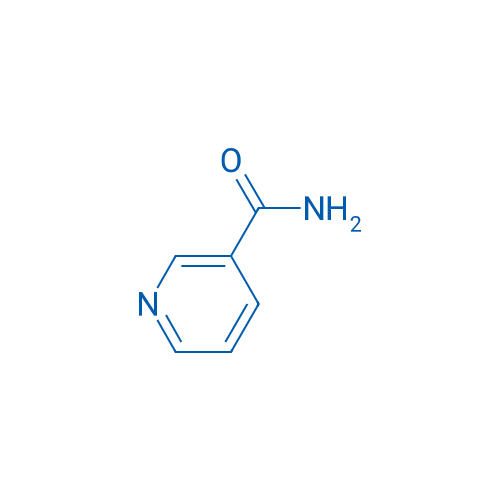
CAS No.: 98-92-0
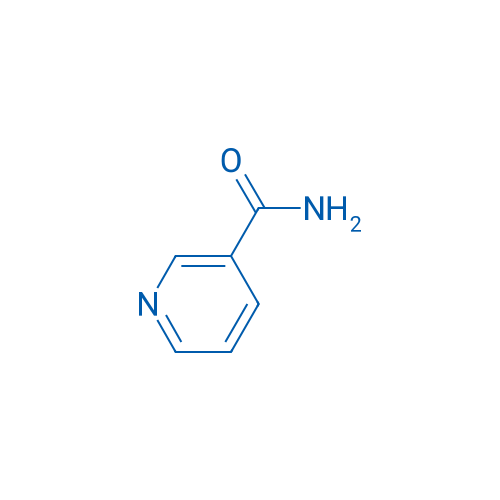
烟酰胺 Catalog No. CSN16796
Synonyms: 烟酰胺;Niacinamide;Nicotinic acid amide
Nicotinamide can act as an inhibitor of sirtuins and it is the active component of NAD and NADP.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
- 细胞研究
- Cell Data
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00203476 Hyperlipidemia ... more >> Hypercholesterolemia Collapse << Phase 4 Completed - United States, Alabama ... more >> Tuscaloosa Research & Education Advancement Corporation Tuscaloosa, Alabama, United States, 35404 Collapse << NCT03615534 Atherogenic Dyslipidemia ... more >> Obesity Associated Disorder Collapse << Phase 4 Completed - Iraq ... more >> Al Kindy College of Medicine, University of Baghdad Baghdad, Iraq, 10045 Lewai S Abdulaziz Baghdad, Iraq, 10045 Collapse << NCT02061267 Metabolic Syndrome Not Applicable Unknown December 2014 Spain ... more >> Instituto de la Grasa, CSIC Seville, Spain, 41012 Collapse << - 更多
- 参考文献
- [1] Bitterman KJ, Anderson RM, et al. Inhibition of silencing and accelerated aging by nicotinamide, a putative negative regulator of yeast sir2 and human SIRT1. J Biol Chem. 2002 Nov 22;277(47):45099-107.
- [2] Otonkoski T, Beattie GM, et al. Nicotinamide is a potent inducer of endocrine differentiation in cultured human fetal pancreatic cells. J Clin Invest. 1993 Sep;92(3):1459-66
- [3] Otonkoski T, Beattie GM, Mally MI, Ricordi C, Hayek A. Nicotinamide is a potent inducer of endocrine differentiation in cultured human fetal pancreatic cells. J Clin Invest. 1993 Sep;92(3):1459-66. doi: 10.1172/JCI116723. PMID: 8104197; PMCID: PMC288291.
- [4] Shetty PK, Galeffi F, Turner DA. Nicotinamide pre-treatment ameliorates NAD(H) hyperoxidation and improves neuronal function after severe hypoxia. Neurobiol Dis. 2014 Feb;62:469-78. doi: 10.1016/j.nbd.2013.10.025. Epub 2013 Oct 31. PMID: 24184921; PMCID: PMC4143422.
- [5] Green KN, Steffan JS, Martinez-Coria H, Sun X, Schreiber SS, Thompson LM, LaFerla FM. Nicotinamide restores cognition in Alzheimer's disease transgenic mice via a mechanism involving sirtuin inhibition and selective reduction of Thr231-phosphotau. J Neurosci. 2008 Nov 5;28(45):11500-10. doi: 10.1523/JNEUROSCI.3203-08.2008. PMID: 18987186; PMCID: PMC2617713.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 98-92-0 | 储存条件 |
|
|||||
| 分子式 | C6H6N2O | 运输 | 蓝冰 | |||||
| 分子量 | 122.12 | 别名 | 烟酰胺;Niacinamide;Nicotinic acid amide | |||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| Escherichia coli BL21 (DE3) cells | - | Function assay | - | Inhibition of catalytically active human SIRT3 (102 to 399 amino acids) expressed in Escherichia coli BL21 (DE3) cells using fluorogenic 7-amino-4-methylcoumarin (AMC)-labeled peptide by fluorescence assay, IC50=6.2 μM | 25275824 |
| human SK-MEL-28 cells | 100 μM | Function assay | 24 h | Reduction in ATP level in human SK-MEL-28 cells at 100 uM maintained in Locke's solution after 24 hrs by luciferase-based assay in absence of GF and glucose | 22835719 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00203476 | Hyperlipidemia ... more >> Hypercholesterolemia Collapse << | Phase 4 | Completed | - | United States, Alabama ... more >> Tuscaloosa Research & Education Advancement Corporation Tuscaloosa, Alabama, United States, 35404 Collapse << |
| NCT03615534 | Atherogenic Dyslipidemia ... more >> Obesity Associated Disorder Collapse << | Phase 4 | Completed | - | Iraq ... more >> Al Kindy College of Medicine, University of Baghdad Baghdad, Iraq, 10045 Lewai S Abdulaziz Baghdad, Iraq, 10045 Collapse << |
| NCT02061267 | Metabolic Syndrome | Not Applicable | Unknown | December 2014 | Spain ... more >> Instituto de la Grasa, CSIC Seville, Spain, 41012 Collapse << |
| NCT00796887 | Ischemic Stroke | Phase 2 | Completed | - | United States, Michigan ... more >> Henry Ford Hospital Detroit, Michigan, United States, 48202 Collapse << |
| NCT03615534 | - | - | Completed | - | - |
| NCT01258491 | Healthy Subjects | Phase 1 | Completed | - | - |
| NCT01010516 | Dyslipidemia | Phase 4 | Unknown | - | Greece ... more >> University of Ioannina Medical School Recruiting Ioannina, Greece, 45 110 Contact: M S Elisaf, MD +302651007509 egepi@cc.uoi.gr Principal Investigator: Moses S Elisaf, MD Sub-Investigator: Evangelos N Liberopoulos, MD Sub-Investigator: Anastazia Kei, MD Collapse << |
| NCT00203476 | - | - | Completed | - | - |
| NCT01426438 | - | - | Completed | - | - |
| NCT01426438 | HIV-1 Infection | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama Therapeutics CRS (5801) Birmingham, Alabama, United States, 35294 United States, California University of Southern California (1201) Los Angeles, California, United States, 90033-1079 UCLA CARE Center CRS (601) Los Angeles, California, United States, 90095 Harbor-UCLA Med. Ctr. CRS (603) Torrance, California, United States, 90502 United States, Colorado University of Colorado Hospital CRS (6101) Aurora, Colorado, United States, 80045 United States, Illinois Northwestern University CRS (2701) Chicago, Illinois, United States, 60611 United States, New Jersey New Jersey Medical School-Adult Clinical Research Ctr. CRS (31477) Newark, New Jersey, United States, 07103 United States, New York NY Univ. HIV/AIDS CRS (401) New York, New York, United States, 10016 United States, North Carolina Unc Aids Crs (3201) Chapel Hill, North Carolina, United States, 27516 Duke Univ. Med. Ctr. Adult CRS (1601) Durham, North Carolina, United States, 27710 Moses H. Cone Memorial Hospital CRS (3203) Greensboro, North Carolina, United States, 27401 United States, Ohio Univ. of Cincinnati CRS (2401) Cincinnati, Ohio, United States, 45267 Case CRS (2501) Cleveland, Ohio, United States, 44106 United States, Washington University of Washington AIDS CRS (1401) Seattle, Washington, United States, 98104 Collapse << |
| NCT03565328 | Heart Failure | Phase 2 | Recruiting | June 1, 2021 | United States, Maryland ... more >> Walter Reed National Military Medical Center Recruiting Bethesda, Maryland, United States, 20889 Contact: Autumn Mains, B.A. 410-627-3494 autumn.mains.ctr@usuhs.edu National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT03392623 | Melasma | Early Phase 1 | Completed | - | - |
| NCT00176020 | Healthy | Phase 1 | Unknown | - | - |
| NCT00986986 | HIV Infections ... more >> Dyslipidemia Endothelial Dysfunction Collapse << | Not Applicable | Completed | - | United States, Hawaii ... more >> University of Hawaii - Hawaii Center for AIDS Honolulu, Hawaii, United States, 96816 Collapse << |
| NCT00880178 | - | - | Completed | - | - |
| NCT00712049 | Dyslipidemia ... more >>Atherosclerosis Collapse << | Phase 4 | Unknown | - | Austria ... more >> Medical University Vienna Recruiting Vienna, Austria, 1090 Contact: Renate Koppensteiner, Prof. Dr. 00431404004671 renate.koppensteiner@meduniwien.ac.at Sub-Investigator: Martin Schillinger, Prof. Dr. Sub-Investigator: Jasmin Amighi, Dr. Sub-Investigator: Schila Sabeti, Dr. Collapse << |
| NCT00986986 | - | - | Completed | - | - |
| NCT01583647 | Hypercholesterolemia, Familial... more >> Heterozygous Familial Hypercholesterolemia Collapse << | Phase 1 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs.) Collapse << | - | - |
| NCT03151239 | Glucose Metabolism Disorders | Not Applicable | Recruiting | June 1, 2020 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Sally Torbitzky, RN, BSN 314-362-9950 storbitz@wustl.edu Contact: Mihoko Yoshino, MD 314-362-8228 myoshino@wustl.edu Principal Investigator: Samuel Klein, MD Collapse << |
| NCT00633698 | Elevated Lipoprotein(a) Levels | Phase 3 | Unknown | March 2010 | Germany ... more >> Institute of Social Medicine, Epidemiology and Health Economics Berlin, Germany, 10098 Collapse << |
| NCT01583647 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs.) Collapse << | - | - |
| NCT00852969 | Chronic Kidney Disease | Phase 4 | Completed | - | United States, Massachusetts ... more >> Tufts Medical Center Boston, Massachusetts, United States, 02111 Collapse << |
| NCT01274559 | Primary Hypercholesterolemia ... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT00852969 | - | - | Completed | - | - |
| NCT00508989 | Sickle Cell Disease | Phase 2 | Completed | - | United States, District of Col... more >>umbia Howard University Hospital Washington, District of Columbia, United States, 20060 United States, Maryland National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01274559 | - | - | Terminated(In HPS2-THRIVE, MK-... more >>0524A did not meet the primary efficacy objective and there was a significant increase in incidence of some types of non-fatal SAEs) Collapse << | - | - |
| NCT01159054 | - | - | Terminated(The funding source ... more >>is not going to fund this anymore. Only two subjects completed the study therefore meaningful analysis not possible.) Collapse << | - | - |
| NCT00608699 | Healthy Subjects ... more >> Dyslipidaemias Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT03163576 | Hyperphosphatemia | Phase 4 | Not yet recruiting | December 2019 | - |
| NCT00913081 | Flushing | Phase 4 | Completed | - | United States, Pennsylvania ... more >> CTRC Univ. of Penn - Andrew Mutch Bldg., 4th floor Phila, Pennsylvania, United States, 19104 University of Pennsylvania Phila, Pennsylvania, United States, 19104 Collapse << |
| NCT01159054 | Dialysis Card... more >>iovascular Disease Atherosclerosis Inflammation Collapse << | Not Applicable | Terminated(The funding source ... more >>is not going to fund this anymore. Only two subjects completed the study therefore meaningful analysis not possible.) Collapse << | - | United States, Massachusetts ... more >> Brigham and Women's Hospital Boston, Massachusetts, United States, 02115 BWH/FH/DCI Outpatient Dialysis Unit Boston, Massachusetts, United States, 02130 DCI Dialysis Unit-Somerville Somerville, Massachusetts, United States Collapse << |
| NCT00533611 | Flushing | Phase 3 | Completed | - | - |
| NCT00913081 | - | - | Completed | - | - |
| NCT01083329 | Obesity | Phase 2 | Completed | - | France ... more >> Centre d'Investigation Clinique, Purpan University Toulouse Hospital Toulouse, France, 31059 Collapse << |
| NCT02458924 | Schizophrenia | Phase 2 Phase 3 | Completed | - | - |
| NCT00300365 | Metabolic Syndrome | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00300365 | - | - | Completed | - | - |
| NCT01391377 | Peripheral Arterial Disease | Not Applicable | Terminated(Tredaptive has been... more >> suspended worldwide) Collapse << | December 2014 | Australia, Victoria ... more >> Baker IDI Heart and diabetes research institute Melbourne, Victoria, Australia, 3004 Collapse << |
| NCT01118598 | Polycystic Ovary Syndrome | Phase 4 | Completed | - | United Kingdom ... more >> Hull & East Yorkshire Hospitals NHS Trust Hull, United Kingdom, HU3 2RW Collapse << |
| NCT01221402 | Aortocoronary Saphenous Vein B... more >>ypass Graft Atherosclerosis Intermediate Saphenous Vein Graft Lesions Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> VA North Texas Healthcare System Dallas, Texas, United States, 75216 Collapse << |
| NCT03176628 | Acute Kidney Injury | Not Applicable | Recruiting | October 31, 2018 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02118 Contact: Petra Simic, MD, PhD 617-724-6700 psimic@bwh.harvard.edu Contact: Katherine E Brock, BS 617-643-9463 kbrock2@mgh.harvard.edu Principal Investigator: Eugene Rhee, MD Sub-Investigator: Ravi I Thadhani, MD, MPH Sub-Investigator: Petra Simic, MD, PhD Collapse << |
| NCT01071525 | Hypercholesterolemia | Phase 3 | Unknown | October 2010 | Israel ... more >> Rambam Health Care Campus Not yet recruiting Haifa, Israel, 31096 Contact: Shadi +972-523591876 s_hamoud@rambam.health.gov.il Contact: Tony Hayek, Prof +972-523782009 t_hayek@rambam.health.gov.il Sub-Investigator: Tony Hayek, Prof Collapse << |
| NCT01308203 | Coronary Artery Disease ... more >> Dyslipidemias Collapse << | Phase 4 | Terminated(Merck has decided t... more >>o discontinue all studies with extended-release niacin/laropiprant.because the HPS2-THRIVE did not meet its primary endpoint) Collapse << | - | Argentina ... more >> Hospital Italiano de Buenos Aires Buenos Aires, Argentina, C1181ACH Collapse << |
| NCT00605930 | Progressive Supranuclear Palsy | Not Applicable | Completed | - | United States, Kentucky ... more >> Frazier Rehab Louisville, Kentucky, United States, 40202 Collapse << |
| NCT00316472 | Hyperphosphatemia | Phase 1 | Completed | - | United States, Missouri ... more >> Washington University Medical Center Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT00485758 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01542138 | Hyperpigmentation | Phase 4 | Completed | - | Mexico ... more >> Dermatology Department. Hospital Central "Dr. Ignacio Morones Prieto" San Luis Potosi, Mexico, 78210 Collapse << |
| NCT02701127 | Acute Kidney Injury | Early Phase 1 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00485758 | - | - | Completed | - | - |
| NCT02942888 | Mild Cognitive Impairment ... more >> NAD Collapse << | Not Applicable | Recruiting | December 2018 | United States, Texas ... more >> South Texas Veterans Healthcare System (STVHCS) Recruiting San Antonio, Texas, United States, 78229 Contact: Joan Hecht 210-617-5300 ext 14654 Joan.hecht@va.gov University of Texas Health San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Becky Powers, MD 210-617-5179 Becky.powers@va.gov Collapse << |
| NCT01619228 | Premature Birth of Newborn | Not Applicable | Terminated(PI left the institu... more >>tion resulting in the study being closed early.) Collapse << | - | United States, Ohio ... more >> Cincinnati Childrens Hospital Medical Center Neonatal Intensive Care Unit Cincinnati, Ohio, United States, 45229 University Hospital Neonatal Intensive Care Unit Cincinnati, Ohio, United States, 45267 Collapse << |
| NCT00493064 | Retinal Vein Occlusion | Phase 2 Phase 3 | Completed | - | United States, California ... more >> Palo Alto Medical Foundation Department of Ophthalmology Palo Alto, California, United States, 94301 Collapse << |
| NCT03685253 | Diabetic Neuropathy Peripheral | Phase 1 Phase 2 | Not yet recruiting | October 1, 2023 | United States, Maryland ... more >> University of Maryland Not yet recruiting Baltimore, Maryland, United States, 21201 Contact: Neda M Ilieva, B.S. 410-328-6583 inmed@som.umaryland.edu Contact: James W Russell, MD (410) 328-3100 jrussell@som.umaryland.edu Principal Investigator: James W Russell, MD Sub-Investigator: Lindsay A Zilliox, MD Collapse << |
| NCT02153879 | Type 2 Diabetes Mellitus ... more >> Dyslipidemia Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Universitari Sant Joan Reus, Tarragona, Spain, 43204 Collapse << |
| NCT02721537 | Concussion, Mild | Not Applicable | Recruiting | June 2019 | United States, Minnesota ... more >> University of Minnesota Center for Magnetic Resonance Research Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Stacy Valenzuela, RAC 612-624-9926 majes014@umn.edu Principal Investigator: Xiao-Hong Zhu, Ph.D. Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Heather Ondler Hinson 507-284-4799 ondlerhinson.heather@mayo.edu Contact: Chuck Barta 507-266-5566 barta.charles@mayo.edu Principal Investigator: Brent A Bauer, MD Collapse << |
| NCT00691210 | Hodgkin's Disease ... more >> Non-Hodgkin's Lymphoma Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Columbia University Medical Center NY, New York, United States, 10022 Collapse << |
| NCT01239992 | Hyperlipoproteinemia ... more >> Metabolic Syndrome Collapse << | Phase 4 | Terminated(negative endpoint s... more >>tudy resulting in withdrawal of study drug) Collapse << | - | Germany ... more >> Medizinische Klinik II, Klinikum der Universitaet Muenchen, Grosshadern Munich, Germany, 81377 Collapse << |
| NCT03626298 | Acne Vulgaris | Phase 4 | Completed | - | - |
| NCT03370848 | Flushing Hype... more >>rlipidemias Dyslipidemias Collapse << | Phase 4 | Completed | - | United States, California ... more >> VA West Los Angeles Medical Center Los Angeles, California, United States, 90073 Collapse << |
| NCT00508885 | Hyperphosphatemia | Phase 1 Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine St. Louis, Missouri, United States, 63110 Collapse << |
| NCT02303483 | Obese | Not Applicable | Completed | - | Denmark ... more >> Medical Research Laboratories, Aarhus University Hospital Aarhus, Denmark, 8000 Collapse << |
| NCT00691210 | - | - | Completed | - | - |
| NCT01239992 | - | - | Terminated(negative endpoint s... more >>tudy resulting in withdrawal of study drug) Collapse << | - | - |
| NCT00953667 | Healthy Volunteers | Not Applicable | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00953667 | - | - | Completed | - | - |
| NCT02003638 | Coronary Artery Disease ... more >> Carotid Artery Disease Peripheral Artery Disease Collapse << | Not Applicable | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01683656 | HIV Atheroscl... more >>erosis Collapse << | Phase 4 | Terminated(Withdrawal of IMP f... more >>rom the market. Data on risk-benefit ratio pending.) Collapse << | July 2014 | Switzerland ... more >> University Hospital Berne Inselspital Berne, BE, Switzerland, 3010 University Hospital Basel Basel, BS, Switzerland, 4031 University Hospitals Genève Geneva, GE, Switzerland, 1211 Kantonsspital St Gallen St Gallen, SG, Switzerland, 9007 EOC Ente Ospedaliero Cantonale, civico Lugano, TI, Switzerland, 6903 CHUV Cantonal University Hospital Vaud Lausanne, VD, Switzerland, 1011 University Hospital Zurich Zurich, ZH, Switzerland Collapse << |
| NCT01984073 | Dyslipidemia | Phase 1 | Recruiting | October 2018 | United States, Pennsylvania ... more >> Hospital of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Laura J Pollan, MPH 215-615-4740 pollan@mail.med.upenn.edu Principal Investigator: Richard L Dunbar, MD Sub-Investigator: Daniel J Rader, MD Presbyterian Hospital Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Laura J Pollan, MPH 215-615-4740 pollan@mail.med.upenn.edu Principal Investigator: Richard L Dunbar, MD Sub-Investigator: Daniel J Rader, MD Collapse << |
| NCT02003638 | - | - | Completed | - | - |
| NCT02322203 | Cardiovascular Disease | Phase 2 | Recruiting | July 3, 2019 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact Office of Patient Recruitment (OPR) 800-411-1222 ext TTY8664111010 prpl@cc.nih.gov Collapse << |
| NCT00930839 | - | - | Completed | - | United States, Virginia ... more >> Eastern Virgnia Medical School, Strelitz Diabetes Center Norfolk, Virginia, United States, 23510f Collapse << |
| NCT03136705 | Healthy | Phase 1 | Completed | - | United States, Illinois ... more >> Center for Translational Metabolism and Health (CTMH), Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00194402 | Dyslipidemia | Phase 4 | Completed | - | United States, Washington ... more >> Northwest Lipid Research Clinic, University of Washington Seattle, Washington, United States, 98104 Collapse << |
| NCT03642990 | Chemotherapy-induced Periphera... more >>l Neuropathy Breast Cancer Metastatic Collapse << | Phase 2 | Not yet recruiting | April 2022 | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Not yet recruiting Iowa City, Iowa, United States, 52242 Contact: Donna L Hammond, PhD 319-335-9595 donna-hammond@uiowa.edu Contact: Marian K Anderson, MA 319-353-4578 marian-andersen@uiowa.edu United States, North Carolina Wake Forest University Not yet recruiting Salem, North Carolina, United States, 27109 Contact: Alexandra Thomas, MD 336-716-7217 althomas@wakehealth.edu Collapse << |
| NCT00664287 | Dyslipidemia | Phase 3 | Withdrawn | - | - |
| NCT02812238 | Atherosclerosis ... more >> Diabetes Coronary Artery Disease Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center Bethesda, Maryland, United States, 20892 Collapse << |
| NCT03754842 | Muscle Injury | Not Applicable | Not yet recruiting | July 1, 2020 | - |
| NCT03743636 | Peripheral Artery Disease | Phase 3 | Recruiting | April 2022 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu Contact: Kathryn Domanchuk, BS 312-503-6438 k-domanchuk@northwestern.edu Principal Investigator: Mary McDermott, MD Collapse << |
| NCT01200784 | Chronic Kidney Disease ... more >> Hemodialysis Hyperphosphatemia Collapse << | Phase 2 | Completed | - | Germany ... more >> Alsfeld, Germany, 36304 Arnstadt, Germany, 99310 Augsburg, Germany, 86157 Berlin Hellersdorf, Germany, 12627 Berlin Kreuzberg, Germany, 10245 Berlin, Germany, 12045 Berlin, Germany, 12203 Berlin, Germany, 12435 Berlin, Germany, 13051 Bielefeld, Germany, 33602 Coburg, Germany, 96450 Darmstadt, Germany, 64295 Dortmund, Germany, 44135 Dülmen, Germany, 48249 Düsseldorf, Germany, 40210 Elsenfeld, Germany, 63820 Erfurt, Germany, 99089 Essen, Germany, 45127 Friedrichsroda, Germany, 99894 Hamburg, Germany, 22297 Hameln, Germany, 31787 Herne, Germany, 44623 Herzberg, Germany, 04916 Iserlohn, Germany, 58638 Jena-Drakendorf, Germany, 07751 Kamen, Germany, 59174 Leverkusen, Germany, 51373 Mannheim, Germany, 68309 Minden, Germany, 32429 Nordhausen, Germany, 99734 Osnabrueck, Germany, 49074 Pfarrkirchen, Germany, 84347 Regensburg, Germany, 93053 Schwabach, Germany, 91126 Tangermünde, Germany, 39590 Wuppertal, Germany, 42283 Collapse << |
| NCT01126073 | Coronary Heart Disease | Phase 4 | Completed | - | Slovenia ... more >> UMC Ljubljana Ljubljana, Slovenia, 1000 Collapse << |
| NCT00580931 | Alzheimer's Disease | Phase 1 Phase 2 | Completed | - | United States, California ... more >> UC Irvine School of Medicine Irvine, California, United States, 92697 Collapse << |
| NCT02258074 | Chronic Kidney Disease | Phase 3 | Active, not recruiting | June 2018 | United States, California ... more >> University of California at San Diego San Diego, California, United States, 92161 United States, Colorado Denver Nephrology Research Denver, Colorado, United States, 80230 United States, District of Columbia George Washington University Washington, District of Columbia, United States, 20037 United States, Illinois NorthShore University Health System Chicago, Illinois, United States, 60201 Northwestern University Chicago, Illinois, United States, 60611 United States, Utah University of Utah Salt Lake City, Utah, United States, 84112 Utah VA Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT03579693 | Chronic Kidney Disease ... more >> Sarcopenia Frailty Collapse << | Phase 2 | Recruiting | April 2020 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98104 Contact: Kidney Research Institute 206-616-8574 info@kri.washington.edu Collapse << |
| NCT01275300 | Healthy Volunteer | Phase 4 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Hospital Phila., Pennsylvania, United States, 19104 Collapse << |
| NCT02300740 | Healthy Participants | Early Phase 1 | Completed | - | Denmark ... more >> Medical Research Laboratories, Aarhus University Hospital Aarhus, Denmark, 8000 Collapse << |
| NCT01250990 | Dyslipidemias | Not Applicable | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT02140814 | Polycystic Kidney Disease | Phase 2 | Completed | - | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Collapse << |
| NCT01763424 | Psoriasis | Phase 2 Phase 3 | Completed | - | Iran, Islamic Republic of ... more >> Skin Diseases and Leishmaniasis Research Center Isfahan, Iran, Islamic Republic of Collapse << |
| NCT01250990 | - | - | Completed | - | - |
| NCT01321034 | Hypercholesterolemia | Phase 4 | Completed | - | Spain ... more >> Hospital San Jorge Huesca, Spain Hospital Universitario Miguel Servet Zaragoza, Spain, 50009 Hospital Royo Villanova Zaragoza, Spain Collapse << |
| NCT02836184 | Hyperphosphatemia | Phase 4 | Unknown | September 2016 | China, Jiangxi ... more >> Jiujiang NO.1 People's Hospital Recruiting Jiujiang, Jiangxi, China, 332000 Collapse << |
| NCT03562468 | Cognitive Function ... more >> Mood Sleep Collapse << | Not Applicable | Active, not recruiting | May 2019 | United States, Florida ... more >> MB Clinical Research Boca Raton, Florida, United States, 33487 Collapse << |
| NCT02835664 | Obesity Insul... more >>in Resistance Collapse << | Not Applicable | Recruiting | December 2018 | Netherlands ... more >> Maastricht University Medical Centre Recruiting Maastricht, Netherlands Contact: Carlijn Remie, Msc Contact: Patrick Schrauwen, Prof.Dr. Collapse << |
| NCT02950441 | Aging | Phase 2 | Recruiting | September 2019 | United Kingdom ... more >> University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, West Midlands, United Kingdom, B15 2TH Contact: Yasir Elhassan, MRCP +441214158705 y.mohamedelhassan@bham.ac.uk Collapse << |
| NCT03462680 | Parkinson's Disease | Not Applicable | Recruiting | September 30, 2019 | United States, Georgia ... more >> Charlie Norwood VA Medical Center, Augusta, GA Recruiting Augusta, Georgia, United States, 30904 Contact: Thomas J Hartney, MD 706-733-0188 ext 2510 Tom.Hartney@va.gov Contact: James P Hill (706) 733-0188 ext 2508 James.Hill5@va.gov Principal Investigator: Chandramohan Wakade, MBBS Collapse << |
| NCT01589809 | Neurodegenerative Disorders | Phase 2 | Active, not recruiting | December 2017 | United Kingdom ... more >> NIHR/Wellcome Trust Imperial CRF London, Hammersmith, United Kingdom, W12 0HS National Hospital for Neurology and Neurosurgery London, United Kingdom, WC1N 3BG Collapse << |
| NCT02558595 | Polycystic Kidney Disease | Phase 2 | Active, not recruiting | December 2018 | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Collapse << |
| NCT02921659 | Aging | Phase 1 Phase 2 | Completed | - | United States, Colorado ... more >> Clinical Translational Research Center Boulder, Colorado, United States, 80309 Collapse << |
| NCT03151707 | Healthy | Phase 4 | Recruiting | July 2019 | United States, Massachusetts ... more >> Cagri Yuksel Recruiting Belmont, Massachusetts, United States, 02478 Contact: Cagri Yuksel, MD 617-855-2779 ayuksel@partners.org Sub-Investigator: Cagri Yuksel, MD Principal Investigator: Dost Ongur, MD, PhD Collapse << |
| NCT03568968 | Parkinson Disease | Not Applicable | Not yet recruiting | October 1, 2021 | - |
| NCT01942291 | Hypoalphalipoproteinemia | Phase 4 | Completed | - | Brazil ... more >> University of Campinas Campinas, SP, Brazil, 13083-887 Collapse << |
| NCT02018965 | HIV | Phase 2 | Completed | - | Canada, Quebec ... more >> Montreal Chest Institute Montreal, Quebec, Canada, H2W1T7 Collapse << |
| NCT03761511 | Friedreich Ataxia | Phase 2 | Not yet recruiting | December 2020 | Germany ... more >> University Hospital RWTH Aachen Not yet recruiting Aachen, Germany, 52074 Contact: Jörg B. Schulz, Univ.-Prof. jschulz@ukaachen.de Collapse << |
| NCT01011699 | Chronic Renal Failure ... more >> Hemodialysis Collapse << | Phase 3 | Terminated(Financial problem) | - | France ... more >> Centre Hospitalier Général Soissons, Aisne, France, 02009 Centre Hospitalier Lisieux, Calvados, France, 14100 ALURAD Limoges, Limousin, France, 87042 Centre Hospitalier Universitaire Reims, Marne, France, 51092 Association Régionale Promotion Dialyse à domicile (ARPDD) Reims, Marne, France Association pour le Développement de l'Hémodialyse Hénin-Beaumont, Nord-Pas de Calais, France, 62110 Polyclinique de la Louvière Lille, Nord, France, 59000 CHRU Lille, Nord, France, 59037 Hôpital Victor Provo Roubaix, Nord, France, 59056 Centre Hospitalier Général Valenciennes, Nord, France, 59322 Centre Hospitalier Général Beauvais, Oise, France, 60000 Clinique Saint Côme Compiegne, Oise, France, 60200 Centre Hospitalier Général Creil, Oise, France, 60100 Clinique du Bois Bernard Bois Bernard, Pas de calais, France, 62320 Centre Hospitalier Boulogne sur mer, Pas de calais, France, 62200 Centre Hospital-Universitaire d'Amiens Amiens, Picardie, France, 80054 Clinique de l'Europe Rouen, Seine maritime, France, 76040 Centre Hospitalier Cambrai, France, 59407 Collapse << |
| NCT03769285 | Non-melanoma Skin Cancer ... more >> Carcinoma, Squamous Cell Carcinoma, Basal Cell Collapse << | Phase 2 | Recruiting | December 2020 | Canada, Ontario ... more >> Toronto General Hospital, University Health Network Recruiting Toronto, Ontario, Canada, M5G 2N2 Contact: Ashley Lau, BMRSc 416-351-3732 ext 2706 ashley.lau@wchospital.ca Collapse << |
| NCT03061474 | Alzheimer's Disease ... more >> Mild Cognitive Impairment Collapse << | Phase 2 | Recruiting | February 28, 2019 | United States, California ... more >> University of California, Irvine Recruiting Irvine, California, United States, 92697 Contact: Beatriz Yanez, R.N 949-824-0008 byanez@uci.edu Contact: Huong Nguyen 949-824-0008 huongtn2@uci.edu Principal Investigator: Aimee Pierce, M.D. University of California, Los Angeles Recruiting Los Angeles, California, United States, 90095 Contact: Celine Ossinalde, MA 310-794-6191 cossinalde@mednet.ucla.edu Principal Investigator: Sarah Kremen, M.D. Collapse << |
| NCT03727646 | Heart Failure,Congestive ... more >> Heart Failure New York Heart Association Class IV Mitochondrial Alteration Collapse << | Early Phase 1 | Recruiting | July 26, 2019 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Kevin D O'Brien, MD 206-529-7802 cardiac@uw.edu Contact: Rong Tian, MD 206 616-5672 rongtian@u.washington.edu Collapse << |
| NCT02213094 | Pregnancy Induced Hypertension... more >> Superimposed Preeclampsia Hypertension Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> University of North Carolina Women's Hospital Chapel Hill, North Carolina, United States, 27599-7516 Collapse << |
| NCT03501433 | Aging Lipemia | Not Applicable | Recruiting | December 2018 | United States, Iowa ... more >> Iowa State University Recruiting Ames, Iowa, United States, 50011 Contact: Rudy Valentine, Ph.D. 515-294-3867 rvalenti@iastate.edu Principal Investigator: Rudy Valentine, Ph.D. Collapse << |
| NCT02416739 | Non-Small-Cell Lung Carcinoma | Phase 2 Phase 3 | Active, not recruiting | June 2020 | Korea, Republic of ... more >> Chonnam National University Hwasun Hospital Hwasun, Chonnam, Korea, Republic of, 58128 Collapse << |
| NCT03423342 | Heart Failure, Systolic | Phase 1 Phase 2 | Recruiting | June 30, 2019 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Kevin D O'Brien, MD 206-529-7802 cardiac@uw.edu Contact: Rong Tian, MD 206 616-5672 rongtian@u.washington.edu Collapse << |
| NCT02689882 | Metabolic Disturbance | Phase 1 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT03432871 | Mitochondrial Diseases ... more >> Mitochondrial Myopathies Progressive External Ophthalmoplegia Progressive Ophthalmoplegia Progressive; Ophthalmoplegia, External Mitochondria DNA Deletion MELAS Mitochondrial Encephalomyopathy, Lactic Acidosis, and Stroke-Like Episodes Mitochondrial Encephalopathy, Lactic Acidosis and Stroke-Like Episodes (MELAS Syndrome) Collapse << | Not Applicable | Recruiting | May 30, 2019 | United Kingdom ... more >> Cambridge University Hospitals NHS Foundation Trust Recruiting Cambridge, United Kingdom, CB20QQ Contact: Zoe McIntyre, MSc 0044 1223 331506 mitopatients@mrc-mbu.cam.ac.uk Contact: Sarah Bird, MSc 0044 1223 331506 mitopatients@mrc-mbu.cam.ac.uk Principal Investigator: Patrick F Chinnery, MD PhD Collapse << |
| NCT02213094 | - | - | Completed | - | - |
| NCT03419364 | Preeclampsia | Phase 2 | Recruiting | December 31, 2019 | United States, North Carolina ... more >> UNC at Chapel Hill Recruiting Chapel Hill, North Carolina, United States, 27599 Contact: Karen Dorman, RN 984-974-9012 kdorman@med.unc.edu Principal Investigator: Kim Boggess, MD Collapse << |
| NCT03260166 | Cutaneous Lupus Erythematosus ... more >> Systemic Lupus Erythematosus Rash Collapse << | Phase 2 | Recruiting | February 2019 | China, Hunan ... more >> The Second Xiangya Hospital of Central South University Recruiting Changsha, Hunan, China, 410011 Contact: Xiangqi Tang, MD, PhD +86-731-84896038 xiangyagcp@126.com Principal Investigator: Qianjin Lu, MD, PhD Sub-Investigator: Hai Long, MD, PhD Collapse << |
| NCT02532816 | Stunting | Not Applicable | Unknown | July 2016 | Indonesia ... more >> in the community (Woja,Manggelewa, Kempo subdistrict) Dompu, West Nusa Tenggara, Indonesia Collapse << |
| NCT02961829 | Chronic Infection ... more >> HIV Collapse << | Not Applicable | Active, not recruiting | March 2020 | Brazil ... more >> CCDI Sao Paulo, SP, Brazil, 04040002 Collapse << |
| NCT03423719 | Overweight and Obesity | Not Applicable | Completed | - | Spain ... more >> UCAM (Universidad Catolica San Antonio de Murcia) Murcia, Spain Collapse << |
| NCT03739242 | Hypercholesterolemia | Not Applicable | Completed | - | Italy ... more >> Policlinico S.Orsola - Malpighi Medicina Interna Borghi Bologna, Italy, 40138 Collapse << |
| NCT01665378 | Anemia Intrau... more >>terine Growth Retardation Preterm Delivery Iron Deficiency Collapse << | Phase 3 | Active, not recruiting | September 2019 | Vietnam ... more >> Thainguyen University of Medicine and Pharmacy Thai Nguyen, Vietnam Collapse << |
| NCT01488656 | Inflammation | Not Applicable | Completed | - | - |
| NCT01643187 | Malnutrition | Phase 2 | Unknown | December 2013 | Guatemala ... more >> APEVIHS Not yet recruiting Retalhuleu, Guatemala Contact: Victor Alfonso +502 42196935 valfonso@apevihs.org Principal Investigator: Victor Alfonso Collapse << |
| NCT02999581 | Dietary Modification | Not Applicable | Completed | - | - |
| NCT00642408 | Multiple Micronutrient Deficie... more >>ncies During Pregnancy Collapse << | Phase 4 | Completed | - | Burkina Faso ... more >> Centre Muraz, 2054, Avenue Mamadou KONATE Bobo-Dioulasso, Burkina Faso, BP 390 Collapse << |
| NCT03325491 | Impaired Mitochondrial Functio... more >>n, Muscle Performance Collapse << | Not Applicable | Recruiting | December 31, 2018 | United Kingdom ... more >> NIHR Exeter Clinical Research Facility Recruiting Exeter, Devon, United Kingdom, EX2 5DW Contact: Colleen Deane, PhD 01392722882 c.s.deane@exeter.ac.uk Collapse << |
| NCT00197730 | HIV Infections ... more >> Pregnancy Complications Collapse << | Phase 3 | Completed | - | Tanzania ... more >> Muhimbili University of Health and Allied Sciences Dar es Salaam, Tanzania Collapse << |
| NCT02280330 | Iodine Deficiency ... more >> Iodine Deficiency Disorder Hypothyroidism Collapse << | Phase 4 | Completed | - | Philippines ... more >> La Trinidad Benguet Day Care Centers La Trinidad, Benguet, Philippines, 2601 Collapse << |
| NCT02792621 | Impaired Mitochondrial Functio... more >>n, Muscle Performance Collapse << | Not Applicable | Recruiting | December 8, 2018 | United Kingdom ... more >> NIHR Exeter Clinical Research Facility Recruiting Exeter, Devon, United Kingdom, EX2 5DW Contact: Anna Steele, PhD 01392 408173 anna.steele@nhs.net Contact: Kim Rownden 01392 408188 kim.rowden@nhs.net Collapse << |
| NCT02952014 | Dietary Modification | Not Applicable | Completed | - | United States, Texas ... more >> Exercise & Sport Nutrition Laboratory College Station, Texas, United States, 77843 Collapse << |
| NCT01111864 | Bacterial Infections | Not Applicable | Completed | - | Kenya ... more >> Kikoneni Clinic Kikoneni, Kwale district, Kenya Collapse << |
| NCT03234452 | Disbiosis Abd... more >>ominal Pain Constipation Diarrhea Stress Collapse << | Not Applicable | Completed | - | Italy ... more >> Centro Ricerche Cliniche di Verona Srl, Verona, (Vr), Italy, 37100 Collapse << |
| NCT00145184 | Tuberculosis | Phase 3 | Terminated(Slow recruitment) | - | Tanzania ... more >> Muhimbili University, College of Health Sciences Dar Es Salaam, Tanzania Collapse << |
| NCT03337360 | Male Subfertility | Not Applicable | Recruiting | April 1, 2021 | Netherlands ... more >> Canisius-Wilhelmina Hospital Not yet recruiting Nijmegen, Gelderland, Netherlands Contact: Cathelijne van Heteren, MD, PhD c.v.heteren@cwz.nl Principal Investigator: Cathelijne van Heteren, MD, PhD Jeroen Bosch Ziekenhuis Not yet recruiting 's-Hertogenbosch, Netherlands, 5200ME Contact: Jan Peter de Bruin, MD PhD j.d.bruin@jbz.nl Principal Investigator: Jan Peter de Bruin, MD PhD Gelre Ziekenhuizen Not yet recruiting Apeldoorn, Netherlands, 7334DZ Contact: Maaike Traas, MD PhD m.traas@gelre.nl Principal Investigator: Maaike Traas, MD PhD Rijnstate Recruiting Arnhem, Netherlands, 6815AD Contact: Annemiek Nap, MD PhD ANap@rijnstate.nl Principal Investigator: Annemiek Nap, MD PhD Maasziekenhuis Pantein Not yet recruiting Boxmeer, Netherlands, 5835DV Contact: Esther Haagen, MD e.haagen@pantein.nl Principal Investigator: Esther Haagen, MD Slingeland Ziekenhuis Recruiting Doetinchem, Netherlands, 7002BL Contact: Rogier Donker, MD PhD R.Donker@slingeland.nl Principal Investigator: Rogier Donker, MD PhD Nij Geertgen Not yet recruiting Elsendorp, Netherlands, 5424SM Contact: Marieke Schoonenberg, MD marieke.schoonenberg@nijgeertgen.nl Contact: Martine Nijs, MSc PhD Martine.Nijs@nijgeertgen.nl Principal Investigator: Marieke Schoonenberg, MD Medisch Centrum Kinderwens Not yet recruiting Leiderdorp, Netherlands Contact: Tessa Cox, MD t.cox@mckinderwens.nl Maastricht UMC+ Not yet recruiting Maastricht, Netherlands, 6229HX Contact: Ron van Golde, MD PhD ron.van.golde@mumc.nl Principal Investigator: Ron van Golde, MD PhD Radboudumc Recruiting Nijmegen, Netherlands, 6500HB Contact: Roos Smits, MSc roos.smits@radboudumc.nl Contact: Kathrin Fleischer, Dr kathrin.fleischer@radboudumc.nl Sub-Investigator: Roos Smits, MSc Principal Investigator: Kathrin Fleischer, Dr Elisabeth-TweeSteden Ziekenhuis Not yet recruiting Tilburg, Netherlands, 5022GC Contact: Jesper Smeenk, MD PhD j.smeenk@etz.nl Principal Investigator: Jesper Smeenk, MD PhD Bernhoven Ziekenhuis Not yet recruiting Uden, Netherlands, 5400AS Contact: Marcel Hoekstra, MD PhD m.hoekstra@bernhoven.nl Principal Investigator: Marcel Hoekstra, MD PhD Máxima Medisch Centrum Not yet recruiting Veldhoven, Netherlands, 5504DB Contact: Jacques Maas, MD PhD Jacques.Maas@mmc.nl Principal Investigator: Jacques Maas, MD PhD Nij Barrahus Not yet recruiting Wolvega, Netherlands Contact: Harold Mous, MD harold.mous@nijbarrahus.nl Collapse << |
| NCT02118402 | Anemia Iron D... more >>eficiency Diarrhea Malaria Respiratory Tract Infections (RTI) Antibiotics Gut Inflammation Prebiotics Collapse << | Not Applicable | Completed | - | Kenya ... more >> Kikoneni Health Center Kikoneni, Kwale County, Kenya Collapse << |
| NCT00170404 | Mycobacterium Tuberculosis | Phase 3 | Completed | - | Tanzania ... more >> Muhimbili University, College of Health Sciences Dar Es Salaam, Tanzania Collapse << |
| NCT01427413 | - | - | Recruiting | December 2018 | Italy ... more >> Cervesi Hospital Recruiting Cattolica, Rimini, Italy, 47841 Contact: Silvia De Stefani, Biotecnology +39 320 1111937 silvia.destefani83@libero.it Contact: Simone Palini, biology +39 339 4572101 simonepalini@yahoo.it Collapse << |
| NCT01088958 | Anemia, Iron-deficiency | Phase 4 | Completed | - | Kenya ... more >> CDC/Kenya Medical Research Institute Kisumu, Kenya Collapse << |
| NCT00728273 | Healthy | Phase 2 | Completed | - | Thailand ... more >> Institute of Nutrition, Mahidol University Nakhon Pathom, Thailand, 73170 Collapse << |
| NCT01538316 | Primary Prevention of Prostate... more >> Cancer Collapse << | Not Applicable | Unknown | April 2014 | Germany ... more >> Institute of Nutritional Medicine, University of Hohenheim Recruiting Stuttgart, Germany, 70599 Contact: Antje Damms Machado, dipl. troph. antje.machado@uni-hohenheim.de Principal Investigator: Stephan C Bischoff, MD, Prof. Department of Urology, University Hospital Tübingen Recruiting Tübingen, Germany, 72076 Contact: Stefan Aufderklamm, MD Principal Investigator: Arnulf Stenzl, MD, Prof. Collapse << |
| NCT03032549 | Dietary Supplements | Not Applicable | Completed | - | - |
| NCT00311298 | Tuberculosis ... more >>HIV Diabetes Collapse << | Phase 3 | Completed | - | Tanzania ... more >> Mwanza Medical Centre, NIMR Mwanza, Tanzania Collapse << |
| NCT02620345 | Fibroids | Phase 4 | Completed | - | - |
| NCT02532218 | Hyperlipidemia | Phase 2 | Unknown | December 2016 | United States, California ... more >> Catalina Reserch Institute, LLC Chino, California, United States, 91710 United States, Florida S&W Clinical Reserch Ft Lauderdale, Florida, United States, 33306 Jacksonville Center for Clinical Research Jacksonvile, Florida, United States, 32216 Progressive Medical Research Port Orange, Florida, United States, 32127 United States, Georgia Sestron Clinical Research 833 Campbell Hill Street Suite 230 Marietta, Georgia, United States, 30060 United States, Indiana Midwest Institute for Clinical Research Indianapolis, Indiana, United States, 46260 United States, Kentucky Louisville Metabolic and Atherosclerosis Research Center Louisville, Kentucky, United States, 40213 United States, New York Rochester clinical Research,Inc Rochester, New York, United States, 14069 United States, Ohio Sterling Research Group Ltd Cincinnati, Ohio, United States, 45219 The Carl and Edyth Lindner Center for Reserch and education at the Christ Hospital Cincinnati, Ohio, United States, 45219 Metabolic and atherosclerosis Research center Cincinnati, Ohio, United States, 45227 IVA reserch Cincinnati, Ohio, United States, 45245 Sterling Research group, Ltd Cincinnati, Ohio, United States, 45246 Ohio Clinical Research-Lyndhurst Lyndhurst, Ohio, United States, 44124 United States, Oklahoma COR Clinical Research Oklahoma City, Oklahoma, United States, 73103 United States, Oregon Willamette Valley Clinical studies Eugene, Oregon, United States, 97404 United States, Virginia Health Research of Hampton Roads - Norfolk Norfolk, Virginia, United States, 23502 National Clinical Research inc Richmond, Virginia, United States, 23294 United States, Washington Raninier Clinical Reserach Renton, Washington, United States, 98057 Collapse << |
| NCT02666417 | Anemia, Iron-Deficiency | Not Applicable | Completed | - | Kenya ... more >> Msambweni District Hopsital Msambweni, Kwale County, Kenya Collapse << |
| NCT02422368 | Thyroid Eye Disease | Phase 2 Phase 3 | Not yet recruiting | June 2022 | Iran, Islamic Republic of ... more >> Private Thyroid eye disease clinic Not yet recruiting Tehran, Iran, Islamic Republic of, 14455 Rassoul Akram Hospital Recruiting Tehran, Iran, Islamic Republic of Contact: Mohsen B Kashkouli, MD Collapse << |
| NCT02464111 | Malnutrition | Not Applicable | Completed | - | United States, California ... more >> USDA, ARS, Western Human Nutrition Research Center Davis, California, United States, 95616 Collapse << |
| NCT03630991 | Acute Myeloid Leukemia ... more >> Acute Myeloid Leukemia Arising From Previous Myelodysplastic Syndrome Chronic Myelogenous Leukemia, BCR-ABL1 Positive Chronic Myelomonocytic Leukemia Myelodysplastic Syndrome Myelodysplastic/Myeloproliferative Neoplasm Secondary Acute Myeloid Leukemia Collapse << | Phase 2 | Recruiting | December 31, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact mohanian@mdanderson.org Collapse << |
