CAS No.: 1428-67-7
DPN Catalog No. CSN15920
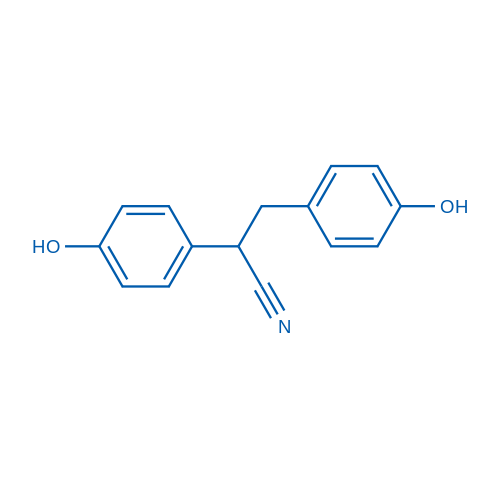
Synonyms: Diarylpropionitrile;2,3-bis (4-Hydroxyphenyl) Propionitrile
DPN is a non-steroidal estrogen receptor β (ER β) selective ligand.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01695629 - - Completed - United States, Michigan ... more >> Kellogg Eye Center Ann Arbor, Michigan, United States, 48105 Collapse << NCT00348894 Pain Phase 2 Terminated(- Study was termina... more >>ted due to insufficient clinical efficacy observed in previous studies conducted in postherpetic neuralgia.) Collapse << - - NCT03743636 Peripheral Artery Disease Phase 3 Recruiting April 2022 United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu Contact: Kathryn Domanchuk, BS 312-503-6438 k-domanchuk@northwestern.edu Principal Investigator: Mary McDermott, MD Collapse << - 更多
- 参考文献
- [1] Suwanna N, Thangnipon W, et al. Neuroprotective effects of diarylpropionitrile against β-amyloid peptide-induced neurotoxicity in rat cultured cortical neurons. Neurosci Lett. 2014 Aug 22;578:44-9.
- [2] Khalaj AJ, Yoon J, et al. Estrogen receptor (ER) β expression in oligodendrocytes is required for attenuation of clinical disease by an Erβ ligand. Proc Natl Acad Sci U S A. 2013 Nov 19;110(47):19125-30.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1428-67-7 | 储存条件 |
|
|
| 分子式 | C15H13NO2 | 运输 | 蓝冰 | |
| 分子量 | 239.27 | 别名 | Diarylpropionitrile;2,3-bis (4-Hydroxyphenyl) Propionitrile |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT01695629 | - | - | Completed | - | United States, Michigan ... more >> Kellogg Eye Center Ann Arbor, Michigan, United States, 48105 Collapse << |
| NCT00348894 | Pain | Phase 2 | Terminated(- Study was termina... more >>ted due to insufficient clinical efficacy observed in previous studies conducted in postherpetic neuralgia.) Collapse << | - | - |
| NCT03743636 | Peripheral Artery Disease | Phase 3 | Recruiting | April 2022 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Mary McDermott, MD 312-503-6419 mdm608@northwestern.edu Contact: Kathryn Domanchuk, BS 312-503-6438 k-domanchuk@northwestern.edu Principal Investigator: Mary McDermott, MD Collapse << |
| NCT02513693 | Neuromuscular Blockade | Phase 4 | Unknown | March 2016 | Czech Republic ... more >> Dept. of Anesthesiology and Intensive Care Medicine, University Hospital Olomouc Recruiting Olomouc, Czech Republic, 775 20 Contact: Milan Adamus, MD,PhD,MBA +420 588 442 705 milan.adamus@seznam.cz Contact: Lenka Doubravská, MD +420 588 445 979 lenadoub@seznam.cz Dept. of Anesthesiology, Perioperative Medicine and Intensive Care, J. E. Purkinje University, Masaryk Hospital Recruiting Usti nad Labem, Czech Republic, 401 13 Contact: Vladimir Cerny, MD,PhD,FCCM +420 602 492 054 cernyvla1960@gmail.com Collapse << |
| NCT01057693 | Diabetic Neuropathy, Painful | Phase 3 | Completed | - | - |
| NCT01057693 | - | - | Completed | - | - |
| NCT03176628 | Acute Kidney Injury | Not Applicable | Recruiting | October 31, 2018 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02118 Contact: Petra Simic, MD, PhD 617-724-6700 psimic@bwh.harvard.edu Contact: Katherine E Brock, BS 617-643-9463 kbrock2@mgh.harvard.edu Principal Investigator: Eugene Rhee, MD Sub-Investigator: Ravi I Thadhani, MD, MPH Sub-Investigator: Petra Simic, MD, PhD Collapse << |
| NCT03501433 | Aging Lipemia | Not Applicable | Recruiting | December 2018 | United States, Iowa ... more >> Iowa State University Recruiting Ames, Iowa, United States, 50011 Contact: Rudy Valentine, Ph.D. 515-294-3867 rvalenti@iastate.edu Principal Investigator: Rudy Valentine, Ph.D. Collapse << |
| NCT01928381 | Diabetic Neuropathy, Painful; ... more >>Diabetic Neuropathies Collapse << | Phase 2 | Completed | - | United States, Florida ... more >> Research Site Clearwater, Florida, United States Research Site Orlando, Florida, United States United States, Georgia Research Site Newnan, Georgia, United States United States, Massachusetts Research Site Brockton, Massachusetts, United States Research Site Natick, Massachusetts, United States Research Site Watertown, Massachusetts, United States United States, North Carolina Research Site Winston-Salem, North Carolina, United States United States, Pennsylvania Research Site Duncansville, Pennsylvania, United States Research Site Philadelphia, Pennsylvania, United States Collapse << |
| NCT01608126 | Cervical Plexus Block Comparis... more >>on Collapse << | Not Applicable | Completed | - | Czech Republic ... more >> Krajská zdravotní, a.s. - Masarykova nemocnice v Ústí nad Labem, o.z. Ústí nad Labem, Czech Republic, 401 13 Collapse << |
| NCT02461225 | - | - | - | - | - |
| NCT02061267 | Metabolic Syndrome | Not Applicable | Unknown | December 2014 | Spain ... more >> Instituto de la Grasa, CSIC Seville, Spain, 41012 Collapse << |
| NCT02619539 | - | - | Completed | - | Czech Republic ... more >> University Hospital in Plzen Plzen, Czech Republic Masaryk Hospital in Usti nad Labem Usti nad Labem, Czech Republic, 40001 Collapse << |
| NCT01566799 | Locally Advanced Malignant Neo... more >>plasm Collapse << | Phase 2 | Unknown | April 2014 | Mexico ... more >> Instituto Nacional Cancerologi Mexico, Distrito Federal, Mexico, 14080 Collapse << |
| NCT02389660 | Depression | Not Applicable | Completed | - | Poland ... more >> University of Social Sciences and Humanities Warsaw, Poland, 03-815 Collapse << |
| NCT00465907 | Lung Cancer | Phase 2 | Unknown | December 2009 | Singapore ... more >> Johns Hopkins Singapore International Medical Center Singapore, Singapore, 308433 Collapse << |
| NCT02064569 | Leber Hereditary Optic Neuropa... more >>thy Collapse << | Phase 1 Phase 2 | Active, not recruiting | June 2020 | France ... more >> CIC du CHNO DES QUINZE-VINGTS Paris, France, 75012 Collapse << |
| NCT00164528 | Treatment Con... more >>trol Collapse << | Phase 1 Phase 2 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01928381 | - | - | Completed | - | - |
| NCT00466778 | - | - | Completed | - | United States, New York ... more >> Michael I. Weintraub, MD Briarcliff Manor, New York, United States, 10510 Collapse << |
| NCT00888758 | Coronary Heart Disease ... more >> Percutaneous Coronary Intervention Collapse << | Phase 4 | Unknown | May 2011 | Czech Republic ... more >> Department of Cardiology, Masaryk hospital and University of JEP Not yet recruiting Ústí nad Labem, Czech Republic, 40113 Contact: Pavel Cervinka, MD,PhD +420477117886 pavel.cervinka@mnul.cz Sub-Investigator: Petr Kala, MD,P hD Sub-Investigator: Ladislav Pesl, MD Collapse << |
| NCT00454948 | Obesity | Phase 1 | Unknown | August 2009 | United States, California ... more >> Recruiting San Jose, California, United States Collapse << |
| NCT03601494 | Diabetic Neuropathies ... more >> Chronic Pain Collapse << | Not Applicable | Completed | - | Egypt ... more >> Emad Zarief Kamel Said Assiut, Egypt, 71111 Collapse << |
| NCT01521598 | Painful Diabetic Neuropathy | Phase 2 | Completed | - | United States, Alabama ... more >> Neurology Clinic, P.C. Northport, Alabama, United States, 35476 United States, Arkansas Principals Research Group Hot Springs, Arkansas, United States, 71901 Clinical Trials, Inc. Little Rock, Arkansas, United States, 72205 United States, California Collaborative Neuroscience Network, Inc. Long Beach, California, United States, 90806 Neurological Research Institute Santa Monica, California, United States, 90404 United States, Florida Renstar Medical Research Ocala, Florida, United States, 33471 Comprehensive Clinical Development St. Petersburg, Florida, United States, 33716 Clinical Research of West Florida, Inc. Tampa, Florida, United States, 33603 United States, Kansas International Clinical Research Institute Leawood, Kansas, United States, 66211 United States, Michigan Michigan Head Pain & Neurological Institute. Ann Arbor, Michigan, United States, 48104 United States, Nebraska Creighton Diabetes Center Omaha, Nebraska, United States, 68131 United States, Oregon Sunstone Medical Research, LLC Medford, Oregon, United States, 97504 United States, Texas Nerve and Muscle Center of Texas Houston, Texas, United States, 77030 Collapse << |
| NCT02423434 | - | - | Enrolling by invitation | August 2018 | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48105 Australia Queensland University of Technology Brisbane, Australia, 4059 Canada, Alberta University of Calgary Calgary, Alberta, Canada, T3B6A8 Canada, Ontario Mount Sinai Hospital and University Health Network Toronto, Ontario, Canada, M5G 2C4 United Kingdom University of Manchester Manchester, United Kingdom, M139PT Collapse << |
| NCT03115970 | - | - | Not yet recruiting | December 31, 2017 | - |
| NCT02214329 | Diabetes | Not Applicable | Unknown | December 2015 | United States, Arizona ... more >> University Medical Center Recruiting Tucson, Arizona, United States, 85724 Contact: David G Armstrong, DPM, MD, PhD 520-626-1349 dga@email.arizona.edu Contact: Bijan Najafi, PhD 520 626 7097 bnajafi@surgery.arizona.edu Principal Investigator: David G Armstrong, DPM, MD, PhD Collapse << |
| NCT02291159 | Stroke Spasti... more >>city Collapse << | Not Applicable | Completed | - | Spain ... more >> San Jorge University Villanueva de Gallego, Zaragoza, Spain, 50830 Collapse << |
| NCT02056431 | - | - | Completed | - | - |
| NCT03440203 | - | - | Completed | - | Japan ... more >> Okayama University Okayama, Japan, 7008558 Collapse << |
| NCT03356041 | Diabetic Peripheral Neuropathy | Not Applicable | Recruiting | March 23, 2018 | Iraq ... more >> Prof. Kawa Dizaye Recruiting Erbil, Iraq Contact: Kawa Dizaye, PhD 009647504452392 kawa.dizaye@hmu.edu.iq Contact: Anfal Al-Mallah, MSc 009647702080031 anfalalmallah@gmail.com Collapse << |
| NCT02056431 | Diabetic Peripheral Neuropathy | Not Applicable | Completed | - | United States, California ... more >> Division of Research Kaiser Permanente Oakland, California, United States, 94612 Collapse << |
