CAS No.: 32986-56-4
妥布霉素 Catalog No. CSN12101
Synonyms: Nebramycin Factor 6;Deoxykanamycin B;NSC 180514
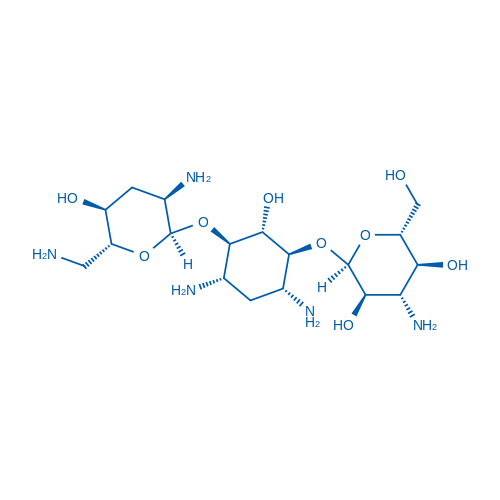
Tobramycin is an aminoglycoside, broad-spectrum antibiotic produced by Streptomyces tenebrarius.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02958514 Dry Eye Syndrome Not Applicable Unknown March 2017 - NCT01319253 - - Completed - United States, Indiana ... more >> Indiana University Indianapolis, Indiana, United States, 46202 Collapse << NCT02334384 Furunculosis Phase 1 Phase 2 Completed - United States, Virginia ... more >> Sentara Norfolk General Hospital Norfolk, Virginia, United States, 23507 Collapse << - 更多
- 参考文献
- [1] Loose M, Naber KG, et al. Urinary concentration and antimicrobial activity of tobramycin in healthy volunteers receiving a single oral dose of a novel formulation for improved absorption. Int J Antimicrob Agents. 2017 Nov 17. pii: S0924-8579(17)30396-5.
- [2] Brogden RN, Pinder RM, Sawyer PR, Speight TM, Avery GS. Tobramycin: a review of its antibacterial and pharmacokinetic properties and therapeutic use. Drugs. 1976;12(3):166–200
- [3] Brogden RN, Pinder RM, Sawyer PR, Speight TM, Avery GS. Tobramycin: a review of its antibacterial and pharmacokinetic properties and therapeutic use. Drugs. 1976;12(3):166–200
- [4] Garmyn A, Vereecken M, De Gussem K, Depondt W, Haesebrouck F, Martel A. Efficacy of Tylosin and Tilmicosin Against Experimental Mycoplasma gallisepticum Infection in Chickens. Avian Dis. 2019;63(2):359–365
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 32986-56-4 | 储存条件 |
|
|||||
| 分子式 | C18H37N5O9 | 运输 | 蓝冰 | |||||
| 分子量 | 467.51 | 别名 | Nebramycin Factor 6;Deoxykanamycin B;NSC 180514;Distobram;Gernebcin;妥布霉素 | |||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02958514 | Dry Eye Syndrome | Not Applicable | Unknown | March 2017 | - |
| NCT01319253 | - | - | Completed | - | United States, Indiana ... more >> Indiana University Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT02334384 | Furunculosis | Phase 1 Phase 2 | Completed | - | United States, Virginia ... more >> Sentara Norfolk General Hospital Norfolk, Virginia, United States, 23507 Collapse << |
| NCT02389036 | Critical Illness ... more >> Sepsis Septic Shock Ventilator Associated Pneumonia Collapse << | Phase 3 | Recruiting | December 2020 | Australia, New South Wales ... more >> The George Institute for Global Health Recruiting Sydney, New South Wales, Australia, 2000 Contact: John Myburgh, | MBBCh PhD 61 2 8052 4348 jmyburgh@georgeinstitute.org.au Collapse << |
| NCT02334384 | - | - | Completed | - | - |
| NCT02526004 | Cystic Fibrosis | Not Applicable | Unknown | October 2016 | Ireland ... more >> University College Cork Recruiting Cork, Ireland Contact: Barry P Plant, MD Principal Investigator: Marcus Mall, MD Principal Investigator: Isabelle Fajac, MD Principal Investigator: Stuart Elborn, MD Principal Investigator: Charles Haworth, MD Principal Investigator: Christopher Goss, MD Principal Investigator: Lieven Dupont, MD Collapse << |
| NCT01570192 | Bacterial Pneumonia | Phase 2 | Terminated(NIAID terminated th... more >>e study due to low subject enrollment) Collapse << | - | United States, California ... more >> InClin, Inc. San Mateo, California, United States, 94403 United States, Florida UFL Department of Medicine: Pulmonary, Critical Care and Sleep Medicine Gainesville, Florida, United States, 32610 United States, Georgia Emory University Atlanta, Georgia, United States, 30322-4250 United States, Illinois Northwestern University Chicago, Illinois, United States, 60611 United States, Iowa JMI Laboratories North Liberty, Iowa, United States, 52317 United States, Missouri Washington University in St. Louis School of Medicine Saint Louis, Missouri, United States, 63130 United States, New York Weill Cornell Medical Center of Cornell University New York, New York, United States, 10065 United States, Ohio Cleveland Clinic Lerner College of Medicine Cleveland, Ohio, United States, 44195 France Institut de Cardiologie, Groupe Hospitalier Pitie-Salpetriere Paris, France, Cedex 13 Germany Hannover Clinical Trial Center GmbH Hannover, Germany, 30625 Spain Hospital Vall d'Hebron Barcelona, Spain, 08035 Collapse << |
| NCT00532961 | Inflammation | Phase 4 | Completed | - | - |
| NCT01044719 | Cystic Fibrosis | Phase 4 | Unknown | April 2011 | United Kingdom ... more >> Department of Cystic Fibrosis, Royal Brompton Hospital Recruiting London, United Kingdom, Sw3 6NP Collapse << |
| NCT00404937 | Post-surgical Inflammation | Phase 3 | Withdrawn | - | - |
| NCT01740947 | Colorectal Cancer | Phase 4 | Terminated(Superiority was no ... more >>longer attainable) Collapse << | - | Netherlands ... more >> Slotervaart Ziekenhuis Amsterdam, Netherlands VU University Medical Center Amsterdam, Netherlands Kennemer Gasthuis Haarlem, Netherlands Spaarne Ziekenhuis Hoofddorp, Netherlands WestFries Gasthuis Hoorn, Netherlands Collapse << |
