| NCT00871910 | Solid Tumors
... more >>Lymphoma, Non-Hodgkin
Multiple Myeloma Collapse << | Phase 1 | Completed | - | - |
| NCT00871546 | Lymphoma, Mantle-Cell
... more >> Leukemia, Lymphocytic, Chronic, B-Cell Collapse << | Phase 2 | Terminated | - | - |
| NCT00798213 | Leukemia, Myeloid, Acute
... more >> Lymphoblastic Leukemia, Acute Collapse << | Phase 2 | Terminated | - | - |
| NCT01096342 | Refractory Multiple Myeloma | Phase 2 | Completed | - | United States, Arizona
... more >>
Mayo Clinic in Arizona
Scottsdale, Arizona, United States, 85259
United States, Florida
Mayo Clinic in Florida
Jacksonville, Florida, United States, 32224-9980
United States, Michigan
Wayne State University/Karmanos Cancer Institute
Detroit, Michigan, United States, 48201
United States, Minnesota
Mayo Clinic
Rochester, Minnesota, United States, 55905
United States, Wisconsin
University of Wisconsin Hospital and Clinics
Madison, Wisconsin, United States, 53792
Singapore
National University Hospital
Singapore, Singapore, 119074 Collapse << |
| NCT00732810 | Breast Neoplasms
... more >> Carcinoma, Non-Small-Cell Lung Collapse << | Phase 2 | Completed | - | - |
| NCT00871663 | Solid Tumors
... more >>Lymphoma, Non-Hodgkin
Multiple Myeloma
Leukemia, Lymphocytic, Chronic. B-Cell Collapse << | Phase 1 | Completed | - | - |
| NCT01096342 | - | - | Completed | - | - |
| NCT00937937 | Acral Lentiginous Melanoma
... more >> Lentigo Maligna Melanoma
Mucosal Melanoma
Nodular Melanoma
Recurrent Melanoma
Stage IV Cutaneous Melanoma AJCC v6 and v7
Superficial Spreading Melanoma Collapse << | Phase 2 | Active, not recruiting | - | - |
| NCT01026324 | Stage IIIB Melanoma
... more >> Stage IIIC Melanoma
Stage IV Melanoma Collapse << | Phase 1
Phase 2 | Terminated(Slow accrual) | - | United States, Massachusetts
... more >>
Dana-Farber Cancer Institute
Boston, Massachusetts, United States, 02115 Collapse << |
| NCT00937937 | - | - | Active, not recruiting | - | - |
| NCT01026324 | - | - | Terminated(Slow accrual) | - | - |
| NCT01580228 | Chronic Lymphocytic Leukemia (... more >>CLL) Collapse << | Phase 3 | Completed | - | - |
| NCT01515176 | - | - | Completed | - | - |
| NCT01515176 | Chronic Lymphocytic Leukemia
... more >> Prolymphocytic Leukemia
Recurrent Small Lymphocytic Lymphoma
Refractory Chronic Lymphocytic Leukemia Collapse << | Phase 1
Phase 2 | Completed | - | United States, Ohio
... more >>
Ohio State University Comprehensive Cancer Center
Columbus, Ohio, United States, 43210 Collapse << |
| NCT01434316 | Advanced Malignant Solid Neopl... more >>asm
BRCA1 Gene Mutation
BRCA2 Gene Mutation Collapse << | Phase 1 | Recruiting | - | United States, Massachusetts
... more >>
Massachusetts General Hospital Cancer Center
Recruiting
Boston, Massachusetts, United States, 02114
Contact: Geoffrey I. Shapiro 617-632-4942 geoffrey_shapiro@dfci.harvard.edu
Principal Investigator: Geoffrey I. Shapiro
Brigham and Women's Hospital
Recruiting
Boston, Massachusetts, United States, 02115
Contact: Scott J. Rodig 617-525-7825 srodig@partners.org
Principal Investigator: Scott J. Rodig
Beth Israel Deaconess Medical Center
Recruiting
Boston, Massachusetts, United States, 02215
Contact: Gerburg M. Wulf 617-667-9925
Sub-Investigator: Gerburg M. Wulf
Dana-Farber Cancer Institute
Recruiting
