CAS No.: 117772-70-0
阿奇霉素 Catalog No. CSN10371
Synonyms: CP-62993 dihydrate;XZ 450 Dihydrate;Azitro
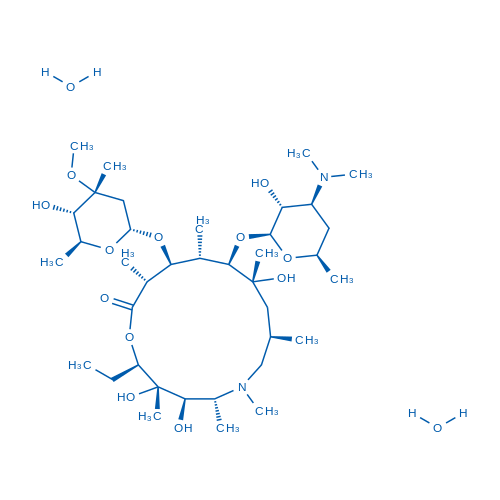
Azithromycin Dihydrate is an antibiotic for inhibition of parasite growth with IC50 of 8.4 μM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00694694 Malaria Phase 3 Completed - Tanzania ... more >> Teule Hospital Muheza, Tanga Region, Tanzania Collapse << NCT01235546 Endometritis ... more >>Wound Infection Abscess Surgical Site Infection Collapse << Not Applicable Completed - United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 United States, Louisiana Ochsner Clinic Foundation New Orleans, Louisiana, United States, 70121 United States, Mississippi University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 United States, New York Columbia University New York, New York, United States, 10032 United States, North Carolina Mission Hospital Asheville, North Carolina, United States, 28801 University of North Carolina Chapel Hill, North Carolina, United States, 27599-7516 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555-0587 University of Texas Health Science Center at Houston Houston, Texas, United States, 77225 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Collapse << NCT02527005 Malaria Pregn... more >>ant HIV Collapse << Phase 1 Completed - Nigeria ... more >> University College Hospital, Ibadan, Oyo, Nigeria, 200001 Collapse << - 更多
- 参考文献
- [1] Menzel M, Akbarshahi H, et al. Azithromycin augments rhinovirus-induced IFNβ via cytosolic MDA5 in experimental models of asthma exacerbation. Oncotarget. 2017 May 9;8(19):31601-31611.
- [2] Vandooren J, Knoops S, et al. Differential inhibition of activity, activation and gene expression of MMP-9 in THP-1 cells by azithromycin and minocycline versus bortezomib: A comparative study. PLoS One. 2017 Apr 3;12(4):e0174853.
- [3] Bakheit AH, Al-Hadiya BM, Abd-Elgalil AA. Azithromycin. Profiles Drug Subst Excip Relat Methodol. 2014;39:1‐40
- [4] Menzel M, Akbarshahi H, Tufvesson E, Persson C, Bjermer L, Uller L. Azithromycin augments rhinovirus-induced IFNβ via cytosolic MDA5 in experimental models of asthma exacerbation. Oncotarget. 2017;8(19):31601‐31611
- [5] Wei S, Mortensen MS, Stokholm J, et al. Short- and long-term impacts of azithromycin treatment on the gut microbiota in children: A double-blind, randomized, placebo-controlled trial. EBioMedicine. 2018;38:265‐272
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 117772-70-0 | 储存条件 |
|
|||||
| 分子式 | C38H76N2O14 | 运输 | 蓝冰 | |||||
| 分子量 | 785.02 | 别名 | CP-62993 dihydrate;XZ 450 Dihydrate;Azitro;CP-62993;Goxal;Odaz;Ribotrex;Toraseptol;Ultreon;XZ-450;Zenstavion;Zithromax;Zmax;阿奇霉素二水合物;阿奇霉素二水物 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00694694 | Malaria | Phase 3 | Completed | - | Tanzania ... more >> Teule Hospital Muheza, Tanga Region, Tanzania Collapse << |
| NCT01235546 | Endometritis ... more >>Wound Infection Abscess Surgical Site Infection Collapse << | Not Applicable | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35233 United States, Louisiana Ochsner Clinic Foundation New Orleans, Louisiana, United States, 70121 United States, Mississippi University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 United States, New York Columbia University New York, New York, United States, 10032 United States, North Carolina Mission Hospital Asheville, North Carolina, United States, 28801 University of North Carolina Chapel Hill, North Carolina, United States, 27599-7516 United States, Texas University of Texas Medical Branch Galveston, Texas, United States, 77555-0587 University of Texas Health Science Center at Houston Houston, Texas, United States, 77225 United States, Utah University of Utah Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT02527005 | Malaria Pregn... more >>ant HIV Collapse << | Phase 1 | Completed | - | Nigeria ... more >> University College Hospital, Ibadan, Oyo, Nigeria, 200001 Collapse << |
| NCT01955252 | Yaws | Phase 3 | Completed | - | Papua New Guinea ... more >> Lihir Medical Centre Londolovit, New ireland province, Papua New Guinea, 034 Collapse << |
| NCT01778634 | Ureaplasma Infections | Phase 2 | Active, not recruiting | December 2019 | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35249-7335 United States, Delaware Christiana Care Health Services Newark, Delaware, United States, 19713 United States, Maryland University of Maryland School of Medicine Baltimore, Maryland, United States, 21201 Mercy Medical Center Baltimore, Maryland, United States, 21202 Johns Hopkins University Baltimore, Maryland, United States, 21287 United States, Tennessee Monroe Carell Jr. Children's Hospital at Vanderbilt Nashville, Tennessee, United States, 37232-9544 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908-0386 Collapse << |
