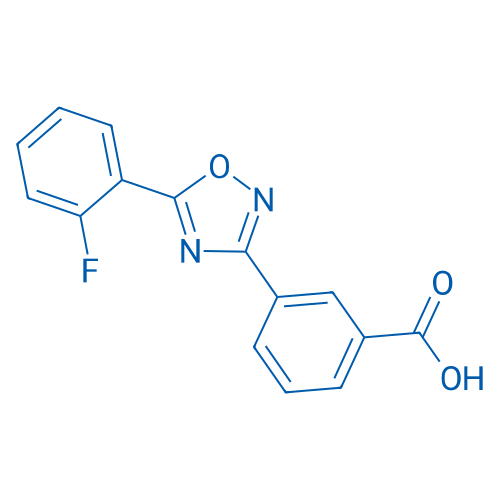
CAS No.: 775304-57-9
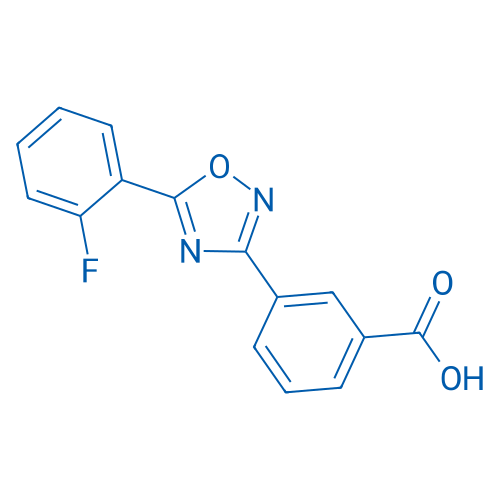
阿塔鲁伦 Catalog No. CSN12266
Synonyms: PTC124;阿塔鲁伦
PTC-124 is nonsense allele inhibitor, used to treat Duchenne muscular dystrophy.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03256799 Cystic Fibrosis Phase 4 Completed - United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 Collapse << NCT02139306 Cystic Fibrosis Phase 3 Unknown November 2016 - NCT01182324 - - Completed - United States, Maryland ... more >> National Human Genome Research Institute (NHGRI), 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << - 更多
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 775304-57-9 | 储存条件 |
|
|||||||||||||
| 分子式 | C15H9FN2O3 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 284.24 | 别名 | PTC124;阿塔鲁伦 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| BAC_MMA∗EGFP | 20 μM | Function Assay | 72 h | results in a significant increase in median peak fluorescence | 23041189 |
| HEK293T | 10 µg/ml | Function Assay | - | restores full-length harmonin a1 (∼80 kDa) in p.R31X-transfected cells | 23027640 |
| ML1 | 3.3/10 μM | Function Assay | 48 h | increases ARSB activity | 22971959 |
| ML2 | 3.3/10 μM | Function Assay | 48 h | increases ARSB activity | 22971959 |
| Mut−/−MUTStop+/− | 20 μM | Function Assay | 72 h | increases the amount of MUT mRNA | 23041189 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03256799 | Cystic Fibrosis | Phase 4 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 Collapse << |
| NCT02139306 | Cystic Fibrosis | Phase 3 | Unknown | November 2016 | - |
| NCT01182324 | - | - | Completed | - | United States, Maryland ... more >> National Human Genome Research Institute (NHGRI), 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02369731 | - | - | Recruiting | April 2022 | Austria ... more >> Krankenhaus der Barmherzigen Schwestern Linz Recruiting Linz, Austria, 4010 Principal Investigator: Veronika Pilshofer, Dr Preyer Children Hospital Recruiting Wien, Austria, 1100 Principal Investigator: Guenther Bernert, Pr France CHU Brest, hôpital Morvan Recruiting Best, France, 29609 Principal Investigator: Ropars Juliette, Dr CHU Bordeaux Pelgrin Recruiting Bordeaux Cedex, France, 33076 Principal Investigator: Caroline Espil-Taris L'Escale Hôpital Femme Mère Enfant- Hospices Civils de Lyon Recruiting Bron, France, 69500 Principal Investigator: Carole Vuillerot, Dr CHU Clermont-Ferrand, hôpital Estaing Recruiting Clermont-Ferrand, France, 63003 Principal Investigator: Catherine Sarret, Dr CHU de Martinique - Hôpital P. Zobda-Quitman Recruiting Fort De France Cedex, France, 97261 Principal Investigator: Rémi Bellance, Dr CHRU de Lille Recruiting Lille Cedex, France, 59037 Principal Investigator: Jean-Marie Cuisset, Dr CHU Marseille Hôpital de la Timone Recruiting Marseille Cedex 5, France, 13385 Principal Investigator: Brigitte Chabrol, Pr CHU Nantes Recruiting Nantes, France, 44000 Principal Investigator: Yan Pereon, Pr Hôpital Armand Trousseau Recruiting Paris, France, 75012 Principal Investigator: Gidaro Teresa, Dr Hôpital Necker Recruiting Paris, France, 75015 Principal Investigator: Isabelle Desguerre, Pr CHU Reims Recruiting Reims, France, 51100 Principal Investigator: Pascal Sabouraud, Dr CHU Saint-Etienne MPR pédiatrique Hôpital Bellevue Recruiting Saint Etienne Cedex, France, 42055 Principal Investigator: Stéphane Chabrier, Dr CHU de la Réunion - GHSR - GH Sud-Réunion Recruiting Saint Pierre, France, 97448 Principal Investigator: Renouil Michel, Dr CHU Strasbourg Recruiting Strasbourg Cedex, France, 67098 Principal Investigator: Vincent Laugel, Pr CHU Toulouse Recruiting Toulouse, France, 31059 Principal Investigator: