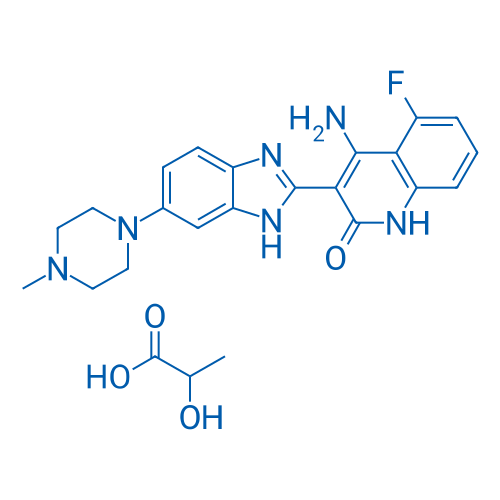
CAS No.: 692737-80-7
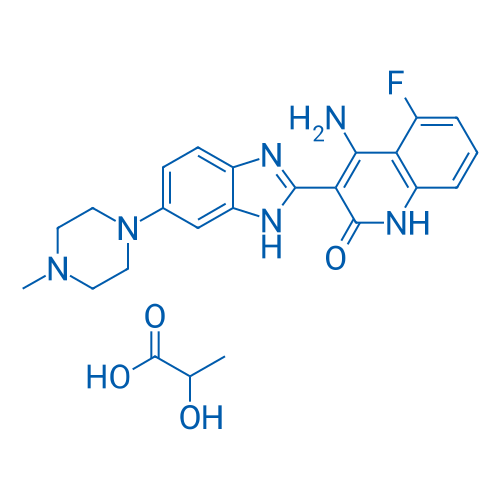
Dovitinib lactate/多韦替尼乳酸盐 Catalog No. CSN12808
Synonyms: CHIR-258 lactate;TKI-258 lactate;多韦替尼乳酸盐
Dovitinib lactate is a potent inhibitor of fibroblast growth factor receptor 3 (FGFR3) with an IC50 of 5 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01678105 Recurrent Adenoid Cystic Carci... more >>noma of the Salivary Glands Metastatic Adenoid Cystic Carcinoma of the Salivary Glands Salivary Gland Cancers ACC Collapse << Phase 2 Completed - Canada, Ontario ... more >> Tom Baker Cancer Centre Calgary, Ontario, Canada, T2N 4N2 Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 London Health Sciences Centre London, Ontario, Canada, N6A 4L6 Ottawa Hospital Regional Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Collapse << NCT01223027 Metastatic Renal Cell Carcinom... more >>a Collapse << Phase 3 Completed - - NCT01496534 Solid Tumors ... more >>Bladder Cancer Collapse << Phase 1 Terminated(toxicity of combina... more >>tion of medications) Collapse << - United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << - 更多
- 参考文献
- [1] Huynh H, Chow PK, et al. Dovitinib demonstrates antitumor and antimetastatic activities in xenograft models of hepatocellular carcinoma. J Hepatol. 2012 Mar;56(3):595-601.
- [2] Trudel S, Li ZH, et al. CHIR-258, a novel, multitargeted tyrosine kinase inhibitor for the potential treatment of t(4;14) multiple myeloma. Blood. 2005 Apr 1;105(7):2941-8.
- [3] Chiu YH, et al. Dovitinib Triggers Apoptosis and Autophagic Cell Death by Targeting SHP-1/p-STAT3 Signaling in Human Breast Cancers. J Oncol. 2019 Aug 14;2019:2024648.
