CAS No.: 110078-46-1
普乐沙福 Catalog No. CSN12664
Synonyms: AMD 3100;JM3100;SID791
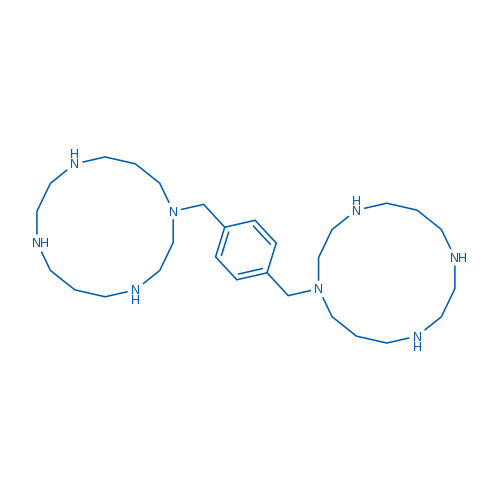
Plerixafor is a chemokine receptor antagonist for CXCR4 and CXCL12-mediated chemotaxis with IC50 of 44 nM and 5.7 nM, respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 CXCL12
IC50:5.7nMCXCR4
IC50:44nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01164475 - - Completed - - NCT01711073 End Stage Liver DIsease Phase 1 Unknown September 2013 United States, New Jersey ... more >> University of Medicine and Dentistry of New Jersey Recruiting Newark, New Jersey, United States, 07101 Contact: Baburao Koneru, MD koneruba@umdnj.edu Collapse << NCT01123499 - - Recruiting - United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: Sandra Maxwell, B.S.N. 301-402-1773 maxwells@niaid.nih.gov Collapse << - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 110078-46-1 | 储存条件 |
|
|||||||||||
| 分子式 | C28H54N8 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 502.78 | 别名 | AMD 3100;JM3100;SID791;SDZ SID 791;Mozobil;JKL 169;普乐沙福 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| CEM-SS cells | - | Function assay | - | Effective concentration of compound against HIV-1 LAI strain in CEM-SS cells, EC50=127 nM | 14698189 |
| CHOK1 cells | - | Function assay | - | Displacement of [125I]SDF1alpha from CXCR4 expressed in CHOK1 cells, IC50=0.81 nM | 17715128 |
| GHOST CXCR4 cell line | - | Function assay | - | Inhibitory concentration of compound against HIV-1 LAI strain in GHOST CXCR4 cell line, IC50=0.95 nM | 14698189 |
| HEK293 cells | - | Function assay | 2 days | Antiviral activity against T20-resistant HIV1 NL4-3 infected in HEK293 cells assessed as inhibition of viral replication after 2 days, IC50=2.3 nM | 19451305 |
| human HL60 cells | - | Function assay | - | Displacement of [125I]SDF1alpha from CXCR4 in human HL60 cells, IC50=15.2 μM | 19188071 |
| human Jurkat cells | - | Function assay | - | Antagonist activity at CXCR4 in human Jurkat cells assessed as inhibition of SDF1-induced cell migration, IC50=27.4 nM | 19188071 |
| human MOLT4 cells | 1000 nM | Function assay | - | Inhibition of Mab 12G5 binding to CXCR4 expressed in human MOLT4 cells at 1000 nM by FACS analysis | 19451305 |
| human MT2 cells | 1 ug/mL | Function assay | 4 days | Antiviral activity against HIV1 3B infected in human MT2 cells assessed as inhibition of viral p24 antigen production at 1 ug/mL after 4 days by ELISA | 21168336 |
| human MT4 cells | - | Function assay | 4 days | Antiviral activity against HIV1 3B infected in human MT4 cells assessed as inhibition of virus replication after 4 days by MTT assay, EC50=4 nM | 20043638 |
| human U87 cells | 1000 nM | Function assay | - | Antagonist activity at CXCR4 in human U87 cells assessed as inhibition of SDF1-induced modulation of cAMP production at 1000 nM by TR-FRET assay | 17958344 |
| MT-4 cells | - | Function assay | - | Effective concentration of compound against HIV-1 IIIB strain in MT-4 cells, EC50=65 nM | 14698189 |
| PBMC cells | - | Function assay | - | Effective concentration of compound against HIV-1 89.6 strain in PBMC cells, EC50=3.8 nM | 14698189 |
| rat IR983F cells | - | Function assay | - | Displacement of [125I]CXCL12 from CXCR4 in rat IR983F cells, IC50=108 nM | 19053768 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01164475 | - | - | Completed | - | - |
| NCT01711073 | End Stage Liver DIsease | Phase 1 | Unknown | September 2013 | United States, New Jersey ... more >> University of Medicine and Dentistry of New Jersey Recruiting Newark, New Jersey, United States, 07101 Contact: Baburao Koneru, MD koneruba@umdnj.edu Collapse << |
| NCT01123499 | - | - | Recruiting | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: Sandra Maxwell, B.S.N. 301-402-1773 maxwells@niaid.nih.gov Collapse << |
| NCT03055247 | Chronic Granulomatous Disease ... more >>X-linked (X-CGD) Collapse << | Phase 2 | Recruiting | August 2018 | Italy ... more >> Ospedale Pediatrico Bambino Gesù Recruiting Rome, Lazio, Italy, 00165 Contact: Franco Locatelli, MD, PhD Sub-Investigator: Andrea Finocchi, MD Ospedale San Raffaele Recruiting Milan, Lombardia, Italy, 20132 Contact: Fabio Ciceri, MD, PhD Sub-Investigator: Alessandro Aiuti, MD, PhD Sub-Investigator: Bernhard Gentner, MD, PhD Sub-Investigator: Maddalena Migliavacca, MD Sub-Investigator: Laura Bellio, MD Collapse << |
| NCT01182675 | Severe Combined Immunodeficien... more >>cy Collapse << | Phase 2 | Terminated(Insufficient eviden... more >>ce of efficacy) Collapse << | - | United States, California ... more >> UCSF Benioff Children's Hospital San Francisco, California, United States, 94143 Collapse << |
| NCT01182675 | - | - | Terminated(Insufficient eviden... more >>ce of efficacy) Collapse << | - | - |
| NCT00914849 | - | - | Completed | - | - |
| NCT00291811 | - | - | - | - | - |
