| NCT03762330 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Not Applicable | Not yet recruiting | January 2021 | - |
| NCT02835716 | - | - | Recruiting | September 2020 | United States, Connecticut
... more >>
Millennium Magnetic Technologies, LLC
Recruiting
Westport, Connecticut, United States, 06880
Contact: Steve Levy, MD 203-423-9494 SL@MilMag.net
Contact: Donald H Marks, MD PhD 9733070364 DHM@MilMag.net Collapse << |
| NCT01730404 | COPD | Phase 2 | Completed | - | Ireland
... more >> Celerion
Belfast, Ireland
United Kingdom
Parexel, early phases
Harrow, United Kingdom
Medicines Evaluation Unit Ltd
Manchester, United Kingdom
Freeman Hospital
Newcastle, United Kingdom
Nottingham University Hospitals NHS Trust
Nottingham, United Kingdom Collapse << |
| NCT00613587 | - | - | Completed | - | United States, Nebraska
... more >>
Creighton University Medical Center
Omaha, Nebraska, United States, 68131 Collapse << |
| NCT02363335 | Healthy Volunteers | Phase 1 | Completed | - | United States, Maryland
... more >>
National Institute of Aging, Clinical Research Unit
Baltimore, Maryland, United States, 21224 Collapse << |
| NCT01580748 | Symptomatic Bronchiectasis | Phase 2 | Unknown | January 2013 | Korea, Republic of
... more >>
Seoul National University Bundang Hospital
Not yet recruiting
Seongnam, Gyeongi-do, Korea, Republic of, 463-707
Contact: Ho Il Yoon, M.D., Ph.D. +82-31-787-7036 dextro@snubh.org Collapse << |
| NCT02340520 | - | - | Completed | - | - |
| NCT00242294 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2
Phase 3 | Completed | - | Japan
... more >> Nycomed Japan and Mitsubishi Tanabe Oharma Corporation
Osaka, Japan Collapse << |
| NCT02340520 | COPD | Phase 3 | Completed | - | United States, Connecticut
... more >>
UConnHealth
Farmington, Connecticut, United States, 06032 Collapse << |
| NCT00108823 | Chronic Obstructive Pulmonary ... more >>Disease, COPD Collapse << | Phase 3 | Completed | - | United States, California
... more >>
ALTANA Pharma
Cities in California, California, United States
United States, Colorado
ALTANA Pharma
Cities in Colorado, Colorado, United States
United States, Florida
ALTANA Pharma
Cities in Florida, Florida, United States
United States, Missouri
ALTANA Pharma
Cities in Missouri, Missouri, United States
United States, New Mexico
ALTANA Pharma
Cities in New Mexico, New Mexico, United States
United States, New York
ALTANA Pharma
Cities in New York, New York, United States
United States, Oregon
ALTANA Pharma
Cities in Oregeon, Oregon, United States
United States, South Carolina
ALTANA Pharma
Cities in South Carolina, South Carolina, United States
United States, Texas
ALTANA Pharma
Cities in Texas, Texas, United States
United States, Washington
ALTANA Pharma
Cities in Washington, Washington, United States
Canada
ALTANA Pharma
Cities in Canada, Canada
France
ALTANA Pharma
Cities in France, France
Poland
ALTANA Pharma
Cities in Poland, Poland
South Africa
ALTANA Pharma
Cities in South Africa, South Africa
Spain
ALTANA Pharma
Cities in Spain, Spain
United Kingdom
ALTANA Pharma
Cities in the United Kingdom, United Kingdom Collapse << |
| NCT01285180 | - | - | Completed | - | - |
| NCT01285167 | - | - | Completed | - | - |