| NCT01235546 | - | - | Completed | - | - |
| NCT01382004 | Yaws Treponem... more >>a Infection Neglected Tropical Disease Collapse << | Phase 3 | Completed | - | Papua New Guinea ... more >> Lihir Medical Centre Kavieng, New Ireland Province, Papua New Guinea, 034 Collapse << |
| NCT02336516 | Haemolytic and Uremic Syndrome | Not Applicable | Recruiting | October 2021 | France ... more >> Arnaud de Villeneuve Hospital Recruiting Montpellier, France, 34295 Montpellier cedex 5 Contact: Marc Fila, MD +33467336428 m-fila@chu-montpellier.fr Contact: Georges Deschenes, MD, PhD +33140031242 georges.deschenes@rdb.aphp.fr Collapse << |
| NCT01778634 | - | - | Active, not recruiting | - | - |
| NCT00237445 | Pneumonia | Phase 4 | Terminated | - | United States, New Jersey ... more >> Sanofi-Aventis Bridgewater, New Jersey, United States, 08807 Collapse << |
| NCT01227395 | - | - | Completed | - | - |
| NCT00356772 | Eye Infections, Bacterial | Phase 1 | Completed | - | - |
| NCT03672630 | Mycobacterium Avium Complex ... more >> Nontuberculous Mycobacterium Infection Collapse << | Phase 2 Phase 3 | Not yet recruiting | February 28, 2023 | - |
| NCT02960503 | Sickle Cell Disease | Phase 1 Phase 2 | Withdrawn(Key personnel moved ... more >>to new location. Therefore, we had insufficient resources to initiate the trial.) Collapse << | October 2018 | United States, Tennessee ... more >> Vanderbilt University Medical Center Nashville, Tennessee, United States, 37203 Collapse << |
| NCT01178762 | - | - | Completed | - | - |
| NCT01227395 | - | - | Completed | - | - |
| NCT01178762 | Chlamydial Conjunctivitis | Not Applicable | Completed | - | Taiwan ... more >> National Taiwan University Hospital Taipei, Taiwan, 100 Collapse << |
| NCT02107274 | Bronchiectasis | Phase 4 | Completed | - | Malaysia ... more >> Respiratory Unit, Taiping Hospital Taiping, Perak, Malaysia, 34000 Collapse << |
| NCT01329588 | Neisseria Gonorrhoeae Infectio... more >>n Chlamydia Trachomatis HIV Collapse << | Phase 4 | Completed | - | Benin ... more >> Dispensaire des infections sexuellement transmissibles Cotonou, Benin Collapse << |
| NCT02107274 | - | - | Completed | - | - |
| NCT00649831 | Bronchitis, Chronic | Phase 3 | Completed | - | France ... more >> Pfizer Investigational Site Anzin, France, 59410 Pfizer Investigational Site Avignon, France, 84 000 Pfizer Investigational Site Avignon, France, 84000 Pfizer Investigational Site Dijon, France, 21000 Pfizer Investigational Site Equeurdreville Hainneville, France, 50120 Pfizer Investigational Site Escaudain, France, 59 124 Pfizer Investigational Site Le Grand Quevilly, France, 76120 Pfizer Investigational Site Les Lilas, France, 93 260 Pfizer Investigational Site Lyon, France, 69001 Pfizer Investigational Site Mantes La Jolie, France, 78200 Pfizer Investigational Site Maromme, France, 76150 Pfizer Investigational Site Marseille, France, 13 008 Pfizer Investigational Site Maubeuge, France, 59600 Pfizer Investigational Site Mont Saint Martin, France, 54350 Pfizer Investigational Site Nancy, France, 54000 Pfizer Investigational Site Paris, France, 75 016 Pfizer Investigational Site Paris, France, 75012 Pfizer Investigational Site Paris, France, 75017 Pfizer Investigational Site Paris, France, 75020 Pfizer Investigational Site Saint Aulaire, France, 19 130 Pfizer Investigational Site Savigny, France, 91600 Pfizer Investigational Site Soissons, France, 02 200 Pfizer Investigational Site Tourcoing, France, 59200 Pfizer Investigational Site Tulette, France, 26 790 Pfizer Investigational Site Valenton, France, 94460 Pfizer Investigational Site Vaux S/ Seine, France, 78740 Pfizer Investigational Site Villejuif, France, 94800 Pfizer Investigational Site Vincennes, France, 94300 Pfizer Investigational Site Vitry, France, 94 400 Pfizer Investigational Site Wattrelos, France, 59150 Pfizer Investigational Site Yerres, France, 91330 Collapse << |
| NCT01009619 | Bronchiolitis Obliterans Syndr... more >>ome Graft Rejection Lymphocytic Bronchiolitis Respiratory Infection Collapse << | Phase 4 | Completed | - | Belgium ... more >> Katholieke Universiteit Leuven and University Hospital Gasthuisberg Leuven, Belgium, B-3000 Collapse << |
| NCT03578679 | Pediculoses | Not Applicable | Recruiting | December 30, 2018 | Senegal ... more >> Institut de Recherche pour le Developpement Recruiting Dakar, Senegal, 18524 Contact: Laurent Vidal, PhD 338493530 ext 221 laurent.vidal@ird.fr Contact: Laurent Gournac, PhD 338493530 ext 221 laurent.gournac@ird.fr Collapse << |
| NCT01103713 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT01009619 | - | - | Completed | - | - |
