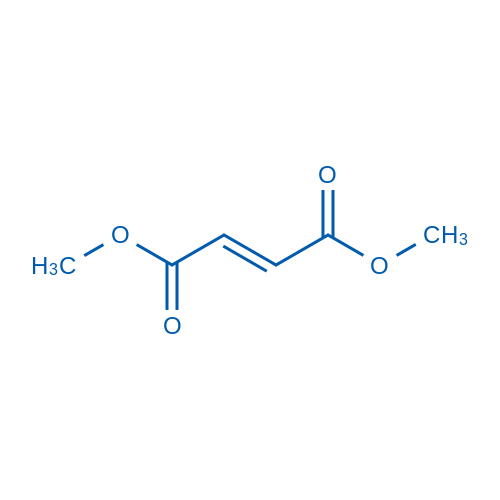
CAS No.: 624-49-7
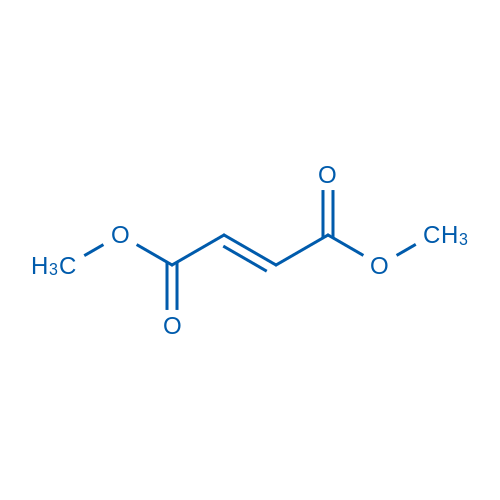
富马酸二甲酯 Catalog No. CSN13701
Synonyms: 富马酸二甲酯
Dimethyl fumarate is a nuclear factor (erythroid-derived)-like 2 (Nrf2) pathway activator which induces upregulation of antioxidant gene expression.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02559765 - - Completed - United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << NCT02634307 Multiple Sclerosis Phase 3 Enrolling by invitation December 2020 - NCT02749396 - - Completed - Finland ... more >> Many locations Multiple Locations, Finland Collapse << - 更多
- 参考文献
- [1] Peng H, Guerau-de-Arellano M, et al. Dimethyl fumarate inhibits dendritic cell maturation via nuclear factor κB (NF-κB) and extracellular signal-regulated kinase 1 and 2 (ERK1/2) and mitogen stress-activated kinase 1 (MSK1) signaling. J Biol Chem. 2012 Aug 10;287(33):28017-26.
- [2] Albrecht P, Bouchachia I, et al. Effects of dimethyl fumarate on neuroprotection and immunomodulation. J Neuroinflammation. 2012 Jul 7;9:163.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 624-49-7 | 储存条件 |
|
|||||||
| 分子式 | C6H8O4 | 运输 | 蓝冰 | |||||||
| 分子量 | 144.13 | 别名 | 富马酸二甲酯 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02559765 | - | - | Completed | - | United Kingdom ... more >> The University of Glasgow Glasgow, United Kingdom, G12 8LL Collapse << |
| NCT02634307 | Multiple Sclerosis | Phase 3 | Enrolling by invitation | December 2020 | - |
| NCT02749396 | - | - | Completed | - | Finland ... more >> Many locations Multiple Locations, Finland Collapse << |
| NCT03723356 | - | - | Recruiting | June 16, 2020 | United States, New York ... more >> New York University School of Medicine Recruiting New York, New York, United States, 10016 Contact: Kathleen Sherman, MD 929-455-5125 kai.sherman@nyumc.org Principal Investigator: Leigh Charvet, MD Collapse << |
| NCT02201849 | Multiple Sclerosis | Phase 1 | Completed | - | United States, Texas ... more >> Alkermes Investigational Site Austin, Texas, United States Collapse << |
| NCT03535298 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Not yet recruiting | September 2023 | United States, Ohio ... more >> Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Tammy Skaramagas, BA 216-445-6724 skaramt1@ccf.org Principal Investigator: Daniel Ontaneda, MD, MSc United Kingdom University of Nottingham Not yet recruiting Nottingham, United Kingdom, NG7 2UH Contact: Sara Wilkins +44 115 9249924 ext 66816 Sara.wilkins@nuh.nhs.uk Principal Investigator: Nikos Evangelou, MD, DPhil Collapse << |
| NCT02241785 | Relapsing Multiple Sclerosis | Phase 4 | Terminated(Business Decision) | - | United States, California ... more >> Research Site Fullerton, California, United States, 92835 United States, Colorado Research Site Aurora, Colorado, United States, 80045 United States, Iowa Research Site Des Moines, Iowa, United States, 50314 United States, Missouri Research Site Saint Louis, Missouri, United States, 63110 United States, New York Research Site Plainview, New York, United States, 11803 United States, North Carolina Research Site Raleigh, North Carolina, United States, 27607-6010 United States, Ohio Research Site Akron, Ohio, United States, 44320 Research Site Cleveland, Ohio, United States, 44195 United States, Tennessee Research Site Knoxville, Tennessee, United States, 37922 United States, Texas Research Site Round Rock, Texas, United States, 78681 United States, Washington Research Site Tacoma, Washington, United States, 98405 Collapse << |
| NCT03193866 | - | - | Recruiting | December 2021 | Sweden ... more >> Fredrik Piehl Recruiting Stockholm, Sweden Contact: Fredrik Piehl, Professor +46 73-671 81 01 Fredrik.Piehl@ki.se Contact: Anna Fogdell-Hahn, PhD +46 70-544 90 60 anna.fogdell-hahn@ki.se Collapse << |
| NCT01726933 | Plaque Psoriasis | Phase 3 | Completed | - | Austria ... more >> Investigatives site names will not be disclosed to avoid competitive information. Specify at least one site as follows: Almirall Investigative Site Various, Austria Germany Investigatives site names will not be disclosed to avoid competitive information. Specify at least one site as follows: Almirall Investigative Site Various, Germany Netherlands Investigatives site names will not be disclosed to avoid competitive information. Specify at least one site as follows: Almirall Investigative Site Various, Netherlands Poland Investigatives site names will not be disclosed to avoid competitive information. Specify at least one site as follows:Almirall Investigative Site # Various, Poland Collapse << |
| NCT03500328 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Not Applicable | Recruiting | October 31, 2022 | - |
| NCT03029897 | Multiple Sclerosis | Not Applicable | Recruiting | October 2018 | France ... more >> CHU Caen Recruiting Caen, France, 14000 Contact: Laure PEYRO SAINT PAUL Collapse << |
| NCT00811005 | Pustular Palmoplantar Psoriasi... more >>s Collapse << | Phase 3 | Unknown | October 2010 | Austria ... more >> Medical University of Vienna; University Clinic of Dermatology; Division of Special and Environmental Dermatology Vienna, Austria, 1180 Collapse << |
| NCT03302442 | - | - | Completed | - | France ... more >> Nantes University Hospital Nantes, France, 44093 Collapse << |