Pascal Cintas, Dr Hôpital d'enfants Gatien de Clocheville Recruiting Tours Cedex 9, France, 37044 Principal Investigator: Lagrue Emmanuelle, Dr CHU Nancy Hôpital de Brabois-Enfants Rue du Morvan Recruiting Vandoeuvre-les-Nancy, France, 54500 Principal Investigator: Emmanuel Raffo, Pr Germany Klinikum Bayreuth GmbH Recruiting Bayreuth, Germany, 95445 Principal Investigator: Almut Hirsch, Dr Charité Universitätsmedizin Berlin Recruiting Berlin, Germany, 13353 Principal Investigator: Claudia Weiss, Dr Universitätsklinikum Erlangen Recruiting Erlangen, Germany, 91054 Principal Investigator: Regina Trollmann, Pr Universitaetsklinikum Essen (AoR) Recruiting Essen, Germany, 45147 Principal Investigator: Ulrike Schara, Pr SPZ Frankfurt Mitte Recruiting Frankfurt am Main, Germany, 60316 Principal Investigator: Jurgen Seeger, Dr Universitaetsklinikum Freiburg Recruiting Freiburg, Germany, 79106 Principal Investigator: Janbernd Kirschner, Pr UKE, Universitätsklinikum Hamburg-Eppendorf Recruiting Hamburg, Germany, 20246 Principal Investigator: Jessica Johannsen, Dr Israel Hadassah Medical Center Recruiting Jerusalem, Israel, 91120 Principal Investigator: Talya Dor, Dr Chaim Sheba Medical Center Ramat Gan, Neuromuscle clinic Recruiting Ramat Gan, Israel, 52621 Principal Investigator: Amir Dori, Dr Sweden Queen Silvia Children's Hospital Recruiting Goteborg, Sweden, 41685 Principal Investigator: Mar Tulinius, Pr Collapse << |
| NCT00237380 | Cystic Fibrosis | Phase 2 | Completed | - | Israel ... more >> Hadassah University Hospital - Mount Scopus Jerusalem, Israel, 91240 Collapse << |
| NCT00458341 | Cystic Fibrosis | Phase 2 | Completed | - | Belgium ... more >> Reine Fabiola Hospital Brussels, Belgium, 1020 UZ Gasthuisberg Leuven Leuven, Belgium, 3000 France Hopital Necker Enfants Malades Paris, France, 75015 Collapse << |
| NCT00264888 | Duchenne Muscular Dystrophy | Phase 2 | Completed | - | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104-4399 United States, Utah University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT03256968 | Cystic Fibrosis | Phase 4 | Active, not recruiting | December 31, 2018 | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 Collapse << |
| NCT00234663 | Cystic Fibrosis | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233-1711 United States, California Stanford University Medical Center Palo Alto, California, United States, 94304-5786 United States, Colorado The Children's Hospital Denver, Colorado, United States, 80218 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Ohio Rainbow Babies and Children's Hospital Cleveland, Ohio, United States, 44106 Collapse << |
| NCT03179631 | Muscular Dystrophy, Duchenne ... more >> Muscular Dystrophies Muscular Disorders, Atrophic Muscular Diseases Musculoskeletal Disease Neuromuscular Diseases Nervous System Diseases Genetic Diseases, X-Linked Genetic Diseases, Inborn Collapse << | Phase 3 | Recruiting | December 31, 2021 | - |
| NCT02409004 | Healthy | Phase 1 | Completed | - | Canada ... more >> Inventiv Quebec, Canada, G1P 0A2 Collapse << |
| NCT00351078 | Cystic Fibrosis | Phase 2 | Completed | - | Israel ... more >> Hadassah University Hospital - Mount Scopus Jerusalem, Israel, 91240 Collapse << |
| NCT02647359 | Aniridia | Phase 2 | Active, not recruiting | December 2019 | United States, Oregon ... more >> Casey Eye Institute, Oregon Health & Science University Portland, Oregon, United States, 97239 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908 Canada, British Columbia University of British Columbia Vancouver, British Columbia, Canada, V5Z3N9 Collapse << |
| NCT00592553 | Duchenne Muscular Dystrophy ... more >> Becker Muscular Dystrophy Collapse << | Phase 2 | Completed | - | - |
