| NCT03662893 | Overactive Bladder | Not Applicable | Completed | - | Turkey
... more >> Ankara Training and Research Hospital
Ankara, Turkey, 06340 Collapse << |
| NCT00547378 | Urinary Incontinence, Urgency-... more >>frequency
Overactive Bladder Collapse << | Phase 4 | Completed | - | - |
| NCT02386072 | - | - | Completed | - | - |
| NCT00892450 | Parkinson's Disease
... more >> Overactive Bladder Collapse << | Not Applicable | Completed | - | United States, Pennsylvania
... more >>
VA Medical Center, Philadelphia
Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00800462 | Spinal Cord Injury
... more >> Neurogenic Detrusor Overactivity Collapse << | Phase 4 | Completed | - | Canada, Manitoba
... more >>
University of Manitoba, Health Sciences Centre
Winnipeg, Manitoba, Canada, R3A1R9
Canada, Ontario
Toronto Rehabilitation Institute, Lyndhurst Centre
Toronto, Ontario, Canada, M4G 3V9 Collapse << |
| NCT03572231 | - | - | Recruiting | December 2019 | Korea, Republic of
... more >>
Site KR410008
Recruiting
Daejeon, Korea, Republic of
Site KR410009
Recruiting
Incheon, Korea, Republic of
Site KR410005
Recruiting
Kangam, Korea, Republic of
Site KR410001
Recruiting
Seoul, Korea, Republic of
Site KR410002
Recruiting
Seoul, Korea, Republic of
Site KR410003
Recruiting
Seoul, Korea, Republic of
Site KR410006
Recruiting
Seoul, Korea, Republic of
Taiwan
Site TW158001
Recruiting
Hualien City, Taiwan Collapse << |
| NCT03543566 | - | - | Recruiting | April 2019 | United States, Alabama
... more >>
UAB Kirklin Clinic
Recruiting
Birmingham, Alabama, United States, 35233
Contact: Lindsay Kissane, MD 205-996-0916 Collapse << |
| NCT01018225 | Nocturia | Phase 4 | Withdrawn(Study was cancelled ... more >>due to lack of enrollment) Collapse << | July 2010 | - |
| NCT00413426 | Healthy | Phase 1 | Completed | - | India
... more >> Novartis Investigative Site
Mumbai, India Collapse << |
| NCT00547378 | - | - | Completed | - | - |
| NCT03602508 | - | - | Active, not recruiting | February 2019 | Australia
... more >> Site AU10000
Sydney, Australia
Korea, Republic of
Site KR82001
Seoul, Korea, Republic of Collapse << |
| NCT00786448 | - | - | Completed | - | Germany
... more >>
Many Locations, Germany Collapse << |
| NCT00921245 | - | - | Completed | - | South Africa
... more >>
Many Locations, South Africa Collapse << |
| NCT00845338 | Multiple Sclerosis
... more >> Overactive Detrusor Collapse << | Phase 2 | Terminated(The enrollment rate... more >> at most centers was unlikely to result in the recruitment of the planned sample size of 40 evaluable subjects.) Collapse << | - | Germany
... more >>
Emmendingen, Baden-Württemberg, Germany, 79312
Heidelberg, Baden-Württemberg, Germany, 69112
Lahr, Baden-Württemberg, Germany, 77933
Tübingen, Baden-Württemberg, Germany, 72076
Villingen-Schwenningen, Baden-Württemberg, Germany, 78054
Planegg, Bayern, Germany, 82152
Oberursel, Hessen, Germany, 61440
Herne, Nordrhein-Westfalen, Germany, 44627
Mönchengladbach, Nordrhein-Westfalen, Germany, 41063
Mülheim, Nordrhein-Westfalen, Germany, 45468
Mainz, Rheinland-Pfalz, Germany, 55131
Halle, Sachsen-Anhalt, Germany, 06112
Bad Berka, Thüringen, Germany, 99437
Berlin, Germany, 10115
Hamburg, Germany, 20246 Collapse << |
| NCT00712322 | Neurogenic Detrusor Overactivi... more >>ty Collapse << | Phase 2 | Terminated(Due to extremely di... more >>fficult enrollment.) Collapse << | - | United States, California
