CAS No.: 50-91-9
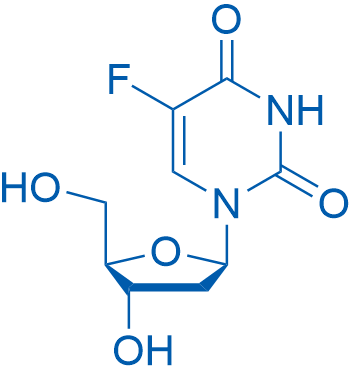
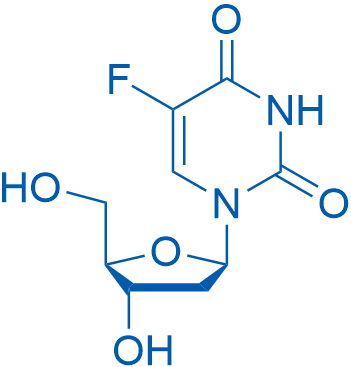
2'-脱氧-5-氟尿苷 Catalog No. CSN18715
Synonyms: 5-Fluorouracil 2'-deoxyriboside;Deoxyfluorouridine;FDUR
Floxuridine is an oncology drug that belongs to antimetabolites with an GI50 of 5.1 μM for the inhibition of PEPT1.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 Thymidylate Synthase
GI50:5.1μM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00296322 - - Completed - - NCT00296322 Stomach Cancer Phase 3 Completed - Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << NCT00296335 Stomach Cancer Phase 3 Completed - Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << - 更多
- 参考文献
- [1] Tsume Y, Hilfinger JM, Amidon GL. Enhanced cancer cell growth inhibition by dipeptide prodrugs of floxuridine: increased transporter affinity and metabolic stability. Mol Pharm. 2008 Sep-Oct;5(5):717-27.
- [2] Mini E, Moroson BA, Bertino JR. Cytotoxicity of floxuridine and 5-fluorouracil in human T-lymphoblast leukemia cells: enhancement by leucovorin. Cancer Treat Rep. 1987 Apr;71(4):381-9.
- [3] Huehls AM, Wagner JM, Huntoon CJ, Geng L, Erlichman C, Patel AG, Kaufmann SH, Karnitz LM. Poly(ADP-Ribose) polymerase inhibition synergizes with 5-fluorodeoxyuridine but not 5-fluorouracil in ovarian cancer cells. Cancer Res. 2011 Jul 15;71(14):4944-54
- [4] Power DG, Kemeny NE. The role of floxuridine in metastatic liver disease. Mol Cancer Ther. 2009 May;8(5):1015-25
- [5] Yeo WS, Arya R, Kim KK, Jeong H, Cho KH, Bae T. The FDA-approved anti-cancer drugs, streptozotocin and floxuridine, reduce the virulence of Staphylococcus aureus. Sci Rep. 2018 Feb 6;8(1):2521
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 50-91-9 | 储存条件 |
|
|||||||
| 分子式 | C9H11FN2O5 | 运输 | 蓝冰 | |||||||
| 分子量 | 246.19 | 别名 | 5-Fluorouracil 2'-deoxyriboside;Deoxyfluorouridine;FDUR;NSC-27640;5-FDU;FdUrd;5-Fluorodeoxyuridine;5-FUDR;NSC 26740;floxuridin;fluorodeoxyuridine;fluorouridine deoxyribose;fluoruridine deoxyribose. FUDF. 5FUDR;FUdR. WR138720;氟尿苷 | |||||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| 786-0 cells | - | Function assay | 48 h | Anticancer activity against human 786-0 cells after 48 hrs by sulforhodamine B assay, GI50=0.1 μM | 19744858 |
| A2780 cells | - | Cytotoxicity assay | 5 days | Cytotoxicity against human A2780 cells after 5 days by MTT assay, IC50=0.026 μM | 22738636 |
| A498 cells | - | Function assay | 48 h | Anticancer activity against human A498 cells after 48 hrs by sulforhodamine B assay, GI50=1.25893 μM | 19744858 |
| A549 cells | - | Function assay | 48 h | Anticancer activity against human A549 cells after 48 hrs by sulforhodamine B assay, GI50=0.01259 μM | 19744858 |
| A549 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human A549 cells after 72 hrs by MTT assay, EC50=9.74 μM | 19917528 |
| A549 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human A549 cells assessed as cell viability after 72 hrs by WST-8 assay, IC50=0.047 μM | 22248856 |
| A549 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human A549 cells after 72 hrs by microplate reader method, IC50=0.0124 μM | 22847019 |
| ACHN cells | - | Function assay | 48 h | Anticancer activity against human ACHN cells after 48 hrs by sulforhodamine B assay, GI50=0.03162 μM | 19744858 |
| BALB/3T3 cell | - | Proliferation assay | 72 h | Antiproliferative activity against mouse BALB/3T3 cells assessed as growth inhibition after 72 hrs by SRB method, IC50=23.9 μM | 25644674 |
| BT549 cells | - | Function assay | 48 h | Anticancer activity against human BT549 cells after 48 hrs by sulforhodamine B assay, GI50=1 μM | 19744858 |
| Caki1 cells | - | Function assay | 48 h | Anticancer activity against human Caki1 cells after 48 hrs by sulforhodamine B assay, GI50=0.03162 μM | 19744858 |
| CCRF-CEM | - | Growth inhibition assay | - | In vitro concentration required for 50% inhibition of growth of human leukemia cell line CCRF-CEM with hPAP (0.2 unit/mL), GI50=0.0006 μM | 11597404 |
| CCRF-CEM cell | - | Growth inhibition assay | - | Tested in vitro for the inhibition of cell growth of human T lymphoblastoid CCRF-CEM cell line (ATCC CCL 119), IC50=0.5 μM | 11909716 |
| CCRFCEM cells | - | Function assay | - | Cytostatic activity against human CCRFCEM cells by MTT assay, IC50=0.29 μM | 17804231 |
| CCRF-CEM cells | - | Growth inhibition assay | 72 h | Cytostatic activity against human CCRF-CEM cells ATCC CCL 119 assessed as growth reduction after 72 hrs, IC50=0.017 μM | 17997319 |
| CCRF-CEM cells | - | Growth inhibition assay | 72 h | Cytostatic activity in human CCRF-CEM cells assessed as inhibition of cell growth after 72 hrs, IC50=0.017 μM | 18078757 |
| CCRF-CEM cells | - | Function assay | 48 h | Anticancer activity against human CCRF-CEM cells after 48 hrs by sulforhodamine B assay, GI50=0.00631 μM | 19744858 |
| CEM cells | - | Function assay | 72 h | Cytostatic activity against human CEM cells expressing human ENT1 transporter after 72 hrs by cell counting in presence of NBMPR, IC50=0.8 μM | 21892829 |
| CEM/0 cells | - | Function assay | 72 h | Cytostatic activity against human CEM/0 cells after 72 hrs by cell counting, IC50=0.022 μM | 21892829 |
