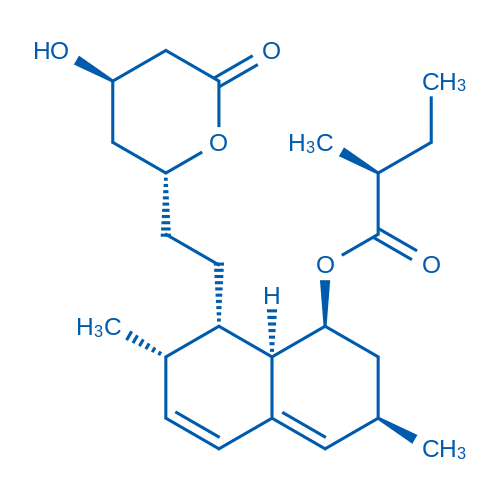
CAS No.: 75330-75-5
洛伐他汀 Catalog No. CSN11335
Synonyms: 洛伐他汀;Mevinolin
Lovastatin is a HMG-CoA reductase used to treat high blood cholesterol and reduce the risk of cardiovascular disease, a naturally occuring fungal metabolite.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01047683 Hypertriglyceridemia Phase 3 Completed - - NCT00256750 Kidney Transplantation ... more >> Chronic Kidney Failure Collapse << Phase 3 Completed - - NCT01047501 Hypertriglyceridemia Phase 3 Completed - - - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 75330-75-5 | 储存条件 |
|
|||||||||||
| 分子式 | C24H36O5 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 404.54 | 别名 | 洛伐他汀;Mevinolin | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| HEP G2 cells | - | Function assay | - | Inhibition of cellular HMG-CoA reductase in cultures of hepatic cells (HEP G2, a human hepatoma cell line), IC50=5e-05 μM | 2153213 |
| HEP G2 cells | - | Function assay | - | Tested for inhibition of cholesterol biosynthesis in HEP G2 cells, IC50=0.029 μM | 7932551 |
| HES 9 cell line | - | Function assay | - | Concentration required to inhibit HMG-CoA reductase by 50% was determined in HES 9 cell line, IC50=0.013 μM | 1527791 |
| human A549 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, IC50=11.4 μM | 23570542 |
| human AU565 cells | - | Growth inhibition assay | 48 h | Growth inhibition of ER-negative, HER2-positive human AU565 cells after 48 hrs by WST-1 assay | 24556504 |
| human HepG2 cells | - | Function assay | 6 h | Lipid lowering activity in human HepG2 cells assessed as decrease in oleic acid elicited lipid accumulation after 6 hrs by oil-red O staining method, IC50=8.3 μM | 26169125 |
| human HS68 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human HS68 cells after 72 hrs by MTT assay, IC50=23.2 μM | 23570542 |
| human HT29 cells | - | Growth inhibition assay | 96 h | Growth inhibition of human HT29 cells after 96 hrs by MTS assay, IC50=46.8 μM | 17472962 |
| human K562 cells | - | Function assay | 48 h | Inhibition of GGTase1 in human K562 cells assessed as reduction of Rap1a protein geranylgeranylation after 48 hrs by Western blotting | 20832326 |
| human LS180 cells | 20 μM | Function assay | - | Inhibition of survivin expression in parent human LS180 cells at 20 uM by immunoblot analysis | 17472962 |
| human MCF7 cells | - | Growth inhibition assay | 48 h | Total growth inhibition of ER-positive, HER2-negative human MCF7 cells after 48 hrs by WST-1 assay | 24556504 |
| human MDA-MB-231 cells | - | Function assay | 24 h | Induction of p21 expression in human PR, ER, HER2-negative human MDA-MB-231 cells at 1 to 10 uM after 24 hrs by western blot analysis | 24556504 |
| human MDA-MB-361 cells | - | Growth inhibition assay | 48 h | Growth inhibition of ER-positive, HER2-positive human MDA-MB-361 cells after 48 hrs by WST-1 assay | 24556504 |
| human MDA-MB-468 cells | - | Function assay | 48 h | Total growth inhibition of PR, ER, HER2-negative human MDA-MB-468 cells after 48 hrs by WST-1 assay | 24556504 |
| human RPMI8226 cells | 20 μM | Apoptosis assay | 48 h | Induction of apoptosis in human RPMI8226 cells assessed as increase in caspase-3 cleavage at 20 uM incubated for 48 hrs by immunoblot method | 25935643 |
| human SW480 cells | - | Growth inhibition assay | 96 h | Growth inhibition of human SW480 cells after 96 hrs by MTS assay, IC50=7.1 μM | 17472962 |
| human SW480 cells | 20 μM | Function assay | - | Inhibition of FBS-stimulated increase in Akt phosphorylation in human SW480 cells at 20 uM by immunoblot analysis | 17472962 |
| mouse MEF cells | - | Cytotoxic assay | 72 h | Cytotoxicity against mouse MEF cells after 72 hrs by MTT assay, IC50=35 μM | 23570542 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01047683 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT00256750 | Kidney Transplantation ... more >> Chronic Kidney Failure Collapse << | Phase 3 | Completed | - | - |
| NCT01047501 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT00256750 | - | - | Completed | - | - |