| NCT00628459 | Anemia Undern... more >>utrition Collapse << | Not Applicable | Completed | - | Burkina Faso ... more >> District Sanitaire de Kongoussi Kongoussi, Burkina Faso Collapse << |
| NCT03442283 | - | - | Active, not recruiting | June 2018 | - |
| NCT00302744 | Anxiety | Phase 1 | Completed | - | United States, California ... more >> Harbor-UCLA Medical Center Torrance, California, United States, 90509 Collapse << |
| NCT01152073 | Hyperlipidemia | Not Applicable | Completed | - | United States, Florida ... more >> Mitchell Karl, M.D. -- Cardiology Boca Raton, Florida, United States, 33486 Collapse << |
| NCT01412580 | - | - | Completed | - | Tanzania ... more >> MUHAS Dar es Salaam, Tanzania Collapse << |
| NCT00623857 | Malaria | Phase 3 | Completed | - | Tanzania ... more >> Kilimanjaro Christian Medical Centre Moshi, Tanzania Collapse << |
| NCT01139398 | Delivery of Health Care | Not Applicable | Completed | - | France ... more >> Biofortis Nantes, France, 44200 Collapse << |
| NCT00064753 | Chronic Kidney Disease ... more >> Cardiovascular Disease Death Collapse << | Phase 2 Phase 3 | Completed | - | - |
| NCT00383669 | HIV Infections | Phase 3 | Completed | - | Tanzania ... more >> Muhimbili University College of Health Sciences Dar es Salaam, Tanzania Collapse << |
| NCT03360435 | - | - | Recruiting | November 18, 2019 | United States, Florida ... more >> University of Florida Recruiting Gainesville, Florida, United States, 32611 Collapse << |
| NCT00197678 | HIV Infections | Phase 3 | Completed | - | United States, Massachusetts ... more >> Harvard School of Public Health Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00479882 | Primary Hypercholesterolemia ... more >> Mixed Dyslipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00361751 | Burns Insulin... more >> Resistance Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Shriners Hospital for CHildren Galveston, Texas, United States, 77550 Collapse << |
| NCT02521116 | Healthy Subjects ... more >> Diabetic Retinopathy Retinal Vein Occlusion Collapse << | Not Applicable | Completed | - | Austria ... more >> Medical University of Vienna Vienna, Austria, 1090 Collapse << |
| NCT00064753 | - | - | Completed | - | - |
| NCT00202228 | HIV Infections ... more >> AIDS Lactic Acidosis Lipodystrophy Collapse << | Phase 4 | Completed | - | Canada, Ontario ... more >> Queen's University Kingston, Ontario, Canada, K7L 3N6 Collapse << |
| NCT00687076 | Peripheral Arterial Disease | Phase 4 | Completed | - | United States, Texas ... more >> Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT02976818 | - | - | Recruiting | April 2019 | Canada ... more >> Institute of Nutrition and Functional Foods (INAF) Recruiting Quebec, Canada, G1V 0A6 Contact: Patrick Couture, MD, FRCP,PhD 418-654-2106 patrick.couture@crchul.ulaval.ca Contact: André Tremblay, PhD 418-656-2131 ext 11417 andre.tremblay@fsaa.ulaval.ca Principal Investigator: Patrick Couture, MD,FRCP,PhD Sub-Investigator: André Tremblay, PhD Collapse << |
| NCT00479882 | - | - | Completed | - | - |
| NCT03707925 | Lung Carcinoid Tumor ... more >> Non-Small Cell Lung Carcinoma Stage IV Lung Cancer AJCC v8 Stage IVA Lung Cancer AJCC v8 Stage IVB Lung Cancer AJCC v8 Collapse << | Not Applicable | Recruiting | October 31, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Roberto F. Casal 713-792-6238 Principal Investigator: Roberto F. Casal Collapse << |
| NCT02592148 | - | - | Completed | - | Germany ... more >> BioTeSys Esslingen, Germany, 73728 Collapse << |
| NCT02782208 | Hypopituitarism ... more >> Insulin Resistance Endocrine System Diseases Glucose Metabolism Disorders Metabolic Diseases Pituitary Diseases Brain Diseases Collapse << | Not Applicable | Unknown | November 2018 | Denmark ... more >> University Hospital of Aarhus Recruiting Aarhus, Denmark, 8000 Contact: Astrid J Hjelholt, MD +4524800664 ajh@clin.au.dk Collapse << |
| NCT01183572 | Anemia | Not Applicable | Completed | - | Tanzania ... more >> Ifakara Health Institute Rufiji, Tanzania Collapse << |
| NCT00755950 | Acute Hepatitis A ... more >> Acute Hepatitis B Acute Hepatitis C Acute Hepatitis E Acute EBV Hepatitis Acute CMV Hepatitis Collapse << | Phase 2 Phase 3 | Terminated(Low enrollment) | - | Egypt ... more >> Alexandria University Hospital Alexandria, Alexandria Governorate, Egypt Tanta Fever Hospital Tanta, Gharbeya Governorate, Egypt Banha Fever Hospital Benha, Kaluobeya Governorate, Egypt Collapse << |
| NCT01064934 | Hyperlipoproteinemia(a) ... more >> Progressive Cardiovascular Disease Collapse << | Not Applicable | Withdrawn | August 2015 | Germany ... more >> Lipidambulanz, Interdisziplinäres Stoffwechsel-Centrum, CVK, Charite Berlin, Germany, 13353 Collapse << |
| NCT00687076 | - | - | Completed | - | - |
| NCT03489200 | Amyotrophic Lateral Sclerosis | Not Applicable | Completed | - | Spain ... more >> Universidad Católica de Valencia San Vicente Màrtir Valencia, Spain Collapse << |
| NCT01515241 | Heterozygous Familial Hypercho... more >>lesterolemia Collapse << | Phase 2 | Completed | - | Canada, Quebec ... more >> Montreal Heart Institute Montreal, Quebec, Canada, H1T1C8 Collapse << |
| NCT00421668 | Diarrheal Illnesses ... more >> Respiratory Illness Growth Faltering Collapse << | Phase 3 | Completed | - | Tanzania ... more >> Muhimbili Uinverstiy College of Health Sciences Dar es Salaam, Tanzania Collapse << |
| NCT03288623 | Oxidative Stress ... more >> Athletes Heart Physical Activity Collapse << | Not Applicable | Recruiting | December 2018 | Italy ... more >> Sapienza University of Rome, Policlinico Umberto I Recruiting Rome, Italy, 00161 Contact: Elena Cavarretta, MD, PhD +390649973154 elena.cavarretta@uniroma1.it Principal Investigator: Elena Cavarretta, MD, PhD Principal Investigator: Giacomo Frati, MD Principal Investigator: Roberto Carnevale, PhD Sub-Investigator: Riccardo Del Vescovo, MD, PhD Sub-Investigator: Sebastiano Sciarretta, MD, PhD Sub-Investigator: Giuseppe Biondi-Zoccai, MD Sub-Investigator: Mariangela Peruzzi, MD, PhD Sub-Investigator: Andrea Serdoz, MD Sub-Investigator: Roberto Ferrara, MD Collapse << |
| NCT01260376 | Diabetes Obes... more >>ity Collapse << | Not Applicable | Completed | - | Denmark ... more >> University Hospital of Aarhus, Norrebrogade Aarhus, Denmark, 8000 University Hospital of Aarhus, Norrebrogade Aarhus, Denmark, 8 Collapse << |
| NCT00000553 | Cardiovascular Diseases ... more >> Coronary Disease Heart Diseases Myocardial Ischemia Collapse << | Phase 3 | Completed | - | - |
| NCT02642159 | Dyslipidemia | Phase 4 | Completed | - | - |
| NCT01075373 | - | - | Unknown | November 2010 | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 10002 Collapse << |
| NCT03579615 | Diabetes Mellitus, Type 1 | Not Applicable | Not yet recruiting | October 1, 2019 | - |
| NCT02863185 | Chronic Kidney Disease | Phase 4 | Recruiting | December 2019 | Korea, Republic of ... more >> Won Suk An Recruiting Busan, Korea, Republic of, 602-715 Contact: Won Suk An, M.D., Ph.D. +82-51-240-2811 anws@dau.ac.kr Dong-A University Not yet recruiting Busan, Korea, Republic of, 602715 Contact: Su Mi Lee, MD +822405549 promise131@hanmail.net Collapse << |
| NCT01071278 | - | - | Completed | - | - |
| NCT02642159 | - | - | Completed | - | - |
| NCT00359281 | Healthy | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01071278 | - | - | Completed | - | - |
| NCT00173069 | - | - | Unknown | - | - |
| NCT00359281 | - | - | Completed | - | - |
| NCT03143946 | Neuroendocrine Tumor | Not Applicable | Recruiting | December 2018 | Netherlands ... more >> University Medical Center Groningen Recruiting Groningen, Netherlands, 9713 GZ Contact: A. M.E. Walenkamp, MD, PhD +31 50 3612821 a.walenkamp@umcg.nl Contact: L. D. de Hosson, MD +31 50 3616161`` l.d.de.hosson@umcg.nl Principal Investigator: A. M.E. Walenkamp, MD, PhD Collapse << |
| NCT03444155 | Healthy Thiam... more >>ine and Niacin Deficiency States Pyridoxine Deficiency Folic Acid Deficiency Anemia, Dietary Vitamin B 12 Deficiency Peroxidase; Defect Polyphenols Oxidative Stress Homocystine; Metabolic Disorder Collapse << | Not Applicable | Completed | - | Austria ... more >> Wonisch Willibald Graz, Styria, Austria, 8036 Collapse << |
| NCT01250340 | Type 2 Diabetes Mellitus | Phase 4 | Unknown | December 2012 | Italy ... more >> Sapienza Università di Roma Recruiting Rome, Italy, 00161 Contact: Francesco Violi, Full Prof +39-06-4461933 francesco.violi@uniroma1.it Contact: Stefania Basili, Ass Prof +39-06-49974678 stefania.basili@uniroma1.it Principal Investigator: Francesco Violi, full Prof Collapse << |
| NCT03310034 | Aging Mitocho... more >>ndrial Function Collapse << | Not Applicable | Recruiting | December 2019 | Netherlands ... more >> Maastricht University Recruiting Maastricht, Limburg, Netherlands, 6229ER Contact: Niels Connell, MSc +31433881587 n.connell@maastrichtuniversity.nl Collapse << |
| NCT00197704 | Tuberculosis | Phase 3 | Completed | - | United States, Massachusetts ... more >> Harvard School of Public Health Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01371955 | - | - | Completed | - | France ... more >> University Hospital of Grenoble Grenoble, France, 38043 Collapse << |
| NCT01030406 | Opioid Abuse | Phase 2 | Completed | - | United States, Utah ... more >> Lifetree Clinical Research Salt Lake City, Utah, United States, 84106 Collapse << |
| NCT01030406 | - | - | Completed | - | - |
| NCT02251678 | Plaque Psoriasis | Phase 1 | Completed | - | United States, Illinois ... more >> NMFF Dermatology Clinic Chicago, Illinois, United States, 60611 NM Lake Forest Hospital/ Women's Center Lake Forest, Illinois, United States, 60045 United States, New Jersey TKL Research Fair Lawn, New Jersey, United States, 07410 United States, Ohio University Hospitals Case Medical Center / Dept of Dermatology Cleveland, Ohio, United States, 44106 Collapse << |
| NCT02549924 | Type 2 Diabetes Mellitus | Phase 2 | Terminated(no financial suppor... more >>t) Collapse << | - | Mexico ... more >> Institute of Experimental and Clinical Therapeutics (INTEC), CUCS, University of Guadalajara Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT02518087 | Cardiopulmonary Bypass ... more >> Cardiac Surgery Associated - Acute Kidney Injury Systemic Inflammatory Response Syndrome Acute Kidney Injury Collapse << | Not Applicable | Recruiting | December 2019 | Spain ... more >> Hospital German Trias i Pujol Recruiting Badalona, Barcelona, Spain, 08916 Contact: Ana Campos, MD PhD anacamgom@gmail.com Sub-Investigator: Marc Ayala Gallardo, MD Sub-Investigator: Marius Sánchez Satorra, MD Hospital Universitari de Bellvitge Recruiting L'Hospitalet de Llobregat, Barcelona, Spain, 08907 Contact: Xosé L. Pérez Fernández, MD +34 93 2607521 josep@bellvitgehospital.cat Principal Investigator: Joan Sabater, MD PhD Principal Investigator: Xosé Pérez, MD Sub-Investigator: Enric Boza, MD Sub-Investigator: Albert Gil, MD Sub-Investigator: Albert Miralles, MD Sub-Investigator: Luisa Rivera, MD Sub-Investigator: Fabrizio Sbraga, MD Sub-Investigator: Jacobo Toscano, MD Principal Investigator: Erika Plata-Menchaca, MD Sub-Investigator: Marcos Potocnik, MD Sub-Investigator: Karina Osorio, MD Hospital de la Santa Creu i Sant Pau Recruiting Barcelona, Spain, 08041 Contact: Jorge Ordoñez, MD PhD jordonez@santpau.cat Contact: Antoni Betbesé, MD PhD Collapse << |
| NCT02537392 | Congenital Heart Disease | Not Applicable | Recruiting | June 2019 | China, Shaanxi ... more >> Xi'an Jiaotong University College of Medicine Recruiting Xi'an, Shaanxi, China, 710061 Contact: Hong Yan, Professor 13991802320 yanhonge@mail.xjtu.edu.cn Contact: Jiaomei Yang, Graduate 15829697385 yjm_violet@163.com Collapse << |
| NCT01810822 | - | - | Completed | - | Brazil ... more >> Faculty of Medicine from University of São Paulo São Paulo, SP, Brazil, 01246-000 Collapse << |
| NCT00160095 | - | - | Completed | - | Netherlands ... more >> Free University Medical Centre Amsterdam Amsterdam, Netherlands, po 7057 1007 MB Rijnstate Ziekenhuis Arnhem Arnhem, Netherlands, po 9555 6800 TA Academic Hospital Maastricht Maastricht, Netherlands, po 5800 6202 AZ Radboud University Nijmegen Medical Centre Nijmegen, Netherlands, 6500 HB Universitair Medisch Centrum Utrecht Utrecht, Netherlands, po 85500 3508 GA Collapse << |
| NCT00168688 | Pregnancy Bir... more >>th Weight Collapse << | Phase 1 | Completed | - | Guinea-Bissau ... more >> Bandim Health Project Bissau, Guinea-Bissau, Apartado 861 Collapse << |
| NCT01684059 | - | - | Completed | - | Iraq ... more >> Babylon university/ college of science Hilla, Iraq, IQ Collapse << |
| NCT02758990 | Obesity Rhini... more >>tis Rhinitis, Allergic Respiratory Sounds Dyspnea Headache Migraine Disorders Arthritis Arthralgia Anxiety Sleep Initiation and Maintenance Disorders Sleep Deprivation Collapse << | Not Applicable | Terminated(Recruiting and fina... more >>ncial constraints) Collapse << | - | United States, Texas ... more >> Verifomics, LLC Dallas, Texas, United States, 75219 Collapse << |
| NCT01474486 | Congestive Heart Failure ... more >> Ischemic Cardiomyopathy Collapse << | Not Applicable | Completed | - | United States, Arizona ... more >> Phoenix VA Health Care System Phoenix, Arizona, United States, 85012 Collapse << |
| NCT01708278 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 1 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT01708278 | - | - | Completed | - | - |
| NCT00699413 | Obesity Diabe... more >>tes Mellitus, Type 2 Collapse << | Phase 4 | Terminated(Sponsor decided to ... more >>withdraw from study.) Collapse << | - | United States, California ... more >> USC - Preventive Medicine Los Angeles, California, United States, 90033 Collapse << |
| NCT01709123 | Diabetes Mellitus, Type 1 | Not Applicable | Unknown | September 2013 | United States, Louisiana ... more >> Louisiana State University Health Sciences Center in Shreveport Recruiting Shreveport, Louisiana, United States, 71130 Contact: Sushil K Jain, Ph.D. 318-675-6086 sjain@lsuhsc.edu Sub-Investigator: Robert McVie, M.D. Sub-Investigator: Tommie Stapleton, RN, CCRC Sub-Investigator: Cynthia Brewer, RN Sub-Investigator: John Rowell, RN, MSN Sub-Investigator: Henry R McKnight, RPH Sub-Investigator: Pat F Bass, M.D. Sub-Investigator: Shikha Mane, M.D. Sub-Investigator: David Micinski, BS Sub-Investigator: Neslihan Gungor, M.D. Collapse << |
| NCT00699413 | - | - | Terminated(Sponsor decided to ... more >>withdraw from study.) Collapse << | - | - |
| NCT00903617 | Dyslipidaemias | Phase 2 | Completed | - | United States, Florida ... more >> GSK Investigational Site Pembroke Pines, Florida, United States, 33026 United States, Indiana GSK Investigational Site Indianapolis, Indiana, United States, 46260 United States, Kentucky GSK Investigational Site Louisville, Kentucky, United States, 40213 United States, Maine GSK Investigational Site Auburn, Maine, United States, 04210 United States, Minnesota GSK Investigational Site Brooklyn Center, Minnesota, United States, 55430 United States, North Carolina GSK Investigational Site Statesville, North Carolina, United States, 28677 United States, Ohio GSK Investigational Site Cincinnati, Ohio, United States, 45246 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78205 United States, Virginia GSK Investigational Site Richmond, Virginia, United States, 23294 United States, Washington GSK Investigational Site Olympia, Washington, United States, 98502 GSK Investigational Site Seattle, Washington, United States, 98104 Collapse << |
| NCT01921010 | Cardiovascular Outcomes | Phase 4 | Completed | - | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States, 90048 Collapse << |
| NCT00859911 | Micronutrient Deficiencies | Not Applicable | Unknown | October 2011 | Bangladesh ... more >> Save the Children Dhaka, Dhaka Division, Bangladesh Collapse << |
| NCT00909974 | Intrauterine Growth Retardatio... more >>n Low Birth Weight Collapse << | Phase 4 | Completed | - | Burkina Faso ... more >> Centre Muraz, 2054, Avenue Mamadou KONATE Bobo-Dioulasso, Burkina Faso Collapse << |
| NCT00269217 | Primary Hypercholesterolemia ... more >> Mixed Hyperlipidemia Collapse << | Phase 3 | Completed | - | - |
| NCT00000482 | Cardiovascular Diseases ... more >> Coronary Disease Heart Diseases Myocardial Infarction Myocardial Ischemia Collapse << | Phase 3 | Completed | - | - |
| NCT01085981 | Female Sexual Dysfunction | Not Applicable | Unknown | July 2010 | United States, Pennsylvania ... more >> East Suburban Obgyn Not yet recruiting Monroeville, Pennsylvania, United States, 15668 Contact: michae j pelekanos, md 412-856-7500 mipel@aol.com Contact: vickie lathom 4128567500 vlathom@wpahs.org Collapse << |
| NCT00590629 | Coronary Artery Disease | Phase 4 | Completed | - | United States, California ... more >> CSMC Los Angeles, California, United States, 90048 Collapse << |
| NCT00903617 | - | - | Completed | - | - |
| NCT01295450 | Lack of Appetite ... more >> Anorexia Collapse << | Phase 3 | Unknown | August 2011 | Brazil ... more >> ABC School of Medicine Not yet recruiting Sao Paulo, Brazil, 09060-650 Contact: Adriana P Albuquerque, Coordenator 55 (11) 4438-3558 adrianaalbuquerque@fmabc.br Principal Investigator: Anete S Grumach, Physician Collapse << |
| NCT00000483 | Cardiovascular Diseases ... more >> Coronary Disease Heart Diseases Myocardial Infarction Myocardial Ischemia Collapse << | Not Applicable | Completed | - | - |
| NCT01368887 | Psoriasis | Phase 2 | Suspended(Sponsor seeking addi... more >>tional financial support before starting the study) Collapse << | April 2014 | United States, Massachusetts ... more >> Boston VA Hospital Boston, Massachusetts, United States, 02130 Brockton VA Hospital Brockton, Massachusetts, United States, 02301 Collapse << |
| NCT00893724 | Neovascular Age-related Macula... more >>r Degeneration Diabetic Macular Edema Collapse << | Not Applicable | Unknown | September 2011 | United States, West Virginia ... more >> Mid-Atlantic Retina Consultations, Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT02450149 | Refractory Solid Tumors | Not Applicable | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, seoul, korea, Republic of, Korea, Republic of, 135-710 Collapse << |
| NCT02425605 | Hepatocellular Carcinoma | Phase 2 | Unknown | January 2018 | Korea, Republic of ... more >> Department of Internal Medicine, Yonsei University College of Medicine Recruiting Seoul, Korea, Republic of, 120-752 Contact: BeomKyung Kim, M.D., Ph.D. +82-2-2228-1930 beomkkim@yuhs.ac Contact: Wonseok Kang, M.D., Ph.D. WSKANG79@yuhs.ac Collapse << |
| NCT02460991 | Hepatocellular Carcinoma ... more >> Hepatoma Liver Cell Carcinoma Collapse << | Phase 3 | Terminated(Slow enrollment) | - | United States, Alabama ... more >> University of Alabama Hospital Birmingham, Alabama, United States, 35233 United States, California University of Southern California Los Angeles, California, United States, 90033 United States, Kentucky University of Louisville Louisville, Kentucky, United States, 40202 United States, Maryland University of Maryland Medical Center Baltimore, Maryland, United States, 21201 United States, Oregon Oregon Health & Science University Portland, Oregon, United States, 97239 United States, Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01936363 | Ovarian Cancer | Phase 2 | Completed | - | - |
| NCT02474290 | Acute Myeloid Leukemia ... more >> Hematopoietic Stem Cell Transplantation Collapse << | Phase 2 Phase 3 | Unknown | August 2018 | China, Guangdong ... more >> Department of Hematology,Nanfang Hospital, Southern Medical University Recruiting Guangzhou, Guangdong, China, 510515 Contact: Li Xuan +86-020-61641613 356135708@qq.com Principal Investigator: Qifa Liu Collapse << |
| NCT02460991 | - | - | Terminated(Slow enrollment) | - | - |
| NCT02537158 | Hepatocellular Carcinoma | Not Applicable | Unknown | July 2018 | China ... more >> Cancer Hospital and Institute, Chinese Academy of Medical Sciences Recruiting Bei jing, China, 100026 Contact: Xinyu Bi, Doctor 86-13911792177 beexy1971@163.com Collapse << |
| NCT02530476 | Leukemia Acut... more >>e Myeloid Leukemia Collapse << | Phase 1 Phase 2 | Active, not recruiting | December 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02504983 | Hepatocellular Carcinoma | Phase 4 | Unknown | July 2018 | Taiwan ... more >> Chang Gung Memorial Hospital Recruiting Taoyuan, Taiwan Contact: Chau-Ting Yeh, MD/PhD +886-3-3281200 ext 8125 chautingy@gmail.com Collapse << |
| NCT02538393 | Biological Availability | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany, 13353 Collapse << |
| NCT01336738 | Diabetes, Type 2 | Phase 2 | Completed | - | - |
| NCT03599193 | Acne Vulgaris | Phase 2 | Completed | - | United States, Texas ... more >> Dr. DuBois Austin, Texas, United States, 73301 Dr. Jones Austin, Texas, United States, 73301 Collapse << |
| NCT01336738 | - | - | Completed | - | - |
| NCT01338870 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT02562755 | Hepatocellular Carcinoma (HCC) | Phase 3 | Recruiting | October 2019 | - |
| NCT02575339 | Hepatocellular Carcinoma ... more >> Liver Cancer HCC Collapse << | Phase 1 Phase 2 | Active, not recruiting | June 2020 | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 University of Illinois Cancer Center Chicago, Illinois, United States, 60612 United States, Indiana Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, North Carolina University of Noth Carolina at Chapel Hill Chapel Hill, North Carolina, United States, 27514 United States, Pennsylvania Penn State Cancer Institute Hershey, Pennsylvania, United States, 17033 United States, Wisconsin University of Wisconsin Madison, Wisconsin, United States, 53705 Collapse << |
| NCT02636426 | Neoplasms | Phase 1 | Completed | - | Netherlands ... more >> VU University Medical Center Amsterdam, Noord-Holland, Netherlands, 1081 HV Collapse << |
| NCT01338870 | - | - | Completed | - | - |
| NCT02627963 | Carcinoma, Renal Cell | Phase 3 | Active, not recruiting | December 2019 | - |
| NCT02642913 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Recruiting | December 2019 | United States, California ... more >> University of California San Francisco Recruiting San Francisco, California, United States, 94143 Contact: Robin K Kelley, MD 415-353-9888 United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Benjamin Tan, MD 314-747-7222 Principal Investigator: Benjamin Tan, MD United States, New York Roswell Park Cancer Institute Recruiting Buffalo, New York, United States, 14263-0001 Contact: Iyer Renuka, MD 716-845-2300 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: James Harding, MD 646-888-4314 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: James Harding, MD 646-888-4314 Contact: Ghassan Abou-Alfa, MD 646-888-4184 Collapse << |
| NCT02645981 | HCC | Phase 2 Phase 3 | Active, not recruiting | August 2019 | China, Jiangsu ... more >> The PLA 81 Hospital Nanjing, Jiangsu, China, 025 China, Sichuan West China Hospital Sichuan University Chengdu, Sichuan, China, 610042 Collapse << |
| NCT02599337 | Fasting | Phase 1 | Completed | - | Canada, Quebec ... more >> Inventiv Health Clinique Inc. Québec, Quebec, Canada, G1P 0A2 Collapse << |
| NCT02794337 | Carcinoma, Hepatocellular | Phase 2 Phase 3 | Recruiting | January 2022 | India ... more >> Advanced Centre for Treatment, Research and Education in Cancer, Tata Memorial Centre Recruiting Navi Mumbai, Maharashtra, India, 410210 Contact: Supriya Chopra, MD,DNB 022-27405000 ext 5510 schopra@actrec.gov.in Advanced Centre of Treatment Research and Education In Cancer,Tata Memorial Centre Recruiting Navi Mumbai, Maharashtra, India, 410210 Contact: Dr Supriya Chopra, MD 91-22-27405000 ext 5491 schopra@actrec.gov.in Principal Investigator: Supriya Chopra, MD Collapse << |
| NCT02834546 | - | - | Recruiting | October 2019 | France ... more >> Institut Bergonié Not yet recruiting Bordeaux, France Principal Investigator: Dominique BECHADE, MD CHU de Limoges Not yet recruiting Limoges, France Principal Investigator: Véronique LOUSTAUD-RATTI, MD CHU de Montpellier Not yet recruiting Montpellier, France Principal Investigator: Eric ASSENAT, MD CHU de Bordeaux Recruiting Pessac, France Contact: Clémence FRON, MD 05 57 65 64 39 ext +33 clemence.fron@chu-bordeaux.fr Contact: Nathalie TERRAS 05 57 62 34 60 ext +33 nathalie.terras@chu-bordeaux.fr Principal Investigator: Clémence FRON, MD Sub-Investigator: Jean-Frédéric BLANC, MD-PhD CHU de Toulouse Not yet recruiting Toulouse, France Principal Investigator: Jean-Marie PERON, MD Collapse << |
| NCT02733809 | Hepatocellular Carcinoma | Phase 4 | Recruiting | December 2024 | Saudi Arabia ... more >> King Saud University Medical City Recruiting Riyadh, Saudi Arabia, 7805 Contact: Weam S Husseim +966541480459 wshussain@ksu.edu.sa Principal Investigator: Dr. Mazen M Hassanain, MBBS FRCSC FACS PhD Sub-Investigator: Prof. Ayman A Abdo, MD, FRCPC, FACP Sub-Investigator: Dr. Khalid A Alswat, MD, ABIM, MRCP,FACP Sub-Investigator: Prof. Waleed K Alhamoudi Principal Investigator: Dr. Shouki M Bazarbashi Collapse << |
| NCT02867280 | Hepatocellular Carcinoma ... more >> Recurrence Collapse << | Phase 3 | Recruiting | June 2023 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510060 Contact: Minshan Cchen, M.D. 8620-87343117 chenmsh@sysucc.org.cn Contact: Li Xu, M.D. xuli@sysucc.org.cn Principal Investigator: Minshan Chen, M.D. Collapse << |
| NCT02971696 | HCC | Phase 3 | Completed | - | Egypt ... more >> El Kahraba Hosital Cairo, N/A = Not Applicable, Egypt, 1234 Ain Shams University Hospital Cairo, Egypt Nasser Institute Cairo, Egypt Collapse << |
| NCT02856126 | Hepatocellular Carcinoma | Phase 3 | Recruiting | August 2018 | China, Guangdong ... more >> Dongguan People's Hospital Recruiting Dongguan, Guangdong, China, 523059 Contact: Wusheng Yu, MD 13827285010 yuwusheng1998@126.com Cancer Center Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Ming Shi, MD 8620-87343115 shiming@mail.sysu.edu.cn The First Affiliated Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Guosheng Tan, MD 13725254145 tgs1976@163.com Guangzhou Twelfth People 's Hospital Recruiting Guangzhou, Guangdong, China, 510620 Contact: Yuanmin Zhou, MD 15521278919 13430288977@139.com Principal Investigator: Jinghua Chen, MD Kaiping Central Hospital Recruiting Kaiping, Guangdong, China, 529300 Contact: Shijie Zhang, MD 13717287622 Shijie_9262511@163.com China, Hunan The First Affiliated Hospital of University Of South China Recruiting Hengyang, Hunan, China, 421001 Contact: Xiaoping Wu, MD 13975486015 wxp19730806@sina.com China, Shanxi The First Affiliated Hospital Of Xi'an Jiaotong University Recruiting Xi'an, Shanxi, China, 710061 Contact: Xin Zheng, MD 13649265446 183421344@qq.com Collapse << |
| NCT03097848 | HCC | Not Applicable | Recruiting | December 31, 2018 | China, Chongqing ... more >> Southwest Hospital Recruiting Chongqing, Chongqing, China, 400038 Contact: kuansheng Ma, MD Collapse << |
| NCT03113604 | - | - | Withdrawn(The study has been s... more >>topped because no one is) Collapse << | - | France ... more >> CHU Amiens Picardie Amiens, Picardie, France, 80054 Collapse << |
| NCT02302729 | Undernutrition | Not Applicable | Completed | - | Guatemala ... more >> APEVIHS (Association for the Prevention and Study of HIV/AIDS) Retalhuleu, Guatemala Collapse << |
| NCT03178656 | Portal Vein Tumor Thrombus | Phase 4 | Recruiting | December 15, 2019 | China, Hubei ... more >> Hepatic surgery center, Tong ji Hospital Recruiting Wuhan, Hubei, China, 430030 Contact: Xiaoping Chen, Doctor 8602783662851 chenxp@medmail.com.cn Contact: Qiong Gong, Doctor 8618086496360 dtjgq486@hust.edu.cn Principal Investigator: peng zhu, doctor Collapse << |
| NCT03140332 | Hepatocellular Carcinoma ... more >> Perfusion Scanner Collapse << | Not Applicable | Recruiting | February 27, 2020 | France ... more >> CHU Amiens Picardie Recruiting Amiens, Picardie, France, 80054 Contact: Vincent HAUTEFEUILLE, Dr +33322668206 hautefeuille.vincent@chu-amiens.fr Collapse << |
| NCT03212625 | Hepatocellular Carcinoma | Phase 4 | Active, not recruiting | January 31, 2019 | Korea, Republic of ... more >> Korea University Guro Hospital Seoul, Korea, Republic of, 08308 Collapse << |
| NCT03236649 | Hepatocellular Carcinoma (HCC) | Phase 3 | Not yet recruiting | August 2019 | - |
| NCT03518502 | Hepatocellular Carcinoma ... more >> Metastasis Collapse << | Phase 4 | Recruiting | February 28, 2022 | Korea, Republic of ... more >> Korea University Ansan Hospital Recruiting Ansan, Gyeonggi-do, Korea, Republic of, 425-707 Contact: Hyung Joon Yim, M.D. 82-31-412-6565 gudwns21@korea.ac.kr Contact: Sang Jun Suh, M.D. 82-31-412-4926 mothpickle@naver.com Principal Investigator: Hyung Joon Yim, M.D. Sub-Investigator: Sang Jun Suh, M.D. Sub-Investigator: Young Kul Jung, M.D. Soonchunghyang University Bucheon Hospital Recruiting Bucheon, Gyeonggi-do, Korea, Republic of Contact: Young-Seok Kim Keimyung University Dongsan Hospital Recruiting Daegu, Korea, Republic of Contact: Woo Jin Chung Chonnam National University Hwasoon Hospital Recruiting Gwangju, Korea, Republic of Contact: Sung-Bum Cho, M.D. Seoul Saint Marry Hospital, the Catholic University of Korea Recruiting Seoul, Korea, Republic of Contact: Si-Hyun Bae Severance Hospital, Yonsei University Recruiting Seoul, Korea, Republic of Contact: Jun Yong Park, M.D. Collapse << |
| NCT03565536 | Thyroid Cancer, Anaplastic | Phase 2 | Recruiting | October 30, 2019 | China, Fujian ... more >> Fujian Medical University Union Hospital Recruiting FuZhou, Fujian, China, 350001 Contact: bo wang, md 13705947900 wangbo@fjmu.edu.cn Collapse << |
| NCT03582618 | Hepatocellular Carcinoma ... more >> Advanced Cancer Collapse << | Phase 2 | Recruiting | June 2021 | United States, California ... more >> Innovative Clinical Research Institute Recruiting Whittier, California, United States, 90603 United States, South Carolina Charleston Hematology Oncology Associates Recruiting Charleston, South Carolina, United States, 29403 Taiwan National Cheng Kung University Hospital Not yet recruiting Tainan, Taiwan, 704 Collapse << |
| NCT02988440 | Hepatocellular Carcinoma | Phase 1 | Recruiting | August 16, 2019 | United States, Michigan ... more >> Karmanos Cancer Institute Recruiting Detroit, Michigan, United States, 48201 Contact: Colette Zack zackc@karmanos.org Principal Investigator: Philip A. Philip Canada, Quebec Novartis Investigative Site Recruiting Montreal, Quebec, Canada, H3T 1E2 France Novartis Investigative Site Terminated Clichy, France, 92110 Germany Novartis Investigative Site Completed Essen, Germany, 45147 Hong Kong Novartis Investigative Site Active, not recruiting Hong Kong, Hong Kong Italy Novartis Investigative Site Recruiting Rozzano, MI, Italy, 20089 Japan Novartis Investigative Site Active, not recruiting Kashiwa, Chiba, Japan, 277-8577 Novartis Investigative Site Active, not recruiting Yokohama city, Kanagawa, Japan, 232 0024 Spain Novartis Investigative Site Completed Pamplona, Navarra, Spain, 31008 Taiwan Novartis Investigative Site Active, not recruiting Taipei, Taiwan, 10002 Collapse << |
| NCT03412773 | Hepatocellular Carcinoma (HCC) | Phase 3 | Recruiting | May 2022 | - |
| NCT03622541 | FLT3-ITD Mutation ... more >> AML Collapse << | Phase 2 | Recruiting | November 2019 | Hong Kong ... more >> The University of Hong Kong Recruiting Hong Kong, Hong Kong Contact: Chunxiao Zhang, SC 852-22554361 chunxiao@hku.hk Collapse << |
| NCT03606590 | Hepatocellular Carcinoma | Phase 2 | Recruiting | December 2020 | Spain ... more >> Vall d'Hebron Institute of Oncology (VHIO) Recruiting Barcelona, Spain Contact: Teresa Macarulla, Dr. +34-932748855 tmacarulla@vhio.net Contact: Raquel García Pelaez +34-934893000 rgarcia@vhio.net Principal Investigator: Teresa Macarulla Mercade, Dr. HM Hospitales - Centro Integral Oncológico Clara Campal Recruiting Madrid, Spain, 28050 Contact: Antonio Cubillo, Dr. acubillo@hmhospitales.com Principal Investigator: acubillo@hmhospitales.com acubillo@hmhospitales.com, Dr. Collapse << |
| NCT03674060 | Healthy, Male | Phase 1 | Not yet recruiting | February 2019 | - |
| NCT03535259 | Hepatocellular Carcinoma, Radi... more >>otherapy, Sorafenib Collapse << | Phase 2 | Recruiting | April 1, 2021 | China, Beijing ... more >> Bo Chen Recruiting Beijing, Beijing, China, 100021 Contact: Bo Chen, MD 008613240000876 chenboo@outlook.com Contact: Yexiong Li, MD 00861087788860 yexiong12@163.com Principal Investigator: Yexiong Li, MD Principal Investigator: Bo Chen, MD Sub-Investigator: Huiying Zeng, MD Sub-Investigator: Liming Wang, MD Collapse << |
| NCT02989311 | Anemia Iron D... more >>eficiency Collapse << | Not Applicable | Completed | - | Kenya ... more >> Msambweni County Referral Hospital Msambweni, Kwale County, Kenya Collapse << |
| NCT00872365 | Preeclampsia ... more >>Pregnancy Micronutrients Exercise Collapse << | Not Applicable | Completed | - | Colombia ... more >> Ana C Plata, MSc Cali, Valle Del Cauca, Colombia Collapse << |
| NCT02866448 | Cardiovascular Disease | Not Applicable | Withdrawn(Funding delayed beyo... more >>nd acceptable start date) Collapse << | - | United Kingdom ... more >> University of Reading Reading, Berkshire, United Kingdom, RG6 6AP Collapse << |
| NCT02481375 | Anemia Iron D... more >>eficiency Hemoglobin Disorder Infection Inflammation Collapse << | Not Applicable | Completed | - | Cambodia ... more >> Kampong Chhnang province Kampong Chhnang, Kampong Chhnang Province, Cambodia Collapse << |
| NCT02691780 | Refractory Solid Tumors | Not Applicable | Active, not recruiting | December 2018 | Korea, Republic of ... more >> Samsung Medical Center Seoul, seoul, korea, Republic of, Korea, Republic of, 135-710 Collapse << |
| NCT01381354 | Multiple Sclerosis | Phase 1 | Completed | - | United States, Iowa ... more >> Univeristy of Iowa/ VA Iowa City VA Medical Center Iowa City, Iowa, United States, 52246 Collapse << |
| NCT02890992 | Hypercholesterolaemia | Phase 2 | Active, not recruiting | February 22, 2019 | United States, Missouri ... more >> Investigational Site Number 8400002 Saint Louis, Missouri, United States, 63110 United States, North Carolina Investigational Site Number 8400005 Charlotte, North Carolina, United States, 28204 United States, Ohio Investigational Site Number 8400001 Cincinnati, Ohio, United States, 45229 Canada Investigational Site Number 1240001 Quebec, Canada, G1V 4W2 Czechia Investigational Site Number 2030001 Brno, Czechia, 62500 Investigational Site Number 2030003 Praha 5 - Motol, Czechia, 15006 Investigational Site Number 2030002 Zlin, Czechia, 76275 France Investigational Site Number 2500001 Bron Cedex, France, 69677 Netherlands Investigational Site Number 5280001 Amsterdam, Netherlands, 1105 AZ Norway Investigational Site Number 5780001 Oslo, Norway, 0373 Russian Federation Investigational Site Number 6430001 Kemerovo, Russian Federation, 650002 Investigational Site Number 6430004 Saint-Petersburg, Russian Federation, 194100 South Africa Investigational Site Number 7100001 Parow, South Africa, 7500 Spain Investigational Site Number 7240004 A Coruna, Spain, 15001 Investigational Site Number 7240001 Madrid, Spain, 28009 Sweden Investigational Site Number 7520001 Stockholm, Sweden, 171 76 Collapse << |
| NCT01147042 | IFN-Gamma Therapy ... more >> CGD Gene Mutation CGD Response to IFNg CGD - Chronic Granulomatous Disease Immunodeficiency Disease Collapse << | Phase 4 | Terminated(Early termination d... more >>ue to only 2 subjects completing trial.) Collapse << | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT01147042 | - | - | Terminated(Early termination d... more >>ue to only 2 subjects completing trial.) Collapse << | - | - |
| NCT02481375 | - | - | Completed | - | - |
| NCT03510715 | Hypercholesterolemia | Phase 3 | Recruiting | June 2020 | United States, California ... more >> Investigational Site Number 8400004 Recruiting Los Angeles, California, United States, 90027 Austria Investigational Site Number 0400001 Recruiting Wien, Austria, 1090 Brazil Investigational Site Number 0760001 Recruiting Sao Paulo, Brazil, 05403-000 Canada Investigational Site Number 1240002 Recruiting Montreal, Canada, H3T 1C5 Investigational Site Number 1240001 Recruiting Quebec, Canada, G1V 4W2 Denmark Investigational Site Number 2080001 Recruiting Viborg, Denmark, 8800 France Investigational Site Number 2500001 Recruiting Bron Cedex, France, 69677 Italy Investigational Site Number 3800001 Recruiting Palermo, Italy, 90127 Mexico Investigational Site Number 4840006 Recruiting Oaxaca, Mexico, 68000 Netherlands Investigational Site Number 5280001 Recruiting Amsterdam, Netherlands, 1105 AZ Norway Investigational Site Number 5780001 Recruiting Oslo, Norway, 0373 Russian Federation Investigational Site Number 6430002 Recruiting Kemerovo, Russian Federation, 650002 Slovenia Investigational Site Number 7050001 Recruiting Ljubljana, Slovenia, 1000 Spain Investigational Site Number 7240001 Recruiting A Coruna, Spain, 15001 Taiwan Investigational Site Number 1580001 Recruiting Taipei, Taiwan, 112 Turkey Investigational Site Number 7920002 Recruiting Ankara, Turkey, 06500 Investigational Site Number 7920001 Recruiting Izmir, Turkey, 35040 Collapse << |
| NCT02428647 | Diarrhea | Not Applicable | Completed | - | Lao People's Democratic Republ... more >>ic National Institute of Public Health Vientiane, Lao People's Democratic Republic Collapse << |
| NCT02768181 | Stunting in Under-2 Children | Not Applicable | Enrolling by invitation | November 2018 | Bangladesh ... more >> ICDDR,B Dhaka, Bangladesh, 1212 Collapse << |
| NCT01271218 | Knee Osteoarthritis | Phase 4 | Completed | - | United States, Texas ... more >> Exercise & Sport Nutrition Lab College Station, Texas, United States, 78843 Collapse << |
| NCT03533712 | Small for Gestational Age at D... more >>elivery Low Birth Weight Prematurity Infant Malnutrition Collapse << | Phase 4 | Not yet recruiting | May 2022 | Burkina Faso ... more >> Houndé district Not yet recruiting Houndé, Tuy, Burkina Faso Contact: Laeticia Celine Toe, MD cellaety@yahoo.fr Collapse << |
| NCT03645980 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Recruiting | August 31, 2022 | Germany ... more >> Universitätsmedizin Mainz, I. Med. Klinik und Poliklinik Recruiting Mainz, Germany, 55131 Contact: Jens U Marquardt, Dr.med. +49 6131 17 ext 6863 marquarj@uni-mainz.de Collapse << |
| NCT01144169 | Renal Cell Carcinoma | Phase 1 | Terminated(barriers to accrual... more >> delay until surgery and additional pre-operative visits) Collapse << | - | United States, Pennsylvania ... more >> UPCI/UPMC Cancer Centers Pittsburgh, Pennsylvania, United States, 15232 UPMC Department of Urology Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00000461 | Cardiovascular Diseases ... more >> Coronary Disease Heart Diseases Myocardial Ischemia Collapse << | Phase 2 | Completed | - | - |
| NCT00089960 | Gastrointestinal Cancer | Phase 2 | Completed | - | - |
| NCT00093873 | Tumors | Phase 1 | Completed | - | - |
| NCT03230396 | Nutrient Pharmacokinetics | Not Applicable | Completed | - | - |
| NCT01482689 | Healthy | Not Applicable | Completed | - | Norway ... more >> University of Oslo Oslo, Norway Collapse << |
| NCT00519714 | Hypertriglyceridemia | Phase 2 Phase 3 | Completed | - | Canada, Quebec ... more >> Institut de Cardiologie de Montreal Montreal, Quebec, Canada, H1T 1C8 Collapse << |
| NCT00360867 | Solid Tumors | Phase 2 | Terminated(Amgen decision) | - | - |
| NCT00860470 | Infant Mortality ... more >> Preterm Birth Low Birth Weight Neonatal Mortality Perinatal Mortality Collapse << | Phase 3 | Completed | - | United States, Maryland ... more >> Johns Hopkins School of Public Health Baltimore, Maryland, United States, 21205 Bangladesh JiVitA Project Office Rangpur, Gaibandha District, Bangladesh Collapse << |
| NCT03287882 | Adolescents A... more >>nemia Pregnancy Empowerment Collapse << | Phase 3 | Recruiting | April 30, 2021 | Pakistan ... more >> Matiari Research and Training Centre Recruiting Matiari, Sindh, Pakistan Contact: Zulfiqar A Bhutta, PhD, MBBS 011922134930051 ext 1054 Collapse << |
| NCT00448786 | Solid Tumors | Phase 1 | Completed | - | - |
| NCT00791752 | Healthy | Phase 1 | Completed | - | Sweden ... more >> Research Site Gotteborg, Sweden Collapse << |
| NCT00804570 | Alcohol Dependence | Phase 2 | Completed | - | United States, Indiana ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Indianapolis, Indiana, United States, 46260 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lafayette, Indiana, United States, 47905 United States, Maryland For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Baltimore, Maryland, United States, 21205 United States, Massachusetts For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Belmont, Massachusetts, United States, 02478 United States, Minnesota For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Rochester, Minnesota, United States, 55905 United States, Nebraska For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Omaha, Nebraska, United States, 68133 United States, New Hampshire For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lebanon, New Hampshire, United States, 03756 United States, New York For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. New York, New York, United States, 10016 United States, North Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chapel Hill, North Carolina, United States, 27599 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Charlotte, North Carolina, United States, 28211 United States, Ohio For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Cincinnati, Ohio, United States, 45237 United States, Oregon For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Portland, Oregon, United States, 97210 United States, Pennsylvania For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Philadelphia, Pennsylvania, United States, 19104 United States, Rhode Island For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Providence, Rhode Island, United States, 02908 United States, South Carolina For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Charleston, South Carolina, United States, 29425 United States, Texas For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. San Antonio, Texas, United States, 78229 United States, Washington For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seattle, Washington, United States, 98104 Collapse << |
| NCT00841048 | Diabetes Mellitus Type 2 | Phase 1 | Completed | - | Sweden ... more >> Research Site Gothenburg, Sweden Collapse << |
