CAS No.: 439081-18-2
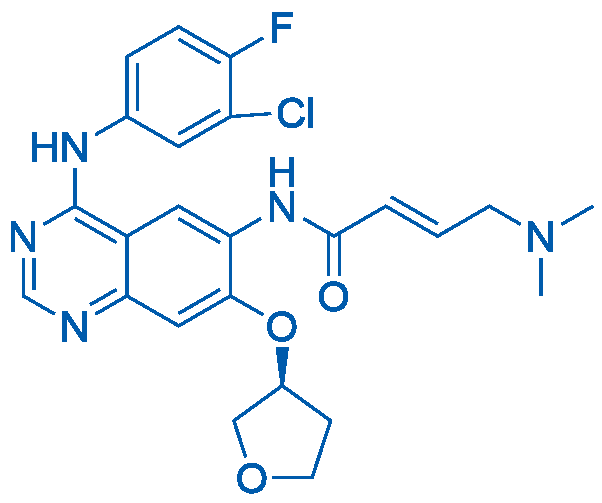
(S)-N-(4-((3-Chloro-4-fluorophenyl)amino)-7-((tetrahydrofuran-3-yl)oxy)quinazolin-6-yl)-4-(dimethylamino)but-2-enamide Catalog No. CSN12371
Synonyms: BIBW 2992;BIBW2992 mixture
Afatinib is an irreversible inhibitor of pan-ErbB inhibitor with IC50 values of 0.4nM, 0.5nM, 10nM, 14nM and 1nM for EGFR (L858R), EGFR (wt), EGFR (L858R/T790M), HER2 and ErbB4, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 EGFR (T790M, L858R)
IC50:10nMEGFR (L858R)
IC50:0.4nMEGFR
IC50:0.5nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice: 20 mg/kg[3] (p.o.)
给药途径 p.o.
动物药代数据 Animal Rats[4] Dose 4 mg/kg (i.v.)
8 mg/kg (p.o.)Administration i.v.
p.o.Cmax 1620 nmol/L (i.v.)
397 nmol/L (p.o.)T1/2 5.22 h (i.v.)
4.54 h (p.o.)AUC0→∞ 2920 nmol·h/L (i.v.)
2600 nmol·h/L (p.o.)CL/F 55.3 ml/min·kg (i.v.)
108 ml/min·kg (p.o.)Vss/F 16.2 L/kg (i.v.)
43.6 L/kg (p.o.)AUC0→24h 2500 nmol·h/L (i.v.)
2540 nmol·h/L (p.o.)Vz/F 25.0 L/kg (i.v.)
42.8 L/kg (p.o.)MRT 4.95 h (i.v.)
6.65 h (p.o.)- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02145637 NSCLC Phase 1 Unknown July 2016 - NCT01649284 - - - - - NCT03054038 Non-Small Cell Lung Carcinoma Phase 1 Recruiting March 2021 United States, California ... more >> City of Hope National Medical Center Recruiting Duarte, California, United States, 91010 Contact: Thomas Fok tfok@coh.org Principal Investigator: Karen Reckamp, MD Stanford Cancer Institute Recruiting Stanford, California, United States, 94035 Contact: Jordan Preiss 650-723-1002 Principal Investigator: Sukhmani Padda, MD United States, Tennessee Vanderbilt-Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << - 更多
- 参考文献
- [1] Li D, Ambrogio L, et al. BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene. 2008 Aug 7;27(34):4702-11.
- [2] Ferrarotto R, Gold KA, et al. Afatinib in the treatment of head and neck squamous cell carcinoma. Expert Opin Investig Drugs. 2014 Jan;23(1):135-43.
- [3] Hu X, Shi S, et al. Blocking autophagy improves the anti-tumor activity of afatinib in lung adenocarcinoma with activating EGFR mutations in vitro and in vivo. Sci Rep. 2017 Jul 4;7(1):4559.
- [4] Pharmacokinetics of afatinib
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 439081-18-2 | 储存条件 |
|
|||||||
| 分子式 | C24H25ClFN5O3 | 运输 | 蓝冰 | |||||||
| 分子量 | 485.94 | 别名 | BIBW 2992;BIBW2992 mixture | |||||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 22RV1 | - | Growth Inhibition Assay | - | IC50=34.1754 μM | SANGER |
| 23132-87 | - | Growth Inhibition Assay | - | IC50=0.31923 μM | SANGER |
| 5637 | - | Growth Inhibition Assay | - | IC50=1.0151 μM | SANGER |
| 639-V | - | Growth Inhibition Assay | - | IC50=31.5243 μM | SANGER |
| 647-V | - | Growth Inhibition Assay | - | IC50=3.98452 μM | SANGER |
| 697 | - | Growth Inhibition Assay | - | IC50=28.6331 μM | SANGER |
| 769-P | - | Growth Inhibition Assay | - | IC50=2.29037 μM | SANGER |
| 786-0 | - | Growth Inhibition Assay | - | IC50=3.06408 μM | SANGER |
| 8305C | - | Growth Inhibition Assay | - | IC50=3.75911 μM | SANGER |
| 8505C | - | Growth Inhibition Assay | - | IC50=13.2067 μM | SANGER |
| 8-MG-BA | - | Growth Inhibition Assay | - | IC50=36.9483 μM | SANGER |
| A101D | - | Growth Inhibition Assay | - | IC50=49.9425 μM | SANGER |
| A172 | - | Growth Inhibition Assay | - | IC50=12.0798 μM | SANGER |
| A204 | - | Growth Inhibition Assay | - | IC50=32.5069 μM | SANGER |
| A2058 | - | Growth Inhibition Assay | - | IC50=15.0409 μM | SANGER |
| A2780 | - | Growth Inhibition Assay | - | IC50=2.06294 μM | SANGER |
| A388 | - | Growth Inhibition Assay | - | IC50=0.36857 μM | SANGER |
| A3-KAW | - | Growth Inhibition Assay | - | IC50=23.1003 μM | SANGER |
| A427 | - | Growth Inhibition Assay | - | IC50=37.9107 μM | SANGER |
| A431 | - | Growth Inhibition Assay | - | IC50=0.48233 μM | SANGER |
| A498 | - | Growth Inhibition Assay | - | IC50=0.40674 μM | SANGER |
| A4-Fuk | - | Growth Inhibition Assay | - | IC50=22.1419 μM | SANGER |
| A549 | - | Growth Inhibition Assay | - | IC50=6.12553 μM | SANGER |
| A673 | - | Growth Inhibition Assay | - | IC50=41.5157 μM | SANGER |
| A704 | - | Growth Inhibition Assay | - | IC50=18.4753 μM | SANGER |
| ABC-1 | - | Growth Inhibition Assay | - | IC50=34.3693 μM | SANGER |
| ACHN | - | Growth Inhibition Assay | - | IC50=0.31994 μM | SANGER |
| ACN | - | Growth Inhibition Assay | - | IC50=0.55094 μM | SANGER |
| AGS | - | Growth Inhibition Assay | - | IC50=0.83648 μM | SANGER |
| ALL-PO | - | Growth Inhibition Assay | - | IC50=11.2512 μM | SANGER |
| AN3-CA | - | Growth Inhibition Assay | - | IC50=28.4395 μM | SANGER |
| AsPC-1 | - | Growth Inhibition Assay | - | IC50=19.6471 μM | SANGER |
| ATN-1 | - | Growth Inhibition Assay | - | IC50=33.1416 μM | SANGER |
| AU565 | - | Growth Inhibition Assay | - | IC50=0.08504 μM | SANGER |
| BALL-1 | - | Growth Inhibition Assay | - | IC50=9.21907 μM | SANGER |
| BB30-HNC | - | Growth Inhibition Assay | - | IC50=0.16576 μM | SANGER |
| BB49-HNC | - | Growth Inhibition Assay | - | IC50=43.2356 μM | SANGER |
| BB65-RCC | - | Growth Inhibition Assay | - | IC50=0.34593 μM | SANGER |
| BCPAP | - | Growth Inhibition Assay | - | IC50=27.137 μM | SANGER |
| BE-13 | - | Growth Inhibition Assay | - | IC50=7.98112 μM | SANGER |
| Becker | - | Growth Inhibition Assay | - | IC50=20.7201 μM | SANGER |
| BEN | - | Growth Inhibition Assay | - | IC50=27.8517 μM | SANGER |
| BFTC-905 | - | Growth Inhibition Assay | - | IC50=29.2705 μM | SANGER |
| BFTC-909 | - | Growth Inhibition Assay | - | IC50=9.06799 μM | SANGER |
| BHT-101 | - | Growth Inhibition Assay | - | IC50=14.241 μM | SANGER |
| BHY | - | Growth Inhibition Assay | - | IC50=0.28655 μM | SANGER |
| BPH-1 | - | Growth Inhibition Assay | - | IC50=0.05988 μM | SANGER |
| BT-20 | - | Growth Inhibition Assay | - | IC50=3.69795 μM | SANGER |
| BT-474 | - | Growth Inhibition Assay | - | IC50=0.04986 μM | SANGER |
| BT-549 | - | Growth Inhibition Assay | - | IC50=6.66889 μM | SANGER |
| BV-173 | - | Growth Inhibition Assay | - | IC50=0.17596 μM | SANGER |
| BxPC-3 | - | Growth Inhibition Assay | - | IC50=10.0972 μM | SANGER |
| C2BBe1 | - | Growth Inhibition Assay | - | IC50=14.764 μM | SANGER |
| C32 | - | Growth Inhibition Assay | - | IC50=9.33064 μM | SANGER |
| C-33-A | - | Growth Inhibition Assay | - | IC50=0.81297 μM | SANGER |
| C3A | - | Growth Inhibition Assay | - | IC50=30.2927 μM | SANGER |
| C8166 | - | Growth Inhibition Assay | - | IC50=33.8854 μM | SANGER |
| Ca9-22 | - | Growth Inhibition Assay | - | IC50=0.08353 μM | SANGER |
| CAKI-1 | - | Growth Inhibition Assay | - | IC50=1.78211 μM | SANGER |
| CAL-120 | - | Growth Inhibition Assay | - | IC50=15.1447 μM | SANGER |
| CAL-12T | - | Growth Inhibition Assay | - | IC50=1.88945 μM | SANGER |
| CAL-27 | - | Growth Inhibition Assay | - | IC50=0.17636 μM | SANGER |
| CAL-33 | - | Growth Inhibition Assay | - | IC50=0.20184 μM | SANGER |
| CAL-39 | - | Growth Inhibition Assay | - | IC50=0.11792 μM | SANGER |
| CAL-51 | - | Growth Inhibition Assay | - | IC50=31.5667 μM | SANGER |
| CAL-54 | - | Growth Inhibition Assay | - | IC50=0.61621 μM | SANGER |
| CAL-62 | - | Growth Inhibition Assay | - | IC50=43.4028 μM | SANGER |
| CAL-72 | - | Growth Inhibition Assay | - | IC50=10.9391 μM | SANGER |
| CAL-85-1 | - | Growth Inhibition Assay | - | IC50=9.87383 μM | SANGER |
| Calu-3 | - | Growth Inhibition Assay | - | IC50=0.81434 μM | SANGER |
| Calu-6 | - | Growth Inhibition Assay | - | IC50=30.2963 μM | SANGER |
| CAMA-1 | - | Growth Inhibition Assay | - | IC50=9.28128 μM | SANGER |
| CAPAN-1 | - | Growth Inhibition Assay | - | IC50=30.5181 μM | SANGER |
| Capan-2 | - | Growth Inhibition Assay | - | IC50=3.46875 μM | SANGER |
| CaR-1 | - | Growth Inhibition Assay | - | IC50=2.28281 μM | SANGER |
| CAS-1 | - | Growth Inhibition Assay | - | IC50=46.7701 μM | SANGER |
| Ca-Ski | - | Growth Inhibition Assay | - | IC50=7.64259 μM | SANGER |
| CCRF-CEM | - | Growth Inhibition Assay | - | IC50=25.967 μM | SANGER |
| CFPAC-1 | - | Growth Inhibition Assay | - | IC50=1.42493 μM | SANGER |
| CGTH-W-1 | - | Growth Inhibition Assay | - | IC50=42.4955 μM | SANGER |
| ChaGo-K-1 | - | Growth Inhibition Assay | - | IC50=32.007 μM | SANGER |
| CHL-1 | - | Growth Inhibition Assay | - | IC50=0.01839 μM | SANGER |
| CHP-212 | - | Growth Inhibition Assay | - | IC50=22.3222 μM | SANGER |
| COLO-320-HSR | - | Growth Inhibition Assay | - | IC50=18.746 μM | SANGER |
| COLO-678 | - | Growth Inhibition Assay | - | IC50=0.80834 μM | SANGER |
| COLO-679 | - | Growth Inhibition Assay | - | IC50=35.388 μM | SANGER |
| COLO-680N | - | Growth Inhibition Assay | - | IC50=44.339 μM | SANGER |
| COLO-684 | - | Growth Inhibition Assay | - | IC50=16.9477 μM | SANGER |
| COLO-741 | - | Growth Inhibition Assay | - | IC50=20.66 μM | SANGER |
| COLO-792 | - | Growth Inhibition Assay | - | IC50=25.2732 μM | SANGER |
| COLO-800 | - | Growth Inhibition Assay | - | IC50=7.41446 μM | SANGER |
| COLO-824 | - | Growth Inhibition Assay | - | IC50=15.1579 μM | SANGER |
| COLO-829 | - | Growth Inhibition Assay | - | IC50=8.80976 μM | SANGER |
| COR-L105 | - | Growth Inhibition Assay | - | IC50=0.21084 μM | SANGER |
| COR-L23 | - | Growth Inhibition Assay | - | IC50=24.1767 μM | SANGER |
| COR-L88 | - | Growth Inhibition Assay | - | IC50=0.64113 μM | SANGER |
| CP50-MEL-B | - | Growth Inhibition Assay | - | IC50=17.0343 μM | SANGER |
| CP66-MEL | - | Growth Inhibition Assay | - | IC50=15.4688 μM | SANGER |
| CTB-1 | - | Growth Inhibition Assay | - | IC50=4.8488 μM | SANGER |
| CTV-1 | - | Growth Inhibition Assay | - | IC50=0.64392 μM | SANGER |
| CW-2 | - | Growth Inhibition Assay | - | IC50=0.46914 μM | SANGER |
| D-263MG | - | Growth Inhibition Assay | - | IC50=33.8378 μM | SANGER |
| D-283MED | - | Growth Inhibition Assay | - | IC50=18.0088 μM | SANGER |
| D-336MG | - | Growth Inhibition Assay | - | IC50=30.2892 μM | SANGER |
| D-392MG | - | Growth Inhibition Assay | - | IC50=35.0565 μM | SANGER |
| D-423MG | - | Growth Inhibition Assay | - | IC50=5.61705 μM | SANGER |
| D-502MG | - | Growth Inhibition Assay | - | IC50=8.29282 μM | SANGER |
| D-542MG | - | Growth Inhibition Assay | - | IC50=1.01064 μM | SANGER |
| D-566MG | - | Growth Inhibition Assay | - | IC50=29.9393 μM | SANGER |
| Daoy | - | Growth Inhibition Assay | - | IC50=24.126 μM | SANGER |
| DB | - | Growth Inhibition Assay | - | IC50=7.61005 μM | SANGER |
| DBTRG-05MG | - | Growth Inhibition Assay | - | IC50=13.0632 μM | SANGER |
| DEL | - | Growth Inhibition Assay | - | IC50=10.3946 μM | SANGER |
| Detroit562 | - | Growth Inhibition Assay | - | IC50=1.70152 μM | SANGER |
| DJM-1 | - | Growth Inhibition Assay | - | IC50=6.92012 μM | SANGER |
| DK-MG | - | Growth Inhibition Assay | - | IC50=30.5456 μM | SANGER |
| DMS-114 | - | Growth Inhibition Assay | - | IC50=40.8374 μM | SANGER |
| DMS-273 | - | Growth Inhibition Assay | - | IC50=32.5322 μM | SANGER |
| DMS-79 | - | Growth Inhibition Assay | - | IC50=40.5889 μM | SANGER |
| DOHH-2 | - | Growth Inhibition Assay | - | IC50=20.5463 μM | SANGER |
| DOK | - | Growth Inhibition Assay | - | IC50=0.03194 μM | SANGER |
| DoTc2-4510 | - | Growth Inhibition Assay | - | IC50=0.32219 μM | SANGER |
| DSH1 | - | Growth Inhibition Assay | - | IC50=0.03314 μM | SANGER |
| DU-145 | - | Growth Inhibition Assay | - | IC50=0.44833 μM | SANGER |
| DU-4475 | - | Growth Inhibition Assay | - | IC50=1.40121 μM | SANGER |
| ECC10 | - | Growth Inhibition Assay | - | IC50=1.55369 μM | SANGER |
| EC-GI-10 | - | Growth Inhibition Assay | - | IC50=0.05912 μM | SANGER |
| EFM-19 | - | Growth Inhibition Assay | - | IC50=37.7967 μM | SANGER |
| EFO-21 | - | Growth Inhibition Assay | - | IC50=11.1045 μM | SANGER |
| EFO-27 | - | Growth Inhibition Assay | - | IC50=0.05182 μM | SANGER |
| EGI-1 | - | Growth Inhibition Assay | - | IC50=28.5928 μM | SANGER |
| EKVX | - | Growth Inhibition Assay | - | IC50=1.01064 μM | SANGER |
| EM-2 | - | Growth Inhibition Assay | - | IC50=14.8576 μM | SANGER |
| EoL-1-cell | - | Growth Inhibition Assay | - | IC50=0.26892 μM | SANGER |
| EPLC-272H | - | Growth Inhibition Assay | - | IC50=4.41533 μM | SANGER |
| ES1 | - | Growth Inhibition Assay | - | IC50=19.9213 μM | SANGER |
| ES3 | - | Growth Inhibition Assay | - | IC50=16.9134 μM | SANGER |
| ES4 | - | Growth Inhibition Assay | - | IC50=0.97836 μM | SANGER |
| ES5 | - | Growth Inhibition Assay | - | IC50=4.83723 μM | SANGER |