| NCT01492361 | Cardiovascular Diseases | Phase 3 | Completed | - | - |
| NCT02523001 | - | - | Completed | - | Italy ... more >> Department of Biomedical Sciences Human Oncology - Clinica Medica "A. Murri" Bari, Italy, 70124 Collapse << |
| NCT00132717 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT03739242 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> Policlinico S.Orsola - Malpighi Medicina Interna Borghi Bologna, Italy, 40138 Collapse << |
| NCT01890967 | - | - | Completed | - | - |
| NCT03510715 | Hypercholesterolemia | Phase 3 | Recruiting | June 2020 | United States, California ... more >> Investigational Site Number 8400004 Recruiting Los Angeles, California, United States, 90027 Austria Investigational Site Number 0400001 Recruiting Wien, Austria, 1090 Brazil Investigational Site Number 0760001 Recruiting Sao Paulo, Brazil, 05403-000 Canada Investigational Site Number 1240002 Recruiting Montreal, Canada, H3T 1C5 Investigational Site Number 1240001 Recruiting Quebec, Canada, G1V 4W2 Denmark Investigational Site Number 2080001 Recruiting Viborg, Denmark, 8800 France Investigational Site Number 2500001 Recruiting Bron Cedex, France, 69677 Italy Investigational Site Number 3800001 Recruiting Palermo, Italy, 90127 Mexico Investigational Site Number 4840006 Recruiting Oaxaca, Mexico, 68000 Netherlands Investigational Site Number 5280001 Recruiting Amsterdam, Netherlands, 1105 AZ Norway Investigational Site Number 5780001 Recruiting Oslo, Norway, 0373 Russian Federation Investigational Site Number 6430002 Recruiting Kemerovo, Russian Federation, 650002 Slovenia Investigational Site Number 7050001 Recruiting Ljubljana, Slovenia, 1000 Spain Investigational Site Number 7240001 Recruiting A Coruna, Spain, 15001 Taiwan Investigational Site Number 1580001 Recruiting Taipei, Taiwan, 112 Turkey Investigational Site Number 7920002 Recruiting Ankara, Turkey, 06500 Investigational Site Number 7920001 Recruiting Izmir, Turkey, 35040 Collapse << |
| NCT03510884 | Hypercholesterolaemia | Phase 3 | Recruiting | March 2022 | - |
| NCT00532311 | Hypercholesterolemia | Phase 3 | Terminated(Overall profile of ... more >>the compound does not offer significant clinical advantage to patients over currently available lipid lowering agents) Collapse << | - | - |
| NCT03065491 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> S. Orsola-Malpighi University Hospital Bologna, BO, Italy, 40138 Collapse << |
| NCT00986583 | Pain | Not Applicable | Completed | - | United States, Ohio ... more >> Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01890967 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| NCT02969070 | Hypercholesterolemia ... more >> Endothelial Dysfunction Collapse << | Phase 3 | Unknown | March 2017 | Italy ... more >> University of Naples "Federico II" Recruiting Naples, Italy, 80131 Contact: Giuseppe Giugliano, MD, PhD +390817462240 giuseppe.giugliano@unina.it Sub-Investigator: Raffaele Izzo, MD Sub-Investigator: Maurizio Galderisi, MD Sub-Investigator: Giuseppe Giugliano, MD, PhD Collapse << |
| NCT00986583 | - | - | Completed | - | - |
| NCT03425630 | Hypercholesterolemia | Not Applicable | Completed | - | - |
| NCT02142569 | Cholesterol Lowering | Phase 2 Phase 3 | Suspended(We were unable to pr... more >>ocure the supplement for this study.) Collapse << | July 2018 | United States, California ... more >> UCLA Center for Human Nutrition, 900 Veteran Avenue, WH 14-187 Los Angeles, California, United States, 90095 Collapse << |
| NCT00726362 | - | - | Completed | - | Taiwan ... more >> Research Site Changhua, Taiwan Research Site Hualien, Taiwan Research Site Kaohsiung, Taiwan Research Site Pingtung, Taiwan Research Site Taichung, Taiwan Research Site Tainan, Taiwan Research Site Taipei, Taiwan Research Site Yun-Lin, Taiwan Collapse << |
| NCT01807078 | Coronary Artery Disease | Phase 4 | Unknown | December 2017 | Italy ... more >> Sapienza University Not yet recruiting Rome, Lazio, Italy, 00161 Contact: Francesco Pelliccia, MD +39064997 ext 123 f.pelliccia@mclink.it Contact: Giuseppe Marazzi, MD +39064997 ext 123 md4151@mclink.it Principal Investigator: Francesco Pelliccia, MD Collapse << |
