981981 | Hypercholesterolemia | Phase 2 | Completed | - | Australia, New South Wales ... more >> SUGiRS Human Nutrition Unit, School of Molecular & Microbial Biosciences, Unviersity of Sydney Sydney, New South Wales, Australia, 2006 Canada, Ontario Human Nutraceutical Research Unit, Department of Human Health and Nutritional Sciences, University of Guelph Guelph, Ontario, Canada, N1G 2W1 Glycemic Index Laboratories, Inc. Toronto, Ontario, Canada, M5C 2X3 Canada, Quebec Nutraceuticals and Functional Foods Institute, Faculte des science de l'agriculture et de l'alimentation, Universite Laval Laval, Quebec, Canada United Kingdom Reading Scientific Services, Ltd (RSSL) Reading, Berkshire, United Kingdom, RG6 6LA Collapse << |
| NCT02133079 | Liver Cancer ... more >>Pancreatic Adenocarcinoma Collapse << | Phase 1 Phase 2 | Active, not recruiting | November 2019 | China ... more >> Cancer Insititute and Hospital,Chinese Academy of Medical Sciences Beijing, China, 100021 Collapse << |
| NCT01982734 | Pharmacokinetics of New Curcum... more >>in Formulations Safety of New Curcumin Formulations Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Collapse << |
| NCT03516903 | Myocardial Infarction, Anterio... more >>r Wall Myocardial Remodeling, Ventricular Collapse << | Phase 2 Phase 3 | Recruiting | July 1, 2021 | Brazil ... more >> Heart Institute (InCor) - University of São Paulo Medical School, São Paulo, Brazil Recruiting São Paulo, Brazil Contact: Aline G. Ferrari, MD +551126615058 alinegferrari@gmail.com Collapse << |
| NCT03414294 | Healthy | Phase 1 | Recruiting | February 27, 2020 | Australia, South Australia ... more >> CMAX, Clinical Research Pty Ltd Recruiting Adelaide, South Australia, Australia, 5000 Collapse << |
| NCT00394953 | Anemia | Phase 3 | Completed | - | - |
| NCT00394953 | - | - | Completed | - | - |
| NCT02119260 | - | - | Completed | - | - |
| NCT02581345 | Chronic Plaque Psoriasis ... more >> Psoriasis Collapse << | Phase 3 | Completed | - | - |
| NCT03140397 | Safety After Oral Intake ... more >> Pharmacokinetics After Oral Intake Immune Cells Activity Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Eberhard Karls University Tuebingen Tübingen, Baden-Württemberg, Germany, 72076 Collapse << |
| NCT02944097 | Safety After Oral Intake ... more >> Pharmacokinetics After Oral Intake Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Collapse << |
| NCT03286777 | Safety of Native vs. Micellar ... more >>6-PN After Oral Intake Pharmacokinetics of Native vs. Micellar 6-PN After Oral Intake PBMC Activity After Native vs. Micellar 6-PN Oral Intake Collapse << | Not Applicable | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Eberhard Karls University Tuebingen Tübingen, Baden-Württemberg, Germany, 72076 Collapse << |
| NCT02581345 | - | - | Completed | - | - |
| NCT02221622 | Mild Cognitive Impairment ... more >> Alzheimer Disease Collapse << | Phase 1 | Completed | - | United States, California ... more >> University of Southern California - Alzheimer Disease Research Center - Healthcare Consultation Center II Los Angeles, California, United States, 90033 Collapse << |
| NCT01241292 | - | - | Completed | - | - |
| NCT02944084 | Safety After Oral Intake ... more >> Pharmacokinetics After Oral Intake Collapse << | Early Phase 1 | Completed | - | Germany ... more >> University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Collapse << |
| NCT02222493 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT03388528 | Mitochondrial Diseases | Not Applicable | Recruiting | September 7, 2018 | United Kingdom ... more >> Grainne Gorman Recruiting Newcastle upon Tyne, Tyne And Wear, United Kingdom, NE2 4HH Contact: David Houghton, PhD 07729221941 david.houghton@ncl.ac.uk Contact: Paula Hynd, BSc 0191 208 3084 paula.hynd@ncl.ac.uk Collapse << |
| NCT03008070 | Non-Alcoholic Steatohepatitis ... more >>(NASH) Collapse << | Phase 2 | Recruiting | January 2020 | - |
| NCT02512900 | Asthma | Phase 1 | Completed | - | Poland ... more >> Research Site Kielce, Poland, 25-040 Research Site Wrocław, Poland, 51-162 Research Site Wrocław, Poland, 53-201 Research Site Łódź, Poland, 71-329 Collapse << |
| NCT02512900 | - | - | Completed | - | - |
| NCT00434512 | AIDS | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Collapse << |
| NCT00815737 | Lung Infection ... more >> Cerebral Hemorrhage Pneumonia Collapse << | Phase 2 Phase 3 | Unknown | - | China, Shanghai ... more >> Zhongshan hospital of Fudan University Shanghai, Shanghai, China, 200032 Collapse << |
| NCT03632876 | Sickle Cell Anemia in Children... more >> Vitamin A Deficiency in Children Collapse << | Not Applicable | Completed | - | United States, Pennsylvania ... more >> Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19146 Collapse << |
| NCT00910962 | Acute Coronary Syndrome | Phase 2 | Completed | - | - |
| NCT00789269 | Pneumonia Acu... more >>te Stroke Collapse << | Phase 2 | Unknown | - | China, Shanghai ... more >> Department of Integrative Medicine, Zhongshan hospital, Fudan University Shanghai, Shanghai, China, 200032 Collapse << |
| NCT03674957 | - | - | Active, not recruiting | March 2019 | Canada, Manitoba ... more >> University of Manitoba Winnipeg, Manitoba, Canada, R3T 6C5 Collapse << |
| NCT02222493 | - | - | Completed | - | - |
| NCT02492958 | Staphylococcal Infections | Phase 2 | Completed | - | Japan ... more >> SOUSEIKAI Sumida Hospital (formerly Medical Co. LTA Sumida Hospital) Sumida-ku, Tokyo, Japan, 130-0004 SOUSEIKAI PS Clinic (formerly Medical Co. LTA PS Clinic) Fukuoka, Japan, 812-0025 Collapse << |
| NCT00910962 | - | - | Completed | - | - |
| NCT01241292 | Multiple Myeloma | Phase 1 | Completed | - | Japan ... more >> Local Institution Nagoya-shi, Aichi, Japan, 4600001 Local Institution Nagoya-shi, Aichi, Japan, 4678602 Local Institution Niigata-shi, Niigata, Japan, 9518566 Collapse << |
| NCT03032861 | Intestinal Diseases | Not Applicable | Completed | - | Brazil ... more >> Sao Paulo State University "Julio de Mesquita Filho", Faculdade de Ciências Farmacêuticas Araraquara, Sao Paulo, Brazil, 14800-903 Collapse << |
| NCT03282955 | Patients Requiring Home Parent... more >>eral Nutrition Collapse << | Phase 4 | Recruiting | September 1, 2019 | France ... more >> Hospices Civiles de Lyon - Centre hospitalier Lyon Sud Recruiting Pierre-Bénite, France Contact: Cécile Chambrier, Dr. Italy Department of Medical and Surgical Science, University of Bologna, Center for Chronic Intestinal Failure, Department of Digestive System, St. Orsola-Malpighi Hospital Recruiting Bologna, Italy Contact: Loris Pironi, Prof. Netherlands Radboud university medical center Recruiting Nijmegen, Netherlands Contact: Geert Wanten, Dr. Poland Stanley Dudrick`s Memorial Hospital Recruiting Skawina, Poland Contact: Stanislaw Klek, Prof. Principal Investigator: Stanislaw Klek, Prof. Samodzielny Publiczny Szpital Kliniczny, im. prof. dr W. Orlowskiego, Centrum Medycznego Ksztalcenia Podyplomowego, Oddzial Kliniczny Zywienia i Chirurgii Recruiting Warszawa, Poland Contact: Jacek Sobocki, Dr. Wojewódzkim Specjalistycznym Szpitalem im. M. Pirogowa w Łodzi , Oddział Chirurgii Ogólnej i Naczyniowej Recruiting Łódź, Poland Contact: Marek Kunecki, Dr. United Kingdom University of East Anglia, Faculty of Medicine and Health Sciences Not yet recruiting Norwich, United Kingdom Contact: Alastair Forbes, Prof. Collapse << |
| NCT01120873 | Metabolic Syndrome | Phase 3 | Completed | - | Taiwan ... more >> Division of Endocrinology and Metabolism, Department of Internal Medicine, Taichung Veterans General Hospital Taichang, Taiwan, 407 Collapse << |
| NCT01072175 | Cancer | Phase 2 | Completed | - | United States, California ... more >> Novartis Investigative Site Los Angeles, California, United States, 90025 Novartis Investigative Site San Francisco, California, United States, 94115 United States, Colorado Novartis Investigative Site Aurora, Colorado, United States, 80045 United States, Connecticut Novartis Investigative Site New Haven, Connecticut, United States, 06520 United States, Florida Novartis Investigative Site Tampa, Florida, United States, 33612 United States, Maryland Novartis Investigative Site Lutherville-Timonium, Maryland, United States, 21093 United States, Massachusetts Novartis Investigative Site Boston, Massachusetts, United States, 02114 Novartis Investigative Site Boston, Massachusetts, United States, 02215 United States, Minnesota Novartis Investigative Site Rochester, Minnesota, United States, 55905 United States, Pennsylvania Novartis Investigative Site Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Novartis Investigative Site Nashville, Tennessee, United States, 37203 Novartis Investigative Site Nashville, Tennessee, United States, 37232 United States, Texas Novartis Investigative Site Houston, Texas, United States, 77030-4009 Australia, New South Wales Novartis Investigative Site Westmead, New South Wales, Australia, 2145 Australia, Victoria Novartis Investigative Site Heidelberg, Victoria, Australia, 3084 Collapse << |
| NCT01817036 | Vitamin D Deficiency | Phase 1 | Completed | - | China, Shanghai ... more >> Institute for Nutritional Sciences, Chinese Academy of Sciences Shanghai, Shanghai, China, 200031 Collapse << |
| NCT02492958 | - | - | Completed | - | - |
| NCT02955602 | Primary Biliary Cirrhosis | Phase 2 | Active, not recruiting | January 2020 | - |
| NCT02778477 | Normal Healthy | Phase 1 | Terminated(Preliminary analysi... more >>s of accrued data failed to indicate any meaningful pharmacodynamic response.) Collapse << | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT02625454 | Fever Oxidati... more >>ve Stress Collapse << | Phase 2 | Recruiting | October 2018 | United States, New York ... more >> Richmond University Medical Center Recruiting Staten Island, New York, United States, 10310 Contact: Nisha A Lakhi, MD 718-818-2109 NLAKHI@RUMCSI.ORG Contact: Michale M Moretti, MD 718-818-4213 MMORETTI@RUMCSI.ORG Collapse << |
| NCT03684044 | Influenza | Phase 3 | Recruiting | July 10, 2021 | - |
| NCT02936596 | Hepatitis, Autoimmune ... more >> Cholangitis Liver Cirrhosis, Biliary Cholestasis Collapse << | Not Applicable | Recruiting | August 2018 | China, Sichuan ... more >> West China Hospital Recruiting Chengdu, Sichuan, China, 610041 Contact: Xiaoli Fan, MM +8618008028017 13980433451@163.com Contact: Li Yang, MD yangli_hx@scu.edu.cn Collapse << |
| NCT00734474 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT02841813 | - | - | Recruiting | December 2019 | China, Shanghai ... more >> Xinhua Hospital, Shanghai Jiao Tong University School of Medicine Recruiting Shanghai, Shanghai, China, 200092 Contact: Jie Zhu, MD, PhD 86-021-2507-8999 ext 6418 jacky284868@163.com Collapse << |
| NCT02927873 | Respiratory Syncytial Virus In... more >>fections Collapse << | Phase 2 | Recruiting | November 17, 2020 | - |
| NCT00117949 | Prostate Cancer | Phase 2 | Completed | - | United States, California ... more >> Advanced Urology Medical Center Anaheim, California, United States, 92801 South Orange County Medical Research Center Laguna Woods,, California, United States, 92653 San Bernardino Urological Associates Medical Group San Bernardino, California, United States, 92404 Western Clinical Research Torrance, California, United States, 90505 United States, Colorado Urology Associate PC` Denver, Colorado, United States, 80210 United States, Florida SW Florida Urological Associates Fort Myers, Florida, United States, 33907 Pinellas Urology, Inc. St. Petersburg, Florida, United States, 33710 United States, Maryland Drs. Werner, Murdock & Francis, PA Greenbelt, Maryland, United States, 20770 United States, Nevada Nevada Urology Associates Reno, Nevada, United States, 89511 United States, Oklahoma Urology Specialists of Oklahoma, Inc. Tulsa, Oklahoma, United States, 74104 United States, Texas Urology Clinics of NorthTexas, PA Dallas, Texas, United States, 75231 Urology San Antonio Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT02342548 | Huntington's Disease | Phase 2 | Terminated(Study terminated on... more >> 15DEC2016 due to study A8241021 showing no significant difference on primary endpoint between PF-02545920 & placebo. No safety concerns.) Collapse << | - | - |
| NCT02397447 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00117949 | - | - | Completed | - | - |
| NCT00734474 | - | - | Completed | - | - |
| NCT01416571 | Influenza | Phase 2 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Mesa, Arizona, United States, 85213 Collapse << |
| NCT02342548 | - | - | Terminated(Study terminated on... more >> 15DEC2016 due to study A8241021 showing no significant difference on primary endpoint between PF-02545920 & placebo. No safety concerns.) Collapse << | - | - |
| NCT02548832 | Mixed Dyslipidemia | Phase 2 | Unknown | July 2016 | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: Esperanza Martínez Abundis, PhD Science (33) 1058-5200 ext 34211 esperanzamartnezabundi@yahoo.com Collapse << |
| NCT01072175 | - | - | Completed | - | - |
| NCT00048932 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01416571 | - | - | Completed | - | - |
| NCT02549924 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(no financial suppor... more >>t) Collapse << | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT00044278 | Epilepsy | Phase 2 | Completed | - | - |
| NCT01332019 | - | - | Completed | - | - |
| NCT02769611 | Heart Failure | Phase 1 Phase 2 | Recruiting | December 2019 | United States, Ohio ... more >> The Lindner Center for Research and Education at The Christ Hospital Recruiting Cincinnati, Ohio, United States, 45219 Contact: Gregory Egnaczyk, MD 513-585-1777 Principal Investigator: Gregory Egnaczyk, MD Collapse << |
| NCT02893943 | Obesity | Not Applicable | Active, not recruiting | - | Canada, Manitoba ... more >> Richardson Centre for Functional Foods and Nutraceuticals Winnipeg, Manitoba, Canada, R3T 2N2 Collapse << |
| NCT00048932 | - | - | Completed | - | - |
| NCT01332019 | Relapsing Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT02708966 | Impaired Glucose Tolerance | Phase 2 | Recruiting | December 2019 | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: Esperanza Martínez-Abundis, PhD 52+ (33) 1058-5200 ext 34211 esperanzamartnezabundi@yahoo.com Collapse << |
| NCT03215043 | Pre Diabetes | Not Applicable | Recruiting | June 28, 2019 | Brazil ... more >> Sao Paulo State University "Julio de Mesquita Filho", Faculdade de Ciências Farmacêuticas Recruiting Araraquara, Sao Paulo, Brazil, 14800-903 Contact: Thais B Cesar, Ph.D. +55 16 3301-6927 tcesar@fcfar.unesp.br Collapse << |
| NCT03304938 | Diabetes Mellitus, Type 2 ... more >> Sleep Disturbance Collapse << | Not Applicable | Recruiting | February 18, 2018 | Iran, Islamic Republic of ... more >> Shiraz University of Medical Sciences Recruiting Shiraz, Fars, Iran, Islamic Republic of Contact: Zeinab Nasiri Lari, M.D. 09173139409 dr.znasiri@gmail.com Collapse << |
| NCT02621060 | Impaired Glucose Tolerance | Phase 2 | Unknown | February 2017 | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02956837 | Respiratory Syncytial Virus In... more >>fections Collapse << | Phase 1 Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Gent, Belgium, 9000 Estonia GSK Investigational Site Tallinn, Estonia, 10117 GSK Investigational Site Tartu, Estonia, 50106 France GSK Investigational Site Clermont-Ferrand, France, 63003 GSK Investigational Site Paris, France, 75679 Germany GSK Investigational Site Wuerzburg, Bayern, Germany, 97070 GSK Investigational Site Hannover, Niedersachsen, Germany, 30159 GSK Investigational Site Goch, Nordrhein-Westfalen, Germany, 47574 Collapse << |
| NCT03516721 | Obesity Adole... more >>scent Obesity Collapse << | Not Applicable | Completed | - | Belgium ... more >> Jessa Ziekenhuis Hasselt, Belgium, 3500 Collapse << |
| NCT03714438 | Glucose Tolerance | Phase 2 | Completed | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03622762 | Diabetic Nephropathy Type 2 | Phase 2 | Recruiting | June 20, 2019 | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: Fernando Grover Paez, PhD (33) 10585200 ext 34215 fgroverp@hotmail.com Collapse << |
| NCT00802464 | Herpes Zoster | Phase 2 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Phoenix, Arizona, United States, 85020 United States, Florida GSK Investigational Site Pembroke Pines, Florida, United States, 33024 United States, Nevada GSK Investigational Site Las Vegas, Nevada, United States, 89130 United States, New Jersey GSK Investigational Site Edison, New Jersey, United States, 08817 United States, North Carolina GSK Investigational Site Raleigh, North Carolina, United States, 27612 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44122 United States, Pennsylvania GSK Investigational Site Pittsburg, Pennsylvania, United States, 15236 Czechia GSK Investigational Site Hradec Kralove, Czechia, 500 01 Spain GSK Investigational Site Barcelona, Spain, 08035 GSK Investigational Site Barcelona, Spain GSK Investigational Site Mahadahonda( Madrid, Spain, 28222 GSK Investigational Site Marid, Spain, 28040 Collapse << |
| NCT01934192 | Gastroparesis | Phase 2 | Terminated | - | United States, Colorado ... more >> GSK Investigational Site Aurora, Colorado, United States, 80045 United States, Georgia GSK Investigational Site Augusta, Georgia, United States, 30909 United States, Kentucky GSK Investigational Site Louisville, Kentucky, United States, 40202 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 Australia, New South Wales GSK Investigational Site Randwick, New South Wales, Australia, 2031 Australia, Queensland GSK Investigational Site Southport, Queensland, Australia, 4215 Australia, South Australia GSK Investigational Site Adelaide, South Australia, Australia, 5000 GSK Investigational Site Woodville, South Australia, Australia, 5011 Canada, Alberta GSK Investigational Site Calgary, Alberta, Canada, T1Y 6J4 GSK Investigational Site Calgary, Alberta, Canada, T2N 2T9 Canada, Ontario GSK Investigational Site Hamilton, Ontario, Canada, L8L 2X2 GSK Investigational Site Kingston, Ontario, Canada, K7L 2V7 GSK Investigational Site Ottawa, Ontario, Canada, K1H 8L6 GSK Investigational Site Ottawa, Ontario, Canada, K1Y 4E9 GSK Investigational Site Toronto, Ontario, Canada, M5G 1X5 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H2X 3J4 GSK Investigational Site Sainte-Foy, Quebec, Canada, G1V 4G5 GSK Investigational Site Sherbrooke, Quebec, Canada, J1H 5N4 Canada GSK Investigational Site Quebec, Canada, G1J 1Z4 Collapse << |
| NCT02956837 | - | - | Completed | - | - |
| NCT02914249 | Obesity | Not Applicable | Completed | - | - |
| NCT01934192 | - | - | Terminated | - | - |
| NCT01223365 | Chronic Pain | Phase 3 | Completed | - | - |
| NCT01223365 | - | - | Completed | - | - |
| NCT00802464 | - | - | Completed | - | - |
| NCT01018017 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Neuss, Nordrhein-Westfalen, Germany, 41460 Collapse << |
| NCT01187888 | Progressive Supranuclear Palsy | Phase 3 | Terminated(IMP used off label ... more >>by phys. in pat. with PSP. Thus no more eligible patients were available for the study(pre-treatm.with Rasagiline=exclusion criterion) Collapse << | - | Germany ... more >> Department of Neurology and Palliative Care Klinikum der Universität München (Hospital of the University of Munich) München, Germany, 81377 Collapse << |
| NCT00298272 | Rheumatoid Arthritis | Phase 2 | Terminated(Sponsor decided to ... more >>end long term extension phase for business reasons unrelated to safety.) Collapse << | - | United States, Alabama ... more >> Research Site Huntsville, Alabama, United States, 35801 United States, Arizona Research Site Paradise Valley, Arizona, United States, 85253 United States, California Research Site Palm Desert, California, United States, 92260 United States, Florida Research Site Jupiter, Florida, United States, 33458 Research Site Sarasota, Florida, United States, 34239 United States, Idaho Research Site Boise, Idaho, United States, 83702 United States, Michigan Research Site Kalamazoo, Michigan, United States, 49048 United States, Missouri Research Site St. Louis, Missouri, United States, 63141 United States, Ohio Research site Chardon, Ohio, United States, 44024 Research Site Mayfield Village, Ohio, United States, 44143 United States, Oklahoma Research Site Oklahoma City, Oklahoma, United States, 73103 Research Site Tulsa, Oklahoma, United States, 74135 United States, Pennsylvania Research Site Duncansville, Pennsylvania, United States, 16635 United States, Texas Research Site Dallas, Texas, United States, 75231 Research Site Houston, Texas, United States, 77074 Research Site San Antonio, Texas, United States, 78217 United States, Utah Research Site Salt Lake City, Utah, United States, 84132 United States, Vermont Research Site Burlington, Vermont, United States, 05401 Collapse << |
| NCT03217604 | Healthy Volunteers | Phase 1 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT00298272 | - | - | Terminated(Sponsor decided to ... more >>end long term extension phase for business reasons unrelated to safety.) Collapse << | - | - |
| NCT01313663 | Lung Cancer, Small Cell | Phase 2 | Terminated | - | - |
| NCT01313663 | - | - | Terminated | - | - |
| NCT02966418 | Hypoxic Preconditioning ... more >> Carotid Artery Stenosis Ischemic Cerebrovascular Disease Collapse << | Not Applicable | Completed | - | China, Tianjin ... more >> Tianjin HuanHu Hospital Tianjin, Tianjin, China, 300350 Collapse << |
| NCT01899703 | Cholestasis, Intrahepatic | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Birmingham, West Midlands, United Kingdom, B15 2WB GSK Investigational Site Cambridge, United Kingdom GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT00976820 | Influenza | Phase 2 | Completed | - | - |
| NCT02000817 | Diabetes Mellitus, Type 1 | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Brussels, Belgium, 1090 GSK Investigational Site Bruxelles, Belgium, 1070 GSK Investigational Site Edegem, Belgium, 2650 GSK Investigational Site Gent, Belgium, 9000 GSK Investigational Site Leuven, Belgium, 3000 GSK Investigational Site Liège, Belgium, 4000 Collapse << |
| NCT02457637 | - | - | Unknown | December 2017 | China, Beijing ... more >> Ditan Hospital of integrated traditional Chinese and Western Medicine Center Enrolling by invitation Beijing, Beijing, China, 100015 302ed Military Hospital of PLA Enrolling by invitation Beijing, Beijing, China, 100039 China, Chongqing Southwest Hospital of Third Military Medical University Recruiting Chongqing, Chongqing, China, 400038 Contact: Baoyan Xu +86-13032396309 xby1977@126.com China, Fujian Fuzhou general hospital of Xiamen university Active, not recruiting Fuzhou, Fujian, China, 350025 China, Guangdong Southern hospital infection department Recruiting Guangzhou, Guangdong, China, 510515 Contact: Jinjun Chen, Professor 13902246336 chjj@smu.edu.cn China, Hebei Affiliated Hospital of the Armed Police Logistics College Active, not recruiting Tianjing, Hebei, China, 300499 China, Hubei Taihe Hospital Enrolling by invitation Shiyan, Hubei, China, 442000 Wuhan Union Hospital of Huazhong University of Science and Technology Enrolling by invitation Wuhan, Hubei, China, 430022 China, Hunan Xiangya hospital of Central South University Enrolling by invitation Changsha, Hunan, China, 410008 China, Jilin The First Affiliated Hospital of Jilin University Enrolling by invitation Changchun, Jilin, China, 130021 China, Shandong The Second Hospital of Shandong University Enrolling by invitation Jinan, Shandong, China, 250033 China, Shanghai Renji hospital of Shanghai Jiao Tong University School of Medical Enrolling by invitation Shanghai, Shanghai, China, 200001 Shanghai Public Health Clinical Center Enrolling by invitation Shanghai, Shanghai, China, 200083 China, Sichuan West China Hospital of Sichuan University Completed Chengdu, Sichuan, China, 610041 China, Xinjiang The First Teaching Hospital of Xinjiang Medical University Enrolling by invitation Wulumuqi, Xinjiang, China, 830054 Collapse << |
| NCT00988533 | - | - | Completed | - | - |
| NCT01953770 | Childhood Acute Lymphoblastic ... more >>Leukemia Collapse << | Not Applicable | Active, not recruiting | June 2018 | - |
| NCT00988533 | Head Lice | Phase 1 | Completed | - | United States, Arkansas ... more >> Children's Investigational Research Program Bentonville, Arkansas, United States, 72712 United States, California Universal BioPharma Research, Inc Dinuba, California, United States, 93618 Impact Clinical Trials Los Angeles, California, United States, 90036 United States, Florida Lice Source Solutions Inc Plantation, Florida, United States, 33313 United States, Mississippi Spence Medical Research L.L.C Picayune, Mississippi, United States, 39466 Collapse << |
| NCT01818544 | Bronchiectasis | Phase 2 | Completed | - | France ... more >> Nice, France, 06000 Perpignan, France, 66025 Germany Donaustauf, Bayern, Germany, 93093 Rüdersdorf, Brandenburg, Germany, 15562 Hannover, Niedersachsen, Germany, 30167 Leipzig, Sachsen, Germany, 04103 Großhansdorf, Schleswig-Holstein, Germany, 22927 Kiel, Schleswig-Holstein, Germany, 24105 Lübeck, Schleswig-Holstein, Germany, 23538 Jena, Thüringen, Germany, 07740 Hamburg, Germany, 20354 Italy Orbassano, Torino, Italy, 10043 Genova, Italy, 16132 Milano, Italy, 20142 Napoli, Italy, 80131 Padova, Italy, 35128 Siena, Italy, 53100 Spain Badalona, Barcelona, Spain, 08916 Salt, Girona, Spain, 17190 Majadahonda, Madrid, Spain, 28222 Madrid, Spain, 28046 United Kingdom Southampton, Hampshire, United Kingdom, SO16 6YD Cottingham, Humberside, United Kingdom, HU16 5JQ Leicester, Leicestershire, United Kingdom, LE3 9QP Liverpool, Merseyside, United Kingdom, L9 7JU Sheffield, South Yorkshire, United Kingdom, S5 7AU Dundee, Tayside, United Kingdom, DD2 1UB Newcastle Upon Tyne, Tyne and Wear, United Kingdom, NE7 7DN South Shields, Tyne and Wear, United Kingdom, NE34 0PL Wolverhampton, West Midlands, United Kingdom, WV10 0QP Bradford, West Yorkshire, United Kingdom, BD9 6RJ Glasgow, United Kingdom, G42 9TY London, United Kingdom, EC1M 6BQ Collapse << |
| NCT03468556 | NASH - Nonalcoholic Steatohepa... more >>titis Collapse << | Phase 2 | Not yet recruiting | June 30, 2019 | - |
| NCT00976820 | - | - | Completed | - | - |
| NCT03453060 | Thrombosis | Phase 1 | Active, not recruiting | November 2018 | United States, Arizona ... more >> Celerion Tempe, Arizona, United States, 85283 Collapse << |
| NCT00870740 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 2 | Completed | - | - |
| NCT00870740 | - | - | Completed | - | - |
| NCT03318731 | Muscle Damage ... more >> Inflammation Collapse << | Not Applicable | Recruiting | September 2018 | United States, Texas ... more >> UMHB Human Performance Lab Recruiting Belton, Texas, United States, 76513 Contact: Stacie L Urbina, MsEd 254-295-4280 ext 4280 surbina@umhb.edu Contact: Lem W Taylor, PhD 254-295-4895 ext 4895 ltyalor@umhb.edu Sub-Investigator: Katelyn B Villa, B.S. Sub-Investigator: Allyson K Bermea Sub-Investigator: Torrey Boggs Sub-Investigator: Matthias R Tinnin, B.S. Sub-Investigator: Javier A Zaragoza Sub-Investigator: Seth M Bates Sub-Investigator: Colin D Wilborn, PhD Sub-Investigator: Cliffa Foster, EdD Collapse << |
| NCT03551522 | NASH - Nonalcoholic Steatohepa... more >>titis Collapse << | Phase 2 | Recruiting | December 31, 2020 | United States, Arizona ... more >> CymaBay Research Site Recruiting Glendale, Arizona, United States, 85306 CymaBay Research Site Recruiting Tucson, Arizona, United States, 85712 United States, California CymaBay Research Site Recruiting Los Angeles, California, United States, 90057 United States, Florida CymaBay Research Site Recruiting Boca Raton, Florida, United States, 33434 CymaBay Research Site Recruiting Lakewood Ranch, Florida, United States, 34211 United States, Mississippi CymaBay Research Site Recruiting Flowood, Mississippi, United States, 39232 United States, Tennessee CymaBay Research Site Recruiting Clarksville, Tennessee, United States, 37040 CymaBay Research Site Recruiting Germantown, Tennessee, United States, 38138 CymaBay Research Site Recruiting Hermitage, Tennessee, United States, 37076 CymaBay Research Site Recruiting Knoxville, Tennessee, United States, 37909 United States, Texas CymaBay Research Site Recruiting Chandler, Texas, United States, 85224 CymaBay Research Site Recruiting Dallas, Texas, United States, 75246 CymaBay Research Site Recruiting Live Oak, Texas, United States, 78233 CymaBay Research Site Recruiting Rollingwood, Texas, United States, 78746 Collapse << |
| NCT03432416 | Healthy Women ... more >> Female Contraception Collapse << | Phase 2 | Recruiting | June 20, 2020 | United States, California ... more >> Essential Access Health Recruiting Los Angeles, California, United States, 30010 Contact: Kieumai Vo 213-386-5614 ext 4592 KVo@essentialaccess.org Contact: Ron Frezeries 213-386-5614 ext 4403 RFrezieres@essentialaccess.org Principal Investigator: Anita Nelson University of California, Davis Recruiting Sacramento, California, United States, 95817 Contact: Courtney Overstreet 916-734-6846 cboverstreet@ucdavis.edu Contact: Aubrey Blanton 916-734-6846 akblanton@ucdavis.edu Principal Investigator: Mitchell Creinin United States, Colorado University of Colorado Denver Recruiting Aurora, Colorado, United States, 80045 Contact: Ines Crato 303-724-5559 INES.CRATO@UCDENVER.EDU Contact: Rebecca Seale 303-724-2013 REBECCA.SEALE@UCDENVER.EDU Principal Investigator: Stephanie Teal United States, Maryland Johns Hopkins Bayview Medical Center Recruiting Baltimore, Maryland, United States, 21224 Contact: Katrina Thaler 410-550-8506 kstouff3@jhmi.edu Contact: Lauren Beal 410-550-4825 lbeal2@jhu.edu Principal Investigator: Anne Burke United States, New York Bellevue Hospital Center Recruiting New York, New York, United States, 10016 Contact: Anna Davis 212-263-6253 anna.davis@nyumc.org Principal Investigator: Treasure Walker Columbia University Recruiting New York, New York, United States, 10032 Contact: Subrina Bisnauth 212-305-8031 Snb2147@cumc.columbia.edu Contact: Connie Colon 212-305-6098 cc3238@cumc.columbia.edu Principal Investigator: Carolyn Westhoff United States, Ohio University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45267 Contact: Deborah Boerschig 513-584-4100 BOERSCDA@UCMAIL.UC.EDU Contact: Tiffany Rupert 513-584-4100 tiffany.rupert@uc.edu Principal Investigator: Michael Thomas United States, Oregon Oregon Health Science University Recruiting Portland, Oregon, United States, 97239 Contact: Ariela Schnyer 503-494-3173 schnyer@ohsu.edu Contact: Alison Edelman 503-494-5949 edelmana@ohsu.edu Principal Investigator: Jeffrey Jensen United States, Pennsylvania University of Pennsylvania School of Medicine Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Andrea Morley 215-615-4202 Andrea.morley@uphs.upenn.edu Contact: Kieran Alessi 215-615-3664 Kieran.Alessi@uphs.upenn.edu Principal Investigator: Kurt Barnhart United States, Utah University of Utah Recruiting Salt Lake City, Utah, United States, 84132 Contact: Jasmin Alcantra 801-213-3796 jasmin.alcantara@hsc.utah.edu Contact: Amy Orr 801-213-2774 amy.orr@hsc.utah.edu Principal Investigator: David Turok United States, Virginia Eastern Virginia Medical School Recruiting Norfolk, Virginia, United States, 23507 Contact: Jackie Rocccazella 757-446-7161 roccazj@evms.edu Contact: Kim Pitts 757-446-8925 pittska@evms.edu Principal Investigator: David Archer Collapse << |
| NCT03033004 | Subcutaneous Fat Disorder | Not Applicable | Completed | - | Brazil ... more >> Centro de Estudos e Formação Avançada Ibramed. Amparo, São Paulo, Brazil Collapse << |
| NCT00457002 | Venous Thrombosis ... more >> Pulmonary Embolism Collapse << | Phase 3 | Completed | - | - |
| NCT03523455 | Iron-deficiency | Not Applicable | Terminated(Unable to recruit q... more >>ualified participants) Collapse << | - | United States, Texas ... more >> UMHB Human Performance Lab Belton, Texas, United States, 76513 Collapse << |
| NCT01097655 | - | - | Completed | - | - |
| NCT01097655 | - | - | Completed | - | - |
| NCT02226354 | Healthy | Phase 1 | Completed | - | Canada, New Brunswick ... more >> Université de Moncton Moncton, New Brunswick, Canada, E1A 3E9 Collapse << |
| NCT01777295 | Hepatitis B | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Antwerpen, Belgium, 2060 Collapse << |
| NCT00457002 | - | - | Completed | - | - |
| NCT03320538 | Peptic Ulcer Disease | Not Applicable | Recruiting | July 1, 2019 | China, Jiangxi ... more >> Jiangxi University of Traditional Chinese Medicine Recruiting Nanchang, Jiangxi, China, 330004 Contact: Xu Zhou, Ph.D. +8618780085060 zhouxu_ebm@hotmail.com Contact: Xiaofan Chen, Ph.D. +86079187118012 841347982@qq.com Collapse << |
| NCT02800668 | - | - | Completed | - | Germany ... more >> Herz- und Diabeteszentrum Bad Oeynhausen, Germany, 32545 Collapse << |
| NCT01489410 | Hepatocellular Carcinoma | Not Applicable | Withdrawn(Insufficient staff) | - | United States, Minnesota ... more >> Masonic Cancer Center, University of Minnesota Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT03268005 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Active, not recruiting | January 29, 2019 | - |
| NCT02926456 | - | - | Completed | - | - |
| NCT01173120 | - | - | Completed | - | - |
| NCT00492544 | Infections, Papillomavirus | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Saitama, Japan, 360-0812 GSK Investigational Site Saitama, Japan, 360-0846 GSK Investigational Site Tokyo, Japan, 136-0073 GSK Investigational Site Tokyo, Japan, 154-0024 Collapse << |
| NCT01173120 | Rheumatoid Arthritis (RA) | Phase 3 | Completed | - | - |
| NCT00492544 | - | - | Completed | - | - |
| NCT03731260 | Indolent Systemic Mastocytosis... more >> Smoldering Systemic Mastocytosis Collapse << | Phase 2 | Not yet recruiting | November 2023 | - |
| NCT01997762 | Gestational Diabetes | Phase 4 | Unknown | December 2016 | Canada, Manitoba ... more >> Manitoba Institute of Child Health Recruiting Winnipeg, Manitoba, Canada, R3E 3P4 Principal Investigator: Shayne P Taback, MD Collapse << |
| NCT02398279 | Schizophrenia ... more >> Schizoaffective Disorder Collapse << | Not Applicable | Completed | - | Turkey ... more >> Hacettepe University Faculty of Medicine Department of Psychiatry Ankara, Turkey, 06100 Collapse << |
| NCT01760148 | - | - | Unknown | March 2014 | China, Jilin ... more >> First Hospital Jilin University Recruiting Changchun, Jilin, China, 130061 Contact: Yu Pan , PhD/MD 0431-88782120 panyu20000@yahoo.com.cn Contact: Xiumei Chi , PhD/MD 0431-88782222 xiumeichi@hotmail.com Principal Investigator: Yu Pan, PhD/MD Collapse << |
| NCT01250652 | Chronic Urticaria | Phase 4 | Completed | - | Bulgaria ... more >> Medical University Sofia, Department of Allergology Sofia, Bulgaria, 1431 Collapse << |
| NCT01518517 | Acute Lymphoblastic Leukemia, ... more >>in Relapse Collapse << | Phase 2 Phase 3 | Completed | - | Belgium ... more >> Hopital Des Enfants Reine Fabiola Bruxelles, Belgium Chr de La Citadelle Liege, Belgium France Chu D'Angers Angers, France Hopital Saint Jacques Besancon, France Hopital Pellegrin Enfants Bordeaux, France Chu Estaing Clermont Ferrand, France Hopital Henri Mondor Creteil, France Chu Grenoble Grenoble, France Chru Lille - Hop Jeanne de Flandres Lille, France Institut Hematologie Oncologie Pediatrique Lyon, France Institut Paoli Calmettes Marseille, France Hopital Mere Enfant Nantes, France Hotel Dieu Nantes, France Hopital de L'Archet 2 Nice, France Hopital Armand Trousseau Paris, France Hopital Robert Debre Paris, France Hopital Saint Louis Paris, France Hopital Haut-Leveque Pessac, France Hopital Lyon Sud Pierre Benite, France Chru Hopital Sud Rennes, France Centre Henri Becquerel Rouen, France Chu Hopital Nord Saint Etienne, France Institut Cancerologie de La Loire Saint Priest En Jarez, France Hopital de Hautepierre Strasbourg, France Chu de Toulouse Enfants Toulouse, France Hotel Dieu Valenciennes, France Hopital Brabois Enfants Vandoeuvre Les Nancy, France Hopital de Brabois Vandoeuvre Les Nancy, France Collapse << |
| NCT02540759 | Healthy | Phase 1 | Completed | - | Canada, New Brunswick ... more >> Université de Moncton Moncton, New Brunswick, Canada, E1A 3E9 Collapse << |
| NCT02226939 | Liver Cirrhosis ... more >> Hepatitis C, Chronic Collapse << | Phase 2 | Completed | - | - |
| NCT02442687 | Nonalcoholic Steatohepatitis | Phase 2 | Completed | - | United States, Alabama ... more >> Digestive Disease Specialists of the Southeast Dothan, Alabama, United States, 36305 United States, Illinois Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 United States, Nevada Digestive Associates Las Vegas, Nevada, United States, 89102 United States, North Carolina Duke University Durham, North Carolina, United States, 27710 United States, Ohio Digestive Disease Specialists Cincinnati, Ohio, United States, 45219 United States, Texas Brook Army Medical Center Houston, Texas, United States, 78234 United States, Virginia University of Virginia Health Systems Charlottesville, Virginia, United States, 22903 Medical College of Virginia Richmond, Virginia, United States, 23298 Collapse << |
| NCT02226952 | Hepatitis C, Chronic | Phase 1 Phase 2 | Completed | - | - |
| NCT01059617 | Influenza | Phase 1 | Completed | - | United States, Massachusetts ... more >> GSK Investigational Site Milford, Massachusetts, United States, 01757 United States, North Carolina GSK Investigational Site Salisbury, North Carolina, United States, 28144 GSK Investigational Site Winston-Salem, North Carolina, United States, 27103 Collapse << |
| NCT00452673 | Advanced Breast Cancer | Phase 1 | Completed | - | United States, California ... more >> City Of Hope National Medical Center Duarte, California, United States, 91010-3000 United States, Illinois Northwestern University Feinberg School Of Medicine Chicago, Illinois, United States, 60611 United States, New York New York Presbyterian Hospital New York, New York, United States, 10065 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109-1023 Italy Local Institution Rozzano, Milano, Italy, 20089 Spain Local Institution Barcelona, Spain, 08035 Local Institution Sevilla, Spain, 41013 Collapse << |
| NCT02616172 | Sensorineural Hearing Loss | Phase 1 Phase 2 | Suspended(Restructuring Cell L... more >>ab) Collapse << | September 2018 | United States, Florida ... more >> Florida Hospital for Children Orlando, Florida, United States, 32803 Collapse << |
| NCT03191383 | Respiratory Syncytial Virus In... more >>fections Collapse << | Phase 2 | Withdrawn(Study was canceled d... more >>ue to instability of the PreF antigen during manufacturing. No safety concern has been identified in past or ongoing studies.) Collapse << | - | United States, Kansas ... more >> GSK Investigational Site Newton, Kansas, United States, 67114 United States, New York GSK Investigational Site Syracuse, New York, United States, 13210 United States, Washington GSK Investigational Site Ellensburg, Washington, United States, 98926 Finland GSK Investigational Site Oulu, Finland, 90220 GSK Investigational Site Seinajoki, Finland, 60100 Spain GSK Investigational Site Aravaca, Spain, 28023 GSK Investigational Site Burgos, Spain, 09006 GSK Investigational Site Madrid, Spain, 28040 GSK Investigational Site Majadahonda (Madrid), Spain, 28222 GSK Investigational Site Santiago de Compostela, Spain, 15706 GSK Investigational Site Santiago, Spain, 15705 GSK Investigational Site Sevilla, Spain, 41014 Collapse << |
| NCT00452673 | - | - | Completed | - | - |
| NCT01104935 | Breast Cancer | Phase 3 | Active, not recruiting | April 30, 2018 | Japan ... more >> Aichi Cancer Center Hospital Nagoya, Aichi, Japan, 464-8681 Collapse << |
| NCT01059617 | - | - | Completed | - | - |
| NCT02663089 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT02122211 | Men Carrying 2 Minor Alleles f... more >>or Choline Dehydrogenase rs12676 Male Infertility Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> UNC Nutrition Research Institute Kannapolis, North Carolina, United States, 28081 Collapse << |
| NCT02155829 | PTSD | Phase 1 Phase 2 | Unknown | - | United States, Maryland ... more >> Walter Reed National Military Medical Center Recruiting Bethesda, Maryland, United States, 20814 Contact: Patricia T Spangler, Ph.D. 240-620-4076 patricia.spangler.ctr@usuhs.edu Contact: Jennifer Bornemann, LCSW-C 301-295-3485 jennifer.bornemann@usuhs.edu Principal Investigator: David M Benedek, MD United States, New York Syracuse VA Medical Center, Center for Integrated Healthcare (116C) Recruiting Syracuse, New York, United States, 13210 Contact: Shannon McKenzie 315-425-3954 Shannon.McKenzie@va.gov Contact: Kyle Possemato, PhD 315-425-3551 Kyle.Possemato@va.gov Principal Investigator: Kyle Possemato, PhD Collapse << |
| NCT01348971 | Coronary Artery Disease | Phase 2 | Completed | - | Sweden ... more >> Karolinska University Hospital Stockholm, Sweden, S-171 76 Collapse << |
| NCT02647554 | Sepsis Septic... more >> Shock Collapse << | Phase 4 | Unknown | August 2018 | China, Beijing ... more >> Peking Union Medical College Hospital Recruiting Beijing, Beijing, China Contact: Bin Du, MD 0086-10-69155036 dubin98@gmail.com Collapse << |
| NCT03732690 | Type 2 Diabetes | Not Applicable | Not yet recruiting | December 30, 2020 | France ... more >> Hôpital PITIE SALPETRIERE - APHP Not yet recruiting Paris, France, 75013 Contact: Karine CLEMENT, MD, PhD Collapse << |
| NCT03395405 | Gastroenteritis Norovirus | Phase 2 | Recruiting | May 22, 2021 | United States, Alabama ... more >> University of Alabama Hospital - Infectious Diseases Not yet recruiting Birmingham, Alabama, United States, 35249-0001 United States, Illinois Northwestern University - Comprehensive Transplant Center Recruiting Chicago, Illinois, United States, 60611-2927 United States, Kansas University of Kansas Medical Center - Infectious Diseases Recruiting Kansas City, Kansas, United States, 66160-8500 United States, Maryland Johns Hopkins Hospital - Medicine - Infectious Diseases Not yet recruiting Baltimore, Maryland, United States, 21287-0005 United States, Michigan University of Michigan - Infectious Disease Clinic at Taubman Center Not yet recruiting Ann Arbor, Michigan, United States, 48109 United States, Nebraska University of Nebraska Medical Center - Infectious Diseases Not yet recruiting Omaha, Nebraska, United States, 68198-5400 United States, Ohio Cincinnati Children's Hospital Medical Center - Infectious Diseases Not yet recruiting Cincinnati, Ohio, United States, 45229-3039 United States, Pennsylvania University of Pittsburgh - Medicine - Infectious Diseases Recruiting Pittsburgh, Pennsylvania, United States, 15213-3403 Children's Hospital of Pittsburgh of UPMC - Pediatric Infectious Diseases Recruiting Pittsburgh, Pennsylvania, United States, 15224-1529 United States, Texas University of Texas Southwestern Medical Center - Internal Medicine Subspecialties Clinic Recruiting Dallas, Texas, United States, 75390-8884 United States, Washington Fred Hutchinson Cancer Research Center - Vaccine and Infectious Diseases Recruiting Seattle, Washington, United States, 98109-4433 University of Washington - Medicine Recruiting Seattle, Washington, United States, 98195-7110 Collapse << |
| NCT02936362 | Dietary Modification ... more >> Bread Gastrointestinal Microbiome Hematologic Tests Body Weight Changes Collapse << | Not Applicable | Completed | - | Israel ... more >> Weizmann Institute of Science Rehovot, Israel Department of Gastroentherology Tel Aviv, Israel, 64239 Collapse << |
| NCT02769091 | Nonalcoholic Steatohepatitis ... more >> Type 2 Diabetes Mellitus Collapse << | Phase 2 | Withdrawn(Business decision) | February 28, 2018 | - |
| NCT01775813 | Obesity Insul... more >>in Resistance Gonadal Dysfunction Type 2 Diabetes Collapse << | Phase 4 | Completed | - | United States, Colorado ... more >> Children's Hospital Colorado Aurora, Colorado, United States, 80045 Collapse << |
| NCT03527875 | Jaundice, Obstructive ... more >> Klatskin Tumor Collapse << | Not Applicable | Not yet recruiting | July 2022 | - |
| NCT00134212 | Septic Shock | Phase 2 | Completed | - | France ... more >> Rennes University Hospital Rennes, France, 35033 Collapse << |
| NCT03708874 | - | - | Recruiting | March 14, 2019 | Turkey ... more >> Turgut Donmez Recruiting Istanbul, Turkey, 34300 Contact: Turgut Donmez, MD 5347400967 surgeont73@hotmail.com Principal Investigator: Turgut Donmez, MD Collapse << |
| NCT03465306 | Search MeSH | Not Applicable | Active, not recruiting | April 20, 2018 | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 03080 Collapse << |
| NCT02927314 | Non-alcoholic Steatohepatitis ... more >>(NASH) Collapse << | Phase 2 | Recruiting | December 2018 | Israel ... more >> Can-Fite Investigational Site #318 Recruiting Jerusalem, Israel Contact: Study Coordinator Can-Fite Investigational Site #319 Recruiting Nazareth, Israel Contact: Study Coordinator Can-Fite Investigational Site #311 Recruiting Petach Tikva, Israel Contact: Study Coordinator Collapse << |
| NCT03394924 | Primary Biliary Cholangitis | Phase 2 | Recruiting | April 2019 | United States, Arkansas ... more >> Arkansas Diagnostic Center Recruiting Little Rock, Arkansas, United States, 72205 Principal Investigator: Alonzo Williams Texas Clinical Research Institute Recruiting Little Rock, Arkansas, United States, 76012 Principal Investigator: Reem Ghalib United States, California Southern California Research Center Recruiting Coronado, California, United States, 92118 Principal Investigator: Tarek Hassanein Cedars-Sinai Medical Center Recruiting Los Angeles, California, United States, 90048 Principal Investigator: Ayoub Walid California Liver Research Institue Recruiting Pasadena, California, United States, 91105 Principal Investigator: Edward Mena Inland Empire Liver Foundation Recruiting Rialto, California, United States, 92377 Contact: Zeid Kayali, MD Principal Investigator: Zeid Kayali, MD United States, Florida Nature Coast Clinical Research Recruiting Inverness, Florida, United States, 34452 Principal Investigator: Paul Hellstern University of Miami Leonard M. Miller School of Medicine Recruiting Miami, Florida, United States, 33136 Principal Investigator: Cynthia Levy United States, Georgia Consultative Gastroenterology Recruiting Atlanta, Georgia, United States, 30312 Principal Investigator: Gary Richter United States, Maryland Mercy Medical Center-McAuley Plaza Recruiting Baltimore, Maryland, United States, 21202 Contact: Olivia Tang Principal Investigator: Paul Thuluvath Digestive Disease Associates Recruiting Catonsville, Maryland, United States, 21228 Principal Investigator: Natarajan Ravendhran United States, Michigan Henry Ford Hospital Recruiting Detroit, Michigan, United States, 48377 Principal Investigator: Reena Salgia United States, Nebraska CHI Health Recruiting Omaha, Nebraska, United States, 68124 Principal Investigator: Sandeep Mukherjee United States, New York Northwell Health Recruiting Manhasset, New York, United States, 11030 Principal Investigator: David Bernstein Mount Sinai Beth Isreal Recruiting New York, New York, United States, 10016 Principal Investigator: Ira Jacobson Weill Cornell Medical College Recruiting New York, New York, United States, 10024 Principal Investigator: Sonal Kumar United States, Pennsylvania University of Pittsburgh Medical Center - Center for Liver Disease Recruiting Pittsburgh, Pennsylvania, United States, 15213 Principal Investigator: Mordechai Rabinovitz United States, Texas The Liver Institute at Methodist Dallas Medical Center Recruiting Dallas, Texas, United States, 75203 Principal Investigator: Maisha Barnes Liver Consultants of Texas Recruiting Dallas, Texas, United States, 76104 Principal Investigator: Manjushree Gautam Baylor Saint Luke's Medical Center Recruiting Houston, Texas, United States, 77030 Principal Investigator: John Vierling American Research Corporation at the Texas Liver Institute Recruiting San Antonio, Texas, United States, 78215 Contact: Eric J Lawitz, MD 210-253-3426 Principal Investigator: Eric Lawitz, MD United States, Virginia Liver Institute of Virginia-Bremo Recruiting Richmond, Virginia, United States, 23602 Principal Investigator: Mitchell Shiffman United States, Washington Swedish First Hill Campus Recruiting Seattle, Washington, United States, 98104 Principal Investigator: Kris Kowdley Collapse << |
| NCT03048487 | - | - | Completed | - | United States, Pennsylvania ... more >> Geisinger Medical Center Danville, Pennsylvania, United States, 17822 Geisinger Community Medical Center, Scranton, PA Scranton, Pennsylvania, United States, 18510 Geisinger Wyoming Valley Medical Center Wilkes-Barre, Pennsylvania, United States, 18711 Collapse << |
| NCT00876733 | - | - | Completed | - | - |
| NCT00876733 | - | - | Completed | - | - |
| NCT03323359 | Hepatectomy C... more >>ancer, Metastatic Hemostasis Collapse << | Not Applicable | Recruiting | March 17, 2019 | Italy ... more >> Policlinico Universitario Agostino Gemelli Recruiting Rome, Italy, 00168 Contact: Pacelli 00390630155133 ext 4477 fabio.pacelli@policlinicogemelli.it Principal Investigator: Fabio FP Pacelli, MD Collapse << |
| NCT03467217 | NAFLD - Nonalcoholic Fatty Liv... more >>er Disease Collapse << | Phase 2 | Recruiting | August 30, 2019 | United States, California ... more >> University of California, San Diego Recruiting San Diego, California, United States, 92103 Contact: Janis Durelle 619-543-5226 jdurelle@ucsd.edu Principal Investigator: Jeffrey Schwimmer, MD University of California, San Francisco Recruiting San Francisco, California, United States, 94143 Contact: Camille Langlois 415-476-1756 Camille.Langlois@ucsf.edu Principal Investigator: Philip Rosenthal, MD United States, Georgia Emory University Recruiting Atlanta, Georgia, United States, 30322 Contact: Miriam Vos, MD, MSPH 404-727-1463 Mvos@emory.edu Contact: Rebecca Cleeton (404) 727-5383 rcleeto@emory.edu Principal Investigator: Miriam Vos, MD, PhD United States, Illinois Northwestern Univ-Ann & Robert H. Lurie Children's Hospital of Chicago Recruiting Chicago, Illinois, United States, 60611-2605 Contact: Joy Ito 312-227-4559 jito@luriechildrens.org Contact: Mary Riordan (312) 227.4558 mriordan@luriechildrens.org Principal Investigator: Mark Fishbein, MD United States, Indiana Indiana University Recruiting Indianapolis, Indiana, United States, 46202 Contact: Ann Klipsch 317-274-9605 aeye@iupui.edu Contact: Laura Walker (317) 944-4490 walkerlk@iu.edu Principal Investigator: Jean Molleston, MD United States, Missouri St. Louis University Recruiting Saint Louis, Missouri, United States, 63104 Contact: Theresa Cattoor, BSN,MA,CCRC 314-977-9335 theresa.cattoor@health.slu.edu Principal Investigator: Ajay Jain, MD United States, New York Columbia University Recruiting New York, New York, United States, 10032 Contact: Elena Reynoso 212-305-6274 er2564@columbia.edu Principal Investigator: Joel Lavine, MD, PhD United States, Ohio Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229-3039 Contact: April Carr 513-803-7744 april.carr@cchmc.org Contact: Kimberlee Bernstein, BS, CCRP (513)636-4406 Kimberlee.bernstein@cchmc.org Principal Investigator: Stavra Xanthakos, MD United States, Texas Texas Children's Hospital Recruiting Houston, Texas, United States, 77030 Contact: Tamir Miloh, MD 832-822-3648 tamiloh@texaschildrens.org Contact: Alicia Lawson (832) 824-3848 alicia.lawson@bcm.edu Principal Investigator: Tamir Miloh, MD United States, Washington University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Melissa Young 206-987-1037 melissa.young@seattlechildrens.org Sub-Investigator: Niviann Blondet, MD Principal Investigator: Karen Murray, MD Collapse << |
| NCT03419364 | Preeclampsia | Phase 2 | Recruiting | December 31, 2019 | United States, North Carolina ... more >> UNC at Chapel Hill Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Karen Dorman, RN 984-974-9012 kdorman@med.unc.edu Principal Investigator: Kim Boggess, MD Collapse << |
| NCT02819219 | Healthy | Not Applicable | Completed | - | - |
| NCT00048035 | Anemia | Phase 2 | Completed | - | United States, Alabama ... more >> Birmingham, Alabama, United States, 35211 United States, California Los Angeles, California, United States, 90095 United States, Illinois Maywood, Illinois, United States, 60153 United States, Kentucky Louisville, Kentucky, United States, 40202-1718 United States, Michigan Detroit, Michigan, United States, 48202-2689 Detroit, Michigan, United States, 48236 United States, Minnesota Brooklyn Center, Minnesota, United States, 55430 United States, Nevada Las Vegas, Nevada, United States, 89106 United States, New Jersey Paterson, New Jersey, United States, 07503 United States, New York Brooklyn, New York, United States, 11203 Mineola, New York, United States, 11501 New York, New York, United States, 10128 United States, Tennessee Nashville, Tennessee, United States, 37232 United States, West Virginia Morgantown, West Virginia, United States, 26506 Collapse << |
| NCT02952742 | Hot Flashes | Phase 2 | Withdrawn(Lack of funding supp... more >>ort) Collapse << | November 2018 | - |
| NCT00048035 | - | - | Completed | - | - |
| NCT03327857 | Steroid-refractory Acute Graft... more >>-versus-Host Disease Collapse << | Phase 1 | Recruiting | June 2020 | United States, California ... more >> City of Hope Recruiting Duarte, California, United States, 91010 Contact: Roxanne Garcia roxgarcia@coh.org Contact: Golnaz Namdar 626-218-0061 gnamdar@coh.org Principal Investigator: Haris Ali United States, Georgia Emory University Not yet recruiting Atlanta, Georgia, United States, 30322 Contact: Shantoria Mayes 404-778-6547 shantoria.mayes@emory.edu Principal Investigator: Edmund Waller United States, Illinois University of Chicago Not yet recruiting Chicago, Illinois, United States, 60637 Principal Investigator: Michael Bishop United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Lynne Bischoff 734-615-5939 lloisros@med.umich.edu Principal Investigator: Sarah Anand, MD United States, Minnesota University of Minnesota Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Amy Hays 612-626-0461 hays0024@umn.edu Principal Investigator: Shernan Holtan, MD United States, Tennessee Vanderbilt-Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Contact: Delia Darst 615-343-7190 delia.h.darst@vumc.org Principal Investigator: Madan Jagasia, MD United States, Washington Fred Hutchinson Recruiting Seattle, Washington, United States, 98109 Contact: Lisa Grimm 206-667-2353 lgrimm@fredhutch.org Principal Investigator: Paul Martin, MD Sub-Investigator: Marco Mielcarek United States, Wisconsin Medical College of Wisconsin Recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Kaylee Meisinger 414-805-8926 kmeisinger@mcw.edu Principal Investigator: Sameem Abedin, MD Sub-Investigator: Mehdi Hamadani, MD Collapse << |
| NCT00374907 | Type 2 Diabetes | Phase 3 | Completed | - | United States, California ... more >> Va San Diego Healthcare System San Diego, California, United States, 92161 United States, Louisiana Pennington Biomedical Research Center Baton Rouge, Louisiana, United States, 70808 United States, Texas Diabetes & Glandular Disease Research Assoc,, Inc. San Antonio, Texas, United States, 78229 Collapse << |
| NCT02541045 | Non-alcoholic Steatohepatitis | Phase 3 | Suspended(lack of resources to... more >> finance the project) Collapse << | August 2019 | Mexico ... more >> Hospital General de México Mexico City, DF, Mexico, 06726 Collapse << |
| NCT00374907 | - | - | Completed | - | - |
| NCT01992900 | Malaria,Falciparum | Phase 2 | Completed | - | Burkina Faso ... more >> Centre Muraz Bobo Dioulasso, Burkina Faso Centre National de Recherche et de Formation en Paludisme Ouagadougou, Burkina Faso Congo, The Democratic Republic of the Kinshasa School of Public Health, School of Medicine, University of Kinshasa Kinshasa, Congo, The Democratic Republic of the Gambia Medical Research Council Fajara, Gambia Mozambique Manica's Health Research Centre Manica, Mozambique Tanzania Bagamoyo Research center, Ifakara Heath Institute Bagamoyo, Tanzania National Insititute for Medical Research Tanga, Tanzania Collapse << |
| NCT03739242 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> Policlinico S.Orsola - Malpighi Medicina Interna Borghi Bologna, Italy, 40138 Collapse << |
| NCT02625207 | Healthy Subjects | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT03656744 | Fatty Liver, Nonalcoholic ... more >> NAFLD Nonalcoholic Fatty Liver Disease Nonalcoholic Steatohepatitis Digestive System Diseases Collapse << | Phase 2 | Recruiting | October 31, 2019 | United States, Florida ... more >> Florida Research Institute Recruiting Lakewood Ranch, Florida, United States, 34211 Contact: Tracy L. Balentine, RN, MS, CCRC 941-727-7772 tracy.balentine@fdhs.com Contact: Johanna Valentin, MBA 941-727-7772 johanna.valentin@fdhs.com United States, Texas Pinnacle Clinical Research Recruiting Austin, Texas, United States, 78746 Contact: Nadege Gunn, MD 512-384-4560 ngunn@pinnacleresearch.com Pinnacle Clinical Research Recruiting San Antonio, Texas, United States, 78229 Contact: Leslie M. DeHart, MSN, FNP-C 210-982-0320 ext 1451 ldehart@pinnacleresearch.com Collapse << |
| NCT03472157 | Surgery Obesi... more >>ty NASH - Nonalcoholic Steatohepatitis Cirrhosis Collapse << | Not Applicable | Recruiting | March 2023 | France ... more >> Hôpital Claude Huriez, CHRU Recruiting Lille, France Principal Investigator: Guillaume Lassailly, MD Collapse << |
| NCT02625207 | - | - | Completed | - | - |
| NCT02912260 | Non-alcoholic Steatohepatitis | Phase 2 | Active, not recruiting | April 2018 | - |
| NCT01679899 | Type 2 Diabetes Mellitus ... more >> Menopause Osteoporosis Osteopenia Collapse << | Phase 4 | Completed | - | Brazil ... more >> Centro de Diabetes Curitiba Curitiba, Parana, Brazil, 80810040 Collapse << |
| NCT01263691 | Bacillus Anthracis (Anthrax) I... more >>nfection Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Miami Research Associates Miami, Florida, United States, 33143 United States, North Carolina North Carolina Clinical Research Raleigh, North Carolina, United States, 27607 United States, Utah Jean Brown Research Salt Lake City, Utah, United States, 84124 Collapse << |