- [4] Konecny GE, et al. Activity of the fibroblast growth factor receptor inhibitors dovitinib (TKI258) and NVP-BGJ398 in human endometrial cancer cells. Mol Cancer Ther. 2013 May;12(5):632-42.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 692737-80-7 | 储存条件 |
|
|||||||
| 分子式 | C24H27FN6O4 | 运输 | 蓝冰 | |||||||
| 分子量 | 482.51 | 别名 | CHIR-258 lactate;TKI-258 lactate;多韦替尼乳酸盐 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01678105 | Recurrent Adenoid Cystic Carci... more >>noma of the Salivary Glands Metastatic Adenoid Cystic Carcinoma of the Salivary Glands Salivary Gland Cancers ACC Collapse << | Phase 2 | Completed | - | Canada, Ontario ... more >> Tom Baker Cancer Centre Calgary, Ontario, Canada, T2N 4N2 Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 London Health Sciences Centre London, Ontario, Canada, N6A 4L6 Ottawa Hospital Regional Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT01223027 | Metastatic Renal Cell Carcinom... more >>a Collapse << | Phase 3 | Completed | - | - |
| NCT01496534 | Solid Tumors ... more >>Bladder Cancer Collapse << | Phase 1 | Terminated(toxicity of combina... more >>tion of medications) Collapse << | - | United States, New York ... more >> Icahn School of Medicine at Mount Sinai New York, New York, United States, 10029 Collapse << |
| NCT01528345 | Metastatic Breast Cancer | Phase 2 | Terminated(Slow and low enroll... more >>ment) Collapse << | - | - |
| NCT01223027 | - | - | Completed | - | - |
| NCT01769547 | Advanced Malignant Pleural Mes... more >>othelioma MPM Collapse << | Phase 2 | Terminated(Stage 1 accrual com... more >>pleted. Study did not proceed to Stage 2 accrual.) Collapse << | - | Canada, Alberta ... more >> Tom Baker Cancer Centre Calgary, Alberta, Canada, T2N 4N2 Canada, Ontario Juravinski Cancer Centre Hamilton, Ontario, Canada, L8V 5C2 Ottawa General Hospital Cancer Centre Ottawa, Ontario, Canada, K1H 8L6 Northeast Cancer Centre, Health Sciences North Sudbury, Ontario, Canada, P3E 5J1 Princess Margaret Hospital Toronto, Ontario, Canada, M5G 1X6 Collapse << |
| NCT01528345 | - | - | Terminated(Slow and low enroll... more >>ment) Collapse << | - | - |
| NCT01888965 | Colorectal Cancer ... more >> Pancreas Cancer Collapse << | Phase 2 | Terminated(Adverse Event issue... more >>s.) Collapse << | - | United States, District of Col... more >>umbia Georgetown University- Lombardi Cancer Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT01888965 | - | - | Terminated(Adverse Event issue... more >>s.) Collapse << | - | - |
| NCT01753713 | Adult Giant Cell Glioblastoma ... more >> Adult Glioblastoma Adult Gliosarcoma Recurrent Adult Brain Tumor Collapse << | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01680796 | Multiple Myeloma | Phase 1 | Withdrawn(The target drug, dov... more >>itinib, failed as a single agent in prior studies in patients with heavily treated multiple myeloma.) Collapse << | - | - |
| NCT01791387 | Clear Cell Renal Cell Carcinom... more >>a Collapse << | Phase 2 | Unknown | June 2015 | New Zealand ... more >> Auckland Hospital Auckland, New Zealand, 1142 Collapse << |
| NCT01484041 | Breast Cancer | Phase 1 Phase 2 | Terminated(Decision by company... more >> to cease development of dovitinib) Collapse << | - | United States, District of Col... more >>umbia Georgetown Lombardi Comprehensive Cancer Center Washington, District of Columbia, United States, 20007 Collapse << |
| NCT01484041 | - | - | Terminated(Decision by company... more >> to cease development of dovitinib) Collapse << | - | - |
| NCT01548924 | Solid Tumors | Phase 1 | Terminated(Drug toxicity) | - | Spain ... more >> Hospital Universitario de Bellvitge Hospitalet de Llobregat, Barcelona, Spain, 08907 Hospital Universitario de Fuenlabrada Fuenlabrada, Madrid, Spain, 28049 MD Anderson Cancer Centre Madrid, Spain Collapse << |
| NCT01831726 | Tumor Pathway Activations Inhi... more >>bited by Dovitinib Collapse << | Phase 2 | Completed | - | - |
| NCT01753713 | - | - | Completed | - | - |