| NCT02231879 | Myelokathexis ... more >> WHIMS Neutropenia Warts Collapse << | Phase 3 | Active, not recruiting | May 31, 2021 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00733824 | Lymphoma, Non-Hodgkin ... more >> Hodgkin Disease Collapse << | Phase 1 Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT00733824 | - | - | Completed | - | - |
| NCT02931071 | Fanconi Anemia | Phase 2 | Unknown | September 2018 | Spain ... more >> Hospital Universitari Vall d'Hebron Recruiting Barcelona, Spain, 08035 Contact: Cristina Díaz de Heredia, MD, PhD +34934893093 crdiaz@vhebron.net Hospital Infantil Universitario Niño Jesus Recruiting Madrid, Spain, 28009 Contact: Julian Sevilla Navarro, MD, PhD 0034 91 5035938 jsevilla.hnjs@salud.madrid.org Collapse << |
| NCT00914849 | Hematologic Neoplasms | Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT02098109 | - | - | Completed | - | - |
| NCT02098109 | Multiple Myeloma ... more >> Lymphoma, Non-Hodgkin Collapse << | Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00241358 | - | - | Completed | - | - |
| NCT01074060 | Refractory Multiple Myeloma ... more >> Stage I Multiple Myeloma Stage II Multiple Myeloma Stage III Multiple Myeloma Collapse << | Phase 1 | Completed | - | United States, California ... more >> City of Hope Duarte, California, United States, 91010 Collapse << |
| NCT00241358 | Leukemia, Myeloid, Acute ... more >> Leukemia, Myelogenous, Chronic Leukemia, Lymphoblastic, Acute Lymphocytic Leukemia, Chronic Myelodysplastic Syndromes Multiple Myeloma Lymphoma, Non-Hodgkin Hodgkin Disease Collapse << | Phase 1 Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00720603 | - | - | - | - | - |
| NCT01288573 | Ewing's Sarcoma/Soft Tissue Sa... more >>rcoma Neuroblastoma Brain Tumors Collapse << | Phase 1 Phase 2 | Completed | - | - |
| NCT01076270 | Accelerated Phase Chronic Myel... more >>ogenous Leukemia Adult Acute Lymphoblastic Leukemia in Remission Adult Acute Myeloid Leukemia in Remission Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(15;17)(q22;q12) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Atypical Chronic Myeloid Leukemia, BCR-ABL Negative Blastic Phase Chronic Myelogenous Leukemia Chronic Phase Chronic Myelogenous Leukemia de Novo Myelodysplastic Syndromes Extranodal Marginal Zone B-cell Lymphoma of Mucosa-associated Lymphoid Tissue Myelodysplastic/Myeloproliferative Neoplasm, Unclassifiable Nodal Marginal Zone B-cell Lymphoma Noncontiguous Stage II Adult Burkitt Lymphoma Noncontiguous Stage II Adult Diffuse Large Cell Lymphoma Noncontiguous Stage II Adult Diffuse Mixed Cell Lymphoma Noncontiguous Stage II Adult Diffuse Small Cleaved Cell Lymphoma Noncontiguous Stage II Adult Immunoblastic Large Cell Lymphoma Noncontiguous Stage II Adult Lymphoblastic Lymphoma Noncontiguous Stage II Grade 1 Follicular Lymphoma Noncontiguous Stage II Grade 2 Follicular Lymphoma Noncontiguous Stage II Grade 3 Follicular Lymphoma Noncontiguous Stage II Mantle Cell Lymphoma Noncontiguous Stage II Marginal Zone Lymphoma Noncontiguous Stage II Small Lymphocytic Lymphoma Previously Treated Myelodysplastic Syndromes Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Recurrent Adult Burkitt Lymphoma Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Adult Diffuse Mixed Cell Lymphoma Recurrent Adult Diffuse Small Cleaved Cell Lymphoma Recurrent Adult Hodgkin Lymphoma Recurrent Adult Immunoblastic Large Cell Lymphoma Recurrent Adult Lymphoblastic Lymphoma Recurrent Cutaneous T-cell Non-Hodgkin Lymphoma Recurrent Grade 1 Follicular Lymphoma Recurrent Grade 2 Follicular Lymphoma Recurrent Grade 3 Follicular Lymphoma Recurrent Mantle Cell Lymphoma Recurrent Marginal Zone Lymphoma Recurrent Mycosis Fungoides/Sezary Syndrome Recurrent Small Lymphocytic Lymphoma Refractory Chronic Lymphocytic Leukemia Refractory Hairy Cell Leukemia Refractory Multiple Myeloma Relapsing Chronic Myelogenous Leukemia Secondary Acute Myeloid Leukemia Secondary Myelodysplastic Syndromes Splenic Marginal Zone Lymphoma Stage I Multiple Myeloma Stage II Multiple Myeloma Stage III Adult Burkitt Lymphoma Stage III Adult Diffuse Large Cell Lymphoma Stage III Adult Diffuse Mixed Cell Lymphoma Stage III Adult Diffuse Small Cleaved Cell Lymphoma Stage III Adult Hodgkin Lymphoma Stage III Adult Immunoblastic Large Cell Lymphoma Stage III Adult Lymphoblastic Lymphoma Stage III Chronic Lymphocytic Leukemia Stage III Grade 1 Follicular Lymphoma Stage III Grade 2 Follicular Lymphoma Stage III Grade 3 Follicular Lymphoma Stage III Mantle Cell Lymphoma Stage III Marginal Zone Lymphoma Stage III Multiple Myeloma Stage III Small Lymphocytic Lymphoma Stage IV Adult Burkitt Lymphoma Stage IV Adult Diffuse Large Cell Lymphoma Stage IV Adult Diffuse Mixed Cell Lymphoma Stage IV Adult Diffuse Small Cleaved Cell Lymphoma Stage IV Adult Hodgkin Lymphoma Stage IV Adult Immunoblastic Large Cell Lymphoma Stage IV Adult Lymphoblastic Lymphoma Stage IV Chronic Lymphocytic Leukemia Stage IV Grade 1 Follicular Lymphoma Stage IV Grade 2 Follicular Lymphoma Stage IV Grade 3 Follicular Lymphoma Stage IV Mantle Cell Lymphoma Stage IV Marginal Zone Lymphoma Stage IV Small Lymphocytic Lymphoma Collapse << | Not Applicable | Terminated(Funding withdrawal) | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT01076270 | - | - | Terminated(Funding withdrawal) | - | - |