| NCT02634801 | Plaque Psoriasis | Phase 3 | Completed | - | Germany ... more >> For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Augsburg, Germany, 86179 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Berlin, Germany, 10783 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bochum, Germany, 44803 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Bonn, Germany, 53105 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Buxtehude, Germany, 21614 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Dresden, Germany, 01307 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Erlangen, Germany, 91054 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Essen, Germany, 45122 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Frankfurt, Germany, 60590 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Friedrichshafen, Germany, 88045 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Gelsenkirchen, Germany, 45883 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hamburg, Germany, 20354 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Hanau, Germany, 63450 For additional information regarding investigative sites for this trial, contact 1-888-545-5972 Mon - Fri, 9 AM to 4 PM or 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri, 9 AM to 5 PM Eastern Time or speak with your personal physician. Kiel, Germany, 24105 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Lübeck, Germany, 23538 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Mannheim, Germany, 68167 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Münster, Germany, 48149 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Selters, Germany, 56242 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Stuttgart, Germany, 70178 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Tübingen, Germany, 72076 For additional information regarding investigative sites for this trial, contact 1-877-CTLILLY (1-877-285-4559, 1-317-615-4559) Mon - Fri from 9 AM to 5 PM Eastern Time (UTC/GMT - 5 hours, EST), or speak with your personal physician. Ulm, Germany, 89081 Collapse << |
| NCT02241785 | - | - | Terminated(Business Decision) | - | - |
| NCT03308994 | - | - | Recruiting | December 31, 2018 | France ... more >> Groupement des Hôpitaux de l'Institut Catholique de Lille (HICL) Recruiting Lille, Hauts-de-France, France, 59020 Contact: Arnaud Kwiatkowski, MD-PhD 33320874901 kwiatkowski.arnaud@ghicl.net University Hospital of Lille Recruiting Lille, Hauts-de-France, France, 59037 Contact: Patrick Vermersch, Professor 33320445765 patrick.vermersch@univ-lille2.fr University Hospital Center of Caen Recruiting Caen, Normandie, France, 14033 Contact: Gilles-Louis Defer, Professor 33231064617 defer-gi@chu-caen.fr Contact: Damien Chevanne, PhD 33231064617 chevanne-d@chu-caen.fr University Hospital of Rouen Recruiting Rouen, Normandie, France, 76031 Contact: Bertrand Bourre, MD 33232888990 Bertrand.Bourre@chu-rouen.fr Collapse << |
| NCT01088165 | Psoriasis Car... more >>diovascular Diseases Diabetes Mellitus, Type 2 Collapse << | Phase 4 | Unknown | May 2014 | Austria ... more >> Medical University Vienna Dpt. of Dermatology Recruiting Vienna, Austria, 1090 Contact: Gregor Holzer, Dr 40400 7702 gregor.holzer@meduniwien.ac.at Contact: Adrian Tanew, ao Univ. Prof. 40400 7701 adrian.tanew@meduniwien.ac.at Principal Investigator: Gregor Holzer, Dr Collapse << |
| NCT03399981 | - | - | Active, not recruiting | December 31, 2023 | United States, Massachusetts ... more >> Research Site Cambridge, Massachusetts, United States, 02142 Collapse << |
| NCT01632449 | Healthy Volunteers | Phase 1 | Completed | - | United States, Minnesota ... more >> Research Site Minneapolis, Minnesota, United States Collapse << |
| NCT01420458 | Healthy Volunteers | Phase 1 | Completed | - | United States, Minnesota ... more >> Research Site Minneapolis, Minnesota, United States Collapse << |
| NCT02634801 | - | - | Completed | - | - |
| NCT03331835 | Psoriasis Vulgaris | Phase 4 | Recruiting | October 2018 | Germany ... more >> Fachklinik Bad Bentheim Klinik für Dermatologie Recruiting Bad Bentheim, Germany, 48455 Charité - Universitätsmedizin Berlin Klinik für Dermatologie, Venerologie und Allergologie Psoriasis Studien Zentrum Recruiting Berlin, Germany, 10117 Rothhaar Studien GmbH Dermatologisches Studienzentrum Recruiting Berlin, Germany, 10783 Hautarztpraxis Dr. Wildfeuer Recruiting Berlin, Germany, 13055 Klinikum Bielefeld Klinik für Dermatologie und Allergologie Recruiting Bielefeld, Germany, 33647 Hauttumorzentrum Ruhr- Universität im St. Josef Hospital Recruiting Bochum, Germany, 44791 Hautarztpraxis Dr. Niesmann und Dr. Othlinghaus Recruiting Bochum, Germany, 44793 Universitätsklinikum Bonn (AöR) Klinik und Poliklinik für Dermatologie und Allergologie Recruiting Bonn, Germany, 53127 Elbe Klinikum Buxtehude Klinik für Dermatologie Recruiting Buxtehude, Germany, 21614 Rosenpark Research Recruiting Darmstadt, Germany, 64283 Universitätsklinikum Carl Gustav Carus Klinik und Poliklinik für Dermatologie Recruiting Dresden, Germany, 01307 Universitätsklinikum Erlangen Hautklinik Recruiting Erlangen, Germany, 91054 Universitätsklinikum Frankfurt Klinik für Dermatologie Recruiting Frankfurt, Germany, 60590 Derma-Study-Center-Friedrichshafen Recruiting Friedrichshafen, Germany, 88045 Gemeinschaftspraxis Rotterdam & Kollegen Facharzt für Haut & Geschlechtskrankheiten Recruiting Gelsenkirchen, Germany, 45883 Universitätsklinikum Hamburg-Eppendorf, Institut für Versorgungsforschung in der Dermatologie und bei Pflegeberufen Recruiting Hamburg, Germany, 20246 SCIderm GmbH Recruiting Hamburg, Germany, 20354 Medizinische Hochschule Hannover Klinik für Dermatologie Allergologie und Venerologie Recruiting Hannover, Germany, 30625 Universitäts-Hautklinik Heidelberg Recruiting Heidelberg, Germany, 69120 Klinik für Dermatologie, Venerologie und Allergologie Universitätsklinikum Schleswig-Holstein, Campus Kiel Psoriasis-Zentrum Recruiting Kiel, Germany, 24105 Exellenzzentrum Entzündungsmedizin (CCIM) Universitätsklinikum Schleswig-Holstein, Campus Lübeck Recruiting Lübeck, Germany, 23538 University Medical Center Mainz Department of Dermatology and Allergy, Clinical Research Center Recruiting Mainz, Germany, 55131 Universitätsklinikum Mannheim der Universität Heidelberg Klinik für Dermatologie, Venerologie und Allergologie Recruiting Mannheim, Germany, 68167 Technische Universität München Klinik und Poliklinik für Dermatologie und Allergologie Recruiting München, Germany, 80802 Klinische Forschung Osnabrück - Klifos Recruiting Osnabrück, Germany, 49074 KLINIKUM VEST GmbH Knappschaftskrankenhaus Recklinghausen Klinik für Dermatologie und Allergologie Recruiting Recklinghausen, Germany, 45657 Gemeinschaftspraxis Weber & Crainic Recruiting Schweinfurt, Germany, 97421 Hautarztpraxis Dres. Leitz Recruiting Stuttgart, Germany, 70178 University Medical Center University of Tübingen Recruiting Tübingen, Germany, 72076 Hautarztpraxis Recruiting Witten, Germany, 58453 Collapse << |