| NCT00286026 | Trachoma | Phase 4 | Withdrawn(Prevalence of infect... more >>ion for screened population too low (<7%) to enroll anyone.) Collapse << | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94143 Collapse << |
| NCT01103713 | Asymptomatic Parasitemia In Pr... more >>egnancy Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | Benin ... more >> Centre de Sante d'AHOUANSORI -AGUE Cotonou, Benin Hôpital Bethesda Cotonou, Benin Kenya Siaya District Hospital Siaya, Kenya Malawi Zomba Central Hospital Zomba, Malawi Tanzania Teule Hospital Muheza, Tanga, Tanzania National Institute for Medical Research (Mwanza Centre)/ Nyamagana District Hospital Mwanza, Tanzania, 1903 Uganda Mulanda Health Centre IV Kampala, Uganda Collapse << |
| NCT00266851 | Asthma | Phase 3 | Completed | - | United States, Illinois ... more >> ANSR Peoria, Illinois, United States, 61602 United States, Kansas AAFP National Research Network Kansas City, Kansas, United States, 66211 United States, Ohio RAP - Cleveland Clinic Cleveland, Ohio, United States, 44130 United States, Oklahoma University of Oklahoma Health Sciences Center (OUHSC) and Oklahoma Physicians Resource/Research Network (OKPRN) Oklahoma City, Oklahoma, United States, 73104 United States, Wisconsin Wisconsin Research and Education Network (WREN) Madison, Wisconsin, United States, 53713 Collapse << |
| NCT00792922 | Trachoma | Phase 4 | Completed | - | United States, California ... more >> UCSF Proctor Foundation San Francisco, California, United States, 94143 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 United Kingdom London School of Hygiene and Tropical Medicine London, United Kingdom, WC1E 7HT Collapse << |
| NCT00347776 | - | - | Completed | - | - |
| NCT00367653 | Malaria | Phase 3 | Completed | - | Burkina Faso ... more >> Pfizer Investigational Site Ouagadougou, West Africa, Burkina Faso, 01 Pfizer Investigational Site Nouna, Burkina Faso Ghana Pfizer Investigational Site Navrongo, West Africa, Ghana Kenya Pfizer Investigational Site Kisumu, Kenya, 40100 Mali Pfizer Investigational Site Bamako, West Africa, Mali Pfizer Investigational Site Bamako, Mali Senegal Pfizer Investigational Site Senegal, West Africa, Senegal Zambia Pfizer Investigational Site Ndola, Zambia Collapse << |
| NCT00347776 | Trichiasis | Phase 4 | Completed | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21205 Collapse << |
| NCT00792922 | - | - | Completed | - | - |
| NCT00796224 | Acute Otitis Media | Phase 2 | Completed | - | Costa Rica ... more >> Pfizer Investigational Site San Jose, Costa Rica, 0000 Pfizer Investigational Site San Jose, Costa Rica Collapse << |
| NCT00796224 | - | - | Completed | - | - |
| NCT00599079 | Mycobacterium Avium Complex Lu... more >>ng Disease Collapse << | Phase 4 | Completed | - | United States, Texas ... more >> The University of Texas Health Science Center at Tyler Tyler, Texas, United States, 75708 Collapse << |
| NCT00610623 | Pneumonia, Ventilator-Associat... more >>ed Pseudomonas Infections Collapse << | Phase 2 | Terminated(The sponsor decided... more >> to stop the study prematurely because of financial issues) Collapse << | - | Belgium ... more >> Intensive Care Unit, Clinique Saint-Pierre Ottignies, Louvain, Belgium, 1340 General Intensive Car Unit, Centre Hospitalier Universitaire de Liège Liège, Belgium, B-4000 Former Serbia and Montenegro Surgical Intensive Care Unit, Clinical Center of Serbia, Institut of Neurosurgery Belgrade, Former Serbia and Montenegro Surgical Intensive Care Unit, Clinical Center of Serbia Belgrade, Former Serbia and Montenegro France Intensive Care Unit, Jean Minjoz University Hospital Besancon, France, 25030 Intensive Car Unit, Calmette University Hospital of Lille Lille, France, 59037 Medical-surgical intensive car unit, Dupuytren Teaching Hospital Limoges, France, 8700 General Intensive Care Unit, Montauban City Hospital Montauban, France, 82013 Medical Intensive Car Unit, Hospital Bichat Paris, France, 75018 Surgical Intensive Car Unit; University Hospital Bichat Paris, France, 75018 General Intensive Care Unit, Hospital Saint-Joseph Paris, France, 75674 Medical Intensive Care Unit, Cochin Hospital Paris, France, 75679 Poland Intensive Care Unit, Wojewodzki Hospital Krakow, Poland Intensive Care Unit, Wojewodzki Hospital Sosnowiec, Poland Intensive Care Unit, Central Hospital Warsaw, Poland Spain Intensive Care Unit, Hospital del Mar Barcelona, Spain, 08003 Internal Medicine, Vall d'Hebron Hospital Barcelona, Spain, 08035 Intensive Care Unit, San Dureta University Hospital Palma de Mallorca, Spain, 07014 Intensive Care Unit, Joan XXIII University Hospital Tarragona, Spain, 43007 Switzerland Surgical and Medical Intensive Care Units, University Hospital Lausanne Lausanne, Vaud, Switzerland Collapse << |