| NCT01460849 | - | - | Active, not recruiting | - | - |
| NCT02819856 | Ototoxicity | Phase 1 Phase 2 | Enrolling by invitation | September 2019 | United States, South Carolina ... more >> Medical University of South Carolina Charleston, South Carolina, United States, 29425 Collapse << |
| NCT00097773 | - | - | Completed | - | - |
| NCT03705962 | Fractures, Open | Phase 3 | Recruiting | July 2020 | United States, Virginia ... more >> University of Virginia Medical Center Recruiting Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT01570192 | - | - | Terminated(NIAID terminated th... more >>e study due to low subject enrollment) Collapse << | - | - |
| NCT01775137 | Long-term Safety of TIP | Phase 4 | Completed | - | United States, Ohio ... more >> Novartis Investigative Site Akron, Ohio, United States, 44308 United States, South Carolina Novartis Investigative Site Charleston, South Carolina, United States, 29425 United States, Texas Novartis Investigative Site Dallas, Texas, United States, 75230 Novartis Investigative Site Houston, Texas, United States, 77030 Argentina Novartis Investigative Site Caba, Buenos Aires, Argentina, C1425DTG Novartis Investigative Site Capital Federal, Buenos Aires, Argentina, C1425EFD Novartis Investigative Site Córdoba, Cordoba, Argentina, X5014AKN Australia, New South Wales Novartis Investigative Site New Lambton Heights, New South Wales, Australia, 2305 Australia, Victoria Novartis Investigative Site Parkville, Victoria, Australia, 3052 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H3T1C5 Germany Novartis Investigative Site Essen, Germany, 45147 Hungary Novartis Investigative Site Budapest, Hungary, 1121 Italy Novartis Investigative Site Firenze, FI, Italy, 50139 Novartis Investigative Site Messina, ME, Italy, 98125 Novartis Investigative Site Verona, VR, Italy, 37126 Novartis Investigative Site Palermo, Italy, 90100 Novartis Investigative Site Roma, Italy, 00161 Mexico Novartis Investigative Site Mexico, Distrito Federal, Mexico, 06720 Novartis Investigative Site Monterrey, Nuevo León, Mexico, 64020 Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Novartis Investigative Site Valencia, Comunidad Valenciana, Spain, 46026 Collapse << |
| NCT02497365 | Bacterial Keratitis | Not Applicable | Unknown | July 2017 | Canada, Quebec ... more >> McGill Academic Eye Center Recruiting Montreal, Quebec, Canada, H4A 3S9 Contact: Michael Ross, MD 51493444074 mrosscan@gmail.com Collapse << |
| NCT00097773 | Cystic Fibrosis ... more >> Pulmonary Disease, Chronic Obstructive Collapse << | Phase 2 | Completed | - | - |
| NCT01775137 | - | - | Completed | - | - |
| NCT01456780 | Meibomian Gland Dysfunction ... more >> Posterior Blepharitis Collapse << | Phase 4 | Completed | - | United States, Massachusetts ... more >> Massachusetts Eye & Ear Infirmary Boston, Massachusetts, United States, 02114 Collapse << |
| NCT01641822 | - | - | Completed | - | - |
| NCT02054156 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT01456780 | - | - | Completed | - | - |
| NCT01641822 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02137161 | Cataract Pseu... more >>doexfoliation Syndrome Collapse << | Phase 4 | Completed | - | Italy ... more >> Arcispedale Santa Maria Nuova IRCSS - Ophthalmology Reggio Emilia, RE, Italy, 42123 Collapse << |
| NCT01116089 | Cystic Fibrosis | Phase 1 | Completed | - | Czech Republic ... more >> University Hospital Brno Bohunice Brno, Czech Republic, 625 00 Moldova, Republic of SMSI Institude of Cardiology Chisinau, Moldova, Republic of, MD-2025 Slovakia University hospital with Health Center Banská Bystrica, Slovakia, 975 17 Fakultná nemocnica s poliklinikou Bratislava (FNsP) Brastislava, Slovakia, 826 06 University Hospital of L. Pasteur, Pneumonology Department Kosice, Slovakia, 041 90 Collapse << |
| NCT00357773 | Conjunctivitis, Bacterial | Phase 3 | Completed | - | - |
| NCT01315678 | Pseudomonas Aeruginosa Infecti... more >>on Collapse << | Phase 3 | Completed | - | - |
| NCT02918409 | Cystic Fibrosis | Phase 4 | Recruiting | January 2021 | United States, Colorado ... more >> National Jewish Health Recruiting Denver, Colorado, United States, 80206 Contact: Katie Poch, BS pochk@njhealth.org Principal Investigator: Milene Saavedra, MD Sub-Investigator: Jerry Nick, MD Collapse << |
| NCT00447577 | Blepharokeratoconjunctivitis | Phase 4 | Completed | - | United States, North Carolina ... more >> Cornerstone Eye Care High Point, North Carolina, United States, 27262 Collapse << |
| NCT01230593 | Chalazion Unspecified Eye, Uns... more >>pecified Eyelid Chalazion Left Eye, Unspecified Eyelid Chalazion Right Eye, Unspecified Eyelid Chalazion Both Eyes Collapse << | Not Applicable | Completed | - | United States, New York ... more >> Elmhurst Hospital Center Elmhurst, New York, United States, 11373 Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Canada, Ontario St. Joseph's Hospital Eye Clinic Stoney Creek, Ontario, Canada, L8G 5E4 Collapse << |