| COLO205 cells | - | Function assay | 48 h | Anticancer activity against human COLO205 cells after 48 hrs by sulforhodamine B assay, GI50=2.51189 μM | 19744858 |
| Colo-357 cells | 100 μM | Function assay | 15 mins | Activity at MRP5 in human Colo-357 cells assessed as 5-FdUMP accumulation at 100 uM after 15 mins | 20930123 |
| DU145 cells | - | Function assay | 48 h | Anticancer activity against human DU145 cells after 48 hrs by sulforhodamine B assay, GI50=0.25119 μM | 19744858 |
| EKVX cells | - | Function assay | 48 h | Anticancer activity against human EKVX cells after 48 hrs by sulforhodamine B assay, GI50=10 μM | 19744858 |
| FM3A cells | - | Function assay | 2 days | Cytostatic activity against mouse FM3A cells after 2 days by coulter counting analysis, IC50=0.0094 μM | 21330014 |
| HCC2998 cells | - | Function assay | 48 h | Anticancer activity against human HCC2998 cells after 48 hrs by sulforhodamine B assay, GI50=1.58489 μM | 19744858 |
| HCT116 cells | - | Function assay | 48 h | Anticancer activity against human HCT116 cells after 48 hrs by sulforhodamine B assay, GI50=0.001 μM | 19744858 |
| HCT15 cells | - | Function assay | 48 h | Anticancer activity against human HCT15 cells after 48 hrs by sulforhodamine B assay, GI50=0.12589 μM | 19744858 |
| HeLa cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HeLa cells after 72 hrs by MTT assay, EC50=10.26 μM | 19917528 |
| HeLa cells | - | Function assay | - | Cytostatic activity against human HeLa cells in presence of 20 uM thymidine, IC50=8.5 μM | 21330014 |
| Hela cells | - | Function assay | 72 h | Cytostatic activity against human HeLa cells after 72 hrs by cell counting, IC50=0.05 μM | 21892829 |
| HeLa cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HeLa cells after 72 hrs by SRB assay, IC50=6.5 μM | 23867603 |
| HepG2 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human HepG2 cells after 72 hrs by MTT assay, EC50=18.84 μM | 19917528 |
| HFF cells | - | Function assay | 72 h | Antiparasitic activity against Toxoplasma gondii ATCC 50839 infected in HFF cells after 72 hrs by beta-galactosidase reporter gene assay, EC50=0.91 μM | 17698618 |
| HL60 cells | - | Growth inhibition assay | 72 h | Cytostatic activity against human HL60 cells ATCC CCL 240 assessed as growth reduction after 72 hrs, IC50=0.012 μM | 17997319 |
| HL60 cells | - | Growth inhibition assay | 72 h | Cytostatic activity in human HL60 cells assessed as inhibition of cell growth after 72 hrs, IC50=0.012 μM | 18078757 |
| HL60 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human HL60 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50=0.24 μM | 25644674 |
| HL-60(TB) cells | - | Function assay | 48 h | Anticancer activity against human HL-60(TB) cells after 48 hrs by sulforhodamine B assay, GI50=0.19953 μM | 19744858 |
| HOP62 cells | - | Function assay | 48 h | Anticancer activity against human HOP62 cells after 48 hrs by sulforhodamine B assay, GI50=0.02512 μM | 19744858 |
| HOP92 cells | - | Function assay | 48 h | Anticancer activity against human HOP92 cells after 48 hrs by sulforhodamine B assay, GI50=0.79433 μM | 19744858 |
| Hs 578T cells | - | Function assay | 48 h | Anticancer activity against human Hs 578T cells after 48 hrs by sulforhodamine B assay, GI50=3.98107 μM | 19744858 |
| HT1080 cells | - | Function assay | - | Cytostatic activity against human HT1080 cells by MTT assay, IC50=0.18 μM | 17804231 |
| HT-29 cells | - | Function assay | 48 h | Anticancer activity against human HT-29 cells after 48 hrs by sulforhodamine B assay, GI50=1.99526 μM | 19744858 |
| human ACHN cells | - | Function assay | - | Anticancer activity against human ACHN cells by SRB assay, GI50=2.1 μM | 20732810 |
| human SR cells | - | Function assay | 48 h | Anticancer activity against human SR cells after 48 hrs by sulforhodamine B assay, GI50=0.01259 μM | 19744858 |
| IGROV1 cells | - | Function assay | 48 h | Anticancer activity against human IGROV1 cells after 48 hrs by sulforhodamine B assay, GI50=2.51189 μM | 19744858 |
| K562 cells | - | Function assay | 48 h | Anticancer activity against human K562 cells after 48 hrs by sulforhodamine B assay, GI50=0.79433 μM | 19744858 |
| KB cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human KB cells after 72 hrs by SRB assay, IC50=8.69 μM | 23867603 |
| KBALB cell | - | Cytotoxicity assay | - | In vitro cell cytotoxicity against KBALB cell line (transformed fibroblast sarcoma cell line), CC50=6.00E-05 μM | 12620076 |
| KBALB-STK cell | - | Cytotoxicity assay | - | In vitro cell cytotoxicity against KBALB-STK cell lines expressed in HSV-1 TK, IC50=8.80E-05 μM | 12620076 |
| KM12 cells | - | Function assay | 48 h | Anticancer activity against human KM12 cells after 48 hrs by sulforhodamine B assay, GI50=2.51189 μM | 19744858 |
| L1210 cells | - | Growth inhibition assay | 72 h | Cytostatic activity against mouse L1210 cells ATCC CCL219 assessed as growth reduction after 72 hrs, IC50=0.012 μM | 17997319 |
| L1210 cells | - | Growth inhibition assay | 72 h | Cytostatic activity in mouse L1210 cells assessed as inhibition of cell growth after 72 hrs, IC50=0.012 μM | 18078757 |
| L1210 cells | - | Function assay | 15 mins | Inhibition of thymidylate synthase in mouse L1210 cells assessed as inhibition of tritium release from [5-3H]deoxyuridine after preincubation for 15 mins by liquid scintillation counting, IC50=0.0006 μM | 21330014 |