| NCT01477944 | - | - | Completed | - | - |
| NCT01263691 | - | - | Completed | - | - |
| NCT03508895 | Hypertension | Not Applicable | Recruiting | March 2020 | Canada, Manitoba ... more >> Richarson Centre for Funtional Foods and Nutraceuticals Recruiting Winnipeg, Manitoba, Canada, R3T 6C5 Contact: Peter JH Jones, PhD 204-474-8883 peter.jones@umanitoba.ca Principal Investigator: Peter JH Jones, PhD Collapse << |
| NCT03121807 | Metastatic Gastric Cancer | Not Applicable | Recruiting | December 1, 2019 | Taiwan ... more >> Chung-Ho Memorial Hospital, Kaohsiung Medical University: Recruiting Kaohsiung, Taiwan, 807 Contact: Jaw-Yuan Wang, PhD cy614112@ms14.hinet.net Contact: Cheng-Jen Ma, MD kmu880402@gmail.com Principal Investigator: Jaw-Yuan Wang, Ph.D. Sub-Investigator: Cheng-Jen Ma, MD Collapse << |
| NCT02425020 | Dietary Modification | Not Applicable | Completed | - | - |
| NCT02868489 | Obesity Quali... more >>ty of Life Collapse << | Not Applicable | Recruiting | March 2018 | United States, California ... more >> Faculty of Medical Offices - LLU Recruiting Loma Linda, California, United States, 92354 Contact: Taimmie Wharton 909 558 1000 ext 22126 TWharton@llu.edu Contact: Tammie Wharton, PhD (c), MS, RD 909 677 9593 fkazzi@llu.edu Collapse << |
| NCT03009292 | Solid Tumor | Phase 1 | Recruiting | December 2019 | China, Beijing ... more >> Beijing Cancer Hospital Recruiting Beijing, Beijing, China Contact: Lin Shen 86100881211226561 China, Heilongjiang Harbin Medical University Cancer Hospital Recruiting Harbin, Heilongjiang, China Contact: Yuxian Bai 8645186298694 Collapse << |
| NCT02330341 | Glucose Intolerance | Phase 2 | Completed | - | Mexico ... more >> Manuel González Ortiz Guadalajara, Jalisco, Mexico, 44140 Collapse << |
| NCT02197130 | Huntington's Disease | Phase 2 | Completed | - | - |
| NCT03432260 | Alcoholic Hepatitis | Phase 2 | Recruiting | March 2019 | United States, California ... more >> DURECT Study Site 0001 Recruiting San Diego, California, United States, 92118 United States, Florida DURECT Study Site 007 Recruiting Miami, Florida, United States, 33136 United States, Georgia DURECT Study Site 0004 Recruiting Atlanta, Georgia, United States, 30309 United States, Illinois DURECT Study Site 0002 Recruiting Chicago, Illinois, United States, 60611 United States, Indiana DURECT Study Site 008 Recruiting Indianapolis, Indiana, United States, 46202 United States, Kentucky DURECT Study Site 0005 Recruiting Louisville, Kentucky, United States, 40202 United States, Texas DURECT Study Site 006 Recruiting San Antonio, Texas, United States, 78215 Collapse << |
| NCT02832414 | Sleep Apnea, Obstructive ... more >> Obesity Collapse << | Not Applicable | Recruiting | - | Spain ... more >> Hospital Universitari de Bellvitge Recruiting L'Hospitalet de Llobregat, Barcelona, Spain Contact: Carmen Monasterio Collapse << |
| NCT02954367 | Dietary Modification | Not Applicable | Unknown | - | - |
| NCT02316717 | Non-alcoholic Steatohepatitis ... more >>(NASH) Collapse << | Phase 2 | Completed | - | - |
| NCT03233048 | Obesity | Not Applicable | Recruiting | July 2020 | United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Julia H Byrne 507-538-0893 byrne.julia@mayo.edu Collapse << |
| NCT03404167 | Gonorrhoea | Phase 1 | Completed | - | United States, Kansas ... more >> Quintiles Phase I Services - Overland Park Overland Park, Kansas, United States, 66211-1553 Collapse << |
| NCT03205150 | Obese Patients With Non-alcoho... more >>lic Steatohepatitis (NASH) Collapse << | Phase 2 | Recruiting | October 1, 2019 | United States, Florida ... more >> Novartis Investigative Site Recruiting Miami, Florida, United States, 33126 United States, Louisiana Novartis Investigative Site Recruiting Baton Rouge, Louisiana, United States, 70808 Novartis Investigative Site Recruiting New Orleans, Louisiana, United States, 70121 United States, Missouri Novartis Investigative Site Recruiting Saint Louis, Missouri, United States, 63110 United States, Tennessee Novartis Investigative Site Recruiting Knoxville, Tennessee, United States, 37920 United States, Texas Novartis Investigative Site Recruiting Live Oak, Texas, United States, 78233 Argentina Novartis Investigative Site Recruiting Caba, Buenos Aires, Argentina, C1056ABJ Novartis Investigative Site Recruiting Ciudad Autonoma de Bs As, Buenos Aires, Argentina, C1119ACN Novartis Investigative Site Recruiting Buenos Aires, Argentina, C1120AAC Canada, Quebec Novartis Investigative Site Recruiting Montreal, Quebec, Canada, H3P 3P1 Hong Kong Novartis Investigative Site Recruiting Hong Kong Island, Hong Kong Israel Novartis Investigative Site Recruiting Haifa, Israel, 343621 Novartis Investigative Site Recruiting Ramat Gan, Israel Novartis Investigative Site Recruiting Tel Aviv, Israel, 64239 Netherlands Novartis Investigative Site Recruiting Leiden, Netherlands, 2333 CL Russian Federation Novartis Investigative Site Recruiting Moscow, Russian Federation, 117198 Taiwan Novartis Investigative Site Recruiting Chia-Yi, Taiwan, 60002 Novartis Investigative Site Recruiting Tainan, Taiwan, 70421 Thailand Novartis Investigative Site Recruiting Bangkok, Thailand, 10330 Novartis Investigative Site Recruiting Chiang Mai, Thailand, 50200 Collapse << |
| NCT02197130 | - | - | Completed | - | - |
| NCT02700334 | Prediabetes I... more >>mpaired Fasting Glucose Impaired Glucose Tolerance Collapse << | Phase 4 | Completed | - | Mexico ... more >> Intstituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03436420 | Non-Alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 2 | Active, not recruiting | August 2019 | United States, Georgia ... more >> Children's Healthcare of Atlanta / Emory University Atlanta, Georgia, United States, 30322 Collapse << |
| NCT03231514 | Ketogenic Dieting | Not Applicable | Completed | - | United States, Texas ... more >> Texas Woman's University Denton, Texas, United States, 76204 Collapse << |
| NCT02565615 | - | - | Completed | - | - |
| NCT03674177 | Respiratory Syncytial Virus In... more >>fections Collapse << | Phase 1 | Recruiting | September 19, 2019 | United States, California ... more >> GSK Investigational Site Withdrawn Glendale, California, United States, 91206 GSK Investigational Site Withdrawn San Diego, California, United States, 92108 United States, Illinois GSK Investigational Site Withdrawn Peoria, Illinois, United States, 61602 United States, Kansas GSK Investigational Site Recruiting Lenexa, Kansas, United States, 66219 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Principal Investigator: Casey Johnson United States, New York GSK Investigational Site Recruiting Rochester, New York, United States, 14609 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Principal Investigator: Matthew G Davis United States, Texas GSK Investigational Site Withdrawn Austin, Texas, United States, 78705 Belgium GSK Investigational Site Withdrawn Leuven, Belgium, 3000 Finland GSK Investigational Site Not yet recruiting Helsinki, Finland, 00260 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Tampere, Finland, FI-331 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Turku, Finland, 20100 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Turku, Finland, 20540 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Germany GSK Investigational Site Not yet recruiting Wuerzburg, Bayern, Germany, 97070 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Withdrawn Neu-Isenburg, Hessen, Germany, 63263 GSK Investigational Site Not yet recruiting Hannover, Niedersachsen, Germany, 30159 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Withdrawn Essen, Nordrhein-Westfalen, Germany, 45355 GSK Investigational Site Not yet recruiting Goch, Nordrhein-Westfalen, Germany, 47574 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Mainz, Rheinland-Pfalz, Germany, 55116 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Hamburg, Germany, 22143 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Centre 877-379-3718 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01427140 | Healthy | Not Applicable | Completed | - | Sweden ... more >> Uppsala University Hospital, Uppsala Science Park Uppsala, Sweden Collapse << |
| NCT02918032 | - | - | Recruiting | October 2020 | United States, California ... more >> Stanford University Recruiting Stanford, California, United States, 94305-2004 Contact: Sean M Wu 650-723-2300 United States, New York Columbia University Recruiting New York, New York, United States, 10027 Contact: Salvatore DiMauro 212-854-1754 Austria University of Graz Recruiting Graz, Steiermark, Austria, 8010 Contact: Rudolf Zechner +43-316-380-0 China, Beijing Peking University First Hospital Recruiting Beijing, Beijing, China, 100034 Contact: Yun Yuan +86-66551122 France Pitié-Salpêtrière Hospital Recruiting Paris, France, 75013 Contact: Pascal Laforêt +33-1-42-16-00-00 Contact: Karim Wahbi +33-1-42-16-00-00 Germany Ludwig-Maximilians University Recruiting Munich, Bayern, Germany, 80539 Contact: Peter Reilich +49-89-2180-0 Italy Sapienza University of Rome Recruiting Rome, Lazio, Italy, 00185 Contact: Marcello Arca +39-6-49911 A.C.O. San Filippo Neri Hospital Recruiting Rome, Lazio, Italy, 00193 Contact: Elena M Pennisi, MD, Ph.D. +39-6-33061 Catholic University Recruiting Milano, Lombardia, Italy, 20123 Contact: Daniela Tavian, MD +39-2-72341 University of Padova Recruiting Padova, Veneto, Italy, 35122 Contact: Corrado Angelini +39-49-827-5111 Japan Graduate School of Osaka University Recruiting Suita-city, Osaka-prefecture, Japan, 565-0874 Contact: Kenichi Hirano, MD, Ph.D. +81-6-6872-8215 National Center of Neurology and Psychiatry Recruiting Kodaira-city, Tokyo, Japan, 187-8551 Contact: Ichizo Nishino +81-42-341-2711 Netherlands Maastricht University Recruiting Maastricht, Limburg, Netherlands, 6211 Contact: Matthijs Hesselink +31-43-388-2222 United Kingdom Harefield Hospital Recruiting Harefield, London, United Kingdom, UB9 6JH Contact: Nick Banner +44-1895-823-737 New Castle University Recruiting Newcastle upon Tyne, Tyne And Wear, United Kingdom, NE1 7RU Contact: Rita Horvath +44-191-208-6000 Collapse << |
| NCT03029390 | Prediabetes I... more >>mpaired Fasting Glucose Impaired Glucose Tolerance Collapse << | Phase 4 | Active, not recruiting | May 2018 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT03444142 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 30, 2018 | Mexico ... more >> Instituto de terapeutica Experimental y Clinica. Centro universitario de Ciencias de la Salud. Universidad de Guadalajara Recruiting Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02230930 | Parkinson's Disease ... more >> Apomorphine-induced Skin Reactions Collapse << | Phase 2 | Recruiting | November 2017 | Netherlands ... more >> Department of Neurology Recruiting Groningen, Netherlands, 9713GZ Contact: Robbert Borgemeester, MD +31 (0) 50-3611519 r.w.k.borgemeester@umcg.nl Sub-Investigator: Robbert Borgemeester, MD Principal Investigator: Teus van Laar, MD PhD Collapse << |
| NCT01252355 | - | - | Terminated(Sponsor decision to... more >> prematurely stop the study, not linked to any safety concern.) Collapse << | - | - |
| NCT01925547 | Metabolic Syndrome, Protection... more >> Against Collapse << | Phase 2 | Completed | - | Germany ... more >> Institute of Biological Chemistry and Nutrition, University of Hohenheim Stuttgart, Baden-Württemberg, Germany, 70599 Collapse << |
| NCT02807324 | - | - | Recruiting | June 2022 | Netherlands ... more >> Maastricht University Medical Center (MUMC+) Recruiting Maastricht, Netherlands, 6202 AZ Contact: Jolien Oben, Msc 043 - 38774145 ext 0031 jolien.oben@mumc.nl Contact: Marc Spaanderman, MD, PhD 043 - 38774774 ext 0031 marc.spaanderman@mumc.nl Collapse << |
| NCT03606694 | Type 2 Diabetes Mellitus | Phase 2 | Not yet recruiting | September 30, 2020 | Mexico ... more >> Instituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02983669 | Fatty Liver F... more >>atty Liver, Nonalcoholic Collapse << | Not Applicable | Completed | - | Iran, Islamic Republic of ... more >> Shahid Motahhari Clinic, Shiraz University of Medical Sciences Shiraz, Fars, Iran, Islamic Republic of Collapse << |
| NCT01252355 | Multiple Sclerosis Relapse | Phase 3 | Terminated(Sponsor decision to... more >> prematurely stop the study, not linked to any safety concern.) Collapse << | - | - |
| NCT02279147 | Systemic Inflammatory Response... more >> Syndrome Jaundice, Obstructive Collapse << | Phase 1 Phase 2 | Unknown | September 2017 | China, Beijing ... more >> CHN Chinese PLA General Hospital Recruiting Beijing, Beijing, China, 100853 Principal Investigator: GU wanqing Collapse << |
| NCT03416127 | Diabetes Mellitus, Type 2 | Phase 2 | Not yet recruiting | May 1, 2019 | Mexico ... more >> Intstituto de Terapeútica Experimental y Clínica. Centro Universitario de Ciencias de la Salud. Universidad de Guadalajara Not yet recruiting Guadalajara, Jalisco, Mexico, 44340 Contact: MANUEL GONZALEZ, PhD +523310585200 ext 34212 uiec@prodigy.net.mx Contact: KARINA PEREZ RUBIO, PhD +523310585200 ext 34212 karina2410@hotmail.com Collapse << |
| NCT03052855 | Depression | Not Applicable | Recruiting | January 31, 2018 | France ... more >> Montpellier Hospital University Recruiting Montpellier, France, 34295 Contact: Frédéric COPPOLA, MD Collapse << |
| NCT03123432 | Gastric Cancer | Not Applicable | Completed | - | Taiwan ... more >> Chung-Ho Memorial Hospital, Kaohsiung Medical University: Kaohsiung, Taiwan, 807 Collapse << |
| NCT01415895 | Back Pain Low... more >>er Back Pain Cervical Pain Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Annette C. Toledano MD North Miami, Florida, United States, 33181 Collapse << |
| NCT01160224 | Asthma | Phase 2 | Completed | - | Canada, Alberta ... more >> GSK Investigational Site Calgary, Alberta, Canada, T2N 4Z6 Canada, Ontario GSK Investigational Site Hamilton, Ontario, Canada, L8N 4A6 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H2X 2P4 GSK Investigational Site Montreal, Quebec, Canada, H4J 1C5 Canada GSK Investigational Site Quebec, Canada, G1V 4G5 Collapse << |
| NCT00320359 | Lung Cancer, Small Cell | Phase 3 | Completed | - | - |
| NCT00403611 | Schistosomiasis Mansoni | Phase 4 | Completed | - | - |
| NCT01892644 | Hemochromatosis ... more >> Myelodysplastic Syndromes Collapse << | Phase 2 | Terminated(Failure to recruit ... more >>patients with hemochromatosis to the Deferasirox arm) Collapse << | - | Norway ... more >> Haukeland University Hospital, Clinical Trial Unit Bergen, Norway, 5021 Collapse << |
| NCT00113308 | Arthritis, Rheumatoid | Phase 3 | Completed | - | - |
| NCT02809781 | Spondylitis S... more >>pondylitis, Ankylosing Ankylosis Arthritis Spondylarthritis Spondylarthropathies Spinal Diseases Musculoskeletal Diseases Bone Diseases Collapse << | Phase 2 Phase 3 | Unknown | December 2018 | China, Guangdong ... more >> Sun Yat-sen Memorial Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510120 Contact: Shen Huiyong, Doctor +8602081332612 shenhuiyong@aliyun.com Collapse << |
| NCT01218204 | Dyslipidaemias | Phase 2 | Completed | - | - |
| NCT00319020 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 3 | Completed | - | United States, Colorado ... more >> The Children's Hospital Cardiac Care Center Denver, Colorado, United States, 80218 United States, New York Columbia University Medical Center New York, New York, United States, 10032 France Hopital Antoine Beclere Clamart, France, 92140 Hopital Necker Paris, France, 75743 CHE de Toulouse Hopital d'Enfants Toulouse, France Germany Deutsches Herzzentrum Berlin, Germany Universitats Kinderklinik Giessen, Germany Italy Policlinico S. Orsola-Malpighi Bologna, Italy, 40138 Netherlands Beatrix Children's Hospital Groningen, Netherlands Switzerland Hopital des Enfants Geneva, Switzerland United Kingdom The Institute of Child Health London, United Kingdom Collapse << |
| NCT02834065 | Cognition Disorders | Not Applicable | Recruiting | July 2021 | United States, Georgia ... more >> Emory Saint Joseph's Hospital Recruiting Atlanta, Georgia, United States, 30342 Contact: Michele Fielding, RN, BSN mfield@emory.edu Principal Investigator: Edward P Chen, MD United States, North Carolina Duke Health Recruiting Durham, North Carolina, United States, 27710 United States, Pennsylvania Penn Presbyterian Medical Center Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Marisa Konig, RN, MSN marisa.konig@uphs.upenn.edu Principal Investigator: Wilson Y Szeto, MD Collapse << |
| NCT01218204 | - | - | Completed | - | - |
| NCT01327547 | HIV Coinfection | Phase 4 | Completed | - | - |
| NCT01016912 | Hepatitis C Infection | Phase 2 | Completed | - | Japan ... more >> Local Institution Hiroshima City, Hiroshima, Japan, 734-0037 Local Institution Sapporo-Shi, Hokkaido, Japan, 060-0033 Local Institution Kawasaki-Shi, Kanagawa, Japan, 2138587 Local Institution Suita-Shi, Osaka, Japan, 5650871 Local Institution Iruma-Gun, Saitama, Japan, 3500495 Local Institution Minato-Ku, Tokyo, Japan, 105-0001 Collapse << |
| NCT00319020 | - | - | Completed | - | - |
| NCT00110812 | HIV Infections | Phase 2 | Completed | - | - |
| NCT02496221 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT01327547 | - | - | Completed | - | - |
| NCT01016912 | - | - | Completed | - | - |
| NCT00802854 | - | - | Completed | - | Korea, Republic of ... more >> Chonbuk National University Hospital Deokjin-gu, Jeollabuk-do, Korea, Republic of, 561-712 Dong-A University Hospital Busan, Korea, Republic of, 602-715 Dong-A University Medical Center (Dong-A University Hospital) Busen, Korea, Republic of, 602-715 Keimyung University Dongsan Medical Center (KUDMC) Daegu, Korea, Republic of, 700-712 Daegu fatima hospital Daegu, Korea, Republic of, 701-724 Daegu Catholic University Medical Center (DCUMC) Daegu, Korea, Republic of, 705-718 Ajou University Hospital Gyeonggi-do, Korea, Republic of, 443-721 Severance Hospital, Yonsei University Health System Seoul, Korea, Republic of, 120-752 Seoul Medical Center Seoul, Korea, Republic of, 131-795 Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT03561584 | Primary Sclerosing Cholangitis | Phase 2 | Recruiting | April 1, 2020 | United States, California ... more >> University of California, Davis Recruiting Davis, California, United States, 95616 Contact: Sandeep Dhaliwal Principal Investigator: Chris Bowlus, MD United States, Florida University of Miami Recruiting Miami, Florida, United States, 33136 Contact: Andrea Elibero Principal Investigator: Cynthia Levy, MD Sub-Investigator: Diane Sabogal, MD United States, Massachusetts Brigham and Women's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Fernanda Quevedo 617-732-9676 squevedo@bwh.harvard.edu Principal Investigator: Joshua Korzenik, MD United States, Michigan Henry Ford Health System Recruiting Detroit, Michigan, United States, 48202 Contact: Donna Rush Principal Investigator: Stuart Gordon, MD United States, North Carolina Duke University Recruiting Durham, North Carolina, United States, 27710 Contact: Stacey Smith Principal Investigator: Andrew Muir United States, Pennsylvania University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Maureen Mccauley Principal Investigator: David Goldberg, MD Collapse << |
| NCT02496221 | - | - | Completed | - | - |
| NCT00802854 | - | - | Completed | - | - |
| NCT01172847 | - | - | Completed | - | - |
| NCT00110812 | - | - | Completed | - | - |
| NCT01863043 | Infant, Premature ... more >> Nutrition, Enteral Collapse << | Not Applicable | Active, not recruiting | February 2019 | United States, Florida ... more >> Neonatal intensive care unit at Shands children's hospital at the Univeristy of Florida Gainesville, Florida, United States, 32504 Collapse << |
| NCT02871570 | Moderate Hepatic Impairment ... more >> Normal Hepatic Function Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> Orlando Clincial Research Center Orlando, Florida, United States, 32809 Collapse << |
| NCT01172847 | Healthy Volunteer | Phase 1 | Completed | - | United States, Arkansas ... more >> Little Rock, Arkansas, United States, 72204 Collapse << |
| NCT01718054 | Sickle Cell Disease | Phase 2 Phase 3 | Unknown | September 2017 | Tanzania ... more >> Muhimbili University of Heath and Allied Sciences (MUHAS) Dar es Salaam, Tanzania Collapse << |
| NCT01571284 | Colorectal Cancer Metastatic | Phase 3 | Completed | - | - |
| NCT01028911 | Alzheimer's Disease | Phase 1 | Terminated(This study terminat... more >>ed 27Apr2010 due to slow recruitment and the need to use the existing information to determine dosing for another study.) Collapse << | - | United States, Kansas ... more >> Pfizer Investigational Site Wichita, Kansas, United States, 67207 Pfizer Investigational Site Wichita, Kansas, United States, 67211 Collapse << |
| NCT01571284 | - | - | Completed | - | - |
| NCT01028911 | - | - | Terminated(This study terminat... more >>ed 27Apr2010 due to slow recruitment and the need to use the existing information to determine dosing for another study.) Collapse << | - | - |
| NCT00575159 | Diabetes Mellitus, Type 1 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site San Diego, California, United States, 92161 Collapse << |
| NCT01652885 | Dermatitis, Atopic | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Anacor Investigational Site Fremont, California, United States, 94538 Anacor Investigational Site San Diego, California, United States, 92123 United States, Indiana Anacor Investigational Site Indianapolis, Indiana, United States, 46256 United States, North Carolina Anacor Investigational Site High Point, North Carolina, United States, 27262 Anacor Investigational Site Winston-Salem, North Carolina, United States, 27104 United States, Texas Anacor Investigational Site Houston, Texas, United States, 77030 Collapse << |
| NCT02261961 | Sarcopenia Ag... more >>ing Immune Function Collapse << | Not Applicable | Active, not recruiting | September 30, 2019 | United States, Arkansas ... more >> Central Arkansas Veterans Healthcare System Eugene J. Towbin Healthcare Center, Little Rock, AR North Little Rock, Arkansas, United States, 72114-1706 Collapse << |
| NCT02809833 | - | - | Completed | - | Germany ... more >> Berlin, Germany, 10117 Collapse << |
| NCT01652885 | - | - | Completed | - | - |
| NCT02871570 | - | - | Completed | - | - |
| NCT01285518 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 United States, Florida Comprehensive Phase One (A Division of Comprehensive NeuroScience, Inc.) Ft. Meyers, Florida, United States, 33901 Elite Research Institute Miami, Florida, United States, 33169 Comprehensive Phase One (A Division of Comprehensive NeuroScience, Inc.) Miramar, Florida, United States, 33025 United States, Texas Cetero Research San Antonio, Texas, United States, 78229 Collapse << |
| NCT02809833 | - | - | Completed | - | - |
| NCT01285518 | - | - | Completed | - | - |
| NCT01577745 | Solid Tumors | Phase 1 | Completed | - | United States, California ... more >> Research Site Los Angeles, California, United States, 90404 United States, Connecticut Research Site New Haven, Connecticut, United States, 06513 United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Michigan Research Site Ann Arbor, Michigan, United States, 48103 United States, Minnesota Research Site Minneapolis, Minnesota, United States United States, New York Research Site New York, New York, United States, 10002 United States, Ohio Research Site Cincinnati, Ohio, United States, 45201 Research Site Cleveland, Ohio, United States, 44105 United States, Texas Research Site Houston, Texas, United States, 77030 United States, Washington Research Site Seattle, Washington, United States, 98112 Research Site Tacoma, Washington, United States, 98405 Collapse << |
| NCT01754714 | Non Alcoholic Fatty Liver Dise... more >>ase Collapse << | Phase 3 | Completed | - | France ... more >> Site Reference ID 93914 Amiens, France, 80054 Site Reference ID 93895 Angers, France, 49933 Site Reference ID 93894 Bobigny, France, 93009 Site Reference ID 93913 Montpellier, France, 34295 Site Reference ID 93916 Nice, France, 06202 Site Reference ID 93915 Paris, France, 75012 Site Reference ID 93893 Paris, France, 75651 Site Reference ID 93896 Pessac, France, 33604 Germany Site Reference ID 93953 Bonn, Germany, 53127 Site Reference ID 93954 Frankfurt, Germany, 60594 Site Reference ID 93935 Freiburg, Germany, 79106 Site Reference ID 93955 Halle, Germany, 06120 Site Reference ID 93917 Hannover, Germany, 30625 Site Reference ID 93933 Homburg, Germany, 66424 Site Reference ID 94015 Leipzig, Germany, 04103 Site Reference ID 93918 Mainz, Germany, 55131 Site Reference ID 94014 Ulm, Germany, 89081 Poland Site reference ID/Investigator # 109455 Bydgoszcz, Poland, 85-030 Site Reference ID 93958 Chorzow, Poland, 41-500 Site Reference ID 93973 Krakow, Poland, 31-531 Site Reference ID 93956 Lodz, Poland, 91-347 Site Reference ID 93957 Myslowice, Poland, 41-400 Site Reference ID 93974 Warsaw, Poland, 02-507 Site Reference ID 93975 Wroclaw, Poland, 50-220 Russian Federation Site reference ID ORG-000905 Krasnoyarsk, Russian Federation, 660022 Site reference ID ORG-000906 Moscow, Russian Federation, 119435 Site reference ID ORG-000900 Nizhniy Novgorod, Russian Federation, 603126 Site reference ID ORG-000907 Novosibirsk, Russian Federation, 630084 Site reference ID ORG-000903 Omsk, Russian Federation, 644043 Site reference ID ORG-000920 Rostov-on-Don, Russian Federation, 344022 Site reference ID ORG-000904 Samara, Russian Federation, 443011 Site reference ID ORG-000901 Stavropol, Russian Federation, 355017 Collapse << |
| NCT01577745 | - | - | Completed | - | - |
| NCT00635089 | Eosinophilic Esophagitis | Phase 3 | Completed | - | - |
| NCT01023958 | - | - | Completed | - | - |
| NCT01023958 | Neoplasms | Phase 2 | Completed | - | United States, California ... more >> 1230.2.5 Boehringer Ingelheim Investigational Site Beverly Hills, California, United States 1230.2.10 Boehringer Ingelheim Investigational Site Los Angeles, California, United States United States, Florida 1230.2.34 Boehringer Ingelheim Investigational Site Miami, Florida, United States 1230.2.29 Boehringer Ingelheim Investigational Site Orlando, Florida, United States United States, Illinois 1230.2.6 Boehringer Ingelheim Investigational Site Chicago, Illinois, United States 1230.2.17 Boehringer Ingelheim Investigational Site Joliet, Illinois, United States United States, Louisiana 1230.2.24 Boehringer Ingelheim Investigational Site Metairie, Louisiana, United States United States, Maryland 1230.2.1 Boehringer Ingelheim Investigational Site Baltimore, Maryland, United States United States, Nevada 1230.2.25 Boehringer Ingelheim Investigational Site Las Vegas, Nevada, United States 1230.2.36 Boehringer Ingelheim Investigational Site Las Vegas, Nevada, United States United States, New Hampshire 1230.2.19 Boehringer Ingelheim Investigational Site Lebanon, New Hampshire, United States United States, New York 1230.2.20 Boehringer Ingelheim Investigational Site New York, New York, United States 1230.2.23 Boehringer Ingelheim Investigational Site New York, New York, United States United States, North Carolina 1230.2.12 Boehringer Ingelheim Investigational Site Charlotte, North Carolina, United States United States, Pennsylvania 1230.2.4 Boehringer Ingelheim Investigational Site Philadelphia, Pennsylvania, United States United States, Texas 1230.2.38 Boehringer Ingelheim Investigational Site Beaumont, Texas, United States 1230.2.41 Boehringer Ingelheim Investigational Site Tyler, Texas, United States 1230.2.43 Boehringer Ingelheim Investigational Site Webster, Texas, United States United States, Virginia 1230.2.44 Boehringer Ingelheim Investigational Site Fairfax, Virginia, United States Taiwan 1230.2.51 Boehringer Ingelheim Investigational Site Tainan, Taiwan 1230.2.50 Boehringer Ingelheim Investigational Site Taipei, Taiwan Collapse << |
| NCT00575159 | - | - | Completed | - | - |
| NCT01036022 | Colitis, Ulcerative | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Bonheiden, Belgium, 2820 GSK Investigational Site Brussels, Belgium, 1200 GSK Investigational Site Gent, Belgium, 9000 Canada, Alberta GSK Investigational Site Edmonton, Alberta, Canada, T6G 2C8 Canada, Ontario GSK Investigational Site Kingston, Ontario, Canada, K7L 5G2 GSK Investigational Site London, Ontario, Canada, N6A 4G5 GSK Investigational Site Vaughan, Ontario, Canada, L4L 4Y7 Canada GSK Investigational Site Quebec, Canada, G1S 4L8 Germany GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Muenster, Nordrhein-Westfalen, Germany, 48149 GSK Investigational Site Ludwigshafen, Rheinland-Pfalz, Germany, 67067 GSK Investigational Site Dresden, Sachsen, Germany, 01307 GSK Investigational Site Leipzig, Sachsen, Germany, 04129 GSK Investigational Site Kiel, Schleswig-Holstein, Germany, 24105 GSK Investigational Site Jena, Thueringen, Germany, 07747 GSK Investigational Site Berlin, Germany, 10117 GSK Investigational Site Hamburg, Germany, 20148 GSK Investigational Site Hamburg, Germany, 20246 Norway GSK Investigational Site Lørenskog, Norway, 1478 GSK Investigational Site Oslo, Norway, 0514 GSK Investigational Site Oslo, Norway, N-0456 GSK Investigational Site Tønsberg, Norway, 3116 Sweden GSK Investigational Site Göteborg, Sweden, SE-416 85 GSK Investigational Site Lund, Sweden, SE-221 85 GSK Investigational Site Stockholm, Sweden, SE-111 86 GSK Investigational Site Stockholm, Sweden, SE-171 76 Collapse << |
| NCT00635089 | - | - | Completed | - | - |
| NCT02613403 | Hepatitis Hep... more >>atitis C Digestive System Diseases Flaviviridae Infections Hepatitis, Viral, Human Liver Diseases RNA Virus Infections Virus Diseases Collapse << | Phase 2 | Terminated | - | - |
| NCT00397891 | Alzheimer Disease | Phase 1 | Completed | - | Japan ... more >> Pfizer Investigational Site Chiba, Japan Pfizer Investigational Site Saitama, Japan Pfizer Investigational Site Shizuoka, Japan Pfizer Investigational Site Tokyo, Japan Collapse << |
| NCT01036022 | - | - | Completed | - | - |
| NCT01754714 | - | - | Completed | - | - |
| NCT01664117 | - | - | Completed | - | Spain ... more >> Vitoria, Alava, Spain, 01009 Elche, Alicante, Spain, 03203 Oviedo, Asturias, Spain, 33006 Zafra, Badajoz, Spain, 06300 Sabadell, Barcelona, Spain, 08208 Viladecans, Barcelona, Spain, 08840 Jerez de la Frontera, Cadiz, Spain, 11407 Vinaroz, Castellon, Spain, 12500 Cenes de la vega, Granada, Spain, 18190 Granda, Granada, Spain, 18190 San Sebastian, Guipuzcoa, Spain, 20080 Ibiza, Islas Baleares, Spain, 07800 Inca, Islas Baleares, Spain, 07300 Manacor (Islas Baleares), Islas Baleares, Spain, 07500 Jaén, Jaen, Spain, 23007 La Coruna, La Coruña, Spain, 15006 Alcala de Henares, Madrid, Spain, 28805 Benalmadena, Malaga, Spain, 29630 Vigo, Pontevedra, Spain, 36214 Gandia, Valencia, Spain, 46700 Alicante, Spain, 03010 Burgos, Spain, 06006 Caceres, Spain, 10310 Cordoba, Spain, 14001 Girona, Spain, 17007 Madrid, Spain, 28006 Madrid, Spain, 28007 Madrid, Spain, 28034 Madrid, Spain, 28222 Madrid, Spain, 28905 Malaga, Spain, 29005 Murcia, Spain, 30008 Palencia, Spain, 34005 Sevilla, Spain, 41013 Sevilla, Spain, 41018 Tenerife, Spain, 38010 Toledo, Spain, 45004 Valencia, Spain, 46015 Valladolid, Spain, 47005 Zamora, Spain, 49022 Zaragoza, Spain, 50009 Collapse << |
| NCT02613403 | - | - | Terminated | - | - |
| NCT00077766 | Anemia | Phase 3 | Completed | - | - |
| NCT00397891 | - | - | Completed | - | - |
| NCT03401060 | Osteoporosis ... more >>Systemic Mastocytosis Collapse << | Phase 3 | Recruiting | March 30, 2023 | France ... more >> Jean Minjoz Hospital, Dermatology department Not yet recruiting Besançon, France, 25030 Contact: Fabien Pelletier, MD fabien.pelletier@univ-fcomte.fr Caen Hospital, Clinical haematology department Not yet recruiting Caen, France, 14033 Contact: Gandhi Damaj, MD damaj-gl@chu-caen.fr Estaing Hospital, Cellular therapy and clinical haematology department Not yet recruiting Clermont-Ferrand, France, 63100 Contact: Richard Lemal, MD richard.lemal@hotmail.fr Lille CHRU Hospital, Internal medicine and clinical immunology department Not yet recruiting Lille, France, 59037 Contact: David Launay, MD La Timone Hospital, Dermatology and cutaneous oncology department Recruiting Marseille, France, 13385 Contact: Caroline Gaudy, MD caroline.gaudy@ap-hm.fr Lapeyronie Hospital, Rheumatology and Immunology department Not yet recruiting Montpellier, France, 34295 Contact: Jacques Morel, MD j-morel@chu-montpellier.fr La Pitié-Salpêtrière Hospital, Internal medicine and clinical immunology department Recruiting Paris, France, 75013 Contact: Stéphane Barete, MD stephane.barete@aphp.fr Cochin Hospital, Rheumatology department Not yet recruiting Paris, France, 75014 Contact: Karine Briot, MD karine.briot@aphp.fr Necker Hospital, Adult haematology department Recruiting Paris, France, 75015 Contact: Laurent Frenzel, MD +33 (0)1 44 49 52 90 Laurent.frenzel@aphp.fr Strasbourg Hospital, Rheumatology department Not yet recruiting Strasbourg, France, 67098 Contact: Rose-Marie Javier, MD rose-marie.javier@chru-strasbourg.fr Toulouse Hospital, Dermatology department Recruiting Toulouse, France, 31060 Contact: Cristina Livideanu, MD livideanu.c@chu-toulouse.fr Collapse << |
| NCT00077766 | - | - | Completed | - | - |
| NCT01371825 | Lysosomal Acid Lipase Deficien... more >>cy Wolman Disease Collapse << | Phase 2 Phase 3 | Active, not recruiting | April 2018 | United States, California ... more >> University of California, Irvine Irvine, California, United States, 92697 United States, New York North Shore Hospital Long Island Jewish Health System Manhasset, New York, United States, 11030 Egypt Ain Shams University Pediatrics Hospital Cairo, Egypt France CHU Grenoble Alpes Grenoble, France, 38700 Hopital Necker Enfants Malades Paris, France Ireland Children's University Hospital, Temple Street Dublin, Ireland Saudi Arabia Dept of Medical Genetics, King Faisal Specialist Hospital Riyadh, Saudi Arabia United Kingdom Evalina Children's Hospital London, United Kingdom St. Mary's Hospital, Central Manchester University Hospitals Manchester, United Kingdom Collapse << |
| NCT01371825 | - | - | Active, not recruiting | - | - |
| NCT01664117 | - | - | Completed | - | - |
| NCT02118792 | Dermatitis, Atopic | Phase 3 | Completed | - | United States, Virginia ... more >> Anacor Investigational Site Henrico, Virginia, United States Collapse << |
| NCT00420056 | Lymphoma, Mantle-Cell | Phase 1 | Completed | - | United States, Massachusetts ... more >> Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 New York Presbyterian Hospital New York, New York, United States, 10021 Weill Medical College of Cornell University - New York Presbyterian Hospital New York, New York, United States, 10021 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT02118792 | - | - | Completed | - | - |
| NCT02715193 | Type 1 Diabetes Mellitus ... more >> Diabetes Collapse << | Phase 1 | Completed | - | United States, California ... more >> San Diego, California, United States United States, Missouri St. Louis, Missouri, United States Collapse << |
| NCT00420056 | - | - | Completed | - | - |
| NCT01235741 | - | - | Terminated(Results from previo... more >>us study DFA102 demonstrated neutralizing activity to metreleptin in invitro assay in 2 participants.) Collapse << | - | - |
| NCT01235741 | Obesity | Phase 2 | Terminated(Results from previo... more >>us study DFA102 demonstrated neutralizing activity to metreleptin in invitro assay in 2 participants.) Collapse << | - | United States, California ... more >> Research Site Greenbrae, California, United States Research Site La Jolla, California, United States United States, Colorado Research Site Denver, Colorado, United States United States, Florida Research Site Winter Park, Florida, United States United States, Illinois Research Site Chicago, Illinois, United States United States, Louisiana Research Site Baton Rouge, Louisiana, United States United States, Maryland Research Site Hyattsville, Maryland, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Missouri Research Site St. Louis, Missouri, United States United States, Montana Research Site Butte, Montana, United States United States, New York Research Site New York, New York, United States United States, Oklahoma Research Site Tulsa, Oklahoma, United States United States, Pennsylvania Research Site Philadelphia, Pennsylvania, United States United States, Texas Research Site Austin, Texas, United States United States, Utah Research Site Salt Lake City, Utah, United States United States, Virginia Research Site Arlington, Virginia, United States Research Site Norfolk, Virginia, United States Research Site Richmond, Virginia, United States Collapse << |
| NCT02751424 | Infections, Bacterial | Phase 1 | Terminated(Study data is curre... more >>ntly under review.) Collapse << | - | Australia, South Australia ... more >> GSK Investigational Site Adelaide, South Australia, Australia, 5000 Collapse << |
| NCT02751424 | - | - | Terminated(Study data is curre... more >>ntly under review.) Collapse << | - | - |
| NCT02038764 | Diabetes Mellitus, Type 1 | Phase 1 | Completed | - | United States, California ... more >> VA San Diego Healthcare System (Drug Shipment) San Diego, California, United States, 92161 Veterans Administration San Diego Healthcare System San Diego, California, United States, 92161 University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado Barbara Davis Center Aurora, Colorado, United States, 80045 United States, Connecticut Yale School of Medicine New Haven, Connecticut, United States, 06510 Yale New Haven Hospital - Investigational Drug Services New Haven, Connecticut, United States, 06511 Yale University School of Medicine New Haven, Connecticut, United States, 06519 United States, Georgia Atlanta Diabetes Associates Atlanta, Georgia, United States, 30318 United States, Illinois Duchossois Center for Advanced Medicine Chicago, Illinois, United States, 60637 The University of Chicago Medical Center Chicago, Illinois, United States, 60637 University of Chicago Clinical Resource Center Chicago, Illinois, United States, 60637 University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Massachusetts Umass Memorial Medical Center Worcester, Massachusetts, United States, 01655 University of Massachusetts Medical School Worcester, Massachusetts, United States, 01655 United States, Minnesota University Of Minnesota Fairview Pharmacy Services Minneapolis, Minnesota, United States, 55454 University Of Minnesota Medical School Minneapolis, Minnesota, United States, 55455 United States, Missouri Barnes- Jewish HOSP Att: Kathryn Vehe Saint Louis, Missouri, United States, 63110 Washington University - Center for Advanced Medicine Saint Louis, Missouri, United States, 63110 Washington University Saint Louis, Missouri, United States, 63110 United States, North Carolina Duke Clinical Research Unit Durham, North Carolina, United States, 27710 Duke University Health Systems (DUHS) Investigational Drug Services Durham, North Carolina, United States, 27710 Collapse << |
| NCT02038764 | - | - | Completed | - | - |
| NCT02509117 | Healthy Subjects | Phase 1 | Completed | - | United States, California ... more >> California Clinical Trials Medical Group, Inc Glendale, California, United States, 91206 Glendale Adventist Medical Center Glendale, California, United States, 91206 Collapse << |
| NCT00365859 | - | - | Completed | - | - |
| NCT00122382 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT02332798 | Schizophrenia | Phase 1 | Completed | - | United States, California ... more >> Collaborative Neuroscience Network, LLC. Long Beach, California, United States, 90806 Collapse << |
| NCT00365859 | Autistic Disorder ... more >> Behavioral Symptoms Collapse << | Phase 3 | Completed | - | - |
| NCT02509117 | - | - | Completed | - | - |
| NCT00122382 | - | - | Completed | - | - |
| NCT02691702 | - | - | Completed | - | - |
| NCT02332798 | - | - | Completed | - | - |
| NCT02793232 | - | - | Completed | - | - |
| NCT02951052 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Active, not recruiting | February 8, 2022 | - |
| NCT02190721 | - | - | Completed | - | - |
| NCT02691702 | Healthy Adult Subjects ... more >> Healthy Elderly Subjects Collapse << | Phase 1 | Completed | - | United States, Florida ... more >> QPS-MRA, LLC (Miami Research Associates) South Miami, Florida, United States, 33143 Qps-Mra, Llc South Miami, Florida, United States, 33143 Collapse << |
| NCT02793232 | Healthy Subjects | Phase 1 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT02190721 | Neutropenia | Phase 2 | Completed | - | - |
| NCT02750930 | - | - | Terminated(The termination was... more >> result of GSK business considerations and not due to quality, safety or efficacy concerns with any albiglutide formulations or study conduct) Collapse << | - | - |
| NCT02750930 | Diabetes Mellitus, Type 2 | Phase 4 | Terminated(The termination was... more >> result of GSK business considerations and not due to quality, safety or efficacy concerns with any albiglutide formulations or study conduct) Collapse << | - | - |
| NCT02528305 | Non-alcoholic Steatohepatitis ... more >> Non-alcoholic Fatty Liver Disease Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Abertay University Dundee, United Kingdom, DD1 1HG Ninewells Hospital Dundee, United Kingdom, DD1 9SY Collapse << |
| NCT01012167 | - | - | Completed | - | - |
| NCT02528305 | - | - | Completed | - | - |
| NCT01012167 | Schizophrenia | Phase 2 | Completed | - | United States, Maryland ... more >> Baltimore VA Medical Center Baltimore, Maryland, United States, 21201 Community Mental Health Centers Baltimore, Maryland, United States, 21201 Keypoint Community Mental Health Centers Baltimore, Maryland, United States, 21222 Maryland Psychiatric Research Center Baltimore, Maryland, United States, 21228 Maryland Psychiatric Research Center Catonsville, Maryland, United States, 21228 Keypoint Mental health Center Dundalk, Maryland, United States, 21222 Collapse << |
| NCT02310750 | Plaque Psoriasis | Phase 1 | Completed | - | United States, California ... more >> Anaheim Clinical Trials, LLC Anaheim, California, United States, 92801 Collapse << |
| NCT01439971 | Hemophilia A | Phase 1 | Completed | - | United States, California ... more >> University of California San Diego Medical Center San Diego, California, United States, 92103-8651 United States, Connecticut New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 United States, Illinois Rush University Medical Center Chicago, Illinois, United States, 60612 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Oregon Doernbecher Children's Hospital Portland, Oregon, United States, 97239 OHSU Investigational Pharmacy Portland, Oregon, United States, 97239 Oregon Health & Science University Portland, Oregon, United States, 97239 United States, Pennsylvania CTRC Philadelphia, Pennsylvania, United States, 19104 Penn Comprehensive Hemophilia Program - Center for Blood Disorders Philadelphia, Pennsylvania, United States, 19104 Belgium Pfizer Clinical Research Unit Bruxelles, Belgium, B-1070 Hungary Semmelweis Egyetem Altalanos Orvostudomanyi Kar/ I.sz. Belgyogyaszati Klinika Budapest, Hungary, 1083 Italy Dipartimento di Medicina Clinica 1-Centro Regionale per le Malattie del Sangue Castelfranco Veneto (TV), Italy, 31033 Servizio Farmacia- Ospedale Castelfranco Veneto Castelfranco Veneto - Treviso, Italy, 31033 Centro Emofilia e Trombosi "Angelo Bianchi Bonomi" U.O.S. Dipartimentale per la Diagnosi e la Terapi Milano, Italy, 20122 Servizio Farmacia Fondazione IRCCS Ca' Granda, Ospedale Maggiore Policlinico Milano, Italy, 20122 Centro Malattie Emorragiche e Trombotiche - Ematologia Vicenza, Italy, 36100 Farmacia Ospedaliera Ospedale San Bortolo Vicenza, Italy, 36100 New Zealand Christchurch Clinical Studies Trust Christchurch, New Zealand, 08011 South Africa Phoenix Pharma (Pty) Ltd Port Elizabeth, Eastern Cape, South Africa, 6001 Turkey Ege University Tip Fakultesi Izmir, Turkey, 35100 United Kingdom Royal Free Hampstead NHS Trust, Royal Free Hospital London, United Kingdom, NW3 2QG Haematology Department London, United Kingdom, W12 0HS Central Manchester Universtiy Hospitals NHS Foundation Trust Manchester, United Kingdom, M13 9WL Collapse << |
| NCT03753854 | Sickle Cell Disease | Not Applicable | Recruiting | October 28, 2021 | France ... more >> Hôpital Edouard Herriot Recruiting Lyon, France, 69003 Contact: Giovanna CANNAS 4.72.11.74.12 ext +33 giovanna.cannas@chu-lyon.fr Principal Investigator: Giovanna CANNAS Sub-Investigator: Solène POUTREL Centre Léon Berard Recruiting Lyon, France, 69008 Contact: Alexandra GAUTHIER 4.69.16.65.67 ext +33 alexandra.gauthier@ihope.fr Principal Investigator: Alexandra GAUTHIER Hôpital de la Croix Rousse Recruiting Lyon, France, 69317 Contact: Emeric STAUFFER, MD +33 4 26 10 92 67 emeric.stauffer@chu-lyon.fr Principal Investigator: Emeric Stauffer, MD Sub-Investigator: Thierry PETITJEAN, PH Collapse << |
| NCT02205333 | Advanced Solid Tumors ... more >> Aggressive B-cell Lymphomas Collapse << | Phase 1 Phase 2 | Terminated(The study was termi... more >>nated early at the sponsor's discretion.) Collapse << | - | United States, Arizona ... more >> Research Site Scottsdale, Arizona, United States, 85259 United States, California Research Site Sacramento, California, United States, 95817 Research Site Santa Monica, California, United States, 90404 United States, District of Columbia Research Site Washington, D.C., District of Columbia, United States, 20007 United States, Florida Research Site Tampa, Florida, United States, 33612 United States, Illinois Research Site Chicago, Illinois, United States, 60611 United States, Michigan Research Site Detroit, Michigan, United States, 48202 United States, Nevada Research Site Las Vegas, Nevada, United States, 89169 United States, New Jersey Research Site Hackensack, New Jersey, United States, 7601 United States, North Carolina Research Site Huntersville, North Carolina, United States, 28078 United States, Oregon Research Site Portland, Oregon, United States, 97213 United States, Tennessee Research Site Memphis, Tennessee, United States, 38120 United States, Texas Research Site Houston, Texas, United States, 77030 Collapse << |
| NCT01439971 | - | - | Completed | - | - |
| NCT03508687 | Familial Partial Lipodystrophy... more >> Hypertriglyceridemia Fatty Liver NASH - Nonalcoholic Steatohepatitis Collapse << | Phase 1 Phase 2 | Recruiting | March 1, 2020 | United States, Michigan ... more >> University of Michigan Recruiting Ann Arbor, Michigan, United States, 48105 Contact: Adam H Neidert, MS 734-615-0539 aneidert@med.umich.edu Contact: Elif A Oral, MD 734-615-7271 eliforal@med.umich.edu Principal Investigator: Elif A Oral, MD Sub-Investigator: Nevin N Ajluni, MD Sub-Investigator: Corey Lager, MD Collapse << |
| NCT01114503 | Graves Ophthalmopathy | Phase 2 | Terminated(Clinical study in G... more >>raves' ophthalmopathy terminated until there is a better understanding of an efficacious dose with Otelixizumab from other clinical studies.) Collapse << | - | United Kingdom ... more >> GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 3BZ Collapse << |
| NCT03715205 | Carcinoma, Non-Small-Cell Lung... more >> Melanoma Collapse << | Phase 4 | Not yet recruiting | January 1, 2026 | - |
| NCT01114503 | - | - | Terminated(Clinical study in G... more >>raves' ophthalmopathy terminated until there is a better understanding of an efficacious dose with Otelixizumab from other clinical studies.) Collapse << | - | - |
| NCT02310750 | - | - | Completed | - | - |
| NCT02228395 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02262754 | - | - | Completed | - | - |
| NCT02262754 | Chronic Low Back Pain | Phase 2 | Completed | - | - |
| NCT02525536 | Neoplasms, Advanced Solid | Phase 1 | Completed | - | - |
| NCT02228395 | - | - | Completed | - | - |
| NCT03267030 | Acute Lymphoblastic Leukemia | Phase 2 | Recruiting | August 1, 2020 | Denmark ... more >> Aahus University hospial, hematological department Recruiting Aarhus, Aarhus C, Denmark, 8000 Contact: Mette Skov Holm, MD, phD Mettehlm@rm.dk Contact: Line Stensig Lynggaard, MD +45 25112022 Line.stensig@rm.dk Aarhus University hospital Recruiting Aarhus, Aarhus N, Denmark, 8200 Contact: Line Stensig Lynggaard, MD +25112022 Line.stensig@rm.dk Contact: Birgitte Klug Albertsen, MD, phd +45 20224643 biralber@rm.dk Aalborg University Hospital, pediatric department Recruiting Aalborg, Denmark Contact: Ruta Tuckuviene Rigshospitalet, Hematological department Recruiting Copenhagen, Denmark, 2100 Contact: Ulrik Malthe Overgaard Rigshospitalet, Child and Adolescent Medicine Recruiting Copenhagen, Denmark Contact: Thomas Frandsen Odense University hospital, pediatric department Recruiting Odense, Denmark Contact: Michael Thude Callesen Estonia Tallin Childrens Hospital Recruiting Tallin, Estonia Contact: Kristi Lepik Tartu University Clinics Not yet recruiting Tartu, Estonia Contact: Sirje Mikkel Finland Childrens Hospital, Helsinki. University Central Hospital Not yet recruiting Helsinki, Finland Contact: Samppa Ryhänen Kuopio University Hospital Not yet recruiting Kuopio, Finland Contact: Tuuli Pöyhönen University Hospital of Oulu Not yet recruiting Oulu, Finland Contact: Riita Niinimäki Tampere University Hospital Recruiting Tampere, Finland Contact: Olli Lohi Turku University Hospital Recruiting Turku, Finland Contact: Päivi Lähteenmäki Lithuania Vilnius University Children's Hospital Recruiting Vilnius, Lithuania Contact: Goda Vaitkeviciene Norway Helse Bergen Not yet recruiting Bergen, Norway Contact: Anne Kristine Lehmann Oslo Universitetssykehus, Rikshospitalet Not yet recruiting Oslo, Norway Contact: Sigurd Liestøl St Olavs Hospital Recruiting Trondheim, Norway Contact: Petter Quist Paulsen Sweden Drottning Silvias Barn- och ungdomssjukhus Not yet recruiting Göteborg, Sweden Contact: cecilia Langenskiöld Universitetssjukhuset Linköping Not yet recruiting Linköping, Sweden Contact: Mikael Behrendtz Skånes Universitets sjukhus Not yet recruiting Lund, Sweden Contact: Anders Castor Astrid Lindgrens Barnsjukhus Karolinska Not yet recruiting Stockholm, Sweden Contact: Arja Harila-Sari arn- och Ungdomscentrum Norrlands Universitetssjukhus Not yet recruiting Umeå, Sweden Contact: Ulrika Norén Nyström Akademiska sjukhuset Uppsala Not yet recruiting Uppsala, Sweden Contact: Natalja Jackmann Collapse << |
| NCT02525536 | - | - | Completed | - | - |
| NCT01673178 | Diabetes Melliuts, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Anaheim Clinical Trials, LLC Anaheim, California, United States, 92801 Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 Diablo Clinical Research, Inc. Walnut Creek, California, United States, 94598 United States, Florida Avail Clinical Research, LLC DeLand, Florida, United States, 32720 Elite Research Institute Miami, Florida, United States, 33169 Miami Research Associates, Inc. South Miami, Florida, United States, 33143 MRA Clinical Research, LLC South Miami, Florida, United States, 33143 United States, Kentucky Central Kentucky Research Associates, Inc. Lexington, Kentucky, United States, 40509 L-MARC Research Center Louisville, Kentucky, United States, 40213 United States, Minnesota Prism Research Saint Paul, Minnesota, United States, 55114 United States, North Carolina High Point Clinical Trials Center, LLC High Point, North Carolina, United States, 27265 Carolina Phase 1 Research Raleigh, North Carolina, United States, 27612 Wake Internal Medicine Consultants Raleigh, North Carolina, United States, 27612 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45227 Community Research Cincinnati, Ohio, United States, 45255 Mercy Hospital Pharmacy Cincinnati, Ohio, United States, 45255 United States, Texas Covance Clinical Research Unit Dallas, Texas, United States, 75247 Collapse << |
| NCT02205333 | - | - | Terminated(The study was termi... more >>nated early at the sponsor's discretion.) Collapse << | - | - |
| NCT02032875 | Hepatitis C | Phase 3 | Completed | - | United States, Florida ... more >> University Of Miami Schiff Center For Liver Diseases Miami, Florida, United States, 33136 United States, Michigan University Of Michigan Health System Ann Arbor, Michigan, United States, 48109 United States, Texas Baylor St. Luke'S Medical Center Houston, Texas, United States, 77030 American Research Corporation San Antonio, Texas, United States, 78215 United States, Washington Harborview Medical Center Seattle, Washington, United States, 98104 Collapse << |
| NCT01673178 | - | - | Completed | - | - |
| NCT02032875 | - | - | Completed | - | - |
| NCT02443740 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02443740 | - | - | Completed | - | - |
| NCT01369225 | Alzheimer's Disease | Phase 1 | Completed | - | United States, Florida ... more >> MD Clinical Hallandale Beach, Florida, United States, 33009 Munroe Regional Medical Center Ocala, Florida, United States, 34471 Renstar Medical Research Ocala, Florida, United States, 34471 Trinity Care Solutions Ocala, Florida, United States, 34471 Advanced Imaging of Ocala Ocala, Florida, United States, 34481 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30308 United States, Maryland Foers Medical Arts Pharmacy Bethesda, Maryland, United States, 20814 CBH Health, LLC Rockville, Maryland, United States, 20850 United States, Michigan Borgess Medical Center Kalamazoo, Michigan, United States, 49048 Borgess Research Institute Kalamazoo, Michigan, United States, 49048 KNI Southwest Michigan Imaging Center, LLC Kalamazoo, Michigan, United States, 49048 United States, Missouri Millennium Psychiatric Associates, LLC Creve Coeur, Missouri, United States, 63141 DePaul Health Center-MRI Department St. Louis, Missouri, United States, 63044 United States, New Jersey Memory Enhancement Center of America, Inc. Eatontown, New Jersey, United States, 07724 Central Jersey Radiology Oakhurst, New Jersey, United States, 07755 Korea, Republic of Seoul National University Hospital, Department Neurology Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-707 Inha University Hospital, Department of Neurology Incheon, Korea, Republic of, 400-711 Samsung Medical Center, Department of Neurology Seoul, Korea, Republic of, 1350710 Korea University Anam Hospital IRB Seoul, Korea, Republic of, 136-705 ASAN Medical Center Seoul, Korea, Republic of, 138-736 Konkuk University Medical Center, Department of Neurology Seoul, Korea, Republic of, 143914 Collapse << |
| NCT02014272 | Healthy Volunteers | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT01806896 | Huntington's Disease | Phase 2 | Completed | - | France ... more >> Centre d'Investigation Clinique (CIC)/ Institut du Cerveau et de la Möelle Epinière (ICM) Paris Cedex 13, France, 75651 Collapse << |
| NCT02059759 | Amyotrophic Lateral Sclerosis | Phase 2 | Completed | - | France ... more >> CHRU de Montpellier - Hôpital Gui de Chauliac Montpellier, France, 34295 Collapse << |
| NCT01369225 | - | - | Completed | - | - |
| NCT02014272 | - | - | Completed | - | - |
| NCT02997878 | Cholangitis, Sclerosing ... more >> Hepatitis, Autoimmune Collapse << | Phase 1 Phase 2 | Not yet recruiting | December 2021 | - |
| NCT02224664 | Parkinson`s Disease | Phase 1 | Completed | - | United States, California ... more >> Orange Coast Memorial Medical Center Fountain Valley, California, United States, 92708 The Parkinson's and Movement Disorder Institute Fountain Valley, California, United States, 92708 Collaborative Neuroscience Network, LLC Long Beach, California, United States, 90806 United States, Colorado Rocky Mountain Movement Disorders Center Englewood, Colorado, United States, 80113 Davita Clinical Research Center Lakewood, Colorado, United States, 80228 United States, Florida MD Clinical Hallandale Beach, Florida, United States, 33009 Compass Research, LLC Orlando, Florida, United States, 32806 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30331 United States, Massachusetts Massachusetts General Hospital -- FOR DRUG SHIPMENT ONLY Boston, Massachusetts, United States, 02114 Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Michigan Quest Research Institute Farmington Hills, Michigan, United States, 48334 United States, New Jersey PRA International Marlton, New Jersey, United States, 08053 United States, North Carolina Carolina Phase I Research, LLC Raleigh, North Carolina, United States, 27612 United States, Oklahoma Lynn Health Science Institute Oklahoma City, Oklahoma, United States, 73112 United States, Texas Neurology Consultants of Dallas, PA Dallas, Texas, United States, 75231 Walnut Hill Medical Center Dallas, Texas, United States, 75231 Belgium Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT00335153 | Advanced Parkinson's Disease | Phase 3 | Completed | - | - |
| NCT02224664 | - | - | Completed | - | - |
| NCT01379703 | - | - | Completed | - | - |
| NCT01602341 | Dermatitis, Atopic | Phase 2 | Completed | - | United States, California ... more >> Anacor Investigational Site Fremont, California, United States United States, Florida Anacor Investigational Site Miami, Florida, United States United States, Kentucky Anacor Investigational Site Lousiville, Kentucky, United States United States, Michigan Anacor Investigational Site Detroit, Michigan, United States United States, New Mexico Anacor Investigational Site Albuquerque, New Mexico, United States United States, New York Anacor Investigational Site Stony Brook, New York, United States United States, North Carolina Anacor Investigational Site High Point, North Carolina, United States United States, Oregon Anacor Investigational Site Portland, Oregon, United States United States, Tennessee Anacor Investigational Site Knoxville, Tennessee, United States United States, Utah Anacor Investigational Site Salt Lake City, Utah, United States United States, Virginia Anacor Investigational Site Norfolk, Virginia, United States Australia, Australian Capital Territory Anacor Investigational Site Phillip, Australian Capital Territory, Australia, 2606 Australia, New South Wales Anacor Investigational Site Kogarah, New South Wales, Australia, 2217 Australia, Queensland Anacor Investigational Site Wooloongabba, Queensland, Australia Australia, Victoria Anacor Investigational Site Box Hill, Victoria, Australia Anacor Investigational Site Parkville, Victoria, Australia Australia, Western Australia Anacor Investigational Site Fremantle, Western Australia, Australia, 6160 Collapse << |