| NCT02248922 | Chronic Lung Infection With Ps... more >>eudomonas Aeruginosa in Cystic Fibrosis Collapse << | Phase 4 | Terminated(Reason for terminat... more >>ion was challenge with enrollment and recruitment. A significant decrease in the eligible patient population was main driver.) Collapse << | - | Germany ... more >> Novartis Investigative Site Dresden, Germany, 01307 Novartis Investigative Site Erlangen, Germany, 91054 Novartis Investigative Site Essen, Germany, 45147 Novartis Investigative Site Frankfurt, Germany, 60590 Novartis Investigative Site Germering, Germany, 82110 Novartis Investigative Site Jena, Germany, 07740 Collapse << |
| NCT02248922 | - | - | Terminated(Reason for terminat... more >>ion was challenge with enrollment and recruitment. A significant decrease in the eligible patient population was main driver.) Collapse << | - | - |
| NCT00547053 | Cystic Fibrosis | Phase 1 | Completed | - | United States, Massachusetts ... more >> Children's Hospital Boston, Massachusetts, United States, 02115 Collapse << |
| NCT01079559 | Infection Ost... more >>eoarthritis of the Knee Collapse << | Phase 3 | Withdrawn | - | Canada, Ontario ... more >> London Health Sciences Centre, University Hospital London, Ontario, Canada, N6A 5W9 Collapse << |
| NCT01155999 | Purulent Bacterial Conjunctivi... more >>tis Collapse << | Phase 3 | Completed | - | France ... more >> Clermont-Ferrand, France, 63000 Collapse << |
| NCT01230593 | - | - | Completed | - | - |
| NCT01155999 | - | - | Completed | - | - |
| NCT03464435 | Vernal Keratoconjunctivitis | Phase 4 | Completed | - | - |
| NCT00420628 | Chalazion Hor... more >>deolum Collapse << | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Pediatric Ophthalmology of Erie Erie, Pennsylvania, United States, 16501 Collapse << |
| NCT01749644 | - | - | Unknown | January 2015 | Israel ... more >> Safra Children's Hospital, Sheba Medical Center Not yet recruiting Ramat Gan, Israel Contact: Ifat Sarouk, MD 97235302884 Collapse << |
| NCT01227915 | Acute Bacterial Conjunctivitis | Phase 3 | Unknown | - | Brazil ... more >> LAL Clínica Pesquisa e Desenvolvimento Ltda Not yet recruiting Valinhos, São Paulo, Brazil Contact: Alexandre Frederico, doctor 55 19 3871-6399 alexandre@lalclinica.com.br Principal Investigator: Jose Roberto de Camargo, doctor Collapse << |
| NCT02489955 | Cystic Fibrosis | Not Applicable | Unknown | - | United Kingdom ... more >> Leeds Teaching Hospitals NHS Trust Recruiting Leeds, United Kingdom Contact: Giles Fitch giles.fitch@nhs.net Collapse << |
| NCT00004829 | Cystic Fibrosis ... more >> Bacterial Infection Collapse << | Phase 3 | Completed | - | - |
| NCT02478710 | Pneumonia, Ventilator-Associat... more >>ed Collapse << | Phase 4 | Unknown | December 2017 | United States, Ohio ... more >> Miami Valley Hospital Recruiting Dayton, Ohio, United States, 45409 Contact: Linda Clark, MSN 937-208-2913 Collapse << |
| NCT02734459 | Bilateral Cataract | Phase 3 | Completed | - | - |
| NCT01844778 | Cystic Fibrosis | Phase 4 | Completed | - | Germany ... more >> Novartis Investigative Site Berlin, Germany, 13353 Novartis Investigative Site Essen, Germany, 45147 Novartis Investigative Site München, Germany, 81241 Novartis Investigative Site Tübingen, Germany, 72076 Ireland Novartis Investigative Site Galway, Ireland Spain Novartis Investigative Site Sevilla, Andalucia, Spain, 41013 Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Novartis Investigative Site Valencia, Comunidad Valenciana, Spain, 46026 Novartis Investigative Site Palma De Mallorca, Islas Baleares, Spain, 07120 Novartis Investigative Site Madrid, Spain, 28046 Switzerland Novartis Investigative Site Basel, Switzerland, 4031 Novartis Investigative Site St. Gallen, Switzerland, 9007 Novartis Investigative Site Zürich, Switzerland, 8032 United Kingdom Novartis Investigative Site Southampton, Hampshire, United Kingdom, SO16 6YD Novartis Investigative Site Penarth, Vale of Glamorgan, United Kingdom, CF64 2XX Novartis Investigative Site Birmingham, West Midlands, United Kingdom, b9 5ss Novartis Investigative Site Bristol, United Kingdom, BS1 3NU Novartis Investigative Site East Yorkshire, United Kingdom, HU16 5JQ Novartis Investigative Site Exeter, United Kingdom, EX2 5DW Novartis Investigative Site Liverpool, United Kingdom, L14 3PE Novartis Investigative Site Newcastle upon Tyne, United Kingdom, NE1 4LP Collapse << |
| NCT00420628 | - | - | Completed | - | - |
| NCT02038803 | - | - | Unknown | - | United States, Illinois ... more >> Rush University Medical Center Recruiting Chicago, Illinois, United States, 60612 Contact: Robert A Balk, MD (312) 942-6744 robert_a_balk@rush.edu Contact: Ellen A Becker, PhD, RRT-NPS (312) 942-5000 ellen_becker@rush.edu Principal Investigator: Robert A Balk, MD Sub-Investigator: Roisin McLaughlin, RRT, RPFT Principal Investigator: Ellen A Becker, PhD,RRT-NPS,RPFT,AE-C, FAARC Collapse << |