| NCT01232296 | Hepatocellular Carcinoma | Phase 2 | Completed | - | China, Jiangsu ... more >> Novartis Investigative Site Nanjing, Jiangsu, China, 210002 China, Shanxi Novartis Investigative Site Xi'an, Shanxi, China, 710032 China, Zhejiang Novartis Investigative Site Hangzhou, Zhejiang, China, 310016 China Novartis Investigative Site Beijing, China, 100039 Hong Kong Novartis Investigative Site Hong Kong, Hong Kong Novartis Investigative Site Shatin, New Territories, Hong Kong Japan Novartis Investigative Site Kashiwa, Chiba, Japan, 277-8577 Novartis Investigative Site Yokohama-city, Kanagawa, Japan, 232-0024 Novartis Investigative Site OsakaSayama, Osaka, Japan, 589-8511 Korea, Republic of Novartis Investigative Site Seoul, Korea, Korea, Republic of, 03722 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 05505 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 06351 Novartis Investigative Site Seoul, Korea, Korea, Republic of, 110 744 Novartis Investigative Site Seoul, Korea, Republic of, 136-705 Singapore Novartis Investigative Site Singapore, Singapore, 308433 Taiwan Novartis Investigative Site Taipei, Taiwan, ROC, Taiwan, 112 Novartis Investigative Site Kuei-Shan Chiang, Taoyuan/ Taiwan ROC, Taiwan, 33305 Novartis Investigative Site Taichung, Taiwan, 40705 Novartis Investigative Site Taipei, Taiwan, 10048 Thailand Novartis Investigative Site Bangkok, Thailand, 10330 Novartis Investigative Site Bangkok, Thailand, 10700 Novartis Investigative Site Chiang Mai, Thailand, 50200 Novartis Investigative Site Khon Kaen, Thailand, 40002 Novartis Investigative Site Songkla, Thailand, 90110 Collapse << |
| NCT02065323 | Prostate Cancer | Phase 2 | Withdrawn(Terminated due to bu... more >>dgetary considerations and length of development.) Collapse << | - | United States, Nevada ... more >> Comprehensive Cancer Centers of Nevada Las Vegas, Nevada, United States, 89148 Collapse << |
| NCT01232296 | - | - | Completed | - | - |
| NCT01831726 | - | - | Completed | - | - |
| NCT01994590 | Prostate Cancer | Phase 2 | Terminated(Sponsor stopped sup... more >>plying study drug) Collapse << | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02116803 | Solid Tumors | Phase 2 Phase 3 | Completed | - | United States, Nevada ... more >> Novartis Investigative Site Las Vegas, Nevada, United States, 89169 United States, New York Novartis Investigative Site Bronx, New York, United States, 10467-2490 Austria Novartis Investigative Site Salzburg, Austria, 5020 Belgium Novartis Investigative Site Wilrijk, Belgium, 2610 Denmark Novartis Investigative Site Copenhagen, Denmark, DK-2100 Italy Novartis Investigative Site Pavia, PV, Italy, 27100 Novartis Investigative Site Torino, TO, Italy, 10153 Japan Novartis Investigative Site Osaka-city, Osaka, Japan, 545-8586 Spain Novartis Investigative Site Palma De Mallorca, Islas Baleares, Spain, 07120 Collapse << |
| NCT01719549 | Gastric Cancer | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 138-736 Collapse << |
| NCT01514526 | Adrenocortical Carcinoma | Phase 2 | Completed | - | Spain ... more >> Complejo Hospitalario Universitario de Santiago Santiago de Compostela, A Coruña, Spain, 15706 Hospital Universitario Fundación de Alcorcón Alcorcón, Madrid, Spain, 28922 Hospital del Mar Barcelona, Spain, 08003 Hospital Universitario Reina Sofía Córdoba, Spain, 14004 Hospital Universitario Central de Asturias Oviedo, Spain, 33006 Complejo Hospitalario de Navarra Pamplona, Spain, 31008 Fundación Instituto Valenciano de Oncología Valencia, Spain, 46009 Collapse << |
| NCT01478373 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Finland ... more >> Novartis Investigative Site HUS, Finland, FIN-00029 France Novartis Investigative Site Bordeaux, France, 33076 Novartis Investigative Site Lille Cedex, France, 59020 Novartis Investigative Site Lyon Cedex, France, 69373 Novartis Investigative Site Reims, France, 51092 Novartis Investigative Site Villejuif Cedex, France, 94805 Germany Novartis Investigative Site Essen, Germany, 45147 Italy Novartis Investigative Site Milano, MI, Italy, 20133 Novartis Investigative Site Roma, RM, Italy, 00168 Novartis Investigative Site Candiolo, TO, Italy, 10060 Novartis Investigative Site Torino, TO, Italy, 10153 Spain Novartis Investigative Site Barcelona, Catalunya, Spain, 08035 Novartis Investigative Site Hospitalet de LLobregat, Catalunya, Spain, 08907 Novartis Investigative Site Palma De Mallorca, Islas Baleares, Spain, 07120 Novartis Investigative Site Barcelona, Spain, 08041 Collapse << |