| NCT02683863 | Multiple Sclerosis | Phase 4 | Completed | - | United States, New York ... more >> Multiple Sclerosis Center of Northeastern New York Latham, New York, United States, 12110 Collapse << |
| NCT02475304 | Psoriatic Arthritis | Phase 2 | Withdrawn(Difficulties to enro... more >>l patients) Collapse << | June 2017 | Sweden ... more >> Department of Rheumatology, Skåne University Hospital Malmö, Sweden, 20502 Collapse << |
| NCT00273364 | Multiple Sclerosis | Phase 2 | Active, not recruiting | December 2018 | United States, Illinois ... more >> Northwestern University, Feinberg School of Medicine Chicago, Illinois, United States, 60611 Collapse << |
| NCT02951533 | Psoriasis | Phase 3 | Active, not recruiting | February 14, 2019 | Germany ... more >> Augsburg, Germany Berlin, Germany Bielefeld, Germany Dresden, Germany Duelmen, Germany Duesseldorf, Germany Erlangen, Germany Essen, Germany Frankfurt, Germany Gera, Germany Hamburg, Germany Heidelberg, Germany Jena, Germany Kiel, Germany Leipzig, Germany Luebeck, Germany Mahlow, Germany Mainz, Germany Memmingen, Germany Muenchen, Germany Muenster, Germany Neu-Ulm, Germany Osnabrueck, Germany Selters, Germany Stuttgart, Germany Tuebingen, Germany Witten, Germany Wuppertal, Germany Collapse << |
| NCT02975349 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 2 | Active, not recruiting | August 12, 2020 | - |
| NCT02776072 | - | - | Completed | - | - |
| NCT02474082 | Psoriasis | Phase 3 | Completed | - | Germany ... more >> Novartis Investigative Site Bad Bentheim, Germany, 48455 Novartis Investigative Site Berlin, Germany, 10117 Novartis Investigative Site Berlin, Germany, 10247 Novartis Investigative Site Berlin, Germany, 10789 Novartis Investigative Site Berlin, Germany, 13187 Novartis Investigative Site Berlin, Germany, 13578 Novartis Investigative Site Bielefeld, Germany, 33647 Novartis Investigative Site Bochum, Germany, 44791 Novartis Investigative Site Bochum, Germany, 44803 Novartis Investigative Site Bonn, Germany, 53105 Novartis Investigative Site Darmstadt, Germany, 64283 Novartis Investigative Site Erlangen, Germany, 91054 Novartis Investigative Site Essen, Germany, 45147 Novartis Investigative Site Frankfurt, Germany, 60590 Novartis Investigative Site Gera, Germany, 07548 Novartis Investigative Site Halle, Germany, 06108 Novartis Investigative Site Hamburg, Germany, 20246 Novartis Investigative Site Hamburg, Germany, 20354 Novartis Investigative Site Hamburg, Germany, 22391 Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site Heidelberg, Germany, 69120 Novartis Investigative Site Kiel, Germany, 24105 Novartis Investigative Site Lubeck, Germany, 23538 Novartis Investigative Site Ludwigshafen, Germany, 67063 Novartis Investigative Site Mannheim, Germany, 68167 Novartis Investigative Site Muenchen, Germany, 81675 Novartis Investigative Site Muenster, Germany, 48149 Novartis Investigative Site Osnabruck, Germany, 49074 Novartis Investigative Site Quedlinburg, Germany, 06484 Novartis Investigative Site Schwerin, Germany, 19055 Novartis Investigative Site Selters, Germany, 56242 Novartis Investigative Site Stade, Germany, 21682 Collapse << |
| NCT01911767 | - | - | Recruiting | May 31, 2028 | United States, Massachusetts ... more >> Research Site Recruiting Cambridge, Massachusetts, United States, 02139-1955 Australia, Victoria Research Site Recruiting Box Hill, Victoria, Australia, 3128 Germany Research Site Recruiting Bochum, Nordrhein Wesfalen, Germany, 44791 Ireland Research Site Recruiting Dublin 4, Dublin, Ireland, DU Italy Research Site Recruiting Firenze, Italy, 50134 Research Site Recruiting Genova, Italy, 16132 Research Site Recruiting Milano, Italy, 20132 Research Site Recruiting Palermo, Italy, 90146 Research Site Recruiting Roma, Italy, 00152 Spain Research Site Recruiting Malaga, Spain, 29010 United Kingdom Research Site Recruiting Salford, Greater Manchester, United Kingdom, M6 8HD Collapse << |
| NCT02217982 | - | - | Terminated(Study did not meet ... more >>required enrollment numbers) Collapse << | - | - |
| NCT02474082 | - | - | Completed | - | - |
| NCT00168701 | Multiple Sclerosis | Phase 2 | Completed | - | - |
| NCT02907177 | Multiple Sclerosis | Phase 3 | Recruiting | March 31, 2020 | - |
| NCT01281111 | Healthy | Phase 1 | Completed | - | United States, Minnesota ... more >> Research Site St. Paul, Minnesota, United States, 55114 Collapse << |
| NCT02099370 | - | - | Completed | - | United States, Florida ... more >> USF Carol and Frank Morsani Center for Advanced Healthcare Tampa, Florida, United States, 33612 Collapse << |