| NCT01186224 | Multiple Myeloma ... more >> Plasma Cell Dyscrasia Lymphoma Lymphoproliferative Disorders Collapse << | Not Applicable | Unknown | May 2012 | United Kingdom ... more >> Dept of Haematology, University of Liverpool Recruiting Liverpool, Merseyside, United Kingdom, L7 8XP Principal Investigator: Richard E Clark, BA, MB, BS Collapse << |
| NCT01027923 | Leukemia, Myeloid, Acute | Phase 1 | Terminated(Withdrawal of suppo... more >>rt from sponsor) Collapse << | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01225419 | Children Cancer, Solid Tumor | Phase 2 | Completed | - | France ... more >> CHU Clermont-Ferrand Clermont-Ferrand, France, 63003 Collapse << |
| NCT00444912 | Non-Hodgkin Lymphoma ... more >> Hodgkin Disease Collapse << | Phase 2 | Completed | - | United States, Georgia ... more >> Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01610999 | Chronic Lymphocytic Leukemia ... more >> Lymphoma Multiple Myeloma Collapse << | Phase 1 | Terminated(Unavailability of s... more >>tudy article) Collapse << | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT00444912 | - | - | Completed | - | - |
| NCT00103610 | Lymphoma, Non-Hodgkin | Phase 3 | Completed | - | - |
| NCT00103662 | Multiple Myeloma | Phase 3 | Completed | - | - |
| NCT00103610 | - | - | Completed | - | - |
| NCT00103662 | - | - | Completed | - | - |
| NCT00822770 | Stem Cell Transplantation ... more >> Leukemia Collapse << | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00822770 | - | - | Completed | - | - |
| NCT01220375 | Myeloma | Phase 2 | Completed | - | Switzerland ... more >> Dep. of Medical Oncology, Bern University Hospital Bern, Switzerland, 3010 Collapse << |
| NCT00943943 | Acute Myelogenous Leukemia ... more >> Leukemia Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00903968 | Multiple Myeloma | Phase 1 Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Cape Cod Hospital Hyannis, Massachusetts, United States, 02601 Milford Hospital Milford, Massachusetts, United States, 01757 Newton-Wellesley Hospital Newton, Massachusetts, United States, 02462 United States, Pennsylvania Cancer Treatment Centers of America (Eastern Regional Medical Center) Philadelphia, Pennsylvania, United States, 19124 Collapse << |
| NCT01916577 | COPD Cystic F... more >>ibrosis Pulmonary Fibrosis Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado Hospital Aurora, Colorado, United States, 80045 Collapse << |
| NCT00903968 | - | - | Completed | - | - |
| NCT01065129 | Myelodysplastic Syndromes | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01767714 | Non-Hodgkin's Lymphoma | Phase 3 | Completed | - | China ... more >> Investigational Site Number 156017 Beijing, China, 100034 Investigational Site Number 156001 Beijing, China, 100044 Investigational Site Number 156005 Beijing, China, 100071 Investigational Site Number 156002 Beijing, China, 100142 Investigational Site Number 156003 Beijing, China, 100730 Investigational Site Number 156020 Chongqing, China, 400037 Investigational Site Number 156016 Fuzhou, China, 350001 Investigational Site Number 156021 Guangzhou, China, 510060 Investigational Site Number 156011 Hangzhou, China, 310003 Investigational Site Number 156018 Nanjing, China, 210029 Investigational Site Number 156009 Shanghai, China, 200025 Investigational Site Number 156010 Suzhou, China, 215006 Investigational Site Number 156008 Tianjin, China, 300020 Investigational Site Number 156013 Wuhan, China, 430022 Investigational Site Number 156015 Xi'An, China, 710038 Investigational Site Number 156022 Zhengzhou, China, 450008 Collapse << |
| NCT01301963 | Refractory Multiple Myeloma | Phase 3 | Terminated(Slow Accrual) | - | United States, Ohio ... more >> Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01236144 | Acute Myeloid Leukaemia ... more >> High Risk Myelodysplastic Syndrome Collapse << | Phase 1 Phase 2 | Completed | - | United Kingdom ... more >> Aberdeen Royal Infirmary Aberdeen, United Kingdom Belfast City Hospital Belfast, United Kingdom Birmingham Heartlands Hospital Birmingham, United Kingdom Addenbrooke's Hospital Cambridge, United Kingdom University Hospital of Wales Cardiff, United Kingdom Castle Hill Hospital Hull, United Kingdom St James's University Hospital Leeds, United Kingdom Royal Liverpool University Hospital Liverpool, United Kingdom St Bartholomew's Hospital London, United Kingdom Christie Hospital Manchester, United Kingdom Manchester Royal Infirmary Manchester, United Kingdom Freeman Hospital Newcastle, United Kingdom Nottingham University Hospital Nottingham, United Kingdom Collapse << |