Boston, Massachusetts, United States, 02215
Contact: Geoffrey I. Shapiro 617-632-4942 geoffrey_shapiro@dfci.harvard.edu
Principal Investigator: Geoffrey I. Shapiro
Massachusetts General Hospital
Recruiting
Charlestown, Massachusetts, United States, 02129
Contact: Jeffrey Supko 617-724-1970 jsupko@partners.org
Principal Investigator: Jeffrey Supko Collapse << |
| NCT01783171 | Pancreatic Adenocarcinoma
... more >> Recurrent Pancreatic Carcinoma
Stage III Pancreatic Cancer AJCC v6 and v7
Stage IV Pancreatic Cancer AJCC v6 and v7
Unresectable Pancreatic Carcinoma Collapse << | Phase 1 | Completed | - | United States, Colorado
... more >>
University of Colorado Cancer Center - Anschutz Cancer Pavilion
Aurora, Colorado, United States, 80045
University of Colorado
Denver, Colorado, United States, 80217-3364
United States, Maryland
Johns Hopkins University/Sidney Kimmel Cancer Center
Baltimore, Maryland, United States, 21287
United States, Wisconsin
University of Wisconsin Hospital and Clinics
Madison, Wisconsin, United States, 53792
Canada, Ontario
University Health Network-Princess Margaret Hospital
Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01624441 | Estrogen Receptor Negative
... more >> HER2/Neu Negative
Male Breast Carcinoma
Progesterone Receptor Negative
Recurrent Breast Carcinoma
Stage IV Breast Cancer AJCC v6 and v7
Triple-Negative Breast Carcinoma Collapse << | Phase 1 | Completed | - | United States, Texas
... more >>
M D Anderson Cancer Center
Houston, Texas, United States, 77030 Collapse << |
| NCT01711528 | Recurrent Plasma Cell Myeloma | Phase 1 | Completed | - | United States, Arizona
... more >>
Mayo Clinic in Arizona
Scottsdale, Arizona, United States, 85259
United States, Minnesota
Mayo Clinic
Rochester, Minnesota, United States, 55905
United States, Texas
M D Anderson Cancer Center
Houston, Texas, United States, 77030
United States, Virginia
Virginia Commonwealth University/Massey Cancer Center
Richmond, Virginia, United States, 23298
United States, Wisconsin
University of Wisconsin Hospital and Clinics
Madison, Wisconsin, United States, 53792
Aspirus UW Cancer Center
Wisconsin Rapids, Wisconsin, United States, 54494 Collapse << |
| NCT01650727 | Chronic Lymphocytic Leukemia
... more >> Small Lymphocytic Lymphoma Collapse << | Phase 1 | Completed | - | - |
| NCT02684617 | rrCLL
rrMM
... more >> rrDLBCL Collapse << | Phase 1 | Recruiting | February 6, 2020 | United States, California
... more >>
Call for Information (Investigational Site 0024)
Recruiting
Duarte, California, United States, 91010
Call for Information (Investigational Site 0036)
Recruiting
Los Angeles, California, United States, 90033
United States, Michigan
Call for Information (Investigational Site 0014)
Recruiting
Detroit, Michigan, United States, 48201
Call for Information (Investigational Site 0028)
Recruiting
Detroit, Michigan, United States, 48202
United States, Minnesota
Call for Information (Investigational Site 0006)
Recruiting
Rochester, Minnesota, United States, 55905
United States, New Jersey
Call for Information (Investigational Site 0003)
Recruiting
New Brunswick, New Jersey, United States, 08903
United States, New York
Call for Information (Investigational Site 0026)
Recruiting
Buffalo, New York, United States, 14263
United States, Ohio