| L1210 mouse leukemia cells | - | Growth inhibition assay | 24 h | Growth inhibition in L1210 mouse leukemia cells after 24 h treatment, IC50=0.0041 μM | 11728193 |
| L1210 mouse leukemia cells | - | Growth inhibition assay | 48 h | Growth inhibition in L1210 mouse leukemia cells after 48 hr treatment, IC50=0.00064 μM | 11728193 |
| L1210 mouse leukemia cells | - | Function assay | 2 h | Thymidylate synthase inhibition in L1210 mouse leukemia cells after 2 hr treatment, IC50=0.0079 μM | 11728193 |
| L1210/0 cells | - | Function assay | 48 h | Cytostatic activity against mouse L1210/0 cells after 48 hrs by cell counting, IC50=0.0009 μM | 21892829 |
| L5178Y cell | - | Growth inhibition assay | - | Comparative inhibition of L5178Y cell growth in vitro (concentration required for 50% inhibition), IC50=0.00076 μM | 6779007 |
| L5178Y cells | - | Function assay | - | Inhibitory concentration of compound was calculated on L5178Y cells by [14C]Leu incorporation, IC50=2 μM | 8246229 |
| LAC cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human LAC cells after 72 hrs by MTT assay, EC50=32.09 μM | 19917528 |
| LLC cells | - | Cytotoxicity assay | 24 h | Cytotoxicity against mouse LLC cells after 24 hrs by resazurin assay, IC50=14.2 μM | 21536448 |
| LLC cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against mouse LLC cells after 72 hrs by resazurin assay, IC50=2 μM | 21536448 |
| LM cells | - | Function assay | 2 h | Thymidylate synthase inhibition in thymidine kinase deficient LM cells after 2 hr treatment, IC50=5.4 μM | 11728193 |
| LMTK cells | - | Cytotoxicity assay | 5 days | Cytotoxicity against thymidine kinase-deficient mouse LMTK cells after 5 days by MTT assay, IC50=4.5 μM | 22738636 |
| LNCaP cells | - | Cytotoxicity assay | - | Cytotoxic concentration in prostate specific antigen (PSA) producing human LNCaP cells, IC50=0.0692 μM | 12161157 |
| LoVo cells | - | Proliferation assay | 72 h | Antiproliferative activity against human LoVo cells assessed as growth inhibition after 72 hrs by SRB method, IC50=19.07 μM | 25644674 |
| LOXIMVI cells | - | Function assay | 48 h | Anticancer activity against human LOXIMVI cells after 48 hrs by sulforhodamine B assay, GI50=0.02512 μM | 19744858 |
| M14 cells | - | Function assay | 48 h | Anticancer activity against human M14 cells after 48 hrs by sulforhodamine B assay, GI50=0.15849 μM | 19744858 |
| MALME-3M cells | - | Function assay | 48 h | Anticancer activity against human MALME-3M cells after 48 hrs by sulforhodamine B assay, GI50=7.94328 μM | 19744858 |
| MCF7 cells | - | Function assay | 48 h | Anticancer activity against human MCF7 cells after 48 hrs by sulforhodamine B assay, GI50=0.00631 μM | 19744858 |
| MCF7 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against human MCF7 cells after 72 hrs by SRB assay, IC50=12.19 μM | 23867603 |
| MDA-MB-231 cells | - | Function assay | 48 h | Anticancer activity against human MDA-MB-231 cells after 48 hrs by sulforhodamine B assay, GI50=3.98107 μM | 19744858 |
| MDA-MB-231 cells | - | Cytotoxicity assay | - | Cytotoxicity against human MDA-MB-231 cells overexpressing urokinase plasminogen activator, IC50=0.21 μM | 20363130 |
| MDA-MB-231 cells | - | Function assay | - | Anticancer activity against human MDA-MB-231 cells by SRB assay, GI50=0.16 μM | 20732810 |
| MDA-MB-435 cells | - | Function assay | 48 h | Anticancer activity against human MDA-MB-435 cells after 48 hrs by sulforhodamine B assay, GI50=3.16228 μM | 19744858 |
| MDA-N cells | - | Function assay | 48 h | Anticancer activity against human MDA-N cells after 48 hrs by sulforhodamine B assay, GI50=1.25893 μM | 19744858 |
| MOLT4 cells | - | Function assay | 48 h | Anticancer activity against human MOLT4 cells after 48 hrs by sulforhodamine B assay, GI50=0.03981 μM | 19744858 |
| NCI/ADR-RES cells | - | Function assay | 48 h | Anticancer activity against human NCI/ADR-RES cells after 48 hrs by sulforhodamine B assay, GI50=1.25893 μM | 19744858 |
| NCI-H226 cells | - | Function assay | 48 h | Anticancer activity against human NCI-H226 cells after 48 hrs by sulforhodamine B assay | 19744858 |
| NCI-H23 | - | Function assay | 48 h | Anticancer activity against human NCI-H23 cells after 48 hrs by sulforhodamine B assay, GI50=10 μM | 19744858 |
| NCI-H322 | - | Function assay | 48 h | Anticancer activity against human NCI-H322M cells after 48 hrs by sulforhodamine B assay, GI50=0.50119 μM | 19744858 |
| NCI-H460 | - | Function assay | 48 h | Anticancer activity against human NCI-H460 cells after 48 hrs by sulforhodamine B assay, GI50=0.50119 μM | 19744858 |
| NCI-H522 | - | Function assay | 48 h | Anticancer activity against human NCI-H522 cells after 48 hrs by sulforhodamine B assay, GI50=0.002 μM | 19744858 |
| OVCAR-3 cells | - | Function assay | 48 h | Anticancer activity against human OVCAR-3 cells after 48 hrs by sulforhodamine B assay, GI50=2.51189 μM | 19744858 |
| OVCAR4 cells | - | Function assay | 48 h | Anticancer activity against human OVCAR4 cells after 48 hrs by sulforhodamine B assay, GI50=10 μM | 19744858 |