| NCT01301963 | - | - | Terminated(Slow Accrual) | - | - |
| NCT01352650 | Acute Myeloid Leukemia | Phase 1 | Completed | - | United States, New York ... more >> Weill Cornell Medical College New York, New York, United States, 10021 Collapse << |
| NCT02006225 | Autologous Stem Cell Transplan... more >>tation Collapse << | Phase 4 | Not yet recruiting | February 2020 | - |
| NCT01977677 | Adult Ependymoblastoma ... more >> Adult Giant Cell Glioblastoma Adult Glioblastoma Adult Gliosarcoma Adult Medulloblastoma Adult Mixed Glioma Adult Oligodendroglial Tumors Adult Pineoblastoma Adult Supratentorial Primitive Neuroectodermal Tumor (PNET) Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Stanford University, School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00741325 | - | - | Completed | - | - |
| NCT01160354 | Acute Myelogenous Leukemia | Phase 1 Phase 2 | Terminated(Slow Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01977677 | - | - | Completed | - | - |
| NCT01158118 | - | - | Completed | - | - |
| NCT01164475 | Non-Hodgkin's Lymphoma | Phase 4 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States, 91010 United States, Colorado University of Colorado Cancer Center Aurora, Colorado, United States, 80045 Canada, Ontario Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of, 135-710 St. Mary's Hospital Seoul, Korea, Republic of, 137-701 Taiwan National Taiwan University Hospital Taipei, Taiwan, 10002 Taipei Veterans General Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT01158118 | Leukemia, Myeloid, Acute ... more >> Myelodysplastic Syndromes Lymphoma, Non-Hodgkin Hodgkin Disease Leukemia, Lymphocytic, Chronic, B-Cell Multiple Myeloma Collapse << | Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00741780 | - | - | Completed | - | - |
| NCT00512252 | Leukemia, Myeloid, Acute | Phase 1 Phase 2 | Completed | - | United States, Missouri ... more >> Washington University St. Louis, Missouri, United States, 63110 Collapse << |
| NCT01738373 | - | - | Completed | - | France ... more >> Marseille, France Collapse << |
| NCT00512252 | - | - | Completed | - | - |
| NCT01579149 | Healthy Volunteer | Phase 1 | Completed | - | United States, Hawaii ... more >> Honolulu, Hawaii, United States Collapse << |
| NCT01206075 | Beta-Thalassemia | Not Applicable | Completed | - | Greece ... more >> George Papanicolaou Hospital Thessaloniki, Greece Collapse << |
| NCT00396383 | Multiple Myeloma | Phase 2 | Terminated(Insufficient cell m... more >>obilization for tandem transplants) Collapse << | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT01339039 | High Grade Glioma: Glioblastom... more >>a (GBM) High Grade Glioma: Gliosarcoma Anaplastic Astrocytoma (AA) Anaplastic Oligodendroglioma (AO) Mixed Anaplastic Oligoastrocytoma (AOA) Collapse << | Phase 1 | Terminated(Low accrual rate) | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02214 Collapse << |
| NCT00396383 | - | - | Terminated(Insufficient cell m... more >>obilization for tandem transplants) Collapse << | - | - |
| NCT00396201 | Hodgkin's Disease | Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine,Division of Bone Marrow Transplantation & Leukemia St Louis, Missouri, United States, 63110-1093 Collapse << |
| NCT03019809 | Wiskott-Aldrich Syndrome ... more >> Hematopoietic Stem Cell Transplantation Graft Failure Collapse << | Phase 2 | Recruiting | July 2019 | Russian Federation ... more >> Dmitry Rogachev Federal Research and Clinical Centre of Paediatric Haematology, Oncology and Immunology Recruiting Moscow, Russian Federation, 117997 Contact: Dmitry Balashov, MD, PhD +7(495)287-6570 ext 6534 bala8@yandex.ru Contact: Michael Maschan, Professor +7(926)287-6570 mmaschan@yandex.ru Sub-Investigator: Michael Maschan, Professor Sub-Investigator: Alexandra Laberko, MD Sub-Investigator: Svetlana Kozlovskaya, MD Sub-Investigator: Elena Gutovskaya, MD Sub-Investigator: Anna Shcherbina, Professor Collapse << |
| NCT02212535 | Major Sickle Cell Syndrome of ... more >>Type SS or Sβ Thalassemia Collapse << | Phase 1 Phase 2 | Completed | - | France ... more >> Hôpital Necker - Enfants Malades Paris, France, 75015 Collapse << |
| NCT00396266 | Multiple Myeloma ... more >> Lymphoma, Non-Hodgkin Collapse << | Phase 2 | Completed | - | Canada, Alberta ... more >> Tom Baker Cancer Center Calgary, Alberta, Canada, T2N 4N2 Canada, British Columbia Vancouver General Hospital, BC Cancer Agency Vancouver, British Columbia, Canada, V5Z 4E3 Collapse << |
| NCT00396201 | - | - | Completed | - | - |
| NCT00396266 | - | - | Completed | - | - |
| NCT01753453 | Multiple Myeloma | Phase 2 | Completed | - | Belgium ... more >> Investigational Site Number 056002 Brugge, Belgium, B-8000 Estonia Investigational Site Number 233001 Tallinn, Estonia, 13419 Lithuania Investigational Site Number 440001 Vilnius, Lithuania, LT-08661 Sweden Investigational Site Number 752001 Stockholm, Sweden, 14186 Investigational Site Number 752002 Umeå, Sweden, 901 85 Collapse << |
