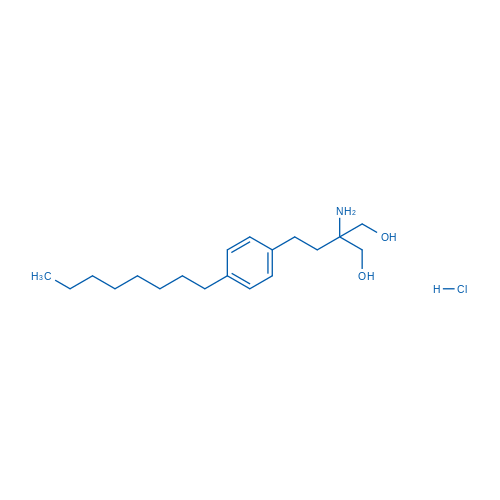
CAS No.: 162359-56-0
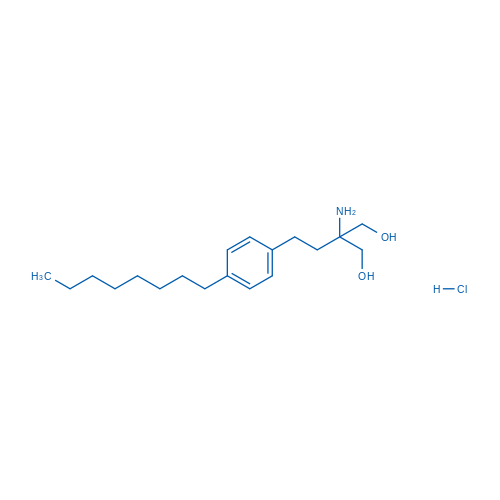
盐酸芬戈莫德 Catalog No. CSN16720
Synonyms: FTY720;Fingolimod (hydrochloride);FTY 720. Fingolimod
Fingolimod HCl is a S1P antagonist with IC50 of 0.033 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 S1PR1
IC50:0.033nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice: 1 mg/kg - 10 mg/kg[5] (i.p.), 0.03 mg/kg - 1 mg/kg[4] (p.o.) Rat: 3 mg/kg - 7.5 mg/kg[6] (i.v.)
给药途径 i.p., p.o., i.v.
动物药代数据 Animal Mice[6] Rats[6] Dogs[6] Monkeys[6] Dose 2.5 mg/kg (p.o. or i.v.) 7.5 mg/kg (p.o.)
4 mg/kg (i.v.)3 mg/kg (p.o.)
1 mg/kg (i.v.)3 mg/kg (p.o.)
0.1 mg/kg (i.v.)Administration p.o.
i.v.p.o.
i.v.p.o.
i.v.F 66% (p.o.) 59% (p.o.) 60.8% (p.o.) 51% (p.o.) T1/2 25 h (p.o.) 29.3 h (p.o.) Tmax 8 h (p.o.) 8 h (p.o.) 9.3 h (p.o.) 9.3 h (p.o.) MRT 31.5 h (i.v.) 32 h (p.o.) 34.7 h (i.v.) t1/2β 24.6 h (i.v.) T1/2β 27.8 h (i.v.) Cmax 95.3 ng/ml (p.o.) 148 ng/ml (p.o.) 165 ng/ml (p.o.) 30.7 ng/ml (p.o.) CL 0.55 L/h/kg (i.v.)
9.17 ml/min/kg (i.v.)0.666 L/h/kg (i.v.)
11.1 ml/min/kg (i.v.)0.219 L/h/kg (i.v.)
3.65 ml/min/kg (i.v.)0.0917 L/h/kg (i.v.)
1.528 ml/min/kg (i.v.)Vss 17.30 L/kg (i.v.) 21 L/kg (p.o.) 7.60 L/kg (i.v.) 17.3 L/kg (i.v.) AUC 3167 ng·h/ml (p.o.)
5442 ng·h/ml (i.v.)6795 ng·h/ml (p.o.)
6027 ng·h/ml (i.v.)8491 ng·h/ml (p.o.)
4658 ng·h/ml (i.v.)1640 ng·h/ml (p.o.)
323 ng·h/ml (i.v.)- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02559765 - - Completed - United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << NCT03500328 Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << Not Applicable Recruiting October 31, 2022 - NCT03535298 Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << Phase 4 Not yet recruiting September 2023 United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Tammy Skaramagas, BA 216-445-6724 skaramt1@ccf.org Principal Investigator: Daniel Ontaneda, MD, MSc United Kingdom University of Nottingham Not yet recruiting Nottingham, United Kingdom, NG7 2UH Contact: Sara Wilkins +44 115 9249924 ext 66816 Sara.wilkins@nuh.nhs.uk Principal Investigator: Nikos Evangelou, MD, DPhil Collapse << - 更多
- 参考文献
- [1] Brinkmann V, Davis MD, et al. The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J Biol Chem. 2002 Jun 14;277(24):21453-7. Epub 2002 Apr 19.
- [2] Oo ML, Thangada S, et al. Immunosuppressive and anti-angiogenic sphingosine 1-phosphate receptor-1 agonists induce ubiquitinylation and proteasomal degradation of the receptor. J Biol Chem. 2007 Mar 23;282(12):9082-9. Epub 2007 Jan 21.
- [3] Coelho RP, Payne SG, et al. The immunomodulator FTY720 has a direct cytoprotective effect in oligodendrocyte progenitors. J Pharmacol Exp Ther. 2007 Nov;323(2):626-35. Epub 2007 Aug 28.
- [4] Chiba K, Kataoka H, et al. Fingolimod (FTY720), sphingosine 1-phosphate receptor modulator, shows superior efficacy as compared with interferon-β in mouse experimental autoimmune encephalomyelitis. Int Immunopharmacol. 2011 Mar;11(3):366-72.
- [5] Blanc CA, Rosen H, et al. FTY720 (fingolimod) modulates the severity of viral-induced encephalomyelitis and demyelination. J Neuroinflammation. 2014 Aug 20;11:138.
- [6] FTY720
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 162359-56-0 | 储存条件 |
|
|||||||
| 分子式 | C19H34ClNO2 | 运输 | 蓝冰 | |||||||
| 分子量 | 343.93 | 别名 | FTY720;Fingolimod (hydrochloride);FTY 720. Fingolimod;Fingolimod HCl;Band name: Gilenia and Gilenya;盐酸芬戈莫德 | |||||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| CHO cells | - | Function assay | - | Displacement of [33P]sphingosine 1 phosphate from human S1P1 receptor expressed in CHO cells, IC50=0.84 μM. | 15615513 |
| human BLIN-1 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against Ph-negative human BLIN-1 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=5.5 μM. | 24273632 |
| human BV173 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against Ph-positive human BV173 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=6.3 μM. | 24273632 |
| human CCRF-CEM cells | - | Cytotoxic assay | 72 h | Cytotoxicity against Ph-negative human CCRF-CEM cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=6.8 μM. | 24273632 |
| human DU145 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human DU145 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=6.5 μM. | 24273632 |
| human HCT116 cells | - | Proliferation assay | 78 h | Antiproliferative activity against human HCT116 cells assessed as growth inhibition after 78 hrs by WST-1 assay, IC50=5 μM. | 21456524 |
| human Jurkat cells | - | Function assay | 18 h | Reversal of inhibition of mitochondrial function in human Jurkat cells after 18 hrs in presence of Z-VAD-fmk | 17400555 |
| human MCF7 cells | - | Proliferation assay | 78 h | Antiproliferative activity against human MCF7 cells assessed as growth inhibition after 78 hrs by WST-1 assay, IC50=5 μM. | 21456524 |
| human NALM6 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against Ph-negative human NALM6 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=9.6 μM. | 24273632 |
| human PC3 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against human PC3 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=9.8 μM. | 24273632 |
| human SK-BR-3 cells | - | Proliferation assay | 78 h | Antiproliferative activity against human SK-BR-3 cells assessed as growth inhibition after 78 hrs by WST-1 assay, IC50= 5 μM. | 21456524 |
| human SUP-B15 cells | - | Cytotoxic assay | 72 h | Cytotoxicity against Ph-positive human SUP-B15 cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=6.8 μM. | 24273632 |
| human SW620 cells | - | Proliferation assay | 78 h | Antiproliferative activity against human SW620 cells assessed as growth inhibition after 78 hrs by WST-1 assay, IC50=5 μM. | 21456524 |
| human U2OS cells | - | Function assay | - | Agonist activity at human S1P1 receptor expressed in human U2OS cells co-expressing eGFP assessed as receptor internalization into cytoplasm using Hoechst dye staining, EC50=0.002 μM. | 22104144 |
| MDA-MB-231 cells | - | Proliferation assay | 78 h | Antiproliferative activity against human MDA-MB-231 cells assessed as growth inhibition after 78 hrs by WST-1 assay, IC50= 5 μM. | 21456524 |
| mouse bone marrow cells | - | Cytotoxic assay | 72 h | Cytotoxicity against BCR-ABL fusion protein 190 expressing mouse bone marrow cells after 72 hrs by vital dye exclusion/flow cytometric analysis, IC50=3.3 μM. | 24273632 |
| mouse MN9D cells | 0.16 μM | Function assay | 72 h | Neuroprotective activity in mouse MN9D cells assessed as blocking of TNF-alpha associated toxicity at 0.16 uM after 72 hrs by Trypan blue staining. | 25050165 |
| mouse MN9D cells | 0.16 μM | Function assay | 24 h | Neuroprotective activity in mouse MN9D cells assessed as stimulation of BDNF expression at 0.16 uM after 24 hrs | 25050165 |
| mouse MN9D cells | 5 μM | Function assay | - | Induction of PP2A catalytic subunit activity in mouse MN9D cells assessed as phosphate level at 5 uM | 25050165 |
| rat PC12 cells | 5 μM | Function assay | 30 to 120 mins | Induction of PP2A catalytic subunit activity in rat PC12 cells assessed as phosphate level at 5 uM measured 30 to 120 mins. | 25050165 |
| Sf9 insect cells | - | Function assay | 1 h | Inhibition of human recombinant S1PL (62 to 568) expressed in Sf9 insect cells using S1P as substrate after 1 hr | 24809814 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02559765 | - | - | Completed | - | United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << |
| NCT03500328 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Not Applicable | Recruiting | October 31, 2022 | - |
| NCT03535298 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Not yet recruiting | September 2023 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Tammy Skaramagas, BA 216-445-6724 skaramt1@ccf.org Principal Investigator: Daniel Ontaneda, MD, MSc United Kingdom University of Nottingham Not yet recruiting Nottingham, United Kingdom, NG7 2UH Contact: Sara Wilkins +44 115 9249924 ext 66816 Sara.wilkins@nuh.nhs.uk Principal Investigator: Nikos Evangelou, MD, DPhil Collapse << |
| NCT00273364 | Multiple Sclerosis | Phase 2 | Active, not recruiting | December 2018 | United States, Illinois ... more >> Northwestern University, Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT01795872 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT03193866 | - | - | Recruiting | December 2021 | Sweden ... more >> Fredrik Piehl Recruiting Stockholm, Sweden Contact: Fredrik Piehl, Professor +46 73-671 81 01 Fredrik.Piehl@ki.se Contact: Anna Fogdell-Hahn, PhD +46 70-544 90 60 anna.fogdell-hahn@ki.se Collapse << |
| NCT02139696 | - | - | Completed | - | Finland ... more >> Turku University Hospital Turku, Finland, 20520 Collapse << |
| NCT03399981 | - | - | Active, not recruiting | December 31, 2023 | United States, Massachusetts ... more >> Research Site Cambridge, Massachusetts, United States, 02142 Collapse << |
| NCT02241785 | - | - | Terminated(Business Decision) | - | - |
| NCT02193217 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Investigational site Leeds, United Kingdom Collapse << |
| NCT02241785 | Relapsing Multiple Sclerosis | Phase 4 | Terminated(Business Decision) | - | United States, California ... more >> Research Site Fullerton, California, United States, 92835 United States, Colorado Research Site Aurora, Colorado, United States, 80045 United States, Iowa Research Site Des Moines, Iowa, United States, 50314 United States, Missouri Research Site Saint Louis, Missouri, United States, 63110 United States, New York Research Site Plainview, New York, United States, 11803 United States, North Carolina Research Site Raleigh, North Carolina, United States, 27607-6010 United States, Ohio Research Site Akron, Ohio, United States, 44320 Research Site Cleveland, Ohio, United States, 44195 United States, Tennessee Research Site Knoxville, Tennessee, United States, 37922 United States, Texas Research Site Round Rock, Texas, United States, 78681 United States, Washington Research Site Tacoma, Washington, United States, 98405 Collapse << |
| NCT02408380 | - | - | Unknown | December 2015 | Canada, Quebec ... more >> McGill University Recruiting Montreal, Quebec, Canada, H3A 2B4 Contact: Leslie Fitz-Gerald, MSc 5143987192 ext 00510 leslie.fitz-gerald@mail.mcgill.ca Principal Investigator: David Haegert, MD Collapse << |
| NCT02776072 | - | - | Completed | - | - |
| NCT01592097 | - | - | Completed | - | United States, California ... more >> Keck School of Medicine of the University of Southern Calfornia Los Angeles, California, United States, 90033 Collapse << |
| NCT02335892 | - | - | Completed | - | United Kingdom ... more >> Novartis Investigative Site Truro, Cornwall, United Kingdom, TR1 3LJ Novartis Investigative Site Swansea, England, United Kingdom, SA6 6NL Novartis Investigative Site Romford, Essex, United Kingdom, RM7 0AG Novartis Investigative Site Basingstoke, Hampshire, United Kingdom, RG24 9NA Novartis Investigative Site Edinburgh, United Kingdom, EH16 4SA Novartis Investigative Site Glasgow, United Kingdom, G51 4TF Novartis Investigative Site Leicester, United Kingdom, LE5 4PW Novartis Investigative Site Liverpool, United Kingdom, L9 7LT Novartis Investigative Site London, United Kingdom, E1 1BB Novartis Investigative Site London, United Kingdom, SW17 0QT Novartis Investigative Site London, United Kingdom, W6 8RF Novartis Investigative Site London, United Kingdom, WC1N 3BG Novartis Investigative Site Norwich, United Kingdom, NR4 7UY Novartis Investigative Site Nottingham, United Kingdom, NG5 1PB Collapse << |