| NCT00246922 | Bronchial Asthma | Phase 3 | Completed | - | Japan
... more >> Nycomed Japan and Mitsubishi Tanabe Pharma Corporation
Osaka, Japan Collapse << |
| NCT00062582 | Chronic Obstructive Pulmonary ... more >>Disease
COPD Collapse << | Phase 3 | Completed | - | - |
| NCT00246935 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | Japan
... more >> Nycomed in Japan and Mitsubishi Tanabe Pharma Corporation
Osaka, Japan Collapse << |
| NCT02187926 | - | - | Completed | - | - |
| NCT00242307 | Bronchial Asthma | Phase 2
Phase 3 | Completed | - | Japan
... more >> Nycomed Japan and Mitsubishi Tanabe Pharma Corporation
Osaka, Japan Collapse << |
| NCT00073177 | Asthma | Phase 3 | Completed | - | - |
| NCT00076076 | Asthma | Phase 3 | Completed | - | - |
| NCT00076089 | Chronic Obstructive Pulmonary ... more >>Disease
COPD Collapse << | Phase 3 | Completed | - | - |
| NCT02051335 | Memory Impairment
... more >> Alzheimer's Disease Collapse << | Phase 1 | Completed | - | United Kingdom
... more >>
London, United Kingdom Collapse << |
| NCT00163475 | Asthma | Phase 3 | Completed | - | Australia
... more >> ALTANA Pharma
Cities in Australia, Australia
Belgium
ALTANA Pharma
Cities in Belgium, Belgium
France
ALTANA Pharma
Cities in France, France
South Africa
ALTANA Pharma
Cities in South Africa, South Africa
Spain
ALTANA Pharma
Cities in Spain, Spain Collapse << |
| NCT02671942 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Recruiting | December 2017 | China, Guangdong
... more >>
First Affiliated Hospital of Guangzhou Medical University
Recruiting
Guangzhou, Guangdong, China, 510120
Contact: Zheng jinping 18928868238 18928868238@163.com Collapse << |
| NCT00163527 | Asthma | Phase 3 | Completed | - | - |
| NCT00297102 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT00297115 | - | - | Completed | - | - |
| NCT02015767 | - | - | Completed | - | - |
| NCT00940329 | Healthy | Phase 1 | Completed | - | Germany
... more >> Nycomed
Konstanz, Germany, 78467 Collapse << |
| NCT00297102 | - | - | Completed | - | - |
| NCT00297115 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT01509677 | COPD
Chronic ... more >>Obstructive Pulmonary Disease Collapse << | Phase 3 | Completed | - | Denmark
... more >>
Kobenhavn NV, Denmark
København NV, Denmark, DK-2400
Germany
Freiburg, Germany
Grosshansdorf, Germany
Hannover, Germany
Heidelberg, Germany
Immenhausen, Germany
Kiel, Germany
Mainz, Germany
Poland
Bialystok, Poland
Krakow, Poland
Lodz, Poland, 90-153
Lodz, Poland
Sweden
Lund, Sweden
United Kingdom
Cottingham, United Kingdom
Dafen, United Kingdom
Leicester, United Kingdom
London, United Kingdom
Manchester, United Kingdom
Norwich, United Kingdom, NR4 7UY Collapse << |
| NCT01664624 | - | - | Completed | - | - |
| NCT00242320 | COPD | Phase 3 | Completed | - | Hong Kong
... more >> Altana Pharma/Nycomed
Hong Kong, Hong Kong, 852
Altana Pharma/Nycomed
Shatin, NT, Hong Kong, Hong Kong
Korea, Republic of
Altana Pharma/Nycomed
Anvang-Si, Gveonggi-Do, Korea, Republic of, 431-070
Altana Pharma/Nycomed
Chungbuk, Korea, Republic of, 361-711
Altana Pharma/Nycomed
Gangwon-do, Korea, Republic of, 220-701
Altana Pharma/Nycomed
Gangwon-Do, Korea, Republic of, 220-711
Altana Pharma/Nycomed
Gwangju, Korea, Republic of, 501-757
