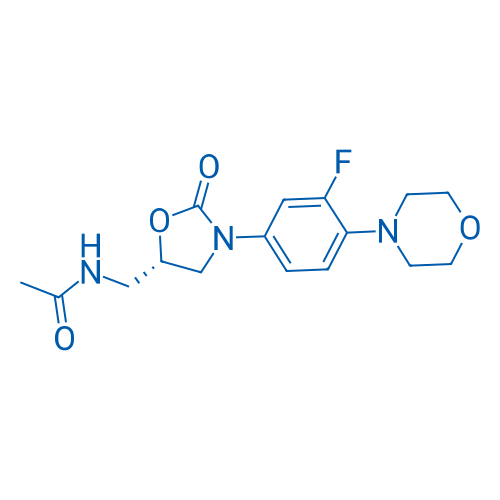
CAS No.: 165800-03-3
吗啉恶酮 Catalog No. CSN16808
Synonyms: PNU-100766;Zyvox;Zyvoxid
Linezolid is a synthetic first-class-oxazolidinone antibiotic active against most Gram-positive bacteria.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01592032 Catheter-Related Infections ... more >> Bacteremia. Collapse << Phase 4 Unknown December 2015 Spain ... more >> Clinica Universidad de Navarra Pamplona, Navarra, Spain, 31008 Collapse << NCT03583333 Hospital-Acquired Bacterial Pn... more >>eumonia Ventilator-Associated Bacterial Pneumonia Collapse << Phase 3 Recruiting April 25, 2021 China, Beijing ... more >> Peking University Third Hospital ( Site 0115) Recruiting Beijing, Beijing, China, 100191 Contact: Study Coordinator +8613910125933 China, Guangdong The First Affiliated Hospital of Guangzhou Medical University ( Site 0123) Recruiting Guangzhou, Guangdong, China, 510120 Contact: Study Coordinator +8613902273260 Guangzhou First People's Hospital ( Site 0101) Recruiting Guangzhou, Guangdong, China, 510180 Contact: Study Coordinator +862081048888 Southern Medical University Nanfang Hospital ( Site 0120) Recruiting Guangzhou, Guangdong, China, 510515 Contact: Study Coordinator +8613119545239 Shenzhen People s Hospital ( Site 0134) Recruiting Shenzhen, Guangdong, China, 518020 Contact: Study Coordinator +8675525533018 China, Jiangsu First Hospital Affiliated to Suzhou University ( Site 0111) Recruiting Suzhou, Jiangsu, China, 215008 Contact: Study Coordinator +8651267780040 Wuxi People's Hospital ( Site 0124) Recruiting Wuxi, Jiangsu, China, 214023 Contact: Study Coordinator +86051085350348 China, Jiangxi Jiangxi Provincial People's Hospital ( Site 0129) Recruiting Nanchang, Jiangxi, China, 330006 Contact: Study Coordinator +8679186895863 The First Affiliated Hospital of Nanchang University ( Site 0132) Recruiting Nanchang, Jiangxi, China, 330006 Contact: Study Coordinator +8613707089183 China, Shanghai Huashan Hospital of Fudan University ( Site 0105) Recruiting Shanghai, Shanghai, China, 200040 Contact: Study Coordinator +8621528899 Shanghai General Hospital ( Site 0125) Recruiting Shanghai, Shanghai, China, 200080 Contact: Study Coordinator 8602163240090 China, Zhejiang The First Affiliated Hospital.Zhejiang University ( Site 0102) Recruiting Hangzhou, Zhejiang, China, 310003 Contact: Study Coordinator +8613505719970 Sir Run Run Shaw Hospital School of Medicine, Zhejiang University ( Site 0110) Recruiting Hangzhou, Zhejiang, China, 310016 Contact: Study Coordinator +86057186006102 The 2nd Affiliated Hospital of Wenzhou Medical University ( Site 0130) Recruiting Wenzhou, Zhejiang, China, 325000 Contact: Study Coordinator +8613857715778 China Aero Space center hospital ( Site 0118) Recruiting Beijing, China, 100049 Contact: Study Coordinator +8613611106725 Beijing Hospital ( Site 0127) Recruiting Beijing, China, 100730 Contact: Study Coordinator +8613701097436 Ruijin Hospital Shanghai Jiao Tong University School of Medicine ( Site 0104) Recruiting Shanghai, China, 200025 Contact: Study Coordinator +8602134186000 Collapse << NCT00035269 Pneumonia, Pneumococcal ... more >> Community Acquired Infections Gram-positive Bacterial Infections Collapse << Phase 3 Completed - - - 更多
- 参考文献
- [1] Swaney SM, Aoki H, et al. The oxazolidinone linezolid inhibits initiation of protein synthesis in bacteria. Antimicrob Agents Chemother. 1998 Dec;42(12):3251-5.
- [2] Shinabarger DL, Marotti KR, et al. Mechanism of action of oxazolidinones: effects of linezolid and eperezolid on translation reactions. Antimicrob Agents Chemother. 1997 Oct;41(10):2132-6.