| NCT01806896 | - | - | Completed | - | - |
| NCT00983437 | Traumatic Brain Injury | Phase 3 | Terminated(Study has been stop... more >>ped by sponsor decision) Collapse << | - | - |
| NCT00335153 | - | - | Completed | - | - |
| NCT01602341 | - | - | Completed | - | - |
| NCT03400033 | Anaemia | Phase 3 | Recruiting | July 7, 2020 | - |
| NCT01233232 | - | - | Completed | - | - |
| NCT01379703 | - | - | Completed | - | - |
| NCT01478607 | Painful Diabetic Peripheral Ne... more >>uropathy (PDPN) Collapse << | Phase 3 | Completed | - | - |
| NCT03211403 | Gout Hyperuri... more >>cemia Collapse << | Phase 1 | Completed | - | Australia, New South Wales ... more >> Atridia Pty Limited Sydney, New South Wales, Australia, 2000 Collapse << |
| NCT01899755 | Psoriasis | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT03493191 | Acute Pain | Phase 1 | Recruiting | September 2018 | Australia ... more >> Linear Clinical Research Limited Recruiting Nedlands, Australia Contact: Sarah Powell +61 (0) 411 062 165 spowell@linear.org.au Collapse << |
| NCT02656225 | Postpartum Anemia | Phase 1 Phase 2 | Unknown | August 2016 | China, Guangdong ... more >> the First Affiliated Hospital, Guangzhou University of Chinese Medicine Recruiting Guangzhou, Guangdong, China, 510405 Contact: Yanfang Li, PhD +86 13928777126 gzyanfangli@hotmail.com Collapse << |
| NCT01233232 | Scientific Terminology Chronic... more >> Obstructive Pulmonary Disease (COPD) Laymen Terminology Chronic Bronchitis and Emphysema Collapse << | Phase 2 | Completed | - | Bulgaria ... more >> Research Site Sofia, Bulgaria Germany Research Site Berlin, Germany Research Site GROßHANSDORF, Germany Hungary Research Site Debrecen, Hungary Research Site Pécs, Hungary Research Site Szeged, Hungary Research Site Százhalombatta, Hungary Ukraine Research Site Kyiv, Ukraine Collapse << |
| NCT00983437 | - | - | Terminated(Study has been stop... more >>ped by sponsor decision) Collapse << | - | - |
| NCT00478738 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Berlin, Germany, 14050 South Africa GSK Investigational Site Bloemfontein, South Africa, 9301 GSK Investigational Site George, South Africa, 6530 GSK Investigational Site Mowbray, South Africa, 7700 Collapse << |
| NCT02027376 | - | - | Completed | - | - |
| NCT02280096 | Multiple Sclerosis | Phase 2 | Unknown | March 2016 | Mexico ... more >> Hospital de Especialidades, CMN Siglo XXI Mexico City, Distrito Federal, Mexico, 06725 Collapse << |
| NCT02027376 | Advanced Breast Cancer | Phase 1 | Completed | - | Spain ... more >> Complejo Hospitalario Universitario A Coruña A Coruña, Spain, 15006 Hospital General Universitario Gregorio Marañón Madrid, Spain, 28007 Hospital Clínico Universitario San Carlos Madrid, Spain, 28040 Hospital Clínico Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Universitario Virgen del Rocío Sevilla, Spain, 41071 Collapse << |
| NCT01802489 | Optic Neuritis ... more >> Multiple Sclerosis Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> John Radcliffe Hospital Oxford, United Kingdom, OX3 9DU Collapse << |
| NCT01922739 | Low Back Pain | Phase 3 | Completed | - | - |
| NCT02281552 | Rheumatoid Arthritis | Phase 3 | Completed | - | Japan ... more >> Japanese Red Cross Nagoya Daiichi Hospital Nagoya, Aichi, Japan, 453-8511 National Hospital Organization Nagoya Medical Center Nagoya, Aichi, Japan, 460-0001 National Hospital Organization Toyohashi Medical Center Toyohashi, Aichi, Japan, 440-8510 Yamada Rheumatology Clinic Matsuyama, Ehime, Japan, 790-0905 St. Mary's Hospital Kurume, Fukuoka, Japan, 830-8543 Gunma Rheumatism Clinic Takasaki-shi, Gunma, Japan, 370-0004 Inoue Hospital Tohrimachi, Takasaki, Gunma, Japan, 370-0053 Hiroshima Red Cross Hospital & Atomic-bomb Survivors Hospital Hiroshima-city, Hiroshima, Japan, 730-8619 National Hospital Organization Asahikawa Medical Center Asahikawa, Hokkaido, Japan, 070-8644 Katayama Orthopaedic Rheumatology Clinic Asahikawa, Hokkaido, Japan, 078-8243 Yoshida orthopaedics and rheumatologyclinic Morioka, Iwate, Japan, 020-0015 Komagamine Rheumatoid Orthopaedic Clinic Morioka, Iwate, Japan, 020-0034 National Hospital Organization Sagamihara National Hospital Sagamihara, Kanagawa, Japan, 252-0392 National Hospital Organization Yokohama Medical Center Yokohama, Kanagawa, Japan, 245-8575 Osaki Citizen Hospital Oosaki, Miyagi, Japan, 989-6183 Tohoku University Hospital/ Department of Hematology and Rheumatology Sendai, Miyagi, Japan, 9808574 Nagano Red Cross Hospital Nagano-shi, Nagano, Japan, 380-8582 Sasebo Chuo Hospital Sasebo, Nagasaki, Japan, 857-1195 National Hospital Organization Ureshino Medical Center Ureshino-shi, Saga, Japan, 843-0393 Soshigayaokura clinic Setagaya-ku, Tokyo, Japan, 157-0073 Showa University Hospital Shinagawa-ku, Tokyo, Japan, 142-8666 Honjo Rheumatism Clinic Takaoka-shi, Toyama, Japan, 933-0874 Miyasato Clinic Shunan, Yamaguchi, Japan, 745-0824 National Hospital Organization Chiba-East Hospital Chiba, Japan, 260-8712 Sugimoto rheumatology and internal medicine clinic Fukui, Japan, 910-0068 National Hospital Organization Kyushu Medical Center Fukuoka, Japan, 810-8563 SHONO Rheumatism Clinic Fukuoka, Japan, 814-0002 Sagawa Akira Rheumatology Clinic Hokkaido, Japan, 060-0001 Matsubara Mayflower Hospital Hyogo, Japan, 673-1462 Yokohama Minami Kyosai Hospital Kanagawa, Japan, 236-0037 Kumamoto Orthopaedic Hospital Kumamoto, Japan, 862-0976 Japanese Red Cross Kyoto Daiichi Hospital Kyoto, Japan, 605-0981 Oribe Clinic of Rheumatology and Internal Medicine Oita, Japan, 870-0823 Otsuka Clinic of Rheumatism and Medicine Oita, Japan, 870-1155 Rabbit Clinic Saitama, Japan, 336-0911 Hirose Clinic Saitama, Japan, 359-1111 Collapse << |
| NCT03680105 | Critical Limb Ischemia | Phase 1 | Recruiting | March 2019 | United States, Texas ... more >> ICON Early Phase Services Recruiting San Antonio, Texas, United States, 78209 Contact: Dennis A Ruff, MD Collapse << |
| NCT03764345 | Hepatitis C, Chronic ... more >> Children, Only Collapse << | Not Applicable | Completed | - | Egypt ... more >> Pediatric Hepatology, Gastroenterology and Nutrition Department, National Liver Institute, Menoufia University Shebin El-Koom, Menofiya, Egypt, 32511 Collapse << |
| NCT00903201 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT02288078 | Rectal Cancer | Phase 2 | Unknown | September 2016 | Japan ... more >> Saiseikai Fukuoka General Hospital Recruiting Fukuoka, Japan, 810-0001 Contact: Yasunori Emi, MD, PhD +81-92-771-8151 emi-y@saiseikai-hp.chuo.fukuoka.jp Collapse << |
| NCT02281552 | - | - | Completed | - | - |
| NCT01317797 | Rheumatoid Arthritis | Phase 1 | Completed | - | Bulgaria ... more >> Nycomed Investigational Site Sofia, Bulgaria Netherlands Nycomed Investigational Site Leids, Netherlands Collapse << |
| NCT00903201 | - | - | Completed | - | - |
| NCT02966665 | Chronic Obstructive Pulmonary ... more >>Disease Pulmonary Artery Hypertension Heart Failure Hypertension Collapse << | Phase 1 | Recruiting | August 2020 | United States, Utah ... more >> George E Wahlen VA Medical Center Recruiting Salt Lake City, Utah, United States, 84132 Contact: Russell Richardson, Ph.D. 801-582-1565 r.richardson@hsc.utah.edu Contact: Ashley Nelson, MD 801-582-1565 ext 4127 ash.nelson@hsc.utah.edu Collapse << |
| NCT01317797 | - | - | Completed | - | - |
| NCT03045887 | Asthma | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 2GG Collapse << |
| NCT02016365 | Transthyretin Amyloidosis ... more >> Cardiomyopathy Collapse << | Phase 2 | Completed | - | Sweden ... more >> Dept of Clinical Medicin, Ptieå Hospital Piteå, Sweden, SE-941 28 Dept of clinical medicin, Skellefteå Hospital Skellefteå, Sweden, SE-931 86 Dept of Clinical Medicine, Umeå University Hospital Umeå, Sweden, SE-90185 Collapse << |
| NCT00656253 | Parkinson's Disease | Phase 2 Phase 3 | Completed | - | China, Beijing ... more >> Department of Neurology, Xuanwu hospital, Capital University of Medical Sciences Beijing, Beijing, China, 100053 China, Shanghai Department of Neurology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine Shanghai, Shanghai, China, 200025 Department of Integrative Medicine, Zhongshan hospital, Fudan University Shanghai, Shanghai, China, 200032 Department of Nuerology, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Shanghai, Shanghai, China, 200032 Tongji Hospital of Tongji University Shanghai, Shanghai, China, 200065 Shanghai Chinese Medical Hospital Shanghai, Shanghai, China, 200071 Department of Nuerology, Xinhua Hospital, Affiliated to Shanghai Jiaotong University School of Medicine Shanghai, Shanghai, China, 200127 The Sixth People's Hospital, Shanghai Jiaotong University Shanghai, Shanghai, China, 200233 Department of Neurology, Yueyang Hospital of Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine Shanghai, Shanghai, China, 200437 China, Zhejiang Department of Neurology, The second people's hospital of Wenzhou Wenzhou, Zhejiang, China, 325000 Collapse << |
| NCT00629161 | Parkinson Disease | Phase 2 Phase 3 | Unknown | - | China, Beijing ... more >> Department of Neurology, Xuanwu hospital, Capital University of Medical Sciences Recruiting Beijing, Beijing, China, 100053 Contact: Li Gao, MD China, Shanghai Department of Neurology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine Recruiting Shanghai, Shanghai, China, 200025 Contact: Qin Xiao, MD Department of Nuerology, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Recruiting Shanghai, Shanghai, China, 200032 Contact: Canxing Yuan, MD Zhongshan hospital, Fudan University Recruiting Shanghai, Shanghai, China, 200032 Contact: Dingfang Cai, MD doctorcn@hotmail.com Tongji Hospital of Tongji University Recruiting Shanghai, Shanghai, China, 200065 Contact: Qing Zhan, MD Shanghai Chinese Medical Hospital Recruiting Shanghai, Shanghai, China, 200071 Contact: Yi Liu, MD Department of Nuerology, Xinhua Hospital, Affiliated to Shanghai Jiaotong University School of Medicine Recruiting Shanghai, Shanghai, China, 200127 Contact: Zhenguo Liu The Sixth People's Hospital, Shanghai Jiaotong University Recruiting Shanghai, Shanghai, China, 200233 Contact: Xiaojiang Sun, MD Department of Neurology, Yueyang Hospital of Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine Recruiting Shanghai, Shanghai, China, 200437 Contact: Hong Zhao, MD China, Zhejiang Department of Neurology, The second people's hospital of Wenzhou Recruiting Wenzhou, Zhejiang, China, 325000 Contact: Liya Han Collapse << |
| NCT01909284 | Low Back Pain | Not Applicable | Unknown | March 2014 | Korea, Republic of ... more >> Daegu Catholic University Medical Center Recruiting Daegu, Kyungsangbukdo, Korea, Republic of Collapse << |
| NCT01615185 | Schizophrenia | Phase 4 | Terminated | - | Taiwan ... more >> Kai-Suan Psychiatric Hospital Kaohsiung, Taiwan, 802 Collapse << |
| NCT01054586 | - | - | Completed | - | - |
| NCT01038609 | Pain | Phase 2 | Completed | - | United States, Arizona ... more >> Site Reference ID/Investigator# 26223 Peoria, Arizona, United States, 85381 United States, Texas Site Reference ID/Investigator# 26302 Austin, Texas, United States, 78705 Site Reference ID/Investigator# 26303 San Marcos, Texas, United States, 78666 United States, Utah Site Reference ID/Investigator# 26304 West Jordan, Utah, United States, 84088 Collapse << |
| NCT01054586 | - | - | Completed | - | - |
| NCT01038609 | - | - | Completed | - | - |
| NCT01922739 | - | - | Completed | - | - |
| NCT03272737 | Frail Elderly Syndrome | Not Applicable | Recruiting | March 4, 2019 | Brazil ... more >> Instituto Israelita de Ensino e Pesquisa Albert Einstein 's (IIEP) Recruiting São Paulo, SP, Brazil, 05652-000 Contact: André L Gonçalves 00551135898237 goncalvesnp@yahoo.com.br Sub-Investigator: Samuel A Souza, Specialist Collapse << |
| NCT01438424 | Hepatitis B Virus ... more >> HBV Collapse << | Phase 2 | Completed | - | - |
| NCT00742885 | - | - | Completed | - | - |
| NCT00920140 | Cancer | Phase 2 | Completed | - | - |
| NCT00920140 | - | - | Completed | - | - |
| NCT00742885 | Influenza | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-8588 GSK Investigational Site Tokyo, Japan, 204-8585 Collapse << |
| NCT02378506 | - | - | Completed | - | - |
| NCT02378506 | Arthritis, Rheumatoid | Phase 3 | Completed | - | Bulgaria ... more >> MHAT Plovdiv Plovdiv, Bulgaria, 4000 DCC "Aleksandrovska" EOOD Sofia, Bulgaria, 1431 UMHAT "Sv. Ivan Rilski" EAD, Sofia Sofia, Bulgaria, 1612 Medical Center - "New rehabilitation center" EOOD Stara Zagora, Bulgaria, 6000 Croatia Chc Rijeka Rijeka, Croatia, 51000 Medicinski Centar Kuna Peric Zagreb, Croatia, 10000 Germany Centrum für innovative Diagnostik und Therapie Frankfurt/Main, Hessen, Germany, 60528 Rheumaforschung - Studienambulanz Dr. Wassenberg Ratingen, Nordrhein-westfalen, Germany, 40882 Schlosspark-Klinik, Innere Medizin II, Rheumatologie Berlin, Germany, 14059 Medizinische Hochschule Hannover Hannover, Germany, 30625 Immunologisches Zentrum Vogelsang-Gommern GmbH Vogelsang-Gommern, Germany, 39245 Greece Rheumatology Department Rion Patras, Achaia, Greece, 26500 University Hospital of Heraklion Heraklion, Crete, Greece, 71110 Rheumatology Unit Thessaloniki, Greece, 56429 Hungary Qualiclinic Kft. Budapest, Hungary, 1036 Pest Megyei Flor Ferenc Korhaz Kistarcsa, Hungary, 2143 Szabolcs-Szatmar-Bereg megyei Nyiregyhaza, Hungary, 4400 Poland Krakowskie Centrum Medyczne sp. Z.O.O Krakow, Poland, 31-501 NZOZ Lecznica MAK-MED s.c. Nadarzyn, Poland, 05-830 Prywatna Praktyka Lekarska Prof. UM dr hab.med. Pawel Hrycaj Poznan, Poland, 61-397 Reumatika-Centrum Reumatologii Warszawa, Poland, 02-691 Serbia Institute of Rheumatology Belgrade, Beograd, Serbia, 11000 Slovakia Reumatologicka ambulancia Bratislava, Slovakia, 841 04 AAGS, s.r.o. Dunajska Streda, Slovakia, 929 01 Reumatologicka ambulancia, MUDr. Zuzana Cizmarikova, s.r.o. Poprad, Slovakia, 058 01 REUMEX s.r.o. Rimavska Sobota, Slovakia, 979 01 REUMA-GLOBAL s.r.o. Trnava, Slovakia, 917 01 Reumatologicka ambulancia Zilina, Slovakia, 010 01 South Africa Panorama Medical Centre Panorama, Cape Town, South Africa, 7500 Wits Clinical Research CMJAH Clinical Trial Site Parktown, Johannesburg, South Africa, 2193 St. Augustines Hospital Durban/Berea, Kwazulu-natal, South Africa, 4001 Collapse << |
| NCT01411215 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01438424 | - | - | Completed | - | - |
| NCT00989235 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT00471237 | Osteoporosis | Phase 2 | Terminated(Terminated for futi... more >>lity by sponsor after a pre-planned interim review of data) Collapse << | - | - |
| NCT01928940 | Solid Tumours | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Shizuoka, Japan, 411-8777 GSK Investigational Site Tokyo, Japan, 104-0045 Collapse << |
| NCT01411215 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | China, Chongqing ... more >> Daping Hospital Chongqing, Chongqing, China, 400038 China, Fujian Fujian Provincial Hospital Fuzhou, Fujian, China, 350001 China, Gansu Lanzhou University Second Hospital Lanzhou, Gansu, China, 730030 China, Guangdong The First Affiliated Hospital of Guangzhou University of Chinese Medicine Guangzhou, Guangdong, China, 510405 China, Heilongjiang No. 199 Haerbin, Heilongjiang, China, 150001 China, Hunan The Second Xiangya Hospital of Central South University Changsha, Hunan, China, 410011 China, Inner Mongolia The First Affiliated Hospital of Baotou Medical College Baotou, Inner Mongolia, China, 014010 China, Jiangsu Jiangsu Province Hospital/Department of Rheumatology Nanjing, Jiangsu, China, 210029 Affiliated Hospital of Nantong University Nantong, Jiangsu, China, 226001 China, Shanxi The Second Hospital of Shanxi Medical University Taiyuan, Shanxi, China, 030001 China, Sichuan Si Chuan Huaxi Hospital/Rheumatology Department Chengdu, Sichuan, China, 610041 China Baotou Central Hospital Baotou city, China Shanghai Changning Guanghua Integrative Medicine Hospital Beijing, China, 200052 Shanghai Jiaotong University Affiliated Third People's Hospital Shanghai, China, 201900 Xinjiang Uygur Autonomous Region People's Hospital Urumqi, China, 830001 Collapse << |
| NCT00471237 | - | - | Terminated(Terminated for futi... more >>lity by sponsor after a pre-planned interim review of data) Collapse << | - | - |
| NCT00989235 | - | - | Completed | - | - |
| NCT02193815 | Psoriasis Vulgaris | Phase 1 | Completed | - | Germany ... more >> Bioskin GmbH Hamburg, Germany, 20095 Collapse << |
| NCT02193815 | - | - | Completed | - | - |
| NCT00387764 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT01928940 | - | - | Completed | - | - |
| NCT00387764 | - | - | Completed | - | - |
| NCT01336621 | Epilepsy | Phase 3 | Completed | - | - |
| NCT02938520 | HIV Infections | Phase 2 Phase 3 | Active, not recruiting | July 7, 2022 | - |
| NCT01396148 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | United States, California ... more >> Children's Center for Cancer and Blood Diseases, Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Massachusetts Children's Hospital Boston Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Czechia Fakultni nemocnice Brno Brno, Czechia, 613 00 FN Brno Brno, Czechia, 625 00 Masarykuv onkologicky ustav Brno, Czechia, 656 33 France CHU de La Timone, Hopital enfants Marseille cedex 5, France, 13385 Hopital d'Enfants de la Timone Marseille cedex 5, France, 13385 Hopital de la Timone Marseille cedex 5, France, 13385 Collapse << |
| NCT01336621 | - | - | Completed | - | - |
| NCT01714817 | Lupus Nephritis | Phase 3 | Completed | - | - |
| NCT01714817 | - | - | Completed | - | - |
| NCT00053495 | - | - | Completed | - | - |
| NCT01396148 | - | - | Completed | - | - |
| NCT01684215 | Neoplasms Bre... more >>ast Neoplasms Collapse << | Phase 2 | Completed | - | Japan ... more >> Aichi Cancer Center Hospital Nagoya, Aichi, Japan, 464-8681 National Cancer Center Hospital East Kashiwa, Chiba, Japan, 277-8577 National Hospital Organization Hokkaido Cancer Center Sapporo, Hokkaido, Japan, 003-0804 Kumamoto Shinto General Hospital Kumamoto-city, Kumamoto, Japan, 862-8655 Saitama Cancer Center Kita-adachi-gun, Saitama, Japan, 362-0806 National Cancer Center Hospital Chuo-Ku, Tokyo, Japan, 104-0045 Chiba Cancer Center Chiba, Japan, 260-8717 National Hospital Organization Shikoku Cancer Center Ehime, Japan, 791-0280 National Hospital Organization Kyushu Cancer Center Fukuoka, Japan, 811-1395 Hiroshima City Hiroshima Citizens Hospital Hiroshima, Japan, 730-8518 Iwate Medical University Hospital Iwate, Japan, 020-8505 Hakuaikai Medical Corporation Sagara Hospital Kagoshima, Japan, 892-0833 Kumamoto University Hospital Kumamoto, Japan, 860-8556 Kumamoto City Hospital Kumamoto, Japan, 862-8505 Kyoto University Hospital Kyoto, Japan, 606-8507 National Hospital Organization Osaka National Hospital Osaka, Japan, 540-0006 Collapse << |
| NCT01001832 | Rheumatoid Arthritis | Phase 2 Phase 3 | Completed | - | Japan ... more >> Local Institution Narita-Shi, Chiba, Japan, 2868523 Local Institution Fukuoka-Shi, Fukuoka, Japan, 8108563 Local Institution Kitakyushu-Shi, Fukuoka, Japan, 8078555 Local Institution Kurume-Shi, Fukuoka, Japan, 8308543 Local Institution Maebashi-Shi, Gunma, Japan, 3718511 Local Institution Takasaki-Shi, Gunma, Japan, 3700053 Local Institution Higashi-Hiroshima-Shi, Hiroshima, Japan, 7390002 Local Institution Sapporo-Shi, Hokkaido, Japan, 0608604 Local Institution Sapporo-Shi, Hokkaido, Japan, 0608648 Local Institution Sapporo-Shi, Hokkaido, Japan, 0630811 Local Institution Sapporo, Hokkaido, Japan, 0630005 Local Institution Kanzaki-Gun, Hyogo, Japan, 6792414 Local Institution Kato-Shi, Hyogo, Japan, 6731462 Local Institution Kobe-Shi, Hyogo, Japan, 6500001 Local Institution Hitachi-Shi, Ibaraki, Japan, 3160035 Local Institution Kagoshima-Shi, Kagoshima, Japan, 8900067 Local Institution Sagamihara-Shi, Kanagawa, Japan, 2520392 Local Institution Yokohama-Shi, Kanagawa, Japan, 2220036 Local Institution Yokohama-Shi, Kanagawa, Japan, 2360037 Local Institution Nagano-Shi, Nagano, Japan, 3808582 Local Institution Kurashiki-Shi, Okayama, Japan, 7128044 Local Institution Hannan-Shi, Osaka, Japan, 5990212 Local Institution Iruma-Gun, Saitama, Japan, 3500495 Local Institution Kawagoe-Shi, Saitama, Japan, 3508550 Local Institution Kitamoto-Shi, Saitama, Japan, 3640026 Local Institution Tokorozawa-Shi, Saitama, Japan, 3591111 Local Institution Hamamatsu-Shi, Shizuoka, Japan, 4308558 Local Institution Shizuoka-Shi, Shizuoka, Japan, 4208623 Local Institution Shimotsuke-Shi, Tochigi, Japan, 3290498 Local Institution Utsunomiya-Shi, Tochigi, Japan, 3291193 Local Institution Bunkyo-Ku, Tokyo, Japan, 1138519 Local Institution Nakano-Ku, Tokyo, Japan, 1648541 Local Institution Shinjuku-Ku, Tokyo, Japan, 1608582 Collapse << |
| NCT01684215 | - | - | Completed | - | - |
| NCT00053495 | Smallpox | Phase 2 | Completed | - | United States, Florida ... more >> Orlando Clinical Research Center Orlando, Florida, United States, 32806 United States, Kansas PRA International Lenexa, Kansas, United States, 66219 United States, Missouri Bio-Kinetic Clinical Applications Springfield, Missouri, United States, 65802 United States, Rhode Island Memorial Hospital of Rhode IslandDivision of Infectious Diseases Pawtucket, Rhode Island, United States, 02860 Collapse << |
| NCT01668784 | Advanced or Metastatic (Medica... more >>lly or Surgically Unresectable) Clear-cell Renal Cell Carcinoma Collapse << | Phase 3 | Active, not recruiting | September 28, 2018 | - |
| NCT01671059 | - | - | Completed | - | - |
| NCT01001832 | - | - | Completed | - | - |
| NCT01512160 | Post-surgical Dental Pain | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | United States, Texas ... more >> Pfizer Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT01671059 | - | - | Completed | - | Serbia ... more >> Institut Za Reumatologiju; Reumatologiju Belgrade, Serbia, 11000 Military Medical Academy; Clinic of Rheumatology Belgrade, Serbia, 11000 Belgrade, Serbia, 11000 Institute of Rheumatology and Cardiovascular Diseases; Rheumatology Niska Banja, Serbia, 18205 Niska Banja, Serbia, 18205 Clinical Center Voivodina; Clinic for Immunology and Nephrology Novi Sad, Serbia, 21000 Special hospital for rheumatic diseases Novi Sad Novi Sad, Serbia, 21000 Novi Sad, Serbia, 21000 Collapse << |
| NCT02373202 | Rheumatoid Arthritis | Phase 3 | Completed | - | Japan ... more >> Investigational Site Number 392010 Asahi-Shi, Japan Investigational Site Number 392001 Asahikawa-Shi, Japan Investigational Site Number 392070 Beppu-Shi, Japan Investigational Site Number 392036 Chiba-Shi, Japan Investigational Site Number 392083 Chuo-Ku, Japan Investigational Site Number 392004 Fukui-Shi, Japan Investigational Site Number 392039 Fukuoka-Shi, Japan Investigational Site Number 392030 Ichinomiya-Shi, Japan Investigational Site Number 392002 Iizuka-Shi, Japan Investigational Site Number 392019 Kagoshima-Shi, Japan Investigational Site Number 392066 Kamakura-Shi, Japan Investigational Site Number 392050 Kato-Shi, Japan Investigational Site Number 392037 Kawachi-Nagano-Shi, Japan Investigational Site Number 392099 Kawasaki-Shi, Japan Investigational Site Number 392013 Kitakyushu-Shi, Japan Investigational Site Number 392097 Kochi-Shi, Japan Investigational Site Number 392065 Kushiro-Shi, Japan Investigational Site Number 392026 Matsuyama-Shi, Japan Investigational Site Number 392034 Miyagi-Gun, Japan Investigational Site Number 392076 Nagoya-Shi, Japan Investigational Site Number 392080 Nagoya-Shi, Japan Investigational Site Number 392046 Narashino-Shi, Japan Investigational Site Number 392059 Oita-Shi, Japan Investigational Site Number 392062 Okayama-Shi, Japan Investigational Site Number 392027 Osaki-Shi, Japan Investigational Site Number 392049 Sagamihara-Shi, Japan Investigational Site Number 392014 Sapporo-Shi, Japan Investigational Site Number 392041 Sapporo-Shi, Japan Investigational Site Number 392073 Sapporo-Shi, Japan Investigational Site Number 392006 Sasebo-Shi, Japan Investigational Site Number 392021 Sendai-Shi, Japan Investigational Site Number 392022 Sendai-Shi, Japan Investigational Site Number 392033 Sendai-Shi, Japan Investigational Site Number 392071 Sendai-Shi, Japan Investigational Site Number 392029 Shizuoka-Shi, Japan Investigational Site Number 392023 Takaoka-Shi, Japan Investigational Site Number 392003 Tomakomai-Shi, Japan Investigational Site Number 392074 Urasoe-Shi, Japan Investigational Site Number 392079 Urayasu-Shi, Japan Investigational Site Number 392048 Yokohama-Shi, Japan Collapse << |
| NCT02373202 | - | - | Completed | - | - |
| NCT03083184 | Tunnelled Hemodialysis Cathete... more >>r Infection Collapse << | Phase 3 | Not yet recruiting | March 2021 | France ... more >> CHU Clermont-Ferrand Not yet recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 0473751081 placarin@chu-clermontferrand.fr Principal Investigator: Julien ANIORT Collapse << |
| NCT00985673 | Influenza | Phase 2 | Completed | - | United States, Georgia ... more >> GSK Investigational Site Stockbridge, Georgia, United States, 30281 United States, North Carolina GSK Investigational Site Raleigh, North Carolina, United States, 27612 United States, Texas GSK Investigational Site Austin, Texas, United States, 78705 GSK Investigational Site Fort Worth, Texas, United States, 76135 Canada, Nova Scotia GSK Investigational Site Halifax, Nova Scotia, Canada, B3K 6R8 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H2K 4L5 GSK Investigational Site Sherbrooke, Quebec, Canada, J1H 4J6 Collapse << |
| NCT01512160 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00903617 | - | - | Completed | - | - |
| NCT02319148 | Healthy Volunteers | Phase 1 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT01366521 | Asthma | Phase 2 | Completed | - | United States, North Carolina ... more >> GSK Investigational Site Winston-Salem, North Carolina, United States, 27103 United States, Pennsylvania GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 Estonia GSK Investigational Site Tallinn, Estonia, 13619 GSK Investigational Site Tartu, Estonia, 51014 France GSK Investigational Site Marseille cedex 20, France, 13915 GSK Investigational Site Montpellier, France, 34295 GSK Investigational Site Pessac Cedex, France, 33604 Germany GSK Investigational Site Gauting, Bayern, Germany, 82131 GSK Investigational Site Frankfurt am Main, Hessen, Germany, 60596 GSK Investigational Site Frankfurt, Hessen, Germany, 60596 GSK Investigational Site Magdeburg, Sachsen-Anhalt, Germany, 39112 GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT01668784 | - | - | Active, not recruiting | - | - |
| NCT01668654 | Epilepsy | Phase 3 | Terminated(FDA placed a clinic... more >>al hold on the Pediatric Program requiring retigabine discontinuation in subjects; early termination allows for timely reporting of results.) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90027 United States, Florida GSK Investigational Site Gulf Breeze, Florida, United States, 32561 GSK Investigational Site Port Charlotte, Florida, United States, 33952 GSK Investigational Site Wellington, Florida, United States, 33414 United States, Minnesota GSK Investigational Site Saint Paul, Minnesota, United States, 55102-2534 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Tennessee GSK Investigational Site Memphis, Tennessee, United States, 38105 United States, Texas GSK Investigational Site Austin, Texas, United States, 78723 GSK Investigational Site Dallas, Texas, United States, 75230-2507 Collapse << |
| NCT01366521 | - | - | Completed | - | - |
| NCT00985673 | - | - | Completed | - | - |
| NCT01668654 | - | - | Terminated(FDA placed a clinic... more >>al hold on the Pediatric Program requiring retigabine discontinuation in subjects; early termination allows for timely reporting of results.) Collapse << | - | - |
| NCT02319148 | - | - | Completed | - | - |
| NCT00404768 | Obstetric Labour, Premature | Phase 2 | Completed | - | - |
| NCT00903617 | Dyslipidaemias | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Pembroke Pines, Florida, United States, 33026 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46260 United States, Kentucky GSK Investigational Site Louisville, Kentucky, United States, 40213 United States, Maine GSK Investigational Site Auburn, Maine, United States, 04210 United States, Minnesota GSK Investigational Site Brooklyn Center, Minnesota, United States, 55430 United States, North Carolina GSK Investigational Site Statesville, North Carolina, United States, 28677 United States, Ohio GSK Investigational Site Cincinnati, Ohio, United States, 45246 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78205 United States, Virginia GSK Investigational Site Richmond, Virginia, United States, 23294 United States, Washington GSK Investigational Site Olympia, Washington, United States, 98502 GSK Investigational Site Seattle, Washington, United States, 98104 Collapse << |
| NCT02370238 | Metastatic Breast Cancer | Phase 2 | Active, not recruiting | February 2019 | - |
| NCT00404768 | - | - | Completed | - | - |
| NCT00048048 | Anemia | Phase 2 | Completed | - | United States, Alabama ... more >> Birmingham, Alabama, United States, 35211 United States, California San Diego, California, United States, 92103-8342 United States, Michigan Detroit, Michigan, United States, 48202-2689 Detroit, Michigan, United States, 48236 United States, Nevada Las Vegas, Nevada, United States, 89106 United States, Oregon Portland, Oregon, United States, 97201-2940 Canada, Alberta Edmonton, Alberta, Canada, T6G 2B7 Canada, British Columbia Vancouver, British Columbia, Canada, V6Z 1Y6 Mexico Mexico City, Mexico, 14000 Monterrey, Mexico, 64710 Poland Gdansk, Poland, 80-211 Krakow, Poland, 31-501 Wroclaw, Poland, 50-417 United Kingdom Belfast, United Kingdom, BT9 7LJ London, United Kingdom, SE22 8PT Collapse << |
| NCT02621931 | Migraine | Phase 3 | Completed | - | - |
| NCT01529346 | - | - | Completed | - | - |
| NCT01529346 | Postoperative Dental Pain | Phase 2 | Completed | - | United States, Texas ... more >> Central Texas Oral Surgery Associates Austin, Texas, United States, 78705 Premier Research Group Limited Austin, Texas, United States, 78705 PPD Development, LP Austin, Texas, United States, 78744 Collapse << |
| NCT00277212 | Bipolar Disorder | Phase 4 | Completed | - | - |
| NCT00048048 | - | - | Completed | - | - |
| NCT00696657 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT00244751 | Cirrhosis, Liver | Phase 2 | Completed | - | - |
| NCT00277212 | - | - | Completed | - | - |
| NCT03432871 | Mitochondrial Diseases ... more >> Mitochondrial Myopathies Progressive External Ophthalmoplegia Progressive Ophthalmoplegia Progressive; Ophthalmoplegia, External Mitochondria DNA Deletion MELAS Mitochondrial Encephalomyopathy, Lactic Acidosis, and Stroke-Like Episodes Mitochondrial Encephalopathy, Lactic Acidosis and Stroke-Like Episodes (MELAS Syndrome) Collapse << | Not Applicable | Recruiting | May 30, 2019 | United Kingdom ... more >> Cambridge University Hospitals NHS Foundation Trust Recruiting Cambridge, United Kingdom, CB20QQ Contact: Zoe McIntyre, MSc 0044 1223 331506 mitopatients@mrc-mbu.cam.ac.uk Contact: Sarah Bird, MSc 0044 1223 331506 mitopatients@mrc-mbu.cam.ac.uk Principal Investigator: Patrick F Chinnery, MD PhD Collapse << |
| NCT00559585 | Rheumatoid Arthritis (RA) | Phase 3 | Completed | - | - |
| NCT00559585 | - | - | Completed | - | - |
| NCT00244751 | - | - | Completed | - | - |
| NCT02621931 | - | - | Completed | - | - |
| NCT01147809 | Thrombocytopaenia | Phase 2 | Completed | - | - |
| NCT02629861 | Migraine | Phase 3 | Completed | - | - |
| NCT00696657 | - | - | Completed | - | - |
| NCT01147809 | - | - | Completed | - | - |
| NCT02629861 | - | - | Completed | - | - |
| NCT02907944 | Substance-related Disorders | Not Applicable | Recruiting | September 2021 | United States, Connecticut ... more >> Hartford Hospital's HIV Clinic Recruiting Hartford, Connecticut, United States, 06115 Contact: Deborah Cornman, PhD 860-486-4645 Haelen Center at Yale New Haven Hospital Recruiting New Haven, Connecticut, United States, 06511 Contact: Evangelia Louizos 203-737-3347 United States, New York SUNY Downstate Medical Center Recruiting Brooklyn, New York, United States, 11203 Contact: Gabriel Rebick, MD 718-270-3745 United States, Rhode Island Immunology Center at Miriam Hospital Recruiting Providence, Rhode Island, United States, 02906 Contact: Philip Chan, MD 401-793-2928 Collapse << |
| NCT02377427 | Asthma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site San Diego, California, United States, 92123 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599-7310 United States, Oklahoma GSK Investigational Site Oklahoma City, Oklahoma, United States, 73112 United States, Oregon GSK Investigational Site Medford, Oregon, United States, 97504 United States, Wisconsin GSK Investigational Site Madison, Wisconsin, United States, 53792-9988 Japan GSK Investigational Site Fukuoka, Japan, 811-1394 GSK Investigational Site Hiroshima, Japan, 720-8520 GSK Investigational Site Hiroshima, Japan, 721-8511 GSK Investigational Site Wakayama, Japan, 646-8558 Poland GSK Investigational Site Lodz, Poland, 90-329 GSK Investigational Site Tarnow, Poland, 33-100 GSK Investigational Site Wroclaw, Poland, 53-201 United Kingdom GSK Investigational Site Glasgow, United Kingdom, G514TF GSK Investigational Site Leicester, United Kingdom, LE27LX GSK Investigational Site Liverpool, United Kingdom, L12 2AP GSK Investigational Site London, United Kingdom, SE5 9RS GSK Investigational Site London, United Kingdom, SW3 6NP GSK Investigational Site Oxford, United Kingdom, OX3 9DU GSK Investigational Site Sheffield, United Kingdom, S10 2TH Collapse << |
| NCT02341482 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01577381 | Age-Related Maculopathy | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT02341482 | - | - | Completed | - | - |
| NCT02377427 | - | - | Completed | - | - |
| NCT01577381 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01819922 | Healthy | Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00313144 | Alpha1-antitrypsin Deficiency | Phase 4 | Terminated(Study terminated ea... more >>rly due to Aralast being phased out of the market.) Collapse << | - | United States, California ... more >> Adupa Rao, MD San Marino, California, United States, 91108 Collapse << |
| NCT01819922 | - | - | Completed | - | - |
| NCT02211352 | Hypertension | Phase 2 | Completed | - | Korea, Republic of ... more >> Daegu Catholic University Medical Center Daegu, Korea, Republic of, 705-718 Collapse << |
| NCT01971476 | - | - | Completed | - | - |
| NCT00195260 | Neoplasms | Phase 1 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmington, Alabama, United States, 35233 United States, Arizona Pfizer Investigational Site Scottsdale, Arizona, United States, 85258 United States, California Pfizer Investigational Site Los Angeles, California, United States, 90033 United States, Florida Pfizer Investigational Site Tampa, Florida, United States, 33612 United States, Georgia Pfizer Investigational Site Atlanta, Georgia, United States, 30341 United States, Indiana Pfizer Investigational Site Indianpolis, Indiana, United States, 46202 United States, Maryland Pfizer Investigational Site Baltimore, Maryland, United States, 21287 United States, Michigan Pfizer Investigational Site Detroit, Michigan, United States, 48202 Pfizer Investigational Site Lansing, Michigan, United States, 48910 United States, New York Pfizer Investigational Site New York, New York, United States, 10016 Pfizer Investigational Site New York, New York, United States, 10032 United States, North Carolina Pfizer Investigational Site Charlotte, North Carolina, United States, 28203 Pfizer Investigational Site Charlotte, North Carolina, United States, 28211 Pfizer Investigational Site UNC Chapel Hill, North Carolina, United States, 27514 Pfizer Investigational Site UNC Chapel Hill, North Carolina, United States, 27759 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44106-1736 United States, Texas Pfizer Investigational Site San Antonio, Texas, United States, 78258 Pfizer Investigational Site Tyler, Texas, United States, 75702 United States, Washington Pfizer Investigational Site Seattle, Washington, United States, 98104 Collapse << |
| NCT01971476 | Leukemia Neop... more >>lasms Collapse << | Phase 1 | Completed | - | Belgium ... more >> UNIV UZ Gent Gent, Belgium, 9000 Czechia University Hospital Motol Prague, Czechia, 150 06 France INS Curie Paris, France, 75248 Germany Universitätsklinikum Köln (AöR) Köln, Germany, 50937 Italy Osp. Pediatrico Bambin Gesù Roma, Italy, 00165 Collapse << |
| NCT00458393 | - | - | Completed | - | - |
| NCT01121536 | Depression | Phase 3 | Terminated(Business decision r... more >>elated to efficacy rather than tolerability limitations, not stopped for any safety reasons.) Collapse << | - | - |
| NCT00313144 | - | - | Terminated(Study terminated ea... more >>rly due to Aralast being phased out of the market.) Collapse << | - | - |
| NCT02716194 | - | - | Completed | - | - |
| NCT00458393 | HIV Infections | Phase 3 | Completed | - | United States, California ... more >> San Francisco Dept. of Public Health iPrEx CRS San Francisco, California, United States, 94102 United States, Illinois Stroger Hospital of Cook County/Core Center IPREX CRS Chicago, Illinois, United States, 60612 United States, Massachusetts Fenway Community Health iPrEx CRS Boston, Massachusetts, United States, 02215 Brazil IPEC/FIOCRUZ iPrEx CRS Rio de Janeiro, Brazil, 21040-900 Projeto Praca Onze, Universidade Federal do Rio de Janeiro iPrEx CRS Rio de Janeiro, Brazil, 21941.590 Universidade de Sao Paulo iPrEx CRS Sao Paulo, Brazil, 05403 Ecuador Fundación Ecuatoriana Equidad, Guayaquil, iPrEx CRS Guayaquil, Guayas, Ecuador Peru Asociación Civil Selva Amazónica, Iquitos, iPrEx CRS Iquitos, Maynas, Peru Investigaciones Médicas en Salud (INMENSA), Lince, iPrEx CRS Lima, Peru South Africa Desmond Tutu HIV Ctr. iPrEx CRS Cape Town, South Africa, 7925 Thailand Research Institute for Health Sciences iPrEx CRS Chiang Mai, Thailand, 50200 Collapse << |
| NCT01779882 | Myeloid Leukemia ... more >> Precursor Myeloid Neoplasms Lymphoid Neoplasms Collapse << | Not Applicable | Completed | - | Switzerland ... more >> University Hospital, Basel Basel, Switzerland, 4031 University Hospital Geneva Geneva, Switzerland, 1205 University Hospital Zurich Zurich, Switzerland, 8091 Collapse << |
| NCT00195260 | - | - | Completed | - | - |
| NCT00490646 | Metastatic Breast Cancer | Phase 2 | Completed | - | France ... more >> Local Institution Caen, France, F-14076 Local Institution Grenoble, France, 38028 Local Institution St Priest En Jarez, France, 42271 Local Institution Toulouse, France, 31300 Greece Local Institution Athens, Greece, 11522 Local Institution Athens, Greece, 12462 Local Institution Pireaus, Greece, 18537 Italy Local Institution Bologna, Italy, 40138 Local Institution Brescia, Italy, 25123 Local Institution Modena, Italy, 41100 Local Institution Napoli, Italy, 80131 Local Institution Roma, Italy, 00144 Local Institution Sora, Italy, 03039 Spain Local Institution Madrid, Spain, 28007 Local Institution Madrid, Spain, 28034 Turkey Local Institution Ankara, Turkey, 06100 Local Institution Ankara, Turkey, 06500 Local Institution Izmir, Turkey, 35100 Local Institution Izmir, Turkey, 35340 Collapse << |
| NCT01121536 | - | - | Terminated(Business decision r... more >>elated to efficacy rather than tolerability limitations, not stopped for any safety reasons.) Collapse << | - | - |
| NCT02716194 | Hemophilia A | Phase 1 | Completed | - | Bulgaria ... more >> UMHAT "Sv. Georgi", EAD Plovdiv, Bulgaria, 4000 Germany Werlhof-Institut Hannover, Niedersachsen, Germany, 30159 Medizinische Hochschule Hannover Hannover, Niedersachsen, Germany, 30625 Vivantes Klinikum im Friedrichshain - Landsberger Allee Berlin, Germany, 10249 Universitaetsklinikum des Saarlandes Homburg, Germany, 66421 Universitaetsklinikum Gießen Marburg, Germany, 35043 Hungary Semmelweis Egyetem AOK I.sz. Belgyogyaszati Klinika Budapest, Hungary, 1083 Italy Presidio Ospedaliero di Castelfranco Veneto Castelfranco Veneto, Treviso, Italy, 31033 Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico Milano, Italy, 20122 Policlinico Umberto I di Roma-Università di Roma La Sapienza Roma, Italy, 00144 Netherlands Radboud University Nijmegen Medical Centre Nijmegen, Netherlands, 6525 GA Erasmus Medisch Centrum Rotterdam, Netherlands, 3015 AA Poland Instytut Hematologii i Transfuzjologii Warszawa, Poland, 02-776 Russian Federation FSBI "Kirov SR Institute of Hematology and Blood Transfusion FMBA" Kirov, Russian Federation, 610027 FSBI "Hematological Research Center" MoH of RF Moscow, Russian Federation, 125167 SBEI HPE "Samara State Medical University" of the MoH of the RF Samara, Russian Federation, 443099 Spain Complejo Hospitalario Universitario A Coruña A Coruña, La Coruña, Spain, 15006 Hospital General Universitario de Alicante Alicante, Spain, 03010 Hospital Universitari Vall d'Hebron Barcelona, Spain, 08035 Hospital Universitario La Paz Madrid, Spain, 28046 Hospital Regional Universitario de Malaga Malaga, Spain, 29010 Hospital Universitario Son Espases Palma de Mallorca, Spain, 07120 United Kingdom Royal Cornwall Hospital Truro, Cornwall, United Kingdom, TR1 3LJ Royal London Hospital London, Greater London, United Kingdom, E1 1BB Royal Free Hospital London, Greater London, United Kingdom, NW3 2QG St Thomas' Hospital Centre for Haemostasis & Thrombosis London, Greater London, United Kingdom, SE1 7EH Manchester Royal Infirmary Manchester, Greater Manchester, United Kingdom, M13 9WL University Hospital of Wales Cardiff, West Glamorgan, United Kingdom, CF14 4XW Collapse << |
| NCT02452047 | Bacterial Infections | Phase 3 | Completed | - | - |
| NCT03090516 | Alzheimer's | Phase 2 Phase 3 | Recruiting | March 10, 2018 | China, Jiangsu ... more >> Ting Wu Recruiting Nanjing, Jiangsu, China, 210000 Contact: Ting Wu, professor 18851122018 wuting80000@126.com Contact: Wanrong Zhang, master 15851875395 zwr10010533@163.com Collapse << |
| NCT00490646 | - | - | Completed | - | - |
| NCT01109524 | - | - | Completed | - | - |
| NCT00818623 | Prostate Cancer | Phase 2 | Completed | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark KAS Glostrup Glostrup, Denmark KAS Herlev Herlev, Denmark Finland Marian Sairaala Helsinki, Finland P-K Keskussairaala Joensuu, Finland Vuorikadun lääkäriasema Kuopio, Finland OYS Oulu, Finland Kirugikeskus Seinäjoki, Finland TAYS Tampere, Finland Hungary Bajcsy-Zsilinszky Hospital, Urology Budapest, Hungary Jahn Ferenc Dél Pesti Hospital, Urology Budapest, Hungary Péterfy Hospital, Urology Budapest, Hungary Bács-Kiskun County Hospital, Urology Kecskemét, Hungary Hospital of Local Gov. Szeged, Urology Szeged, Hungary MÁV Hospital, Urology Szolnok, Hungary Norway Sentralsykehuset i Rogland Stavanger, Norway Romania CF2 Hospital - Bucharest, Urology Bucharest, Romania Dr. Th Burghele Hospital Bucharest, Romania Fundeni Hospital - Bucharest, Urology Bucharest, Romania County Hospital - Timisoara, Urology Timisoara, Romania Russian Federation City Hospital #1, State Med Univ/Urology Moscow, Russian Federation Institute of Urology of MoH Moscow, Russian Federation Moscow City Hospital #60, Urology Moscow, Russian Federation City Hospital #15, Urology Department St. Petersburg, Russian Federation City Hospital #26, Urology Department St. Petersburg, Russian Federation Sweden Sahlgrenska Universitetssjukehuset Göteborg, Sweden Helsingborgs Lasaret Helsingborg, Sweden Universitetssjukehuset, MAS Malmö, Sweden Akademiska Sjukhuset Uppsala Uppsala, Sweden University Hospital, Örebro Örebro, Sweden Collapse << |
| NCT02452047 | - | - | Completed | - | - |
| NCT00819741 | - | - | Completed | - | - |
| NCT01251809 | Acute Lymphoblastic Leukaemia | Phase 1 Phase 2 | Terminated | - | Germany ... more >> Charité Campus Benjamin Franklin Hämatologie, Onkologie, Transfusionsmedizin Medizinische Klinik III Berlin, Germany, 12200 Charité University Hospital Campus Virchow Berlin, Germany, 13353 Universität Bonn, Medizinische Klinik & Poliklinik III Bonn, Germany, 53105 Städtisches Klinikum Braunschweig Medizinische Klinik III Braunschweig, Germany, 38114 Klinikum Carl Gustav Carus der Technischen Universität Dresden, Germany, 01307 St. Johannes-Hospital Duisburg, Germany, 47166 Universität Erlangen-Nürnberg Med. Klinik V/Hämatologie Erlangen, Germany, 91054 Universitätsklinikum Essen Westdeutsches Tumorzentrum Essen, Germany, 45147 Universitätsklinikum Frankfurt Medizinische Klinik II Frankfurt, Germany, 60590 Universitätsmedizin Göttingen Hämatologie / Onkologie Göttingen, Germany, 37075 Katholisches Krankenhaus Hagen gGmbH Klinik für Hämatologie und Onkologie Hagen, Germany, 58095 Asklepios Klinik St. Georg Hämatologie & Stammzelltransplantation Hamburg, Germany, 20099 Asklepios Klinik Altona II. Medizinische Abteilung Hamburg, Germany, 22763 Evangelisches Krankenhaus Medizinische Klinik Hämatologie/Onkologie Hamm, Germany, 59063 Medical University Hannover Hannover, Germany, 30625 Universitätsklinikum Heidelberg Heidelberg, Germany, 69120 Universitätsklinikum Schleswig-Holstein Kiel, Germany, 24116 Universität Leipzig José-Carreras-Haus Abt. Hämatologie / Onkologie Leipzig, Germany, 04103 Universitätsmedizin Mainz III. Medizinische Klinik Mainz, Germany, 55131 Klinikum Schwabing, Klinik für Hämatologie, Onkologie, Immunologie, Palliativmedizin, Infektiologie & Tropenmedizin München, Germany, 80804 Klinikum Rechts der Isar der TU München III. Medizinische Klinik München, Germany, 81675 Universitätsklinikum Münster Münster, Germany, 48129 Klinikum Nürnberg, 5. Medizinische Klinik Nürnberg, Germany, 90419 Klinikum Oldenburg Innere Medizin II Oldenburg, Germany, 26133 Klinikum Ernst von Bergmann, Zentrum für Hämatologie, Onkologie und Strahlenheilkunde Potsdam, Germany, 14467 Klinikum der Universität Regensburg Regensburg, Germany, 93053 Universität Rostock, Zentrum für Innere Medizin, Klinik III Rostock, Germany, 18057 Robert Bosch-Krankenhaus Abt. Hämatologie / Onkologie Stuttgart, Germany, 70376 Universitätsklinik Ulm Klinik für Innere Medizin III Zentrum für Innere Medizin Ulm, Germany, 89070 Klinikum der Universität Würzburg Würzburg, Germany, 97070 Collapse << |
| NCT00818623 | - | - | Completed | - | - |
| NCT00633464 | Triple Negative Locally Advanc... more >>ed Non-resectable Breast Cancer Metastatic Breast Cancer Collapse << | Phase 2 | Completed | - | Austria ... more >> Local Institution Graz, Austria, 8036 Local Institution Wien, Austria, 1090 Czech Republic Local Institution Brno, Czech Republic, 656 53 Local Institution Prague 5, Czech Republic, 150 06 Local Institution Praha 2, Czech Republic, 128 08 France Local Institution Bayonne, France, 64100 Local Institution Dijon Cedex, France, 21079 Local Institution Lyon, France, 69008 Local Institution Paris Cedex 13, France, 75651 Local Institution Saint Brieuc, France, 22015 Local Institution Saint Herblain Cedex, France, 44805 Local Institution Toulouse Cedex, France, 31052 Greece Local Institution Thessaloniki, Greece, 54642 Italy Local Institution Napoli, Italy, 80131 Poland Local Institution Gdansk, Poland, 80952 Local Institution Olsztyn, Poland, 10-513 Spain Local Institution Barcelona, Spain, 08036 Local Institution Barcelona, Spain, 08208 Local Institution Barcelona, Spain, 08221 Collapse << |
| NCT00983957 | Chronic Hepatitis C | Phase 1 | Completed | - | United States, Arizona ... more >> MDS Pharma Services (US), Inc Tempe, Arizona, United States, 85283 United States, Texas Covance Clinical Research Unit, Inc. Austin, Texas, United States, 78752 Canada, Quebec Local Institution St. Laurent, Quebec, Canada, H4R2N6 Collapse << |
| NCT00633464 | - | - | Completed | - | - |
| NCT00819741 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 4 | Completed | - | China, Fujian ... more >> Novo Nordisk Investigational Site Fuzhou, Fujian, China, 350001 China, Jiangsu Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210009 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210012 Novo Nordisk Investigational Site Nanjing, Jiangsu, China, 210029 Novo Nordisk Investigational Site Suzhou, Jiangsu, China, 215004 Novo Nordisk Investigational Site Wuxi, Jiangsu, China, 214023 China, Jiangxi Novo Nordisk Investigational Site Nanchang, Jiangxi, China, 330006 China, Shanghai Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200001 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200025 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200072 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200092 Novo Nordisk Investigational Site Shanghai, Shanghai, China, 200120 China, Zhejiang Novo Nordisk Investigational Site Hangzhou, Zhejiang, China, 310003 Novo Nordisk Investigational Site Wenzhou, Zhejiang, China, 325000 China Novo Nordisk Investigational Site Hangzhou, China, 310016 Novo Nordisk Investigational Site Nantong, China, 226001 Novo Nordisk Investigational Site Shanghai, China, 200040 Collapse << |
| NCT03041961 | Healthy Young Men | Not Applicable | Completed | - | United Kingdom ... more >> Department of Life Sciences and Medecine London, Central London, United Kingdom, SE1 9NH Collapse << |
| NCT03649438 | Relapsed Neuroblastoma ... more >> Metastatic Pheochromocytoma Collapse << | Early Phase 1 | Not yet recruiting | December 31, 2021 | United States, Texas ... more >> The University of Texas Southwestern Medical Center Not yet recruiting Dallas, Texas, United States, 75235 Contact: Tanya Watt, MD 214-456-6363 Tanya.Watt@UTSouthwestern.edu Principal Investigator: Tanya Watt, MD Collapse << |
| NCT01109524 | Lung Neoplasms ... more >> Carcinoma Cancer of the Lung Non-Small-Cell Lung Carcinoma Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Donald W. Hill M.D., P.C. Casa Grande, Arizona, United States, 85122 United States, California Beverly Hills Cancer Center Beverly Hills, California, United States, 90211 Cancer Care Institute Los Angeles, California, United States, 90036 Northern California Hematology & Oncology Oakland, California, United States, 94609 Sharp Clinical Oncology Research San Diego, California, United States, 92123 United States, Florida Broward Oncology Associates, P.A. Ft. Lauderdale, Florida, United States, 33308 Elite Research Institute Miami, Florida, United States, 33125 United States, Illinois Edward H. Kaplan, MD & Associates Skokie, Illinois, United States, 60076 United States, Minnesota Fairview Southdale Medical Oncology Clinic Edina, Minnesota, United States, 55435 United States, North Dakota Mid Dakota Clinic, Pc Bismarck, North Dakota, United States, 58501 United States, Pennsylvania Temple University Hospital Philadelphia, Pennsylvania, United States, 19140 United States, Texas University Medical Center Lubbock, Texas, United States, 79415 United States, Washington Columbia Basin Hematology and Oncology Kennewick, Washington, United States, 99336 Canada, New Brunswick The Moncton Hospital Moncton, New Brunswick, Canada, E1C 6Z8 Canada, Newfoundland and Labrador Local Institution Grand Falls-Windsor, Newfoundland and Labrador, Canada, A2A 2E2 Canada, Ontario The Credit Valley Hospital Mississauga, Ontario, Canada, L5M 2N1 Sudbury Regional Hospital Sudbury, Ontario, Canada, P3E 5J1 Thunder Bay Regional Health Sciences Centre (Regional Cancer Care) Thunder Bay, Ontario, Canada, P7B 6V4 Toronto East General Hospital Toronto, Ontario, Canada, M4C 3E7 Canada, Quebec Centre de sante et de services sociaux de Rimouski-Neigette Rimouski, Quebec, Canada, G5L 5T1 Puerto Rico Ponce School of Medicine (Caimed Center) Ponce, Puerto Rico, 00716 Fundacion de Investigacion de Diego San Juan, Puerto Rico, 00927 Collapse << |
| NCT03624556 | Down Syndrome ... more >> Fragile X Syndrome Collapse << | Not Applicable | Recruiting | September 1, 2019 | Spain ... more >> IMIM (Institut Hospital del Mar d'Investigacions Mèdiques) Recruiting Barcelona, Spain, 08003 Contact: Rafael De la Torre Fornell, PhD 0034933160484 rtorre@imim.es Principal Investigator: Rafael de la Torre, PhD Collapse << |
| NCT00958568 | Treatment Resistant Depression | Phase 3 | Completed | - | - |
| NCT00983957 | - | - | Completed | - | - |
| NCT01894022 | Hypertension | Phase 3 | Terminated | - | - |
| NCT00958568 | - | - | Completed | - | - |
| NCT01925274 | - | - | Terminated(Enrollment to the s... more >>tudy was terminated on 11Nvo2014 due to slow recruitment. There were no safety or efficacy issues that contributed to this decision.) Collapse << | - | - |
| NCT01792635 | Diabetes Mellitus, Type 2 | Phase 2 | Terminated(The study was termi... more >>nated prematurely on 16 May 2014 due to a safety concern.) Collapse << | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 Collapse << |
| NCT00048568 | - | - | Completed | - | - |
| NCT01925274 | Metastatic Colorectal Cancer | Phase 2 | Terminated(Enrollment to the s... more >>tudy was terminated on 11Nvo2014 due to slow recruitment. There were no safety or efficacy issues that contributed to this decision.) Collapse << | - | United States, California ... more >> CBCC Global Research, Inc. at Comprehensive Blood and Cancer Center Bakersfield, California, United States, 93309 Drug Management Only: UCLA West Medical Pharmacy, Att: Steven L Wong, Pharm D Los Angeles, California, United States, 90095-7349 Drug Management Only: UCLA West Medical Pharmacy Los Angeles, California, United States, 90095-7349 UCLA West Medical Pharmacy Los Angeles, California, United States, 90095-7349 Regulatory Management Only: TRIO-US Central Administration Los Angeles, California, United States, 90095 TRIO-US Central Administration (Regulatory Management only) Los Angeles, California, United States, 90095 TRIO_US Los Angeles, California, United States, 90095 West Valley Hematology/Oncology Med Group Northridge, California, United States, 91328 United States, Missouri Siteman Cancer Center - West County Creve Coeur, Missouri, United States, 63141 Barnes-Jewish Hospital Saint Louis, Missouri, United States, 63110 Drug Shipping Address: Washington University Infusion Center Pharmacy Saint Louis, Missouri, United States, 63110 Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Siteman Cancer Center - South County Saint Louis, Missouri, United States, 63129 Siteman Cancer Center - St Peters Saint Peters, Missouri, United States, 63376 United States, Nevada Regulatory Office: Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89014 Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89052 Comprehensive Cancer Centers of Nevada Henderson, Nevada, United States, 89074 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89128 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89148 Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89169 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 United States, Washington Kadlec Clinic Hematology and Oncology Kennewick, Washington, United States, 99336 Kadlec Medical Center Richland, Washington, United States, 99352 Outpatient Imaging Center Richland, Washington, United States, 99352 Spokane Valley Cancer Center Spokane Valley, Washington, United States, 99216 Medical Oncology Associates, PS Spokane, Washington, United States, 99208 Japan Aichi cancer center central hospital Nagoya, Aichi, Japan, 464-8681 National Cancer Center Hospital East Kashiwa, Chiba, Japan, 277-8577 Korea, Republic of National Cancer Center Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Seoul National University Hospital / Department of Internal Medicine Seoul, Korea, Republic of, 110-744 Samsung Medical Center Seoul, Korea, Republic of, 135-710 Asan Medical Center Seoul, Korea, Republic of, 138-736 Spain Hospital General Universitario Gregorio Marañón Madrid, Spain, 28009 Collapse << |
| NCT02969044 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT01894022 | - | - | Terminated | - | - |
| NCT01792635 | - | - | Terminated(The study was termi... more >>nated prematurely on 16 May 2014 due to a safety concern.) Collapse << | - | - |
| NCT02969044 | - | - | Completed | - | - |
| NCT00048568 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT00893789 | Traumatic Brain Injury | Phase 3 | Terminated(Study has been stop... more >>ped by sponsor decision.) Collapse << | - | - |
| NCT00893789 | - | - | Terminated(Study has been stop... more >>ped by sponsor decision.) Collapse << | - | - |
| NCT01929863 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT01050998 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT01929863 | - | - | Completed | - | - |
| NCT01970865 | ALK-positive Non Small Cell Lu... more >>ng Cancer (NSCLC) and ROS1-positive NSCLC Collapse << | Phase 2 | Active, not recruiting | February 19, 2019 | - |
| NCT02807415 | - | - | Recruiting | June 2019 | Germany ... more >> Justus-Liebig-University Recruiting Gießen, Germany, 35385 Contact: Lutz Nährlich, MD 004964198557620 lutz.naehrlich@paediat.med.uni-giessen.de Contact: Claudia Rückes-Nilges, Dipl-Biol 004964198556945 claudia.rueckes-nilges@paediat.med.uni-giessen.de Principal Investigator: Lutz Nährlich, MD Sub-Investigator: Stefan Kuhnert, MD Hannover Medical School Recruiting Hannover, Germany, 30625 Contact: Burkhard Tümmler, MD PhD 00495115322920 tuemmler.burkhard@mh-hannover.de Contact: Christian Dopfer, MD 00495115326111 dopfer.christian@mh-hannover.de Principal Investigator: Burkhard Tümmler, MD PhD Sub-Investigator: Christian Dopfer, MD University of Heidelberg Recruiting Heidelberg, Germany, 69120 Contact: Marcus Mall, MD 00496221564502 Marcus.Mall@med.uni-heidelberg.de Contact: Simon Gräber, MD 004962215638926 Simon.Graeber@med.uni-heidelberg.de Principal Investigator: Marcus Mall, MD Sub-Investigator: Simon Gräber, MD Sub-Investigator: Susanne Dittrich, MD Collapse << |
| NCT01536379 | - | - | Completed | - | - |
| NCT00190684 | Attention Deficit Hyperactivit... more >>y Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT01536379 | Transplantation, Organ | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 0QQ GSK Investigational Site London, United Kingdom, SE1 9RT Collapse << |