| NCT00998049 | Multiple Myeloma ... more >> Refractory Multiple Myeloma Stage I Multiple Myeloma Stage II Multiple Myeloma Stage III Multiple Myeloma Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00322842 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Collapse << | Phase 2 | Completed | - | Germany ... more >> University of Cologne Cologne, Germany Carl Gustav Carus University Hospital Dresden, Germany University of Heidelberg Heidelberg, Germany Collapse << |
| NCT02221492 | Lymphoma | Phase 2 | Completed | - | Japan ... more >> Investigational Site Number 392005 Chiba-Shi, Japan Investigational Site Number 392011 Fukuoka-Shi, Japan Investigational Site Number 392014 Fukuyama-Shi, Japan Investigational Site Number 392010 Hamamatsu-Shi, Japan Investigational Site Number 392006 Kamogawa-Shi, Japan Investigational Site Number 392003 Kobe-Shi, Japan Investigational Site Number 392008 Kurashiki-Shi, Japan Investigational Site Number 392015 Ota-Shi, Japan Investigational Site Number 392004 Sapporo-Shi, Japan Investigational Site Number 392001 Shibuya-Ku, Japan Investigational Site Number 392009 Suwa-Shi, Japan Investigational Site Number 392007 Toyohashi-Shi, Japan Collapse << |
| NCT00322842 | - | - | Completed | - | - |
| NCT00322491 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> University of Arkansas for Medical Sciences Little Rock, Arkansas, United States United States, California UCLA School of Medicine Loa Angeles, California, United States United States, Illinois Loyola University Medical Center Maywood, Illinois, United States United States, Iowa University of Iowa Iowa City, Iowa, United States United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States Mayo Clinic Rochester, Minnesota, United States United States, New York Roswell Park Cancer Institute Buffalo, New York, United States United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States Collapse << |
| NCT00322491 | - | - | Completed | - | - |
| NCT02221479 | Multiple Myeloma | Phase 2 | Completed | - | Japan ... more >> Investigational Site Number 392105 Kamogawa-Shi, Japan Investigational Site Number 392103 Kyoto-Shi, Japan Investigational Site Number 392102 Nagoya-Shi, Japan Investigational Site Number 392104 Okayama-Shi, Japan Investigational Site Number 392101 Shibuya-Ku, Japan Collapse << |
| NCT00322387 | Lymphoma, Non-Hodgkin ... more >> Multiple Myeloma Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States United States, Indiana Indiana Blood and Marrow Transplantation Beech Grove, Indiana, United States United States, New York University of Rochester Medical Center Rochester, New York, United States United States, Oregon Oregon Health and Science University Portland, Oregon, United States United States, Washington Fred Hutchinson Cancer Research Center Seattle, Washington, United States Collapse << |
| NCT00322387 | - | - | Completed | - | - |
| NCT00998049 | - | - | Completed | - | - |
| NCT00075335 | Healthy | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01037517 | Multiple Myeloma ... more >> Lymphoma Collapse << | Phase 2 | Completed | - | Canada, Manitoba ... more >> CancerCare Manitoba Winnipeg, Manitoba, Canada, R3E 0V9 Collapse << |
| NCT01818284 | Blood And Marrow Transplantati... more >>on Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00396331 | - | - | Completed | - | - |
| NCT02678533 | Fanconi Anemia | Phase 1 Phase 2 | Recruiting | March 2021 | France ... more >> Hôpital necker-Enfants malades Recruiting Paris, France, 75015 Contact: Francois LEFRERE, MD +33 1 44 49 52 74 francois.lefrere@nck.aphp.fr Collapse << |
| NCT00396331 | Autologous Stem Cell Transplan... more >>tation Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope National Medical Center` Duarte, California, United States, 91010 United States, Florida H. Lee Moffitt Cancer Center Tampa, Florida, United States, 33612-9497 United States, Georgia Blood & Marrow Transplant Group of Georgia Atlanta, Georgia, United States, 30342 United States, Mississippi University of Mississippi Medical Center, Div of Hematology Jackson, Mississippi, United States, 39216 United States, Missouri Kansas City Cancer Centers Kansas City, Missouri, United States, 64111 United States, New Jersey Hackensack University Medical Center Hackensack, New Jersey, United States, 07601 United States, Virginia Virginia Commonwealth University - Massey Cancer Center Richmond, Virginia, United States, 23298-0037 United States, Wisconsin University of Wisconsin, Blood and Bone Marrow Transplant Madison, Wisconsin, United States, 53792-5156 Collapse << |