| ES7 | - | Growth Inhibition Assay | - | IC50=19.6292 μM | SANGER |
| ES8 | - | Growth Inhibition Assay | - | IC50=17.6087 μM | SANGER |
| ESS-1 | - | Growth Inhibition Assay | - | IC50=18.5205 μM | SANGER |
| ETK-1 | - | Growth Inhibition Assay | - | IC50=1.37456 μM | SANGER |
| EW-1 | - | Growth Inhibition Assay | - | IC50=45.9689 μM | SANGER |
| EW-11 | - | Growth Inhibition Assay | - | IC50=9.17653 μM | SANGER |
| EW-13 | - | Growth Inhibition Assay | - | IC50=14.0352 μM | SANGER |
| EW-16 | - | Growth Inhibition Assay | - | IC50=27.2947 μM | SANGER |
| EW-18 | - | Growth Inhibition Assay | - | IC50=0.68698 μM | SANGER |
| EW-22 | - | Growth Inhibition Assay | - | IC50=41.8697 μM | SANGER |
| EW-24 | - | Growth Inhibition Assay | - | IC50=21.3945 μM | SANGER |
| EW-3 | - | Growth Inhibition Assay | - | IC50=1.07883 μM | SANGER |
| FADU | - | Growth Inhibition Assay | - | IC50=0.54404 μM | SANGER |
| FTC-133 | - | Growth Inhibition Assay | - | IC50=11.1407 μM | SANGER |
| G-361 | - | Growth Inhibition Assay | - | IC50=26.1027 μM | SANGER |
| G-401 | - | Growth Inhibition Assay | - | IC50=1.67413 μM | SANGER |
| G-402 | - | Growth Inhibition Assay | - | IC50=35.9867 μM | SANGER |
| GAK | - | Growth Inhibition Assay | - | IC50=25.2963 μM | SANGER |
| GAMG | - | Growth Inhibition Assay | - | IC50=3.91314 μM | SANGER |
| GB-1 | - | Growth Inhibition Assay | - | IC50=25.4069 μM | SANGER |
| GCIY | - | Growth Inhibition Assay | - | IC50=1.08104 μM | SANGER |
| GCT | - | Growth Inhibition Assay | - | IC50=20.3026 μM | SANGER |
| GI-1 | - | Growth Inhibition Assay | - | IC50=22.7658 μM | SANGER |
| GI-ME-N | - | Growth Inhibition Assay | - | IC50=31.0571 μM | SANGER |
| GMS-10 | - | Growth Inhibition Assay | - | IC50=2.03517 μM | SANGER |
| GOTO | - | Growth Inhibition Assay | - | IC50=31.8506 μM | SANGER |
| GP5d | - | Growth Inhibition Assay | - | IC50=6.97394 μM | SANGER |
| GR-ST | - | Growth Inhibition Assay | - | IC50=28.6696 μM | SANGER |
| GT3TKB | - | Growth Inhibition Assay | - | IC50=8.67964 μM | SANGER |
| H9 | - | Growth Inhibition Assay | - | IC50=0.07132 μM | SANGER |
| HAL-01 | - | Growth Inhibition Assay | - | IC50=8.5191 μM | SANGER |
| HC-1 | - | Growth Inhibition Assay | - | IC50=5.51839 μM | SANGER |
| HCC1187 | - | Growth Inhibition Assay | - | IC50=20.6734 μM | SANGER |
| HCC1395 | - | Growth Inhibition Assay | - | IC50=14.9338 μM | SANGER |
| HCC1419 | - | Growth Inhibition Assay | - | IC50=0.36408 μM | SANGER |
| HCC1569 | - | Growth Inhibition Assay | - | IC50=2.58297 μM | SANGER |
| HCC1806 | - | Growth Inhibition Assay | - | IC50=0.69666 μM | SANGER |
| HCC1937 | - | Growth Inhibition Assay | - | IC50=12.7564 μM | SANGER |
| HCC1954 | - | Growth Inhibition Assay | - | IC50=11.0754 μM | SANGER |
| HCC2218 | - | Growth Inhibition Assay | - | IC50=0.00987 μM | SANGER |
| HCC2998 | - | Growth Inhibition Assay | - | IC50=5.52615 μM | SANGER |
| HCC38 | - | Growth Inhibition Assay | - | IC50=42.9618 μM | SANGER |
| HCC70 | - | Growth Inhibition Assay | - | IC50=0.57661 μM | SANGER |
| HCE-4 | - | Growth Inhibition Assay | - | IC50=9.21558 μM | SANGER |
| HCE-T | - | Growth Inhibition Assay | - | IC50=0.88428 μM | SANGER |
| HCT-116 | - | Growth Inhibition Assay | - | IC50=4.41007 μM | SANGER |
| HCT-15 | - | Growth Inhibition Assay | - | IC50=22.7441 μM | SANGER |
| HDLM-2 | - | Growth Inhibition Assay | - | IC50=0.00572 μM | SANGER |
| HD-MY-Z | - | Growth Inhibition Assay | - | IC50=48.786 μM | SANGER |
| HEC-1 | - | Growth Inhibition Assay | - | IC50=3.5895 μM | SANGER |
| HEL | - | Growth Inhibition Assay | - | IC50=7.71203 μM | SANGER |
| H-EMC-SS | - | Growth Inhibition Assay | - | IC50=32.9678 μM | SANGER |
| HGC-27 | - | Growth Inhibition Assay | - | IC50=31.5873 μM | SANGER |
| HH | - | Growth Inhibition Assay | - | IC50=16.5162 μM | SANGER |
| HL-60 | - | Growth Inhibition Assay | - | IC50=4.86611 μM | SANGER |
| HLE | - | Growth Inhibition Assay | - | IC50=0.35046 μM | SANGER |
| HN | - | Growth Inhibition Assay | - | IC50=0.13077 μM | SANGER |
| HO-1-N-1 | - | Growth Inhibition Assay | - | IC50=0.27853 μM | SANGER |
| HOP-62 | - | Growth Inhibition Assay | - | IC50=11.6764 μM | SANGER |
| HOP-92 | - | Growth Inhibition Assay | - | IC50=26.1226 μM | SANGER |
| HOS | - | Growth Inhibition Assay | - | IC50=39.6273 μM | SANGER |
| HPAF-II | - | Growth Inhibition Assay | - | IC50=41.6709 μM | SANGER |
| Hs-578-T | - | Growth Inhibition Assay | - | IC50=45.0294 μM | SANGER |
| HSC-2 | - | Growth Inhibition Assay | - | IC50=0.16837 μM | SANGER |
| HSC-3 | - | Growth Inhibition Assay | - | IC50=0.21198 μM | SANGER |
| HSC-4 | - | Growth Inhibition Assay | - | IC50=0.01748 μM | SANGER |
| HT | - | Growth Inhibition Assay | - | IC50=40.6639 μM | SANGER |
| HT-1197 | - | Growth Inhibition Assay | - | IC50=4.52669 μM | SANGER |
| HT-1376 | - | Growth Inhibition Assay | - | IC50=1.01513 μM | SANGER |
| HT-144 | - | Growth Inhibition Assay | - | IC50=27.4032 μM | SANGER |
| HT-29 | - | Growth Inhibition Assay | - | IC50=3.07309 μM | SANGER |
| HT-3 | - | Growth Inhibition Assay | - | IC50=1.28753 μM | SANGER |
| HT55 | - | Growth Inhibition Assay | - | IC50=2.5738 μM | SANGER |
| HTC-C3 | - | Growth Inhibition Assay | - | IC50=27.2561 μM | SANGER |
| HuCCT1 | - | Growth Inhibition Assay | - | IC50=10.6033 μM | SANGER |
| HuH-7 | - | Growth Inhibition Assay | - | IC50=28.4028 μM | SANGER |
| HuO-3N1 | - | Growth Inhibition Assay | - | IC50=5.24043 μM | SANGER |
| HuO9 | - | Growth Inhibition Assay | - | IC50=11.8774 μM | SANGER |
| HuP-T3 | - | Growth Inhibition Assay | - | IC50=16.6576 μM | SANGER |
| HuP-T4 | - | Growth Inhibition Assay | - | IC50=0.6976 μM | SANGER |
| HUTU-80 | - | Growth Inhibition Assay | - | IC50=41.0569 μM | SANGER |
| IGR-1 | - | Growth Inhibition Assay | - | IC50=28.8874 μM | SANGER |
| IGROV-1 | - | Growth Inhibition Assay | - | IC50=0.25952 μM | SANGER |
| IST-MEL1 | - | Growth Inhibition Assay | - | IC50=11.4559 μM | SANGER |
| IST-MES1 | - | Growth Inhibition Assay | - | IC50=18.4428 μM | SANGER |
| IST-SL1 | - | Growth Inhibition Assay | - | IC50=26.6591 μM | SANGER |
| J82 | - | Growth Inhibition Assay | - | IC50=15.057 μM | SANGER |
| JAR | - | Growth Inhibition Assay | - | IC50=19.299 μM | SANGER |
| JEG-3 | - | Growth Inhibition Assay | - | IC50=22.378 μM | SANGER |
| J-RT3-T3-5 | - | Growth Inhibition Assay | - | IC50=6.869 μM | SANGER |
| JVM-2 | - | Growth Inhibition Assay | - | IC50=0.88299 μM | SANGER |
| JVM-3 | - | Growth Inhibition Assay | - | IC50=7.89599 μM | SANGER |
| K5 | - | Growth Inhibition Assay | - | IC50=1.58357 μM | SANGER |
| KALS-1 | - | Growth Inhibition Assay | - | IC50=23.1414 μM | SANGER |
| KARPAS-299 | - | Growth Inhibition Assay | - | IC50=20.3422 μM | SANGER |
| KARPAS-45 | - | Growth Inhibition Assay | - | IC50=0.01671 μM | SANGER |
| KASUMI-1 | - | Growth Inhibition Assay | - | IC50=0.00911 μM | SANGER |
| KE-37 | - | Growth Inhibition Assay | - | IC50=25.7315 μM | SANGER |
| KG-1 | - | Growth Inhibition Assay | - | IC50=24.1928 μM | SANGER |
| KGN | - | Growth Inhibition Assay | - | IC50=32.633 μM | SANGER |
| KINGS-1 | - | Growth Inhibition Assay | - | IC50=5.60944 μM | SANGER |
| KLE | - | Growth Inhibition Assay | - | IC50=4.40819 μM | SANGER |
| KM12 | - | Growth Inhibition Assay | - | IC50=22.356 μM | SANGER |
| KM-H2 | - | Growth Inhibition Assay | - | IC50=34.1779 μM | SANGER |
| KMOE-2 | - | Growth Inhibition Assay | - | IC50=19.2242 μM | SANGER |
| KNS-42 | - | Growth Inhibition Assay | - | IC50=35.5826 μM | SANGER |
| KNS-62 | - | Growth Inhibition Assay | - | IC50=3.94731 μM | SANGER |
| KNS-81-FD | - | Growth Inhibition Assay | - | IC50=19.5468 μM | SANGER |
| KOSC-2 | - | Growth Inhibition Assay | - | IC50=3.83451 μM | SANGER |
| KP-4 | - | Growth Inhibition Assay | - | IC50=42.8832 μM | SANGER |
| KP-N-YN | - | Growth Inhibition Assay | - | IC50=37.6195 μM | SANGER |
| KP-N-YS | - | Growth Inhibition Assay | - | IC50=40.6768 μM | SANGER |
| KS-1 | - | Growth Inhibition Assay | - | IC50=4.50889 μM | SANGER |
| KU-19-19 | - | Growth Inhibition Assay | - | IC50=0.77464 μM | SANGER |
| KU812 | - | Growth Inhibition Assay | - | IC50=0.15456 μM | SANGER |
| KURAMOCHI | - | Growth Inhibition Assay | - | IC50=7.94811 μM | SANGER |
| KY821 | - | Growth Inhibition Assay | - | IC50=0.0206 μM | SANGER |
| KYSE-140 | - | Growth Inhibition Assay | - | IC50=0.0833 μM | SANGER |
| KYSE-150 | - | Growth Inhibition Assay | - | IC50=42.1782 μM | SANGER |
| KYSE-180 | - | Growth Inhibition Assay | - | IC50=0.39089 μM | SANGER |
| KYSE-270 | - | Growth Inhibition Assay | - | IC50=2.02732 μM | SANGER |
| KYSE-410 | - | Growth Inhibition Assay | - | IC50=1.65672 μM | SANGER |
| KYSE-450 | - | Growth Inhibition Assay | - | IC50=0.05978 μM | SANGER |
| KYSE-510 | - | Growth Inhibition Assay | - | IC50=19.8748 μM | SANGER |
| KYSE-520 | - | Growth Inhibition Assay | - | IC50=5.20839 μM | SANGER |
| KYSE-70 | - | Growth Inhibition Assay | - | IC50=30.5114 μM | SANGER |
| L-363 | - | Growth Inhibition Assay | - | IC50=16.4577 μM | SANGER |
| L-428 | - | Growth Inhibition Assay | - | IC50=24.5791 μM | SANGER |
| LAMA-84 | - | Growth Inhibition Assay | - | IC50=4.1702 μM | SANGER |
| LAN-6 | - | Growth Inhibition Assay | - | IC50=40.0112 μM | SANGER |
| LB1047-RCC | - | Growth Inhibition Assay | - | IC50=2.74888 μM | SANGER |
| LB2241-RCC | - | Growth Inhibition Assay | - | IC50=0.10884 μM | SANGER |
| LB2518-MEL | - | Growth Inhibition Assay | - | IC50=49.8723 μM | SANGER |
| LB373-MEL-D | - | Growth Inhibition Assay | - | IC50=7.76771 μM | SANGER |
| LB771-HNC | - | Growth Inhibition Assay | - | IC50=35.1658 μM | SANGER |
| LB831-BLC | - | Growth Inhibition Assay | - | IC50=38.6039 μM | SANGER |
| LC-2-ad | - | Growth Inhibition Assay | - | IC50=21.2729 μM | SANGER |
| LCLC-103H | - | Growth Inhibition Assay | - | IC50=36.7803 μM | SANGER |
| LCLC-97TM1 | - | Growth Inhibition Assay | - | IC50=4.53686 μM | SANGER |
| LK-2 | - | Growth Inhibition Assay | - | IC50=13.7639 μM | SANGER |
| LN-405 | - | Growth Inhibition Assay | - | IC50=22.5913 μM | SANGER |
| LNCaP-Clone-FGC | - | Growth Inhibition Assay | - | IC50=15.4248 μM | SANGER |
| LoVo | - | Growth Inhibition Assay | - | IC50=2.10797 μM | SANGER |
| LOXIMVI | - | Growth Inhibition Assay | - | IC50=5.63873 μM | SANGER |
| LS-1034 | - | Growth Inhibition Assay | - | IC50=6.20706 μM | SANGER |
| LS-123 | - | Growth Inhibition Assay | - | IC50=3.74284 μM | SANGER |
| LS-411N | - | Growth Inhibition Assay | - | IC50=5.79441 μM | SANGER |
| LS-513 | - | Growth Inhibition Assay | - | IC50=4.31217 μM | SANGER |
| LU-134-A | - | Growth Inhibition Assay | - | IC50=21.1026 μM | SANGER |
| LU-135 | - | Growth Inhibition Assay | - | IC50=33.8829 μM | SANGER |
| LU-139 | - | Growth Inhibition Assay | - | IC50=0.3051 μM | SANGER |
| LU-65 | - | Growth Inhibition Assay | - | IC50=0.12633 μM | SANGER |
| LU-99A | - | Growth Inhibition Assay | - | IC50=37.5731 μM | SANGER |
| LXF-289 | - | Growth Inhibition Assay | - | IC50=2.63565 μM | SANGER |
| M14 | - | Growth Inhibition Assay | - | IC50=44.1332 μM | SANGER |
| MC116 | - | Growth Inhibition Assay | - | IC50=3.95489 μM | SANGER |
| MCF7 | - | Growth Inhibition Assay | - | IC50=16.4723 μM | SANGER |
| MC-IXC | - | Growth Inhibition Assay | - | IC50=11.6728 μM | SANGER |
| MDA-MB-157 | - | Growth Inhibition Assay | - | IC50=2.21828 μM | SANGER |
| MDA-MB-175-VII | - | Growth Inhibition Assay | - | IC50=0.03416 μM | SANGER |
| MDA-MB-231 | - | Growth Inhibition Assay | - | IC50=28.2795 μM | SANGER |
| MDA-MB-361 | - | Growth Inhibition Assay | - | IC50=0.72662 μM | SANGER |
| MDA-MB-415 | - | Growth Inhibition Assay | - | IC50=23.9499 μM | SANGER |
| MDA-MB-453 | - | Growth Inhibition Assay | - | IC50=11.9515 μM | SANGER |
| ME-180 | - | Growth Inhibition Assay | - | IC50=1.61096 μM | SANGER |
| MEG-01 | - | Growth Inhibition Assay | - | IC50=4.99403 μM | SANGER |
| MEL-HO | - | Growth Inhibition Assay | - | IC50=30.1423 μM | SANGER |
| Mewo | - | Growth Inhibition Assay | - | IC50=34.1349 μM | SANGER |
| MFE-280 | - | Growth Inhibition Assay | - | IC50=16.0488 μM | SANGER |
| MFH-ino | - | Growth Inhibition Assay | - | IC50=37.3371 μM | SANGER |
| MFM-223 | - | Growth Inhibition Assay | - | IC50=16.3965 μM | SANGER |
| MG-63 | - | Growth Inhibition Assay | - | IC50=5.48762 μM | SANGER |