... more >>
University of California at San Diego
San Diego, California, United States, 92123
United States, Massachusetts
Children's Hospital, Karp 8210
Boston, Massachusetts, United States, 02115
United States, Michigan
Children's Hospital of Michigan
Detroit, Michigan, United States, 48201-2119
United States, Missouri
Washington University Pediatric Urology
Saint Louis, Missouri, United States, 63110
United States, New York
Pediatric Urology Associates, PC
Lake Success, New York, United States, 11042
Pediatric Urology Associates PC
Tarrytown, New York, United States, 10591
United States, Ohio
Pediatric & Adolescent Urology, Inc Akron Children's Hospital
Akron, Ohio, United States, 44302
Cincinnati Children's Hospital Medical Center
Cincinnati, Ohio, United States, 45229
United States, Oregon
Oregon Health & Science University
Portland, Oregon, United States, 97239
Argentina
Warner Chilcott Investigational Study Site
Buenos Aires, Argentina, C1181ACH
Colombia
Fundacion Hospital Infantil, Universitario De San Jose
Bogota, Colombia
Fundación Valle de Lili
Cali, Colombia
Hospital Pablo Tobon Uribe
Medellín, Colombia Collapse << |
| NCT00127270 | Urinary Incontinence | Phase 4 | Completed | - | - |
| NCT00171145 | Overactive Bladder Syndrome | Phase 3 | Completed | - | United States, New Jersey
... more >>
Novartis
East Hanover, New Jersey, United States, 07936-108 Collapse << |
| NCT01229280 | Bioequivalency | Phase 1 | Unknown | February 2011 | Uruguay
... more >> Center for Clinical Pharmacology Research (CCPR) Bdbeq S.A. Hospital Italiano.
Not yet recruiting
Montevideo, Uruguay, 11600
Contact: Francisco E. Estevez-Carrizo, M.D. +59824876288 francisco.estevez@bdbeq.com.uy
Contact: Mónica Cedrés, Pharm. B. +59824876288 mcderes@bdbeq.com.uy
Principal Investigator: Susana Parrillo, M.D. Collapse << |
| NCT00170768 | Healthy Volunteers | Phase 2 | Completed | - | United States, District of Col... more >>umbia
Washington Neuropsychological Institute LLC Georgetown
Washington, District of Columbia, United States, 20016 Collapse << |
| NCT00413790 | Healthy | Phase 4 | Completed | - | United States, Arizona
... more >>
Investigative Site
Scottsdale, Arizona, United States, 85251
Investigative Site
Tempe, Arizona, United States, 85282
United States, California
Investigative Site
San Diego, California, United States, 92103
United States, Florida
Investigative Site
Jacksonville, Florida, United States, 32216
Investigative Site
Jupiter, Florida, United States, 33458
United States, Kansas
Investigative Site
Overland Park, Kansas, United States, 66215
United States, Kentucky
Investigative Site
Madisonville, Kentucky, United States, 42431
United States, Massachusetts
Investigative Site
Wellesley Hills, Massachusetts, United States, 02481
United States, New Jersey
Investigative Site
Hackensack, New Jersey, United States, 07601
United States, New York
Investigative Site
Buffalo, New York, United States, 14202
United States, Texas
Investigative Site
Austin, Texas, United States, 78752
Investigative Site
Dallas, Texas, United States, 75235 Collapse << |
| NCT00703703 | Healthy | Phase 1 | Completed | - | United States, Arizona
... more >>
Investigative Site
Phoenix, Arizona, United States
Investigative Site