| NCT00629681 | Painful Diabetic Neuropathy an... more >>d Post Herpetic Neuralgia Collapse << | Phase 4 | Completed | - | Germany ... more >> Pfizer Investigational Site Aachen, Germany, 52064 Pfizer Investigational Site Albstadt, Germany, 72458 Pfizer Investigational Site Berlin, Germany, 10117 Pfizer Investigational Site Berlin, Germany, 10435 Pfizer Investigational Site Berlin, Germany, 13053 Pfizer Investigational Site Bielefeld, Germany, 33604 Pfizer Investigational Site Celle, Germany, 29221 Pfizer Investigational Site Deggingen, Germany, 73326 Pfizer Investigational Site Duisburg, Germany, 47051 Pfizer Investigational Site Erbach, Germany, 64711 Pfizer Investigational Site Erfurt, Germany, 99089 Pfizer Investigational Site Frankfurt, Germany, 60311 Pfizer Investigational Site Gera, Germany, 07548 Pfizer Investigational Site Goeppingen, Germany, 73033 Pfizer Investigational Site Hamburg, Germany, 21073 Pfizer Investigational Site Hamburg, Germany, 22149 Pfizer Investigational Site Hamburg, Germany, 22607 Pfizer Investigational Site Hemsbach, Germany, 69502 Pfizer Investigational Site Hildesheim, Germany, 31134 Pfizer Investigational Site Holle, Germany, 31188 Pfizer Investigational Site Itzehoe, Germany, 25524 Pfizer Investigational Site Karlsruhe, Germany, 76133 Pfizer Investigational Site Karlsruhe, Germany, 76199 Pfizer Investigational Site Katzhuette, Germany, 98746 Pfizer Investigational Site Leer, Germany, 26789 Pfizer Investigational Site Leipzig, Germany, 04103 Pfizer Investigational Site Limburgerhof, Germany, 67117 Pfizer Investigational Site Ludwigshafen, Germany, 67061 Pfizer Investigational Site Ludwigshafen, Germany, 67069 Pfizer Investigational Site Luebeck, Germany, 23552 Pfizer Investigational Site Luenen, Germany, 44534 Pfizer Investigational Site Mainz, Germany, 55116 Pfizer Investigational Site Marl, Germany, 45768 Pfizer Investigational Site Muenchen, Germany, 81479 Pfizer Investigational Site Muenster, Germany, 48129 Pfizer Investigational Site Osnabrueck, Germany, 49078 Pfizer Investigational Site Recklinghausen, Germany, 45657 Pfizer Investigational Site Senftenberg, Germany, 01968 Pfizer Investigational Site Sinsheim, Germany, 74889 Pfizer Investigational Site Surwold, Germany, 26903 Pfizer Investigational Site Unterhaching, Germany, 82008 Pfizer Investigational Site Veitsbronn, Germany, 90587 Pfizer Investigational Site Weimar, Germany, 99425 Pfizer Investigational Site Wiesbaden, Germany, 65191 Pfizer Investigational Site Witten, Germany, 58452 Pfizer Investigational Site Zwoenitz, Germany, 08297 Collapse << |
| NCT02433132 | Cystic Fibrosis ... more >> Atypical Form of Cystic Fibrosis Collapse << | Not Applicable | Recruiting | September 2018 | France ... more >> Centre Hospitalier Intercommunal de Créteil Recruiting Creteil, France, 94010 Collapse << |
| NCT01893125 | Diabetic Peripheral Neuropathy | Phase 2 | Completed | - | Hungary ... more >> Petz Aladar County Teaching Hospital Gyor, Hungary, H-9002 Collapse << |
| NCT03077893 | Diabetic Peripheral Neuropathy | Not Applicable | Completed | - | United States, Arizona ... more >> Associated Foot & Ankle Specialists, LLC Phoenix, Arizona, United States, 85015 Collapse << |
| NCT01990092 | Diabetic Peripheral Neuropathy... more >> B Vitamin Deficiency Collapse << | Not Applicable | Unknown | March 2018 | - |
| NCT01056315 | - | - | Terminated(Trial was stopped d... more >>ue to difficult enrolment) Collapse << | - | - |
| NCT01056315 | Painful Diabetic Neuropathy | Phase 2 | Terminated(Trial was stopped d... more >>ue to difficult enrolment) Collapse << | - | - |
| NCT01556152 | Painful Diabetic Neuropathy | Phase 2 | Withdrawn(Due to new scientifi... more >>c evidence assessment, the study design was revised before the study started recruitment) Collapse << | - | Germany ... more >> Site 2 Mainz, Germany United Kingdom Site 1 Manchester, United Kingdom Collapse << |
| NCT00603265 | Peripheral Neuropathy ... more >> Neuropathic Pain Collapse << | Phase 2 | Completed | - | - |
| NCT01726413 | Painful Diabetic Peripheral Ne... more >>uropathy Collapse << | Phase 2 | Completed | - | Czech Republic ... more >> NeuroHelp s.r.o Olomouc, Prague, Czech Republic, 772 00 DADO Medical s.r.o Prague, Czech Republic, 12000 DADO Medical s.r.o Ricany, Czech Republic Germany Institute for Clinical Research and Development( IKFE-CRO GmbH BahnhofstraBe 8A) Mainz, Germany, 55116 India Bangalore Clinisearch Bangalore, Karnataka, India, 560043 K.L.E.S Dr. Prabhakar Kore Hospital & Medical research Centre Belgaum, Karnataka, India, 590010 Jnana Sanjeevani Medical Centre Bangalore, Karntaka, India, 560078 TOTALL Diabetes Hormone Institute Indore, Madhya Pradesh, India, 452010 Jehangir Clinical development Centre Pvt Ltd Pune, Maharashtra, India, 411001 Getwell Hospital and Research Centre Nagpur, Maharastra, India, 440012 MV Hospital for Diabetes (P) Ltd Chennai, Tamil Nadu, India, 600 013 Kovai Diabetes Speciality Centre and Hospital Coimbatore, Tamil Nadu, India, 641 009 Arthur Asirvathma Hospital Madhurai, Tamil Nadu, India, 625020 Maulana Azad Medical College & Associate Hospitals New Delhi, India, 110002 United Kingdom ICON Manchester CPU Manchester, UK, United Kingdom, M15 6SH Collapse << |
| NCT01040962 | - | - | Completed | - | United States, Kansas ... more >> University of Kansas Medical Center Kansas City, Kansas, United States, 66160 United States, Missouri North Kansas City Hospital Kansas City, Missouri, United States, 64116 Collapse << |
| NCT00375960 | Pain Diabetic... more >> Neuropathies Collapse << | Phase 2 | Completed | - | United States, California ... more >> Diablo Clinical Research, Inc. Walnut Creek, California, United States, 94598 United States, Florida Clinical Research of West Florida, Inc. Clearwater, Florida, United States, 33765 United States, Massachusetts Translational Pain Research Group, Brigham & Women's Hospital Boston, Massachusetts, United States, 02115 United States, Michigan American Center for Clinical Trials Southfield, Michigan, United States, 48034 United States, Texas Diabetes & Glandular Disease Research Associates Inc. San Antonio, Texas, United States, 78299-4801 Canada, Ontario LMC Endocrinology Centres Ltd. Thornhill, Ontario, Canada, L4J 8L7 Collapse << |
| NCT03769675 | Painful Diabetic Neuropathy | Not Applicable | Recruiting | December 2021 | United States, Minnesota ... more >> Mayo Clinic in Rochester Recruiting Rochester, Minnesota, United States, 55905 Contact: Anita D Stoltenberg, RRT, MHA 507-422-0582 Stoltenberg.Anita@mayo.edu Contact: Lavonne M Liedl, RRT 866-265-9263 Liedl.Lavonne@mayo.edu Principal Investigator: Narayan R Kissoon, M.D. Collapse << |
| NCT01175928 | Diabetic Polyneuropathy | Phase 1 Phase 2 | Unknown | September 2012 | United States, Massachusetts ... more >> NormaTec Newton Center, Massachusetts, United States, 02459 Collapse << |
| NCT00044421 | Diabetic Neuropathies ... more >> Diabetes Mellitus Collapse << | Phase 3 | Completed | - | - |
| NCT00603265 | - | - | Completed | - | - |
| NCT00891683 | Diabetic Peripheral Neuropathy... more >> Chronic Pain Collapse << | Phase 2 | Completed | - | United States, California ... more >> Neurological Research Institute Santa Monica, California, United States, 90404 United States, Ohio Radiant Research Cincinnatti, Ohio, United States, 45249 Wells Institute for Health Awareness Kettering, Ohio, United States, 45429 United States, Pennsylvania Altoona Center for Clinical Research Duncansville, Pennsylvania, United States, 16635 United States, Texas UT Southwestern Medical Center Dallas, Texas, United States, 75390 Bulgaria Multiprofile Hospital for Active Treatment - Internal Department Byala, Bulgaria, 7100 University Multiprofile Hospital for Active Treatment - Clinic of Endocrinology and Metabolic Diseases Pleven, Bulgaria, 5800 University Multiprofile Hospital for Active Treatment - Clinic of Endocrinology and Metabolic Diseases Plovdiv, Bulgaria, 4002 Multiprofile Hospital for Active Treatment - Therapeutical and Endocrinology Department Ruse, Bulgaria, 7002 University Multiprofile Hospital Treatment Stara Zagora Stara Zagora, Bulgaria, 6003 Canada, Quebec Clinique d'Endocrinologie de l'Outaouais Hull, Quebec, Canada, J8V 2P5 Centre de Recherche Clinique de Laval Laval, Quebec, Canada, H7T 2P5 Canada Hopital de l'Enfant Jesus Quebec, Canada, G1J 1Z4 Romania Medical Center "Dr. Negrisanu" SRL Timisoara, Transylvania, Romania, 300456 S.C. Nicodiab SRL Bucharest, Romania, 010496 National Clinical Institute of Diabetes, Nutrition and Metabolic Diseases Bucharest, Romania, 020045 National Institute of Diabetes Nutrition and Metabolic Diseases Bucharest, Romania, 020475 Mosilor Diabetes Mellitus and Obesity Medical Bucharest, Romania, 020859 Emergency Clinical County Hospital Cluj County Cluj Napoca, Romania, 4000006 St. Spiridon Emergency Clinical County Hospital Iasi, Romania, 700111 Collapse << |
| NCT03106376 | Healthy | Not Applicable | Not yet recruiting | December 2020 | United States, Massachusetts ... more >> McLean Hospital Belmont, Massachusetts, United States, 02478 Collapse << |
| NCT01681290 | Diabetic Peripheral Neuropathy | Phase 2 | Completed | - | - |
| NCT01087203 | Diabetic Peripheral Neuropathy | Phase 2 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT03727646 | Heart Failure,Congestive ... more >> Heart Failure New York Heart Association Class IV Mitochondrial Alteration Collapse << | Early Phase 1 | Recruiting | July 26, 2019 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Kevin D O'Brien, MD 206-529-7802 cardiac@uw.edu Contact: Rong Tian, MD 206 616-5672 rongtian@u.washington.edu Collapse << |
| NCT03686423 | - | - | Not yet recruiting | December 2022 | United States, New York ... more >> New York University School of Medicine Not yet recruiting New York, New York, United States, 10016 Contact: Danielle Costanzo 212-263-0380 danielle.costanzo@nyumc.org Principal Investigator: Ryan Brown, PhD Collapse << |
| NCT00712439 | Diabetic Peripheral Neuropathy | Phase 2 | Completed | - | - |
| NCT00870454 | Diabetic Neuropathies | Phase 2 | Completed | - | - |