| MHH-ES-1 | - | Growth Inhibition Assay | - | IC50=27.4447 μM | SANGER |
| MHH-NB-11 | - | Growth Inhibition Assay | - | IC50=16.0839 μM | SANGER |
| MHH-PREB-1 | - | Growth Inhibition Assay | - | IC50=10.1999 μM | SANGER |
| MIA-PaCa-2 | - | Growth Inhibition Assay | - | IC50=28.7959 μM | SANGER |
| MKN1 | - | Growth Inhibition Assay | - | IC50=3.47108 μM | SANGER |
| MKN28 | - | Growth Inhibition Assay | - | IC50=32.7838 μM | SANGER |
| MKN45 | - | Growth Inhibition Assay | - | IC50=21.1551 μM | SANGER |
| MKN7 | - | Growth Inhibition Assay | - | IC50=2.35335 μM | SANGER |
| ML-2 | - | Growth Inhibition Assay | - | IC50=0.24802 μM | SANGER |
| MLMA | - | Growth Inhibition Assay | - | IC50=22.747 μM | SANGER |
| MMAC-SF | - | Growth Inhibition Assay | - | IC50=19.2607 μM | SANGER |
| MN-60 | - | Growth Inhibition Assay | - | IC50=7.28089 μM | SANGER |
| MOLT-13 | - | Growth Inhibition Assay | - | IC50=0.23975 μM | SANGER |
| MOLT-16 | - | Growth Inhibition Assay | - | IC50=17.8934 μM | SANGER |
| MOLT-4 | - | Growth Inhibition Assay | - | IC50=7.02135 μM | SANGER |
| Mo-T | - | Growth Inhibition Assay | - | IC50=18.8204 μM | SANGER |
| MPP-89 | - | Growth Inhibition Assay | - | IC50=22.8661 μM | SANGER |
| MS-1 | - | Growth Inhibition Assay | - | IC50=30.8889 μM | SANGER |
| MSTO-211H | - | Growth Inhibition Assay | - | IC50=45.9482 μM | SANGER |
| MV-4-11 | - | Growth Inhibition Assay | - | IC50=3.87994 μM | SANGER |
| MZ1-PC | - | Growth Inhibition Assay | - | IC50=30.393 μM | SANGER |
| MZ2-MEL | - | Growth Inhibition Assay | - | IC50=16.8403 μM | SANGER |
| MZ7-mel | - | Growth Inhibition Assay | - | IC50=34.6487 μM | SANGER |
| NB10 | - | Growth Inhibition Assay | - | IC50=22.2371 μM | SANGER |
| NB12 | - | Growth Inhibition Assay | - | IC50=25.2947 μM | SANGER |
| NB13 | - | Growth Inhibition Assay | - | IC50=42.6217 μM | SANGER |
| NB14 | - | Growth Inhibition Assay | - | IC50=6.43836 μM | SANGER |
| NB17 | - | Growth Inhibition Assay | - | IC50=31.5185 μM | SANGER |
| NB5 | - | Growth Inhibition Assay | - | IC50=1.41317 μM | SANGER |
| NB69 | - | Growth Inhibition Assay | - | IC50=1.02945 μM | SANGER |
| NB7 | - | Growth Inhibition Assay | - | IC50=2.29676 μM | SANGER |
| NBsusSR | - | Growth Inhibition Assay | - | IC50=3.18254 μM | SANGER |
| NCI-H1048 | - | Growth Inhibition Assay | - | IC50=2.49158 μM | SANGER |
| NCI-H1092 | - | Growth Inhibition Assay | - | IC50=16.8316 μM | SANGER |
| NCI-H1155 | - | Growth Inhibition Assay | - | IC50=19.7238 μM | SANGER |
| NCI-H1299 | - | Growth Inhibition Assay | - | IC50=6.63171 μM | SANGER |
| NCI-H1304 | - | Growth Inhibition Assay | - | IC50=26.9108 μM | SANGER |
| NCI-H1355 | - | Growth Inhibition Assay | - | IC50=34.3889 μM | SANGER |
| NCI-H1395 | - | Growth Inhibition Assay | - | IC50=20.6697 μM | SANGER |
| NCI-H1437 | - | Growth Inhibition Assay | - | IC50=18.1641 μM | SANGER |
| NCI-H1563 | - | Growth Inhibition Assay | - | IC50=9.76031 μM | SANGER |
| NCI-H1573 | - | Growth Inhibition Assay | - | IC50=0.7363 μM | SANGER |
| NCI-H1581 | - | Growth Inhibition Assay | - | IC50=29.3145 μM | SANGER |
| NCI-H1623 | - | Growth Inhibition Assay | - | IC50=0.08025 μM | SANGER |
| NCI-H1648 | - | Growth Inhibition Assay | - | IC50=0.44182 μM | SANGER |
| NCI-H1650 | - | Growth Inhibition Assay | - | IC50=9.60946 μM | SANGER |
| NCI-H1651 | - | Growth Inhibition Assay | - | IC50=25.597 μM | SANGER |
| NCI-H1666 | - | Growth Inhibition Assay | - | IC50=1.80948 μM | SANGER |
| NCI-H1693 | - | Growth Inhibition Assay | - | IC50=3.84181 μM | SANGER |
| NCI-H1703 | - | Growth Inhibition Assay | - | IC50=9.34375 μM | SANGER |
| NCI-H1755 | - | Growth Inhibition Assay | - | IC50=41.8725 μM | SANGER |
| NCI-H1770 | - | Growth Inhibition Assay | - | IC50=0.1028 μM | SANGER |
| NCI-H1793 | - | Growth Inhibition Assay | - | IC50=14.2087 μM | SANGER |
| NCI-H1838 | - | Growth Inhibition Assay | - | IC50=0.60299 μM | SANGER |
| NCI-H1975 | - | Growth Inhibition Assay | - | IC50=0.61728 μM | SANGER |
| NCI-H1993 | - | Growth Inhibition Assay | - | IC50=8.33631 μM | SANGER |
| NCI-H2009 | - | Growth Inhibition Assay | - | IC50=3.61216 μM | SANGER |
| NCI-H2029 | - | Growth Inhibition Assay | - | IC50=43.5581 μM | SANGER |
| NCI-H2030 | - | Growth Inhibition Assay | - | IC50=14.6448 μM | SANGER |
| NCI-H2052 | - | Growth Inhibition Assay | - | IC50=21.8963 μM | SANGER |
| NCI-H2087 | - | Growth Inhibition Assay | - | IC50=49.9682 μM | SANGER |
| NCI-H209 | - | Growth Inhibition Assay | - | IC50=0.2046 μM | SANGER |
| NCI-H2122 | - | Growth Inhibition Assay | - | IC50=34.3471 μM | SANGER |
| NCI-H2126 | - | Growth Inhibition Assay | - | IC50=0.51926 μM | SANGER |
| NCI-H2170 | - | Growth Inhibition Assay | - | IC50=0.03728 μM | SANGER |
| NCI-H2228 | - | Growth Inhibition Assay | - | IC50=32.6602 μM | SANGER |
| NCI-H226 | - | Growth Inhibition Assay | - | IC50=49.7785 μM | SANGER |
| NCI-H2291 | - | Growth Inhibition Assay | - | IC50=5.98943 μM | SANGER |
| NCI-H23 | - | Growth Inhibition Assay | - | IC50=37.4196 μM | SANGER |
| NCI-H2342 | - | Growth Inhibition Assay | - | IC50=17.557 μM | SANGER |
| NCI-H2405 | - | Growth Inhibition Assay | - | IC50=17.1974 μM | SANGER |
| NCI-H28 | - | Growth Inhibition Assay | - | IC50=7.16405 μM | SANGER |
| NCI-H292 | - | Growth Inhibition Assay | - | IC50=0.0609 μM | SANGER |
| NCI-H358 | - | Growth Inhibition Assay | - | IC50=0.5321 μM | SANGER |
| NCI-H441 | - | Growth Inhibition Assay | - | IC50=3.59841 μM | SANGER |
| NCI-H446 | - | Growth Inhibition Assay | - | IC50=37.4862 μM | SANGER |
| NCI-H460 | - | Growth Inhibition Assay | - | IC50=46.523 μM | SANGER |
| NCI-H510A | - | Growth Inhibition Assay | - | IC50=31.4006 μM | SANGER |
| NCI-H520 | - | Growth Inhibition Assay | - | IC50=24.8861 μM | SANGER |
| NCI-H526 | - | Growth Inhibition Assay | - | IC50=0.07823 μM | SANGER |
| NCI-H596 | - | Growth Inhibition Assay | - | IC50=12.0369 μM | SANGER |
| NCI-H630 | - | Growth Inhibition Assay | - | IC50=1.45582 μM | SANGER |
| NCI-H650 | - | Growth Inhibition Assay | - | IC50=11.2418 μM | SANGER |
| NCI-H661 | - | Growth Inhibition Assay | - | IC50=16.9694 μM | SANGER |
| NCI-H69 | - | Growth Inhibition Assay | - | IC50=0.21451 μM | SANGER |
| NCI-H720 | - | Growth Inhibition Assay | - | IC50=0.05325 μM | SANGER |
| NCI-H727 | - | Growth Inhibition Assay | - | IC50=8.1042 μM | SANGER |
| NCI-H747 | - | Growth Inhibition Assay | - | IC50=5.42472 μM | SANGER |
| NCI-H810 | - | Growth Inhibition Assay | - | IC50=34.7278 μM | SANGER |
| NCI-H82 | - | Growth Inhibition Assay | - | IC50=1.54006 μM | SANGER |
| NCI-N87 | - | Growth Inhibition Assay | - | IC50=0.02773 μM | SANGER |
| NCI-SNU-1 | - | Growth Inhibition Assay | - | IC50=3.69735 μM | SANGER |
| NCI-SNU-5 | - | Growth Inhibition Assay | - | IC50=1.239 μM | SANGER |
| NEC8 | - | Growth Inhibition Assay | - | IC50=14.5472 μM | SANGER |
| NH-12 | - | Growth Inhibition Assay | - | IC50=7.72808 μM | SANGER |
| NKM-1 | - | Growth Inhibition Assay | - | IC50=3.18744 μM | SANGER |
| NMC-G1 | - | Growth Inhibition Assay | - | IC50=12.9234 μM | SANGER |
| no-10 | - | Growth Inhibition Assay | - | IC50=16.6343 μM | SANGER |
| no-11 | - | Growth Inhibition Assay | - | IC50=3.53564 μM | SANGER |
| NOMO-1 | - | Growth Inhibition Assay | - | IC50=34.5703 μM | SANGER |
| NOS-1 | - | Growth Inhibition Assay | - | IC50=11.7109 μM | SANGER |
| NTERA-S-cl-D1 | - | Growth Inhibition Assay | - | IC50=5.54537 μM | SANGER |
| NUGC-3 | - | Growth Inhibition Assay | - | IC50=0.38373 μM | SANGER |
| NY | - | Growth Inhibition Assay | - | IC50=3.53655 μM | SANGER |
| OAW-28 | - | Growth Inhibition Assay | - | IC50=7.94578 μM | SANGER |
| OAW-42 | - | Growth Inhibition Assay | - | IC50=3.50026 μM | SANGER |
| OC-314 | - | Growth Inhibition Assay | - | IC50=15.4714 μM | SANGER |
| OCI-AML2 | - | Growth Inhibition Assay | - | IC50=8.95481 μM | SANGER |
| OCUB-M | - | Growth Inhibition Assay | - | IC50=3.76187 μM | SANGER |
| OE19 | - | Growth Inhibition Assay | - | IC50=1.71512 μM | SANGER |
| OE33 | - | Growth Inhibition Assay | - | IC50=13.786 μM | SANGER |
| OMC-1 | - | Growth Inhibition Assay | - | IC50=2.08169 μM | SANGER |
| ONS-76 | - | Growth Inhibition Assay | - | IC50=41.4709 μM | SANGER |
| OS-RC-2 | - | Growth Inhibition Assay | - | IC50=9.0219 μM | SANGER |
| OVCAR-3 | - | Growth Inhibition Assay | - | IC50=2.67167 μM | SANGER |
| OVCAR-4 | - | Growth Inhibition Assay | - | IC50=10.8835 μM | SANGER |
| OVCAR-5 | - | Growth Inhibition Assay | - | IC50=13.1593 μM | SANGER |
| OVCAR-8 | - | Growth Inhibition Assay | - | IC50=15.8861 μM | SANGER |
| P12-ICHIKAWA | - | Growth Inhibition Assay | - | IC50=25.4785 μM | SANGER |
| P30-OHK | - | Growth Inhibition Assay | - | IC50=35.8752 μM | SANGER |
| PA-1 | - | Growth Inhibition Assay | - | IC50=49.8335 μM | SANGER |
| PANC-03-27 | - | Growth Inhibition Assay | - | IC50=2.16631 μM | SANGER |
| PANC-08-13 | - | Growth Inhibition Assay | - | IC50=1.16416 μM | SANGER |
| PANC-10-05 | - | Growth Inhibition Assay | - | IC50=14.6247 μM | SANGER |
| PC-14 | - | Growth Inhibition Assay | - | IC50=0.05668 μM | SANGER |
| PC-3 | - | Growth Inhibition Assay | - | IC50=19.9604 μM | SANGER |
| PFSK-1 | - | Growth Inhibition Assay | - | IC50=13.06 μM | SANGER |
| QIMR-WIL | - | Growth Inhibition Assay | - | IC50=16.7105 μM | SANGER |
| Ramos-2G6-4C10 | - | Growth Inhibition Assay | - | IC50=14.0202 μM | SANGER |
| RCC10RGB | - | Growth Inhibition Assay | - | IC50=18.0559 μM | SANGER |
| RCM-1 | - | Growth Inhibition Assay | - | IC50=2.52938 μM | SANGER |
| RD | - | Growth Inhibition Assay | - | IC50=29.5233 μM | SANGER |
| RERF-LC-MS | - | Growth Inhibition Assay | - | IC50=17.5508 μM | SANGER |
| RH-18 | - | Growth Inhibition Assay | - | IC50=18.0035 μM | SANGER |
| RKO | - | Growth Inhibition Assay | - | IC50=14.5075 μM | SANGER |
| RMG-I | - | Growth Inhibition Assay | - | IC50=7.39296 μM | SANGER |
| RO82-W-1 | - | Growth Inhibition Assay | - | IC50=32.1604 μM | SANGER |
| RPMI-2650 | - | Growth Inhibition Assay | - | IC50=11.5885 μM | SANGER |
| RPMI-7951 | - | Growth Inhibition Assay | - | IC50=3.0759 μM | SANGER |
| RPMI-8226 | - | Growth Inhibition Assay | - | IC50=4.11631 μM | SANGER |
| RPMI-8866 | - | Growth Inhibition Assay | - | IC50=27.7562 μM | SANGER |
| RS4-11 | - | Growth Inhibition Assay | - | IC50=12.1725 μM | SANGER |
| RT-112 | - | Growth Inhibition Assay | - | IC50=22.8616 μM | SANGER |
| RVH-421 | - | Growth Inhibition Assay | - | IC50=35.7645 μM | SANGER |
| RXF393 | - | Growth Inhibition Assay | - | IC50=36.3785 μM | SANGER |
| S-117 | - | Growth Inhibition Assay | - | IC50=36.3008 μM | SANGER |
| Saos-2 | - | Growth Inhibition Assay | - | IC50=19.1162 μM | SANGER |
| SAS | - | Growth Inhibition Assay | - | IC50=0.48398 μM | SANGER |
| SBC-1 | - | Growth Inhibition Assay | - | IC50=0.18959 μM | SANGER |
| SCC-15 | - | Growth Inhibition Assay | - | IC50=1.39451 μM | SANGER |
| SCC-25 | - | Growth Inhibition Assay | - | IC50=0.84171 μM | SANGER |
| SCC-4 | - | Growth Inhibition Assay | - | IC50=8.44059 μM | SANGER |
| SCC-9 | - | Growth Inhibition Assay | - | IC50=37.7838 μM | SANGER |
| SCH | - | Growth Inhibition Assay | - | IC50=18.6456 μM | SANGER |
| SF126 | - | Growth Inhibition Assay | - | IC50=23.6924 μM | SANGER |
| SF268 | - | Growth Inhibition Assay | - | IC50=29.5931 μM | SANGER |
| SF295 | - | Growth Inhibition Assay | - | IC50=26.0425 μM | SANGER |
| SF539 | - | Growth Inhibition Assay | - | IC50=9.23562 μM | SANGER |
| SH-4 | - | Growth Inhibition Assay | - | IC50=32.1858 μM | SANGER |
| SHP-77 | - | Growth Inhibition Assay | - | IC50=29.6182 μM | SANGER |
| SiHa | - | Growth Inhibition Assay | - | IC50=23.9327 μM | SANGER |
| SJRH30 | - | Growth Inhibition Assay | - | IC50=30.2108 μM | SANGER |
| SJSA-1 | - | Growth Inhibition Assay | - | IC50=27.0176 μM | SANGER |
| SKG-IIIa | - | Growth Inhibition Assay | - | IC50=3.52237 μM | SANGER |
| SK-HEP-1 | - | Growth Inhibition Assay | - | IC50=30.6326 μM | SANGER |
| SK-LU-1 | - | Growth Inhibition Assay | - | IC50=40.6568 μM | SANGER |
| SK-MEL-1 | - | Growth Inhibition Assay | - | IC50=13.7135 μM | SANGER |
| SK-MEL-2 | - | Growth Inhibition Assay | - | IC50=32.6527 μM | SANGER |
| SK-MEL-24 | - | Growth Inhibition Assay | - | IC50=11.2366 μM | SANGER |
| SK-MEL-28 | - | Growth Inhibition Assay | - | IC50=19.3363 μM | SANGER |