- [3] Perry CM, Jarvis B. Linezolid: a review of its use in the management of serious gram-positive infections [published correction appears in Drugs. 2003;63(19):2126]. Drugs. 2001;61(4):525–551
- [4] Clemett D, Markham A. Linezolid. Drugs. 2000;59(4):815–828
- [5] Santini A, Ronchi D, Garbellini M, Piga D, Protti A. Linezolid-induced lactic acidosis: the thin line between bacterial and mitochondrial ribosomes. Expert Opin Drug Saf. 2017;16(7):833–843
- [6] Li Y, Xu W. Efficacy and safety of linezolid compared with other treatments for skin and soft tissue infections: a meta-analysis. Biosci Rep. 2018;38(1):BSR20171125. Published 2018 Feb 13
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 165800-03-3 | 储存条件 |
|
|||||||
| 分子式 | C16H20FN3O4 | 运输 | 蓝冰 | |||||||
| 分子量 | 337.35 | 别名 | PNU-100766;Zyvox;Zyvoxid;Zyvoxam;U-100766;利奈唑胺 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01592032 | Catheter-Related Infections ... more >> Bacteremia. Collapse << | Phase 4 | Unknown | December 2015 | Spain ... more >> Clinica Universidad de Navarra Pamplona, Navarra, Spain, 31008 Collapse << |
| NCT03583333 | Hospital-Acquired Bacterial Pn... more >>eumonia Ventilator-Associated Bacterial Pneumonia Collapse << | Phase 3 | Recruiting | April 25, 2021 | China, Beijing ... more >> Peking University Third Hospital ( Site 0115) Recruiting Beijing, Beijing, China, 100191 Contact: Study Coordinator +8613910125933 China, Guangdong The First Affiliated Hospital of Guangzhou Medical University ( Site 0123) Recruiting Guangzhou, Guangdong, China, 510120 Contact: Study Coordinator +8613902273260 Guangzhou First People's Hospital ( Site 0101) Recruiting Guangzhou, Guangdong, China, 510180 Contact: Study Coordinator +862081048888 Southern Medical University Nanfang Hospital ( Site 0120) Recruiting Guangzhou, Guangdong, China, 510515 Contact: Study Coordinator +8613119545239 Shenzhen People s Hospital ( Site 0134) Recruiting Shenzhen, Guangdong, China, 518020 Contact: Study Coordinator +8675525533018 China, Jiangsu First Hospital Affiliated to Suzhou University ( Site 0111) Recruiting Suzhou, Jiangsu, China, 215008 Contact: Study Coordinator +8651267780040 Wuxi People's Hospital ( Site 0124) Recruiting Wuxi, Jiangsu, China, 214023 Contact: Study Coordinator +86051085350348 China, Jiangxi Jiangxi Provincial People's Hospital ( Site 0129) Recruiting Nanchang, Jiangxi, China, 330006 Contact: Study Coordinator +8679186895863 The First Affiliated Hospital of Nanchang University ( Site 0132) Recruiting Nanchang, Jiangxi, China, 330006 Contact: Study Coordinator +8613707089183 China, Shanghai Huashan Hospital of Fudan University ( Site 0105) Recruiting Shanghai, Shanghai, China, 200040 Contact: Study Coordinator +8621528899 Shanghai General Hospital ( Site 0125) Recruiting Shanghai, Shanghai, China, 200080 Contact: Study Coordinator 8602163240090 China, Zhejiang The First Affiliated Hospital.Zhejiang University ( Site 0102) Recruiting Hangzhou, Zhejiang, China, 310003 Contact: Study Coordinator +8613505719970 Sir Run Run Shaw Hospital School of Medicine, Zhejiang University ( Site 0110) Recruiting Hangzhou, Zhejiang, China, 310016 Contact: Study Coordinator +86057186006102 The 2nd Affiliated Hospital of Wenzhou Medical University ( Site 0130) Recruiting Wenzhou, Zhejiang, China, 325000 Contact: Study Coordinator +8613857715778 China Aero Space center hospital ( Site 0118) Recruiting Beijing, China, 100049 Contact: Study Coordinator +8613611106725 Beijing Hospital ( Site 0127) Recruiting Beijing, China, 100730 Contact: Study Coordinator +8613701097436 Ruijin Hospital Shanghai Jiao Tong University School of Medicine ( Site 0104) Recruiting Shanghai, China, 200025 Contact: Study Coordinator +8602134186000 Collapse << |
| NCT00035269 | Pneumonia, Pneumococcal ... more >> Community Acquired Infections Gram-positive Bacterial Infections Collapse << | Phase 3 | Completed | - | - |
| NCT02609646 | - | - | Recruiting | December 2019 | Italy ... more >> Ospedale Maurizio Bufalini, UO Anestesia Terapia Intensiva Recruiting Cesena, Emilia Romagna, Italy, 47023 Contact: Marco Benni, MD Principal Investigator: Marco Benni, MD Ospedale S. Giovanni di Dio ASL 10, Servizio Anestesia e Rianimazione Not yet recruiting Firenze, FI, Italy, 50143 Principal Investigator: Valerio Mangani, MD Presidio Ospedaliero "San Leopoldo Mandic" Recruiting Merate, Lecco, Italy, 23807 Contact: Davide Guzzon, MD Principal Investigator: Davide Guzzon, MD Ospedale A. Manzoni, U.O. Anestesia e Rianimazione 1 Recruiting Lecco, Lombardia, Italy, 23900 Contact: Giuseppe Nattino, MD Principal Investigator: Giuseppe Nattino, MD Sub-Investigator: Rita Ciceri, MD Ospedale San Giovanni Bosco, Servizio Anestesia e Rianimazione B-DEA Recruiting Torino, Piemonte, Italy, 10148 Contact: Daniela Silengo, MD Principal Investigator: Daniela Silengo, MD Fondazione IRCCS Policlinico San Matteo Recruiting Pavia, PV, Italy, 27100 Contact: Alessandro Amatu, MD Principal Investigator: Alessandro Amatu, MD Ospedale Giovanni Paolo II Not yet recruiting Olbia, Sardegna, Italy, 07026 Contact: Maria Giovanna Dessena, MD 0789552984 Principal Investigator: Maria Giovanna Dessena, MD Ospedale del Mugello Recruiting Borgo San Lorenzo, Toscana, Italy, 50032 Contact: Rossella Carlà, MD Principal Investigator: Rossella Carlà, MD Ospedale Santa Maria Nuova, Anestesia e Rianimazione Recruiting Firenze, Toscana, Italy, 50122 Contact: Massimo Barattini, MD Principal Investigator: Massimo Barattini, MD Ospedale Alta Val d'Elsa, Terapia Intensiva Not yet recruiting Poggibonsi, Toscana, Italy, 53036 Contact: Andrea Veneziani, MD Principal Investigator: Andrea Veneziani, MD Collapse << |
| NCT00754273 | - | - | Completed | - | United States, Kentucky ... more >> University of Kentucky Medical Center Lexington, Kentucky, United States, 40536 Collapse << |
| NCT00671814 | Healthy Subjects | Phase 1 | Completed | - | United States, Wisconsin ... more >> Covance Clinical Research Unit Madison, Wisconsin, United States, 53704 Collapse << |
| NCT00925093 | Neurosurgery | Phase 1 | Withdrawn(Primary investigator... more >> left institution) Collapse << | December 2013 | - |