| OVCAR5 cells | - | Function assay | 48 h | Anticancer activity against human OVCAR5 cells after 48 hrs by sulforhodamine B assay, GI50=6.30957 Μm | 19744858 |
| OVCAR8 cells | - | Function assay | 48 h | Anticancer activity against human OVCAR8 cells after 48 hrs by sulforhodamine B assay, GI50=0.12589 μM | 19744858 |
| PC3 cells | - | Function assay | 48 h | Anticancer activity against human PC3 cells after 48 hrs by sulforhodamine B assay, GI50=0.31623 μM | 19744858 |
| PC3 cells | - | Function assay | - | Anticancer activity against human PC3 cells by SRB assay, GI50=4.97 μM | 20732810 |
| RAW264.7 cells | - | Cytotoxicity assay | 72 h | Cytotoxicity against mouse RAW264.7 cells after 72 hrs by resazurin assay, IC50=30 μM | 21536448 |
| RPMI8266 cells | - | Function assay | 48 h | Anticancer activity against human RPMI8266 cells after 48 hrs by sulforhodamine B assay, GI50=0.79433 μM | 19744858 |
| RXF393 cells | - | Function assay | 48 h | Anticancer activity against human RXF393 cells after 48 hrs by sulforhodamine B assay, GI50=3.98107 μM | 19744858 |
| SF268 cells | - | Function assay | 48 h | Anticancer activity against human SF268 cells after 48 hrs by sulforhodamine B assay, GI50=10 μM | 19744858 |
| SF295 cells | - | Function assay | 48 h | Anticancer activity against human SF295 cells after 48 hrs by sulforhodamine B assay, GI50=0.01259 μM | 19744858 |
| SF539 cells | - | Function assay | 48 h | Anticancer activity against human SF539 cells after 48 hrs by sulforhodamine B assay, GI50=0.03981 μM | 19744858 |
| SK-MEL-2 cells | - | Function assay | 48 h | Anticancer activity against human SK-MEL-2 cells after 48 hrs by sulforhodamine B assay, GI50=10 μM | 19744858 |
| SK-MEL-28 cells | - | Function assay | 48 h | Anticancer activity against human SK-MEL-28 cells after 48 hrs by sulforhodamine B assay, GI50=1.99526 μM | 19744858 |
| SK-MEL-5 cells | - | Function assay | 48 h | Anticancer activity against human SK-MEL-5 cells after 48 hrs by sulforhodamine B assay, GI50=0.19953 μM | 19744858 |
| SKOV3 cells | - | Function assay | 48 h | Anticancer activity against human SKOV3 cells after 48 hrs by sulforhodamine B assay, GI50=1.99526 μM | 19744858 |
| SN12C cells | - | Function assay | 48 h | Anticancer activity against human SN12C cells after 48 hrs by sulforhodamine B assay, GI50=0.19953 μM | 19744858 |
| SNB19 cells | - | Function assay | 48 h | Anticancer activity against human SNB19 cells after 48 hrs by sulforhodamine B assay, GI50=1.99526 μM | 19744858 |
| SNB75 cells | - | Function assay | 48 h | Anticancer activity against human SNB75 cells after 48 hrs by sulforhodamine B assay, GI50=0.19953 μM | 19744858 |
| SW620 cells | - | Function assay | 48 h | Anticancer activity against human SW620 cells after 48 hrs by sulforhodamine B assay, GI50=6.30957 μM | 19744858 |
| SW707 cells | - | Proliferation assay | 72 h | Antiproliferative activity against human SW707 cells assessed as growth inhibition after 72 hrs by SRB method, IC50=23.86 μM | 25644674 |
| T47D cells | - | Function assay | 48 h | Anticancer activity against human T47D cells after 48 hrs by sulforhodamine B assay, GI50=1.25893 μM | 19744858 |
| TK10 cells | - | Function assay | 48 h | Anticancer activity against human TK10 cells after 48 hrs by sulforhodamine B assay, GI50=5.01187 μM | 19744858 |
| TSU cells | - | Cytotoxicity assay | - | Cytotoxic concentration in non prostate specific antigen (PSA) producing human TSU cells, IC50=0.058 μM | 12161157 |
| U251 cells | - | Function assay | 48 h | Anticancer activity against human U251 cells after 48 hrs by sulforhodamine B assay, GI50=0.12589 μM | 19744858 |
| UACC257 cells | - | Function assay | 48 h | Anticancer activity against human UACC257 cells after 48 hrs by sulforhodamine B assay, GI50=3.16228 μM | 19744858 |
| UACC62 cells | - | Function assay | 48 h | Anticancer activity against human UACC62 cells after 48 hrs by sulforhodamine B assay, GI50=0.03981 μM | 19744858 |
| UO31 cells | - | Function assay | 48 h | Anticancer activity against human UO31 cells after 48 hrs by sulforhodamine B assay, GI50=0.12589 μM | 19744858 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00296322 | - | - | Completed | - | - |
| NCT00296322 | Stomach Cancer | Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT00296335 | Stomach Cancer | Phase 3 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT00296335 | - | - | Completed | - | - |
| NCT01815359 | Appendix Cancer ... more >> Colorectal Cancer Collapse << | Phase 2 | Recruiting | March 2019 | United States, Florida ... more >> University of Miami Recruiting Miami, Florida, United States Contact: Floriano Marchetti, MD 305-243-9110 United States, Missouri Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Sean Glasglow, MD 314-747-7222 Principal Investigator: Benjamin Tan, MD United States, New Jersey Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Garrett Nash, MD, MPH 212-639-8668 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Garrett Nash, MD, MPH 212-639-8668 Memorial Sloan Kettering Bergen Recruiting Montvale, New Jersey, United States, 07645 Contact: Garrett Nash, MD,MPH 212-639-8668 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Garrett Nash, MD, MPH 212-639-8668 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Garrett Nash, MD, MPH 212-639-8668 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Garrett Nash, MD, MPH 212-639-8668 Contact: Andrea Cercek, MD 646-888-4189 Principal Investigator: Garrett Nash, MD, MPH Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Garrett Nash, MD, MPH 212-639-8668 Collapse << |