| NCT01478373 | - | - | Completed | - | - |
| NCT01266070 | Von Hippel-Lindau Syndrome | Phase 2 | Terminated(Trial met toxicity ... more >>stopping rule) Collapse << | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01440959 | Gastrointestinal Stromal Tumor... more >>s Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Asan Medical Center, University of Ulsan College of Medicine Seoul,, Korea, Republic of, 138-736 Collapse << |
| NCT00790426 | Urothelial Cancer | Phase 2 | Completed | - | - |
| NCT01440959 | - | - | Completed | - | - |
| NCT02116803 | - | - | Completed | - | - |
| NCT01262027 | Breast Cancer | Phase 2 | Active, not recruiting | December 2017 | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01596647 | Advanced Solid Tumors, Excludi... more >>ng Breast Cancer Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> University of Kansas Cancer Center Medical Center Kansas City, Kansas, United States, 66160 United States, Michigan Henry Ford Hospital Henry Ford Detroit, Michigan, United States, 48202-2689 United States, Minnesota University of Minnesota Minneapolis, Minnesota, United States, 55455 United States, Nevada Comprehensive Cancer Centers Las Vegas, Nevada, United States, 89169 United States, New Jersey Cancer Institute of New Jersey Dept of Cancer Institute of NJ New Brunswick, New Jersey, United States, 08901 Collapse << |
| NCT01262027 | - | - | Active, not recruiting | - | - |
| NCT00243763 | Multiple Myeloma | Phase 1 | Terminated | - | United States, Arizona ... more >> Mayo Clinic - Arizona Scottsdale, Arizona, United States, 85259 United States, Florida H. Lee Moffitt Cancer Center and Research Institute Tampa, Florida, United States, 33612 United States, Georgia Emory University Atlanta, Georgia, United States, 30322 United States, Massachusetts Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 United States, Minnesota Mayo Clinic - Minnesota Rochester, Minnesota, United States, 55905 Collapse << |
| NCT00279773 | Acute Myeloid Leukemia | Phase 1 | Terminated(Study was stopped d... more >>ue to time dependent drug accumulation) Collapse << | - | United States, Texas ... more >> The University of Texas, M.D. Anderson Cancer Center Houston, Texas, United States, 77020 Collapse << |
| NCT01700270 | Advanced Solid Tumors, Excludi... more >>ng Breast Cancer Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Montefiore Medical Center Montefiore Medical Center (SC) Bronx, New York, United States, 10467 United States, Texas Cancer Therapy & Research Center / UT Health Science Center SC San Antonio, Texas, United States, 78229 Denmark Novartis Investigative Site Copenhagen, Denmark, DK-2100 Netherlands Novartis Investigative Site Amsterdam, Netherlands, 1066 CX Switzerland Novartis Investigative Site Chur, Switzerland, 7000 Novartis Investigative Site Genève, Switzerland, 1211 Collapse << |
| NCT01524692 | - | - | Completed | - | - |
| NCT00958971 | Metastatic Breast Cancer | Phase 2 | Completed | - | - |
| NCT01266070 | - | - | Terminated(Trial met toxicity ... more >>stopping rule) Collapse << | - | - |
| NCT00831792 | Prostate Cancer | Phase 2 | Completed | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT01058434 | Relapsed or Refractory Multipl... more >>e Myeloma Collapse << | Phase 2 | Completed | - | - |
| NCT01417143 | Adenoid Cystic Carcinoma | Phase 2 | Completed | - | Korea, Republic of ... more >> Department of Internal Medicine, Seoul National University Hospital Seoul, Korea, Republic of, 110-744 Collapse << |
| NCT00303251 | Melanoma | Phase 1 Phase 2 | Completed | - | United States, Kentucky ... more >> James Graham Brown Cancer Center Louisville, Kentucky, United States, 40202 United States, Pennsylvania University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232 United States, Texas MD Anderson Cancer Houston, Texas, United States, 77030 Collapse << |
| NCT01270906 | Neoplasms Can... more >>cer Tumors Collapse << | Phase 1 | Terminated | - | United Kingdom ... more >> Novartis Investigative Site Glasgow, United Kingdom Novartis Investigative Site Sutton, United Kingdom Collapse << |