| NCT00990990 | Tuberculosis | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT03032380 | Healthcare-associated Pneumoni... more >>a (HCAP) Hospital Acquired Pneumonia (HAP) Ventilator Associated Pneumonia (VAP) Collapse << | Phase 3 | Recruiting | February 1, 2019 | - |
| NCT01967225 | Skin Diseases, Infectious | Phase 3 | Completed | - | Japan ... more >> Nagakute, Aichi, Japan, 480-1195 Nagoya, Aichi, Japan, 455-8530 Nagoya, Aichi, Japan, 457-8510 Toyoake, Aichi, Japan, 470-1192 Yoshida, Fukui, Japan, 910-1193 Kasuga, Fukuoka, Japan, 816-0864 Kitakyushu, Fukuoka, Japan, 802-0077 Miyako-gun, Fukuoka, Japan, 800-0344 Sapporo, Hokkaido, Japan, 006-8555 Sapporo, Hokkaido, Japan, 060-0061 Amagasaki, Hyogo, Japan, 660-8511 Kobe, Hyogo, Japan, 650-0017 Inashiki-gun, Ibaraki, Japan, 300-0395 Tsukuba, Ibaraki, Japan, 305-8576 Kamakura, Kanagawa, Japan, 247-8533 Sagamihara, Kanagawa, Japan, 252-0375 Yokohama, Kanagawa, Japan, 231-8682 Koshi, Kumamoto, Japan, 861-1196 Tsu, Mie, Japan, 514-1101 Sendai, Miyagi, Japan, 983-8520 Nakagami-gun, Okinawa, Japan, 901-2393 Shimajiri, Okinawa, Japan, 901-0493 Hamamatsu, Shizuoka, Japan, 430-0929 Iwata, Shizuoka, Japan, 438-8550 Numazu, Shizuoka, Japan, 410-8555 Meguro-ku, Tokyo, Japan, 152-8902 Musashimurayama, Tokyo, Japan, 208-0011 Ota-ku, Tokyo, Japan, 143-0013 Ota-ku, Tokyo, Japan, 143-8541 Ota-ku, Tokyo, Japan, 145-0065 Setagaya-ku, Tokyo, Japan, 158-8531 Shinagawa, Tokyo, Japan, 141-8625 Shinjuku-ku, Tokyo, Japan, 162-8655 Tachikawa, Tokyo, Japan, 190-0014 Yonago, Tottori, Japan, 683-8605 Shimonoseki, Yamaguchi, Japan, 750-8520 Kofu, Yamanashi, Japan, 400-8506 Fukuoka, Japan, 810-0001 Gifu, Japan, 500-8513 Kochi, Japan, 781-8555 Kumamoto, Japan, 860-0008 Nagasaki, Japan, 852-8501 Osaka, Japan, 534-0021 Shizuoka, Japan, 420-8527 Shizuoka, Japan, 424-8636 Toyama, Japan, 930-0194 Collapse << |
| NCT02679573 | Community Acquired Bacterial P... more >>neumonia Collapse << | Phase 3 | Completed | - | - |
| NCT00501150 | Gram-positive Bacterial Infect... more >>ions Staphylococcal Infections Collapse << | Not Applicable | Completed | - | United Kingdom ... more >> Imperial College London London, United Kingdom, w12 0nn Collapse << |
| NCT00326287 | Pneumonia | Phase 3 | Completed | - | - |
| NCT00229008 | Pneumonia | Phase 3 | Completed | - | - |
| NCT01967225 | - | - | Completed | - | - |
| NCT00035854 | Bacterial Infections | Phase 3 | Completed | - | - |
| NCT00210964 | Pneumonia | Phase 3 | Completed | - | - |
| NCT01819935 | - | - | Completed | - | - |
| NCT01819935 | - | - | Completed | - | - |
| NCT00087490 | Skin/Soft Tissue Infections ... more >> Methicillin Resistant Staphylococcus Aureus (MRSA) Collapse << | Phase 4 | Completed | - | - |
| NCT01554995 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul,, Korea, Republic of, 138736 Collapse << |
| NCT01283581 | Skin and Subcutaneous Tissue B... more >>acterial Infections Collapse << | Phase 2 | Completed | - | - |
| NCT01283581 | - | - | Completed | - | - |
| NCT02426918 | Bacterial Infections | Phase 2 | Completed | - | United States, California ... more >> Dream Team Clinical Research, LLC Anaheim, California, United States, 91776 Physician Alliance Research Center Anaheim, California, United States, 92804 Southbay Pharma Research Buena Park, California, United States, 90620 eStudySite - Chula Vista Chula Vista, California, United States, 91911 eStudySite - La Mesa La Mesa, California, United States, 91942 Long Beach Clinical Trials LLC Long Beach, California, United States, 90806 Alliance Research Long Beach, California, United States, 90813 Central Valley Research, LLC Modesto, California, United States, 95350 eStudySite - Oceanside Oceanside, California, United States, 92056 Olive View - UCLA Medical Center Sylmar, California, United States, 91342 United States, Florida Shands Burn Center at the University of Florida Gainesville, Florida, United States, 32610 Central Florida Internists Orlando, Florida, United States, 32811 Triple O Research Institute West Palm Beach, Florida, United States, 33401 United States, Georgia Columbus Regional Research Columbus, Georgia, United States, 31904 United States, Michigan Beaumont Infectious Disease Services Royal Oak, Michigan, United States, 48073 United States, Montana Mercury Street Medical Group PLLC Butte, Montana, United States, 59701 United States, Nevada eStudySite - Las Vegas Las Vegas, Nevada, United States, 89109 United States, New Jersey South Jersey Infectious Disease Somers Point, New Jersey, United States, 08244 Holy Name Medical Center Teaneck, New Jersey, United States, 07666 United States, Ohio ID Clinical Research, Ltd. Toledo, Ohio, United States, 43608 United States, Texas East Montgomery County Clinic Houston, Texas, United States, 77070 Tidwell Medical Center Splendora, Texas, United States, 77372 Collapse << |
| NCT00087490 | - | - | Completed | - | - |
| NCT01757236 | Hip Prosthetic Joint Infection | Phase 2 | Unknown | April 2016 | France ... more >> CHRU de Tours Recruiting Tours, Indre et Loire, France, 37044 Contact: Louis BERNARD +33 (0) 2 47 47 97 74 l.bernard@chu-tours.fr Principal Investigator: Louis BERNARD CHU de Nantes Recruiting Nantes, France Contact: David BOUTOILLE Principal Investigator: David BOUTOILLE CHU de Poitiers Not yet recruiting Poitiers, France Contact: Gwenaël LE MOAL Principal Investigator: Gwenaël LE MOAL Centre de Chirurgie Orthopédique et de la Main Recruiting Strasbourg, France, 67400 Contact: Jeannot GAUDIAS Jeannot.GAUDIAS@chru-strasbourg.fr Principal Investigator: Jeannot GAUDIAS CH de Tourcoing Not yet recruiting Tourcoing, France, 59208 Contact: Eric SENNEVILLE esenneville@ch-tourcoing.fr Principal Investigator: Eric SENNEVILLE Italy Azienda Opedaliera Universitaria San Maria della Misericordia Recruiting Udine, Italy, 33100 Contact: Matteo BASSETTI mattba@tin.it Principal Investigator: Matteo BASSETTI Spain Hospital Clinic of Barcelona Recruiting Barcelona, Spain, 08036 Contact: Alex SORIANO ASORIANO@clinic.ub.es Principal Investigator: Alex SORIANO Hospital General Universitario Gregorio Maranon Not yet recruiting Madrid, Spain, 28007 Contact: Mar SANCHEZ SOMOLINOS Principal Investigator: Mar SANCHEZ SOMOLINOS Hospital Son Llatzer Not yet recruiting Palma de Mallorca, Spain, 07198 Contact: Bartolome LLADO FERRER Principal Investigator: Bartolome LLADO FERRER Collapse << |