| NCT01401075 | Gastric Cancer | Phase 4 | Completed | - | Korea, Republic of ... more >> ASAN Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01312857 | Metastatic Colorectal Cancer | Phase 2 | Recruiting | March 2019 | United States, New Jersey ... more >> Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Nancy Kemeny, MD 646-888-4180 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Nancy Kemeny, MD 646-888-4180 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Nancy Kemeny, MD, PhD 646-888-4180 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Nancy Kemeny, MD 646-888-4180 Contact: Michael D'Angelica, MD 212-639-3226 Principal Investigator: Nancy Kemeny, MD Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Nancy Kemeny, MD 646-888-4180 Contact: Michael D'Angelica, MD 212-639-3226 Principal Investigator: Nancy Kemeny, MD Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Nancy Kemeny, MD 646-888-4180 Collapse << |
| NCT03693807 | Colorectal Cancer ... more >> Cholangiocarcinoma Collapse << | Phase 2 | Recruiting | September 2020 | United States, New Jersey ... more >> Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Nancy Kemeny, MD 646-888-4180 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Nancy Kemeny, MD 646-888-4180 Memorial Sloan Kettering Bergen Recruiting Montvale, New Jersey, United States, 07645 Contact: Nancy Kemeny, MD 646-888-4180 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Nancy Kemeny, MD, PhD 646-888-4180 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Nancy Kemeny, MD 646-888-4180 Principal Investigator: Nancy Kemeny, MD Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Nancy Kemeny, MD 646-888-4180 Principal Investigator: Nancy Kemeny, MD Memorial Sloan Kettering Rockville Centre Recruiting Rockville Centre, New York, United States, 11570 Contact: Nancy Kemeny, MD 646-888-4180 Collapse << |
| NCT01348412 | Colorectal Cancer ... more >> Liver Metastases Collapse << | Phase 2 | Completed | - | France ... more >> Centre Georges François Leclerc Dijon, France, 21000 Collapse << |
| NCT00020501 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 3 | Completed | - | United States, Maryland ... more >> Warren Grant Magnuson Clinical Center - NCI Clinical Studies Support Bethesda, Maryland, United States, 20892-1182 Collapse << |
| NCT03069950 | Colorectal Adenocarcinoma Meta... more >>static to the Liver Collapse << | Phase 2 | Recruiting | February 2020 | United States, New Jersey ... more >> Memorial Sloan Kettering Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Andrea Cercek, MD 646-888-4189 Principal Investigator: Andrea Cercek, MD Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Andrea Cercek, MD 646-888-4189 United States, New York Memorial Sloan Kettering Commack Recruiting Commack, New York, United States, 11725 Contact: Andrea Cercek, MD Principal Investigator: Andera Cercek, MD Memoral Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Andrea Cercek, MD 646-888-4189 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Andrea Cercek, MD 646-888-4189 Contact: Michael D'Angelica, MD 212-639-3226 Principal Investigator: Andrea Cercek, MD Collapse << |
| NCT00244348 | Colorectal Cancer ... more >> Metastasis Liver Cancer Collapse << | Phase 1 Phase 2 | Withdrawn(Investigator has lef... more >>t institution) Collapse << | - | United States, Wisconsin ... more >> Medical College of Wisconsin/ Froedtert Hospital Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT02788201 | Urothelial Carcinoma ... more >> Bladder Cancer Urinary Bladder Neoplasms Collapse << | Phase 2 | Recruiting | July 1, 2020 | United States, Maryland ... more >> National Institutes of Health Clinical Center Recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT00408551 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Unknown | - | United States, Indiana ... more >> Center for Cancer Care at Goshen General Hospital Recruiting Goshen, Indiana, United States, 46526 Contact: Clinical Trials Office - Center for Cancer Care at Goshen Gene 574-535-2858 Collapse << |
| NCT01042691 | Unresectable Colorectal Liver ... more >>Metastases Collapse << | Phase 1 | Completed | - | United States, Pennsylvania ... more >> UPMC Presbyterian Pittsburgh, Pennsylvania, United States, 15213 UPMC Cancer Centers, Hillman Cancer Center Pittsburgh, Pennsylvania, United States, 15232 UPMC Shadyside Pittsburgh, Pennsylvania, United States, 15232 Collapse << |
| NCT00019760 | Recurrent Colon Cancer ... more >> Liver Metastases Stage IV Rectal Cancer Recurrent Rectal Cancer Stage IV Colon Cancer Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> Surgery Branch Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00643877 | Colorectal Neoplasms | Phase 3 | Unknown | December 2014 | China ... more >> Zhongshan Hospital, Fudan University Recruiting Shanghai, China, 200032 Principal Investigator: jianmin xu, MD Collapse << |