| NCT02097849 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Research Site Gilbert, Arizona, United States, 85234 United States, Colorado Research Site Thornton, Colorado, United States, 80233 United States, Florida Research Site Fort Lauderdale, Florida, United States, 33312 Research Site Sarasota, Florida, United States, 34243 United States, Indiana Research Site Indianapolis, Indiana, United States, 46202 United States, Kentucky Research Site Lexington, Kentucky, United States, 40513 United States, Maine Research Site Auburn, Maine, United States, 04210 United States, New York Research Site New York, New York, United States, 10016 United States, North Carolina Research Site Charlotte, North Carolina, United States, 28203 United States, Ohio Research Site Akron, Ohio, United States, 44320 Research Site Cleveland, Ohio, United States, 44195 Research Site Dayton, Ohio, United States, 45417 United States, Texas Research Site Round Rock, Texas, United States, 78761 Research Site San Antonio, Texas, United States, 78258 Collapse << |
| NCT02117050 | Multiple Sclerosis | Phase 4 | Terminated(recruitment challen... more >>ges) Collapse << | - | United States, Massachusetts ... more >> Call EMD Serono Medical Information for information on recruiting sites Boston, Massachusetts, United States Collapse << |
| NCT02097849 | - | - | Completed | - | - |
| NCT02218879 | - | - | Terminated | - | United States, Connecticut ... more >> Yale University New Haven, Connecticut, United States, 06519 Collapse << |
| NCT00810836 | Rheumatoid Arthritis | Phase 2 | Completed | - | Australia, Queensland ... more >> Research Site Woolloongabba, Queensland, Australia, 4102 Australia, South Australia Research Site Woodville, South Australia, Australia, 5011 Canada, Ontario Research Site Sarnia, Ontario, Canada, N7T4X3 Canada Research Site New Market, Canada Research Site Ottawa, Canada Research Site Rothesay, Canada Czech Republic Research Site Active, not recruiting, Czech Republic Research Site Pardubice, Czech Republic Research Site Uh. Hradiste, Czech Republic Research Site Zlin, Czech Republic India Research Site Bangalore, India Research Site Hyderabaad, India Research Site Hyderabad, India Research Site Lucknow, India Poland Research Site Bialystock, Poland Research Site Grodzisk Mazowiecki, Poland Research Site Lublin, Poland Research Site Poznan, Poland Research Site Torun, Poland Research Site Warsaw, Poland Research Site Warszawa, Poland Slovakia Research Site Banska Bysterica, Slovakia Research Site Bratislava, Slovakia Research Site Kosice, Slovakia Research Site Piestany, Slovakia Collapse << |
| NCT01352988 | Lupus Erythematosus, Cutaneous | Phase 2 | Completed | - | Germany ... more >> Department of Dermatology, University Hospital Muenster Muenster, Germany, 48149 Collapse << |
| NCT03255382 | Psoriasis | Phase 3 | Completed | - | Germany ... more >> Universitaetsklinik Heidelberg /ID# 161014 Heidelberg, Baden-Wuerttemberg, Germany, 69120 Universitaetsklinikum Erlangen /ID# 161035 Erlangen, Bayern, Germany, 91054 Universitatsklinikum Frankfurt /ID# 161036 Frankfurt, Hessen, Germany, 60590 Universitatsklinikum Munster /ID# 165739 Munster, Niedersachsen, Germany, 48149 Johannes Wesling Klin Minden /ID# 161015 Minden, Nordrhein-Westfalen, Germany, 32429 CMS3 Company for Medical Study /ID# 161103 Selters (Westerwald), Rheinland-Pfalz, Germany, 56242 Univ Hosp Schleswig-Holstein /ID# 160995 Kiel, Schleswig-Holstein, Germany, 24105 Medizinisches Versorgungszentrum DermaKiel GmbH /ID# 161102 Kiel, Schleswig-Holstein, Germany, 24148 Charité Universitätsmedizin Campus Mitte /ID# 165621 Berlin, Germany, 10117 ISA GmbH /ID# 165619 Berlin, Germany, 10789 Gemeinschaftspraxis /ID# 161037 Blankenfeld-mahlow, Germany, 15831 Hautzentrum Niesmann Othlingha /ID# 161034 Bochum, Germany, 44803 Universitaetsklinikum Bonn /ID# 165618 Bonn, Germany, 53113 Hautklinik Klinikum Darmstadt /ID# 164940 Darmstadt, Germany, 64297 Universitaetklinikum Dresden /ID# 160983 Dresden, Germany, 01307 Univ Klinik Eppendorf Hamburg /ID# 161038 Hamburg, Germany, 20246 SCIderm GmbH /ID# 160994 Hamburg, Germany, D-20354 Klinik fur Dermatologie /ID# 161101 Leipzig, Germany, 4103 Univ Johannes Gutenberg /ID# 161104 Mainz, Germany, 55131 TU Uniklinik Munchen /ID# 160996 Munich, Germany, 80802 Universitatsklinikum Tubingen /ID# 165620 Tuebingen, Germany, 72076 Hoffmann, Witten, DE /ID# 165622 Witten, Germany, 58453 Centroderm Wuppertal /ID# 165615 Wuppertal, Germany, 42287 Collapse << |
| NCT01945359 | - | - | Completed | - | United States, Utah ... more >> Rocky Mountain MS Research Group Salt Lake City, Utah, United States, 84103 Collapse << |
| NCT01453426 | Healthy | Phase 1 | Completed | - | Australia, Victoria ... more >> Research Site Melbourne, Victoria, Australia China, Hong Kong Research Site Hong Kong, Hong Kong, China Collapse << |
| NCT03284970 | - | - | Completed | - | United States, Massachusetts ... more >> Neurology Center of New England Foxboro, Massachusetts, United States, 02035 Collapse << |
| NCT02117050 | - | - | Terminated(recruitment challen... more >>ges) Collapse << | - | - |
| NCT02823951 | - | - | Completed | - | - |
| NCT00837785 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 1 | Completed | - | Germany ... more >> Research Site Berlin, Germany Collapse << |
| NCT02410278 | - | - | Completed | - | - |
| NCT02217982 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Terminated(Study did not meet ... more >>required enrollment numbers) Collapse << | - | United States, Utah ... more >> Rocky Mountain MS Research Group Salt Lake City, Utah, United States, 84103 Collapse << |
| NCT00451451 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02410278 | Multiple Sclerosis | Phase 4 | Completed | - | - |
| NCT00451451 | - | - | Completed | - | - |
| NCT01568112 | Healthy | Phase 3 | Completed | - | United States, Minnesota ... more >> Research Site St Paul, Minnesota, United States Collapse << |
| NCT02343159 | - | - | Terminated(Sponsor Decision) | - | - |