| NCT00082329 | Healthy | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02200380 | For Donors Re... more >>lated Donors Giving Peripheral Blood Stem Cells (PBSC) to a Sibling For Recipients Acute Myelogenous Leukemia (AML) Acute Lymphoblastic Leukemia (ALL) Myelodysplastic Syndrome (MDS) Chronic Myelogenous Leukemia (CML) Non-Hodgkins Lymphoma (NHL) Hodgkins Disease (HD) Chronic Lymphocytic Leukemia (CLL) Collapse << | Phase 2 | Terminated | - | United States, California ... more >> UCLA Medical Center Los Angeles, California, United States, 90095 United States, Georgia Emory University-Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Indiana Indiana Blood and Marrow Transplant Indianapolis, Indiana, United States, 46237 United States, Iowa University of Iowa Iowa City, Iowa, United States, 52242 United States, North Carolina Wake Forest Baptist Health Winston-Salem, North Carolina, United States, 27157 United States, Ohio Ohio State University Arthur G. James Cancer Hospital and Richard J. Solove Research Institute Columbus, Ohio, United States, 43210 United States, Virginia University of Virginia Medical Center Charlottesville, Virginia, United States, 22908 Virginia Commonwealth University Medical Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT00838357 | Lymphoma (Non-Hodgkin's Lympho... more >>ma) Hodgkin's Disease or Multiple Myeloma Front Line Mobilization Transplantation Collapse << | Phase 3 | Completed | - | France ... more >> Hôpital du Haut Lévêque Bordeaux, France Hôpital Lyon Sud Lyon, France Institut Paoli Calmettes Marseille, France CHU Hotel-Dieu Université de Nantes Nantes, France Hôpital Saint-Louis Paris, France Institut Gustave Roussy Villejuif, France Germany Charité - Campus Benjamin Franklin Berlin, Germany Klinikum der Universität zu Köln Cologne, Germany Universitätsklinikum Carl Gustav Carus Dresden, Germany Universitätsklinikum Heidelberg Heidelberg, Germany Klinikum Nürnberg Nord Nürnberg, Germany Universitätsklinik Würzburg Würzburg, Germany Italy L. & A. Seragnoli, University of Bologna Bologna, Italy Ospedale Ferrarotto Catania, Italy Azienda Ospedaliera S. Martino Genova, Italy Netherlands VU Medisch Centrum Amsterdam, Netherlands Spain Hospital Santa Creu y Sant Pau Barcelona, Spain Hospital Carlos-Haya Malaga, Spain Hospital Universitario de Salamanca Salamanca, Spain Hospital la Fe Valencia, Spain Sweden Karolinska Universitetssjukhuset Huddinge Stockholm, Sweden Akademiska Sjukhuset Uppsala, Sweden United Kingdom Gartnavel Hospital Glasgow, United Kingdom St James's University Hospital Leeds, United Kingdom King's college Hospital London, United Kingdom Nottingham University NHS Trust Nottingham, United Kingdom Collapse << |
| NCT01058993 | Neutropenia | Phase 1 | Completed | - | United States, Washington ... more >> University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT00075335 | - | - | Completed | - | - |
| NCT01058993 | - | - | Completed | - | - |
| NCT01373229 | - | - | Completed | - | - |
| NCT01164345 | Non-Hodgkin's Lymphoma ... more >> Hodgkin's Lymphoma Stem Cell Mobilization Autologous Stem Cell Transplantation Collapse << | Phase 2 | Completed | - | Israel ... more >> Chaim Sheba Medical Center Tel-Hashomer, Israel Collapse << |
| NCT00082329 | - | - | Completed | - | - |
| NCT00395967 | Multiple Myeloma ... more >> Lymphoma, Non-Hodgkin's Collapse << | Phase 2 | Terminated(Enrollment terminat... more >>ed in 2005 to focus on Phase 3 study enrollment.) Collapse << | - | United States, North Carolina ... more >> Duke University Medical Center - Adult BMT Program Durham, North Carolina, United States, 27705 Collapse << |
| NCT00395967 | - | - | Terminated(Enrollment terminat... more >>ed in 2005 to focus on Phase 3 study enrollment.) Collapse << | - | - |
| NCT02069080 | Cancer | Early Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01373229 | Leukemia, Lymphocytic, Chronic... more >>, B-Cell Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT02522572 | Transplants and Implants | Phase 1 Phase 2 | Recruiting | December 2018 | United States, Ohio ... more >> The Christ Hospital Recruiting Cincinnati, Ohio, United States, 45219 Contact: E. Steve Woodle, MD 513-558-6001 Woodlees@uc.edu Sub-Investigator: Adele Rike-Shields, PharmD Sub-Investigator: Michael Cardi, MD Principal Investigator: Ervin S Woodle, MD University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45267 Contact: E. Steve Woodle, MD 513-558-6001 Woodlees@uc.edu Sub-Investigator: Rita R Alloway, PharmD Principal Investigator: Ervin S Woodle, MD Sub-Investigator: Amit Govil, MD Collapse << |
| NCT00990054 | Acute Myeloid Leukemia | Phase 1 | Completed | - | United States, California ... more >> Duarte, California, United States United States, Massachusetts Boston, Massachusetts, United States United States, Michigan Ann Arbor, Michigan, United States United States, Missouri St. Louis, Missouri, United States United States, New York Rochester, New York, United States United States, Ohio Cleveland, Ohio, United States United States, Texas Houston, Texas, United States United States, Washington Seattle, Washington, United States Collapse << |