| NCT01709812 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Withdrawn | July 2013 | - |
| NCT01490840 | Fatigue in Multiple Sclerosis | Phase 4 | Terminated(The study stopped d... more >>ue to slow enrollment and low participant compliance.) Collapse << | - | Germany ... more >> Novartis Investigative Site Aachen, Germany, 52062 Novartis Investigative Site Alzenau, Germany, 63755 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Bad Mergentheim, Germany, 97980 Novartis Investigative Site Bamberg, Germany, 96052 Novartis Investigative Site Bayreuth, Germany, 95445 Novartis Investigative Site Berlin, Germany, 10437 Novartis Investigative Site Berlin, Germany, 10625 Novartis Investigative Site Berlin, Germany, 10691 Novartis Investigative Site Berlin, Germany, 12587 Novartis Investigative Site Berlin, Germany, 13347 Novartis Investigative Site Bielefeld, Germany, 33647 Novartis Investigative Site Bochum, Germany, 44787 Novartis Investigative Site Böblingen, Germany, 71032 Novartis Investigative Site Dortmund, Germany, 44135 Novartis Investigative Site Erbach, Germany, 64711 Novartis Investigative Site Erlangen, Germany, 91054 Novartis Investigative Site Frankfurt, Germany, 65929 Novartis Investigative Site Hamburg, Germany, 22179 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Jena, Germany, 07740 Novartis Investigative Site Kassel, Germany, 34121 Novartis Investigative Site Leipzig, Germany, 04299 Novartis Investigative Site Potsdam, Germany, 14471 Novartis Investigative Site Prien, Germany, 83209 Novartis Investigative Site Regensburg, Germany, 93053 Novartis Investigative Site Siegen, Germany, 57076 Novartis Investigative Site Sinsheim, Germany, 74889 Novartis Investigative Site Stuttgart, Germany, 70174 Novartis Investigative Site Stuttgart, Germany, 70178 Novartis Investigative Site Teupitz, Germany, 15755 Novartis Investigative Site Unterhaching, Germany, 82008 Collapse << |
| NCT02720107 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Ostfildern, Baden-Wuerttemberg, Germany, 73760 Novartis Investigative Site Altenholz-Stift, Germany, 24161 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Berlin, Germany, 10713 Novartis Investigative Site Berlin, Germany, 12163 Novartis Investigative Site Berlin, Germany, 13347 Novartis Investigative Site Böblingen, Germany, 71032 Novartis Investigative Site Celle, Germany, 29223 Novartis Investigative Site Dortmund, Germany, 44137 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Erbach, Germany, 64711 Novartis Investigative Site Frankfurt, Germany, 60313 Novartis Investigative Site Göttingen, Germany, 37073 Novartis Investigative Site Jena, Germany, 07740 Novartis Investigative Site Kiel, Germany, 24105 Novartis Investigative Site Klingenmünster, Germany, 76889 Novartis Investigative Site Lappersdorf, Germany, 93138 Novartis Investigative Site Leverkusen, Germany, 51375 Novartis Investigative Site Mönchengladbach, Germany, 41239 Novartis Investigative Site München, Germany, 81829 Novartis Investigative Site Neuburg an der Donau, Germany, 86633 Novartis Investigative Site Siegen, Germany, 57076 Novartis Investigative Site Singen, Germany, 78224 Novartis Investigative Site Troisdorf, Germany, 53844 Novartis Investigative Site Ulm, Germany, 89073 Novartis Investigative Site Unterhaching, Germany, 82008 Collapse << |
| NCT01490840 | - | - | Terminated(The study stopped d... more >>ue to slow enrollment and low participant compliance.) Collapse << | - | - |
| NCT01704183 | - | - | Completed | - | Finland ... more >> Neuro Center, Kuopio University Hospital Kuopio, Finland Department of Neurology, Mikkeli Central Hospital Mikkeli, Finland Collapse << |
| NCT02277964 | - | - | Completed | - | Switzerland ... more >> University Hospital Basel, Switzerland Basel, Switzerland, 4031 Collapse << |
| NCT02141022 | Multiple Sclerosis ... more >> Cognitive Deficits Gilenya Modifying Therapy for MS Collapse << | Not Applicable | Completed | - | United States, New York ... more >> Stony Brook University Stony Brook, New York, United States, 11794 Collapse << |
| NCT02307838 | Multiple Sclerosis, Relapsing ... more >>Forms of Multiple Sclerosis Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Novartis Investigative Site Ottawa, Ontario, Canada, K1H 8L6 Novartis Investigative Site Toronto, Ontario, Canada, M5B 1N9 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H2L 4M1 Novartis Investigative Site Montreal, Quebec, Canada, H3A 2B4 Denmark Novartis Investigative Site Copenhagen, Denmark, DK-2100 Novartis Investigative Site Glostrup, Denmark, DK-2600 France Novartis Investigative Site Lille, France, 59037 Novartis Investigative Site Marseille, France, 13385 Germany Novartis Investigative Site Würzburg, Germany, 97080 Italy Novartis Investigative Site Genova, GE, Italy, 16132 Novartis Investigative Site Milano, MI, Italy, 20132 Novartis Investigative Site Roma, RM, Italy, 00189 Novartis Investigative Site Gallarate, VA, Italy, 21013 Poland Novartis Investigative Site Warszawa, Poland, 02-097 Novartis Investigative Site Warszawa, Poland, 02-957 Portugal Novartis Investigative Site Coimbra, Portugal, 3000-075 Novartis Investigative Site Lisboa, Portugal, 1150-314 Spain Novartis Investigative Site Malaga, Andalucia, Spain, 29010 Novartis Investigative Site Sevilla, Andalucia, Spain, 41009 Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Novartis Investigative Site L'Hospitalet de Llobregat, Catalunya, Spain, 08907 Novartis Investigative Site Valencia, Comunidad Valenciana, Spain, 46026 Novartis Investigative Site Madrid, Spain, 28040 Switzerland Novartis Investigative Site Basel, Switzerland, 4031 Novartis Investigative Site Zuerich, Switzerland, 8091 United Kingdom Novartis Investigative Site Newcastle Upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT02307877 | - | - | Active, not recruiting | December 2019 | United States, Colorado ... more >> University of Colorado Hospital Aurora, Colorado, United States, 80045 Collapse << |
| NCT00239863 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT01705236 | Relapsing Remitting Multiple S... more >>clerosis RRMS Collapse << | Phase 4 | Active, not recruiting | January 28, 2019 | Germany ... more >> Novartis Investigative Site Bochum, Germany, 44791 Novartis Investigative Site Bonn, Germany, 53105 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Duesseldorf, Germany, 40225 Novartis Investigative Site Hannover, Germany, 30623 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Leipzig, Germany, 04103 Novartis Investigative Site Rostock, Germany, 18057 Novartis Investigative Site Ulm, Germany, 89081 Switzerland Novartis Investigative Site Zuerich, Switzerland, 8091 Collapse << |
| NCT00239785 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT02307838 | - | - | Completed | - | - |
| NCT00239798 | Renal Transplantation | Phase 2 | Completed | - | - |
| NCT00239902 | Renal Transplantation | Phase 2 | Completed | - | - |
| NCT00098735 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT00099749 | Kidney Transplantation | Phase 2 Phase 3 | Completed | - | United States, Illinois ... more >> Loyola University Medical Center Maywood, Illinois, United States, 60153 SIU School of Medicine Springfield, Illinois, United States, 62781 United States, New Jersey St. Barnabas Medical Center Livingston, New Jersey, United States, 07039 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Rhode Island Rhode Island Hospital Providence, Rhode Island, United States, 02903 Collapse << |
| NCT01285479 | - | - | Recruiting | December 13, 2030 | - |
| NCT00099801 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT00239811 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT02769689 | Multiple Sclerosis | Phase 4 | Unknown | November 2017 | France ... more >> CHU Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 04 73 75 11 95 placarin@chu-clermontferrand.fr Principal Investigator: Pierre CLAVELOU Collapse << |
| NCT01333501 | Multiple Sclerosis | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Alzenau, Germany, 63755 Novartis Investigative Site Bamberg, Germany, 96049 Novartis Investigative Site Leipzig, Germany, 04103 Novartis Investigative Site Würzburg, Germany, 97080 Italy Novartis Investigative Site Ancona, AN, Italy, 60126 Novartis Investigative Site Bari, BA, Italy, 70124 Novartis Investigative Site Bergamo, BG, Italy, 24128 Novartis Investigative Site Montichiari, BS, Italy, 25018 Novartis Investigative Site Chieti, CH, Italy, 66100 Novartis Investigative Site Catania, CT, Italy, 95122 Novartis Investigative Site Catania, CT, Italy, 95123 Novartis Investigative Site Firenze, FI, Italy, 50134 Novartis Investigative Site Genova, GE, Italy, 16132 Novartis Investigative Site Pozzilli, IS, Italy, 86077 Novartis Investigative Site Messina, ME, Italy, 98121 Novartis Investigative Site Milano, MI, Italy, 20122 Novartis Investigative Site Milano, MI, Italy, 20132 Novartis Investigative Site Cefalù, PA, Italy, 90015 Novartis Investigative Site Pavia, PV, Italy, 27100 Novartis Investigative Site Roma, RM, Italy, 00133 Novartis Investigative Site Roma, RM, Italy, 00161 Novartis Investigative Site Roma, RM, Italy, 00179 Novartis Investigative Site Roma, RM, Italy, 00189 Novartis Investigative Site Gallarate, VA, Italy, 21013 Novartis Investigative Site Napoli, Italy, 80131 Novartis Investigative Site Napoli, Italy, 80138 Collapse << |
| NCT01981161 | - | - | Recruiting | November 2017 | France ... more >> CHU Besançon Not yet recruiting Besancon, France, 25000 Contact: Eric Berger, MD lrumbach@chu-besancon.fr Principal Investigator: Eric Berger, MD CHU Bordeaux Not yet recruiting Bordeaux, France, 33 000 Contact: Bruno Brochet, MD PHD bruno.brochet@chu-bordeaux.fr Principal Investigator: bruno brochet, MD PHD CHU Caen Not yet recruiting Caen, France, 14033 Contact: Gilles-Louis Defer, MD PHD defer-gi@chu-caen.fr Principal Investigator: Gilles-Louis Defer, MD PHD Chu Lille Not yet recruiting Lille, France, 59037 Contact: Patrick Vermersch, MD PHD Principal Investigator: Patrick Vermersch, MD PHD Chu La Timone Not yet recruiting Marseille, France, 13385 Contact: Jean Pelletier, MD PHD Principal Investigator: Jean Pelletier, MD PHD CHU Nancy Not yet recruiting Nancy, France, 54035 Contact: Marc Debouverie, MD PHD Principal Investigator: Marc Debouverie, MD PHD CHU Nantes Recruiting Nantes, France, 44093 Contact: David Laplaud, MD david.laplaud@chu-nantes.fr Principal Investigator: David Laplaud, MD CHU Nice Not yet recruiting Nice, France, 06002 Contact: Christine Lebrun-Frenay, MD Principal Investigator: Christine Lebrun-Frenay, MD CHU Nîmes Not yet recruiting Nimes, France, 30029 Contact: Pierre Labauge, MD PHD Principal Investigator: Labauge Pierre, MD PHD CHRU Strasbourg Not yet recruiting Strasbourg, France, 67098 Contact: Jérôme De Seze, MD pHD Principal Investigator: Jerome De Seze, MD PHD Germany Université de Muenter Not yet recruiting Munster, Germany Contact: H Wiendl, MD PHD Principal Investigator: H Wiendl, MD PHD Spain Hôpital Vall D'Hebron Not yet recruiting Barcelone, Spain Contact: Xavier Montalban, MD PHD Principal Investigator: XAVIER Montalban, MD PHD Collapse << |