| NCT00804570 | - | - | Completed | - | - |
| NCT00957580 | Leukemia, Myeloid, Acute ... more >> Hematologic Neoplasms Collapse << | Phase 2 | Terminated(The trial has been ... more >>terminated due to an estimated low probability of clinical benefit based on limited anti-leukemic effects observed in safety run-in (Part 1)) Collapse << | - | United States, Illinois ... more >> Northwestern University Chicago, Illinois, United States United States, Maryland Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Baltimore, Maryland, United States United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States Dana Farber Cancer Institute Boston, Massachusetts, United States United States, Texas University of Texas - MD Anderson Cancer Center Houston, Texas, United States France Hospital Hotel Dieu, Service D'Hematologie Nantes, Cedex, France Hospital Edouard Herriot, Service d'Hematologie Clinique Lyon Cedex, France Hospital Saint Louis, Service des Maladies du Sang Paris, France CHU du Haut-Leveque, Service des Maladies du Sang Unite de Recherche Clinique Pessac, France Collapse << |
| NCT00860470 | - | - | Completed | - | - |
| NCT03510884 | Hypercholesterolaemia | Phase 3 | Recruiting | March 2022 | - |
| NCT00982865 | Solid Tumors ... more >>Cancer Collapse << | Phase 1 | Completed | - | Australia ... more >> Westmead Hospital Westmead, Australia Belgium Jules Bordet Institute Brussels, Belgium, B-1000 Ghent University Hospital Ghent, Belgium France Institute Bergonié Bordeaux, France Hôpital Beaujon Paris, France, 92118 Hopital Saint Louis Paris, France Centre Eugène Marquis Rennes, France Institute Claudius Regaud Toulouse, France, 03 31052 Netherlands Netherlands Cancer Institute - Antonie van Leeuwenhoek Hospital Amsterdam, Netherlands Collapse << |
| NCT00799747 | Healthy | Phase 1 | Completed | - | Japan ... more >> Research Site Fukuoka, Japan Collapse << |
| NCT00982865 | - | - | Completed | - | - |
| NCT01096004 | Obesity | Phase 1 | Completed | - | Sweden ... more >> Research Site Goteborg, Sweden Collapse << |
| NCT01085331 | - | - | Terminated(Conduct the randomi... more >>zed Phase 2 part not recommended due to the achieved MTD for pimasertib (45mg/day) in combination with FOLFIRI) Collapse << | - | - |
| NCT01173471 | Raised Intraocular Pressure | Phase 2 | Completed | - | United States, California ... more >> Research Site Newport Beach, California, United States United States, Georgia Research Site Atlanta, Georgia, United States Research Site Morrow, Georgia, United States United States, Kansas Research Site Overland Park, Kansas, United States United States, North Carolina Research Site Charlotte, North Carolina, United States Research Site Durham, North Carolina, United States United States, Tennessee Research Site Memphis, Tennessee, United States Sweden Research Site Lund, Sweden Research Site Molndal, Sweden Research Site Stockholm, Sweden United Kingdom Research Site Nottingham, United Kingdom Collapse << |
| NCT00419146 | Schizophrenia ... more >> Schizophreniform Disorders Schizoaffective Disorder Psychotic Disorders Collapse << | Phase 2 Phase 3 | Completed | - | Norway ... more >> Aker University Hospital Oslo, Norway, 0320 Collapse << |
| NCT01386866 | Advanced Solid Tumors | Phase 1 | Completed | - | - |
| NCT01085331 | Metastatic Colorectal Cancer | Phase 1 Phase 2 | Terminated(Conduct the randomi... more >>zed Phase 2 part not recommended due to the achieved MTD for pimasertib (45mg/day) in combination with FOLFIRI) Collapse << | - | Belgium ... more >> Research Site Leuven, Belgium Italy Research Site Napoli, Italy Spain Research Site Barcelona, Spain Collapse << |
| NCT00957580 | - | - | Terminated(The trial has been ... more >>terminated due to an estimated low probability of clinical benefit based on limited anti-leukemic effects observed in safety run-in (Part 1)) Collapse << | - | - |
| NCT03060772 | Alcoholism | Phase 2 | Recruiting | March 20, 2019 | United States, Georgia ... more >> Atlanta VA Medical Center Recruiting Decatur, Georgia, United States, 30033 Contact: David Guidot, MD 404-321-6111 ext 6935 Principal Investigator: David Guidot, MD Sub-Investigator: Ashish Mehta, MD, MSc Collapse << |
| NCT00232531 | Atherosclerosis | Not Applicable | Unknown | - | United Kingdom ... more >> Oxford University Oxford, United Kingdom Collapse << |
| NCT01173471 | - | - | Completed | - | - |
| NCT01693068 | N-Ras Mutated Locally Advanced... more >> or Metastasis Malignant Cutaneous Melanoma Collapse << | Phase 2 | Completed | - | - |
| NCT01668017 | - | - | Terminated(The sponsor decided... more >> not to conduct the expansion part of trial (part 2)) Collapse << | - | - |
| NCT01936363 | - | - | Completed | - | - |
| NCT01713036 | - | - | Completed | - | - |
| NCT01992874 | Neoplasms | Phase 1 | Completed | - | United States, Massachusetts ... more >> Please Contact U.S. Medical Information Located in Rockland, Massachusetts, United States Collapse << |
| NCT01985191 | Neoplasm Malignant | Phase 1 | Completed | - | France ... more >> Investigational Site Number 250001 Villejuif Cedex, France, 94805 Netherlands Investigational Site Number 528001 Amsterdam, Netherlands, 1066 CX Investigational Site Number 528003 Rotterdam, Netherlands, 3075 EA Investigational Site Number 528002 Utrecht, Netherlands, 3584 CX Collapse << |
| NCT01693068 | - | - | Completed | - | - |
| NCT01668017 | Advanced Solid Tumors ... more >> Hepatocellular Carcinoma Collapse << | Phase 1 | Terminated(The sponsor decided... more >> not to conduct the expansion part of trial (part 2)) Collapse << | - | Japan ... more >> Please contact Merck Serono Co., Ltd for recruiting locations in, Japan Collapse << |
| NCT02017444 | Idiopathic Intracranial Hypert... more >>ension Collapse << | Phase 2 | Completed | - | United Kingdom ... more >> University Hospital Birmingham (Queen Elizabeth Hospital) Birmingham, West Midlands, United Kingdom, B15 2TH Collapse << |
| NCT01992874 | - | - | Completed | - | - |
| NCT01713036 | Locally Advanced or Metastatic... more >> Solid Tumors Collapse << | Phase 1 | Completed | - | Hungary ... more >> Research Site Budapest, Hungary Collapse << |
| NCT00619060 | - | - | Completed | - | - |
| NCT01129258 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 Pfizer Investigational Site Fountain Valley, California, United States, 92708 United States, Florida Pfizer Investigational Site Miami, Florida, United States, 33169 United States, Texas Pfizer Investigational Site San Antonio, Texas, United States, 78229 Collapse << |
| NCT00197548 | Pregnancy Pre... more >>mature Birth Infant, Low Birth Weight Pregnancy Outcomes Collapse << | Phase 3 | Completed | - | - |
| NCT01369277 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00765284 | - | - | Completed | - | United States, Texas ... more >> Diabetes and Glandular Research Associates San Antonio, Texas, United States, 78229 Collapse << |
| NCT01469065 | Diabetes Mellitus, Type 2 | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Chula Vista, California, United States, 91911 United States, Florida Pfizer Investigational Site Miami, Florida, United States, 33169 Japan Pfizer Investigational Site Hachioji-shi, Tokyo, Japan Collapse << |
| NCT01369602 | Type 2 Diabetes Mellitus ... more >> Healthy Collapse << | Phase 1 | Completed | - | United States, California ... more >> Pfizer Investigational Site Anaheim, California, United States, 92801 United States, Florida Pfizer Investigational Site DeLand, Florida, United States, 32720 Collapse << |
| NCT01102673 | Diabetes Mellitus ... more >> Diabetes Mellitus, Type 2 Glucose Metabolism Disorders Collapse << | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01469481 | Healthy | Phase 1 | Completed | - | United States, Washington ... more >> Pfizer Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT00619060 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Arizona Cancer Center at University of Arizona Health Sciences Center Tucson, Arizona, United States, 85724-5024 Collapse << |
| NCT01071291 | Dyslipidemia | Phase 1 | Completed | - | - |
| NCT01388829 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT01104519 | Dyslipidemia | Phase 1 | Completed | - | - |
| NCT03711682 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | El Salvador ... more >> Unidad Comunitaria de Salud Familiar Intermedia de Comasagua (UCSFI-Comasagua) Comasagua, La Libertad, El Salvador, 0504 Collapse << |
| NCT03313297 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | April 1, 2019 | United Kingdom ... more >> Leeds Teaching Hospitals Trust Recruiting Leeds, United Kingdom, LS9 7TF Contact: Ana Tiganescu, PhD 00441133438336 a.tiganescu@leeds.ac.uk Collapse << |
| NCT01469065 | - | - | Completed | - | - |
| NCT01131689 | Hepatocellular Carcinoma | Phase 1 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01141478 | Carcinoma, Hepatocellular | Not Applicable | Unknown | June 2018 | United States, California ... more >> Loma Linda University Medical Center Recruiting Loma Linda, California, United States, 92354 Collapse << |
| NCT01135056 | Hepatocellular Carcinoma | Phase 3 | Active, not recruiting | July 31, 2018 | - |
| NCT01189370 | Renal Cell Cancer | Phase 2 | Completed | - | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 Collapse << |
| NCT01182272 | Liver Cancer | Phase 2 | Unknown | - | France ... more >> Hopital Beaujon Recruiting Clichy, France, 92110 Contact: Contact Person 33-1-4087-5614 sandrine.faivre@bjn.aphp.fr Collapse << |
| NCT01158287 | Esophageal Cancer ... more >> Gastric Cancer Collapse << | Phase 2 | Completed | - | Ireland ... more >> Bon Secours Hospital Cork, Ireland Cork University Hospital Cork, Ireland Adelaide and Meath Hospital, Dublin Incorporating the National Children's Hospital Dublin, Ireland, 24 Mater Misericordiae University Hospital Dublin, Ireland, 7 St. James's Hospital Dublin, Ireland, 8 Beaumont Hospital Dublin, Ireland, 9 University College Hospital Galway, Ireland Waterford Regional Hospital Waterford, Ireland Collapse << |
| NCT01171651 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Korea, Republic of ... more >> Pusan National University Hospital Busan, Korea, Republic of Pusan National University Yangsan Hospital Yangsan, Korea, Republic of Collapse << |
| NCT01170104 | Hepatocellular Carcinoma | Phase 2 | Unknown | June 2013 | Korea, Republic of ... more >> Chung-Ang University Hospital Recruiting Dongjak, Seoul, Korea, Republic of, 156-755 Contact: Joung-Soon Jang, M.D. 82-2-6299-1427 alsaba@daum.net Contact: In Gyu Hwang, M.D. 82-2-748-9786 higyu72@yahoo.co.kr Principal Investigator: Joung-Soon Jang, M.D. Sub-Investigator: Hyung Joon Kim, M.D. Sub-Investigator: Hyun Woong Lee, M.D. Sub-Investigator: In Gyu Hwang, M.D. Collapse << |
| NCT01159327 | Small Cell Lung Cancer | Phase 2 | Terminated(low enrolling parti... more >>cipants) Collapse << | - | Korea, Republic of ... more >> National Cancer Center, Korea Goyang-si, Gyeonggi-do, Korea, Republic of Collapse << |
| NCT01203787 | Hepatocellular Carcinoma | Phase 4 | Completed | - | United States, Florida ... more >> University of Florida Hepatology Gainesville, Florida, United States, 32610-0277 Mayo Clinic Jacksonville, Florida, United States, 32224 Florida Hospital Transplant Center Orlando, Florida, United States, 32804 Tampa General Hospital Tampa, Florida, United States, 33606 United States, Illinois Loyola University Medical Center Maywood, Illinois, United States, 60153 United States, Michigan Henry Ford Health System Detroit, Michigan, United States, 48202 United States, Pennsylvania Drexel University College of Medicine Philadelphia, Pennsylvania, United States, 19107 United States, Texas University of Texas Health Science Center Houston Houston, Texas, United States, 77030 Brooke Army Medical Center San Antonio, Texas, United States, 78234 Collapse << |
| NCT01189370 | - | - | Completed | - | - |
| NCT01204177 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Hong Kong ... more >> Shatin, New Territories, Hong Kong Hong Kong, Hong Kong Korea, Republic of Jung-gu, Daegu Gwang''yeogsi, Korea, Republic of, 700-721 Goyang-si, Gyeonggido, Korea, Republic of, 410-769 Busan, Korea, Republic of, 602-739 Seoul, Korea, Republic of, 110-744 Seoul, Korea, Republic of, 120-752 Seoul, Korea, Republic of, 135-710 Seoul, Korea, Republic of, 138-736 Singapore Singapore, Singapore, 228510 Singapore, Singapore, 258499 Taiwan Kaohsiung, Taiwan, 833 Tainan, Taiwan, 736 Tainan, Taiwan Taipei, Taiwan, 100 Collapse << |
| NCT01203787 | - | - | Completed | - | - |
| NCT01215266 | Bladder Cancer | Phase 2 | Terminated(Slow recruitment) | - | - |
| NCT01223027 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT01232296 | - | - | Completed | - | - |
| NCT01271504 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Completed | - | United States, Arizona ... more >> Tucson, Arizona, United States, 85715 United States, Florida Fort Myers, Florida, United States, 33905 Saint Petersburg, Florida, United States, 33705 United States, North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Ohio Cincinnati, Ohio, United States, 45267 Toledo, Ohio, United States, 43623 United States, Tennessee Nashville, Tennessee, United States, 37203 Belgium Brussels, Belgium, 1070 Bruxelles, Belgium, 1200 Leuven, Belgium, 3000 Italy Rozzano, Milano, Italy, 20089 Modena, Italy, 40124 Pavia, Italy, 27100 Spain Madrid, Spain, 28007 Madrid, Spain, 28034 Madrid, Spain, 28046 Madrid, Spain, 28050 Ukraine Dnipropetrovsk, Ukraine, 49102 Donetsk, Ukraine, 83092 Kharkiv, Ukraine, 61037 Lviv, Ukraine, 79031 United Kingdom London, Greater London, United Kingdom, WC1E 6BT Manchester, Greater Manchester, United Kingdom, M20 4BX Glasgow, Strathclyde, United Kingdom, G12 OYN Collapse << |
| NCT01328223 | - | - | Unknown | December 2012 | Taiwan ... more >> Shang-Wen Chen Recruiting Taichung, Taiwan, 404 Contact: Shang-Wen Chen, MD 886-4-22052121 ext 7450 vincent1680616@yahoo.com.tw Sub-Investigator: Yu-Cheng Kuo Sub-Investigator: Ji-An Liang, MD Li-Ching Lin Recruiting Tainan, Taiwan, 700 Contact: Li-Ching Lin, MD liching51@yahoo.com.tw Jeng-Fong Chiou Recruiting Taipei, Taiwan, 100 Contact: Jeng-Fong Chiou, MD;PhD sjfchiou@xuite.net Collapse << |
| NCT01232296 | Hepatocellular Carcinoma | Phase 2 | Completed | - | China, Jiangsu ... more >> Novartis Investigative Site Nanjing, Jiangsu, China, 210002 China, Shanxi Novartis Investigative Site Xi'an, Shanxi, China, 710032 China, Zhejiang Novartis Investigative Site Hangzhou, Zhejiang, China, 310016 China Novartis Investigative Site Beijing, China, 100039 Hong Kong Novartis Investigative Site Hong Kong, Hong Kong Novartis Investigative Site Shatin, New Territories, Hong Kong Japan Novartis Investigative Site Kashiwa, Chiba, Japan, 277-8577 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 232-0024 Novartis Investigative Site OsakaSayama, Osaka, Japan, 589-8511 Korea, Republic of Novartis Investigative Site Seoul, Korea, Korea, Republic of, 03722 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 05505 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 06351 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 110 744 Novartis Investigative Site Seoul, Korea, Republic of, 136-705 Singapore Novartis Investigative Site Singapore, Singapore, 308433 Taiwan Novartis Investigative Site Taipei, Taiwan, ROC, Taiwan, 112 Novartis Investigative Site Kuei-Shan Chiang, Taoyuan/ Taiwan ROC, Taiwan, 33305 Novartis Investigative Site Taichung, Taiwan, 40705 Novartis Investigative Site Taipei, Taiwan, 10048 Thailand Novartis Investigative Site Bangkok, Thailand, 10330 Novartis Investigative Site Bangkok, Thailand, 10700 Novartis Investigative Site Chiang Mai, Thailand, 50200 Novartis Investigative Site Khon Kaen, Thailand, 40002 Novartis Investigative Site Songkla, Thailand, 90110 Collapse << |
| NCT01319942 | - | - | Unknown | June 2013 | Taiwan ... more >> Chi-Mei Hospital Recruiting Tainan, Taiwan, 600 Contact: Li-Ching Lin, MD 886-6-2812811 ext 53501 liching51@yahoo.com.tw Principal Investigator: Li-Ching Lin, MD Taipei Medical University Hospital Recruiting Taipei, Taiwan, 100 Contact: Jeng-Fong Chiou, MD;PhD 886-2-27372181 ext 2130 sjfchiou@xuite.net Principal Investigator: Jen-Fong Chiou, MD;PhD Collapse << |
| NCT01276210 | Tumors Metastatic to Brain | Phase 1 | Completed | - | United States, Tennessee ... more >> Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << |
| NCT01334710 | - | - | Terminated(safety issue observ... more >>ed on another hepatocellular study using OSI906) Collapse << | - | - |
| NCT01334710 | Liver Cancer | Phase 2 | Terminated(safety issue observ... more >>ed on another hepatocellular study using OSI906) Collapse << | - | United States, Georgia ... more >> Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Michigan University of Michigan Ann Arbor, Michigan, United States, 48109 United States, Ohio Ohio State University Columbus, Ohio, United States, 43210 Collapse << |
| NCT01338857 | neurofibromatosis1 (NF1) ... more >> Recurrent or Progressive Optic Pathway Gliomas (OPG) Recurrent or Progressive Low-grade Glioma Collapse << | Phase 2 | Terminated(Sorafenib ineffecti... more >>ve for tx of recurrent or progressive PLGA) Collapse << | - | United States, New York ... more >> New York University Stephen D. Hassenfeld Children's Center for Cancer & Blood Disorders New York, New York, United States, 10016 Collapse << |
| NCT01115322 | Acne Vulgaris | Phase 1 | Completed | - | United States, Arizona ... more >> HillTop Research Corporation Scottsdale, Arizona, United States, 85251 Collapse << |
| NCT01338857 | - | - | Terminated(Sorafenib ineffecti... more >>ve for tx of recurrent or progressive PLGA) Collapse << | - | - |
| NCT00779896 | Cutaneous T-Cell Lymphoma | Phase 1 Phase 2 | Unknown | December 2012 | Canada, Quebec ... more >> McGill University Recruiting Montreal, Quebec, Canada, H2W 1S6 Contact: Penny Chipman 514-398-1444 penny.chipman@mcgill.ca Contact: Crystal Lameira 514-398-2229 crystal.lameira@mcgill.ca Collapse << |
| NCT01223027 | - | - | Completed | - | - |
| NCT00278382 | Anaplastic Large Cell Lymphoma... more >> Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Mantle Cell Lymphoma Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> University of Maryland Greenebaum Cancer Center Baltimore, Maryland, United States, 21201-1595 Collapse << |
| NCT01342627 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(low enrollment rate... more >>) Collapse << | - | Italy ... more >> Centro di Riferimento Oncologico Aviano, Pordenone, Italy, 33081 Collapse << |
| NCT00783965 | Skin Carcinoma | Phase 2 | Completed | - | United States, California ... more >> Children's Hospital Oakland Research Institute Oakland, California, United States, 94609-1693 Children's Hospital Oakland Research Institiute Oakland, California, United States, 94609 United States, New York Herbert Irving Comprehensive Cancer Center at Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00303966 | Refractory Chronic Lymphocytic... more >> Leukemia Stage I Chronic Lymphocytic Leukemia Stage II Chronic Lymphocytic Leukemia Stage III Chronic Lymphocytic Leukemia Stage IV Chronic Lymphocytic Leukemia Collapse << | Phase 2 | Terminated(Slow accrual) | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637-1470 Collapse << |
| NCT00330421 | Metastatic Ewing Sarcoma/Perip... more >>heral Primitive Neuroectodermal Tumor Metastatic Osteosarcoma Recurrent Adult Soft Tissue Sarcoma Recurrent Ewing Sarcoma/Peripheral Primitive Neuroectodermal Tumor Recurrent Osteosarcoma Stage I Adult Soft Tissue Sarcoma Stage II Adult Soft Tissue Sarcoma Stage III Adult Soft Tissue Sarcoma Stage IV Adult Soft Tissue Sarcoma Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00783965 | - | - | Completed | - | - |
| NCT00303966 | - | - | Terminated(Slow accrual) | - | - |
| NCT00301847 | Kidney Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Cancer Research Center Chicago, Illinois, United States, 60637-1470 Decatur Memorial Hospital Cancer Care Institute Decatur, Illinois, United States, 62526 Evanston Northwestern Health Care - Evanston Hospital Evanston, Illinois, United States, 60201-1781 Ingalls Cancer Care Center at Ingalls Memorial Hospital Harvey, Illinois, United States, 60426 Cardinal Bernardin Cancer Center at Loyola University Medical Center Maywood, Illinois, United States, 60153 Oncology Hematology Associates of Central Illinois, PC - Peoria Peoria, Illinois, United States, 61615-7828 Central Illinois Hematology Oncology Center Springfield, Illinois, United States, 62701 United States, Indiana Fort Wayne Medical Oncology and Hematology Fort Wayne, Indiana, United States, 46885-5099 CCOP - Northern Indiana CR Consortium South Bend, Indiana, United States, 46601 United States, Michigan Oncology Care Associates, PLLC Saint Joseph, Michigan, United States, 49085 United States, Missouri David C. Pratt Cancer Center at St. John's Mercy Saint Louis, Missouri, United States, 63141 United States, Wisconsin Medical College of Wisconsin Cancer Center Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00287495 | Kaposi's Sarcoma ... more >> HHV-8 KSHV Collapse << | Phase 1 | Terminated | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00285857 | Breast Cancer | Phase 2 | Terminated(Slow accrual) | - | United States, California ... more >> Stanford University Cancer Center Stanford, California, United States, 94305 Collapse << |
| NCT00330421 | - | - | Completed | - | - |
| NCT00285857 | - | - | Terminated(Slow accrual) | - | - |
| NCT00373373 | Acute Myeloid Leukemia | Phase 2 | Completed | - | Germany ... more >> Charité Campus Benjamin Franklin, Med. Klinik III Berlin, Germany, 12203 Klinikum Chemnitz GmbH, Klinik für Innere Medizin III Chemnitz, Germany, 09113 Universitätsklinikum Carl Gustav Carus der TU Dresden, Medizinische Klinik I Dresden, Germany, 01307 St. Johannes Hospital, Medizinische Klinik II Duisburg, Germany, 47166 Universitätsklinikum Essen, Zentrum für Innere Medizin, Medizinische Klinik und Poliklinik für Hämatologie Essen, Germany, 45147 Klinikum der J. W. Goethe-Universität Frankfurt am Main, Medizinische Klinik II Frankfurt / Main, Germany, 60590 Allgemeines Krankenhaus St. Georg, Hämatologische Abteilung Hamburg, Germany, 20099 Universitätsklinikum Heidelberg, Med. Klinik V Heidelberg, Germany, D-69120 Klinikum der Universität zu Köln, Klinik I für Innere Medizin Köln, Germany, 50937 Klinikum der Johannes Gutenberg Universität, 3. Medizinische Klinik und Poliklinik Mainz, Germany, 55101 Philipps Universität, Abteilung für Hämatologie, Onkologie und Immunologie Marburg, Germany, 35043 Klinik für Hämatologie und Onkologie Klinikum Minden Minden, Germany, 32423 TU München, Medizinische Klinik III München, Germany, 81675 Universitätsklinikum Münster, Medizinische Klinik A Münster, Germany, 48149 Klinikum Nürnberg, 5. Medizinische Klinik Einheit für Knochenmarktransplantation Nürnberg, Germany, 90419 Universität Regensburg, Abteilung für Hämatologie und Internistische Onkologie Regensburg, Germany, 93042 Robert-Bosch Krankenhaus Stuttgart Stuttgart, Germany, 70376 Julius-Maximilians-Universität Würzburg, Medizinische Klinik und Poliklinik II Würzburg, Germany, 97979 Collapse << |
| NCT00390325 | Multiple Endocrine Neoplasia ... more >> Recurrent Thyroid Gland Carcinoma Thyroid Gland Medullary Carcinoma Collapse << | Phase 2 | Active, not recruiting | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Ohio Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00398814 | Renal Cancer ... more >>Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT00406601 | Sarcoma, Soft Tissue | Phase 2 | Completed | - | Italy ... more >> Istituto Clinico Humanitas Rozzano, Milan, Italy, 20089 Collapse << |
| NCT00405366 | Kidney Cancer | Early Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Collapse << |
| NCT00389285 | Carcinoma, Renal Cell | Phase 1 Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama Birmingham, Alabama, United States, 35294 United States, Arizona Premiere Oncology of Arizona Scottsdale, Arizona, United States, 85260 United States, New Hampshire Dartmouth-Hitchcock Medical Center Lebanon, New Hampshire, United States, 03756 Collapse << |
| NCT00427349 | Gastrointestinal Carcinoid Tum... more >>or Islet Cell Tumor Neoplastic Syndrome Collapse << | Phase 2 | Completed | - | - |
| NCT00411671 | Lung Cancer | Phase 2 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01017120 | Acne Vulgaris | Phase 3 | Completed | - | United States, California ... more >> The Laser Institute for Dermatology Santa Monica, California, United States, 90404 United States, Florida FXM Research Corporation Miami, Florida, United States, 33175 United States, Georgia Atlanta Dermatology & Vein Research Center, PC Alpharetta, Georgia, United States, 30022 Gwinnett Clinical Research Center, Inc. Snellville, Georgia, United States, 30078 United States, Indiana Hudson Dermatology Evansville, Indiana, United States, 47714 United States, Kentucky Dermatology Specialists Louisville, Kentucky, United States, 40202 United States, Michigan Henry Ford Medical Center Detroit, Michigan, United States, 48202 Somerset Skin Centre Troy, Michigan, United States, 48084 United States, Nebraska Skin Specialists, P.C. Omaha, Nebraska, United States, 68144 United States, New York Dermatology Associates of Rochester, PC Rochester, New York, United States, 14623 United States, Rhode Island Clinical Partners, LLC Johnston, Rhode Island, United States, 02919 United States, Texas Suzanne Bruce and Associates, PA Houston, Texas, United States, 77056 Progressive Clinical Research San Antonio, Texas, United States, 78229 United States, Virginia Virginia Clinical Research, Inc. Norfolk, Virginia, United States, 23507 Canada, British Columbia Guildford Dermatology Specialist Surrey, British Columbia, Canada, V3R 6A7 Canada, Manitoba Dermadvances Research Winnipeg, Manitoba, Canada, R3C 1R4 Canada, Ontario Lynderm Research, Inc. Markham, Ontario, Canada, L3P 1A8 North Bay Dermatology Centre North Bay, Ontario, Canada, P1B3Z7 Collapse << |
| NCT01017146 | Acne Vulgaris | Phase 3 | Completed | - | - |
| NCT00445042 | Carcinoma, Renal Cell | Phase 2 | Completed | - | United States, Texas ... more >> Methodist Hospital - Baylor College of Medicine Houston, Texas, United States, 77030 Collapse << |
| NCT00478114 | Renal Cell Carcinoma | Phase 3 | Completed | - | Thailand ... more >> Siriraj Hospital, Department of Medicine Bangkoknoi, Bangkok, Thailand, 10700 Collapse << |
| NCT01017146 | - | - | Completed | - | - |
| NCT00427349 | - | - | Completed | - | - |
| NCT00480389 | Renal Cell Carcinoma ... more >> Metastatic Disease Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital, University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01017120 | - | - | Completed | - | - |
| NCT00490685 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Italy ... more >> Istituto Clinico Humanitas Rozzano, Milan, Italy, 20089 Collapse << |
| NCT01019603 | Acne Vulgaris | Phase 1 | Completed | - | United States, Texas ... more >> DermResearch, Inc. Austin, Texas, United States, 78759 Collapse << |
| NCT00492752 | Carcinoma, Hepatocellular | Phase 3 | Completed | - | China, Anhui ... more >> Hefei, Anhui, China, 230022 China, Guangdong Guangzhou, Guangdong, China, 510060 Guangzhou, Guangdong, China, 510515 China, Hubei Wuhan, Hubei, China, 430030 China, Jiangsu Nanjing, Jiangsu, China, 210003 Nanjing, Jiangsu, China, 210009 China, Liaoning Dalian, Liaoning, China, 116011 Dalian, Liaoning, China, 116027 China, Zhejiang Hangzhou, Zhejiang, China, 310016 China Beijing, China, 100021 Beijing, China, 100039 Chongqing, China, 400038 Shanghai, China, 200003 Shanghai, China, 200032 Tianjin, China Korea, Republic of Seoul, Seoul Teugbyeolsi, Korea, Republic of, 152-703 Daegu, Korea, Republic of, 702-701 Seoul, Korea, Republic of, 138-736 Taiwan Changhua, Taiwan, 500 Tainan, Taiwan, 736 Taipei, Taiwan, 10016 Taipei, Taiwan, 251 Taoyuan, Taiwan, 333 Collapse << |
| NCT00494299 | Carcinoma, Hepatocellular | Phase 3 | Completed | - | - |
| NCT00492986 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT00411671 | - | - | Completed | - | - |
| NCT00496756 | Kidney Cancer | Phase 2 | Terminated(Low accrual rate.) | - | United States, Nebraska ... more >> UNMC Eppley Cancer Center at the University of Nebraska Medical Center Omaha, Nebraska, United States, 68198-6805 Collapse << |
| NCT01114841 | Acne Vulgaris | Phase 1 | Completed | - | United States, Arizona ... more >> HillTop Research Corporation Scottsdale, Arizona, United States, 85251 Collapse << |
| NCT01112787 | Acne Vulgaris | Phase 1 | Completed | - | United States, Arizona ... more >> HillTop Research Corporation Scottsdale, Arizona, United States, 85251 Collapse << |
| NCT00510289 | Myelodysplastic Syndromes ... more >> Leukemia, Myelomonocytic, Chronic Collapse << | Phase 2 | Terminated(Early closure of st... more >>udy due to poor response) Collapse << | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00522301 | Fallopian Tube Cancer ... more >> Ovarian Cancer Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Terminated | - | United States, New York ... more >> Memorial Sloan - Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00492752 | - | - | Completed | - | - |
| NCT01119651 | Acne Vulgaris | Phase 1 | Completed | - | United States, Florida ... more >> HillTop Research Corporation Saint Petersburg, Florida, United States, 33710 Collapse << |
| NCT00522301 | - | - | Terminated | - | - |
| NCT00510289 | - | - | Terminated(Early closure of st... more >>udy due to poor response) Collapse << | - | - |
| NCT00525161 | - | - | Terminated(Slow accrual and lo... more >>ss of funding) Collapse << | - | - |
| NCT00532025 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Terminated | - | Germany ... more >> SIRN investigational trial site Essen, Germany SIRN investigational trial site Halle (Saale), Germany SIRN investigational trial site Loewenstein, Germany SIRN investigational trial site Johannes Gutenberg Universität Mainz, Germany SIRN investigational trial site Katholisches Klinikum Mainz, Germany SIRN investigational trial site Rotenburg (Wuemme), Germany Collapse << |
| NCT00548899 | Breast Cancer | Phase 2 | Completed | - | Germany ... more >> St. Gertrauden Krankenhaus Berlin, Germany, 10713 Klinikum der J. W. Goethe Universität Frankfurt am Main, Germany, 60590 Henriettenstiftung Hannover, Germany, 30559 Elisabeth Krankenhaus Kassel, Germany, 34177 Universitätsklinikum Schleswig-Holstein Campus Kiel Kiel, Germany, 24105 Klinikum der Universität zu Köln Köln, Germany, 50924 Klinikum Offenbach Offenbach, Germany, 63069 Klinikum Südstadt Rostock, Germany, 18059 Dr.-Horst-Schmidt-Kliniken GmbH Wiesbaden, Germany, 65199 Collapse << |
| NCT00544609 | Bladder Cancer | Phase 1 | Completed | - | Spain ... more >> Hospital Clínic i Provincial de Barcelona Barcelona, Spain Hospital de la Santa Creu i Sant Pau Barcelona, Spain Institut Català d' Oncología Barcelona, Spain Collapse << |
| NCT00494299 | - | - | Completed | - | - |
| NCT00557830 | Renal Cell Carcinoma | Phase 2 | Terminated(The study was close... more >>d to enrollment when it became clear that enrollment was too slow to complete the full enrollment target within the time frame allowed.) Collapse << | - | United States, Arkansas ... more >> Clopton Clinic Jonesboro, Arkansas, United States, 72401 United States, California Wilshire Oncology Medical Group, Inc. La Verne, California, United States, 91750 United States, Florida Advanced Medical Specialties Miami, Florida, United States, 33176 United States, Georgia Northeast Georgia Cancer Care Athens, Georgia, United States, 30607 Peachtree Hematology Oncology Consultants Atlanta, Georgia, United States, 30309 Central Georgia Cancer Care Macon, Georgia, United States, 31201 Northwest Georgia Oncology Centers Marietta, Georgia, United States, 30060 United States, Illinois Mid-Illinois Hematology and Oncology Associates, Ltd. Normal, Illinois, United States, 61761 United States, Montana Hematology Oncology Centers of the Northern Rockies Billings, Montana, United States, 59101 United States, North Carolina Gaston Hematology and Oncology Gastonia, North Carolina, United States, 28054 United States, Oregon Pacific Oncology, PC Beaverton, Oregon, United States, 97006 United States, Pennsylvania The Lancaster Cancer Center, Ltd Lancaster, Pennsylvania, United States, 17605 United States, Tennessee The West Clinic Memphis, Tennessee, United States, 38120 Collapse << |
| NCT00557830 | - | - | Terminated(The study was close... more >>d to enrollment when it became clear that enrollment was too slow to complete the full enrollment target within the time frame allowed.) Collapse << | - | - |
| NCT00543335 | Lung Cancer | Phase 1 | Terminated(Lack of Accrual.) | - | United States, Texas ... more >> UT MD . Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00525161 | Breast Cancer | Phase 2 | Terminated(Slow accrual and lo... more >>ss of funding) Collapse << | - | United States, Kentucky ... more >> University of Kentucky Lexington, Kentucky, United States, 40536 Collapse << |
| NCT00586495 | Carcinoma, Renal Cell | Phase 2 | Completed | - | Japan ... more >> Asahi, Chiba, Japan, 289-2511 Matsuyama, Ehime, Japan, 791-0280 Kurume, Fukuoka, Japan, 830-0011 Isesaki, Gunma, Japan, 372-0817 Maebashi, Gunma, Japan, 371-8511 Sapporo, Hokkaido, Japan, 003-0804 Sapporo, Hokkaido, Japan, 060-8543 Sapporo, Hokkaido, Japan, 060-8648 Sunagawa, Hokkaido, Japan, 073-0196 Tsukuba, Ibaraki, Japan, 305-8576 Morioka, Iwate, Japan, 020-8505 Tsu, Mie, Japan, 514-8507 Natori, Miyagi, Japan, 981-1293 Kashihara, Nara, Japan, 634-8522 Kurashiki, Okayama, Japan, 710-8602 Osakasayama, Osaka, Japan, 589-8511 Suita, Osaka, Japan, 565-0871 Hidaka, Saitama, Japan, 350-1298 Irima-gun, Saitama, Japan, 350-0495 Tokorozawa, Saitama, Japan, 359-8513 Hamamatsu, Shizuoka, Japan, 431-3192 Sunto, Shizuoka, Japan, 411-8777 Utsunomiya, Tochigi, Japan, 320-0834 Arakawa-ku, Tokyo, Japan, 116-8567 Bunkyo-ku, Tokyo, Japan, 113-8655 Chuo-ku, Tokyo, Japan, 104-0045 Itabashi-ku, Tokyo, Japan, 173-0003 Shinjuku-ku, Tokyo, Japan, 162-8666 Ube, Yamaguchi, Japan, 755-8505 Akita, Japan, 010-8543 Chiba, Japan, 260-8677 Chiba, Japan, 260-8717 Fukuoka, Japan, 812-0033 Fukuoka, Japan, 812-8582 Kagoshima, Japan, 890-8520 Kyoto, Japan, 602-8566 Kyoto, Japan, 606-8507 Okayama, Japan, 700-8558 Osaka, Japan, 537-8511 Tokushima, Japan, 770-8503 Yamagata, Japan, 990-9585 Collapse << |
| NCT00586105 | - | - | Completed | - | - |
| NCT00606866 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Completed | - | United States, Illinois ... more >> The University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00586495 | - | - | Completed | - | - |
| NCT00595985 | Gastric Cancer | Phase 2 | Terminated(low response rate, ... more >>no evidence of PFS or OS improved.) Collapse << | January 2009 | China, Shanghai ... more >> Fudan University Cancer Hospital ShangHai, Shanghai, China, 200032 Collapse << |
| NCT00609401 | Metastatic Disease ... more >> Renal Cell Carcinoma Collapse << | Phase 2 | Completed | - | Italy ... more >> Istituto Tumori Milano, Italy Collapse << |
| NCT00618384 | Hepatocellular Carcinoma | Phase 2 | Terminated | - | Germany ... more >> Universitätsklinikum Tübingen Innere Medizin I Tübingen, BW, Germany, 72076 Medizinische Universitätsklinik Ulm Innere Medizin I Ulm, BW, Germany, 89081 Klinikum der Universität Großhardern Muenchen, BY, Germany, 81377 Klinikum der Johann-Goethe-Universität Frankfurt, HE, Germany, 60590 Universitätsklinikum Hamburg-Eppendorf Zentrum für Innere Medizin Hamburg, HH, Germany, 20246 Johannes-Gutenberg-Universität Medizinische Klinik Mainz, RP, Germany, 55131 Collapse << |
| NCT00586105 | Carcinoma, Renal Cell | Phase 3 | Completed | - | China, Jiangsu ... more >> Nanjing, Jiangsu, China, 210003 China Beijing, China, 100021 Shanghai, China, 200032 Shanghai, China, 200127 Taiwan Tainan, Taiwan, 70428 Taipei, Taiwan, 10002 Taipei, Taiwan, 112 Taoyuan, Taiwan, 333 Collapse << |
| NCT00609934 | Renal Cell Carcinoma | Phase 1 Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hosptial Toronto, Ontario, Canada, M5G2M9 Collapse << |
| NCT00610246 | Cancer | Phase 1 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00618982 | - | - | Completed | - | - |
| NCT00619242 | Esophageal Cancer | Not Applicable | Terminated(Low accrual) | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00619242 | - | - | Terminated(Low accrual) | - | - |
| NCT00657254 | Neoplasm Meta... more >>stasis Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02115-6084 Belgium Bruxelles - Brussel, Belgium, 1000 Canada, Ontario Hamilton, Ontario, Canada, L8V 5C2 Toronto, Ontario, Canada, M5G 2M9 Germany Essen, Nordrhein-Westfalen, Germany, 45147 Herne, Nordrhein-Westfalen, Germany, 44625 Collapse << |
| NCT00625378 | Neoplasms | Phase 3 | Active, not recruiting | June 30, 2020 | - |
| NCT00618982 | Carcinoma, Renal Cell | Phase 2 | Completed | - | France ... more >> Bordeaux, France, 33000 La Roche Sur Yon, France, 85925 Marseille, France, 13385 Nantes, France, 44805 Paris Cedex 10, France, 75475 Tours, France, 37044 Germany Tübingen, Baden-Württemberg, Germany, 72076 Marburg, Hessen, Germany, 35043 Hannover, Niedersachsen, Germany, 30625 Mainz, Rheinland-Pfalz, Germany, 55131 Jena, Thüringen, Germany, 07740 Italy Aviano, Pordenone, Italy, 33081 Milano, Italy, 20133 Pavia, Italy, 27100 Poland Olsztyn, Poland, 10-226 Warszawa, Poland, 02-781 Warszawa, Poland, 04-141 Wroclaw, Poland, 50 - 556 United Kingdom Greater Manchester, Manchester, United Kingdom, M20 4BX Cardiff, South Glamorgan, United Kingdom, CF14 7TB Glasgow, United Kingdom, G12 0YN London, United Kingdom, SW3 6JJ Collapse << |
| NCT00622479 | Carcinoma, Renal Cell | Phase 1 | Terminated | - | United States, Arizona ... more >> Scottsdale, Arizona, United States, 85259 United States, Florida Jacksonville, Florida, United States, 32224 United States, Illinois Maywood, Illinois, United States, 60153 United States, Michigan Detroit, Michigan, United States, 48201 United States, Minnesota Rochester, Minnesota, United States, 55905 United States, New Jersey Hackensack, New Jersey, United States, 07601 United States, New York Stony Brook, New York, United States, 11794-944 Collapse << |
| NCT00692770 | Carcinoma, Hepatocellular | Phase 3 | Completed | - | - |
| NCT00663741 | Gastric Cancer | Phase 1 | Completed | - | Japan ... more >> Kashiwa, Chiba, Japan, 277-8577 Kobe, Hyogo, Japan, 650-0017 Chuo-ku, Tokyo, Japan, 104-0045 Collapse << |
| NCT00661375 | Carcinoma, Renal Cell | Phase 2 | Completed | - | - |
| NCT00692770 | - | - | Completed | - | - |
| NCT00712790 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Completed | - | Korea, Republic of ... more >> Seoul National University Bundang Hospital Seoul, Korea, Republic of Malaysia Selayang Hospital Selangor, Malaysia Myanmar Yangon GI & Liver Centre Yangon, Myanmar Singapore Singapore General Hospital Singapore, Singapore, 169608 National Cancer Centre Singapore, Singapore, 169610 Collapse << |
| NCT00771147 | - | - | Completed | - | Belgium ... more >> Many Locations, Belgium Collapse << |
| NCT00754923 | Lung Cancer | Phase 2 | Terminated | - | United States, Ohio ... more >> Ohio State University Columbus, Ohio, United States, 43210-1240 Collapse << |
| NCT00780169 | Metastatic Colorectal Cancer | Phase 1 | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT00727532 | Hereditary Clear Cell Renal Ce... more >>ll Carcinoma Kidney Cancer Collapse << | Not Applicable | Terminated(Low Accrual) | June 2015 | United States, Illinois ... more >> Robert H. Lurie Comprehensive Cancer Center at Northwestern University Chicago, Illinois, United States, 60611-3013 Collapse << |
| NCT00754923 | - | - | Terminated | - | - |
| NCT00768937 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Completed | - | Austria ... more >> Abteilung Gastroenterologie und Hepatologie, Medizinische Universität und AKH Wien Vienna, Austria, 1090 Collapse << |
| NCT00785226 | Advanced Cancer | Phase 1 Phase 2 | Completed | - | United States, California ... more >> USC/Norris Comprehensive Cancer Center and LAC/USC Medical Center Los Angeles, California, United States, 90033 United States, Nevada Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89169 United States, New York New York Oncology Hematology, PC Albany, New York, United States, 12206 Roswell Park Cancer Institute Buffalo, New York, United States, 14263 James P. Wilmot Cancer Center Rochester, New York, United States, 14642 United States, Pennsylvania UPenn Philadelphia, Pennsylvania, United States, 19104 United States, South Carolina Greenville Hospital System University Medical Center, (ITOR) Greenville, South Carolina, United States, 29605 United States, Texas Texas Oncology - Baylor Charles A. Simmons Cancer Center Dallas, Texas, United States, 75246 Texas Oncology-Tyler Tyler, Texas, United States, 75702 Canada, Ontario Toronto General Hospital Toronto, Ontario, Canada, M5G 2N2 Collapse << |
| NCT00791778 | Ovarian Neoplasms | Phase 2 | Completed | - | - |
| NCT00794235 | Melanoma Stage III or IV ... more >> No Prior Chemotherapy Collapse << | Phase 2 | Completed | - | Switzerland ... more >> Department of Dermatology, University Hospital Zurich Zurich, Switzerland, 8091 Collapse << |
| NCT00772694 | Testicular Cancer | Phase 2 | Unknown | December 2011 | Poland ... more >> Chemotherapy Unit, Dept of Urology, Instituite of Oncology Recruiting Warsaw, Poland, 02781 Principal Investigator: Iwona A Skoneczna, MD Sub-Investigator: Agnieszka Chaladaj-Kujawska, MD Collapse << |
| NCT00810394 | - | - | Completed | - | - |
| NCT00827177 | Advanced Solid Tumors | Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02111 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 United States, Tennessee Nashville, Tennessee, United States, 37232 Italy Rozzano, Italy, 20089 Collapse << |
| NCT00812175 | - | - | Completed | - | - |
| NCT00829231 | Liver Disease | Phase 1 | Completed | - | United States, Florida ... more >> Miami, Florida, United States, 33014 Orlando, Florida, United States, 32809 United States, Tennessee Knoxville, Tennessee, United States, 37920 Collapse << |
| NCT00791778 | - | - | Completed | - | - |
| NCT00810394 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California Davis Cancer Center Sacramento, California, United States, 95817 Collapse << |
| NCT00801606 | Tuberculosis | Phase 3 | Completed | - | India ... more >> Department of Pediatrics, All India Institute of Medical Sciences New Delhi, Delhi, India, 110029 Collapse << |