| NCT00004103 | Gastric Cancer | Phase 2 | Completed | - | United States, New York ... more >> NYU Cancer Institute at New York University Medical Center New York, New York, United States, 10016 Collapse << |
| NCT02529774 | Resected Liver Metastases From... more >> Colorectal Cancer Collapse << | Phase 2 Phase 3 | Not yet recruiting | September 2021 | China, Shanghai ... more >> Fudan University Shanghai Cancer Center China Not yet recruiting Shanghai, Shanghai, China, 200032 Contact: Ye Xu, Dr. +86 21 64175590 xu_shirley021@163.com Contact: Hongtu Zheng, Dr. +86 180 1731 7711 zht5863@126.com Collapse << |
| NCT00008294 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT03678428 | Metastatic Colorectal Cancer ... more >> Liver Metastases Collapse << | Phase 3 | Not yet recruiting | December 31, 2022 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Guangzhou, Guangdong, China, 510060 Collapse << |
| NCT02402972 | Colorectal Cancer ... more >> Metastasis Collapse << | Phase 3 | Recruiting | February 2022 | China ... more >> Zhongshan Hospital, Fudan University Recruiting Shanghai, China, 200032 Contact: Wenju Chang, MD 86-21-13764476150 Principal Investigator: jianmin xu, MD Collapse << |
| NCT00448682 | Gastric Cancer | Phase 2 | Terminated | - | United States, Florida ... more >> University of Miami Sylvester Comprehensive Cancer Center - Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00006038 | Esophageal Cancer ... more >> Gastric Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00002783 | Gastric Cancer | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00448682 | - | - | Terminated | - | - |
| NCT00522509 | - | - | Unknown | - | Italy ... more >> Department of Surgery - Liver Unit, Scientific Institute San Raffaele, Milan, Italy, 20132 Collapse << |
| NCT00448760 | Esophageal Cancer | Phase 2 | Completed | - | United States, Florida ... more >> University of Miami Sylvester Comprehensive Cancer Center - Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00448760 | - | - | Completed | - | - |
| NCT01525069 | Cholangiocarcinoma ... more >> Liver Neoplasms Collapse << | Phase 1 | Recruiting | June 30, 2022 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: William Chapman, M.D. 314-362-7792 chapmanw@wustl.edu Contact: Tracey Guthrie 314-747-4404 guthriet@wustl.edu Sub-Investigator: William Chapman, M.D. Sub-Investigator: M.B. Majella Doyle, M.D. Sub-Investigator: William Hawkins, M.D. Sub-Investigator: Jeffrey Lowell, M.D. Sub-Investigator: Joel Picus, M.D. Sub-Investigator: Steven Strasberg, M.D. Sub-Investigator: Rama Suresh, M.D. Sub-Investigator: Andrea Wang-Gillam, M.D., Ph.D. Sub-Investigator: Kathryn J. Fowler, M.D. Sub-Investigator: Benjamin Tan, M.D. Sub-Investigator: John Kotyk, M.D. Collapse << |
| NCT00001449 | Neoplasms | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00059930 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 1 | Active, not recruiting | June 2019 | United States, New Jersey ... more >> Cancer Institute of New Jersey at UMDNJ - Robert Wood Johnson Medical School New Brunswick, New Jersey, United States, 08903 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00002716 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 3 | Completed | - | United States, Iowa ... more >> CCOP - Cedar Rapids Oncology Project Cedar Rapids, Iowa, United States, 52403-1206 CCOP - Iowa Oncology Research Association Des Moines, Iowa, United States, 50309-1016 John Stoddard Cancer Center at Iowa Methodist Medical Center Des Moines, Iowa, United States, 50309 Mercy Cancer Center at Mercy Medical Center-Des Moines Des Moines, Iowa, United States, 50314 Iowa Lutheran Hospital Des Moines, Iowa, United States, 50316-2301 United States, Nebraska Midlands Cancer Center at Midlands Community Hospital Papillion, Nebraska, United States, 68128-4157 United States, New Mexico MBCCOP - University of New Mexico HSC Albuquerque, New Mexico, United States, 87131 United States, Ohio MetroHealth Medical Center Cleveland, Ohio, United States, 44109 United States, Pennsylvania Penn State Cancer Institute at Milton S. Hershey Medical Center Hershey, Pennsylvania, United States, 17033-0850 Fox Chase Cancer Center Philadelphia, Pennsylvania, United States, 19111-2497 United States, Wisconsin CCOP - St. Vincent Hospital Cancer Center, Green Bay Green Bay, Wisconsin, United States, 54307-3453 Australia, New South Wales Westmead Hospital Westmead, New South Wales, Australia, 2145 Peru Instituto de Enfermedades Neoplasicas Lima, Peru, 34 Puerto Rico San Juan City Hospital San Juan, Puerto Rico, 00936-7344 Collapse << |
| NCT03500874 | Colorectal Cancer ... more >> Liver Metastases HAI Collapse << | Phase 3 | Recruiting | December 31, 2024 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510060 Contact: Yuhong Li, MD +86(20)87343135 kyk@sysucc.org.cn Contact: Yuhong Li, MD +86(20)87342490 liyh@sysucc.org.cn Collapse << |
| NCT00026234 | Adenocarcinoma of the Colon ... more >> Adenocarcinoma of the Rectum Liver Metastases Recurrent Colon Cancer Recurrent Rectal Cancer Stage IV Colon Cancer Stage IV Rectal Cancer Collapse << | Phase 2 | Completed | - | United States, Minnesota ... more >> North Central Cancer Treatment Group Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00005049 | Colorectal Cancer ... more >> Gastric Cancer Gastrointestinal Carcinoid Tumor Gastrointestinal Stromal Tumor Ovarian Cancer Peritoneal Cavity Cancer Small Intestine Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> NYU School of Medicine's Kaplan Comprehensive Cancer Center New York, New York, United States, 10016 Collapse << |
