| NCT02367066 | Type 2 Diabetes Mellitus | Phase 1 | Completed | - | United States, California
... more >>
Research Site
Chula Vista, California, United States Collapse << |
| NCT00766415 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | Germany
... more >> Research Site
Frieburg, Germany
Research Site
GROßHANSDORF, Germany
Research Site
Hannover, Germany
Netherlands
Research Site
Amsterdam, Netherlands
United Kingdom
Research Site
Leicester, United Kingdom
Research Site
London, United Kingdom
Research Site
Manchester, United Kingdom
Research Site
Newcastle upon Tyne, United Kingdom Collapse << |
| NCT00766415 | - | - | Completed | - | - |
| NCT01196689 | Asthma | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT00758589 | Asthma | Phase 2 | Completed | - | Argentina
... more >> Research Site
Quilmes, Buenos Aires, Argentina
Research Site
San Miguel de Tucuman, Tucuman, Argentina
Research Site
Buenos Aires, Argentina
Research Site
Santa Fe, Argentina
Brazil
Research Site
Porto Alegre, Brasil, Brazil
Research Site
Rio de Janeiro, RJ, Brazil
Research Site
Santo Andre, SP, Brazil
Research Site
Sao Paulo, SP, Brazil
Costa Rica
Research Site
Barrio Los Yoses, San Jose, Costa Rica
Research Site
San Francisco de Dos Rios, San Jose, Costa Rica
Poland
Research Site
Bialystok, Poland
Research Site
Bydgoszcz, Poland
Research Site
Gdalsk, Poland
Research Site
Kielce, Poland
Research Site
Krakow, Poland
Research Site
Lublin, Poland
Research Site
Poznal, Poland
Research Site
Szczecin, Poland
Research Site
Tarnow, Poland
Research Site
Turek, Poland
Research Site
Wroclaw, Poland Collapse << |
| NCT00698282 | Healthy | Phase 1 | Completed | - | United Kingdom
... more >> Research Site
Berkshire, United Kingdom Collapse << |
| NCT00758589 | - | - | Completed | - | - |
| NCT01254461 | Drug Interaction | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT01058447 | Healthy | Phase 1 | Completed | - | - |
| NCT00918398 | Healthy
Postm... more >>enopausal
Surgically Sterile Women Collapse << | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT00690482 | - | - | Completed | - | - |
| NCT01197794 | Asthma Patients | Phase 2 | Completed | - | - |
| NCT02367066 | - | - | Completed | - | - |
| NCT01265641 | Asthma | Phase 1 | Completed | - | United Kingdom
... more >> Researche Site
London, UK, United Kingdom Collapse << |
| NCT01110525 | Oral Contraceptives | Phase 1 | Completed | - | Sweden
... more >> Research Site
Lulea, Sweden
Research Site
Uppsala, Sweden Collapse << |
| NCT01199341 | Pharmakokinetic | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT00859352 | Healthy Male Volunteer | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT00690482 | Moderate to Severe COPD | Phase 2 | Completed | - | Bulgaria
... more >> Research Site
Pleven, Bulgaria
Research Site
Russe, Bulgaria
Research Site
Sofia, Bulgaria
Research Site
Varna, Bulgaria
Denmark
Research Site
Arhus C, Denmark
Research Site
Hellerup, Denmark
Research Site
Hvidovre, Denmark
Research Site
Vaerlose, Denmark
Poland
Research Site
Krakow, Poland
Research Site
Lublin, Poland
Research Site
Ostrow Wielkopolski, Poland
Research Site
Przemysl, Poland
Research Site
Rzeszow, Poland
Slovakia
Research Site
Bojnice, Slovakia
Research Site
Liptovsky Hradok, Slovakia
Research Site
Poprad, Slovakia
Research Site
Spisska Nova Ves, Slovakia
Research Site
Zilina, Slovakia
Sweden
Research Site
Lund, Sweden
Research Site
Stockholm, Sweden
Research Site
Uppsala, Sweden Collapse << |
| NCT01311635 | Healthy | Phase 1 | Completed | - | Sweden
... more >> Research Site
Uppsala, Sweden Collapse << |
| NCT02031679 | Chronic Idiopathic Urticaria | Phase 2 | Completed | - | United States, Maryland
... more >>
Johns Hopkins Asthma and Allergy Center
Baltimore, Maryland, United States, 21224-6821 Collapse << |
| NCT02031679 | - | - | Completed | - | - |
| NCT01197794 | - | - | Completed | - | - |
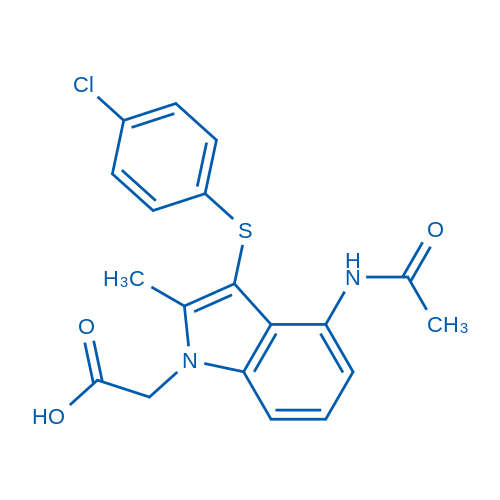

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