| NCT02090413 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Ireland ... more >> Research Site Cork, Ireland Research Site Dublin, Ireland, Dublin 4 Research Site Dublin, Ireland, Dublin 7 Research Site Dublin, Ireland, Dublin 9 United Kingdom Research Site Basingstoke, United Kingdom, RG24 9NA Research Site Birmingham, United Kingdom, B15 2TH Research Site Cardiff, United Kingdom, CF14 4XW Research Site Edinburgh, United Kingdom, EHA 2XU Research Site Exeter, United Kingdom, EX2 5DW Research Site Glasgow, United Kingdom, G51 4TH Research Site Leicester, United Kingdom, LE5 4PW Research Site Liverpool, United Kingdom, L9 7LJ Research Site London, United Kingdom, SE5 9RS Research Site London, United Kingdom, SW17 0QT Research Site London, United Kingdom, WC1N 3BG Research Site Newcastle upon Tyne, United Kingdom, NE1 4LP Research Site Norwich, United Kingdom, NR4 7UY Research Site Nottingham, United Kingdom, NG7 2UH Research Site Plymouth, United Kingdom, PL6 8BX Research Site Salford, United Kingdom, M6 8HD Research Site Swansea, United Kingdom, SA6 6NL Collapse << |
| NCT02343159 | Multiple Sclerosis | Phase 4 | Terminated(Sponsor Decision) | - | - |
| NCT03093324 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Recruiting | June 2019 | - |
| NCT02419638 | - | - | Withdrawn(Collaborative decisi... more >>on between study PI and sponsor.) Collapse << | - | United States, Massachusetts ... more >> Partners MS Center, 1 Brookline Place Suite 225 Brookline, Massachusetts, United States, 02445 Collapse << |
| NCT00420212 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02090413 | - | - | Completed | - | - |
| NCT01568112 | - | - | Completed | - | - |
| NCT02955693 | Psoriasis | Phase 1 | Completed | - | United Kingdom ... more >> Almirall Investigational Site #1 London, Harrow, United Kingdom, HA1 3UJ Collapse << |
| NCT00420212 | - | - | Completed | - | - |
| NCT02283853 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Recruiting | May 30, 2025 | - |
| NCT02428231 | - | - | Terminated(Sponsor decision) | - | - |
| NCT02746744 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 3 | Recruiting | August 2021 | Sweden ... more >> Danderyd hospital Recruiting Stockholm, Sweden Contact: Anders Svenningsson, MD, PhD Umeå University Recruiting Umeå, Sweden Contact: Jonatan Saltzer, MD, PhD Collapse << |
| NCT01903291 | - | - | Completed | - | - |
| NCT02428231 | Multiple Sclerosis | Phase 3 | Terminated(Sponsor decision) | - | United States, Arizona ... more >> Research Site Gilbert, Arizona, United States Research Site Phoenix, Arizona, United States United States, California Research Site Long Beach, California, United States United States, Florida Research Site Miami, Florida, United States United States, Indiana Research Site Avon, Indiana, United States Research Site Franklin, Indiana, United States United States, Kansas Research Site Overland Park, Kansas, United States United States, Maine Research Site Lewiston, Maine, United States United States, North Carolina Research Site Asheville, North Carolina, United States Research Site Cary, North Carolina, United States Research Site Charlotte, North Carolina, United States Research Site Wilmington, North Carolina, United States United States, Ohio Research Site Cincinnati, Ohio, United States United States, Texas Research Site Dallas, Texas, United States United States, Washington Research Site Wenatchee, Washington, United States Belgium Research Site Leuven, Belgium Research Site Wilrijk, Belgium Czech Republic Research Site Prague, Czech Republic Italy Research Site Merano, Bolzano, Italy Research Site Montichiari, Italy Collapse << |
| NCT01915901 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Daytona Beach, Florida, United States, 32117 United States, Texas Research Site Dallas, Texas, United States, 75247 United States, Wisconsin Research Site Madison, Wisconsin, United States, 53704 Collapse << |
| NCT02428218 | Relapsing-Remitting Multiple S... more >>clerosis Relapsing Forms of Multiple Sclerosis Collapse << | Phase 3 | Withdrawn(Feasibility) | January 2027 | - |
| NCT00835770 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Active, not recruiting | September 28, 2020 | - |
| NCT02171208 | Healthy | Phase 1 | Completed | - | United States, Florida ... more >> Research Site Daytona Beach, Florida, United States, 32117 United States, Indiana Research Site Evansville, Indiana, United States, 47710 Collapse << |
| NCT02546440 | Cutaneous T Cell Lymphoma | Phase 2 | Unknown | September 2017 | Germany ... more >> University Medical Center Recruiting Mannheim, Germany, 68167 Contact: Jan P Nicolay, MD 00496213832280 jan.nicolay@umm.de Contact: Jochen Utikal, MD 00496213832280 jochen.utikal@umm.de Collapse << |
| NCT02337426 | Adult Brain Glioblastoma ... more >> Adult Giant Cell Glioblastoma Adult Gliosarcoma Collapse << | Phase 1 | Completed | - | United States, Virginia ... more >> Virginia Commonwealth University/Massey Cancer Center Richmond, Virginia, United States, 23298 Collapse << |
| NCT01930708 | Relapsing-Remitting Multiple S... more >>clerosis Multiple Sclerosis Collapse << | Phase 4 | Active, not recruiting | April 30, 2019 | - |
| NCT02471560 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Completed | - | Norway ... more >> Research site Drammen, Norway, 3019 Research site Haukeland, Norway, 5021 Research Site Lillehammer, Norway, 2609 Research site Lørenskog, Norway, 1478 Research site Molde, Norway, 6412 Research site Oslo, Norway, 0450 Research site Stavanger, Norway, 4011 Collapse << |
| NCT01069913 | Healthy | Phase 1 | Completed | - | - |