| NCT01119846 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Arizona ... more >> GSK Investigational Site Phoenix, Arizona, United States, 85013 United States, California GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27705 United States, Ohio GSK Investigational Site Columbus, Ohio, United States, 43212 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 United States, Washington GSK Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT02565628 | Idiopathic Parkinson's Disease | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 United States, Florida MD Clinical Hallandale Beach, Florida, United States, 33009 QPS-MRA, LLC (Broward Research Group) Hollywood, Florida, United States, 33024 Qps-Mra Llc South Miami, Florida, United States, 33143 QPS-MRA, LLC (Miami research Associates) South Miami, Florida, United States, 33143 QPS-MRA, LLC (MRA Clinical Research) South Miami, Florida, United States, 33143 United States, Georgia Atlanta Center for Medical Research Atlanta, Georgia, United States, 30331 United States, New Jersey CRI Worldwide, LLC Marlton, New Jersey, United States, 08053 United States, Ohio CTI Clinical Research Center Cincinnati, Ohio, United States, 45255 Collapse << |
| NCT00190684 | - | - | Completed | - | - |
| NCT02565628 | - | - | Completed | - | - |
| NCT00453479 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Netherlands ... more >> GSK Investigational Site Eindhoven, Netherlands, 5623 EJ GSK Investigational Site Harderwijk, Netherlands, 3844 DG GSK Investigational Site Utrecht, Netherlands, 3584 CJ GSK Investigational Site Zuidlaren, Netherlands, 9471 GP Collapse << |
| NCT00698841 | - | - | Completed | - | - |
| NCT01970865 | - | - | Active, not recruiting | - | - |
| NCT00453479 | - | - | Completed | - | - |
| NCT01050998 | - | - | Completed | - | - |
| NCT00822900 | - | - | Terminated(futility low condit... more >>ional power to demonstrate benefit of progesterone) Collapse << | - | - |
| NCT02202161 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21225 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78209 Collapse << |
| NCT00822900 | Traumatic Brain Injury | Phase 3 | Terminated(futility low condit... more >>ional power to demonstrate benefit of progesterone) Collapse << | - | - |
| NCT01119846 | - | - | Completed | - | - |
| NCT01573923 | Liver Cirrhosis | Phase 1 Phase 2 | Unknown | October 2015 | China, Beijing ... more >> The 302 Hospital of Chinese People's Liberation Army Recruiting Beijing, Beijing, China Principal Investigator: Hanwei Li, MD, PhD China, Gansu the First Affiliated Hospital of Lanzhou University Recruiting Lanzhou, Gansu, China Contact: Xun Li, MD, PhD Principal Investigator: Xun Li, MD China, Hainan Hainan BOAO Life infinity international anti-aging medical center Recruiting Qionghai, Hainan, China, 571434 Contact: Jiang Shu Jiang.st@hotmail.com China, Jiangsu The first people's hospital of Lianyungang Recruiting Lianyungang, Jiangsu, China, 222002 Contact: Hui Shi, MD Principal Investigator: Hui Shi, MD China, Shanghai Tongji Hospital of Tongji University Recruiting Shanghai, Shanghai, China Contact: Liming Chen, MD, PhD Principal Investigator: Jianwei Lu, PhD China, Shanxi The 323 Hospital of Chinese People's Liberation Army Recruiting Xi'an, Shanxi, China, 710054 Contact: Liming Wang, MD 86-29-84756502 wanglm@fmmu.edu.cn Principal Investigator: Liming Wang, MD Collapse << |
| NCT02202161 | - | - | Completed | - | - |
| NCT00698841 | Advanced Cancer | Phase 2 | Completed | - | United States, Alabama ... more >> Northwest Alabama Cancer Center Muscle Shoals, Alabama, United States, 35661 United States, Arizona Donald W. Hill, MD Casa Grande, Arizona, United States, 85222 United States, California Compassionate Cancer Care Medical Group, Inc Corona, California, United States, 92879 Compassionate Cancer Care Medical Group Inc Fountain Valley, California, United States, 92708 Pacific Shores Medical Group Long Beach, California, United States, 90813 Desert Hospital Comprehensive Cancer Center Palm Springs, California, United States, 92262 Compassionate Cancer Care Medical Group, Inc Riverside, California, United States, 92501 American Institute Research Whittier, California, United States, 90603 United States, District of Columbia Georgetown University Medical Center Washington, District of Columbia, United States, 20007 United States, Florida Baptist Cancer Institute Jacksonville, Florida, United States, 32207 Ocala Oncology Center Ocala, Florida, United States, 34471 United States, Louisiana Brinz, Burroff, Gurtler, & Russo Metairie, Louisiana, United States, 70006 United States, Oklahoma Cancer Specialists Of Oklahoma Oklahoma City, Oklahoma, United States, 73112 United States, Rhode Island Pharma Resource East Providence, Rhode Island, United States, 02915 United States, Texas Austin Cancer Centers Austin, Texas, United States, 78759 Puerto Rico Local Institution San Juan, Puerto Rico, 00910 Collapse << |
| NCT01488097 | - | - | Completed | - | - |
| NCT00358449 | Oesophagitis, Eosinophilic | Phase 2 | Completed | - | - |
| NCT00477334 | Genital Herpes | Phase 4 | Completed | - | - |
| NCT01488097 | Cholesterol Ester Storage Dise... more >>ase (CESD) Lysosomal Acid Lipase Deficiency LAL-Deficiency Collapse << | Phase 2 | Completed | - | United States, California ... more >> Eureka, California, United States, 95501-320 Sacramento, California, United States, 95817 San Francisco, California, United States, 94143 United States, Minnesota Minneapolis, Minnesota, United States, 55455 United States, New York New York, New York, United States, 10029 Canada, Ontario Sudbury, Ontario, Canada Czechia Prague, Czechia France Paris, France United Kingdom Leeds, United Kingdom Salford, United Kingdom Collapse << |
| NCT02130193 | - | - | Completed | - | - |
| NCT00358449 | - | - | Completed | - | - |
| NCT02130193 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Birmingham, Alabama, United States, 35294 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80206 United States, Iowa GSK Investigational Site Iowa City, Iowa, United States, 52242 United States, Pennsylvania GSK Investigational Site Oaks, Pennsylvania, United States, 19456 GSK Investigational Site Philadelphia, Pennsylvania, United States, 19140 United States, South Carolina GSK Investigational Site Spartanburg, South Carolina, United States, 29303 Germany GSK Investigational Site Frankfurt, Hessen, Germany, 60596 GSK Investigational Site Neu isenburg, Hessen, Germany, 63263 GSK Investigational Site Dueren, Nordrhein-Westfalen, Germany, 52349 GSK Investigational Site Essen, Nordrhein-Westfalen, Germany, 45359 GSK Investigational Site Magdeburg, Sachsen-Anhalt, Germany, 39112 GSK Investigational Site Leipzig, Sachsen, Germany, 04207 GSK Investigational Site Leipzig, Sachsen, Germany, 04275 GSK Investigational Site Grosshansdorf, Schleswig-Holstein, Germany, 22927 GSK Investigational Site Berlin, Germany, 10787 GSK Investigational Site Berlin, Germany, 10789 Collapse << |
| NCT00896363 | Depressive Disorder | Phase 2 | Completed | - | Russian Federation ... more >> GSK Investigational Site Ekaterinburg, Russian Federation, 620030 GSK Investigational Site Kemerovo, Russian Federation, 650036 GSK Investigational Site Lipetsk Region, Russian Federation, 399083 GSK Investigational Site Moscow, Russian Federation, 119992 GSK Investigational Site Nizhny Novgorod, Russian Federation, 603107 GSK Investigational Site Saint Petersburg, Russian Federation, 190005 GSK Investigational Site Saint Petersburg, Russian Federation, 191180 GSK Investigational Site Saint-Petersburg, Russian Federation GSK Investigational Site Saratov, Russian Federation, 410060 GSK Investigational Site Smolensk, Russian Federation, 214 019 GSK Investigational Site St-Petersburg, Russian Federation, 197341 GSK Investigational Site St. Petersburg, Russian Federation, 190121 GSK Investigational Site St. Petersburg, Russian Federation, 194044 GSK Investigational Site St.Petersburg, Russian Federation, 193167 GSK Investigational Site Tomsk, Russian Federation, 634014 Collapse << |
| NCT00477334 | - | - | Completed | - | - |
| NCT01376323 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Anniston, Alabama, United States, 36207 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 GSK Investigational Site Miramar, Florida, United States, 33025 France GSK Investigational Site Nantes cedex 01, France, 44093 GSK Investigational Site Pierre-Bénite Cedex, France, 69495 GSK Investigational Site Rennes Cedex, France, 35046 GSK Investigational Site Rueil-Malmaison, France, 92502 Spain GSK Investigational Site Alicante, Spain, 03114 GSK Investigational Site Badalona, Spain, 08916 GSK Investigational Site Granada, Spain, 18004 GSK Investigational Site Madrid, Spain, 28046 United Kingdom GSK Investigational Site Edinburgh, Midlothian, United Kingdom, EH4 2XU GSK Investigational Site Cambridge, United Kingdom, CB2 0GG GSK Investigational Site Coventry, United Kingdom, CV2 2DX GSK Investigational Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT00896363 | - | - | Completed | - | - |
| NCT00451321 | Diabetes Mellitus, Type 1 | Phase 2 | Terminated | - | United States, Alabama ... more >> GSK Investigational Site Birmingham, Alabama, United States, 35294 United States, California GSK Investigational Site Walnut Creek, California, United States, 94598 United States, Colorado GSK Investigational Site Aurora, Colorado, United States, 80045 United States, District of Columbia GSK Investigational Site Washington, D.C., District of Columbia, United States, 20037 United States, Florida GSK Investigational Site Jacksonville, Florida, United States, 32207 GSK Investigational Site Pinellas Park, Florida, United States, 33781 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60637 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21201 United States, Massachusetts GSK Investigational Site Worcester, Massachusetts, United States, 1655 United States, Michigan GSK Investigational Site Kalamazoo, Michigan, United States, 49048 United States, Minnesota GSK Investigational Site Minneapolis, Minnesota, United States, 55455 United States, Mississippi GSK Investigational Site Gulfport, Mississippi, United States, 39501 United States, Nebraska GSK Investigational Site Omaha, Nebraska, United States, 68131 United States, Ohio GSK Investigational Site Mentor, Ohio, United States, 44060 United States, South Dakota GSK Investigational Site Rapid City, South Dakota, United States, 57701 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78229-4801 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M4G 3E8 Collapse << |
| NCT01462565 | Hypertension, Pulmonary | Phase 4 | Completed | - | United States, Florida ... more >> GSK Investigational Site Miami Beach, Florida, United States, 33140 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60637 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21205 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02118-2393 United States, Ohio GSK Investigational Site Columbus, Ohio, United States, 43221 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75390-8550 Canada GSK Investigational Site Quebec, Canada, G1V 4G5 Netherlands GSK Investigational Site Amsterdam, Netherlands, 1081 HV Collapse << |
| NCT00239356 | Schizophrenia | Phase 3 | Completed | - | Canada, Ontario ... more >> Local Institution Ottawa, Ontario, Canada Canada, Quebec Local Institution Sherbrooke, Quebec, Canada Croatia Local Institution Rijeka, Croatia Local Institution Zagreb, Croatia Czech Republic Local Institution Hradec Kralove, Czech Republic Local Institution Prague 6, Czech Republic France Local Institution Nantes Orvault, France Local Institution Rennes Cedex, France Local Institution Uzes, France Hungary Local Institution Budapest, Hungary Local Institution Gyor, Hungary Netherlands Local Institution Vught, Netherlands Poland Local Institution Krakow, Poland Local Institution Poznan, Poland Romania Local Institution Bucharest, Romania Russian Federation Local Institution St. Petersburg, Russian Federation South Africa Local Institution Westdene, Free State, South Africa Local Institution Johannesburg, Gauteng, South Africa Local Institution Cape Town, Western Cape, South Africa United Kingdom Local Institution Antrim, United Kingdom Collapse << |
| NCT01376323 | - | - | Completed | - | - |
| NCT01734993 | Rheumatoid Arthritis | Phase 3 | Completed | - | France ... more >> Bordeaux, France, 33076 Montpellier, France, 34295 Nantes, France, 44035 Paris, France, 75679 Strasbourg, France, 67098 Toulouse, France, 31059 Collapse << |
| NCT01734993 | - | - | Completed | - | - |
| NCT00239356 | - | - | Completed | - | - |
| NCT01404312 | Tuberculosis ... more >>HIV Infections Collapse << | Phase 3 | Completed | - | - |
| NCT00451321 | - | - | Terminated | - | - |
| NCT01462565 | - | - | Completed | - | - |
| NCT01673490 | - | - | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | - |
| NCT01404312 | - | - | Completed | - | - |
| NCT00833989 | Ischaemic Attack, Transient | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90095-6984 GSK Investigational Site Orange, California, United States, 92868-4280 United States, Colorado GSK Investigational Site Fort Collins, Colorado, United States, 80524 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48201 United States, Oregon GSK Investigational Site Portland, Oregon, United States, 97239 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M4N 3M5 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H3T 1E2 Germany GSK Investigational Site Ulm, Baden-Wuerttemberg, Germany, 89081 GSK Investigational Site Wiesbaden, Hessen, Germany, 65199 GSK Investigational Site Celle, Niedersachsen, Germany, 29223 GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Essen, Nordrhein-Westfalen, Germany, 45122 GSK Investigational Site Koeln, Nordrhein-Westfalen, Germany, 50937 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Leipzig, Sachsen, Germany, 04103 Collapse << |
| NCT01673490 | Prostatic Hyperplasia | Phase 4 | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | Vietnam ... more >> GSK Investigational Site Ho Chi Minh, Vietnam Collapse << |
| NCT02114203 | Phase 1 Sickle Cell | Phase 1 | Completed | - | United States, Illinois ... more >> University of Illinois Hospital and Health Sciences System Chicago, Illinois, United States, 60612-5836 University of Illinois at Chicago Clinical Research Center Chicago, Illinois, United States, 60612 University of Illinois Hospital and Health Sciences System Chicago, Illinois, United States, 60612 United States, Massachusetts Boston Medical Center E7E Boston, Massachusetts, United States, 02118 Boston Medical Center Boston, Massachusetts, United States, 02118 Boston University Medical Center Boston, Massachusetts, United States, 02118 United States, New York Interfaith Medical Center Brooklyn, New York, United States, 11213 Interfaith Medical Center Brooklyn, New York, United States, 11238 United States, North Carolina UNC Hospitals' Investigational Drug Service Pharmacy Chapel Hill, North Carolina, United States, 27514 UNC School of Medicine Clinical and Translational Research Center Chapel Hill, North Carolina, United States, 27599 United States, Virginia Investigational Drug Services Richmond, Virginia, United States, 23298 Virginia Commonwealth University Richmond, Virginia, United States, 23298 Belgium Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Italy Fondazione IRCCS Ca'Granda Ospedale Maggiore Policlinico Milano, Italy, 20122 A.O.O.R Villa Sofia - V. Cervello Palermo, Italy, 90146 Netherlands Centre for Human Drug Research Leiden, Netherlands, 2333 CL United Kingdom Royal Liverpool and Broadgreen University Hospital Trust Liverpool, Merseyside, United Kingdom, L7 8XP Guy's and St Thomas' NHS Foundation Trust London, United Kingdom, SE1 9RT King's College Hospital NHS Foundation Trust London, United Kingdom, SE5 9RS Imperial College Healthcare NHS Trust London, United Kingdom, W12 0HS Central Manchester University Hospitals NHS Foundation Trust Manchester, United Kingdom, M13 9WL Manchester Royal Infirmary Manchester, United Kingdom, M13 9WL Oxford University Hospitals NHS Trust Oxford, United Kingdom, OX3 7LE Collapse << |
| NCT02563899 | Hyperhidrosis | Phase 2 | Completed | - | United States, North Carolina ... more >> GSK Investigational Site Raleigh, North Carolina, United States, 27612 United States, Texas GSK Investigational Site Austin, Texas, United States, 78759 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H2K 4L5 Collapse << |
| NCT02114203 | - | - | Completed | - | - |
| NCT00833989 | - | - | Completed | - | - |
| NCT02563899 | - | - | Completed | - | - |
| NCT01844063 | Liver Failure | Phase 1 Phase 2 | Recruiting | January 2019 | China, Guangdong ... more >> Qi Zhang Recruiting Guangzhou, Guangdong, China, 510630 Principal Investigator: Qi Zhang, Doctor Collapse << |
| NCT00420199 | - | - | Completed | - | - |
| NCT01587924 | - | - | Completed | - | - |
| NCT00135356 | HIV-Associated Lipodystrophy S... more >>yndrome Collapse << | Phase 4 | Completed | - | - |
| NCT01587924 | Anaemia | Phase 2 | Completed | - | - |
| NCT03069989 | Idiopathic Pulmonary Fibrosis | Phase 1 | Active, not recruiting | May 23, 2019 | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, SW3 6HP GSK Investigational Site London, United Kingdom, WC1E 6JF GSK Investigational Site London, United Kingdom Collapse << |
| NCT01240915 | Colitis, Ulcerative | Phase 2 | Completed | - | - |
| NCT03136380 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Tokyo, Japan, 162-0053 Collapse << |
| NCT03447314 | Neoplasms | Phase 1 | Recruiting | February 14, 2020 | United States, Missouri ... more >> GSK Investigational Site Not yet recruiting Saint Louis, Missouri, United States, 63110 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Texas GSK Investigational Site Recruiting Dallas, Texas, United States, 75230 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Houston, Texas, United States, 77030 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Washington GSK Investigational Site Recruiting Tacoma, Washington, United States, 98405 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Netherlands GSK Investigational Site Not yet recruiting Amsterdam, Netherlands, 1066 CX Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Spain GSK Investigational Site Not yet recruiting Barcelona, Spain, 08035 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03287310 | Asthma | Phase 1 | Recruiting | September 24, 2019 | Germany ... more >> GSK Investigational Site Recruiting Hannover, Niedersachsen, Germany, 30625 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Berlin, Germany, 14050 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United Kingdom GSK Investigational Site Recruiting Harrow, Middlesex, United Kingdom, HA1 3UJ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Cambridge, United Kingdom, CB2 2GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting London, United Kingdom, NW10 7EW Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Manchester, United Kingdom, M23 9QZ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00135356 | - | - | Completed | - | - |
| NCT01240915 | - | - | Completed | - | - |
| NCT02229227 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00420199 | Active Rheumatoid Arthritis | Phase 3 | Completed | - | Belgium ... more >> Local Institution Bruxelles, Belgium, 1200 Local Institution Yvoir, Belgium, 5530 Germany Local Institution Berlin, Germany, 14059 Local Institution Berlin, Germany, BE-10117 Netherlands Local Institution Amsterdam, Netherlands, 1105 AZ Spain Local Institution Barcelona, Spain, 08036 Local Institution Barcelona, Spain, 08907 Sweden Local Institution Stockholm, Sweden, 171 76 United Kingdom Local Institution London, Greater London, United Kingdom, SE19RT Local Institution Leeds, North Yorkshire, United Kingdom, LS7 4SA Collapse << |
| NCT02229227 | - | - | Completed | - | - |
| NCT02798978 | Cancer | Phase 1 | Completed | - | Germany ... more >> GSK Investigational Site Berlin, Germany, 14050 Collapse << |
| NCT03590613 | Autoimmune Diseases | Phase 1 | Active, not recruiting | September 21, 2018 | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 812-0025 Collapse << |
| NCT01376167 | Malaria, Vivax | Phase 2 | Completed | - | Bangladesh ... more >> GSK Investigational Site Bandarban, Bangladesh Brazil GSK Investigational Site Manaus, Amazonas, Brazil, 69040-000 GSK Investigational Site Porto Velho, Rondônia, Brazil, 76812-329 Cambodia GSK Investigational Site Oddar Meancheay Province, Cambodia Ethiopia GSK Investigational Site Gondar, Ethiopia GSK Investigational Site Jimma, Ethiopia India GSK Investigational Site Bikaner, India GSK Investigational Site Chennai, India, 600016 GSK Investigational Site Lucknow, India, 226003 GSK Investigational Site Secunderabad, India, 500 003 Peru GSK Investigational Site Iquitos, Loreto, Peru, Iqui 01 Philippines GSK Investigational Site Rio Tuba, Bataraza, Philippines, 5306 Thailand GSK Investigational Site Bangkok, Thailand, 10400 GSK Investigational Site Tak, Thailand, 63110 Collapse << |
| NCT02653339 | Rhinitis, Allergic, Perennial | Not Applicable | Unknown | August 2018 | China, Beijing ... more >> Peking Union Medical College Hospital traditional Chinese medicine department Not yet recruiting Beijing, Beijing, China, 100730 Contact: Xiaoyang Zhang, Doctor (+86)13910769068 zhangxymz@163.com Collapse << |
| NCT00371150 | Hepatitis B Infection | Phase 4 | Completed | - | - |
| NCT01376167 | - | - | Completed | - | - |
| NCT00371150 | - | - | Completed | - | - |
| NCT00383149 | Metastatic Pancreatic Cancer | Phase 2 | Completed | - | United States, District of Col... more >>umbia Georgetn Univ Lombardi Can Ctr Washington, District of Columbia, United States, 20007 United States, Florida Mayo Clinic Jacksonville Jacksonville, Florida, United States, 32224 University Of Miami Miami, Florida, United States, 33136 United States, Michigan University Of Michigan Ann Arbor, Michigan, United States, 48109 Wayne State University Detroit, Michigan, United States, 48201 United States, Missouri Ellis Fischel Cancer Center Columbia, Missouri, United States, 65203 United States, South Carolina Cancer Centers Of The Carolinas Greenville, South Carolina, United States, 29615 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 United States, West Virginia West Virginia University Morgantown, West Virginia, United States, 26506 Collapse << |
| NCT01920568 | Fractures, Bone | Phase 3 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China, 510060 GSK Investigational Site Guangzhou, Guangdong, China, 510120 GSK Investigational Site Guangzhou, Guangdong, China, 510515 China, Hubei GSK Investigational Site Wuhan, Hubei, China, 430030 China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210002 GSK Investigational Site Nanjing, Jiangsu, China, 210009 China, Jilin GSK Investigational Site Changchun, Jilin, China, 130012 China, Sichuan GSK Investigational Site Chengdu, Sichuan, China, 610041 China, Zhejiang GSK Investigational Site Hangzhou, Zhejiang, China, 310003 China GSK Investigational Site Beijing, China, 100021 GSK Investigational Site Beijing, China, 100034 GSK Investigational Site Beijing, China, 100036 GSK Investigational Site Beijing, China, 100071 GSK Investigational Site Fuzhou, China, 350001 GSK Investigational Site Hangzhou, China, 310016 GSK Investigational Site Harbin, China GSK Investigational Site Shanghai, China, 200025 GSK Investigational Site Shanghai, China, 200032 GSK Investigational Site Shanghai, China, 200070 GSK Investigational Site Shanghai, China, 200080 GSK Investigational Site Shanghai, China, 200233 GSK Investigational Site Tianjin, China, 300060 Singapore GSK Investigational Site Singapore, Singapore, 119074 Taiwan GSK Investigational Site Taichung, Taiwan, 404 GSK Investigational Site Taoyuan, Taiwan, 333 Collapse << |
| NCT01945775 | - | - | Active, not recruiting | - | - |
| NCT00383149 | - | - | Completed | - | - |
| NCT01945775 | Breast Neoplasms ... more >> BRCA 1 Gene Mutation BRCA 2 Gene Mutation Collapse << | Phase 3 | Active, not recruiting | September 22, 2019 | - |
| NCT01920568 | - | - | Completed | - | - |
| NCT01936103 | Acute Anterior Myocardial Infa... more >>rction Collapse << | Phase 4 | Unknown | July 2014 | China, Liaoning ... more >> Shenyang Northen Hospital Recruiting Shenyang, Liaoning, China, 110016 Contact: Wang Geng, Dr 13309886393 Collapse << |
| NCT01299610 | Dermatitis, Atopic | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Berlin, Germany, 10117 Collapse << |
| NCT01990859 | Melanoma | Phase 2 | Completed | - | Japan ... more >> Local Institution Fukuoka-shi, Fukuoka, Japan, 8128582 Local Institution Kumamoto-shi, Kumamoto, Japan, 8608556 Local Institution Matsumoto-shi, Nagano, Japan, 3908621 Local Institution Sunto-gun, Shizuoka, Japan, 4118777 Local Institution Chuo-ku, Tokyo, Japan, 1040045 Local Institution Chuo-shi, Yamanashi, Japan, 4093898 Collapse << |
| NCT01990859 | - | - | Completed | - | - |
| NCT01299610 | - | - | Completed | - | - |
| NCT03367195 | Gastroesophageal Reflux Diseas... more >>e (GERD) Collapse << | Phase 3 | Active, not recruiting | December 2018 | Indonesia ... more >> Division of Gastroenterohepatology Department of Internal Medicine Faculty of Medicine, University of Padjajaran Dr. Hasan Sadikin Hospital Bandung, Indonesia Division of Gastroenterology Department of Internal Medicine Faculty of Medicine, University of Indonesia Dr. Cipto Mangunkusumo National General Hospital Jakarta, Indonesia, 10430 Division of Gastroenterohepatology Department of Internal Medicine Faculty of Medicine, University of Diponegoro Dr. Kariadi Hospital Semarang, Indonesia Division of Gastroenterohepatology Department of Internal Medicine Faculty of Medicine, University of Airlangga Dr. Soetomo Hospital Surabaya, Indonesia Division of Gastroenterohepatology Department of Internal Medicine Faculty of Medicine, University of Gadjah Mada Dr. Sardjito Hospital Yogyakarta, Indonesia Collapse << |
| NCT03557281 | Tuberculosis | Phase 2 | Not yet recruiting | September 13, 2020 | - |
| NCT02283047 | Hypertension ... more >>Overweight Sedentary Collapse << | Not Applicable | Completed | - | Spain ... more >> Faculty of Physical Activity and Sport Sciences Vitoria-gasteiz, Araba/alava, Spain, 01007 Collapse << |
| NCT00077610 | - | - | Completed | - | - |
| NCT03479892 | Metabolism and Nutrition Disor... more >>der Obesity Collapse << | Phase 1 | Recruiting | May 4, 2019 | United States, Texas ... more >> Novo Nordisk Investigational Site Recruiting San Antonio, Texas, United States, 78209 Collapse << |
| NCT00077610 | Anemia | Phase 3 | Completed | - | - |
| NCT02911701 | Pre-Eclampsia ... more >> HELLP Syndrome Eclampsia Collapse << | Phase 4 | Active, not recruiting | June 2018 | United States, New Mexico ... more >> University of New Mexico Health Sciences Center Albuquerque, New Mexico, United States Collapse << |
| NCT03520842 | KRAS Gene Mutation ... more >> Metastatic Malignant Neoplasm in the Brain Recurrent Non-Small Cell Lung Carcinoma Stage IV Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 2 | Recruiting | June 1, 2020 | United States, California ... more >> Stanford University School of Medicine Recruiting Palo Alto, California, United States, 94304 Contact: Vivian Tsang 650-723-0186 tsangv@stanford.edu Principal Investigator: Sukhmani Padda Collapse << |
| NCT02262169 | Non-Bleeding Peptic Ulcers | Phase 3 | Recruiting | March 2019 | Indonesia ... more >> Division of Gastroenterohepatology Department of Internal Medicine Faculty of Medicine, University of Udayana Sanglah General Hospital Recruiting Denpasar, Bali, Indonesia, 80114 Contact: IDN Wibawa Prof. DR. Dr., SpPD-KGEH agusbobwibawa@yahoo.com Principal Investigator: IDN Wibawa Prof. DR. Dr., SpPD-KGEH Sub-Investigator: Nyoman Purwadi dr., Sp.PD-KGEH Sub-Investigator: I Gusti Agung Suryadarma dr., Sp.PD-KGEH Sub-Investigator: I Ketut Mariadi dr., Sp.PD Sub-Investigator: Gde Somayana dr., Sp.PD Sub-Investigator: Putu Prathiwi Primadharsini dr., M. Biomed, Sp.PD Sub-Investigator: Anak Agung Ayu Ngurah Susraini dr., Sp.PA (K) Sub-Investigator: Herman Saputra dr., Sp.PA (K) Collapse << |
| NCT02505126 | Alcoholic Intoxication, Chroni... more >>c Collapse << | Not Applicable | Unknown | - | France ... more >> CHU de DIJON Recruiting Dijon, France, 21079 Contact: Benoît TROJAK benoît.trojak@chu-dijon.fr Collapse << |
| NCT02518672 | Cystic Fibrosis | Phase 2 | Terminated | - | Canada, Quebec ... more >> Centre Hospitalier Universitaire de Sherbrooke Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT00306202 | Leukemia | Phase 1 | Active, not recruiting | October 4, 2022 | Austria ... more >> Local Institution Vienna, Austria, 1090 France Local Institution Nantes, France, 44093 Local Institution Paris Cedex 10, France, 75475 Local Institution Paris Cedex 12, France, 75571 Germany Local Institution Berlin, Germany, 13353 Local Institution Frankfurt, Germany, 60590 Local Institution Hannover, Germany, 30625 Italy Local Institution Monza (mi), Italy, 20052 Netherlands Local Institution Rotterdam, Netherlands, 3015 GJ United Kingdom Local Institution Manchester, Greater Manchester, United Kingdom, M27 4HA Local Institution Bristol, Somerset, United Kingdom, BS2 8BJ Local Institution Sutton, Surrey, United Kingdom, SM2 5PT Local Institution Birmingham, West Midlands, United Kingdom, B4 6NH Collapse << |
| NCT00254293 | Rheumatoid Arthritis | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Orlando Clinical Research Center Orlando, Florida, United States, 32806 United States, Georgia Winship Cancer Institute, Emory University Atlanta, Georgia, United States, 30322 United States, Louisiana New Orleans Center For Clinical Research New Orleans, Louisiana, United States, 70119 United States, Minnesota Davita Clinical Research Minneapolis, Minnesota, United States, 55404 United States, North Carolina The Arthritis Clinic & Carolina Bone & Joint Charlotte, North Carolina, United States, 28210 Collapse << |
| NCT03335488 | Urea Cycle Disorder | Phase 4 | Recruiting | March 31, 2020 | United States, New York ... more >> Mount Sinai School of Medicine Recruiting New York, New York, United States, 10029 Contact: Dr. George Diaz, M.D., PhD. 212-241-0858 George.Diaz@mssm.edu Contact: Luca Fierro 212 659 1477 luca.fierro@mssm.edu Principal Investigator: Dr. George Diaz, M.D., PhD. United States, Ohio University Hospitals Case Medical Center Recruiting Cleveland, Ohio, United States, 44106-6005 Contact: Dr. Jirair Bedoyan, MD 216-844-7124 Jirair.Bedoyan@UHhospitals.org Contact: Audrey Lynn 216 844 7124 audrey.lynn@UHhospitals.org United States, Pennsylvania Children's Hospital of Pittsburgh of UPMC Recruiting Pittsburgh, Pennsylvania, United States, 15224 Contact: Dr. Gerard Vockley, MD, PhD 412-692-7530 Jennifer.baker@chp.edu Contact: Jennifer Baker 412-692-7530 Jennifer.baker@chp.edu Principal Investigator: Dr. Gerard Vockley, MD, PhD United States, Utah University of Utah Recruiting Salt Lake City, Utah, United States, 84132 Contact: Dr. Nicola Longo, M.D., PhD. 801-587-3605 Nicola.Longo@hsc.utah.edu Contact: Carrie Bailey 801 587 3605 Carrie.Bailey@hsc.utah.edu Principal Investigator: Dr. Nicola Longo, M.D., PhD. Belgium Cliniques Universitaires Saint-Luc Recruiting Brussels, Belgium, 1200 Contact: Prof. Etienne Sokal, MD 32 2 764 13 87 etienne.sokal@uclouvain.be Contact: Julia Versavau 32 2 764 19 33 julia.versavau@uclouvain.be Principal Investigator: Prof. Etienne Sokal, MD Spain Hospital Materno-Infantil (HRU Carlos Haya) Recruiting Málaga, Andalucia, Spain, 29006 Contact: Dr. Javier Blasco-Alonso, MD 34 951 29 21 91 javierblascoalonso@yahoo.es Contact: Almudena Frias 34 951 29 21 91 Principal Investigator: Dr. Javier Blasco-Alonso Hospital Clinico Universitario de Santiago Recruiting Santiago de Compostela, Galicia, Spain, 15706 Contact: Dr. Maria Luz Couce-Pico, MD 34 981 950 162 maria.luz.couce.pico@sergas.es Contact: Maria Teresa Oreiro-Garcia 34 981 955 627 maria.teresa.oreiro.garcia@sergas.es Principal Investigator: Dr. Maria Luz Couce-Pico, MD Hospital Universitario de Cruces Recruiting Barakaldo, Vizcaya, Spain, 48903 Contact: Dr. Luis Jose Aldamiz-Echevar Azuara, MD 34 94 600 6327 LUISJOSE.ALDAMIZ-ECHEVARAZUARA@osakidetza.net Contact: Fernando Olatz 34 94 600 6327 fernando.andradelodeiro@osakidetza.eus Principal Investigator: Dr. Luis Jose Aldamiz-Echevar Azuara, MD Switzerland Universitätsspital, Inselspital Bern Recruiting Bern, Switzerland, 3010 Contact: Matthias Gautchshi,, MD 41 (0)31 632 01 47 Matthias.Gautschi@insel.ch Contact: Alexandra Nuoffer +41 (0)31 632 51 37 alexandra.nuoffer@insel.ch Principal Investigator: Dr. Matthias Gautchshi, MD University Children's Hospital Recruiting Zurich, Switzerland, 75, 8032 Contact: Prof Johannes Häberle, MD 41- 44-266-7111 johannes.haeberle@kispi.uzh.ch Contact: Ms. Doris Anzengruber 41- 44-266-7722 Doris.Anzengruber@kispi.uzh.ch Principal Investigator: Prof Johannes Häberle, MD Collapse << |
| NCT02058420 | Osteoporosis | Phase 1 Phase 2 | Active, not recruiting | December 2018 | United States, Texas ... more >> Texas Tech University Health Sciences Center Lubbock, Texas, United States, 79430 Collapse << |
| NCT03372603 | Cough | Phase 2 | Active, not recruiting | April 22, 2019 | United Kingdom ... more >> GSK Investigational Site Manchester, Lancashire, United Kingdom, M23 9LT GSK Investigational Site Belfast, United Kingdom, BT9 7BL GSK Investigational Site Cottingham, United Kingdom, HU16 5JQ GSK Investigational Site North Shields, United Kingdom, NE29 8NH Collapse << |
| NCT00306202 | - | - | Active, not recruiting | - | - |
| NCT02302456 | Immediate Postpartum Hemorrhag... more >>e Collapse << | Phase 3 | Completed | - | France ... more >> Angers University Hospital Angers, France, 49933 Collapse << |
| NCT00323882 | Prostate Cancer ... more >> Neoplasm Metastasis Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> The Angeles Clinic and Research Institute Los Angeles, California, United States, 11818 The Angeles Clinic and Research Institute Los Angeles, California, United States, 90025 UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 United States, Connecticut Yale University School of Medicine New Haven, Connecticut, United States, 06520 United States, Michigan Josephine Ford Cancer Center-Downriver Brownstown, Michigan, United States, 48183 Henry Ford Medical Center-Fairlane Dearborn, Michigan, United States, 48126 Henry Ford Hospital Detroit, Michigan, United States, 48202-2689 Henry Ford Medical Center-West Bloomfield West Bloomfield, Michigan, United States, 48322 United States, Missouri Washington University School of Medicine St. Louis, Missouri, United States, 63110 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, North Carolina Carolina BioOncology Institute Huntersville, North Carolina, United States, 28078 United States, Oregon Oregon Health and Science University Portland, Oregon, United States, 97239-3098 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT00323882 | - | - | Completed | - | - |
| NCT00254293 | - | - | Completed | - | - |
| NCT03541005 | Overweight and Obesity | Phase 3 | Recruiting | December 1, 2019 | Cuba ... more >> National Institute of Endocrinology Recruiting Havana, La Habana, Cuba, 10400 Contact: Eduardo Cabrera Rode, Dr. diabetes@infomed.sld.cu Sub-Investigator: Ileana Cubas Dueñas, Dr. Sub-Investigator: Ana I. Conesa González, Dr. Sub-Investigator: José Hernández, Dr. Principal Investigator: Judith Parlá Sardiñas, Dr. Sub-Investigator: Silvia Marín Julia, Dr. Sub-Investigator: Yuri Arnold Domínguez, Dr. Sub-Investigator: Antonio D. Reyes Rodríguez, PD Sub-Investigator: Aimee Álvarez Álvarez, MD Sub-Investigator: Ragmila Echevarría Valdés, BSc Sub-Investigator: Janet Rodríguez Acosta, BSc Sub-Investigator: Liudmila Jorge Espinosa, CPhT Sub-Investigator: Kenia Enrique Lugo, BNurs Sub-Investigator: Masiel Peña Roque, BNurs Sub-Investigator: Rocío Montes de Oca Somoano, Dr. Collapse << |
| NCT03483454 | Pediatric Obesity | Not Applicable | Completed | - | United States, Pennsylvania ... more >> Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT02925546 | Heart Failure, Congestive | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT00087607 | Hepatitis C, Chronic | Phase 4 | Completed | - | - |
| NCT03729791 | Schizophrenia | Not Applicable | Not yet recruiting | August 2019 | Korea, Republic of ... more >> Seoul National University Hospital Not yet recruiting Seoul, Korea, Republic of Contact: Tae Young Lee, MD 82236687663 leetaey@gmail.com Collapse << |
| NCT00087607 | - | - | Completed | - | - |
| NCT01526655 | Endometrial Cancer | Not Applicable | Unknown | August 2016 | Sweden ... more >> Department of Obstetrics and Gynecology, Ryhov Central Hospital Not yet recruiting Jönköping, Jönköpings län, Sweden, 551 85 Contact: Laila Falknäs, MD +4636321000 Laila.Falknas@lj.se Contact: Johan Skoglund, MD +4636321000 Johan.Skoglund@lj.se Principal Investigator: Laila Falknäs, MD Sub-Investigator: Johan Skoglund, MD Department of Obstetrics and Gynecology, University Hospital Recruiting Linköping, Östergötland, Sweden, 58185 Contact: Preben Kjölhede, MD, PhD +46101030000 ext 3187 Preben.Kjolhede@regionostergotland.se Contact: Evelyn Lundin, MD +46101030000 Evelyn.Lundin@regionostergotland.se Principal Investigator: Preben Kjölhede, MD, PhD Sub-Investigator: Evelyn Lundin, MD Sub-Investigator: Shefqet Halili, MD Sub-Investigator: Peter Lukas, MD Sub-Investigator: Gulnara Kassymova, MD Sub-Investigator: Ninnie Borendal Wodlin, MD, PhD Collapse << |
| NCT00611455 | Arthritis, Rheumatoid | Phase 3 | Terminated(Ofatumumab IV trial... more >>s in RA were prematurely terminated because GSK refocused clinical development of autoimmune indications on the subcutaneous delivery.) Collapse << | - | - |
| NCT03479086 | Alcohol Dependence | Phase 3 | Recruiting | November 1, 2020 | Australia, New South Wales ... more >> Drug Health Services, Royal Prince Alfred Hospital Recruiting Sydney, New South Wales, Australia, 2050 Contact: Kirsten C Morley, PhD +61295153636 kirsten.morley@sydney.edu.au Contact: Central Intake 95157611 sydneyalcoholtreatmentgroup@gmail.com Principal Investigator: Paul Haber, MBBS Collapse << |
| NCT00611455 | - | - | Terminated(Ofatumumab IV trial... more >>s in RA were prematurely terminated because GSK refocused clinical development of autoimmune indications on the subcutaneous delivery.) Collapse << | - | - |
| NCT02780739 | Cognitive Ability, General | Not Applicable | Completed | - | United States, Illinois ... more >> University of Illinois at Urbana-Champaign Urbana, Illinois, United States, 61801 Collapse << |
| NCT01890330 | Metabolic Syndrome | Not Applicable | Completed | - | Canada, Manitoba ... more >> St. Boniface General Hospital - I.H. Asper Clinical Research Institute Winnipeg, Manitoba, Canada, R2H 2A6 Collapse << |
| NCT03714685 | Healthy Males | Phase 1 | Not yet recruiting | April 2019 | - |
| NCT01810705 | Acute Myeloid Leukemia | Phase 2 | Completed | - | France ... more >> Hôpital l'Archet 1 Nice, Alpes Maritimes, France, 06200 Institut Paoli Calmettes Marseille, Bouche Du Rhone, France, 13000 Hôpital JEAN MINJOZ Besancon, Doubs, France, 25000 Hopital Morvan Brest, Finistere, France, 29200 Hôpital Haut-Lévèque Pessac, Gironde, France, 33600 CHRU de Nîmes Nimes, Guard, France, 30000 Hopital de Hautepierre Strasbourg, Haut Rhin, France, 67000 Hopital De Purpan CHU Toulouse Toulouse, Haute Garonne, France, 31500 Hopital Région d'Annecy Pringy, Haute Savoie, France, 74000 Hôpital Saint Eloi Montpellier, Hérault, France, 34295 Hôtel Dieu - CHU de NANTES Nantes, Loire Atlantique, France, 44200 Institut de Cancérologie de la Loire Saint-priest-en-jarez, Loire, France, 42270 Chu D'Angers Angers, Maine Et Loire, France, 49000 Hopital de Brabois Vandoeuvre Les Nancy, Meurthe Et Moselle, France, 54500 Hôpital Claude-Huriez Lille, Nord, France, 59800 CHU Estaing Clermont-ferrand, Puy De Dome, France, 63000 hopital de Perpignan Perpignan, Pyrénées Orientales, France, 66100 Centre Léon Bérard Lyon, Rhone Alpes, France, 69008 Centre hospitalier Lyon Sud Pierre Benite, Rhone Alpes, France, 69310 Centre Henri Becquerel Rouen, Seine Maritime, France, 76100 Groupe Hospitalier Sud Amiens, Somme, France, 80090 Collapse << |
| NCT00859898 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00603525 | - | - | Terminated(Ofatumumab IV trial... more >>s in RA were prematurely terminated because GSK refocused clinical development of autoimmune indications on the subcutaneous delivery.) Collapse << | - | - |
| NCT00859898 | - | - | Completed | - | - |
| NCT00603525 | Arthritis, Rheumatoid | Phase 3 | Terminated(Ofatumumab IV trial... more >>s in RA were prematurely terminated because GSK refocused clinical development of autoimmune indications on the subcutaneous delivery.) Collapse << | - | - |
| NCT00950859 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 2 | Completed | - | - |
| NCT00601172 | - | - | Completed | - | - |
| NCT00736879 | - | - | Completed | - | - |
| NCT00601172 | Nausea and Vomiting, Chemother... more >>apy-Induced Collapse << | Phase 3 | Completed | - | - |
| NCT01677325 | NAFLD( Non-alcoholic Fatty Liv... more >>er Disease ) Collapse << | Phase 1 | Completed | - | - |
| NCT00392925 | Overweight Ob... more >>esity Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Research Site Deland, Florida, United States United States, Illinois Research Site Chicago, Illinois, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Kansas Research Site Overland Park, Kansas, United States United States, Louisiana Research Site Baton Rouge, Louisiana, United States United States, Michigan Research Site Detroit, Michigan, United States United States, Minnesota Research Site Edina, Minnesota, United States United States, Montana Research Site Butte, Montana, United States United States, Ohio Research Site Cincinnati, Ohio, United States United States, Oregon Research Site Portland, Oregon, United States United States, Utah Research Site Salt Lake City, Utah, United States United States, Washington Research Site Olympia, Washington, United States Collapse << |
| NCT00736879 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00364793 | HIV Infections | Phase 1 Phase 2 | Completed | - | Argentina ... more >> Local Institution Buenos Aires, Argentina, 1425 Local Institution Capital Federal, Argentina, 1425 Colombia Local Institution Cali, Colombia Mexico Local Institution Df, Distrito Federal, Mexico, 06720 Local Institution Guadalajara, Jalisco, Mexico, 44280 Local Institution Guadalajara, Jalisco, Mexico, 44520 Local Institution Morelia, Michioacan, Mexico, 58000 Local Institution Monterrey, Nuevo Leon, Mexico, 64460 Local Institution Colima, Mexico, 28019 Local Institution Puebla, Mexico, 72000 Local Institution San Luis Potosi, Mexico, 78240 Panama Local Institution Ciudad De Panama, Panama, 0816-00383 South Africa Local Institution Bloemfontein, Free State, South Africa, 9301 Local Institution Westdene, Gauteng, South Africa, 2092 Local Institution Cape Town, Western Cape, South Africa, 7505 Thailand Local Institution Bangkok, Thailand, 10330 Local Institution Bangkok, Thailand, 10700 Collapse << |
| NCT00950859 | - | - | Completed | - | - |
| NCT00392925 | - | - | Completed | - | - |
| NCT02060162 | - | - | Active, not recruiting | June 2021 | Zambia ... more >> Centre for Infectious Disease Research in Zambia Lusaka, Zambia Collapse << |
| NCT03340935 | Malignant Neoplasm ... more >> Cancer Collapse << | Not Applicable | Recruiting | June 30, 2018 | Italy ... more >> Fondazione IRCCS Istituto Nazionale dei Tumori Recruiting Milan, Italy, 20133 Contact: Claudio Vernieri, M.D., Ph.D. +39 02 23903066 claudio.vernieri@istitutotumori.mi.it Collapse << |
| NCT03709680 | Solid Tumors ... more >>Ewing Sarcoma Rhabdoid Tumor Rhabdomyosarcoma Neuroblastoma Medulloblastoma Collapse << | Phase 1 | Not yet recruiting | September 27, 2023 | - |
| NCT03355664 | Malaria, Falciparum | Phase 3 | Recruiting | March 31, 2020 | Cambodia ... more >> Siem Pang Health Center Recruiting Siem Pang, Stung Treng, Cambodia, 1803 Contact: Rupam Tripura, M.D. (66) 2 203 6333 Rupam@tropmedres.ac, Contact: James Callery (66) 2 203 6333 James.C@tropmedres.ac Pailin Referral Hospital Not yet recruiting Pailin, Cambodia, 2401 Contact: Rupam. , Tripura, M.D. (66) 2 203 6333 Rupam@tropmedres.ac Contact: Thomas J Peto, PhD. (66) 2 203 6333 Tom@tropmedres.ac Vietnam Hospital for Tropical Diseases of Khanh Hoa, Not yet recruiting Van Ninh, Khanh Hoa, Vietnam Phuoc Long Hospital Not yet recruiting Phuoc Long, Phuoc, Vietnam Contact: Rob van der Pluijm, M.D. (66) 2 203 6333 Rob@tropmedres.ac Collapse << |
| NCT00089973 | Neoplasms, Breast | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Jacksonville, Florida, United States, 32224 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599 United States, Pennsylvania GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 Belgium GSK Investigational Site Bruxelles, Belgium, 1000 Malaysia GSK Investigational Site Bandar Tun Razak, Cheras, Malaysia, 56000 GSK Investigational Site Bandar Tun Razak, Cheras, Malaysia, 59100 Singapore GSK Investigational Site Singapore, Singapore, 119074 GSK Investigational Site Singapore, Singapore, 169610 United Kingdom GSK Investigational Site Southampton, Hampshire, United Kingdom, SO16 6YD GSK Investigational Site Manchester, Lancashire, United Kingdom, M20 4BX GSK Investigational Site Newcastle Upon Tyne, Northumberland, United Kingdom, NE4 6BE GSK Investigational Site London, United Kingdom, W12 0NN Collapse << |
| NCT00364793 | - | - | Completed | - | - |
| NCT00089973 | - | - | Completed | - | - |
| NCT02652585 | Alcoholism | Phase 3 | Completed | - | Germany ... more >> Klinik und Poliklinik für Psychiatrie und Psychotherapie Dresden, Germany, 01307 Collapse << |
| NCT02451371 | Schizophrenia | Phase 1 Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00123487 | Myeloid Leukemia, Chronic, Acc... more >>elerated Phase Leukemia, Lymphoblastic, Acute, Philadelphia-Positive Collapse << | Phase 3 | Completed | - | - |
| NCT03649412 | Autoimmune Diseases | Phase 1 | Recruiting | April 24, 2019 | United Kingdom ... more >> GSK Investigational Site Recruiting Nottingham, United Kingdom, NG11 6JS Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00123487 | - | - | Completed | - | - |
| NCT01720719 | Fatty Liver D... more >>yslipidemias Diabetes Mellitus Collapse << | Phase 4 | Unknown | December 2016 | China, Shanghai ... more >> Department of Endocrinology and Metabolism, Zhongshan Hospital, Fudan University Recruiting Shanghai, Shanghai, China, 200032 Contact: Xin Gao, doctor 862164041990 ext 8021 gao.xin@zs-hospital.sh.cn Contact: Hongmei Yan, doctor 13761666976 yan.hongmei@zs-hospital.sh.cn Principal Investigator: Xin Gao, doctor Sub-Investigator: hongmei Yan, doctor Sub-Investigator: Mingfeng Xia, doctor Collapse << |
| NCT00423891 | Hepatitis B, Chronic | Phase 1 Phase 2 | Completed | - | - |
| NCT03452475 | Plasmodium Falciparum | Phase 3 | Not yet recruiting | December 1, 2020 | - |
| NCT03613571 | Amyotrophic Lateral Sclerosis | Phase 2 | Enrolling by invitation | June 1, 2019 | Sweden ... more >> Sahlgrenska University Hospital Gothenburg, Sweden Collapse << |
| NCT00569803 | Transplantation | Phase 1 | Completed | - | United States, Texas ... more >> Ppd Development Austin, Texas, United States, 78744 Collapse << |
| NCT01508936 | Eosinophilic Asthma | Phase 3 | Completed | - | - |
| NCT00423891 | - | - | Completed | - | - |
| NCT01227928 | Neoplasms, Ovarian | Phase 2 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210009 China, Liaoning GSK Investigational Site Shenyang, Liaoning, China, 110022 China, Shandong GSK Investigational Site Jinan, Shandong, China, 250012 China, Sichuan GSK Investigational Site Chengdu, Sichuan, China, 610041 China, Zhejiang GSK Investigational Site Hangzhou, Zhejiang, China, 310006 GSK Investigational Site Hangzhou, Zhejiang, China, 310022 China GSK Investigational Site Beijing, China, 100021 GSK Investigational Site Beijing, China, 100044 GSK Investigational Site Beijing, China, 100853 GSK Investigational Site Shanghai, China, 200032 Hong Kong GSK Investigational Site Hong Kong, Hong Kong Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 135-710 Taiwan GSK Investigational Site Taipei, Taiwan, 104 GSK Investigational Site Taipei, Taiwan, 112 Collapse << |
| NCT01227928 | - | - | Completed | - | - |
| NCT00569803 | - | - | Completed | - | - |
| NCT01285323 | Eosinophilic Asthma | Phase 3 | Completed | - | - |
| NCT03584529 | Gynaecological Disease ... more >> Vitamin D Deficiency Collapse << | Not Applicable | Not yet recruiting | July 1, 2021 | China, Zhejiang ... more >> The Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University Not yet recruiting Wenzhou, Zhejiang, China, 325000 Contact: TING LI feclinicalresearch@163.com Collapse << |
| NCT03181165 | Diet Modification | Not Applicable | Recruiting | July 1, 2020 | Canada, British Columbia ... more >> University of British Columbia Recruiting Kelowna, British Columbia, Canada, V1V1V7 Contact: Jonathan Little, PhD 2508079876 jonathan.little@ubc.ca Contact: Cody Durrer, MSc 2508079122 cdurrer@live.com Collapse << |
| NCT01285323 | - | - | Completed | - | - |
| NCT00424255 | Neoplasms, Head and Neck | Phase 3 | Completed | - | - |
| NCT01287039 | Eosinophilic Asthma | Phase 3 | Completed | - | - |
| NCT01662999 | - | - | Completed | - | - |
| NCT01508936 | - | - | Completed | - | - |
| NCT01774721 | Non-small Cell Lung Cancer Wit... more >>h EGFR-Activating Mutations Collapse << | Phase 3 | Active, not recruiting | March 29, 2019 | - |
| NCT01662999 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Icon Clinical Pharmacology Omaha, Nebraska, United States, 68154 Collapse << |
| NCT02258126 | Non-alcoholic Fatty Liver Dise... more >>ase Obesity Metabolic Syndrome Collapse << | Not Applicable | Completed | - | Spain ... more >> Pediatric Endocrinology Unit of the University Hospital of Araba (HUA) Vitoria-Gasteiz, Araba, Spain, 01009 Collapse << |
| NCT01287039 | - | - | Completed | - | - |
| NCT01774721 | - | - | Active, not recruiting | - | - |
| NCT03124641 | Hepatocellular Injury ... more >> Liver Transplant Collapse << | Not Applicable | Recruiting | June 2019 | Belgium ... more >> Ghent University Hospital and Medical School Not yet recruiting Ghent, Belgium Contact: Xavier Rogiers, Professor Xavier.Rogiers@uzgent.be Czechia Department of Transplantation Surgery, Institute for Clinical and Experimental Medicine Recruiting Praha, Czechia Contact: Jiří Froněk, Professor jifr@medicon.cz Germany Department of Surgery and Transplantation, University Hospital RWTH Aachen Recruiting Aachen, Germany, 52074 Contact: Georg Lurje, M.D. +492418035048 glurje@ukaachen.de Romania Fundeni Clinical Institute Center of General Surgery & Liver Transplantation Not yet recruiting Bucharest, Romania Contact: Irinel Popescu, Professor irinel.popescu220@gmail.com Contact: Florin Botea, MD boteaflorin@yahoo.com Collapse << |
| NCT01584648 | Melanoma | Phase 3 | Active, not recruiting | October 15, 2018 | - |
| NCT00261846 | Chronic Myeloid Leukemia | Phase 2 | Completed | - | - |
| NCT00726323 | - | - | Completed | - | - |
| NCT00424255 | - | - | Completed | - | - |
| NCT03077048 | Renal Insufficiency, Chronic | Phase 1 Phase 2 | Completed | - | Czechia ... more >> Thomayer Hospital Clinical - Pharmacology Unit (CPU) Prague, Czechia, 140 59 Collapse << |
| NCT01195662 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01584648 | - | - | Active, not recruiting | - | - |
| NCT01079806 | Chronic Hepatitis B Virus, Ped... more >>iatric Collapse << | Phase 3 | Completed | - | - |
| NCT00726323 | Carcinoma, Renal Cell | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Greenbrae, California, United States, 94904-2007 GSK Investigational Site San Francisco, California, United States, 94115 GSK Investigational Site Stanford, California, United States, 94305 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, Maryland GSK Investigational Site Bethesda, Maryland, United States, 20892 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02115 United States, Michigan GSK Investigational Site Detroit, Michigan, United States, 48201 United States, New Jersey GSK Investigational Site New Brunswick, New Jersey, United States, 08901 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44195 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37232 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78229 Collapse << |
| NCT00261846 | - | - | Completed | - | - |
| NCT01079806 | - | - | Completed | - | - |
| NCT01195662 | - | - | Completed | - | - |
| NCT00371683 | Deep Vein Thrombosis ... more >> Pulmonary Embolism Collapse << | Phase 3 | Completed | - | - |
| NCT00673387 | Overweight Ob... more >>esity Collapse << | Phase 2 | Completed | - | - |
| NCT02753231 | Obesity Metab... more >>olic Syndrome X Lipid Metabolism Disorders Body Composition, Beneficial Mental Health Wellness 1 Collapse << | Not Applicable | Completed | - | - |
| NCT00371683 | - | - | Completed | - | - |
| NCT00873041 | Non-transfusion Dependent Thal... more >>assemia Collapse << | Phase 2 | Completed | - | United States, California ... more >> Children's Hospital & Research Center Oakland Oakland, California, United States, 94609-1809 United States, Illinois Children's Memorial Hospital/Division of Hematology/Oncology Chicago, Illinois, United States, 60614-3394 United States, New York New York Presbyterian Hospital/Weill Medical College of Cornell University New York, New York, United States, 10021 Greece Novartis Investigative Site Athens, Greece Novartis Investigative Site Patras, Greece Novartis Investigative Site Thessaloniki, Greece Italy Novartis Investigative Site Cagliari, Italy Novartis Investigative Site Genova, Italy Novartis Investigative Site Milano, Italy Novartis Investigative Site Napoli, Italy Novartis Investigative Site Rome, Italy Lebanon Novartis Investigative Site Beirut, Lebanon Malaysia Novartis Investigative Site Ampang Selangor, Malaysia Novartis Investigative Site Kuala Lumpur, Malaysia Taiwan Novartis Investigative Site Taipei, Taiwan Thailand Novartis Investigative Site Bangkok, Thailand Turkey Novartis Investigative Site Adana, Turkey Novartis Investigative Site Ankara, Turkey Novartis Investigative Site Istanbul, Turkey Novartis Investigative Site Izmir, Turkey United Kingdom Novartis Investigative Site London, United Kingdom Collapse << |
| NCT00673387 | - | - | Completed | - | - |
| NCT00873041 | - | - | Completed | - | - |
| NCT00114777 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT01153763 | Melanoma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90025 GSK Investigational Site Los Angeles, California, United States, 90095 GSK Investigational Site San Francisco, California, United States, 94115 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37232 United States, Texas GSK Investigational Site Houston, Texas, United States, 77030-4009 Australia, New South Wales GSK Investigational Site Newcastle, New South Wales, Australia, 2300 GSK Investigational Site Westmead, New South Wales, Australia, 2145 Australia, Western Australia GSK Investigational Site Nedlands, Western Australia, Australia, 6009 France GSK Investigational Site Bordeaux, France, 33075 GSK Investigational Site Boulogne-Billancourt, France, 92100 GSK Investigational Site Lille, France, 59037 GSK Investigational Site Marseille Cedex 5, France, 13385 GSK Investigational Site Montpellier, France, 34295 GSK Investigational Site Paris Cedex 10, France, 75475 GSK Investigational Site Villejuif, France, 94805 Germany GSK Investigational Site Essen, Nordrhein-Westfalen, Germany, 45122 GSK Investigational Site Kiel, Schleswig-Holstein, Germany, 24105 GSK Investigational Site Luebeck, Schleswig-Holstein, Germany, 23538 GSK Investigational Site Berlin, Germany, 10117 Italy GSK Investigational Site Napoli, Campania, Italy, 80131 GSK Investigational Site Genova, Liguria, Italy, 16132 GSK Investigational Site Padova, Veneto, Italy, 35128 Collapse << |
| NCT01153763 | - | - | Completed | - | - |
| NCT00394251 | Breast Cancer | Phase 2 | Completed | - | - |
| NCT03601546 | - | - | Not yet recruiting | August 31, 2020 | France ... more >> CHU Nimes Nîmes, France, 30029 Vietnam Hospital Viet Tiep Not yet recruiting Hai Phong, Vietnam Contact: Vu Hai Vinh, MD +84.(0)9.84.77.70.03 vinhvuhai@gmail.com Principal Investigator: Vu Hai Vinh Collapse << |
| NCT00983801 | Stomach Neoplasms | Phase 2 | Completed | - | Hong Kong ... more >> Local Institution Hong Kong, Hong Kong, B Japan Local Institution Nagoya, Aichi, Japan, 4648681 Local Institution Sunto-Gun, Shizuoka, Japan, 4118777 Korea, Republic of Local Institution Seoul, Korea, Republic of, 135-710 Local Institution Seoul, Korea, Republic of, 136-705 Local Institution Seoul, Korea, Republic of, 138-736 Singapore Local Institution Singapore, Singapore, 169610 Taiwan Local Institution Taipei, Taiwan, 112 Local Institution Taoyuan Hsien, Taiwan, 333 Collapse << |
| NCT00394251 | - | - | Completed | - | - |
| NCT02478151 | End-stage Liver Disease | Phase 1 | Enrolling by invitation | April 2020 | Canada, Ontario ... more >> Toronto General Hospital Toronto, Ontario, Canada, M5G 2N2 Collapse << |
| NCT01231620 | Influenza, Human | Phase 3 | Completed | - | - |
| NCT01525238 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Alabama ... more >> The Children Hospital Of Alabama Birmingham, Alabama, United States, 35233 United States, California Axis Clinical Trials Los Angeles, California, United States, 90036 United States, Florida Nemours Childrens Hospital Orlando, Florida, United States, 32827 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Kentucky Kosair Charities Pediatric Clinical Research Unit Louisville, Kentucky, United States, 40202 United States, Louisiana Lsuhsc-Shreveport Shreveport, Louisiana, United States, 71103 United States, Missouri Childrens Mercy Hospital Kansas City, Missouri, United States, 64108 United States, New York Women And Children'S Hopsital Of Buffalo Buffalo, New York, United States, 14222 United States, Ohio Promedica Toledo Children'S Hospital Toledo, Ohio, United States, 43606 Mercy Children'S Hospital Toledo, Ohio, United States, 43608 United States, Pennsylvania Children'S Hospital Of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Children'S Hospital Of Pittsburgh Of Upmc Pittsburgh, Pennsylvania, United States, 15224 United States, Tennessee Methodist Le Bonheur Hlthcare Memphis, Tennessee, United States, 38103 United States, Texas Christus Santa Rosa Childrens Hospital San Antonio, Texas, United States, 78207 Mexico Local Institution Guadalajara, Jalisco, Mexico, 44150 Local Institution Monterrey, Nuevo Leon, Mexico, 64460 Local Institution Veracruz, Mexico, 91910 Collapse << |