| SK-MEL-3 | - | Growth Inhibition Assay | - | IC50=16.4787 μM | SANGER |
| SK-MES-1 | - | Growth Inhibition Assay | - | IC50=1.11487 μM | SANGER |
| SK-N-AS | - | Growth Inhibition Assay | - | IC50=21.8276 μM | SANGER |
| SK-N-DZ | - | Growth Inhibition Assay | - | IC50=28.6885 μM | SANGER |
| SK-NEP-1 | - | Growth Inhibition Assay | - | IC50=1.17513 μM | SANGER |
| SK-N-FI | - | Growth Inhibition Assay | - | IC50=28.433 μM | SANGER |
| SK-OV-3 | - | Growth Inhibition Assay | - | IC50=8.71329 μM | SANGER |
| SK-PN-DW | - | Growth Inhibition Assay | - | IC50=35.4369 μM | SANGER |
| SK-UT-1 | - | Growth Inhibition Assay | - | IC50=13.1083 μM | SANGER |
| SN12C | - | Growth Inhibition Assay | - | IC50=2.91964 μM | SANGER |
| SNU-387 | - | Growth Inhibition Assay | - | IC50=40.4553 μM | SANGER |
| SNU-449 | - | Growth Inhibition Assay | - | IC50=3.65826 μM | SANGER |
| SNU-C2B | - | Growth Inhibition Assay | - | IC50=2.68616 μM | SANGER |
| SW1116 | - | Growth Inhibition Assay | - | IC50=1.01287 μM | SANGER |
| SW13 | - | Growth Inhibition Assay | - | IC50=0.16216 μM | SANGER |
| SW1417 | - | Growth Inhibition Assay | - | IC50=4.12147 μM | SANGER |
| SW1463 | - | Growth Inhibition Assay | - | IC50=15.0485 μM | SANGER |
| SW1573 | - | Growth Inhibition Assay | - | IC50=11.7295 μM | SANGER |
| SW1710 | - | Growth Inhibition Assay | - | IC50=8.18515 μM | SANGER |
| SW1990 | - | Growth Inhibition Assay | - | IC50=16.1755 μM | SANGER |
| SW48 | - | Growth Inhibition Assay | - | IC50=1.25432 μM | SANGER |
| SW620 | - | Growth Inhibition Assay | - | IC50=5.24964 μM | SANGER |
| SW626 | - | Growth Inhibition Assay | - | IC50=11.5755 μM | SANGER |
| SW684 | - | Growth Inhibition Assay | - | IC50=19.1326 μM | SANGER |
| SW756 | - | Growth Inhibition Assay | - | IC50=6.91176 μM | SANGER |
| SW780 | - | Growth Inhibition Assay | - | IC50=25.3264 μM | SANGER |
| SW837 | - | Growth Inhibition Assay | - | IC50=12.9028 μM | SANGER |
| SW872 | - | Growth Inhibition Assay | - | IC50=42.6331 μM | SANGER |
| SW900 | - | Growth Inhibition Assay | - | IC50=7.82121 μM | SANGER |
| SW948 | - | Growth Inhibition Assay | - | IC50=46.4462 μM | SANGER |
| SW954 | - | Growth Inhibition Assay | - | IC50=1.20214 μM | SANGER |
| SW962 | - | Growth Inhibition Assay | - | IC50=5.62032 μM | SANGER |
| SW982 | - | Growth Inhibition Assay | - | IC50=18.2671 μM | SANGER |
| T-24 | - | Growth Inhibition Assay | - | IC50=34.0205 μM | SANGER |
| T47D | - | Growth Inhibition Assay | - | IC50=3.62217 μM | SANGER |
| T84 | - | Growth Inhibition Assay | - | IC50=11.3907 μM | SANGER |
| T98G | - | Growth Inhibition Assay | - | IC50=27.5846 μM | SANGER |
| TCCSUP | - | Growth Inhibition Assay | - | IC50=28.6418 μM | SANGER |
| TE-1 | - | Growth Inhibition Assay | - | IC50=1.81475 μM | SANGER |
| TE-10 | - | Growth Inhibition Assay | - | IC50=2.68078 μM | SANGER |
| TE-11 | - | Growth Inhibition Assay | - | IC50=26.7752 μM | SANGER |
| TE-12 | - | Growth Inhibition Assay | - | IC50=1.80414 μM | SANGER |
| TE-5 | - | Growth Inhibition Assay | - | IC50=5.4085 μM | SANGER |
| TE-6 | - | Growth Inhibition Assay | - | IC50=0.40246 μM | SANGER |
| TE-8 | - | Growth Inhibition Assay | - | IC50=1.91377 μM | SANGER |
| TE-9 | - | Growth Inhibition Assay | - | IC50=3.84382 μM | SANGER |
| TGBC11TKB | - | Growth Inhibition Assay | - | IC50=47.1777 μM | SANGER |
| TGBC1TKB | - | Growth Inhibition Assay | - | IC50=30.1238 μM | SANGER |
| TGBC24TKB | - | Growth Inhibition Assay | - | IC50=1.6823 μM | SANGER |
| TI-73 | - | Growth Inhibition Assay | - | IC50=0.14715 μM | SANGER |
| TK10 | - | Growth Inhibition Assay | - | IC50=0.26738 μM | SANGER |
| U031 | - | Growth Inhibition Assay | - | IC50=5.25424 μM | SANGER |
| U-118-MG | - | Growth Inhibition Assay | - | IC50=36.8847 μM | SANGER |
| U251 | - | Growth Inhibition Assay | - | IC50=40.4054 μM | SANGER |
| U-266 | - | Growth Inhibition Assay | - | IC50=16.2299 μM | SANGER |
| U-2-OS | - | Growth Inhibition Assay | - | IC50=17.5307 μM | SANGER |
| U-87-MG | - | Growth Inhibition Assay | - | IC50=4.10481 μM | SANGER |
| UACC-257 | - | Growth Inhibition Assay | - | IC50=41.7369 μM | SANGER |
| UACC-62 | - | Growth Inhibition Assay | - | IC50=11.2368 μM | SANGER |
| UACC-893 | - | Growth Inhibition Assay | - | IC50=0.11885 μM | SANGER |
| UMC-11 | - | Growth Inhibition Assay | - | IC50=26.4514 μM | SANGER |
| UM-UC-3 | - | Growth Inhibition Assay | - | IC50=9.9503 μM | SANGER |
| VA-ES-BJ | - | Growth Inhibition Assay | - | IC50=35.4649 μM | SANGER |
| VM-CUB-1 | - | Growth Inhibition Assay | - | IC50=4.86819 μM | SANGER |
| VMRC-RCZ | - | Growth Inhibition Assay | - | IC50=8.01697 μM | SANGER |
| WM-115 | - | Growth Inhibition Assay | - | IC50=11.7588 μM | SANGER |
| YAPC | - | Growth Inhibition Assay | - | IC50=11.9259 μM | SANGER |
| YKG-1 | - | Growth Inhibition Assay | - | IC50=1.05419 μM | SANGER |
| ZR-75-30 | - | Growth Inhibition Assay | - | IC50=0.09545 μM | SANGER |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02145637 | NSCLC | Phase 1 | Unknown | July 2016 | - |
| NCT01649284 | - | - | - | - | - |
| NCT03054038 | Non-Small Cell Lung Carcinoma | Phase 1 | Recruiting | March 2021 | United States, California ... more >> City of Hope National Medical Center Recruiting Duarte, California, United States, 91010 Contact: Thomas Fok tfok@coh.org Principal Investigator: Karen Reckamp, MD Stanford Cancer Institute Recruiting Stanford, California, United States, 94035 Contact: Jordan Preiss 650-723-1002 Principal Investigator: Sukhmani Padda, MD United States, Tennessee Vanderbilt-Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Collapse << |
| NCT01085136 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | - |
| NCT00730925 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | Belgium ... more >> 1200.41.32003 Boehringer Ingelheim Investigational Site Antwerpen, Belgium 1200.41.32007 Boehringer Ingelheim Investigational Site Charleroi, Belgium 1200.41.32001 Boehringer Ingelheim Investigational Site Jette, Belgium 1200.41.32011 Boehringer Ingelheim Investigational Site Leuven, Belgium 1200.41.32008 Boehringer Ingelheim Investigational Site Liège, Belgium 1200.41.32006 Boehringer Ingelheim Investigational Site Namur, Belgium Spain 1200.41.34001 Boehringer Ingelheim Investigational Site Badalona (Barcelona), Spain Collapse << |
| NCT02491775 | - | - | Terminated(Changes in treatmen... more >>t plan affecting drug therapy choices) Collapse << | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00525148 | - | - | Completed | - | - |
| NCT01441596 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Phase 2 | Completed | - | - |
| NCT01085136 | - | - | Completed | - | - |
| NCT01679405 | Metastatic Disease | Phase 1 | Terminated(2016/04/26) | December 2016 | Germany ... more >> I. Medizinische Klinik und Poliklinik der Universitätsmedizin Mainz, Germany, 55131 Collapse << |
| NCT00525148 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | - |
| NCT01441596 | - | - | Completed | - | - |
| NCT00727506 | Glioma | Phase 2 | Completed | - | - |
| NCT00730925 | - | - | Completed | - | - |
| NCT01271725 | Breast Neoplasms | Phase 2 | Completed | - | Hong Kong ... more >> 1200.98.85202 Boehringer Ingelheim Investigational Site Hong Kong, Hong Kong 1200.98.85201 Boehringer Ingelheim Investigational Site Shatin, Hong Kong India 1200.98.91011 Boehringer Ingelheim Investigational Site Amravati, India 1200.98.91009 Boehringer Ingelheim Investigational Site Maharashtra, India 1200.98.91010 Boehringer Ingelheim Investigational Site Maharashtra, India 1200.98.91004 Boehringer Ingelheim Investigational Site Nagpur, India 1200.98.91001 Boehringer Ingelheim Investigational Site Pune, India 1200.98.91005 Boehringer Ingelheim Investigational Site Thiruvananthapuram, India Poland 1200.98.48002 Boehringer Ingelheim Investigational Site Gdansk, Poland Russian Federation 1200.98.07014 Boehringer Ingelheim Investigational Site Kazan, Russian Federation 1200.98.07013 Boehringer Ingelheim Investigational Site Krasnodar, Russian Federation 1200.98.07004 Boehringer Ingelheim Investigational Site Moscow, Russian Federation 1200.98.07012 Boehringer Ingelheim Investigational Site Pyatigorsk, Russian Federation 1200.98.07008 Boehringer Ingelheim Investigational Site Samara, Russian Federation 1200.98.07011 Boehringer Ingelheim Investigational Site Sochi, Russian Federation 1200.98.07003 Boehringer Ingelheim Investigational Site Stavropol, Russian Federation 1200.98.07006 Boehringer Ingelheim Investigational Site Yaroslavl, Russian Federation Taiwan 1200.98.88603 Boehringer Ingelheim Investigational Site Taichung, Taiwan 1200.98.88607 Boehringer Ingelheim Investigational Site Taichung, Taiwan 1200.98.88601 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1200.98.88602 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1200.98.88604 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1200.98.88605 Boehringer Ingelheim Investigational Site Taipei, Taiwan United Kingdom 1200.98.44009 Boehringer Ingelheim Investigational Site Barnstaple, United Kingdom 1200.98.44001 Boehringer Ingelheim Investigational Site Bournemouth, United Kingdom 1200.98.44004 Boehringer Ingelheim Investigational Site Exeter, United Kingdom 1200.98.44005 Boehringer Ingelheim Investigational Site London, United Kingdom Collapse << |
| NCT02372006 | Neuroectodermal Tumors ... more >> Rhabdomyosarcoma Collapse << | Phase 1 Phase 2 | Recruiting | August 15, 2019 | - |
| NCT01396265 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1200.152.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01396265 | - | - | Completed | - | - |
| NCT00727506 | - | - | Completed | - | - |
| NCT01152437 | Colorectal Neoplasms | Phase 2 | Completed | - | United Kingdom ... more >> 1200.74.44001 Boehringer Ingelheim Investigational Site Bournemouth, United Kingdom 1200.74.44005 Boehringer Ingelheim Investigational Site Bristol, United Kingdom 1200.74.44006 Boehringer Ingelheim Investigational Site Cambridge, United Kingdom 1200.74.44003 Boehringer Ingelheim Investigational Site Glasgow, United Kingdom 1200.74.44009 Boehringer Ingelheim Investigational Site London, United Kingdom 1200.74.44012 Boehringer Ingelheim Investigational Site Manchester, United Kingdom 1200.74.44007 Boehringer Ingelheim Investigational Site Northwood, United Kingdom 1200.74.44013 Boehringer Ingelheim Investigational Site Nottingham, United Kingdom 1200.74.44011 Boehringer Ingelheim Investigational Site Poole, United Kingdom 1200.74.44010 Boehringer Ingelheim Investigational Site Sheffield, United Kingdom 1200.74.44008 Boehringer Ingelheim Investigational Site Southampton, United Kingdom 1200.74.44004 Boehringer Ingelheim Investigational Site Sutton, Surrey, United Kingdom 1200.74.44002 Boehringer Ingelheim Investigational Site Truro, United Kingdom Collapse << |
| NCT01152437 | - | - | Completed | - | - |
| NCT01426958 | - | - | Completed | - | - |
| NCT01298063 | Liver Diseases ... more >> Healthy Collapse << | Phase 1 | Completed | - | Germany ... more >> 1200.86.1 Boehringer Ingelheim Investigational Site Kiel, Germany Collapse << |
| NCT02541903 | Penile Squamous Cell Carcinoma... more >> (PSCC) Collapse << | Phase 2 | Recruiting | October 2019 | United States, Alabama ... more >> University of Alabama at Birmingham Recruiting Birmingham, Alabama, United States, 35294 Contact: Pam Dixon, RN, BSN, OCN 205-975-9875 pamdixon@uab.edu Principal Investigator: Lisle Nabell, MD United States, California University of Southern California Recruiting Los Angeles, California, United States, 90033 Contact: Tanya Dorff, MD 323-865-3900 dorff@usc.edu United States, Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Cherie Perez 713-563-1602 GUClinicalTrials@mdanderson.org Principal Investigator: John Araujo, MD Collapse << |
| NCT01298063 | - | - | Completed | - | - |
| NCT00796549 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | Italy ... more >> 1200.40.39011 Boehringer Ingelheim Investigational Site Arezzo, Italy 1200.40.39007 Boehringer Ingelheim Investigational Site Aviano (PN), Italy 1200.40.39013 Boehringer Ingelheim Investigational Site Faenza (RA), Italy 1200.40.39003 Boehringer Ingelheim Investigational Site Genova, Italy 1200.40.39010 Boehringer Ingelheim Investigational Site Livorno, Italy 1200.40.39012 Boehringer Ingelheim Investigational Site Lugo (RA), Italy 1200.40.39008 Boehringer Ingelheim Investigational Site Modena, Italy 1200.40.39005 Boehringer Ingelheim Investigational Site Monza (MI), Italy 1200.40.39006 Boehringer Ingelheim Investigational Site Padova, Italy 1200.40.39002 Boehringer Ingelheim Investigational Site Perugia, Italy 1200.40.39004 Boehringer Ingelheim Investigational Site Prato, Italy 1200.40.39009 Boehringer Ingelheim Investigational Site Ravenna, Italy 1200.40.39001 Boehringer Ingelheim Investigational Site Rozzano (MI), Italy Collapse << |