| NCT02555215 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 3 | Completed | - | United States, California ... more >> Research Site Loma Linda, California, United States, 92354 Belgium Research Site Ghent, Belgium, 9000 Bulgaria Research Site Sofia, Bulgaria, 1113 Czechia Research Site Hradec Kralove, Czechia, 50333 Germany Research Site Muenchen, Bayern, Germany, 80337 Research Site Göttingen, Niedersachsen, Germany, 37075 Kuwait Research Site Kuwait City, Kuwait, 15462 Latvia Research Site Riga, Latvia, LV-1004 Lebanon Research Site Beirut, Lebanon, 1107 2020 Poland Research Site Gdansk, Poland, 80-952 Research Site Poznan, Poland, 60-355 Turkey Research Site Ankara, Turkey, 06100 Collapse << |
| NCT02519413 | - | - | Completed | - | United States, Alabama ... more >> Research site Homewood, Alabama, United States, 35209 United States, California Research Site Newport Beach, California, United States, 92663 United States, Georgia Research Site Atlanta, Georgia, United States, 30309 United States, New York Research Site Buffalo, New York, United States, 14203 Research Site Patchogue, New York, United States, 11772 United States, North Carolina Research Site Huntersville, North Carolina, United States, 28078 United States, Pennsylvania Research Site Philadelphia, Pennsylvania, United States, 19125 United States, Washington Research Site Seattle, Washington, United States, 98101 Research Site Seattle, Washington, United States, 98104 United States, Wisconsin Research Site Milwaukee, Wisconsin, United States, 53215 Collapse << |
| NCT02159573 | - | - | Completed | - | - |
| NCT01873417 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 4 | Completed | - | - |
| NCT01873417 | - | - | Completed | - | - |
| NCT03101735 | - | - | Active, not recruiting | March 31, 2020 | Greece ... more >> University General Hospital AHEPA (B') Thessaloniki, Greece, 54636 Collapse << |
| NCT01838668 | Relapsing-Remitting Multiple S... more >>clerosis Multiple Sclerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02430532 | Multiple Sclerosis, Secondary ... more >>Progressive Collapse << | Phase 3 | Terminated(Sponsor Decision) | - | United States, California ... more >> Research Site Long Beach, California, United States, 90806 Research Site San Francisco, California, United States, 94143 United States, Florida Research Site Tampa, Florida, United States, 33634 Research Site Vero Beach, Florida, United States, 32960 United States, North Carolina Research Site Charlotte, North Carolina, United States, 28207 United States, Pennsylvania Research Site Willow Grove, Pennsylvania, United States, 19001 United States, Texas Research Site Round Rock, Texas, United States, 78681 Belgium Research Site Bruxelles, Belgium, 1200 Czech Republic Research Site Brno, Czech Republic, 656 91 Research Site Hradec Kralove, Czech Republic, 500 05 Netherlands Research Site Sittard-Geleen, Netherlands, 6162 BG Poland Research Site Gdansk, Poland, 80-803 Research Site Katowice, Poland Research Site Krakow, Poland, 31-505 Research Site Lodz, Poland, 93-121 Research Site Plewiska, Poland, 62-064 Research Site Poznan, Poland, 61-853 Slovakia Research Site Banska Bystrica, Slovakia, 97404 Collapse << |
| NCT02430532 | - | - | Terminated(Sponsor Decision) | - | - |
| NCT02739542 | Multiple Sclerosis (MS) | Phase 4 | Recruiting | March 2022 | United States, California ... more >> Keck School of Medicine - USC - Department of Neurology Recruiting Los Angeles, California, United States, 90089 Contact: Rama Kishan Koppula 323-442-6021 rk_198@usc.edu Principal Investigator: Lilyana Amezcua, MD United States, Colorado University of Colorado - Denver Recruiting Aurora, Colorado, United States, 80045 Contact: Brianna Blume 303-724-6386 BRIANNA.BLUME@UCDENVER.EDU Principal Investigator: Timothy Vollmer, MD United States, Florida University of South Florida Recruiting Tampa, Florida, United States, 33612 Contact: Angela Aungst 813-974-6378 aaungst@health.usf.edu Principal Investigator: Derrick Robertson, MD United States, Maryland Johns Hopkins University - Neurology Recruiting Baltimore, Maryland, United States, 21287 Contact: Madiha Qutab 410-614-9201 mqutab1@jhmi.edu Principal Investigator: Ellen Mowry, MD United States, Minnesota Mayo Clinic Department of Neurology Recruiting Rochester, Minnesota, United States, 55905 Contact: Sandra Looney 507-538-4107 looney.sandra@mayo.edu Principal Investigator: Orhun Kantarci, MD United States, Missouri Washington University Department of Neurology Recruiting Saint Louis, Missouri, United States, 63110 Contact: Courtney Dula 314-362-3402 dulac@wustl.edu Principal Investigator: Robert Naismith, MD United States, Nevada Cleveland Clinic - Lou Ruvo Center for Brain Health Recruiting Las Vegas, Nevada, United States, 89106 Contact: Miranda Staples 702-701-7669 STAPLEM@ccf.org Principal Investigator: Le Hua, MD United States, New York MS Clinical Care and Research Center, Dept of Neurology, Columbia University Recruiting New York, New York, United States, 10032 Contact: Eva Gelernt 212-342-4649 erg2144@cumc.columbia.edu Principal Investigator: Wendy Vargas, MD United States, Ohio Ohio Health Neurology Recruiting Columbus, Ohio, United States, 43214 Contact: Susan Foster 614-566-1268 Susan.Foster@ohiohealth.com Principal Investigator: Aaron Boster, MD United States, Oklahoma Oklahoma Medical Research Foundation, MS Center of Excellence Recruiting Oklahoma City, Oklahoma, United States, 73104 Contact: Micki Moore 405-271-6241 micki-moore@omrf.org Principal Investigator: Gabriel Pardo, MD United States, Texas MS Treatment Center of Dallas Recruiting Dallas, Texas, United States, 75246 Contact: Victoria Stokes 214-818-2539 victoria.stokes@bswhealth.org Principal Investigator: Annette Okai, MD UT Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390-8806 Contact: Cindy Daniel, CCRP 214-645-9165 cindy.daniel@utsouthwestern.edu Contact: Jose Santoyo, BS 214-645-0567 jose.santoyo@utsouthwestern.edu