| NCT00108433 | Bacteremia Gr... more >>am-Positive Bacterial Infections Collapse << | Phase 3 | Terminated(See Detailed Descri... more >>ption) Collapse << | - | United States, Maryland ... more >> Pfizer Investigational Site Baltimore, Maryland, United States, 21201-1524 Pfizer Investigational Site Baltimore, Maryland, United States, 21201 Pfizer Investigational Site Baltimore, Maryland, United States, 21230 Colombia Pfizer Investigational Site Barranquilla, Atlantico, Colombia Pfizer Investigational Site Bogota, Cundinamarca, Colombia, 0 Pfizer Investigational Site Bogota, D.C, Colombia India Pfizer Investigational Site Hyderbad, Andhra Pradesh, India, 500 082 Pfizer Investigational Site New Delhi, Delhi, India, 110 044 Pfizer Investigational Site Bangalore, Karnataka, India, 560 034 Pfizer Investigational Site Bangalore, Karnataka, India, 560 054 Pfizer Investigational Site Chandigarh, Punjab, India, 160 012 Pfizer Investigational Site Chennai, Tamil Nadu, India, 600 004 Israel Pfizer Investigational Site Tel-Aviv, Israel, 64239 Italy Pfizer Investigational Site Imperia, Italy, 18100 Poland Pfizer Investigational Site Czestochowa, Poland, 42-200 Slovakia Pfizer Investigational Site Banska Bystrica, Slovakia, 975 17 Pfizer Investigational Site Nitra, Slovakia, 950 01 Collapse << |
| NCT02619994 | Tuberculosis, Multidrug-Resist... more >>ant Collapse << | Phase 2 | Recruiting | December 2019 | Korea, Republic of ... more >> Division of Pulmonology and Critical Care Medicine, Department of Internal Medicine and Lung Institute of Medical Research Center, Seoul National University College of Medicine Recruiting Seoul, Korea, Republic of, 110-744 Contact: Jae-Joon Yim, MD +82-2-2072-2059 yimjj@snu.ac.kr Collapse << |
| NCT00108433 | - | - | Terminated(See Detailed Descri... more >>ption) Collapse << | - | - |
| NCT00037050 | Bacterial Infections ... more >> Gram-Positive Bacterial Infections Bacteremia Collapse << | Phase 3 | Completed | - | - |
| NCT00303550 | Skin Diseases, Bacterial | Phase 3 | Completed | - | United States, Illinois ... more >> Edward Hospital & Health Services Naperville, Illinois, United States, 60540 United States, Wyoming Wyoming Medical Center Casper, Wyoming, United States, 82601 Collapse << |
| NCT02067390 | - | - | Recruiting | July 2020 | United States, Illinois ... more >> Northwestern Medicine Recruiting Chicago, Illinois, United States, 60611 Contact: Scheetz mscheetz@nmh.org Principal Investigator: Marc H Scheetz, PharmD, MSc Collapse << |
| NCT00646958 | - | - | Completed | - | - |
| NCT01339091 | Abscess Wound... more >> Infection Surgical Site Infection Cellulitis Collapse << | Phase 3 | Completed | - | - |
| NCT02732327 | - | - | Terminated(No longer aligned w... more >>ith the revised clinical development plan and commercial strategy) Collapse << | - | - |
| NCT00646958 | Infectious Skin Diseases ... more >> Bacterial Skin Diseases Staphylococcal Skin Infections Streptococcal Infections Abscess Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Drug Research & Analysis Corp Montgomery, Alabama, United States United States, California OWENS Pharma Research Center Long Beach, California, United States Healthcare Partners Medical Group Los Angeles, California, United States HealthCare Partners Medical Group Pasadena, California, United States Olive View- UCLA Center, Dept. Emergency Medicine Sylmar, California, United States United States, Florida South Florida Clinical Research Atlantis, Florida, United States United States, Georgia Southeast Regional Research Group Columbus, Georgia, United States Contemporary Medicine, LLC Hinesville, Georgia, United States United States, Indiana Infectious Disease of Indiana, PSC. Indianapolis, Indiana, United States United States, Louisiana Clinical Trials of America, Inc. Shreveport, Louisiana, United States United States, Michigan Arnold Markowitz, MD., PC Keego Harbor, Michigan, United States United States, Montana Mercury Street Medical Group, LLC Butte, Montana, United States United States, New York Jamaica Hospital Medical Center Jamaica, New York, United States United States, North Carolina ALL-TRIALS Clinical Research, LLC Winston-Salem, North Carolina, United States United States, Ohio Summa Health System Akron, Ohio, United States United States, Pennsylvania Brandywine Clinical Research Downington, Pennsylvania, United States Warminster Medical Associates, P.C. Warminster, Pennsylvania, United States United States, Tennessee McKenzie Medical Center McKenzie, Tennessee, United States United States, Utah J. Lewis Research, Inc. Salt Lake City, Utah, United States Collapse << |
| NCT01431339 | Abscess Wound... more >> Infection Surgical Site Infection Cellulitis Collapse << | Phase 3 | Completed | - | - |
| NCT03716024 | Infectious Skin Disease ... more >> Bacterial Skin Disease Collapse << | Phase 2 | Completed | - | - |
| NCT00035425 | Febrile Neutropenia | Phase 3 | Completed | - | - |
| NCT01431339 | - | - | Completed | - | - |
| NCT01039610 | Infections, Bacterial | Phase 1 | Withdrawn(Internal decision to... more >> progress alternate molecule with more preferable profile) Collapse << | August 2010 | - |
| NCT01339091 | - | - | Completed | - | - |
| NCT00949130 | Acute Bacterial Skin and Skin ... more >>Structure Infections Collapse << | Phase 2 | Completed | - | United States, California ... more >> Sharp Chula Vista Research Office Chula Vista, California, United States, 91911 Fountain Valley Regional Hospital and Medical Center Fountain Valley, California, United States, 92708 Sharp Grossmont Research Office LaMesa, California, United States, 91942 Tri-City Oceanside Office Oceanside, California, United States, 92056 United States, Georgia Southeast Regional Research Group Columbus, Georgia, United States, 31904 Southeast Regional Research Group Savannah, Georgia, United States, 31406 United States, Idaho Idaho Falls Infectious Diseases Idaho Falls, Idaho, United States, 83404 United States, Minnesota Hennepin County Medical Center Minneapolis, Minnesota, United States, 55415 Infectious Diseaes MPLS-LTD Minneapolis, Minnesota, United States, 55422 United States, Montana Mercury Street Medical Group Butte, Montana, United States, 59701 United States, Pennsylvania RPS Infectious Diseases West Reading, Pennsylvania, United States, 19611 Guatemala Roosevelt Hospital Guatemala, Guatemala, 1011 UNICAR Guatemala, Guatemala, 1011 Military Health Center Guatemala, Guatemala Private Hospital Quetzaltenango, Guatemala Collapse << |