| NCT01403896 | Malignant Lymphoma, Stem Cell ... more >>Type Collapse << | Phase 2 | Completed | - | Canada, Nova Scotia ... more >> Capital Health District Authority Halifax, Nova Scotia, Canada Collapse << |
| NCT03226691 | Sickle Cell Disease | Phase 1 | Recruiting | February 28, 2027 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov United States, Tennessee St. Jude Childrens Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Akshay Sharma Not Listed Akshay.Sharma@stjude.org Collapse << |
| NCT01095757 | - | - | Completed | - | - |
| NCT00445302 | Renal Impairment | Phase 1 | Completed | - | United States, California ... more >> Apex Research of Riverside Santa Ana, California, United States, 92705 United States, Minnesota Prism Research, 1000 Westgate Dr. suite 149 St. Paul, Minnesota, United States, 55114 United States, Nebraska Creighton University Medical Center Omaha, Nebraska, United States, 68131 Collapse << |
| NCT00694590 | Chronic Lymphocytic Leukemia (... more >>CLL) Small Lymphocytic Lymphoma (SLL) Collapse << | Phase 1 | Completed | - | United States, California ... more >> UCSD Moores Cancer Center La Jolla, California, United States United States, Ohio Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States United States, Texas UTMD Anderson Cancer Center Houston, Texas, United States Collapse << |
| NCT00901225 | Multiple Myeloma ... more >> Non-Hodgkins Lymphoma Hodgkins Disease Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27705 Collapse << |
| NCT01095757 | Myeloma Lymph... more >>oma Collapse << | Phase 2 | Completed | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01068301 | Acute Lymphoblastic Leukemia ... more >> Acute Myeloid Leukemia Chronic Myeloid Leukemia Myelodysplastic Syndrome Non-Hodgkin's Lymphoma Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT00445302 | - | - | Completed | - | - |
| NCT01700608 | - | - | Completed | - | Italy ... more >> Fondazione IRCCS Istituto Nazionale dei Tumori Milan, Italy, 20133 Collapse << |
| NCT00901225 | - | - | Completed | - | - |
| NCT03746080 | Glioblastoma ... more >>Glioblastoma With Primitive Neuronal Component Gliosarcoma Malignant Glioma Oligodendroglial Component Present Collapse << | Phase 2 | Recruiting | January 11, 2027 | United States, California ... more >> Stanford Cancer Institute Palo Alto Recruiting Palo Alto, California, United States, 94304 Contact: Lawrence Recht 650-725-8630 lrecht@stanford.edu Principal Investigator: Lawrence Recht Collapse << |
| NCT01655875 | Pediatric Acute Myeloblastic L... more >>eukemia, Relapsed Pediatric Acute Lymphoblastic Leukemia, Relapsed Collapse << | Phase 1 | Terminated(lack of patient enr... more >>ollment) Collapse << | - | United States, Georgia ... more >> Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 Collapse << |
| NCT02193191 | Sickle Cell Disease | Phase 1 | Recruiting | July 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Farid Boulad, MD 212-639-6684 Contact: Susan Prockop, MD 212-639-6715 Principal Investigator: Farid Boulad, MD Weill Cornell Medical College Recruiting New York, New York, United States, 10065 Contact: Tsiporah Shore, MD 212-746-2646 Collapse << |
| NCT03277209 | Pancreas Cancer | Phase 1 | Active, not recruiting | December 2020 | United States, New York ... more >> Weill Cornell Medical College New York, New York, United States, 10065 United Kingdom Cambridge University Hospitals NHS Foundation Trust Cambridge, United Kingdom, CB2 0QQ Collapse << |
| NCT03244930 | Lymphoma, Non-Hodgkin ... more >> Lymphoma, Hodgkin Myeloma Stem Cell Transplant Complications Collapse << | Phase 2 | Recruiting | June 10, 2018 | Mexico ... more >> Servicio de Hematología Hospital Universitario "Dr. José Eleuterio Gonzalez", Universidad Autónoma de Nuevo Leon Recruiting Monterrey, Nuevo Leon, Mexico, 64460 Contact: Perla Colunga Pedraza, MD +528110761973 colunga.perla@gmail.com Contact: Guillermo Sotomayor Duque, MD Collapse << |
| NCT02861287 | - | - | Completed | - | France ... more >> Institut Paoli-Calmettes Marseille, Bouches-du Rhône, France, 13009 Collapse << |
| NCT01280955 | Failure of Bone Marrow Graft | Phase 1 Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27705 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT01042717 | Multiple Myeloma ... more >> Non-Hodgkins Lymphoma Collapse << | Not Applicable | Unknown | December 2011 | United States, New York ... more >> Mount Sinai School of Medicine Recruiting New York, New York, United States, 10029 Contact: Patricia A Shi, MD 212-241-9237 patricia.shi@mssm.edu Contact: Luis M Isola, MD 212-241-6021 luis.isola@msnyuhealth.org Principal Investigator: Patricia A Shi, MD Sub-Investigator: Luis M Isola, MD Collapse << |