Principal Investigator: Darin T Okuda, MD Neurology Center of San Antonio Recruiting San Antonio, Texas, United States, 78258 Contact: Tina Clements 210-490-0016 ext 23 ncsastudycoordinator@hotmail.com Principal Investigator: Ann Bass, MD United States, Vermont University of Vermont Medical Center - MS Research Division - Department of Neurology Recruiting Burlington, Vermont, United States, 05401 Contact: Jane Low 802-847-0983 jane.low@uvmhealth.org Principal Investigator: Andrew Solomon, MD United States, Washington Swedish Medical Center Recruiting Seattle, Washington, United States, 98122 Contact: Caryl Tongco 206-215-3565 caryl.tongco@swedish.org Principal Investigator: Pavle Repovic, MD MultiCare Institute for Research and Innovation Recruiting Tacoma, Washington, United States, 98405 Contact: Jarrod Monroe 253-403-7449 jarrod.monroe@multicare.org Principal Investigator: Stacy Donlon, MD Collapse << |
| NCT01815723 | Plaque Psoriasis | Phase 3 | Withdrawn | June 2017 | Canada, Ontario ... more >> Probity Medical Research Waterloo, Ontario, Canada, N2J 1C4 Collapse << |
| NCT02410200 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 2 | Completed | - | United States, California ... more >> Research Site San Bernardino, California, United States, 92408 Belgium Research Site Gent, Belgium, B-9000 Bulgaria Research Site Sofia, Bulgaria, B-1113 Czechia Research Site Hradec kralove, Czechia, 500 05 Germany Research Site Munchen, Bayern, Germany, 80337 Research Site Gottingen, Niedersachsen, Germany, 37075 Kuwait Research Site Dasman, Kuwait City, Kuwait, 15462 Latvia Research Site Riga, Latvia, LV-1004 Lebanon Research Site Beirut, Lebanon, 1107 2020 Poland Research Site Gdansk, Poland, 80-952 Research Site Poznan, Poland, 60-355 Turkey Research Site Ankara, Turkey, 06100 Collapse << |
| NCT02410200 | - | - | Completed | - | - |
| NCT01156311 | - | - | Completed | - | - |
| NCT01924832 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Research Site Nottingham, United Kingdom, NJ116JS Collapse << |
| NCT01156311 | Relapsing-Remitting Multiple S... more >>clerosis Multiple Sclerosis Collapse << | Phase 2 | Completed | - | United States, Arizona ... more >> Research Site Gilbert, Arizona, United States Research Site Phoenix, Arizona, United States United States, Connecticut Research Site Danbury, Connecticut, United States United States, Georgia Research Site Atlanta, Georgia, United States United States, Indiana Research Site Fort Wayne, Indiana, United States United States, Maryland Research Site Baltimore, Maryland, United States United States, Massachusetts Research Site Boston, Massachusetts, United States United States, Minnesota Research Site Golden Valley, Minnesota, United States United States, New Jersey Research Site Teaneck, New Jersey, United States United States, New York Research Site Patchogue, New York, United States United States, Ohio Research Site Cleveland, Ohio, United States Research Site Dayton, Ohio, United States United States, Oregon Research Site Portland, Oregon, United States United States, Tennessee Research Site Cordova, Tennessee, United States Research Site Franklin, Tennessee, United States United States, Wisconsin Research Site Milwaukee, Wisconsin, United States Collapse << |
| NCT02525874 | Multiple Sclerosis, Relapsing-... more >>Remitting Multiple Sclerosis Collapse << | Phase 3 | Completed | - | - |
| NCT02644083 | - | - | Terminated | - | United States, Oregon ... more >> Oregon Health & Science University Portland, Oregon, United States, 97239 Collapse << |
| NCT02472938 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Withdrawn(Sponsor Decision) | July 2020 | - |
| NCT02736279 | - | - | Recruiting | December 2020 | United States, Washington ... more >> EvergreenHealth MS Center Recruiting Kirkland, Washington, United States, 98034 Contact: Shalom E Kilcup 425-899-5369 SEKilcup@evergreenhealth.com Principal Investigator: Virginia I Simnad, MD, M.Sc Sub-Investigator: Theodore R Brown, MD, MPH Collapse << |
| NCT02323269 | - | - | Terminated(109MS415 ImPROve st... more >>udy was terminated due to patient enrollment challenges and feasibility . The decision was not a result of safety concerns.) Collapse << | - | Canada, Alberta ... more >> Research Site Edmonton, Alberta, Canada, T6G 2G3 Canada, British Columbia Research Site Burnaby, British Columbia, Canada, V5G 2X6 Canada, New Brunswick Research Site St. John, New Brunswick, Canada, E2L 4L2 Canada, Newfoundland and Labrador Research Site St. John's, Newfoundland and Labrador, Canada, A1B 3V6 Canada, Nova Scotia Research Site Halifax, Nova Scotia, Canada, B3H 4K4 Research Site Sydney, Nova Scotia, Canada, B1P 1P3 Canada, Ontario Research Site Cambridge, Ontario, Canada, N1R 7L6 Research Site London, Ontario, Canada, N6A 5A5 Canada, Quebec Research Site Gatineau, Quebec, Canada, J9J 0A5 Research Site Montreal, Quebec, Canada, H3A 2B4 Collapse << |
| NCT03092544 | Multiple Sclerosis | Phase 4 | Active, not recruiting | December 2018 | United States, New York ... more >> Multiple Sclerosis Center of Northeastern New York Latham, New York, United States, 12110 Collapse << |
| NCT03345940 | Relapsing Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Recruiting | November 30, 2020 | Italy ... more >> Fondazione IRCCS Istituto Neurologico C. Besta, Neuroimmunology Unit Recruiting Milan, Italy, 20133 Contact: Silvia Rossi, MD, PhD +39022394 ext 2471 silvia.rossi@istituto-besta.it Contact: Renato Mantegazza, MD +39022394 ext 2321 renato.mantegazza@istituto-besta.it Principal Investigator: Silvia Rossi, MD, PhD Collapse << |
| NCT02686684 | - | - | Completed | - | - |