| NCT00299520 | Complicated Skin and Skin Stru... more >>cture Infection Collapse << | Phase 3 | Completed | - | United States, California ... more >> e-Study Site National City, California, United States, 91950 United States, Idaho VA Medical Center Boise, Idaho, United States, 83702 United States, Indiana Infectious Disease of Indiana Indianapolis, Indiana, United States, 46260 United States, Maryland Judith Stone. M.D. Cumberland, Maryland, United States, 21502 United States, Ohio ID Clinical Research, Ltd. Toledo, Ohio, United States, 43608 Collapse << |
| NCT02732327 | Neoplasms Feb... more >>rile Neutropenia Collapse << | Phase 2 | Terminated(No longer aligned w... more >>ith the revised clinical development plan and commercial strategy) Collapse << | - | United States, Texas ... more >> MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00177723 | - | - | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00519025 | - | - | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00396084 | - | - | Completed | - | - |
| NCT00633152 | - | - | Completed | - | - |
| NCT00396084 | Tuberculosis | Phase 1 Phase 2 | Completed | - | United States, California ... more >> San Francisco General Hospital - Pulmonary and Critical Care Medicine San Francisco, California, United States, 94110-3518 Brazil Universidade Federal do Espirito Santo - Duke Hubert-Yeargan Center Vitória, Espírito Santo, Brazil, 29040-091 Collapse << |
| NCT00711854 | MRSA Infection | Phase 4 | Completed | - | Switzerland ... more >> Geneva University Hospitals Geneva, Switzerland, 1211 Collapse << |
| NCT02975570 | Tuberculosis Multi Drug Resist... more >>ant Active Collapse << | Phase 3 | Withdrawn(The study could not ... more >>be conducted since funding was not obtained.) Collapse << | August 2022 | - |
| NCT00865280 | Skin Diseases, Infectious | Phase 3 | Terminated(Terminated) | - | United States, California ... more >> Paratek Recruiting Site Fountain Valley, California, United States, 92708 Parateck Recruiting Site La Mesa, California, United States, 91942 Paratek Recruiting Site Oceanside, California, United States, 92056 Paratek Recruiting Site San Diego, California, United States, 92114 Paratek Recruiting Site San Jose, California, United States, 95154 United States, Georgia Parateck Recruiting Site Columbus, Georgia, United States, 31904 Paratek Recruiting Site Savannah, Georgia, United States, 31406 Collapse << |
| NCT00633152 | Bacterial Infection | Phase 2 | Completed | - | United States, California ... more >> Investigational Site Buena Park, California, United States, 96020 Investigational Site Long Beach, California, United States, 90813 Investigational Site Los Angeles, California, United States, 90015 Investigational Site Rolling Hills Estate, California, United States, 90274 Investigational Site San Diego, California, United States, 92114 United States, Florida Investigational Site Atlantis, Florida, United States, 33462 United States, Georgia Investigational Site Columbus, Georgia, United States, 31904 Investigational Site Savannah, Georgia, United States, 31405 United States, Minnesota Investigational Site Minneapolis, Minnesota, United States, 55422 United States, Montana Investigational Site Butte, Montana, United States, 59701 United States, New Jersey Investigational Site Somers Point, New Jersey, United States, 08244 United States, Ohio Investigational Site Columbus, Ohio, United States, 43215 Investigational Site Toledo, Ohio, United States, 43608 Collapse << |
| NCT00876850 | Skin Structures and Soft Tissu... more >>e Infections Collapse << | Phase 3 | Withdrawn(Terminated) | August 2010 | - |
| NCT01363271 | - | - | Completed | - | - |
| NCT00572559 | Pneumonia, Ventilator-Associat... more >>ed Collapse << | Phase 4 | Completed | - | - |
| NCT01295801 | - | - | Unknown | September 2011 | China, Beijing ... more >> Chinese PLA General Hospital Recruiting Beijing, Beijing, China, 100853 Collapse << |
| NCT00150332 | Infections, Nosocomial | Phase 3 | Completed | - | Japan ... more >> Pfizer Investigational Site Kamogawa, Chiba, Japan, 296-0041 Pfizer Investigational Site Onga-gun, Fukuoka, Japan, 807-0051 Pfizer Investigational Site Sappopro, Hokkaido, Japan, 060-8648 Pfizer Investigational Site Sapporo, Hokkaido, Japan, 006-8555 Pfizer Investigational Site Moriya, Ibaraki, Japan, 302-0118 Pfizer Investigational Site Yokohama, Kanagawa, Japan, 236-0051 Pfizer Investigational Site Isahaya, Nagasaki, Japan, 854-8501 Pfizer Investigational Site Kurashiki, Okayama, Japan, 701-0912 Pfizer Investigational Site Kurashiki, Okayama, Japan Pfizer Investigational Site Suita, Osaka, Japan, 565-8565 Pfizer Investigational Site Iruma-gun, Saitama, Japan, 350-0495 Pfizer Investigational Site Kodaira, Tokyo, Japan, 187-0004 Pfizer Investigational Site Fukuoka, Japan, 811-0213 Pfizer Investigational Site Fukuoka, Japan, 813-0025 Pfizer Investigational Site Hiroshima, Japan, 730-8619 Pfizer Investigational Site Hiroshima, Japan, 734-8530 Pfizer Investigational Site Kyoto, Japan, 615-8256 Pfizer Investigational Site Nagasaki, Japan, 852-8501 Pfizer Investigational Site Okayama, Japan, 700-8505 Pfizer Investigational Site Okayama, Japan, 703-8275 Collapse << |
| NCT00084266 | Methicillin Resistant Staphylo... more >>coccus Aureus (MRSA) Collapse << | Phase 4 | Completed | - | - |
| NCT02559310 | Community Acquired Pneumonia | Phase 3 | Completed | - | - |