Altana Pharma/Nycomed
Gyeonggi-do, Korea, Republic of, 420-767
Altana Pharma/Nycomed
Jiniu-Si. Gveongsangnam-Do, Korea, Republic of, 660-702
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 135-720
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 136-705
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 137-701
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 138-736
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 140-757
Altana Pharma/Nycomed
Seoul, Korea, Republic of, 158-710
Altana Pharma/Nycomed
Uijongbu-city, Gveonggi-Do, Korea, Republic of, 480-130
Altana Pharma/Nycomed
Ulsan, Korea, Republic of, 682-060
Malaysia
Altana Pharma/Nycomed
Jalan Rasah, Seremban, Malaysia, 70300
Altana Pharma/Nycomed
Kota Bharu / Kelantan, Malaysia, 16150
Altana Pharma/Nycomed
Kota Kinabalu, Sabah, Malaysia, 88586
Altana Pharma/Nycomed
Kuala Lumpur, Malaysia, 50590
Altana Pharma/Nycomed
Kuala Lumpur, Malaysia, 56000
Altana Pharma/Nycomed
Kuala Lumpur, Malaysia, 59100
Philippines
Altana Pharma/Nycomed
Manila, Philippines, 1000
Altana Pharma/Nycomed
Quezon City, Philippines, 1100
Altana Pharma/Nycomed
Quezon City, Philippines, 1102
Altana Pharma/Nycomed
Quezon City, Philippines, 850
Altana Pharma/Nycomed
Quezon City, Philippines, 870
Taiwan
Altana Pharma/Nycomed
Kaohsiung, Taiwan, 886-807
Altana Pharma/Nycomed
Taipei City 114, Taiwan, 886-114
Altana Pharma/Nycomed
Taipei, Taiwan, 886-114
Altana Pharma/Nycomed
Taipei, Taiwan
Altana Pharma/Nycomed
Tau-Yuan, Taiwan, 886-333 Collapse << |
| NCT00313209 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT00430729 | Chronic Obstructive Pulmonary ... more >>Disease
COPD Collapse << | Phase 3 | Completed | - | Australia
... more >> ALTANA Pharma
Cities in Australia, Australia
Austria
ALTANA Pharma
Cities in Austria, Austria
Canada
ALTANA Pharma
Cities in Canada, Canada
France
ALTANA Pharma
Cities in France, France
Hungary
ALTANA Pharma
Cities in Hungary, Hungary
Italy
ALTANA Pharma
Cities in Italy, Italy
Netherlands
ALTANA Pharma
Cities in the Netherlands, Netherlands
Poland
ALTANA Pharma
Cities in Poland, Poland
Portugal
ALTANA Pharma
Cities in Portugal, Portugal
Russian Federation
ALTANA Pharma
Cities in the Russian Federation, Russian Federation
South Africa
ALTANA Pharma
Cities in South Africa, South Africa
Spain
ALTANA Pharma
Cities in Spain, Spain
Switzerland
ALTANA Pharma
Cities in Switzerland, Switzerland
United Kingdom
ALTANA Pharma
Cities in the United Kingdom, United Kingdom Collapse << |
| NCT02051335 | - | - | Completed | - | - |
| NCT00746434 | Plaque Psoriasis | Phase 2 | Withdrawn(Study withdrawn due ... more >>to business decisions. No subjects were treated.) Collapse << | September 2009 | - |
| NCT01473758 | Chronic Obstructive Pulmonary ... more >>Disease With (Acute) Exacerbation Collapse << | Phase 2 | Terminated(Company decision No... more >> Safety or Efficacy Concerns) Collapse << | - | United Kingdom
... more >> Academic Unit of Respiratory Medicine, Royal Free Hospital, Jadwiga A. Wedzicha
London, United Kingdom, NW3 2PF
London, United Kingdom Collapse << |
| NCT00313209 | - | - | Completed | - | - |
| NCT01433666 | Dementia | Phase 2 | Completed | - | Netherlands
... more >> Maastricht University, Faculty of Psychology and Neuroscience