| NCT00837148 | Sarcoma Synov... more >>ial Sarcoma Leiomyosarcoma Malignant Peripheral Nerve Sheath Tumor Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00813293 | Hepatocellular Cancer | Phase 2 | Active, not recruiting | December 2017 | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00846131 | Hepatocellular Carcinoma | Phase 1 | Completed | - | United States, Illinois ... more >> Northwestern Memorial Hospital Chicago, Illinois, United States, 60611 Collapse << |
| NCT00844168 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Localized Resectable Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Collapse << | Phase 1 | Completed | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT00848640 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Completed | - | Egypt ... more >> Egyptian Foundation For Cancer Research Cairo, Egypt, 11553 Collapse << |
| NCT00854620 | - | - | Completed | - | - |
| NCT00854620 | Carcinoma, Renal Cell ... more >> Kidney Disease Kidney Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT00882869 | Advanced Hepatocellular Carcin... more >>oma Collapse << | Phase 1 Phase 2 | Completed | - | China, New Territories, Hong K... more >>ong Tuen Mun Hospital Tuen Mun, New Territories, Hong Kong, China China Queen Elizabeth Hospital Hong Kong, China Queen Mary Hospital Hong Kong, China Collapse << |
| NCT00863746 | Carcinoma Non... more >>-Small-Cell Lung Collapse << | Phase 3 | Completed | - | - |
| NCT00887107 | Thyroid Cancer | Phase 2 | Completed | - | Netherlands ... more >> Leiden University Medical Center Leiden, Netherlands, 2300 RC Collapse << |
| NCT00889057 | Osteosarcoma | Phase 2 | Completed | - | Italy ... more >> I.R.C.C. - Unit of Medical Oncology Candiolo, Torino, Italy, 10060 Istituti Ortopedici Rizzoli - Unit of Chemotherapy of Muscoloskeletal Tumors Bologna, Italy, 40136 Istituto Nazionale Tumori - Unit of Medical Oncology Milano, Italy, 20133 Ospedale Infantile Regina Margherita - Unit of Paediatric Oncoematology Torino, Italy, 10126 Collapse << |
| NCT00892424 | Liver Metastasis ... more >> Cancer Collapse << | Phase 1 Phase 2 | Active, not recruiting | November 2019 | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00893373 | Acute Myeloid Leukemia (AML) | Phase 2 | Completed | - | Germany ... more >> Sozialstiftung Bamberg Klinikum am Bruderwald Bamberg, Germany, 96049 Klinikum Bayreuth Bayreuth, Germany, 95445 Charite Campus Benjamin Franklin Berlin, Germany Ev. Diakonie-Krankenhaus gGmbH Bremen Bremen, Germany, 28239 University Hospital Dresden Dresden, Germany, 01307 Klinikum Frankfurt (Oder) GmbH Frankfurt (Oder), Germany, 15236 Westpfalz-Klinikum GmbH Kaiserslautern, Germany, 67655 Agaplesion Diakoniekrankenhaus Rotenburg Rotenburg, Germany, 27356 Robert-Bosch-Krankenhaus Stuttgart, Germany, 70376 Collapse << |
| NCT00892658 | Hepatocellular Carcinoma | Phase 1 | Active, not recruiting | December 2019 | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00954278 | Carcinoma, Non Small Cell Lung | Phase 1 | Active, not recruiting | December 2018 | United States, Wisconsin ... more >> University of Wisconsin - Madison Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00917462 | Esophageal Cancer ... more >> Gastroesophageal Junction Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01616654 | Acne Vulgaris | Phase 2 | Completed | - | - |
| NCT00984282 | Thyroid Neoplasms | Phase 3 | Completed | - | - |
| NCT00997022 | Hepatocellular Cancer | Phase 1 | Completed | - | United States, California ... more >> University of Southern California Los Angeles, California, United States, 90089 University of California, Los Angeles Los Angeles, California, United States, 90095 United States, New York Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00943449 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Germany ... more >> ONKOPLUS Beratung und Hilfe für Menschen mit Krebs Berlin, Germany, 14195 Universitätsklinikum Essen (Innere Klinik/Tumorforschung und Zentrum für Innere Medizin) Essen, Germany, 45147 Medizinische Fakultät der Martin-Luther-Universität Halle-Wittenberg (Klinik und Poliklinik für Innere Medizin I) Halle, Germany, 06120 Universitätsklinikum Hamburg-Eppendorf (I. Medizinische Klinik und Poliklinik) Hamburg, Germany, 20246 Universitätsklinikum Heidelberg (Medizinische Universitätsklinik/Innere Medizin IV/Nationales Centrum für Tumorerkrankungen) Heidelberg, Germany, 69120 Johannes Gutenberg-Universität Mainz (I. Medizinische Klinik und Poliklinik) Mainz, Germany, 55131 Klinikum rechts der Isar der Technischen Universität München (Medizinische Klinik und Poliklinik II/Gastroenterologie) Munich, Germany, 81675 Universitätsklinikum Tübingen (Medizinische Klinik/Innere Medizin I) Tübingen, Germany, 72076 Italy l'Azienda Ospedaliero-Universitaria di Bologna-Policlinico S. Orsola Malpighi Bologna, Italy, 40138 l'Università degli Studi di Genova (Dipartimento di Medicina Interna e Specialità Mediche) Genova, Italy, 16132 Istituto Europea di Oncologia EIO Milano, Italy, 20141 A.O.R.N. Monaldi-Cotugno-CTO Napoli, Italy, 80131 l'Azienda Ospedaliera di Padova and l'Istituto Oncologico Veneto IOV Padova, Italy l'Ente "Azienda Ospedaliera Universitaria Policlinico Paolo Giaccone" Palermo, Italy, 12990127 L'Istituto Clinico Humanitas-Humanitas Mirasole S.p.A. Rozzano-Milano, Italy, 20089 Collapse << |
| NCT01005875 | Hepatocellular Carcinoma ... more >> Liver Cancer Collapse << | Not Applicable | Terminated(Sponsor(Bayer)did n... more >>ot wish to continue with study due to slow accrual. Therefore, there is insufficient data and will not be any study results/outcomes.) Collapse << | - | United States, Alabama ... more >> UAB Birmingham, Alabama, United States, 35294 Collapse << |
| NCT00984282 | - | - | Completed | - | - |
| NCT01005875 | - | - | Terminated(Sponsor(Bayer)did n... more >>ot wish to continue with study due to slow accrual. Therefore, there is insufficient data and will not be any study results/outcomes.) Collapse << | - | - |
| NCT01029418 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Terminated(The phase II portio... more >>n was not conducted due to funding issue.) Collapse << | - | Singapore ... more >> National University Hospital Singapore Singapore, Singapore, 119074 National Cancer Centre Singapore Singapore, Singapore, 169610 Collapse << |
| NCT00837148 | - | - | Completed | - | - |
| NCT00863746 | - | - | Completed | - | - |
| NCT00999843 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Unknown | October 2012 | China, Shanghai ... more >> Fudan University Shanghai Cancer Center Recruiting Shanghai, Shanghai, China, 200032 Collapse << |
| NCT01030783 | Advanced Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT01016483 | Pancreatic Adenocarcinoma | Phase 1 Phase 2 | Completed | - | United States, Massachusetts ... more >> For Recruiting Locations in the United States, please Contact U.S. Medical Information Rockland, Massachusetts, United States Germany For Recruiting Locations outside the United States, Please contact the Merck KGaA Communication Center Darmstadt, Germany Collapse << |
| NCT01076010 | Advanced Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT01098760 | Hepatocellular Carcinoma | Phase 4 | Completed | - | Taiwan ... more >> Chia Yi, Taiwan, 613 Hualien county, Taiwan, 970 Kaohsiung City, Taiwan, 807 Kaohsiung City, Taiwan, 8330 Taichung City, Taiwan Taipei City,, Taiwan Taoyuan, Taiwan, 33305 Collapse << |
| NCT01033240 | Advanced Hepatocellular Carcin... more >>oma Liver Cancer Hepatic Cancer Liver Neoplasms Collapse << | Phase 2 | Completed | - | United States, California ... more >> Kenmar Research Group Los Angeles, California, United States, 90057 United States, District of Columbia Georgetown-Lombardi Cancer Center Washington DC, District of Columbia, United States, 20007 United States, New York The Mount Sinai Medical Center New York, New York, United States, 10029 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 Japan Kurume University Hospital Kurume-shi, Fukuoka, Japan, 830-0011 Hiroshima University Hiroshima-city, Hiroshima, Japan Kanazawa University Kanazawa, Ishikawa, Japan Kinki University Hospital Osaka, Osaka-sayama, Japan, 589-8511 Osaka Med Center Cancer and Cardiovascular Disease Higashinari-ku, Osaka, Japan, 537-8511 Yamaguchi University Hospital Ube, Yamaguchi, Japan Chiba University Hospital Chiba, Japan, 260-8677 Okayama University Hospital Okayama, Japan, 700-8558 Musashino Red-Cross Hospital Tokyo, Japan, 180-8610 Korea, Republic of Samsung Medical Center Gangnam-gu, Seoul, Korea, Republic of, 135-710 Keimyung University Dongsan Hospital Daegu, Korea, Republic of, 700-712 Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Severance Hospital Seoul, Korea, Republic of, 120-752 Korea University Anam Hospital Seoul, Korea, Republic of, 136-705 Catholic Univ. of Korea, Seoul St. Mary's Hospital Seoul, Korea, Republic of, 137-701 Asan Medical Center Seoul, Korea, Republic of, 138-736 Taiwan Chang Gung Memorial Hospital Putz, Chiayi, Taiwan Kaohiung Chang Gung Hospital Kaohsiung, Niaosung Hsiang, Taiwan Chi-Mei Medical Center Liouying, Tainan County, Taiwan, 736 Changhua Christian Hospital Changhua, Taiwan, 500 Kaohslung Medical University Hospital Kaohsiung, Taiwan, 807 National Cheng-Kung University Hospital Tainan city, Taiwan, 704 National Taiwan University Hospital Taipei, Taiwan Chang Gung Medical Foundation-Linkuo Taoyuan, Taiwan, 33305 Collapse << |
| NCT01126645 | Hepatocellular Carcinoma | Phase 2 | Unknown | March 2018 | Germany ... more >> University of Magdeburg Magdeburg, Germany, 39120 Collapse << |
| NCT01041482 | Renal Cell Carcinoma | Phase 2 | Unknown | November 2013 | China, Beijing ... more >> Jianhui Ma Recruiting Beijing, Beijing, China, 100730 Contact: Jianhui Ma Jianhui Ma Jianhui Ma, Doctor 01086108878 Collapse << |
| NCT01016483 | - | - | Completed | - | - |
| NCT01377025 | Uveal Melanoma | Phase 2 | Unknown | June 2017 | Germany ... more >> Universitätsmedizin Berlin, Charité Campus Benjamin Franklin Berlin, Germany, 12203 Universitätsklinikum Erlangen Erlangen, Germany, 91052 Univesitätsklinikum Essen Essen, Germany, 45147 Collapse << |
| NCT01398501 | Acute Myeloid Leukemia | Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02214 Collapse << |
| NCT01128998 | Advanced Solid Tumors | Phase 1 | Completed | - | Taiwan ... more >> National Cheng-Kung University Hospital Tainan City, Taiwan, 704 Collapse << |
| NCT01405573 | Advanced Adult Hepatocellular ... more >>Carcinoma Collapse << | Phase 3 | Terminated(lack of enrolment) | - | Italy ... more >> Azienda Ospedaliera G. Rummo Benevento, BN, Italy Policlinico Giaccone Palermo, PA, Italy, 90127 S. Orsola-Malpighi Bologna, Italy Ospedale Ramazzini di Carpi Carpi, Italy Osp. Civile Infermi Faenza, Italy Azienda Ospedaliera Careggi Firenze, Italy IRCCS-Azienda Ospedaliera Universitaria San Martino-IST Genova, Italy AO C. Poma Mantova, Italy Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori Meldola, Italy A.O.U. G.Martino- Policlinico Universita di Messina Messina, Italy Istituto Nazionale Tumori Milano, Italy A.O.U. Federico II Napoli, Italy AOU II Università di Napoli Napoli, Italy Istituto Nazionale dei Tumori Napoli, Italy Ospedale Cardarelli Napoli, Italy Azienda Ospedaliera Universitaria di Padova Padova, Italy Istituto Oncologico Veneto Padova, Italy A.O. Ospedali Riuniti Villa Sofia-Cervello Palermo, Italy Azienda Ospedaliero Universitaria di Parma Parma, Italy Ospedale Guglielmo da Saliceto Piacenza, Italy, Italy AO S. Carlo Potenza, Italy Policlinico Universitario Tor Vergata Roma, Italy Oncologia IRCCS - Casa Sollilevo Sofferenza S. Giovanni Rotondo, Italy Azienda Ospedaliera Universitaria Arcispedal, U.O. di Oncologia Clinica Sant'Anna di Ferrara, Italy A.O. Trevigilio - Caravaggio Trevigilio, Italy Collapse << |
| NCT01409499 | Hepatectomy H... more >>epatocellular Carcinoma Sorafenib Collapse << | Phase 4 | Completed | - | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Guangzhou, Guangdong, China, 510060 Collapse << |
| NCT01411423 | - | - | Completed | - | Japan ... more >> Multiple Locations, Japan Collapse << |
| NCT01411436 | - | - | Completed | - | Japan ... more >> Many locations, Japan Collapse << |
| NCT01412671 | - | - | Completed | - | Japan ... more >> Multiple Locations, Japan Collapse << |
| NCT02160665 | Plaque Psoriasis | Phase 3 | Completed | - | United States, Illinois ... more >> Altman Dermatology Associates Arlington Heights, Illinois, United States, 60005 Collapse << |
| NCT01425216 | Keloids | Phase 2 | Terminated(IND application was... more >> not approved by the US FDA - Project was never started) Collapse << | - | United States, New York ... more >> Michael H. Tirgan, MD New York, New York, United States, 10023 Collapse << |
| NCT01470495 | HCC Radiofreq... more >>uency Ablation Sorafenib Collapse << | Not Applicable | Recruiting | December 2018 | China, Chongqing ... more >> Institute of hepatobiliary surgery,southwest hospital Recruiting Chongqing, Chongqing, China, 400038 Contact: feng xiaobin, MD +86-23-13228683243 fengxiaobin200708@yahoo.com.cn Principal Investigator: feng xiaobin, MD Collapse << |
| NCT02160678 | Acne Vulgaris | Phase 3 | Completed | - | United States, Illinois ... more >> Altman Dermatology Associates Arlington Hts., Illinois, United States, 60005 Collapse << |
| NCT02235480 | Nail Psoriasis | Phase 2 | Completed | - | Germany ... more >> Investigational site 3 Berlin, Germany Investigational site 4 Berlin, Germany Investigational 2 Hamburg, Germany Investigational site 1 Hamburg, Germany Collapse << |
| NCT01480817 | Hepatocellular Carcinoma | Phase 2 | Terminated(Difficulty of parti... more >>cipant enrollment.) Collapse << | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT01444807 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | September 2019 | Italy ... more >> Istituto Tumori Milano Recruiting Milan, Mi, Italy, 20156 Contact: Giuseppe Procopio, MD +390223902557 Giuseppe.Procopio@istitutotumori.mi.it Contact: Elena Verzoni, MD +390223902557 elena.verzoni@istitutotumori.mi.it Principal Investigator: Giuseppe Procopio, MD Cinzia Ortega Recruiting Alba, Italy Alessandra Bearz Recruiting Aviano, Italy Alfredo Berruti Recruiting Brescia, Italy Saverio Cinieri Recruiting Brindisi, Italy Francesco Atzori Recruiting Cagliari, Italy Rodolfo Passalaqua Recruiting Cremona, Italy Francesco Di Costanzo Recruiting Firenze, Italy Vincenzo Emanuele Chiuri Recruiting Lecce, Italy Alessandra Mosca Recruiting Novara, Italy Vittorio Gebbia Recruiting Palermo, Italy Enrico Cortesi Recruiting Roma, Italy Francesco Cognetti Recruiting Roma, Italy Contact: Michele Milella Franco Morelli Recruiting San Giovanni Rotondo, Italy Collapse << |
| NCT01482442 | Liver Carcinoma | Phase 3 | Completed | - | France ... more >> CHU Amiens #2 Amiens, France, 80054 CHU Angers #3 Angers, France, 49933 CHRU Besançon Hôpital Jean Minjoz #21 Besançon, France, 25030 Hôpital Jean Verdier #25 Bondy, France, 93140 Hôpital Côte de Nacre #4 Caen, France, 14033 Hôpital Antoine Béclère #29 Clamart, France, 92140 Hopital beaujon #1 Clichy, France, 92110 Henri Mondor #24 Créteil, France, 94000 CHU Dijon Hôpital Bocage #22 Dijon, France, 21000 CHU Grenoble #5 Grenoble, France, 38043 Hôpital Edouard Herriot #6 Lyon, France, 69003 Lyon La croix Rousse #27 Lyon, France, 69004 Institut Paoli Calmettes #7 Marseille, France, 13009 CHU Marseille Hôpital La Timone #23 Marseille, France, 13385 Hôpital Saint Eloi #8 Montpellier, France, 34295 Hôpital de Brabois #9 Nancy, France, 54511 Hotel Dieu #10 Nantes, France, 44093 Hôpital de L'Archet #11 Nice, France, 06002 Hôpital Européen Georges Pompidou #13 Paris, France, 75908 Hôpital Haut Leveque #14 Pessac, France, 33604 CHU Poitiers La Milétrie Poitiers, France, 86021 CHU Robert Debré #28 Reims, France, 51100 CHU Saint Etienne Hôpital Nord #17 Saint-Priest en Jarez, France, 42270 Hôpital de Hautepierre #18 Strasbourg, France, 67098 Paul Brousse #19 Villejuif, France, 94275 Institut Gustave Roussy #20 Villejuif, France, 94805 Collapse << |
| NCT02218034 | Acne Vulgaris | Phase 1 | Completed | - | United States, Texas ... more >> College Station, Texas, United States Collapse << |
| NCT02267746 | Acne Vulgaris | Phase 3 | Completed | - | United States, California ... more >> Encino, California, United States, 91436 Center for Dermatology Clinical Research Fremont, California, United States, 94538 United States, Florida Moore Clinical Research Brandon, Florida, United States, 33511 LCC Medical Research Institute Miami, Florida, United States, 33126 International Dermatology Research Miami, Florida, United States, 33144 FXM Research Corp Miami, Florida, United States, 33175 FXM Research Miramar Miramar, Florida, United States, 33027 Moore Clinical Research Tampa, Florida, United States, 33609 Moore Clinical Research Tampa, Florida, United States, 33618 Atlantic Clinical Research Collaborative Wellington, Florida, United States, 33414 United States, Rhode Island Omega Medical Research Warwick, Rhode Island, United States, 02886 United States, Texas Research Across America Dallas, Texas, United States, 75234 Research Across America Plano, Texas, United States, 75093 United States, Utah Dermatology Research Center Salt Lake City, Utah, United States, 84117 Belize FXM International Belize City, Belize Collapse << |
| NCT01605734 | PHENYTOIN/SORAFENIB [VA Drug I... more >>nteraction] Carcinoma, Hepatocellular Collapse << | Phase 2 | Unknown | December 2015 | China, Shandong ... more >> Shandong Medical Imaging Research Institute Not yet recruiting Jinan, Shandong, China, 250021 Contact: Jun Tang, MD +8613964031909 tangjun7925032@163.com Principal Investigator: Jun Tang, MD Qilu Hospital of Shandong University Not yet recruiting Jinan, Shandong, China, 250114 Contact: Caixia Li, MD +8613505312972 Lilyshanda88@hotmail.com Principal Investigator: Caixia Li, MD Shandong Cancer Hospital and Institute Jinan, Shandong, China, 250117 the Affiliated Hospital of Medical College Qingdao University Not yet recruiting Qingdao, Shandong, China, 266003 Contact: Zixiang Li, MD +8618669739263 zixiangli@yahoo.com.cn Principal Investigator: Zixiang Li, MD Yantai Yuhuangding Hospital Not yet recruiting Yantai, Shandong, China, 264000 Contact: Yanbo Zheng, MD +8613805356068 13805356068@163.com Principal Investigator: Yanbo Zheng Collapse << |
| NCT01761266 | Hepatocellular Carcinoma (HCC) | Phase 3 | Active, not recruiting | March 2019 | - |
| NCT00712855 | Hepatocellular Carcinoma | Phase 1 | Completed | - | United States, California ... more >> Cedars-Sinai Health System Los Angeles, California, United States, 90048 United States, Colorado University of Colorado Cancer Center Aurora, Colorado, United States, 80045 United States, Florida University of Florida Gainesville, Florida, United States, 32610 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Pennsylvania University of Pennsylvania- Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00680732 | Malaria Malar... more >>ia in Pregnancy Collapse << | Phase 4 | Completed | - | Burkina Faso ... more >> Centre Muraz Bobo-Dioulasso, Burkina Faso, BP 390 Collapse << |
| NCT01761266 | - | - | Active, not recruiting | - | - |
| NCT01844947 | Urothelial Carcinoma ... more >> Bladder Cancer Renal Pelvis Cancer Ureter Cancer Urethra Cancer Collapse << | Phase 1 | Completed | - | Denmark ... more >> Department of Oncology, Aarhus University Hospital Aarhus, Denmark, DK-8200 Department of Oncology, Rigshospitalet Copenhagen, Denmark, DK-2100 Sweden Department of Oncology, Karolinska University Hospital Stockholm, Sweden, SE-171 76 Collapse << |
| NCT01640665 | Metastatic Breast Cancer ... more >> Gastrointestinal Tumors Collapse << | Phase 1 | Completed | - | United States, Connecticut ... more >> Yale Cancer Center New Haven, Connecticut, United States, 06519 Collapse << |
| NCT01498952 | Unresectable or Metastatic Hep... more >>atocellular Carcinoma (HCC) Collapse << | Phase 1 | Completed | - | United States, California ... more >> Research Site Oxnard, California, United States United States, Colorado Research Site Golden Springs, Colorado, United States United States, Nevada Research Site Las Vegas, Nevada, United States Collapse << |
| NCT00767468 | Liver Cancer | Phase 1 | Terminated(Funding unavailable... more >>) Collapse << | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Duke Comprehensive Cancer Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00825734 | Metastatic Breast Cancer | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Ft. Myers, Florida, United States, 33916 United States, Indiana Providence Medical Group Terre Haute, Indiana, United States, 47802 RHHP/ Hope Cancer Center Terre Haute, Indiana, United States, 47802 United States, Kentucky Baptist Hospital East Louisville, Kentucky, United States, 40207 United States, Louisiana Hematology Oncology Clinic, LLP Baton Rouge, Louisiana, United States, 70806 United States, Maine Mercy Hospital Portland, Maine, United States, 04101 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 National Capital Clinical Research Consortium Bethesda, Maryland, United States, 20817 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63044 United States, New Hampshire Portsmouth Regional Hospital Portsmouth, New Hampshire, United States, 03801 United States, New Jersey Hematology-Oncology Associates of Northern NJ Morristown, New Jersey, United States, 07962 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, South Carolina South Carolina Oncology Associates Columbia, South Carolina, United States, 29210 Spartanburg Regional Medical Center Spartanburg, South Carolina, United States United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00825734 | - | - | Completed | - | - |
| NCT01903694 | Child-Pugh A Hepatocellular Ca... more >>rcinoma Collapse << | Phase 3 | Completed | - | France ... more >> CHU de Dijon Dijon, France, 21079 Collapse << |
| NCT01634893 | Refractory or Relapsed Solid T... more >>umors Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Cancer Therapy and Research Center at UTHSCSA San Antonio, Texas, United States, 78229 Collapse << |
| NCT00922584 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | China, Guangdong ... more >> WuYiLong Guangzhou, Guangdong, China, 510080 Collapse << |
| NCT02129322 | Hepatocellular Carcinoma | Phase 2 | Not yet recruiting | December 2019 | China ... more >> First affiliated hospital, Zhejiang University Not yet recruiting Hangzhou, China Contact: Weijiafang fang Principal Investigator: Weijia fang, MD Collapse << |
| NCT02143401 | Cirrhosis Hep... more >>atitis B Infection Hepatitis C Infection Metastatic Malignant Solid Neoplasm Recurrent Hepatocellular Carcinoma Recurrent Malignant Solid Neoplasm Refractory Malignant Neoplasm Stage IV Hepatocellular Carcinoma AJCC v7 Unresectable Solid Neoplasm Collapse << | Phase 1 | Recruiting | - | United States, Arizona ... more >> Mayo Clinic in Arizona Recruiting Scottsdale, Arizona, United States, 85259 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, California University of California Davis Comprehensive Cancer Center Recruiting Sacramento, California, United States, 95817 Contact: Site Public Contact 916-734-3089 Principal Investigator: Edward J. Kim United States, Colorado University of Colorado Hospital Recruiting Aurora, Colorado, United States, 80045 Contact: Site Public Contact 720-848-0650 Principal Investigator: Sarah L. Davis United States, Florida Mayo Clinic in Florida Recruiting Jacksonville, Florida, United States, 32224-9980 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, Iowa University of Iowa/Holden Comprehensive Cancer Center Recruiting Iowa City, Iowa, United States, 52242 Contact: Site Public Contact 800-237-1225 Principal Investigator: Mohammed M. Milhem United States, Maryland University of Maryland/Greenebaum Cancer Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Site Public Contact 800-888-8823 Principal Investigator: Edward A. Sausville United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Site Public Contact 855-776-0015 Principal Investigator: Brian A. Costello United States, New York Columbia University/Herbert Irving Cancer Center Recruiting New York, New York, United States, 10032 Contact: Site Public Contact 212-305-6361 nr2616@cumc.columbia.edu Principal Investigator: Gulam A. Manji United States, Pennsylvania Thomas Jefferson University Hospital Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Site Public Contact 215-955-6084 Principal Investigator: James A. Posey United States, Texas M D Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Site Public Contact 877-312-3961 Principal Investigator: Ahmed O. Kaseb Collapse << |
| NCT01128322 | Hypertension | Phase 2 | Completed | - | Korea, Republic of ... more >> Hallym University Sacred Heart Hospital Anyang, Korea, Republic of Soonchunhyang University Hospital in Bucheon Bucheon, Korea, Republic of Kosin University Gospel Hospital Busan, Korea, Republic of Chungbuk National Uiversity Hospital Cheongju, Korea, Republic of Daegu Catholic University Medical Center Daegu, Korea, Republic of Keimyung University Dongsan Medical Center Daegu, Korea, Republic of DonGuk University International Hospital Goyang, Korea, Republic of National Health Insurance Corporation Ilsan Hospital Goyang, Korea, Republic of Chonnam National University Hospital Gwangju, Korea, Republic of Gachon University Gil Medical Center Inchon, Korea, Republic of Chonbuk National University Hospital Jeonju, Korea, Republic of Pusan National University Hospital Pusan, Korea, Republic of Seoul National University Bundang Hospital Seongnam, Korea, Republic of Ewha Womans University Hospital Seoul, Korea, Republic of Gangnam Severance Hospital Seoul, Korea, Republic of Kandong Sacred Heart Hospital Seoul, Korea, Republic of Korea University Guro Hospital Seoul, Korea, Republic of Kyung Hee University Medical Center Seoul, Korea, Republic of Seoul St. Mary's Hospital, The catholic University of Korea Seoul, Korea, Republic of Severance Hospital Seoul, Korea, Republic of SMG-SNU Boramae Medical Center Seoul, Korea, Republic of Soon Chun Hyang University Hospital Seoul, Korea, Republic of St. Paul's Hospital, The catholic University of Korea Seoul, Korea, Republic of Ajou University Hospital Suwon, Korea, Republic of Uijeongbu St. Mary's Hospital, The catholic University of Korea Uijeongbu, Korea, Republic of Wonju Christian Hospital Wonju, Korea, Republic of Collapse << |
| NCT02196857 | Leukemia | Phase 2 | Active, not recruiting | February 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02292173 | Hepatocellular Cancer ... more >> Liver Cancer Collapse << | Phase 1 | Active, not recruiting | April 2020 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT02406508 | Hepatocellular Carcinoma (HCC) | Phase 2 | Withdrawn(No enrollment in the... more >> study) Collapse << | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute at University of South Florida Tampa, Florida, United States, 33612 United States, New York Montefiore Medical Center New York, New York, United States, 10467 Collapse << |
| NCT01264705 | Hepatocellular Carcinoma ... more >> Liver Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> UT Southwestern Medical Center Dallas, Texas, United States, 75390 Collapse << |
| NCT02436902 | Hepatocellular Carcinoma | Phase 3 | Recruiting | August 2019 | China, Guangxi ... more >> Department of Hepatobilliary Surgery, Affiliated Tumor of Guangxi University Recruiting Nanning, Guangxi, China, 530021 Contact: Bang-De Xiang, MD,PhD 86-771-5330855 xiangbangde@163.com Collapse << |
| NCT02150317 | Advanced Hepatocellular Carcin... more >>oma Collapse << | Not Applicable | Unknown | December 2016 | China, Shanghai ... more >> Eastern Hepatobiliary Surgery Hospital Recruiting Shanghai, Shanghai, China, 200438 Contact: Ai-jun Li, M.D. 0086-021-81875531 yinlei409@hotmail.com Contact: Lei Yin, M.D. 0086-021-81875532 dermatology2008@126.com Collapse << |
| NCT01303341 | Advanced Malignant Solid Neopl... more >>asm Recurrent Melanoma Refractory Malignant Solid Neoplasm Stage III Cutaneous Melanoma AJCC v7 Stage IIIA Cutaneous Melanoma AJCC v7 Stage IIIB Cutaneous Melanoma AJCC v7 Stage IIIC Cutaneous Melanoma AJCC v7 Stage IV Cutaneous Melanoma AJCC v6 and v7 Collapse << | Phase 1 | Active, not recruiting | - | United States, New Jersey ... more >> Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 Collapse << |
| NCT01306058 | Hepatoma Live... more >>r Neoplasms Adenoma, Liver Cell Carcinoma, Hepatocellular Liver Neoplasms, Experimental Collapse << | Phase 1 Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02330783 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Recruiting | December 2018 | China, Guangdong ... more >> Sun Yat-sen university cancer center Not yet recruiting Guangzhou, Guangdong, China, 510060 Contact: Fangjian Zhou, MD 0086-20-87343088 zhoufj@sysucc.org.cn Principal Investigator: Fangjian Zhou, MD China Peking University First Hospital Recruiting Beijing, China, 100034 Contact: Zhisong He, MD 0086-10-83572211 wyj7074@sohu.com Principal Investigator: Zhisong He, MD Beijing Cancer Hospital Recruiting Beijing, China, 100142 Contact: Xinan Sheng, MD 0086-10-88196951 doctor_sheng@126.com Principal Investigator: Jun Guo, MD,PHD Sub-Investigator: Xinan Sheng, MD Cancer Hospital, Fudan University Not yet recruiting Shanghai, China, 200032 Contact: Dingwei Ye, MD 0086-21-64175590 dwye@163.com Principal Investigator: Dingwei Ye, MD Collapse << |
| NCT02529761 | Hepatocellular Carcinoma | Not Applicable | Unknown | October 2018 | China, Shaanxi ... more >> Xijing Hospital of digestive disease, Fourth Military Medical University Recruiting Xi'an, Shaanxi, China, 710032 Contact: Han hangh@fmmu.edu.cn Principal Investigator: Guohong Han, MD,Ph.D Collapse << |
| NCT01306058 | - | - | Completed | - | - |
| NCT01356043 | Healthy Male Volunteers | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01370343 | Healthy Drug ... more >>Interaction Collapse << | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT02672488 | Hepatocellular Carcinoma | Phase 2 | Unknown | December 2018 | China ... more >> Tianjin Medical University Cancer Institute and Hospital Recruiting Tianjin, China, 300060 Contact: TI ZHANG, MD +862223340123 ext 3092 zhangti@tjmuch.com Contact: Huikai Li, MD +862223340123 ext 3091 lihuikai@tjmuch.com Collapse << |
| NCT02908165 | Hepatocellular Carcinoma | Phase 2 | Withdrawn(A future study will ... more >>be taking place using the same patient population.) Collapse << | June 2020 | United States, Connecticut ... more >> Smilow Cancer Center New Haven, Connecticut, United States, 06510 Collapse << |
| NCT01258608 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | - |
| NCT02748304 | Hepatocellular Carcinoma | Not Applicable | Recruiting | June 2021 | China ... more >> Huashan hospital Recruiting Shanghai, China, 200040 Contact: Lu Lu, MD 862152887175 lulu@huashan.org.cn Collapse << |
| NCT02961998 | Hepatocellular Carcinoma | Phase 4 | Completed | - | - |
| NCT01501253 | Hypertension | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul St. Mary's hospital, The catholic university of Korea Seoul, Korea, Republic of Collapse << |
| NCT03037437 | Hepatocellular Cancer | Phase 2 | Recruiting | December 2018 | United States, Texas ... more >> University of Texas Health Cancer Center Recruiting San Antonio, Texas, United States, 78229 Contact: Epp Goodwin 210-450-5798 goodwine@uthscsa.edu Collapse << |
| NCT01634295 | Hypertension | Phase 3 | Completed | - | Korea, Republic of ... more >> Seoul national university Bundang Hospital Bundang-gu, Gyeonggi-do, Korea, Republic of, 463-707 Collapse << |
| NCT03211416 | Advanced Adult Hepatocellular ... more >>Carcinoma Child-Pugh Class A Stage III Hepatocellular Carcinoma Stage IIIA Hepatocellular Carcinoma Stage IIIB Hepatocellular Carcinoma Stage IIIC Hepatocellular Carcinoma Stage IV Hepatocellular Carcinoma Stage IVA Hepatocellular Carcinoma Stage IVB Hepatocellular Carcinoma Collapse << | Phase 1 Phase 2 | Recruiting | October 11, 2019 | United States, New York ... more >> Roswell Park Cancer Institute Recruiting Buffalo, New York, United States, 14263 Contact: Roswell Park 877-275-7724 ASKRPCI@roswellpark.org Principal Investigator: Renuka V. Iyer Collapse << |
| NCT02624700 | Breast Cancer ... more >> Metastatic Breast Cancer Recurrent Breast Cancer Collapse << | Phase 2 | Recruiting | April 1, 2025 | United States, Virginia ... more >> Virginia Commonwealth University/Massey Cancer Center Recruiting Richmond, Virginia, United States, 23298 Contact: Alison Ryan, NP 804-628-1936 masseysiit@vcu.edu Contact: Andrew S Poklepovic, MD 804-628-2321 andrew.poklepovic@vcuhealth.org Principal Investigator: Andrew S Poklepovic, MD Collapse << |
| NCT01983735 | Hypertension | Phase 3 | Unknown | August 2014 | Korea, Republic of ... more >> The catholic university of Korea Seoul St. Mary's hospital Not yet recruiting Seoul, Korea, Republic of Contact: Ho-Joong Yoon, Ph.D 82-2-2258-6029 younhj@catholic.ac.kr Principal Investigator: Ho-Joong Yoon, Ph.D Collapse << |
| NCT01897038 | Carcinoma, Hepatocellular | Phase 1 | Completed | - | United States, Florida ... more >> Sarasota, Florida, United States, 34232 United States, Maryland Baltimore, Maryland, United States, 21231 United States, New York New York, New York, United States, 10065 United States, Tennessee Nashville, Tennessee, United States, 37203 United States, Texas Houston, Texas, United States, 77030 Hong Kong Pokfulam, Hong Kong Singapore Singapore, Singapore, 119228 Singapore, Singapore, 169610 Taiwan Tainan, Taiwan, 00704 Taipei, Taiwan, 100 Collapse << |
| NCT03775395 | Hepatocellular Carcinoma | Phase 3 | Recruiting | December 1, 2021 | China, Guangdong ... more >> Cancer Center Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Ming Shi, MD 8620-87343115 shiming@mail.sysu.edu.cn Collapse << |
| NCT02024087 | Advanced Adult Hepatocellular ... more >>Carcinoma Collapse << | Phase 1 Phase 2 | Completed | - | United States, Illinois ... more >> The University of Chicago Medical Center Chicago, Illinois, United States United States, Kansas University of Kansas Medical Center (KUMC) Westwood, Kansas, United States United States, Massachusetts Beth Israel Deaconess Medical Center (BIDMC) Boston, Massachusetts, United States United States, New Jersey Hackensack University Medical Center Hackensack, New Jersey, United States United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States University of Rochester Rochester, New York, United States United States, Ohio University of Cincinnati Cancer Institute Cincinnati, Ohio, United States Collapse << |
| NCT02069145 | Hepatocellular Cancer ... more >> Liver Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> USC/Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 United States, Colorado University of Colorado Cancer Center Aurora, Colorado, United States, 80045 United States, Indiana Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, Massachusetts Massachussetts General Hospital Boston, Massachusetts, United States, 02114 United States, New York Mount Sinai Medical Center New York, New York, United States, 10029 United States, Pennsylvania Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT01131546 | Essential Hypertension | Phase 4 | Completed | - | China ... more >> Peking University People's Hospital Beijing, China Collapse << |
| NCT02250833 | Hypertension | Phase 1 | Completed | - | Korea, Republic of ... more >> Kyungpook National University Hospital Daegu, samdeok-dong, 2-ga 50, samdeok-dong, 2-ga 50 Jung-gu, Korea, Republic of, 700-721 Collapse << |
| NCT01708915 | - | - | Completed | - | - |
| NCT02560779 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Recruiting | November 2020 | United States, Alabama ... more >> University of Alabama Recruiting Birmingham, Alabama, United States, 35294-3300 United States, California City of Hope Withdrawn Duarte, California, United States, 91010 United States, Maryland National Cancer Institute Withdrawn Bethesda, Maryland, United States, 20892 United States, Ohio University Hospitals Recruiting Cleveland, Ohio, United States, 44106 United States, Texas MD Anderson Recruiting Houston, Texas, United States, 77030-4009 Collapse << |
| NCT02989870 | HepatoCellular Carcinoma ... more >> Unresectable HepatoCellular Carcinoma Liver Cancer Collapse << | Phase 1 | Withdrawn(No Interest. Investi... more >>gator was unable to enroll any participants on study.) Collapse << | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00000539 | Arterial Occlusive Diseases ... more >> Cardiovascular Diseases Heart Diseases Vascular Diseases Diabetes Mellitus Collapse << | Phase 3 | Completed | - | - |
| NCT02358824 | Hypertension | Phase 1 | Completed | - | Korea, Republic of ... more >> Kyungpook National University Hospital Daegu, samdeok-dong, 2-ga 50, samdeok-dong, 2-ga 50 Jung-gu, Korea, Republic of, 700-721 Collapse << |
| NCT03657550 | Hypertension | Phase 1 | Completed | - | United States, California ... more >> Collaborative Neuroscience Network, LLC Long Beach, California, United States, 90806 Collapse << |
| NCT01708915 | Acute Low Back Pain | Phase 3 | Completed | - | Germany ... more >> 69.52.49007 Boehringer Ingelheim Investigational Site Bad Lippspringe, Germany 69.52.49003 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49004 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49026 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49032 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49033 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49041 Boehringer Ingelheim Investigational Site Berlin, Germany 69.52.49002 Boehringer Ingelheim Investigational Site Einbeck, Germany 69.52.49024 Boehringer Ingelheim Investigational Site Essen, Germany 69.52.49031 Boehringer Ingelheim Investigational Site Essen, Germany 69.52.49036 Boehringer Ingelheim Investigational Site Essen, Germany 69.52.49005 Boehringer Ingelheim Investigational Site Everswinkel, Germany 69.52.49039 Boehringer Ingelheim Investigational Site Fürth, Germany 69.52.49018 Boehringer Ingelheim Investigational Site Goch, Germany 69.52.49025 Boehringer Ingelheim Investigational Site Großheirath-Rossach, Germany 69.52.49022 Boehringer Ingelheim Investigational Site Haag, Germany 69.52.49009 Boehringer Ingelheim Investigational Site Hamburg, Germany 69.52.49019 Boehringer Ingelheim Investigational Site Hamburg, Germany 69.52.49015 Boehringer Ingelheim Investigational Site Kaarst, Germany 69.52.49042 Boehringer Ingelheim Investigational Site Karlsruhe-Rüppurr, Germany 69.52.49027 Boehringer Ingelheim Investigational Site Köln, Germany 69.52.49035 Boehringer Ingelheim Investigational Site Köln, Germany 69.52.49040 Boehringer Ingelheim Investigational Site Köln, Germany 69.52.49013 Boehringer Ingelheim Investigational Site Köthen, Germany 69.52.49014 Boehringer Ingelheim Investigational Site Künzing, Germany 69.52.49034 Boehringer Ingelheim Investigational Site Ludwigshafen, Germany 69.52.49028 Boehringer Ingelheim Investigational Site Meßkirch, Germany 69.52.49030 Boehringer Ingelheim Investigational Site Meßkirch, Germany 69.52.49029 Boehringer Ingelheim Investigational Site Münster, Germany 69.52.49012 Boehringer Ingelheim Investigational Site Neunkirchen, Germany 69.52.49020 Boehringer Ingelheim Investigational Site Rodgau, Germany 69.52.49037 Boehringer Ingelheim Investigational Site Siegen, Germany 69.52.49016 Boehringer Ingelheim Investigational Site Stockach, Germany 69.52.49010 Boehringer Ingelheim Investigational Site Straßkirchen, Germany 69.52.49021 Boehringer Ingelheim Investigational Site Villingen-Schwenningen, Germany 69.52.49023 Boehringer Ingelheim Investigational Site Wangen, Germany 69.52.49011 Boehringer Ingelheim Investigational Site Weilburg, Germany Collapse << |
| NCT02300311 | - | - | Completed | - | - |
| NCT02756026 | Micronutrient Deficiency | Not Applicable | Completed | - | Bangladesh ... more >> International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b) Dhaka, Bangladesh Collapse << |
| NCT01115647 | Moderate Acute Malnutrition | Not Applicable | Completed | - | Burkina Faso ... more >> 18 Health Centres in Houndé District Houndé, Burkina Faso Collapse << |
| NCT02300311 | Low Back Pain | Phase 3 | Completed | - | Russian Federation ... more >> 69.53.53201 Boehringer Ingelheim Investigational Site Kyiv, Russian Federation 69.53.53102 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation 69.53.53103 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation 69.53.53105 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation 69.53.53106 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation 69.53.53107 Boehringer Ingelheim Investigational Site St. Petersburg, Russian Federation Ukraine 69.53.53202 Boehringer Ingelheim Investigational Site Kyiv, Ukraine 69.53.53203 Boehringer Ingelheim Investigational Site Kyiv, Ukraine Collapse << |
| NCT02567981 | Micronutrient Deficiency | Not Applicable | Recruiting | September 2020 | El Salvador ... more >> FUSAL Recruiting Santa Elena, El Salvador Contact: María J Dominguez, BA (503) 2241-6900 Collapse << |
| NCT03597061 | Weight, Body ... more >>Diet Habit Collapse << | Not Applicable | Not yet recruiting | July 31, 2019 | United States, Ohio ... more >> University of Cincinnati Not yet recruiting Cincinnati, Ohio, United States, 45219 Collapse << |
| NCT01473914 | End Stage Renal Disease | Phase 3 | Completed | - | Canada, Alberta ... more >> University of Calgary Calgary, Alberta, Canada, T2N 1N4 University of Alberta Edmonton, Alberta, Canada, T6G 2V2 Collapse << |
| NCT03544671 | Vitamin D Deficiency ... more >> 25-Hydroxyvitamin D3 Deficiency, Selective Collapse << | Not Applicable | Completed | - | Mexico ... more >> Marta Rivera Pasquel Cuernavaca, Morelos, Mexico, 62100 Collapse << |
| NCT00152893 | HIV Infections | Phase 2 | Completed | - | Canada, Ontario ... more >> University Health Network - Toronto General Hospital Toronto, Ontario, Canada, M5G 2C4 Collapse << |
| NCT03431597 | Micronutrient Deficiencies ... more >> Hyperhomocysteinemia Collapse << | Phase 2 | Active, not recruiting | December 2018 | Gambia ... more >> Keneba Field Station Banjul, Gambia Collapse << |