| NCT02552277 | Diabetic Nephropathies ... more >> Peripheral Nervous System Diseases Collapse << | Phase 2 | Completed | - | United States, California ... more >> Collaborative Neuroscience Network, LLC- Southland Neurologic Associates - Los Alamitos Garden Grove, California, United States, 92845 SDS Clinial Trials, Inc Orange, California, United States, 92868 United States, Florida Compass Research, LLC Orlando, Florida, United States, 32806 Infinity Clinical Research, LLC Sunrise, Florida, United States, 33351 United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 United States, Texas Nerve And Muscle Center Of Texas Houston, Texas, United States, 77099 Endeavor Clinical Trials PA San Antonio, Texas, United States, 78229 Collapse << |
| NCT02689882 | Metabolic Disturbance | Phase 1 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01455415 | - | - | Completed | - | - |
| NCT01496365 | Diabetic Peripheral Neuropathy | Phase 2 | Completed | - | - |
| NCT01247558 | - | - | Completed | - | United States, Delaware ... more >> HealthCore, Inc. Wilmington, Delaware, United States, 19801 Collapse << |
| NCT01455415 | Painful Diabetic Peripheral Ne... more >>uropathy Collapse << | Phase 3 | Completed | - | - |
| NCT03423342 | Heart Failure, Systolic | Phase 1 Phase 2 | Recruiting | June 30, 2019 | United States, Washington ... more >> University of Washington Recruiting Seattle, Washington, United States, 98195 Contact: Kevin D O'Brien, MD 206-529-7802 cardiac@uw.edu Contact: Rong Tian, MD 206 616-5672 rongtian@u.washington.edu Collapse << |
| NCT03415256 | Diabetic Neuropathies | Not Applicable | Recruiting | December 2019 | United States, California ... more >> Loma Linda University Recruiting Loma Linda, California, United States, 92350 Contact: Everett Lohman, DSc 909-558-4300 ext 83171 elohman@llu.edu Collapse << |
| NCT00123136 | Diabetic Neuropathies | Phase 4 | Unknown | - | United States, New York ... more >> Michael I. Weintraub MD 325 S. Highland Avenue Recruiting Briarcliff, New York, United States, 10510 Sub-Investigator: Susan Wolert, Data Collapse << |
| NCT02129231 | Oxidative Stress ... more >> Diabetic Polyneuropathy Collapse << | Phase 2 | Completed | - | Mexico ... more >> Cardiovascular Research Unit Guadalajara, Jalisco, Mexico, 44340 Collapse << |
| NCT01035281 | Diabetic Neuropathies | Phase 3 | Unknown | April 2011 | Canada, Alberta ... more >> Hotchkiss Brain Institute Recruiting Calgary, Alberta, Canada, T2N4N1 Contact: Cory Toth, MD 4032208831 corytoth@shaw.ca Collapse << |
| NCT01002235 | Painful Diabetic Neuropathy | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Diablo Clinical Research Hospital Walnut Creek, California, United States, 94598 United States, Illinois Northwestern Memorial Hospital Chicago, Illinois, United States Collapse << |
| NCT03568968 | Parkinson Disease | Not Applicable | Not yet recruiting | October 1, 2021 | - |
| NCT03482167 | Mild Cognitive Impairment | Phase 1 Phase 2 | Not yet recruiting | March 2022 | United States, Delaware ... more >> Neurovascular Aging Laboratory Not yet recruiting Newark, Delaware, United States, 19713 Contact: Joshua Hobson, MS 302-831-8137 chs-novalab@udel.edu Principal Investigator: Christopher R Martens, Ph.D. Collapse << |
| NCT02063126 | Chronic Fatigue Syndrome | Phase 2 Phase 3 | Completed | - | Spain ... more >> Vall Hebron University Hospital Barcelona, Spain, 08035 Collapse << |
| NCT00835757 | - | - | Completed | - | United States, Michigan ... more >> University of Michigan Ann Arbor, Michigan, United States, 48109 Collapse << |
| NCT02010151 | Out of Hospital Cardiac Arrest | Not Applicable | Recruiting | December 2018 | Korea, Republic of ... more >> Seoul Metropolitan City Recruiting Seoul, Korea, Republic of Sub-Investigator: Kyoung Jun Song, MD Sub-Investigator: Eui Jung Lee, MD Sub-Investigator: Joo Yeong Kim, MD Sub-Investigator: Yu Jin Lee, MD Sub-Investigator: Ju Ok Park, MD Sub-Investigator: Ki Jeong Hong, MD Sub-Investigator: So Young Ha, MD Sub-Investigator: Kwang Soo Bae, MD Sub-Investigator: Jeong Eun Kim, MPH Collapse << |
| NCT01793350 | Diabetic Neuropathy, Painful | Phase 1 Phase 2 | Withdrawn | - | United Kingdom ... more >> Royal Hallamshire Hospital Sheffield, South Yorkshire, United Kingdom, S10 2JF Ipswich Hospital Ipswich, Suffolk, United Kingdom, IP4 5PD Birmingham Heartlands Hospital Birmingham, West Midlands, United Kingdom, B9 5SS MAC Clinical Research Manchester, United Kingdom, M32 0UT Manchester Royal Infirmary Manchester, United Kingdom Collapse << |
| NCT03331614 | Painful Diabetic Neuropathy | Not Applicable | Recruiting | April 30, 2018 | United States, New Jersey ... more >> Sean D Rosenblum DPM Recruiting Lodi, New Jersey, United States, 07055 Contact: Jonathan Rosenblum, DPM 720-744-3222 diabfootman@gmail.com Contact: Evan Renov clinicaltrials@flowaid.com Principal Investigator: Sean Rosenblum, DPM Collapse << |
| NCT00407511 | Diabetic Peripheral Neuropathi... more >>c Pain (DPN) Postherpetic Neuralgia (PHN) HIV-related Neuropathic Pain (HIV) Chemotherapy Induced Neuropathic Pain Collapse << | Phase 4 | Completed | - | Colombia ... more >> Pfizer Investigational Site Bogota, Cundinamarca, Colombia Ecuador Pfizer Investigational Site Quito, Pichincha, Ecuador Mexico Pfizer Investigational Site Tijuana, B. C., Mexico, 22010 Pfizer Investigational Site Mexicali, B.c., Mexico, 21100 Pfizer Investigational Site Mexico, Distrito Federal, Mexico, 14080 Pfizer Investigational Site Acapulco, Guerrero, Mexico, 39670 Pfizer Investigational Site México, Monterrey, NL, Mexico, 64460 Pfizer Investigational Site Merida, Yucatan, Mexico, 97000 Peru Pfizer Investigational Site Lima, Peru, 27 Pfizer Investigational Site Lima, Peru, L13 Venezuela Pfizer Investigational Site Caracas, Distrito Capital, Venezuela, 1020 Collapse << |
| NCT02115932 | Diabetic Neuropathies ... more >> Peripheral Nervous System Diseases Collapse << | Not Applicable | Completed | - | Singapore ... more >> National University of Singapore; National University Hospital Singapore, Singapore Collapse << |
| NCT03450200 | Diabetic Peripheral Neuropathy | Not Applicable | Completed | - | Japan ... more >> Okayama University Okayama, Japan, 7008558 Collapse << |
| NCT01041859 | - | - | Completed | - | - |
| NCT01041859 | Diabetic Peripheral Neuropathy | Phase 3 | Completed | - | - |
| NCT00134524 | Diabetic Neuropathies ... more >> Pain Neuralgia Collapse << | Phase 3 | Unknown | January 2008 | United States, Arizona ... more >> AMRI Arizona Tucson, Arizona, United States, 85711 United States, Michigan AMRI MI Sterling Heights, Michigan, United States, 48313 United States, North Carolina AMRI NC Mocksville, North Carolina, United States, 27208 United States, Ohio Amri NW Oh Toledo, Ohio, United States, 48313 United States, Washington AMRI WA Renton, Washington, United States, 98055 Collapse << |
| NCT03224598 | Dermatosis Papulosa Nigra | Phase 2 | Active, not recruiting | December 2018 | United States, District of Col... more >>umbia Aclaris Investigational Site Washington, District of Columbia, United States, 20037 United States, New York Aclaris Investigational Site New York, New York, United States, 10155 Collapse << |
| NCT03151707 | Healthy | Phase 4 | Recruiting | July 2019 | United States, Massachusetts ... more >> Cagri Yuksel Recruiting Belmont, Massachusetts, United States, 02478 Contact: Cagri Yuksel, MD 617-855-2779 ayuksel@partners.org Sub-Investigator: Cagri Yuksel, MD Principal Investigator: Dost Ongur, MD, PhD Collapse << |
| NCT00476151 | - | - | Completed | - | - |
| NCT00407511 | - | - | Completed | - | - |
| NCT03767478 | Diabetic Neuropathies | Not Applicable | Recruiting | December 31, 2020 | United Kingdom ... more >> Imperial College London Recruiting London, United Kingdom, W6 8RF Contact: Tristan R Lane, PhD FRCS tristan.lane@imperial.ac.uk Collapse << |
| NCT00476151 | Diabetic Peripheral Neuropathy... more >> Neuralgia Collapse << | Phase 2 | Completed | - | India ... more >> Multiple Centers New Delhi, India Collapse << |
| NCT02950441 | Aging | Phase 2 | Recruiting | September 2019 | United Kingdom ... more >> University Hospitals Birmingham NHS Foundation Trust Recruiting Birmingham, West Midlands, United Kingdom, B15 2TH Contact: Yasir Elhassan, MRCP +441214158705 y.mohamedelhassan@bham.ac.uk Collapse << |
| NCT02721537 | Concussion, Mild | Not Applicable | Recruiting | June 2019 | United States, Minnesota ... more >> University of Minnesota Center for Magnetic Resonance Research Recruiting Minneapolis, Minnesota, United States, 55455 Contact: Stacy Valenzuela, RAC 612-624-9926 majes014@umn.edu Principal Investigator: Xiao-Hong Zhu, Ph.D. Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Heather Ondler Hinson 507-284-4799 ondlerhinson.heather@mayo.edu Contact: Chuck Barta 507-266-5566 barta.charles@mayo.edu Principal Investigator: Brent A Bauer, MD Collapse << |
| NCT02215252 | Diabetic Neuropathy, Painful | Phase 2 | Completed | - | - |
| NCT00614341 | Diabetic Peripheral Neuropathy | Phase 4 | Completed | - | United States, Georgia ... more >> Nephrology and Rheumatology Associates Augusta, Georgia, United States, 30909 Collapse << |
| NCT00487981 | - | - | Terminated(Insufficient Data C... more >>ollected) Collapse << | - | - |
| NCT00156078 | Diabetic Neuropathy, Painful | Phase 4 | Completed | - | - |
| NCT02215252 | - | - | Completed | - | - |
| NCT01063868 | Diabetic Neuropathy, Painful ... more >> Diabetic Polyneuropathy Collapse << | Phase 3 | Terminated(Business decision) | - | - |
| NCT00487981 | Diabetic Neuropathy ... more >> Pain Peripheral Neuropathy Collapse << | Phase 4 | Terminated(Insufficient Data C... more >>ollected) Collapse << | - | United States, Indiana ... more >> Deaconess Pain Management Center Evansville, Indiana, United States, 47747 Collapse << |
| NCT00643760 | Neuropathy, Diabetic | Phase 2 | Completed | - | - |
| NCT00643760 | - | - | Completed | - | - |
| NCT03219320 | Diabetic Peripheral Neuropathy | Phase 2 | Recruiting | December 30, 2018 | - |
| NCT00710203 | Facial Dermatoses ... more >> Seborrheic Keratoses Collapse << | Not Applicable | Completed | - | United States, California ... more >> University of California, Davis Department of Dermatology Sacramento, California, United States, 95816 Collapse << |