| NCT01154569 | Bariatric Surgery | Not Applicable | Completed | - | Canada, Alberta ... more >> University of Alberta Hospital Edmonton, Alberta, Canada, T6G2B7 Collapse << |
| NCT01202331 | Trachoma Chla... more >>mydia Collapse << | Phase 4 | Completed | - | Ethiopia ... more >> The Carter Center, Ethiopia Addis Ababa, Ethiopia Collapse << |
| NCT01270074 | Cystic Fibrosis ... more >> Bronchiectasis Collapse << | Phase 3 | Recruiting | December 2020 | Australia, New South Wales ... more >> Sydney Children's Hospital Recruiting Sydney, New South Wales, Australia Principal Investigator: Adam Jaffe, FRACP Westmead Children's Hospital Recruiting Sydney, New South Wales, Australia Principal Investigator: Peter Cooper, FRACP Australia, Queensland Mater Children's Hospital Recruiting Brisbane, Queensland, Australia Principal Investigator: Carolyn Dakin, FRACP Royal Children's Hospital Recruiting Brisbane, Queensland, Australia Principal Investigator: Claire Wainwright, FRACP Australia, South Australia Women's and Children's Hospital Recruiting Adelaide, South Australia, Australia Principal Investigator: Andrew Tai, FRACP Australia, Victoria Monash Medical Centre Recruiting Melbourne, Victoria, Australia Principal Investigator: David Armstrong, FRACP Royal Children's Hospital Recruiting Melbourne, Victoria, Australia Contact: Philip J Robinson, MD PhD Fracp phil.robinson@rch.org.au Principal Investigator: Philip J Robinson, MD PhD FRACP Australia, Western Australia Princess Margaret Hospital for Children Recruiting Perth, Western Australia, Australia, 6008 Principal Investigator: Barry S Clements, MBBS New Zealand Starship Hospital Recruiting Auckland, New Zealand Principal Investigator: Cass Byrnes, FRACP Christchurch Hospital Not yet recruiting Christchurch, New Zealand Principal Investigator: Philip Pattemore, FRACP Collapse << |
| NCT01327625 | Graft vs Host Disease ... more >> Bronchiolitis Obliterans Collapse << | Not Applicable | Terminated(Preliminary reports... more >> of this study was too bad.) Collapse << | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01767506 | - | - | Completed | - | - |
| NCT01617083 | PANS PANDAS ... more >> Obsessive Compulsive Disorder (OCD) Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Rothman Center for Neuropsychiatry Saint Petersburg, Florida, United States, 33701 Collapse << |
| NCT01531218 | Diarrhea Feve... more >>r Vomiting Abdominal Pain Collapse << | Phase 3 | Completed | - | Denmark ... more >> Department of Infectious Diseases, Aalborg Hospital Aalborg, Denmark, 9000 Collapse << |
| NCT01919996 | Pharyngitis T... more >>onsillitis Collapse << | Phase 2 | Terminated(See Termination Sta... more >>tement in the Detailed Description below) Collapse << | - | United States, California ... more >> Martel Eye Medical Group Rancho Cordova, California, United States, 95670 United States, Illinois Ann & Robert H. Lurie Children's Hospital of Chicago Chicago, Illinois, United States, 60611 Outpatient Center in Lincoln Park Chicago, Illinois, United States, 60614 Infant Welfare Society of Chicago Chicago, Illinois, United States, 60647 United States, Utah Murray Pediatrics Murray, Utah, United States, 84107 Daynes Eye and Lasik Salt Lake City, Utah, United States, 84124 Collapse << |
| NCT01767506 | Trachoma | Not Applicable | Completed | - | United States, Maryland ... more >> Johns Hopkins University Baltimore, Maryland, United States, 21205 Collapse << |
| NCT01617083 | - | - | Completed | - | - |
| NCT01783340 | Malaria, Falciparum | Phase 1 Phase 2 | Withdrawn(No funding) | April 2014 | Netherlands ... more >> Leiden University Medical Centre Leiden, Netherlands, 2333ZA Collapse << |
| NCT02048007 | Childhood Mortality | Phase 4 | Enrolling by invitation | December 2019 | United States, California ... more >> UCSF Proctor Foundation San Francisco, California, United States, 94143-0944 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 Malawi College of Medicine at the University of Malawi, Blantyre Blantyre, Malawi Niger The Carter Center, Niger Niamey, Niger Tanzania Kongwa Trachoma Project Kongwa, Tanzania United Kingdom London School of Hygiene & Tropical Medicine London, United Kingdom Collapse << |
| NCT02047981 | Childhood Mortality | Phase 4 | Completed | - | United States, California ... more >> UCSF Proctor Foundation San Francisco, California, United States, 94143 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21205 Malawi College of Medicine at the University of Malawi, Blantyre Blantyre, Malawi Niger The Carter Center, Niger Niamey, Niger Tanzania Kongwa Trachoma Project Kongwa, Tanzania United Kingdom London School of Hygiene & Tropical Medicine London, United Kingdom Collapse << |