| NCT01141075 | Amino Acid Metabolism, Inborn ... more >>Errors Collapse << | Phase 2 | Terminated | - | Belgium ... more >> ZNA Queen Paola Child Hospital and Provincial Centre for Metabolic Disorders Antwerp, Belgium France Hôpital Edouard Herriot Lyon, France Necker-Enfants Malades Hospital Paris, France Germany University Children's Hospital Duesseldorf, Germany Italy Istituti Clinici di Perfezionamento, Milano Milan, Italy Federico II University Naples, Italy University Hospital, Department of Pediatrics Padova, Italy Switzerland University Children's Hospital Zürich, Switzerland United Kingdom Great Ormand Street Hospital London, United Kingdom Collapse << |
| NCT00947193 | Hemophilia A ... more >>Hemophilia B Collapse << | Phase 2 | Suspended | May 2012 | United States, Illinois ... more >> The Bleeding and Clotting Disorders Institute Peoria, Illinois, United States, 61614 United States, Indiana St. Vincent Indianapolis Hospital Indianapolis, Indiana, United States, 46260 United States, Massachusetts New England Hemophilia Center Worcester, Massachusetts, United States, 01605 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt Hemostatis and Thrombosis Clinic Nashville, Tennessee, United States, 37232 United States, Washington Puget Sound Blood Center Seattle, Washington, United States, 98104 Canada, British Columbia St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z 1Y6 France Hôpital Cardiologique Lille Cedex, France Hôpital Edouard Herriot Lyon Cedex, France Hôpital Necker Enfants Malades Paris, France Italy Azienda Ospedaliero-Universitaria Careggi Viale G.B. Morgagni Firenze, Italy A.Bianchi Bonomi Hemophilia and Thrombosis Center Milano, Italy Collapse << |
| NCT01140451 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT01826487 | Muscular Dystrophy, Duchenne ... more >> Muscular Dystrophies Muscular Disorders, Atrophic Muscular Diseases Musculoskeletal Diseases Neuromuscular Diseases Nervous System Diseases Genetic Diseases, X-Linked Genetic Diseases, Inborn Collapse << | Phase 3 | Completed | - | - |
| NCT00759876 | Duchenne Muscular Dystrophy | Phase 2 | Terminated | - | United States, Ohio ... more >> Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Utah University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00803205 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02758626 | Epilepsy | Phase 2 | Active, not recruiting | December 2022 | United States, New York ... more >> New York University School of Medicine New York, New York, United States, 10016 Collapse << |
| NCT01247207 | Duchenne Muscular Dystrophy | Phase 3 | Enrolling by invitation | December 31, 2018 | United States, California ... more >> University of California-Davis Sacramento, California, United States, 95817 United States, Colorado The Children's Hospital Aurora, Colorado, United States, 80045 United States, Florida Child Neurology Center of NW Florida Gulf Breeze, Florida, United States, 32561 United States, Iowa University of Iowa Children's Hospital Iowa City, Iowa, United States, 52242 United States, Kansas University of Kansas Medical Center Kansas City, Kansas, United States, 66160 United States, Massachusetts Children's Hospital of Boston/Harvard Medical School Boston, Massachusetts, United States, 02115 United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States, 55455 United States, Missouri Washington University Medical School Saint Louis, Missouri, United States, 63110 United States, New York Columbia University Medical School New York, New York, United States, 10032 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Cincinnati Childrens Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Oregon