| NCT03707652 | Increase in Blood Levels of Ni... more >>cotinamide Adenine Dinucleotide (NAD+) Collapse << | Not Applicable | Completed | - | United States, Florida ... more >> Life Extension Clinical Research, Inc. Fort Lauderdale, Florida, United States, 33308 Collapse << |
| NCT01063868 | - | - | Terminated(Business decision) | - | - |
| NCT01474772 | Diabetic Peripheral Neuropathy | Phase 3 | Completed | - | - |
| NCT00933998 | Diabetic Peripheral Neuropathy | Not Applicable | Completed | - | United States, South Carolina ... more >> Carolina Musculoskeletal Institute Aiken, South Carolina, United States, 29841 Collapse << |
| NCT00710203 | - | - | Completed | - | - |
| NCT01474772 | - | - | Completed | - | - |
| NCT03186027 | Chronic Fatigue Syndrome | Not Applicable | Recruiting | December 31, 2018 | Spain ... more >> Vall d'Hebron University Hospital Recruiting Barcelona, Spain, 08035 Contact: Jose Alegre, MD; PhD +34 934893000 ext 4927 jalegre@vhebron.net Contact: Jesus Castro, PhD +34 934893000 ext 4927 jesus.castro@vhir.org Collapse << |
| NCT00726713 | Type 2 Diabetic Peripheral Neu... more >>ropathy (DPN) Collapse << | Phase 4 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham School of Medicine Birmingham, Alabama, United States, 35294 United States, Louisiana Tulane University Health Sciences Center New Orleans, Louisiana, United States, 70112 United States, Nebraska Omaha VA Medical Center Omaha, Nebraska, United States, 68105 United States, Texas Dallas Diabetes and Endocrine Center Dallas, Texas, United States, 75230 dgd Research, Inc. San Antonio, Texas, United States, 78229 Scott and White Hospital & Clinic Temple, Texas, United States, 76504 Collapse << |
| NCT00501202 | Diabetic Neuropathy, Painful ... more >> Diabetic Neuralgia Diabetic Polyneuropathy Diabetic Mononeuropathy Collapse << | Phase 2 | Completed | - | - |
| NCT02541838 | Diabetes Complications ... more >> Diabetic Neuropathies Collapse << | Not Applicable | Completed | - | United States, Maryland ... more >> PTRS Research Lab Baltimore, Maryland, United States, 21201 VA Maryland Health Care System Baltimore, Maryland, United States, 21201 Collapse << |
| NCT00726713 | - | - | Completed | - | - |
| NCT02332005 | Diabetic Peripheral Neuropathy | Phase 2 Phase 3 | Completed | - | Russian Federation ... more >> City Hospital No 40 of the Kurortny District St Petersburg, Sestroretsk, Russian Federation, 197706 Northern State Medical University Arkhangelsk, Russian Federation, 163000 Health Services Severstal Cherepovets, Russian Federation, 162600 Clinic of Neurology Ekaterinburg, Russian Federation, 620014 Kemerovo Regional Clinical Hospital Kemerovo, Russian Federation, 650066 Endocrinology Dispensary Moscow, Russian Federation, 119034 Morozovskaya Children City Hospital of Moscow Moscow, Russian Federation, 119049 I M Sechenov First Moscow State Medical University Moscow, Russian Federation, 119435 IM Sechenov First Moscow State Medical University Moscow, Russian Federation, 119435 IM Sechenov First Moscow State Medical University Moscow, Russian Federation, 119991 City Clinical Hospital No 71 Moscow, Russian Federation, 121374 Central Clinical Hospital No 1 of JSC Russian Railway Moscow, Russian Federation, 125367 City Clinical Hospital No 50 Moscow, Russian Federation, 127206 The Federal Bureau of Medical and Social Expertise Moscow, Russian Federation, 127486 Perm State Medical Academy Perm, Russian Federation, 614107 VA Baranov Respublical Hospital Petrozavodsk, Russian Federation, 185019 Rostov State Medical University Rostov-on-Don, Russian Federation, 344022 City Polyclinic No 20 Saratov, Russian Federation, 410053 Imc Sogaz St Petersburg, Russian Federation, 191186 Medical Center Reavita St Petersburg, Russian Federation, 194295 City Hospital of the Holy Martyr Elizabeth St Petersburg, Russian Federation, 195257 Nikolaev Hospital St Petersburg, Russian Federation, 198510 Bashkir State Medical University Ufa, Russian Federation, 450000 Central City Clinical Hospital Ulyanovsk, Russian Federation, 432057 State Medical University Volgograd, Russian Federation, 400131 Regional Clinical Hospital Yaroslavl, Russian Federation, 150000 NV Solovyov Clinical Emergency Hospital Yaroslavl, Russian Federation, 150003 Collapse << |
| NCT02606747 | - | - | Unknown | June 2016 | China, Beijing ... more >> Peking University First Hospital Recruiting Beijing, Beijing, China, 100000 Contact: Geheng Yuan, M.D +8613811235488 139197109@qq.com Collapse << |
| NCT03310034 | Aging Mitocho... more >>ndrial Function Collapse << | Not Applicable | Recruiting | December 2019 | Netherlands ... more >> Maastricht University Recruiting Maastricht, Limburg, Netherlands, 6229ER Contact: Niels Connell, MSc +31433881587 n.connell@maastrichtuniversity.nl Collapse << |
| NCT02942888 | Mild Cognitive Impairment ... more >> NAD Collapse << | Not Applicable | Recruiting | December 2018 | United States, Texas ... more >> South Texas Veterans Healthcare System (STVHCS) Recruiting San Antonio, Texas, United States, 78229 Contact: Joan Hecht 210-617-5300 ext 14654 Joan.hecht@va.gov University of Texas Health San Antonio Recruiting San Antonio, Texas, United States, 78229 Contact: Becky Powers, MD 210-617-5179 Becky.powers@va.gov Collapse << |
| NCT03069729 | - | - | Recruiting | October 1, 2019 | United States, New Mexico ... more >> VisionQuest Biomedical LLC Recruiting Albuquerque, New Mexico, United States, 87106 Contact: Maria Vahtel, MA 505-508-1994 ext 1015 mvahtel@visionquest-bio.com Contact: Niranchana Manivannan, PhD 505-505-1994 ext 1019 NManivannan@visionquest-bio.com Principal Investigator: Peter Soliz, PhD Principal Investigator: Mark Burge, MD Collapse << |
| NCT02429986 | Chronic Heart Failure and ... more >> Complex Sleep Apnea Syndrome Obstructive Sleep Apnea Syndrome and Idiopathic Central Sleep Apnea Syndrome Idiopathic Induced Periodic Breathing Central Sleep Apnea Syndrome Collapse << | Not Applicable | Completed | - | France ... more >> Polyclinic Saint-Privat Boujan sur Libron, France, 34480 CHU Dijon Dijon, France, 21079 Hôpital Nord Marseille, France, 13015 Arnaud de Villeneuve University Hospital Montpellier, France, 34295 Hôpital Pitié Salpetrière Paris, France, 75651 Collapse << |
| NCT02495727 | Muscular Atrophy | Not Applicable | Completed | - | United Kingdom ... more >> University of Bath Bath, Avon, United Kingdom, BA2 7AY Collapse << |
| NCT02137746 | Prostate Cancer | Phase 2 | Completed | - | Czechia ... more >> Hradec Kralove, Czechia Jablonec nad Nisou, Czechia Novy Jicin, Czechia Olomouc, Czechia Prague 4, Czechia Prague 5, Czechia Uherske Hradiste, Czechia Usti nad Labem, Czechia Collapse << |
| NCT02107391 | Prostate Cancer | Phase 2 | Completed | - | Czechia ... more >> Jablonec nad Nisou, Czechia Jihlava, Czechia Liberec, Czechia Mnisek pod Brdy, Czechia Olomouc, Czechia Plzen, Czechia Praha 10, Czechia Praha 4, Czechia Praha 5, Czechia Praha 8, Czechia Usti nad Labem, Czechia Zlin, Czechia Collapse << |
| NCT00937521 | Meningococcal Meningitis ... more >> Meningococcal Infections Collapse << | Phase 2 | Completed | - | Argentina ... more >> Hospital Privado de Córdoba CMC SA Naciones Unidas 346, Cordoba, Argentina, X5016KHE Chile Universidad de Chile, Av Independencia 1027 Comuna de Independencia, Santiago, Chile Consultorio Manuel Bustos Lo Cruzat 486, Quilicura, Santiago, Chile Czech Republic Samostatna ordinace praktickeho lekare pro deti a dorost O. Kubina 17, Boskovice, Czech Republic, 680 01 Samostatna ordinace praktickeho lekare pro deti a dorost Neklez 3, Brno, Czech Republic, 628 00 Samostatna ordinace praktickeho lekare pro deti a dorost Pernštýnská 127/l, Chlumec nad Cidlinou, Czech Republic, 503 51 Zdravotní středisko Vaclavska 4186, Chomutov, Czech Republic, 430 03 Nemocnice Decin, Detske oddělení U nemocnice 1, Děčín, Czech Republic, 405 01 Fakulta vojenskeho zdravotnictvi UO Trebešská 1575, Hradec Králové, Czech Republic, 50001 Samostatna ordinace praktickeho lekare pro deti a dorost Masarykova 389, Humpolec, Czech Republic, 396 01 Samostatna ordinace praktickeho lekare pro deti a dorost Ruských legii 352, Jindřichův Hradec, Czech Republic, 377 01 Samostatna ordinace praktickeho lekare pro deti a dorost Hrnčířská 1401, Lipník nad Bečvou, Czech Republic, 751 31 Oblastni nemocnice Nachod, Destske oddělení Purkyňova 446, Náchod, Czech Republic, 547 01 Samostatna ordinace praktickeho lekare pro deti a dorost U lékárny 306, Odolena Voda, Czech Republic, 250 70 Prakticky lekar pro deti a dorost Dvouletky 54, Ostrava, Czech Republic, 700 30 KHS Ostrava, Protiepidemické oddělení Na Bělidle 7, Ostrava, Czech Republic, 702 00 Nemocnice Pardubice, Destske odděleni Kyjevská 44, Pardubice, Czech Republic, 532 03 Fakultni nemocnice Bory E. Beneše 13, Plzeň, Czech Republic, 305 99 Samostatna ordinace praktickeho lekare pro deti a dorost Chrudimska 2a, Praha 3, Czech Republic, 130 00 Samostatna ordinace praktickeho lekare pro deti a dorost Kladenská 53, Praha 6, Czech Republic, 160 00 Samostatna ordinace praktickeho lekare pro deti a dorost Velka Michalska 22, Znojmo, Czech Republic, 669 00 Hungary Házi Gyermekorvosi szolgálat Honvéd u.2., Bordány, Hungary, 6795 Medszolg 2000 Bt, 6723, Szeged, Dandár u.4 Ányos u.4., Budapest, Hungary, 1031 Erzsébet Kórház Gyermekosztály Hodmezovasarhely, dr. Imre József u.2., Hungary Baby Box Bt,, 6724, Szeged, Kossuth Lajos sgt.109 Szeged, Kossuth Lajos sgt.109, Hungary Dr. Bán Mariann és Társa Bt. Kando Kalman u.1, Miskolc, Hungary, 3534 Futurnest Kft Selyemrét u.1., Miskolc, Hungary, 3527 Ped-Med Kft. , 3434 Mályi, Fő u.12. Fő u.12., Mályi, Hungary, 3434 S.K. Sipka és Kovács Eü. Bt. Csongrádi sgt. 63., Szeged, Hungary, 6723 Oszila Kft. 6723, Szeged, Debreceni u.10-14. Debreceni u.10-14., Szeged, Hungary, 6723 Győriné dr. Bari Eszter egyéni vállalkozó Csongrad, Szentháromság tér 10, Hungary Vas Megyei Markusovszky Kórház, Gyermekosztály Markusovszky u. 1-3, Szombathely, Hungary, 9700 Italy Dipartimento di Neonatologia e Terapia Intensiva Neonatale, "Ospedale dei Bambini", Presidio Ospedaliero dell'Azienda Ospedaliera Spedali Civili di Brescia P.le Spedali di Brescia,1, Brescia, Italy, 25125 Dipartimento di Pediatria dell'Università degli Studi di Firenze Viale Pieraccini n. 24, Firenze, Italy, 50139 Università degli Studi di Messina, Pad. NI - A.O.U. Policlinico G.Martino Via Consolare Valeria, 1, Messina, Italy, 98125 Fondazione IRCCS dell'Ospedale Maggiore Policlinico, Mangiagalli e Regina Elena di Milano Via Commenda, 9, Milano, Italy, 20122 Pediatria dell'Ospedale Sacco di Milano Via G.B.Grossi 74, Milano, Italy, 20157 Dipartimento di Pediatria Azienda Ospedaliera di Padova Via Giustiniani, 3, Padova, Italy, 35128 Collapse << |