| NCT01333501 | - | - | Completed | - | - |
| NCT01647880 | Multiple Sclerosis | Phase 2 Phase 3 | Terminated(delayed recruitment... more >>) Collapse << | - | Germany ... more >> Charité-Universitätsmedizin Berlin Berlin, Germany Sankt Josefs Krankenhaus Potsdam Neurologie Potsdam, Germany Collapse << |
| NCT01892722 | Multiple Sclerosis | Phase 3 | Active, not recruiting | March 15, 2023 | - |
| NCT00099736 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT01892722 | - | - | Active, not recruiting | - | - |
| NCT00239876 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT01436643 | - | - | Terminated(Due to slow enrollm... more >>ent the study was terminated early) Collapse << | - | - |
| NCT01127750 | Relapsing Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01317004 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Italy ... more >> Novartis Investigative Site Ancona, AN, Italy, 60126 Novartis Investigative Site Ponderano, BI, Italy, 13900 Novartis Investigative Site Caltanissetta, CL, Italy, 93100 Novartis Investigative Site Cuneo, CN, Italy, 12100 Novartis Investigative Site Como, CO, Italy, 22100 Novartis Investigative Site Catania, CT, Italy, 95122 Novartis Investigative Site Foggia, FG, Italy, 71100 Novartis Investigative Site Castelfiorentino, FI, Italy, 50051 Novartis Investigative Site Milano, MI, Italy, 20122 Novartis Investigative Site Milano, MI, Italy, 20133 Novartis Investigative Site San Donato Milanese, MI, Italy, 20097 Novartis Investigative Site Modena, MO, Italy, 41100 Novartis Investigative Site Palermo, PA, Italy, 90129 Novartis Investigative Site Palermo, PA, Italy, 90146 Novartis Investigative Site Pisa, PI, Italy, 56126 Novartis Investigative Site Legnago, VR, Italy, 37045 Novartis Investigative Site Novara, Italy, 28100 Collapse << |
| NCT01436643 | Depression Re... more >>lapsing-remitting Multiple Sclerosis Collapse << | Phase 4 | Terminated(Due to slow enrollm... more >>ent the study was terminated early) Collapse << | - | Germany ... more >> Novartis Investigative Site Achim, Germany, 28832 Novartis Investigative Site Altenholz-Stift, Germany, 24161 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Bad Honnef, Germany, 53604 Novartis Investigative Site Baesweiler, Germany, 52499 Novartis Investigative Site Berlin, Germany, 12621 Novartis Investigative Site Bielefeld, Germany, 33602 Novartis Investigative Site Bielefeld, Germany, 33647 Novartis Investigative Site Bochum, Germany, 44787 Novartis Investigative Site Bremerhaven, Germany, 27574 Novartis Investigative Site Butzbach, Germany, 35510 Novartis Investigative Site Grevenbroich, Germany, 41515 Novartis Investigative Site Heidenheim, Germany, 89518 Novartis Investigative Site Klingenmünster, Germany, 76889 Novartis Investigative Site Leipzig, Germany, 04275 Novartis Investigative Site Merzig, Germany, 66663 Novartis Investigative Site Nienburg, Germany, 31582 Novartis Investigative Site Oberhausen, Germany, 46045 Novartis Investigative Site Oldenburg, Germany, 26122 Novartis Investigative Site Potsdam, Germany, 14471 Novartis Investigative Site Schwalmstadt-Treysa, Germany, 34613 Novartis Investigative Site Stadtroda, Germany, 07646 Novartis Investigative Site Tübingen, Germany, 72076 Novartis Investigative Site Weil am Rhein, Germany, 79576 Novartis Investigative Site Zwickau, Germany, 08060 Collapse << |
| NCT01534182 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Russian Federation ... more >> Novartis Investigative Site Arkhangelsk, Russia, Russian Federation, 163045 Novartis Investigative Site Barnaul, Russian Federation, 656024 Novartis Investigative Site Belgorod, Russian Federation, 308007 Novartis Investigative Site Kazan, Russian Federation, 420021 Novartis Investigative Site Kemerovo, Russian Federation, 650066 Novartis Investigative Site Khanty-Mansiysk, Russian Federation, 628012 Novartis Investigative Site Kirov, Russian Federation, 610014 Novartis Investigative Site Krasnodar, Russian Federation, 350086 Novartis Investigative Site Kursk, Russian Federation, 305007 Novartis Investigative Site Moscow, Russian Federation, 119992 Novartis Investigative Site Moscow, Russian Federation, 127018 Novartis Investigative Site N.Novgorod, Russian Federation, 603126 Novartis Investigative Site Nizhniy Novgorod, Russian Federation, 603076 Novartis Investigative Site Nizhny Novgorod, Russian Federation, 603155 Novartis Investigative Site Novosibirsk, Russian Federation, 630087 Novartis Investigative Site Perm, Russian Federation, 614990 Novartis Investigative Site Saransk, Russian Federation, 430032 Novartis Investigative Site Saratov, Russian Federation, 410030 Novartis Investigative Site Smolensk, Russian Federation, 214019 Novartis Investigative Site St. Petersburg, Russian Federation, 197376 Novartis Investigative Site Tomsk, Russian Federation, 634050 Novartis Investigative Site Tumen, Russian Federation, 625048 Novartis Investigative Site Tver, Russian Federation, 170036 Novartis Investigative Site Ufa, Russian Federation, 450000 Novartis Investigative Site Ulyanovsk, Russian Federation, 432063 Novartis Investigative Site Yaroslavl, Russian Federation, 150030 Collapse << |
| NCT00416845 | Healthy | Phase 1 | Completed | - | United States, New Jersey ... more >> Novartis Investigative Site East Hanover, New Jersey, United States, 07936-1080 Collapse << |
| NCT00785083 | Asthma | Phase 2 | Completed | - | United Kingdom ... more >> Novartis Investigator Site Manchester, United Kingdom Collapse << |
| NCT01534182 | - | - | Completed | - | - |
| NCT00731692 | Primary Progressive Multiple S... more >>clerosis Collapse << | Phase 3 | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT01317004 | - | - | Completed | - | - |
| NCT00731692 | - | - | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT01442194 | - | - | Recruiting | December 26, 2022 | - |
| NCT02225977 | - | - | Completed | - | United States, California ... more >> University of Southern California, Department of Neurology Los Angeles, California, United States, 90033 Collapse << |
| NCT03243721 | Multiple Sclerosis | Not Applicable | Active, not recruiting | December 2019 | United States, Ohio ... more >> Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02799199 | - | - | Completed | - | - |
| NCT00289978 | - | - | Completed | - | - |
| NCT00289978 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Completed | - | - |
| NCT03257358 | Relapsing Multiple Sclerosis | Phase 4 | Recruiting | July 1, 2019 | - |
| NCT01199861 | Relapsing Multiple Sclerosis | Phase 3 | Completed | - | Belgium ... more >> Novartis Investigative Site Aalst, Belgium, 9300 Novartis Investigative Site Bruxelles, Belgium, 1200 Novartis Investigative Site Leuven, Belgium, 3000 Novartis Investigative Site Liege, Belgium, 4000 Novartis Investigative Site Wilrijk, Belgium, 2610 Canada, Ontario Novartis Investigative Site Nepean, Ontario, Canada, K2G 6E2 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H3A 2B4 Canada Novartis Investigative Site Sherbrooke, Canada, JiH 5N4 Finland Novartis Investigative Site Seinajoki, Finland, 60220 Novartis Investigative Site Turku, Finland, 20100 France Novartis Investigative Site Caen, France, 14033 Novartis Investigative Site Rennes, France, 35043 Novartis Investigative Site St Herblain, France, 44800 Novartis Investigative Site Toulouse, France, 31059 Guatemala Novartis Investigative Site Guatemala City, Guatemala, 01010 Novartis Investigative Site Guatemala City, Guatemala, 01014 Poland Novartis Investigative Site Katowice, Poland, 40-594 Novartis Investigative Site Lodz, Poland, 90-153 Spain Novartis Investigative Site Madrid, Spain, 28029 Novartis Investigative Site Madrid, Spain, 28040 Novartis Investigative Site Sevilla, Spain, 41009 Novartis Investigative Site Valencia, Spain, 46009 Switzerland Novartis Investigational Site Basel, Switzerland, 4031 United Kingdom Novartis Investigative Site Nottingham, United Kingdom, NG7 2UH Novartis Investigative Site Stoke-on-Trent, United Kingdom, ST4 7LN Collapse << |
| NCT01623596 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01623596 | - | - | Completed | - | - |
| NCT01199861 | - | - | Completed | - | - |
| NCT01310166 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT00731523 | Renal Insufficiency | Phase 1 | Completed | - | Russian Federation ... more >> Novartis Investigator Site Moscow, Russian Federation Collapse << |
| NCT01585298 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT01811290 | - | - | Active, not recruiting | December 2018 | United States, Washington ... more >> MS Center at Evergreen Health Kirkland, Washington, United States, 98034 Collapse << |
| NCT01621269 | Multiple Sclerosis | Phase 4 | Withdrawn | June 2014 | - |
| NCT00340834 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00537082 | Multiple Sclerosis | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative site Sapporo-shi, Hokkaido, Japan Novartis Investigative Site Chiba, Japan, 276-8524 Novartis Investigative Site Ehime, Japan, 791-0295 Novartis Investigative Site Fukuoka, Japan, 807-8555 Novartis Investigative Site Gunma, Japan, 371-8511 Novartis Investigative Site Hyogo, Japan, 650-0017 Novartis Investigative Site Ibaraki, Japan, 305-8576 Novartis Iwate, Japan, 020-8505 Novartis Investigative Site Kanagawa, Japan, 259-1193 Novartis Investigative Site Kyoto, Japan, 604-8453 Novartis Investigative Site Kyoto, Japan, 616-8255 Novartis Investigative Site Niigata, Japan, 951-8520 Novartis Investigative Site Osaka, Japan, 556-0016 Novartis Investigative Site Osaka, Japan, 589-8511 Novartis Investigative Site Tochigi, Japan, 329-0498 Novartis Investigative Site Tokyo, Japan, 145-0065 Novartis Investigative Site Tokyo, Japan, 162-8666 Novartis Investigative Site Wakayama, Japan, 641-8510 Collapse << |
| NCT00333138 | Multiple Sclerosis | Phase 2 | Completed | - | Canada ... more >> Novartis Investigational site Montreal, Canada Novartis Investigational site Ottawa, Canada Novartis Investigational site Toronto, Canada Novartis Investigational site Vancouver, Canada Denmark Novartis Investigational site Copenhagen, Denmark Finland Novartis Investigational site Helsinki, Finland Novartis Investigational site Turku, Finland France Novartis Investigational site Lille, France Novartis Investigational site Marseille, France Germany Novartis Investigational site Schwendi, Germany Novartis Investigational site Wurzburg, Germany Italy Novartis Investigational site Gallarate, Italy Novartis Investigational site Genova, Italy Novartis Investigational site Milano, Italy Novartis Investigational site Roma, Italy Poland Novartis Investigational site Warszawa, Poland Portugal Novartis Investigational site Coimbra, Portugal Novartis Investigational site Lisboa, Portugal Spain Novartis Investigational site Barcelona, Spain Novartis Investigational site Madrid, Spain Novartis Investigational site Malaga, Spain Novartis Investigational site Sevilla, Spain Novartis Investigational site Valencia, Spain Switzerland Novartis Investigational site Basel, Switzerland Novartis Investigational site Zurich, Switzerland United Kingdom Novartis Investigational site Newcastle upon Tyne, United Kingdom Collapse << |