| NCT03086486 | Tuberculosis, Pulmonary ... more >> Tuberculosis, Multidrug-Resistant Tuberculosis, MDR Tuberculosis Extensively Drug-Resistant Tuberculosis Pre-XDR-TB Collapse << | Phase 3 | Recruiting | January 2022 | Georgia ... more >> National Center for Tuberculosis and Lung Diseases Recruiting Tbilisi, Georgia, 0101 Contact: Lali Mikiashvili Principal Investigator: Lali Mikiashvili Moldova, Republic of Institute of Phthisiopneumology Chiril Draganiuc Not yet recruiting Chisinau, Moldova, Republic of, 2025 Contact: Elena Tudor, MD Principal Investigator: Elena Tudor, MD Russian Federation Central TB Research Institute of the Federal Agency of Scientific Organizations Moscow Not yet recruiting Moscow, Russian Federation, 107564 Contact: Tatevik Bagdasaryan Principal Investigator: Tatevik Bagdasaryan Research Institute of Phthisiopulmonology of I.M. Sechenov First Moscow State Medical University Not yet recruiting Moscow, Russian Federation Contact: Anastasia Samoilova Principal Investigator: Anastasia Samoilova FSBI "Saint-Petersburg Research Institute of Phthisiopulmonology" Recruiting Saint Petersburg, Russian Federation, 191036 Contact: Petr Yablonskiy, MD Principal Investigator: Petr Yablonskiy, MD Ural Research Institute of Phthisiopulmonology Not yet recruiting Yekaterinburg, Russian Federation, 620039 Contact: Sergey Skornyakov, MD Principal Investigator: Sergey Skornyakov, MD South Africa Tshepong Hospital Recruiting Klerksdorp, North - West, South Africa, 2574 Contact: Ebrahim Variava Principal Investigator: Ebrahim Variava King DinuZulu Hospital Complex Recruiting Durban, South Africa, 4015 Contact: Nosipho Ngubane Principal Investigator: Nosipho Ngubane Clinical HIV Research Unit (CHRU) Sizwe Tropical Diseases Hospital Recruiting Johannesburg, South Africa, 2131 Contact: Pauline Howell, MD Principal Investigator: Pauline Howell, MD Empilweni TB Hospital Not yet recruiting Johannesburg, South Africa, 2194 Contact: Francesca Conradie, MBBS Principal Investigator: Francesca Conradie, MBBS Collapse << |
| NCT03220074 | Non-tuberculous Mycobacterial ... more >>Diseases Collapse << | Phase 4 | Not yet recruiting | June 15, 2021 | Thailand ... more >> Siriraj Hospital Not yet recruiting Bangkoknoi, Bangkok, Thailand, 10700 Contact: Yupin Suputtamongkol, MD 66 2 419 7203 yupin.sup@mahidol.ac.th Contact: nasikarn Angkasekwinai, MD 66818708766 nasikarn@gmail.com Sub-Investigator: Pakpoom Phoompoung, MD Sub-Investigator: Niphon Chirapapisan, MD Sub-Investigator: Suporn Foonglada, PhD Sub-Investigator: Peeraya Krongtip, MSc Sub-Investigator: Ekkarat Wongsawat, MSc Collapse << |
| NCT00147511 | Gram-Positive Bacterial Infect... more >>ions Skin and Connective Tissue Diseases Collapse << | Phase 4 | Completed | - | - |
| NCT00084266 | - | - | Completed | - | - |
| NCT00475891 | - | - | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Medical Center Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02991131 | - | - | Terminated(Company decision fo... more >>r 18299 TERMINATION status) Collapse << | - | Russian Federation ... more >> Multiple Locations, Russian Federation Singapore Multiple Locations, Singapore Collapse << |
| NCT02279875 | Pulmonary Tuberculosis | Phase 2 | Completed | - | South Africa ... more >> TASK Clinical Research Centre Bellville, Cape Town, South Africa, 7530 University of Cape Town Lung Institute (Pty) Ltd Mowbray, Cape Town, South Africa, 7700 Collapse << |
| NCT00948142 | Skin Diseases, Bacterial | Phase 2 | Completed | - | United States, California ... more >> Chula Vista, California, United States, 91911 La Mesa, California, United States, 91942 Los Angeles, California, United States, 90015 Oceanside, California, United States, 92056 Oxnard, California, United States, 93030 Pasadena, California, United States, 91105 Santa Ana, California, United States, 92701 Torrance, California, United States, 90501 Torrance, California, United States, 90509 United States, Georgia Columbus, Georgia, United States, 31904 Savannah, Georgia, United States, 31406 United States, Illinois Springfield, Illinois, United States, 62701 United States, Michigan Detroit, Michigan, United States, 48202 United States, Montana Butte, Montana, United States, 59701 United States, New Jersey Somers Point, New Jersey, United States, 08244 United States, Ohio Akron, Ohio, United States, 44304 Collapse << |
| NCT01994460 | Pulmonary Tuberculosis Without... more >> Resistance to Rifampicin Collapse << | Phase 2 | Unknown | June 2016 | Korea, Republic of ... more >> Seoul National University Bundang Hospital Recruiting Seongnam, Kyunggi, Korea, Republic of, 463-707 Contact: Jong Sun Park, MD 031-787-7054 jspark.im@gmail.com Principal Investigator: Jong Sun Park, MD National Medical Center Recruiting Seoul, Korea, Republic of, 100-799 Contact: Ji Yeon Lee, MD 82222607284 jedidiah125@gmail.com Principal Investigator: Ji Yeon Lee, MD SMG-SNU Boramae Medical Center Recruiting Seoul, Korea, Republic of, 156-707 Contact: Deog Kyeom Kim, MD 02-870-2228 kimdkmd@gmail.com Principal Investigator: Deog Kyeom Kim, MD Collapse << |
| NCT02269644 | Community Acquired Pneumonia | Phase 3 | Withdrawn | December 2016 | United States, Montana ... more >> Mercury Street Medical Group Butte, Montana, United States, 59701 Collapse << |
| NCT02066402 | Bacterial Infections | Phase 3 | Completed | - | - |
| NCT01209078 | Skin Infections, Bacterial | Phase 2 | Completed | - | United States, Alabama ... more >> GSK Investigational Site Anniston, Alabama, United States, 36207 United States, California GSK Investigational Site Chula Vista, California, United States, 91911 GSK Investigational Site La Mesa, California, United States, 91942 GSK Investigational Site Long Beach, California, United States, 90813 GSK Investigational Site Oceanside, California, United States, 92056 United States, Hawaii GSK Investigational Site Honolulu, Hawaii, United States, 96813 United States, Pennsylvania GSK Investigational Site West Reading, Pennsylvania, United States, 19611 Collapse << |
| NCT02570490 | Acute Bacterial Skin and Skin ... more >>Structure Infections Collapse << | Phase 3 | Completed | - | - |
| NCT03126890 | - | - | Recruiting | December 2020 | Taiwan ... more >> National Taiwan University Hospital Recruiting Taipei, Test2, Taiwan, test3 Contact: Shu-Wen Lin 02 - 33668782 shuwenlin@ntu.edu.tw Collapse << |