Maastricht, Limburg, Netherlands, 6200 MD Collapse << |
| NCT01313494 | COPD | Phase 3 | Completed | - | China
... more >>
Beijing, China
Changsha, China
Chengdu, China
Chongqing, China
Fuzhou, China
Guangzhou, China
Hangzhou, China
Nanjing, China
Nanning, China
Qingdao, China
Shanghai, China
Shenyang, China
Shijiazhuang, China
Hong Kong
Hong Kong, Hong Kong
Singapore
Singapore, Singapore Collapse << |
| NCT01140542 | Diabetes Mellitus Type 2 | Phase 2 | Completed | - | - |
| NCT01480661 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | Belgium
... more >> Antwerp University Hospital
Edegem, Antwerp, Belgium, 2650
ZNA Middelheim
Antwerp, Belgium, 2020 Collapse << |
| NCT00424268 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01473758 | - | - | Terminated(Company decision No... more >> Safety or Efficacy Concerns) Collapse << | - | - |
| NCT01329029 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01664624 | Type 2 Diabetes | Phase 1 | Completed | - | United States, California
... more >>
Chula Vista, California, United States
United States, Florida
Orlando, Florida, United States Collapse << |
| NCT00746382 | Dermatitis | Phase 2 | Withdrawn(Study withdrawn due ... more >>to business decisions. No subjects were treated.) Collapse << | September 2009 | - |
| NCT01703260 | Nonalcoholic Steatohapatitis | Phase 2 | Terminated(Company decision; N... more >>o safety or efficacy concerns (see detailed description)) Collapse << | - | United States, California
... more >>
Coronado, California, United States, 92118
Coronado, California, United States
United States, Maryland
Annapolis, Maryland, United States, 21401
Baltimore, Maryland, United States, 21202 Collapse << |
| NCT01595750 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Unknown | August 2012 | Spain
... more >> Spanish Research Center for Respiratory Diseases
Not yet recruiting
Bunyola, Balearic Island, Spain, 07110
Contact: Paloma Vaquer 0034971011772 pvaquer@ciberes.org Collapse << |
| NCT01329029 | - | - | Completed | - | - |
| NCT00424268 | - | - | Completed | - | - |
| NCT01856764 | - | - | Completed | - | - |
| NCT02187250 | PCOS
Obesity | Phase 4 | Completed | - | Slovenia
... more >> University Medical Center Ljubljana
Ljubljana, Slovenia, 1000 Collapse << |
| NCT01313494 | - | - | Completed | - | - |
| NCT01856764 | Atopic Dermatitis | Phase 2 | Completed | - | Germany
... more >>
Berlin, Germany
Gera, Germany
Hamburg, Germany Collapse << |
| NCT01701934 | Chronic Obstructive Pulmonary ... more >>Disease
Metabolic Syndrome Collapse << | Phase 2 | Terminated(The recruitment of ... more >>the study was prematurely stopped in July 2014 for the following reason; no more study medication.) Collapse << | December 2014 | Canada, British Columbia
... more >>
St. Paul's Hospital
Vancouver, British Columbia, Canada, V6Z 1Y6
Canada, Quebec
Centre hospitalier de l'Université de Montréal
Montréal, Quebec, Canada, H2W 1T8
Montreal Chest Institute
Montréal, Quebec, Canada, H2X 2P4
Institut universitaire de cardiologie et de pneumologie de Québec
Québec, Quebec, Canada, G1V 4G5
Hôpital régional de Saint-Jérôme
Saint-Jérôme, Quebec, Canada, J7Z 5T3 Collapse << |
| NCT01572948 | COPD | Not Applicable | Completed | - | United States, Alabama