| NCT00183911 | Gastric Adenocarcinoma ... more >> Gastric Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> U.S.C. / Norris Comprehensive Cancer Center Los Angeles, California, United States, 90033 Collapse << |
| NCT03366155 | Colorectal Cancer ... more >> Liver Metastases Colorectal Adenocarcinoma Colorectal Cancer With Hepatic Metastases Colorectal Carcinoma Collapse << | Phase 2 | Not yet recruiting | December 30, 2022 | United States, Maryland ... more >> National Institutes of Health Clinical Center Not yet recruiting Bethesda, Maryland, United States, 20892 Contact: For more information at the NIH Clinical Center contact National Cancer Institute Referral Office 888-624-1937 Collapse << |
| NCT01862315 | Intrahepatic Cholangiocarcinom... more >>a Peripheral Cholangiocarcinoma Cholangiolar Carcinoma Cholangiocellular Carcinoma Collapse << | Phase 2 | Recruiting | May 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Recruiting Basking Ridge, New Jersey, United States Contact: William Jarnagin, MD 212-639-7601 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: William R. Jarnagin, MD 212-639-7601 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Recruiting Commack, New York, United States, 11725 Contact: William Jarnagin, MD 212-639-7601 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: William Jarnagin, MD 212-639-7601 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: William R Jarnagin, MD 212-639-7601 Contact: Nancy Kemeny, MD 646-888-4180 Principal Investigator: William R Jarnagin, MD Memorial Sloan Kettering at Mercy Medical Center Recruiting Rockville Centre, New York, United States Contact: William Jarnagin, MD 212-639-7601 Collapse << |
| NCT00645710 | Liver Metastases ... more >> Recurrent Colon Cancer Recurrent Rectal Cancer Stage IV Colon Cancer Stage IV Rectal Cancer Collapse << | Phase 1 Phase 2 | Completed | - | United States, California ... more >> City of Hope Duarte, California, United States, 91010 Collapse << |
| NCT01692704 | Cholangiocellular Carcinoma | Phase 1 Phase 2 | Completed | - | Switzerland ... more >> University Hospital Zurich, Department of Oncology Zurich, ZH, Switzerland, 8091 Collapse << |
| NCT00449163 | Colorectal Cancer | Phase 2 | Terminated(Study terminated by... more >> University of Miami Institutional Review Board) Collapse << | - | United States, Florida ... more >> University of Miami Miami, Florida, United States, 33136 Collapse << |
| NCT00200200 | Hepatic Metastases ... more >> Colon Cancer Rectal Cancer Collapse << | Phase 2 | Active, not recruiting | November 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan-Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan-Kettering Cancer Center 1275 York Avenue New York, New York, United States, 10021 Memorial Sloan-Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memoral Sloan Kettering Cancer Center at Phelps Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT00858338 | Gastric Cancer ... more >> Stomach Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Bellevue Hospital New York, New York, United States, 10016 NYU Cancer Center New York, New York, United States, 10016 NYU Tisch Hospital New York, New York, United States, 10016 Collapse << |
| NCT00449163 | - | - | Terminated(Study terminated by... more >> University of Miami Institutional Review Board) Collapse << | - | - |
| NCT00004142 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas - MD Anderson Cancer Center Houston, Texas, United States, 77030-4009 Collapse << |
| NCT02102789 | Metastatic Colorectal Cancer | Phase 3 | Recruiting | December 2024 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510060 Contact: Yu-hong Li, MD, Ph D liyh@sysucc.org.cn Principal Investigator: Yu-hong Li, MD, Ph D Collapse << |
| NCT00410956 | Liver Cancer | Phase 2 | Active, not recruiting | July 2019 | United States, New York ... more >> New York Weill Cornell Cancer Center at Cornell University New York, New York, United States, 10021 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00002842 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010-3000 Collapse << |
| NCT00003753 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT03493061 | Colo-rectal Cancer ... more >> Liver Metastases Collapse << | Phase 2 | Recruiting | December 30, 2022 | China, Guangdong ... more >> Sun Yat-sen University Cancer Center Recruiting Guangzhou, Guangdong, China, 510060 Contact: Li Yuhong, MD 020-87342487 liyh@sysucc.org.cn Principal Investigator: Li Yuhong, MD Collapse << |
| NCT00001576 | Colorectal Neoplasm ... more >> Liver Neoplasm Neoplasm Metastasis Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Cancer Institute (NCI) Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00492999 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Active, not recruiting | May 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States Memoral Sloan Kettering Cancer Center@Phelps Memorial Hospital Sleepy Hollow, New York, United States Collapse << |