| NCT00452244 | Lung Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> National Cancer Center, Korea Goyang-si, Gyeonggi-do, Korea, Republic of Collapse << |
| NCT00145574 | Hypercholesterolemia | Phase 4 | Completed | - | - |
| NCT00145574 | - | - | Completed | - | - |
| NCT00046358 | Alzheimer Disease | Phase 4 | Completed | - | United States, Maryland ... more >> National Institute of Mental Health (NIMH) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02518503 | - | - | Completed | - | - |
| NCT02603276 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> S. Orsola-Malpighi University Hospital Bologna, Italy, 40038 Collapse << |
| NCT02518516 | - | - | Completed | - | Canada, British Columbia ... more >> Dept. of Anesthesiology, Pharmacology & Therapeutics (APT), University of British Columbia Vancouver, British Columbia, Canada, V6T 1Z3 Collapse << |
| NCT00125125 | Dyslipidemia | Phase 4 | Completed | - | United States, New Jersey ... more >> Novartis East Hanover, New Jersey, United States, 07936 Germany Investigative Centers, Germany Collapse << |
| NCT01087632 | Metabolic Syndrome | Not Applicable | Completed | - | Italy ... more >> Federico II University - Department of Internal Medicine Naples, Italy Collapse << |
| NCT00730132 | - | - | Completed | - | - |
| NCT03216811 | Hypercholesterolemia | Phase 2 | Completed | - | Italy ... more >> University Hospital of Ferrara Cona, Ferrara, Italy, 44124 Collapse << |
| NCT00000463 | Cardiovascular Diseases ... more >> Coronary Disease Heart Diseases Myocardial Ischemia Collapse << | Phase 3 | Completed | - | - |
| NCT02492464 | Atherosclerosis | Not Applicable | Completed | - | Italy ... more >> S. Orsola-Malpighi University Hospital Bologna, BO, Italy, 40138 Collapse << |
| NCT00730132 | - | - | Completed | - | - |
| NCT01269762 | Healthy Volunteer | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan Collapse << |
| NCT03470376 | Hypercholesterolemia ... more >> Inflammation Atherosclerosis Collapse << | Phase 4 | Completed | - | - |
| NCT02658838 | LOVASTATIN/TICAGRELOR [VA Drug... more >> Interaction] Collapse << | Phase 4 | Unknown | September 2017 | China, Beijing ... more >> Beijing Anzhen Hosipital Beijing, Beijing, China, 100000 Collapse << |
| NCT01569854 | - | - | Withdrawn(Insufficient recruit... more >>ment) Collapse << | March 2022 | United States, Ohio ... more >> Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00718796 | Cardiovascular Disease | Phase 3 | Completed | - | Canada, Ontario ... more >> Canadian College of Naturopathic Medicine Toronto, Ontario, Canada, M2K 1E2 Collapse << |
| NCT01385020 | Healthy Subjects | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan Collapse << |
| NCT00000477 | Atherosclerosis ... more >> Cardiovascular Diseases Coronary Disease Heart Diseases Hypercholesterolemia Myocardial Ischemia Collapse << | Phase 2 | Completed | - | - |
| NCT01569867 | - | - | Recruiting | March 2022 | United States, Ohio ... more >> The Ohio State University Medical Center Recruiting Columbus, Ohio, United States, 43210 Principal Investigator: Joseph P Kitzmiller, MD PhD Sub-Investigator: Wolfgang Sadee, Dr rer nat Sub-Investigator: Mitch Phelps, PhD Sub-Investigator: Rebecca Jackson, MD Collapse << |
| NCT02689934 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> Centro Dislipidemie Dip. Cardiotoracovasc ASST Grande Ospedale Metropolitano Niguarda Cà Granda Milano, Mi, Italy, 20162 Collapse << |
| NCT00092846 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT02964884 | Neurofibromatosis Type 1 ... more >> Learning Disability Reading Disability NF1 Collapse << | Phase 2 | Recruiting | June 2022 | United States, Tennessee ... more >> Vanderbilt University Recruiting Nashville, Tennessee, United States Contact: Laura E. Cutting 615-875-1054 Collapse << |