| NCT00795145 | Bacterial Infections | Phase 1 | Completed | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT02333799 | Pulmonary Tuberculosis | Phase 3 | Active, not recruiting | October 2021 | South Africa ... more >> Task Applied Science - Brooklyn Chest Hospital Ysterplaat, Cape Town, South Africa, 7405 King DinuZulu Hospital Complex Sydenham, Durban, South Africa, 4001 Sizwe Tropical Disease Hospital Sandringham, Johannesburg, South Africa, 2131 THINK: Tuberculosis & HIV Investigative Network of KwaZulu-Natal (Durban) Pietermaritzburg, Kwazulu-Natal, South Africa, 3200 Collapse << |
| NCT03643497 | - | - | Recruiting | December 31, 2021 | China ... more >> Beijing Children's Hospital of Capital Medical University Recruiting Beijing, China Contact: Adong Shen, Master 13370115087 shenad16@hotmail.com Collapse << |
| NCT00852540 | Skin Infections, Bacterial | Phase 3 | Completed | - | - |
| NCT02066402 | - | - | Completed | - | - |
| NCT00795145 | - | - | Completed | - | - |
| NCT00255996 | Burns | Phase 1 | Completed | - | France ... more >> Pfizer Investigational Site Nantes, France, 44093 Pfizer Investigational Site Paris, France, 75015 Pfizer Investigational Site Paris, France, 75679 Collapse << |
| NCT01421511 | Skin and Subcutaneous Tissue B... more >>acterial Infections Collapse << | Phase 3 | Completed | - | - |
| NCT01209078 | - | - | Completed | - | - |
| NCT00852540 | - | - | Completed | - | - |
| NCT01564758 | - | - | Completed | - | - |
| NCT01564758 | - | - | Completed | - | - |
| NCT01421511 | - | - | Completed | - | - |
| NCT00811980 | - | - | Completed | - | - |
| NCT00664313 | Multi-drug Resistant Tuberculo... more >>sis Extensively Drug Resistant Tuberculosis Collapse << | Phase 1 Phase 2 | Completed | - | South Africa ... more >> King George V Hospital Durban, South Africa Collapse << |
| NCT01356472 | Methicillin-resistant Staphylo... more >>coccus Aureus Pneumonia, Ventilator-Associated Collapse << | Phase 4 | Unknown | - | - |
| NCT02019420 | Pneumonia | Phase 3 | Completed | - | - |
| NCT01040585 | - | - | Withdrawn | March 2011 | - |
| NCT00359632 | Optic Nerve Diseases | Phase 3 | Terminated(Study was stopped d... more >>ue to poor enrollment on 28 Feb 2012. Reason for termination was not due to safety concerns.) Collapse << | - | United States, Arkansas ... more >> St. Bernards Research Center Jonesboro, Arkansas, United States, 72401 United States, Florida Triple O Research Institute, PA West Palm Beach, Florida, United States, 33401 United States, Louisiana Ochsner Clinic Foundation New Orleans, Louisiana, United States, 70121 United States, Michigan Henry Ford Health System Detroit, Michigan, United States, 48202 United States, Minnesota University of Minnesota, Department of Medicine/Division of Infectious Diseases Minneapolis, Minnesota, United States, 55455 United States, Pennsylvania Drexel University College of Medicine, Partnership Comprehensive Care Practice Philadelphia, Pennsylvania, United States, 19102 Associates in Infectious Disease and Tropical Medicine Pittsburgh, Pennsylvania, United States, 15206 Italy Azienda Ospedaliera Universitaria di San Martino Genova, Italy, 16132 Ospedale San Martino, Clinica Malattie Infettive Genova, Italy, 16132 Università di Genova Genova, Italy, 16132 Clinica Malattie Infettive, Azienda Ospedaliero Universitaria Santa Maria della Misericordia Udine, Italy, 33100 Sweden Infektionskliniken 1-73, Karolinska Universitetssjukhuset Huddinge Stockholm, Sweden, 141 86 Collapse << |
| NCT00359632 | - | - | Terminated(Study was stopped d... more >>ue to poor enrollment on 28 Feb 2012. Reason for termination was not due to safety concerns.) Collapse << | - | - |
| NCT00727844 | - | - | Completed | - | - |
| NCT00727844 | Pulmonary Tuberculosis ... more >> Multidrug Resistant Tuberculosis Extensively Drug Resistant Tuberculosis Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> National Masan Tuberculosis Hospital Changwon, Korea, Republic of National Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02087566 | Renal Failure ... more >> Impaired Renal Function Collapse << | Not Applicable | Completed | - | - |
| NCT01170221 | Skin and Subcutaneous Tissue B... more >>acterial Infections Collapse << | Phase 3 | Completed | - | - |
| NCT01128530 | Complicated Skin and Skin Stru... more >>cture Infections Collapse << | Phase 2 | Completed | - | United States, California ... more >> Furiex Research Site Anaheim, California, United States, 92804 Furiex Research Site Buena Park, California, United States, 90620 Furiex Research Site Chula Vista, California, United States, 91911 Furiex Research Site Fountain Valley, California, United States, 92708 Furiex Research Site La Mesa, California, United States, 91942 Furiex Research Site Long Beach, California, United States, 90813 Furiex Research Site Oceanside, California, United States, 92056 Furiex Research Site Santa Ana, California, United States, 92701 United States, Florida Furiex Research Site Fort Myers, Florida, United States, 33912 Furiex Research Site Kissimmee, Florida, United States, 34741 Furiex Research Site St. Cloud, Florida, United States, 34769 United States, Georgia Furiex Research Site Columbus, Georgia, United States, 31904 Furiex Research Site Savannah, Georgia, United States, 31406 United States, Idaho Furiex Research Site Idaho Falls, Idaho, United States, 83404 United States, Illinois Furiex Research Site Libertyville, Illinois, United States, 60048 United States, Louisiana Furiex Research Site Baton Rouge, Louisiana, United States, 70809 Furiex Research Site New Orleans, Louisiana, United States, 70112 Furiex Research Site Sulphur, Louisiana, United States, 70663 United States, Michigan Furiex Research Site Detroit, Michigan, United States, 48202 Furiex Research Site Keego Harbor, Michigan, United States, 48320 United States, Montana Furiex Research Site Butte, Montana, United States, 59701 United States, Ohio Furiex Research Site Toledo, Ohio, United States, 43608 United States, Pennsylvania Furiex Research Site Philadelphia, Pennsylvania, United States, 19107 United States, Texas Furiex Research Site Houston, Texas, United States, 77002 Furiex Research Site Houston, Texas, United States, 77005 Furiex Research Site Sugar Land, Texas, United States, 77498 Furiex Research Site Webster, Texas, United States, 77598 Collapse << |