| NCT01919996 | - | - | Terminated(See Termination Sta... more >>tement in the Detailed Description below) Collapse << | - | - |
| NCT02176057 | Trachoma | Phase 2 Phase 3 | Withdrawn(Nepal Ministry of He... more >>alth implemented public drug treatment prior to the proposed start date) Collapse << | - | Nepal ... more >> Geta Eye Hospital Geta, Kailali, Nepal, 10900 Collapse << |
| NCT02196493 | Chronic Cough | Phase 3 | Unknown | December 2016 | United Kingdom ... more >> Respiratory Research Unit Recruiting Nottingham, Nottinghamshire, United Kingdom, NG5 1PB Contact: Matthew Martin, MBChB 01158231935 Principal Investigator: Tim Harrison, MD, FRCP Sub-Investigator: Matthew Martin, MBChB MRCP Collapse << |
| NCT02414399 | Pneumonia Dia... more >>rrhea Malaria Co-infection Death Malnutrition Collapse << | Phase 4 | Recruiting | December 2020 | Kenya ... more >> Kisii Teaching and Referral Hospital, Homa Bay District Hospital, St. Paul's Mission Hospital Recruiting Kisii And Homa Bay Counties, Kenya Contact: Benson Singa, MBChB, MPH singabo2008@gmail.com Collapse << |
| NCT02677701 | Cystic Fibrosis | Phase 4 | Recruiting | March 31, 2020 | - |
| NCT03523156 | Trachoma | Phase 4 | Not yet recruiting | December 2020 | Ethiopia ... more >> Not yet recruiting Amhara, Ethiopia Contact: Demelash Gessesse 251 920 258 272 Contact: Zerihun Tadesse 251-91-140-1498 Zerihun.Tadesse@cartercenter.org Collapse << |
| NCT01464840 | - | - | Completed | - | - |
| NCT03045198 | Cystic Fibrosis | Phase 4 | Enrolling by invitation | December 2018 | - |
| NCT03338244 | Childhood Mortality | Phase 4 | Recruiting | July 31, 2019 | United States, California ... more >> UCSF Proctor Foundation Active, not recruiting San Francisco, California, United States, 94143-0944 Niger The Carter Center Recruiting Niamey, Niger Contact: Mohamed Salissou Kane, PhD +227 20 73 28 57 mohamedsalissoukane@yahoo.fr Collapse << |
| NCT03335072 | Trachoma | Phase 4 | Not yet recruiting | July 1, 2022 | United States, California ... more >> UCSF Proctor Foundation Active, not recruiting San Francisco, California, United States, 94143 Ethiopia The Carter Center Not yet recruiting Addis Ababa, Ethiopia Contact: Zerihun Tadesse, MD +251-116-631863 Zerihun.Tadesse@ucsf.edu Collapse << |
| NCT02557958 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Early Phase 1 | Active, not recruiting | December 2020 | - |
| NCT03568643 | Uncomplicated Severe Acute Mal... more >>nutrition Collapse << | Phase 4 | Not yet recruiting | May 2020 | - |
| NCT02630394 | Sickle Cell Disease ... more >> Acute Chest Syndrome Collapse << | Phase 1 | Withdrawn(Principal Investigat... more >>or has moved to another institution) Collapse << | - | United States, Mississippi ... more >> University of Mississippi Medical Center Jackson, Mississippi, United States, 39216 United States, Tennessee Vanderbilt University Nashville, Tennessee, United States Collapse << |
| NCT03736629 | Asthma Chronic | Phase 2 | Not yet recruiting | February 2020 | United States, Illinois ... more >> Northwestern University Not yet recruiting Chicago, Illinois, United States, 60201 Contact: Lew Smith, MD University of Chicago Not yet recruiting Chicago, Illinois, United States, 60637 Contact: Steve White, MD Collapse << |
| NCT03187834 | Child Development | Phase 4 | Active, not recruiting | September 1, 2019 | United States, California ... more >> UCSF Proctor Foundation San Francisco, California, United States, 94143 Burkina Faso Centre de Recherche en Santé de Nouna Nouna, Burkina Faso Collapse << |
| NCT01602055 | Fasting | Phase 1 | Completed | - | Indonesia ... more >> PT Clinisindo Laboratories Jakarta, Indonesia Collapse << |
| NCT01008761 | Asthma | Not Applicable | Unknown | December 2013 | Canada, Alberta ... more >> Alberta Children's Hospital Recruiting Calgary, Alberta, Canada, T3B 6A8 Contact: Janielee Williamson, RN 403-955-3186 janie.williamson@albertahealthservices.ca Contact: Paula Finnson 403-955-7873 paula.finnson@albertahealthservices.ca Principal Investigator: David Johnson, MD Sub-Investigator: Sheldon Spier, MD Sub-Investigator: James D Kellner, MD Sub-Investigator: Candice Bjornson, MD Sub-Investigator: Michael Surrette, PhD Sub-Investigator: Alberto Nettle-Aguire, PhD Stollery Children's Hospital Recruiting Edmonton, Alberta, Canada, T6G 2V7 Contact: Piush Mandhane, MD piush.mandhane@albertahealthservices.ca Principal Investigator: Piush Mandhane, MD Sub-Investigator: William Craig, MD Sub-Investigator: Darryl Adamko, MD Sub-Investigator: Bonita Lee, MD Sub-Investigator: Carina Majaesic, MD Collapse << |