| NCT02048943 | Duct Cell Adenocarcinoma of th... more >>e Pancreas Recurrent Pancreatic Cancer Stage III Pancreatic Cancer Stage IV Pancreatic Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Withdrawn(Lack of funding) | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT00715182 | Advanced/ Metastatic Renal Cel... more >>l Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> Novartis Investigative Site San Francisco, California, United States United States, North Carolina Novartis Investigative Site Durham, North Carolina, United States, 27710 United States, Washington Novartis Investigative Site Seattle, Washington, United States, 98109-1023 France Novartis Investigative Site Bordeaux Cedex, France, 33075 Novartis Investigative Site Villejuif Cedex, France, 94805 Germany Novartis Investigative Site Hannover, Germany, 30625 Novartis Investigative Site München, Germany, 81675 Netherlands Novartis Investigative Site Rotterdam, Netherlands, 3075 EA Spain Novartis Investigative Site Madrid, Spain, 28041 Taiwan Novartis Investigative Site Taipei, Taiwan, ROC, Taiwan, 112 Novartis Investigative Site Taichung, Taiwan, 40705 Novartis Investigative Site Taipei, Taiwan, 10002 Collapse << |
| NCT01443481 | Solid Tumors ... more >>Hepatic Impairment Collapse << | Phase 1 | Completed | - | United States, California ... more >> University of California at Los Angeles Dept. of UCLA (4) Los Angeles, California, United States, 90095 United States, North Carolina Duke University Medical Center DUMC Durham, North Carolina, United States, 27710 United States, Texas Cancer Therapy & Research Center / UT Health Science Center SC San Antonio, Texas, United States, 78229 Belgium Novartis Investigative Site Gent, Belgium, 9000 Germany Novartis Investigative Site Frankfurt, Germany, 60590 Novartis Investigative Site Hannover, Germany, 30625 Italy Novartis Investigative Site Milano, MI, Italy, 20133 Novartis Investigative Site Rozzano, MI, Italy, 20089 Novartis Investigative Site Verona, VR, Italy, 37126 Netherlands Novartis Investigative Site Amsterdam, Netherlands, 1066 CX Novartis Investigative Site Maastricht, Netherlands, 5800 Singapore Novartis Investigative Site Singapore, Singapore, 119228 Collapse << |
| NCT01421004 | Advanced Solid Tumors ... more >> Excluding Breast Cancer Collapse << | Phase 1 | Completed | - | United States, California ... more >> City of Hope National Medical Center SC-2 Duarte, California, United States, 91010-3000 University of California at Los Angeles UCLA LeConte Location Los Angeles, California, United States, 90095 University of California San Francisco UCSF (SC) San Francisco, California, United States, 94101 United States, Florida Florida Cancer Specialists Sarasota Office Fort Myers, Florida, United States, 33901 United States, Illinois Rush University Medical Center Rush 3 Chicago, Illinois, United States, 60612 United States, Missouri Washington University School of Medicine SC St. Louis, Missouri, United States, 63110 United States, New York Montefiore Medical Center Montefiore Medical Center (SC) Bronx, New York, United States, 10467 United States, Oklahoma University of Oklahoma Health Sciences Center OUHSC - SC Oklahoma City, Oklahoma, United States, 73104 United States, Pennsylvania University of Pittsburgh Cancer Institute Pittsburgh, Pennsylvania, United States, 15232-1305 United States, Tennessee Sarah Cannon Research Institute Sarah Cannon Research (SC) Nashville, Tennessee, United States, 37203 United States, Texas Sammons Cancer Center - Texas Oncology SC-2 Dallas, Texas, United States, 78246 Cancer Therapy & Research Center / UT Health Science Center InstituteForDrugDevelopment(5) San Antonio, Texas, United States, 78229 United States, Utah University of Utah / Huntsman Cancer Institute Huntsman Salt Lake City, Utah, United States, 84112 United States, Wisconsin University of Wisconsin Univ Wisc Madison, Wisconsin, United States, 53792 Collapse << |
| NCT01379534 | - | - | Completed | - | - |