| NCT01170221 | - | - | Completed | - | - |
| NCT01561469 | - | - | Completed | - | - |
| NCT01561469 | - | - | Completed | - | - |
| NCT00666276 | - | - | Completed | - | - |
| NCT03308227 | - | - | Not yet recruiting | April 30, 2018 | - |
| NCT01544673 | Gram Positive Infection | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00691392 | Multi-Drug Resistant Tuberculo... more >>sis Extensively Drug Resistant Tuberculosis Collapse << | Phase 1 Phase 2 | Completed | - | South Africa ... more >> King George V Hospital Durban, KwaZulu Natal, South Africa Collapse << |
| NCT01521364 | Multidrug-resistant Tuberculos... more >>is Extensively Drug-resistant Tuberculosis Collapse << | Phase 4 | Completed | - | Netherlands ... more >> Tuberculosis Center Beatrixoord Haren, Groningen, Netherlands Collapse << |
| NCT00625703 | Cystic Fibrosis | Phase 2 | Unknown | July 2009 | United States, Texas ... more >> University of Texas Southwestern Medical Center at Dallas Recruiting Dallas, Texas, United States, 75390 Contact: Jane Siegel, MD 214-648-3720 jane.siegel@utsouthwestern.edu Contact: Chonnamet Techasaensiri, MD 214-648-3720 chonnamet.techasensiri@utsouthwestern.edu Sub-Investigator: Roberto Santos, MD Sub-Investigator: Claude Prestidge, MD Sub-Investigator: Michael E Brown, MD Sub-Investigator: Chonnamet Techasaensiri, MD Sub-Investigator: Pablo J. Sanchez, MD Sub-Investigator: Steven Copenhaver, MD Sub-Investigator: Andrew Gelfand, MD Collapse << |
| NCT02378480 | Bacterial Infections ... more >> Skin Structures and Soft Tissue Infections Collapse << | Phase 3 | Completed | - | - |
| NCT01521364 | - | - | Completed | - | - |
| NCT00666276 | - | - | Completed | - | - |
| NCT01619410 | Skin Diseases, Bacterial ... more >> Abscess Collapse << | Not Applicable | Completed | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 United States, Illinois University of Chicago Medical Center Chicago, Illinois, United States, 60637 Collapse << |
| NCT01055769 | Healthy Volunteers | Phase 1 | Completed | - | China ... more >> Pfizer Investigational Site Shanghai, China, 200040 Collapse << |
| NCT02877927 | Bacterial Infections ... more >> Skin Structures and Soft Tissue Infections Collapse << | Phase 3 | Completed | - | - |
| NCT02877927 | - | - | Completed | - | - |
| NCT03405064 | Acute Bacterial Skin and Skin ... more >>Structure Infections Collapse << | Phase 3 | Recruiting | November 2018 | India ... more >> GMERS Medical College & General Hospital Recruiting Vadodara, Gujarat, India, 390021 Contact: Bhavin Patel, MD 91-9898216643 gmers.trials@spearsmind.com Mysore Medical College and Research Institute Recruiting Mysore, Karnataka, India, 570001 Contact: Mohan Kakola, MD 91-9890318105 dryunus29@rediffmail.com Govt. Medical College Recruiting Kozhikode, Kerala, India, Kozhikode, Contact: Kozhikode, , MD 91-8589951229 researchatcmc@gmail.com TNMC & BYL Nair Hospital Recruiting Mumbai, Maharashtra, India, 400008 Contact: Jayashri Pandya, MD 91-9833774575 smruti63@hotmail.com G.S. Medical College & KEM Hospital Recruiting Mumbai, Maharashtra, India, 400012 Contact: Milind Prabhakar More, MD 91-9820541126 drmilindpmore@gmail.com B.J. Medical College and Sassoon General Hospital Recruiting Pune, Maharashtra, India, 411001, Contact: Lata Bhoir, MD 91-9764132444 drlatabhoir@gmail.com Oyster & Pearl Hospital Recruiting Pune, Maharashtra, India, 411005 Contact: Shekhar Takale, MD +91 9960444711 drshekhar7@gmail.com Principal Investigator: Shekhar Takale, MD KEM Hospital Research Center Not yet recruiting Pune, Maharashtra, India, 411011 Contact: Bharat Kalambe, MD 9822174858 bkalambe@hotmail.com Noble Hospital Not yet recruiting Pune, Maharashtra, India, 411013 Contact: Umesh Paprunia, MD 91-9860172720 umeshpaprunia@gmail.com Datta Meghe Institute of Medical Sciences (Deemed University) Jawaharlal Nehru Medical College Recruiting Wardha, Maharashtra, India, 442004 Contact: Chandrashekhar Mahakalkar, MD 91-9822369277 cmahakalkar@rediffmail.com M.V. Hospital & Research Centre Recruiting Lucknow, Uttar Pradesh, India, 226003 Contact: SandeepKumar Gupta, MD 8400388966 sandeepkumar.gupta@rediffmail.com Principal Investigator: SandeepKumar Gupta, MD Ajanta Research Centre Recruiting Lucknow, Uttar Pradesh, India, 2260055 Contact: Anil Khanna, MD +91 9335242509 dr_khanna@yahoo.com Principal Investigator: Anil Khanna, MD Om Surgical & Maternity Home Recruiting Varanasi, Uttar Pradesh, India, 22100 Contact: Pankaj Srivastava, MD 91-9415226817 omresearchcenter@gmail.com Collapse << |
| NCT02269319 | Bacterial Infections | Phase 2 | Completed | - | - |
| NCT01224626 | - | - | Completed | - | - |
| NCT01805284 | Pneumonia | Phase 4 | Unknown | December 2015 | Belgium ... more >> Ghent University Hospital Recruiting Ghent, Belgium, 9000 Contact: Stijn Blot, Ph.D, prof. Stijn.Blot@ugent.be Principal Investigator: Dirk Vogelaers, Ph.D., M.D. Collapse << |
| NCT03747497 | Skin Diseases, Bacterial ... more >> Gram-Positive Bacterial Infections Collapse << | Phase 2 | Not yet recruiting | June 30, 2019 | - |
| NCT03140410 | - | - | Active, not recruiting | January 2, 2018 | France ... more >> CHU Jean Minjoz Besançon, France, 25000 Collapse << |
| NCT01055769 | - | - | Completed | - | - |
| NCT03537495 | Tuberculosis, Meningeal ... more >> Linezolid Collapse << | Phase 2 | Not yet recruiting | July 2019 | Indonesia ... more >> Hasan Sadikin General Hospital Bandung, Jawa Barat, Indonesia, 40161 Collapse << |
| NCT01224626 | - | - | Completed | - | - |
| NCT02778828 | Tuberculosis | Not Applicable | Active, not recruiting | December 31, 2018 | France ... more >> Sanatorium du Petit Fontainebleau Centre Médical de Bligny Briis-sous-Forges, Essonne, France, 91640 Collapse << |
| NCT01200654 | Methicillin-Resistant Staphylo... more >>coccus AureuS Collapse << | Phase 4 | Completed | - | - |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