| NCT00340834 | - | - | Completed | - | - |
| NCT00537082 | - | - | Completed | - | - |
| NCT02939079 | Multiple Sclerosis | Phase 2 Phase 3 | Completed | - | - |
| NCT00333138 | - | - | Completed | - | - |
| NCT02021162 | - | - | Active, not recruiting | October 2018 | United States, New York ... more >> Buffalo Neuroimaging Analysis Center Buffalo, New York, United States, 14203 Collapse << |
| NCT03345940 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Recruiting | November 30, 2020 | Italy ... more >> Fondazione IRCCS Istituto Neurologico C. Besta, Neuroimmunology Unit Recruiting Milan, Italy, 20133 Contact: Silvia Rossi, MD, PhD +39022394 ext 2471 silvia.rossi@istituto-besta.it Contact: Renato Mantegazza, MD +39022394 ext 2321 renato.mantegazza@istituto-besta.it Principal Investigator: Silvia Rossi, MD, PhD Collapse << |
| NCT01281657 | - | - | Completed | - | - |
| NCT01790269 | - | - | Terminated | - | Germany ... more >> Charité Universitätsmedizin Berlin Berlin, Germany, 10117 Collapse << |
| NCT02232061 | Multiple Sclerosis | Phase 4 | Recruiting | July 6, 2020 | Belgium ... more >> Novartis Investigative Site Recruiting Gent, Belgium, 9000 Novartis Investigative Site Recruiting Hasselt, Belgium, 3500 Novartis Investigative Site Withdrawn Verviers, Belgium, 4800 Germany Novartis Investigative Site Recruiting Ravensburg, Germany, 88212 Novartis Investigative Site Recruiting Trier, Germany, 54292 Italy Novartis Investigative Site Recruiting Napoli, Italy, 80131 Collapse << |
| NCT02342704 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Terminated(Business Decision) | - | - |
| NCT01498887 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01216072 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 4 | Completed | - | - |
| NCT02342704 | - | - | Terminated(Business Decision) | - | - |
| NCT01216072 | - | - | Completed | - | - |
| NCT01633112 | Relapsing-remitting Multiple S... more >>clerosis (RRMS) Collapse << | Phase 4 | Completed | - | - |
| NCT01625182 | Chronic Inflammatory Demyelina... more >>ting Polyradiculoneuropathy Collapse << | Phase 3 | Completed | - | - |
| NCT01499667 | Relapsing Remitting Multiple S... more >>clerosis (RRMS) Collapse << | Phase 3 | Terminated(Based on recent pub... more >>lications, determination of natalizumub washout period was no longer relevant.) Collapse << | - | - |
| NCT01625182 | - | - | Completed | - | - |
| NCT01499667 | - | - | Terminated(Based on recent pub... more >>lications, determination of natalizumub washout period was no longer relevant.) Collapse << | - | - |
| NCT02373098 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novartis Investigative Site Istanbul, Turkey, 34093 Novartis Investigative Site Mecidiyekoy/Istanbul, Turkey, 34394 Novartis Investigative Site Uskudar / Istanbul, Turkey, 34668 Collapse << |
| NCT01420055 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Completed | - | France ... more >> Novartis Investigative Site Agen Cedex 9, France, 47923 Novartis Investigative Site Amiens Cedex 1, France, 80054 Novartis Investigative Site Angers Cedex 1, France, 49033 Novartis Investigative Site Auxerre, France, 89000 Novartis Investigative Site Avignon, France, 84000 Novartis Investigative Site Bayonne, France, 64109 Novartis Investigative Site Beziers, France, 34525 Novartis Investigative Site Bobigny Cedex, France, 93009 Novartis Investigative Site Brest, France, 29240 Novartis Investigative Site Calais, France, 62107 Novartis Investigative Site Castelnau-le-Lez, France, 34170 Novartis Investigative Site Chambray les Tours, France, 37175 Novartis Investigative Site Cherbourg, France, 50100 Novartis Investigative Site Clermont-Ferrand Cedex 1, France, 63003 Novartis Investigative Site Créteil, France, 94010 Novartis Investigative Site Dijon, France, 21034 Novartis Investigative Site Epinal, France, 88021 Novartis Investigative Site Grenoble Cedex 9, France, 38042 Novartis Investigative Site Hagueneau, France, 67500 Novartis Investigative Site La Rochelle cedex 1, France, 17019 Novartis Investigative Site La Seyne sur mer, France, 83500 Novartis Investigative Site Le Kremlin Bicetre, France, 94275 Novartis Investigative Site Le Mans Cedex 09, France, 72037 Novartis Investigative Site Limoges, France Novartis Investigative Site Lyon, France, 69275 Novartis Investigative Site Marseille, France, 13008 Novartis Investigative Site Marseille, France, 13012 Novartis Investigative Site Metz, France, 57085 Novartis Investigative Site Montpellier, France, 34000 Novartis Investigative Site Montpellier, France, 34295 Novartis Investigative Site Nimes, France, 32900 Novartis Investigative Site Orsay, France, 91401 Novartis Investigative Site Paris, France, 75005 Novartis Investigative Site Poitiers, France, 86021 Novartis Investigative Site Pontoise, France, F-95300 Novartis Investigative Site Reims, France, 51092 Novartis Investigative Site Rennes Cedex, France, 35033 Novartis Investigative Site Roanne, France, 42328 Novartis Investigative Site Saint Aubin sur Scie, France, 76550 Novartis Investigative Site Saint Jean, France, 31243 Novartis Investigative Site Sainte Foy les Lyon, France, 69110 Novartis Investigative Site Saumur, France, 49400 Novartis Investigative Site Strasbourg Cedex, France, 67091 Novartis Investigative Site Talant, France, 21240 Novartis Investigative Site Toulouse, France, 31059 Novartis Investigative Site Tourcoing, France, 59200 Novartis Investigative Site Tours Cedex, France, 37044 Collapse << |
| NCT00355134 | - | - | Completed | - | - |
| NCT00355134 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01497262 | Multiple Sclerosis | Phase 3 | Completed | - | Argentina ... more >> Novartis Investigative Site Caba, Buenos Aires, Argentina, 1425 Novartis Investigative Site Guaymallen, Mendoza, Argentina, M5507XAD Novartis Investigative Site San Miguel de Tucuman, Tucumán, Argentina, T4000DPB Novartis Investigative Site San Miguel de Tucumán, Tucumán, Argentina, 4000 Novartis Investigative Site Cordoba, Argentina, X5004CDT Novartis Investigative Site Salta, Argentina, A4400ANG Novartis Investigative Site Salta, Argentina, A4400BKZ Brazil Novartis Investigative Site Rio de Janeiro, RJ, Brazil, 20270-004 Novartis Investigative Site Rio de Janeiro, RJ, Brazil, 21941-590 Novartis Investigative Site Porto Alegre, RS, Brazil, 90610-000 Colombia Novartis Investigative Site Barranquilla, Atlantico, Colombia Novartis Investigative Site Bogotá, Cundinamarca, Colombia, 110111 Novartis Investigative Site Bogotá, Colombia, 00000 Novartis Investigative Site Bogotá, Colombia Novartis Investigative Site Cali, Colombia Jordan Novartis Investigative Site Amman, Jordan, 11942 Novartis Investigative Site Irbid, Jordan, 22110 Malaysia Novartis Investigative Site Kuala Lumpur, Malaysia, 50586 Novartis Investigative Site Kuala Lumpur, Malaysia, 59100 Novartis Investigative Site Penang, Malaysia, 10990 Mexico Novartis Investigative Site Mexico, Distrito Federal, Mexico, 06700 Novartis Investigative Site México, Distrito Federal, Mexico, 10700 Novartis Investigative Site Monterrey, Nuevo León, Mexico, 64060 Novartis Investigative Site San Nicolas De Los Garza, Nuevo León, Mexico, 66480 Novartis Investigative Site San Luis Potosí, Mexico, 78240 Panama Novartis Investigative Site Panama City, Panamá, Panama Peru Novartis Investigative Site La Perla, Callao, Peru, 04 Novartis Investigative Site Jesus Maria, Lima, Peru, 11 Novartis Investigative Site San Isidro, Lima, Peru, 27 Collapse << |
| NCT00662649 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00662649 | - | - | Completed | - | - |
| NCT01497262 | - | - | Completed | - | - |
| NCT02325440 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Unknown | April 2016 | Germany ... more >> Universitaetsklinikum Muenster, Department of Neurology Recruiting Muenster, Germany, 48149 Contact: Luisa Klotz, PD Dr. med. +49 251 83444 ext 52 luisa.klotz@ukmuenster.de Principal Investigator: Luisa Klotz, PD Dr. med. Collapse << |
| NCT01201356 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02048072 | - | - | Completed | - | - |
| NCT02048072 | Multiple Sclerosis ... more >> Autonomic Nervous System Dysfunction Collapse << | Phase 4 | Completed | - | Switzerland ... more >> Cantonal Hospital St. Gallen St. Gallen, SG, Switzerland, 9000 Collapse << |
| NCT03216915 | - | - | Recruiting | December 31, 2019 | Germany ... more >> University Medical Hospital Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Contact: Christoph Heesen, MD +49 (0) 40 7410 - 52124 heesen@uke.de Contact: Ruken Özge Akbulak, MD +49 (0) 40 7410 - 59471 r.akbulak@uke.de Collapse << |
| NCT01757691 | Acute Demylelinating Optic Neu... more >>ritis Collapse << | Phase 2 | Terminated(Discontinuation of ... more >>this study was based on Novartis decision to discontinue development of fingolimod for the treatment of ADON) Collapse << | - | United States, Florida ... more >> Novartis Investigative Site St. Petersburg, Florida, United States, 33713 Spain Novartis Investigative Site Majadahonda, Madrid, Spain, 28222 Collapse << |
| NCT00670449 | Multiple Sclerosis | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative Site Chiba, Japan, 276-8524 Novartis Investigative Site Ehime, Japan, 791-0295 Novartis Investigative Site Fukuoka, Japan, 807-8555 Novartis Investigative Site Gunma, Japan, 371-8511 Novartis Investigative Site Hyogo, Japan, 650-0017 Novartis Investigative Site Ibaraki, Japan, 305-8576 Novartis Investigative Site Kanagawa, Japan, 259-1193 Novartis Investigative Site Kyoto, Japan, 604-8453 Novartis Investigative Site Kyoto, Japan, 616-8255 Novartis Investigative Site Morioka, Japan, 020-8505 Novartis Investigative Site Niigata, Japan, 951-8520 Novartis Investigative Site Osaka, Japan, 556-0016 Novartis Investigative Site Osaka, Japan, 589-8511 Novartis Investigative Site Osaka, Japan Novartis Investigative Site Sapporo, Japan, 060-8648 Novartis Investigative Site Tochigi, Japan, 329-0498 Novartis Investigative Site Tokyo, Japan, 113-8519 Novartis Investigative Site Tokyo, Japan, 145-0065 Novartis Investigative Site Tokyo, Japan, 162-8666 Novartis Investigative Site Wakayama, Japan, 641-8510 Collapse << |
| NCT01757691 | - | - | Terminated(Discontinuation of ... more >>this study was based on Novartis decision to discontinue development of fingolimod for the treatment of ADON) Collapse << | - | - |
| NCT01791192 | Acute Noninfectious Posterior,... more >> Intermediate, or Pan Uveitis Collapse << | Phase 2 | Withdrawn | October 2014 | - |
| NCT01779700 | Schizophrenia | Phase 2 | Active, not recruiting | December 2017 | United States, Indiana ... more >> Center for NeuroImaging Indianapolis, Indiana, United States, 46202 Prevention and Recovery Center Indianapolis, Indiana, United States, 46202 Larue D Carter Memorial Hospital Indianapolis, Indiana, United States, 46222 Collapse << |