| NCT01525238 | - | - | Completed | - | - |
| NCT00114777 | - | - | Completed | - | - |
| NCT00983801 | - | - | Completed | - | - |
| NCT01610492 | Glomerulonephritis, Membranous | Phase 2 | Completed | - | United Kingdom ... more >> GSK Investigational Site Exeter, Devon, United Kingdom, EX2 5DW GSK Investigational Site Stevenage, Hertfordshire, United Kingdom, SG1 4AB GSK Investigational Site Cambridge, United Kingdom, CB2 0QQ GSK Investigational Site Glasgow, United Kingdom, G11 6NT GSK Investigational Site Manchester, United Kingdom, M13 9WL GSK Investigational Site Reading, United Kingdom, RG1 5AN GSK Investigational Site Salford, United Kingdom, M6 8HD GSK Investigational Site Whitechapel, London, United Kingdom, E1 1BB Collapse << |
| NCT01520909 | Idiopathic Thrombocytopenic Pu... more >>rpura Collapse << | Phase 3 | Completed | - | - |
| NCT00924170 | - | - | Active, not recruiting | - | - |
| NCT00924170 | Adult T-Cell Leukemia (ATL) | Phase 1 Phase 2 | Active, not recruiting | January 1, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01231620 | - | - | Completed | - | - |
| NCT01766050 | Transplant Rejection | Phase 4 | Completed | - | United States, Texas ... more >> Healthcare Discoveries, Llc D/B/A Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT01610492 | - | - | Completed | - | - |
| NCT02031224 | Chronic Kidney Disease | Phase 4 | Completed | - | Romania ... more >> "Dr Carol Davila" Teaching Hospital of Nephrology Bucharest, Romania, 010731 Collapse << |
| NCT01520909 | - | - | Completed | - | - |
| NCT03356977 | Atopic Dermatitis | Phase 4 | Recruiting | February 23, 2019 | - |
| NCT01766050 | - | - | Completed | - | - |
| NCT03156478 | Type2 Diabetes ... more >> Life Style Diet Habit Physical Activity Empowerment Collapse << | Not Applicable | Recruiting | December 2037 | Finland ... more >> University of Eastern Finland Recruiting Kuopio, Finland Contact: Jussi Pihlajamäki, MD, PhD +358 50 344 0187 jussi.pihlajamaki@uef.fi Collapse << |
| NCT00680901 | Neoplasms, Gastrointestinal Tr... more >>act Collapse << | Phase 3 | Active, not recruiting | December 31, 2020 | - |
| NCT00372112 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Camperdown, New South Wales, Australia, 2050 Australia, Western Australia GSK Investigational Site Nedlands, Western Australia, Australia, 6009 Bulgaria GSK Investigational Site Ruse, Bulgaria, 7000 GSK Investigational Site Sofia, Bulgaria, 1431 GSK Investigational Site Sofia, Bulgaria, 1606 Germany GSK Investigational Site Weinheim, Baden-Wuerttemberg, Germany, 69469 GSK Investigational Site Hannover, Niedersachsen, Germany, 30159 GSK Investigational Site Geesthacht, Schleswig-Holstein, Germany, 21502 Netherlands GSK Investigational Site Breda, Netherlands, 4819 EV GSK Investigational Site Hoorn, Netherlands, 1624 NP New Zealand GSK Investigational Site Auckland, New Zealand, 1005 GSK Investigational Site Tauranga, New Zealand Romania GSK Investigational Site Bucharest, Romania, 050159 GSK Investigational Site Iasi, Romania, 700506 Collapse << |
| NCT03539601 | Atopic Dermatitis | Phase 4 | Recruiting | May 25, 2020 | United States, Arkansas ... more >> Dermatology Trial Associates Recruiting Bryant, Arkansas, United States, 72022 United States, California California Dermatology & Clinical Research Institute Recruiting Encinitas, California, United States, 92024 United States, Florida Park Avenue Dermatology Recruiting Orange Park, Florida, United States, 32073 Forward Clinical Trials, Inc. Recruiting Tampa, Florida, United States, 33624 United States, Kentucky DS Research Recruiting Louisville, Kentucky, United States, 40241 United States, North Carolina M3-Wake Research, Inc. Recruiting Raleigh, North Carolina, United States, 27612 United States, Texas Arlington Research Center Inc Recruiting Arlington, Texas, United States, 76011 United States, Virginia Virginia Clinical Research, Inc Recruiting Norfolk, Virginia, United States, 23502 Collapse << |
| NCT01225822 | - | - | Completed | - | - |
| NCT02722044 | - | - | Completed | - | - |
| NCT00372112 | - | - | Completed | - | - |
| NCT01947998 | - | - | Recruiting | December 31, 2019 | United Kingdom ... more >> Recruiting Multiple Locations, United Kingdom Collapse << |
| NCT02722044 | Rheumatoid Arthritis | Phase 3 | Completed | - | United States, Michigan ... more >> Henry Ford Health System Detroit, Michigan, United States, 48202 United States, North Carolina Medication Management, LLC Greensboro, North Carolina, United States, 27408 United States, Oklahoma Lynn Health Science Institute Oklahoma City, Oklahoma, United States, 73112 United States, Pennsylvania Altoona Center for Clinical Research, PC Duncansville, Pennsylvania, United States, 16635 United States, Texas Austin Regional Clinic, PA Austin, Texas, United States, 78731 Accurate Clinical Management Houston, Texas, United States, 77004 Accurate Clinical Research, Inc. Houston, Texas, United States, 77034 Collapse << |
| NCT01439373 | Hepatitis C, Chronic | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Anaheim, California, United States, 92801 GSK Investigational Site Chula Vista, California, United States, 91911 GSK Investigational Site Coronado, California, United States, 92118 GSK Investigational Site La Mesa, California, United States, 91942 GSK Investigational Site Oceanside, California, United States, 92056 United States, Florida GSK Investigational Site Miramar, Florida, United States, 33025 United States, Nevada GSK Investigational Site Las Vegas, Nevada, United States, 89109 United States, Oklahoma GSK Investigational Site Tulsa, Oklahoma, United States, 74104 United States, Texas GSK Investigational Site Houston, Texas, United States, 77004 Puerto Rico GSK Investigational Site San Juan, Puerto Rico, 00927 Collapse << |
| NCT00680901 | - | - | Active, not recruiting | - | - |
| NCT03460652 | ADHD | Phase 3 | Recruiting | March 2019 | United States, Arkansas ... more >> Woodland International Research Recruiting Little Rock, Arkansas, United States, 72211 Contact: Lacey Earls 501-221-8681 lacey@woodlandintlresearchgrp.com Principal Investigator: Jim Aukstuolis, MD United States, Colorado MCB Clinical Research Centers Recruiting Colorado Springs, Colorado, United States, 80910 Contact: Nichole Gonzalez 719-634-6576 ngonzalez@mcbcrc.com Principal Investigator: Andrew Sedillo, MD United States, Florida Meridien Research Recruiting Bradenton, Florida, United States, 34201 Contact: Margaret Bell 941-756-8680 mbell@meridienresearch.net Principal Investigator: Andrew Cutler, MD Clinical Neuroscience Solutions, Inc. Recruiting Jacksonville, Florida, United States, 32256 Contact: Sandeep Jandir 904-281-5757 sjandir@cnshealthcare.com Principal Investigator: Nandita Jones, MD Innovative Clinical Research, Inc. Recruiting Lauderhill, Florida, United States, 33319 Contact: Kadian Watson 954-990-6326 kwatson@segaltrials.com Principal Investigator: Rishi Kakar, MD Meridien Research Recruiting Maitland, Florida, United States, 32751 Contact: Sondra Chapman 407-644-1165 schapman@meridienresearch.net Principal Investigator: Andrea Marraffino, MD Clinical Neuroscience Solutions, Inc. Recruiting Orlando, Florida, United States, 32801 Contact: Rebecca Klipper 407-425-5100 rklipper@cnshealthcare.com Principal Investigator: Robert Molpus, MD United States, Illinois Capstone Clinical Research Recruiting Libertyville, Illinois, United States, 60048 Contact: Kelly Burns 847-549-7214 kburns@capstoneclinical.com Principal Investigator: Michael Greenbaum, MD United States, Kansas Psychiatric Associates Recruiting Overland Park, Kansas, United States, 66211 Contact: Glynis Murphy, PhD 913-438-8221 research@murphyclinic.com Principal Investigator: William Murphy, MD United States, Nevada Center for Psychiatry and Behavioral Medicine, Inc. Recruiting Las Vegas, Nevada, United States, 89128 Contact: Jordan Karbins 702-838-0742 jordankabins@gmail.com Principal Investigator: Ann Childress, MD United States, New Jersey Hassman Research Institute Recruiting Berlin, New Jersey, United States, 08009 Contact: Vania Dasilva 856-753-7335 vdasilva@hritrials.com Principal Investigator: Howard Hassman, MD United States, Ohio Neuro-Behavioral Clinical Research Recruiting Canton, Ohio, United States, 44718 Contact: Natalie Kuemerle 330-493-1118 nkuemerle@nb-cr.com Principal Investigator: Shishuka Malhotra, MD Midwest Clinical Research Center, LLC Recruiting Dayton, Ohio, United States, 45417 Contact: Rebecca Vernon 937-424-1050 rvernon@midwestclinical.com Principal Investigator: Otto Dueno, MD United States, Oregon Oregon Center for Clinical Investigations, Inc. Recruiting Portland, Oregon, United States, 97214 Contact: Christina Friesen 503-276-6224 christina.friesen@occi.org Principal Investigator: Beal Essink, MD Oregon Center for Clinical Investigations, Inc. Recruiting Salem, Oregon, United States, 97301 Contact: Emily Valdez 503-540-0100 emily.valdez@occi.org Principal Investigator: Drissana Tran, MD United States, Tennessee Clinical Neuroscience Solutions, Inc. Recruiting Memphis, Tennessee, United States, 38119 Contact: Kelly Iskiwitz, BS 901-843-1045 kiskiwitz@cnshealthcare.com Principal Investigator: Valerie Arnold, MD United States, Texas FutureSearch Trials Recruiting Austin, Texas, United States, 78731 Contact: Erin Ashmore 214-369-2600 ext 109 erina@fstrials.com Principal Investigator: Michael Downing, MD Bayou City Research, Ltd. Recruiting Houston, Texas, United States, 77007 Contact: Kathy Harpst 832-251-7000 ext 302 Principal Investigator: Matthew Brams, MD Houston Clinical Trials Recruiting Houston, Texas, United States, 77098 Contact: Janice Roggenkamp 713-527-8448 jroggenkamp@houstonclintrials.com Principal Investigator: Alain Katic, MD Collapse << |
| NCT00486863 | Schistosomiasis | Phase 2 | Completed | - | Philippines ... more >> Research Institute for Tropical Medicine - Health Compound Muntinlupa City, National Capital Region, Philippines, 1781 Collapse << |
| NCT01225822 | Venous Thromboembolism | Phase 2 | Completed | - | - |
| NCT01439373 | - | - | Completed | - | - |
| NCT03160976 | Subcutaneous Fat | Not Applicable | Not yet recruiting | October 25, 2017 | Brazil ... more >> Federal University of Health Science of Porto Alegre Not yet recruiting Porto Alegre, Rio Grande do Sul, Brazil, 90050170 Contact: Rodrigo DM Plentz, PhD 55 51 998419044 roplentz@yahoo.com.br Collapse << |
| NCT00819234 | Obesity | Phase 2 | Completed | - | - |
| NCT00486863 | - | - | Completed | - | - |
| NCT00927082 | Hepatitis B, Chronic | Phase 4 | Completed | - | - |
| NCT00374322 | - | - | Completed | - | - |
| NCT00819234 | - | - | Completed | - | - |
| NCT01286779 | - | - | Completed | - | - |
| NCT01286779 | Hemophilia B | Phase 3 | Completed | - | - |
| NCT00374322 | Neoplasms, Breast | Phase 3 | Completed | - | - |
| NCT00546364 | - | - | Terminated(Slow Accrual) | - | - |
| NCT02293902 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT00927082 | - | - | Completed | - | - |
| NCT00920907 | Advanced Melanoma | Phase 1 | Completed | - | United States, California ... more >> The Angeles Clinic & Research Inst. Los Angeles, California, United States, 90025 California Pacific Medical Center San Francisco, California, United States, 94115 United States, Florida H Lee Moffitt Cancer Center Tampa, Florida, United States, 33612-9416 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 11065 United States, North Carolina Carolinas Medical Center Charlotte, North Carolina, United States, 28204 United States, Pennsylvania St Luke'S Hospital And Health Network Bethlehem, Pennsylvania, United States, 18015 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109-1023 Collapse << |
| NCT00546364 | Breast Neoplasms | Phase 2 | Terminated(Slow Accrual) | - | - |
| NCT00284141 | Pulmonary Diseases ... more >> Neoplasms, Lung Collapse << | Phase 2 | Completed | - | United States, New Jersey ... more >> Sanofi-Aventis Administrative Office Bridgewater, New Jersey, United States, 08807 Canada Sanofi-Aventis Administrative Office Laval, Canada France Sanofi-Aventis Administrative Office Paris, France Collapse << |
| NCT03667430 | Safety Issues | Not Applicable | Completed | - | - |
| NCT00920907 | - | - | Completed | - | - |
| NCT01300234 | Hepatitis B | Phase 3 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China, 510060 GSK Investigational Site Guangzhou, Guangdong, China, 510515 GSK Investigational Site Guangzhou, Guangdong, China, 510630 China, Hubei GSK Investigational Site Wuhan, Hubei, China, 430030 China, Hunan GSK Investigational Site Changsha, Hunan, China, 410008 China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210003 GSK Investigational Site Nanjing, Jiangsu, China, 210029 China, Jilin GSK Investigational Site Changchun, Jilin, China, 130021 China, Sichuan GSK Investigational Site Chengdu, Sichuan, China, 610041 China, Zhejiang GSK Investigational Site Hangzhou, Zhejiang, China, 310003 China GSK Investigational Site Beijing, China, 100015 GSK Investigational Site Beijing, China, 100044 GSK Investigational Site Beijing, China, 100050 GSK Investigational Site Chongqing, China, 400038 GSK Investigational Site Fuzhou, China, 350025 GSK Investigational Site Jinan, China, 250021 GSK Investigational Site Shanghai, China, 200001 GSK Investigational Site Shanghai, China, 200025 GSK Investigational Site Shanghai, China, 200040 GSK Investigational Site Shanghai, China, 201508 Collapse << |
| NCT02293902 | - | - | Completed | - | - |
| NCT03188497 | Acute Toxic Effects ... more >> Tumor Responses Collapse << | Phase 1 | Completed | - | China, Guangdong ... more >> the Fifth Hospital Affiliated to Sun Yat-Sen University Zhuhai, Guangdong, China, 519000 Collapse << |
| NCT01491165 | Other Surgical Procedures | Phase 2 Phase 3 | Unknown | June 2014 | China ... more >> Yihua An Recruiting Beijing, China Contact: Yihua An, doctor 0086-10-88276848 doctoran2010@hotmail.com Collapse << |
| NCT03585413 | Obesity Gastr... more >>ic Bypass NAFLD Collapse << | Phase 3 | Not yet recruiting | August 2020 | Germany ... more >> St. Franziskus-Hospital Not yet recruiting Cologne, Germany, 50825 Collapse << |
| NCT00284141 | - | - | Completed | - | - |
| NCT03725059 | Breast Cancer | Phase 3 | Not yet recruiting | January 24, 2031 | - |
| NCT01805466 | C.Surgical Procedure; Cardiac | Not Applicable | Completed | - | Italy ... more >> Anthea Hospital Bari, Italy Maria Cecilia Hospital Cotignola, Italy, 48010 Città di Lecce Hospital Lecce, Italy ICLAS Rapallo Rapallo, Italy Collapse << |
| NCT00976560 | Depressive Disorder, Major | Phase 2 | Completed | - | United States, Illinois ... more >> GSK Investigational Site Hoffman Estates, Illinois, United States, 60169 GSK Investigational Site Park Ridge, Illinois, United States, 60068 United States, New York GSK Investigational Site New York, New York, United States, 10128 United States, Ohio GSK Investigational Site Columbus, Ohio, United States, 43210 Bulgaria GSK Investigational Site Pazardzhik, Bulgaria, 4400 GSK Investigational Site Plovdiv, Bulgaria, 4000 GSK Investigational Site Ruse, Bulgaria GSK Investigational Site Sofia, Bulgaria, 1113 Estonia GSK Investigational Site Tartu, Estonia, 50417 Germany GSK Investigational Site Huettenberg, Hessen, Germany, 35625 GSK Investigational Site Schwerin, Mecklenburg-Vorpommern, Germany, 19053 GSK Investigational Site Achim, Niedersachsen, Germany, 28832 GSK Investigational Site Westerstede, Niedersachsen, Germany, 26655 GSK Investigational Site Bielefeld, Nordrhein-Westfalen, Germany, 33647 GSK Investigational Site Dresden, Sachsen, Germany, 01097 GSK Investigational Site Berlin, Germany, 10629 Russian Federation GSK Investigational Site Moscow, Russian Federation, 107076 GSK Investigational Site Moscow, Russian Federation, 125367 GSK Investigational Site Samara, Russian Federation, 443016 GSK Investigational Site Smolensk, Russian Federation, 214 019 GSK Investigational Site St-Petersburg, Russian Federation Collapse << |
| NCT03176433 | Liver Transplantation | Phase 1 Phase 2 | Recruiting | July 31, 2018 | United Kingdom ... more >> Addenbrooke's Hospital Recruiting Cambridge, United Kingdom Contact: Christopher Watson King's College Hospital Not yet recruiting London, United Kingdom Contact: Wayel Jassem Royal Free Hospital Recruiting London, United Kingdom Contact: Charles Imber Collapse << |
| NCT02594735 | Dermatomyositis | Phase 4 | Recruiting | December 2018 | United States, District of Col... more >>umbia 2300 M Street, 9th floor. Medical Faculty Associates, The George Washington University. Recruiting Washington, District of Columbia, United States, 20037 Contact: Rodolfo V Curiel, MD 202-750-0377 rcuriel@mfa.gwu.edu Contact: Hassan Awal 202-741-2389 hawal@mfa.gwu.edu Collapse << |
| NCT01300234 | - | - | Completed | - | - |
| NCT00022737 | Childhood Acute Lymphoblastic ... more >>Leukemia in Remission Recurrent Childhood Acute Lymphoblastic Leukemia Collapse << | Phase 3 | Completed | - | United States, California ... more >> Children's Oncology Group Arcadia, California, United States, 91006-3776 Collapse << |
| NCT02876952 | Myocardial Infarction | Not Applicable | Recruiting | June 2018 | Spain ... more >> Department of Physical Education and Sport. Faculty of Education and Sport-Physical Activity and Sport Section. University of the Basque Country Recruiting Vitoria-gasteiz, Araba/álava, Spain, 01007 Contact: SARA MALDONADO-MARTIN, PhD sara.maldonado@ehu.eus Sub-Investigator: Juan Jose GOIRIENA, PhD Collapse << |
| NCT03559985 | Neuropathic Pain | Phase 2 | Recruiting | September 30, 2019 | France ... more >> CHU Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 0473751195 placarin@chu-clermontferrand.fr Principal Investigator: Gisèle PICKERING Sub-Investigator: Christian DUALE Sub-Investigator: Claude DUBRAY Sub-Investigator: Gilles DUCHEIX Collapse << |
| NCT03603639 | Photosensitive Epilepsy | Phase 2 | Recruiting | February 11, 2019 | United States, Arkansas ... more >> Clinical Trials, Inc. and Arkansas Epilepsy Program Recruiting Little Rock, Arkansas, United States, 72205 United States, Idaho Consultants in Epilepsy & Neurology, PLLC Recruiting Boise, Idaho, United States, 83702 United States, Maryland Johns Hopkins University- School of Medicine Not yet recruiting Baltimore, Maryland, United States, 21287 United States, Missouri Washington University Hospital Not yet recruiting Saint Louis, Missouri, United States, 63110 United States, Pennsylvania Unniversity of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01933945 | - | - | Completed | - | - |
| NCT00262925 | Recurrent Adult Acute Lymphobl... more >>astic Leukemia Collapse << | Phase 2 | Terminated(slow accrual) | - | - |
| NCT03528642 | Anaplastic Astrocytoma, IDH-Mu... more >>tant Diffuse Astrocytoma, IDH-Mutant IDH1 Gene Mutation IDH2 Gene Mutation Collapse << | Phase 1 | Not yet recruiting | August 31, 2020 | - |
| NCT02700958 | Atherosclerosis ... more >> Stable Angina Peripheral Artery Disease Contrast-induced Nephropathy Collapse << | Not Applicable | Completed | - | Estonia ... more >> Tartu University Hospital Tartu, Tartu County, Estonia, 50406 Collapse << |
| NCT03578991 | Patients Aged 65-85 (Both Incl... more >>uded) Diagnosed of Mild Cognitive Impairment Diagnosed of Type 2 Diabetes in Active Treatment (Hypoglycemic Agents) for a Period ≥5 Years Collapse << | Not Applicable | Not yet recruiting | May 2020 | Spain ... more >> Fundació Privada Hospital Asil de Granollers (HAG) Not yet recruiting Granollers, Barcelona, Spain, 08402 Contact: Sergio Ariño, MD, PhD Principal Investigator: Sergio Ariño, MD, PhD Sub-Investigator: Diana Navarro Llobet, PhD Sub-Investigator: Gemma Molist Señé, PhD Sub-Investigator: Juliana Valencia Robledo, MD Clínica Universitària de la Fundació Universitària del Bages (FUB) Not yet recruiting Manresa, Barcelona, Spain, 08242 Contact: Xavier Gironès García, PhD Principal Investigator: Xavier Gironès García, PhD Sub-Investigator: Lluïsa Sort García, management Sub-Investigator: Antonia Puiggrós Binefa, PhD Sub-Investigator: Daniel García, PhD Sub-Investigator: Ester Colillas Malet, PhD Sub-Investigator: Núria Obradors Rial, PhD Sub-Investigator: Marina Mateu Capell, PhD Sub-Investigator: Montserrat Soler Sellarés, PhD Sub-Investigator: Sara Sevilla Martínez, Nurse Sub-Investigator: Jordi Franch Pareja PhD, PhD Fundació Althaia (FA) Not yet recruiting Manresa, Barcelona, Spain, 08243 Contact: María Cruz Crespo Maraver, PhD Principal Investigator: María Cruz Crespo Maraver, PhD Sub-Investigator: Jesús Montesinos, BMBS Sub-Investigator: Anna Arnau Bartés, PhD Sub-Investigator: Irantzu Elorz, BCh Sub-Investigator: Eduard Pons Villanueva, BMBS Sub-Investigator: Jordi Aligué Capsada, BMBS Sub-Investigator: Jordi Giménez Salinas, BMBS Sub-Investigator: Judit Bonet Álvarez, BMBS Fundació Recerca Mútua Terrassa (FMT) Not yet recruiting Terrassa, Barcelona, Spain, 08221 Contact: María José Barahona Constanzo, MD, PhD Principal Investigator: María José Barahona Constanzo, MD, PhD Sub-Investigator: Laura Casas, PhD Sub-Investigator: Carmen Quiros Lopez, MD, PhD Sub-Investigator: Jerzy Krupinski, MD, PhD Sub-Investigator: Laura Buguñá Hoffmann, Enginyer Sub-Investigator: Verónica Perea Castilla, MD Sub-Investigator: Nuria Alonso Carril, Nurse Sub-Investigator: Jessica Molina Seguín, MD Sub-Investigator: Marta Almería Vivo, Psychologist Sub-Investigator: Sonia Arribas Pardo, Psychologist Sub-Investigator: Dolors Badenes Guia, Psychologist Sub-Investigator: Maria Teresa Buongiorno, MD Sub-Investigator: Andreu Simó Servat, MD Consorci Sanitari de Terrassa (CST) Not yet recruiting Terrassa, Barcelona, Spain, 08227 Contact: Maite Franco Romero Principal Investigator: Maite Franco Romero, Graduate. Sub-Investigator: Ramón Roca Puig, MD, PhD Sub-Investigator: Clara Romero Rascón, PhD Sub-Investigator: Ció Tor Figueras, Graduate. Sub-Investigator: Antonio Rey, BMBS Sub-Investigator: María José Sender, BMBS Sub-Investigator: Laia Pijuan, BMBS Sub-Investigator: Julia Grau, BMBS Sub-Investigator: Laura Palenzuela, BMBS Consorci Hospitalari de Vic (CHV) Not yet recruiting Vic, Barcelona, Spain, 08500 Contact: Joan Carles Rovira Pascual, MD Sub-Investigator: Núria Roger Casals, MD, PhD Sub-Investigator: Emi Chirveches, PhD Sub-Investigator: Joan Espaulella Panicot, MD, PhD Sub-Investigator: Isabel Ramon Bofarull, MD Sub-Investigator: Antoni Casals Pascual, MD, PhD Sub-Investigator: Francesc Escabila, MD Sub-Investigator: Anna Bartés Plans, MSc Sub-Investigator: Jordina Muñoz Padrós, MSc Sub-Investigator: Laura Espinosa Lopez Sub-Investigator: Emma Puigoriol Juvanteny Mind the byte Not yet recruiting Barcelona, Spain, 08007 Contact: David Bermúdez Principal Investigator: David Bermúdez, PhD Sub-Investigator: Oscar Villacañas Pérez, PhD Sub-Investigator: Laia Casafont, management Parc Sanitari Pere Virgili Barcelona, Spain, 08023 LambdaLoopers Not yet recruiting Barcelona, Spain, 08034 Contact: Adrià Batlle Principal Investigator: Adrià Batlle University Hospital Vall d'Hebron - Vall d'Hebron Institute of Research Not yet recruiting Barcelona, Spain, 08035 Contact: Rafael Simó Canonge, MD, PhD Contact: Cristina Hernaández Pascual, MD, PhD Sub-Investigator: Andreea Ciudin, MD, PhD Collapse << |
| NCT03685552 | Gastrointestinal Symptoms | Not Applicable | Completed | - | United States, Utah ... more >> The Hughes Center for Research and Innovation Lehi, Utah, United States, 84043 Collapse << |
| NCT01306942 | Metastatic Breast Cancer | Phase 1 Phase 2 | Active, not recruiting | June 2018 | Spain ... more >> Instituto Catalán de Oncología de Barcelona (Hospital Duran i Reynalds) Hospitalet de Llobregat, Barcelona, Spain, 08907 Hospital Alvaro Cunqueiro Vigo, Pontevedra, Spain, 36204 Complejo Hospitalario Universitario A Coruña A Coruña, Spain, 15006 Hospital Clinic i Provincial Barcelona, Spain, 08036 Hospital General Universitario Gregorio Marañón Madrid, Spain, 28007 Hospital Clínico Universitario Virgen de la Victoria Málaga, Spain, 29010 Hospital Universitario Virgen del Rocío Sevilla, Spain, 41013 Instituto Valenciano de Oncología Valencia, Spain, 46009 Collapse << |
| NCT00002798 | Childhood Acute Erythroleukemi... more >>a (M6) Childhood Acute Megakaryocytic Leukemia (M7) Childhood Acute Monoblastic Leukemia (M5a) Childhood Acute Monocytic Leukemia (M5b) Childhood Acute Myeloblastic Leukemia With Maturation (M2) Childhood Acute Myeloblastic Leukemia Without Maturation (M1) Childhood Acute Myelomonocytic Leukemia (M4) Childhood Myelodysplastic Syndromes Chronic Myelomonocytic Leukemia de Novo Myelodysplastic Syndromes Refractory Anemia Refractory Anemia With Excess Blasts Refractory Anemia With Excess Blasts in Transformation Refractory Anemia With Ringed Sideroblasts Secondary Myelodysplastic Syndromes Untreated Childhood Acute Myeloid Leukemia and Other Myeloid Malignancies Collapse << | Phase 3 | Completed | - | United States, California ... more >> Children's Oncology Group Arcadia, California, United States, 91006-3776 Collapse << |
| NCT00262925 | - | - | Terminated(slow accrual) | - | - |
| NCT03025152 | Gastric Cancer ... more >> Gastrointestinal Disease Collapse << | Not Applicable | Recruiting | July 2020 | China, Jiangxi ... more >> Jiangxi University of Traditional Chinese Medicine Recruiting Nanchang, Jiangxi, China, 330004 Contact: Xu Zhou +8618780085060 zhouxu_ebm@hotmail.com Contact: Xiaofan Chen +86079187118012 841347982@qq.com Collapse << |
| NCT03227861 | HIV-1 | Phase 3 | Active, not recruiting | July 31, 2019 | United States, Arizona ... more >> Spectrum Medical Group Phoenix, Arizona, United States, 85012 United States, California The Office of Franco Felizarta, MD Bakersfield, California, United States, 93301 Jeffrey Goodman Clinic - DBA Los Angeles Gay and Lesbian Center Los Angeles, California, United States, 90028 United States, District of Columbia Whitman Walker Health Washington, District of Columbia, United States, 20009 United States, Florida Midway Immunology and Research Center Fort Pierce, Florida, United States, 34982 University of Miami Miami, Florida, United States, 33136 Orlando Immunology Center Orlando, Florida, United States, 32803 United States, Georgia Chatham County Health Department Savannah, Georgia, United States, 31401 United States, Illinois The Ruth M. Rothstein CORE Center Chicago, Illinois, United States, 60612 United States, New Jersey Saint Michael's Medical Center- Infectious Disease Newark, New Jersey, United States, 07102 United States, New Mexico Southwest CARE Center Albuquerque, New Mexico, United States, 87109 Southwest CARE Center Santa Fe, New Mexico, United States, 87505 United States, Texas North Texas Infectious Diseases Consultants Dallas, Texas, United States, 75246 Tarrant County Infectious Disease Associates Fort Worth, Texas, United States, 76104 Therapeutic Concepts - Donald R Watkins Foundation Houston, Texas, United States, 77004 Gordon Crofoot, MD Houston, Texas, United States, 77098 Collapse << |
| NCT00098839 | Recurrent Childhood Acute Lymp... more >>hoblastic Leukemia Collapse << | Phase 1 Phase 2 | Completed | - | - |
| NCT00098839 | - | - | Completed | - | - |
| NCT01644396 | Moderate to Severe Plaque Psor... more >>iasis Collapse << | Phase 4 | Completed | - | Russian Federation ... more >> Site Reference ID/Investigator# 67547 Ekaterinburg, Russian Federation, 620076 Site Reference ID/Investigator# 78433 Kazan, Russian Federation, 420012 Site Reference ID/Investigator# 67542 Moscow, Russian Federation, 107076 Site Reference ID/Investigator# 67546 Saratov, Russian Federation, 410028 Site Reference ID/Investigator# 78417 Smolensk, Russian Federation, 214018 Site Reference ID/Investigator# 78413 St. Petersburg, Russian Federation, 190013 Site Reference ID/Investigator# 67545 St. Petersburg, Russian Federation, 194044 Collapse << |
| NCT03031470 | Ischemia-reperfusion Injury in... more >> Liver Transplant Early Allograft Dysfunction Collapse << | Phase 2 | Completed | - | Belarus ... more >> Healthcare Organization "9th City Clinical Hospital" Minsk, Belarus, 220045 Russian Federation State Budgetary Health Institution "Scientific Research Institute - Regional Clinical Hospital # 1 n.a. professor S.V. Ochapovskiy" of the Ministry of Health of the Krasnodar Territory Krasnodar, Krasnodar Territory, Russian Federation, 350086 State Budgetary Educational Institution of Higher Professional Education "First Saint Petersburg State Medical University n.a. I.P. Pavlov" of the Ministry of Health of the Russian Federation St. Petersburg, Saint Petersburg, Russian Federation, 197022 Federal State Budgetary Institution "Academician V.I. Shumakov Federal Research Center of Transplantology and Artificial Organs" Ministry of Health of the Russian Federation Moscow, Russian Federation, 123182 Federal State Budgetary Institution "State Research Centre of the Russian Federation - Federal Medical Biophysical Centre n.a. A.I. Burnazyan" Moscow, Russian Federation, 123182 State Budgetary Health Institution of Moscow "Scientific Research Institute of Emergency n.a. N.V. Sklifosovskiy of Moscow Healthcare Department" Moscow, Russian Federation, 129090 State Budgetary Health Institution of Novosibirsk Region "State Novosibirsk Regional Clinical Hospital" Novosibirsk, Russian Federation, 630087 Collapse << |
| NCT03067961 | Salmonella Typhi Infection | Not Applicable | Active, not recruiting | August 15, 2019 | United Kingdom ... more >> Oxford Vaccine Group, Centre for Clinical Vaccinology and Tropical Medicine Oxford, Oxfordshire, United Kingdom, OX3 7LE Collapse << |
| NCT03622112 | Asthma | Phase 2 | Not yet recruiting | September 6, 2019 | - |
| NCT03169959 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00537654 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Indiana ... more >> GSK Investigational Site Evansville, Indiana, United States, 47714 United States, Texas GSK Investigational Site Austin, Texas, United States, 78752 Collapse << |
| NCT02660736 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Texas ... more >> GSK Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT03582137 | Parkinson Disease | Phase 2 | Not yet recruiting | June 2019 | - |
| NCT02729051 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT03483090 | Osteoarthritis, Knee | Phase 2 | Recruiting | July 30, 2020 | Australia, Queensland ... more >> University of the Sunshine Coast Clinical Trials Centre, Morayfield Health Hub Recruiting Morayfield, Queensland, Australia, 4506 Contact: Jessica Baird +61 7 5456 3797 trials@usc.edu.au Contact: Georgina Street +61 7 5456 3798 gstreet@usc.edu.au Principal Investigator: Susan Thackwray University of the Sunshine Coast Clinical Trials Centre, USC Health Clinics Recruiting Sippy Downs, Queensland, Australia, 4556 Contact: Jessica Baird +61 7 5456 3797 trials@usc.edu.au Contact: Georgina Street +61 7 5456 3798 gstreet@usc.edu.au Principal Investigator: Susan Thackwray Australia, South Australia CSIRO Nutrition and Health Research Clinic Recruiting Adelaide, South Australia, Australia, 5000 Contact: Bianca Benassi-Evans Australia, Victoria Emeritus Research Recruiting Camberwell, Victoria, Australia, 3124 Contact: Edwin Young +61 3 950 96166 edwinyoung@emeritusresearch.com Contact: Teresa Ringeri +61 3 950 96166 teresaringeri@emeritusresearch.com Principal Investigator: Andrew Ostor Collapse << |
| NCT03395704 | Hereditary Hemochromatosis | Phase 2 | Recruiting | December 2018 | United States, Arkansas ... more >> Investigative Site Recruiting North Little Rock, Arkansas, United States, 72117 United States, California Investigative Site Recruiting Palo Alto, California, United States, 94305 Investigative Site Recruiting Rialto, California, United States, 92377 Investigational Site Recruiting San Diego, California, United States, 91942 United States, Michigan Investigative Site Recruiting Wyoming, Michigan, United States, 49519 United States, Mississippi Investigative Site Recruiting Jackson, Mississippi, United States, 39216 United States, New York Investigative Site Recruiting East Setauket, New York, United States, 11733 Investigative Site Recruiting Manhasset, New York, United States, 11030 Investigative Site Recruiting New York, New York, United States, 10029 United States, Texas Investigative Site Recruiting Dallas, Texas, United States, 75246 Investigative Site Recruiting Fort Worth, Texas, United States, 76104 Investigative Site Recruiting Houston, Texas, United States, 77058 Investigative Site Recruiting San Antonio, Texas, United States, 78215 United States, Washington Investigative Site Recruiting Seattle, Washington, United States, 98104 Australia, New South Wales Investigative Site Recruiting Westmead, New South Wales, Australia, 2145 Australia, Queensland Investigative Site Recruiting Brisbane, Queensland, Australia, 4120 Investigative Site Recruiting Herston, Queensland, Australia, 4029 Australia, Victoria Investigative Site Recruiting Melbourne, Victoria, Australia, 3004 France Investigative Site Recruiting Bondy, France, 93140 Investigative Site Recruiting Orléans, France, 45000 Investigative Site Recruiting Pessac, France, 33604 Investigative Site Recruiting Rennes Cedex 9, France, 35033 United Kingdom Investigative Site Recruiting Bradford, England, United Kingdom, BD9 6RJ Investigative Site Recruiting Newcastle Upon Tyne, England, United Kingdom, NE7 7DN Investigative Site Recruiting Portsmouth, England, United Kingdom, PO6 3LY Collapse << |
| NCT01644396 | - | - | Completed | - | - |
| NCT03493386 | Anaemia | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-0017 Collapse << |
| NCT03555890 | Rhinitis | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 812-0025 Collapse << |
| NCT02322450 | Essential Hypertension | Phase 2 | Completed | - | France ... more >> Hôpital Arthur Gardiner Dinard, France, 35800 Hôpital Cardiologique, CHRU de Lille Lille, France, 59037 Hospices Civils de Lyon - Hôpital de la Croix Rousse Lyon, France, 69317 Hôpital Européen Georges Pompidou Paris, France, 75015 Collapse << |
| NCT03525678 | Multiple Myeloma | Phase 2 | Recruiting | June 30, 2020 | - |
| NCT00873093 | - | - | Completed | - | - |
| NCT03472742 | - | - | Recruiting | July 31, 2021 | Japan ... more >> Niigata University Medical & Dental Hospital Recruiting Niigata, Japan, 951-8510 Contact: Division of Gastroenterology and Hepatology Collapse << |
| NCT03136991 | Cardiovascular Disease | Phase 1 | Completed | - | United States, California ... more >> Research Site Glendale, California, United States, 91206 Collapse << |
| NCT02832362 | Common Cold | Phase 1 | Completed | - | Canada, Quebec ... more >> GSK Investigational Site Quebec City, Quebec, Canada, G1P 0A2 Collapse << |
| NCT02780882 | Ectopic ACTH Syndrome | Phase 2 | Withdrawn(Due to stringent inc... more >>lusion/exclusion criteria, study investigator deemed it not feasible.) Collapse << | June 2019 | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States, 90048 Collapse << |
| NCT00873093 | B-cell Adult Acute Lymphoblast... more >>ic Leukemia B-cell Childhood Acute Lymphoblastic Leukemia Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Lymphoblastic Lymphoma Recurrent Childhood Acute Lymphoblastic Leukemia Recurrent Childhood Lymphoblastic Lymphoma T-cell Adult Acute Lymphoblastic Leukemia T-cell Childhood Acute Lymphoblastic Leukemia Collapse << | Phase 2 | Completed | - | - |
| NCT03677596 | Leukemia Prec... more >>ursor b-Cell Lymphoblastic Leukemia-Lymphoma ACUTE LYMPHOBLASTIC LEUKEMIA Collapse << | Phase 4 | Not yet recruiting | May 16, 2025 | - |
| NCT03019042 | Gastrointestinal Disease | Not Applicable | Active, not recruiting | December 2019 | China, Jiangxi ... more >> Jiangxi University of Traditional Chinese Medicine Nanchang, Jiangxi, China, 330004 Collapse << |
| NCT03329573 | Anxiety Disorders | Phase 1 | Completed | - | China ... more >> GSK Investigational Site Shanghai, China, 201508 Collapse << |
| NCT03511105 | Healthy Volunteers | Phase 1 | Recruiting | February 3, 2019 | Germany ... more >> GSK Investigational Site Recruiting Hannover, Niedersachsen, Germany, 30625 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03326232 | Gestational Diabetes | Not Applicable | Recruiting | July 2018 | United States, Virginia ... more >> Eastern Virginia Medical School Recruiting Norfolk, Virginia, United States, 23507 Contact: Joanne Audouin, MS 757-446-5121 audouij@evms.edu Contact: Andrew Lane, MD 864-608-4134 laneas@evms.edu Principal Investigator: Malgorzata Mlynarczyk, MD, PhD Sub-Investigator: Andrew Lane, MD Sub-Investigator: Margarita de Veciana, MD Sub-Investigator: Alfred Abuhamad, MD Collapse << |
| NCT03329547 | Infections, Bacterial | Phase 1 | Withdrawn(Change in commercial... more >> strategy) Collapse << | February 2, 2018 | - |
| NCT03393806 | Asthma | Phase 2 | Recruiting | October 10, 2019 | France ... more >> GSK Investigational Site Not yet recruiting Brest Cedex, France, 29609 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Marseille, France, 13015 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Montpellier cedex 5, France, 34295 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Netherlands GSK Investigational Site Recruiting Amsterdam, Netherlands, 1105 AZ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United Kingdom GSK Investigational Site Recruiting Wythenshawe, Greater Manchester, United Kingdom, M23 9LT Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Leicester, Leicestershire, United Kingdom, LE3 9QP Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Liverpool, Merseyside, United Kingdom, L7 8XP Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Birmingham, United Kingdom, B9 5SS Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Bradford, United Kingdom, BD9 6RJ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Edgbaston, United Kingdom, B15 2GW Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03149848 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom Collapse << |
| NCT02478463 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Active, not recruiting | September 10, 2019 | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21287-5554 United States, Pennsylvania GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00258050 | Neoplasms, Breast | Phase 1 | Completed | - | United States, New Hampshire ... more >> GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, North Carolina GSK Investigational Site Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT03485092 | Heart Failure ... more >> Diabetes Mellitus Collapse << | Phase 4 | Recruiting | February 28, 2020 | United Kingdom ... more >> Queen Elizabeth University Hospital Recruiting Glasgow, Scotland, United Kingdom, G51 4TF Contact: Mathew Lee, MBChB 0141 232 7600 matthewlee1@nhs.net Contact: Ann Wright, RCN 0141 232 7600 ann.wright@nhs.net Principal Investigator: Mark Petrie, MBChB Collapse << |
| NCT03299049 | HIV Infections | Phase 3 | Active, not recruiting | March 11, 2022 | - |
| NCT03562117 | Infections, Bacterial | Phase 1 | Recruiting | April 10, 2019 | United States, Florida ... more >> GSK Investigational Site Recruiting Miami, Florida, United States, 33136 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Orlando, Florida, United States, 32809 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT02893488 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT02924454 | Drug Overdose ... more >> Overdose of Beta-adrenergic Blocking Drug Blood Pressure Collapse << | Phase 4 | Completed | - | Denmark ... more >> Bispebjerg University Hospital Copenhagen Copenhagen NW, Capital Region of Denmark, Denmark, 2400 Collapse << |
| NCT03266172 | Autoimmune Diseases | Phase 1 | Recruiting | November 13, 2018 | United Kingdom ... more >> GSK Investigational Site Recruiting Nottingham, United Kingdom, NG11 6JS Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT02837380 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | China ... more >> GSK Investigational Site Shanghai, China, 200030 Collapse << |
| NCT00549679 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Estonia ... more >> GSK Investigational Site Tallinn, Estonia, 13419 Finland GSK Investigational Site Tampere, Finland, 33520 Germany GSK Investigational Site Gauting, Bayern, Germany, 82131 GSK Investigational Site Berlin, Germany, 10717 GSK Investigational Site Hamburg, Germany, 22291 Netherlands GSK Investigational Site Hoorn, Netherlands, 1624 NP GSK Investigational Site Horn, Netherlands, 6085 NM GSK Investigational Site Veldhoven, Netherlands, 5504 DB Russian Federation GSK Investigational Site Barnaul, Russian Federation, 656 045 GSK Investigational Site Moscow, Russian Federation, 105 077 GSK Investigational Site St. Petersburg, Russian Federation, 197022 GSK Investigational Site Yaroslavl, Russian Federation, 150003 Slovakia GSK Investigational Site Bratislava, Slovakia, 826 06 GSK Investigational Site Kosice, Slovakia, 041 90 GSK Investigational Site Martin, Slovakia, 036 59 Collapse << |
| NCT03562195 | Asthma | Phase 3 | Recruiting | April 2, 2021 | China, Guangdong ... more >> GSK Investigational Site Not yet recruiting Guangzhou, Guangdong, China, 510120 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com China, Jilin GSK Investigational Site Not yet recruiting Changchun, Jilin, China, 130041 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com China, Xinjiang GSK Investigational Site Not yet recruiting Urumqi, Xinjiang, China, 830054 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com China, Zhejiang GSK Investigational Site Recruiting Wenzhou, Zhejiang, China, 323027 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com China GSK Investigational Site Not yet recruiting Guangzhou, China, 511400 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Shanghai, China Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT02899026 | Polymyalgia Rheumatica | Phase 3 | Withdrawn(GSK decision to retu... more >>rn rights to sirukumab to Janssen and discontinue sirukumab development in polymyalgia rheumatica.) Collapse << | August 26, 2021 | - |
| NCT03450759 | Healthy Volunteers | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom, HA1 3UJ Collapse << |
| NCT02802514 | Diabetes Mellitus | Phase 4 | Terminated | - | United States, Massachusetts ... more >> GSK Investigational Site Boston, Massachusetts, United States, 02114 Collapse << |
| NCT02502812 | Cardiovascular Disease | Phase 1 | Completed | - | India ... more >> GSK Investigational Site Telangana, India, 500 013 Collapse << |
| NCT03639311 | HIV Infections | Phase 2 | Not yet recruiting | November 30, 2020 | United States, Florida ... more >> GSK Investigational Site Not yet recruiting Fort Lauderdale, Florida, United States, 33316 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Texas GSK Investigational Site Not yet recruiting Dallas, Texas, United States, 75246 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT02924688 | Asthma | Phase 3 | Active, not recruiting | February 21, 2019 | - |
| NCT03258762 | Toxoplasmosis | Phase 1 | Completed | - | Australia, Victoria ... more >> GSK Investigational Site Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT02578953 | Prostatic Hyperplasia | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-0017 Collapse << |
| NCT02666287 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT02751294 | Malaria, Vivax | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02794519 | Asthma | Phase 2 | Withdrawn(This study has suspe... more >>nded participant recruitment.) Collapse << | October 4, 2016 | United States, California ... more >> GSK Investigational Site Long Beach, California, United States, 90808 United States, Florida GSK Investigational Site Aventura, Florida, United States, 33180 United States, Ohio GSK Investigational Site Cincinnati, Ohio, United States, 45231 United States, Virginia GSK Investigational Site Richmond, Virginia, United States, 23225 GSK Investigational Site Richmond, Virginia, United States, 23229 Spain GSK Investigational Site Alcorcón (Madrid), Spain, 28922 GSK Investigational Site Gerona, Spain, 17005 GSK Investigational Site Santiago De Compostela. La Coruña., Spain, 15706 Collapse << |
| NCT03728868 | Healthy Individuals | Not Applicable | Recruiting | October 1, 2019 | Sweden ... more >> Geriatric Medicine, Sahlgrenska University Hospital, Mölndal Recruiting Gothenburg, Västra Götaland, Sweden, 43180 Contact: Maria Falkdal, Nurse +4631 34 310 67 maria.falkdal@gu.se Contact: Linda Jonsson, Nurse Collapse << |
| NCT03055195 | Dermatitis, Atopic | Phase 2 | Terminated(Study reached pre-d... more >>etermined futility criteria following interim analysis. No safety concerns were noted.) Collapse << | - | - |
| NCT03345407 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Active, not recruiting | January 10, 2019 | - |
| NCT02706535 | Drug Interactions | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02635347 | Liver Failure ... more >> Carcinoma, Hepatocellular Collapse << | Not Applicable | Completed | - | United States, New Jersey ... more >> Rutgers University - University Hospital Newark, New Jersey, United States, 07101-0820 Collapse << |
| NCT03575962 | HIV Infections | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT03408392 | Infections, Bacterial | Phase 1 | Completed | - | South Africa ... more >> GSK Investigational Site Bloemfontein, Free State, South Africa, 9300 Collapse << |
| NCT02698553 | Depressive Disorder, Major | Phase 1 | Completed | - | China ... more >> GSK Investigational Site Shanghai, China, 200030 Collapse << |
| NCT03417830 | Amyloidosis | Phase 1 | Terminated(Change in benefit/r... more >>isk profile.) Collapse << | - | Sweden ... more >> GSK Investigational Site Uppsala, Sweden, SE-751 85 GSK Investigational Site Uppsala, Sweden, SE-751 Collapse << |
| NCT03697824 | Neoplasms | Phase 2 | Not yet recruiting | July 17, 2022 | - |
| NCT03393975 | Congenital Thrombotic Thromboc... more >>ytopenic Purpura Collapse << | Phase 3 | Active, not recruiting | January 21, 2021 | United Kingdom ... more >> University College London Hospitals London, Greater London, United Kingdom, NW1 2PG Collapse << |
| NCT03250689 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Active, not recruiting | December 3, 2018 | United Kingdom ... more >> GSK Investigational Site Dundee, United Kingdom, DD1 9SY Collapse << |
| NCT02964507 | Neoplasms | Phase 2 | Recruiting | June 1, 2020 | - |
| NCT02466204 | Infertility | Phase 4 | Completed | - | Vietnam ... more >> My Duc Hospital Ho Chi Minh City, Vietnam, 70000 Research Center for Genetics and Reproductive Health Ho Chi Minh City, Vietnam, 70000 Collapse << |
| NCT03286842 | HER2-ve Metastatic Breast Canc... more >>er Germline BRCA1/2 Mutations Somatic BRCA1/2 Mutations Collapse << | Phase 3 | Recruiting | November 30, 2020 | - |
| NCT01457989 | - | - | Completed | - | - |
| NCT03000686 | Healthy Volunteers | Phase 1 | Recruiting | June 14, 2019 | Germany ... more >> GSK Investigational Site Recruiting Hannover, Niedersachsen, Germany, 30625 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03726593 | Plasmodium Falciparum Malaria ... more >>(Drug Resistant) Collapse << | Phase 4 | Recruiting | June 30, 2021 | Cambodia ... more >> Kratie Referral Hospital Not yet recruiting Kratie, Cambodia Contact: Somaly Kieng, M.D +855 72 971 755 Contact: Chanthap Lon, M.D 855 23 881 845 chanthapl@afrims.org Sub-Investigator: Darapiseth Sea, MD Sub-Investigator: Dysoley Lek, MD Anlong Veng Referral Hospital Recruiting Ânlóng Vêng, Cambodia Contact: Phan Kong, M.A 016 51 09 09 Principal Investigator: Chanthap Lon, MD Sub-Investigator: Darapiseth Sea, MD Sub-Investigator: Dysoley Lek, MD Collapse << |
| NCT02802501 | Malaria, Vivax | Phase 3 | Recruiting | November 29, 2019 | Indonesia ... more >> GSK Investigational Site Recruiting Jakarta, Indonesia, 10430 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03649165 | Neurofibromatosis Type 1 (NF1)... more >>-Related Plexiform Neurofibromas (PNs) Healthy Participants Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Research Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02668146 | Essential Tremor | Phase 2 | Unknown | June 2017 | United States, California ... more >> VA Greater Los Angeles Los Angeles, California, United States, 90073 Collapse << |
| NCT03741088 | Carcinoma, Hepatocellular ... more >> Liver Metastases Collapse << | Not Applicable | Recruiting | February 21, 2019 | Spain ... more >> Clinica Diagonal Recruiting Esplugues De Llobregat, Barcelona, Spain, 08950 Contact: Joan Vidal-Jove 0034 932 05 32 13 jvidal@khuab.com Principal Investigator: Joan Vidal-Jove Hospital universitario Mutua Terrassa Recruiting Terrassa, Barcelona, Spain, 08221 Contact: Joan Vidal-Jove 0034 937 36 50 50 jvidal@mutuaterrassa.es Principal Investigator: Joan Vidal-Jove Hospital Universitario Vall d´Hebrón Recruiting Barcelona, Spain, 08035 Contact: Xavier Serres 0034 93 489 30 00 xavierserresc@gmail.com Principal Investigator: Xavier Serres Collapse << |
| NCT03645434 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Recruiting | September 23, 2019 | Germany ... more >> Research Site Not yet recruiting Berlin, Germany, 14050 Research Site Withdrawn Frankfurt, Germany, 60596 Research Site Not yet recruiting Grosshansdof, Germany, 22927 Research Site Not yet recruiting Wiesbaden, Germany, 65187 United Kingdom Research Site Recruiting London, United Kingdom, HA1 3UJ Research Site Not yet recruiting Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT03398421 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT03462251 | Breast Cancer ... more >> Hormone Receptor Positive Tumor HER 2 Negative Breast Cancer Collapse << | Phase 3 | Recruiting | June 30, 2025 | Germany ... more >> Gemeinschaftspraxis für Hämatologie und Onkologie Recruiting Ravensburg, Baden-Wuerttemberg, Germany, 88212 Contact: Thomas Decker, Prof. +49 751 3661970 thomas.decker@onkonet.eu Collapse << |
| NCT03552393 | Anemia Renal ... more >>Insufficiency, Chronic Collapse << | Phase 2 | Recruiting | October 28, 2021 | - |
| NCT02787304 | Non-Alcoholic Steatohepatitis | Phase 2 | Terminated | - | - |
| NCT00976560 | - | - | Completed | - | - |
| NCT02246699 | Overweight and Obese Volunteer... more >>s. Collapse << | Not Applicable | Completed | - | France ... more >> Clinical Nutrition Center Naturalpha (CNCN) Lille, France, 59120 Collapse << |
| NCT02032888 | - | - | Completed | - | - |
| NCT00157248 | Atrial Fibrillation ... more >> Stroke Collapse << | Phase 2 | Terminated | - | - |
| NCT01843621 | Dengue | Phase 1 Phase 2 | Completed | - | Thailand ... more >> Department of Pediatrics, Phramongkutklao Hospital Phayathai, Bangkok, Thailand, 10400 Collapse << |
| NCT00157248 | - | - | Terminated | - | - |
| NCT02032888 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00908037 | Purpura, Thrombocytopaenic, Id... more >>iopathic Collapse << | Phase 2 | Completed | - | - |
| NCT01843621 | - | - | Completed | - | - |
| NCT03054194 | Healthy Participants | Phase 1 | Completed | - | United States, Texas ... more >> Eisai Trial Site San Antonio, Texas, United States, 78217 Collapse << |
| NCT00908037 | - | - | Completed | - | - |
| NCT02795754 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00256867 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01884675 | - | - | Terminated | - | - |
| NCT02758431 | Hypogonadism | Not Applicable | Recruiting | January 2021 | United States, Colorado ... more >> University of Colorado CCTSI CTRC Recruiting Denver, Colorado, United States, 80045 Contact: Kerrie Moreau, PhD 303-724-1914 kerrie.moreau@ucdenver.edu Collapse << |
| NCT00778622 | - | - | Completed | - | - |
| NCT02627898 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00778622 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | China, Beijing ... more >> Local Institution Beijing, Beijing, China, 100028 Local Institution Beijing, Beijing, China, 100034 Local Institution Beijing, Beijing, China, 100044 Local Institution Beijing, Beijing, China, 100088 Local Institution Beijing, Beijing, China, 100730 Local Institution Beijing, Beijing, China, 101100 Local Institution Beijing, Beijing, China, 200016 China, Guangdong Local Institution Guangdong Province, Guangdong, China, 510180 Local Institution Guangdong Province, Guangdong, China, 528000 Local Institution Guangdong, Guangdong, China, 510180 China, Shanghai Local Institution Shanghai, Shanghai, China, 200003 Local Institution Shanghai, Shanghai, China, 200092 Local Institution Shanghai, Shanghai, China, 201100 Local Institution Shanghai, Shanghai, China, 201200 Collapse << |
| NCT01935804 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 2 | Completed | - | Saudi Arabia ... more >> Center of Excellence for Osteoporosis Research, King Abdulaziz University Jeddah, Makkah, Saudi Arabia, 21589 Collapse << |
| NCT02629627 | Metabolic Syndrome | Phase 2 | Completed | - | - |
| NCT01884675 | Hypertension | Phase 3 | Terminated | - | - |
| NCT01435382 | Hypercholesterolemia ... more >> Dyslipidemias Hyperlipidemias Lipid Metabolism Disorders Metabolic Diseases Collapse << | Phase 1 | Completed | - | United States, California ... more >> Profil Institute for Clinical Research, Inc. Chula Vista, California, United States, 91911 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 United States, Kansas Vince and Associates Clinical Research Overland Park, Kansas, United States, 66212 United States, Maryland PAREXEL International - Baltimore Early Phase Clinical Unit Baltimore, Maryland, United States, 21225 United States, Michigan Jasper Clinic, Inc. Kalamazoo, Michigan, United States, 49007 United States, Minnesota Prism Research Saint Paul, Minnesota, United States, 55114 United States, Ohio Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45212 Collapse << |
| NCT01435382 | - | - | Completed | - | - |
| NCT01389596 | - | - | Completed | - | - |
| NCT01389596 | Epilepsy, Partial Seizures | Phase 3 | Completed | - | - |
| NCT02133807 | Atherosclerosis ... more >> Coronary Disease Carotid Artery Diseases Collapse << | Phase 3 | Completed | - | Russian Federation ... more >> Russian Cardiology Research and Production Center Moscow, Russian Federation, 121552 Collapse << |
| NCT01712074 | - | - | Terminated(The study was termi... more >>nated October 23, 2015 as pre-specified, interim analysis futility criteria were met. The termination was not due to safety concerns.) Collapse << | - | - |
| NCT03008954 | Overweight Ob... more >>esity Collapse << | Not Applicable | Completed | - | Czechia ... more >> Hradec Králové Hradec, Czechia, 500 00 Charles University Prague, Czechia, 121 08 Denmark University of Cophenhagen Copenhagen, Denmark, DK-1958 Italy Federico University Hospital Naples, Italy, 80131 Policlinico Umberto I Rome, Italy, 00161 Collapse << |
| NCT01712074 | Alzheimer's Disease | Phase 2 | Terminated(The study was termi... more >>nated October 23, 2015 as pre-specified, interim analysis futility criteria were met. The termination was not due to safety concerns.) Collapse << | - | - |
| NCT01473407 | Chronic Kidney Disease ... more >> Chronic Renal Failure Collapse << | Phase 3 | Completed | - | - |
| NCT02655510 | Alcoholic Hepatitis | Phase 1 Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic in Rochester Rochester, Minnesota, United States, 55905 Collapse << |
| NCT03021291 | Overweight and Obesity | Not Applicable | Completed | - | United States, California ... more >> Arternis Institute for Clinical Research San Diego, California, United States, 92103 Clinical Trial Investigators Tustin, California, United States, 92780 United States, Colorado University of Colorado Aurora, Colorado, United States, 80045 United States, Florida Westside Center for Clinical Research Jacksonville, Florida, United States, 32205 United States, Louisiana Pennington Biomedical Research Baton Rouge, Louisiana, United States, 70808 United States, Massachusetts Boston University Medical Center Boston, Massachusetts, United States, 02118 United States, New York Cornell Weill Medical College New York, New York, United States, 10065 United States, Ohio Radiant Research Cincinnati, Ohio, United States, 45249 Aventiv Research Columbus, Ohio, United States, 43213 United States, Pennsylvania Geisinger Health System Danville, Pennsylvania, United States, 17822 United States, Texas Texas Diabetes and Endocrinology Round Rock, Texas, United States, 78681 Czechia Health & Care SRO Prague, Czechia, 182 00 Denmark University of Cophenhagen Copenhagen, Denmark, DK-1958 Italy IRCCS Policlinico San Donato Milan, Italy, 20097 University of Rome Rome, Italy, 00186 Spain University of Navarra Pamplona, Spain, 31008 Collapse << |
| NCT01473407 | - | - | Completed | - | - |
| NCT03587142 | Gastroparesis | Phase 2 | Not yet recruiting | June 2020 | United States, Kentucky ... more >> University of Louisville Not yet recruiting Louisville, Kentucky, United States, 40202 Principal Investigator: Thomas Abell, MD United States, Maryland Johns Hopkins Hospital Not yet recruiting Baltimore, Maryland, United States, 21287 Principal Investigator: Pankaj J Pasricha, MD United States, Massachusetts Massachusetts General Hospital Not yet recruiting Boston, Massachusetts, United States, 02114 Principal Investigator: Braden Kuo, MD United States, North Carolina Wake Forest University Health Sciences Not yet recruiting Winston-Salem, North Carolina, United States, 27157 Principal Investigator: Kenneth Koch, MD United States, Pennsylvania Temple University Not yet recruiting Philadelphia, Pennsylvania, United States, 19140 Principal Investigator: Henry P Parkman, MD United States, Texas Texas Tech University Health Science Center Not yet recruiting El Paso, Texas, United States, 79905 Principal Investigator: Richard W McCallum, MD Collapse << |