| NCT02269969 | Burns Infecti... more >>on Collapse << | Phase 1 Phase 2 | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N3M5 Collapse << |
| NCT01844778 | - | - | Completed | - | - |
| NCT00834171 | - | - | Completed | - | United States, Virginia ... more >> Maclean, Virginia, United States Collapse << |
| NCT00834171 | - | - | Completed | - | - |
| NCT00783757 | Breast Cancer | Not Applicable | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT00794586 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT02033486 | Breast Cancer | Not Applicable | Recruiting | January 2019 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Principal Investigator: David A Boas, PhD Collapse << |
| NCT00576251 | Ocular Inflammation Associated... more >> With Blepharaconjunctivitis Collapse << | Phase 3 | Completed | - | United States, Texas ... more >> Houston Houston, Texas, United States, 77090 Collapse << |
| NCT01082367 | Treatment of Early Pulmonary I... more >>nfections With P. Aeruginosa in Cystic Fibrosis Patients Collapse << | Phase 3 | Completed | - | Canada, Ontario ... more >> Novartis Investigative Site Toronto, Ontario, Canada, M5G 1X8 Canada, Quebec Novartis Investigative Site Montreal, Quebec, Canada, H3T1C5 Egypt Novartis Investigative Site Alexandria, Egypt, 21131 France Novartis Investigative Site Bordeaux, France, 33076 Novartis Investigative Site Paris, France, 75019 Germany Novartis Investigative Site Dresden, Germany, 01307 Greece Novartis Investigative Site Thessaloniki, GR, Greece, 546 29 Novartis Investigative Site Thessaloniki, GR, Greece, 546 42 Novartis Investigative Site Patras, Greece, 265 00 Hungary Novartis Investigative Site Debrecen, Hungary, 4032 Novartis Investigative Site Debrecen, Hungary, 4043 Novartis Investigative Site Kaposvar, Hungary, 7400 Novartis Investigative Site Szeged, Hungary, 6725 Italy Novartis Investigative Site Milano, MI, Italy, 20122 Romania Novartis Investigative Site Bucuresti, Romania, 20395 Russian Federation Novartis Investigative Site Kazan, Russian Federation, 420012 Novartis Investigative Site Voronezh, Russian Federation, 394036 Novartis Investigative Site Yaroslavl, Russian Federation, 150003 Switzerland Novartis Investigative Site Zürich, Switzerland, 8032 Collapse << |
| NCT01082367 | - | - | Completed | - | - |
| NCT01400750 | Cystic Fibrosis | Phase 4 | Completed | - | Belgium ... more >> Department of pediatrics, CF center Uuiversity Hospital Leuven Leuven, Belgium, 3000 Collapse << |
| NCT02449031 | - | - | Active, not recruiting | July 31, 2023 | - |
| NCT00006280 | Cystic Fibrosis | Phase 2 | Completed | - | United States, California ... more >> Stanford University/Lucille Packard Children's Health Services at Stanford Palo Alto, California, United States, 94304 United States, Colorado The Children's Hospital Denver, Colorado, United States, 80218 United States, Maryland Johns Hopkins Hospital Baltimore, Maryland, United States, 21287 United States, Massachusetts Children's Hospital Boston, Massachusetts, United States, 02115 United States, North Carolina University of North Carolina - Chapel Hill Chapel Hill, North Carolina, United States, 27599 United States, Ohio Children's Hospital Medical Center Cincinnati, Ohio, United States, 45229 Rainbow Babies and Children's Hospital Cleveland, Ohio, United States, 44106 United States, Texas Baylor College of Medicine Houston, Texas, United States, 77030 United States, Washington Children's Hospital and Regional Medical Center Seattle, Washington, United States, 98105 Collapse << |
| NCT00198523 | Eye Infections ... more >> Postoperative Complications Collapse << | Phase 3 | Completed | - | United States, Kansas ... more >> Donald E. Beahm, MD Great Bend, Kansas, United States, 67530 United States, Massachusetts Cornea Consultants/Laser Eye Consultants of Boston Boston, Massachusetts, United States, 02114 United States, Michigan Great Lakes Eye Care St. Joseph, Michigan, United States, 49085 United States, New Hampshire Eyesight Ophthalmic Services, PA Portsmouth, New Hampshire, United States, 03801 United States, Texas Texan Eye Care PA Austin, Texas, United States, 78705 Houston Eye Associates Houston, Texas, United States, 77025 Collapse << |
| NCT00576251 | - | - | Completed | - | - |
| NCT01288170 | Cystic Fibrosis | Not Applicable | Unknown | - | France ... more >> Centre de ressources et de compétences pour la mucovisidose, enfants Recruiting Bron, France, 69500 Principal Investigator: Gabriel Bellon, Professor Centre de ressources et de compétences pour la mucovisidose, adultes Not yet recruiting Pierre-Bénite, France, 69495 Sub-Investigator: Isabelle Durieu, Professor Collapse << |
| NCT00705159 | Conjunctivitis | Phase 4 | Completed | - | United States, Pennsylvania ... more >> Pediatric Ophthalmology of Erie Erie, Pennsylvania, United States, 16501 Collapse << |
| NCT03739528 | Cataract | Phase 3 | Recruiting | March 2019 | - |