| NCT02090348 | Relapsing-Remitting Multiple S... more >>clerosis Multiple Sclerosis Collapse << | Phase 4 | Withdrawn(The study was withdr... more >>awn for business reasons. The decision to stop the TECNERGY study was not a result of any safety or efficacy concerns.) Collapse << | October 2017 | - |
| NCT02579681 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 3 | Completed | - | Italy ... more >> Research site Bari, Italy Research site Cagliari, Italy Research site Catania, Italy Research site Cefalu, Italy Research site Chieti, Italy Research site Ferrara, Italy Research site Fidenza, Italy Research site Firenze, Italy Research site Gallarate, Italy Research site Genova, Italy Research site L'aquila, Italy Research site Milano, Italy Research site Napoli, Italy Research site Orbassano, Italy Research site Padova, Italy Research site Palermo, Italy Research site Pavia, Italy Research site Roma, Italy Collapse << |
| NCT03526224 | - | - | Active, not recruiting | November 2019 | United States, New York ... more >> Buffalo Neuroimaging Analysis Center Buffalo, New York, United States, 14203 Collapse << |
| NCT02125604 | - | - | Completed | - | - |
| NCT02959658 | Primary Progressive Multiple S... more >>clerosis Collapse << | Phase 2 | Recruiting | December 2019 | Denmark ... more >> Danish Multiple Sclerosis Center, Department of neurology Recruiting Copenhagen, Denmark, 2100 Contact: Finn Sellebjerg, Professor finn.thorup.sellebjerg@regionh.dk Contact: Jacob Lando Talbot, MD jacob.lando.talbot@regionh.dk Collapse << |
| NCT02901106 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Recruiting | August 2023 | France ... more >> Fondation Ophtalmique Adolphe de Rothschild Recruiting Paris, France, 75019 Contact: Laurence Salomon, MD +33 1 48 03 64 31 lsalomon@fo-rothschild.fr Principal Investigator: Jennifer Aboab Sub-Investigator: Karen Lecouturier Collapse << |
| NCT02981082 | Systemic Sclerosis ... more >> Pulmonary; Hypertension Collapse << | Phase 1 | Recruiting | May 2020 | United States, California ... more >> Stanford University Not yet recruiting Stanford, California, United States, 94305 Contact: Dina Jean 650-725-9861 Dina.jean@stanford.edu Principal Investigator: Roham Zamanian, MD United States, Colorado National Jewish Not yet recruiting Denver, Colorado, United States, 80206 Contact: Kris Eliopoulos 303-270-2622 eliopoulosk@njhealth.org Principal Investigator: Patricia George, MD United States, Maryland John Hopkins Not yet recruiting Baltimore, Maryland, United States, 21205 Contact: Blessing Enobun 410-502-4362 benobun1@jhu.edu Principal Investigator: Paul Hassoun, MD United States, Massachusetts Boston University Recruiting Boston, Massachusetts, United States, 02118 Contact: Eric Stratton 617-358-6777 eas@bu.edu Contact: Kim Finch 617-358-6785 kimtobin@bu.edu Principal Investigator: Robert Simms, MD United States, Michigan University of Michigan Not yet recruiting Ann Arbor, Michigan, United States, 48109 Contact: Ann Green 866-963-3587 johnsann@med.umich.edu Contact: Cindy Alsamarraie 866-963-3587 sindi@med.umich.edu Principal Investigator: Vallerie McLaughlin, MD United States, Pennsylvania University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Dana Ivanco, BA, CCRC 412-648-7040 des2@pitt.edu Contact: Maureen Laffoon, BS 412-648-7871 laffoonm@pitt.edu Principal Investigator: Robert A Lafyatis, MD Collapse << |
| NCT02784834 | Chronic Lymphocytic Leukemia ... more >> Small Lymphocytic Lymphoma Collapse << | Phase 1 | Active, not recruiting | July 2020 | United States, California ... more >> UC San Diego Moores Cancer Center La Jolla, California, United States, 92093 Collapse << |
| NCT02125604 | Relapsing-Remitting Multiple S... more >>clerosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Research Site Augsburg, Germany Research Site Bamburg, Germany Research Site Bayreuth, Germany Research Site Berlin, Germany Research Site Bochum, Germany Research Site Bonn, Germany Research Site Erbach, Germany Research Site Erlangen, Germany Research Site Freiburg, Germany Research Site Hamburg, Germany Research Site Leipzig, Germany Research Site Marburg, Germany Research Site Minden, Germany Research Site Mittweida, Germany Research Site Munchen, Germany Research Site Munster, Germany Research Site Osnabruck, Germany Research Site Potsdam, Germany Research Site Siegen, Germany Collapse << |
| NCT02047097 | - | - | Recruiting | February 28, 2024 | - |
| NCT02438137 | Obstructive Sleep Apnea ... more >> OSA Sleep Apnea Collapse << | Phase 2 | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02461069 | Multiple Sclerosis, Relapsing-... more >>Remitting Collapse << | Phase 4 | Completed | - | Germany ... more >> Neurologisches Studienzentrum Dr. Schmidt/Dr. Neudecker/ Dr. Viehbahn/Dr. Kronenberger Bonn, Germany, 53111 Neurologische Gemeinschaftspraxis im Bienenkorbhaus Frankfurt am Main, Germany, 60313 Neurologische Univ.-Klinik Heidelberg, Germany, 69120 Klinik und Poliklinik für Neurologie, Universitätsklinikum Mainz Mainz, Germany, 55131 University Hospital Muenster, Department of Neurology Muenster, Germany, 48149 MVZ-Neurologie Klinikum Osnabrück GmbH Osnabrück, Germany, 49076 Collapse << |
| NCT02438137 | - | - | Completed | - | - |
| NCT02969304 | - | - | Completed | - | Germany ... more >> Research Site Leipzig, Saxony, Germany, 04109 Collapse << |
| NCT02675413 | Multiple Sclerosis ... more >> Multiple Sclerosis, Relapsing-Remitting Collapse << | Phase 4 | Withdrawn(Principal Investigat... more >>or decided to withdraw.) Collapse << | - | United States, Missouri ... more >> Washington University (John L. Trotter MS Center) St. Louis, Missouri, United States, 63110 United States, Washington Swedish Neuroscience Institute Seattle, Washington, United States, 98122 Collapse << |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