| NCT00937521 | - | - | Completed | - | - |
| NCT00226499 | Varicella | Phase 3 | Completed | - | - |
| NCT01335971 | COPD | Phase 2 | Completed | - | United States, Maryland ... more >> Johns Hopkins School of Medicine Baltimore, Maryland, United States, 21224 United States, New York University at Baffalo, The State University of New York Buffalo, New York, United States, 14215 United States, Pennsylvania Temple University Philadelphia, Pennsylvania, United States, 19122 Collapse << |
| NCT01335971 | - | - | Completed | - | - |
| NCT00484198 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00922987 | - | - | Completed | - | - |
| NCT00922987 | - | - | Completed | - | - |
| NCT01021332 | Lower Urinary Tract Symptoms ... more >> Benign Prostatic Hyperplasia Collapse << | Phase 3 | Completed | - | - |
| NCT01021332 | - | - | Completed | - | - |
| NCT01018511 | Benign Prostatic Hyperplasia ... more >> Lower Urinary Tract Symptoms Collapse << | Phase 3 | Completed | - | - |
| NCT02087098 | - | - | Completed | - | - |
| NCT01920711 | Heart Failure With Preserved E... more >>jection Fraction Collapse << | Phase 3 | Active, not recruiting | May 31, 2019 | - |
| NCT02993406 | Cardiovascular Diseases ... more >> Statin Adverse Reaction Collapse << | Phase 3 | Recruiting | March 2022 | - |
| NCT02250573 | Haemophilia B | Phase 3 | Completed | - | Poland ... more >> Samodzielny Publiczny Zaklad Opieki Zdrowotnej nad Matka I Dzieckiem, ul. Krysiewicza 7/8. Poznan, Poland, 61-825 Romania Institutul National de Haemtologie, 2-8 Constantin Caracas Str. Bucharest, Romania Spitalul de urgenta pentru copii "Louis Turcana", Str. losef Nemoianu 2. Timisoara, Romania Russian Federation Kirov Research Institute of Haematology, 72 Krasnoarmeyskaya ul. Kirov, Russian Federation, 610027 Haematology Centre, Russian Academy if Medical Sciences, 4a Novozykovsky Proezed. Moscow, Russian Federation, 125167 Collapse << |
| NCT03105687 | Hypertension ... more >>Primary Hypertension Non-Adherence, Medication Non-Adherence, Patient Collapse << | Not Applicable | Completed | - | Slovakia ... more >> Lekáreň Needham Banská Bystrica, Slovakia Lekáreň V KAUFLANDE BB, s.r.o. Banská Bystrica, Slovakia Lekáreň DANUBIA Bratislava, Slovakia Lekáreň Dr.Max 12 Bratislava, Slovakia Lekáreň Poliklinika Šustekova Bratislava, Slovakia Lekáreň GREEN-STRAP Dubnica nad Váhom, Slovakia Lekáreň Námestie Matice Slovenskej, Dr. Max Dubnica nad Váhom, Slovakia Lekáreň Dr.Max 90, Nemocnica s poliklinkou Sv.Lukáša Galanta, Slovakia Lekáreň REDMOON Hnúšťa, Slovakia Lekáreň 17 Kežmarok, Slovakia Lekáreň Námestie Osloboditeľov, Dr. Max Liptovský Mikuláš, Slovakia Lekáreň RED-MARKET s.r.o. Malacky, Slovakia Lekáreň PRED NEMOCNICOU Martin, Slovakia Lekáreň, OC Tulip Martin, Slovakia Lekáreň Námestie Slobody, Dr. Max Piešťany, Slovakia Lekáreň HEALTHSTORE Prešov, Slovakia Lekáreň Dr.Max 22 Rimavská Sobota, Slovakia Lekáreň Dr.Max 36 Rožňava, Slovakia Lekáreň OC Madaras Dr. Max Spišská Nová Ves, Slovakia Lekáreň EURO FARMÁCIA s.r.o. Trenčín, Slovakia Lekáreň SD5 s.r.o. Trenčín, Slovakia Lekáreň CASTILION Vranov nad Topľou, Slovakia Železničná Lekáreň Zvolen, Slovakia Lekáreň Bernolákova, Dr. Max Žilina, Slovakia Collapse << |
| NCT03663569 | - | - | Recruiting | May 28, 2019 | - |
| NCT01006135 | - | - | Completed | - | - |
| NCT01018511 | - | - | Completed | - | - |
| NCT02174627 | Anemia | Phase 3 | Completed | - | - |
| NCT00119743 | Hepatitis A | Phase 3 | Terminated | - | Czech Republic ... more >> GSK Investigational Site Brno, Czech Republic, 628 00 GSK Investigational Site Ceske Budejovice, Czech Republic, 370 71 GSK Investigational Site Decin, Czech Republic, 405 01 GSK Investigational Site Frydek-Mistek, Czech Republic, 738 02 GSK Investigational Site Havlickuv Brod, Czech Republic, 580 22 GSK Investigational Site Hlinsko v Cechach, Czech Republic, 539 01 GSK Investigational Site Jicin, Czech Republic, 506 01 GSK Investigational Site Jindrichuv Hradec, Czech Republic, 377 01 GSK Investigational Site Karvina, Czech Republic, 734 01 GSK Investigational Site Litomerice, Czech Republic, 412 01 GSK Investigational Site Nachod, Czech Republic, 547 01 GSK Investigational Site Ostrava, Czech Republic, 728 92 GSK Investigational Site Pardubice, Czech Republic, 532 03 GSK Investigational Site Praha 2, Czech Republic, 120 00 GSK Investigational Site Praha 4, Czech Republic, 140 00 GSK Investigational Site Praha 5, Czech Republic, 150 06 GSK Investigational Site Praha 6, Czech Republic, 160 00 GSK Investigational Site Praha 8, Czech Republic, 180 00 GSK Investigational Site Praha 9, Czech Republic, 190 00 GSK Investigational Site Usti nad Labem, Czech Republic, 400 01 GSK Investigational Site Usti nad Labem, Czech Republic, 400 78 GSK Investigational Site Znojmo, Czech Republic, 669 00 Slovakia GSK Investigational Site Dolny Kubin, Slovakia, 026 01 GSK Investigational Site Dubnica Nad Vahom, Slovakia, 018 41 GSK Investigational Site Kostany Nad Turcom, Slovakia, 038 41 GSK Investigational Site Liptovsky Hradok, Slovakia, 033 01 GSK Investigational Site Liptovsky Mikulas, Slovakia, 031 01 GSK Investigational Site Martin, Slovakia, 036 01 GSK Investigational Site Namestovo, Slovakia, 029 01 GSK Investigational Site Nitra, Slovakia, 949 11 GSK Investigational Site Nova Dubnica, Slovakia, 018 51 GSK Investigational Site Nove Mesto nad Vahom, Slovakia, 915 01 GSK Investigational Site Nove Zamky, Slovakia, 940 01 GSK Investigational Site Povazska Bystrica, Slovakia, 017 01 GSK Investigational Site Puchov, Slovakia, 020 01 GSK Investigational Site Ruzomberok, Slovakia, 034 01 GSK Investigational Site Sturovo, Slovakia, 943 01 GSK Investigational Site Sucany, Slovakia, 038 42 GSK Investigational Site Surany, Slovakia, 942 18 GSK Investigational Site Trencin, Slovakia, 911 01 GSK Investigational Site Zlate Moravce, Slovakia, 953 01 Collapse << |
| NCT01035255 | Heart Failure With Reduced Eje... more >>ction Fraction Collapse << | Phase 3 | Terminated(Early termination w... more >>as approved due to compelling efficacy of LCZ696 in patients with HF & reduced EF after final pre-specified interim analysis 28-Mar-2014.) Collapse << | - | - |
| NCT01327001 | - | - | Completed | - | - |
| NCT01006135 | - | - | Completed | - | - |
| NCT01764633 | - | - | Completed | - | - |
| NCT01035255 | - | - | Terminated(Early termination w... more >>as approved due to compelling efficacy of LCZ696 in patients with HF & reduced EF after final pre-specified interim analysis 28-Mar-2014.) Collapse << | - | - |
| NCT01130883 | - | - | Completed | - | - |
| NCT01991795 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | December 28, 2018 | - |
| NCT01764633 | Dyslipidemia | Phase 3 | Completed | - | - |
| NCT01108796 | - | - | Completed | - | - |
| NCT01130883 | - | - | Completed | - | - |
| NCT01108796 | - | - | Completed | - | - |
| NCT02678611 | Safety: Healthy Subjects | Phase 1 | Completed | - | Canada, Ontario ... more >> KGK Synergize Inc. London, Ontario, Canada, N6A 5R8 Collapse << |
| NCT00619177 | - | - | Completed | - | - |
| NCT01536236 | Diabetic Polyneuropathy | Not Applicable | Withdrawn | October 2014 | United States, Missouri ... more >> Midwest Neurosurgery Associates Kansas City, Missouri, United States, 64132 Collapse << |
| NCT01939158 | Infections, Meningococcal | Phase 3 | Active, not recruiting | February 12, 2020 | - |
| NCT01108185 | - | - | Completed | - | - |
| NCT01055977 | - | - | Completed | - | - |
| NCT00619177 | - | - | Completed | - | - |
| NCT01079195 | - | - | Completed | - | - |
| NCT01108185 | - | - | Completed | - | - |
| NCT02433366 | - | - | Completed | - | - |
| NCT01327846 | Atherosclerosis | Phase 3 | Active, not recruiting | April 1, 2020 | - |
| NCT02433366 | - | - | Completed | - | - |
| NCT01079195 | - | - | Completed | - | - |
| NCT01127139 | - | - | Completed | - | - |
| NCT00792909 | Infections, Streptococcal | Phase 3 | Completed | - | Slovakia ... more >> GSK Investigational Site Dlha nad Oravou, Slovakia, 027 55 GSK Investigational Site Dolny Kubin, Slovakia, 026 01 GSK Investigational Site Ruzomberok, Slovakia, 034 01 Sweden GSK Investigational Site Umeå, Sweden, SE-901 85 GSK Investigational Site Örebro, Sweden, SE-701 16 Collapse << |
| NCT01127139 | - | - | Completed | - | - |
| NCT02402309 | Sjögren-Larsson Syndrome | Phase 2 | Completed | - | United States, Nebraska ... more >> University of Nebraska Medical Center Omaha, Nebraska, United States, 68198 United States, Pennsylvania Penn State Hershey Medical Center - Department of Dermatology Hershey, Pennsylvania, United States, 17033 United States, Virginia Pediatric and General Dermatology Fairfax, Virginia, United States, 22031 Collapse << |