| NCT01473420 | - | - | Completed | - | - |
| NCT01223352 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 3 | Completed | - | - |
| NCT01473420 | Chronic Renal Failure ... more >> Chronic Kidney Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01223352 | - | - | Completed | - | - |
| NCT02552602 | HIV | Not Applicable | Unknown | February 2016 | - |
| NCT00570063 | Schizophrenia | Phase 2 | Terminated | - | United States, California ... more >> California Clinical Trials Medical Group, Inc. Glendale, California, United States, 91206 Glendale Adventist Medical Center Glendale, California, United States, 91206 Telecare-Cresta Loma Lemon Grove, California, United States, 91945 California Clinical Trials Medical Group Paramount, California, United States, 90723 LaPaz Geropsychiatric Center Paramount, California, United States, 90723 California Clinical Trials Medical Group San Diego, California, United States, 92123 United States, Connecticut Connecticut Mental Health Center-Yale University, Clinical Neuroscience Research Unit New Haven, Connecticut, United States, 06519 United States, New Jersey CRI Worldwide, LLC Willingboro, New Jersey, United States, 08046 United States, Oklahoma St. Anthony Hospital Oklahoma City, Oklahoma, United States, 73101 IPS Research Company Oklahoma City, Oklahoma, United States, 73103 Collapse << |
| NCT02130557 | Leukemia, Myelogenous, Chronic... more >>, Breakpoint Cluster Region-Abelson Proto-oncogene (BCR-ABL) Positive Collapse << | Phase 3 | Active, not recruiting | April 4, 2020 | - |
| NCT02800967 | Hypertension ... more >>Overweight Collapse << | Not Applicable | Unknown | June 2017 | Serbia ... more >> Centre of Research Excellence in Nutrition and Metabolism, Institute for Medical Research, University of Belgrade Belgrade, Serbia, 11000 Collapse << |
| NCT02130557 | - | - | Active, not recruiting | - | - |
| NCT00425061 | Asthma | Phase 2 | Completed | - | - |
| NCT00425061 | - | - | Completed | - | - |
| NCT01943006 | Thrombophlebitis ... more >> Phlebitis Collapse << | Phase 3 | Completed | - | Thailand ... more >> Siriraj Hospital Bangkok, Thailand, 107000 Chulalongkorn Hospital Bangkok, Thailand Rajvithi Hospital Bangkok, Thailand Bamrasnaradua Infectious Diseases Institute Nonthaburi, Thailand Collapse << |
| NCT03527277 | Cardiovascular Risk Factor ... more >> Type2 Diabetes Mellitus Insulin Sensitivity Metabolic Syndrome Collapse << | Not Applicable | Recruiting | March 31, 2023 | United States, California ... more >> University of California, Davis Recruiting Davis, California, United States, 95616 Contact: Kimber Stanhope, Ph.D. 530-752-3720 klstanhope@ucdavis.edu Contact: Vivien Lee, B.S. 530-752-2714 vilee@ucdavis.edu Collapse << |
| NCT02072824 | Partial Onset Seizures | Phase 3 | Completed | - | - |
| NCT00886821 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, Florida ... more >> Cetero Research Miami Gardens, Florida, United States, 33169 United States, Texas Cetero Research - San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00570063 | - | - | Terminated | - | - |
| NCT00531752 | - | - | Completed | - | - |
| NCT00531752 | Attention Deficit Hyperactivit... more >>y Disorder Collapse << | Phase 2 | Completed | - | United States, California ... more >> Bay Area Research Institute Lafayette, California, United States, 94549 Pharmacology Research Institute Los Alamitos, California, United States, 90720 Pharmacology Research Institute Newport Beach, California, United States, 92660 United States, Illinois The University of Illinois at Chicago, Institute for Juvenile Research Chicago, Illinois, United States, 60608 United States, Massachusetts Massachusetts General Hospital, ADHD Program Cambridge, Massachusetts, United States, 02138 United States, New York New York University School of Medicine New York, New York, United States, 10010 United States, Virginia NeuroScience, Inc Herndon, Virginia, United States, 20170 Collapse << |
| NCT02194946 | End-Stage Renal Disease | Phase 1 Phase 2 | Enrolling by invitation | December 2018 | - |
| NCT02657759 | HYPERCHOLESTEROLEMIA | Not Applicable | Unknown | May 2016 | France ... more >> Biofortis Recruiting Saint-Herblain, France, 44800 Contact: Patrice CHAUVEAU + 33 (0)2 40 20 45 65 patrice.chauveau@mxns.com Principal Investigator: David GENDRE, M.D. Collapse << |
| NCT01994291 | Macular Edema, Diabetic | Phase 2 | Terminated | - | - |
| NCT03478631 | Hypertension ... more >>Blood Pressure, High Collapse << | Phase 3 | Not yet recruiting | August 2020 | Canada, Ontario ... more >> Clinical Nutrition and Risk Factor Modification Centre, St. Michael's Hospital Not yet recruiting Toronto, Ontario, Canada, M5B 1W8 Contact: Elena Jovanovski, MSc 416-864-6060 ext 2597 JovanovskiE@smh.ca Contact: Dandan Li, MSc (c) 416-864-6060 ext 5527 LiDand@smh.ca Principal Investigator: Vladimir Vuksan, PhD Sub-Investigator: John Sievenpiper (Qualified Investigator), MD, PhD Sub-Investigator: Verma Subodh, MD,FRCSC,PhD Sub-Investigator: David Jenkins, MD, PhD, DSc Sub-Investigator: Alexandra Jenkins, PhD, RD Sub-Investigator: Elena Jovanovski, MSc, PhD (c) Collapse << |
| NCT02072824 | - | - | Completed | - | - |
| NCT00886821 | - | - | Completed | - | - |
| NCT00528879 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01994291 | - | - | Terminated | - | - |
| NCT00528879 | - | - | Completed | - | - |
| NCT01234337 | - | - | Completed | - | - |
| NCT02007512 | - | - | Active, not recruiting | - | - |
| NCT01234337 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT02007512 | Breast Cancer | Phase 2 | Active, not recruiting | March 29, 2019 | - |
| NCT02650193 | - | - | Completed | - | - |
| NCT02650193 | Non-metastatic Breast Cancer | Phase 2 | Completed | - | Hungary ... more >> CRU Hungary Kft.,CRU Early Phase Unit, Miskolci Semmelweis Kórház és Egyetemi Oktató Kórház Miskolc, BAZ Megye, Hungary, 3529 Debreceni Egyetem Klinikai Központ, ÁOK, Onkológiai Klinika Debrecen, Hajdú-bihar Megye, Hungary, 4032 Országos Onkológiai Intézet Budapest, Hungary, 1122 Spain Hospital Universitario Arnau de Vilanova Lleida, Cataluna, Spain, 25198 Hospital Universitario de Fuenlabrada, Servicio de oncologia Fuenlanbrada, Madrid, Spain, 28942 START Madrid - CIOCC, Unidad de fases 1 planta 3, Hospital Universitario HM Sanchinarro Madrid, Spain, 28050 Hospital Arnau de Vilanova. planta 6, unidad de Oncología Valencia, Spain, 46015 Collapse << |
| NCT00408005 | T Acute Lymphoblastic Leukemia... more >> T Lymphoblastic Lymphoma Collapse << | Phase 3 | Completed | - | - |
| NCT00557193 | Acute Lymphoblastic Leukemia ... more >> Acute Undifferentiated Leukemia Childhood T Acute Lymphoblastic Leukemia Untreated Childhood Acute Lymphoblastic Leukemia Collapse << | Phase 3 | Completed | - | - |
| NCT03355079 | Cancer-related Malnutrition | Phase 4 | Recruiting | July 2020 | France ... more >> Hôpital Cochin Recruiting Paris, France, 75014 Collapse << |
| NCT00720109 | Adult B Acute Lymphoblastic Le... more >>ukemia With t(9;22)(q34;q11.2); BCR-ABL1 Childhood B Acute Lymphoblastic Leukemia With t(9;22)(q34;q11.2); BCR-ABL1 Untreated Adult Acute Lymphoblastic Leukemia Untreated Childhood Acute Lymphoblastic Leukemia Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT00557193 | - | - | Completed | - | - |
| NCT02101853 | B Acute Lymphoblastic Leukemia | Phase 3 | Recruiting | - | - |
| NCT02757625 | ICU Sedation | Phase 3 | Completed | - | Japan ... more >> Asahikawa Medical University Hospital Asahikawa, Hokkaido, Japan, 078-8510 Hyogo Prefectural Kobe Children's Hospital Kobe, Hyogo, Japan, 650-0047 Shikoku Medical Center for Children and Adults Zentsuji, Kagawa, Japan, 765-8507 Osaka Women's and Children's Hospital Izumi, Osaka, Japan, 594-1101 Osaka Medical College Hospital Takatsuki, Osaka, Japan, 569-8686 Jichi Medical University Hospital Shimotsuke, Tochigi, Japan, 329-0498 Juntendo University Hospital Bunkyo-ku, Tokyo, Japan, 113-8431 Tokyo Metropolitan Children's Medical Center Fuchu, Tokyo, Japan, 183-8561 University Hospital, Kyoto Prefectural University of Medicine Kyoto, Japan, 602-8566 Okayama University Hospital Okayama, Japan, 700-8558 Osaka City General Hospital Osaka, Japan, 534-0021 Shizuoka Children's Hospital Shizuoka, Japan, 420-8660 Collapse << |
| NCT01371981 | Acute Myeloid Leukemia ... more >> Leukemia Cutis Myeloid Neoplasm Myeloid Sarcoma Untreated Adult Acute Myeloid Leukemia Untreated Childhood Myeloid Neoplasm Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT02521493 | Blasts 5 Percent or More of Bo... more >>ne Marrow Nucleated Cells Childhood Acute Myeloid Leukemia Childhood Myelodysplastic Syndrome Cytopenia Down Syndrome Myeloid Leukemia Associated With Down Syndrome Myeloproliferative Neoplasm Trisomy 21 Trisomy 21 Mosaicism Collapse << | Phase 3 | Recruiting | - | - |
| NCT02757625 | - | - | Completed | - | - |
| NCT02956447 | Hyperprolactinemia ... more >> Hypogonadism Collapse << | Phase 2 | Recruiting | February 2021 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Isabella McDonald 617-726-8484 mghkisspeptinresearch@partners.org Collapse << |
| NCT03591471 | Henoch-Schönlein Purpura Nephr... more >>itis Collapse << | Phase 1 Phase 2 | Recruiting | December 2018 | China, Henan ... more >> Children's Hospital, The First Affiliated Hospital of HUTCM Recruiting Zhengzhou, Henan, China, 450000 Contact: Ying Ding, Director +86-13503840642 dingying3236@sina.com Contact: Xia Zhang, secretary +86-13838276039 zhangxia_125@163.com Principal Investigator: Ying Ding, Director The First Affiliated Hospital of Henan University of Traditional Chinese Medicine Recruiting Zhengzhou, Henan, China, 450000 Contact: Ying Ding, MD +86-371-66221361 dingying3236@sina.com Contact: Xia zhang, MD +86-371-13213210370 ardar123@sina.com Principal Investigator: Jiansheng Li, MD Collapse << |
| NCT00121667 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00797108 | Pneumonia, Bacterial | Phase 2 | Completed | - | United States, California ... more >> eStudySite, Inc. Chula Vista, California, United States, 91911 Sharp Chula Vista Medical Center Chula Vista, California, United States, 91911 eStudySite, Inc. Oceanside, California, United States, 92056 Tri-City Medical Center Oceanside, California, United States, 92056 United States, Illinois Medical Arts Associates, Ltd Moline, Illinois, United States, 61265 Trinity Medical Center Rock Island, Illinois, United States, 61201 United States, Minnesota Infectious Disease Minneapolis Limited Minneapolis, Minnesota, United States, 55422 United States, Ohio Summa Health System Akron, Ohio, United States, 44304 Summa Health System Akron, Ohio, United States, 44309 Summa Health System Akron, Ohio, United States, 44310 United States, Utah Utah Valley Pulmonary Clinic Provo, Utah, United States, 84604 Utah Valley Regional Medical Center Provo, Utah, United States, 84604 Australia, Queensland Infection Management Services, Building 17, Level 1 Brisbane, Queensland, Australia, 4102 Canada, Ontario Hamilton Health Sciences - General Site Hamilton, Ontario, Canada, L8L 2X2 Hamilton Health Sciences- McMaster Site Hamilton, Ontario, Canada, L8N 3Z5 Hamilton Health Sciences - Henderson Site Hamilton, Ontario, Canada, L8V 1C3 Korea, Republic of Asan Medical Center, Division of Infectious Diseases Seoul, Korea, Republic of, 138-736 Poland Oddzial Chorob Wewnetrznych i Gastroenterologii Bialystok, Poland, 15-003 Oddzial Chorob Pluc Brzesko, Poland, 32-800 Kliniczny Oddzial Gruzlicy i Chorob Pluc Krakow, Poland, 30-901 Oddzial Kliniczny Pulmonologii i Alergologii Lodz, Poland, 90-153 Oddzial Pulmonologiczny III Poznan, Poland, 60-569 Oddzial Pulmonologiczny Proszowice, Poland, 32-100 II Oddzial Chorob Wewnetrznych Warszawa, Poland, 03-401 Collapse << |
| NCT00797108 | - | - | Completed | - | - |
| NCT00720109 | - | - | Completed | - | - |
| NCT01889238 | - | - | Active, not recruiting | - | - |
| NCT00121641 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00121667 | - | - | Completed | - | - |
| NCT02418650 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Clinical Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT02538926 | B Acute Lymphoblastic Leukemia... more >> B Lymphoblastic Lymphoma Recurrent Adult Acute Lymphoblastic Leukemia Recurrent B Lymphoblastic Lymphoma Recurrent T Lymphoblastic Leukemia/Lymphoma Refractory B Lymphoblastic Lymphoma Refractory T Lymphoblastic Lymphoma T Acute Lymphoblastic Leukemia T Lymphoblastic Lymphoma Collapse << | Phase 2 | Withdrawn(Drugs unavailable) | July 30, 2021 | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT00121641 | - | - | Completed | - | - |
| NCT01628120 | Chronic Renal Failure Requirin... more >>g Hemodialysis Collapse << | Phase 3 | Completed | - | - |
| NCT01628107 | - | - | Completed | - | - |
| NCT01628120 | - | - | Completed | - | - |
| NCT01006122 | Excessive Daytime Sleepiness ... more >> Narcolepsy Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Pfizer Investigational Site Phoenix, Arizona, United States, 85006 Pfizer Investigational Site Phoenix, Arizona, United States, 85051 Pfizer Investigational Site Tucson, Arizona, United States, 85712 United States, California Pfizer Investigational Site Los Angeles, California, United States, 90048 Pfizer Investigational Site Orange, California, United States, 92868 United States, Florida Pfizer Investigational Site Brandon, Florida, United States, 33511 Pfizer Investigational Site St. Petersburg, Florida, United States, 33707 United States, Georgia Pfizer Investigational Site Atlanta, Georgia, United States, 30342 Pfizer Investigational Site Macon, Georgia, United States, 31201 United States, Illinois Pfizer Investigational Site Vernon Hills, Illinois, United States, 60061 United States, Kentucky Pfizer Investigational Site Crestview Hills, Kentucky, United States, 41017 Pfizer Investigational Site Louisville, Kentucky, United States, 40217 United States, Mississippi Pfizer Investigational Site Hattiesburg, Mississippi, United States, 39401 Pfizer Investigational Site Hattiesburg, Mississippi, United States, 39402 United States, North Carolina Pfizer Investigational Site Durham, North Carolina, United States, 27710 United States, Ohio Pfizer Investigational Site Dublin, Ohio, United States, 43017 United States, Pennsylvania Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19114 Pfizer Investigational Site Philadelphia, Pennsylvania, United States, 19139 United States, South Carolina Pfizer Investigational Site Columbia, South Carolina, United States, 29201 United States, Texas Pfizer Investigational Site Austin, Texas, United States, 78731 Pfizer Investigational Site Houston, Texas, United States, 77063 Collapse << |
| NCT01889238 | Advanced, Androgen Receptor Po... more >>sitive Triple Negative Breast Cancer Collapse << | Phase 2 | Active, not recruiting | June 30, 2019 | - |
| NCT00942968 | Cancer | Phase 4 | Completed | - | - |
| NCT01006122 | - | - | Completed | - | - |
| NCT02366637 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Terminated(Study terminated on... more >> 7 April 2015 for business reasons. No safety and/or efficacy concerns contributed to the termination of the study) Collapse << | - | United Kingdom ... more >> Medicines Evaluation Unit Ltd Wythenshawe, Manchester, United Kingdom, M23 9QZ Nottingham University Hospital NHS Trust Nottingham, Nottinghamshire, United Kingdom, NG5 1PB University Hospitals Birmingham NHS Foundation Trust Birmingham, United Kingdom, B15 2WB Heart of England NHS Foundation Trust Birmingham, United Kingdom, B9 5SS Respiratory Medicine, Bradford Institute of Health Research Bradford, United Kingdom, BD9 6RJ Collapse << |
| NCT00565812 | Osteoarthritis | Phase 2 | Completed | - | - |
| NCT01821131 | Above Optimal Plasma LDL Conce... more >>ntrations Collapse << | Not Applicable | Active, not recruiting | September 2018 | Canada, Manitoba ... more >> Asper Clinical Research Institute Winnipeg, Manitoba, Canada, R2H 2A6 Canada, Ontario Glycemic Index Laboratories Inc. Toronto, Ontario, Canada, M5C 2N8 Collapse << |
| NCT01628107 | Chronic Renal Failure Requirin... more >>g Hemodialysis Collapse << | Phase 3 | Completed | - | - |
| NCT02484768 | Restless Legs Syndrome | Phase 2 | Withdrawn(as per GCP sponsor/c... more >>ompany decision) Collapse << | - | - |
| NCT00974311 | Castration-Resistant Prostate ... more >>Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00431236 | Nausea and Vomiting, Chemother... more >>apy-Induced Collapse << | Phase 3 | Completed | - | - |
| NCT00942968 | - | - | Completed | - | - |
| NCT01726777 | Type 2 Diabetes Mellitus ... more >> Vitamin D Deficiency Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> University of Guelph Guelph, Ontario, Canada, N1G 2W1 Glycemic Index Laboratories Toronto, Ontario, Canada, M5C 2N8 Canada, Quebec Institut de recherches cliniques de Montréal Montreal, Quebec, Canada, H2W 1R7 Collapse << |
| NCT02790658 | Healthy | Not Applicable | Completed | - | - |
| NCT00974311 | - | - | Completed | - | - |
| NCT02366637 | - | - | Terminated(Study terminated on... more >> 7 April 2015 for business reasons. No safety and/or efficacy concerns contributed to the termination of the study) Collapse << | - | - |
| NCT01910259 | Secondary Progressive Multiple... more >> Sclerosis Collapse << | Phase 2 | Active, not recruiting | July 2018 | United Kingdom ... more >> Anne Rowling Regenerative Neurology Clinic, Royal Infirmary of Edinburgh Edinburgh, United Kingdom, EH16 4SA Gartnavel Royal Hospital, 1055 Great Western Road Glasgow, United Kingdom, G12 OXH Brighton and Sussex University Hospitals Haywards Heath, United Kingdom, RH16 4EX St James's University Hospital Leeds, United Kingdom, LS9 7TF The Walton Centre Liverpool, United Kingdom, L9 7LJ The National hospital for Neurology and Neurosurgery, University College London London, United Kingdom, WC1N 3BG The Royal Victoria Infirmary Newcastle, United Kingdom, NE1 4LP Queens Medical Centre Nottingham, United Kingdom, NG7 2UH John Radcliffe Hospital, Oxford University Hospitals NHS Trust Oxford, United Kingdom, OX3 9DU Derriford Hospital Plymouth, United Kingdom, PL6 8DH Royal Hallamshire Hospital Sheffield, United Kingdom, S10 2JF University Hospital of North Staffordshire Stoke-on-Trent, United Kingdom, ST4 7LN Royal Cornwall Hospital Truro, United Kingdom, TR1 3LJ Collapse << |
| NCT00107718 | Melanoma | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90089 GSK Investigational Site San Francisco, California, United States, 94115 GSK Investigational Site Santa Monica, California, United States, 90404-2104 United States, Connecticut GSK Investigational Site New Haven, Connecticut, United States, 06520 United States, Florida GSK Investigational Site Jacksonville, Florida, United States, 32209 GSK Investigational Site Miami Beach, Florida, United States, 33140 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60637 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46202 United States, Maryland GSK Investigational Site Lutherville-Timonium, Maryland, United States, 21093 United States, New York GSK Investigational Site New York, New York, United States, 10016 United States, Ohio GSK Investigational Site Toledo, Ohio, United States, 43614-5809 United States, Pennsylvania GSK Investigational Site Pittsburgh, Pennsylvania, United States, 15213-2584 Australia, New South Wales GSK Investigational Site Waratah, New South Wales, Australia, 2298 GSK Investigational Site Westmead, New South Wales, Australia, 2145 Australia, Queensland GSK Investigational Site Douglas, Queensland, Australia, 4814 GSK Investigational Site South Brisbane, Queensland, Australia, 4101 GSK Investigational Site Woolloongabba, Queensland, Australia, 4102 Australia, Tasmania GSK Investigational Site Hobart, Tasmania, Australia, 7000 Australia, Victoria GSK Investigational Site East Melbourne, Victoria, Australia, 3002 Australia, Western Australia GSK Investigational Site Nedlands, Western Australia, Australia, 6009 Collapse << |
| NCT00565812 | - | - | Completed | - | - |
| NCT03693300 | Non-small Cell Lung Cancer (NS... more >>CLC) Collapse << | Phase 2 | Not yet recruiting | December 30, 2022 | United States, Tennessee ... more >> Research Site Not yet recruiting Knoxville, Tennessee, United States, 37920 France Research Site Not yet recruiting Saint Priest en Jarez, France, 42270 United Kingdom Research Site Not yet recruiting Leeds, United Kingdom, LS9 7TF Collapse << |
| NCT03470376 | Hypercholesterolemia ... more >> Inflammation Atherosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT02655315 | Parkinson Disease | Phase 2 | Recruiting | February 2021 | Austria ... more >> Medizinische Universitat Innsbruck Recruiting Innsbruck, Austria Principal Investigator: Werner Poewe, MD, PhD Czechia Univerzita Karlova V Praze Recruiting Prague, Czechia Principal Investigator: Evzen Ruzicka, MD,PhD Charles University Recruiting Praha, Czechia Principal Investigator: Evzen Ruzicka, MD France CHU Pellegrin Recruiting Bordeaux, France Principal Investigator: Wassilios Meissner, MD,PhD Hôpital Wertheimer Recruiting Bron, France Principal Investigator: Stéphane Thobois, MD,PhD Hôpital Montpied Recruiting Clermont-Ferrand, France Principal Investigator: Frank Durif, MD,PhD Hôpital Salengro, CHRU Recruiting Lille, France CHU la TIMONE Recruiting Marseille, France Principal Investigator: Alexandre Eusebio, MD, PhD AP-HP, Hôpital Pitié-Salpêtrière Recruiting Paris, France Principal Investigator: Jean-Christophe Corvol, MD, PhD CHU de Strasbourg, Hôpital de Hautepierre Recruiting Strasbourg, France Principal Investigator: Christine Tranchant, MD,PhD Chu Purpan Recruiting Toulouse, France Principal Investigator: Olivier Rascol, MD,PhD Germany University Hospital, Saarland University Not yet recruiting Homburg, Germany Principal Investigator: Stefanie Behnke, MD Christian-albrechts universität zu kiel Recruiting Kiel, Germany Principal Investigator: Daniela Berg, MD,PhD Klinik und Poliklinik für Neurologie der Universitätsmedizin Rostock Recruiting Rostock, Germany Principal Investigator: Uwe Walter, MD,PhD Netherlands Acadamic central center, Amsterdam Recruiting Amsterdam, Netherlands Principal Investigator: Rob De Bie, MD Radboud university medical center Recruiting Nijmegen, Netherlands Principal Investigator: Bart Post, MD Portugal Centro Hospitalar e universitario de Coimbra Not yet recruiting Coimbra, Portugal Principal Investigator: Cristina Januario, MD Centro Hospitalar do Alto Ave Recruiting Guimarães, Portugal Principal Investigator: Miguel Gago, MD Centro Hospitalar Lisboa Norte Not yet recruiting Lisbon, Portugal Principal Investigator: Joaquim Ferreira, MD Spain Hospital Clinic Universitari de Barcelona Recruiting Barcelona, Spain Principal Investigator: Eduard Tolosa, MD,PhD Hospital de Bellvitge Not yet recruiting Barcelona, Spain Principal Investigator: Matilde Calopa, MD,Phd Hospital de la Santa Creu i Sant Pau Recruiting Barcelona, Spain Principal Investigator: Jaime Kulisevsky, MD,PhD United Kingdom Cambridge University Hospital Not yet recruiting Cambridge, United Kingdom Principal Investigator: Paul Worth, MD University of Glasgow Terminated Glasgow, United Kingdom Newcastle University Not yet recruiting Newcastle, United Kingdom Principal Investigator: David Burn, MD,PhD Collapse << |
| NCT02737306 | Graft Vs Host Disease | Phase 2 | Recruiting | December 2019 | United States, Florida ... more >> University of Miami Sylvester Comprehensive Cancer Center Recruiting Miami, Florida, United States, 33136 United States, Illinois Loyola University Medical Center Cardinal Bernardin Cancer Center Recruiting Maywood, Illinois, United States, 60153 United States, Michigan Barbara Ann Karmanos Cancer Institute Recruiting Detroit, Michigan, United States, 48201 United States, Minnesota University of Minnesota Recruiting Minneapolis, Minnesota, United States, 55409 United States, North Carolina Wake Forest Baptist Health Recruiting Winston-Salem, North Carolina, United States, 27157 United States, Pennsylvania University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 United States, Texas Texas Transplant Institute Methodist Hospital Recruiting San Antonio, Texas, United States, 78229 United States, West Virginia West Virginia University Medicine Recruiting Morgantown, West Virginia, United States, 26506 Collapse << |
| NCT00281684 | Toothache | Phase 2 | Completed | - | Italy ... more >> GSK Investigational Site Verona, Veneto, Italy, 37134 Korea, Republic of GSK Investigational Site Seoul, Korea, Republic of, 110-768 United Kingdom GSK Investigational Site Croydon, Surrey, United Kingdom, CR7 7YE GSK Investigational Site Leeds, United Kingdom, LS2 9NG GSK Investigational Site Manchester, United Kingdom, M13 9WL Collapse << |
| NCT03635567 | Cervical Cancer | Phase 3 | Recruiting | November 23, 2022 | - |
| NCT03528681 | Hyperkalemia | Phase 3 | Not yet recruiting | October 7, 2019 | - |
| NCT00569062 | Depressive Disorder, Major | Phase 2 | Terminated(Negative data from ... more >>a GSK PoC study in RA combined with evidence of greater sensitivity of toll-like receptor pathways to p38 inhibition.) Collapse << | - | Estonia ... more >> GSK Investigational Site Tartu, Estonia, 50417 India GSK Investigational Site Bangalore, India, 560029 GSK Investigational Site Bangalore, India, 560034 GSK Investigational Site Ludhiana, India, 141001 GSK Investigational Site Manipal,, India, 576 104 GSK Investigational Site Mumbai, India, 400010 GSK Investigational Site Pune, India, 411004 Russian Federation GSK Investigational Site Moscow, Russian Federation, 107076 GSK Investigational Site Moscow, Russian Federation, 115522 GSK Investigational Site Moscow, Russian Federation, 119992 GSK Investigational Site Moscow, Russian Federation, 123367 GSK Investigational Site Nizhny Novgorod, Russian Federation, 603115 GSK Investigational Site Smolensk, Russian Federation, 214 019 GSK Investigational Site St-Petersburg, Russian Federation GSK Investigational Site St.Petersburg, Russian Federation, 193167 Collapse << |
| NCT01641393 | Exocrine Pancreatic Insufficie... more >>ncy: Cystic Fibrosis Collapse << | Phase 3 | Completed | - | - |
| NCT00549744 | Asthma | Phase 2 | Completed | - | New Zealand ... more >> GSK Investigational Site Wellington, New Zealand, 6035 South Africa GSK Investigational Site George, Eastern Cape, South Africa, 6529 GSK Investigational Site Bloemfontein, South Africa, 9301 Collapse << |
| NCT01817166 | Multiple Sclerosis | Phase 3 | Recruiting | January 2022 | France ... more >> CHU d'Amiens - Hôpital Nord Not yet recruiting Amiens Cedex 1, France, 80054 Sub-Investigator: Olivier Godefroy, MD, PhD Sub-Investigator: Abdullatif Al Khedr, MD CHU de Lyon - Hôpital Pierre Wertheimer Recruiting Bron, France, 69677 Sub-Investigator: Christian Confavreux, MD, PHD Sub-Investigator: Sandra Vukusic, MD, PhD CHU de Caen - Hôpital Côte de Nacre Recruiting Caen Cedex 9, France, 14033 Sub-Investigator: Gilles Defer, MD, PhD Sub-Investigator: Nathalie Derache Belpalme, MD CHU de Clermont Ferrand - Hôpital Gabriel-Montpied Recruiting Clermont Ferrand, France, 63003 Sub-Investigator: Pierre Clavelou, MD, PhD Sub-Investigator: Frédéric Thaite, MD CH Sud Francilien Recruiting Corbeil-Essonnes, France, 91100 Sub-Investigator: Ivania Patry, MD Clinique des Cèdres - Capio Not yet recruiting Cornebarrieu, France, 31700 Sub-Investigator: Claude Mekies, MD CHU de Dijon Recruiting Dijon, France, 21079 Sub-Investigator: Thibault Moreau, MD, PhD CHU de Martinique - Hôpital Pierre Zobda-Quitman Not yet recruiting Fort-de-France, France, 97261 Sub-Investigator: Philippe Cabre, MD CHU de Grenoble - Hôpital A Michallon Recruiting Grenoble, France, 38043 Sub-Investigator: Olivier Casez, MD CHRU de Lille - Hôpital Roger Salengro Recruiting Lille, France, 59037 Sub-Investigator: Patrick Vermersch, MD, PhD Sub-Investigator: Hélène Zephyr, MD Sub-Investigator: Olivier Outteryck, MD CHU de Limoges - Hôpital Dupuytren Recruiting Limoges, France, 87042 Sub-Investigator: Laurent Magy, MD, PhD Sub-Investigator: Alexis Montcuquet, MD Groupe Hospitalier de l'Institut Catholique de Lille Recruiting Lomme Cedex, France, 59462 Sub-Investigator: Patrick Hautecoeur, MD Sub-Investigator: Cécile Donzé-Parret, MD CHU de Montpellier - Hôpital Gui de Chauliac Recruiting Montpellier, France, 34295 Sub-Investigator: William Camu, MD, PhD Sub-Investigator: Bertrand Carlander, MD Sub-Investigator: Mahmoud Charif, MD Sub-Investigator: Pierre Labauge, MD, PhD CHU de Nancy - Hôpital Central Recruiting Nancy, France, 54035 Sub-Investigator: Marc Debouverie, MD, PhD Sub-Investigator: Sophie Pittion-Vouyovitch, MD CHU de Nantes - Hôtel-Dieu Recruiting Nantes, France, 44093 Sub-Investigator: Sandrine Wiertlewski, MD Sub-Investigator: David Laplaud, MD CHU de Nice - Hôpital Pasteur Recruiting Nice, France, 06002 Sub-Investigator: Christine Lebrun-Fresnay, MD CHU de Nîmes - Hôpital Universitaire Carémeau Recruiting Nîmes Cedex 9, France, 30029 Principal Investigator: Eric Thouvenot, MD, PhD Sub-Investigator: Giovanni Castelnovo, MD APHP - Hôpital Saint-Antoine Not yet recruiting Paris Cedex 12, France, 75571 Sub-Investigator: Bruno Stankoff, MD, PhD Sub-Investigator: Claire Giannesini, MD MAILLART Elisabeth - La Pitié Salpétrière Not yet recruiting Paris, France, 75013 Contact: MAILLART Elisabeth, PH Fondation Ophtalmologique Adolphe Rothschild Recruiting Paris, France, 75019 Sub-Investigator: Olivier Gout, MD CH de Pau Not yet recruiting Pau, France, 64000 Sub-Investigator: Mickaël Bonnan, MD CH de Perpignan - Hôpital Saint Jean Recruiting Perpignan, France, 66046 Sub-Investigator: Nicolas Gaillard, MD Sub-Investigator: Anais Dutray, MD CH de Cornouaille - Site Quimper - Hôpital Laennec Recruiting Quimper, France, 29107 Sub-Investigator: Marc Coustans, MD CHU de Reims - Hôpital Maison Blanche Recruiting Reims, France, 51092 Sub-Investigator: Ayman Tourbah, MD CHU de Rennes - Hôpital PontChaillou Recruiting Rennes, France, 35033 Sub-Investigator: Gilles Edan, MD, PhD Sub-Investigator: Emmanuelle Le Page, MD Sub-Investigator: Véronique Deburghgraeve, MD CHU de Rouen - Hôpital Charles Nicolle Recruiting Rouen, France, 76031 Sub-Investigator: Didier Hannequin, MD, PhD Sub-Investigator: Bertrand Bourre, MD CH de Poissy - Saint-Germain-en-Laye Recruiting Saint-Germain-en-Laye, France, 78100 Sub-Investigator: Olivier Heinzlef, MD CH de Saumur Recruiting Saumur Cedex, France, 49403 Sub-Investigator: Richard Devy, MD CHRU de Strasbourg - Hôpital Civil Recruiting Strasbourg Cedex, France, 67091 Sub-Investigator: Jérôme De Sèze, MD, PhD CHRU de Toulouse - Hôpital Purpan Recruiting Toulouse Cedex 9, France, 31059 Sub-Investigator: David Brassat, MD, PhD CHRU de Tours - Hôpital Bretonneau Recruiting Tours, France, 37044 Sub-Investigator: Philippe Corcia, MD, PhD Sub-Investigator: Anne-Marie Guennoc, MD CH de Versailles - Hôpital Mignot Recruiting Versailles, France, 78000 Sub-Investigator: Fernando Pico, MD, PhD Sub-Investigator: Chantal Nifle, MD CH de Vichy - Jacques Larin Not yet recruiting Vichy, France, 03207 Sub-Investigator: Renato Colamarino, MD Collapse << |
| NCT02461992 | - | - | Completed | - | - |
| NCT02461992 | Hemophilia A | Phase 1 | Completed | - | China ... more >> Phase I Unit, Clinical Pharmacology Research Center, Peking Union Medical College Hospital Beijing, China, 100032 Hematology Department,Beijing Children's Hospital, Capital Medical University Beijing, China, 100045 Collapse << |
| NCT02278861 | Actinic Keratosis | Phase 2 | Completed | - | Brazil ... more >> Dermatology Outpatient Clinic - Hospital das Clínicas - Federal University of Goias (UFG) Goiânia, Goiás, Brazil, 74605-020 Collapse << |
| NCT01891240 | - | - | Unknown | April 2018 | Ireland ... more >> Cork University Maternity Hospital, University College Cork Recruiting Wilton, Cork, Ireland Contact: Louise Kenny +353 (0)21 420 5023 l.kenny@ucc.ie Netherlands Erasmus Medical Center Rotterdam Recruiting Rotterdam, Netherlands Contact: Hans Duvekot j.j.duvekot@erasmusmc.nl Sweden Karolinska University Hospital Huddinge, Karolinska Institute Recruiting Stockholm, Sweden Contact: Karolina Kublickiene +46 735 930988 karolina.kublickiene@ki.se United Kingdom Liverpool Women's Hospital, University of Liverpool Recruiting Liverpool, Merseyside, United Kingdom Contact: Zarko Alfirevic +44 (0)151 795 9550 Zarko@liverpool.ac.uk Keele University School of Medicine, Shrewsbury and Telford Hospital NHS Trust Completed Shrewsbury, Shropshire, United Kingdom Keele University School of Medicine, University Hospital of North Midlands Completed Stoke-on-Trent, Staffordshire, United Kingdom Collapse << |
| NCT00111787 | Neoplasms, Breast | Phase 2 | Completed | - | - |
| NCT02566031 | COPD | Phase 4 | Completed | - | Korea, Republic of ... more >> Novartis Investigative Site Chuncheon, Gangwon-do, Korea, Republic of, 24253 Novartis Investigative Site Wonju, Gangwon-Do, Korea, Republic of, 26426 Novartis Investigative Site Anyang-si, Gyeonggi-do, Korea, Republic of, 14068 Novartis Investigative Site Guri-si, Gyeonggi-do, Korea, Republic of, 471-701 Novartis Investigative Site Seongnam-si, Gyeonggi-do, Korea, Republic of, 463-712 Novartis Investigative Site Suwon-si, Gyeonggi-do, Korea, Republic of, 16499 Novartis Investigative Site Jeonju-si, Jeollabuk-do, Korea, Republic of, 561-712 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 02559 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 06351 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 08308 Novartis Investigative Site Koyang, Kyunggi, Korea, Republic of, 410-719 Novartis Investigative Site Gwangju, Korea, Republic of, 61469 Novartis Investigative Site Incheon, Korea, Republic of, 403-720 Novartis Investigative Site Incheon, Korea, Republic of, 405 760 Novartis Investigative Site Jeju, Korea, Republic of, 63241 Novartis Investigative Site Seoul, Korea, Republic of, 03722 Novartis Investigative Site Seoul, Korea, Republic of, 04763 Novartis Investigative Site Seoul, Korea, Republic of, 05030 Novartis Investigative Site Seoul, Korea, Republic of, 06273 Novartis Investigative Site Seoul, Korea, Republic of, 06591 Novartis Investigative Site Seoul, Korea, Republic of, 110-746 Novartis Investigative Site Seoul, Korea, Republic of, 150-713 Novartis Investigative Site Seoul, Korea, Republic of, 156-707 Novartis Investigative Site Ulsan, Korea, Republic of, 44033 Collapse << |
| NCT02178592 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Active, not recruiting | December 31, 2019 | - |
| NCT02799264 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT02424539 | Rhinitis, Allergic, Perennial ... more >>and Seasonal Collapse << | Phase 4 | Completed | - | China, Fujian ... more >> GSK Investigational Site Xiamen, Fujian, China, 361003 China, Guangdong GSK Investigational Site Shenzhen, Guangdong, China, 518038 China, Hunan GSK Investigational Site Changsha, Hunan, China, 410005 GSK Investigational Site Changsha, Hunan, China, 410011 China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210029 China, Shanxi GSK Investigational Site Taiyuan, Shanxi, China, 030016 China, Zhejiang GSK Investigational Site Wenzhou, Zhejiang, China, 323027 China GSK Investigational Site Beijing, China, 100034 GSK Investigational Site Beijing, China GSK Investigational Site Changsha, China GSK Investigational Site Chongqing, China, 400014 GSK Investigational Site Fuzhou, China, 350025 GSK Investigational Site Hangzhou, China, 310052 GSK Investigational Site Shanghai, China, 200040 GSK Investigational Site Shanghai, China, 200092 GSK Investigational Site Shanghai, China, 200127 Collapse << |
| NCT00390845 | Pain, Neuropathic | Phase 2 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Broadmeadow, New South Wales, Australia, 2292 GSK Investigational Site Randwick, New South Wales, Australia, 2031 Germany GSK Investigational Site Wuerzburg, Bayern, Germany, 97080 GSK Investigational Site Hannover, Niedersachsen, Germany, 30625 GSK Investigational Site Bochum, Nordrhein-Westfalen, Germany, 44789 GSK Investigational Site Koeln, Nordrhein-Westfalen, Germany, 50937 Russian Federation GSK Investigational Site Moscow, Russian Federation, 117292 United Kingdom GSK Investigational Site Cambridge, Cambridgeshire, United Kingdom, CB2 0QQ GSK Investigational Site Glasgow, Lanarkshire, United Kingdom, G12 0YN GSK Investigational Site Solihull, West Midlands, United Kingdom, B91 2JL GSK Investigational Site Liverpool, United Kingdom, L9 7LJ GSK Investigational Site London, United Kingdom, W12 0NN GSK Investigational Site London, United Kingdom, WC1X 8LD Collapse << |
| NCT02689414 | Abdominal Aortic Aneurysm ... more >> Peripheral Artery Disease Carotid Artery Stenosis Collapse << | Not Applicable | Completed | - | Estonia ... more >> Tartu University Hospital Tartu, Tartu County, Estonia, 50406 Collapse << |
| NCT03665506 | - | - | Recruiting | June 2021 | Germany ... more >> Onkologische Schwerpunktpraxis Kurfürstendamm Recruiting Berlin, Germany, 10707 Contact: Ingo Schwaner, MD +49 30 887742570 ingo.schwaner@onkologie-kurfuerstendamm.de Collapse << |
| NCT02147587 | - | - | Completed | - | - |
| NCT03610334 | Healthy Volunteers | Phase 1 | Recruiting | July 2019 | France ... more >> Centre d'Investigation Clinique - Centre de Pharmacologie Clinique et d'Evaluations Thérapeutiques (CIC-CPCET) Recruiting Marseille, France, 13385 Contact: Christine Audebert, MD 491384641 ext +33 christine.audebert@ap-hm.fr Contact: Laurence Attolini, PhD 491388003 ext +33 laurence.attolini@ap-hm.fr Principal Investigator: Christine Audebert, MD Sub-Investigator: Marie-Noelle Lefebvre, MD Sub-Investigator: Frank Rouby, MD Collapse << |
| NCT02639975 | Glaucoma | Phase 1 | Completed | - | - |
| NCT02147587 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT01302938 | Overactive Bladder | Phase 4 | Terminated(Stop date for rando... more >>mization 31/5/2012. Recruitment terminated due to lack of recruitment. No new safety issues were identified.) Collapse << | - | United States, California ... more >> Mytrus, Inc. San Francisco, California, United States, 94104 University of California San Francisco San Francisco, California, United States, 94121 Collapse << |
| NCT00562965 | Lymphoma, Follicular | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT02956746 | Human Rabies | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> inVentiv Clinical Research Facility, 1951 NW 7th Ave. Suit 450 Miami, Florida, United States, 33136 Collapse << |
| NCT01302938 | - | - | Terminated(Stop date for rando... more >>mization 31/5/2012. Recruitment terminated due to lack of recruitment. No new safety issues were identified.) Collapse << | - | - |
| NCT02503644 | Scleroderma, Diffuse ... more >> Diffuse Cutaneous Systemic Sclerosis Collapse << | Phase 2 | Active, not recruiting | November 2018 | - |
| NCT00562965 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT02324777 | Osteoarthritis, Knee | Phase 2 | Recruiting | June 2019 | Canada, Nova Scotia ... more >> Pain Management Unit, Queen Elizabeth II Health Sciences Centre Recruiting Halifax, Nova Scotia, Canada, B3H 1V7 Contact: Melissa Todd (902) 473-7475 melissa.todd@nshealth.ca Principal Investigator: Mary Lynch, MD Canada, Quebec Montreal General Hospital - McGill University Health Centre Recruiting Montreal, Quebec, Canada, H3G 1A4 Contact: Mark Ware, MRCP(UK) 514 934-1934 ext 42784 mark.ware@mcgill.ca Collapse << |
| NCT01233869 | - | - | Completed | - | - |
| NCT00381394 | Leishmaniasis, Visceral | Phase 2 | Completed | - | India ... more >> GSK Investigational Site Muzaffarpur, India, 842001 GSK Investigational Site Muzaffarpur, India GSK Investigational Site Patna, India, 800007 Collapse << |
| NCT00381680 | B-cell Childhood Acute Lymphob... more >>lastic Leukemia L1 Childhood Acute Lymphoblastic Leukemia L2 Childhood Acute Lymphoblastic Leukemia Intermediate Risk Recurrent Childhood Acute Lymphoblastic Leukemia Collapse << | Phase 3 | Completed | - | - |
| NCT01703624 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02231021 | Type 2 Diabetes Mellitus | Phase 4 | Recruiting | November 2018 | Korea, Republic of ... more >> Seoul St Mary's Hospital, The Catholic University of Korea Recruiting Seoul, Korea, Republic of, 137-701 Principal Investigator: Kun-Ho Yoon, MD, PhD Sub-Investigator: Yoon-Hee Choi, MD, PhD Sub-Investigator: Hye-Kyung Yang, MD Collapse << |
| NCT00016302 | T-cell Childhood Acute Lymphob... more >>lastic Leukemia Untreated Childhood Acute Lymphoblastic Leukemia Collapse << | Not Applicable | Completed | - | United States, California ... more >> Children's Oncology Group Arcadia, California, United States, 91006-3776 Collapse << |
| NCT00256867 | - | - | Completed | - | - |
| NCT03272230 | Apathy Fronto... more >>temporal Dementia Parkinson Disease Depressive Symptoms Collapse << | Not Applicable | Recruiting | July 31, 2019 | France ... more >> ICM / Pitié-Salpêtrière Hospital AP-HP / CIC Recruiting Paris, France, 75013 Contact: Armelle Ramettilacroux, Master armelle.ramettilacroux@icm-institute.org Collapse << |
| NCT00700401 | - | - | Completed | - | Brazil ... more >> Brasilia, DF, Brazil, 70335900 Vitoria, ES, Brazil, 29043-260 Sao Luis, MA, Brazil, 65020560 Rio de Janeiro, RJ, Brazil, 20020-022 Porto Alegre, RS, Brazil, 90020-090 Porto Alegre, RS, Brazil, 90035-003 Campinas, SP, Brazil, 13060-803 Campinas, SP, Brazil, 13083-888 Ribeirao Preto, SP, Brazil, 14049-900 Santo Andre, SP, Brazil, 09060-650 Sao Paulo, SP, Brazil, 04040-003 Sorocaba, SP, Brazil, 18047-600 Collapse << |
| NCT01233869 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 2 | Completed | - | - |
| NCT00381680 | - | - | Completed | - | - |
| NCT00700401 | - | - | Completed | - | - |
| NCT02210091 | Hemophilia A | Phase 3 | Completed | - | - |
| NCT00087568 | Hepatitis C, Chronic | Phase 4 | Completed | - | - |
| NCT02210091 | - | - | Completed | - | - |
| NCT00316693 | Infections, Papillomavirus | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Kagoshima, Japan, 892-0824 GSK Investigational Site Tokyo, Japan, 160-0017 GSK Investigational Site Tokyo, Japan, 183-0056 Collapse << |
| NCT00087568 | - | - | Completed | - | - |
| NCT00798707 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00863798 | Major Depressive Disorder | Phase 3 | Completed | - | United States, Alabama ... more >> Pfizer Investigational Site Birmingham, Alabama, United States, 35216 United States, California Pfizer Investigational Site Encino, California, United States, 91316 Pfizer Investigational Site Newport Beach, California, United States, 92660 Pfizer Investigational Site Redlands, California, United States, 92374 Pfizer Investigational Site Upland, California, United States, 91786 United States, Colorado Pfizer Investigational Site Aurora, Colorado, United States, 80045 Pfizer Investigational Site Denver, Colorado, United States, 80204 United States, Connecticut Pfizer Investigational Site Cromwell, Connecticut, United States, 06416 United States, Florida Pfizer Investigational Site Maitland, Florida, United States, 32751 Pfizer Investigational Site North Miami, Florida, United States, 33161 Pfizer Investigational Site South Miami, Florida, United States, 33143 United States, Indiana Pfizer Investigational Site Indianapolis, Indiana, United States, 46260 United States, Missouri Pfizer Investigational Site St. Louis, Missouri, United States, 63139 United States, New York Pfizer Investigational Site New York, New York, United States, 10128 Pfizer Investigational Site Staten Island, New York, United States, 10312 United States, Ohio Pfizer Investigational Site Toledo, Ohio, United States, 43623 United States, Oregon Pfizer Investigational Site Eugene, Oregon, United States, 97401 Pfizer Investigational Site Portland, Oregon, United States, 97210 Pfizer Investigational Site Salem, Oregon, United States, 97301 United States, Pennsylvania Pfizer Investigational Site Media, Pennsylvania, United States, 19063 United States, Virginia Pfizer Investigational Site Herndon, Virginia, United States, 20170 Pfizer Investigational Site Midlothian, Virginia, United States, 23112 United States, Wisconsin Pfizer Investigational Site Middleton, Wisconsin, United States, 53562 Pfizer Investigational Site Waukesha, Wisconsin, United States, 53188 Collapse << |
| NCT00707239 | Pneumonia, Bacterial | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00707239 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT03093441 | Healthy | Not Applicable | Active, not recruiting | December 8, 2018 | United States, Michigan ... more >> Oakland University Rochester, Michigan, United States, 48309 Collapse << |
| NCT00683800 | Vasomotor Symptoms | Phase 3 | Completed | - | - |
| NCT00798707 | - | - | Completed | - | - |
| NCT00316693 | - | - | Completed | - | - |
| NCT01226563 | Acute Myocardial Infarction ... more >> Congestive Heart Failure ST-Elevation Myocardial Infarction Collapse << | Not Applicable | Completed | - | - |
| NCT03738761 | Hypertension | Phase 4 | Recruiting | April 2019 | Armenia ... more >> Unimed medical center Not yet recruiting Abovyan, Armenia Center of Preventive Cardiology Not yet recruiting Yerevan, Armenia Croatia Thalassotherapia Opatija, Specijalna bolnica za medicinsku rehabilitaciju, bolesti srca, pluća i reumatizma, Klinika za liječenje, rehabilitaciju i prevenciju bolesti srca i krvnih žila Recruiting Opatija, Croatia KBC Rijeka - Sušak, Klinika za internu medicinu, Zavod za nefrologiju, dijalizu i transplantaciju bubrega Recruiting RIjeka, Croatia KB Dubrava, Klinika za unutarnje bolesti, Zavod za nefrologiju i dijalizu Recruiting Zagreb, Croatia KB Merkur, Klinika za unutarnje bolesti, Zavod za kardiologiju Recruiting Zagreb, Croatia KBC Sestre milosrdnice, Klinika za bolesti srca i krvnih žila, Zavod za bolesti krvnih žila Recruiting Zagreb, Croatia KBC Zagreb, Klinika za bolesti srca i krvnih žila Recruiting Zagreb, Croatia KBC Zagreb, Klinika za unutarnje bolesti, Zavod za nefrologiju, arterijsku hipertenziju, dijalizu i transplantaciju Recruiting Zagreb, Croatia Poliklinika za prevenciju kardiovaskularnih bolesti i rehabilitaciju SRČANA Recruiting Zagreb, Croatia Hungary Biatros Kft. Recruiting Biatorbágy, Hungary AD SANITATEM Bt. Recruiting Pilisvörösvár, Hungary Poland Przychodnia Kardiologiczna w Zespole Przychodni Specjalistycznych DIAB-END-COR Active, not recruiting Krakow, Poland Niepubliczny Zakład Opieki Zdrowotnej Neuromed M.i M. Nastaj Spółka Partnerska Recruiting Lublin, Poland Poradnia Kardiologiczna, Nowodworskie Centrum Medyczne Active, not recruiting Nowy Dwór Mazowiecki, Poland Poradnia Kardiologiczna Samodzielnego Publicznego Zespołu Zakładów Opieki Zdrowotnej im. Marszałka Józefa Piłsudskiego w Płońsku Active, not recruiting Płońsk, Poland Poradnia Kardiologiczna w Niepublicznym Zakładzie Opieki Zdrowotnej Euro-Klinika Sp. z o.o. w Siemianowicach Śląskich Active, not recruiting Siemianowice Śląskie, Poland Poradnia internistyczno-kardiologiczna w Niepublicznym Zakładzie Opieki Zdrowotnej AURUM Active, not recruiting Warsaw, Poland Poradnia Kardiologiczna w Przychodni Rejonowej - Specjalistycznej (III) Samodzielnego Zespołu Publicznych Zakładów Lecznictwa Otwartego Warszawa - Ochota Recruiting Warsaw, Poland Przychodnia ACTIV-MED. Recruiting Warsaw, Poland Russian Federation Federal State Budgetary Scientific Institution "Scientific Research Institute of Complex Problems of Cardiovascular Diseases" Recruiting Kemerovo, Russian Federation Federal State budget organization National medical research center of cardiology of the Ministry of healthcare of the Russian Federation Recruiting Moscow, Russian Federation State Budgetary Institution of Healthcare of Moscow "City Clinical Hospital № 29 named after N.E. Bauman of Department of Healthcare of Moscow city" (SBIH "CCH № 29 Bauman") Not yet recruiting Moscow, Russian Federation Federal State Budgetary Institution "V.A. Almazov National Medical Research Center" of the Ministry of Health of the Russian Federation (V.A. Almazov NMRC) Recruiting Saint Petersburg, Russian Federation Federal State Budgetary Educational Institution Higher Education "Volgograd State Medical University" of the Ministry of Healthcare of the Russian Federation (FSBEI HE VolgSMU MOH Russia) Not yet recruiting Volgograd, Russian Federation Serbia Klinički centar Srbije Recruiting Belgrade, Serbia Klinički centar Niš Recruiting Niš, Serbia Inštitut za kardiovaskularne bolesti Vojvodine Recruiting Sremska Kamenica, Serbia Slovenia Univerzitetna klinika za pljučne bolezni in alergijo Golnik Recruiting Golnik, Slovenia Splošna bolnišnica Jesenice Recruiting Jesenice, Slovenia Zdravstveni Zavod CELJENJE Recruiting Koper, Slovenia Ordinacija interne medicine Verboten Kopriva Renata Recruiting Litija, Slovenia Cardial, d. o. o. Recruiting Ljubljana, Slovenia MC Medicor, d. d. Recruiting Ljubljana, Slovenia Intermed, d. o. o. Recruiting Maribor, Slovenia Srce in ožilje, d. o. o. Recruiting Maribor, Slovenia Collapse << |
| NCT02752867 | Cognitive Impairment | Not Applicable | Recruiting | April 2018 | China, Beijing ... more >> Department of Neurology,Dongfang Hospital,Beijing University of Chinese Medicine Recruiting Beijing, Beijing, China, 100078 Contact: Yunling Zhang, PhD,MD 86 10 67689634 Collapse << |
| NCT00359177 | Vomiting | Phase 1 | Completed | - | United States, Florida ... more >> GSK Investigational Site Gainesville, Florida, United States, 32608 GSK Investigational Site Orlando, Florida, United States, 32809 Collapse << |
| NCT00863798 | - | - | Completed | - | - |
| NCT00683800 | - | - | Completed | - | - |
| NCT02471573 | Infertility | Not Applicable | Completed | - | Vietnam ... more >> My Duc Hospital, IVFMD Ho Chi Minh City, Ho Chi Minh, Vietnam Collapse << |
| NCT02148471 | - | - | Completed | - | Canada, Ontario ... more >> Toronto General Hospital Toronto, Ontario, Canada, M5G 1Z5 Collapse << |
| NCT02423694 | Perimenopause ... more >> Depression Collapse << | Not Applicable | Unknown | - | China, Guangdong ... more >> Guangdong Provincial Hospital of Traditional Chinese Medicine Recruiting Guangzhou, Guangdong, China, 510120 Contact: Wenbin Fu, MD fuwenbin@139.com Contact: Zhaofeng Li qdlzfcmd@126.com Collapse << |
| NCT03657186 | Overweight We... more >>ight Loss Collapse << | Not Applicable | Recruiting | August 2019 | Germany ... more >> Analyze & Realize Recruiting Berlin, Germany, 10369 Contact: Udo Bongartz, Dr. med. Barbara Grube Recruiting Berlin, Germany, 10709 Contact: Barbara Grube Jörg Förstermann Recruiting Berlin, Germany, 14059 Contact: Jörg Förstermann Collapse << |
| NCT02164552 | - | - | Unknown | December 2014 | Saudi Arabia ... more >> Center of Excellence for Osteoporosis Research, King Abdulaziz University Recruiting Jeddah, Makkah, Saudi Arabia, 21589 Contact: Veronica B Orbacedo, BSc 0096612640000 ext 25574 vorbacedo@yahoo.com Contact: Ramia Al-sobhi, BSc 0096612640000 ext 25528 ceor@kau.edu.sa Sub-Investigator: Ahmed Y Ali, MD Sub-Investigator: Ghazi Y Refai, MD Sub-Investigator: Mohammed H Qari, FRCPA Sub-Investigator: Heba S Kary, MD Collapse << |
| NCT02210806 | Asthma | Phase 2 | Completed | - | United States, California ... more >> Amphastar Site 0001 San Jose, California, United States, 95117 United States, Oregon Amphastar Site 0025 Medford, Oregon, United States, 97504 United States, Texas Amphastar Site 0030 New Braunfels, Texas, United States, 78130 Amphastar Site 0032 San Antonio, Texas, United States, 78229 Collapse << |
| NCT02484027 | Infarction, Anterior Cerebral ... more >>Artery Collapse << | Phase 4 | Unknown | September 2018 | China, Jiangsu ... more >> Department of Neurology, Second Affiliated Hospital of Soochow University Not yet recruiting Suzhou, Jiangsu, China, 215004 Contact: Chun-Feng Liu, Ph.D, M.D. 00 86 512 67783307 liucf@suda.edu.cn Principal Investigator: Yong-Jun Cao, Ph.D, M.D. Principal Investigator: Wan-Qing Zhai, M.D Sub-Investigator: Xia Zhang, M.D. Collapse << |
| NCT02922140 | Acute Coronary Syndrome | Not Applicable | Unknown | August 2017 | Egypt ... more >> Ain Shams University Cairo, Egypt Collapse << |
| NCT02606097 | Gastrointestinal Stromal Tumou... more >>r (GIST) Collapse << | Phase 2 | Unknown | - | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Taoyuan, Taiwan, 333 Contact: Chun-Nan Yeh, MD 886-3-3281200 ext 3219 yehchunnan@gmail.com Collapse << |
| NCT02419105 | Hypogonadism ... more >>Pre-frail Seniors Collapse << | Phase 3 | Recruiting | March 2022 | Switzerland ... more >> Dept. of Geriatrics and Aging Research, University of Zurich / Geriatric Clinic University Hospital Zurich Recruiting Zürich, ZH, Switzerland, 8037 Contact: Heike A Bischoff-Ferrari, MD, DrPH (+41)44 2552699 Heike.Bischoff@usz.ch Principal Investigator: Heike A Bischoff-Ferrari, MD, DrPH Sub-Investigator: Andreas Egli Linder, MD Collapse << |
| NCT00814762 | AIDS | Phase 1 | Completed | - | Germany ... more >> GSK Investigational Site Erlangen, Bayern, Germany, 91054 GSK Investigational Site Muenchen, Bayern, Germany, 80335 GSK Investigational Site Koeln, Nordrhein-Westfalen, Germany, 50937 GSK Investigational Site Berlin, Germany, 12157 GSK Investigational Site Hamburg, Germany, 20099 GSK Investigational Site Hamburg, Germany, 20246 Collapse << |
| NCT00744497 | Prostatic Neoplasms | Phase 3 | Completed | - | - |
| NCT01021553 | - | - | Completed | - | - |
| NCT00111800 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00528372 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01021553 | Premature Ejaculation | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site San Diego, California, United States, 92120 GSK Investigational Site San Jose, California, United States, 95128 United States, Indiana GSK Investigational Site Fort Wayne, Indiana, United States, 46825 United States, New York GSK Investigational Site New York, New York, United States, 10016 United States, Pennsylvania GSK Investigational Site Bala-Cynwyd, Pennsylvania, United States, 19004 GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 Netherlands GSK Investigational Site Amsterdam, Netherlands, 1081 HV GSK Investigational Site Utrecht, Netherlands, 3584 CJ Collapse << |
| NCT00861614 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT01842607 | Asthma | Phase 3 | Completed | - | - |
| NCT00111800 | - | - | Completed | - | - |
| NCT00861614 | - | - | Completed | - | - |
| NCT00744497 | - | - | Completed | - | - |
| NCT00528372 | - | - | Completed | - | - |
| NCT00555880 | - | - | Completed | - | - |
| NCT01842607 | - | - | Completed | - | - |
| NCT00423319 | Deep Vein Thrombosis ... more >> Pulmonary Embolism Collapse << | Phase 3 | Completed | - | - |
| NCT00423319 | - | - | Completed | - | - |
| NCT00383708 | Acromegaly | Phase 3 | Completed | - | Czechia ... more >> University Hospital, Charles University Hradec Kralove, Czechia, 500 05 Charles University Prague, Czechia, 120 00 PRAHA 2 Denmark Aarhus Kommunehospital Aarhus, Denmark France Groupe Hospitalier Henri Mondor- Albert Chenevier Créteil Cedex, France Hôpital Bicêtre Le Kremlin-Bicêtre, France, 94275 Clinique Marc Linquette Lille Cedex, France, 59037 Hôpital de la Timone Marseille Cedex, France, 13385 CHU de Rangueil Toulouse, France, 31054 Germany Charite Campus Mitte Berlin, Germany, 10117 Klinikum Johann Wolfgang Goethe-Universität Frankfurt, Germany, 605090 Medizinische Klinik Innenstadt Munchen, Germany, 80336 Greece Anticancer Hospital Metaxa Piraeus Piraeus, Greece, 18537 Italy Universitá degli Studi di Milano Milano, Italy, 20122 University Federico II Napoli, Italy, 80131 Universitá di Torino Torino, Italy, 10126 Netherlands Leiden University Medical Center Leiden, Netherlands, 2300 RC Dept. of Internal Medicine Erasmus MC Rotterdam, Netherlands, 3015 GD Spain Hospital General de Alicante Alicante, Spain, 03012 Clínica Puerta de Hierro Madrid, Spain, 28035 Hospital Clínico Universitario de Santiago de Compostela Santiago de Compostela, Spain, 15706 Sweden Sahlgrenska University Hospital Göteborg, Sweden, 413 45 Uppsala University Hospital Uppsala, Sweden, 75185 United Kingdom Christie Hospital and Holt Radium Institute Manchester, United Kingdom, M20 4BX Royal Hallamshire Hospital Sheffield, United Kingdom Collapse << |
| NCT00555880 | Hypotension, Orthostatic | Phase 4 | Completed | - | United States, Indiana ... more >> Fort Wayne Neurological Center Fort Wayne, Indiana, United States, 46805 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Minnesota Minnesota VA Medical Center Minneapolis, Minnesota, United States, 55417 United States, Missouri Forest Park Neurophysiology Saint Louis, Missouri, United States, 63139 United States, New York Mount Sinai Medical Center New York, New York, United States, 10029 United States, North Carolina Raleigh Neurology Associates Raleigh, North Carolina, United States, 27607 United States, Ohio University Hospitals of Cleveland Cleveland, Ohio, United States, 44106 United States, South Carolina Absher Neurology, PA Greenville, South Carolina, United States, 29615 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States, 37212 United States, Texas Diabetes and Glandular Disease research Associates, PA San Antonio, Texas, United States, 78229 Collapse << |
| NCT00383708 | - | - | Completed | - | - |
| NCT00430677 | Systemic Lupus Erythematosus | Phase 2 Phase 3 | Terminated(Terminated due to f... more >>ailure to meet the primary efficacy endpoint in the Short-term Period) Collapse << | - | - |
| NCT00557856 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, South Carolina Medical University of South Carolina, Hollings Cancer Center Charleston, South Carolina, United States, 29425 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37212-3505 Vanderbilt University Medical Center Nashville, Tennessee, United States, 37232 Italy S.C Diagnostica Radiologica 2 Milano, Italy, 20133 S.C. Chirurgia Generale Indirizzo Oncologico 1 (epato-gastro-pancreatica) Milano, Italy, 20133 S.C. Medicina Oncologica I, Fondazione IRCCS Istituto Nazionale Tumori Milano, Italy, 20133 Dipartimento di Medicina Milano, Italy, 20141 UO di Oncologia ed Ematologia, Istituto Clinico Humanitas-Humanitas Cancer Center Rozzano (MI), Italy, 20089 Korea, Republic of Seoul National University Hospital / Department of Internal Medicine Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT03425994 | - | - | Recruiting | January 2020 | Taiwan ... more >> National Taiwan University Hospital Recruiting Taipei, Taiwan, 100 Contact: Chien-Ching Hung, MD, PhD +886-2-23123456 ext 67552 hcc0401@ntu.edu.tw Contact: Yu-Shan Huang +886-953-939-523 b101091021@gmail.com Collapse << |