| NCT01069705 | Pulmonary Infections ... more >> Pseudomonas Aeruginosa Collapse << | Phase 3 | Completed | - | Bulgaria ... more >> Novartis Investigative Site Pleven, Bulgaria Novartis Investigative Site Polvdiv, Bulgaria Novartis Investigative Site Sofia, Bulgaria Novartis Investigative Site Varna, Bulgaria Estonia Novartis Investigative Site Tallin, Estonia Novartis Investigative Site Tartu, Estonia Latvia Novartis Investigative Site Riga, Latvia Lithuania Novartis Investigative Site Kaunas, Lithuania Novartis Investigative Site Vilnius, Lithuania Romania Novartis Investigative Site Bucharest, Romania Novartis Investigative Site Timisoara, Romania Russian Federation Novartis Investigative Site Kazan, Russian Federation Novartis Investigative Site Moscow, Russian Federation Novartis Investigative Site Saint Petersburg, Russian Federation Novartis Investigative Site Samara, Russian Federation Novartis Investigative Site Yaroslavl, Russian Federation South Africa Novartis Investigative Site Durban, South Africa Collapse << |
| NCT00982930 | Pseudomonas Aeruginosa ... more >> Cystic Fibrosis Collapse << | Phase 3 | Completed | - | Estonia ... more >> Novartis Investigative Site Tallin, Estonia Russian Federation Novartis Investigative Site Yaroslavl, Russian Federation Collapse << |
| NCT02113397 | - | - | Terminated(Insufficient enroll... more >>ment) Collapse << | - | United States, New Hampshire ... more >> Dartmouth-Hitchcock Medical Center Lebanon, New Hampshire, United States, 03756 Collapse << |
| NCT01460836 | - | - | Completed | - | - |
| NCT02712983 | Non-cystic Fibrosis Bronchiect... more >>asis Collapse << | Phase 2 | Recruiting | October 16, 2019 | - |
| NCT00992355 | Cataract | Not Applicable | Completed | - | Greece ... more >> Department of Ophthalmology, Veroia General Hospital Veroia, Greece, 59100 Collapse << |
| NCT01028027 | Conjunctivitis ... more >> Keratitis Blepharitis Collapse << | Phase 3 | Completed | - | Singapore ... more >> Bausch & Lomb Inc Singapore, Singapore, 556741 Collapse << |
| NCT01270347 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT00774072 | Cystic Fibrosis ... more >> Pseudomonas Aeruginosa Collapse << | Phase 2 | Completed | - | Germany ... more >> Universitäts-Kinderklinik Tübingen, Baden-Würtemberg, Germany, 72076 Mukoviszidosezentrum der Friedrich-Schiller-Universität Jena, Thüringen, Germany, 07745 Collapse << |
| NCT01028027 | - | - | Completed | - | - |
| NCT01111383 | Cystic Fibrosis | Phase 3 | Completed | - | France ... more >> CHR Clemenceau Caen, France, 14 033 Hopital Arnaud de Villeneuve, Clinique des maladies respiratoires Montpellier, France, 34 295 Hopital Necker Paris, France, 75 015 Poland Specjalistyczny ZOZ nad Matka i Dzieckiem, Poradnia Leczenia Mukowiscydozy Gdansk, Poland, 80-308 I Oddzial Chorob Dzieciecych, Wojewodzki Specjalistyczny Szpital Dzieciecy Kielce, Poland, 25-381 Oddzial Kliniczny Interny Dzieciecej i Alergologii, Wojewodzki Szpital Specjalistyczny Lodz, Poland, 93-513 Dzieciecy Szpital Kliniczny Akademii Medycznej, Klinika Chorob Pluc I Reumatologii Lublin, Poland, 20-093 Klinika Pneumonologii, Alergologii Dzieciecej i Immunologii Klinicznej Szpital Kliniczny Uniwersytetu Medycznego w Poznaniu Poznan, Poland, 60-572 Klinika Pneumonologii i Mukowiscydozy, Instytut Gruzlicy i Chorob Pluc w Rabce Zdroj Rabka Zdroj, Poland, 34-700 Poradnia Mukowiscydozy Wojewodzkiej, Przychodni Specjalistycznej dla Dzieci, Szpitala Wojewodzkiego Nr 2 Rzeszow, Poland, 35-301 Klinika Pediatrii Instytut Matki I Dziecka Warszawa, Poland, 01-211 Ukraine Dnipropetrovsk City Children Clinical Hospital # 2 Dnipropetrovsk, Ukraine, 49101 Donetsk Regional Children Clinical Hospital Donetsk, Ukraine, 83052 Kriviy Rig City Clinical Hospital # 8 Kriviy Rig, Ukraine, 50047 Institute of Phthysiology and Pulmonology n.a., F.G.Yanovskiy of the Academy of Medical Science of Ukraine Kyiv, Ukraine, 03680 Institute of Pediatrics, Obstetrics and Gynecology of the Academy of Medical Science of Ukraine Kyiv, Ukraine, 04050 Lviv Regional Children Specialized Clinical Hospital Lviv, Ukraine, 79035 Odesa Regional Children Clinical Hospital Odesa, Ukraine, 65031 Simferopol Central District Clinical Hospital Simferopol, Ukraine, 95033 Zaporizhya Regional Clinical Children Hospital Zaporizhya, Ukraine Collapse << |
| NCT00391976 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT03502070 | Cystic Fibrosis | Phase 4 | Recruiting | June 27, 2019 | United States, Oklahoma ... more >> Novartis Investigative Site Recruiting Oklahoma City, Oklahoma, United States, 73112 Collapse << |
| NCT00391976 | - | - | Completed | - | - |
| NCT00105469 | Bacterial Conjunctivitis | Phase 3 | Completed | - | - |