Shriners Hospital for Children-Portland Portland, Oregon, United States, 97239 United States, Pennsylvania Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 United States, Texas Southwestern University Dallas, Texas, United States, 75207 United States, Utah University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT03648827 | Duchenne Muscular Dystrophy | Phase 2 | Not yet recruiting | February 29, 2020 | United States, Arizona ... more >> Phoenix Childrens Hospital Not yet recruiting Phoenix, Arizona, United States, 85016 Contact: Nakia Croft 602-933-0641 ncroft@phoenixchildrens.com Principal Investigator: Saunder Bernes United States, California University of California, Los Angeles (UCLA) Not yet recruiting Los Angeles, California, United States, 90025 Contact: Emilie Douine 310-267-2416 edouine@mednet.ucla.edu Principal Investigator: Stanley Nelson University of California (UC) Davis Medical Center Not yet recruiting Sacramento, California, United States, 95817 Contact: Omaid Sarwary 916-734-0968 omsarwary@ucdavis.edu Principal Investigator: Craig McDonald United States, Illinois Rush University Medical Center Not yet recruiting Chicago, Illinois, United States, 60612 Contact: Susan Rohde 312-942-0079 susan_rohde@rush.edu Principal Investigator: Peter Heydemann United States, Kansas University of Kansas Medical Center Not yet recruiting Kansas City, Kansas, United States, 66160 Contact: Katie Roath Principal Investigator: Jeffrey Statland United States, Minnesota University of Minnesota Not yet recruiting Minneapolis, Minnesota, United States, 55455 Contact: Natalya Alassy 612-626-4690 burla019@umn.edu Principal Investigator: Peter Karachunski United States, New York Columbia University College of Physicians & Surgeons Not yet recruiting New York, New York, United States, 10032 Contact: Ameneh Onativia 212-342-3679 am2959@cumc.columbia.edu Principal Investigator: Darryl De Vivo United States, Texas Texas Children's Hospital Not yet recruiting Houston, Texas, United States, 77030 Contact: Monica Garza 832-822-1255 mxgarza9@texaschildrens.org Principal Investigator: Tim Lotze University of Texas Heath Science Center at San Antonio Not yet recruiting San Antonio, Texas, United States, 78229-3900 Contact: Yogeet Kaur 210-567-8222 kaury@uthscsa.edu Principal Investigator: Ratna Bhavaraju-Sanka United States, Virginia Children's Hospital of the King's Daughters Not yet recruiting Norfolk, Virginia, United States, 23507 Contact: Terrie Karras Conklin 757-469-9123 terrie.conklin@chkd.org Principal Investigator: Crystal Proud Collapse << |
| NCT00847379 | Duchenne Muscular Dystrophy ... more >> Becker Muscular Dystrophy Collapse << | Phase 2 Phase 3 | Terminated | - | - |
| NCT02456103 | Cystic Fibrosis | Phase 3 | Terminated(CF Data from CF stu... more >>dy 021 did not meet end points) Collapse << | - | - |
| NCT02819557 | Duchenne Muscular Dystrophy | Phase 2 | Completed | - | United States, Florida ... more >> Child Neuro NWF Gulf Breeze, Florida, United States, 32561 United States, Illinois Rush University Medical Center Chicago, Illinois, United States, 60612 United States, Massachusetts Children's Hospital Boston Boston, Massachusetts, United States, 02115 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 United States, Texas Children's Medical Center Dallas Dallas, Texas, United States, 75390-8843 United States, Utah University of Utah Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT01009294 | Duchenne Muscular Dystrophy ... more >> Becker Muscular Dystrophy Collapse << | Phase 2 | Terminated | - | United States, California ... more >> University of California-Davis Davis, California, United States, 95616 United States, Massachusetts Children's Hospital of Boston Boston, Massachusetts, United States, 02115 United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States, 55455 United States, Missouri Washington University Medical School Saint Louis, Missouri, United States, 63110 United States, Ohio Nationwide Children's Hospital Columbus, Ohio, United States, 43205 United Kingdom University of Newcastle Newcastle upon Tyne, United Kingdom, NE1 3BZ Collapse << |