| NCT03340285 | Cardiovascular Risk Factor | Not Applicable | Not yet recruiting | April 2018 | - |
| NCT00000512 | Cardiovascular Diseases ... more >> Coronary Arteriosclerosis Coronary Disease Heart Diseases Myocardial Ischemia Collapse << | Phase 3 | Completed | - | - |
| NCT00000469 | Cardiovascular Diseases ... more >> Carotid Stenosis Cerebral Arteriosclerosis Cerebrovascular Disorders Heart Diseases Vascular Diseases Collapse << | Phase 2 | Completed | - | - |
| NCT02862470 | - | - | Active, not recruiting | July 7, 2020 | - |
| NCT03504501 | Impaired Synaptic Plasticity ... more >> Impaired Cognition Collapse << | Phase 2 | Recruiting | June 2019 | Germany ... more >> Technical University Munich Recruiting Munich, Germany, 81377 Contact: Volker Mall, Prof. +49 (0)89 71009-233 volker.mall@kbo.de Contact: Nikolai Jung, Dr. +49 (0)89 71009-236 nikolai.jung@tum.de Collapse << |
| NCT00741013 | Lung Inflammation | Early Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00004346 | Cerebrotendinous Xanthomatosis | Phase 2 | Unknown | - | United States, Oregon ... more >> Oregon Health and Science University Recruiting Portland, Oregon, United States, 97239-3098 Contact: Sonja Connor 503-494-7775 connors@ohsu.edu Collapse << |
| NCT00116870 | Atherosclerosis ... more >> Coronary Artery Disease Collapse << | Phase 2 Phase 3 | Completed | - | United States, California ... more >> Atherosclerosis Research Unit, Division of Cardiovascular Medicine, Department of Medicine Los Angeles, California, United States, 90033 Collapse << |
| NCT01250535 | Healthy Volunteers ... more >> Drug Drug Interactions Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> University of North Carolina, Clinical and Translational Research Center (CTRC) Chapel Hill, North Carolina, United States, 27599 Collapse << |
| NCT03763175 | Irritable Bowel Syndrome With ... more >>Constipation Collapse << | Phase 2 | Not yet recruiting | January 1, 2020 | United States, California ... more >> Cedars-Sinai Medical Center Not yet recruiting Los Angeles, California, United States, 90048 Contact: Tahli Singer-Englar 310-423-3792 tahli.singerenglar@cshs.org Collapse << |
| NCT01361217 | - | - | Completed | - | - |
| NCT00062556 | Intermittent Claudication ... more >> Peripheral Vascular Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00345657 | Hyperlipidemia ... more >> Mixed Hyperlipidemia Dyslipidemia Collapse << | Phase 4 | Completed | - | United States, Oklahoma ... more >> Family Medical Care of Tulsa Tulsa, Oklahoma, United States, 74136 Collapse << |
| NCT00741013 | - | - | Completed | - | - |
| NCT01976936 | Stroke Rhabdo... more >>myolysis Jaundice Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California, Los Angeles Stroke Network Los Angeles, California, United States, 90024 United States, Florida Jackson Memorial Hospital Miami, Florida, United States, 33136 University of Miami Miller School of Medicine Miami, Florida, United States, 33136 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Massachusetts Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 United States, New York Mount Sinai School of Medicine New York, New York, United States, 10029 United States, Pennsylvania The Hospital of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01361217 | Drug-Drug Interaction ... more >> Healthy Volunteers Collapse << | Not Applicable | Completed | - | - |
| NCT00721305 | HIV Seropositivity | Phase 2 | Completed | - | Colombia ... more >> Group of Immunovirology, Research Universitary Center, University of Antioquia Medellin, Antioquia, Colombia Collapse << |
| NCT00071266 | Intermittent Claudication ... more >> Peripheral Vascular Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00902668 | Breast Cancer ... more >> Radiation Toxicity Collapse << | Phase 2 | Terminated(Slow accrual) | - | - |
| NCT00902668 | - | - | Terminated(Slow accrual) | - | - |
| NCT00689806 | Severe Persistent Asthma | Phase 1 Phase 2 | Withdrawn(Study never opened) | - | - |
| NCT02998151 | Fragile X Syndrome | Phase 2 | Enrolling by invitation | July 2021 | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 Collapse << |