| NCT01586169 | Parasitic Diseases ... more >> Bacterial Diseases Collapse << | Not Applicable | Completed | - | Mali ... more >> Centre National D'Appui a la lutte contre la Maladie Bamako, Mali Collapse << |
| NCT01915082 | Disorder Related to Lung Trans... more >>plantation Collapse << | Phase 4 | Completed | - | Belgium ... more >> University Hospitals Leuven Leuven, Belgium, B-3000 Collapse << |
| NCT01464840 | - | - | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Birmingham, Alabama, United States, 35249 Collapse << |
| NCT00643539 | Tonsillitis | Phase 4 | Completed | - | France ... more >> Pfizer Investigational Site Ancenis, France, 44150 Pfizer Investigational Site Asnieres Sur Seine, France, 92600 Pfizer Investigational Site Auch, France, 32000 Pfizer Investigational Site Boulogne, France, 92100 Pfizer Investigational Site Brest, France, 29200 Pfizer Investigational Site Brive, France, 19100 Pfizer Investigational Site Cannes La Boca, France, 06150 Pfizer Investigational Site Chalons En Champagne, France, 51000 Pfizer Investigational Site Chambery, France, 73000 Pfizer Investigational Site Champigny sur Marne, France, 94500 Pfizer Investigational Site Combs La Ville, France, 77380 Pfizer Investigational Site Dijon, France, 21000 Pfizer Investigational Site Draguignan, France, 33170 Pfizer Investigational Site Draguignan, France, 83300 Pfizer Investigational Site Essey Les Nancy, France, 54270 Pfizer Investigational Site Lagny, France, 77400 Pfizer Investigational Site Les Sables D`olonne, France, 85100 Pfizer Investigational Site Les Ulis, France, 91940 Pfizer Investigational Site Levallois Perret, France, 92300 Pfizer Investigational Site Marseille, France, 13006 Pfizer Investigational Site Marseille, France, 13013 Pfizer Investigational Site Marseille, France, 13014 Pfizer Investigational Site Massy, France, 91300 Pfizer Investigational Site Meudon La Foret, France, 92360 Pfizer Investigational Site Meysieu, France, 69330 Pfizer Investigational Site Millery, France, 69390 Pfizer Investigational Site Nogent Sur Marne, France, 94130 Pfizer Investigational Site Paris, France, 75004 Pfizer Investigational Site Paris, France, 75005 Pfizer Investigational Site Paris, France, 75015 Pfizer Investigational Site Paris, France, 75019 Pfizer Investigational Site Paris, France, 75020 Pfizer Investigational Site Saint Marcel, France, 71380 Pfizer Investigational Site Saint Quentin, France, 02100 Pfizer Investigational Site Salon de Provence, France, 13300 Pfizer Investigational Site Sartrouville, France, 78500 Pfizer Investigational Site Selestat, France, 67600 Pfizer Investigational Site Strasbourg, France, 67100 Pfizer Investigational Site Thionville, France, 57100 Pfizer Investigational Site Tours, France, 37000 Pfizer Investigational Site Tresses, France, 33370 Pfizer Investigational Site Vandoeuvre Les Nancy, France, 54500 Pfizer Investigational Site Varois Et Chaignot, France, 21490 Pfizer Investigational Site Vence, France, 06140 Pfizer Investigational Site Versailles, France, 78000 Collapse << |
| NCT02307825 | Chronic Rhinosinusitis ... more >> High-risk Patient Collapse << | Phase 4 | Completed | - | Canada, Quebec ... more >> Centre Hospitalier de l'Université de Montréal (CHUM) Montreal, Quebec, Canada, H2W 1T8 Collapse << |
| NCT03682640 | Diabetes Mellitus, Type 1 ... more >> Diabetes Mellitus Metabolic Disease Glucose Metabolism Disorders Endocrine System Diseases Juvenile Diabetes Insulin Dependent Diabetes Type 1 Diabetes Mellitus Inflammation Diet Modification Collapse << | Phase 2 | Recruiting | December 31, 2021 | Sweden ... more >> The Queen Silvia Children's Hospital / Sahlgrenska University Hospital Recruiting Gothenburg, Sweden Contact: Gun Forsander Collapse << |
| NCT01071161 | Cough Chronic... more >> Obstructive Pulmonary Disease Chronic Bronchitis Collapse << | Phase 3 | Completed | - | Netherlands ... more >> Isala Klinieken Zwolle, Ov, Netherlands, 8011 JW Collapse << |
| NCT01667692 | Peptic Ulcer ... more >>Dyspepsia H-pylori Collapse << | Phase 4 | Completed | - | Iran, Islamic Republic of ... more >> Behnam Baghianimoghadam Yazd, Iran, Islamic Republic of, 8916863411 Collapse << |
| NCT00431964 | - | - | Completed | - | - |
| NCT02972957 | Influenza | Phase 4 | Recruiting | December 2019 | Gambia ... more >> Medical Research Council unit The Gambia Recruiting Banjul, Gambia Contact: Thushan de Silva, FRCP +220-4495442 tdesilva@mrc.gm Collapse << |
| NCT00431964 | Cystic Fibrosis | Phase 4 | Completed | - | - |
| NCT03090048 | Acne Vulgaris | Early Phase 1 | Not yet recruiting | May 31, 2019 | Canada, British Columbia ... more >> Manchanda Mc Not yet recruiting Surrey, British Columbia, Canada, V3W2T2 Contact: VINOD TAWAR, MD 604 754 2235 TAWAR.VINOD@YAHOO.COM Collapse << |