... more >>
UAB Lung Health Center
Birmingham, Alabama, United States, 35294 Collapse << |
| NCT01703260 | - | - | Terminated(Company decision; N... more >>o safety or efficacy concerns (see detailed description)) Collapse << | - | - |
| NCT01888952 | Advanced B-cell Lymphoid Malig... more >>nancies Collapse << | Early Phase 1 | Completed | - | United States, Texas
... more >>
Ctrc @ Uthscsa
San Antonio, Texas, United States, 78229 Collapse << |
| NCT01572948 | - | - | Completed | - | - |
| NCT01888952 | - | - | Completed | - | - |
| NCT01973998 | COPD | Phase 3 | Completed | - | United States, Pennsylvania
... more >>
Temple University Hospital
Philadelphia, Pennsylvania, United States, 19140 Collapse << |
| NCT02097992 | - | - | Completed | - | - |
| NCT02079844 | Schizophrenia | Phase 1 | Completed | - | United Kingdom
... more >>
Denmark Hill, London, United Kingdom Collapse << |
| NCT02037672 | PCOS
Obesity | Phase 4 | Completed | - | Slovenia
... more >> University Medical Center Ljubljana
Ljubljana, Slovenia, 1000 Collapse << |
| NCT01443845 | COPD
COPD Exa... more >>cerbation
Lung Diseases
Respiratory Disorders
Pulmonary Disease
Chronic Obstructive Lung Diseases
Chronic Obstructive Airway Disease Collapse << | Phase 4 | Completed | - | - |
| NCT01765192 | Asthma | Phase 2 | Completed | - | Germany
... more >>
Berlin, Germany
Grosshansdorf, Germany
Hamburg, Germany
Hannover, Germany
Mainz, Germany
Schwerin, Germany
Hungary
Budapest, Hungary
Nyiregyhaza, Hungary
Szarvas, Hungary
Torokbalint, Hungary
South Africa
Bloemfontein, South Africa
Cape Town, South Africa
Pretoria, South Africa Collapse << |
| NCT03073798 | - | - | Completed | - | - |
| NCT01765192 | - | - | Completed | - | - |
| NCT02079844 | - | - | Completed | - | - |
| NCT02451540 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 2 | Terminated(No new investigatio... more >>nal product can be delivered to the site.) Collapse << | - | Belgium
... more >> University Hospital of Antwerp
Edegem, Antwerp, Belgium, 2650 Collapse << |
| NCT01630200 | Chronic Obstructive Pulmonary ... more >>Disease and Allied Conditions Collapse << | Phase 2 | Unknown | January 2016 | Austria
... more >> Deparment for Respiratory and Critical Care Medicine, Otto Wangner Hospital
Vienna, Austria, 1140 Collapse << |
| NCT01443845 | - | - | Completed | - | - |
| NCT03073798 | COPD
Chronic ... more >>Bronchitis
Emphysema Collapse << | Phase 4 | Completed | - | - |
| NCT03458546 | Lymphoma, B-Cell | Phase 1 | Recruiting | January 30, 2020 | United States, Texas
... more >>
Mays Cancer Center, UT Health San Antonio
Recruiting
San Antonio, Texas, United States, 78229
Contact: Epp Goodwin 210-450-1000 Collapse << |
| NCT01365533 | Allergic Asthma | Phase 2 | Completed | - | Canada
... more >> Nycomed investigational site
Quebec, Canada Collapse << |
| NCT02165826 | - | - | Completed | - | - |
| NCT01830959 | Sarcoidosis | Phase 4 | Unknown | October 2016 | United States, Ohio
... more >>
University of Cincinnati
Cincinnati, Ohio, United States, 45267 Collapse << |
| NCT02097992 | COPD | Phase 1
Phase 2 | Completed | - | United States, Florida
... more >>
University of Miami Divison of Pulmonary and Critical Care Medicine