| NCT01030055 | Neoplasms Can... more >>cer Tumors Collapse << | Phase 1 | Completed | - | United States, California ... more >> City of Hope National Medical Center Duarte, California, United States, 91010 University of California at Los Angeles Los Angeles, California, United States, 90095 United States, North Carolina Duke University Medical Center Cancer Dept Durham, North Carolina, United States, 27710 United States, Utah University of Utah / Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT01515969 | Non-small Cell Lung Cancer (NS... more >>CLC), Recurrent Non-small Cell Lung Cancer (NSCLC), Stage IV Collapse << | Phase 1 | Terminated(Patient safety - Un... more >>acceptable toxicity) Collapse << | - | United States, California ... more >> Stanford University Stanford, California, United States, 94305 Collapse << |
| NCT01155713 | Neoplasm Canc... more >>er Tumors Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Scottsdale Healthcare/TGen Clinical Research Service TGen Clinical Research Service Scottsdale, Arizona, United States, 85258 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27710 United States, Tennessee Sarah Cannon Research Institute Sarah Cannon Research Instit Nashville, Tennessee, United States, 37203 United States, Utah University of Utah / Huntsman Cancer Institute Salt Lake City, Utah, United States, 84112 Collapse << |
| NCT00669097 | Advanced Solid Malignancies | Phase 1 | Completed | - | Netherlands ... more >> Novartis Investigative Site Amsterdam, Netherlands Collapse << |
| NCT01524692 | Adenoid Cystic Carcinoma | Phase 2 | Completed | - | United States, Virginia ... more >> University of Virginia Health System Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT01972750 | First or Second Recurrence of ... more >>Glioblastoma Collapse << | Phase 1 | Unknown | November 2016 | Germany ... more >> Department of Neurology and Center of Integrated Oncology, University Hospital Bonn Bonn, Germany, 53105 Collapse << |
| NCT01576380 | Adenocarcinoma, Scirrhous ... more >> Linitis Plastica Stomach Neoplasms Stomach Diseases Neoplasms by Site Neoplasms Collapse << | Phase 2 | Completed | - | Japan ... more >> Novartis Investigative Site Nagoya, Aichi, Japan, 464-8681 Novartis Investigative Site Kashiwa, Chiba, Japan, 277-8577 Novartis Investigative Site Matsuyama, Ehime, Japan, 791-0280 Novartis Investigative Site Sapporo-city, Hokkaido, Japan, 060-8648 Novartis Investigative Site Takatsuki, Osaka, Japan, 569-8686 Novartis Investigative Site Sunto-gun, Shizuoka, Japan, 411-8777 Novartis Investigative Site Chuo-ku, Tokyo, Japan, 104-0045 Novartis Investigative Site Koto, Tokyo, Japan, 135-8550 Collapse << |
| NCT01497392 | Adenocarcinoma of the Pancreas... more >> Stage III Pancreatic Cancer Stage IV Pancreatic Cancer Unspecified Adult Solid Tumor, Protocol Specific Collapse << | Phase 1 | Completed | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT01379534 | Solid Tumors and Advanced Endo... more >>metrial Cancer Endometrial Cancer Second-line Treatment VEGF Collapse << | Phase 2 | Completed | - | - |
| NCT02108782 | Gastrinoma Gl... more >>ucagonoma Insulinoma Pancreatic Polypeptide Tumor Recurrent Islet Cell Carcinoma Somatostatinoma Collapse << | Phase 2 | Withdrawn | - | - |
| NCT01471548 | Advanced Solid Tumors | Phase 1 | Completed | - | Japan ... more >> Novartis Investigative Site Takatsuki, Osaka, Japan, 569-8686 Novartis Investigative Site Hidaka, Saitama, Japan, 350-1241 Collapse << |
| NCT02720926 | Colorectal Cancer ... more >> Gastric Cancer Collapse << | Phase 1 | Terminated(Unafavourable toxic... more >>ity profile) Collapse << | - | Singapore ... more >> National Cancer Centre singapore Singapore, Singapore Collapse << |
| NCT01741116 | Hormone Refractory Prostate Ca... more >>ncer Collapse << | Phase 2 | Unknown | June 2016 | Korea, Republic of ... more >> Korean Cancer Study Group Recruiting Seoul, Chongro-ku, Korea, Republic of, 110999 Contact: So-Yeon Kang 82-2-3276-3511 datacenter1@kcsg.org Korea University Anam Hospital Recruiting Seoul, Seongbuk-gu, Inchon-ro, Korea, Republic of, 136-705 Contact: Kyong Hwa Park, MD, phD +82-2-920-5983 ext 5980 khpark@korea.ac.kr Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
