CAS No.: 248281-84-7
拉喹莫德 Catalog No. CSN10659
Synonyms: ABR-215062;LAQ;SAIK-MS
Laquinimod is an orally administered neuroimmunomodulator initially developed for the treatment of multiple sclerosis.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01404117 Relapsing Multiple Sclerosis Phase 2 Withdrawn(Study is being redes... more >>igned) Collapse << January 2014 - NCT00605215 Multiple Sclerosis Phase 3 Completed - - NCT01707992 Multiple Sclerosis Phase 3 Completed - - - 更多
- 参考文献
- [1] Yang JS, Xu LY, et al. Laquinimod (ABR-215062) suppresses the development of experimental autoimmune encephalomyelitis, modulates the Th1/Th2 balance and induces the Th3 cytokine TGF-beta in Lewis rats. J Neuroimmunol. 2004 Nov;156(1-2):3-9.
- [2] Zou LP, Abbas N, et al. Suppression of experimental autoimmune neuritis by ABR-215062 is associated with altered Th1/Th2 balance and inhibited migration of inflammatory cells into the peripheral nerve tissue. Neuropharmacology. 2002 Apr;42(5):731-9.
- [3] Schulze-Topphoff U, Shetty A, Varrin-Doyer M, Molnarfi N, Sagan SA, Sobel RA, Nelson PA, Zamvil SS. Laquinimod, a quinoline-3-carboxamide, induces type II myeloid cells that modulate central nervous system autoimmunity. PLoS One. 2012;7(3):e33797
- [4] Li Z, Chen J, Lei L, Jiang N, Zhu Y, Jia Y, Zhuo Y, Su W. Laquinimod Inhibits Inflammation-Induced Angiogenesis in the Cornea. Front Med (Lausanne). 2020 Nov 10;7:598056
- [5] Zhang X, Jin J, Xie A. Laquinimod inhibits MMP+ induced NLRP3 inflammasome activation in human neuronal cells. Immunopharmacol Immunotoxicol. 2020 Jun;42(3):264-271
- [6] Gentile A, Musella A, De Vito F, Fresegna D, Bullitta S, Rizzo FR, Centonze D, Mandolesi G. Laquinimod ameliorates excitotoxic damage by regulating glutamate re-uptake. J Neuroinflammation. 2018 Jan 5;15(1):5
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 248281-84-7 | 储存条件 |
|
|||||||
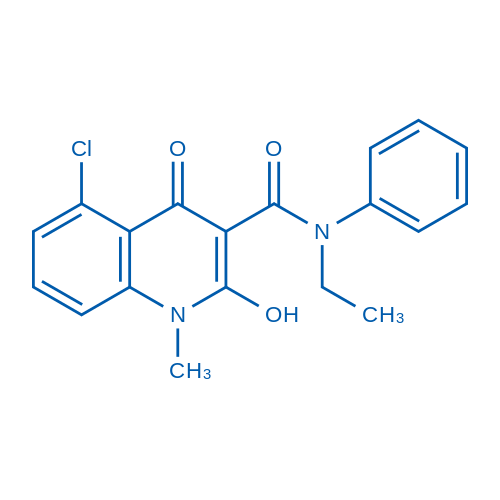
| 分子式 | C19H17ClN2O3 | 运输 | 蓝冰 | |||||||
| 分子量 | 356.80 | 别名 | ABR-215062;LAQ;SAIK-MS;TV-5600;拉喹莫德 | |||||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