| NCT00462280 | - | - | Completed | - | - |
| NCT00853580 | Neurofibromatosis Type 1 | Phase 2 | Completed | - | United States, Alabama ... more >> The University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, California Children's Hospital Los Angeles Los Angeles, California, United States, 90027 United States, District of Columbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Illinois University of Chicago Chicago, Illinois, United States, 60637 United States, Maryland NIH Bethesda, Maryland, United States, 20892 United States, Massachusetts Children' Hospital Boston Boston, Massachusetts, United States, 02115 United States, Missouri Washington University - St. Louis Saint Louis, Missouri, United States, 63110 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19096 United States, Texas Childrens Medical Center - Univ. of Texas SW Medical Center Dallas, Texas, United States, 75235 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Australia, New South Wales The Children's Hospital at Westmead Westmead, New South Wales, Australia, 2145 Collapse << |
| NCT01110642 | Syndromic Ichthyoses ... more >> CHILD Syndrome Smith Lemli Opitz Syndrome Conradi Syndrome Collapse << | Phase 2 | Withdrawn(Study was withdrawn ... more >>due to lack of eligible population for study) Collapse << | - | - |
| NCT00243880 | Stroke | Phase 1 | Completed | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT03178526 | Alveolar Bone Loss, Chronic Pe... more >>riodontitis, Lovestatin Gel, Regeneration Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT00352599 | Neurofibromatosis 1 | Phase 1 | Completed | - | United States, California ... more >> Semel Institute for Neuroscience and Human Behavior Los Angeles, California, United States, 90095 Collapse << |
| NCT00462280 | Precancerous Condition ... more >> Stage 0 Melanoma Stage I Melanoma Stage II Melanoma Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California Medical Center At Irvine-Orange Campus Orange, California, United States, 92868 United States, Florida H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Utah Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT02563860 | Rett Syndrome | Phase 2 | Completed | - | United States, New York ... more >> Montefiore Medical center Bronx, New York, United States, 10467 Collapse << |
| NCT02389868 | Blood Platelets Proteome | Phase 2 | Completed | - | Canada, Quebec ... more >> Centre de Recherche du CHUS Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT00302952 | Rheumatoid Arthritis | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham, Alabama, United States, 35294 United States, California University of California, San Francisco San Francisco, California, United States, 94143 United States, Colorado University of Colorado Aurora, Colorado, United States, 80095 United States, Illinois University of Chicago Medical Center Chicago, Illinois, United States, 60637 United States, Michigan Justus J. Fiechtner, MD, PLLC Lansing, Michigan, United States, 48910 United States, New York Feinstein Institute for Medical Research NS-LIJ Health System Manhasset, New York, United States, 11030 University of Rochester Rochester, New York, United States, 14642 United States, North Carolina Carolina Bone and Joint Charlotte, North Carolina, United States, 29425 Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Oklahoma Oklahoma Medical Research Foundation Oklahoma City, Oklahoma, United States, 73104 United States, Pennsylvania Altoona Center for Clinical Research Duncansville, Pennsylvania, United States, 16635 University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15261 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29425 United States, Texas Baylor Research Institute Dallas, Texas, United States, 75231 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT00853580 | - | - | Completed | - | - |
| NCT00302952 | - | - | Completed | - | - |
| NCT00584012 | - | - | Terminated(Funding issues) | - | - |