| NCT00430677 | - | - | Terminated(Terminated due to f... more >>ailure to meet the primary efficacy endpoint in the Short-term Period) Collapse << | - | - |
| NCT00489359 | - | - | Completed | - | - |
| NCT01762761 | Purpura, Thrombocytopenic, Idi... more >>opathic and Hepatitis C Collapse << | Phase 3 | Active, not recruiting | November 15, 2018 | China, Fujian ... more >> Novartis Investigative Site Fuzhou, Fujian, China, 350001 China, Guangdong Novartis Investigative Site Guangzhou, Guangdong, China, 510080 Novartis Investigative Site Guangzhou, Guangdong, China, 510515 Novartis Investigative Site Zhongshan, Guangdong, China, 528403 China, Hunan Novartis Investigative Site Changsha, Hunan, China, 410013 China, Jiangsu Novartis Investigative Site Nanjing, Jiangsu, China, 210029 China, Jiangxi Novartis Investigative Site Nanchang, Jiangxi, China, 330006 China, Shandong Novartis Investigative Site Jianan, Shandong, China, 250012 China, Zhejiang Novartis Investigative Site Hangzhou, Zhejiang, China, 310003 China Novartis Investigative Site Beijing, China, 100034 Novartis Investigative Site Beijing, China, 100044 Novartis Investigative Site Beijing, China, 100083 Novartis Investigative Site Beijing, China, 100730 Novartis Investigative Site Chengdu, China, 610041 Novartis Investigative Site Jiang Su Province, China, 215006 Novartis Investigative Site Shanghai, China, 200025 Novartis Investigative Site Shanghai, China Novartis Investigative Site Tianjin, China, 300020 Collapse << |
| NCT01762761 | - | - | Active, not recruiting | - | - |
| NCT02339155 | Infections, Bacterial | Phase 4 | Completed | - | United States, Florida ... more >> GSK Investigational Site Edgewater, Florida, United States, 32132 United States, Kansas GSK Investigational Site Overland Park, Kansas, United States, 66211 United States, Tennessee GSK Investigational Site Knoxville, Tennessee, United States, 37920 Collapse << |
| NCT01134055 | - | - | Completed | - | - |
| NCT01268566 | Glioblastoma Multiforme | Phase 2 | Completed | - | United States, Arizona ... more >> Research Site Tucson, Arizona, United States United States, California Research Site Los Angeles, California, United States Research Site Stanford, California, United States United States, Illinois Research Site Chicago, Illinois, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Michigan Research Site Detroit, Michigan, United States United States, New York Research Site New York, New York, United States United States, Ohio Research Site Canton, Ohio, United States United States, Pennsylvania Research Site Pittsburgh, Pennsylvania, United States United States, Tennessee Research Site Nashville, Tennessee, United States United States, Texas Research Site San Antonio, Texas, United States United States, Washington Research Site Seattle, Washington, United States Collapse << |
| NCT00557856 | - | - | Completed | - | - |
| NCT01268566 | - | - | Completed | - | - |
| NCT00489359 | Ovarian Cancer ... more >> Primary Peritoneal Cancer Collapse << | Phase 1 Phase 2 | Completed | - | Argentina ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bahia Blanca, Argentina, B8000HXM For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Buenos Aires, Argentina, C1199ACK For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ramos Mejia, Argentina, B1704ESN For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Salta, Argentina, 4400 Canada, Alberta For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Calgary, Alberta, Canada, T2N 4N2 Canada, Ontario For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Toronto, Ontario, Canada, M5G 2M9 Germany For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Berlin, Germany, 13353 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bonn, Germany, 53127 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chemnitz, Germany, D-09116 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Duesseldorf, Germany, 40489 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Erlangen, Germany, D-91054 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Essen, Germany, DE-45145 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 22081 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Jena, Germany, D-07743 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kiel, Germany, D-24105 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mainz, Germany, 55131 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tübingen, Germany, 72076 Poland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gdansk, Poland, 80-402 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Olsztyn, Poland, 10-228 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Warsaw, Poland, 00909 Sweden For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Göteborg, Sweden, 416 85 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lund, Sweden, 22241 Collapse << |
| NCT00867087 | Lymphoma, B-Cell | Phase 2 | Completed | - | - |
| NCT03165812 | Diabetes Mellitus, Type 2 ... more >> Overweight Collapse << | Not Applicable | Recruiting | April 2019 | Malaysia ... more >> Hospital Serdang Recruiting Kajang, Selangor, Malaysia, 43400 Contact: Tikfu Gee, MBBS, MS +60 18-984 2300 groupesantesoin@gmail.com Principal Investigator: Tikfu Gee, MBBS, MS Sub-Investigator: Ng Ooi Chuan, MBBS, MRCP Sub-Investigator: Ching Siew Mooi, MFamMed Sub-Investigator: Subashini a/p Chellappah Thambiah, MBBS, Mpath Sub-Investigator: Intan Nureslyna Samsudin, Mpath Sub-Investigator: Zubaidah Hanipah, MD, MS Sub-Investigator: Lim Shu Yu, MS Hospital Kuala Lumpur Recruiting Kuala Lumpur, Wilayah Persekutuan Kuala Lumpur, Malaysia, 50586 Contact: Tikfu Gee, MBBS, MS +6018 984 2300 groupesantesoin@gmail.com Principal Investigator: Tikfu Gee, MBBS, MS Sub-Investigator: Ng Ooi Chuan, MBBS, MRCP Sub-Investigator: Ching Siew Mooi, MFamMed Sub-Investigator: Subashini a/p Chellappah Thambiah, MBBS, Mpath Sub-Investigator: Intan Nureslyna Samsudin, Mpath Sub-Investigator: Zubaidah Hanipah, MD, MS Sub-Investigator: Lim Shu Yu, MS Sub-Investigator: Raflis Ruzairee Awang, MBChB, MS Collapse << |
| NCT02339155 | - | - | Completed | - | - |
| NCT01134055 | Macular Degeneration | Phase 2 | Completed | - | - |
| NCT00867087 | - | - | Completed | - | - |
| NCT02216123 | Malaria, Vivax | Phase 3 | Completed | - | Brazil ... more >> GSK Investigational Site Manaus, Amazonas, Brazil, 69040-000 Colombia GSK Investigational Site Cali, Colombia GSK Investigational Site Monteria, Colombia Ethiopia GSK Investigational Site Gondar, Ethiopia GSK Investigational Site Jimma, Ethiopia Peru GSK Investigational Site Iquitos, Loreto, Peru, Iqui 01 Thailand GSK Investigational Site Bangkok, Thailand, 10400 GSK Investigational Site Mae Sot, Thailand, 63110 GSK Investigational Site Tak, Thailand, 63170 Vietnam GSK Investigational Site Ho Chi Minh, Vietnam Collapse << |
| NCT01568112 | Healthy | Phase 3 | Completed | - | United States, Minnesota ... more >> Research Site St Paul, Minnesota, United States Collapse << |
| NCT00710684 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT00710684 | - | - | Completed | - | - |
| NCT03014674 | Asthma | Phase 3 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Germany GSK Investigational Site Berlin, Germany, 14050 United Kingdom GSK Investigational Site Harrow, Middlesex, United Kingdom, HA1 3UJ Collapse << |
| NCT03068065 | Non-Alcoholic Fatty Liver Dise... more >>ase Type2 Diabetes Collapse << | Phase 4 | Completed | - | China, Jiangsu ... more >> at Division of Endocrinology, the Affiliated Drum Tower Hospital of Nanjing University Nanjing, Jiangsu, China, 210008 Collapse << |
| NCT02216123 | - | - | Completed | - | - |
| NCT00035932 | HIV Infections | Phase 3 | Completed | - | - |
| NCT01568112 | - | - | Completed | - | - |
| NCT00455533 | Breast Cancer | Phase 2 | Completed | - | - |
| NCT03014674 | - | - | Completed | - | - |
| NCT00107653 | Hepatitis C, Chronic | Phase 4 | Completed | - | - |
| NCT03630627 | Healthy Volunteers | Phase 1 | Recruiting | August 20, 2020 | United States, California ... more >> PAREXEL International Recruiting Glendale, California, United States, 91206 Contact 888-228-7425 Collapse << |
| NCT00550420 | Alzheimer's Disease | Phase 3 | Terminated(Based on preliminar... more >>y parent study results) Collapse << | - | - |
| NCT00550420 | - | - | Terminated(Based on preliminar... more >>y parent study results) Collapse << | - | - |
| NCT03351751 | Healthy Volunteers | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00035932 | - | - | Completed | - | - |
| NCT01262677 | Partial Seizures ... more >> Epilepsies, Partial Collapse << | Phase 3 | Completed | - | - |
| NCT00455533 | - | - | Completed | - | - |
| NCT02422797 | - | - | Active, not recruiting | - | - |
| NCT02422797 | HIV Infections | Phase 3 | Active, not recruiting | August 12, 2021 | - |
| NCT02014584 | Alopecia | Phase 3 | Completed | - | Chile ... more >> GSK Investigational Site Santiago, Región Metro De Santiago, Chile, 7580206 Hong Kong GSK Investigational Site Pokfulam, Hong Kong GSK Investigational Site Shatin, Hong Kong Korea, Republic of GSK Investigational Site Chungcheongnam-do, Korea, Republic of GSK Investigational Site Daejeon, Korea, Republic of, 301-721 GSK Investigational Site Gwangju, Korea, Republic of, 501-757 GSK Investigational Site Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Incheon, Korea, Republic of, 400-711 GSK Investigational Site Jeonju-si, Jeollabuk-do, Korea, Republic of, 561-712 GSK Investigational Site Kangwon-Do, Korea, Republic of, 220-701 GSK Investigational Site Pusan, Korea, Republic of, 602-739 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 134-090 GSK Investigational Site Seoul, Korea, Republic of, 143-729 GSK Investigational Site Seoul, Korea, Republic of, 156-755 Singapore GSK Investigational Site Singapore, Singapore, 169608 Taiwan GSK Investigational Site Taipei, Taiwan, 106 GSK Investigational Site Taipei, Taiwan Collapse << |
| NCT01262677 | - | - | Completed | - | - |
| NCT02772016 | Thalassemia | Early Phase 1 | Unknown | - | China, Guangdong ... more >> the first affiliated hospital of Guangzhou University of Chinese Medicine Recruiting Guangzhou, Guangdong, China, 510405 Contact: Yanfang Li, PhD +86-20-36598857 gzyanfangli@hotmail.com Collapse << |
| NCT02014584 | - | - | Completed | - | - |
| NCT02641054 | Idiopathic Parkinson Disease | Phase 2 | Completed | - | France ... more >> Clevexel Pharma Maisons-Alfort, France, 94700 Collapse << |
| NCT00428090 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT00107653 | - | - | Completed | - | - |
| NCT00509028 | Asthma | Phase 3 | Completed | - | Japan ... more >> Research Site Takizawa, Iwate, Japan Collapse << |
| NCT01174446 | Hemophilia B | Phase 3 | Completed | - | - |
| NCT00509028 | - | - | Completed | - | - |
| NCT01174446 | - | - | Completed | - | - |
| NCT01697956 | Allergic Rhinitis | Phase 3 | Completed | - | United States, California ... more >> Teva Investigational Site 10305 Long Beach, California, United States United States, Georgia Teva Investigational Site 10304 Stockbridge, Georgia, United States United States, Minnesota Teva Investigational Site 10300 Plymouth, Minnesota, United States United States, Pennsylvania Teva Investigational Site 10302 Normal, Pennsylvania, United States United States, Texas Teva Investigational Site 10301 New Braunfels, Texas, United States Teva Investigational Site 10303 San Antonio, Texas, United States Collapse << |
| NCT02429791 | HIV Infections | Phase 3 | Active, not recruiting | August 13, 2021 | - |
| NCT01405196 | - | - | Completed | - | - |
| NCT00428090 | - | - | Completed | - | - |
| NCT01943864 | Cancer | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 277-8577 GSK Investigational Site Osaka, Japan, 537-8511 GSK Investigational Site Shizuoka, Japan, 411-8777 GSK Investigational Site Tokyo, Japan, 104-0045 GSK Investigational Site Tokyo, Japan, 181-8611 Collapse << |
| NCT01697956 | - | - | Completed | - | - |
| NCT02271984 | Healthy | Phase 1 | Completed | - | Germany ... more >> Please contact the Merck KGaA Communication Center Darmstadt, Germany Collapse << |
| NCT02429791 | - | - | Active, not recruiting | - | - |
| NCT01405196 | Lupus Erythematosus, Systemic | Phase 2 | Completed | - | - |
| NCT02271984 | - | - | Completed | - | - |
| NCT01424514 | Rhinitis | Phase 2 | Completed | - | Canada, Ontario ... more >> GSK Investigational Site Mississauga, Ontario, Canada, L4W 1N2 Collapse << |
| NCT00077623 | Anemia | Phase 3 | Completed | - | - |
| NCT01943864 | - | - | Completed | - | - |
| NCT00077623 | - | - | Completed | - | - |
| NCT00817063 | Eczema | Phase 3 | Completed | - | - |
| NCT01424514 | - | - | Completed | - | - |
| NCT00817063 | - | - | Completed | - | - |
| NCT00272779 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00790933 | Ulcerative Colitis ... more >> Crohn's Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01636778 | Hepatitis C, Chronic | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Aichi, Japan, 467-8602 GSK Investigational Site Fukui, Japan, 918-8503 GSK Investigational Site Fukuoka, Japan, 803-8505 GSK Investigational Site Fukuoka, Japan, 830-0011 GSK Investigational Site Gunma, Japan, 370-0829 GSK Investigational Site Hyogo, Japan, 663-8501 GSK Investigational Site Ibaraki, Japan, 317-0077 GSK Investigational Site Kagawa, Japan, 760-0017 GSK Investigational Site Kagawa, Japan, 760-8557 GSK Investigational Site Kagoshima, Japan, 899-5112 GSK Investigational Site Kanagawa, Japan, 213-8587 GSK Investigational Site Nagasaki, Japan, 856-8562 GSK Investigational Site Oita, Japan, 879-5593 GSK Investigational Site Osaka, Japan, 540-0006 GSK Investigational Site Tokyo, Japan, 105-8470 GSK Investigational Site Wakayama, Japan, 646-8558 Collapse << |
| NCT00768989 | - | - | Terminated(Efficacy endpoint m... more >>et, but overall experimental dosing regimen not considered optimal to support further clinical development in this population.) Collapse << | - | - |
| NCT00272779 | - | - | Completed | - | - |
| NCT00768989 | HIV | Phase 2 | Terminated(Efficacy endpoint m... more >>et, but overall experimental dosing regimen not considered optimal to support further clinical development in this population.) Collapse << | - | United States, Arizona ... more >> Southwest Center For HIV/AIDS Phoenix, Arizona, United States, 85006 United States, Connecticut Yale University School Of Medicine New Haven, Connecticut, United States, 06510 United States, District of Columbia Dupont Circle Physicians Group Washington, District of Columbia, United States, 20009 United States, Florida Orlando Immunology Center Orlando, Florida, United States, 32803 United States, New York The Aaron Diamond AIDS Research Center New York, New York, United States, 10016 United States, Ohio University Of Cincinnati Cincinnati, Ohio, United States, 45267 United States, Texas Tarrant County Infectious Disease Associates Ft Worth, Texas, United States, 76104 Therapeutic Concepts, P.A. Houston, Texas, United States, 77004 Diversified Medical Practices, P.A. Houston, Texas, United States, 77057 Argentina Local Institution Buenos Aires, Bs As, Buenos Aires, Argentina, 1141 Local Institution Capital Federal, Buenos Aires, Argentina, 1264 Local Institution Mar Del Plata, Buenos Aires, Argentina, B7600CTO Local Institution Rosario, Santa Fe, Argentina, 2000 France Local Institution Nantes Cedex 01, France, 44035 Local Institution Paris Cedex 10, France, 75475 Local Institution Paris Cedex 20, France, 75970 Collapse << |
| NCT00495469 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT02034916 | Breast Neoplasms ... more >> BRCA 1 Gene Mutation BRCA 2 Gene Mutation Collapse << | Phase 2 | Completed | - | - |
| NCT00874770 | Hepatitis C Infection | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama Liver & Digestive Specialists (Alds) Montgomery, Alabama, United States, 36116 United States, Colorado University Of Colorado Denver & Hospital Aurora, Colorado, United States, 80045 United States, Connecticut Yale University School Of Medicine New Haven, Connecticut, United States, 06520 United States, Maryland Mercy Medical Center Baltimore, Maryland, United States, 21202 United States, Massachusetts Llc Dba The Research Institute Springfield, Massachusetts, United States, 01107 United States, New York Veterans Affairs Medical Center Bronx, New York, United States, 10468 United States, North Carolina Carolinas Center For Liver Disease Statesville, North Carolina, United States, 28677 United States, Oklahoma Options Health Research, Llc Tulsa, Oklahoma, United States, 74104 Healthcare Research Consultants Tulsa, Oklahoma, United States, 74135 United States, Texas North Texas Research Institute Arlington, Texas, United States, 76012 United States, Virginia Metropolitan Research Fairfax, Virginia, United States, 22031 France Local Institution Creteil, France, 94010 Local Institution Paris Cedex 14, France, 75679 Local Institution Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT01636778 | - | - | Completed | - | - |
| NCT03070548 | Advanced Solid Tumors | Phase 1 | Completed | - | Hungary ... more >> PRA Magyarorszag Kft, Fazis I-es Klinikai Farmakologiai Vizsgalohely Budapest, Hungary, 1077 Collapse << |
| NCT00874770 | - | - | Completed | - | - |
| NCT01114620 | Influenza | Phase 4 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-8588 Collapse << |
| NCT03070548 | - | - | Completed | - | - |
| NCT00495469 | - | - | Completed | - | - |
| NCT02034916 | - | - | Completed | - | - |
| NCT03397641 | Atrial Fibrillation | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Clinical Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT01318993 | Crohn's Disease | Phase 3 | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT01114620 | - | - | Completed | - | - |
| NCT01318993 | - | - | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT03744663 | Opioid Use | Phase 2 | Not yet recruiting | November 2019 | United States, North Carolina ... more >> Wake Forest Baptist Medical Center Not yet recruiting Winston-Salem, North Carolina, United States, 27157 Contact: Ernesto Aranda-Aguirre, MD earandaa@wakehealth.edu Collapse << |
| NCT01362348 | Macular Degeneration | Phase 2 | Terminated(Lack of efficacy id... more >>entified during a preliminary analysis) Collapse << | - | United States, Arizona ... more >> GSK Investigational Site Phoenix, Arizona, United States, 85014 United States, Florida GSK Investigational Site Miami, Florida, United States, 33143 GSK Investigational Site Winter Haven, Florida, United States, 33880 United States, Georgia GSK Investigational Site Decatur, Georgia, United States, 30030 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02114 United States, Michigan GSK Investigational Site Grand Rapids, Michigan, United States, 49525 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44195 United States, Texas GSK Investigational Site Austin, Texas, United States, 78705 GSK Investigational Site Houston, Texas, United States, 77030 France GSK Investigational Site Paris cedex 10, France, 75475 GSK Investigational Site Paris cedex 12, France, 75571 Germany GSK Investigational Site Muenchen, Bayern, Germany, 80336 GSK Investigational Site Bonn, Nordrhein-Westfalen, Germany, 53127 GSK Investigational Site Muenster, Nordrhein-Westfalen, Germany, 48145 GSK Investigational Site Leipzig, Sachsen, Germany, 04103 Collapse << |
| NCT01090492 | Raynaud's Disease ... more >> Peripheral Vascular Disease Collapse << | Phase 2 | Completed | - | - |
| NCT01362348 | - | - | Terminated(Lack of efficacy id... more >>entified during a preliminary analysis) Collapse << | - | - |
| NCT00793871 | Gastrointestinal Neoplasms, Ga... more >>strointestinal Stromal Tumors Collapse << | Phase 4 | Completed | - | China, Jiangsu ... more >> Nanjing Bayi Hospital Nanjing, Jiangsu, China, 210002 China Beijing Cancer Hospital Beijing, China, 100035 307 Hospital of PLA Beijing, China, 100071 Cancer Institute & Hospital Chinese Academy of Medical Sciences and PUMC Bejing, China, 100021 Collapse << |
| NCT03511820 | - | - | Completed | - | Taiwan ... more >> Changhua Christian Hospital Changhua, Taiwan, 500 Kaohsiung Veterans General Hospital Kaohsiung, Taiwan, 81362 Kaohsiung Chang Gung Memorial Hospital Kaohsiung, Taiwan, 833 Keelung Chang Gung Memorial Hospital Keelung, Taiwan, 204 China Medical University Hospital Taichung City, Taiwan, 40447 Tri-Service General Hospital Taipei city, Taiwan, 114 Taipei Veterans General Hospital Taipei, Taiwan, 11212 Taipei Mucinipal Wanfang Hospital Taipei, Taiwan, 116 Chang Guang Memorial Hospital at LinKou Taoyuan, Taiwan, 333 Collapse << |
| NCT03627494 | Muscular Dystrophies | Phase 1 | Recruiting | June 30, 2019 | United States, Maryland ... more >> GSK Investigational Site Recruiting Baltimore, Maryland, United States, 21225 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00793871 | - | - | Completed | - | - |
| NCT02691325 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02293161 | Autoimmune Diseases | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 2GG Collapse << |
| NCT02593539 | Activated PI3K-delta Syndrome | Phase 2 | Recruiting | December 11, 2019 | United Kingdom ... more >> GSK Investigational Site Recruiting Cambridge, United Kingdom, CB2 0GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00897390 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, Nebraska ... more >> Mds Pharma Services Lincoln, Nebraska, United States, 68502 Collapse << |
| NCT01090492 | - | - | Completed | - | - |
| NCT03358407 | Inflammatory Bowel Diseases | Phase 1 | Recruiting | September 16, 2019 | United Kingdom ... more >> GSK Investigational Site Recruiting Cambridge, Cambridgeshire, United Kingdom, CB2 2GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01001169 | - | - | Completed | - | - |
| NCT03426995 | Arthritis, Rheumatoid | Phase 1 | Recruiting | July 29, 2019 | United Kingdom ... more >> GSK Investigational Site Recruiting Cambridge, United Kingdom, CB2 2GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00897390 | - | - | Completed | - | - |
| NCT00534313 | Psoriatic Arthritis | Phase 2 | Terminated(Lack of efficacy wi... more >>th respect to skin-related lesions in the short-term phase) Collapse << | - | - |
| NCT01001169 | Influenza | Phase 2 | Completed | - | Japan ... more >> GSK Investigational Site Tokyo, Japan, 157-8535 Collapse << |
| NCT00371345 | Breast Cancer ... more >> Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Ucsf-Comprehensive Cancer Center San Francisco, California, United States, 94143 United States, Florida Mayo Clinic Florida Jacksonville, Florida, United States, 32224 United States, Massachusetts Dana-Farber Cancer Inst Boston, Massachusetts, United States, 02115 United States, New York Montefiore Medical Center Bronx, New York, United States, 10461 United States, North Carolina University Of North Carolina At Chapel Hill Chapel Hill, North Carolina, United States, 275997305 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 United States, Texas University Of Texas Md Anderson Cancer Ctr Houston, Texas, United States, 77030 Argentina Local Institution Haedo, Buenos Aires, Argentina, 1684 Local Institution Buenos Aires, Argentina, 1019 Local Institution Buenos Aires, Argentina, 1185 Belgium Local Institution Bruxelles, Belgium, 1000 Local Institution Bruxelles, Belgium, 1200 France Local Institution Dijon Cedex, France, 21079 Local Institution Paris, France, 75231 Local Institution Saint Herblain Cedex, France, 44805 Local Institution Toulouse Cedex 3, France, 31052 Italy Local Institution Modena, Italy, 41100 Peru Local Institution Arequipa, Peru Local Institution Lima, Peru, 34 Local Institution Lima, Peru, LIMA 11 Spain Local Institution Barcelona, Spain, 08035 Local Institution Lleida, Spain, 25198 Local Institution Madrid, Spain, 28041 Collapse << |
| NCT03245619 | Pancreatitis, Acute Necrotizin... more >>g Collapse << | Phase 1 | Terminated(Sponsor decision pe... more >>nding review of data.) Collapse << | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 0GG Collapse << |
| NCT03544281 | Multiple Myeloma | Phase 2 | Recruiting | November 28, 2022 | United States, Alabama ... more >> GSK Investigational Site Recruiting Birmingham, Alabama, United States, 35294 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01920594 | Surgical Procedures | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Birmingham, Alabama, United States, 35233 United States, California GSK Investigational Site Los Angeles, California, United States, 90048 United States, Colorado GSK Investigational Site Aurora, Colorado, United States, 80045 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30322 United States, Michigan GSK Investigational Site Ann Arbor, Michigan, United States, 48109-5864 United States, Minnesota GSK Investigational Site Rochester, Minnesota, United States, 55905 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44195 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 GSK Investigational Site Pittsburgh, Pennsylvania, United States, 308902 United States, Texas GSK Investigational Site Houston, Texas, United States, 77030 United States, Virginia GSK Investigational Site Charlottesville, Virginia, United States, 22908 GSK Investigational Site Richmond, Virginia, United States, 23298 Canada, Alberta GSK Investigational Site Calgary, Alberta, Canada, T2N 2T9 Canada, Quebec GSK Investigational Site Quebec City, Quebec, Canada, G1V 4G5 Collapse << |
| NCT00319254 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | United States, California ... more >> Pfizer Investigational Site Duarte, California, United States, 91010-3000 Pfizer Investigational Site Santa Monica, California, United States, 90404 United States, Florida Pfizer Investigational Site Tampa, Florida, United States, 33612 United States, Ohio Pfizer Investigational Site Cleveland, Ohio, United States, 44195 Australia, New South Wales Pfizer Investigational Site Darlinghurst, New South Wales, Australia, 2010 France Pfizer Investigational Site Dijon, France, 21034 Pfizer Investigational Site Saint-Herblain, France, 44805 Hong Kong Pfizer Investigational Site Pokfulam, Hong Kong Malta Pfizer Investigational Site Floriana, Malta, VLT 14 Poland Pfizer Investigational Site Lodz, Poland, 90-553 Pfizer Investigational Site Wroclaw, Poland, 51-124 Russian Federation Pfizer Investigational Site Moscow, Russian Federation, 115478 Ukraine Pfizer Investigational Site Dnipropetrovsk, Ukraine, 49102 Pfizer Investigational Site Sumy, Ukraine, 40005 Pfizer Investigational Site Uzhgorod, Ukraine, 88014 Collapse << |
| NCT00319254 | - | - | Completed | - | - |
| NCT00534313 | - | - | Terminated(Lack of efficacy wi... more >>th respect to skin-related lesions in the short-term phase) Collapse << | - | - |
| NCT02204371 | Telangiectasia, Hereditary Hem... more >>orrhagic Collapse << | Phase 2 | Terminated(The study was termi... more >>nated as the asset was sold to another company who decided not to pursue this indication with this drug.) Collapse << | - | United States, California ... more >> GSK Investigational Site Los Angeles, California, United States, 90095 United States, Georgia GSK Investigational Site Augusta, Georgia, United States, 30912-3135 United States, Missouri GSK Investigational Site Saint Louis, Missouri, United States, 63110 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44195 United States, Utah GSK Investigational Site Salt Lake City, Utah, United States, 84132 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M5B 1W8 Collapse << |
| NCT00371345 | - | - | Completed | - | - |
| NCT02204371 | - | - | Terminated(The study was termi... more >>nated as the asset was sold to another company who decided not to pursue this indication with this drug.) Collapse << | - | - |
| NCT00555906 | Multiple Myeloma | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern Medical Faculty Foundation Chicago, Illinois, United States, 60611 Northwestern Memorial Hospital/Main Labs Chicago, Illinois, United States, 60611 United States, Kansas University of Kansas Medical Center Kansas City, Kansas, United States, 66160 University of Kansas Cancer Center and Medical Pavilion Westwood, Kansas, United States, 66205 United States, Maryland University of Maryland Baltimore, Maryland, United States, 21201 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Missouri Washington University School of Medicine St. Louis, Missouri, United States, 63110-1093 Barnes-Jewish Hospital St. Louis, Missouri, United States, 63110 Washington University School of Medicine St. Louis, Missouri, United States, 63110 United States, Nebraska Nebraska Methodist Hospital Omaha, Nebraska, United States, 68114 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 New York Presbyterian, Weill Cornell Medical College New York, New York, United States, 10065 United States, Pennsylvania University of Pennsylvania Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 United States, South Carolina Hollings Cancer Center Charleston, South Carolina, United States, 29425 Medical University of South Carolina Charleston, South Carolina, United States, 29425 Czech Republic Vseobecna fakultni nemocnice v Praze Praha 2, Czech Republic, 12808 Germany Universitaetsklinikum Heidelberg Heidelberg, Germany, 69120 Klinikum Johannes-Gutenberg -Universitaet, III. Medizinische Klinik und Poliklinik Mainz, Germany, 55131 Collapse << |
| NCT01494038 | HIV Infections ... more >> Tuberculosis Collapse << | Phase 4 | Completed | - | Botswana ... more >> Gaborone CRS Gaborone, Botswana Molepolole CRS Gaborone, Botswana Haiti Les Centres GHESKIO Clinical Research Site (GHESKIO-INLR) CRS Port-au-Prince, Haiti, HT-6110 India Byramjee Jeejeebhoy Medical College (BJMC) CRS Pune, Maharashtra, India, 411001 South Africa Soweto IMPAACT CRS Johannesburg, Gauteng, South Africa, 1862 Desmond Tutu TB Centre - Stellenbosch University (DTTC-SU) CRS Cape Town, Western Cape Province, South Africa, 7505 Fam-Cru Crs Cape Town, Western Cape Province, South Africa, 7505 Tanzania Kilimanjaro Christian Medical Centre (KCMC) Moshi, Tanzania Thailand Chiang Mai University HIV Treatment (CMU HIV Treatment) CRS Chiang Mai, Thailand, 50200 Uganda MU-JHU Research Collaboration (MUJHU CARE LTD) CRS Kampala, Uganda Zimbabwe Seke North CRS Chitungwiza, Zimbabwe St Mary's CRS Chitungwiza, Zimbabwe Harare Family Care CRS Harare, Zimbabwe Collapse << |
| NCT00555906 | - | - | Completed | - | - |
| NCT03359473 | Cachexia | Phase 2 | Recruiting | November 3, 2019 | United States, California ... more >> GSK Investigational Site Not yet recruiting Torrance, California, United States, 90502 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Florida GSK Investigational Site Recruiting Jacksonville, Florida, United States, 32216 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, North Carolina GSK Investigational Site Recruiting High Point, North Carolina, United States, 27262 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, South Carolina GSK Investigational Site Recruiting Spartanburg, South Carolina, United States, 29303 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Germany GSK Investigational Site Recruiting Frankfurt, Hessen, Germany, 60596 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Grosshansdorf, Schleswig-Holstein, Germany, 22927 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01468077 | Rheumatoid Arthritis | Phase 2 | Completed | - | Denmark ... more >> Alborg, Denmark, 9000 Frederiksberg, Denmark, 2000 Hellerup, Denmark, 2900 Holbæk, Denmark, 4300 Odense, Denmark, 5000 Silkeborg, Denmark, 8600 Svendborg, Denmark, 5700 Iceland Reykjavik, Iceland, 108 Collapse << |
| NCT01920594 | - | - | Completed | - | - |
| NCT01494038 | - | - | Completed | - | - |
| NCT03457727 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT01468077 | - | - | Completed | - | - |
| NCT00979602 | Influenza | Phase 3 | Completed | - | - |
| NCT01035749 | Influenza | Phase 2 | Completed | - | Estonia ... more >> GSK Investigational Site Tartu, Estonia, 50106 Slovakia GSK Investigational Site Cifer, Slovakia, 919 43 GSK Investigational Site Dolny Kubin, Slovakia, 026 01 GSK Investigational Site Dunajska Streda, Slovakia, 929 01 GSK Investigational Site Nova Dubnica, Slovakia, 018 51 GSK Investigational Site Nove Mesto nad Vahom, Slovakia, 915 01 GSK Investigational Site Puchov, Slovakia, 020 01 GSK Investigational Site Ruzomberok, Slovakia, 034 01 GSK Investigational Site Trencin, Slovakia, 911 01 Collapse << |
| NCT00089505 | - | - | Completed | - | - |
| NCT00089505 | HIV Infections | Phase 3 | Completed | - | Botswana ... more >> The Gaborone BHP Study Clinic Bontleng, Gaborone, Botswana Molepolole BHP Study Clinic, Scottish Livingstone Hospital Bontleng Gaborone, Botswana Kenya Moi University International Clnical Trials Unit Eldoret, Kenya, 30100 KMRI / Walter Reed Project Clinical Research Center Kericho, Kenya Malawi University of North Carolina Project (UNC Project) Lilongwe, Malawi, (265) 175-5056 South Africa University of KwaZulu Natal Durban, KZN, South Africa, 4013 Chris Hani Baragwanath Hospital, Johannesburg Johannesburg, South Africa University of Witwatersrand Johannesburg, South Africa Uganda Joint Clinical Research Centre (JCRC) Kampala, Uganda Zambia Centre for Infectious Disease Research in Zambia (CIDRZ) Lusaka, Zambia Zimbabwe University of Zimbabwe Avondale, Harare, Zimbabwe Collapse << |
| NCT00979602 | - | - | Completed | - | - |
| NCT02216097 | Post-Traumatic Stress Disorder | Phase 2 | Terminated(The study stopped b... more >>ased on Pfizer portfolio prioritization and not due to safety and/or efficacy concern or change in benefitrisk assessment of PF-04457845.) Collapse << | - | United States, New York ... more >> Clinical and Translational Science Institute (CTSI) New York, New York, United States, 10016 NYU CTSI Research Pharmacy (Drug Shipment Address) New York, New York, United States, 10016 NYU School of Medicine New York, New York, United States, 10016 Collapse << |
| NCT02723955 | Cancer | Phase 1 | Recruiting | May 18, 2020 | United States, California ... more >> GSK Investigational Site Recruiting Duarte, California, United States, 91010 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, New York GSK Investigational Site Recruiting New York, New York, United States, 10016 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting New York, New York, United States, 10032 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Pennsylvania GSK Investigational Site Recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Tennessee GSK Investigational Site Recruiting Nashville, Tennessee, United States, 37203 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Nashville, Tennessee, United States, 37232-6307 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Australia, Victoria GSK Investigational Site Recruiting Heidelberg, Victoria, Australia, 3084 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Melbourne, Victoria, Australia, 3000 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Australia, Western Australia GSK Investigational Site Recruiting Nedlands, Western Australia, Australia, 6009 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Canada, Ontario GSK Investigational Site Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com France GSK Investigational Site Not yet recruiting Bordeaux Cedex, France, 33076 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Lyon cedex 08, France, 69373 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Paris, France, 75005 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Villejuif cedex, France, 94805 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Italy GSK Investigational Site Recruiting Siena, Toscana, Italy, 53100 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Netherlands GSK Investigational Site Recruiting Amsterdam, Netherlands, 1066 CX Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Spain GSK Investigational Site Recruiting Barcelona, Spain, 08035 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Barcelona, Spain, 08036 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Madrid, Spain, 28040 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Málaga, Spain, 29010 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Sevilla, Spain, 41009 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01035749 | - | - | Completed | - | - |
| NCT02216097 | - | - | Terminated(The study stopped b... more >>ased on Pfizer portfolio prioritization and not due to safety and/or efficacy concern or change in benefitrisk assessment of PF-04457845.) Collapse << | - | - |
| NCT00733980 | Depressive Disorder, Major | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Cerritos, California, United States, 90703 GSK Investigational Site San Diego, California, United States, 92108 GSK Investigational Site Torrance, California, United States, 90502 United States, Florida GSK Investigational Site Jacksonville, Florida, United States, 32216 GSK Investigational Site Miami, Florida, United States, 33125 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30322 GSK Investigational Site Marietta, Georgia, United States, 30060 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60612 GSK Investigational Site Skokie, Illinois, United States, 60076 United States, Louisiana GSK Investigational Site Shreveport, Louisiana, United States, 71104 United States, Maryland GSK Investigational Site Rockville, Maryland, United States, 20852 United States, New Mexico GSK Investigational Site Albuquerque, New Mexico, United States, 87109 United States, North Carolina GSK Investigational Site Raleigh, North Carolina, United States, 27609 United States, Ohio GSK Investigational Site Cincinnati, Ohio, United States, 45219-0516 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee GSK Investigational Site Memphis, Tennessee, United States, 38119 United States, Wisconsin GSK Investigational Site Brown Deer, Wisconsin, United States, 53223 GSK Investigational Site Madison, Wisconsin, United States, 53719 Collapse << |
| NCT01229436 | - | - | Completed | - | - |
| NCT01229436 | Dupuytren's Contracture | Phase 3 | Completed | - | Denmark ... more >> Pfizer Investigational Site Copenhagen, Denmark, 1402 K Pfizer Investigational Site Silkeborg, Denmark, 8600 France Pfizer Investigational Site Montpellier cedex 5, France, 34295 Pfizer Investigational Site Nice, France, 06000 Pfizer Investigational Site Paris, France, 75016 Pfizer Investigational Site Paris, France, Cedex 15 75908 Germany Pfizer Investigational Site Bad Neustadt, Germany, 97616 Pfizer Investigational Site Berlin, Germany, 10627 Pfizer Investigational Site Bonn, Germany, 53123 Pfizer Investigational Site Muenster, Germany, 48149 Pfizer Investigational Site Nurnberg, Germany, 90471 Pfizer Investigational Site Tubingen, Germany, 72076 Hungary Pfizer Investigational Site Miskolc, Hungary, 3526 Italy Pfizer Investigational Site Sesto San Giovanni, Milano, Italy, 20099 Pfizer Investigational Site Modena, Italy, 41124 Pfizer Investigational Site Savona, Italy, 17100 Spain Pfizer Investigational Site Elche, Alicante, Spain, 03203 Pfizer Investigational Site Hospitalet De Llobregat, Barcelona/ Spain, Spain, 08907 Pfizer Investigational Site Barcelona, Spain, 08022 Pfizer Investigational Site Barcelona, Spain, 08036 Pfizer Investigational Site Málaga, Spain, 29010 Sweden Pfizer Investigational Site Malmö, Sweden, SE-205 02 Pfizer Investigational Site Stockholm, Sweden, SE 118 83 Pfizer Investigational Site Uppsala, Sweden, 751 85 United Kingdom Pfizer Investigational Site Derby, United Kingdom, DE22 3NE Pfizer Investigational Site Glasgow, United Kingdom, G4 0SF Pfizer Investigational Site Norwich, United Kingdom, NR4 7UY Pfizer Investigational Site Southampton, United Kingdom, SO16 6YD Collapse << |
| NCT01727791 | Healthy Lactating Women | Phase 4 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT01727791 | - | - | Completed | - | - |
| NCT00612456 | - | - | Completed | - | - |
| NCT03593785 | Hypercholesterolemia | Phase 1 | Recruiting | July 23, 2019 | United States, California ... more >> Research Site Recruiting Glendale, California, United States, 91206 Collapse << |
| NCT00733980 | - | - | Completed | - | - |
| NCT01347866 | Advanced Cancer | Phase 1 | Terminated(Refer to Detailed D... more >>escription for documentaion of Termination Statement.) Collapse << | - | United States, California ... more >> Ronald Reagan UCLA Medical Center Los Angeles, California, United States, 90095-6984 Ronald Reagan UCLA Medical Center Los Angeles, California, United States, 90095 UCLA Medical Center Los Angeles, California, United States, 90095 UCLA Oncology Center Los Angeles, California, United States, 90095 Santa Monica - UCLA Medical Center and Orthopaedic Hospital Santa Monica, California, United States, 90404 UCLA Santa Monica Hematology Oncology Santa Monica, California, United States, 90404 United States, Colorado Anschutz Cancer Pavilion Aurora, Colorado, United States, 80045 University of Colorado Denver (CTRC) Aurora, Colorado, United States, 80045 University of Colorado Hospital Anschutz Inpatient Pavilion Aurora, Colorado, United States, 80045 University of Colorado Hospital Aurora, Colorado, United States, 80045 United States, South Carolina Medical University of South Carolina, Hollings Cancer Center Charleston, South Carolina, United States, 29425 Medical University of South Carolina Charleston, South Carolina, United States, 29425 MUSC, Investigational Drug Services Charleston, South Carolina, United States, 29425 MUSC Health East Cooper Mount Pleasant, South Carolina, United States, 29464 MUSC Specialty Care-North North Charleston, South Carolina, United States, 29406 Canada, Ontario Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Italy Ospedale San Raffaele Milano, Italy, 20132 Spain Hospital General Vall d'Hebron Barcelona, Spain, 08035 Collapse << |
| NCT01607372 | Asthma and Rhinitis | Phase 2 | Completed | - | Canada, Ontario ... more >> GSK Investigational Site Toronto, Ontario, Canada, M5V 2T3 Collapse << |
| NCT00612456 | Macular Degeneration | Phase 2 | Completed | - | - |
| NCT03435276 | Healthy Volunteers | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Harrow, United Kingdom, HA1 3UJ Collapse << |
| NCT00643201 | Venous Thrombosis | Phase 3 | Completed | - | - |
| NCT03480022 | Pre Diabetes ... more >>Polycystic Ovary Syndrome Obesity Android Collapse << | Phase 3 | Recruiting | November 15, 2020 | United States, Louisiana ... more >> Woman's Hospital Recruiting Baton Rouge, Louisiana, United States, 70817 Contact: Karen Elkind-Hirsch, PhD. 225-231-5278 Contact: Ericka Seidemann, MS 225-231-5275 ericka.seidemann@womans.org Principal Investigator: Karen Elkind-Hirsch, Ph.D. Sub-Investigator: Renee Harris, M.D. Sub-Investigator: Drake Belanger, MD Sub-Investigator: Neil Chappell, M.D. Collapse << |
| NCT00643201 | - | - | Completed | - | - |
| NCT01051063 | Leukaemia, Myelocytic, Acute | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21201 United States, Massachusetts GSK Investigational Site Worcester, Massachusetts, United States, 01655 United States, Tennessee GSK Investigational Site Nashville, Tennessee, United States, 37232 France GSK Investigational Site Angers cedex 09, France, 49933 GSK Investigational Site Grenoble cedex 9, France, 38043 GSK Investigational Site Lille, France, 59037 GSK Investigational Site Marseille cedex 9, France, 13273 GSK Investigational Site Nantes cedex 1, France, 44093 GSK Investigational Site Paris cedex 10, France, 75475 GSK Investigational Site Pessac cedex, France, 33604 GSK Investigational Site Pierre-Bénite cedex, France, 69495 Germany GSK Investigational Site Freiburg, Baden-Wuerttemberg, Germany, 79106 GSK Investigational Site Heidelberg, Baden-Wuerttemberg, Germany, 69120 GSK Investigational Site Ulm, Baden-Wuerttemberg, Germany, 89081 GSK Investigational Site Erlangen, Bayern, Germany, 91054 GSK Investigational Site Wuerzburg, Bayern, Germany, 97080 GSK Investigational Site Rostock, Mecklenburg-Vorpommern, Germany, 18057 GSK Investigational Site Oldenburg, Niedersachsen, Germany, 26133 GSK Investigational Site Muenster, Nordrhein-Westfalen, Germany, 48149 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Dresden, Sachsen, Germany, 01307 GSK Investigational Site Berlin, Germany, 12200 Collapse << |
| NCT03485365 | Pain, Inflammatory | Phase 1 | Recruiting | November 27, 2019 | United Kingdom ... more >> GSK Investigational Site Recruiting Cambridge, United Kingdom, CB2 0GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01051063 | - | - | Completed | - | - |
| NCT00683904 | Non-small Cell Lung Cancer (NS... more >>CLC) Collapse << | Phase 1 | Completed | - | Japan ... more >> Local Institution Chuo-Ku, Tokyo, Japan, 104-0045 Collapse << |
| NCT02254304 | Relapsing Multiple Sclerosis ... more >> Clinically Isolated Syndrome Collapse << | Phase 4 | Completed | - | Germany ... more >> Please contact the Merck KGaA Communication Center Darmstadt, Germany Collapse << |
| NCT02254304 | - | - | Completed | - | - |
| NCT01347866 | - | - | Terminated(Refer to Detailed D... more >>escription for documentaion of Termination Statement.) Collapse << | - | - |
| NCT00683904 | - | - | Completed | - | - |
| NCT02972905 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-0017 Collapse << |
| NCT03422172 | HIV Infections | Phase 1 | Active, not recruiting | April 28, 2020 | China, Zhejiang ... more >> GSK Investigational Site Hangzhou, Zhejiang, China, 310000 China GSK Investigational Site Beijing, China, 100069 GSK Investigational Site Shanghai, China, 201508 Collapse << |
| NCT00678392 | Kidney Neoplasms | Phase 3 | Completed | - | - |
| NCT01525550 | Well-differentiated Pancreatic... more >> Neuroendocrine Tumor Collapse << | Phase 4 | Completed | - | - |
| NCT02980666 | Short Bowel Syndrome | Phase 3 | Recruiting | April 12, 2019 | Japan ... more >> Kyushu University Hospital Recruiting Fukuoka-shi, Fukuoka-ken, Japan, 812-8582 Contact: Toshiharu Matsuura +819264111151 matsuura@pedsurg.med.kyushu-u.ac.jp Principal Investigator: Toshiharu Matsuura, MD Tsukuba University Hospital Recruiting Tsukuba, Ibaraki-Ken, Japan, 305-8576 Contact: Kouji Masumoto +181298533900 kmasu@md.tsukuba.ac.jp Principal Investigator: Kouji Masumoto, MD Kagoshima University Recruiting Kagoshima-shi, Kagoshima-Ken, Japan, 890-8520 Contact: Tatsuru Kaji +81992755111 tatu@m2.kufm.kagoshima-u.ac.jp Principal Investigator: Tatsuru Kaji, MD Tohoku University Hospital Recruiting Sendai-shi, Miyagi-Ken, Japan, 980-8574 Contact: Motoshi Wada +81227177000 Principal Investigator: Motoshi Wada, MD Showa University Recruiting Shinagawa-ku, Tokyo-To, Japan, 142-8555 Contact: Masahiro Chiba +81337848000 Principal Investigator: Masahiro Chiba, MD Collapse << |
| NCT02637115 | Metabolic Syndrome x ... more >> Glucose Metabolism Disorders Dyslipidemias Obesity Collapse << | Not Applicable | Completed | - | Belgium ... more >> Cliniques universitaires Saint-Luc Brussels, Belgium, 1200 Collapse << |
| NCT00678392 | - | - | Completed | - | - |
| NCT03044353 | Amyloidosis | Phase 2 | Suspended(Pending data review.... more >>) Collapse << | September 23, 2021 | United States, Massachusetts ... more >> GSK Investigational Site Boston, Massachusetts, United States, 02115 United Kingdom GSK Investigational Site Cambridge, United Kingdom, CB2 0GG GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT01205165 | Hepatitis B, Chronic | Phase 4 | Completed | - | - |
| NCT01712061 | - | - | Completed | - | - |
| NCT01205165 | - | - | Completed | - | - |
| NCT01712061 | Diabetic Nephropathy | Phase 2 | Completed | - | - |
| NCT01718587 | Liver Cirrhosis | Phase 1 Phase 2 | Unknown | July 2014 | China ... more >> Yihua An Beijing, China Collapse << |
| NCT00348140 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT00348140 | - | - | Completed | - | - |
| NCT02603172 | Amyloidosis | Phase 1 | Terminated(A new study will be... more >> initiated in order to specifically investigate cardiac safety) Collapse << | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 2GG Collapse << |
| NCT02903966 | Colitis, Ulcerative | Phase 2 | Recruiting | May 7, 2019 | United States, California ... more >> GSK Investigational Site Recruiting Moreno Valley, California, United States, 92555 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Terminated Oceanside, California, United States, 92056 GSK Investigational Site Recruiting San Diego, California, United States, 92115 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Louisiana GSK Investigational Site Not yet recruiting Marrero, Louisiana, United States, 70072 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Michigan GSK Investigational Site Recruiting Chesterfield, Michigan, United States, 48047 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, New York GSK Investigational Site Recruiting Great Neck, New York, United States, 11021 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Washington GSK Investigational Site Not yet recruiting Seattle, Washington, United States, 98195 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Germany GSK Investigational Site Recruiting Ulm, Baden-Wuerttemberg, Germany, 89081 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Frankfurt, Hessen, Germany, 60431 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Kiel, Schleswig-Holstein, Germany, 24105 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Netherlands GSK Investigational Site Recruiting Amsterdam, Netherlands, 1105 AZ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Poland GSK Investigational Site Recruiting Katowice, Poland, 40-660 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Ksawerow, Poland, 95-054 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Wroclaw, Poland Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Russian Federation GSK Investigational Site Recruiting Rostov-on-Don, Russian Federation, 344091 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting St Pertersburg, Russian Federation, 196247 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Stavropol, Russian Federation, 355018 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Ufa, Russian Federation, 450071 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Sweden GSK Investigational Site Recruiting Stockholm, Sweden, SE-171 76 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Stockholm, Sweden, SE-182 88 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Uppsala, Sweden, SE-752 37 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United Kingdom GSK Investigational Site Recruiting Stockport, Cheshire, United Kingdom, SK2 7JE Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Belfast, United Kingdom, BT16 1RH Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Recruiting Dudley, United Kingdom, DY1 2HQ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Edgbaston, Birmingham, United Kingdom, B15 2GW Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT02542813 | Postpartum Hemorrhage | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, United Kingdom, CB2 2GG Collapse << |
| NCT00723957 | Advanced/Metastatic Non-Small ... more >>Cell Lung Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT03034967 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | - |
| NCT02257359 | Embryo Transfer | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT00723957 | - | - | Completed | - | - |
| NCT03274648 | BehçEt Disease | Not Applicable | Not yet recruiting | January 2018 | - |
| NCT01009060 | Schizophrenia | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Garden Grove, California, United States, 92845 GSK Investigational Site Los Angeles, California, United States, 90073 GSK Investigational Site National City, California, United States, 91950 GSK Investigational Site Sacramento, California, United States, 95817 GSK Investigational Site San Diego, California, United States, 92102 GSK Investigational Site Torrance, California, United States, 90502 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60611 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46222 United States, Maryland GSK Investigational Site Catonsville, Maryland, United States, 21228 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02114 GSK Investigational Site Jamaica Plain, Massachusetts, United States, 02130 United States, New Jersey GSK Investigational Site Mount Laurel, New Jersey, United States, 08054 United States, New York GSK Investigational Site New York, New York, United States, 10016 GSK Investigational Site New York, New York, United States, 10032 United States, North Carolina GSK Investigational Site Butner, North Carolina, United States, 27509 Collapse << |
| NCT02927457 | Lupus Erythematosus, Cutaneous | Phase 1 | Completed | - | Germany ... more >> GSK Investigational Site Tuebingen, Baden-Wuerttemberg, Germany, 72076 GSK Investigational Site Erlangen, Bayern, Germany, 91054 GSK Investigational Site Bonn, Nordrhein-Westfalen, Germany, 53127 GSK Investigational Site Muenster, Nordrhein-Westfalen, Germany, 48149 GSK Investigational Site Wuppertal, Nordrhein-Westfalen, Germany, 42283 GSK Investigational Site Mainz, Rheinland-Pfalz, Germany, 55131 GSK Investigational Site Berlin, Germany, 10117 Collapse << |
| NCT03305419 | Autoimmune Diseases | Phase 1 | Recruiting | October 22, 2018 | United Kingdom ... more >> GSK Investigational Site Recruiting Cambridge, United Kingdom, CB2 2GG Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT00853112 | Hypertension, Pulmonary | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00853112 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01009060 | - | - | Completed | - | - |
| NCT00968526 | - | - | Completed | - | - |
| NCT00424294 | Arthritis, Rheumatoid | Phase 2 | Terminated(See Detailed Descri... more >>ption field) Collapse << | - | - |
| NCT00663208 | Chronic Hepatitis C | Phase 2 | Completed | - | United States, California ... more >> Advanced Clinical Res Inst Anaheim, California, United States, 92801 West Coast Clinical Trials, Llc Cypress, California, United States, 90630 United States, Connecticut Yale University School of Medicine New Haven, Connecticut, United States, 06510 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 Orlando Clinical Research Center Orlando, Florida, United States, 32809 United States, Maryland Parexel International Corporation Baltimore, Maryland, United States, 21225 United States, Texas Alamo Medical Research San Antonio, Texas, United States, 78215 Puerto Rico Local Institution Santurce, Puerto Rico, 00909 Collapse << |
| NCT00968526 | Influenza | Phase 3 | Completed | - | Belgium ... more >> GSK Investigational Site Wilrijk, Belgium, 2610 Collapse << |
| NCT00424294 | - | - | Terminated(See Detailed Descri... more >>ption field) Collapse << | - | - |
| NCT03027726 | Overweight Children With Type ... more >>2 Diabetes Risk Collapse << | Not Applicable | Completed | - | Spain ... more >> Pediatric Endocrinology Unit of the University Hospital of Araba (HUA) Vitoria-Gasteiz, Araba, Spain, 01009 Collapse << |
| NCT00759564 | - | - | Completed | - | - |
| NCT00993967 | - | - | Completed | - | - |
| NCT00663208 | - | - | Completed | - | - |
| NCT01175135 | Schizophrenia | Phase 2 | Completed | - | - |
| NCT00993967 | Freidreich's Ataxia | Phase 3 | Completed | - | Austria ... more >> Universitätsklinik Innsbruck Innsbruck, Austria Belgium Hôpital Erasme - Univeristé Libre de Bruxelles Bruxelles, Belgium, 1070 France Hôpital de la Salpétrière - INSERM U679, Neurologie et Thérapeutique expérimentale Paris, France, 75651 Germany HELIOS Klinikum Berlin Berlin, Germany, 13125 Neurologische Universitätsklinik und Ploklinik - Universitätsklimikum Bonn Bonn, Germany, 53105 Klinik II, Neuropädiatrie und Muskelerkrankungen - Universitätsklinik Freiburg Freiburg, Germany, 79106 Zentrum für Neurologische Medizin Göttingen, Germany, 37073 UKE Hamburg Neuropädiatrie - Zentrum für Frauen, Kinder und Jugendmedizin Hamburg, Germany, 20246 Neurologische Klinik - Klinikum Grosshadern München, Germany, 81377 Neurologische Universitätsklinik und Poliklinik Tübingen, Germany, 72076 Netherlands University medical Center Groningen Groningen, Netherlands, 9700 RB Collapse << |
| NCT00094653 | Melanoma Meta... more >>stases Collapse << | Phase 3 | Completed | - | - |
| NCT00759564 | Pneumonia | Phase 1 | Completed | - | United States, Indiana ... more >> Centurion Clinical Research Indianapolis, Indiana, United States, 46260 Belgium Pfizer Clinical Research Unit Bruxelles, Belgium, 1070 Collapse << |
| NCT00442416 | Anemia | Phase 3 | Terminated(Strategic decision ... more >>unrelated to safety or efficacy) Collapse << | - | - |
| NCT01525550 | - | - | Completed | - | - |
| NCT01192191 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 811-2201 GSK Investigational Site Fukuoka, Japan, 819-8555 GSK Investigational Site Fukushima, Japan, 964-0871 GSK Investigational Site Gunma, Japan, 371-0048 GSK Investigational Site Hiroshima, Japan, 732-0057 GSK Investigational Site Hokkaido, Japan, 001-0901 GSK Investigational Site Hokkaido, Japan, 064-0915 GSK Investigational Site Hokkaido, Japan, 070-8644 GSK Investigational Site Hyogo, Japan, 651-0073 GSK Investigational Site Ibaraki, Japan, 300-0053 GSK Investigational Site Ibaraki, Japan, 310-0015 GSK Investigational Site Ishikawa, Japan, 920-8610 GSK Investigational Site Kagawa, Japan, 760-0073 GSK Investigational Site Kagawa, Japan, 763-8502 GSK Investigational Site Kanagawa, Japan, 239-0821 GSK Investigational Site Kyoto, Japan, 601-1495 GSK Investigational Site Kyoto, Japan, 615-8087 GSK Investigational Site Miyagi, Japan, 981-8563 GSK Investigational Site Miyagi, Japan, 984-8560 GSK Investigational Site Nagano, Japan, 390-0303 GSK Investigational Site Nagano, Japan, 390-0832 GSK Investigational Site Nagano, Japan, 390-8601 GSK Investigational Site Nagano, Japan, 391-0011 GSK Investigational Site Oita, Japan, 870-0921 GSK Investigational Site Oita, Japan, 876-0047 GSK Investigational Site Okayama, Japan, 701-0304 GSK Investigational Site Osaka, Japan, 530-0012 GSK Investigational Site Osaka, Japan, 545-8586 GSK Investigational Site Osaka, Japan, 576-0016 GSK Investigational Site Osaka, Japan, 589-0022 GSK Investigational Site Tokyo, Japan, 185-0014 GSK Investigational Site Tokyo, Japan, 187-0024 GSK Investigational Site Toyama, Japan, 930-0194 GSK Investigational Site Wakayama, Japan, 641-8510 GSK Investigational Site Yamanashi, Japan, 400-0031 Collapse << |