| NCT03066453 | Cystic Fibrosis | Phase 3 | Recruiting | February 2021 | France ... more >> CRCM pédiatrique - CHU d'Amiens Hôpital Nord Not yet recruiting Amiens, France Principal Investigator: Claire POULET, MD CRCM mixte - CHU de Caen Hôpital Côte de Nacre Not yet recruiting Caen, France Principal Investigator: Jacques BROUARD, MD,PhD CRCM mixte - CH de Dunkerque Not yet recruiting Dunkerque, France Principal Investigator: Manuela SCALBERT, MD CRCM mixte - CH de Lens Not yet recruiting Lens, France Principal Investigator: Sarah MITHA, MD CRCM adulte - CHRU de Lille Hôpital Calmette Recruiting Lille, France Principal Investigator: Anne Prévotat, MD CRCM pédiatrique - CHRU de Lille Hôpital Jeanne de Flandres Not yet recruiting Lille, France Principal Investigator: Nathalie WIZLA, MD CRCM mixte - CHU de Rouen Hôpital Charles Nicolle Not yet recruiting Rouen, France Principal Investigator: Stéphane DOMINIQUE, MD Collapse << |
| NCT01103401 | Cataract | Not Applicable | Completed | - | - |
| NCT00257790 | Neutropenia F... more >>ever Cancer Collapse << | Phase 4 | Completed | - | - |
| NCT02178540 | Cystic Fibrosis | Phase 4 | Completed | - | United States, Arizona ... more >> Novartis Investigative Site Phoenix, Arizona, United States, 85016 United States, California Novartis Investigative Site Long Beach, California, United States, 90806 Novartis Investigative Site Los Angeles, California, United States, 90027 Novartis Investigative Site Ventura, California, United States, 93003 United States, District of Columbia Novartis Investigative Site Washington, District of Columbia, United States, 20010-2120 United States, Florida Novartis Investigative Site Jacksonville, Florida, United States, 32207 United States, Illinois Novartis Investigative Site Chicago, Illinois, United States, 60611 United States, New York Novartis Investigative Site New York, New York, United States, 10595 United States, Ohio Novartis Investigative Site Toledo, Ohio, United States, 43606 United States, Oklahoma Novartis Investigative Site Oklahoma City, Oklahoma, United States, 73104 Novartis Investigative Site Oklahoma City, Oklahoma, United States, 73112 United States, Oregon Novartis Investigative Site Portland, Oregon, United States, 97239 United States, Texas Novartis Investigative Site Houston, Texas, United States, 77030 Collapse << |
| NCT02657473 | Non-CF Bronchiectasis | Phase 2 Phase 3 | Recruiting | August 2021 | Netherlands ... more >> North West Clinics Recruiting Alkmaar, Noord-Holland, Netherlands, 1815JD Contact: Lotte Terpstra, researcher 0031725184444 ext 5819 l.c.terpstra@nwz.nl Collapse << |
| NCT00705159 | - | - | Completed | - | - |
| NCT00125346 | Cystic Fibrosis | Phase 3 | Terminated(per Data Monitoring... more >> Committee recommendation) Collapse << | - | - |
| NCT00105469 | - | - | Completed | - | - |
| NCT00757237 | - | - | Completed | - | - |
| NCT00757237 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT02178540 | - | - | Completed | - | - |
| NCT01519661 | Pulmonary Infections ... more >> Pseudomonas Aeruginosa in Cystic Fibrosis Collapse << | Phase 4 | Completed | - | - |
| NCT01337219 | Cystic Fibrosis | Phase 1 Phase 2 | Unknown | - | France ... more >> Centre de Ressource et de Compétence Mucoviscidose Pédiatrique Centre de Référence Mucoviscidose Recruiting Lyon, France, 69500 Contact: Gabriel Bellon, Pr 04 27 85 59 82 ext 0033 gabriel.bellon@chu-lyon.fr Principal Investigator: Gabriel Bellon, Pr Collapse << |
| NCT03721328 | Prosthetic Joint Infection | Phase 2 | Recruiting | April 2019 | United States, Colorado ... more >> Sky Ridge Hospital Not yet recruiting Lone Tree, Colorado, United States, 80124 Contact: Kim Martin, Phd 303-261-7891 kim.martin@boulderclinicalscience.com United States, Indiana Ortho Indy Not yet recruiting Indianapolis, Indiana, United States, 46278 Contact: Molly Miller 317-802-2851 molmiller@orthoindy.com Principal Investigator: Edward Helman, MD United States, Michigan Covenant Hospital Recruiting Saginaw, Michigan, United States, 48604 Contact: Dianne Androsuk 989-583-5173 dianne.androsuk@chs-mi.com Principal Investigator: Brian de Beaubian, MD United States, North Carolina Ortho Carolina Research Institute Recruiting Charlotte, North Carolina, United States, 28207 Contact: Amy Roznowski 704-323-2263 amy.roznowski@orthocarolina.com Contact: Christi Cadd 704-323-2263 Christi.Cadd@orthocarolina.com Principal Investigator: Bryan Springer, MD United States, Pennsylvania University of Pittsburgh Not yet recruiting Pittsburgh, Pennsylvania, United States, 15261 Contact: Tina Vita, BSN 412-864-3654 vitatm@upmc.edu Principal Investigator: Kenneth Urish, MD Collapse << |
| NCT01953367 | Cystic Fibrosis | Phase 1 | Completed | - | Germany ... more >> Inamed GmbH Gauting, Germany, 82131 Collapse << |
| NCT01519661 | - | - | Completed | - | - |
| NCT02212587 | Cystic Fibrosis ... more >> Burkholderia Cepacia Infection Collapse << | Phase 1 | Completed | - | Canada, Ontario ... more >> St. Michael's Hospital Toronto, Ontario, Canada, M5B 1W8 Hospital for Sick Children Toronto, Ontario, Canada, M5G 1X8 Collapse << |