| NCT00431067 | - | - | Completed | - | - |
| NCT00796549 | - | - | Completed | - | - |
| NCT00950742 | Breast Neoplasms | Phase 1 | Completed | - | United Kingdom ... more >> 1200.68.44001 Boehringer Ingelheim Investigational Site Brighton, United Kingdom 1200.68.44003 Boehringer Ingelheim Investigational Site Cambridge, United Kingdom 1200.68.44005 Boehringer Ingelheim Investigational Site Guildford, United Kingdom 1200.68.44004 Boehringer Ingelheim Investigational Site Newcastle upon Tyne, United Kingdom 1200.68.44002 Boehringer Ingelheim Investigational Site Truro, United Kingdom Collapse << |
| NCT01426958 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1200.151.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT00431067 | Breast Neoplasms | Phase 2 | Completed | - | United States, Arizona ... more >> 1200.11.3 Boehringer Ingelheim Investigational Site Scottsdale, Arizona, United States United States, California 1200.11.7 Boehringer Ingelheim Investigational Site Encinitas, California, United States 1200.11.4 Boehringer Ingelheim Investigational Site Santa Monica, California, United States United States, Florida 1200.11.2 Boehringer Ingelheim Investigational Site Tampa, Florida, United States United States, Massachusetts 1200.11.1 Boehringer Ingelheim Investigational Site Boston, Massachusetts, United States United States, North Carolina 1200.11.5 Boehringer Ingelheim Investigational Site Chapel Hill, North Carolina, United States United Kingdom 1200.11.4401 Boehringer Ingelheim Investigational Site Bournemouth, United Kingdom 1200.11.4402 Boehringer Ingelheim Investigational Site Crownhill, Plymouth, United Kingdom 1200.11.4406 Boehringer Ingelheim Investigational Site Guildford, United Kingdom 1200.11.4405 Boehringer Ingelheim Investigational Site London, United Kingdom 1200.11.4404 Boehringer Ingelheim Investigational Site Poole, United Kingdom 1200.11.4403 Boehringer Ingelheim Investigational Site Truro, United Kingdom Collapse << |
| NCT00950742 | - | - | Completed | - | - |
| NCT00875433 | Neoplasms | Phase 2 | Completed | - | United Kingdom ... more >> 1200.24.4403 Boehringer Ingelheim Investigational Site Guildford, United Kingdom 1200.24.4402 Boehringer Ingelheim Investigational Site London, United Kingdom 1200.24.4404 Boehringer Ingelheim Investigational Site London, United Kingdom 1200.24.4401 Boehringer Ingelheim Investigational Site Sutton, United Kingdom Collapse << |
| NCT00875433 | - | - | Completed | - | - |
| NCT01931306 | - | - | - | - | Korea, Republic of ... more >> The Catholic University of Korea, Bucheon St.Mary's Hospital Bucheon, Korea, Republic of, 14647 Gachon University Gil Medical Center Incheon, Korea, Republic of, 405-760 Chonnam National University Hwasun Hospital Jeonnam, Korea, Republic of, 519-763 Gyeongsang National University Hospital Jinju, Korea, Republic of, 660-702 The Catholic University of Korea, Seoul St.Mary's Hospital Seoul, Korea, Republic of, 06591 Severance Hospital Seoul, Korea, Republic of, 120-752 Samsung Medical Center Seoul, Korea, Republic of, 135-710 SMG-SNU Boramae Medical Center Seoul, Korea, Republic of, 156-707 Collapse << |
| NCT00998296 | Neoplasms | Phase 1 | Completed | - | France ... more >> 1239.14.3301A Boehringer Ingelheim Investigational Site Villejuif Cedex, France Collapse << |
| NCT01251653 | - | - | Completed | - | France ... more >> 1200.93.33002 Boehringer Ingelheim Investigational Site Dijon, France 1200.93.33001 Boehringer Ingelheim Investigational Site Saint-Herblain cedex, France 1200.93.33003 Boehringer Ingelheim Investigational Site Toulouse, France Collapse << |
| NCT02506517 | Solid Tumors | Phase 2 | Recruiting | December 2018 | Canada, Ontario ... more >> Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Albiruni Razak, M.D. 416-586-4800 ext 3883 Principal Investigator: Albiruni Razak, M.D. Collapse << |
| NCT01647711 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | United States, Colorado ... more >> Boehringer Ingelheim Investigational Site Aurora, Colorado, United States United States, Massachusetts Boehringer Ingelheim Investigational Site Boston, Massachusetts, United States Collapse << |
| NCT01649271 | Breast Neoplasms ... more >> Stomach Neoplasms Collapse << | Phase 1 | Completed | - | France ... more >> CTR Georges-François Leclerc Dijon, France, 21079 CTR René Gauducheau, Onco, St Herblain Saint Herblain Cedex, France, 44805 INS Claudius Regaud Toulouse, France, 31059 Collapse << |
| NCT00998296 | - | - | Completed | - | - |
| NCT02597946 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | China ... more >> Hunan Province Tumor Hospital Changsha, China, 410013 First Affiliated Hospital of Guangzhou Medical University Guangzhou, China, 510120 Zhejiang Cancer Hospital Hangzhou, China, 310022 The Second Affiliated Hospital to Nanchang University Nanchang, China, 330006 First Hospital Affiliated with Nanjing Medical University Nanjing, China, 210029 Zhongshan Hospital Fudan University Shanghai, China, 200032 Shanghai Pulmonary Hospital Shanghai, China, 200433 Henan Cancer Hospital Zhengzhou, China, 450008 Malaysia University Malaya Medical Centre Kuala Lumpur, Malaysia, 59100 Collapse << |
| NCT01647711 | - | - | Completed | - | - |
| NCT03157089 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Recruiting | October 9, 2020 | United States, California ... more >> University of California Davis Recruiting Sacramento, California, United States, 95817 Contact: Jonathan Riess +001 (916) 734-3772 jwriess@ucdavis.edu United States, District of Columbia Johns Hopkins Sidney Kimmel Cancer Center at Sibley Memorial Hospital Recruiting Washington, District of Columbia, United States, 20016 Contact: Benjamin Levy +001 (202) 660-6500 blevy11@jhmi.edu United States, Florida Florida Hospital Recruiting Orlando, Florida, United States, 32804 Contact: Tarek Mekhail +001 (407) 303-3235 tarek.mekhail.md@flhosp.org United States, Kentucky Baptist Health Medical Group Recruiting Lexington, Kentucky, United States, 40503 Contact: Firas Badin +001 (859) 276-0414 firas.badin@bhsi.com United States, Nevada Comprehensive Cancer Centers of Nevada Recruiting Las Vegas, Nevada, United States, 89128 Contact: Brian Vicuna +001 (702) 952-2140 brian.vicuna@usoncology.com United States, New York Weill Cornell Medical College Recruiting New York, New York, United States, 10021 Contact: Ronald Scheff +001 (646) 962-2066 rjs2002@med.cornell.edu United States, Washington Cancer Care Northwest Centers, PS Recruiting Spokane Valley, Washington, United States, 99216 Contact: Srivalli Gopaluni +001 (509) 228-1000 srivalli.gopaluni@ccnw.net France HOP Côte de Nacre Recruiting Caen, France, 14033 Contact: Jeannick Madelaine +33 (0)2 31 06 46 76 Madelaine-j@chu-caen.fr HOP Le Mans Recruiting Le Mans, France, 72037 Contact: Olivier Molinier +33 (0)2 43 43 24 50 omolinier@ch-lemans.fr HOP Nord Recruiting Marseille, France, 13015 Contact: Fabrice Barlesi +33 (0)4 91 38 56 28 fabrice.barlesi@ap-hm.fr HOP Nord Laënnec Recruiting Nantes, France, 44093 Contact: Jaafar Bennouna +33 (0)2 40 67 99 93 Jaafar.Bennouna@univ-nantes.fr HOP Sainte Musse Recruiting Toulon, France, 83100 Contact: Clarisse Audigier-Valette +33 (0)4 94 14 56 10 clarisse.audigier-valette@ch-toulon.fr Korea, Republic of Severance Hospital Recruiting Seoul, Korea, Republic of, 03722 Contact: Hye Ryun Kim 82 2 2228 8125 nobelg@yuhs.ac Asan Medical Center Recruiting Seoul, Korea, Republic of, 05505 Contact: Sang-We Kim 82 2 3010 3215 swkim@amc.seoul.kr Spain Hospital Vall d'Hebron Recruiting Barcelona, Spain, 08035 Contact: Enriqueta Felip +34932746077 efelip@vhio.net Complejo Hospitalario Universitario Insular - Materno Infantil Recruiting Las Palmas de Gran Canaria, Spain, 35016 Contact: Delvys Rodríguez Abreu +34928441738 drodabr@gobiernodecanarias.org Hospital Universitario 12 de Octubre Recruiting Madrid, Spain, 28041 Contact: Luis Paz-Ares Rodríguez +34913908349 lpazaresr@seom.org Hospital Clínico Universitario Lozano Blesa Recruiting Zaragoza, Spain, 50009 Contact: Dolores Isla +34976765746 lola.isla@gmail.com Turkey Hacettepe Universitesi Tip Fakultesi, Onkoloji ABD Active, not recruiting Ankara, Turkey, 06230 Istanbul Universitesi Cerrahpasa Tip Fakultesi Completed Istanbul, Turkey, 34098 Collapse << |
| NCT01251653 | - | - | Completed | - | - |
| NCT02768337 | Lung Cancer B... more >>reast Cancer Brain Cancer Advanced Breast Cancer Advanced Lung Cancer Collapse << | Phase 1 Phase 2 | Recruiting | December 2021 | United Kingdom ... more >> Cambridge University Hospitals NHS Foundation Trust Recruiting Cambridge, England, United Kingdom, CB2 2QQ University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, United Kingdom The Beatson West of Scotland Cancer Centre Recruiting Glasgow, United Kingdom Clatterbridge Cancer Centre Recruiting Liverpool, United Kingdom The Christie NHS Foundation Trust Recruiting Manchester, United Kingdom Oxford University Hospitals NHS Foundation Trust Recruiting Oxford, United Kingdom Collapse << |
| NCT00716417 | Neoplasms | Phase 1 | Completed | - | Belgium ... more >> 1200.37.3202 Boehringer Ingelheim Investigational Site Bruxelles, Belgium 1200.37.3201 Boehringer Ingelheim Investigational Site Edegem, Belgium 1200.37.3203 Boehringer Ingelheim Investigational Site Gent, Belgium Collapse << |
| NCT01649271 | - | - | Completed | - | - |
| NCT00716417 | - | - | Completed | - | - |
| NCT01156545 | Non-small Cell Lung Cancer | Phase 2 | Active, not recruiting | December 2019 | Korea, Republic of ... more >> National Cancer Center Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Collapse << |
| NCT01732640 | Squamous Cell Carcinoma of the... more >> Head and Neck Collapse << | Phase 1 Phase 2 | Terminated(Slow accrual and hi... more >>gh levels of toxicity lead to early termination.) Collapse << | - | United States, Maryland ... more >> Johns Hopkins Sidney Kimmel Comprehensive Cancer Center Baltimore, Maryland, United States, 21287 United States, Tennessee Vanderbilt Ingram Cancer Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT03695510 | Head and Neck Neoplasms | Phase 2 | Not yet recruiting | April 2021 | Taiwan ... more >> National Taiwan University Hospital Not yet recruiting Taipei, Taiwan, 100 Contact: Ruey-Long Hong, MD PhD +886-2-23123456 rlhong@ntu.edu.tw Collapse << |
| NCT01523587 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | - |
| NCT01732640 | - | - | Terminated(Slow accrual and hi... more >>gh levels of toxicity lead to early termination.) Collapse << | - | - |
| NCT01932229 | NSCLC | Phase 2 | Unknown | - | Israel ... more >> Sheba Medical Center Recruiting Ramat Gat, Israel, 52621 Contact: Dina Vornstein Diana.Vorenshtein@sheba.health.gov.il Principal Investigator: Jair Bar, MD-PhD Collapse << |
| NCT01836341 | Non-small Cell Lung Cancer | Phase 1 | Withdrawn | April 2015 | - |
| NCT02020577 | Neoplasms | Phase 1 | Completed | - | France ... more >> 1200.122.33001 Boehringer Ingelheim Investigational Site Villejuif Cedex, France Spain 1200.122.34001 Boehringer Ingelheim Investigational Site Madrid, Spain Collapse << |
| NCT01003899 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | Korea, Republic of ... more >> 1200.72.8201 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1200.72.8202 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1200.72.8203 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of Collapse << |
| NCT03652233 | Recurrent Squamous Cell Carcin... more >>oma of the Head or Neck Platinum- and Cetuximab-Refractory Squamous Cell Carcinoma of the Head or Neck Metastatic Squamous Cell Carcinoma of the Head or Neck Squamous Cell Carcinoma Collapse << | Phase 1 | Not yet recruiting | October 2022 | United States, Tennessee ... more >> Vanderbilt-Ingram Cancer Center Not yet recruiting Nashville, Tennessee, United States, 37232 Contact: Clinical Trials Information Program 800-811-8480 cip@vanderbilt.edu Principal Investigator: Mike Gibson, MD, PhD Collapse << |