| NCT00005860 | Cancer | Phase 1 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT00002842 | - | - | Completed | - | - |
| NCT00063960 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 2 | Completed | - | United States, Arkansas ... more >> Arkansas Cancer Research Center at University of Arkansas for Medical Sciences Little Rock, Arkansas, United States, 72205 United States, Kentucky Central Baptist Hospital Lexington, Kentucky, United States, 40503-9985 United States, Massachusetts Massachusetts General Hospital Cancer Center Boston, Massachusetts, United States, 02114 United States, Minnesota University of Minnesota Cancer Center Minneapolis, Minnesota, United States, 55455 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 United States, North Carolina Lineberger Comprehensive Cancer Center at University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599-7213 Comprehensive Cancer Center at Wake Forest University Winston-Salem, North Carolina, United States, 27157-1082 United States, Oklahoma Integris Oncology Services Oklahoma City, Oklahoma, United States, 73112 United States, Rhode Island University Medical Group Providence, Rhode Island, United States, 02908-4735 Collapse << |
| NCT00361842 | Colorectal Neoplasms | Phase 2 | Completed | - | United States, California ... more >> California Cancer Center Greenbrae, California, United States, 94904 United States, District of Columbia Lombardi Comprehensive Cancer Research Institute, Georgetown University Medical Center Washington DC, District of Columbia, United States, 20057 United States, Florida NW Oncology & Hematology Associates Coral Springs, Florida, United States, 33065 Broward Oncology Associates Fort Lauderdale, Florida, United States, 33308 United States, Georgia St. Joseph's/Candler Health System Inc. Savannah, Georgia, United States, 31405 United States, North Carolina Presbyterian Hospital Charlotte,, North Carolina, United States, 28204 United States, Ohio Gabrail Cancer Center Canton, Ohio, United States, 44718 United States, Oklahoma Cancer Care Oklahoma Oklahoma City, Oklahoma, United States Cancer Care Oklahoma Tulsa, Oklahoma, United States United States, South Carolina South Carolina Oncology Association Columbia, South Carolina, United States, 29210 United States, Tennessee Sarah Cannon Research Institute Nashville, Tennessee, United States, 37203 Canada, Alberta Cross Cancer Institute Edmonton, Alberta, Canada, T6G1Z2 Canada, Quebec Sir Mortimer B. Davis Jewish General Hospital Montreal, Quebec, Canada, H3T1E2 Collapse << |
| NCT00587067 | - | - | Completed | - | - |
| NCT00587067 | Hepatic Cancer | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT01938729 | Intrahepatic Cholangiocarcinom... more >>a Peripheral Cholangiocarcinoma Cholangiolar Carcinoma Cholangiocellular Carcinoma) (ICC) Collapse << | Phase 1 | Active, not recruiting | September 2019 | United States, New Jersey ... more >> Memorial Sloan Kettering Cancer Center at Basking Ridge Basking Ridge, New Jersey, United States United States, New York Memorial Sloan Kettering Cancer Center at Commack Commack, New York, United States, 11725 Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Memorial Sloan Kettering Cancer Center at Mercy Medical Center Rockville Centre, New York, United States, 11570 Memorial Sloan Kettering Cancer Center Sleepy Hollow Sleepy Hollow, New York, United States, 10591 Collapse << |
| NCT00848783 | Gastric Cancer ... more >> Gastric Adenocarcinoma Esophageal Cancer Collapse << | Phase 2 | Terminated(Due to slow accrual... more >>) Collapse << | - | United States, California ... more >> Norris Cancer Center Los Angeles, California, United States, 90033 United States, New York Bellevue Hospital New York, New York, United States, 10016 NYU Cancer Center New York, New York, United States, 10016 Collapse << |
| NCT00848783 | - | - | Terminated(Due to slow accrual... more >>) Collapse << | - | - |
| NCT00268463 | Colorectal Cancer ... more >> Metastatic Cancer Collapse << | Phase 3 | Terminated(The study was termi... more >>nated due to low accrual.) Collapse << | - | - |
| NCT00695201 | Colon Cancer ... more >>Rectal Cancer Collapse << | Phase 1 | Active, not recruiting | August 2019 | United States, New York ... more >> Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT00268463 | - | - | Terminated(The study was termi... more >>nated due to low accrual.) Collapse << | - | - |
| NCT01749397 | Stage IV Fallopian Tube Cancer... more >> AJCC v6 and v7 Stage IV Ovarian Cancer AJCC v6 and v7 Stage IV Primary Peritoneal Cancer AJCC v7 Collapse << | Phase 1 | Active, not recruiting | - | United States, Arizona ... more >> Mayo Clinic in Arizona Scottsdale, Arizona, United States, 85259 United States, Maryland Johns Hopkins University/Sidney Kimmel Cancer Center Baltimore, Maryland, United States, 21287 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, Missouri Siteman Cancer Center at Washington University Saint Louis, Missouri, United States, 63110 Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| Thymidylate Synthase | GI50:5.1μM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