Call for Information (Investigational Site 0002)
Recruiting
Columbus, Ohio, United States, 43210
United States, Pennsylvania
Call for Information (Investigational Site 0023)
Recruiting
Hershey, Pennsylvania, United States, 17033
United States, Texas
Call for Information (Investigational Site 0017)
Recruiting
Dallas, Texas, United States, 75246
United States, Washington
Call for Information (Investigational Site 0011)
Recruiting
Seattle, Washington, United States, 98116
United States, Wisconsin
Call for Information (Investigational Site 0018)
Recruiting
Madison, Wisconsin, United States, 53792
Australia
Merck Sharp & Dohme
Recruiting
North Ryde, Australia
Contact: Australian Medical Information Centre 61 2 8988 8428 Collapse << |
| NCT03484520 | Acute Myeloid Leukemia (AML) | Phase 1 | Recruiting | June 2, 2021 | United States, Arizona
... more >>
Mayo Clinic Arizona /ID# 201705
Not yet recruiting
Phoenix, Arizona, United States, 85054
United States, Arkansas
University of Arkansas /ID# 200016
Not yet recruiting
Little Rock, Arkansas, United States, 72205
United States, California
Cedars-Sinai Medical Ctr /ID# 202245
Not yet recruiting
Los Angeles, California, United States, 90048
David Geffen School of Medicin /ID# 200015
Not yet recruiting
Los Angeles, California, United States, 90095
United States, Illinois
The University of Chicago /ID# 200017
Recruiting
Chicago, Illinois, United States, 60637
United States, Maryland
Univ Maryland School Medicine /ID# 204015
Not yet recruiting
Baltimore, Maryland, United States, 21201
United States, North Carolina
Wake Forest Baptist Medical Center /ID# 200288
Not yet recruiting
Winston-Salem, North Carolina, United States, 27157-0001
United States, Ohio
The Ohio State University - Columbus /ID# 200668
Not yet recruiting
Columbus, Ohio, United States, 43210
United States, Texas
Univ TX, MD Anderson /ID# 205215
Not yet recruiting
Houston, Texas, United States, 77030
Australia, Queensland
Gold Coast University Hospital /ID# 202759
Not yet recruiting
Southport, Queensland, Australia, 4215
Australia, Tasmania
Royal Hobart Hospital /ID# 202763
Not yet recruiting
Hobart, Tasmania, Australia, 7000
Australia, Victoria
Monash Medical Centre /ID# 202762
Not yet recruiting
Melbourne, Victoria, Australia, 3168
Spain
Hospital Universitario y Politecnico La Fe /ID# 202318
Recruiting
Valencia, Valenciana, Spain, 46026
Hospital Ramon y Cajal /ID# 201729
Recruiting
Madrid, Spain, 28036
Hospital Clinico Univ de Salamanca /ID# 201728
Recruiting
Salamanca, Spain, 37007 Collapse << |
| NCT01676753 | Advanced or Metastatic Breast ... more >>Cancer
Triple Negative Breast Cancer Collapse << | Phase 1 | Recruiting | August 2019 | United States, California
... more >>
UCSF Helen Diller Family Comprehensive Cancer Center
Recruiting
San Francisco, California, United States, 94143
Contact: A. Jo Chien, MD 877-827-3222 cancertrials@ucsf.edu
Principal Investigator: A. Jo Chien, MD
Principal Investigator: Andrei Goga, MD, PhD
Sub-Investigator: Michelle Melisko, MD
Sub-Investigator: Mark Moasser, MD
Sub-Investigator: Pamela N Munster, MD
Sub-Investigator: Hope Rugo, MD
Sub-Investigator: John Park, MD
Sub-Investigator: Melanie Majure, MD Collapse << |
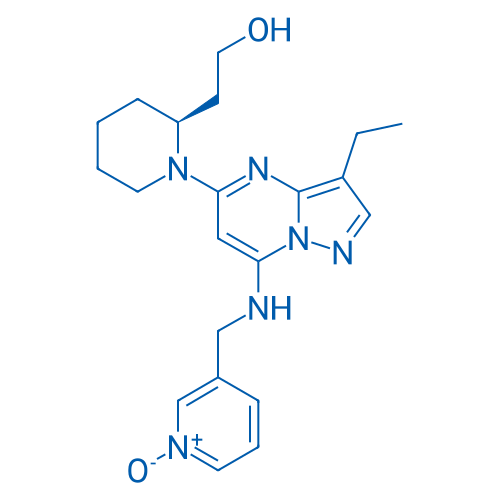

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