| NCT00044512 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | United States, California ... more >> Los Angeles, California, United States, 90057 United States, New York New York, New York, United States, 10021-6007 Belgium Bruxelles - Brussel, Belgium, 1000 Bruxelles - Brussel, Belgium, 1070 Bruxelles - Brussel, Belgium, 1090 Gent, Belgium, 9000 Leuven, Belgium, 3000 France Lille Cedex, France, 59020 Marseille, France, 13005 Paris, France, 75020 Rennes Cedex, France, 35062 Saint Herblain, France, 44805 Israel Haifa, Israel, 31096 Jerusalem, Israel, 91120 Petach Tikva, Israel, 49100 Rehovot, Israel, 76100 Tel Aviv, Israel, 64239 Tel Hashomer, Israel, 52621 Italy Rozzano, Milano, Italy, 20089 Forlì, Italy, 47100 Milano, Italy, 20122 Pisa, Italy, 56126 Verona, Italy, 37126 Collapse << |
| NCT00133744 | Perinatal Mortality ... more >> Stillbirth Neonatal Mortality Collapse << | Phase 3 | Completed | - | China, Hebei ... more >> Fengrun Maternal and Child Health Institute Fengrun, Hebei, China, 064000 Laoting Maternal and Child Health Institute Laoting, Hebei, China, 063600 Mancheng Maternal and Child Health Institute Mancheng, Hebei, China, 072150 Xianghe Maternal and Child Health Institute Xianghe, Hebei, China, 065400 Yuanshi Maternal and Child Health Institute Yuanshi, Hebei, China, 051130 Collapse << |
| NCT00044512 | - | - | Completed | - | - |
| NCT00064350 | Lung Cancer | Phase 2 | Completed | - | - |
| NCT00073307 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT00155506 | - | - | Completed | - | - |
| NCT00079612 | Carcinoma, Renal Cell | Phase 2 | Completed | - | United States, Illinois ... more >> Chicago, Illinois, United States, 60637 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas Houston, Texas, United States, 77030 San Antonio, Texas, United States, 78229 United Kingdom London, Greater London, United Kingdom, SW3 6JJ Glasgow, Strathclyde, United Kingdom, G11 6NT Sutton, Surrey, United Kingdom, SM2 5PT Collapse << |
| NCT00489086 | Neoplastic Syndrome | Phase 2 | Completed | - | United States, California ... more >> Children's Hospital and Research Center Oakland Oakland, California, United States, 94609 United States, New York Herbert Irving Comprehensive Cancer Center at Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00093613 | Adult Anaplastic Astrocytoma ... more >> Adult Anaplastic Oligodendroglioma Adult Giant Cell Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 1 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Florida H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Georgia Emory University/Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Maryland Adult Brain Tumor Consortium Baltimore, Maryland, United States, 21231-1000 Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 United States, Massachusetts Massachusetts General Hospital Cancer Center Boston, Massachusetts, United States, 02114 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, North Carolina Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 United States, Ohio Cleveland Clinic Foundation Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania/Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00064350 | - | - | Completed | - | - |
| NCT00093626 | Primary Peritoneal Carcinoma ... more >> Recurrent Ovarian Carcinoma Collapse << | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Gynecologic Oncology Group Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT01368328 | Type 2 Diabetes | Phase 3 | Unknown | September 2011 | Brazil ... more >> Goiania Municipal Health Departament Goiânia, Goiás, Brazil, 74000000 Collapse << |
| NCT00096512 | Metastatic Squamous Neck Cance... more >>r With Occult Primary Squamous Cell Carcinoma Recurrent Metastatic Squamous Neck Cancer With Occult Primary Recurrent Salivary Gland Cancer Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Nasopharynx Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Nasopharynx Stage IVA Salivary Gland Cancer Stage IVA Squamous Cell Carcinoma of the Larynx Stage IVA Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVA Squamous Cell Carcinoma of the Oropharynx Stage IVA Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVA Verrucous Carcinoma of the Larynx Stage IVA Verrucous Carcinoma of the Oral Cavity Stage IVB Salivary Gland Cancer Stage IVB Squamous Cell Carcinoma of the Larynx Stage IVB Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVB Squamous Cell Carcinoma of the Oropharynx Stage IVB Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVB Verrucous Carcinoma of the Larynx Stage IVB Verrucous Carcinoma of the Oral Cavity Stage IVC Salivary Gland Cancer Stage IVC Squamous Cell Carcinoma of the Larynx Stage IVC Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVC Squamous Cell Carcinoma of the Oropharynx Stage IVC Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVC Verrucous Carcinoma of the Larynx Stage IVC Verrucous Carcinoma of the Oral Cavity Tongue Cancer Untreated Metastatic Squamous Neck Cancer With Occult Primary Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Southwest Oncology Group (SWOG) Research Base San Antonio, Texas, United States, 78245 Collapse << |
| NCT00489086 | - | - | Completed | - | - |
| NCT00098254 | - | - | Completed | - | - |
| NCT00098540 | Recurrent Non-small Cell Lung ... more >>Cancer Stage IIIB Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> North Central Cancer Treatment Group Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00073307 | - | - | Completed | - | - |
| NCT00101400 | Breast Neoplasms ... more >> Breast Cancer Collapse << | Phase 2 | Completed | - | Germany ... more >> Stuttgart, Baden-Württemberg, Germany, 70199 München, Bayern, Germany, 80637 Frankfurt, Hessen, Germany, 60590 Italy Bologna, Italy, 40138 Milano, Italy, 20133 Milano, Italy, 20162 Parma, Italy, 43100 Collapse << |
| NCT00101400 | - | - | Completed | - | - |
| NCT00098254 | Non-Small-Cell Lung Carcinoma | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00197756 | - | - | Completed | - | United States, Massachusetts ... more >> Harvard School of Public Health Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00101413 | - | - | Completed | - | - |
| NCT00101413 | Cancer Carcin... more >>oma, Non-Small Cell Lung Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Houston, Texas, United States, 77030 Germany Großhansdorf, Schleswig-Holstein, Germany, 22927 Collapse << |
| NCT00105443 | Carcinoma, Hepatocellular | Phase 3 | Completed | - | - |
| NCT00101894 | Rectal Cancer ... more >> Colon Cancer Collapse << | Phase 1 | Completed | - | - |
| NCT00110994 | Cancer Melano... more >>ma Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Tucson, Arizona, United States, 85724 United States, Colorado Aurora, Colorado, United States, 80045 United States, Florida Lakeland, Florida, United States, 33805 United States, Illinois Park Ridge, Illinois, United States, 60068 United States, Massachusetts Boston, Massachusetts, United States, 02114 Boston, Massachusetts, United States, 02115-6084 Boston, Massachusetts, United States, 02215 United States, Missouri St. Louis, Missouri, United States, 63110-1093 United States, Nebraska Omaha, Nebraska, United States, 68114 United States, North Carolina Charlotte, North Carolina, United States, 28203 United States, Pennsylvania Pittsburgh, Pennsylvania, United States, 15232 United States, South Carolina Hilton Head Island, South Carolina, United States, 29926-2739 United States, Tennessee Nashville, Tennessee, United States, 37232-6307 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT00105443 | - | - | Completed | - | - |
| NCT00107432 | Epithelial Mesothelioma ... more >> Recurrent Malignant Mesothelioma Sarcomatous Mesothelioma Stage IA Malignant Mesothelioma Stage IB Malignant Mesothelioma Stage II Malignant Mesothelioma Stage III Malignant Mesothelioma Stage IV Malignant Mesothelioma Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> Cancer and Leukemia Group B Chicago, Illinois, United States, 60606 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00111020 | Carcinoma, Renal Cell | Phase 3 | Completed | - | - |
| NCT00112671 | - | - | Completed | - | - |
| NCT00112905 | Adenocarcinoma of the Bladder ... more >> Distal Urethral Cancer Metastatic Transitional Cell Cancer of the Renal Pelvis and Ureter Proximal Urethral Cancer Recurrent Bladder Cancer Recurrent Transitional Cell Cancer of the Renal Pelvis and Ureter Recurrent Urethral Cancer Regional Transitional Cell Cancer of the Renal Pelvis and Ureter Squamous Cell Carcinoma of the Bladder Stage III Bladder Cancer Stage IV Bladder Cancer Transitional Cell Carcinoma of the Bladder Urethral Cancer Associated With Invasive Bladder Cancer Collapse << | Phase 2 | Terminated | - | United States, Massachusetts ... more >> Eastern Cooperative Oncology Group Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00112905 | - | - | Terminated | - | - |
| NCT00118170 | Extranodal Marginal Zone B-cel... more >>l Lymphoma of Mucosa-associated Lymphoid Tissue Nodal Marginal Zone B-cell Lymphoma Recurrent Adult Burkitt Lymphoma Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Adult Diffuse Mixed Cell Lymphoma Recurrent Adult Diffuse Small Cleaved Cell Lymphoma Recurrent Adult Grade III Lymphomatoid Granulomatosis Recurrent Adult Immunoblastic Large Cell Lymphoma Recurrent Adult Lymphoblastic Lymphoma Recurrent Grade 1 Follicular Lymphoma Recurrent Grade 2 Follicular Lymphoma Recurrent Grade 3 Follicular Lymphoma Recurrent Mantle Cell Lymphoma Recurrent Marginal Zone Lymphoma Recurrent Small Lymphocytic Lymphoma Refractory Multiple Myeloma Splenic Marginal Zone Lymphoma Stage II Multiple Myeloma Stage III Adult Burkitt Lymphoma Stage III Adult Diffuse Large Cell Lymphoma Stage III Adult Diffuse Mixed Cell Lymphoma Stage III Adult Diffuse Small Cleaved Cell Lymphoma Stage III Adult Immunoblastic Large Cell Lymphoma Stage III Adult Lymphoblastic Lymphoma Stage III Grade 1 Follicular Lymphoma Stage III Grade 2 Follicular Lymphoma Stage III Grade 3 Follicular Lymphoma Stage III Mantle Cell Lymphoma Stage III Marginal Zone Lymphoma Stage III Multiple Myeloma Stage III Small Lymphocytic Lymphoma Stage IV Adult Burkitt Lymphoma Stage IV Adult Diffuse Large Cell Lymphoma Stage IV Adult Diffuse Mixed Cell Lymphoma Stage IV Adult Diffuse Small Cleaved Cell Lymphoma Stage IV Adult Immunoblastic Large Cell Lymphoma Stage IV Adult Lymphoblastic Lymphoma Stage IV Grade 1 Follicular Lymphoma Stage IV Grade 2 Follicular Lymphoma Stage IV Grade 3 Follicular Lymphoma Stage IV Mantle Cell Lymphoma Stage IV Marginal Zone Lymphoma Stage IV Small Lymphocytic Lymphoma Unspecified Adult Solid Tumor, Protocol Specific Waldenström Macroglobulinemia Collapse << | Phase 1 | Completed | - | United States, Illinois ... more >> Cancer and Leukemia Group B (CALGB) Research Base Chicago, Illinois, United States, 60604-1104 Collapse << |
| NCT00110994 | - | - | Completed | - | - |
| NCT00119249 | Stage III Melanoma ... more >> Stage IV Melanoma Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Montefiore Medical Center Bronx, New York, United States, 10467-2490 Collapse << |
| NCT00112671 | Metastatic Transitional Cell C... more >>ancer of the Renal Pelvis and Ureter Recurrent Bladder Cancer Recurrent Transitional Cell Cancer of the Renal Pelvis and Ureter Regional Transitional Cell Cancer of the Renal Pelvis and Ureter Stage III Bladder Cancer Stage IV Bladder Cancer Transitional Cell Carcinoma of the Bladder Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Phase 2 Consortium Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00119639 | Kidney Diseases | Phase 1 | Completed | - | - |
| NCT00121264 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Michigan ... more >> Barbara Ann Karmanos Cancer Institute Detroit, Michigan, United States, 48201 Collapse << |
| NCT00648986 | Brittle Nails | Phase 4 | Completed | - | United States, New York ... more >> Columbia University Medical Center New York, New York, United States, 10032 Collapse << |
| NCT00126568 | Anaplastic Thyroid Cancer ... more >> Recurrent Thyroid Cancer Collapse << | Phase 2 | Terminated | - | United States, Ohio ... more >> Case Western Reserve University Cleveland, Ohio, United States, 44106 Collapse << |
| NCT00126659 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 2 | Terminated | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00126659 | - | - | Terminated | - | - |
| NCT00131911 | - | - | Completed | - | - |
| NCT00131911 | Gastrinoma Gl... more >>ucagonoma Insulinoma Metastatic Gastrointestinal Carcinoid Tumor Neuroendocrine Tumor Pancreatic Polypeptide Tumor Recurrent Gastrointestinal Carcinoid Tumor Recurrent Islet Cell Carcinoma Somatostatinoma WDHA Syndrome Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00131989 | Adult Acute Basophilic Leukemi... more >>a Adult Acute Eosinophilic Leukemia Adult Acute Megakaryoblastic Leukemia (M7) Adult Acute Minimally Differentiated Myeloid Leukemia (M0) Adult Acute Monocytic Leukemia (M5b) Adult Acute Myeloblastic Leukemia With Maturation (M2) Adult Acute Myeloblastic Leukemia Without Maturation (M1) Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With t(15;17)(q22;q12) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Myelomonocytic Leukemia (M4) Adult Erythroleukemia (M6a) Adult Pure Erythroid Leukemia (M6b) Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21287-8936 Collapse << |
| NCT00131937 | Anaplastic Large Cell Lymphoma... more >> Angioimmunoblastic T-cell Lymphoma Hepatosplenic T-cell Lymphoma Peripheral T-cell Lymphoma Recurrent Adult Diffuse Large Cell Lymphoma Recurrent Adult Immunoblastic Large Cell Lymphoma Recurrent Adult T-cell Leukemia/Lymphoma Recurrent Cutaneous T-cell Non-Hodgkin Lymphoma Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Eastern Cooperative Oncology Group Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00126568 | - | - | Terminated | - | - |
| NCT00131937 | - | - | Completed | - | - |
| NCT00217620 | Sarcoma | Phase 2 | Completed | - | - |
| NCT00182689 | Extensive Stage Small Cell Lun... more >>g Cancer Recurrent Small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | United States, Oregon ... more >> SWOG Portland, Oregon, United States, 97239 Collapse << |
| NCT00217646 | Adult Acute Basophilic Leukemi... more >>a Adult Acute Eosinophilic Leukemia Adult Acute Megakaryoblastic Leukemia Adult Acute Monoblastic Leukemia Adult Acute Monocytic Leukemia Adult Acute Myeloid Leukemia With Inv(16)(p13.1q22); CBFB-MYH11 Adult Acute Myeloid Leukemia With Maturation Adult Acute Myeloid Leukemia With Minimal Differentiation Adult Acute Myeloid Leukemia With t(16;16)(p13.1;q22); CBFB-MYH11 Adult Acute Myeloid Leukemia With t(8;21)(q22;q22); RUNX1-RUNX1T1 Adult Acute Myeloid Leukemia With t(9;11)(p22;q23); MLLT3-MLL Adult Acute Myeloid Leukemia Without Maturation Adult Acute Myelomonocytic Leukemia Adult Acute Promyelocytic Leukemia With t(15;17)(q22;q12); PML-RARA Adult Erythroleukemia Adult Pure Erythroid Leukemia Alkylating Agent-Related Acute Myeloid Leukemia Blastic Phase de Novo Myelodysplastic Syndrome Previously Treated Myelodysplastic Syndrome Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Secondary Acute Myeloid Leukemia Secondary Myelodysplastic Syndrome Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00182689 | - | - | Completed | - | - |
| NCT00245102 | Adult Angiosarcoma ... more >> Adult Epithelioid Sarcoma Adult Leiomyosarcoma Adult Malignant Fibrous Histiocytoma Adult Neurofibrosarcoma Adult Synovial Sarcoma Ovarian Sarcoma Recurrent Adult Soft Tissue Sarcoma Recurrent Uterine Sarcoma Stage III Adult Soft Tissue Sarcoma Stage III Uterine Sarcoma Stage IV Adult Soft Tissue Sarcoma Stage IV Uterine Sarcoma Uterine Carcinosarcoma Uterine Leiomyosarcoma Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00238212 | Adenocarcinoma of the Extrahep... more >>atic Bile Duct Adenocarcinoma of the Gallbladder Adenocarcinoma With Squamous Metaplasia of the Gallbladder Cholangiocarcinoma of the Extrahepatic Bile Duct Cholangiocarcinoma of the Gallbladder Recurrent Extrahepatic Bile Duct Cancer Recurrent Gallbladder Cancer Squamous Cell Carcinoma of the Gallbladder Unresectable Extrahepatic Bile Duct Cancer Unresectable Gallbladder Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Southwest Oncology Group San Antonio, Texas, United States, 78245 Collapse << |
| NCT00253578 | Refractory Multiple Myeloma | Phase 2 | Completed | - | United States, Texas ... more >> Southwest Oncology Group San Antonio, Texas, United States, 78245 Collapse << |
| NCT00245102 | - | - | Completed | - | - |
| NCT00238121 | - | - | Completed | - | - |
| NCT00238121 | Recurrent Uterine Sarcoma ... more >> Stage III Uterine Sarcoma Stage IV Uterine Sarcoma Uterine Carcinosarcoma Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 University of Southern California Los Angeles, California, United States, 90033-0804 United States, Illinois Decatur Memorial Hospital Decatur, Illinois, United States, 62526 Central Illinois Hematology Oncology Center Springfield, Illinois, United States, 60702 Canada, Ontario Juravinski Cancer Centre at Hamilton Health Sciences Hamilton, Ontario, Canada, L8V 5C2 Cancer Centre of Southeastern Ontario at Kingston General Hospital Kingston, Ontario, Canada, K7L 5P9 University Health Network-Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00259129 | Neoplasms | Phase 1 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02115 United States, Texas San Antonio, Texas, United States, 78229 Collapse << |
| NCT00254267 | Advanced Gastrointestinal Stro... more >>mal Tumor Collapse << | Phase 2 | Completed | - | - |
| NCT00217620 | - | - | Completed | - | - |
| NCT01502410 | Childhood Hepatocellular Carci... more >>noma Papillary Thyroid Cancer Previously Treated Childhood Rhabdomyosarcoma Recurrent Childhood Liver Cancer Recurrent Childhood Rhabdomyosarcoma Recurrent Thyroid Cancer Recurrent Wilms Tumor and Other Childhood Kidney Tumors Collapse << | Phase 2 | Completed | - | - |
| NCT01502410 | - | - | Completed | - | - |
| NCT01510756 | - | - | Terminated(slow enrollment) | - | - |
| NCT01510756 | Chronic Lymphocytic Leukemia | Phase 2 | Terminated(slow enrollment) | - | United States, California ... more >> UCSD Medical Center La Jolla, California, United States, 92093 Collapse << |
| NCT01578109 | Acute Myeloid Leukemia in Remi... more >>ssion Acute Myeloid Leukemia With FLT3/ITD Mutation Collapse << | Not Applicable | Active, not recruiting | - | United States, Maryland ... more >> University of Maryland/Greenebaum Cancer Center Baltimore, Maryland, United States, 21201 Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01539681 | - | - | Completed | - | Italy ... more >> Many Locations, Italy Collapse << |
| NCT01561833 | T Cell Lymphoma | Phase 1 | Completed | - | United States, Connecticut ... more >> Yale Cancer Center New Haven, Connecticut, United States, 06510 Collapse << |
| NCT01585870 | Neoplasms | Phase 1 | Completed | - | France ... more >> Toulouse, France, 31052 Germany Freiburg, Baden-Württemberg, Germany, 79106 Heidelberg, Baden-Württemberg, Germany, 69120 Berlin, Germany, 10117 Collapse << |
| NCT01507064 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Italy ... more >> UOSC Epatologia - Cardarelli Hospital Napoli, Italy, 80131 Collapse << |
| NCT01618253 | Hepatocellular Carcinoma ... more >> Hepatocellular Cancer Hepatoma Liver Cancer Collapse << | Phase 1 | Withdrawn(Closed due to poor a... more >>ccrual.) Collapse << | June 2016 | United States, Wisconsin ... more >> Froedtert Memorial Lutheran Hospital Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT01613846 | Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT01624285 | Adult Primary Hepatocellular C... more >>arcinoma Localized Resectable Adult Primary Liver Cancer Localized Unresectable Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Collapse << | Phase 2 | Active, not recruiting | July 2020 | - |
| NCT01666756 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Advanced Adult Hepatocellular Carcinoma BCLC Stage B Adult Hepatocellular Carcinoma BCLC Stage C Adult Hepatocellular Carcinoma Collapse << | Phase 1 | Active, not recruiting | January 2020 | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 Collapse << |
| NCT00197743 | HIV Infections ... more >> Disease Transmission, Vertical Collapse << | Phase 3 | Completed | - | United States, Massachusetts ... more >> Harvard School of Public Health Boston, Massachusetts, United States, 02115 Collapse << |
| NCT02525822 | Acne Vulgaris | Phase 2 | Unknown | June 2016 | - |
| NCT01714609 | - | - | Completed | - | - |
| NCT01714609 | Clinically Significant Portal ... more >>Hypertension Collapse << | Phase 2 | Completed | - | United States, Connecticut ... more >> Yale Cancer Center New Haven, Connecticut, United States, 06520 VA Medical Center West Haven West Haven, Connecticut, United States, 06516 United States, Massachusetts Brigham & Womens Boston, Massachusetts, United States, 02115 United States, New York New York University Langone Medical Center New York, New York, United States, 10016 United States, Pennsylvania University of Pennsylvania School of Medicine Philadelphia, Pennsylvania, United States, 19104 United States, Texas The University of Texas Health Science Center at Houston Houston, Texas, United States, 77030 Collapse << |
| NCT00469755 | Acne Vulgaris | Phase 4 | Completed | - | United States, California ... more >> Therapeutics Clinical Research San Diego, California, United States, 92123 United States, Michigan Henry Ford Medical Center-Dept. of Dermatology Detroit, Michigan, United States, 48202 United States, Minnesota Minnesota Clinical Study Center Fridley, Minnesota, United States, 55432 United States, New York State University of New York Downstate Medical Center-Dept. of Dermatology Brooklyn, New York, United States, 11203 United States, Ohio Dermatology Research Associates Cincinnati, Ohio, United States, 45230 United States, Oregon Phoebe Rich, MD & Associates Portland, Oregon, United States, 97210 United States, Pennsylvania Milton S. Hershey Medical Center Center-Div. of Dermatology Hershey, Pennsylvania, United States, 17033-0850 United States, Texas DermResearch, Inc. Austin, Texas, United States, 78759 J & S Studies, Inc. Bryan, Texas, United States, 77802 Stephens & Associates Carrollton, Texas, United States, 75006 United States, Utah Dermatology Research Center Salt Lake City, Utah, United States, 84124 United States, Virginia Virginia Clinical Research, Inc. Norfolk, Virginia, United States, 23507 Collapse << |
| NCT01234337 | Breast Cancer | Phase 3 | Completed | - | - |
| NCT00090545 | - | - | Completed | - | - |
| NCT00090545 | Prostate Cancer | Phase 2 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00095459 | Neoplasms | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00114244 | Stage IV Pancreatic Cancer | Phase 2 | Completed | - | United States, California ... more >> City of Hope Duarte, California, United States, 91010 Collapse << |
| NCT00095966 | Adenocarcinoma of the Pancreas... more >> Recurrent Pancreatic Cancer Stage II Pancreatic Cancer Stage III Pancreatic Cancer Stage IV Pancreatic Cancer Collapse << | Phase 2 | Completed | - | United States, Illinois ... more >> University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637-1470 Collapse << |
| NCT00114244 | - | - | Completed | - | - |
| NCT00901901 | - | - | Completed | - | - |
| NCT00126503 | - | - | Completed | - | - |
| NCT00126620 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> Margaret and Charles Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00707174 | Cancer Lentig... more >>o Maligna Collapse << | Not Applicable | Completed | - | United States, Utah ... more >> Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00096395 | Recurrent Ovarian Epithelial C... more >>ancer Recurrent Primary Peritoneal Cavity Cancer Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Phase 2 Consortium Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00707174 | - | - | Completed | - | - |
| NCT00217399 | Recurrent Breast Cancer ... more >> Stage IV Breast Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, District of Col... more >>umbia Lombardi Comprehensive Cancer Center at Georgetown University Washington, District of Columbia, United States, 20057 Collapse << |
| NCT00244972 | Advanced Malignant Neoplasm | Phase 1 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00126503 | Chromophobe Renal Cell Carcino... more >>ma Clear Cell Renal Cell Carcinoma Papillary Renal Cell Carcinoma Recurrent Renal Cell Carcinoma Sarcomatoid Renal Cell Carcinoma Stage IV Renal Cell Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Dana-Farber Harvard Cancer Center Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Pennsylvania University of Pennsylvania/Abramson Cancer Center Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00217399 | - | - | Completed | - | - |
| NCT02411955 | Acne Vulgaris | Phase 1 | Completed | - | - |
| NCT00829049 | Acne Vulgaris | Phase 4 | Terminated(Study enrollment wa... more >>s terminated due to company decision before the target enrollment of 220 patients was reached.) Collapse << | - | United States, California ... more >> Fremont, California, United States Collapse << |
| NCT00834210 | Acne Vulgaris | Phase 4 | Completed | - | United States, California ... more >> Fremont, California, United States Collapse << |
| NCT00829049 | - | - | Terminated(Study enrollment wa... more >>s terminated due to company decision before the target enrollment of 220 patients was reached.) Collapse << | - | - |
| NCT00834210 | - | - | Completed | - | - |
| NCT00326898 | Stage I Renal Cell Cancer ... more >> Stage II Renal Cell Cancer Stage III Renal Cell Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00436215 | Ovarian Neoplasm ... more >> Fallopian Tube Cancer Primary Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00326495 | - | - | Completed | - | - |
| NCT00436579 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Terminated | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00445588 | Adult Giant Cell Glioblastoma ... more >> Adult Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, Florida Moffitt Cancer Center Tampa, Florida, United States, 33612 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21287 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States, 02114 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, North Carolina Wake Forest University Health Sciences Winston-Salem, North Carolina, United States, 27157 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Pennsylvania University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00454194 | Lung Cancer | Phase 2 | Completed | - | - |
| NCT00436215 | - | - | Completed | - | - |
| NCT01053000 | ACTINIC KERATOSES | Not Applicable | Completed | - | United States, Florida ... more >> Roger Stewart MD PA Fort Lauderdale, Florida, United States, 33308 Collapse << |
| NCT01053000 | - | - | Completed | - | - |
| NCT00326495 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00466232 | Small Cell Carcinoma ... more >> Lung Cancer Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Park Nicollet Institute St. Louis Park, Minnesota, United States, 55416 Collapse << |
| NCT00326898 | - | - | Completed | - | - |
| NCT00466752 | Adenocarcinoma of the Prostate... more >> Stage II Prostate Cancer Stage III Prostate Cancer Collapse << | Phase 2 | Completed | - | United States, Washington ... more >> Fred Hutchinson Cancer Research Center/University of Washington Cancer Consortium Seattle, Washington, United States, 98109 Collapse << |
| NCT01094717 | Psoriasis | Not Applicable | Terminated(lack of efficacy an... more >>d lack of funding) Collapse << | - | United States, Utah ... more >> University of Utah Department of Dermatology Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT00454194 | - | - | Completed | - | - |
| NCT00445588 | - | - | Completed | - | - |
| NCT00492297 | Melanoma | Phase 2 | Completed | - | France ... more >> Bordeaux, France, 33000 Lyon Cedex, France, 39373 Toulouse, France, 31052 Villejuif, France, 94805 United Kingdom Cambridge, Cambridgeshire, United Kingdom, CB2 0QQ Southampton, Hampshire, United Kingdom, SO16 6YD Northwood, Middlesex, United Kingdom, HA6 2RN Sutton, Surrey, United Kingdom, SM2 5PT London, United Kingdom, NW3 2QG London, United Kingdom, SE1 7EH London, United Kingdom, SW3 6JJ Manchester, United Kingdom, M20 4BX Collapse << |
| NCT00498836 | Malignant Melanoma | Phase 2 | Completed | - | United States, Pennsylvania ... more >> University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 United States, Texas Cancer Therapy and Research Center, Institute for Drug Development San Antonio, Texas, United States, 78229 Norway The Norwegian Radium Hospital Oslo, Norway, NO-0310 Sweden Lund University Hospital Lund, Sweden, SE-221 85 Umeå University Hospital Umeå, Sweden Uppsala University Hospital Uppsala, Sweden, SE-751 85 Collapse << |
| NCT00526799 | Ovarian Cancer | Phase 1 Phase 2 | Terminated(Study closed to acc... more >>rual due unfavorable interim analysis) Collapse << | - | United States, Illinois ... more >> Medical & Surgical Specialists, LLC Galesburg, Illinois, United States, 61401 United States, Indiana Oncology Hematology Associates of SW Indiana Evansville, Indiana, United States, 47714 Fort Wayne Oncology & Hematology, Inc Fort Wayne, Indiana, United States, 46815 Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 St. Vincent Hospital Cynecologic Oncology Indianapolis, Indiana, United States, 46260 Arnett Cancer Care Lafayette, Indiana, United States, 47904 Medical Consultants, P.C. Muncie, Indiana, United States, 47303 United States, New York Schwartz Gynecologic Oncology, PLLC Brightwaters, New York, United States, 11718 Collapse << |
| NCT00510250 | Cancer of the Cervix | Phase 1 Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00466752 | - | - | Completed | - | - |
| NCT00536575 | Multiple Myeloma | Phase 1 Phase 2 | Completed | - | United States, Maryland ... more >> Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00526799 | - | - | Terminated(Study closed to acc... more >>rual due unfavorable interim analysis) Collapse << | - | - |
| NCT00536575 | - | - | Completed | - | - |
| NCT00565968 | Melanoma (Skin) | Phase 1 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00562523 | Neoplasms | Phase 1 | Completed | - | United States, Florida ... more >> Tampa, Florida, United States, 33612 United States, Pennsylvania Philadelphia, Pennsylvania, United States, 19111-2497 Collapse << |
| NCT00576056 | Carcinoma, Hepatocellular | Phase 2 | Terminated(Bayer Healthcare is... more >> no supplying the study drug) Collapse << | - | United States, Pennsylvania ... more >> University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00600015 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33901 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 Wellstar Cancer Research Marietta, Georgia, United States, 30060 United States, Kansas Kansas City Cancer Centers Overland Park, Kansas, United States, 66210 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 United States, Michigan Grand Rapids Clinical Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Nebraska Methodist Cancer Center Omaha, Nebraska, United States, 68114 United States, North Carolina Cancer Care of Western North Carolina Asheville, North Carolina, United States, 28801 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 Mid Ohio Oncology/Hematology, Inc./ The Mark H. Zangmeister Center Columbus, Ohio, United States, 43219 United States, South Carolina South Carolina Oncology Associates, PA Columbia, South Carolina, United States, 29210 United States, Tennessee Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Family Cancer Center Collierville, Tennessee, United States, 38017 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 United States, Texas Coastal Bend Cancer Center Corpus Christi, Texas, United States, 78463 United States, Virginia Virginia Cancer Institute Richmond, Virginia, United States, 23235 Collapse << |
| NCT00600015 | - | - | Completed | - | - |
| NCT00492297 | - | - | Completed | - | - |
| NCT00602862 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Center Nijmegen, Gelderland, Netherlands, 6500 HB Collapse << |
| NCT00576056 | - | - | Terminated(Bayer Healthcare is... more >> no supplying the study drug) Collapse << | - | - |
| NCT00609804 | - | - | Completed | - | - |
| NCT00613145 | Refractory Malignancy | Phase 1 | Completed | - | United States, Tennessee ... more >> Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00605566 | Neuroendocrine Tumors | Phase 2 | Completed | - | Canada, Ontario ... more >> Princess Margaret Hospital Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00619541 | Hepatocellular Carcinoma | Phase 2 | Completed | - | Italy ... more >> Dipartimento di Oncologia, dei Trapianti e delle Nuove Tecnologie in Medicina ... Indirizzo: via Roma, 55 - 56100 Pisa Tel. 050-2218690 - Fax. 050-2218685 Pisa, Italy, 56100 Collapse << |
| NCT00619996 | Prostate Cancer | Phase 2 | Completed | - | Italy ... more >> Dipartimento di Oncologia, dei Trapianti e delle Nuove Tecnologie in Medicina ... Indirizzo: via Roma, 55 - 56100 Pisa Tel. 050-2218690 - Fax. 050-2218685 Pisa, Italy, 56100 Collapse << |
| NCT00621686 | Brain and Central Nervous Syst... more >>em Tumors Collapse << | Phase 2 | Completed | - | - |
| NCT00621686 | - | - | Completed | - | - |
| NCT00632541 | Metastatic Breast Cancer | Phase 2 | Terminated(Significant Toxicit... more >>ies Experienced) Collapse << | - | United States, Illinois ... more >> Medical & Surgical Specialists, LLC Galesburg, Illinois, United States, 61401 United States, Indiana Oncology Hematology Associates of SW Indiana Evansville, Indiana, United States, 47714 Fort Wayne Oncology & Hematology, Inc Fort Wayne, Indiana, United States, 46815 Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 Quality Cancer Center (MCGOP) Indianapolis, Indiana, United States, 46202 Arnett Cancer Care Lafayette, Indiana, United States, 47904 Horizon Oncology Center Lafayette, Indiana, United States, 47905 Medical Consultants, P.C. Muncie, Indiana, United States, 47303 Northern Indiana Cancer Research Consortium South Bend, Indiana, United States, 46601 United States, Ohio Ireland Cancer Center - University Hospitals of Cleveland Cleveland, Ohio, United States, 44106 Collapse << |
| NCT00635791 | Malignant Solid Tumour ... more >> Renal Cell Carcinoma Non Small Cell Lung Carcinoma Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado Cancer Center Aurora, Colorado, United States, 80045 Collapse << |
| NCT00609804 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33901 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 Wellstar Cancer Research Marietta, Georgia, United States, 30060 United States, Indiana Providence Medical Group Terre Haute, Indiana, United States, 47802 United States, Kentucky Norton Cancer Institute Louisville, Kentucky, United States, 40207 United States, Louisiana Hematology Oncology Clinic, LLP Baton Rouge, Louisiana, United States, 70806 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 National Capital Clinical Research Consortium Bethesda, Maryland, United States, 20817 United States, Mississippi Jackson Oncology Associates Jackson, Mississippi, United States, 39202 United States, Missouri St. Louis Cancer Care Chesterfield, Missouri, United States, 63017 Research Medical Center Kansas City, Missouri, United States, 64132 United States, Nebraska Nebraska Methodist Cancer Center Omaha, Nebraska, United States, 68114 United States, New Jersey Hematology-Oncology Associates of Northern NJ Morristown, New Jersey, United States, 07960 United States, Tennessee Associates in Hematology Oncology Chattanooga, Tennessee, United States, 37404 Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00661180 | Chronic Myelogenous Leukemia | Phase 2 | Completed | - | United States, Oregon ... more >> Portland, Oregon, United States, 97239 France Lyon Cedex, France, 69437 Paris, France, 75010 Poitiers Cedex, France, 86021 Collapse << |
| NCT00627835 | Locally Advanced Squamous Cell... more >> Carcinomas of the Head and Neck (SCCHN) Collapse << | Phase 1 | Withdrawn(Site decided not to ... more >>open this study) Collapse << | - | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Collapse << |
| NCT00632541 | - | - | Terminated(Significant Toxicit... more >>ies Experienced) Collapse << | - | - |
| NCT00694291 | Prostate Cancer | Phase 2 | Withdrawn | - | United States, Pennsylvania ... more >> Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111 Collapse << |
| NCT00661830 | - | - | Completed | - | - |
| NCT00699374 | Carcinoma, Hepatocellular | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01279629 | - | - | Suspended | - | - |
| NCT00699374 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00759928 | Refractory Solid Tumors | Phase 1 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33916 United States, Tennessee Tennessee Oncology Nashville, Tennessee, United States, 37203 Collapse << |
| NCT00722969 | Non-Small Cell Lung Cancer | Phase 2 | Unknown | April 2009 | Netherlands ... more >> VU university medical center Amsterdam, Netherlands, 1007 MB Netherlands Cancer Institute Amsterdam, Netherlands University Medical Center Groningen Groningen, Netherlands University Hospital Maastricht Maastricht, Netherlands Collapse << |
| NCT00703365 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Thailand ... more >> Department of Medicine, Siriraj Hospital Bangkoknoi, Bangkok, Thailand, 10700 Medical Oncology Unit, Chulalongkorn Hospital Patumwan, Bangkok, Thailand, 10330 Horizon Regional Cancer Center, Bamrungrad Hospital Sukhumvit 3, Bangkok, Thailand, 10110 Collapse << |
| NCT00788099 | Advanced Solid Tumors ... more >> Lymphomas Collapse << | Phase 1 | Completed | - | United States, New Jersey ... more >> The Cancer Institute of New Jersey (CINJ) New Brunswick, New Jersey, United States, 08901 France Institut Gustave Roussy Villejuif, France, 94800 Collapse << |
| NCT00732914 | Renal Cell Carcinoma | Phase 3 | Completed | - | Germany ... more >> Urologische Klinik Mannheim, Baden-Wuerttemberg, Germany, 68167 Collapse << |
| NCT00801801 | - | - | Terminated(Sponsors withdrew f... more >>unding; preliminary efficacy data was not encouraging.) Collapse << | - | - |
| NCT00789763 | Pancreatic Cancer | Phase 1 | Completed | - | Spain ... more >> Hospital de La Santa Creu I Sant Pau Barcelona, Spain, 08025 Hospital Clinic I Provincial Barcelona, Spain, 08036 Hospital Universitario La Paz Madrid, Spain, 28046 Hospital Universitario La Fe Valencia, Spain, 46009 Collapse << |
| NCT00815295 | Squamous Cell Cancer | Phase 1 Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Health System Durham, North Carolina, United States, 27710 Collapse << |
| NCT00801801 | Non Squamous Cell Lung Cancer ... more >> Non Small Cell Lung Cancer Collapse << | Phase 2 | Terminated(Sponsors withdrew f... more >>unding; preliminary efficacy data was not encouraging.) Collapse << | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35294 - 0104 Collapse << |
| NCT00815295 | - | - | Completed | - | - |
| NCT00661830 | Adenocarcinoma | Phase 2 | Completed | - | Germany ... more >> Klinikum der Johannes Gutenberg-Universität Mainz, I. Med. Klinik Mainz, Rheinland-Pfalz, Germany, 55131 Universitätsklinikum Jena, Klinik für Innere Medizin, Innere Medizin II D-07740 Jena, Germany Universitätsklinikum Hamburg-Eppendorf, I. Med. Klinik, Zentrum für Innere Medizin, Martinistr. 3 D-20248 Hamburg, Germany Klinikum Fulda gAG, Tumorklinik, Pacelliallee 4 D-36043 Fulda, Germany Klinikum der Johann-Wolfgang Goethe-Universität, Innere Medizin I, Theodor-Stern-Kai 7 D-60590 Frankfurt, Germany Universitätsklinikum des Saarlandes, Klinik für Innere Medizin II, Kirrberger Str., Gebäude 41 D-66421 Homburg/Saar, Germany Klinikum der Universität München, Medizinische Klinik II, Marchioninistr. 15 D-81377 München, Germany Klinikum rechts der Isar, TU München, II. Medizinische Klinik und Poliklinik, Ismaningerstr. 22 D-81675 München, Germany II. Med. Klinik, Leopoldina-Krankenhaus der Stadt Schweinfurt, Gustav-Adolf-Str. 8 D-97422 Schweinfurt, Germany Klinikum Esslingen Esslingen, Germany, 73730 Universitätsklinikum Halle, Innere Medizin I Halle, Germany, 06120 Collapse << |
| NCT00823290 | Hepatocellular Carcinoma | Phase 1 | Unknown | December 2010 | Germany ... more >> Department of Medicine 1, University Hospital Erlangen Recruiting Erlangen, Germany, 91054 Contact: Matthias Ocker, MD +49-6421-58 ext 68931 Matthias.Ocker@staff.uni-marburg.de Contact: Susanne Gahr, MD +49-9131-85 ext 35000 susanne.gahr@uk-erlangen.de Principal Investigator: Deike Strobel, MD Collapse << |
| NCT00837876 | - | - | Completed | - | - |
| NCT00862082 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Terminated(PR104 plus sorafeni... more >>b was poorly tolerated in patients with advanced HCC) Collapse << | - | United States, Arizona ... more >> University of Arizona Tucson, Arizona, United States, 85724 United States, California Moores UCSD Cancer Center La Jolla, California, United States, 92093 University of California, Irvine Orange, California, United States, 92868 Sharp Clinical Oncology Research San Diego, California, United States, 92123 Pacific Oncology/Hematology San Francisco, California, United States, 94115 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Indiana Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 United States, Maryland Sidney Kimmel Comprehensive Cancer Center At Johns Hopkins Baltimore, Maryland, United States, 21231 United States, New York Columbia University Medical Center New York, New York, United States, 10032 Hong Kong Prince of Wales Hospital Shatin, New Territories, Hong Kong Singapore Singapore General Hospital Singapore, Singapore Taiwan Chang Gung Memorial Hospital Kaohsiung, Taiwan Kaohsiung Medical University Chung-Ho Memorial Hospital Kaohsiung, Taiwan Chi Mei Medical Center, Liouying T'ai-nan, Taiwan China Medical University Hospital Taichung City, Taiwan Cathay General Hospital Taipei City, Taiwan, 10630 National Taiwan University Hospital Taipei, Taiwan Collapse << |
| NCT00844688 | Hepatocellular Carcinoma | Phase 2 | Unknown | December 2009 | Pakistan ... more >> Combined Military Hospital Recruiting Rawalpindi, Punjab, Pakistan, 46000 Principal Investigator: Naeem Naqi, MBBS,FCPS Collapse << |
| NCT00837876 | Pancreatic Cancer | Phase 2 | Completed | - | United States, Kentucky ... more >> Purchase Cancer Group - Paducah Paducah, Kentucky, United States, 42002 United States, Tennessee Erlanger Cancer Center at Erlanger Hospital - Baroness Chattanooga, Tennessee, United States, 37403 Baptist Regional Cancer Center at Baptist Riverside Knoxville, Tennessee, United States, 37901 Vanderbilt-Ingram Cancer Center - Cool Springs Nashville, Tennessee, United States, 37064 Vanderbilt-Ingram Cancer Center at Franklin Nashville, Tennessee, United States, 37064 Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232-6838 Collapse << |
