CAS No.: 117570-53-3
2,5-己酮可可碱 Catalog No. CSN10852
Synonyms: ASA-404;NSC-640488;VadimezanVadimezan
DMXAA is an apoptosis inducer with anti-vascular activity. It is mSTING agonist, selective for mouse STING over human STING. It Induces IFN-β and cytokine production from bone marrow-derived dendritic cells.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
动物研究 剂量 Mice: 17.5 mg/kg, 20 mg/kg[3] (i.p.); 30 mg/kg[4] (p.o.)
Rat: 100 mg/kg - 350 mg/kg[5] (i.p.)给药途径 i.p., p.o.
动物药代数据 Animal Mice[4] Dose 30 mg/kg (p.o.)
25 mg/kg (i.p. or i.v.)Administration p.o.
i.p. or i.v.Cmax 358 ± 25 μmol/l (p.o.)
570 ± 21 μmol/l (i.p.)
470 ± 12 μmol/l (i.v.)T1/2 9.2 h (p.o.)
2.7 h (i.p.)
3.3 h (i.v.)AUC 1392 μmol·h/l (p.o.)
1715 μmol·h/l (i.p.)
1595 μmol·h/l (i.v.)- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01031212 Tumors Phase 1 Withdrawn(The investigator has... more >> left the institution (UCSF) prior to study start-up) Collapse << June 2013 United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 Collapse << NCT00662597 Non-Small Cell Lung Cancer Phase 3 Terminated - - NCT01057342 Lung Cancer Phase 2 Completed - Switzerland ... more >> Saint Claraspital AG Basel, Switzerland, CH-4016 Universitaetsspital-Basel Basel, Switzerland, CH-4031 Istituto Oncologico della Svizzera Italiana - Ospedale San Giovanni Bellinzona, Switzerland, CH-6500 Inselspital Bern Bern, Switzerland, CH-3010 Spitalzentrum Biel Biel, Switzerland, CH-2501 Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland, CH-1011 Kantonsspital Olten Olten, Switzerland, CH-4600 Onkologie Schaffhausen Schaffhausen, Switzerland, CH-8200 Kantonsspital - St. Gallen St. Gallen, Switzerland, CH-9007 Regionalspital Thun, Switzerland, 3600 Kantonsspital Winterthur Winterthur, Switzerland, CH-8400 Klinik Hirslanden Zurich, Switzerland, CH-8032 Collapse << - 更多
- 参考文献
- [1] Phillips RM. Inhibition of DT-diaphorase (NAD(P)H:quinone oxidoreductase, EC 1.6.99.2) by 5,6-dimethylxanthenone-4-acetic acid (DMXAA) and flavone-8-acetic acid (FAA): implications for bioreductive drug development. Biochem Pharmacol. 1999 Jul 15;58(2):303-10.
- [2] Buchanan CM, Shih JH, Astin JW, Rewcastle GW, Flanagan JU, Crosier PS, Shepherd PR. DMXAA (Vadimezan, ASA404) is a multi-kinase inhibitor targeting VEGFR2 in particular. Clin Sci (Lond). 2012 May 1;122(10):449-57.
- [3] Liu JJ, Ching LM, Goldthorpe M, Sutherland R, Baguley BC, Kirker JA, McKeage MJ. Antitumour action of 5,6-dimethylxanthenone-4-acetic acid in rats bearing chemically induced primary mammary tumours. Cancer Chemother Pharmacol. 2007 Apr;59(5):661-9.
- [4] Zhao L, Kestell P, et al. Oral activity and pharmacokinetics of 5,6-dimethylxanthenone-4-acetic acid (DMXAA) in mice. Cancer Chemother Pharmacol. 2002 Jan;49(1):20-6.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 117570-53-3 | 储存条件 |
|
|||||||||||||
| 分子式 | C17H14O4 | 运输 | 蓝冰 | |||||||||||||
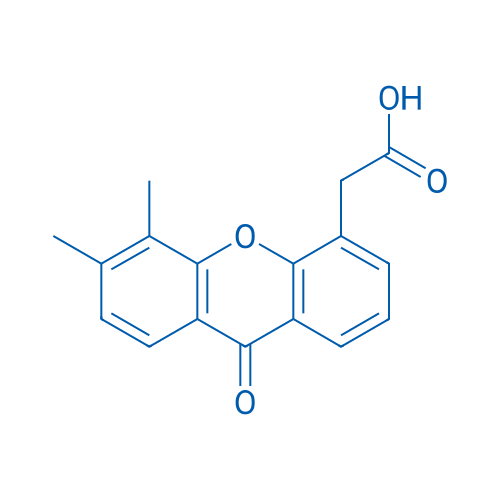
| 分子量 | 282.29 | 别名 | ASA-404;NSC-640488;VadimezanVadimezan;5,6-Dimethylxanthenone-4-acetic Acid;DMXAA | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