| NCT01557400 | Duchenne Muscular Dystrophy ... more >> Becker Muscular Dystrophy Dystrophinopathy Collapse << | Phase 3 | Active, not recruiting | March 2018 | Australia, Melbourne ... more >> Royal Children's Hospital Parkville, Melbourne, Australia Australia Institute For Neuromuscular Research, The Children's Hospital at Westmead Westmead, Australia Belgium University Hospital KU Leuven Leuven, Belgium Canada, Alberta Alberta Children's Hospital Calgary, Alberta, Canada Canada, British Columbia British Columbia Children's Hospital Vancouver, British Columbia, Canada Canada, Ontario Children's Hospital of Western Ontario London, Ontario, Canada France Hôpital d'Enfants CHU Timone Marseille, France Laboratoire d'Exploration Fonctionnelles Nantes, France Groupe Hospitalier La Pitie-Salpetriere Paris, France Germany University of Essen - Clinic for Children Essen, Germany University Hospital Freiburg, Germany Israel Hadassah Medical Center, Hebrew University Hospital Jerusalem, Israel Italy Ospedale Maggiore Policlinico in Milan Milan, Italy Ospedale Pediatrico Bambino Gesu Rome, Italy U.O. Complessa di Neuropsichiatria Infantile Rome, Italy Spain Hospital Sant Joan de déu Barcelona, Spain Hospital Universitari La Fe Valencia, Spain Sweden Queen Silvia Children's Hospital Göteborg, Sweden Astrid Lindgren Pediatric Hospital Stockholm, Sweden United Kingdom Great Ormond Street Hospital London, United Kingdom University of Newcastle Institute of Human Genetics Newcastle Upon Tyne, United Kingdom Collapse << |
| NCT02107859 | Cystic Fibrosis | Phase 3 | Terminated(CF Data from CF stu... more >>dy 021 did not meet end points.) Collapse << | - | United States, Alabama ... more >> University of Alabama-Birmingham Birmingham, Alabama, United States, 35233 United States, California Miller Children's Hospital Long Beach Long Beach, California, United States, 90806 United States, Colorado Denver Children's Hospital Aurora, Colorado, United States, 80045 United States, Illinois Children's Hospital Chicago Chicago, Illinois, United States, 60614 United States, Massachusetts Children's Hospital Boston Boston, Massachusetts, United States, 02115 United States, New York Beth Israel Medical Center New York, New York, United States, 10003 United States, Ohio Rainbow Babies & Children's Hospital Cleveland, Ohio, United States, 44106 Belgium Hôpital Universitaire des Enfants Reine Fabiola Brussels, Belgium University Hospital Brussels Brussels, Belgium University Hospital Leuven Leuven, Belgium France Hôpital Necker - Enfants Malades Paris, France Hôpital des Enfants Toulouse, France, 31059 Israel Hadassah University Hospital - Mount Scopus Jerusalem, Israel, 91240 Italy Università La Sapienza Roma, Italy Azienda Ospedaliera di Verona Verona, Italy Spain Hospital Universitario La Paz Madrid, Spain Sweden Karolinska University Hospital, Huddinge Stockholm, Sweden Collapse << |
| NCT02090959 | Muscular Dystrophy, Duchenne ... more >> Muscular Dystrophies Muscular Disorders, Atrophic Muscular Diseases Musculoskeletal Diseases Neuromuscular Diseases Nervous System Diseases Genetic Diseases, X-Linked Genetic Diseases, Inborn Collapse << | Phase 3 | Active, not recruiting | June 2018 | - |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