| NCT00866320 | - | - | Completed | - | - |
| NCT00862082 | - | - | Terminated(PR104 plus sorafeni... more >>b was poorly tolerated in patients with advanced HCC) Collapse << | - | - |
| NCT00867321 | Liver Cancer | Phase 1 Phase 2 | Completed | - | - |
| NCT00872014 | Advanced Hepatocellular Carcin... more >>oma Inoperable Hepatocellular Carcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT00869570 | Colorectal Cancer | Phase 1 Phase 2 | Completed | - | Hungary ... more >> Szent Laszlo Korhaz Budapest, Hungary, 1097 Switzerland Saint Claraspital AG Basel, Switzerland, CH-4016 Universitaetsspital-Basel Basel, Switzerland, CH-4031 Istituto Oncologico della Svizzera Italiana - Ospedale Regionale Bellinzona e Valli Bellinzona, Switzerland, 6500 Inselspital, Bern Bern, Switzerland, CH-3010 Spitalzentrum Biel Biel, Switzerland, CH-2501 Kantonsspital Bruderholz Bruderholz, Switzerland, CH-4101 Kantonsspital Graubuenden Chur, Switzerland, CH-7000 Hopital Cantonal Universitaire de Geneva HUG Geneva, Switzerland, CH-1211 Kantonsspital Luzern Luzern, Switzerland, 6000 OnkoZentrum Luzern at Klinik St. Anna Luzern, Switzerland, 6006 Kantonsspital - St. Gallen St. Gallen, Switzerland, CH-9007 SpitalSTS AG Simmental-Thun-Saanenland Thun, Switzerland, 3600 Kantonsspital Winterthur Winterthur, Switzerland, CH-8400 Onkozentrum - Klinik im Park Zurich, Switzerland, 8002 Onkozentrum Hirslanden Zurich, Switzerland, CH-8008 UniversitaetsSpital Zuerich Zurich, Switzerland, CH-8091 Stadtspital Triemli Zürich, Switzerland, 8063 Collapse << |
| NCT00873002 | Liver Cancer | Phase 1 | Terminated(Dose Limiting Toxic... more >>ity) Collapse << | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT00867321 | - | - | Completed | - | - |
| NCT00095693 | Anaplastic Thyroid Cancer ... more >> Insular Thyroid Cancer Recurrent Thyroid Cancer Stage III Follicular Thyroid Cancer Stage III Papillary Thyroid Cancer Stage IV Follicular Thyroid Cancer Stage IV Papillary Thyroid Cancer Collapse << | Phase 2 | Terminated | - | United States, Ohio ... more >> Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00866320 | Kidney Cancer | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106 United States, Texas Baylor Sammons Cancer Center Dallas, Texas, United States, 75246 Collapse << |
| NCT00875745 | Leukemia, Myeloid, Acute ... more >> Leukemia, Promyelocytic, Acute Myelodysplastic Syndromes Collapse << | Phase 1 | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00901901 | Carcinoma, Hepatocellular | Phase 3 | Completed | - | - |
| NCT00964743 | - | - | Terminated(Low Accrual) | - | - |
| NCT00964743 | Neoplastic Meningitis | Not Applicable | Terminated(Low Accrual) | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT00884416 | Glioblastoma ... more >>Gliosarcoma Anaplastic Astrocytoma Anaplastic Oligoastrocytoma Anaplastic Oligodendroglioma Collapse << | Phase 1 | Completed | - | Switzerland ... more >> Geneva University Hospital (Hopitaux Universitaires de Geneve), Department of Oncology Geneva, GE, Switzerland, 1211 Collapse << |
| NCT00971126 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Terminated(Two patients in the... more >> first dose level be counted as reaching DLT. DSMB recommend terminated early this trial.) Collapse << | - | Taiwan ... more >> Ghang-Gung Memorial Hospital at Chia-Yi Chia-Yi, Taiwan, 613 Collapse << |
| NCT01004003 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Austria ... more >> 1199.37.43001 Boehringer Ingelheim Investigational Site Wien, Austria 1199.37.43002 Boehringer Ingelheim Investigational Site Wien, Austria France 1199.37.33001 Boehringer Ingelheim Investigational Site Paris, France 1199.37.33002 Boehringer Ingelheim Investigational Site Paris, France Germany 1199.37.49008 Boehringer Ingelheim Investigational Site Berlin, Germany 1199.37.49009 Boehringer Ingelheim Investigational Site Erlangen, Germany 1199.37.49002 Boehringer Ingelheim Investigational Site Freiburg, Germany 1199.37.49001 Boehringer Ingelheim Investigational Site Hannover, Germany 1199.37.49010 Boehringer Ingelheim Investigational Site Heidelberg, Germany 1199.37.49005 Boehringer Ingelheim Investigational Site Jena, Germany 1199.37.49004 Boehringer Ingelheim Investigational Site Magdeburg, Germany 1199.37.49003 Boehringer Ingelheim Investigational Site München, Germany 1199.37.49006 Boehringer Ingelheim Investigational Site Tübingen, Germany Hungary 1199.37.36001 Boehringer Ingelheim Investigational Site Debrecen, Hungary Netherlands 1199.37.31002 Boehringer Ingelheim Investigational Site Leiden, Netherlands 1199.37.31001 Boehringer Ingelheim Investigational Site Utrecht, Netherlands Poland 1199.37.48002 Boehringer Ingelheim Investigational Site Olsztyn, Poland 1199.37.48003 Boehringer Ingelheim Investigational Site Warsaw, Poland 1199.37.48001 Boehringer Ingelheim Investigational Site Warszawa, Poland Romania 1199.37.40002 Boehringer Ingelheim Investigational Site Bucharest, Romania 1199.37.40003 Boehringer Ingelheim Investigational Site Cluj-Napoca, Romania United Kingdom 1199.37.44001 Boehringer Ingelheim Investigational Site Edgbaston, Birmingham, United Kingdom 1199.37.44005 Boehringer Ingelheim Investigational Site Glasgow, United Kingdom 1199.37.44008 Boehringer Ingelheim Investigational Site Liverpool, United Kingdom 1199.37.44002 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.37.44003 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.37.44006 Boehringer Ingelheim Investigational Site Manchester, United Kingdom 1199.37.44004 Boehringer Ingelheim Investigational Site Nottingham, United Kingdom Collapse << |
| NCT01005797 | Renal Cancer ... more >>Non Small Cell Lung Cancer (NSCLC) Soft Tissue Sarcoma Collapse << | Phase 1 | Completed | - | United States, South Carolina ... more >> Medical University of South Carolina Hollings Cancer Center Charleston, South Carolina, United States, 29425 Collapse << |
| NCT00984425 | Solid Tumors | Phase 1 | Completed | - | Italy ... more >> Istituto Clinco Humanitas Rozzano, Milano, Italy, 20089 Collapse << |
| NCT01004003 | - | - | Completed | - | - |
| NCT01032850 | - | - | Terminated(Low accrual rate) | - | - |
| NCT01032850 | Hepatocellular Carcinoma | Phase 2 | Terminated(Low accrual rate) | - | United States, New Mexico ... more >> University of New Mexico Cancer Center @ Lovelace Medical Center Albuquerque, New Mexico, United States, 87102 University of New Mexico Cancer Center Albuquerque, New Mexico, United States, 87106 Cancer Center at Presbyterian Hospital Albuquerque, New Mexico, United States, 87110 Collapse << |
| NCT01047891 | Ovarian Cancer | Phase 2 | Completed | - | Germany ... more >> Charité Campus Virchow-Klinikum Berlin, Germany, 13355 Collapse << |
| NCT01078961 | Melanoma | Phase 1 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01075555 | Liver Cancer | Phase 3 | Completed | - | France ... more >> Hopital Du Bocage Dijon, France, 21000 Collapse << |
| NCT01075113 | Liver Cancer | Phase 1 | Active, not recruiting | January 31, 2023 | United States, Virginia ... more >> Hunter Holmes McGuire VA Medical Center Richmond, Virginia, United States, 23249 Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01093222 | - | - | Completed | - | - |
| NCT01100242 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(Low accrual) | - | United States, New Mexico ... more >> University of New Mexico Cancer Center Albuquerque, New Mexico, United States, 87106 The Cancer Center at Presbyterian Albuquerque, New Mexico, United States, 87110 Memorial Medical Center- Cancer Center Las Cruces, New Mexico, United States, 88011 Collapse << |
| NCT01230697 | - | - | Unknown | September 2011 | Italy ... more >> Alfredo Berruti Recruiting Orbassano (To), Turin, Italy, 10043 Contact: Elisa Bellini, MD (+39) 011 9026 ext 526 elisa.bellini3@tin.it Principal Investigator: Alfredo Berruti, PHD Sub-Investigator: Isabella Chiappino, MD Sub-Investigator: Cinzia Ortega, MD Sub-Investigator: Mario Pirisi, PHD Sub-Investigator: Antonino Picciotto, PHD Sub-Investigator: Camillo Porta, MD Sub-Investigator: Anna Pia, MD Sub-Investigator: Elena Bianco, MD Collapse << |
| NCT01093222 | Extrahepatic Bile Duct Adenoca... more >>rcinoma Gallbladder Adenocarcinoma Gallbladder Adenocarcinoma With Squamous Metaplasia Hilar Cholangiocarcinoma Recurrent Extrahepatic Bile Duct Carcinoma Recurrent Gallbladder Carcinoma Undifferentiated Gallbladder Carcinoma Unresectable Extrahepatic Bile Duct Carcinoma Unresectable Gallbladder Carcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT02678611 | Safety: Healthy Subjects | Phase 1 | Completed | - | Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << |
| NCT01234337 | - | - | Completed | - | - |
| NCT01246986 | Carcinoma, Hepatocellular | Phase 2 | Active, not recruiting | December 3, 2018 | - |
| NCT01235416 | Histologically or Cytologicall... more >>y Documented Solid Tumors Collapse << | Phase 1 | Completed | - | - |
| NCT01100242 | - | - | Terminated(Low accrual) | - | - |
| NCT01217034 | Hepatocellular Carcinoma ... more >> Unresectable Hepatocellular Carcinoma Carcinoma, Hepatocellular Liver Neoplasm Collapse << | Phase 2 | Active, not recruiting | March 2018 | Japan ... more >> Kinki University Hospital Osaka-Sayama, Osaka, Japan, 589-8511 Collapse << |
| NCT00387413 | Hyperalgesia | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Cambridge, Cambridgeshire, United Kingdom, CB2 0QQ Collapse << |
| NCT01357486 | Hepatocellular Carcinoma ... more >> CHILD B Collapse << | Phase 2 | Completed | - | France ... more >> CH d'Abbeville Abbeville, France, 80142 CH Pays d'Aix Aix-en-provence, France, 13616 CH d'Auxerre Auxerre, France, 89011 CH de la Côte Basque Bayonne, France, 64109 AP-HP- Hôpital Jean-Verdier Bondy, France, 93143 CHU de Bordeaux Bordeaux, France, 33075 CH Duchenne Boulogne Sur Mer, France, 62321 CH de Béziers Béziers, France, 34525 AP-HP Hôpital Henri Mondor Creteil, France, 94010 CHU Le Bocage Dijon, France, 21079 CH Départemental Vendée La Roche-sur-yon, France, 85925 CH Le Mans Le Mans, France, 72037 CH de Bretagne Sud Lorient, France, 56100 Hôpital privé Jean Mermoz Lyon, France, 69008 AP-HM Hôpital de la Timone Marseille, France, 13385 CH de Meaux Meaux, France, 77104 CH Mont de Marsan Mont-de-marsan, France, 40024 CHU de Nancy Hôpital Brabois Nancy, France, 54511 CHU de Nantes Hôpital de l'Hotel Dieu Nantes, France, 44093 CHU Nîmes Nimes, France, 30029 CHR d'Orléans - Hôpital La Source Orleans, France, 45067 Groupe Hospitalier Paris Saint Joseph Paris, France, 75014 CH Perpignan Perpignan, France, 66046 CHU de Bordeaux, Hôpital du Haut Lévèque Pessac, France, 33604 CH de la Région d'Annecy Pringy, France, 74374 Centre Eugène Marquis Rennes, France, 35042 Clinique Mathilde Rouen, France, 76000 Clinique Armoricaine de Radiologie Saint Brieuc, France, 22015 CH Saint-Malo Saint Malo, France, 35400 Centre René Gauducheau CLCC Nantes Atlantique Saint-herblain, France, 44805 CH Gaston Ramon Sens, France, 89100 Centre Régional de Lutte contre le Cancer Centre Paul Strauss Strasbourg, France, 67065 Hôpitaux Universitaires de Strasbourg Hôpital civil Strasbourg, France, 67091 Hôpitaux Universitaires de Strasbourg, Hôpital Hautepierre Strasbourg, France, 67098 CHRU de Tours Tours, France, 37044 Collapse << |
| NCT01254890 | Leukemia | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> UT MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00414388 | Prostate Cancer | Phase 1 Phase 2 | Completed | - | United States, Illinois ... more >> Onocology Specialists, S.C Niles, Illinois, United States, 60714 Oncology Specialists, S.C Park Ridge, Illinois, United States, 60068 Collapse << |
| NCT01254890 | - | - | Completed | - | - |
| NCT00414388 | - | - | Completed | - | - |
| NCT01290926 | Colorectal Cancer | Phase 2 | Completed | - | Belgium ... more >> Cliniques Universitaires Saint Luc Woluwe Saint Lambert, Bruxelles, Belgium, 1200 Universiteit Ziekenhuis Antwerpen Antwerpen, Belgium Institut Jules Bordet Brussels, Belgium, 1000 Hopital Erasme Brussels, Belgium, 1070 Entité Jolimontoise La Louviere, Belgium Hopitaux St Joseph Liege, Belgium, 4000 CHU Ambroise Paré Mons, Belgium, 7000 Collapse << |
| NCT01378377 | Advanced Solid Tumor | Phase 1 | Terminated(The trial was stopp... more >>ed due to the toxicities observed with the combination of pimasertib and temsirolimus.) Collapse << | - | United States, California ... more >> Cedars-Sinai Medical Center Los Angeles, California, United States United States, Massachusetts For Recruiting Locations in the US contact US Medical Information in Rockland, Massachusetts, United States United States, Texas The University of Texas MD Anderson Cancer Center Houston, Texas, United States Collapse << |
| NCT01450384 | Unspecified Adult Solid Tumor | Phase 1 | Completed | - | United States, Virginia ... more >> Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01376453 | Rectal Cancer | Phase 1 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 Collapse << |
| NCT01418729 | Advanced Hepatocarcinoma | Phase 2 | Completed | - | Spain ... more >> Hospital Donostia, Instituto Biodonostia San Sebastian, Gipuzkoa, Spain, 20014 Collapse << |
| NCT01471353 | Colorectal Cancer Metastatic | Phase 2 | Completed | - | United States, Florida ... more >> UF Health Cancer Center Gainesville, Florida, United States, 32610 Collapse << |
| NCT01471353 | - | - | Completed | - | - |
| NCT01497444 | Kidney Cancer ... more >> Liver Cancer Collapse << | Phase 1 | Active, not recruiting | - | United States, Arizona ... more >> Mayo Clinic Scottsdale Scottsdale, Arizona, United States, 85259-5499 United States, Minnesota Mayo Clinic Cancer Center Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01378377 | - | - | Terminated(The trial was stopp... more >>ed due to the toxicities observed with the combination of pimasertib and temsirolimus.) Collapse << | - | - |
| NCT01481870 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Unknown | December 2015 | Japan ... more >> Yamagata University Faculty of Medicine Yamagata, Japan, 990-9585 Collapse << |
| NCT01728948 | - | - | Completed | - | China ... more >> Many Locations, China Collapse << |
| NCT01556815 | PHENYTOIN/SORAFENIB [VA Drug I... more >>nteraction] Liver Neoplasms Carcinoma, Hepatocellular Digestive System Neoplasms Neoplasms by Site Liver Diseases Adenocarcinoma Carcinoma Neoplasms, Glandular and Epithelial Neoplasms by Histologic Type DOXORUBICIN/TRASTUZUMAB [VA Drug Interaction] HBV Collapse << | Phase 2 | Unknown | May 2015 | China, Shandong ... more >> Shandong Cancer Hospital and Institute Not yet recruiting Jinan, Shandong, China, 250117 Contact: Wenbo Shao, MD, Ph D +8653167626412 shaowenbomd@gmail.com Principal Investigator: Jinlong Song, MD Sub-Investigator: Wenbo Shao, MD, Ph D Collapse << |
| NCT01730937 | Adult Primary Hepatocellular C... more >>arcinoma Advanced Adult Primary Liver Cancer Recurrent Adult Primary Liver Cancer Collapse << | Phase 3 | Recruiting | June 2025 | - |
| NCT01539018 | Advanced Hepatocellular Carcin... more >>oma Collapse << | Phase 2 | Terminated(After interim resul... more >>ts data showed no evidence of a difference between both groups) Collapse << | - | Egypt ... more >> Ain shams university Cairo, Egypt Cairo University Hospitals Cairo, Egypt National cancer institute Cairo, Egypt NHTMRI Cairo, Egypt National Liver Institute Monofeiya, Egypt Collapse << |
| NCT01736878 | Medullary Thyroid Carcinoma | Phase 2 | Withdrawn(Study was withdrawn ... more >>due to unanticipated hassle in patient recruitment.) Collapse << | - | Germany ... more >> University Hospital Ulm, Clinic for Nuclear Medicine Ulm, Baden-Wuerttemberg, Germany, D-89081 Collapse << |
| NCT01829035 | Advanced Adult Hepatocellular ... more >>Carcinoma Collapse << | Phase 3 | Completed | - | Korea, Republic of ... more >> National Cancer Center, Korea Seoul, Korea, Republic of Collapse << |
| NCT01724606 | Breast Cancer ... more >> Brain Metastases Collapse << | Phase 1 | Recruiting | November 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Recruiting Basking Ridge, New Jersey, United States Contact: Andrew Seidman, MD 646-888-5445 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Andrew Seidman, MD 646-888-5445 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Recruiting Commack, New York, United States, 11725 Contact: Andrew Seidman, MD 646-422-4333 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Andrew Seidman, MD 646-888-5445 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Andrew Seidman, MD 646-888-5445 Contact: Kathryn Beal, MD 212-639-5159 Principal Investigator: Andrew Seidman, MD Memorial Sloan Kettering at Mercy Medical Center Recruiting Rockville Centre, New York, United States Contact: Andrew Seidman, MD 646-888-5445 Collapse << |
| NCT01801163 | Hepatocellular Carcinoma | Phase 1 | Withdrawn(Trial had no patient... more >>s accrued while open) Collapse << | - | United States, Indiana ... more >> Indiana University Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01839630 | - | - | Completed | - | China ... more >> Multiple Locations, China Collapse << |
| NCT01849588 | Hepatocellular Cancer | Phase 4 | Terminated(Slow Accrual) | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02214 Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01900002 | Liver Cancer | Phase 2 | Active, not recruiting | September 2020 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01893099 | Ocular Melanoma | Phase 1 | Completed | - | Switzerland ... more >> Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland Collapse << |
| NCT01901692 | Hepatocellular Carcinoma | Phase 2 Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 05505 Collapse << |
| NCT01849588 | - | - | Terminated(Slow Accrual) | - | - |
| NCT01906216 | Hepatocellular Carcinoma | Phase 2 Phase 3 | Recruiting | December 2018 | China, Shaanxi ... more >> Xijing Hospital of Digestive Disease Recruiting Xi`an, Shaanxi, China, 710032 Contact: Wei Bai, MD +8615991771343 Contact: Yan Zhao, MD +8613571884093 Collapse << |
| NCT01932385 | Liver Cancer | Phase 2 Phase 3 | Completed | - | China, Shandong ... more >> Qingdao Central Hospital, Qingdao Cancer Hospital Qingdao, Shandong, China, 266042 Collapse << |
| NCT01915602 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | - |
| NCT01933945 | - | - | Completed | - | - |
| NCT01934829 | Hepatocellular Carcinoma | Phase 2 | Completed | - | China, Beijing ... more >> 301 Military Hospital, Beijing, China Beijing, Beijing, China, 100039 302 Military Hospital, Beijing, China Beijing, Beijing, China, 100039 China, Fujian Union Hospital of Fujian Medical University, Fujian, China Fuzhou, Fujian, China, 350001 China, Guangdong Guangdong Provincial People's Hospital, Guangdong, China Guangzhou, Guangdong, China, 510080 The Third Affiliated Hospital of Sun Yat-sen University, Guangdong, China Guangzhou, Guangdong, China, 510630 China, Heilongjiang Heilongjiang Provincial Cancer Hospital, Heilongjiang, China Haerbin, Heilongjiang, China, 150081 China, Jiangsu The 81 Hospital of the Chinese People's Liberation Army, Nanjing, China Nanjing, Jiangsu, China, 210002 China, Jilin Jilin Provincial Tumor Hospital, Jilin, China Changchun, Jilin, China, 130012 China, Shanghai Zhongshan Hospital, Fudan University, Shanghai Shanghai, Shanghai, China, 200032 Eastern Hepatobiliary Surgery Hospital of the Second Military Medical University, Shanghai, China Shanghai, Shanghai, China, 200438 China, Tianjin Tianjin Cancer Hospital, Tianjin, China Tianjin, Tianjin, China, 300060 Collapse << |
| NCT02849873 | Acne | Phase 1 | Completed | - | United States, Alabama ... more >> Valeant Site 01 Birmingham, Alabama, United States, 35201 Collapse << |
| NCT02981498 | Hepatocellular Carcinoma | Phase 2 | Recruiting | April 2017 | China, Guangdong ... more >> Cancer Center, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Ming Shi, MD Shiming@sysu.edu.cn Collapse << |
| NCT00752063 | Advanced Hepatocellular Carcin... more >>oma Metastatic Hepatocellular Carcinoma Collapse << | Phase 2 | Unknown | December 2008 | China ... more >> Queen Mary Hospital Recruiting Hong Kong, China Contact: Ida Choi, MPhil (852)28553635 cychoia@hkucc.hku.hk Principal Investigator: Ronnie Poon, MD Collapse << |
| NCT00747799 | Nasopharyngeal Neoplasms | Phase 2 | Completed | - | China, Guangdong ... more >> Cancer Center of Sun-Yat Sen University Guangzhou, Guangdong, China, 510000 Collapse << |
| NCT00808145 | Hepatocellular Carcinoma | Phase 2 | Withdrawn(Lack of enrollment, ... more >>many screen failures and non eligible study participants) Collapse << | - | United States, Massachusetts ... more >> Lahey Clinic Burlington, Massachusetts, United States, 01805 Collapse << |
| NCT00764972 | Metastatic Breast Cancer | Phase 1 Phase 2 | Unknown | December 2011 | Canada, Quebec ... more >> Jewish General Hospital Recruiting Montreal, Quebec, Canada, H3T 1E2 Contact: Lawrence Panasci 514-340-8248 lpanasci@hotmail.com Contact: Cristiano Ferrario 514-340-8248 cristianoferrario@gmail.com Principal Investigator: Lawrence Panasci Collapse << |
| NCT00828074 | Breast Cancer | Phase 1 Phase 2 | Completed | - | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010-3000 South Pasadena Cancer Center Pasadena, California, United States, 91030 Collapse << |
| NCT00828074 | - | - | Completed | - | - |
| NCT00758381 | Locally Advanced Pancreatic Ca... more >>ncer Collapse << | Phase 2 | Unknown | August 2009 | Italy ... more >> A.O. Treviglio-Caravaggio, P.le Ospedale n1 Recruiting Treviglio, Bergamo, Italy, 24047 Sub-Investigator: sandro Barni, MD Ospedale S.Orsola Fatebenefratelli Active, not recruiting Brescia, BS, Italy, 15100 A.O. Ospedale S.Martino Recruiting Genova, GE, Italy, 16132 Sub-Investigator: Alberto Sobrero, MD A.O. san Paolo Recruiting Milano, MI, Italy, 20100 Sub-Investigator: Paolo Foa, MD Casa di Cura Igea Not yet recruiting Milano, MI, Italy, 20100 Sub-Investigator: Gianfranco Pancera, MD Ospedale S.Carlo Borromeo Not yet recruiting Milano, MI, Italy, 20123 Sub-Investigator: Donata Tabiadon, MD A.O. S.Gerardo Recruiting Monza, MI, Italy, 20052 Sub-Investigator: Paolo Bidoli, MD A.O. Universitaria Ospedali Riuniti Umberto I Recruiting Ancona, Italy, 60020 Principal Investigator: Stefano Cascinu, M.Pr Ospedali Riuniti, Largo Barozzi, 1 Recruiting Bergamo, Italy, 24128 Principal Investigator: Roberto Labianca, MD A.O.Policlinico S.Orsola Malpighi Active, not recruiting Bologna, Italy, 40138 A.O. Careggi-Università, Viale Pieraccini, 17 Recruiting Firenze, Italy, 50139 Sub-Investigator: Francesco Di Costanzo, MD Ospedale Galliera Active, not recruiting Genova, Italy, 16132 A.O. Carlo Poma - Via Albertoni, 1 Recruiting Mantova, Italy, 46100 Sub-Investigator: Enrico Aitini, MD A.O. Cà Granda, Piazza Ospedale Maggiore, 3 Recruiting Milano, Italy, 20162 Sub-Investigator: Salvatore Siena, MPr Policlinico di modena Active, not recruiting Modena, Italy, 41100 Università Campus Biomedico, Via Emilio Longoni, 83 Active, not recruiting Roma, Italy, 00155 A.O. S.Giovanni Calabita Fatebenefratelli Active, not recruiting Roma, Italy, 00186 Collapse << |
| NCT00828594 | Hepatocellular Carcinoma | Phase 1 | Terminated | - | United States, California ... more >> City of Hope Medical Center Duarte, California, United States, 91010 UCLA Department of Medicine Los Angeles, California, United States, 90005 United States, North Carolina Duke University Durham, North Carolina, United States, 27710 Korea, Republic of Novartis Investigative Site Seoul, Korea, Republic of Netherlands Novartis Investigative Site Amsterdam, Netherlands Spain Novartis Investigative Site Barcelona, Spain Novartis Invstigative Site Madrid, Spain Taiwan Novartis Investigative Site Tainan, Taiwan Collapse << |
| NCT00791544 | Liver Carcinoma | Phase 1 Phase 2 | Terminated(Company decision to... more >> discontinue the AVE1642 development program, not due to any safety or efficacy concerns) Collapse << | - | France ... more >> Sanofi-Aventis Administrative Office Paris, France Collapse << |
| NCT00844883 | Hepatocellular Carcinoma ... more >> Hepatoma Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> The Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 Collapse << |
| NCT00786110 | Adrenocortical Carcinoma | Phase 2 | Unknown | October 2010 | Italy ... more >> Department of Clinical and Biological Sciences, University of Turin Recruiting Orbassano, Italy Contact: Paola Perotti : +390119026 ext Ext. 017 Principal Investigator: Alfredo Berruti Azienda Ospedaliera di Padova Not yet recruiting Padova, Italy Contact: Franco Mantero Sub-Investigator: Franco Mantero Azienda Ospedaliera Università di Palermo Not yet recruiting Palermo, Italy Sub-Investigator: Vittorio Gebbia Policlinico Universitario Campus Biomedico- Roma Not yet recruiting Roma, Italy Contact: Daniele Santini Sub-Investigator: Daniele Santini Ist. Clin.Humanitas Not yet recruiting Rozzano, Italy Contact: Carlo Carnaghi Sub-Investigator: Carlo Carnaghi Collapse << |
| NCT00844883 | - | - | Completed | - | - |
| NCT00855218 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | - |
| NCT00839111 | Colorectal Neoplasms | Phase 2 | Unknown | November 2010 | China, Shanghai ... more >> Cancer Hospital,Fudan University Recruiting Shanghai, Shanghai, China, 200032 Contact: Yanfei Liu, Master +862164175590 ext 1107 Collapse << |
| NCT00875615 | Liver Cancer | Phase 2 | Completed | - | United States, Florida ... more >> University of Miami Sylvester Comprehensive Cancer Center - Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00870532 | Non-Small Cell Lung Cancer | Phase 1 | Completed | - | Singapore ... more >> National Cancer Centre, Singapore Singapore, Singapore, 169610 Collapse << |
| NCT00875615 | - | - | Completed | - | - |
| NCT00880542 | Sarcoma | Phase 2 | Terminated(The study lost fund... more >>ing.) Collapse << | - | United States, California ... more >> Jonsson Comprehensive Cancer Center at UCLA Los Angeles, California, United States, 90095-1781 Collapse << |
| NCT00881751 | - | - | Completed | - | - |
| NCT00881751 | Liver Cancer | Phase 2 | Completed | - | United States, California ... more >> USC/Norris Comprehensive Cancer Center and Hospital Los Angeles, California, United States, 90033-0804 California Pacific Medical Center San Francisco, California, United States, 94115 United States, New York Columbia University/ New York Presbyterian Hospital New York, New York, United States, 10032 United States, South Carolina Hollings Cancer Center at Medical University of South Carolina Charleston, South Carolina, United States, 29425 United States, Tennessee Tennessee Oncology, PLLCat Sarah Cannon Cancer Center Nashville, Tennessee, United States, 37203 United States, Virginia UVA Cancer Center Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT00855218 | - | - | Completed | - | - |
| NCT00908167 | Acute Myeloid Leukemia ... more >> Infantile Leukemia (Both AML and ALL) Myelodysplastic Syndrome Myelodysplastic/Myeloproliferative Neoplasms Biphenotypic Leukemia Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT00919061 | - | - | Completed | - | - |
| NCT00919061 | Extrahepatic Bile Duct Cancer ... more >> Gallbladder Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00933777 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Non-Small Cell Lung Cancer Collapse << | Phase 1 | Completed | - | Germany ... more >> Center for Integrated Oncology, Dep.I of Internal Medicine, University Hospital Cologne Cologne, Germany Collapse << |
| NCT00880542 | - | - | Terminated(The study lost fund... more >>ing.) Collapse << | - | - |
| NCT00941863 | Carcinoma | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Philadelphia, Pennsylvania, United States, 19104 United States, Tennessee Nashville, Tennessee, United States, 37232 United States, Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00941967 | Liver Cancer | Phase 2 | Unknown | - | France ... more >> Centre Regional de Lutte Contre le Cancer - Centre Val d'Aurelle Recruiting Montpellier, France, 34295 Contact: Eric Assenat, MD 33-4-6761-2593 Collapse << |
| NCT00933816 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Completed | - | Japan ... more >> Kurume University Medical Center Kurume, Fukuoka, Japan, 839-0863 Ogaki Municipal Hospital Oogaki, Gifu, Japan, 503-8502 Kinki University School of Medicine Osaka-Sayama, Osaka, Japan, 589-8511 Collapse << |
| NCT00954135 | Breast Cancer | Phase 2 | Unknown | December 2010 | Italy ... more >> Breast Unit Azienda Ospedaliera Istituti Ospitalieri Cremona, Italy Azienda Ospedaliera S.Luigi Orbassano Orbassano, Italy Collapse << |
| NCT00942682 | Neuroendocrine Tumor ... more >> Carcinoid Tumor Pancreatic Neuroendocrine Tumor Collapse << | Phase 1 | Completed | - | United States, Massachusetts ... more >> Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00955721 | Cholangiocarcinoma ... more >> Biliary Tract Cancer Gallbladder Cancer Collapse << | Phase 1 Phase 2 | Terminated(Lack of Funding) | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00955721 | - | - | Terminated(Lack of Funding) | - | - |
| NCT01005199 | Liver Cancer | Phase 2 | Completed | - | Austria ... more >> Medizinische Universität Wien Wien, Austria, 1090 Hungary Szent Laszlo Korhaz Budapest, Hungary, 1097 Switzerland Saint Claraspital AG Basel, Switzerland, CH-4016 Clinical Cancer Research Center at University Hospital Basel Basel, Switzerland, CH-4031 Istituto Oncologico della Svizzera Italiana - Ospedale Regionale Bellinzona e Valli Bellinzona, Switzerland, 6500 Inselspital Bern Bern, Switzerland, CH-3010 Kantonsspital Bruderholz Bruderholz, Switzerland, CH-4101 Hopital Cantonal Universitaire de Geneve Geneva, Switzerland, CH-1211 Centre Hospitalier Universitaire Vaudois Lausanne, Switzerland, CH-1011 Kantonsspital Liestal Liestal, Switzerland, CH-4410 CHCVS - Hôpital de Sion Sion, Switzerland, 1950 Kantonsspital - St. Gallen St. Gallen, Switzerland, CH-9007 Regionalspital Thun, Switzerland, 3600 City Hospital Triemli Zurich, Switzerland, CH-8063 UniversitaetsSpital Zuerich Zurich, Switzerland, CH-8091 Collapse << |
| NCT01008566 | Adult Hepatocellular Carcinoma... more >> Advanced Adult Hepatocellular Carcinoma Localized Non-Resectable Adult Liver Carcinoma Recurrent Adult Liver Carcinoma Collapse << | Phase 1 | Completed | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 USC / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 United States, Pennsylvania Penn State Hershey Cancer Institute-Clinical Trials Office Hershey, Pennsylvania, United States, 17033-0850 University of Pittsburgh Cancer Institute (UPCI) Pittsburgh, Pennsylvania, United States, 15232 University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00303797 | Refractory Chronic Lymphocytic... more >> Leukemia Refractory Multiple Myeloma Stage III Multiple Myeloma Stage IV Chronic Lymphocytic Leukemia Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00941863 | - | - | Completed | - | - |
| NCT01344525 | Obesity Weigh... more >>t Loss Collapse << | Not Applicable | Recruiting | May 2029 | Germany ... more >> University of Heidelberg Recruiting Heidelberg, Germany, 69120 University of Hohenheim Recruiting Stuttgart, Germany, 70599 University of Tübingen Recruiting Tübingen, Germany, 72076 Collapse << |
| NCT00989469 | Metastatic Colorectal Cancer | Phase 1 Phase 2 | Completed | - | France ... more >> Centre Oscar Lambret Lille, France Hopital Saint Eloi Montpellier, France Centre Rene Gauducheau Nantes, France Centre Antoine Lacassagne Nice, France CHU Robert Debre Reims, France Collapse << |
| NCT01620216 | Acute Myeloid Leukemia Arising... more >> From Previous Myelodysplastic Syndrome Chronic Myelomonocytic Leukemia Myelodysplastic Syndrome Recurrent Adult Acute Lymphoblastic Leukemia Recurrent Adult Acute Myeloid Leukemia Refractory Adult Acute Lymphoblastic Leukemia Collapse << | Phase 2 | Recruiting | March 30, 2019 | United States, Maryland ... more >> National Heart Lung and Blood Institute Not yet recruiting Bethesda, Maryland, United States, 20824 Contact: Christopher S. Hourigan 301-803-8029 hourigan@nih.gov Principal Investigator: Christopher S. Hourigan United States, Oregon OHSU Knight Cancer Institute Not yet recruiting Portland, Oregon, United States, 97239 Contact: Marc Loriaux 503-494-9383 loriauxm@ohsu.edu Principal Investigator: Marc Loriaux United States, Texas UT Southwestern/Simmons Cancer Center-Dallas Recruiting Dallas, Texas, United States, 75390 Contact: Robert H. Collins 214-648-4155 robert.collins@utsouthwestern.edu Principal Investigator: Robert H. Collins United States, Utah Huntsman Cancer Institute/University of Utah Not yet recruiting Salt Lake City, Utah, United States, 84112 Contact: Tibor J. Kovacsovics 801-585-3229 tibor.kovacsovics@hci.utah.edu Principal Investigator: Tibor J. Kovacsovics Collapse << |
| NCT01008917 | Hepatocellular Carcinoma | Phase 1 | Completed | - | United States, California ... more >> University of California San Francisco San Francisco, California, United States, 94143 United States, Illinois Robert H. Lurie Comprehensive Cancer Center of Northwestern University Chicago, Illinois, United States, 60611 Collapse << |
| NCT00865709 | Metastatic Colorectal Cancer | Phase 2 | Completed | - | - |
| NCT00826540 | Recurrent Colon Cancer ... more >> Recurrent Rectal Cancer Stage IV Colon Cancer Stage IV Rectal Cancer Collapse << | Phase 2 | Completed | - | - |
| NCT00826540 | - | - | Completed | - | - |
| NCT03058913 | - | - | Recruiting | December 2025 | United States, Illinois ... more >> Outpatient Endovascular Interventional Society (OEIS) Recruiting Hoffman Estates, Illinois, United States, 60169 Contact: Jeff Carr 903-521-6004 jeffcarr@me.com Collapse << |
| NCT01574313 | Vertigo Menie... more >>re Disease Collapse << | Phase 4 | Completed | - | Taiwan ... more >> i Mei Medical Center Tainan, Taiwan, Taiwan Tainan, Taiwan, 70014 Collapse << |
| NCT02728050 | Acute Biphenotypic Leukemia ... more >> Acute Myeloid Leukemia de Novo Myelodysplastic Syndrome High Risk Myelodysplastic Syndrome Myelodysplastic Syndrome Myeloproliferative Neoplasm Collapse << | Phase 1 Phase 2 | Recruiting | - | United States, Washington ... more >> Fred Hutch/University of Washington Cancer Consortium Recruiting Seattle, Washington, United States, 98109 Contact: Anna Halpern 206-314-5944 halpern2@uw.edu Principal Investigator: Anna Halpern Collapse << |
| NCT00865709 | - | - | Completed | - | - |
| NCT02721173 | Acne Vulgaris | Phase 4 | Completed | - | India ... more >> AIIMS, Bhubaneswar Bhubaneswar, Odisha, India, 751019 Collapse << |
| NCT00096200 | Recurrent Fallopian Tube Carci... more >>noma Recurrent Ovarian Carcinoma Recurrent Primary Peritoneal Carcinoma Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Moffitt Cancer Center at Tampa General Hospital Tampa, Florida, United States, 33612 Moffitt Cancer Center Tampa, Florida, United States, 33612 United States, Ohio Case Western Reserve University Cleveland, Ohio, United States, 44106 Lake University Ireland Cancer Center Mentor, Ohio, United States, 44060 United States, Virginia Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT00096200 | - | - | Completed | - | - |
| NCT00111007 | Melanoma | Phase 3 | Completed | - | - |
| NCT00110019 | - | - | Completed | - | - |
| NCT00281957 | Recurrent Melanoma ... more >> Stage IV Melanoma Collapse << | Phase 2 | Completed | - | - |
| NCT00110019 | Mucosal Melanoma ... more >> Recurrent Melanoma Stage IIIA Skin Melanoma Stage IIIB Skin Melanoma Stage IIIC Skin Melanoma Stage IV Skin Melanoma Collapse << | Phase 3 | Completed | - | - |
| NCT00300885 | Carcinoma, Non-Small Cell Lung | Phase 3 | Terminated(Based on the result... more >>s of the interim analysis, it was determined that the study would not meet its primary efficacy endpoint and the study was terminated early.) Collapse << | - | - |
| NCT00281957 | - | - | Completed | - | - |
| NCT00329641 | Ciliary Body and Choroid Melan... more >>oma, Medium/Large Size Extraocular Extension Melanoma Iris Melanoma Metastatic Intraocular Melanoma Recurrent Intraocular Melanoma Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Southwest Oncology Group San Antonio, Texas, United States, 78245 Collapse << |
| NCT03031821 | Prostate Cancer ... more >> Metabolic Syndrome Collapse << | Phase 3 | Not yet recruiting | June 1, 2023 | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Cancer Centre Not yet recruiting Vancouver, British Columbia, Canada, V5Z 4E6 Contact: Bernie Eigl, MD 604-877-6000 bernie.eigl@bccancer.bc.ca Principal Investigator: Bernie Eigl, MD Collapse << |
| NCT00329641 | - | - | Completed | - | - |
| NCT00111007 | - | - | Completed | - | - |
| NCT00369070 | Advanced Non-squamous NSCLC | Phase 2 | Terminated | - | - |
| NCT00390611 | - | - | Completed | - | - |
| NCT00300885 | - | - | Terminated(Based on the result... more >>s of the interim analysis, it was determined that the study would not meet its primary efficacy endpoint and the study was terminated early.) Collapse << | - | - |
| NCT00474786 | Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT00417248 | Non-Small Cell Lung Cancer | Phase 1 Phase 2 | Terminated(Negative sorafenib ... more >>results from ESCAPE trial and safety concerns of regimen) Collapse << | - | United States, Illinois ... more >> Medical & Surgical Specialists, LLC Galesburg, Illinois, United States, 61401 United States, Indiana Fort Wayne Oncology & Hematology, Inc Fort Wayne, Indiana, United States, 46815 Center for Cancer Care at Goshen Health System Goshen, Indiana, United States, 46527 Indiana University Cancer Center Indianapolis, Indiana, United States, 46202 Horizon Oncology Center Lafayette, Indiana, United States, 47905 Medical Consultants, P.C. Muncie, Indiana, United States, 47303 Northern Indiana Cancer Research Consortium South Bend, Indiana, United States, 46601 United States, Ohio Oncology Partners Network Cincinnati, Ohio, United States, 45247 Collapse << |
| NCT00390611 | Ovarian Cancer | Phase 2 | Completed | - | United States, Arkansas ... more >> Northeast Arkansas Clinic Jonesboro, Arkansas, United States, 72401 United States, Florida Florida Cancer Specialists Fort Myers, Florida, United States, 33901 Holy Cross Hospital Ft. Lauderdale, Florida, United States, 33308 Gulfcoast Oncology Associates St. Petersburg, Florida, United States, 33705 United States, Georgia Medical College of Georgia Cancer Specialists Augusta, Georgia, United States, 30912 United States, Indiana Providence Medical Group Terre Haute, Indiana, United States, 47802 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 National Capital Clinical Research Consortium Bethesda, Maryland, United States, 20817 United States, Michigan St. Joseph Mercy Hospital Ann Arbor, Michigan, United States, 48106 Grand Rapids Clinical Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, Oklahoma University of Oklahoma Oklahoma City, Oklahoma, United States, 73104 United States, South Carolina South Carolina Oncology Associates, PA Columbia, South Carolina, United States, 29210 United States, Tennessee Tennessee Valley Clinical Research Chattanooga, Tennessee, United States, 37411 Family Cancer Center Collierville, Tennessee, United States, 38017 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 United States, Virginia Peninsula Cancer Center Newport News, Virginia, United States, 23601 Collapse << |
| NCT00483301 | Malignant Melanoma | Phase 2 | Completed | - | United States, California ... more >> Dr. Steven O'Day Los Angeles, California, United States, 90025 United States, Florida Mt. Sinai Medical Center Miami Beach, Florida, United States, 33140 Collapse << |
| NCT00493636 | Breast Cancer | Phase 2 | Completed | - | - |
| NCT00474786 | - | - | Completed | - | - |
| NCT00417248 | - | - | Terminated(Negative sorafenib ... more >>results from ESCAPE trial and safety concerns of regimen) Collapse << | - | - |
| NCT00494182 | Metastatic Head and Neck Squam... more >>ous Cell Carcinoma Metastatic Squamous Cell Carcinoma of the Hypopharynx Metastatic Squamous Cell Carcinoma of the Larynx Metastatic Squamous Cell Carcinoma of the Oral Cavity Metastatic Squamous Cell Carcinoma of the Oropharynx Recurrent Head and Neck Squamous Cell Carcinoma Recurrent Hypopharyngeal Squamous Cell Carcinoma Recurrent Laryngeal Squamous Cell Carcinoma Recurrent Oral Cavity Squamous Cell Carcinoma Recurrent Oropharyngeal Squamous Cell Carcinoma Collapse << | Phase 2 | Active, not recruiting | April 30, 2019 | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00493636 | - | - | Completed | - | - |
| NCT00547443 | Lung Cancer | Phase 1 Phase 2 | Terminated(Withdrawn as the sp... more >>onsor has stopped the drug for NSCLC population) Collapse << | - | United States, Texas ... more >> Arlington Cancer Center - Arlington Arlington, Texas, United States, 76012-2510 Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center - Dallas Dallas, Texas, United States, 75390 Collapse << |
| NCT00572078 | SOLID TUMORS | Phase 1 | Completed | - | United States, Indiana ... more >> Indiana University Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00558636 | - | - | Terminated(Terminated early wh... more >>en the results from Study NCT00300885 showed an overall lack of efficacy in NSCLC and increased mortality in subjects with squamous subtype.) Collapse << | - | - |
| NCT00562913 | Cancer | Phase 1 | Completed | - | Canada, Alberta ... more >> Edmonton, Alberta, Canada, T6G 1Z2 Canada, Quebec Montreal, Quebec, Canada, H3T 1E2 Collapse << |
| NCT00573755 | Breast Cancer | Phase 2 | Terminated(lack of participant... more >> accrual) Collapse << | - | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 55259 United States, Florida Mayo Clinic in Florida Jacksonville, Florida, United States, 32224 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00573755 | - | - | Terminated(lack of participant... more >> accrual) Collapse << | - | - |
| NCT00589550 | - | - | Terminated(low accrual) | - | - |
| NCT00606125 | Neoplasms | Phase 1 | Completed | - | United States, Connecticut ... more >> New Haven, Connecticut, United States, 06511-5991 United States, Texas San Antonio, Texas, United States, 78229-3307 Collapse << |