| NCT01444469 | Asthma | Phase 4 | Completed | - | United Kingdom ... more >> Barnsley Hospital NHS Foundation Barnsley, England, United Kingdom, S75 2EP Countess of Chester Hospital NHS Foundation Trust Liverpool, England, United Kingdom, CH2 1UL Surrey & Sussex Healthcare NHS Trust Surrey, England, United Kingdom, RH1 5RH Heart of England NHS Foundation Trust Birmingham, United Kingdom, B9 5SS Blackpool Teaching Hospitals NHS Foundation Trust Blackpool, United Kingdom, FY3 8NR University of Glasgow Glasgow, United Kingdom, G12 0YN University Hospitals of Leicester NHS Foundation Trust Leicester, United Kingdom, LE39QP Imperial College Healthcare NHS Trust London, United Kingdom, W2 1PG University Hospital of South Manchester Foundation Trust Manchester, United Kingdom, M23 9QZ Newcastle upon Tyne Hospitals NHS Foundation Trust Newcastle, United Kingdom, NE7 7DN Nottingham University Hospitals NHS Trust Nottingham, United Kingdom, NG5 1PB Portsmouth Hospitals NHS Trust Portsmouth, United Kingdom, PO6 3LY University Hospital of North Tees Stockton-on-Tees, United Kingdom, TS19 8PE Sherwood Forest Hospitals NHS Foundation Trust Sutton in Ashfield, United Kingdom, NG17 4JL Collapse << |
| NCT02288832 | Low Risk of Preterm Delivery | Phase 2 Phase 3 | Completed | - | France ... more >> Assistance Publique Hopitaux de Marseille Marseille, France, 13354 Hôpital Nord Assistance Publique Hôpitaux de Marseille Marseille, France, 13915 Collapse << |
| NCT00682656 | Cutaneous Leishmaniasis | Phase 2 Phase 3 | Terminated(Efficacy issues on ... more >>test arm) Collapse << | - | Brazil ... more >> Núcleo de Medicina Tropical University of Brasília - Health Center Corte de Pedras Presidente Tancredo Neves, Bahia, Brazil, 45416-000 University Estadual de Montes Claros Montes Claros, MG, Brazil, 39401-002 Centro de Pesquisas René Rachou - Fiocruz Belo Horizonte, Minas Gerais, Brazil, 30190-002 Collapse << |
| NCT00137007 | Pneumonia | Phase 4 | Completed | - | - |
| NCT00879983 | Healthy Volunteers | Phase 1 | Completed | - | China ... more >> Pfizer Investigational Site Shanghai, China, 200040 Collapse << |
| NCT00939185 | - | - | Completed | - | - |
| NCT03235141 | Bacterial Conjunctivitis | Phase 1 | Recruiting | December 30, 2017 | China, Beijing ... more >> Beijing Tongren Hospital Recruiting Beijing, Beijing, China, 100005 Contact: Feng Wu 010-58268422 trdrug@126.com Collapse << |
| NCT00830206 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute, Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00939185 | - | - | Completed | - | - |
| NCT00035347 | Community-Acquired Pneumonia (... more >>CAP) Collapse << | Phase 4 | Completed | - | - |
| NCT01102244 | Blepharitis | Phase 3 | Completed | - | United States, Massachusetts ... more >> Ora Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00830206 | - | - | Completed | - | - |
| NCT01109160 | Lymphocytic Bronchi(Oli)Tis Po... more >>st-lung Transplantation Collapse << | Phase 4 | Completed | - | Belgium ... more >> University Hospital Gasthuisberg Leuven, Vlaams-Brabant, Belgium, B-3000 Collapse << |
| NCT00834756 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St. Charles, Missouri, United States, 63301 Collapse << |
| NCT00834756 | - | - | Completed | - | - |
| NCT00875966 | Infections | Phase 1 | Completed | - | - |
| NCT00834132 | - | - | Completed | - | - |
| NCT00834132 | Healthy | Phase 1 | Completed | - | United States, Missouri ... more >> Gateway Medical Research, Inc. St. Charles, Missouri, United States, 63301 Collapse << |
| NCT00578955 | Blepharoconjunctivitis | Phase 3 | Completed | - | - |
| NCT00754949 | Blepharoconjunctivitis | Phase 3 | Withdrawn | - | - |
| NCT00367120 | Sinusitis | Phase 4 | Completed | - | - |
| NCT00830336 | Healthy | Phase 1 | Completed | - | United States, North Dakota ... more >> PRACS Institute Ltd. Fargo, North Dakota, United States, 58104 Collapse << |
| NCT00644774 | Healthy | Phase 4 | Completed | - | United States, Arizona ... more >> Scottsdale, Arizona, United States, 85251 United States, Florida St. Petersburg, Florida, United States, 33710 United States, Ohio Miamiville, Ohio, United States, 45147 Collapse << |
| NCT00564447 | Bacterial Infections ... more >> Eye Infections Collapse << | Phase 4 | Completed | - | - |
| NCT00830336 | - | - | Completed | - | - |
| NCT00865670 | Infection | Phase 1 | Completed | - | - |