| NCT01578330 | Multiple Sclerosis ... more >> Relapsing-Remitting Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novartis Investigative Site Altunizade, Turkey, 34662 Novartis Investigative Site Ankara, Turkey, 06100 Novartis Investigative Site Ankara, Turkey, 06500 Novartis Investigative Site Atakum / Samsun, Turkey, 55139 Novartis Investigative Site Bursa, Turkey, 16059 Novartis Investigative Site Fatih / Istanbul, Turkey, 34098 Novartis Investigative Site Izmir, Turkey, 35040 Novartis Investigative Site Kocaeli, Turkey, 41380 Novartis Investigative Site Mecidiyekoy/Istanbul, Turkey, 34394 Novartis Investigative Site Trabzon, Turkey, 61080 Novartis Investigative Site Uskudar / Istanbul, Turkey, 34668 Collapse << |
| NCT02575365 | Cognition Bra... more >>in Volume Loss Collapse << | Phase 4 | Terminated | - | Turkey ... more >> Novartis Investigative Site Bursa, Turkey, 16059 Novartis Investigative Site Kocaeli, Turkey, 41380 Novartis Investigative Site Kutahya, Turkey, 43000 Collapse << |
| NCT02002390 | Stroke Vascul... more >>ar Accident Cerebral Stroke Ischemic Cerebrovascular Accident Stroke, Acute Collapse << | Phase 2 | Completed | - | China, Tianjin ... more >> Tianjin Medical University General Hospital Tianjin, Tianjin, China, 300052 Collapse << |
| NCT00670449 | - | - | Completed | - | - |
| NCT02061137 | Rett's Syndrome | Phase 1 Phase 2 | Completed | - | Switzerland ... more >> Department of Neuropediatrics - University Children's Hospital Basel, Switzerland, 4056 Collapse << |
| NCT01578330 | - | - | Completed | - | - |
| NCT01941004 | Multiple Sclerosis (Relapsing ... more >>Remitting) Collapse << | Phase 3 | Withdrawn | May 2017 | - |
| NCT01786174 | Amyotrophic Lateral Sclerosis | Phase 2 | Completed | - | United States, California ... more >> University of California, Irvine Orange, California, United States, 92868 United States, Georgia Georgia Regents University Augusta, Georgia, United States, 30912 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Texas Methodist Neurological Institute Houston, Texas, United States, 77030 Collapse << |
| NCT02490930 | Glioblastoma ... more >>Anaplastic Astrocytoma Collapse << | Early Phase 1 | Completed | - | United States, Maryland ... more >> The Johns Hopkins University Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01786174 | - | - | Completed | - | - |
| NCT01755871 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Terminated(lack of recruitment... more >>) Collapse << | - | Germany ... more >> Heinrich Heine Universität Düsseldorf Düsseldorf, Nord-Rhein Westfahlen, Germany, 40225 Collapse << |
| NCT02956200 | Stroke Inflam... more >>mation Collapse << | Phase 2 | Unknown | December 2018 | China, Zhejiang ... more >> The second affiliated hospital of Zhejiang University Hangzhou, Zhejiang, China, 310000 Collapse << |
| NCT03757338 | Healthy Volunteers | Phase 1 | Completed | - | New Zealand ... more >> Zenith Clinical Site Dunedin, New Zealand, 9010 Collapse << |
| NCT02137707 | - | - | Completed | - | Canada, British Columbia ... more >> University of British Columbia Vancouver, British Columbia, Canada, V6T 1Z3 Canada, Nova Scotia Dalhousie University Multiple Sclerosis Research Unit Halifax, Nova Scotia, Canada Canada, Ontario Ottawa General Hospital Ottawa, Ontario, Canada, K1H 8L6 Canada, Quebec Clinique Neuro Rive-Sud Greenfield Park, Quebec, Canada Montreal Neurological Institute Montreal, Quebec, Canada, H3A 2B4 Centre hospitalier de l'Universite de Montreal (CHUM) Montréal, Quebec, Canada Collapse << |
| NCT02559765 | - | - | Completed | - | United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << |
| NCT03193866 | - | - | Recruiting | December 2021 | Sweden ... more >> Fredrik Piehl Recruiting Stockholm, Sweden Contact: Fredrik Piehl, Professor +46 73-671 81 01 Fredrik.Piehl@ki.se Contact: Anna Fogdell-Hahn, PhD +46 70-544 90 60 anna.fogdell-hahn@ki.se Collapse << |
| NCT01795872 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT03535298 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Not yet recruiting | September 2023 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Tammy Skaramagas, BA 216-445-6724 skaramt1@ccf.org Principal Investigator: Daniel Ontaneda, MD, MSc United Kingdom University of Nottingham Not yet recruiting Nottingham, United Kingdom, NG7 2UH Contact: Sara Wilkins +44 115 9249924 ext 66816 Sara.wilkins@nuh.nhs.uk Principal Investigator: Nikos Evangelou, MD, DPhil Collapse << |
| NCT00273364 | Multiple Sclerosis | Phase 2 | Active, not recruiting | December 2018 | United States, Illinois ... more >> Northwestern University, Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT02139696 | - | - | Completed | - | Finland ... more >> Turku University Hospital Turku, Finland, 20520 Collapse << |
| NCT03399981 | - | - | Active, not recruiting | December 31, 2023 | United States, Massachusetts ... more >> Research Site Cambridge, Massachusetts, United States, 02142 Collapse << |
| NCT02241785 | Relapsing Multiple Sclerosis | Phase 4 | Terminated(Business Decision) | - | United States, California ... more >> Research Site Fullerton, California, United States, 92835 United States, Colorado Research Site Aurora, Colorado, United States, 80045 United States, Iowa Research Site Des Moines, Iowa, United States, 50314 United States, Missouri Research Site Saint Louis, Missouri, United States, 63110 United States, New York Research Site Plainview, New York, United States, 11803 United States, North Carolina Research Site Raleigh, North Carolina, United States, 27607-6010 United States, Ohio Research Site Akron, Ohio, United States, 44320 Research Site Cleveland, Ohio, United States, 44195 United States, Tennessee Research Site Knoxville, Tennessee, United States, 37922 United States, Texas Research Site Round Rock, Texas, United States, 78681 United States, Washington Research Site Tacoma, Washington, United States, 98405 Collapse << |
| NCT03500328 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Not Applicable | Recruiting | October 31, 2022 | - |
| NCT02776072 | - | - | Completed | - | - |
| NCT02241785 | - | - | Terminated(Business Decision) | - | - |
| NCT02408380 | - | - | Unknown | December 2015 | Canada, Quebec ... more >> McGill University Recruiting Montreal, Quebec, Canada, H3A 2B4 Contact: Leslie Fitz-Gerald, MSc 5143987192 ext 00510 leslie.fitz-gerald@mail.mcgill.ca Principal Investigator: David Haegert, MD Collapse << |
| NCT02335892 | - | - | Completed | - | United Kingdom ... more >> Novartis Investigative Site Truro, Cornwall, United Kingdom, TR1 3LJ Novartis Investigative Site Swansea, England, United Kingdom, SA6 6NL Novartis Investigative Site Romford, Essex, United Kingdom, RM7 0AG Novartis Investigative Site Basingstoke, Hampshire, United Kingdom, RG24 9NA Novartis Investigative Site Edinburgh, United Kingdom, EH16 4SA Novartis Investigative Site Glasgow, United Kingdom, G51 4TF Novartis Investigative Site Leicester, United Kingdom, LE5 4PW Novartis Investigative Site Liverpool, United Kingdom, L9 7LT Novartis Investigative Site London, United Kingdom, E1 1BB Novartis Investigative Site London, United Kingdom, SW17 0QT Novartis Investigative Site London, United Kingdom, W6 8RF Novartis Investigative Site London, United Kingdom, WC1N 3BG Novartis Investigative Site Norwich, United Kingdom, NR4 7UY Novartis Investigative Site Nottingham, United Kingdom, NG5 1PB Collapse << |
| NCT01592097 | - | - | Completed | - | United States, California ... more >> Keck School of Medicine of the University of Southern Calfornia Los Angeles, California, United States, 90033 Collapse << |
| NCT01490840 | Fatigue in Multiple Sclerosis | Phase 4 | Terminated(The study stopped d... more >>ue to slow enrollment and low participant compliance.) Collapse << | - | Germany ... more >> Novartis Investigative Site Aachen, Germany, 52062 Novartis Investigative Site Alzenau, Germany, 63755 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Bad Mergentheim, Germany, 97980 Novartis Investigative Site Bamberg, Germany, 96052 Novartis Investigative Site Bayreuth, Germany, 95445 Novartis Investigative Site Berlin, Germany, 10437 Novartis Investigative Site Berlin, Germany, 10625 Novartis Investigative Site Berlin, Germany, 10691 Novartis Investigative Site Berlin, Germany, 12587 Novartis Investigative Site Berlin, Germany, 13347 Novartis Investigative Site Bielefeld, Germany, 33647 Novartis Investigative Site Bochum, Germany, 44787 Novartis Investigative Site Böblingen, Germany, 71032 Novartis Investigative Site Dortmund, Germany, 44135 Novartis Investigative Site Erbach, Germany, 64711 Novartis Investigative Site Erlangen, Germany, 91054 Novartis Investigative Site Frankfurt, Germany, 65929 Novartis Investigative Site Hamburg, Germany, 22179 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Jena, Germany, 07740 Novartis Investigative Site Kassel, Germany, 34121 Novartis Investigative Site Leipzig, Germany, 04299 Novartis Investigative Site Potsdam, Germany, 14471 Novartis Investigative Site Prien, Germany, 83209 Novartis Investigative Site Regensburg, Germany, 93053 Novartis Investigative Site Siegen, Germany, 57076 Novartis Investigative Site Sinsheim, Germany, 74889 Novartis Investigative Site Stuttgart, Germany, 70174 Novartis Investigative Site Stuttgart, Germany, 70178 Novartis Investigative Site Teupitz, Germany, 15755 Novartis Investigative Site Unterhaching, Germany, 82008 Collapse << |
| NCT02720107 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Ostfildern, Baden-Wuerttemberg, Germany, 73760 Novartis Investigative Site Altenholz-Stift, Germany, 24161 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Berlin, Germany, 10713 Novartis Investigative Site Berlin, Germany, 12163 Novartis Investigative Site Berlin, Germany, 13347 Novartis Investigative Site Böblingen, Germany, 71032 Novartis Investigative Site Celle, Germany, 29223 Novartis Investigative Site Dortmund, Germany, 44137 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Erbach, Germany, 64711 Novartis Investigative Site Frankfurt, Germany, 60313 Novartis Investigative Site Göttingen, Germany, 37073 Novartis Investigative Site Jena, Germany, 07740 Novartis Investigative Site Kiel, Germany, 24105 Novartis Investigative Site Klingenmünster, Germany, 76889 Novartis Investigative Site Lappersdorf, Germany, 93138 Novartis Investigative Site Leverkusen, Germany, 51375 Novartis Investigative Site Mönchengladbach, Germany, 41239 Novartis Investigative Site München, Germany, 81829 Novartis Investigative Site Neuburg an der Donau, Germany, 86633 Novartis Investigative Site Siegen, Germany, 57076 Novartis Investigative Site Singen, Germany, 78224 Novartis Investigative Site Troisdorf, Germany, 53844 Novartis Investigative Site Ulm, Germany, 89073 Novartis Investigative Site Unterhaching, Germany, 82008 Collapse << |
| NCT02193217 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 1 | Completed | - | United Kingdom ... more >> Investigational site Leeds, United Kingdom Collapse << |
| NCT01490840 | - | - | Terminated(The study stopped d... more >>ue to slow enrollment and low participant compliance.) Collapse << | - | - |