| NCT00685685 | Healthy | Phase 1 | Completed | - | Canada ... more >> Algorithme Pharma Montreal, Canada, H7V 4B4 Collapse << |
| NCT00700921 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | United States, Colorado ... more >> National Jewish Medical and Research Center Denver, Colorado, United States, 80206 Collapse << |
| NCT00684723 | Healthy | Phase 1 | Completed | - | Canada, Quebec ... more >> Algorithme Pharma Montreal, Quebec, Canada Collapse << |
| NCT00584012 | Any Cancer Br... more >>east Cancer Collapse << | Phase 1 | Terminated(Funding issues) | - | United States, Iowa ... more >> Holden Comprehensive Cancer Center Iowa City, Iowa, United States, 52242 Collapse << |
| NCT00685685 | - | - | Completed | - | - |
| NCT00583102 | Acute Myeloid Leukemia | Phase 1 Phase 2 | Terminated(Slow accrual, PI le... more >>ft institution) Collapse << | - | United States, Iowa ... more >> Holden Comprehensive Cancer Center Iowa City, Iowa, United States, 52242 Collapse << |
| NCT00963664 | Melanoma Mali... more >>gnant Melanoma Collapse << | Phase 2 | Withdrawn(Modifications will b... more >>e necessary before full IRB approval will be secured.) Collapse << | December 2016 | United States, Tennessee ... more >> NeoPlas Innovation Nashville, Tennessee, United States, 37027 Collapse << |
| NCT00684723 | - | - | Completed | - | - |
| NCT00285857 | Breast Cancer | Phase 2 | Terminated(Slow accrual) | - | United States, California ... more >> Stanford University Cancer Center Stanford, California, United States, 94305 Collapse << |
| NCT02603770 | Lipid Metabolism Disorder | Phase 1 | Completed | - | United States, California ... more >> NRC Research Institute Orange, California, United States, 92868 Collapse << |
| NCT00583102 | - | - | Terminated(Slow accrual, PI le... more >>ft institution) Collapse << | - | - |
| NCT00580970 | Prostate Cancer | Phase 2 | Completed | - | United States, Virginia ... more >> Hunter Holmes McGuire Veterans Administration Medical Center Richmond, Virginia, United States, 23249 Massey Cancer Center/Virginia Commonwealth University Richmond, Virginia, United States, 23298 Southside Regional Medical Center Richmond, Virginia, United States, 23805 Collapse << |
| NCT02642653 | Fragile X Syndrome ... more >> Genetic Diseases Collapse << | Phase 4 | Completed | - | United States, California ... more >> UC Davis MIND Institute Sacramento, California, United States, 95817 Collapse << |
| NCT00580970 | - | - | Completed | - | - |
| NCT00585052 | Ovarian Cancer | Phase 2 | Terminated(Slow accrual, PI le... more >>ft the institution) Collapse << | - | United States, Iowa ... more >> Holden Comprehensive Cancer Center Iowa City, Iowa, United States, 52327 Collapse << |
| NCT00585052 | - | - | Terminated(Slow accrual, PI le... more >>ft the institution) Collapse << | - | - |
| NCT01346670 | Healthy Volunteer | Phase 4 | Completed | - | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT01527669 | Healthy Subjects | Phase 4 | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan Collapse << |
| NCT01478828 | Prostate Cancer | Not Applicable | Suspended(The study was stoppe... more >>d due to an unanticipated serious adverse event.) Collapse << | April 2016 | United States, Maryland ... more >> The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States, 21231 Collapse << |
| NCT02680379 | Fragile X Syndrome | Phase 2 | Completed | - | Canada, Quebec ... more >> Centre de Recherche du CHUS Sherbrooke, Quebec, Canada, J1H 5N4 Collapse << |
| NCT03242499 | Parkinson Disease | Phase 2 | Recruiting | December 31, 2019 | Taiwan ... more >> National Taiwan University Hospital Recruiting Taipei, Taiwan, 100 Contact: Chin-Hsien Lin, MD,PhD 886-2-3123456 ext 65335 chlin@ntu.edu.tw Principal Investigator: Chin-Hsien Lin, MD,PhD Collapse << |
| NCT01478828 | - | - | Suspended(The study was stoppe... more >>d due to an unanticipated serious adverse event.) Collapse << | - | - |
| NCT00285857 | - | - | Terminated(Slow accrual) | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