Tempe, Arizona, United States
United States, Arkansas
Investigative Site
Little Rock, Arkansas, United States
United States, California
Investigative Site
San Diego, California, United States
United States, District of Columbia
Investigative Site
Washington, District of Columbia, United States
United States, Florida
Investigative Site
Fort Myers, Florida, United States
Investigative Site
Jacksonville, Florida, United States
Investigative Site
Jupiter, Florida, United States
Investigative Site
Miami, Florida, United States
Investigative Site
Orlando, Florida, United States
United States, Kansas
Investigative Site
Overland Park, Kansas, United States
Investigative Site
Topeka, Kansas, United States
United States, Maryland
Investigative Site
Riverdale, Maryland, United States
United States, Massachusetts
Investigative Site
Wellesley Hills, Massachusetts, United States
United States, Missouri
Investigative Site
Springfield, Missouri, United States
United States, New Jersey
Investigative Site
Hackensack, New Jersey, United States
United States, North Carolina
Investigative Site
Burlington, North Carolina, United States
Investigative Site
Greensboro, North Carolina, United States
United States, Oklahoma
Investigative Site
Oklahoma City, Oklahoma, United States
United States, Tennessee
Investigative Site
Knoxville, Tennessee, United States
United States, West Virginia
Investigative Site
Morgantown, West Virginia, United States Collapse << |
| NCT00170755 | Overactive Bladder Syndrome | Phase 3 | Completed | - | United States, New Jersey
... more >>
Novartis
East Hanover, New Jersey, United States, 07936-108 Collapse << |
| NCT02143570 | Overactive Bladder | Phase 3 | Completed | - | Chile
... more >> Hospital Carlos Van Buren
Valparaiso, Chile, 2341131 Collapse << |
| NCT00171184 | Overactive Bladder | Phase 4 | Completed | - | United States, Texas
... more >>
Scott Department of Urology Baylor College of Medicine
Houston, Texas, United States, 77030 Collapse << |
| NCT01227811 | Bioequivalency | Phase 1 | Unknown | March 2011 | Uruguay
... more >> Center for Clinical Pharmacology Research Bdbeq S.A.; Hospital Italiano de Montevideo..
Not yet recruiting
Montevideo, Uruguay, 11600
Contact: Susana Parrillo, MD +59824876288 ext 201 sparrillo@bdbeq.com.uy
Contact: Mónica Cedrés, Pharm. B. +59824876288 ext 202 mcedres@bdbeq.com.uy Collapse << |
| NCT01189071 | Overactive Bladder
... more >> Renal Colic
Pain, Postoperative Collapse << | Not Applicable | Terminated(Co-Investigator lef... more >>t institution. No data analysis occurred.) Collapse << | - | United States, Missouri
... more >>
University of Missouri Healthcare
Columbia, Missouri, United States, 65212 Collapse << |
| NCT00366002 | Overactive Bladder (OAB) | Phase 4 | Completed | - | - |
| NCT01189071 | - | - | Terminated(Co-Investigator lef... more >>t institution. No data analysis occurred.) Collapse << | - | - |
| NCT00800462 | Spinal Cord Injury
... more >> Neurogenic Detrusor Overactivity Collapse << | Phase 4 | Completed | - | Canada, Manitoba
... more >>
University of Manitoba, Health Sciences Centre
Winnipeg, Manitoba, Canada, R3A1R9
Canada, Ontario
Toronto Rehabilitation Institute, Lyndhurst Centre
Toronto, Ontario, Canada, M4G 3V9 Collapse << |
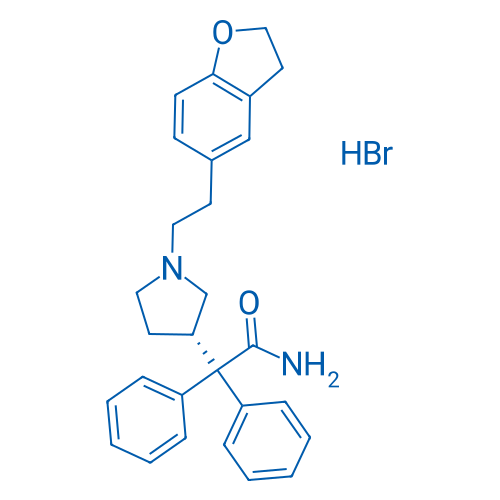

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