| NCT00726986 | Lung Cancer | Phase 2 | Terminated(Extreme toxicity) | - | United States, New York ... more >> Columbia Presbyterian New York, New York, United States, 10032 United States, Ohio Lake/University Ireland Cancer Center Cleveland, Ohio, United States, 44060 Case Medical Center, University Hospitals Seidman Cancer Center, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44106-5065 MetroHealth Medical Center Cleveland, Ohio, United States, 44109 CCF-Fairview Hospital Cleveland, Ohio, United States, 44111 UHHS Chagrin Highlands Medical Center Cleveland, Ohio, United States, 44122 Southwest General Health Center Cleveland, Ohio, United States, 44130 UHHS Westlake Medical Center Cleveland, Ohio, United States, 44145 Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 UH-Monarch Mayfield Heights, Ohio, United States, 44124 UH-Firelands Sandusky, Ohio, United States, 44870 Collapse << |
| NCT00822848 | Sarcoma | Phase 1 | Completed | - | United States, Oregon ... more >> OHSU Knight Cancer Institute Portland, Oregon, United States, 97239-3098 Collapse << |
| NCT00558636 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Terminated(Terminated early wh... more >>en the results from Study NCT00300885 showed an overall lack of efficacy in NSCLC and increased mortality in subjects with squamous subtype.) Collapse << | - | China, Guangdong ... more >> Guangzhou, Guangdong, China, 510060 Guangzhou, Guangdong, China, 510515 China, Hong Kong Shatin, Hong Kong, China China, Jiangsu Nanjing, Jiangsu, China, 210003 China, Zhejiang Hangzhou, Zhejiang, China, (310022), Hangzhou, Zhejiang, China, 310016 China Beijing, China, 100021 Beijing, China, 100142 Beijing, China, 100730 Chongqing, China, 400038 Shanghai, China, 200032 Shanghai, China, 200433 India Mumbai, Maharashtra, India, 400012 New- Delhi, India, 110008 Korea, Republic of Gyeonggi-do, Korea, Republic of, 410-769 Seoul, Korea, Republic of, 136-705 Singapore Singapore, Singapore, 119228 Singapore, Singapore, 169610 Taiwan Changhua, Taiwan, 500 Taipei, Taiwan, 100 Thailand Bangkok, Thailand, 10330 Bangkok, Thailand Collapse << |
| NCT00726986 | - | - | Terminated(Extreme toxicity) | - | - |
| NCT00861419 | Advanced Solid Tumors | Phase 1 | Completed | - | - |
| NCT00981162 | Acinar Cell Adenocarcinoma of ... more >>the Pancreas Duct Cell Adenocarcinoma of the Pancreas Recurrent Pancreatic Cancer Stage IV Pancreatic Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado at Denver Health Sciences Center Aurora, Colorado, United States, 80045 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT00889343 | Colorectal Neoplasms | Phase 2 | Terminated | - | - |
| NCT00542971 | AML Acute Mye... more >>loid Leukemia Myelodysplastic Disorders Collapse << | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> The University of Texas M.D. Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00623402 | Melanoma | Phase 2 | Completed | - | Germany ... more >> Dpt. of Dermatology, Humboldt University Berlin, Germany, 10117 Dept. of Dermatology, Elbe Klinikum Buxtehude, Germany, 21614 Dpt. of Dermatology, University of Hannover Hannover, Germany, 30449 Dpt. of Dermatology, University of Homburg/Saar Homburg/Saar, Germany, 66421 Dpt. of Dermatology; UK-SH Campus Kiel, Germany Kiel, Germany, D-24105 Dpt. of Dermatology, University of Cologne Koeln, Germany, D-50937 Dpt. of Dermatology, University of Mannheim Mannheim, Germany, 68163 Dpt. of Dermatology, Ludwig-Maximilian-University München, Germany, 80337 Dpt. of Dermatology, University of Tübingen Tübingen, Germany, 72076 Dpt. of Dermatology, University of Würzburg Würzburg, Germany, 97080 Collapse << |
| NCT00589550 | Kidney Cancer | Phase 1 | Terminated(low accrual) | - | United States, Ohio ... more >> Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00542971 | - | - | Completed | - | - |
| NCT01171482 | Carcinoma, Hepatocellular | Phase 2 | Terminated(There were problems... more >> with national reimbursement policies.) Collapse << | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Korea, Republic of Collapse << |
| NCT01194869 | - | - | Terminated(Slow accrual, avail... more >>ability of other clinical options) Collapse << | - | - |
| NCT00667589 | Hand-foot Skin Reaction ... more >> Rash Collapse << | Phase 2 | Terminated(Study was halted du... more >>e to poor subject accrual.) Collapse << | - | United States, Illinois ... more >> Northwestern University Feinberg School of Medicine, Department of Dermatology Chicago, Illinois, United States, 60611 Collapse << |
| NCT00667589 | - | - | Terminated(Study was halted du... more >>e to poor subject accrual.) Collapse << | - | - |
| NCT00924807 | Prostate Cancer | Phase 1 Phase 2 | Terminated(Sponsor closed the ... more >>trial) Collapse << | - | United States, New York ... more >> St.Luke's-Roosevelt Hospital Center New York, New York, United States, 10019 Collapse << |
| NCT01194869 | Breast Cancer | Phase 2 | Terminated(Slow accrual, avail... more >>ability of other clinical options) Collapse << | - | United States, Georgia ... more >> Grady Memorial Hospital Atlanta, Georgia, United States, 30303 Emory University Hospital Midtown Atlanta, Georgia, United States, 30308 Emory University Winship Cancer Institute Atlanta, Georgia, United States, 30322 Collapse << |
| NCT02050919 | Pleomorphic Rhabdomyosarcoma ... more >> Stage IIB Adult Soft Tissue Sarcoma AJCC v7 Stage III Adult Soft Tissue Sarcoma AJCC v7 Stage IV Adult Soft Tissue Sarcoma AJCC v7 Collapse << | Phase 2 | Active, not recruiting | December 31, 2018 | United States, Oregon ... more >> OHSU Knight Cancer Institute Portland, Oregon, United States, 97239 Collapse << |
| NCT00924807 | - | - | Terminated(Sponsor closed the ... more >>trial) Collapse << | - | - |
| NCT01616186 | Locally Metastatic Malignant N... more >>eoplasm Collapse << | Phase 2 | Withdrawn | April 2016 | - |
| NCT00722072 | Breast Cancer | Phase 2 | Terminated(Sponsor pulled fund... more >>ing and low accrual) Collapse << | - | United States, Oregon ... more >> OHSU Knight Cancer Institute Portland, Oregon, United States, 97239-3098 Collapse << |
| NCT01861314 | Acute Myeloid Leukemia ... more >> Recurrent Adult Acute Myeloid Leukemia Secondary Acute Myeloid Leukemia Therapy-Related Acute Myeloid Leukemia Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 1 | Active, not recruiting | - | United States, Ohio ... more >> Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT00722072 | - | - | Terminated(Sponsor pulled fund... more >>ing and low accrual) Collapse << | - | - |
| NCT01222676 | Bladder Cancer | Phase 2 | Unknown | - | Italy ... more >> Fondazione Istituto Nazionale dei Tumori Recruiting Milan, Italy, 20133 Contact: Contact Person 39-2-2390-2359 roberto.salvioni@istitutotumori.mi.it Collapse << |
| NCT03406793 | Diarrhea Stun... more >>ting Collapse << | Not Applicable | Recruiting | January 30, 2020 | Bangladesh ... more >> Icddr,B Recruiting Dhaka, Bangladesh Contact: Munir Islam, MBBS, PhD 880 2 982 7001-10 ext 2352 mislam@icddrb.org Collapse << |
| NCT03434379 | Carcinoma, Hepatocellular | Phase 3 | Recruiting | June 29, 2022 | - |
| NCT02259647 | Hepatocellular Carcinoma | Not Applicable | Unknown | June 2016 | India ... more >> Institute of Liver & Biliary Sciences Recruiting New Delhi, Delhi, India, 110070 Contact: Dr Devraj R, MD 011-46300000 drdevurgowda@gmail.com Principal Investigator: Dr Devraja R, MD Collapse << |
| NCT01817751 | Glioblastoma ... more >>Recurrent Adult Brain Neoplasm Malignant Glioma WHO Grade III Glioma Collapse << | Phase 2 | Recruiting | March 31, 2019 | United States, Virginia ... more >> Virginia Commonwealth University/Massey Cancer Center Recruiting Richmond, Virginia, United States, 23298 Contact: Alison A Ryan, NP 804-628-1936 masseysiit@vcu.edu Principal Investigator: Mark G Malkin, MD Collapse << |
| NCT01991990 | Healthy Volunteer | Phase 4 | Completed | - | United Kingdom ... more >> Cambridge, United Kingdom, CB2 0QQ Collapse << |
| NCT03517995 | Bladder Cancer ... more >> Bladder Tumor Urothelial Carcinoma Collapse << | Phase 2 | Not yet recruiting | July 2022 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Kayoko Kennedy 813-745-1947 kayoko.kennedy@moffitt.org Contact: Nagi Kumar, Ph.D. 813-745-6885 nagi.kumar@moffitt.org James A. Haley Veteran's Administration Hospital Not yet recruiting Tampa, Florida, United States, 33612 Contact: Cesar Ercole, M.D. 813-910-4088 cesar.ercole1@va.gov Principal Investigator: Cesar Ercole, M.D. Collapse << |
| NCT02210962 | Schizophrenia | Phase 4 | Unknown | February 2015 | Poland ... more >> Department of Affective and Psychotic Disorders Medical University of Lodz Lodz, Poland, 92216 Collapse << |
| NCT01991990 | - | - | Completed | - | - |
| NCT03226340 | Essential Hypertension | Phase 4 | Recruiting | March 31, 2018 | Korea, Republic of ... more >> Hallym University Medical Center Recruiting Anyang, Dongan-gu, Korea, Republic of, 14068 Contact: Sang-Ho Jo 82-31-380-3722 sophi5neo@gmail.com Kyung Hee University Hospital Recruiting Seoul, Dongdaemun-gu, Korea, Republic of, 02447 Contact: Soo-Joong Kim 82-2-958-8180 soojoong@dreamwiz.com Hallym University Medical Center Recruiting Seoul, Gangdong-gu,, Korea, Republic of, 05355 Contact: Kyoo-Rok Han 82-2-2224-2405 krheart@hallym.or.kr Samsung Medical Center Recruiting Seoul, Gangnam-Gu, Korea, Republic of, 06351 Contact: Sung-Ji Park 82-2-3410-0887 tyche.park@gmail.com Korea Univ. Guro Hospital Recruiting Seoul, Guro, Korea, Republic of, 08308 Contact: Eung-ju Kim, MD,PhD 82-8-2626-3022 withnoel@empas.com Seoul National University Hospital Recruiting Seoul, Jongno-gu, Korea, Republic of, 03080 Contact: Hyun-Jai Cho 82-2-2072-3931 hyunjaicho@snu.ac.kr Hanyang University Medical Center Recruiting Seoul, Seongdong-gu, Korea, Republic of, 04763 Contact: Jin-Ho Shin 82-2-2290-8308 jhs2003@hanyang.ac.kr Asan Medical Center Recruiting Seoul, Songpa-gu, Korea, Republic of, 05505 Contact: Jong-Min Song 82-2-3010-3158 jmsong@amc.seoul.kr Ajou Univ. Medical Center Recruiting Suwon-si, Yeong-tong, Korea, Republic of, 16499 Contact: Joon-han Shin, MD,PhD 82-31-219-5710 shinjh@ajou.ac.kr Seoul National University Bundang Hospital Recruiting Seongnam, Korea, Republic of, 463707 Contact: Dong-Ju Choi, MD, PhD 82-31-787-7007 djchoi@snubh.org Contact: Jin Joo Park, MD 82-31-787 ext 7074 jinjooparkmd@gmail.com Collapse << |
| NCT00289900 | - | - | Completed | - | - |
| NCT03493802 | - | - | Recruiting | November 1, 2020 | United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Kathleen R Leistikow, ACRC 507-266-1316 leistikow.kathleen@mayo.edu Contact: Charles D Madsen, CCRP 507-266-9391 madsen.charles@mayo.edu Principal Investigator: Maria V Irazabal, M.D. Collapse << |
| NCT00098618 | Clear Cell Renal Cell Carcinom... more >>a Papillary Renal Cell Carcinoma Recurrent Renal Cell Cancer Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 2 | Terminated | - | United States, North Carolina ... more >> Duke University Medical Center Durham, North Carolina, United States, 27710 Collapse << |
| NCT00289900 | Mixed Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT00101907 | Lung Cancer P... more >>ancreatic Cancer Esophageal Cancer Collapse << | Phase 1 | Terminated(Study terminated pe... more >>r recommendation of Data Review Team) Collapse << | - | - |
| NCT00101114 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Cancer Stage III Renal Cell Cancer Stage IV Renal Cell Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Southwest Oncology Group San Antonio, Texas, United States, 78245 Collapse << |
| NCT00101907 | - | - | Terminated(Study terminated pe... more >>r recommendation of Data Review Team) Collapse << | - | - |
| NCT00117637 | Carcinoma, Renal Cell | Phase 2 | Completed | - | - |
| NCT00121251 | Recurrent Renal Cell Carcinoma... more >> Stage III Renal Cell Cancer AJCC v7 Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 1 Phase 2 | Completed | - | United States, New York ... more >> Montefiore Medical Center - Moses Campus Bronx, New York, United States, 10467 Laura and Isaac Perlmutter Cancer Center at NYU Langone New York, New York, United States, 10016 Mount Sinai Hospital New York, New York, United States, 10029 Weill Medical College of Cornell University New York, New York, United States, 10065 Collapse << |
| NCT00253370 | Adenocarcinoma of the Gastroes... more >>ophageal Junction Metastatic Gastric Cancer Advanced Unresectable Gastric Cancer Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Eastern Cooperative Oncology Group Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00126594 | - | - | Completed | - | - |
| NCT00108953 | - | - | Completed | - | - |
| NCT00253370 | - | - | Completed | - | - |
| NCT00126594 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Carcinoma Stage IV Renal Cell Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00108953 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | - |
| NCT00265798 | Gastrointestinal Stromal Tumor | Phase 2 | Active, not recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 United States, Illinois University of Chicago Comprehensive Cancer Center Chicago, Illinois, United States, 60637 Decatur Memorial Hospital Decatur, Illinois, United States, 62526 Central Illinois Hematology Oncology Center Springfield, Illinois, United States, 62702 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00255658 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> Cancer Therapy and Research Center at The UT Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00265798 | - | - | Active, not recruiting | - | - |
| NCT00322400 | Locally Recurrent and Metastat... more >>ic Breast Cancer Collapse << | Phase 1 | Completed | - | - |
| NCT00117637 | - | - | Completed | - | - |
| NCT00329719 | Adult Glioblastoma ... more >> Adult Gliosarcoma Recurrent Adult Brain Neoplasm Collapse << | Phase 1 Phase 2 | Completed | - | - |
| NCT00352859 | Carcinoma, Renal Cell | Phase 4 | Terminated | - | United States, Missouri ... more >> Kansas City, Missouri, United States Collapse << |
| NCT00356681 | Breast Neoplasms ... more >> Breast Tumors Breast Cancer Locally Recurrent and Metastatic Breast Cancer Collapse << | Phase 2 | Terminated(Sponsor decision to... more >> close study) Collapse << | - | - |
| NCT00349206 | Melanoma Recu... more >>rrent Melanoma Stage III Melanoma Stage IV Melanoma Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00392821 | Kidney Cancer | Phase 1 Phase 2 | Completed | - | United States, Florida ... more >> Florida Hospital Cancer Institute Orlando, Florida, United States, 32804 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 Wellstar Cancer Research Marietta, Georgia, United States, 30060 United States, Kentucky Baptist Hospital East Louisville, Kentucky, United States, 40207 Norton Cancer Institute Louisville, Kentucky, United States, 40207 United States, Missouri Research Medical Center Kansas City, Missouri, United States, 64132 United States, New Jersey Hematology Oncology Associates of Northern NJ Morristown, New Jersey, United States, 07960 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, South Carolina South Carolina Oncology Associates, PA Columbia, South Carolina, United States, 29210 Spartanburg Regional Medical Center Spartanburg, South Carolina, United States, 29303 United States, Tennessee Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 Collapse << |
| NCT00430235 | Adenocarcinoma of the Prostate | Phase 2 | Completed | - | Canada, British Columbia ... more >> BC Cancer Agency - Vancouver Centre Vancouver, British Columbia, Canada, V5Z 4E6 Collapse << |
| NCT00392821 | - | - | Completed | - | - |
| NCT00418496 | Renal Cancer ... more >>Melanoma Collapse << | Phase 1 | Completed | - | United States, Ohio ... more >> Ohio State University Columbus, Ohio, United States, 43210 Collapse << |
| NCT00329719 | - | - | Completed | - | - |
| NCT00448149 | Carcinoma, Renal Cell | Phase 1 Phase 2 | Completed | - | United States, Texas ... more >> The Methodist Hospital Research Institute Houston, Texas, United States, 77030 Collapse << |
| NCT00449280 | Advanced Solid Tumor | Phase 1 | Completed | - | United States, Illinois ... more >> University of Chicago Chicago, Illinois, United States, 60637 Collapse << |
| NCT00449033 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | - |
| NCT00461851 | Bladder Cancer | Phase 2 | Completed | - | United States, Connecticut ... more >> Yale University, Comprehensive Cancer Center New Haven, Connecticut, United States, 06520 Collapse << |
| NCT00384969 | Renal Cell Carcinoma | Phase 1 | Completed | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94115 Collapse << |
| NCT00461851 | - | - | Completed | - | - |
| NCT00473486 | Carcinoma, Non-Small-Cell Lung | Phase 1 Phase 2 | Terminated(Sorafenib administe... more >>red in the combination with pemetrexed-carboplatin appears to enhance thrombocytopenia compared to historical data.) Collapse << | - | Germany ... more >> University of Muenster, Dept. of Medicine, Hematology / Oncology Muenster, Germany, 48145 Collapse << |
| NCT00474929 | Lymphoma Mult... more >>iple Myeloma and Plasma Cell Neoplasm Collapse << | Phase 1 Phase 2 | Active, not recruiting | January 2019 | United States, Iowa ... more >> Holden Comprehensive Cancer Center at University of Iowa Iowa City, Iowa, United States, 52242-1002 United States, Minnesota Mayo Clinic Cancer Center Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00496301 | Carcinoma, Renal Cell | Phase 2 | Completed | - | Spain ... more >> Hospital del Mar Barcelona, Spain, 08003 Hospital Vall d´Hebron Barcelona, Spain, 08035 Hospital de Basurto Bilbao, Spain, 48013 Hospital Santiago de Compostela Coruña, Spain, 15706 Hospital Josep Trueta Gerona, Spain, 17007 Hospital Juan Ramón Jiménez Huelva, Spain, 21005 Hospital Clínico Virgen de la Victoria Málaga, Spain, 29010 Clínica Universitaria de Navarra Pamplona, Spain, 31008 Hospital Virgen Macarena Sevilla, Spain, 41009 Hospital Xeral Cies Vigo, Spain, 36204 Collapse << |
| NCT00478374 | Hepatocellular Carcinoma | Phase 1 | Completed | - | Switzerland ... more >> Inselspital Bern, Switzerland, 3010 Collapse << |
| NCT00541840 | Soft Tissue Sarcoma | Phase 1 Phase 2 | Unknown | April 2010 | Spain ... more >> Grupo Geis Recruiting Madrid, Spain, 28001 Contact: Xavier Garcia del Muro, MD 915775281 info@grupogeis.org Principal Investigator: Xavier Garcia del Muro, MD Collapse << |
| NCT00565370 | Advanced Gastric Cancer | Phase 1 Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT00544817 | - | - | Completed | - | - |
| NCT00597493 | Recurrent Glioblastoma Multifo... more >>rme Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> Duke University Health System Durham, North Carolina, United States, 27710 Collapse << |
| NCT00565370 | - | - | Completed | - | - |
| NCT00449033 | - | - | Completed | - | - |
| NCT00474929 | - | - | Active, not recruiting | - | - |
| NCT00597493 | - | - | Completed | - | - |
| NCT00544817 | Glioblastoma Multiforme | Phase 2 | Completed | - | United States, Florida ... more >> Florida Cancer Specialists Fort Myers, Florida, United States, 33901 United States, Georgia Northeast Georgia Medical Center Gainesville, Georgia, United States, 30501 United States, Maryland Center for Cancer and Blood Disorders Bethesda, Maryland, United States, 20817 United States, Michigan Grand Rapids Clinical Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Nebraska Methodist Cancer Center Omaha, Nebraska, United States, 68114 United States, Ohio Oncology Hematology Care Cincinnati, Ohio, United States, 45242 United States, South Carolina Spartanburg Regional Medical Center Spartanburg, South Carolina, United States, 29303 United States, Tennessee Tennessee Oncology Nashville, Tennessee, United States, 37203 United States, Texas South Texas Oncology and Hematology San Antonio, Texas, United States, 78258 United States, Virginia Virginia Cancer Institute Richmond, Virginia, United States, 23235 Collapse << |
| NCT00607438 | Breast Cancer | Phase 2 | Terminated(Low accrual) | - | United States, Ohio ... more >> Veeda Oncology Columbus, Ohio, United States, 43215 Collapse << |
| NCT00499525 | Breast Cancer | Phase 2 | Active, not recruiting | December 2021 | - |
| NCT00622466 | Breast Cancer | Phase 2 | Unknown | December 2015 | United States, Texas ... more >> University of Texas Southwestern Medical Center Dallas, Texas, United States, 75390 Collapse << |
| NCT00634634 | Metastatic Breast Cancer | Phase 1 Phase 2 | Active, not recruiting | May 2019 | United States, District of Col... more >>umbia Georgetown University Hospital Washington, District of Columbia, United States, 20007 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, New Jersey Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08901 Cooper Hospital/University Medical Center Voorhees, New Jersey, United States, 08690 Collapse << |
| NCT00602576 | Melanoma (Skin) | Phase 2 | Unknown | - | United States, Pennsylvania ... more >> Abramson Cancer Center of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104-4283 Collapse << |
| NCT00647426 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center of University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT00634751 | - | - | Completed | - | - |
| NCT00639262 | Brain Metastases ... more >> Primary Brain Tumors Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00678288 | Carcinoma, Renal Cell | Phase 2 | Terminated | - | Austria ... more >> Salzburg, Austria, 5020 Wien, Austria, 1090 France Avignon, France, 84000 Bayonne, France, 64100 Bordeaux, France, 33000 Marseille, France, 13015 Marseille, France, 13385 Nantes, France, 44020 Paris, France, 75014 Paris, France, 75651 Reims, France, 51100 Strasbourg, France, 67000 Toulouse, France, 31052 Vandoeuvre Les Nancy, France, 54000 Ireland Cork, Ireland Dublin, Ireland, 24 Italy Aviano, Pordenone, Italy, 33081 Legnago, Verona, Italy, 37045 Napoli, Italy, 80131 Pavia, Italy, 27100 Perugia, Italy, 06156 Reggio Emilia, Italy, 42100 Poland Gdansk, Poland, 80-219 Lublin, Poland, 20-090 Warszawa, Poland, 04-141 Wroclaw, Poland, 50 - 556 Spain Barcelona, Spain, 08035 Madrid, Spain, 28041 Madrid, Spain, 28046 Oviedo, Spain, 33006 Pamplona, Spain, 31008 Valencia, Spain, 46009 United Kingdom Northwood, Middlesex, United Kingdom, HA6 2RN Newcastle Upon Tyne, Tyne and Wear, United Kingdom, NE4 6BE London, United Kingdom, SW3 6JJ Collapse << |
| NCT00665990 | Refractory Solid Tumors ... more >> Leukemia Collapse << | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT00696696 | - | - | Completed | - | - |
| NCT00634751 | Pancreatic Neoplasms ... more >> Bile Duct Neoplasms Collapse << | Phase 1 Phase 2 | Completed | - | United States, Wisconsin ... more >> University of Wisconsin Madison, Wisconsin, United States, 53792 Collapse << |
| NCT00703638 | Breast Cancer ... more >> Colorectal Cancer Head and Neck Cancer Lung Cancer Mesothelioma Pancreatic Cancer Prostate Cancer Sarcoma Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Masonic Cancer Center, University of Minnesota Minneapolis, Minnesota, United States, 55455 Collapse << |
| NCT00678288 | - | - | Terminated | - | - |
| NCT00714948 | - | - | Terminated(Lack of accrual) | - | - |
| NCT00714948 | Bladder Cancer ... more >> URINARY BLADDER Collapse << | Phase 2 | Terminated(Lack of accrual) | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00696696 | Pancreatic Cancer | Phase 2 | Completed | - | United States, California ... more >> Desert Regional Medical Center Palm Springs, California, United States, 92262 United States, New York Bellevue Hospital New York, New York, United States, 10016 New York University Cancer Center New York, New York, United States, 10016 Collapse << |
| NCT01015833 | Advanced Adult Hepatocellular ... more >>Carcinoma Recurrent Hepatocellular Carcinoma Stage III Hepatocellular Carcinoma AJCC v7 Stage IIIA Hepatocellular Carcinoma AJCC v7 Stage IIIB Hepatocellular Carcinoma AJCC v7 Stage IIIC Hepatocellular Carcinoma AJCC v7 Stage IV Hepatocellular Carcinoma AJCC v7 Stage IVA Hepatocellular Carcinoma AJCC v7 Stage IVB Hepatocellular Carcinoma AJCC v7 Unresectable Hepatocellular Carcinoma Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01025453 | Thyroid Cancer | Phase 2 | Completed | - | United States, New Jersey ... more >> Memorial Sloan-Kettering at Basking Ridge Basking Ridge, New Jersey, United States, 07920 United States, New York Memorial Sloan-Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan-Kettering at Mercy Medical Center Rockville Centre, New York, United States Memorial Sloan-Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01091207 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135710 Collapse << |
| NCT01015833 | - | - | Active, not recruiting | - | - |
| NCT00424385 | Prostate Cancer | Phase 1 | Completed | - | United States, Illinois ... more >> Oncology Specialists, S.C Niles, Illinois, United States, 60714 Oncology Specialists, S.C Park Ridge, Illinois, United States, 60068 Collapse << |
| NCT00734526 | Glioblastoma ... more >>Gliosarcoma Collapse << | Phase 1 | Terminated | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01141309 | Thyroid Cancer | Phase 2 | Active, not recruiting | June 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01025453 | - | - | Completed | - | - |
| NCT00424385 | - | - | Completed | - | - |
| NCT01214343 | Advanced Hepatocellular Carcin... more >>oma Carcinoma Carcinoma, Hepatocellular Liver Neoplasms Neoplasms Collapse << | Phase 3 | Unknown | September 2013 | Japan ... more >> National Cancer Center Hospital East Recruiting Kashiwa, Chiba, Japan, 277-8577 Contact: Masafumi Ikeda, Dr. Principal Investigator: Masafumi Ikeda, Dr. Kurume University Medical Center Recruiting Kurume, Fukuoka, Japan, 839-0863 Contact: Masatoshi Tanaka, Professor Principal Investigator: Masatoshi Tanaka, Professor Ogaki Municipal Hospital Recruiting Ogaki, Gifu, Japan, 503-8502 Contact: Takashi Kumada, Dr Principal Investigator: Takashi Kumada, Dr. Sapporo Medical University Recruiting Sapporo, Hokkaido, Japan, 060-8556 Contact: Junji Kato, Dr. Principal Investigator: Junji Kato, Dr. Sapporo-Kosei General Hospital Recruiting Sapporo, Hokkaido, Japan, 060-8556 Contact: Takumi Ohmura, Dr. Principal Investigator: Takumi Ohmura, Dr. Japanese Red Cross Takamatsu Hospital Recruiting Takamatsu, Kagawa, Japan, 760-0017 Contact: Chikara Ogawa, Dr. Principal Investigator: Chikara Ogawa, Dr. Mie University Hospital Recruiting Tsu, Mie, Japan, 514-8507 Contact: Katsuya Shiraki, Dr. Principal Investigator: Katsuya Shiraki, Dr. National Hospital Organization Nagasaki Medical Center Recruiting Ohmura, Nagasaki, Japan, 856-8562 Contact: Hiromi Ishibashi, Dr. Principal Investigator: Hiromi Ishibashi, Dr. Kawasaki Medical School Hospital Recruiting Kurashiki, Okayama, Japan, 701-0192 Contact: Keisuke Hino, Dr. Principal Investigator: Keisuke Hino, Dr. Ikeda Municipal Hospital Recruiting Ikeda, Osaka, Japan, 563-8510 Contact: Yasuharu Imai, Dr. Principal Investigator: Yasuharu Imai, Dr. Kinki University Hospital Recruiting Osaka-Sayama, Osaka, Japan, 589-8511 Contact: Masatoshi Kudo, Professor +81-72-366-0221 ext 3149 m-kudo@med.kindai.ac.jp Contact: Kazuomi Ueshima, Dr. +81-72-366-0221 ext 3525 kaz-ues@med.kindai.ac.jp Principal Investigator: Masatoshi Kudo, Professor Sub-Investigator: Kazuomi Ueshima, Dr. Principal Investigator: Kazuto Nishio, Professor Osaka University Hospital Recruiting Suita, Osaka, Japan, 565-0871 Contact: Hiroaki Nagano, Professor Principal Investigator: Hiroaki Nagano, Professor Kyorin University Hospital Recruiting Mitaka, Tokyo, Japan, 181-8611 Contact: Junji Furuse, Professor Principal Investigator: Junji Furuse, Professor Musashino Red Cross Hospital Recruiting Musashino, Tokyo, Japan, 180-8610 Contact: Namiki Izumi, Dr. Principal Investigator: Namiki Izumi, Dr. Juntendo University Nerima Hospital Recruiting Nerima, Tokyo, Japan, 177-0033 Contact: Shigehiro Kokubu, Dr. Principal Investigator: Shigehiro Kokubu, Dr. Yamaguchi University Hospital Recruiting Ube, Yamaguchi, Japan, 755-8505 Contact: Isao Sakaida, Prof Principal Investigator: Isao Sakaida, Professor Chiba University Hospital Recruiting Chiba, Japan, 260-8677 Contact: Fumihiko Kanai, Dr. Principal Investigator: Fumihiko Kanai, Dr. Gifu Municipal Hospital Recruiting Gifu, Japan, 500-8513 Contact: Eiichi Tomita, Dr. Principal Investigator: Eiichi Tomita, Dr. Hiroshima City Hospital Recruiting Hiroshima, Japan, 730-8518 Contact: Yoshiyuki Kobayashi, Dr. Principal Investigator: Yoshiyuki Kobayashi, Dr. Hiroshima University Hospital Recruiting Hiroshima, Japan, 734-8551 Contact: Hiroshi Aikata, Dr. Principal Investigator: Hiroshi Aikata, Dr. Kumamoto University Hospital Recruiting Kumamoto, Japan, 860-8556 Contact: Yutaka Sasaki, Professor Principal Investigator: Yutaka Sasaki, Professor Kyoto University Hospital Recruiting Kyoto, Japan, 606-8507 Contact: Etsuro Hatano, Professor Principal Investigator: Etsuro Hatano, Professor Center for Gastroenterological and Hepatological Diseases Recruiting Miyazaki, Japan, 880-0003 Contact: Hidemori Sakamoto, Dr. Principal Investigator: Hidemori Sakamoto, Dr. Saiseikai Niigata Dai-ni Hospital Recruiting Niigata, Japan, 950-1104 Contact: Toru Ishikawa, Dr. Principal Investigator: Toru Ishikawa, Dr. Niigata University Medical and Dental Hospital Recruiting Niigata, Japan, 951-8520 Contact: Kouhei Akazawa Principal Investigator: Kouhei Akazawa Okayama University Hospital Recruiting Okayama, Japan, 700-8558 Contact: Kazuhide Yamamoto, Professor Principal Investigator: Kazuhide Yamamoto, Professor Osaka Red Cross Hospital Recruiting Osaka, Japan, 543-8555 Contact: Ikuo Osaki, Dr. Principal Investigator: Ikuo Osaki, Dr. The University of Tokushima Faculty of Medicine Recruiting Tokushima, Japan, 770-8503 Contact: Tetsuji Takayama, Dr. Principal Investigator: Tetsuji Takayama, Dr. Kyoundo Hospital Recruiting Tokyo, Japan, 101-0062 Contact: Shuntaro Obi, Dr. Principal Investigator: Shuntaro Obi, Dr. National Cancer Center Hospital Recruiting Tokyo, Japan, 104-0045 Contact: Takushi Okusaka, Dr. Principal Investigator: Takushi Okusaka, Dr. Collapse << |
| NCT00375310 | Pancreatic Cancer ... more >> Adenocarcinoma of the Pancreas Collapse << | Phase 1 | Completed | - | United States, Indiana ... more >> Indiana University Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01226056 | Advanced Solid Tumors | Phase 1 Phase 2 | Suspended(toxicity (protocol a... more >>mendment under approval)) Collapse << | December 2012 | Italy ... more >> Istituto Europeo di Oncologia, Milano, Italy, 20141 Collapse << |
| NCT01187212 | Malignant Neoplasm of Stomach ... more >> Effects of Chemotherapy Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Songpa-gu, Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01262482 | Advanced or Metastatic Gastric... more >> or Gastroesophageal Junction Adenocarcinoma (Relapsed After a Cisplatin Based Treatment) Collapse << | Phase 2 | Completed | - | Spain ... more >> Hospital Clinic de Barcelona Barcelona, Spain Hospital Sant Pau Barcelona, Spain H. Josep Trueta Girona, Spain Centro Oncológico M.D. Anderson Spain Madrid, Spain Hospital de Fuenlabrada Madrid, Spain Hospital La Paz Madrid, Spain Hospital Althaia Manresa, Spain Clínica Universitaria de Navarra Pamplona, Spain Hospital Parc Taulí Sabadell, Spain Hospital General de Valencia Valencia, Spain Collapse << |
| NCT01245582 | Hepatic Neoplasm Malignant | Phase 3 | Withdrawn | August 2015 | - |
| NCT01253070 | - | - | Active, not recruiting | - | - |
| NCT01263951 | Differentiated Thyroid Cancer | Phase 2 | Active, not recruiting | - | United States, Pennsylvania ... more >> Hospital of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01253070 | Acute Myeloid Leukemia ... more >> Acute Myeloid Leukemia (Megakaryoblastic) With t(1;22)(p13.3;q13.1); RBM15-MKL1 Acute Myeloid Leukemia With a Variant RARA Translocation Acute Myeloid Leukemia With Inv(3) (q21.3;q26.2) or t(3;3) (q21.3;q26.2); GATA2, MECOM Acute Myeloid Leukemia With t(6;9) (p23;q34.1); DEK-NUP214 Acute Myeloid Leukemia With t(9;11)(p21.3;q23.3); MLLT3-KMT2A Acute Myeloid Leukemia With Variant MLL Translocations Untreated Adult Acute Myeloid Leukemia Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01320111 | Metastatic Breast Cancer | Phase 2 | Completed | - | Germany ... more >> Germany Multiple Sites All Over Germany Recruiting Multiple Sites, Germany Contact: iOMEDICO AG Freiburg, Germany, 79108 Collapse << |
| NCT01272557 | Liver Carcinoma | Phase 2 | Unknown | December 2014 | Germany ... more >> Medizinische Klinik und Poliklinik, Heinrich-Heine-Universität Düsseldorf, Germany, 40225 Martin-Luther-University Halle-Wittenberg Halle (Saale), Germany, D-06099 Universitätsklinikum des Saarlande Homburg/Saar, Germany, 66421 Ortenau Klinikum Lahr-Ettenheim Lahr, Germany, 77933 Universitätsklinikum Leipzig Leipzig, Germany, 04013 Klinikum Ludwigsburg Ludwigsburg, Germany, 71640 Collapse << |
| NCT01335074 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Withdrawn | - | Germany ... more >> University Hospital Giessen and Marburg Marburg, Germany, 35043 Collapse << |
| NCT01324076 | Liver Cancer | Phase 3 | Unknown | - | United Kingdom ... more >> Queen Elizabeth Hospital at University Hospital of Birmingham NHS Trust Recruiting Birmingham, England, United Kingdom, B15 2TH Contact: Contact Person 44-121-472-1311 Bristol Royal Infirmary Recruiting Bristol, England, United Kingdom, BS2 8HW Contact: Contact Person 44-117-923-0000 Aintree University Hospital Recruiting Liverpool, England, United Kingdom, L9 7AL Contact: Contact Person 44-151-525-5980 Royal Free Hospital Recruiting London, England, United Kingdom, NW3 2QG Contact: Contact Person 44-20-7794-0500 King's College Hospital Recruiting London, England, United Kingdom, SE5 9RS Contact: Contact Person 44-20-3299-9000 Royal Marsden - London Recruiting London, England, United Kingdom, SW3 6JJ Contact: Contact Person 44-20-7352-8171 Queen's Medical Centre Recruiting Nottingham, England, United Kingdom, NG7 2UH Contact: Contact Person 44-115-924-9924 Southampton General Hospital Recruiting Southampton, England, United Kingdom, SO16 6YD Contact: Contact Person 44-23-8079-8751 Collapse << |
| NCT01686204 | Intestinal Barrier Function ... more >> Chronic Inflammation Collapse << | Not Applicable | Completed | - | United States, Wisconsin ... more >> Department of Food Science, Babcock Hall Madison, Wisconsin, United States, 53706 Collapse << |
| NCT01348503 | Hepatocellular Carcinoma | Phase 1 | Terminated(Treatment was ineff... more >>ective) Collapse << | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT00544167 | Breast Cancer | Not Applicable | Completed | - | United States, Florida ... more >> Integrated Community Oncology Network Jacksonville, Florida, United States, 32256 Florida Hospital Cancer Institute Orlando, Florida, United States, 32804 United States, Kentucky Consultants in Blood Disorders and Cancer Louisville, Kentucky, United States, 40207 United States, Michigan Grand Rapids Clinical Oncology Program Grand Rapids, Michigan, United States, 49503 United States, Nebraska Methodist Cancer Center Omaha, Nebraska, United States, 68114 United States, Tennessee Chattanooga Oncology Hematology Associates Chattanooga, Tennessee, United States, 37404 Tennessee Oncology, PLLC Nashville, Tennessee, United States, 37023 United States, Virginia Peninsula Cancer Institute Newport News, Virginia, United States, 23601 Collapse << |
| NCT00543504 | Advanced Cancer | Phase 1 | Active, not recruiting | October 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00573690 | Unspecified Adult Solid Tumor,... more >> Protocol Specific Collapse << | Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Collapse << |
| NCT00687674 | - | - | Terminated(Due to study design... more >> (and toxicity), this trial closed to accrual prior to opening the phase II portion.) Collapse << | - | - |
| NCT00687674 | Multiple Myeloma and Plasma Ce... more >>ll Neoplasm Collapse << | Phase 1 Phase 2 | Terminated(Due to study design... more >> (and toxicity), this trial closed to accrual prior to opening the phase II portion.) Collapse << | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00378703 | Clear Cell Renal Cell Carcinom... more >>a Recurrent Renal Cell Carcinoma Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 2 | Completed | - | - |
| NCT00134069 | Recurrent Colon Cancer ... more >> Recurrent Rectal Cancer Stage III Colon Cancer Stage III Rectal Cancer Stage IV Colon Cancer Stage IV Rectal Cancer Collapse << | Phase 1 | Completed | - | United States, Colorado ... more >> University of Colorado at Denver Health Sciences Center Aurora, Colorado, United States, 80045 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21287-8936 Collapse << |
| NCT01004978 | Hepatocellular Carcinoma ... more >> Unresectable Hepatocellular Carcinoma Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01183663 | Advanced Cancers | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00378703 | - | - | Completed | - | - |
| NCT00533585 | Lung Cancer | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01187199 | Advanced Cancer | Phase 1 | Active, not recruiting | August 2019 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01042041 | Hepatocellular Carcinoma ... more >> Liver Cancer Localized Unresectable Liver Cancer Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> Abramson Cancer Center of The University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01371981 | Acute Myeloid Leukemia ... more >> Leukemia Cutis Myeloid Neoplasm Myeloid Sarcoma Untreated Adult Acute Myeloid Leukemia Untreated Childhood Myeloid Neoplasm Collapse << | Phase 3 | Active, not recruiting | - | - |
| NCT01383343 | Recurrent Colon Carcinoma ... more >> Recurrent Rectal Carcinoma Stage IVA Colon Cancer Stage IVA Rectal Cancer Stage IVB Colon Cancer Stage IVB Rectal Cancer Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 United States, Florida Mayo Clinic in Florida Jacksonville, Florida, United States, 32224-9980 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT02015728 | Recurrent Childhood Brain Tumo... more >>r Collapse << | Not Applicable | Unknown | December 2017 | United States, Washington ... more >> Seattle Children's Seattle, Washington, United States, 98105 Collapse << |
| NCT02559778 | Neuroblastoma | Phase 2 | Recruiting | September 2026 | - |
| NCT00544167 | - | - | Completed | - | - |
| NCT00387751 | Recurrent Melanoma ... more >> Stage III Skin Melanoma Stage IV Skin Melanoma Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> Cancer Therapy and Research Center at The UT Health Science Center at San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT01664182 | Stage III Renal Cell Cancer AJ... more >>CC v7 Stage IV Renal Cell Cancer AJCC v7 Collapse << | Phase 2 | Active, not recruiting | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 USC / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 University of California Davis Comprehensive Cancer Center Sacramento, California, United States, 95817 City of Hope South Pasadena South Pasadena, California, United States, 91030 United States, Michigan Wayne State University/Karmanos Cancer Institute Detroit, Michigan, United States, 48201 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 Metro Minnesota Community Oncology Research Consortium Saint Louis Park, Minnesota, United States, 55416 United States, New Jersey Rutgers Cancer Institute of New Jersey New Brunswick, New Jersey, United States, 08903 United States, North Carolina UNC Lineberger Comprehensive Cancer Center Chapel Hill, North Carolina, United States, 27599 United States, Pennsylvania Penn State Milton S Hershey Medical Center Hershey, Pennsylvania, United States, 17033-0850 University of Pittsburgh Cancer Institute (UPCI) Pittsburgh, Pennsylvania, United States, 15232 United States, Virginia VCU Massey Cancer Center at Hanover Medical Park Mechanicsville, Virginia, United States, 23116 Collapse << |
| NCT03111810 | Iatrogenic Cushing's Disease | Phase 2 | Recruiting | January 2019 | United Kingdom ... more >> University of Oxford Recruiting Oxford, United Kingdom Contact: Jeremy W Tomlinson, MD PhD 44(0)1865 857359 jeremy.tomlinson@ocdem.ox.ac.uk Contact: Nadia Othonos, MD Nantia.Othonos@ocdem.ox.ac.uk Collapse << |
| NCT00387751 | - | - | Completed | - | - |
| NCT02013089 | Gastrointestinal Cancers | Not Applicable | Unknown | December 2017 | China, Guangdong ... more >> Gastrointestinal Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510655 Contact: Yanghong Deng, PhD 008613925106525 13925106525@163.com Principal Investigator: Yanhong Deng, PhD Collapse << |
| NCT03132454 | Hematopoietic/Lymphoid Cancer ... more >> Acute Myeloid Leukemia Acute Lymphoblastic Leukemia Collapse << | Phase 1 | Recruiting | July 2021 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact tkadia@mdanderson.org Collapse << |
| NCT02270788 | Acute Myeloid Leukemia | Phase 1 | Completed | - | United States, Tennessee ... more >> St. Jude Children's Research Hospital Memphis, Tennessee, United States, 38105 Collapse << |
| NCT03247088 | Acute Myeloid Leukemia | Phase 1 Phase 2 | Recruiting | July 2022 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Clinical Research Operations CR_Study_Registration@mdanderson.org Collapse << |
| NCT01754987 | - | - | Completed | - | - |
| NCT02779283 | Acute Lymphoblastic Leukemia ... more >> Acute Myeloid Leukemia Collapse << | Phase 1 | Recruiting | June 1, 2019 | United States, Oregon ... more >> OHSU Knight Cancer Institute Recruiting Portland, Oregon, United States, 97239 Contact: Stephen E. Spurgeon 503-494-8950 spurgeos@ohsu.edu Principal Investigator: Stephen E. Spurgeon Collapse << |
| NCT02010749 | - | - | Completed | - | Philippines ... more >> Asian Hospital and Medical Center Filinvest Corporate City, Alabang,, Muntinlupa City, Philippines, 1780 Collapse << |
| NCT01754987 | Metastatic Hepatocellular Carc... more >>inoma Advanced Liver Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, Pennsylvania ... more >> Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Collapse << |
| NCT00713609 | Acne Vulgaris | Phase 2 | Completed | - | United States, Arkansas ... more >> Dermatology Research of Arkansas Little Rock, Arkansas, United States, 72205 United States, California Center for Dermatology Cosmetic and Laser Surgery Fremont, California, United States, 94538 Center for Dermatology and Laser Surgery Sacramento, California, United States, 95819 United States, Colorado Boulder Medical Center, P.C. Boulder, Colorado, United States, 80304 United States, Florida Dermatology Associates Research Coral Gables, Florida, United States, 33134 United States, Georgia MedaPhase, Inc. Newnan, Georgia, United States, 30263 United States, Maryland Callender Center for Clinical Research Mitchellville, Maryland, United States, 20721 United States, Michigan Grekin Skin Institute Warren, Michigan, United States, 48088 United States, North Carolina University of North Carolina Chapel Hill Chapel Hill, North Carolina, United States, 27599 Dermatology Consulting Services High Point, North Carolina, United States, 27262 United States, Pennsylvania MS Hershey Medical Center Hershey, Pennsylvania, United States, 17033 United States, Tennessee Rivergate Dermatology & Skin Care Center Goodlettsville, Tennessee, United States, 37072 The Skin Wellness Center, PC Knoxville, Tennessee, United States, 37922 United States, Texas Dermatology Treatment & Research Center Dallas, Texas, United States, 75230 Suzanne Bruce and Associates, PA Houston, Texas, United States, 77056 Dermatology Clinical Research Center of San Antonio San Antonio, Texas, United States, 78229 Collapse << |