| NCT03341741 | Cystic Fibrosis With Pulmonary... more >> Manifestations Collapse << | Phase 3 | Completed | - | - |
| NCT00634192 | Pseudomonas Infections | Phase 3 | Completed | - | Germany ... more >> Novartis Investigator Site Berlin, Germany Novartis Investigator Site Frankfurt, Germany Novartis Investigator site Halle/Saale, Germany Novartis Investigator Site Hamburg, Germany Novartis Investigator Site Hannover, Germany Novartis Investigator Site Heidelberg, Germany Novartis Investigator Site Koeln, Germany Novartis Investigator Site Munich, Germany Collapse << |
| NCT00918957 | - | - | Completed | - | - |
| NCT00420836 | Cystic Fibrosis | Phase 1 | Completed | - | - |
| NCT00885365 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT01677403 | Bronchiectasis | Phase 4 | Unknown | December 2014 | China, Shandong ... more >> Shandong University Recruiting Jinan, Shandong, China, 250100 Collapse << |
| NCT00918957 | Cystic Fibrosis | Phase 3 | Completed | - | Bulgaria ... more >> Novartis Investigative Site Pleven, Bulgaria Novartis Investigative Site Plovdiv, Bulgaria Novartis Investigative Site Sofia, Bulgaria Novartis Investigative Site Varna, Bulgaria Egypt Novartis Investigative Site Alexandria, Egypt Novartis Investigative Site Giza, Egypt Estonia Novartis Investigative Site Tallin, Estonia Novartis Investigative Site Tartu, Estonia India Novartis Investigative Site Chandigarh, India Novartis Investigative Site Hyderabad, India Novartis Investigative Site New Delhi, India Novartis Investigative Site Vellore, India Latvia Novartis Investigative Site Riga, Latvia Lithuania Novartis Investigative Site Kaunas, Lithuania Novartis Investigative Site Vilnius, Lithuania Romania Novartis Investigative Site Bucuresti, Romania Novartis Investigative Site Timisoara, Romania Russian Federation Novartis Investigative Site Kazan, Russian Federation Novartis Investigative Site Moscow, Russian Federation Novartis Investigative Site Saint Petersburg, Russian Federation Novartis Investigative Site Samara, Russian Federation Novartis Investigative Site Voronezh, Russian Federation Novartis Investigator Site Yaroslavi, Russian Federation South Africa Novartis Investigative Site Durban, South Africa Collapse << |
| NCT02440828 | Ventilator Associated Pneumoni... more >>a (VAP) Collapse << | Phase 4 | Unknown | July 2018 | Netherlands ... more >> Erasmus MC Recruiting Rotterdam, Netherlands, 3000CA Contact: Rogier Hoek, MD +31-10-7034744 r.hoek@erasmusmc.nl Contact: Menno Van der eerden, MD, PhD +31-10-7034744 m.vandereerden@erasmusmc.nl Spain Hospital Clinic Not yet recruiting Barcelona, Spain, 08036 Contact: A Torres, MD PhD Prof 93 227 5779 ATORRES@clinic.ub.es Collapse << |
| NCT02677701 | Cystic Fibrosis | Phase 4 | Recruiting | March 31, 2020 | - |
| NCT02015663 | Cystic Fibrosis | Phase 4 | Terminated | - | United States, Alabama ... more >> Novartis Investigative Site Mobile, Alabama, United States, 36608-1128 United States, California Novartis Investigative Site Los Angeles, California, United States, 90027 Novartis Investigative Site Sacramento, California, United States, 95819 Novartis Investigative Site Ventura, California, United States, 93003 United States, Florida Novartis Investigative Site Altamonte Springs, Florida, United States, 32701 Novartis Investigative Site Jacksonville, Florida, United States, 32207 Novartis Investigative Site Miami, Florida, United States, 33136 Novartis Investigative Site Orlando, Florida, United States, 32806 Novartis Investigative Site Pensacola, Florida, United States, 32504 United States, Illinois Novartis Investigative Site Glenview, Illinois, United States, 60025 United States, Indiana Novartis Investigative Site Indianapolis, Indiana, United States, 46202 United States, Iowa Novartis Investigative Site Iowa City, Iowa, United States, 52242 United States, Missouri Novartis Investigative Site St. Louis, Missouri, United States, 63104 United States, New York Novartis Investigative Site New Hyde Park, New York, United States, 11040 Novartis Investigative Site New York, New York, United States, 10595 United States, Ohio Novartis Investigative Site Akron, Ohio, United States, 44308 United States, Oklahoma Novartis Investigative Site Oklahoma City, Oklahoma, United States, 73112 United States, Oregon Novartis Investigative Site Portland, Oregon, United States, 92772 United States, Pennsylvania Novartis Investigative Site Hershey, Pennsylvania, United States, 17033-8050 United States, Texas Novartis Investigative Site Austin, Texas, United States, 78723 United States, Utah Novartis Investigative Site Salt Lake City, Utah, United States, 84132 Collapse << |
| NCT00399945 | Cystic Fibrosis | Phase 1 | Completed | - | United Kingdom ... more >> Stoke on Trent, United Kingdom Collapse << |
| NCT02015663 | - | - | Terminated | - | - |
| NCT00388505 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT00388505 | - | - | Completed | - | - |
| NCT00695435 | - | - | Completed | - | - |