| NCT01705236 | Relapsing Remitting Multiple S... more >>clerosis RRMS Collapse << | Phase 4 | Active, not recruiting | January 28, 2019 | Germany ... more >> Novartis Investigative Site Bochum, Germany, 44791 Novartis Investigative Site Bonn, Germany, 53105 Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Duesseldorf, Germany, 40225 Novartis Investigative Site Hannover, Germany, 30623 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Leipzig, Germany, 04103 Novartis Investigative Site Rostock, Germany, 18057 Novartis Investigative Site Ulm, Germany, 89081 Switzerland Novartis Investigative Site Zuerich, Switzerland, 8091 Collapse << |
| NCT02277964 | - | - | Completed | - | Switzerland ... more >> University Hospital Basel, Switzerland Basel, Switzerland, 4031 Collapse << |
| NCT01709812 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Withdrawn | July 2013 | - |
| NCT02307838 | Multiple Sclerosis, Relapsing ... more >>Forms of Multiple Sclerosis Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Novartis Investigative Site Ottawa, Ontario, Canada, K1H 8L6 Novartis Investigative Site Toronto, Ontario, Canada, M5B 1N9 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H2L 4M1 Novartis Investigative Site Montreal, Quebec, Canada, H3A 2B4 Denmark Novartis Investigative Site Copenhagen, Denmark, DK-2100 Novartis Investigative Site Glostrup, Denmark, DK-2600 France Novartis Investigative Site Lille, France, 59037 Novartis Investigative Site Marseille, France, 13385 Germany Novartis Investigative Site Würzburg, Germany, 97080 Italy Novartis Investigative Site Genova, GE, Italy, 16132 Novartis Investigative Site Milano, MI, Italy, 20132 Novartis Investigative Site Roma, RM, Italy, 00189 Novartis Investigative Site Gallarate, VA, Italy, 21013 Poland Novartis Investigative Site Warszawa, Poland, 02-097 Novartis Investigative Site Warszawa, Poland, 02-957 Portugal Novartis Investigative Site Coimbra, Portugal, 3000-075 Novartis Investigative Site Lisboa, Portugal, 1150-314 Spain Novartis Investigative Site Malaga, Andalucia, Spain, 29010 Novartis Investigative Site Sevilla, Andalucia, Spain, 41009 Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Novartis Investigative Site L'Hospitalet de Llobregat, Catalunya, Spain, 08907 Novartis Investigative Site Valencia, Comunidad Valenciana, Spain, 46026 Novartis Investigative Site Madrid, Spain, 28040 Switzerland Novartis Investigative Site Basel, Switzerland, 4031 Novartis Investigative Site Zuerich, Switzerland, 8091 United Kingdom Novartis Investigative Site Newcastle Upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT02307877 | - | - | Active, not recruiting | December 2019 | United States, Colorado ... more >> University of Colorado Hospital Aurora, Colorado, United States, 80045 Collapse << |
| NCT00098735 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT02307838 | - | - | Completed | - | - |
| NCT01704183 | - | - | Completed | - | Finland ... more >> Neuro Center, Kuopio University Hospital Kuopio, Finland Department of Neurology, Mikkeli Central Hospital Mikkeli, Finland Collapse << |
| NCT00239785 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT00239863 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT02141022 | Multiple Sclerosis ... more >> Cognitive Deficits Gilenya Modifying Therapy for MS Collapse << | Not Applicable | Completed | - | United States, New York ... more >> Stony Brook University Stony Brook, New York, United States, 11794 Collapse << |
| NCT00239798 | Renal Transplantation | Phase 2 | Completed | - | - |
| NCT00099749 | Kidney Transplantation | Phase 2 Phase 3 | Completed | - | United States, Illinois ... more >> Loyola University Medical Center Maywood, Illinois, United States, 60153 SIU School of Medicine Springfield, Illinois, United States, 62781 United States, New Jersey St. Barnabas Medical Center Livingston, New Jersey, United States, 07039 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Rhode Island Rhode Island Hospital Providence, Rhode Island, United States, 02903 Collapse << |
| NCT00239902 | Renal Transplantation | Phase 2 | Completed | - | - |
| NCT01285479 | - | - | Recruiting | December 13, 2030 | - |
| NCT00239811 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT02769689 | Multiple Sclerosis | Phase 4 | Unknown | November 2017 | France ... more >> CHU Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 04 73 75 11 95 placarin@chu-clermontferrand.fr Principal Investigator: Pierre CLAVELOU Collapse << |
| NCT00239876 | Renal Transplantation | Phase 3 | Completed | - | - |
| NCT00099736 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT01333501 | Multiple Sclerosis | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Alzenau, Germany, 63755 Novartis Investigative Site Bamberg, Germany, 96049 Novartis Investigative Site Leipzig, Germany, 04103 Novartis Investigative Site Würzburg, Germany, 97080 Italy Novartis Investigative Site Ancona, AN, Italy, 60126 Novartis Investigative Site Bari, BA, Italy, 70124 Novartis Investigative Site Bergamo, BG, Italy, 24128 Novartis Investigative Site Montichiari, BS, Italy, 25018 Novartis Investigative Site Chieti, CH, Italy, 66100 Novartis Investigative Site Catania, CT, Italy, 95122 Novartis Investigative Site Catania, CT, Italy, 95123 Novartis Investigative Site Firenze, FI, Italy, 50134 Novartis Investigative Site Genova, GE, Italy, 16132 Novartis Investigative Site Pozzilli, IS, Italy, 86077 Novartis Investigative Site Messina, ME, Italy, 98121 Novartis Investigative Site Milano, MI, Italy, 20122 Novartis Investigative Site Milano, MI, Italy, 20132 Novartis Investigative Site Cefalù, PA, Italy, 90015 Novartis Investigative Site Pavia, PV, Italy, 27100 Novartis Investigative Site Roma, RM, Italy, 00133 Novartis Investigative Site Roma, RM, Italy, 00161 Novartis Investigative Site Roma, RM, Italy, 00179 Novartis Investigative Site Roma, RM, Italy, 00189 Novartis Investigative Site Gallarate, VA, Italy, 21013 Novartis Investigative Site Napoli, Italy, 80131 Novartis Investigative Site Napoli, Italy, 80138 Collapse << |
| NCT01892722 | Multiple Sclerosis | Phase 3 | Active, not recruiting | March 15, 2023 | - |
| NCT01333501 | - | - | Completed | - | - |
| NCT01436643 | Depression Re... more >>lapsing-remitting Multiple Sclerosis Collapse << | Phase 4 | Terminated(Due to slow enrollm... more >>ent the study was terminated early) Collapse << | - | Germany ... more >> Novartis Investigative Site Achim, Germany, 28832 Novartis Investigative Site Altenholz-Stift, Germany, 24161 Novartis Investigative Site Aschaffenburg, Germany, 63739 Novartis Investigative Site Bad Honnef, Germany, 53604 Novartis Investigative Site Baesweiler, Germany, 52499 Novartis Investigative Site Berlin, Germany, 12621 Novartis Investigative Site Bielefeld, Germany, 33602 Novartis Investigative Site Bielefeld, Germany, 33647 Novartis Investigative Site Bochum, Germany, 44787 Novartis Investigative Site Bremerhaven, Germany, 27574 Novartis Investigative Site Butzbach, Germany, 35510 Novartis Investigative Site Grevenbroich, Germany, 41515 Novartis Investigative Site Heidenheim, Germany, 89518 Novartis Investigative Site Klingenmünster, Germany, 76889 Novartis Investigative Site Leipzig, Germany, 04275 Novartis Investigative Site Merzig, Germany, 66663 Novartis Investigative Site Nienburg, Germany, 31582 Novartis Investigative Site Oberhausen, Germany, 46045 Novartis Investigative Site Oldenburg, Germany, 26122 Novartis Investigative Site Potsdam, Germany, 14471 Novartis Investigative Site Schwalmstadt-Treysa, Germany, 34613 Novartis Investigative Site Stadtroda, Germany, 07646 Novartis Investigative Site Tübingen, Germany, 72076 Novartis Investigative Site Weil am Rhein, Germany, 79576 Novartis Investigative Site Zwickau, Germany, 08060 Collapse << |
| NCT01127750 | Relapsing Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01981161 | - | - | Recruiting | November 2017 | France ... more >> CHU Besançon Not yet recruiting Besancon, France, 25000 Contact: Eric Berger, MD lrumbach@chu-besancon.fr Principal Investigator: Eric Berger, MD CHU Bordeaux Not yet recruiting Bordeaux, France, 33 000 Contact: Bruno Brochet, MD PHD bruno.brochet@chu-bordeaux.fr Principal Investigator: bruno brochet, MD PHD CHU Caen Not yet recruiting Caen, France, 14033 Contact: Gilles-Louis Defer, MD PHD defer-gi@chu-caen.fr Principal Investigator: Gilles-Louis Defer, MD PHD Chu Lille Not yet recruiting Lille, France, 59037 Contact: Patrick Vermersch, MD PHD Principal Investigator: Patrick Vermersch, MD PHD Chu La Timone Not yet recruiting Marseille, France, 13385 Contact: Jean Pelletier, MD PHD Principal Investigator: Jean Pelletier, MD PHD CHU Nancy Not yet recruiting Nancy, France, 54035 Contact: Marc Debouverie, MD PHD Principal Investigator: Marc Debouverie, MD PHD CHU Nantes Recruiting Nantes, France, 44093 Contact: David Laplaud, MD david.laplaud@chu-nantes.fr Principal Investigator: David Laplaud, MD CHU Nice Not yet recruiting Nice, France, 06002 Contact: Christine Lebrun-Frenay, MD Principal Investigator: Christine Lebrun-Frenay, MD CHU Nîmes Not yet recruiting Nimes, France, 30029 Contact: Pierre Labauge, MD PHD Principal Investigator: Labauge Pierre, MD PHD CHRU Strasbourg Not yet recruiting Strasbourg, France, 67098 Contact: Jérôme De Seze, MD pHD Principal Investigator: Jerome De Seze, MD PHD Germany Université de Muenter Not yet recruiting Munster, Germany Contact: H Wiendl, MD PHD Principal Investigator: H Wiendl, MD PHD Spain Hôpital Vall D'Hebron Not yet recruiting Barcelone, Spain Contact: Xavier Montalban, MD PHD Principal Investigator: XAVIER Montalban, MD PHD Collapse << |
| NCT01892722 | - | - | Active, not recruiting | - | - |
| NCT01436643 | - | - | Terminated(Due to slow enrollm... more >>ent the study was terminated early) Collapse << | - | - |
| NCT00099801 | Kidney Transplantation | Phase 3 | Completed | - | - |
| NCT01647880 | Multiple Sclerosis | Phase 2 Phase 3 | Terminated(delayed recruitment... more >>) Collapse << | - | Germany ... more >> Charité-Universitätsmedizin Berlin Berlin, Germany Sankt Josefs Krankenhaus Potsdam Neurologie Potsdam, Germany Collapse << |
| NCT01534182 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Russian Federation ... more >> Novartis Investigative Site Arkhangelsk, Russia, Russian Federation, 163045 Novartis Investigative Site Barnaul, Russian Federation, 656024 Novartis Investigative Site Belgorod, Russian Federation, 308007 Novartis Investigative Site Kazan, Russian Federation, 420021 Novartis Investigative Site Kemerovo, Russian Federation, 650066 Novartis Investigative Site Khanty-Mansiysk, Russian Federation, 628012 Novartis Investigative Site Kirov, Russian Federation, 610014 Novartis Investigative Site Krasnodar, Russian Federation, 350086 Novartis Investigative Site Kursk, Russian Federation, 305007 Novartis Investigative Site Moscow, Russian Federation, 119992 Novartis Investigative Site Moscow, Russian Federation, 127018 Novartis Investigative Site N.Novgorod, Russian Federation, 603126 Novartis Investigative Site Nizhniy Novgorod, Russian Federation, 603076 Novartis Investigative Site Nizhny Novgorod, Russian Federation, 603155 Novartis Investigative Site Novosibirsk, Russian Federation, 630087 Novartis Investigative Site Perm, Russian Federation, 614990 Novartis Investigative Site Saransk, Russian Federation, 430032 Novartis Investigative Site Saratov, Russian Federation, 410030 Novartis Investigative Site Smolensk, Russian Federation, 214019 Novartis Investigative Site St. Petersburg, Russian Federation, 197376 Novartis Investigative Site Tomsk, Russian Federation, 634050 Novartis Investigative Site Tumen, Russian Federation, 625048 Novartis Investigative Site Tver, Russian Federation, 170036 Novartis Investigative Site Ufa, Russian Federation, 450000 Novartis Investigative Site Ulyanovsk, Russian Federation, 432063 Novartis Investigative Site Yaroslavl, Russian Federation, 150030 Collapse << |