| NCT00915486 | Diabetic Foot Ulcer | Phase 2 | Completed | - | Czech Republic ... more >> Diabetologické centrum, I. Interní klinika FN Hradec Králové, Fakultní nemocnice Hradec Králové (21) Hradec Králové, Czech Republic, 500 05 Interní klinika 2. Lékařské fakulty UK a FN Motol, Fakultní nemocnice Motol (22) Praha, Czech Republic, 150 06 Lékařský dum Ormiga Angiologická a diabetologická ambulance (20) Zlín, Czech Republic, 760 01 Oddelení diabetologie - Podiatricka ambulance Krajská zdravotní a.s.- Masarykova nemocnice v Ústí nad Labem o.z. (23) Ústí nad Labem, Czech Republic, 401 13 Germany Klinikum Sindelfingen-Böblingen (01) Böblingen, Germany, 71031 SRH Klinikum Karlsbad-Langensteinbach (04) Karlsbad, Germany, 76307 Klinikum Stuttgart Bürgerhospital (03) Stuttgart, Germany, 70191 Universitätsklinik Tübingen Chirugische Poliklinik (02) Tübingen, Germany, 72076 Hungary Budai Irgalmasrendi Közhasznú Non-Profit Kft (35) Budapest, Hungary, 1027 Fõvárosi Önkormányzat Egyesített Szent István és Szent László Kórház - Rendelõintézet Sebészeti Osztály (32) Budapest, Hungary, 1097 Fővárosi Önkormányzat Szent Imre Kórház Operativ Szakmák Mátrix Szervezete Általános Sebészeti Profil (33) Budapest, Hungary, 1115 Esztergom Város Önkormányzat Vaszary Kolos Kórház Sebészeti Osztály (34) Esztergom, Hungary, 2500 Kaposi Mór Oktató Kórház (38) Kaposvar, Hungary, 7400 Bács-Kiskun Megyei Önkormányzat Kórháza Szegedi Tudományegyetem Általános Orvostudományi Kar Oktató Kórháza, 2. Belgyógyászat (31) Kecskemét, Hungary, 6000 Borsod-Abaúj-Zemplén Megyei Kórház és Egyetemi Oktató Kórház Érsebészeti Osztály (30) Miskolc, Hungary, 3526 PTE Klinikai Központ Érsebészeti Klinikai Tanszék (36) Pécs, Hungary, 7623 SZTE Szent-Györgyi Albert Klinikai Centrum (37) Szeged, Hungary, 6720 Romania Institutul Naţional de Diabet, Nutriţie şi Boli Metabolice "Prof. Dr. N. C. Paulescu" (42) Bucharest, Romania, 020475 Spitalul Clinic Judeţean de Urgenţă Cluj (43) Cluj-Napoca, Romania, 400006 Spitalul Clinic Judeţean Mureş, Clinica de Dermatologie (40) Tg Mures, Romania, 540342 Cabinet Medical Individual DermaMed (41) Tg. Mures,, Romania, 540530 Spitalul Clinic Judetean de Urgenta Timisoara (45) Timisoara, Romania, 300723 Russian Federation Moscow City Clinical Hospital # 13 (11) Moscow, Russian Federation, 115280 Moscow State University of Public Health "City Clinical (17) Moscow, Russian Federation, 115432 Department of Endocrinology and Diabetes, Russian State Medical University, Moscow City Clinical Hospital # 1 (15) Moscow, Russian Federation, 117049 Department of Diabetic Foot Endocrinology dispensary (12) Moscow, Russian Federation, 119034 Endocrinology Clinic of the State Educational Institute of High Professional Education (18) Moscow, Russian Federation, 119992 Federal State Institution "Federal Bureau of Medical Social Expertise" (10) Moscow, Russian Federation, 123448 Moscow Clinical Hospital # 81 (14) Moscow, Russian Federation, 127644 Serbia Klinički centar Srbije, Institut za endokrinologiju, dijabetes i bolesti metabolizma (51) Belgrade, Serbia, 11 000 Clinical Centre Kragujevac (54) Kragujevac, Serbia, 34 000 Clinical Centre Nis (50) Nis, Serbia, 18000 Clinical Centre of Vojvodina (53) Novi Sad, Serbia, 21 000 Health Centre Valjevo (52) Valjevo, Serbia, 14 000 Collapse << |
| NCT02973581 | Vitreoretinal Surgeries ... more >> Postoperative Nausea and Vomiting Postoperative Pain Collapse << | Not Applicable | Completed | - | Poland ... more >> Medical University of Silesia Sosnowiec, Silesia, Poland, 41-200 Collapse << |
| NCT00375674 | Kidney Neoplasms | Phase 3 | Completed | - | - |
| NCT01582347 | Opioid Related Disorder | Phase 2 | Completed | - | Austria ... more >> Prof. Dr. Fleischhacker Austria, Austria, 6020 Dr. Lindenbauer Linz, Austria, 4020 Prof. Dr. Wurst Salzburg, Austria, 5020 Prof. Wolzt Wien, Austria, 1090 Czech Republic Dr. Vehak Prague, Czech Republic, 1400 Dr. Stankova Usti nad Labem, Czech Republic, 40113 Germany Dr. Tietje Bremen, Germany, 28719 Prof. Scherbaum Essen, Germany, 45147 Dr. Weber Kassel, Germany, 34117 PD. Dr. Pogarell Munich, Germany, 80336 Dr. Rechenmacher Oldenburg, Germany, 26121 Dr. Boniakowski Regensburg, Germany, 93051 Dr. Issler Stuttgart, Germany, 70197 Sweden Dr. Kilaidakis Orebro, Sweden, 70185 Dr. Georgieva Stockholm, Sweden, 17176 Collapse << |
| NCT00679380 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT01116674 | - | - | Completed | - | United States, Georgia ... more >> Children's Healthcare of Atlanta at Egleston - Pediatric Cardiac Intensive Care Unit Atlanta, Georgia, United States, 30308 Children's Healthcare of Atlanta at Egleston - Pediatric Intensive Care Unit Atlanta, Georgia, United States, 30308 Medical Center of Central Georgia - Pediatric Intensive Care Unit Macon, Georgia, United States, 31201 Collapse << |
| NCT00835978 | - | - | Completed | - | - |
| NCT00910091 | - | - | Completed | - | - |
| NCT00792909 | - | - | Completed | - | - |
| NCT00679380 | - | - | Completed | - | - |
| NCT00920582 | Type 1 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00910091 | Endometrial Cancer | Phase 2 | Completed | - | - |
| NCT00835978 | Carcinoma, Renal Cell | Phase 2 | Completed | - | - |
| NCT00375674 | - | - | Completed | - | - |
| NCT01352793 | Meningitis, Meningococcal | Phase 3 | Completed | - | - |
| NCT00988208 | - | - | Completed | - | - |
| NCT01302054 | Urinary Bladder, Overactive | Phase 4 | Completed | - | - |
| NCT01302054 | - | - | Completed | - | - |
| NCT01829321 | Ulcerative Colitis | Phase 2 | Completed | - | Belgium ... more >> Imelda Bonheiden, Belgium UZ Gent Gent, Belgium AZ Groeninge Kortrijk, Belgium UZ Leuven Leuven, Belgium Czech Republic Hepato-Gastroenterologie HK, s.r.o. Hradec Kralove, Czech Republic Fakulní nemocnice Olomouc Olomouc, Czech Republic Homolka Hospitál Prague, Czech Republic Nemocnice Slaný Slany, Czech Republic Krajská zdravotní a.s., Masarykova nemocnice v Ústí nad Labem T. Bati a.s. Usti nad Labem, Czech Republic Krajská nemocnice Tomáše Bati - Zlin Zlin, Czech Republic Nemocnice Znojmo Znojmo, Czech Republic Latvia Latvijas Jūras Medicīnas Centrs Riga, Latvia Rīgas slimnīca Bikor Holim Riga, Latvia SIA Gremošanas slimību centrs "Gastro" Riga, Latvia Slovakia Univerzitná nemocnica Bratislava Bratislava, Slovakia Fakultná Nemocnica Nitra Nitra, Slovakia Collapse << |
| NCT01243424 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00988208 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT01117441 | Leukemia | Phase 3 | Active, not recruiting | December 2021 | - |
| NCT00547651 | Small Cell Lung Cancer | Phase 3 | Completed | - | - |
| NCT01352793 | - | - | Completed | - | - |
| NCT01620255 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT00385697 | Type 1 Diabetes Mellitus | Phase 2 Phase 3 | Completed | - | - |
| NCT01346709 | - | - | Completed | - | - |
| NCT01620255 | - | - | Completed | - | - |
| NCT02118584 | Ulcerative Colitis | Phase 3 | Recruiting | April 8, 2025 | - |
| NCT02100696 | Ulcerative Colitis | Phase 3 | Recruiting | December 26, 2020 | - |
| NCT02136069 | Ulcerative Colitis | Phase 3 | Recruiting | March 30, 2023 | - |
| NCT00947882 | - | - | Completed | - | - |
| NCT03201419 | Nocturia | Phase 2 | Recruiting | November 22, 2019 | - |
| NCT01598831 | Severe Sepsis ... more >> Coagulopathy Collapse << | Phase 3 | Active, not recruiting | July 2019 | - |
| NCT00947882 | Lower Urinary Tract Symptoms (... more >>LUTS) Collapse << | Phase 2 | Completed | - | - |
| NCT02663908 | Prostate Cancer | Phase 3 | Recruiting | December 2020 | - |
| NCT00151892 | - | - | Completed | - | - |
| NCT00508547 | - | - | Recruiting | September 30, 2029 | - |
| NCT02576574 | First Line Non-Small Cell Lung... more >> Cancer Collapse << | Phase 3 | Active, not recruiting | September 5, 2024 | - |
| NCT01771809 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT00299013 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT00151892 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT01469221 | - | - | Terminated(Business reason) | - | - |
| NCT01469221 | Bladder Cancer | Phase 3 | Terminated(Business reason) | - | United States, California ... more >> Tower Research Institute Los Angeles, California, United States, 90048 United States, Florida Department of Urology, University of Miami Miller School of Medicine Miami, Florida, United States, 33136 United States, New Jersey Somerset Urological Associates, PA Somerville, New Jersey, United States, 08876 United States, New York Associated Medical Professionals of New York, PLLC Oneida, New York, United States, 13421 Male and Female Urology Staten Island, New York, United States, 10314 Associated Medical Professionals of New York, PLLC Syracuse, New York, United States, 13210 United States, North Carolina Alliance Urology Specialists Greensboro, North Carolina, United States, 27403 Czechia Nemocnice Jablonec nad Nisou, Urologické oddělení Jablonec nad Nisou, Czechia, 466 60 Fakultní Thomayerova nemocnice s poliklinikou Urologické oddělení Prague, Czechia, 14059 Poland Szpital Miejski im. Prof. E. Michałowskiego Katowice, Poland, 40-073 CenterMed Kraków sp z o.o. Kraków, Poland, 31-530 Wojewódzki Szpital Specjalistyczny im. Stefana Kardynała Wyszyńskiego Samodzielny Publiczny Zakład Opieki Zdrowotnej w Lublinie, Oddział Urologii i Onkologii Urologicznej Lublin, Poland, 20-718 Nieubliczny Zakład Opieki Zdrowotnej Europejskie Centrum Zdrowia Otwock im. Fryderyka Chopina Nieubliczny Zakład Opieki Zdrowotnej Otwock, Poland, 05-400 Niepubliczny Zakład Opieki Zdrowotnej Pabianickie Centrum Medyczne Pabianice, Poland, 95-200 Samodzielny Publiczny Zakład Opieki Zdrowotnej, Wojewódzki Szpital Specjalistyczny nr 3 Rybnik, Poland, 44-200 Wojewódzki Szpital Specjalistyczny W Siedlcach Siedlce, Poland, 08-110 Pomorska Akademia Medyczna Szczecin, Poland, 70-111 Wojewódzki Szpital Specjalistyczny im Janusza Korczaka Słupsk, Poland, 76-200 Centrum Medycznego Kształcenia Podyplomowego, Samodzielny Publiczny Szpital Kliniczny im. Prof. W. Orłowskiego Warszawa, Poland, 00-416 Wojskowy Instytut Medyczny, Centralny Szpital Kliniczny MON Warszawa, Poland, 00-909 Szpital Bielański im. ks. J. Popiełuszki Samodzielny Publiczny Zakład Opieki Zdrowotnej Warszawa, Poland, 01-809 Szpital Kliniczny Dzieciątka Jezus - Centrum Leczenia Obrażeń Warszawa, Poland, 02-005 Międzyleski Szpital Specjalistyczny w Warszawie Oddział Urologii Warszawa, Poland, 04-749 Wojewódzki Szpital Specjalistyczny we Wrocławiu, Ośrodek Badawczo Rozwojowy Wrocław, Poland, 51-124 Niepubliczny Zakład Opieki Zdrowotnej-Centrum Medyczne Szpital Świętej Rodziny sp z o.o. Łódź, Poland, 90-302 Slovakia Univerzitná nemocnica L. Pasteura Košice Univerzitná nemocnica L. Pasteura Košice Košice, Slovakia, 041 66 Univerzitná nemocnica Martin Urologická klinika Martin, Slovakia, 036 59 Fakultná nemocnica s poliklinikou J. A. Reimana Prešov Urologická klinika Prešov, Slovakia, 081 81 Fakultná nemocnica s poliklinikou Žilina Urologické oddelenie Žilina, Slovakia, 012 07 Collapse << |