Miami, Florida, United States, 33136 Collapse << |
| NCT01862029 | Obesity | Phase 1
Phase 2 | Completed | - | United States, Maryland
... more >>
National Institutes of Health Clinical Center, 9000 Rockville Pike
Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02165826 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | Slovakia
... more >>
Bardejov, Slovakia
Spisska Nova Ves, Slovakia Collapse << |
| NCT01862029 | - | - | Completed | - | - |
| NCT02068456 | - | - | Completed | - | Korea, Republic of
... more >>
Seoul, Korea, Republic of Collapse << |
| NCT01354782 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 1 | Completed | - | China
... more >> Nycomed Investigational Site
Beijing, China, 100032 Collapse << |
| NCT01849341 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Completed | - | Spain
... more >> Hospital de Jerez
Jerez de la Frontera, Cádiz, Spain
Hospital Universitario Reina Sofía
Córdoba, Spain
H.U. Virgen de las Nieves
Granada, Spain
Hospital Universitario Virgen Macarena
Sevilla, Spain Collapse << |
| NCT01745848 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Completed | - | United States, Pennsylvania
... more >>
Emphysema COPD Research Center
Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02018432 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 4 | Unknown | December 2015 | Korea, Republic of
... more >>
Department of Pulmonary and Critical Care Medicine and Clinical Research Center for Chronic Obstructive Airway Diseases, Asan Medical Center, University of Ulsan College of Medicine
Not yet recruiting
Seoul, Korea, Republic of, 138-736
Contact: Sei Won Lee, MD +82-3010-3990 iseiwon@gmail.com Collapse << |
| NCT01745848 | - | - | Completed | - | - |
| NCT03428334 | Non-cystic Fibrosis Bronchiect... more >>asis Collapse << | Phase 2 | Recruiting | August 31, 2019 | Hong Kong
... more >> Queen Mary Hospital
Recruiting
Hong Kong, Hong Kong
Contact: James CM Ho, MD Collapse << |
| NCT03381573 | - | - | Recruiting | January 31, 2020 | United States, Massachusetts
... more >>
Quintiles, Inc
Recruiting
Cambridge, Massachusetts, United States, 02139
Finland
Epid Research
Recruiting
Espoo, Finland
Germany
Leibniz-Institut Für Epidemiologie Und Präventionsforschung-Bips Gmbh
Recruiting
Bremen, Germany Collapse << |
| NCT03532490 | Asthma
Obesit... more >>y Collapse << | Phase 2 | Recruiting | April 2020 | United States, Arizona
... more >>
University of Arizona
Active, not recruiting
Tucson, Arizona, United States, 85724
United States, Illinois
Northwestern University Feinberg School of Medicine
Active, not recruiting
Chicago, Illinois, United States, 60611
United States, New York
Mount Sinai Icahn School of Medicine
Active, not recruiting
New York, New York, United States, 10029
United States, North Carolina
Duke University
Recruiting
Durham, North Carolina, United States, 27705
Contact: Cathy Foss, BS 919-479-0861 catherine.foss@duke.edu
United States, Vermont
University of Vermont
Recruiting
Colchester, Vermont, United States, 05446
Contact: Kathy Meehan 802-847-8839 kathy.meehan@uvmhealth.org Collapse << |
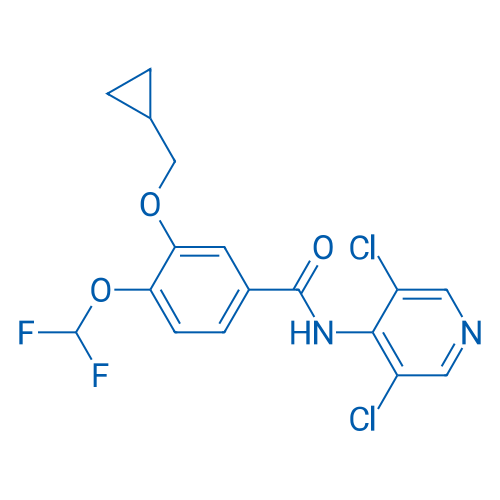
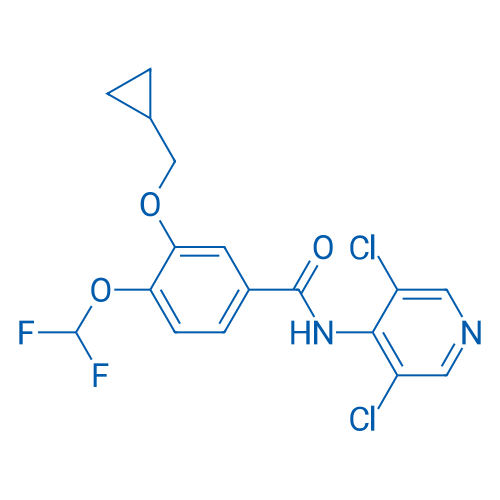

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