| NCT00416845 | Healthy | Phase 1 | Completed | - | United States, New Jersey ... more >> Novartis Investigative Site East Hanover, New Jersey, United States, 07936-1080 Collapse << |
| NCT01317004 | - | - | Completed | - | - |
| NCT00785083 | Asthma | Phase 2 | Completed | - | United Kingdom ... more >> Novartis Investigator Site Manchester, United Kingdom Collapse << |
| NCT01317004 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Italy ... more >> Novartis Investigative Site Ancona, AN, Italy, 60126 Novartis Investigative Site Ponderano, BI, Italy, 13900 Novartis Investigative Site Caltanissetta, CL, Italy, 93100 Novartis Investigative Site Cuneo, CN, Italy, 12100 Novartis Investigative Site Como, CO, Italy, 22100 Novartis Investigative Site Catania, CT, Italy, 95122 Novartis Investigative Site Foggia, FG, Italy, 71100 Novartis Investigative Site Castelfiorentino, FI, Italy, 50051 Novartis Investigative Site Milano, MI, Italy, 20122 Novartis Investigative Site Milano, MI, Italy, 20133 Novartis Investigative Site San Donato Milanese, MI, Italy, 20097 Novartis Investigative Site Modena, MO, Italy, 41100 Novartis Investigative Site Palermo, PA, Italy, 90129 Novartis Investigative Site Palermo, PA, Italy, 90146 Novartis Investigative Site Pisa, PI, Italy, 56126 Novartis Investigative Site Legnago, VR, Italy, 37045 Novartis Investigative Site Novara, Italy, 28100 Collapse << |
| NCT01442194 | - | - | Recruiting | December 26, 2022 | - |
| NCT01534182 | - | - | Completed | - | - |
| NCT00289978 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02799199 | - | - | Completed | - | - |
| NCT02225977 | - | - | Completed | - | United States, California ... more >> University of Southern California, Department of Neurology Los Angeles, California, United States, 90033 Collapse << |
| NCT00731692 | Primary Progressive Multiple S... more >>clerosis Collapse << | Phase 3 | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT03243721 | Multiple Sclerosis | Not Applicable | Active, not recruiting | December 2019 | United States, Ohio ... more >> Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01310166 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT00289978 | - | - | Completed | - | - |
| NCT00731692 | - | - | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT01199861 | Relapsing Multiple Sclerosis | Phase 3 | Completed | - | Belgium ... more >> Novartis Investigative Site Aalst, Belgium, 9300 Novartis Investigative Site Bruxelles, Belgium, 1200 Novartis Investigative Site Leuven, Belgium, 3000 Novartis Investigative Site Liege, Belgium, 4000 Novartis Investigative Site Wilrijk, Belgium, 2610 Canada, Ontario Novartis Investigative Site Nepean, Ontario, Canada, K2G 6E2 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H3A 2B4 Canada Novartis Investigative Site Sherbrooke, Canada, JiH 5N4 Finland Novartis Investigative Site Seinajoki, Finland, 60220 Novartis Investigative Site Turku, Finland, 20100 France Novartis Investigative Site Caen, France, 14033 Novartis Investigative Site Rennes, France, 35043 Novartis Investigative Site St Herblain, France, 44800 Novartis Investigative Site Toulouse, France, 31059 Guatemala Novartis Investigative Site Guatemala City, Guatemala, 01010 Novartis Investigative Site Guatemala City, Guatemala, 01014 Poland Novartis Investigative Site Katowice, Poland, 40-594 Novartis Investigative Site Lodz, Poland, 90-153 Spain Novartis Investigative Site Madrid, Spain, 28029 Novartis Investigative Site Madrid, Spain, 28040 Novartis Investigative Site Sevilla, Spain, 41009 Novartis Investigative Site Valencia, Spain, 46009 Switzerland Novartis Investigational Site Basel, Switzerland, 4031 United Kingdom Novartis Investigative Site Nottingham, United Kingdom, NG7 2UH Novartis Investigative Site Stoke-on-Trent, United Kingdom, ST4 7LN Collapse << |
| NCT01199861 | - | - | Completed | - | - |
| NCT01585298 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT01623596 | - | - | Completed | - | - |
| NCT00731523 | Renal Insufficiency | Phase 1 | Completed | - | Russian Federation ... more >> Novartis Investigator Site Moscow, Russian Federation Collapse << |
| NCT01623596 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01621269 | Multiple Sclerosis | Phase 4 | Withdrawn | June 2014 | - |
| NCT00537082 | - | - | Completed | - | - |
| NCT01811290 | - | - | Active, not recruiting | December 2018 | United States, Washington ... more >> MS Center at Evergreen Health Kirkland, Washington, United States, 98034 Collapse << |
| NCT00333138 | Multiple Sclerosis | Phase 2 | Completed | - | Canada ... more >> Novartis Investigational site Montreal, Canada Novartis Investigational site Ottawa, Canada Novartis Investigational site Toronto, Canada Novartis Investigational site Vancouver, Canada Denmark Novartis Investigational site Copenhagen, Denmark Finland Novartis Investigational site Helsinki, Finland Novartis Investigational site Turku, Finland France Novartis Investigational site Lille, France Novartis Investigational site Marseille, France Germany Novartis Investigational site Schwendi, Germany Novartis Investigational site Wurzburg, Germany Italy Novartis Investigational site Gallarate, Italy Novartis Investigational site Genova, Italy Novartis Investigational site Milano, Italy Novartis Investigational site Roma, Italy Poland Novartis Investigational site Warszawa, Poland Portugal Novartis Investigational site Coimbra, Portugal Novartis Investigational site Lisboa, Portugal Spain Novartis Investigational site Barcelona, Spain Novartis Investigational site Madrid, Spain Novartis Investigational site Malaga, Spain Novartis Investigational site Sevilla, Spain Novartis Investigational site Valencia, Spain Switzerland Novartis Investigational site Basel, Switzerland Novartis Investigational site Zurich, Switzerland United Kingdom Novartis Investigational site Newcastle upon Tyne, United Kingdom Collapse << |
| NCT03257358 | Relapsing Multiple Sclerosis | Phase 4 | Recruiting | July 1, 2019 | - |
| NCT00340834 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00537082 | Multiple Sclerosis | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative site Sapporo-shi, Hokkaido, Japan Novartis Investigative Site Chiba, Japan, 276-8524 Novartis Investigative Site Ehime, Japan, 791-0295 Novartis Investigative Site Fukuoka, Japan, 807-8555 Novartis Investigative Site Gunma, Japan, 371-8511 Novartis Investigative Site Hyogo, Japan, 650-0017 Novartis Investigative Site Ibaraki, Japan, 305-8576 Novartis Iwate, Japan, 020-8505 Novartis Investigative Site Kanagawa, Japan, 259-1193 Novartis Investigative Site Kyoto, Japan, 604-8453 Novartis Investigative Site Kyoto, Japan, 616-8255 Novartis Investigative Site Niigata, Japan, 951-8520 Novartis Investigative Site Osaka, Japan, 556-0016 Novartis Investigative Site Osaka, Japan, 589-8511 Novartis Investigative Site Tochigi, Japan, 329-0498 Novartis Investigative Site Tokyo, Japan, 145-0065 Novartis Investigative Site Tokyo, Japan, 162-8666 Novartis Investigative Site Wakayama, Japan, 641-8510 Collapse << |
| NCT00333138 | - | - | Completed | - | - |
| NCT02939079 | Multiple Sclerosis | Phase 2 Phase 3 | Completed | - | - |
| NCT02021162 | - | - | Active, not recruiting | October 2018 | United States, New York ... more >> Buffalo Neuroimaging Analysis Center Buffalo, New York, United States, 14203 Collapse << |
| NCT01281657 | - | - | Completed | - | - |
| NCT01790269 | - | - | Terminated | - | Germany ... more >> Charité Universitätsmedizin Berlin Berlin, Germany, 10117 Collapse << |
| NCT02232061 | Multiple Sclerosis | Phase 4 | Recruiting | July 6, 2020 | Belgium ... more >> Novartis Investigative Site Recruiting Gent, Belgium, 9000 Novartis Investigative Site Recruiting Hasselt, Belgium, 3500 Novartis Investigative Site Withdrawn Verviers, Belgium, 4800 Germany Novartis Investigative Site Recruiting Ravensburg, Germany, 88212 Novartis Investigative Site Recruiting Trier, Germany, 54292 Italy Novartis Investigative Site Recruiting Napoli, Italy, 80131 Collapse << |
| NCT00340834 | - | - | Completed | - | - |
| NCT03345940 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Recruiting | November 30, 2020 | Italy ... more >> Fondazione IRCCS Istituto Neurologico C. Besta, Neuroimmunology Unit Recruiting Milan, Italy, 20133 Contact: Silvia Rossi, MD, PhD +39022394 ext 2471 silvia.rossi@istituto-besta.it Contact: Renato Mantegazza, MD +39022394 ext 2321 renato.mantegazza@istituto-besta.it Principal Investigator: Silvia Rossi, MD, PhD Collapse << |
| NCT02342704 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Terminated(Business Decision) | - | - |
| NCT01216072 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01498887 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01216072 | - | - | Completed | - | - |
| NCT02342704 | - | - | Terminated(Business Decision) | - | - |
| NCT01625182 | Chronic Inflammatory Demyelina... more >>ting Polyradiculoneuropathy Collapse << | Phase 3 | Completed | - | - |
| NCT01625182 | - | - | Completed | - | - |
| NCT01633112 | Relapsing-remitting Multiple S... more >>clerosis (RRMS) Collapse << | Phase 4 | Completed | - | - |
| NCT01499667 | Relapsing Remitting Multiple S... more >>clerosis (RRMS) Collapse << | Phase 3 | Terminated(Based on recent pub... more >>lications, determination of natalizumub washout period was no longer relevant.) Collapse << | - | - |