| NCT01817738 | Prostate Cancer | Phase 1 Phase 2 | Terminated(Follow up period af... more >>ter primary analysis was prematurely stopped because more mature data will not impact the study outcome) Collapse << | - | - |
| NCT02660580 | Psoriasis Pla... more >>que Type Psoriasis Moderate to Severe Plaque Psoriasis Collapse << | Phase 3 | Completed | - | - |
| NCT02580058 | Ovarian Cancer | Phase 3 | Active, not recruiting | December 19, 2018 | - |
| NCT01769196 | Idiopathic Pulmonary Fibrosis | Phase 2 | Terminated(The Study was termi... more >>nated due to lack of efficacy.) Collapse << | - | - |
| NCT02596893 | Crohn Disease | Phase 3 | Terminated(Study terminated by... more >> Sponsor following a recommendation from external DMC based on lack of emerging benefit; there were no new emergent safety findings) Collapse << | - | - |
| NCT01435356 | Urinary Bladder Neoplasms | Phase 2 | Terminated | - | - |
| NCT03607422 | Atopic Dermatitis | Phase 3 | Recruiting | June 8, 2022 | - |
| NCT03371082 | Diabetes Mellitus, Type 1 | Phase 3 | Recruiting | January 31, 2020 | - |
| NCT01298219 | Opioid-induced Bowel Dysfuncti... more >>on Collapse << | Phase 3 | Completed | - | - |
| NCT03464136 | Crohn Disease | Phase 3 | Recruiting | October 22, 2020 | - |
| NCT01769196 | - | - | Terminated(The Study was termi... more >>nated due to lack of efficacy.) Collapse << | - | - |
| NCT01619085 | Idiopathic Pulmonary Fibrosis | Phase 3 | Active, not recruiting | March 4, 2019 | - |
| NCT01619085 | - | - | Active, not recruiting | - | - |
| NCT03226899 | Gout Chronic ... more >>Kidney Disease (CKD) Collapse << | Phase 4 | Recruiting | December 2022 | - |
| NCT01923051 | - | - | Active, not recruiting | December 2019 | Czechia ... more >> University Hospital Brno Brno, Czechia, 625 00 Hospital Ceske Budejovice Ceské Budějovice, Czechia University Hospital Hradec Kralove Hradec Kralove, Czechia, 500 05 Hospital Jihlava Jihlava, Czechia, 586 33 Regional Hospital Liberec Liberec, Czechia, 460 63 Regional hospital Mlada Boleslav Mlada Boleslav, Czechia University Hospital Olomouc Olomouc, Czechia, 775 20 University Hospital Ostrava Ostrava, Czechia, 708 52 University Hospital Plzen Plzen, Czechia, 305 99 Thomayer Hospital Prague, Czechia, 140 59 University Hospital in Motol Prague, Czechia, 150 06 Hospital Na Bulovce Prague, Czechia, 180 81 Masaryk Hospital in Usti nad Labem Usti nad Labem, Czechia, 401 13 Tomas Bata Regional Hospital Zlin, Czechia, 760 01 Collapse << |
| NCT02517021 | Chemotherapy-Induced Nausea an... more >>d Vomiting Collapse << | Phase 3 | Completed | - | - |
| NCT02517021 | - | - | Completed | - | - |
| NCT01480479 | Glioblastoma ... more >>Small Cell Glioblastoma Giant Cell Glioblastoma Gliosarcoma Glioblastoma With Oligodendroglial Component Collapse << | Phase 3 | Completed | - | - |
| NCT03585270 | Aneurysmal Subarachnoid Hemorr... more >>hage Collapse << | Phase 3 | Not yet recruiting | January 19, 2021 | - |
| NCT02850965 | Psoriasis | Phase 3 | Completed | - | - |
| NCT02654587 | Non Small Cell Lung Cancer | Phase 3 | Recruiting | December 2021 | - |
| NCT01439100 | Parkinson's Disease With Sever... more >>e Pain Collapse << | Phase 3 | Completed | - | Czech Republic ... more >> Fakultní nemocnice u sv. Anny v Brně Neurologická klinika Brno, Czech Republic Poliklinika Choceň Neuroligická ambulance Chocen, Czech Republic Fakultní nemocnice Plzeň Neurologická klinika Plzeň-Lochotín, Czech Republic Neurologická ambulance Policka, Czech Republic CTC Rychnov nad Kněžnou s.r.o. Rychnov nad Kněžnou, Czech Republic Germany Neurologie Berlin Berlin-Steglitz, Germany Ruhr Universität Bochum St. Josef-Hospital Bochum, Germany Universitätsmedizin Göttingen Georg-August-Universität Göttingen, Germany Zentrum für Altersmedizin Haag i. OB, Germany Paracelsus-Elena-Klinik Kassel, Germany Uniklinik Leipzig Leipzig, Germany Philipps-Universität Marburg, Germany Asklepios Fachklinikum Abteilung für Neurologie Stadtroda, Germany Uniklinik Ulm Ulm, Germany Hungary Szent János Kórháza és Észak-budai Egyesített Kórházaik Budapest, Hungary Kenézy Kórház-Rendelőintézet Egészségügyi Szolgáltató Kft. Debrecen, Hungary Szent Pantaleon Kórház-Rendelőintézet Dunaújváros Dunaújváros, Hungary Vaszary Kolos Kórház Esztergom Esztergom, Hungary Petz Aladár Megyei Oktató Kórház Győr, Hungary Bács-Kiskun Megyei Kórháza Kecskemét, Hungary Poland NZOZ Synapsa Kielce, Poland Krakowska Akademia Neurologii Sp. z o.o. Krakow, Poland Romania Spitalul Clinic de Neuropsihiatrie Craiova, Jud. Dolj, Romania Spain Hospital Clínic i Provincial de Barcelona Barcelona, Spain USP Institut Universitari Dexeus Barcelona, Spain Hospital Universtario La Paz Madrid, Spain Hospital General de Catalunya Sant Cugat, Barcelona, Spain United Kingdom Fairfield General Hospital Pennine Acute NHS Trust Bury Great Manchester, United Kingdom King's College Hospital NHS Foundation Trust London, United Kingdom Royal Preston Hospital Preston, United Kingdom City General Hospital, Pharmacy Dept, Newcastle Road Stoke on Trent, United Kingdom Collapse << |
| NCT01289002 | - | - | Unknown | April 2015 | - |
| NCT01984242 | Renal Cell Carcinoma | Phase 2 | Active, not recruiting | August 31, 2019 | - |
| NCT00910247 | Epilepsy | Phase 3 | Completed | - | - |
| NCT03090100 | Psoriasis | Phase 3 | Completed | - | - |
| NCT01984242 | - | - | Active, not recruiting | - | - |
| NCT02788474 | Idiopathic Pulmonary Fibrosis | Phase 4 | Completed | - | - |
| NCT02048618 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT02656329 | Heart Failure | Phase 3 | Suspended(Suspended - Recruitm... more >>ent rate) Collapse << | August 2021 | - |
| NCT00910247 | - | - | Completed | - | - |
| NCT01302067 | Overactive Bladder | Phase 4 | Completed | - | - |
| NCT00558220 | Diffuse Large B-Cell Lymphoma.... more >> Primary Mediastinal B-Cell Lymphoma Follicular Lymphoma Grade III Collapse << | Phase 2 | Completed | - | Czech Republic ... more >> University Hospital Brno-Bohunice Brno, Czech Republic, 625 00 Hospital Chomutov Chomutov, Czech Republic, 430 12 University Hospital Hradec Králové Hradec Králové, Czech Republic, 500 05 University Hospital Královské Vinohrady Prague, Czech Republic, 100 34 General University Hospital Prague, Czech Republic, 128 08 University Hospital Motol Prague, Czech Republic, 150 00 Hospital Ústí nad Labem Usti nad Labem, Czech Republic, 401 13 Hospital České Budějovice České Budějovice, Czech Republic Collapse << |
| NCT03057951 | Heart Failure | Phase 3 | Recruiting | June 1, 2020 | - |
| NCT01091662 | Epilepsy | Phase 3 | Completed | - | - |
| NCT01091662 | - | - | Completed | - | - |
| NCT01302067 | - | - | Completed | - | - |
| NCT03578809 | ST Elevation Myocardial Infarc... more >>tion Collapse << | Phase 2 | Recruiting | November 24, 2020 | - |
| NCT01947907 | - | - | Completed | - | - |
| NCT01702454 | Influenza | Phase 3 | Completed | - | Czechia ... more >> GSK Investigational Site Decin, Czechia, 405 01 GSK Investigational Site Jindrichuv Hradec, Czechia, 37701 GSK Investigational Site Lipnik nad Becvou, Czechia, 75131 GSK Investigational Site Nachod, Czechia, 547 01 GSK Investigational Site Odolena voda, Czechia, 25070 GSK Investigational Site Ostrava - Poruba, Czechia, 70800 GSK Investigational Site Pardubice, Czechia, 532 03 GSK Investigational Site Praha 6, Czechia, 1600 GSK Investigational Site Tabor, Czechia, 390 02 Poland GSK Investigational Site Debica, Poland, 39-200 GSK Investigational Site Katowice, Poland, 40-018 GSK Investigational Site Siemianowice Slaskie, Poland, 41-103 Spain GSK Investigational Site Antequera/Málaga, Spain, 29200 GSK Investigational Site Blanes (Girona), Spain, 17300 GSK Investigational Site Castellón, Spain, 12004 GSK Investigational Site Castellón, Spain, 12530 GSK Investigational Site Centelles (Barcelona), Spain, 08540 GSK Investigational Site Madrid, Spain, 28046 GSK Investigational Site Paiporta, Valencia, Spain, 46200 GSK Investigational Site Quart De Poblet, Valencia, Spain, 46930 GSK Investigational Site Santiago de Compostela, Spain, 15706 GSK Investigational Site Sevilla, Spain, 41014 GSK Investigational Site Valencia, Spain, 46011 GSK Investigational Site Valencia, Spain, 46024 United Kingdom GSK Investigational Site St Austell, Cornwall, United Kingdom, PL26 7RL GSK Investigational Site Coventry, Warwickshire, United Kingdom, CV6 4DD GSK Investigational Site Belfast, United Kingdom, BT7 2EB GSK Investigational Site Bristol, United Kingdom, BS2 8AE GSK Investigational Site Exeter, United Kingdom, EX2 5DW GSK Investigational Site Gloucester, United Kingdom, GL1 3NN GSK Investigational Site London, United Kingdom, SW17 0QT GSK Investigational Site Oxford, United Kingdom, OX3 7LJ GSK Investigational Site Southampton, United Kingdom, SO16 6YD Collapse << |
| NCT01491854 | Short Children Born Small for ... more >>Gestational Age (SGA) Collapse << | Not Applicable | Active, not recruiting | June 30, 2031 | Czechia ... more >> Sandoz Investigational Site Hradec Králové, Czechia Sandoz Investigational Site Prague, Czechia Sandoz Investigational Site Ústí nad Labem, Czechia Georgia Sandoz Investigational Site Tiblisi, Georgia Germany Sandoz Investigational Site Munich, Germany Sandoz Investigational Site Sankt Augustin, Germany Hungary Sandoz Investigational Site Budapest, Hungary Sandoz Investigational Site Györ, Hungary Sandoz Investigational Site Miskolc, Hungary Sandoz Investigational Site Szeged, Hungary Poland Sandoz Investigational Site Bydgoszcz, Poland Sandoz Investigational Site Gdańsk, Poland Sandoz Investigational Site Katowice, Poland Sandoz Investigational Site Kielce, Poland Sandoz Investigational Site Kraków, Poland Sandoz Investigational Site Lódz, Poland Sandoz Investigational Site Poznań, Poland Sandoz Investigational Site Rzeszów, Poland Sandoz Investigational Site Szczecin, Poland Sandoz Investigational Site Warszawa, Poland Sandoz Investigational Site Wroclaw, Poland Sandoz Investigational Site Wrocław, Poland Sandoz Investigational Site Zabrze, Poland Romania Sandoz Investigational Site Bucharest, Romania Sandoz Investigational Site Cluj Napoca, Romania Sandoz Investigational Site Craiova, Romania Sandoz Investigational Site Iasi, Romania Collapse << |