| NCT00360100 | Otitis Media | Phase 3 | Completed | - | United States, Kentucky ... more >> Pfizer Investigational Site Bardstown, Kentucky, United States Pfizer Investigational Site Springfield, Kentucky, United States Argentina Pfizer Investigational Site Cordoba, Argentina Chile Pfizer Investigational Site Santiago, RM, Chile Pfizer Investigational Site Independencia, Santiago, Chile Pfizer Investigational Site Providencia, Santiago, Chile Costa Rica Pfizer Investigational Site San Jose, Costa Rica Dominican Republic Pfizer Investigational Site Santo Domingo, DN, Dominican Republic Guatemala Pfizer Investigational Site Ciudad Guatemala, Guatemala Pfizer Investigational Site Guatemala, Guatemala Panama Pfizer Investigational Site Ciudad de Panama, Panama Collapse << |
| NCT00629980 | Blepharitis | Phase 4 | Completed | - | United States, California ... more >> Sall Research Medical Center Artesia, California, United States, 90701 North Valley Eye Medical Group Mission Hills, California, United States, 91345 United States, Florida Center for Excellence in Eye Care Miami, Florida, United States, 33176 United States, Kentucky Kentuckiana Institute for Eye Research Louisville, Kentucky, United States, 40207 United States, Missouri Tauber Eye Center Kansas City, Missouri, United States, 64111 Silverstein Eye Centers Kansas City, Missouri, United States, 64133 United States, New York South Shore Eye Care, LLP Wantagh, New York, United States, 11793 United States, Tennessee Toyos Clinic Jackson, Tennessee, United States, 38301 United States, Texas Corona Research Consultants, Inc. El Paso, Texas, United States, 79904 Collapse << |
| NCT00649935 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT01408082 | Blepharitis | Phase 3 | Completed | - | - |
| NCT00998309 | - | - | Completed | - | - |
| NCT00564447 | - | - | Completed | - | - |
| NCT00105469 | - | - | Completed | - | - |
| NCT00650273 | Healthy | Phase 1 | Completed | - | United States, West Virginia ... more >> Kendle International Inc. Morgantown, West Virginia, United States, 26505 Collapse << |
| NCT00998309 | - | - | Completed | - | - |
| NCT00105534 | Bacterial Conjunctivitis | Phase 3 | Completed | - | - |
| NCT00629941 | Blepharitis | Phase 4 | Completed | - | United States, Maine ... more >> Central Maine Eye Care Lewiston, Maine, United States, 04240 United States, Massachusetts Ophthalmic Research Associates Andover, Massachusetts, United States, 01810 Collapse << |
| NCT00105534 | - | - | Completed | - | - |
| NCT00656539 | Blepharitis | Phase 4 | Completed | - | United States, New York ... more >> Ophthalmic Consultants of Long Island Lynbrook, New York, United States, 11563 Collapse << |
| NCT00105469 | Bacterial Conjunctivitis | Phase 3 | Completed | - | - |
| NCT00575367 | Bacterial Infections ... more >> Eye Infections Collapse << | Phase 4 | Completed | - | United States, New York ... more >> Mount Sinai School of Medicine New York, New York, United States, 10029 Collapse << |
| NCT00575380 | - | - | Completed | - | - |
| NCT00629590 | Blepharitis | Phase 4 | Completed | - | United States, New York ... more >> South Shore Eye Care Wantagh, New York, United States, 11793 Collapse << |
| NCT01797107 | Meibomian Gland Dysfunction | Phase 2 | Withdrawn(Lack of enrollment) | - | United States, Pennsylvania ... more >> Philadelphia Eye Associates Philadelphia, Pennsylvania, United States, 19148 Collapse << |
| NCT00575380 | Bacterial Infections ... more >> Eye Infections Cataract Extraction Collapse << | Phase 4 | Completed | - | United States, Arizona ... more >> Cornea Consultants of Arizone Phoenix, Arizona, United States, 85032 United States, Arkansas Eye Care Arkansas Little Rock, Arkansas, United States, 72205 United States, California North Valley Eye Medical Group Mission Hills, California, United States, 91345 United States, Florida Center for Excellence in Eye Care Miami, Florida, United States, 33176 Eye Center of North Florida Panama City, Florida, United States, 32405 United States, Georgia Coastal Research Associates, LLC Atlanta, Georgia, United States, 30339 United States, Kentucky Kentuckiana Institute for Eye Research Louisville, Kentucky, United States, 40207 United States, Missouri Ophthalmology Associates St. Louis, Missouri, United States, 63131 United States, New York Ophthalmic Consultants of Long Island Rockville Centre, New York, United States, 11563 United States, Pennsylvania Eye Care Specialists Kingston, Pennsylvania, United States, 18704 United States, South Carolina Medical University of South Carolina Charleston, South Carolina, United States, 29464 Glaucoma Consultants and Center for Eye Research Mount Pleasant, South Carolina, United States, 29464 United States, Texas Corona Research Consultants El Paso, Texas, United States, 79904 Collapse << |
| NCT00575367 | - | - | Completed | - | - |
| NCT00866216 | Infection | Phase 1 | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