| NCT01420055 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Completed | - | France ... more >> Novartis Investigative Site Agen Cedex 9, France, 47923 Novartis Investigative Site Amiens Cedex 1, France, 80054 Novartis Investigative Site Angers Cedex 1, France, 49033 Novartis Investigative Site Auxerre, France, 89000 Novartis Investigative Site Avignon, France, 84000 Novartis Investigative Site Bayonne, France, 64109 Novartis Investigative Site Beziers, France, 34525 Novartis Investigative Site Bobigny Cedex, France, 93009 Novartis Investigative Site Brest, France, 29240 Novartis Investigative Site Calais, France, 62107 Novartis Investigative Site Castelnau-le-Lez, France, 34170 Novartis Investigative Site Chambray les Tours, France, 37175 Novartis Investigative Site Cherbourg, France, 50100 Novartis Investigative Site Clermont-Ferrand Cedex 1, France, 63003 Novartis Investigative Site Créteil, France, 94010 Novartis Investigative Site Dijon, France, 21034 Novartis Investigative Site Epinal, France, 88021 Novartis Investigative Site Grenoble Cedex 9, France, 38042 Novartis Investigative Site Hagueneau, France, 67500 Novartis Investigative Site La Rochelle cedex 1, France, 17019 Novartis Investigative Site La Seyne sur mer, France, 83500 Novartis Investigative Site Le Kremlin Bicetre, France, 94275 Novartis Investigative Site Le Mans Cedex 09, France, 72037 Novartis Investigative Site Limoges, France Novartis Investigative Site Lyon, France, 69275 Novartis Investigative Site Marseille, France, 13008 Novartis Investigative Site Marseille, France, 13012 Novartis Investigative Site Metz, France, 57085 Novartis Investigative Site Montpellier, France, 34000 Novartis Investigative Site Montpellier, France, 34295 Novartis Investigative Site Nimes, France, 32900 Novartis Investigative Site Orsay, France, 91401 Novartis Investigative Site Paris, France, 75005 Novartis Investigative Site Poitiers, France, 86021 Novartis Investigative Site Pontoise, France, F-95300 Novartis Investigative Site Reims, France, 51092 Novartis Investigative Site Rennes Cedex, France, 35033 Novartis Investigative Site Roanne, France, 42328 Novartis Investigative Site Saint Aubin sur Scie, France, 76550 Novartis Investigative Site Saint Jean, France, 31243 Novartis Investigative Site Sainte Foy les Lyon, France, 69110 Novartis Investigative Site Saumur, France, 49400 Novartis Investigative Site Strasbourg Cedex, France, 67091 Novartis Investigative Site Talant, France, 21240 Novartis Investigative Site Toulouse, France, 31059 Novartis Investigative Site Tourcoing, France, 59200 Novartis Investigative Site Tours Cedex, France, 37044 Collapse << |
| NCT01499667 | - | - | Terminated(Based on recent pub... more >>lications, determination of natalizumub washout period was no longer relevant.) Collapse << | - | - |
| NCT02373098 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novartis Investigative Site Istanbul, Turkey, 34093 Novartis Investigative Site Mecidiyekoy/Istanbul, Turkey, 34394 Novartis Investigative Site Uskudar / Istanbul, Turkey, 34668 Collapse << |
| NCT00355134 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT01497262 | - | - | Completed | - | - |
| NCT00355134 | - | - | Completed | - | - |
| NCT00662649 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00662649 | - | - | Completed | - | - |
| NCT01201356 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02048072 | Multiple Sclerosis ... more >> Autonomic Nervous System Dysfunction Collapse << | Phase 4 | Completed | - | Switzerland ... more >> Cantonal Hospital St. Gallen St. Gallen, SG, Switzerland, 9000 Collapse << |
| NCT01497262 | Multiple Sclerosis | Phase 3 | Completed | - | Argentina ... more >> Novartis Investigative Site Caba, Buenos Aires, Argentina, 1425 Novartis Investigative Site Guaymallen, Mendoza, Argentina, M5507XAD Novartis Investigative Site San Miguel de Tucuman, Tucumán, Argentina, T4000DPB Novartis Investigative Site San Miguel de Tucumán, Tucumán, Argentina, 4000 Novartis Investigative Site Cordoba, Argentina, X5004CDT Novartis Investigative Site Salta, Argentina, A4400ANG Novartis Investigative Site Salta, Argentina, A4400BKZ Brazil Novartis Investigative Site Rio de Janeiro, RJ, Brazil, 20270-004 Novartis Investigative Site Rio de Janeiro, RJ, Brazil, 21941-590 Novartis Investigative Site Porto Alegre, RS, Brazil, 90610-000 Colombia Novartis Investigative Site Barranquilla, Atlantico, Colombia Novartis Investigative Site Bogotá, Cundinamarca, Colombia, 110111 Novartis Investigative Site Bogotá, Colombia, 00000 Novartis Investigative Site Bogotá, Colombia Novartis Investigative Site Cali, Colombia Jordan Novartis Investigative Site Amman, Jordan, 11942 Novartis Investigative Site Irbid, Jordan, 22110 Malaysia Novartis Investigative Site Kuala Lumpur, Malaysia, 50586 Novartis Investigative Site Kuala Lumpur, Malaysia, 59100 Novartis Investigative Site Penang, Malaysia, 10990 Mexico Novartis Investigative Site Mexico, Distrito Federal, Mexico, 06700 Novartis Investigative Site México, Distrito Federal, Mexico, 10700 Novartis Investigative Site Monterrey, Nuevo León, Mexico, 64060 Novartis Investigative Site San Nicolas De Los Garza, Nuevo León, Mexico, 66480 Novartis Investigative Site San Luis Potosí, Mexico, 78240 Panama Novartis Investigative Site Panama City, Panamá, Panama Peru Novartis Investigative Site La Perla, Callao, Peru, 04 Novartis Investigative Site Jesus Maria, Lima, Peru, 11 Novartis Investigative Site San Isidro, Lima, Peru, 27 Collapse << |
| NCT02048072 | - | - | Completed | - | - |
| NCT03216915 | - | - | Recruiting | December 31, 2019 | Germany ... more >> University Medical Hospital Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Contact: Christoph Heesen, MD +49 (0) 40 7410 - 52124 heesen@uke.de Contact: Ruken Özge Akbulak, MD +49 (0) 40 7410 - 59471 r.akbulak@uke.de Collapse << |
| NCT00670449 | Multiple Sclerosis | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative Site Chiba, Japan, 276-8524 Novartis Investigative Site Ehime, Japan, 791-0295 Novartis Investigative Site Fukuoka, Japan, 807-8555 Novartis Investigative Site Gunma, Japan, 371-8511 Novartis Investigative Site Hyogo, Japan, 650-0017 Novartis Investigative Site Ibaraki, Japan, 305-8576 Novartis Investigative Site Kanagawa, Japan, 259-1193 Novartis Investigative Site Kyoto, Japan, 604-8453 Novartis Investigative Site Kyoto, Japan, 616-8255 Novartis Investigative Site Morioka, Japan, 020-8505 Novartis Investigative Site Niigata, Japan, 951-8520 Novartis Investigative Site Osaka, Japan, 556-0016 Novartis Investigative Site Osaka, Japan, 589-8511 Novartis Investigative Site Osaka, Japan Novartis Investigative Site Sapporo, Japan, 060-8648 Novartis Investigative Site Tochigi, Japan, 329-0498 Novartis Investigative Site Tokyo, Japan, 113-8519 Novartis Investigative Site Tokyo, Japan, 145-0065 Novartis Investigative Site Tokyo, Japan, 162-8666 Novartis Investigative Site Wakayama, Japan, 641-8510 Collapse << |
| NCT02325440 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Unknown | April 2016 | Germany ... more >> Universitaetsklinikum Muenster, Department of Neurology Recruiting Muenster, Germany, 48149 Contact: Luisa Klotz, PD Dr. med. +49 251 83444 ext 52 luisa.klotz@ukmuenster.de Principal Investigator: Luisa Klotz, PD Dr. med. Collapse << |
| NCT00670449 | - | - | Completed | - | - |
| NCT01757691 | - | - | Terminated(Discontinuation of ... more >>this study was based on Novartis decision to discontinue development of fingolimod for the treatment of ADON) Collapse << | - | - |
| NCT01941004 | Multiple Sclerosis (Relapsing ... more >>Remitting) Collapse << | Phase 3 | Withdrawn | May 2017 | - |
| NCT01791192 | Acute Noninfectious Posterior,... more >> Intermediate, or Pan Uveitis Collapse << | Phase 2 | Withdrawn | October 2014 | - |
| NCT01578330 | Multiple Sclerosis ... more >> Relapsing-Remitting Collapse << | Phase 4 | Completed | - | Turkey ... more >> Novartis Investigative Site Altunizade, Turkey, 34662 Novartis Investigative Site Ankara, Turkey, 06100 Novartis Investigative Site Ankara, Turkey, 06500 Novartis Investigative Site Atakum / Samsun, Turkey, 55139 Novartis Investigative Site Bursa, Turkey, 16059 Novartis Investigative Site Fatih / Istanbul, Turkey, 34098 Novartis Investigative Site Izmir, Turkey, 35040 Novartis Investigative Site Kocaeli, Turkey, 41380 Novartis Investigative Site Mecidiyekoy/Istanbul, Turkey, 34394 Novartis Investigative Site Trabzon, Turkey, 61080 Novartis Investigative Site Uskudar / Istanbul, Turkey, 34668 Collapse << |
| NCT01779700 | Schizophrenia | Phase 2 | Active, not recruiting | December 2017 | United States, Indiana ... more >> Center for NeuroImaging Indianapolis, Indiana, United States, 46202 Prevention and Recovery Center Indianapolis, Indiana, United States, 46202 Larue D Carter Memorial Hospital Indianapolis, Indiana, United States, 46222 Collapse << |
| NCT01578330 | - | - | Completed | - | - |
| NCT02002390 | Stroke Vascul... more >>ar Accident Cerebral Stroke Ischemic Cerebrovascular Accident Stroke, Acute Collapse << | Phase 2 | Completed | - | China, Tianjin ... more >> Tianjin Medical University General Hospital Tianjin, Tianjin, China, 300052 Collapse << |
| NCT01786174 | Amyotrophic Lateral Sclerosis | Phase 2 | Completed | - | United States, California ... more >> University of California, Irvine Orange, California, United States, 92868 United States, Georgia Georgia Regents University Augusta, Georgia, United States, 30912 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Texas Methodist Neurological Institute Houston, Texas, United States, 77030 Collapse << |
| NCT02956200 | Stroke Inflam... more >>mation Collapse << | Phase 2 | Unknown | December 2018 | China, Zhejiang ... more >> The second affiliated hospital of Zhejiang University Hangzhou, Zhejiang, China, 310000 Collapse << |
| NCT02575365 | Cognition Bra... more >>in Volume Loss Collapse << | Phase 4 | Terminated | - | Turkey ... more >> Novartis Investigative Site Bursa, Turkey, 16059 Novartis Investigative Site Kocaeli, Turkey, 41380 Novartis Investigative Site Kutahya, Turkey, 43000 Collapse << |
| NCT01757691 | Acute Demylelinating Optic Neu... more >>ritis Collapse << | Phase 2 | Terminated(Discontinuation of ... more >>this study was based on Novartis decision to discontinue development of fingolimod for the treatment of ADON) Collapse << | - | United States, Florida ... more >> Novartis Investigative Site St. Petersburg, Florida, United States, 33713 Spain Novartis Investigative Site Majadahonda, Madrid, Spain, 28222 Collapse << |
| NCT01786174 | - | - | Completed | - | - |
| NCT02490930 | Glioblastoma ... more >>Anaplastic Astrocytoma Collapse << | Early Phase 1 | Completed | - | United States, Maryland ... more >> The Johns Hopkins University Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01755871 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Terminated(lack of recruitment... more >>) Collapse << | - | Germany ... more >> Heinrich Heine Universität Düsseldorf Düsseldorf, Nord-Rhein Westfahlen, Germany, 40225 Collapse << |
| NCT03757338 | Healthy Volunteers | Phase 1 | Completed | - | New Zealand ... more >> Zenith Clinical Site Dunedin, New Zealand, 9010 Collapse << |
| NCT02137707 | - | - | Completed | - | Canada, British Columbia ... more >> University of British Columbia Vancouver, British Columbia, Canada, V6T 1Z3 Canada, Nova Scotia Dalhousie University Multiple Sclerosis Research Unit Halifax, Nova Scotia, Canada Canada, Ontario Ottawa General Hospital Ottawa, Ontario, Canada, K1H 8L6 Canada, Quebec Clinique Neuro Rive-Sud Greenfield Park, Quebec, Canada Montreal Neurological Institute Montreal, Quebec, Canada, H3A 2B4 Centre hospitalier de l'Universite de Montreal (CHUM) Montréal, Quebec, Canada Collapse << |
| NCT02061137 | Rett's Syndrome | Phase 1 Phase 2 | Completed | - | Switzerland ... more >> Department of Neuropediatrics - University Children's Hospital Basel, Switzerland, 4056 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| S1PR1 | IC50:0.033nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