| NCT00695435 | Dry Eye | Phase 1 | Completed | - | United States, Texas ... more >> Contact Alcon Call Center For Trial Locations Fort Worth, Texas, United States, 76134 Collapse << |
| NCT01608555 | Cystic Fibrosis | Phase 4 | Completed | - | Italy ... more >> IRCCS Ospedale Maggiore Policlinico via F. Sforza 35 Milan, Italy, 20122 Collapse << |
| NCT00885365 | - | - | Completed | - | - |
| NCT02888730 | Cystic Fibrosis ... more >> Rhinosinusitis Lung Diseases Collapse << | Phase 3 | Recruiting | January 2019 | France ... more >> Centre Hospitalier Universitaire de Clermont ferrand Not yet recruiting Clermont Ferrand, France, 63003 Contact: Laurent Gilain, MD PhD lgilain@chu-clermontferrand.fr Principal Investigator: Laurent Gilain, MD PhD Sub-Investigator: Andre Labbe, MD PhD Sub-Investigator: Thierry Mom, MD Sub-Investigator: Nicolas Saroul, MD Sub-Investigator: Marie Christine Heraud, MD Sub-Investigator: Sylvie Montcouquiol, MD Centre Hospitalier Intercommunal de Créteil Recruiting Creteil, France, 94000 Contact: Sandrine LACOUR, PhD 33 1 57 02 23 98 sandrine.lacour@chicreteil.fr Principal Investigator: Virginie Escabasse, MD PhD Sub-Investigator: Andre Coste, MD PhD Sub-Investigator: Ralph Epaud, MD Sub-Investigator: Bruno Housset, MD PhD Sub-Investigator: Laurence Bassinet, MD Centre Hospitalier Universitaire de la Timone Not yet recruiting Marseille, France, 13385 Contact: Justin Michel, MD justin.michel@ap-hm.fr Principal Investigator: Justin Michel, MD Sub-Investigator: Jean Christophe Dubus, MD PhD Sub-Investigator: Richard Nicollas, MD Sub-Investigator: Martine Reynaud Gaubert, MD PhD Sub-Investigator: Patrick Dessi, MD PhD Sub-Investigator: Laurent Mely, MD Sub-Investigator: Nadine Dufeu, MD Centre Hospitalier Universitaire De Nantes Not yet recruiting Nantes, France, 44093 Contact: Olivier Malard, MD PhD olivier.malard@chu-nantes.fr Principal Investigator: Olivier Malard, MD PhD Sub-Investigator: Alain Haloun, MD Sub-Investigator: Valérie David, MD Centre Hosiptalier de Nice Not yet recruiting Nice, France, 06002 Contact: Sylvie Leroy, MD leroy.s2@chu-nice.fr Principal Investigator: Sylvie Leroy, MD Sub-Investigator: Sonanda Bailleux, MD Sub-Investigator: Laurent Castillo, MD PhD Sub-Investigator: carole Bailly, MD Centre Hospitalier Universitaire de Toulouse Not yet recruiting Toulouse, France, 31059 Contact: Josiane Percodani, MD percodani.j@chu-toulouse.fr Principal Investigator: Josiane Percodani, MD Sub-Investigator: Elie Serrano, MD PhD Sub-Investigator: Marie Noelle Calmels, MD Sub-Investigator: Marlène Murris, MD Sub-Investigator: Laurent Tetu, MD Sub-Investigator: François Bremont, MD Sub-Investigator: Géraldine Labouret, MD Sub-Investigator: Marie Mittaine, MD Collapse << |
| NCT01207245 | Cystic Fibrosis | Phase 4 | Completed | - | United Kingdom ... more >> Nottingham University Hospitals NHS Trust Nottingham, Nottinghamshire, United Kingdom, NG7 2UH Collapse << |
| NCT03715322 | Bronchiectasis Adult ... more >> Pseudomonas Infections Collapse << | Phase 3 | Not yet recruiting | June 2020 | China, Guangdong ... more >> The First Affiliated Hospital of Guangzhou Medical University Not yet recruiting Guangzhou, Guangdong, China, 510120 Contact: Nan-shan Zhong, MD +86-13609003622 nanshan@vip.163.com Principal Investigator: Wei-jie Guan, PhD Principal Investigator: Guo-jun Zhang, MD Sub-Investigator: Yong-hua Gao, PhD Principal Investigator: Jie-ming Qu, MD Principal Investigator: Jin-fu Xu, MD Principal Investigator: Yuan-lin Song, MD Principal Investigator: Jian-ping Zhao, MD Principal Investigator: Xiao-nan Tao, MD Principal Investigator: Zong-an Liang, MD Principal Investigator: Ping Chen, MD Principal Investigator: Qi-chang Lin, MD Principal Investigator: Yi-jiang Huang, MD Principal Investigator: Xue-dong Liu, MD Principal Investigator: Xing-xiang Xu, MD Principal Investigator: Wei Zhang, MD Principal Investigator: Chang-jun Du, MD Principal Investigator: Xing-lin Gao, MD Collapse << |
| NCT02035488 | Bronchiectasis | Phase 1 Phase 2 | Completed | - | Netherlands ... more >> University Medical Center Groningen Groningen, Netherlands Collapse << |
| NCT03485456 | Cystic Fibrosis | Phase 1 Phase 2 | Recruiting | May 2019 | Netherlands ... more >> University Medical Center Groningen Recruiting Groningen, Netherlands Contact: Anne Akkerman-Nijland, Drs. Collapse << |
| NCT01938417 | - | - | Completed | - | - |
| NCT02207426 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Clinical Nottingham, United Kingdom, NG11 6JS Collapse << |
| NCT02102152 | Bronchiectasis With Chronic In... more >>fection With Pseudomonas Aeruginosa Collapse << | Not Applicable | Unknown | May 2016 | Israel ... more >> Pulmonary Institute, Rabin Medical Center, Beilinson Campus Not yet recruiting Petach Tikva, Israel, 49100 Principal Investigator: Mordechai Kramer, Prof Sub-Investigator: Leonardo Fuks, Dr Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