| NCT01003899 | - | - | Completed | - | - |
| NCT01919879 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | France ... more >> Institute de Cancérologie de la Loire Nantes, France, 44805 Collapse << |
| NCT02216617 | HNSCC | Phase 1 | Completed | - | France ... more >> ICM Val d'Aurelle, Montpellier Montpellier, France, 34298 Collapse << |
| NCT01861223 | NSCLC | Phase 1 Phase 2 | Unknown | May 2016 | Korea, Republic of ... more >> Samsung Medical Center Recruiting Seoul, Korea, Republic of Contact: Myung-Ju Ahn Sub-Investigator: Jong-Mu Sun Collapse << |
| NCT02183883 | Non-small Cell Lung Cancer | Phase 2 | Recruiting | November 2023 | United Kingdom ... more >> Aberdeen Royal Infirmary (NHS Grampian) Recruiting Aberdeen, United Kingdom Principal Investigator: Gillian Price Heart of England NHS Foundation Trust Recruiting Birmingham, United Kingdom Principal Investigator: Shobhit Baijal Beatson West of Scotland Cancer Centre (NHS Greater Glasgow & Clyde) Recruiting Glasgow, United Kingdom Principal Investigator: Nicola Steele Barnet and Chase Farm Hospitals (Royal Free London NHS Foundation Trust) Recruiting London Borough Of Barnet, United Kingdom Principal Investigator: Tanya Ahmad University College London Hospitals NHS Foundation Trust Recruiting London, United Kingdom, NW1 2BU Principal Investigator: Martin Forster Cr Uk & Ucl Ctc Active, not recruiting London, United Kingdom, W1T 4TJ The Christie NHS Foundation Trust Recruiting Manchester, United Kingdom Principal Investigator: Matthew Krebs Collapse << |
| NCT02020577 | - | - | Completed | - | - |
| NCT00748709 | - | - | Terminated | - | - |
| NCT02780687 | Urologic Neoplasms | Phase 2 | Recruiting | March 30, 2020 | France ... more >> INS Bergonié Recruiting Bordeaux, France, 33076 CTR Léon Bérard Recruiting Lyon, France, 69373 INS Cancérologie du Gard Recruiting Nîmes, France, 30029 HOP Saint-Louis Recruiting Paris, France, 75010 HOP Cochin Recruiting Paris, France, 75014 HOP Européen G. Pompidou Recruiting Paris, France, 75015 HOP Foch Recruiting Suresnes, France, 92150 INS Claudius Regaud Recruiting Toulouse, France, 31059 INS Gustave Roussy Recruiting Villejuif, France, 94805 Italy Ospedale San Donato di Arezzo Recruiting Arezzo, Italy, 52100 A.O. San Camillo Forlanini Recruiting Roma, Italy, 00152 Spain Hospital Germans Trias i Pujol Recruiting Badalona, Spain, 08916 Hospital del Mar Recruiting Barcelona, Spain, 08003 Hospital Santa Creu i Sant Pau Recruiting Barcelona, Spain, 08025 Hospital Clínic de Barcelona Recruiting Barcelona, Spain, 08036 Hospital Vall d'Hebron Recruiting Barcelona, Spain, 08038 Hospital Universitario de Elche Recruiting Elche, Spain, 03202 Hospital Universitari de Girona Doctor Josep Trueta Recruiting Girona, Spain, 17007 Hospital Duran i Reynals Recruiting L'Hospitalet de Llobregat, Spain, 08908 Hospital Universitario Lucus Augusti Recruiting Lugo, Spain, 27003 Hospital Ramón y Cajal Recruiting Madrid, Spain, 28034 Hospital Clínico San Carlos Recruiting Madrid, Spain, 28040 Hospital Universitario 12 de Octubre Recruiting Madrid, Spain, 28041 Hospital La Paz Recruiting Madrid, Spain, 28046 CIO Clara Campal Recruiting Madrid, Spain, 28050 Hospital Son Espases Recruiting Palma de Mallorca, Spain, 07010 CS Parc Taulí Recruiting Sabadell, Spain, 08208 Hospital Virgen Macarena Recruiting Sevilla, Spain, 41009 Hospital Virgen del Rocío Recruiting Sevilla, Spain, 41013 Instituto Valenciano de Oncología Recruiting Valencia, Spain, 46009 Collapse << |
| NCT01523587 | - | - | Completed | - | - |
| NCT01325428 | - | - | Completed | - | - |
| NCT01553942 | Lung Cancer | Phase 2 | Recruiting | December 2020 | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02215 Contact: Geoffrey Oxnard, MD 617-632-6049 goxnard@partners.org Principal Investigator: Geoffrey Oxnard, MD Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02215 Contact: Lecia V Sequist, MD MPH 617-724-4000 lvsequist@partners.org Principal Investigator: Lecia V Sequist, MD MPH Collapse << |
| NCT02438722 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 2 Phase 3 | Active, not recruiting | February 2022 | - |
| NCT02450656 | Colorectal Neoplasms ... more >> Gastrointestinal Neoplasms Pancreatic Neoplasms Carcinoma, Non-Small-Cell Lung Collapse << | Phase 1 Phase 2 | Recruiting | December 2019 | Netherlands ... more >> UMC St. Radboud Nijmegen Recruiting Nijmegen, Gelderland, Netherlands, 6525GA Contact: Carla ML van Herpen, MD, PhD +31243610353 Carla.vanHerpen@radboudumc.nl Principal Investigator: Carla ML van Herpen, MD, PhD Netherlands Cancer Institute - Antoni van Leeuwenhoek Hospital Recruiting Amsterdam, Netherlands, 1066CX Contact: F Opdam, MD, PhD Principal Investigator: F Opdam, MD, PhD Collapse << |
| NCT00748709 | Neoplasms | Phase 2 | Terminated | - | United States, California ... more >> 1200.26.3 Boehringer Ingelheim Investigational Site Los Angeles, California, United States United States, Colorado 1200.26.11 Boehringer Ingelheim Investigational Site Denver, Colorado, United States United States, Indiana 1200.26.9 Boehringer Ingelheim Investigational Site Indianapolis, Indiana, United States United States, Massachusetts 1200.26.1 Boehringer Ingelheim Investigational Site Boston, Massachusetts, United States United States, Nevada 1200.26.13 Boehringer Ingelheim Investigational Site Las Vegas, Nevada, United States United States, New York 1200.26.4 Boehringer Ingelheim Investigational Site Albany, New York, United States 1200.26.2 Boehringer Ingelheim Investigational Site New York, New York, United States United States, Ohio 1200.26.7 Boehringer Ingelheim Investigational Site Kettering, Ohio, United States United States, Texas 1200.26.12 Boehringer Ingelheim Investigational Site Dallas, Texas, United States 1200.26.8 Boehringer Ingelheim Investigational Site Tyler, Texas, United States United States, Virginia 1200.26.6 Boehringer Ingelheim Investigational Site Norfolk, Virginia, United States United States, Washington 1200.26.10 Boehringer Ingelheim Investigational Site Vancouver, Washington, United States Taiwan 1200.26.88603 Boehringer Ingelheim Investigational Site Tainan, Taiwan 1200.26.88601 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1200.26.88602 Boehringer Ingelheim Investigational Site Tao-Yuan, Taiwan Collapse << |
| NCT01074177 | Non-small Cell Lung Cancer ... more >> EGFR Mutations Collapse << | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01074177 | - | - | Completed | - | - |
| NCT01325428 | Breast Neoplasms | Phase 2 | Completed | - | United States, California ... more >> 1200.89.10001 Boehringer Ingelheim Investigational Site Los Angeles, California, United States United States, North Carolina 1200.89.10005 Boehringer Ingelheim Investigational Site Durham, North Carolina, United States Australia, Victoria 1200.89.61002 Boehringer Ingelheim Investigational Site East Bentleigh, Victoria, Australia Australia, Western Australia 1200.89.61003 Boehringer Ingelheim Investigational Site Perth, Western Australia, Australia Hong Kong 1200.89.85201 Boehringer Ingelheim Investigational Site Hong Kong, Hong Kong Korea, Republic of 1200.89.82001 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1200.89.82002 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of Thailand 1200.89.66002 Boehringer Ingelheim Investigational Site Bangkok, Thailand 1200.89.66004 Boehringer Ingelheim Investigational Site Bangkok, Thailand 1200.89.66003 Boehringer Ingelheim Investigational Site Chiangmai, Thailand 1200.89.66001 Boehringer Ingelheim Investigational Site Hat-Yai, Songkhla, Thailand Tunisia 1200.89.21601 Boehringer Ingelheim Investigational Site Ariana, Tunisia 1200.89.21602 Boehringer Ingelheim Investigational Site Sousse, Tunisia United Kingdom 1200.89.44002 Boehringer Ingelheim Investigational Site Bournemouth, United Kingdom 1200.89.44001 Boehringer Ingelheim Investigational Site London, United Kingdom 1200.89.44003 Boehringer Ingelheim Investigational Site London, United Kingdom Collapse << |
| NCT01214616 | Neoplasms | Phase 1 | Completed | - | Japan ... more >> 1200.84.003 Boehringer Ingelheim Investigational Site Chuo-ku, Osaka, Osaka, Japan 1200.84.004 Boehringer Ingelheim Investigational Site Kashiwa, Chiba, Japan 1200.84.001 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1200.84.002 Boehringer Ingelheim Investigational Site Sakyo-ku, Kyoto, Kyoto, Japan Collapse << |
| NCT02716311 | Non Small Cell Lung Cancer | Phase 2 | Recruiting | January 2020 | France ... more >> Centre Hospitalier du Pays d'Aix Not yet recruiting Aix-en-Provence, France Contact: Stéphanie MARTINEZ contact@ifct.fr Clinique de L'Europe Not yet recruiting Amiens, France Contact: Olivier CARRE, Dr Angers - CHU Recruiting Angers, France Contact: José Hureaux, Dr Principal Investigator: José Hureaux, Dr Annecy - CH Recruiting Annecy, France, 74374 Contact: Chantal DECROISETTE, Dr Bordeaux - Institut Bergonié Not yet recruiting Bordeaux, France Contact: Sylvestre LE MOULEC, Dr contact@ifct.fr Principal Investigator: LE MOULEC Sylvestre, Dr Bordeaux - Polyclinique Nord Recruiting Bordeaux, France Contact: Nadine DOHOLLOU, Dr contact@ifct.fr Principal Investigator: Nadine DOHOLLOU, Dr Boulogne - Ambroise Paré Recruiting Boulogne-Billancourt, France Contact: Etienne GIROUX-LEPRIEUR, MD Principal Investigator: Etienne GIROUX-LEPRIEUR, MD Clermont-Ferrand - CHU Not yet recruiting Clermont-Ferrand, France Contact: Patrick MERLE, MD Principal Investigator: Patrick MERLE, MD CH Recruiting Colmar, France Contact: Lionel MOREAU contact@ifct.fr CHRU Grenoble Recruiting Grenoble, France Contact: Denis MORO-SIBILOT contact@ifct.fr Centre Hospitalier - Pneumologie Recruiting Le Mans, France, 72000 Contact: Olivier Molinier, Dr CHRU de Lille Recruiting Lille, France Contact: Alexis CORTOT, Pr contact@ifct.fr Lille - Polyclinique de la Louvière Not yet recruiting Lille, France Contact: Benjamin HURET, Dr contact@ifct.fr Principal Investigator: Benjamin HURET, Dr Lyon - Hôpital Privé Jean Mermoz Not yet recruiting Lyon, France Contact: Pierre Bombaron, MD contact@ifct.fr Principal Investigator: Pierre BOMBARON, MD Institut Paoli Calmette Recruiting Marseille, France Contact: Anne MADROSZYK, Dr Montpellier - GCS Centre de Cancérologie du Grand Montpellier Not yet recruiting Montpellier, France Contact: Catherine Becht, Dr Principal Investigator: Catherine Becht, Dr CH de Mulhouse Recruiting Mulhouse, France Contact: Didier DEBIEUVRE contact@ifct.fr Nantes - ICO René Gauducheau Recruiting Nantes, France Contact: Judith RAIMBOURG, Dr contact@ifct.fr Principal Investigator: Judith RAIMBOURG, Dr Nevers - CH Not yet recruiting Nevers, France, 58033 Contact: Dominique Herman, Dr Centre Antoine Lacassagne Recruiting Nice, France Contact: Josiane OTTO, MD contact@ifct.fr Orléans - Hôpital de la Source Recruiting Orléans, France Contact: Hugues MOREL, Dr contact@ifct.fr Principal Investigator: Hugues MOREL, Dr AP-HP Hopital Tenon - Pneumologie Recruiting Paris, France, 75020 Contact: Jacques Cadranel, Pr +33.1.56.01.65.31 contact@ifct.fr Principal Investigator: Jacques Cadranel, pr Paris - APHP Bichat Not yet recruiting Paris, France Contact: Valérie Gounant, Dr contact@ifct.fr Principal Investigator: Valérie Gounant, Dr Paris - APHP Saint-Louis Recruiting Paris, France Contact: Ludovic DOUCET, MD contact@ifct.fr Principal Investigator: Ludovic DOUCET, MD Pau - CH Recruiting Pau, France, 64046 Contact: Aldo RENAULT, Dr contact@ifct.fr Pontoise - CH Not yet recruiting Pontoise, France Contact: Cécile DURAND-MATRINGE, MD contact@ifct.fr Principal Investigator: Cécile DURAND-MATRINGE, MD Centre Hospitalier Recruiting Saint-Quentin, France Contact: Isabelle RAULT, MD contact@ifct.fr Nouvel Hopital Civil - Pneumologie Recruiting Strasbourg, France, 63000 Contact: Charlotte Leduc, Dr Principal Investigator: Charlotte Leduc, Dr Centre Hospitalier Intercommunal Recruiting Toulon, France Contact: Clarisse AUDIGIER-VALETTE, Dr HIA Saint Anne Recruiting Toulon, France Contact: Henri BERARD, Dr contact@ifct.fr Tours - CHU Not yet recruiting Tours, France, 37000 Contact: Eric Pichon, Dr contact@ifct.fr CH de Villefranche - Pneumologie Recruiting Villefranche, France Contact: Luc ODIER contact@ifct.fr Principal Investigator: Luc ODIER Villeneuve d'Ascq - Hôpital Privé Not yet recruiting Villeneuve-d'Ascq, France Contact: Benjamin HURET, MD Principal Investigator: Benjamin HURET, MD Collapse << |