| NCT01175135 | - | - | Completed | - | - |
| NCT00442416 | - | - | Terminated(Strategic decision ... more >>unrelated to safety or efficacy) Collapse << | - | - |
| NCT01192191 | - | - | Completed | - | - |
| NCT00094653 | - | - | Completed | - | - |
| NCT00819156 | Prostate Cancer | Phase 2 | Completed | - | - |
| NCT01475461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01517373 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00674817 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | New Zealand ... more >> GSK Investigational Site Wellington, New Zealand, 6035 Thailand GSK Investigational Site Chiangmai, Thailand, 50200 GSK Investigational Site Khon Kaen, Thailand, 40002 United Kingdom GSK Investigational Site Manchester, United Kingdom, M23 9LT Collapse << |
| NCT01517373 | - | - | Completed | - | - |
| NCT01121393 | Carcinoma, Non-Small-Cell Lung... more >> Adenocarcinoma Collapse << | Phase 3 | Completed | - | China ... more >> Beijing Chao-Yang Hospital Beijing, China, 100020 307 Hospital of PLA Beijing, China, 100071 Peking Union Medical College Hospital Beijing, China, 100730 Beijing Chest Hospital Beijing, China, 101149 First Hospital of Jilin University Changchun, China, 130021 Xiangya Hospital, Central South University Changsha, China, 410008 Hunan Province Tumor Hospital Changsha, China West China Hospital Chengdu, China, 610041 Fujian Provincial Tumor Hospital Fuzhou, China, 350014 Guangdong General Hospital Guangzhou, China, 510080 Guangzhou Institute of Respiratory Disease Guangzhou, China, 510120 NanFang Hosptial Guangzhou, China, 510515 The Third Affiliated Hospital of Harbin Medical University Haerbin, China, 150081 Zhejiang Cancer Hospital Hangzhou, China, 310022 Hubei Cancer Hospital HongShan, China Yunnan Provincial Tumor Hospital Kunming, China, 650118 Lin Yi Tumor Hospital Linyi, China, 276001 The Affiliated Cancer Hospital, Guangxi Medical University Nan Ning, China the 81th Hospital of PLA Nanjing, China, 210002 Jiangsu Cancer Hospital Nanjing, China, 210009 The affiliated hospital of medicalcollege qingdao university Qingdao, China, 266101 Shanghai Changzheng Hospital Shanghai, China, 200003 Shanghai Chest Hospital Shanghai, China, 200030 Zhongshan Hospital Fudan University Shanghai, China, 200032 Shanghai Pulmonary Hospital Shanghai, China, 200433 Changhai Hospital Shanghai, China, 200443 The First Hospital of Chinese Medical University Shenyang, China, 110001 Hebei Provincial Tumor Hospital Shijiazhuang, China Tangdu Hospital Xi'An, China, 710038 Northern Jiangsu People's Hospital Yangzhou, China, 225001 Korea, Republic of Kosin University Gospel Hospital Busan, Korea, Republic of, 602-702 Chungbuk National University Hospital Cheongju, Korea, Republic of, 361711 Yeungnam University Medical Center Daegu, Korea, Republic of, 705-717 Konkuk University Medical Center Seoul, Korea, Republic of, 143-729 Korea University Guro Hospital Seoul, Korea, Republic of, 152-703 Thailand Songklanagarind Hospital Songkla, Thailand, 90110 Collapse << |
| NCT03101722 | Presbycusis T... more >>innitus Cognitive Impairment Collapse << | Not Applicable | Enrolling by invitation | December 1, 2021 | China, Shanghai ... more >> Huadong Hospital Shanghai, Shanghai, China, 200040 Jinshan Zhong Ren geriatric care hospital Shanghai, Shanghai, China, 200040 Jiuting Community Health Service Shanghai, Shanghai, China, 200040 Collapse << |
| NCT01475461 | - | - | Completed | - | - |
| NCT00632424 | Advanced Solid Tumors | Phase 1 | Terminated(Business Objectives... more >> Changed) Collapse << | - | United States, California ... more >> Stanford University Stanford, California, United States, 94305 United States, District of Columbia Georgetown University Medical Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT00674817 | - | - | Completed | - | - |
| NCT00632424 | - | - | Terminated(Business Objectives... more >> Changed) Collapse << | - | - |
| NCT00053482 | Smallpox | Phase 2 | Completed | - | United States, Kansas ... more >> PRA International Lenexa, Kansas, United States, 66219 United States, Missouri Bio-Kinetic Clinical Applications Springfield, Missouri, United States, 65802 United States, Rhode Island Memorial Hospital of Rhode Island Division of Infectious Diseases Pawtucket, Rhode Island, United States, 02860 Collapse << |
| NCT01121393 | - | - | Completed | - | - |
| NCT02294734 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Edegem, Belgium, 2650 GSK Investigational Site Erpent, Belgium, 5101 Denmark GSK Investigational Site Aalborg, Denmark, DK-9100 GSK Investigational Site Hellerup, Denmark, 2900 GSK Investigational Site Hvidovre, Denmark, 2650 GSK Investigational Site Kobenhavn, Denmark, DK-2400 GSK Investigational Site Odense, Denmark, 5000 Odense C Netherlands GSK Investigational Site Almelo, Netherlands, 7609 PP GSK Investigational Site Eindhoven, Netherlands, 5623 EJ GSK Investigational Site Zutphen, Netherlands, 7207 AE Romania GSK Investigational Site Bucharest, Romania, 050159 GSK Investigational Site Timisoara, Romania, 300310 Russian Federation GSK Investigational Site Ekaterinburg, Russian Federation, 620109 GSK Investigational Site Kemerovo, Russian Federation, 650002 GSK Investigational Site Moscow, Russian Federation, 105 077 GSK Investigational Site Saint Petersburg, Russian Federation, 194291 GSK Investigational Site Vladimir, Russian Federation, 600023 GSK Investigational Site Yaroslavl, Russian Federation, 150003 Collapse << |
| NCT00819156 | - | - | Completed | - | - |
| NCT00053482 | - | - | Completed | - | - |
| NCT01342211 | Hypercholesterolemia ... more >> Dyslipidemia Collapse << | Phase 2 | Completed | - | - |
| NCT00366080 | Narcolepsy | Phase 2 | Terminated(Study was terminate... more >>d based on the interim results of a futility test.) Collapse << | - | Austria ... more >> GSK Investigational Site Innsbruck, Austria, A-6020 GSK Investigational Site Vienna, Austria, A-1190 Finland GSK Investigational Site Espoo, Finland, 02980 Germany GSK Investigational Site Regensburg, Bayern, Germany, 93053 GSK Investigational Site Schwalmstadt, Hessen, Germany, 34613 GSK Investigational Site Schwerin, Mecklenburg-Vorpommern, Germany, 19053 GSK Investigational Site Bochum, Nordrhein-Westfalen, Germany, 44789 GSK Investigational Site Berlin, Germany, 14050 Netherlands GSK Investigational Site Leiden, Netherlands, 2333 CL Spain GSK Investigational Site Barcelona, Spain, 08035 GSK Investigational Site Barcelona, Spain, 08036 United Kingdom GSK Investigational Site Cambridge, Cambridgeshire, United Kingdom, CB3 8RE GSK Investigational Site Edinburgh, United Kingdom, EH3 6HP GSK Investigational Site London, United Kingdom, W1G 6BF Collapse << |
| NCT02294734 | - | - | Completed | - | - |
| NCT00964158 | Influenza | Phase 3 | Completed | - | Spain ... more >> GSK Investigational Site Bilbao, Spain, 48013 GSK Investigational Site Burgos, Spain, 09005 GSK Investigational Site Madrid, Spain, 28046 GSK Investigational Site Móstoles/Madrid, Spain, 28935 GSK Investigational Site Sevilla, Spain, 41013 Collapse << |
| NCT01063062 | Rheumatoid Arthritis | Phase 3 | Completed | - | Egypt ... more >> Alexandria University Hospital; Rheumatology Alexandria, Egypt Alexandria University Alexandria, Egypt Banha Educational Hospital; Rheumatology Banha, Egypt Kasr El Ainy Hospital; Rheumatology Cairo, Egypt, 11562 Ain Shams University Hospital; Rheumatology Cairo, Egypt Bab El Sheereya Hospital; Rheumatology Cairo, Egypt El Hussein University Hospital; Rheumatology Cairo, Egypt Kasr El Ainy Hospital Cairo, Egypt Manial Specialized Hospital; Rheumatology Cairo, Egypt Collapse << |
| NCT01342211 | - | - | Completed | - | - |
| NCT00272987 | Neoplasms, Breast | Phase 3 | Active, not recruiting | December 31, 2018 | - |
| NCT00964158 | - | - | Completed | - | - |
| NCT03669757 | Psoriasis Vulgaris | Phase 1 | Recruiting | February 2019 | Germany ... more >> Investigational Site Recruiting Schwerin, Mecklenburg-Vorpommern, Germany, 19055 Investigational Site Recruiting Hamburg, Germany, 20095 Collapse << |
| NCT01063062 | - | - | Completed | - | - |
| NCT02683746 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT02287779 | Non-Alcoholic Steatohepatitis | Phase 1 | Completed | - | United States, Tennessee ... more >> New Orleans Center for Clinical Research Knoxsville, Tennessee, United States, 37920 Collapse << |
| NCT02683746 | - | - | Completed | - | - |
| NCT00272987 | - | - | Active, not recruiting | - | - |
| NCT02609048 | Primary Biliary Cirrhosis (PBC... more >>) Collapse << | Phase 2 | Terminated(Study stopped early... more >> after review of safety and efficacy demonstrated efficacy proof-of-concept and need for dose reduction.) Collapse << | - | - |
| NCT01134939 | - | - | Completed | - | - |
| NCT01134939 | - | - | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site 23 Aachen, Germany Boehringer Ingelheim Investigational Site 10 Berlin, Germany Boehringer Ingelheim Investigational Site 11 Berlin, Germany Boehringer Ingelheim Investigational Site 14 Berlin, Germany Boehringer Ingelheim Investigational Site 19 Berlin, Germany Boehringer Ingelheim Investigational Site 26 Berlin, Germany Boehringer Ingelheim Investigational Site 28 Berlin, Germany Boehringer Ingelheim Investigational Site 33 Berlin, Germany Boehringer Ingelheim Investigational Site 4 Berlin, Germany Boehringer Ingelheim Investigational Site 5 Berlin, Germany Boehringer Ingelheim Investigational Site 8 Berlin, Germany Boehringer Ingelheim Investigational Site 9 Berlin, Germany Boehringer Ingelheim Investigational Site 31 Chemnitz, Germany Boehringer Ingelheim Investigational Site 29 Dortmund, Germany Boehringer Ingelheim Investigational Site 27 Düsseldorf, Germany Boehringer Ingelheim Investigational Site 17 Frankfurt, Germany Boehringer Ingelheim Investigational Site 1 Frankfurt, Germany Boehringer Ingelheim Investigational Site 2 Freiburg, Germany Boehringer Ingelheim Investigational Site 34 Giessen, Germany Boehringer Ingelheim Investigational Site 22 Hamburg, Germany Boehringer Ingelheim Investigational Site 6 Hamburg, Germany Boehringer Ingelheim Investigational Site 12 Hannover, Germany Boehringer Ingelheim Investigational Site 16 Hannover, Germany Boehringer Ingelheim Investigational Site 32 Karlsruhe, Germany Boehringer Ingelheim Investigational Site 15 Koblenz, Germany Boehringer Ingelheim Investigational Site 25 Köln, Germany Boehringer Ingelheim Investigational Site 21 Leipzig, Germany Boehringer Ingelheim Investigational Site 20 München, Germany Boehringer Ingelheim Investigational Site 24 München, Germany Boehringer Ingelheim Investigational Site 3 München, Germany Boehringer Ingelheim Investigational Site 7 München, Germany Boehringer Ingelheim Investigational Site 13 Münster, Germany Boehringer Ingelheim Investigational Site 18 Osnabrück, Germany Boehringer Ingelheim Investigational Site 30 Stuttgart, Germany Collapse << |
| NCT02287779 | - | - | Completed | - | - |
| NCT01342926 | Atrophy, Geographic | Phase 2 | Completed | - | - |
| NCT01245140 | Psoriasis | Phase 2 | Completed | - | France ... more >> GSK Investigational Site Paris Cedex 10, France, 75475 Germany GSK Investigational Site Muenchen, Bayern, Germany, 80337 GSK Investigational Site Witten, Nordrhein-Westfalen, Germany, 58453 GSK Investigational Site Berlin, Germany, 10117 GSK Investigational Site Berlin, Germany, 10827 GSK Investigational Site Hamburg, Germany, 20354 Netherlands GSK Investigational Site Nijmegen, Netherlands, 6525 GA United Kingdom GSK Investigational Site London, United Kingdom, SE1 7EH Collapse << |
| NCT01245140 | - | - | Completed | - | - |
| NCT01642407 | Pulmonary Arterial Hypertensio... more >>n Hypertension, Pulmonary Collapse << | Phase 4 | Completed | - | Japan ... more >> Kitasato University Hospital Sagamihara, Kanagawa, Japan, 252-0375 Osaka University Hospital Suita, Osaka, Japan, 565-0871 Toho University Omori Medical Center Ota-ku, Tokyo, Japan, 143-8541 National Center for Child Health and Development Setagaya-ku, Tokyo, Japan, 157-8535 Shizuoka Children's Hospital Shizuoka, Japan, 420-8660 Collapse << |
| NCT01642407 | - | - | Completed | - | - |
| NCT02213263 | Follicular Lymphoma | Phase 3 | Completed | - | - |
| NCT02213263 | - | - | Completed | - | - |
| NCT01342926 | - | - | Completed | - | - |
| NCT03184987 | Asthma | Phase 3 | Active, not recruiting | June 24, 2019 | Japan ... more >> GSK Investigational Site Gifu, Japan, 509-6134 GSK Investigational Site Gunma, Japan, 372-0831 GSK Investigational Site Gunma, Japan, 373-0807 GSK Investigational Site Hiroshima, Japan, 732-0052 GSK Investigational Site Kagawa, Japan, 762-8550 GSK Investigational Site Kanagawa, Japan, 236-0004 GSK Investigational Site Kyoto, Japan, 607-8062 GSK Investigational Site Okinawa, Japan, 901-2121 GSK Investigational Site Tokyo, Japan, 103-0027 GSK Investigational Site Tokyo, Japan, 134-0083 GSK Investigational Site Tokyo, Japan, 157-0066 GSK Investigational Site Tokyo, Japan, 169-0073 GSK Investigational Site Toyama, Japan, 937-0042 Collapse << |
| NCT01706926 | Rheumatoid Arthritis | Phase 2 | Completed | - | - |
| NCT03681067 | Crohn Disease | Phase 1 Phase 2 | Not yet recruiting | August 2020 | United Kingdom ... more >> University Hospitals Birmingham NHS Foundation Trust Not yet recruiting Birmingham, United Kingdom, B15 2TH Contact: Marietta Iacucci +44 (0)121 3718119 m.iacucci@bham.ac.uk Contact: Manpreet K Wilkhu +44(0)121 3718165 m.k.wilkhu@bham.ac.uk Collapse << |
| NCT01706926 | - | - | Completed | - | - |
| NCT01154101 | Psoriasis | Phase 2 | Completed | - | United States, Missouri ... more >> GSK Investigational Site Saint Louis, Missouri, United States, 63117 United States, New York GSK Investigational Site New York, New York, United States, 10016 GSK Investigational Site New York, New York, United States, 10065 United States, Oregon GSK Investigational Site Portland, Oregon, United States, 97223 United States, Pennsylvania GSK Investigational Site Philadelphia, Pennsylvania, United States, 19103 United States, Rhode Island GSK Investigational Site Johnston, Rhode Island, United States, 02919 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75246 United States, Washington GSK Investigational Site Seattle, Washington, United States, 98101 Collapse << |
| NCT00975884 | Influenza | Phase 3 | Completed | - | France ... more >> GSK Investigational Site Clermont-Ferrand, France, 63003 GSK Investigational Site Lille, France, 59037 GSK Investigational Site Paris Cedex 18, France, 75877 GSK Investigational Site Paris, France, 75679 GSK Investigational Site Poitiers, France, 86000 Collapse << |
| NCT01017575 | Hepatitis C Infection | Phase 2 | Completed | - | Japan ... more >> Local Institution Chiba-Shi, Chiba, Japan Local Institution Kurume-Shi, Fukuoka, Japan, 8300011 Local Institution Okayama-Shi, Okayama, Japan, 7008558 Local Institution Osaka-Shi, Osaka, Japan, 5438555 Local Institution Osaka-Shi, Osaka, Japan, 545-8586 Local Institution Musashino-Shi, Tokyo, Japan, 180-0023 Collapse << |
| NCT01128621 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Chula Vista, California, United States, 91910 United States, Florida GSK Investigational Site Miami, Florida, United States, 33169 Collapse << |
| NCT01362296 | Lung Cancer, Non-Small Cell | Phase 2 | Completed | - | - |
| NCT03486873 | Solid Tumors | Phase 3 | Recruiting | May 10, 2028 | - |
| NCT01154101 | - | - | Completed | - | - |
| NCT00095173 | Juvenile Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT01128621 | - | - | Completed | - | - |
| NCT00095173 | - | - | Completed | - | - |
| NCT03682848 | HIV Infections | Phase 3 | Not yet recruiting | December 29, 2023 | - |
| NCT01865266 | Ventilator-associated Bacteria... more >>l Pneumonia Mechanical Ventilation Complication Collapse << | Phase 2 Phase 3 | Unknown | June 2016 | China ... more >> Shanghai Minhang Central Hospital Not yet recruiting Shanghai, China, 201199 Contact: Yu YueTian, MD 13818227011 fishyyt@sina.com Contact: Liu ChunYan, MD 13917836226 doctorlcy@sina.com Principal Investigator: Yu YueTian, MD Sub-Investigator: Mao EnQiang, MD Collapse << |
| NCT01362296 | - | - | Completed | - | - |
| NCT02670642 | Gastroesophageal Reflux | Phase 3 | Completed | - | - |
| NCT01017575 | - | - | Completed | - | - |
| NCT03315091 | Solid Tumours | Phase 1 | Completed | - | France ... more >> Research Site Bordeaux Cedex, France, 33075 Research Site Saint Herblain, France, 44805 Netherlands Research Site Amsterdam, Netherlands, 1066 CX Research Site Amsterdam, Netherlands, 1081 HV Research Site Maastricht, Netherlands, 6229 HX United Kingdom Research Site Glasgow, United Kingdom, G12 0YN Research Site Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00527735 | Lung Cancer S... more >>mall Cell Lung Cancer Carcinoma, Non-Small-Cell Lung Collapse << | Phase 2 | Completed | - | - |
| NCT02145442 | Overweight Ob... more >>esity Collapse << | Phase 2 Phase 3 | Completed | - | Cuba ... more >> National Institute of Endocrinology Plaza de la Revolución, Havana, Cuba, 10400 Collapse << |
| NCT02455011 | Type 2 Diabetes Mellitus ... more >> Diabetes Collapse << | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Miami, Florida, United States United States, Texas San Antonio, Texas, United States United States, Washington Renton, Washington, United States Collapse << |
| NCT00527735 | - | - | Completed | - | - |
| NCT01058395 | Traumatic Brain Injury | Phase 1 Phase 2 | Completed | - | United States, Michigan ... more >> Oakwood Dearborn Hospital Dearborn, Michigan, United States, 48124 Oakwood Southshore Hospital Trenton, Michigan, United States, 48183 Collapse << |
| NCT01396187 | - | - | Completed | - | - |
| NCT01058395 | - | - | Completed | - | - |
| NCT03441984 | HIV Infections | Phase 1 | Completed | - | United States, Texas ... more >> GSK Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT00814489 | Streptococcus Pneumoniae ... more >> Haemophilus Influenzae Collapse << | Phase 1 | Completed | - | Sweden ... more >> GSK Investigational Site Karlskrona, Sweden, SE-371 41 Collapse << |
| NCT03330847 | Metastatic Triple Negative Bre... more >>ast Cancer Collapse << | Phase 2 | Recruiting | January 20, 2021 | - |
| NCT01396187 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site Miami, Florida, United States, 33169 Pfizer Investigational Site South Miami, Florida, United States, 33143 United States, Kansas Pfizer Investigational Site Overland Park, Kansas, United States, 66212 United States, Texas Pfizer Investigational Site San Antonio, Texas, United States, 78229 Collapse << |
| NCT00031512 | Enteroviral Sepsis | Phase 2 | Completed | - | - |
| NCT03615534 | Atherogenic Dyslipidemia ... more >> Obesity Associated Disorder Collapse << | Phase 4 | Completed | - | Iraq ... more >> Al Kindy College of Medicine, University of Baghdad Baghdad, Iraq, 10045 Lewai S Abdulaziz Baghdad, Iraq, 10045 Collapse << |
| NCT02571088 | Sickle Cell Hemoglobin C Disea... more >>se Hemoglobin S Disease Collapse << | Not Applicable | Completed | - | France ... more >> Hopital Avicenne Bobigny, France, 93000 Centre hospitalier sud francilien Corbeil-essonnes, France, 91100 CHU Henri MONDOR Creteil, France, 94010 CHU Kremlin-Bicêtre Le Kremlin Bicetre, France, 93270 Hopital Europeen Georges POMPIDOU Paris, France, 75015 Hopital Necker Paris, France, 75015 Hopital Tenon Paris, France, 75020 Centre hospitalier de Saint-Denis Saint-denis, France, 93200 CHU de SAINT-ETIENNE Saint-etienne, France, 42000 Collapse << |
| NCT00814489 | - | - | Completed | - | - |
| NCT03222206 | Non Alcoholic Fatty Liver ... more >> Osteo Arthritis Collapse << | Phase 4 | Active, not recruiting | February 22, 2019 | Korea, Republic of ... more >> Korea University Guro Hospital Seoul, Korea, Republic of, 08308 Collapse << |
| NCT02158351 | - | - | Unknown | October 2014 | Italy ... more >> Stefano Ginanni Corradini Recruiting Rome, Italy, 00161 Contact: Stefano Ginanni Corradini, MD, PhD +39 0649972086 stefano.corradini@uniroma1.it Collapse << |
| NCT03029247 | Anaemia | Phase 2 | Recruiting | June 5, 2019 | United States, California ... more >> GSK Investigational Site Recruiting La Mesa, California, United States, 91942 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Colorado GSK Investigational Site Not yet recruiting Lakewood, Colorado, United States, 80228 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Florida GSK Investigational Site Terminated Miami, Florida, United States, 33176 GSK Investigational Site Not yet recruiting Orlando, Florida, United States, 32809 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Illinois GSK Investigational Site Recruiting Chicago, Illinois, United States, 60643 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com United States, Minnesota GSK Investigational Site Completed Minneapolis, Minnesota, United States, 55404 United States, Texas GSK Investigational Site Not yet recruiting San Antonio, Texas, United States, 78215 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03207243 | Asthma | Phase 1 Phase 2 | Active, not recruiting | May 23, 2019 | - |
| NCT01266967 | Melanoma and Brain Metastases | Phase 2 | Completed | - | - |
| NCT03615534 | - | - | Completed | - | - |
| NCT02995005 | Hepatitis B | Phase 1 Phase 2 | Recruiting | December 31, 2019 | Thailand ... more >> Shoklo Malaria Research Unit Recruiting Mae Sot, Thailand Contact: Rose McGready +66 5554 5021 SMRU@tropmedres.ac Collapse << |
| NCT02717507 | Cancer Survivor | Phase 2 | Recruiting | - | - |
| NCT03766399 | Asthma | Phase 1 | Recruiting | December 5, 2019 | United Kingdom ... more >> Research Site Recruiting Harrow, United Kingdom, HA1 3UJ Research Site Not yet recruiting Manchester, United Kingdom, M23 9GP Collapse << |
| NCT01163851 | Hypercholesterolemia ... more >> Dyslipidemia Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Premier Research Group, Limited Peoria, Arizona, United States, 85381 Dedicated Phase 1, Inc. Phoenix, Arizona, United States, 85013 Premier Research Group Limited Phoenix, Arizona, United States, 85027 United States, Kansas Vince and Associates Clinical Research Overland Park, Kansas, United States, 66211 Vince and Associates Clinical Research Overland Park, Kansas, United States, 66212 Collapse << |
| NCT03446612 | Anaemia | Phase 2 | Not yet recruiting | December 2, 2019 | United Kingdom ... more >> GSK Investigational Site Not yet recruiting Cambridge, Cambridgeshire, United Kingdom, CB2 0QQ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com GSK Investigational Site Not yet recruiting Edinburgh, United Kingdom, EH16 4TJ Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT01266967 | - | - | Completed | - | - |
| NCT03420092 | Coronary Artery Disease | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom, HA1 3UJ Collapse << |
| NCT02193360 | Primary Biliary Cirrhosis | Phase 1 Phase 2 | Unknown | December 2017 | Netherlands ... more >> Amsterdam Medical Center Active, not recruiting Amsterdam, Netherlands, 1100 DD Erasmus University Medical Center Recruiting Rotterdam, Netherlands Contact: H R van Buuren, MD mdlpoli@erasmusmc.nl Principal Investigator: H R van Buuren, MD United Kingdom University Hospital Birmingham Recruiting Birmingham, United Kingdom Contact: G Hirschfield, MD G.Hirschfield@bham.ac.uk Royal Free Hospital Recruiting London, United Kingdom Contact: D Thorburn, MD douglas.thorburn@nhs.net Newcastle upon Tyne Hospitals Recruiting Newcastle upon Tyne, United Kingdom Contact: D Jones, MD 0191 213 7210 Collapse << |
| NCT03458650 | Healthy | Phase 1 | Completed | - | China, Shanghai ... more >> Huashan Hospital, Fudan University Shanghai, Shanghai, China, 200040 Collapse << |
| NCT03739710 | Neoplasms | Phase 2 | Not yet recruiting | February 9, 2022 | - |
| NCT02909959 | Autism Spectrum Disorder ... more >> Autistic Disorder Neurodevelopmental Disorder Childhood Developmental Disorders, Pervasive Collapse << | Phase 2 | Recruiting | November 2019 | United States, North Carolina ... more >> Carolina Institute for Developmental Disabilities, University of North Carolina School of Medicine Recruiting Carrboro, North Carolina, United States, 27510 Contact: Laura Politte, M.D. 919-445-4160 laura.politte@cidd.unc.edu Contact: Morgan Parlier, MSW 919-843-8122 morgan.parlier@cidd.unc.edu Collapse << |
| NCT03455075 | Healthy Men ... more >> Male Contraception Collapse << | Phase 2 | Recruiting | June 2020 | United States, California ... more >> Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center Recruiting Torrance, California, United States, 90509 Contact: Xiaodan Han 310-222-1865 xhan@labiomed.org Principal Investigator: Christina Wang United States, Washington University of Washington Medical Center & Health Sciences Recruiting Seattle, Washington, United States, 98195 Contact: Stephanie Page, MD, PhD 206-616-1818 page@uw.edu Collapse << |
| NCT01163851 | - | - | Completed | - | - |
| NCT00816400 | Cancer | Phase 1 | Completed | - | United States, Colorado ... more >> Research Site Denver, Colorado, United States United States, Indiana Research Site Indianapolis, Indiana, United States United States, Nevada Research Site Las Vegas, Nevada, United States United States, Texas Research Site Dallas, Texas, United States United States, Virginia Research Site Norfolk, Virginia, United States Collapse << |
| NCT00816400 | - | - | Completed | - | - |
| NCT03633227 | Liver Cirrhosis, Biliary | Phase 4 | Recruiting | June 1, 2020 | United States, California ... more >> Inland Empire Liver Foundation Recruiting Rialto, California, United States, 92377 Contact: Darlene Romo 909-883-2999 dromo@ieliverfoundation.com Contact: Nazly Amaya 909-883-2999 namaya@ieliverfoundation.com Principal Investigator: Zeid Kayali, MD United States, Texas American Research Corporation at Texas Liver Institute Recruiting San Antonio, Texas, United States, 78215 Contact: Nicole Forsyth 210-253-3426 nforsyth@txliver.com Contact: Elisha Moreno 210-253-3426 emoreno@txliver.com Principal Investigator: Eric Lawitz, MD Collapse << |
| NCT02640495 | - | - | Withdrawn(Due to the relative ... more >>low incidence of severe malaria in Southeast Asia.) Collapse << | - | - |
| NCT02006628 | Schizophrenia ... more >> Schizophrenia-related Psychotic Disorder Collapse << | Phase 2 | Completed | - | Poland ... more >> Investigator 7 Bytomska, Poland Investigator 4 Gdansk, Poland Investigator 9 Katowice, Poland Investigator 5 Kielce, Poland Investigator 10 Lublin, Poland Investigator 8 Wrocław, Poland Romania Investigator 11 Bucharest, Romania Investigator 12 Bucharest, Romania Investigator 15 Bucharest, Romania Investigator 14 Sibiu, Romania Investigator 16 Targoviste, Romania Investigator 13 Targu Mures, Romania United Kingdom Investigator 2 Chertsey, United Kingdom Investigator 1 Coventry, United Kingdom Investigator 3 London, United Kingdom Collapse << |
| NCT02314936 | Functional Constipation | Phase 2 | Completed | - | Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << |
| NCT03568942 | Infections, Bacterial | Phase 2 | Recruiting | December 14, 2018 | United States, California ... more >> GSK Investigational Site Recruiting La Mesa, California, United States, 91942 Contact: US GSK Clinical Trials Call Center 877-379-3718 GSKClinicalSupportHD@gsk.com Contact: EU GSK Clinical Trials Call Center +44 (0) 20 8990 4466 GSKClinicalSupportHD@gsk.com Collapse << |
| NCT03700424 | Acute Coronary Syndrome | Phase 2 | Not yet recruiting | December 2019 | - |
| NCT01862263 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | Mexico ... more >> Novartis Investigative Site Mexico, Distrito Federal, Mexico, 06700 Novartis Investigative Site Mexico, Distrito Federal, Mexico, 07300 Novartis Investigative Site Mexico, Distrito Federal, Mexico, 14050 Novartis Investigative Site Metepec, Estado de México, Mexico, 52140 Novartis Investigative Site Celaya, Guanajuato, Mexico, 38000 Novartis Investigative Site Guadalajara, Jalisco, Mexico, 44150 Novartis Investigative Site Guadalajara, Jalisco, Mexico, 44600 Novartis Investigative Site Guadalajara, Jalisco, Mexico, 44670 Novartis Investigative Site Monterrey, Nuevo Leon, Mexico, 64710 Novartis Investigative Site Monterrey, Nuevo León, Mexico, 64020 Novartis Investigative Site Cancun, Quintana Roo, Mexico, 77500 Novartis Investigative Site Culiacán, Sinaloa, Mexico, 80000 Novartis Investigative Site Puebla, Mexico, 72190 Collapse << |
| NCT02500186 | Diabetes Melltius, Type 2 | Not Applicable | Completed | - | - |
| NCT03104569 | Cholangitis | Not Applicable | Recruiting | April 1, 2019 | China, Gansu ... more >> The first hospital of Lanzhou university Recruiting Lanzhou, Gansu, China, 730000 Contact: Wence Zhou, M. D. +8609318356919 zhouwc129@163.com Contact: Wenbo Meng, M. D. +8609318356213 mengwb@lzu.edu.cn China, Shaanxi The first affiliated hospital of Xi 'an jiaotong university Not yet recruiting Xi'an, Shaanxi, China, 710061 Contact: Hao Sun, M. D. +86 13891813691 sunhaoxjfy@126.com China, Tianjin Tianjin Nankai Hospital Not yet recruiting Tianjin, Tianjin, China, 300100 Contact: Zhenyu Wang, M. D. +86 15302021661 Wangzytj@163.com Collapse << |
| NCT03580655 | Advanced Systemic Mastocytosis... more >> Aggressive Systemic Mastocytosis Systemic Mastocytosis With an Associated Hematologic Neoplasm Mast Cell Leukemia Collapse << | Phase 2 | Recruiting | November 2023 | United States, California ... more >> Stanford Cancer Institute Recruiting Stanford, California, United States, 94305 United States, Massachusetts Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 United States, New York Herbert Irving Comprehensive Cancer Center Recruiting New York, New York, United States, 10032 United States, Utah Huntsman Cancer Institute Recruiting Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02868177 | Prediabetes T... more >>ype 2 Diabetes Hypertriglyceridemia Obesity Disease Insulin Resistance Collapse << | Phase 2 Phase 3 | Active, not recruiting | August 2019 | France ... more >> Institut Pasteur de Lille Lille, France, 59019 Biofortis Mérieux NutriSciences Clinical Investigation Center Saint Herblain, France, 44800 Ireland Atlantia Food Clinical Trials Cork, Ireland, T23 R50R Serbia Clinical center of Kragujevac, / Poseidon CRO Kragujevac, Serbia Clinical Center of Vojvodina / Poseidon CRO Novi Sad, Serbia, 21000 Slovenia University Medical Centre Ljubljana / Poseidon CRO Ljubljana, Slovenia Collapse << |
| NCT02590887 | Healthy Volunteers | Phase 1 Phase 2 | Unknown | - | Spain ... more >> Hospital Universitario Virgen del Rocío Seville, Spain, 41013 Collapse << |
| NCT03583281 | Obesity Infla... more >>mmation Collapse << | Not Applicable | Recruiting | April 2022 | Canada, Manitoba ... more >> I.H. Asper Clinical Research Institute Recruiting Winnipeg, Manitoba, Canada, R2H 2A6 Contact: Carla G Taylor, PhD 204-258-1361 ctaylor@sbrc.ca Contact: Peter Zahradka, PhD 204-235-3507 pzahradka@sbrc.ca Principal Investigator: Carla G Taylor, PhD Sub-Investigator: Peter Zahradka, PhD Sub-Investigator: Harold M Aukema, PhD Collapse << |
| NCT02314936 | - | - | Completed | - | - |
| NCT02966834 | Cholestasis | Phase 2 | Recruiting | April 1, 2020 | - |
| NCT00369317 | Childhood Acute Basophilic Leu... more >>kemia Childhood Acute Eosinophilic Leukemia Childhood Acute Erythroleukemia (M6) Childhood Acute Megakaryocytic Leukemia (M7) Childhood Acute Minimally Differentiated Myeloid Leukemia (M0) Childhood Acute Monoblastic Leukemia (M5a) Childhood Acute Monocytic Leukemia (M5b) Childhood Acute Myeloblastic Leukemia With Maturation (M2) Childhood Acute Myeloblastic Leukemia Without Maturation (M1) Childhood Acute Myelomonocytic Leukemia (M4) Childhood Myelodysplastic Syndromes de Novo Myelodysplastic Syndromes Secondary Acute Myeloid Leukemia Secondary Myelodysplastic Syndromes Untreated Childhood Acute Myeloid Leukemia and Other Myeloid Malignancies Collapse << | Phase 3 | Completed | - | - |
| NCT03523858 | Progressive Multiple Sclerosis... more >> (PMS) Collapse << | Phase 3 | Recruiting | January 31, 2024 | - |
| NCT02258529 | - | - | Terminated | - | - |
| NCT00855465 | Pulmonary Hypertension | Phase 3 | Completed | - | - |
| NCT00369317 | - | - | Completed | - | - |
| NCT00397215 | Influenza | Phase 2 | Completed | - | Belgium ... more >> GSK Investigational Site Dour, Belgium, 7370 GSK Investigational Site Gozée, Belgium, 6534 GSK Investigational Site Libramont, Belgium, 6800 GSK Investigational Site Mont-Godinne, Belgium, 5530 GSK Investigational Site Sprimont, Belgium, 4140 GSK Investigational Site Tessenderlo, Belgium, 3980 GSK Investigational Site Watermael-Boitsfort, Belgium, 1170 Italy GSK Investigational Site Genova, Liguria, Italy, 16132 GSK Investigational Site Milano, Lombardia, Italy, 20127 GSK Investigational Site Monserrato Cagliari, Sardegna, Italy, 09042 GSK Investigational Site Sassari, Sardegna, Italy, 07100 GSK Investigational Site Ragusa, Sicilia, Italy, 97100 Collapse << |
| NCT02743598 | HIV Infection ... more >> Diabetes Mellitus Type 2 Obesity Overweight Metabolic Syndrome Collapse << | Phase 4 | Recruiting | May 2019 | United States, Pennsylvania ... more >> Cherie Vaz Recruiting Philadelphia, Pennsylvania, United States, 19140 Contact: Cherie Vaz, MD Dipl.ABOM cherie.vaz@tuhs.temple.edu Collapse << |
| NCT01393964 | Multiple Myeloma | Phase 1 | Completed | - | United States, Georgia ... more >> Winship Cancer Institute, Emory University Atlanta, Georgia, United States, 30322 United States, Indiana Investigative Clinical Research Of Indiana, Llc Indianapolis, Indiana, United States, 46260 United States, Iowa University Of Iowa Hospitals And Clinics Iowa City, Iowa, United States, 52242 United States, Maryland University Of Maryland Baltimore, Maryland, United States, 21201 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Missouri Washington University School Of Medicine Saint Louis, Missouri, United States, 63110 United States, New York Weill Cornell Medical College New York, New York, United States, 10021 Mount Sinai Medical Center New York, New York, United States, 10029 United States, Tennessee Tennessee Oncology, Pllc Nashville, Tennessee, United States, 37203 United States, Texas The University Of Texas M.D. Anderson Cancer Center Houston, Texas, United States, 77030 United States, Washington Va Puget Sound Health Care System Seattle, Washington, United States, 98108 Collapse << |
| NCT02258529 | Follicular Lymphoma ... more >> Small Lymphocytic Lymphoma Collapse << | Phase 2 | Terminated | - | United States, California ... more >> Pacific Shores Medical Group Long Beach, California, United States, 90813 United States, Florida Florida Cancer Specialists Fort Myers, Florida, United States, 33916 United States, Maryland St. Agnes Hospital Baltimore, Maryland, United States, 21229 United States, South Dakota Prarie Lakes Health Care Systems, Inc. Watertown, South Dakota, United States, 57201 United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37203 United States, Washington Northwest Medical Specialties, PLLC Tacoma, Washington, United States, 98405 Collapse << |
| NCT00397215 | - | - | Completed | - | - |
| NCT02776033 | Psoriasis | Phase 2 | Completed | - | Canada, Ontario ... more >> GSK Investigational Site Markham, Ontario, Canada, L3P1X2 GSK Investigational Site Peterborough, Ontario, Canada, K9J 5K2 GSK Investigational Site Waterloo, Ontario, Canada, N2J 1C4 Canada, Quebec GSK Investigational Site Montreal, Quebec, Canada, H2K 4L5 Collapse << |
| NCT02854631 | Alcoholic Hepatitis (AH) | Phase 2 | Completed | - | - |
| NCT01434212 | - | - | Unknown | December 2011 | China, Jilin ... more >> First Hospital Jilin University Changchun, Jilin, China, 130061 Collapse << |
| NCT01393964 | - | - | Completed | - | - |
| NCT00855465 | - | - | Completed | - | - |
| NCT02900443 | Autoimmune Hepatitis | Phase 4 | Recruiting | December 2019 | Netherlands ... more >> Vrije Universiteit Medisch Centrum Recruiting Amsterdam, Netherlands, 1081 HV Contact: Karin MJ van Nieuwkerk, MD, PhD Principal Investigator: Karin MJ van Nieuwkerk, MD, PhD Leiden University Medical Centre Recruiting Leiden, Netherlands, 2333ZA Contact: Lida Beneken Kolmer 071-526 1188 A.Beneken_Kolmer@lumc.nl Principal Investigator: Bart van Hoek, MD, PhD Sint Antonius Hospital Recruiting Nieuwegein, Netherlands, 3430 EM Contact: Robert C Verdonk, MD, PhD Principal Investigator: Robert C Verdonk, MD, PhD Radboud University Medical Centre Recruiting Nijmegen, Netherlands, 6525GA Contact: Simon Pape, MD +31-6-52621030 Simon.Pape@radboudumc.nl Principal Investigator: Joost PH Drenth, MD, PhD Sub-Investigator: Simon Pape, MD Collapse << |
| NCT01978691 | Obesity Hyper... more >>glycemia Insulin Resistance Collapse << | Phase 2 | Completed | - | Finland ... more >> VL-Medi Helsinki, Finland, 00101 Kerava healthcare center Kerava, Finland, 04200 FinnMedi Oy Tampere, Finland, 33520 CRST - Clinical Research Services Turku Turku, Finland, 20521 Collapse << |
| NCT00883753 | Rheumatoid Arthritis | Phase 3 | Completed | - | - |
| NCT03004846 | Psoriasis | Phase 2 | Completed | - | Germany ... more >> GSK Investigational Site Hamburg, Germany, 20095 Collapse << |
| NCT02584283 | Liver Failure ... more >> End Stage Liver Disease Biliary Tract Diseases Collapse << | Phase 3 | Recruiting | October 2019 | Belgium ... more >> Ghent University Hospital Not yet recruiting Gent, De Pintelaan 185, Belgium, 9000 Contact: Roberto I Troisi, MD, PhD +3293325519 roberto.troisi@ugent.be University Hospitals Leuven Recruiting Leuven, Herestraat 49, Belgium, 3000 Contact: Diethard Monbaliu, MD, PhD +3216348727 diethard.monbaliu@uzleuven.be Netherlands Leiden Universtiy Medical Center Not yet recruiting Leiden, Zuid-Holland, Netherlands, 2333 ZA Contact: Bart van Hoek, MD, PhD +31715299756 b.van_hoek@lumc.nl University Medical Center Groningen Recruiting Groningen, Netherlands, 9700 RB Contact: Rianne van Rijn, MD +31652724616 r.van.rijn@umcg.nl Contact: Robert J Porte, MD, PhD +31503619027 r.j.porte@umcg.nl Erasmus Medical Center Recruiting Rotterdam, Netherlands, 3015 CE Contact: Jeroen de Jonge, MD, PhD +31633330449 j.dejonge.1@umcg.nl Collapse << |
| NCT00883753 | - | - | Completed | - | - |
| NCT03296098 | Contraception ... more >> Healthy Female Collapse << | Phase 2 | Not yet recruiting | December 2020 | United States, California ... more >> Essential Access Not yet recruiting Los Angeles, California, United States, 90010 Contact: Anita L. Nelson, MD 213-386-5614 ext 4403 nelsona@cfhc.org Contact: Kieumai Vo 213-386-5614 ext 4592 kvo@essentialaccess.org Principal Investigator: Anita L. Nelson, MD Principal Investigator: Ron Freizeres University of California, Davis Not yet recruiting Sacramento, California, United States, 95817 Contact: Mitchell D Creinin, MD 916-734-6886 Contact: Courtney Overstreet 916-734-6227 cboverstreet@ucdavis.edu University of California, San Francisco Not yet recruiting San Francisco, California, United States, 94110 Contact: Abby Sokoloff, MPH 415-206-8715 abby.sokoloff@ucsf.edu Principal Investigator: Phillip Darney, MD United States, Colorado University of Colorado Not yet recruiting Denver, Colorado, United States, 80045 Contact: Ines Crato 303-724-5559 ines.crato@ucdenver.edu Principal Investigator: Stephanie Teal, MD United States, Hawaii University of Hawaii Not yet recruiting Honolulu, Hawaii, United States, 96826 Contact: Bliss Kaneshiro Contact: Tiana Fontanilla, MPH 808-203-6598 United States, Illinois University of Chicago Not yet recruiting Chicago, Illinois, United States, 60637 Contact: Sadia Haider Contact: Kate Lewis 773-834-5437 klewis2@uchicago.edu United States, Maryland Johns Hopkins Bayview Medical Center Not yet recruiting Baltimore, Maryland, United States, 21224 Contact: Anne E. Burke, MD, MPH 410-550-0335 aburke@jhmi.edu Contact: Katrina Thaler, MPH 410-550-8506 kstouff3@jhmi.edu Principal Investigator: Anne E. Burke, MD, MPH United States, Massachusetts Planned Parenthood League of Massachusetts Not yet recruiting Boston, Massachusetts, United States, 02215 Contact: Jennifer Fortin jfortin@pplm.org Principal Investigator: Alisa Goldberg, MD United States, New York New York University, School of Medicine Not yet recruiting New York, New York, United States, 10016 Contact: Treasure Walker Contact: Anna Davis 212-263-6253 anna.davis@nyumc.org Columbia University Not yet recruiting New York, New York, United States, 10032 Contact: Carolyn Westhoff, MD 212-305-4805 clw3@columbia.edu Contact: Claudia Roca 212-342-2986 cr479@mail.cumc.columbia.edu Principal Investigator: Carolyn Westhoff, MD United States, Ohio University of Cincinnati Not yet recruiting Cincinnati, Ohio, United States, 45267 Contact: Michael Thomas, MD 513-584-4130 Contact: Tiffany Rupert 513-584-4130 rupertts@ucmail.uc.edu University Hospitals of Cleveland MacDonald Women's Hospital Not yet recruiting Cleveland, Ohio, United States, 44106 Contact: James H. Liu, MD 440-720-3250 james.liu@uhhs.com Contact: Bridget Ermlich 216-844-8091 bridget.ermlich@uhhospitals.org Principal Investigator: James H. Liu, MD United States, Oregon Oregon Health & Science University Not yet recruiting Portland, Oregon, United States, 97239 Contact: Jeffrey Jensen Contact: Sumi Maristany 503-494-0585 maristan@ohsu.edu United States, Pennsylvania University of Pennsylvania Medical Center Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Kurt T. Barnhart, MD 215-662-2974 kbarnhart@obgyn.upenn.edu Contact: Andrea Morley 215-615-4202 andrea.morley@uphs.upenn.edu Principal Investigator: Kurt T. Barnhart, MD Univeristy of Pittsburgh School of Medicine Not yet recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Beatrice Chen, MD 412-641-5496 chenba@upmc.edu Contact: Jenelle Mock 412-641-4791 mockj@upmc.edu Principal Investigator: Beatrice Chen, MD United States, Rhode Island Women & Infants Hospital of Rhode Island Not yet recruiting Providence, Rhode Island, United States, 02905 Contact: Rebecca Allen Contact: Mindy Marshall mamarshall@wihri.org United States, Utah University of Utah Not yet recruiting Salt Lake City, Utah, United States, 84106 Contact: David Turok, MD 801-213-2774 Contact: Amy Orr 801-213-2774 amy.orr@hsc.utah.edu United States, Virginia Eastern Virginia Medical School (EVMS) Not yet recruiting Norfolk, Virginia, United States, 23507 Contact: David E Archer, MD 757-446-7422 Contact: Erin Maple, MPH 757-446-7971 mapleel@evms.edu Collapse << |
| NCT02989753 | Hypercholesterolemia ... more >> Hypertriglyceridemia Collapse << | Phase 1 Phase 2 | Completed | - | France ... more >> Biofortis Mérieux NutriSciences Clinical Investigation Center Saint Herblain, France, 44800 Collapse << |
| NCT00994344 | HIV Infections | Phase 4 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Hospital Universitario de Canarias Santa Cruz de Tenerife, Canarias, Spain, 38320 Collapse << |
| NCT00622700 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00751881 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT03567499 | Amyloidosis | Phase 1 | Withdrawn(The decision to end ... more >>the study was made for GSK portfolio reasons. No subjects were randomised into the study.) Collapse << | May 3, 2019 | United Kingdom ... more >> GSK Investigational Site London, United Kingdom, NW10 7EW Collapse << |
| NCT00622700 | - | - | Completed | - | - |
| NCT00751881 | - | - | Completed | - | - |
| NCT02873559 | Muscle Weakness | Phase 2 Phase 3 | Recruiting | January 2019 | Denmark ... more >> Karsten Overgaard Recruiting Hellerup, Danmark, Denmark, DK-2900 Contact: Karsten Overgaard, neurologist +45 2617 2611 karstenovergaard20@hotmail.com Contact: Rune Rasmussen, PhD neuropsy +45 2875 7500 rsr@sund.ku.dk Principal Investigator: Mette Midttun, M.D. DMSci Medical Dept. O, Geriatric Section Recruiting Herlev, Denmark, 2730 Contact: Mette Midttun, MD, DMSc +45 38689188 mette.midttun@regionh.dk Principal Investigator: Mette Midttun, MD, DMSc Collapse << |
| NCT01791842 | Fibrous Dysplasia of Bone | Phase 2 | Unknown | May 2015 | France ... more >> Hopital Edouard Herriot - service de rhumatologie Recruiting Lyon, France, 69003 Contact: Roland Chapurlat, Professor +33472117481 Principal Investigator: Roland CHAPURLAT, Professor Sub-Investigator: Deborah GENSBURGER, Dr Hôpital Lariboisière - service de rhumatologie Not yet recruiting Paris, France, 75010 Contact: Philippe ORCEL, Professor Principal Investigator: Philippe ORCEL, Professor Collapse << |
| NCT03333447 | - | - | Recruiting | December 2019 | France ... more >> Beaujon Hospital Recruiting Clichy, France, 92110 Contact: BELMATOUG NADIA, DOCTOR +33 1 40 87 52 86 nadia.belmatoug@aphp.fr Contact: BENGHERBIA MONIA, CRA +33 1 71 11 46 08 monia.bengherbia@aphp.fr Collapse << |
| NCT01218464 | Liver Failure ... more >> Mesenchymal Stem Cells Collapse << | Phase 1 Phase 2 | Unknown | March 2014 | China, Beijing ... more >> Beijing 302 Hospital Recruiting Beijing, Beijing, China, 100039 Contact: Ming Shi, Professor 86-10-63879735 ext 2015.12 shiming302@sina.com Collapse << |
| NCT03452111 | Healthy Men ... more >> Male Contraception Collapse << | Phase 2 | Recruiting | September 2021 | United States, California ... more >> Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center Recruiting Torrance, California, United States, 90509 Contact: Xiaodan Han 310-222-1865 xhan@labiomed.org Principal Investigator: Christina Wang United States, Kansas University of Kansas Recruiting Kansas City, Kansas, United States, 66160 Contact: Katie Glavin 913-588-8721 kglavin@kumc.edu Principal Investigator: Ajay Nangia, MBBS, FACS United States, Washington University of Washington Medical Center & Health Sciences Recruiting Seattle, Washington, United States, 98195 Contact: Stephanie Page, MD, PhD 206-616-1818 page@uw.edu Chile Chilean Institute of Reproductive Medicine Not yet recruiting Santiago, Chile Contact: Veronica Reyes vreyes@icmer.org Principal Investigator: Gabriela Noe, MS Italy University of Bologna Not yet recruiting Bologna, Italy Contact: Giulia Gava gava.giulia@hotmail.it Principal Investigator: Cristina Meriggiola Kenya Kenyatta National Hospital Not yet recruiting Nairobi, Kenya Contact: Sheila Eshiwani seshiwanin@gmail.com Principal Investigator: John Kinuthia, MBChB, MPH Sweden Karolinska Institutet Not yet recruiting Stockholm, Sweden Contact: Eva Broberg 46(8)517 721 28 eva.k.broberg@karolinska.se Principal Investigator: Kristina Gemzell Danielsson, MD United Kingdom University of Edinburgh Not yet recruiting Edinburgh, United Kingdom Contact: Anne Saunderson 0131 242 2669 A.Saunderson@ed.ac.uk Principal Investigator: Richard Anderson, MD, PhD University of Manchester Not yet recruiting Manchester, United Kingdom Contact: Kate Barugh 0161 27 66201 Kate.Barugh@cmft.nhs.uk Principal Investigator: Cheryl Fitzgerald, MD Collapse << |
| NCT03183193 | Non-Alcoholic Fatty Liver Dise... more >>ase Obese Overweight Collapse << | Not Applicable | Recruiting | December 2019 | Spain ... more >> Centre for Nutrition Research, University of Navarra Recruiting Pamplona, Navarra, Spain, 31008 Contact: M. Angeles Zulet, PhD +34948425600 ext 806317 mazulet@unav.es Contact: Itziar Abete, PhD +34948425600 ext 806357 iabetego@unav.es Collapse << |
| NCT02189005 | Diabetes Mellitus ... more >> Pre-Diabetes Hyperglycemia Metabolic Syndrome Collapse << | Phase 3 | Completed | - | India ... more >> Dia Care- Diabetes Care and Hormone Clinic Ahmedabad, Gujarat, India, 380015 St. Johns College and Hospital Bangalore, Karnataka, India, 560034 Totall Diabetes and Hormone Institution Indore, Madhya Pradesh, India, 452010 Bhatia Hospital Mumbai, Maharashtra, India, 411007 Inamdar Multispeciality Hospital Pune, Maharashtra, India, 411040 Diabetes Care Centre Jaipur, Rajasthan, India, 302018 Collapse << |
| NCT02777229 | HIV-1 Infection | Phase 3 | Active, not recruiting | July 2018 | Cameroon ... more >> Cité verte Hospital Yaoundé, Cameroon Hopital Central Yaoundé, Cameroon Military Hospital Yaoundé, Cameroon Collapse << |
| NCT03003338 | Hepatitis C, Chronic | Phase 4 | Unknown | December 2018 | Germany ... more >> Hannover Medical School Recruiting Hannover, Germany, 30625 Contact: Heiner Wedemeyer, Prof. Dr. Contact: Markus Cornberg, Prof. Dr. Principal Investigator: Heiner Wedemeyer, Prof. Dr. Collapse << |
| NCT00985088 | Influenza | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Huntsville, Alabama, United States, 35802 United States, Florida GSK Investigational Site Jacksonville, Florida, United States, 32216 GSK Investigational Site Miami, Florida, United States, 33143 United States, Idaho GSK Investigational Site Meridian, Idaho, United States, 83642 United States, Kansas GSK Investigational Site Lenexa, Kansas, United States, 66219 United States, Montana GSK Investigational Site Missoula, Montana, United States, 59801 United States, Nevada GSK Investigational Site Las Vegas, Nevada, United States, 89104 United States, New Jersey GSK Investigational Site Edison, New Jersey, United States, 08817 United States, New York GSK Investigational Site Rochester, New York, United States, 14609 United States, Ohio GSK Investigational Site Cleveland, Ohio, United States, 44122 Canada, British Columbia GSK Investigational Site Surrey, British Columbia, Canada, V3R 8P8 Canada, Nova Scotia GSK Investigational Site Truro, Nova Scotia, Canada, B2N 1L2 Canada, Ontario GSK Investigational Site Toronto, Ontario, Canada, M9W 4L6 Canada, Quebec GSK Investigational Site Pointe-Claire, Quebec, Canada, H9R 4S3 Collapse << |
| NCT00985088 | - | - | Completed | - | - |
| NCT03734393 | Hiv | Phase 2 | Not yet recruiting | June 30, 2022 | United States, Alabama ... more >> University of Alabama at Birmingham Not yet recruiting Birmingham, Alabama, United States, 35294 Contact: Jared White, MD jaredwhite@uabmc.edu Principal Investigator: Jared White, MD United States, California University of California, San Diego Not yet recruiting San Diego, California, United States, 92103 Contact: Saima Aslam, MD saslam@ucsd.edu Principal Investigator: Saima Aslam, MD University of California, San Francisco Not yet recruiting San Francisco, California, United States, 94193 Contact: Jennifer Price, MD Principal Investigator: Jennifer Price, MD United States, District of Columbia MedStar Georgetown Transplant Institute Not yet recruiting Washington, District of Columbia, United States, 20007 Contact: Coleman Smith, MD coleman.i.smith@gunet.georgetown.edu Principal Investigator: Coleman Smith, MD United States, Florida University of Miami Not yet recruiting Miami, Florida, United States, 33136 Contact: Michele Morris, MD 305-243-4598 mmorris2@med.miami.edu Principal Investigator: Michele Morris, MD United States, Georgia Emory University Not yet recruiting Atlanta, Georgia, United States, 30322 Contact: Valentina Stosor, MD 312-695-5085 v-stosor@northwestern.edu Principal Investigator: Valentina Stosor, MD United States, Illinois Northwestern University Not yet recruiting Chicago, Illinois, United States, 60611 Contact: Valentina Stosor, MD v-stosor@morthwestern.edu Principal Investigator: Valentina Stosor, MD Rush University Medical Center Not yet recruiting Chicago, Illinois, United States, 60612 Contact: Carlos A. Santos, MD, MPHS 312-563-6372 carlos_a_santos@rush.edu Principal Investigator: Carlos A. Santos, MD, MPHS United States, Louisiana Ochsner Medical Center Not yet recruiting New Orleans, Louisiana, United States, 70121 Contact: Jonathan Hand, MD Principal Investigator: Jonathan Hand, MD United States, Maryland University of Maryland, Institute of Human Virology Not yet recruiting Baltimore, Maryland, United States, 21201 Contact: Jennifer Husson, MD 410-706-8614 jhusson@ihv.umaryland.edu Principal Investigator: Jennifer Husson, MD Johns Hopkins University Not yet recruiting Baltimore, Maryland, United States, 21205 Contact: Christine Durand, MD 410-955-5684 cdurand2@jhmi.edu Principal Investigator: Christine Durand, MD United States, Massachusetts Massachusetts General Hospital Not yet recruiting Boston, Massachusetts, United States, 02114 Contact: Nahel Elias, MD 617-726-0174 elias.nahel@mgh.harvard.edu Principal Investigator: Nahel Elias, MD United States, Minnesota University of Minnesota Not yet recruiting Minneapolis, Minnesota, United States, 55455 Contact: Timothy Pruett, MD Principal Investigator: Timothy Pruett, MD United States, New York Icahn School of Medicine at Mount Sinai Not yet recruiting New York, New York, United States, 10029 Contact: Sander Florman, MD 212-659-8313 sander.florman@mountsinai.org Principal Investigator: Sander Florman, MD Columbia University Medical Center Not yet recruiting New York, New York, United States, 10032 Contact: Marcus Pereira, MD 212-305-3839 mp2323@cumc.columbia.edu Principal Investigator: Marcus Pereira, MD New York University School of Medicine Not yet recruiting New York, New York, United States, 11016 Contact: Sapna Mehta, MD sapna.mehta@nyumc.org Contact: Elaina Weldon, MSN,NP elaina.weldon@nyumc.org Principal Investigator: Sapna Mehta, MD United States, Ohio University of Cincinnati Not yet recruiting Cincinnati, Ohio, United States, 45267 Contact: Senu Apewokin, MD Principal Investigator: Senu Apewokin, MD United States, Pennsylvania University of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Emily Blumberg, MD 215-662-7066 blumbere@pennmedicine.upenn.edu Principal Investigator: Emily Blumberg, MD UPMC - University of Pittsburgh Medical Center Not yet recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Ghady Haidar, MD 412-648-6212 haidarg@upmc.edu Principal Investigator: Ghady Haidar, MD Collapse << |
| NCT01913938 | - | - | Recruiting | December 2018 | Germany ... more >> Department of Anaesthesiology, University Hospital Ulm Recruiting Ulm, Germany, 89075 Contact: Manfred E Weiss, MD 49 (0)731-500-60226 manfred.weiss@uniklinik-ulm.de Contact: Elisabeth M Schneider, PhD 49 (0)731-500-60319 marion.schneider@uni-ulm.de Department of Internal Medicine III, Comprehensive Infectious Disease Center, University Hospital Ulm, Germany Recruiting Ulm, Germany, 89075 Contact: Martin Bommer, MD +49 (731) 500-45530 Martin.Bommer@uniklinik-ulm.de Contact: Elisabeth M Schneider, PhD +49 (731) 500-60080 marion.schneider@uni-ulm.de Collapse << |
| NCT01057810 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT01318096 | HBV Coinfection ... more >> HIV Infections Collapse << | Not Applicable | Unknown | September 2013 | China, Yunnan Provice ... more >> Yunnan Provincial Hospital of Infectious Diseases/Yunnan AIDS Care Center Not yet recruiting Kunming, Yunnan Provice, China, 650301 Contact: Cheng Xi Wang, M.D. 86 871 8728060 wxch62597@foxmail.com Principal Investigator: Cheng Xi Wang, M.D. Collapse << |
| NCT03345901 | Diabetic Retinopathy ... more >> Diabetic Macular Edema Collapse << | Phase 3 | Recruiting | January 2023 | - |
| NCT03092765 | Primary Biliary Cholangitis | Phase 2 | Active, not recruiting | October 2018 | Japan ... more >> EA Pharma trial site Nagoya, Aichi, Japan EA Pharma trial site Matsudo, Chiba, Japan EA Pharma trial site Touon, Ehime, Japan EA Pharma trial site Yoshida, Fukui, Japan EA Pharma trial site Kurume, Fukuoka, Japan EA Pharma trial site Maebashi, Gunma, Japan EA Pharma trial site #1 Sapporo, Hokkaido, Japan EA Pharma trial site #2 Sapporo, Hokkaido, Japan EA Pharma trial site Kobe, Hyogo, Japan EA Pharma trial site Nishinomiya, Hyogo, Japan EA Pharma trial site Inashiki, Ibaraki, Japan EA Pharma trial site Morioka, Iwate, Japan EA Pharma trial site Kita, Kagawa, Japan EA Pharma trial site Takamatsu, Kagawa, Japan EA Pharma trial site #1 Yokohama, Kanagawa, Japan EA Pharma trial site #2 Yokohama, Kanagawa, Japan EA Pharma trial site Sendai, Miyagi, Japan EA Pharma trial site Matsumoto, Nagano, Japan EA Pharma trial site Oomura, Nagasaki, Japan EA Pharma trial site Kashihara, Nara, Japan EA Pharma trial site Nakagami, Okinawa, Japan EA Pharma trial site Hirakata, Osaka, Japan EA Pharma trial site #1 Suita, Osaka, Japan EA Pharma trial site #2 Suita, Osaka, Japan EA Pharma trial site Ageo, Saitama, Japan EA Pharma trial site Iruma, Saitama, Japan EA Pharma trial site Shimotsuga, Tochigi, Japan EA Pharma trial site Bunkyo, Tokyo, Japan EA Pharma trial site Itabashi, Tokyo, Japan EA Pharma trial site #1 Minato, Tokyo, Japan EA Pharma trial site #2 Minato, Tokyo, Japan EA Pharma trial site Musashino, Tokyo, Japan EA Pharma trial site Shinjuku, Tokyo, Japan EA Pharma trial site #1 Fukuoka, Japan EA Pharma trial site #2 Fukuoka, Japan EA Pharma trial site Fukushima, Japan EA Pharma trial site #1 Hiroshima, Japan EA Pharma trial site #2 Hiroshima, Japan EA Pharma trial site Kagoshima, Japan EA Pharma trial site Kumamoto, Japan EA Pharma trial site Kyoto, Japan EA Pharma trial site Niigata, Japan EA Pharma trial site Okayama, Japan EA Pharma trial site Osaka, Japan EA Pharma trial site Saga, Japan EA Pharma trial site Yamagata, Japan Collapse << |
| NCT01057810 | - | - | Completed | - | - |
| NCT02494141 | Polycystic Kidney, Autosomal D... more >>ominant Collapse << | Phase 4 | Recruiting | July 2020 | United States, Colorado ... more >> University of Colorado Anschutz Medical Campus Recruiting Aurora, Colorado, United States, 80045 Contact: Kristen L Nowak, Ph.D. 303-724-4842 Kristen.Nowak@ucdenver.edu Contact: Laurel Thur 303-724-9508 Laurel.Thur@ucdenver.edu Collapse << |
| NCT03237689 | Hiv | Early Phase 1 | Recruiting | August 31, 2022 | United States, Maryland ... more >> Johns Hopkins University School of Medicine Recruiting Baltimore, Maryland, United States, 21224 Contact: Leah Rubin lrubin1@jhu.edu Collapse << |
| NCT02606669 | Obesity Fasti... more >>ng Collapse << | Not Applicable | Completed | - | Singapore ... more >> Khoo Teck Puat Hospital Singapore, Singapore, 768828 Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