| NCT00335764 | Adult Giant Cell Glioblastoma ... more >> Adult Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> University of California at Los Angeles Los Angeles, California, United States, 90095 University of California San Francisco San Francisco, California, United States, 94115 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 United States, Pennsylvania University of Pittsburgh Pittsburgh, Pennsylvania, United States, 15232 United States, Texas M D Anderson Cancer Center Houston, Texas, United States, 77030 United States, Wisconsin University of Wisconsin Hospital and Clinics Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01016977 | Acne Vulgaris | Phase 4 | Completed | - | - |
| NCT00335764 | - | - | Completed | - | - |
| NCT00452387 | - | - | Terminated(Early stopping rule... more >>) Collapse << | - | - |
| NCT01016977 | - | - | Completed | - | - |
| NCT00939627 | Metastatic Squamous Neck Cance... more >>r With Occult Primary Squamous Cell Carcinoma Recurrent Metastatic Squamous Neck Cancer With Occult Primary Recurrent Salivary Gland Cancer Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Nasopharynx Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Nasopharynx Stage IVA Salivary Gland Cancer Stage IVA Squamous Cell Carcinoma of the Larynx Stage IVA Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVA Squamous Cell Carcinoma of the Oropharynx Stage IVA Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVA Verrucous Carcinoma of the Larynx Stage IVA Verrucous Carcinoma of the Oral Cavity Stage IVB Salivary Gland Cancer Stage IVB Squamous Cell Carcinoma of the Larynx Stage IVB Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVB Squamous Cell Carcinoma of the Oropharynx Stage IVB Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVB Verrucous Carcinoma of the Larynx Stage IVB Verrucous Carcinoma of the Oral Cavity Stage IVC Salivary Gland Cancer Stage IVC Squamous Cell Carcinoma of the Larynx Stage IVC Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVC Squamous Cell Carcinoma of the Oropharynx Stage IVC Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVC Verrucous Carcinoma of the Larynx Stage IVC Verrucous Carcinoma of the Oral Cavity Tongue Cancer Untreated Metastatic Squamous Neck Cancer With Occult Primary Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Georgia Emory University/Winship Cancer Institute Atlanta, Georgia, United States, 30322 United States, Maryland Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 Suburban Hospital Bethesda, Maryland, United States, 20814 United States, Montana Billings Clinic Billings, Montana, United States, 59107-7000 United States, New York Montefiore Medical Center Bronx, New York, United States, 10467-2490 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Tennessee Vanderbilt-Ingram Cancer Center Nashville, Tennessee, United States, 37232 Collapse << |
| NCT00949182 | Liver Cancer | Phase 2 | Completed | - | United States, New Jersey ... more >> Rutgers University Hospital Newark, New Jersey, United States, 07101 Collapse << |
| NCT00939627 | - | - | Completed | - | - |
| NCT00713609 | - | - | Completed | - | - |
| NCT01011010 | Liver Cancer | Phase 1 | Completed | - | United States, North Carolina ... more >> Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7295 Wake Forest University Comprehensive Cancer Center Winston-Salem, North Carolina, United States, 27157-1096 Collapse << |
| NCT00956436 | Hepatocellular Carcinoma | Phase 1 | Completed | - | United States, Colorado ... more >> Resesarch Site Denver, Colorado, United States United States, Florida Resesarch Site Ocoee, Florida, United States United States, Indiana Resesarch Site Indianapolis, Indiana, United States United States, Massachusetts Resesarch Site Boston, Massachusetts, United States United States, New York Resesarch Site New York, New York, United States United States, Virginia Resesarch Site Norfolk, Virginia, United States Singapore Resesarch Site Singapore, Singapore Taiwan Resesarch Site Tainan, Taiwan Resesarch Site Taipei, Taiwan Resesarch Site Tao-Yuan, Taiwan United Kingdom Resesarch Site Edgbaston, Birmingham, United Kingdom Collapse << |
| NCT01069328 | Carcinoma, Non-Small-Cell Lung... more >> Lung Cancer Collapse << | Phase 1 | Completed | - | United States, Minnesota ... more >> Rochester, Minnesota, United States, 55905 United States, Texas Houston, Texas, United States, 77030 Collapse << |
| NCT01390818 | Locally Advanced Solid Tumor ... more >> Metastatic Solid Tumor Breast Cancer Non Small Cell Lung Cancer Melanoma Colorectal Cancer Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Pinnacle Oncology Hematology Scottsdale, Arizona, United States, 85258 United States, California Cedars Sinai Medical Center Los Angeles, California, United States, 90048 United States, Massachusetts Massachusetts General Hospital Boston, Massachusetts, United States United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37205 United States, Texas Cancer Therapy and Research Center San Antonio, Texas, United States, 78229 Italy Merck Serono Research Site Milan, Italy Spain Merck Serono Research Site Madrid, Spain Collapse << |
| NCT01534260 | Acute Myeloid Leukemia | Phase 1 Phase 2 | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01518413 | Rhabdomyosarcoma and Other Sof... more >>t Tissue Sarcomas Ewing's Sarcoma Family of Tumors Osteosarcoma Neuroblastoma Brain Tumors Collapse << | Phase 1 | Completed | - | United States, District of Col... more >>umbia Children's National Medical Center Washington, District of Columbia, United States, 20010 United States, Maryland National Cancer Institute Bethesda, Maryland, United States, 20892 United States, Massachusetts Children's Hospital Boston/Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Pennsylvania The Children's Hospital of Philadelphia Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT01531361 | Advanced Malignant Neoplasm ... more >> BRAF Gene Mutation Metastatic Malignant Neoplasm Recurrent Malignant Neoplasm Refractory Malignant Neoplasm Collapse << | Phase 1 | Active, not recruiting | February 28, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT00452387 | Metastatic Prostate Cancer | Phase 2 | Terminated(Early stopping rule... more >>) Collapse << | - | United States, California ... more >> Wilshire Oncology Medical Group, Inc. La Verne, California, United States, 91750 United States, Georgia Peachtree Hematology Oncology Consultants Atlanta, Georgia, United States, 30309 Central Georgia Cancer Care Macon, Georgia, United States, 31201 Northwest Georgia Oncology Centers Marietta, Georgia, United States, 30060 United States, Montana Hematology Oncology Centers of the Northern Rockies, PC Billings, Montana, United States, 59101 United States, Ohio Mid-Ohio Oncology/Hematology, Inc. Columbus, Ohio, United States, 43213 United States, Pennsylvania Lancaster Cancer Center Lancaster, Pennsylvania, United States, 17605 Pennsylvania Oncology Hematmology Associates Philadelphia, Pennsylvania, United States, 19106 United States, Tennessee The West Clinic Memphis, Tennessee, United States, 38120 United States, Virginia Cancer Specialists of Tidewater Chesapeake, Virginia, United States, 23320 Collapse << |
| NCT01608139 | Advanced Cancers | Phase 1 | Withdrawn | - | - |
| NCT01715441 | Metastatic Colorectal Cancer P... more >>atients With KRAS Mutated Tumors Collapse << | Phase 2 | Unknown | September 2015 | France ... more >> Hôpital AVICENNE Recruiting Bobigny, France, 93009 Contact: Thomas APARICIO, MD Thomas.aparicio@avc.aphp.fr Principal Investigator: Thomas APARICIO, MD Centre François Baclesse Recruiting Caen, France, 14076 Contact: Marie-Pierre GALAIS, MD Principal Investigator: Marie-Pierre GALAIS, MD Centre Oscar Lambret Recruiting Lille, France, 59020 Contact: Antoine ADENIS, MD a-adenis@o-lambret.fr Principal Investigator: Antoine ADENIS, MD Centre Léon Bérard Recruiting Lyon, France, 69373 Contact: Françoise DESSEIGNE, MD Principal Investigator: Françoise DESSEIGNE, MD Hôpital LA TIMONE Recruiting Marseille, France, 13365 Contact: Jean-François SEITZ, MD Jean-françois.seitz@ap-hm.fr Principal Investigator: Jean-François SEITZ, MD CRLC Val d'Aurelle-Paul Lamarque Recruiting Montpellier, France, 34298 Contact: Emmanuelle SAMALIN, MD Principal Investigator: Emmanuelle SAMALIN, MD C.H.U. de REIMS Recruiting Reims, France, 51092 Contact: Olivier BOUCHE, MD Obouche@chu-reims.fr Principal Investigator: Olivier BOUCHE, MD CHU Charles Nicolle Recruiting Rouen, France, 76038 Contact: Frédéric Di FIORE, MD 33 2.32.88.81.01 Frederic.Di-Fiore@chu-rouen.fr Principal Investigator: Frédéric Di FIORE, MD Institut de Cancérologie de l'Ouest - René Gauducheau Recruiting St. Herblain, France, 44805 Contact: Jaafa BENNOUNA, MD Jaafa.bennouna@ico.unicancer.fr Principal Investigator: Jaafa BENNOUNA, MD Collapse << |
| NCT01534260 | - | - | Completed | - | - |
| NCT01687673 | Hepatocellular Carcinoma | Phase 2 | Active, not recruiting | September 2017 | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94143 United States, Illinois Robert H Lurie Comprehensive Cancer Center Chicago, Illinois, United States, 60611 Collapse << |
| NCT01658878 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Active, not recruiting | December 26, 2019 | - |
| NCT01762150 | Renal Cell Carcinoma | Phase 2 | Completed | - | China, Beijing ... more >> Beijing Cancer Hospital Beijing, Beijing, China, 100142 China, Guangdong Sun Yat-sen university cancer center Guangzhou, Guangdong, China, 510060 China, Liaoning Shenyang general hospital of Shenyang military command Shenyang, Liaoning, China, 110016 China, Shanxi Xijing Hospital Xi'an, Shanxi, China, 710032 China, Sichuan West China Hospital Chengdu, Sichuan, China, 610041 Collapse << |
| NCT01775501 | Hepatocellular Cancer | Phase 2 | Active, not recruiting | December 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02125 Brigham and Women's Hospital Boston, Massachusetts, United States, 02215 Collapse << |
| NCT01434602 | Brain Tumor G... more >>lioblastoma Anaplastic Glioma Collapse << | Phase 1 Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01804374 | Metastatic Osteosarcoma ... more >> Relapsed Osteosarcoma Collapse << | Phase 2 | Completed | - | Italy ... more >> Fondazione del Piemonte per l'Oncologia IRCC Candiolo Candiolo, Torino, Italy, 10060 Collapse << |
| NCT01840592 | Hepatocellular Carcinoma | Phase 2 | Active, not recruiting | April 2019 | United States, New Jersey ... more >> Memorial Sloan Kettering Cancer Center at Basking Ridge Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering West Harrison Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memorial Sloan Kettering Cancer Center at Phelps Memorial Hospital Center Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT01932177 | Metastatic or Locally Advanced... more >> Solid Tumors Collapse << | Phase 1 | Unknown | October 2015 | France ... more >> Hospices Civils de Lyon Recruiting Lyon, France, 69002 Collapse << |
| NCT01390818 | - | - | Completed | - | - |
| NCT02035527 | Metastatic Squamous Neck Cance... more >>r With Occult Primary Squamous Cell Carcinoma Recurrent Metastatic Squamous Neck Cancer With Occult Primary Recurrent Salivary Gland Cancer Recurrent Squamous Cell Carcinoma of the Hypopharynx Recurrent Squamous Cell Carcinoma of the Larynx Recurrent Squamous Cell Carcinoma of the Lip and Oral Cavity Recurrent Squamous Cell Carcinoma of the Nasopharynx Recurrent Squamous Cell Carcinoma of the Oropharynx Recurrent Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Recurrent Verrucous Carcinoma of the Larynx Recurrent Verrucous Carcinoma of the Oral Cavity Salivary Gland Squamous Cell Carcinoma Stage IV Squamous Cell Carcinoma of the Hypopharynx Stage IV Squamous Cell Carcinoma of the Nasopharynx Stage IVA Salivary Gland Cancer Stage IVA Squamous Cell Carcinoma of the Larynx Stage IVA Oral Cavity Squamous Cell Carcinoma Stage IVA Squamous Cell Carcinoma of the Oropharynx Stage IVA Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVA Verrucous Carcinoma of the Larynx Stage IVA Verrucous Carcinoma of the Oral Cavity Stage IVB Salivary Gland Cancer Stage IVB Squamous Cell Carcinoma of the Larynx Stage IVB Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVB Squamous Cell Carcinoma of the Oropharynx Stage IVB Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVB Verrucous Carcinoma of the Larynx Stage IVB Verrucous Carcinoma of the Oral Cavity Stage IVC Salivary Gland Cancer Stage IVC Squamous Cell Carcinoma of the Larynx Stage IVC Squamous Cell Carcinoma of the Lip and Oral Cavity Stage IVC Squamous Cell Carcinoma of the Oropharynx Stage IVC Squamous Cell Carcinoma of the Paranasal Sinus and Nasal Cavity Stage IVC Verrucous Carcinoma of the Larynx Stage IVC Verrucous Carcinoma of the Oral Cavity Tongue Cancer Untreated Metastatic Squamous Neck Cancer With Occult Primary Collapse << | Phase 1 | Completed | - | United States, Ohio ... more >> Arthur G. James Cancer Hospital and Solove Research Institute at Ohio State University Medical Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT01159301 | Adult Acute Basophilic Leukemi... more >>a Adult Acute Eosinophilic Leukemia Adult Acute Megakaryoblastic Leukemia (M7) Adult Acute Minimally Differentiated Myeloid Leukemia (M0) Adult Acute Monoblastic Leukemia (M5a) Adult Acute Monocytic Leukemia (M5b) Adult Acute Myeloblastic Leukemia With Maturation (M2) Adult Acute Myeloblastic Leukemia Without Maturation (M1) Adult Acute Myeloid Leukemia With 11q23 (MLL) Abnormalities Adult Acute Myeloid Leukemia With Del(5q) Adult Acute Myeloid Leukemia With Inv(16)(p13;q22) Adult Acute Myeloid Leukemia With t(16;16)(p13;q22) Adult Acute Myeloid Leukemia With t(8;21)(q22;q22) Adult Acute Myelomonocytic Leukemia (M4) Adult Erythroleukemia (M6a) Adult Pure Erythroid Leukemia (M6b) Blastic Phase Chronic Myelogenous Leukemia Recurrent Adult Acute Myeloid Leukemia Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Terminated | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT01265576 | HCC | Phase 2 | Unknown | April 2016 | United States, California ... more >> Vicus Clinical Site Berkeley, California, United States, 94704 United States, Georgia Vicus Clinical Site Atlanta, Georgia, United States, 30318 United States, New Jersey Vicus Clinical Site New Brunswick, New Jersey, United States Vicus Clinical Site Newark, New Jersey, United States, 07103 Vicus Clinical Site Paterson, New Jersey, United States, 07503 United States, New York Vicus Clinical Site Buffalo, New York, United States, 14263 Vicus Clinical Site New York, New York, United States, 10016 United States, Pennsylvania Vicus Clinical Site Philadelphia, Pennsylvania, United States United States, Texas Vicus Clinical Site Houston, Texas, United States, 77024 Vicus Clinical Site Houston, Texas, United States, 77030 Collapse << |
| NCT01851408 | Melanoma Recu... more >>rrent Melanoma Stage III Melanoma Stage IV Melanoma Collapse << | Phase 2 | Withdrawn(No progression of Ph... more >>ase 1 trial to Phase 2.) Collapse << | - | - |
| NCT02143726 | Refractory Hurthle Cell Thyroi... more >>d Cancer Collapse << | Phase 2 | Recruiting | - | United States, Florida ... more >> Mayo Clinic in Florida Suspended Jacksonville, Florida, United States, 32224-9980 United States, Illinois Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Site Public Contact 312-695-1301 cancer@northwestern.edu Principal Investigator: Maria Matsangou United States, Iowa Siouxland Regional Cancer Center Recruiting Sioux City, Iowa, United States, 51101 Contact: Site Public Contact 712-252-0088 Principal Investigator: Donald B. Wender United States, Michigan University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Site Public Contact 800-865-1125 Principal Investigator: Francis P. Worden United States, Minnesota Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Site Public Contact 855-776-0015 Principal Investigator: Ashish V. Chintakuntlawar United States, Nebraska Nebraska Methodist Hospital Recruiting Omaha, Nebraska, United States, 68114 Contact: Site Public Contact 402-354-5144 Principal Investigator: Yungpo B. Su United States, New Jersey Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Bergen Recruiting Montvale, New Jersey, United States, 07645 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Site Public Contact 212-639-7592 Principal Investigator: Eric J. Sherman Memorial Sloan Kettering Sleepy Hollow Suspended Sleepy Hollow, New York, United States, 10591 United States, North Carolina Southeastern Medical Oncology Center-Clinton Recruiting Clinton, North Carolina, United States, 28328 Contact: Site Public Contact 919-587-9077 ecooke@cancersmoc.com Principal Investigator: James N. Atkins Southeastern Medical Oncology Center-Goldsboro Recruiting Goldsboro, North Carolina, United States, 27534 Contact: Site Public Contact 919-587-9077 ecooke@cancersmoc.com Principal Investigator: James N. Atkins Wayne Memorial Hospital Recruiting Goldsboro, North Carolina, United States, 27534 Contact: Site Public Contact 919-731-6687 ecooke@cancersmoc.com Principal Investigator: James N. Atkins Southeastern Medical Oncology Center-Jacksonville Recruiting Jacksonville, North Carolina, United States, 28546 Contact: Site Public Contact 910-353-0824 ecooke@cancersmoc.com Principal Investigator: James N. Atkins Southeastern Medical Oncology Center-Wilson Suspended Wilson, North Carolina, United States, 27893 United States, Ohio Ohio State University Comprehensive Cancer Center Recruiting Columbus, Ohio, United States, 43210 Contact: Site Public Contact 800-293-5066 Jamesline@osumc.edu Principal Investigator: Bhavana Konda United States, Pennsylvania Fox Chase Cancer Center Recruiting Philadelphia, Pennsylvania, United States, 19111 Contact: Site Public Contact 215-728-4790 Principal Investigator: Jessica R. Bauman United States, Wisconsin Mercy Health System Recruiting Janesville, Wisconsin, United States, 53547 Contact: Site Public Contact 800-928-1103 Principal Investigator: Emily G. Robinson Collapse << |
| NCT02298348 | Neuroblastoma | Phase 1 | Active, not recruiting | May 2019 | United States, California ... more >> Children's Hospital Los Angeles Los Angeles, California, United States, 90027-0700 UCSF Comprehensive Cancer Center San Francisco, California, United States, 94143 United States, Colorado Children Hospital of Colorado Aurora, Colorado, United States, 80045 United States, Georgia Children's Healthcare of Atlanta Atlanta, Georgia, United States, 30322 United States, Illinois University of Chicago, Comer Children's Hospital Chicago, Illinois, United States, 60637 United States, Massachusetts Childrens Hospital Boston, Dana-Farber Cancer Institute. Boston, Massachusetts, United States, 02115 United States, Michigan C.S Mott Children's Hospital Ann Arbor, Michigan, United States, 48109 United States, North Carolina University of North Carolina Chapel Hill, North Carolina, United States, 27599 United States, Ohio Cincinnati Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229-3039 United States, Texas Cook Children's Healthcare System Fort Worth, Texas, United States, 76104 United States, Washington Children's Hospital and Regional Medical Center - Seattle Seattle, Washington, United States, 98105 Canada, Ontario Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT02576509 | Hepatocellular Carcinoma | Phase 3 | Active, not recruiting | July 14, 2020 | - |
| NCT02349867 | Pancreatic Adenocarcinoma ... more >> Stage IA Pancreatic Cancer Stage IB Pancreatic Cancer Stage IIA Pancreatic Cancer Stage IIB Pancreatic Cancer Stage III Pancreatic Cancer Recurrent Pancreatic Carcinoma Collapse << | Phase 1 | Recruiting | May 31, 2022 | United States, Virginia ... more >> Virginia Commonwealth University/Massey Cancer Center Recruiting Richmond, Virginia, United States, 23298 Contact: Andrew Poklepovic, MD 804-628-2321 andrew.poklepovic@vcuhealth.org Contact: Katherine Thompson, CRN 804-628-0960 masseysiit@vcu.edu Principal Investigator: Andrew Poklepovic, MD Collapse << |
| NCT02959125 | Pregnancy Pro... more >>tein-Energy Malnutrition Anemia Collapse << | Not Applicable | Active, not recruiting | December 20, 2017 | Indonesia ... more >> Demak District Health Office Demak, Central Java, Indonesia, 50566 Public Health Center of Karangawen 2 Demak, Jawa Tengah, Indonesia Collapse << |
| NCT02599090 | Glioblastoma ... more >>Gliosarcoma Collapse << | Phase 2 | Withdrawn(Multi phase study di... more >>d not progress to Phase II. Phase I registered as NCT00734526.) Collapse << | - | - |
| NCT02716766 | Carcinoma, Hepatocellular ... more >> Secondary Collapse << | Phase 2 | Active, not recruiting | December 2020 | Hong Kong ... more >> Queen Mary Hospital Hong Kong, Hong Kong Collapse << |
| NCT02774187 | Hepatocellular Carcinoma | Phase 3 | Active, not recruiting | August 2018 | China, Guangdong ... more >> Dongguan People 's Hospital Dongguan, Guangdong, China, 523059 Cancer Center Sun Yat-sen University Guangzhou, Guangdong, China, 510060 The First Affiliated Hospital of Sun Yat-sen University Guangzhou, Guangdong, China, 510060 Guangzhou Twelfth People 's Hospita Guangzhou, Guangdong, China, 510620 The Kaiping Center's Hospital Kaiping, Guangdong, China, 529300 China, Hunan First Affiliated Hospital of University Of South China Hengyang, Hunan, China, 421001 China, Shanxi First Affiliated Hospital Of Xi'an Jiaotong University Xi'an, Shanxi, China, 710061 Collapse << |
| NCT02747537 | Pediatric Solid Tumors | Phase 2 | Recruiting | February 29, 2020 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Alok Kothari, M.D. 314-454-6018 alok@wustl.edu Principal Investigator: Alok Kothari, M.D. Collapse << |
| NCT03009461 | HepatoCellular Carcinoma ... more >> Portal Vein Thrombosis Collapse << | Phase 2 | Recruiting | December 2019 | China, Beijing ... more >> Peking University Cancer Hospital Recruiting Beijing, Beijing, China, 100142 Contact: Xiaodong Wang, MD 008610-88196476 wangxde@gmail.com Sub-Investigator: Guang Cao, MD Sub-Investigator: Hui Chen, MD Sub-Investigator: Jianhai Guo, MD Sub-Investigator: Haifeng Xu, MD Sub-Investigator: Peng Liu, MD Sub-Investigator: Jungang Hu, MD Collapse << |
| NCT03170895 | AML FLT3-ITD ... more >>Mutation Collapse << | Phase 2 | Recruiting | October 1, 2021 | Hong Kong ... more >> The University of Hong Kong Recruiting Hong Kong, Hong Kong Contact: Chunxiao Zhang, SC 852-22554361 chunxiao@hku.hk Collapse << |
| NCT03164382 | Liver Cancer ... more >>Hepatic Carcinoma Collapse << | Phase 3 | Recruiting | December 31, 2022 | China, Guangdong ... more >> Minimally Invasive Interventional Division, Department of Medical Imaging and Interventional Radiology, Sun Yat-sen University Cancer Center, Recruiting Guangzhou, Guangdong, China, 500060 Contact: Ming Zhao, MD 86-20-87343272 zhaoming@sysucc.org.cn Collapse << |
| NCT00943943 | Acute Myelogenous Leukemia ... more >> Leukemia Collapse << | Phase 1 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01683149 | Solid Tumor | Phase 1 | Completed | - | United States, California ... more >> Childrens Hospital Los Angeles Los Angeles, California, United States, 90027 United States, Connecticut Connecticut Childrens Medical Center Hartford, Connecticut, United States, 06106 United States, Delaware Nemours/Alfred I. duPont Hospital for Children, Delaware Wilmington, Delaware, United States, 19803 United States, Florida University of Florida, Gainesville Gainesville, Florida, United States, 32610 Nemours Children's Clinic, Jacksonville Jacksonville, Florida, United States, 32207 University of Miami, Sylvester Comprehensive Cancer Center Miami, Florida, United States, 33136 All Children's Hospital, St. Petersburg St. Petersburg, Florida, United States, 33701 United States, New York Montefiore Medical Center, Children's Hospital at Montefiore Bronx, New York, United States, 10467 United States, Utah Primary Children's Medical Center/Utah Salt Lake City, Utah, United States, 84113 Collapse << |
| NCT01009593 | Hepatocellular Carcinoma Non-r... more >>esectable Hepatocellular Carcinoma Recurrent Carcinoma, Hepatocellular Liver Diseases Neoplasms by Histologic Type Digestive System Neoplasms Carcinoma Liver Neoplasms Neoplasms Neoplasms by Site Digestive System Diseases Adenocarcinoma Neoplasms, Glandular and Epithelial Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT03468231 | Hepatocellular Carcinoma | Phase 3 | Recruiting | March 9, 2020 | China, Guangdong ... more >> Dongguan People's Hospital Recruiting Dongguan, Guangdong, China, 523059 Contact: Wusheng Yu, MD 13827285010 yuwusheng1998@126.com Cancer Center Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Ming Shi, MD 86-2087343115 shiming@mail.sysu.edu.cn Principal Investigator: Ming Shi, MD The First Affiliated Hospital, Sun Yat-sen University Recruiting Guangzhou, Guangdong, China, 510060 Contact: Guosheng Tan, MD 13725254145 tgs1976@163.com Guangzhou Twelfth People 's Hospital Recruiting Guangzhou, Guangdong, China, 510620 Contact: Yuanmin Zhou, MD 15521278919 13430288977@139.com Principal Investigator: Jinghua Chen, MD Kaiping Central Hospital Recruiting Kaiping, Guangdong, China, 529300 Contact: Shijie Zhang, MD 13717287622 Shijie_9262511@163.com China, Hunan First Affiliated Hospital of University Of South China Recruiting Hengyang, Hunan, China, 421001 Contact: Xiaoping Wu, MD 13975486015 wxp19730806@sina.com China, Shanxi The First Affiliated Hospital Of Xi'an Jiaotong University Recruiting Xi'an, Shanxi, China, 710061 Contact: Xin Zheng, MD 13649265446 183421344@qq.com Collapse << |
| NCT03722498 | Hepatocellular Carcinoma | Phase 2 | Recruiting | November 1, 2018 | China, Guangdong ... more >> Ming Shi Recruiting Guangzhou, Guangdong, China Contact: Ming Shi, MD (8620)-87343938 shiming@sysu.edu.cn Principal Investigator: Ming Shi, MD Collapse << |
| NCT03439891 | Stage III Hepatocellular Carci... more >>noma AJCC v8 Stage IIIA Hepatocellular Carcinoma AJCC v8 Stage IIIB Hepatocellular Carcinoma AJCC v7 Stage IIIC Hepatocellular Carcinoma AJCC v7 Stage IV Hepatocellular Carcinoma AJCC v8 Stage IVA Hepatocellular Carcinoma AJCC v8 Stage IVB Hepatocellular Carcinoma AJCC v8 Collapse << | Phase 2 | Recruiting | December 31, 2020 | United States, California ... more >> University of California, San Francisco Recruiting San Francisco, California, United States, 94158 Contact: Robin K. Kelley 877-827-3222 cancertrials@ucsf.edu Principal Investigator: Robin K. Kelley Collapse << |
| NCT03755791 | Hepatocellular Carcinoma | Phase 3 | Recruiting | December 1, 2021 | United States, California ... more >> Exelixis Clinical Site #2 Recruiting Rialto, California, United States, 92377 Contact: Exelixis 888-393-5494 druginfo@exelixis.com United States, Nevada Exelixis Clinical Site #1 Recruiting Las Vegas, Nevada, United States, 89169 Contact: Exelixis 888-393-5494 druginfo@exelixis.com Collapse << |
| NCT02047175 | Hypertension ... more >>Hyperlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> The catholic university of Korea, seoul ST.Mary's hospital Seoul, Gangnam-gu, Korea, Republic of Collapse << |
| NCT01259193 | Hepatocellular Carcinoma | Phase 2 | Unknown | July 2012 | China, Shanghai ... more >> Liver Cancer Institute Recruiting Shanghai, Shanghai, China, 200032 Contact: Zheng-Gang Ren, Ph.D 0086-021-64041990 ext 2137 ren.zhenggang@zs-hospital.sh.cn Contact: Lan Zhang, Master 0086-021-64041990 ext 2171 zhang.lan@zs-hospital.sh.cn Principal Investigator: Zheng-Gang Ren, Ph.D Sub-Investigator: Jubo Zhang, PhD Collapse << |
| NCT01833299 | Hepatocellular Carcinoma ... more >> Face Cancer Collapse << | Phase 4 | Unknown | December 2014 | China ... more >> Cancer Centre of Sun Yat-Sen University Recruiting Guangzhou, China, 510060 Contact: min-shan chen, M.D.,Ph.D. 87343117 Chminsh@mail.sysu.edu.cn Principal Investigator: min-shan chen, M.D.,Ph.D. Collapse << |
| NCT01842230 | Essential Hypertension ... more >> Hyperlipidemia Collapse << | Phase 1 | Completed | - | Korea, Republic of ... more >> Severance Hospital Seoul, Korea, Republic of Collapse << |
| NCT01047683 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT03648333 | Healthy Male Subjects | Phase 1 | Completed | - | - |
| NCT00816426 | Tuberculosis | Phase 1 | Completed | - | Korea, Republic of ... more >> Pusan National Unversity Hospital (PNUH) Busan, Korea, Republic of Asan Medical Center Seoul, Korea, Republic of National Medical Center Seoul, Korea, Republic of Collapse << |
| NCT01047501 | Hypertriglyceridemia | Phase 3 | Completed | - | - |
| NCT01492361 | Cardiovascular Diseases | Phase 3 | Completed | - | - |
| NCT01558193 | Aggression | Not Applicable | Completed | - | United Kingdom ... more >> Swansea University Swansea, Wales, United Kingdom, SA2 8PP Collapse << |
| NCT03383510 | Sustainability ... more >> Microbiota Pesticides Organic Food Collapse << | Not Applicable | Completed | - | Sweden ... more >> Mälardalens University Västerås, Sweden, 721 23 Collapse << |
| NCT03430284 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 31, 2020 | China, Shandong ... more >> Shandong Provincial Hospital Not yet recruiting Jinan, Shandong, China, 250021 Contact: Shanshan Shao +8653168776094 shaoshanshan11@126.com Shandong Provincial Hospital Recruiting Jinan, Shandong, China Collapse << |
| NCT00145106 | Acne Vulgaris | Phase 4 | Completed | - | United States, Pennsylvania ... more >> KGL, Inc Broomall, Pennsylvania, United States, 19008 Collapse << |
| NCT02495727 | Muscular Atrophy | Not Applicable | Completed | - | United Kingdom ... more >> University of Bath Bath, Avon, United Kingdom, BA2 7AY Collapse << |
| NCT02886715 | Acne Vulgaris | Phase 3 | Completed | - | United States, Arizona ... more >> Fougera Investigational Site Tucson, Arizona, United States, 85712 United States, Arkansas Fougera Investigational Site Conway, Arkansas, United States, 72034 Fougera Investigational Site Hot Springs, Arkansas, United States, 71913 United States, California Fougera Investigational Site Anaheim, California, United States, 92801 Fougera Investigational Site La Mesa, California, United States, 91942 Fougera Investigational Site Long Beach, California, United States, 90806 Fougera Investigational Site Long Beach, California, United States, 90813 Fougera Investigational Site Los Angeles, California, United States, 90010 Fougera Investigational Site North Hollywood, California, United States, 91606 Fougera Investigational Site San Diego, California, United States, 92108 Fougera Investigational Site San Ramon, California, United States, 94582 Fougera Investigational Site Upland, California, United States, 91786 Fougera Investigational Site West Covina, California, United States, 91790 United States, Florida Fougera Investigational Site Coral Gables, Florida, United States, 33134 Fougera Investigational Site Miami Gardens, Florida, United States, 33169 Fougera Investigational Site Miami, Florida, United States, 33015 Fougera Investigational Site Miami, Florida, United States, 33175 Fougera Investigational Site Miramar, Florida, United States, 33027 Fougera Investigational Site Sweetwater, Florida, United States, 33172 United States, Indiana Fougera Investigational Site Plainfield, Indiana, United States, 46168 United States, North Carolina Fougera Investigational Site High Point, North Carolina, United States, 27262 United States, Pennsylvania Fougera Investigational Site Hazleton, Pennsylvania, United States, 18201 Fougera Investigational Site Upper Saint Clair, Pennsylvania, United States, 15241 United States, Tennessee Fougera Investigational Site Chattanooga, Tennessee, United States, 37421 United States, Texas Fougera Investigational Site Austin, Texas, United States, 78759 Collapse << |
| NCT01982097 | - | - | Completed | - | Russian Federation ... more >> Multiple Locations, Russian Federation Collapse << |
| NCT02886702 | - | - | Completed | - | - |
| NCT02886715 | - | - | Completed | - | - |
| NCT02886702 | Plaque Psoriasis | Phase 3 | Completed | - | - |
| NCT01988493 | Carcinoma, Hepatocellular | Phase 1 Phase 2 | Active, not recruiting | October 18, 2018 | - |
| NCT02938494 | Acne Vulgaris | Phase 2 | Completed | - | United States, California ... more >> Valeant Site 03 Encinitas, California, United States, 92024 Valeant Site 04 Fremont, California, United States, 94538 Valeant Site 06 Los Angeles, California, United States, 90036 Valeant Site 14 Sacramento, California, United States, 95819 Valeant Site 01 Santa Monica, California, United States, 90404 United States, Colorado Valeant Site 05 Colorado Springs, Colorado, United States, 80915 United States, Florida Valeant Site 08 Coral Gables, Florida, United States, 33146 Valeant Site 10 Miami, Florida, United States, 33175 Valeant Site 09 Miramar, Florida, United States, 33027 United States, Kentucky Valeant Site 12 Louisville, Kentucky, United States, 40202 Valeant Site 13 Louisville, Kentucky, United States, 40217 United States, Maryland Valeant Site 02 Rockville, Maryland, United States, 20850 United States, Minnesota Valeant Site 07 Fridley, Minnesota, United States, 55432 United States, North Carolina Valeant Site 16 Chapel Hill, North Carolina, United States, 27517 Valeant Site 15 High Point, North Carolina, United States, 27262 United States, Pennsylvania Valeant Site 11 Philadelphia, Pennsylvania, United States, 19103 Collapse << |
| NCT02021929 | Hepatopulmonary Syndrome | Phase 2 | Terminated | - | United States, Arizona ... more >> Mayo Clinic Arizona Phoenix, Arizona, United States, 85054 United States, Illinois Northwestern University Feinberg School of Medicine Chicago, Illinois, United States, 60611 United States, Minnesota Mayo Clinic - Rochester Rochester, Minnesota, United States, 55905 United States, New York Columbia University-NewYork-Presbyterian Hospital New York, New York, United States, 10032 United States, Pennsylvania University of Pennsylvania - Perelman Center Philadelphia, Pennsylvania, United States, 19104 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29424 United States, Texas University of Texas Health Science Center at Houston Medical School Houston, Texas, United States, 77030 Collapse << |
| NCT02072486 | Advanced Adult Hepatocellular ... more >>Carcinoma Localized Non-Resectable Adult Hepatocellular Carcinoma Stage III Childhood Hepatocellular Carcinoma Stage IIIA Hepatocellular Carcinoma Stage IIIB Hepatocellular Carcinoma Stage IIIC Hepatocellular Carcinoma Stage IV Childhood Hepatocellular Carcinoma Stage IVA Hepatocellular Carcinoma Stage IVB Hepatocellular Carcinoma Collapse << | Not Applicable | Active, not recruiting | December 3, 2018 | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT02066181 | Desmoid-Type Fibromatosis | Phase 3 | Active, not recruiting | - | - |
| NCT02114658 | Thyroid Carcinoma | Phase 2 | Completed | - | Japan ... more >> Kobe, Hyogo, Japan, 650-0011 Matsumoto, Nagano, Japan, 390-8621 Bunkyo-ku, Tokyo, Japan, 113-8603 Koto-ku, Tokyo, Japan, 135-8550 Osaka, Japan, 545-8586 Collapse << |
| NCT02178358 | Hepatocellular Carcinoma | Phase 2 | Active, not recruiting | December 31, 2018 | China ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Beijing, China, 100036 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bengbu, China, 233004 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Changchun, China, 130012 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hangzhou, China, 310016 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hefei, China, 230022 Hong Kong For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hong Kong, Hong Kong For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kowloon, Hong Kong Korea, Republic of For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Incheon, Korea, Republic of, 405-760 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Seoul, Korea, Republic of, 135 720 Taiwan For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gwei Shan Township, Taiwan, 333 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Liouying/Tainan, Taiwan, 736 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Puzih City, Taiwan, 613 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tainan, Taiwan, 70403 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taipei, Taiwan, 11217 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Taoyuan, Taiwan, 33378 Thailand For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bangkok, Thailand, 10330 Collapse << |
| NCT02084732 | Thyroid Cancer | Phase 2 | Unknown | June 2018 | Colombia ... more >> Instituto Nacional de Cancerología Recruiting Bogotá, Cundinamarca, Colombia, 00000 Contact: Yeinnyer C. Muleth, BSc 3340106 ymuleth@cancer.gov.co Principal Investigator: Luis F Fierro, Md Sub-Investigator: Gloria Garavito, Md Sub-Investigator: Andres A Cuellar, Md Sub-Investigator: Jose A. Carreño, Md Sub-Investigator: Juan S Castillo, Md Collapse << |
| NCT02220088 | Hepatocellular Carcinoma | Phase 2 Phase 3 | Completed | - | China, Guangdong ... more >> Cancer Center Sun Yat-sen University Guangzhou, Guangdong, China, 510060 Collapse << |
| NCT02185560 | - | - | Recruiting | June 30, 2021 | Japan ... more >> Recruiting Multiple Locations, Japan Collapse << |
| NCT02187081 | HCC | Not Applicable | Recruiting | December 2018 | China, Chongqing ... more >> Southwest Hospital Recruiting Chongqing, Chongqing, China, 400038 Contact: Xiaobin Feng Principal Investigator: Kuansheng Ma, Ph.D Collapse << |
| NCT02227914 | Advanced Hepatocellular Carcin... more >>oma Collapse << | Phase 1 Phase 2 | Withdrawn(Study stopped due to... more >> administrative reasons) Collapse << | - | United States, California ... more >> Lahey Hospital & Medical Center Burlington, California, United States United States, Colorado Rocky Mountain Cancer Centers Denver, Colorado, United States United States, Florida University of Miami Hospital & Clinics Miami, Florida, United States United States, Illinois The University of Chicago Medical Center Chicago, Illinois, United States United States, Ohio The Ohio State University, Martha Morehouse Medical Plaza Columbus, Ohio, United States United States, Wisconsin University of Wisconsin Comprehensive Cancer Center Madison, Wisconsin, United States Collapse << |
| NCT02279719 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Active, not recruiting | - | - |
| NCT02240433 | Hepatocellular Carcinoma | Phase 1 | Active, not recruiting | February 11, 2019 | Japan ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Chiba, Japan, 277 8577 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Kanagawa, Japan, 241-8515 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Suita-shi, Japan, 565-0871 Collapse << |
| NCT03170596 | Atrophic Post Acne Scarring | Not Applicable | Completed | - | India ... more >> Dermatology OPD, New OPD Building, Level 5C, Postgraduate Institute of Medical Education and Research Chandigarh, India, 160012 Collapse << |
| NCT03292640 | Acne Vulgaris | Phase 3 | Completed | - | United States, California ... more >> Center for Dermatology Clinical Research Inc. Fremont, California, United States, 94538 Collapse << |
| NCT02288507 | Hepatocellular Cancer | Phase 1 | Withdrawn(Closed due to no acc... more >>rual) Collapse << | - | United States, Hawaii ... more >> University of Hawaii Honolulu, Hawaii, United States, 96813 Collapse << |
| NCT03263624 | Nail Psoriasis | Phase 4 | Not yet recruiting | November 1, 2018 | - |
| NCT02311205 | Hepatocellular Carcinoma ... more >> Metastasis Collapse << | Phase 2 | Active, not recruiting | June 2018 | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of Collapse << |
| NCT03341910 | Acne Vulgaris | Phase 2 | Completed | - | United States, Florida ... more >> International Dermatology Inc. Miami, Florida, United States, 33144 Collapse << |
| NCT02122003 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Terminated(availability of new... more >> and more promising therapeutic agents than expected in the experimentation in question) Collapse << | - | Italy ... more >> Istituto Tumori Milan, Mi, Italy, 20156 Collapse << |
| NCT02332031 | Drug Interactions | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany, 13353 Collapse << |
| NCT02358395 | Hepatocellular Carcinoma | Phase 1 | Completed | - | Japan ... more >> 4 Sites Tokyo,etc, Japan Collapse << |
| NCT02400788 | Hepatocellular Carcinoma | Phase 1 Phase 2 | Completed | - | Japan ... more >> Chiba, Japan Ishikawa, Japan Kanagawa, Japan Kochi, Japan Kyoto, Japan Osaka, Japan Saga, Japan Shizuoka, Japan Tokyo, Japan Korea, Republic of Busan, Korea, Republic of Daegu, Korea, Republic of Seoul, Korea, Republic of Collapse << |
| NCT02323906 | Carcinoma, Hepatocellular | Phase 1 | Terminated(Slow enrollment and... more >> business decision. No safety concerns were identified for the study drugs.) Collapse << | - | United States, California ... more >> University of California San Francisco San Francisco, California, United States, 94115 United States, Florida University of Florida College of Med Gainesville, Florida, United States, 32610-0277 Moffitt Cancer Center Tampa, Florida, United States, 33612 United States, Indiana Indiana University Cancer Center Indianapolis, Indiana, United States, 46202-528 United States, Michigan Henry Ford Hospital Detroit, Michigan, United States, 48202 United States, South Carolina Greenville Hospital System Greenville, South Carolina, United States, 29605 United States, Utah University of Utah Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 Collapse << |
| NCT02354898 | Advanced Solid Tumors ... more >> Hepatocellular Carcinoma Collapse << | Phase 1 | Completed | - | Japan ... more >> 5 Sites Chiba, Etc., Japan Collapse << |
| NCT00500045 | Central Retinal Vein Occlusion... more >> Branch Retinal Vein Occlusion Collapse << | Phase 2 Phase 3 | Completed | - | United States, California ... more >> Palo Alto Medical Foundation Department of Ophthalmology Palo Alto, California, United States, 94301 Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