| NCT01214616 | - | - | Completed | - | - |
| NCT03370770 | - | - | Completed | - | Austria ... more >> Otto-Wagner Hospital Vienna, Austria, 1140 Collapse << |
| NCT02191891 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Japan ... more >> National Hospital Organization Kyushu Cancer Center Fukuoka, Fukuoka, Japan, 811-1395 Korea, Republic of Chungbuk National University Hospital Cheongju, Korea, Republic of, 361-771 Severance Hospital, Yonsei University Health System Seoul, Korea, Republic of, 120-752 Samsung Medical Center Seoul, Korea, Republic of, 135-710 Asan Medical Center Seoul, Korea, Republic of, 138-736 Singapore National Cancer Centre Singapore, Singapore, 169610 Taiwan Chang Gung Memorial Hospital Chiayi Chiayi, Taiwan, 613 Kaohsiung Chang Gung Memorial Hospital Kaohsiung, Taiwan, 83301 NCKUH Tainan, Taiwan, 704 National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT02470065 | Non Small Cell Lung Cancer | Phase 2 | Suspended(Study never started) | January 2021 | - |
| NCT02979977 | Squamous Cell Cancers of the H... more >>ead and Neck Collapse << | Phase 2 | Recruiting | January 2020 | United States, Connecticut ... more >> Yale Cancer Center Recruiting New Haven, Connecticut, United States, 06520-8028 Contact: Clinical Trials Office 203-785-5702 Principal Investigator: Aarti Bhatia, MD United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Hospital Not yet recruiting Baltimore, Maryland, United States, 21287 Contact: Hyunseok Kang, MD, MPH United States, Pennsylvania Fox Chase Cancer Center Not yet recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: Jessica Bauman, MD Collapse << |
| NCT02451553 | Advanced Malignant Solid Neopl... more >>asm Bile Duct Carcinoma Recurrent Malignant Solid Neoplasm Recurrent Pancreatic Carcinoma Stage III Pancreatic Cancer AJCC v6 and v7 Stage IVA Pancreatic Cancer Stage IVB Pancreatic Cancer Collapse << | Phase 1 | Recruiting | - | United States, Indiana ... more >> Indiana University/Melvin and Bren Simon Cancer Center Recruiting Indianapolis, Indiana, United States, 46202 Contact: Sandra Wilson 317-274-3512 slhooper@iu.edu Principal Investigator: Safi Shahda United States, Washington Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Elena G. Chiorean 206-288-6248 gchiorea@uw.edu Principal Investigator: Elena G. Chiorean Collapse << |
| NCT02876081 | SMALL CELL LUNG CARCINOMA | Phase 2 | Withdrawn(medical decision) | December 2018 | France ... more >> Groupe Hospitalier Paris Saint Joseph Paris, Ile-de-France, France, 75014 Collapse << |
| NCT01480141 | Lung Cancer | Phase 2 | Withdrawn(PI left MUSC, no pat... more >>ient enrollments) Collapse << | - | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT02975141 | Metastatic Pancreatic Cancer | Phase 1 | Active, not recruiting | June 2019 | Germany ... more >> Ludwig-Maximilians - University of Munich Munich, Germany, 81377 Collapse << |
| NCT01721525 | Head and Neck Cancer ... more >> Squamous Cell Cancer Collapse << | Phase 1 | Completed | - | United States, New Jersey ... more >> Memorial Sloan Kettering Cancer Center at Basking Ridge Basking Ridge, New Jersey, United States, 07939 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01814553 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | United States, California ... more >> 1200.167.01009 Boehringer Ingelheim Investigational Site Santa Rosa, California, United States United States, Florida 1200.167.01020 Boehringer Ingelheim Investigational Site Orlando, Florida, United States 1200.167.01018 Boehringer Ingelheim Investigational Site Port St. Lucie, Florida, United States 1200.167.01012 Boehringer Ingelheim Investigational Site St. Petersburg, Florida, United States United States, Illinois 1200.167.01007 Boehringer Ingelheim Investigational Site Skokie, Illinois, United States 1200.167.01008 Boehringer Ingelheim Investigational Site Skokie, Illinois, United States United States, New Jersey 1200.167.01001 Boehringer Ingelheim Investigational Site Morristown, New Jersey, United States United States, Oregon 1200.167.01014 Boehringer Ingelheim Investigational Site Corvallis, Oregon, United States United States, Tennessee 1200.167.01002 Boehringer Ingelheim Investigational Site Chattanooga, Tennessee, United States 1200.167.01006 Boehringer Ingelheim Investigational Site Chattanooga, Tennessee, United States 1200.167.01005 Boehringer Ingelheim Investigational Site Nashville, Tennessee, United States United States, Virginia 1200.167.01003 Boehringer Ingelheim Investigational Site Richmond, Virginia, United States Collapse << |
| NCT01999985 | Lung Cancer N... more >>on-small Cell Lung Cancer (NSCLC) Collapse << | Phase 1 | Active, not recruiting | March 2019 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01531764 | Carcinoma Breast Stage IV | Phase 2 | Terminated(The recruitment was... more >> discontinued because of failure to meet expected enrolment goals) Collapse << | - | Germany ... more >> Klinikum St. Marien Amberg Amberg, Bayern, Germany, 92224 Hämato-Onkologische Schwerpunktpraxis München, Bayern, Germany, 80638 Caritas-Krankenhaus, Onkologisches Zentrum Regensburg Regenburg, Bayern, Germany, 93053 Gynäkologische Praxis Hildesheim, Niedersachsen, Germany, 31134 Schwerpunktpraxis für Hämatologie und Onkologie Bottrop, Nordrhein-Westfalen, Germany, 46236 Universitätsklinikum Frauenklinik Düsseldorf Düsseldorf, Nordrhein-Westfalen, Germany, 40225 Otto-von-Guericke-Universität Frauenklinik Magdeburg Magdeburg, Sachsen-Anhalt, Germany, 39108 Klinikum Chemnitz gGmbH Chemnitz, Sachsen, Germany, 09116 Praxisklinik Krebsheilkunde für Frauen / Brustzentrum Berlin, Germany, 10367 Onkologisches Zentrum Süd, Vivantes Tumorzentrum Berlin, Germany, 12351 Internistische Praxisgemeinschaft Eppendorf Hamburg, Germany, 20249 OncoResearch Lerchenfled UG Hamburg, Germany, 22081 Collapse << |
| NCT01466660 | Lung Neoplasms | Phase 2 | Active, not recruiting | October 16, 2018 | - |
| NCT01466660 | - | - | Active, not recruiting | - | - |
| NCT02271906 | Lung Cancer | Phase 2 | Terminated(Poor Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01743365 | Gastric Cancer ... more >> Gastroesophageal Junction Cancer Collapse << | Phase 2 | Active, not recruiting | June 2019 | Greece ... more >> 2nd Dept of Internal Medicine, Agios Savvas Cancer Hospital Athens, Greece, 11522 Dept of Medical Oncology, 251 Air Force Hospital Athens, Greece, 11525 2nd Dept of Internal Medicine, General Hospital of Athens "Hippokratio" Athens, Greece, 11527 Oncology Section, Dept of Clinical Therapeutics, General Hospital of Athens "Alexandra" Athens, Greece, 11528 Division of Oncology, 2nd Dept of Internal Medicine, University Hospital "Attikon" Athens, Greece, 12462 2nd Dept of Medical Oncology, Agii Anargiri Cancer Hospital Athens, Greece, 14564 3rd Dept of Medical Oncology, Agii Anargiri Cancer Hospital Athens, Greece, 14564 3rd Dept of Medical Oncology, Hygeia Hospital Athens, Greece, 15123 1st Dept of Medical Oncology, Metropolitan Hospital Athens, Greece, 18547 2nd Dept of Medical Oncology, Metropolitan Hospital Athens, Greece, 18547 Dept of Medical Oncology, University Hospital of Heraklion Heraklion, Greece, 71110 Dept of Medical Oncology, Ioannina University Hospital Ioannina, Greece, 45110 Division of Oncology, Dept of Internal Medicine, University Hospital of Patras Patras, Greece, 26504 Dept of Medical Oncology, Papageorgiou General Hospital Thessaloniki, Greece, 56429 Dept of Medical Oncology, Thermi Clinic S.A Thessaloniki, Greece, 57001 Collapse << |
| NCT01783587 | Squamous Cell Carcinoma of the... more >> Head and Neck Collapse << | Phase 1 | Active, not recruiting | February 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, New York Mount Sinai Medical Center/Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT01415011 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Active, not recruiting | September 2018 | United Kingdom ... more >> Royal Bournemouth Hospital Bournemouth, United Kingdom Beatson West of Scotland Cancer Centre Glasgow, United Kingdom James Paget University Hospital Great Yarmouth, United Kingdom East Kent Hospitals Kent, United Kingdom Maidstone Hospital Kent, United Kingdom St James's University Hospital Leeds, United Kingdom The Royal Marsden Hospitals London Borough of Sutton, United Kingdom Barnet & Chase Farm Hospitals London, United Kingdom Charing Cross Hospital London, United Kingdom Guy's Hospital London, United Kingdom University College Hospital London, United Kingdom Musgrove Park Hospital Somerset, United Kingdom King's Mill Hospital Sutton in Ashfield, United Kingdom Weston General Hospital Weston Super Mare, United Kingdom York Hospital York, United Kingdom Collapse << |
| NCT01853826 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Active, not recruiting | March 31, 2019 | - |
| NCT01542437 | Non-Small Cell Lung Cancer ... more >> EGFR HER-2 Collapse << | Phase 2 | Active, not recruiting | June 2017 | Mexico ... more >> National Cancer Institute of Mexico Mexico city, Distrito Federal, Mexico, 14080 Collapse << |
| NCT03727724 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Active, not recruiting | May 2021 | Netherlands ... more >> Antoni van Leeuwenhoek Amsterdam, North-Holland, Netherlands, 1066 CX VU Medical Center Amsterdam, Netherlands, 1007 MB University Medical Center Groningen Groningen, Netherlands, 9713 GZ Maastricht UMC+ Maastricht, Netherlands, 6229 HX Collapse << |
| NCT02044380 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | Canada, Ontario ... more >> Boehringer Ingelheim Investigational Site Barrie, Ontario, Canada Boehringer Ingelheim Investigational Site London, Ontario, Canada Boehringer Ingelheim Investigational Site Newmarket, Ontario, Canada Boehringer Ingelheim Investigational Site Toronto, Ontario, Canada Canada, Quebec Boehringer Ingelheim Investigational Site Montreal, Quebec, Canada Collapse << |
| NCT01538381 | Carcinoma, Squamous Cell of He... more >>ad and Neck Collapse << | Phase 2 | Completed | - | Belgium ... more >> Institut Jules Bordet Brussels, Belgium, 1000 Cliniques Universitaires St. Luc Brussels, Belgium, 1200 U.Z. Leuven - Campus Gasthuisberg Leuven, Belgium, 3000 Italy Istituto Nazionale Per Lo Studio E La Cura Dei Tumori Milano, Italy, 20133 Collapse << |
| NCT01320280 | Refractory Cancer | Phase 2 | Completed | - | Germany ... more >> University Medical Center Hamburg-Eppendorf Hamburg, Germany, 20246 Collapse << |
| NCT01746251 | Non Small Cell Lung Cancer | Phase 2 | Active, not recruiting | August 2019 | United States, California ... more >> Stanford University Palo Alto, California, United States, 94304 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan Kettering New York, New York, United States, 10065 Collapse << |
| NCT02747953 | Non-small Cell Lung Cancer | Phase 2 | Completed | - | - |
| NCT02208843 | Carcinoma, Non-Small-Cell Lung | Phase 4 | Completed | - | Egypt ... more >> 1200.217.2201 Boehringer Ingelheim Investigational Site Alexandria, Egypt 1200.217.2202 Boehringer Ingelheim Investigational Site Cairo, Egypt 1200.217.2203 Boehringer Ingelheim Investigational Site Cairo, Egypt Malaysia 1200.217.6001 Boehringer Ingelheim Investigational Site Nilai, Malaysia Philippines 1200.217.6301 Boehringer Ingelheim Investigational Site Baguio City, Philippines 1200.217.6303 Boehringer Ingelheim Investigational Site Taguig, Philippines Poland 1200.217.4803 Boehringer Ingelheim Investigational Site Gdansk, Poland 1200.217.4805 Boehringer Ingelheim Investigational Site Szczecin-Zdunowo, Poland 1200.217.4801 Boehringer Ingelheim Investigational Site Warsaw, Poland Romania 1200.217.4004 Boehringer Ingelheim Investigational Site Braila, Romania 1200.217.4002 Boehringer Ingelheim Investigational Site Bucharest, Romania 1200.217.4005 Boehringer Ingelheim Investigational Site Craiova, Romania 1200.217.4003 Boehringer Ingelheim Investigational Site Iasi, Romania Serbia 1200.217.3801 Boehringer Ingelheim Investigational Site Belgrade, Serbia 1200.217.3802 Boehringer Ingelheim Investigational Site Belgrade, Serbia 1200.217.3804 Boehringer Ingelheim Investigational Site Kragujevac, Serbia 1200.217.3803 Boehringer Ingelheim Investigational Site Sremska Kamenica, Serbia Thailand 1200.217.6601 Boehringer Ingelheim Investigational Site Bangkok, Thailand 1200.217.6603 Boehringer Ingelheim Investigational Site Bangkok, Thailand 1200.217.6604 Boehringer Ingelheim Investigational Site Bangkok, Thailand 1200.217.6602 Boehringer Ingelheim Investigational Site Songkhla, Thailand Collapse << |