| NCT01696461 | Related Donors Donating PBSC t... more >>o a Family Member Acute Myelogenous Leukemia Acute Lymphoblastic Leukemia Myelodysplastic Syndrome Chronic Myelogenous Leukemia Non-Hodgkin's Lymphoma Hodgkin's Disease Chronic Lymphocytic Leukemia Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center Tampa, Florida, United States United States, Georgia Emory University Atlanta, Georgia, United States United States, Illinois University of Chicago Chicago, Illinois, United States United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States Mayo Clinic Rochester, Minnesota, United States United States, Missouri Washington University Saint Louis, Missouri, United States United States, North Carolina Duke University Durham, North Carolina, United States United States, Ohio Cleveland Clinic Cleveland, Ohio, United States Ohio State University Columbus, Ohio, United States United States, West Virginia West Virginia University Morgantown, West Virginia, United States United States, Wisconsin Medical College of Wisconsin Milwaukee, Wisconsin, United States Collapse << |
| NCT02179970 | Pancreatic Adenocarcinoma Meta... more >>static Ovarian Serous Adenocarcinoma Colorectal Cancer Metastatic Collapse << | Phase 1 | Recruiting | March 2019 | United Kingdom ... more >> Addenbrookes Hospital Recruiting Cambridge, United Kingdom, CB2 0QQ Contact: Lisa Bax lisa.bax@addenbrookes.nhs.uk Principal Investigator: Bristi Basu, Dr Collapse << |
| NCT02056210 | Diabetes | Phase 4 | Completed | - | Italy ... more >> Policlinico Universitario, Azienda Ospedaliera di Padova Padova, Italy, 35128 Collapse << |
| NCT02790957 | Diabetes Woun... more >>ds Critical Limb Ischemia Collapse << | Phase 2 | Recruiting | February 2020 | Italy ... more >> University Hospital of Padova Recruiting Padova, Italy, 35128 Contact: Gian Paolo Fadini, MD PhD +39-049-8214318 gianpaolo.fadini@unipd.it Collapse << |
| NCT02989701 | Sickle Cell Disease Without Cr... more >>isis Collapse << | Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston Childrens Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT03547830 | Chronic Granulomatous Disease | Phase 2 | Recruiting | January 1, 2023 | Russian Federation ... more >> Dmitry Rogachev National Research Center of Pediatric Hematology, Oncology and Immunology Recruiting Moscow, Russian Federation, 117997 Contact: Zhanna Shekhovtsova, MD 4956647078 ext 7538 zhanna.shekhovtsova@fccho-moscow.ru Contact: Eugene Pashanov, PhD +79262205578 e.pashanov@gmail.com Principal Investigator: Dmitry Balashov, MD Collapse << |
| NCT01141543 | - | - | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01149863 | Autologous Transplantation | Phase 2 | Completed | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT01149863 | - | - | Completed | - | - |
| NCT01280955 | - | - | Completed | - | - |
| NCT03664830 | Sickle Cell Disease | Phase 1 | Recruiting | September 2020 | United States, California ... more >> City of Hope Medical Center Recruiting Duarte, California, United States, 91010 Contact: Joseph Rosenthal, MD 626-256-4673 ext 88442 jrosenthal@coh.org Principal Investigator: Joseph Rosenthal, MD Collapse << |
| NCT01455025 | Acute Myeloid Leukemia | Phase 1 | Terminated(no recruitment on t... more >>ime) Collapse << | - | France ... more >> Xavier THOMAS Lyon, France, 69437 Collapse << |
| NCT01954914 | Blood Stem Cell Harvest Failur... more >>e Collapse << | Phase 2 | Completed | - | Germany ... more >> Cellex Gesellschaft für Zellgewinnung mbH Dresden Dresden, Germany, 01307 Universitätsklinikum Dresden, Medizinische Klinik I Dresden, Germany, 01307 Cellex Gesellschaft für Zellgewinnung mbH Köln Köln, Germany, 50670 Collapse << |
| NCT01319864 | Relapsed/Refractory AML ... more >> Relapsed/Refractory ALL Secondary AML/MDS Acute Leukemia of Ambiguous Lineage AML ALL Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Phoenix Children's Hospital Phoenix, Arizona, United States, 85016 United States, Colorado The Children's Hospital of Denver Denver, Colorado, United States, 80045 United States, Georgia Children's Healthcare of Atlanta/Emory University Atlanta, Georgia, United States, 30322 United States, Maryland Johns Hopkins Medical Center Baltimore, Maryland, United States, 21231 United States, Missouri The Children's Mercy Hospital and Clinics Kansas City, Missouri, United States, 64108 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Penn State Hershey Children's Hospital Hershey, Pennsylvania, United States, 17033 Canada, Alberta Alberta Children's Hospital Calgary, Alberta, Canada, T3B 6A8 Collapse << |
| NCT01339572 | Non-Hodgkin's Lymphoma ... more >> Multiple Myeloma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Shands Cancer Hospital at the University of Florida Gainesville, Florida, United States, 32608 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| CXCL12 | IC50:5.7nM | |
| CXCR4 | IC50:44nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