| NCT02044380 | - | - | Completed | - | - |
| NCT02096718 | - | - | Completed | - | - |
| NCT02096718 | Renal Insufficiency | Phase 1 | Completed | - | Germany ... more >> 1200.216.1 Boehringer Ingelheim Investigational Site Kiel, Germany Collapse << |
| NCT01814553 | - | - | Completed | - | - |
| NCT01824823 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 2 | Active, not recruiting | December 2020 | - |
| NCT01824823 | - | - | Active, not recruiting | - | - |
| NCT02629523 | Lung Neoplasms ... more >> EGFR Gene Mutation Collapse << | Phase 2 | Active, not recruiting | March 2019 | Korea, Republic of ... more >> Chonnam National University Hwasun Hospital Hwasun, Jeonnam, Korea, Republic of, 58128 Collapse << |
| NCT01953913 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | China ... more >> 307 Hospital of PLA Beijing, China, 100071 Beijing Cancer Hospital Beijing, China, 100142 Chinese PLA General Hospital Beijing, China, 100853 Jilin Province Cancer Hospital Changchun, China Guangdong General Hospital Guangzhou, China, 510080 Zhejiang Cancer Hospital Hangzhou, China, 310022 Lin Yi Tumor Hospital Linyi, China, 276002 Jiangsu Cancer Hospital Nanjing, China, 210000 Shanghai Chest Hospital Shanghai, China, 200030 Fudan University Shanghai Cancer Center Shanghai, China, 200032 Shanghai Pulmonary Hospital Shanghai, China, 200433 Tianjin Medical University Cancer Institute and Hospital Tianjin, China, 300060 Hong Kong Queen Mary Hospital Hong Kong, Hong Kong Prince of Wales Hospital Shatin, Hong Kong India Vikram Hospital Bangalore, India, 560052 HCG Hospital Bengaluru, India, 560027 P VS Hospital Pvt Ltd Calicut, India, 673002 V S Hospital Chennai, India, 600031 Max Super Speciality Hospital, Delhi Delhi, India, 110092 Global Hospitals Hyderabad, India, 500004 Basavatarakam Indo - American Cancer Hospital & Research Ins Hyderabad, India, 500034 SEAROC Cancer Centre Jaipur, India, 302004 B. P. Poddar Hospital & Medial Research Ltd Kolkata,West Bengal, India, 700053 Asirvatham Multispeciality Hospital Madurai, India, 625 020 Curie Manavata Cancer Centre Maharashtra, India, 422 004 Shatabdi Superspeciality Hospital Maharashtra, India, 422005 Prince Aly Khan Hospital Mumbai, India, 400010 Ruby Hall Clinic Pune, India, 411001 Singapore National Cancer Centre Singapore, Singapore, 169610 Taiwan E-Da Hospital Kaohsiung, Taiwan, 824 NCKUH Tainan, Taiwan, 704 Tri-Service General Hospital Taipei, Taiwan, 11490 Chang-Gung Memorial Hospital, Linkou Taoyuan County, Taiwan, 333 Collapse << |
| NCT02501603 | Gastric Cancer ... more >> Gastroesophageal Junction Cancer Collapse << | Phase 2 | Recruiting | December 2018 | Korea, Republic of ... more >> Severance Hospital, Yonsei University Health System, Yonsei Cancer Center Recruiting Seoul, Korea, Republic of, 120-752 Contact: Hyosong Kim, M.D 82-2-2228-8124 hyosong77@yuhs.ac Collapse << |
| NCT02122172 | Distal Urethral Cancer ... more >> Proximal Urethral Cancer Recurrent Bladder Cancer Recurrent Urethral Cancer Stage III Bladder Cancer Stage III Urethral Cancer Stage IV Bladder Cancer Stage IV Urethral Cancer Ureter Cancer Collapse << | Phase 2 | Recruiting | June 12, 2019 | United States, Illinois ... more >> University of Chicago Recruiting Chicago, Illinois, United States, 60637 Contact: Peter H. O'Donnell 773-702-7564 podonnel@medicine.bsd.uchicago.edu Principal Investigator: Peter H. O'Donnell Decatur Memorial Hospital Recruiting Decatur, Illinois, United States, 62526 Contact: James L. Wade 217-876-6600 JLWADE3@sbcglobal.net Principal Investigator: James L. Wade NorthShore University Health System Completed Evanston, Illinois, United States, 60201 Collapse << |
| NCT02491099 | HER2/Neu+ Uterine Serous Carci... more >>noma Collapse << | Phase 2 | Recruiting | June 2021 | United States, Arizona ... more >> University of Arizona Cancer Center Recruiting Tucson, Arizona, United States, 85724 Contact: Setsuko Chambers, M.D. 520-626-0950 schambers@uacc.arizona.edu Contact: Elizabeth Collins, BSHCA 520-694-9067 jenniecollins@email.arizona.edu United States, Connecticut Yale New Haven Hospital Recruiting New Haven, Connecticut, United States, 06510 Contact: Alessandro D. Santin, M.D. 203-737-4450 alessandro.santin@yale.edu Contact: Lisa Baker, R.N. 203-785-6398 lisa.baker@yale.edu United States, Massachusetts Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Whitfield Growdon, MD 617-724-4800 wgrowdon@partners.org Contact: Louisa Bauer, RN 617-643-5415 LJBAUER@mgh.harvard.edu Principal Investigator: Whitfield Growdon, MD Collapse << |
| NCT02274012 | HER-2 Positive Gastric Cancer ... more >> Gastrooesophageal Cancer Esophageal Cancer Collapse << | Phase 2 | Withdrawn(Former PI left insti... more >>tution and had plans to continue at another institution.) Collapse << | - | - |
| NCT01522768 | Esophageal Cancer ... more >> Gastric Cancer Collapse << | Phase 2 | Recruiting | February 2019 | United States, California ... more >> University of Southern California Recruiting Los Angeles, California, United States, 90033 Contact: Syma Iqbal, MD 323-865-3907 Principal Investigator: Syma Iqbal, MD United States, Massachusetts Massachusetts General Hospital Cancer Center Recruiting Boston, Massachusetts, United States, 02114 Contact: Janet Murphy, MD 617-724-5600 United States, New Jersey Memoral Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States Contact: Yelena Janjigian, MD Principal Investigator: Yelena Janjigian, MD Memoral Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Yelena Janjigian, MD 646-888-4186 Memorial Sloan Kettering Bergen Recruiting Montvale, New Jersey, United States, 07645 Contact: Yelena Janjigian, MD 646-888-4186 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Yelena Janjigian, MD Principal Investigator: Yelena Janjigian, MD Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Yelena Janjigian, MD 646-888-4186 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Yelena Janjigian, MD 646-888-4186 Contact: David Ilson, MD 646-888-4183 Principal Investigator: Yelena Janjigian, MD Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States Contact: Yelena Janjigian, MD Principal Investigator: Yelena Janjigian, MD Collapse << |
| NCT02369484 | NSCLC | Phase 2 | Completed | - | Germany ... more >> Universitätsklinikum Köln Köln, Germany Netherlands NKI-AVL Amsterdam, Netherlands Spain Vall d'Hebron University Hospital Barcelona, Spain Switzerland CHUV Lausanne, Switzerland USZ Zürich, Switzerland Collapse << |
| NCT02364609 | Recurrent Non-Small Cell Lung ... more >>Carcinoma Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 1 | Active, not recruiting | December 2018 | United States, California ... more >> University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 Collapse << |
| NCT02695290 | - | - | Terminated | - | - |
| NCT02514174 | Carcinoma, Non-Small-Cell Lung... more >> ErbB Receptors Collapse << | Phase 4 | Active, not recruiting | December 18, 2018 | United States, Arizona ... more >> Mayo Clinic-Phoenix Scottsdale, Arizona, United States, 85250 United States, California Pacific Cancer Medical Center, Inc. Anaheim, California, United States, 92801 City of Hope Duarte, California, United States, 91010 Compassionate Care Research Group, Inc. at Compassionate Cancer Care Medical Group, Inc. Fountain Valley, California, United States, 92708 Los Angeles Hematology Oncology Medical Group Los Angeles, California, United States, 90017 United States, Florida University of Miami Miami, Florida, United States, 33136 United States, Illinois Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 United States, Massachusetts Baystate Health D'Amour Center for Cancer Care Springfield, Massachusetts, United States, 01199 United States, New York Montefiore Medical Center Bronx, New York, United States, 10467 United States, Pennsylvania Lankenau Medical Center Wynnewood, Pennsylvania, United States, 19096 Collapse << |
| NCT02695290 | Carcinoma, Non-Small-Cell Lung... more >> ErbB Receptors Collapse << | Phase 4 | Terminated | - | United States, California ... more >> 1200.208.10032 Boehringer Ingelheim Investigational Site Fountain Valley, California, United States Collapse << |
| NCT03711422 | Non-small Cell Lung Cancer ... more >> Leptomeningeal Disease Central Nervous System Metastases Collapse << | Phase 1 | Not yet recruiting | September 30, 2020 | - |
| NCT01427478 | Head and Neck Squamous Cell Ca... more >>rcinoma Collapse << | Phase 3 | Active, not recruiting | September 2021 | France ... more >> Centre Paul Papin Angers, France, 49933 Institut Sainte-Catherine Avignon, France, 84000 CHU Bordeaux - Hôpital Saint-André Bordeaux, France, 33075 Polyclinique de Bordeaux Nord Bordeaux, France, 33300 CHRU Brest - Hôpital Morvan Brest, France, 29609 Centre François Baclesse Caen, France, 14076 CHIC Créteil Créteil, France, 94010 Centre Guillaume le Conquérant Le Havre, France, 76600 Centre Hospitalier Bretagne Sud Lorient, France, 56100 Centre Léon Bérard Lyon, France, 69373 AP-HM La Timone Adultes Marseille, France, 13386 Cedex Centre Antoine Lacassagne Nice, France, 06189 CHU Poitiers Poitiers, France, 86021 Centre Eugène Marquis Rennes, France, 44229 Centre Henri Becquerel Rouen, France, 76038 Institut de Cancérologie de la Loire Saint Priest en Jarez, France, 42270 Institut de Cancérologie de l'Ouest Saint-Herblain, France, 44805 Pôle Hospitalier Mutualiste- Centre Etienne Dolet Saint-Nazaire, France, 44600 Strasbourg Oncologie Libérale Strasbourg, France, 67000 Hopitaux du Léman Thonon-les-bains, France, 74203 Clinique Pasteur Bâtiment l'Atrium Toulouse, France, 31076 Institut Claudius Regaud Toulouse, France, 71000 CHU TOURS (Hôpital Bretonneau) Tours, France, 37044 Centre de Radiothérapie Marie Curie Valence, France, 26000 Institut de Cancérologie de lorraine (ICL) Vandoeuvre-les-Nancy, France, 54519 cedex Institut Gustave Roussy Villejuif, France, 94805 Collapse << |
| NCT01415674 | Carcinoma, Squamous Cell of He... more >>ad and Neck Collapse << | Phase 2 | Completed | - | France ... more >> CHU d'Angers Angers, France Institut de cancérologie de l'Ouest - Site Paul Papin Angers, France Centre François Baclesse Caen, France, 14000 Centre Léon Bérard Lyon, France, 69373 Institut de cancérologie de l'Ouest - Site René Gauducheau Nantes Saint-herblain, France Chu de Nantes Nantes, France Centre Antoine Lacassagne Nice, France, 06189 Institut Curie Paris, France, 75248 Institut Claudius Regaud Toulouse, France, 31052 Centre Alexis Vautrin Vandoeuvre les Nancy, France, 54511 Institut Gustave Roussy Villejuif, France, 94800 Collapse << |
| NCT02511847 | Triple Negative Breast Cancer | Phase 2 | Unknown | July 2017 | Taiwan ... more >> National Taiwan University Hospital Recruiting Taipei City, Taiwan, 110 Contact: Chiun-Sheng Huang 886-23123456 ext 65080 Collapse << |
| NCT02625168 | Lung Cancer | Phase 2 | Completed | - | Hong Kong ... more >> Department of Clinical Oncology, Queen Mary Hospital Hong Kong, Hong Kong Collapse << |
| NCT03486509 | Squamous Cell Carcinoma | Phase 2 | Not yet recruiting | July 1, 2020 | - |
| NCT02423525 | Brain Cancer | Phase 1 | Recruiting | December 2020 | United States, California ... more >> John Wayne Cancer Institute Recruiting Santa Monica, California, United States, 90404 Contact: Jaya Gill, RN 310-582-7437 jaya.gill@providence.org Principal Investigator: Santosh Kesari, MD, PhD Collapse << |
| NCT03083678 | Chordoma | Phase 2 | Recruiting | December 2019 | Italy ... more >> Istituto Nazionale dei Tumori: Fondazione IRCCS Not yet recruiting Milan, Italy Contact: S Stacchiotti, MD Netherlands Leiden University Medical Center Recruiting Leiden, Netherlands Contact: H Gelderblom, MD Prof United Kingdom University College London Hospital Not yet recruiting London, United Kingdom Contact: S Strauss, MD Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| EGFR (T790M, L858R) | IC50:10nM | |
| EGFR (L858R) | IC50:0.4nM | |
| EGFR | IC50:0.5nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