| NCT01822899 | - | - | Completed | - | - |
| NCT02369471 | Epilepsy Foca... more >>l Seizures Collapse << | Phase 2 | Completed | - | Czech Republic ... more >> Havířov, Czech Republic Hradec Králové, Czech Republic Rychnov nad Kněžnou, Czech Republic Spain Barcelona, Spain United Kingdom Birmingham, United Kingdom Dundee, United Kingdom Glasgow, United Kingdom Great Yarmouth, United Kingdom Stoke-on-Trent, United Kingdom Collapse << |
| NCT01822899 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01947907 | Growth Hormone Deficiency (GHD... more >>) Collapse << | Phase 2 | Completed | - | - |
| NCT01702454 | - | - | Completed | - | - |
| NCT01548404 | Atopic Dermatitis | Phase 2 | Completed | - | Czechia ... more >> Nachod, Czechia Svitavy, Czechia Usti nad Labem, Czechia France Nice, France Pierre Bénite, France Toulouse, France Germany Berlin, Germany Bonn, Germany Frankfurt, Germany Gera, Germany Heidelberg, Germany Kiel, Germany Münster, Germany Hungary Kaposvar, Hungary Szeged, Hungary Szekszard, Hungary Szolnok, Hungary Poland Gdansk, Poland Lodz, Poland Lublin, Poland Warszawa, Poland Collapse << |
| NCT00091637 | Acute Myocardial Infarction | Phase 3 | Completed | - | - |
| NCT01548404 | - | - | Completed | - | - |
| NCT01754974 | Hepatitis C Virus (HCV) | Phase 3 | Completed | - | Czech Republic ... more >> Local Institution Hradec Kralove, Czech Republic, 500 05 Local Institution Praha 4, Czech Republic, 140 00 Local Institution Usti Nad Labem, Czech Republic, 400 01 Korea, Republic of Local Institution Chungcheongnam-do, Korea, Republic of, 330-930 Local Institution Daegu, Korea, Republic of, 700-821 Local Institution Gangwon-do, Korea, Republic of, 200-704 Local Institution Gyeonggi-do, Korea, Republic of, 420-717 Local Institution Gyeonggi-do, Korea, Republic of, 463-707 Local Institution Gyeongsangnam-do, Korea, Republic of, 626-770 Local Institution Incheon, Korea, Republic of, 403-720 Local Institution Seoul, Korea, Republic of, 156-755 Mexico Local Institution Mexico, Distrito Federal, Mexico, 07760 Local Institution Mexico City, Estado De Mexico, Mexico, 06700 Local Institution Guadalajara, Jalisco, Mexico, 44500 Local Institution Guadalajara, Jalisco, Mexico, 44650 Local Institution Distrito Federal, Mexico, 03720 Collapse << |
| NCT00596752 | - | - | Completed | - | - |
| NCT00596752 | Peripheral Arterial Occlusive ... more >>Disease Collapse << | Phase 4 | Completed | - | - |
| NCT02630680 | - | - | Completed | - | Czechia ... more >> Ustredni vojenska nemocnice Praha, Czechia, 169 02 Krajská zdravotní, a.s., nemocnice Teplice Teplice, Czechia, 415 01 Ústeckoorlická nemocnice Ústí nad Orlicí, Czechia, 562 01 Germany MVZ Ortenau Achern, Innere Medizin Achern, Germany, 77855 Gemeinschaftspraxis Dres. Klausmann Aschaffenburg, Germany, 63739 Klinikum Aschaffenburg-Alzenau, Standort Aschaffenburg Aschaffenburg, Germany, 63739 Chefarzt der Medizinischen Klinik III, Westpfalz Klinikum GmbH, Standort I Kaiserslautern Kaiserslautern, Germany, 67655 MVZ Innere Medizin Marburg, Dres. Drude und Partner Marburg, Germany, 35037 Medizinisches Versorgungszentrum Münster, Germany, 48155 Praxisgemeinschaft Innere am Stadtpark Nürnberg, Germany, 90409 Gastro-Praxis Wiesbaden (Gastrodata) Wiesbaden, Germany, 65189 Netherlands Erasmus MC Rotterdam, Netherlands, 3015 CE Elisabeth-Tweesteden Ziekenhuis Tilburg, Netherlands, 5042 AD Poland Copernicus Medical Entity Gdańsk, Poland, 80-462 Instytut Medycyny Lublin, Poland, 20-090 Oncology center - Institutte Warsaw, Poland Collapse << |
| NCT01013675 | Influenza Vaccines | Phase 2 | Completed | - | Czech Republic ... more >> Site Reference ID/Investigator# 45005 Benatky nad Jizerou, Czech Republic, 294 71 Site Reference ID/Investigator# 45004 Hradec Kralove, Czech Republic, 500 03 Estonia Site Reference ID/Investigator# 45007 Paide, Estonia, 72713 Site Reference ID/Investigator# 45012 Saku, Estonia, 75501 Site Reference ID/Investigator# 45009 Tallinn, Estonia, 10617 Site Reference ID/Investigator# 45010 Tallinn, Estonia, 10617 Site Reference ID/Investigator# 45013 Tartu, Estonia, 51014 Lithuania Site Reference ID/Investigator# 45017 Kaunas, Lithuania, 47144 Site Reference ID/Investigator# 45015 Kaunas, Lithuania, 48259 Site Reference ID/Investigator# 45016 Kaunas, Lithuania, 49456 Site Reference ID/Investigator# 45018 Kaunas, Lithuania, 50009 Site Reference ID/Investigator# 45019 Klaipeda, Lithuania, 92231 Collapse << |
| NCT02680197 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | Czechia ... more >> Clinical Trial Site 203-002 Karlovy Vary, Czechia, 36017 Clinical Trial Site 203-003 Kralupy nad Vltavou, Czechia, 27801 Clinical Trial Site 203-001 Melnik, Czechia, 25063 Clinical Trial Site 203-007 Mlada Boleslav, Czechia, 29350 Clinical Trial Site 203-005 Moravský Krumlov, Czechia, 67201 Clinical Trial Site 203-006 Teplice, Czechia, 41501 Clinical Trial Site 203-004 Varnsdorf, Czechia, 40447 Germany Clinical Trial Site 276-003 Bonn, Germany, 53111 Clinical Trial Site 276-004 Cottbus, Germany, 03053 Clinical Trial Site 276-006 Freiburg, Germany, 579106 Clinical Trial Site 276-002 Grosshansdorf, Germany, 22927 Clinical Trial Site 276-005 Stuttgart, Germany, 70174 Clinical Trial Site 276-001 Wiesbaden, Germany, 65187 Hungary Clinical Trail Site 348-006 Budapest, Hungary, 1113 Clinical Trial Site 348-003 Budapest, Hungary, 1126 Clinical Trial Site 348-004 Budapest, Hungary, 1126 Clinical Trial Site 348-001 Deszk, Hungary, 6772 Clinical Trial Site 348-005 Komarom, Hungary, 2900 Clinical Trial Site 348-002 Létavértes, Hungary, 4283 Clinical Trail Site 348-008 Miskolc, Hungary, 3519 Clinical Trail Site 348-009 Seregélyes, Hungary, 8111 Clinical Trail Site 348-007 Szombathely, Hungary, 3700 Romania Clinical Trial Site 642-005 Bacau, Romania, 600252 Clinical Trial Site 642-003 Brasov, Romania, 500283 Clinical Trial Site 642-007 Bucharest, Romania, 030303 Clinical Trial Site 642-008 Bucharest, Romania, 030303 Clinical Trial Site 642-004 Cluj-Napoca, Romania, 400445 Clinical Trial Site 642-001 Codlea, Romania, 505100 Clinical Trial Site 642-002 Miercurea Ciuc, Romania Clinical Trial Site 642-006 Suceava, Romania, 720224 Collapse << |
| NCT01104753 | - | - | Completed | - | Czech Republic ... more >> Hospital Brandýs n/Lab. Brandys nad Labem, Czech Republic Faculty Hospital U Sv. Anny Brno Brno, Czech Republic Hospital Břeclav Břeclav, Czech Republic Investigational site Chomutov, Czech Republic Hospital Chrudim Chrudim, Czech Republic Hospital. Frýdek- Místek Frýdek- Místek, Czech Republic Investigational site Hodonín, Czech Republic Investigational site Hradec Králové, Czech Republic Hospital Karviná Karviná, Czech Republic Investigational site Litoměřice, Czech Republic Hospitál Mladá Bolesl Mladá Boleslav, Czech Republic Faculty Hospital Olomouc Olomouc, Czech Republic Investigational site Ostrava, Czech Republic Vítkovická Hospital Ostrava, Czech Republic Gastromedic s.r.o.,ul. Pardubice, Czech Republic Faculty Hospital Bpry Plzeň Plzeň, Czech Republic Faculty Hospital Lochotín Plzeň Plzeň, Czech Republic Faculty Hospital Motol, Praha 5 Praha, Czech Republic NMSB, Praha 1 Praha, Czech Republic Poliklinika Prosek, Praha Praha, Czech Republic Thomayerova hospital., Praha 4 Praha, Czech Republic VFN, Praha 2 Praha, Czech Republic ÚVN, Praha 6 Praha, Czech Republic Investigational site Přerov, Czech Republic Hospital Svitavy Svitavy, Czech Republic Provincial hospital, Kpt. Tábor, Czech Republic Hospital T. Bati Zlín Zlín, Czech Republic Hospital Znojmo Znojmo, Czech Republic Investigational site Ústí n.Labem, Czech Republic Investigational site České Budějovice, Czech Republic Collapse << |
| NCT03547271 | Meningococcal Infections | Phase 3 | Not yet recruiting | September 2021 | - |
| NCT03281122 | Heart Failure | Phase 1 | Active, not recruiting | May 21, 2019 | Czechia ... more >> Local Institution Praha 2, Czechia, 12808 Local Institution Usti nad Labem, Czechia, 401 13 Netherlands Local Institution Deventer, Netherlands, 7416 SE Local Institution Groningen, Netherlands, 9713 GZ Local Institution Groningen, Netherlands, 9728 NZ Local Institution Haarlem, Netherlands, 2035 RC Local Institution Sneek, Netherlands, 8601 ZR Poland Local Institution Lublin, Poland, 20-954 Local Institution Wroclaw, Poland, 50-981 Spain Local Institution Madrid, Spain, 28034 Local Institution Madrid, Spain, 28040 Complejo Hospitalario Universitario de Santiago Santiago de Compostela, Spain, 15706 Local Institution Valencia, Spain, 46010 Local Institution Vigo, Spain, 36312 United Kingdom Local Institution Dundee, United Kingdom, DD1 9SY Local Institution Edinburgh, United Kingdom, EH16 4SB Local Institution London, United Kingdom, SE1 1YR Collapse << |
| NCT02294786 | Cancer | Phase 2 | Terminated(study was stopped d... more >>ue to futility by the DMC in September 2016) Collapse << | - | - |
| NCT03635411 | Recovery of Function | Phase 1 | Not yet recruiting | January 2020 | - |
| NCT01867021 | Influenza | Phase 4 | Completed | - | Czech Republic ... more >> Site 71 - Ordinace všeobecného lékaře Kbel 163, Benátky nad Jizerou, Czech Republic, 294 71 Site 70 - Vaccination and Travel Medicine Centre Poliklinika II., Bratri Stefanu 895, Hradec Kralove, Czech Republic, 500 03 Philippines Site 61 - Research Institute for Tropical Medicine DOH Compound Filinvest Corporate City, Alabang Muntinlupa City, Philippines Site 60 - De La Salle Health Sciences Institute, Room 6210 and 6206 De La Salle Angelo King Medical Research Center Congressional Road, Dasmarinas City, Cavite, Philippines, 4114 Site 63 - Our Lady of Lourdes Hospital, 46 P. Sanchez Street, Sta. Mesa, Manila, Philippines Site 62 - Research Institute for Tropical Medicine DOH Compound, Filinvest Corporate City Alabang, Muntinlupa City, Philippines South Africa Site 33 - TREAD Research , Room 41, 8th Floor, Department of Cardiology, Tygerberg Hospital, Francie van Zijl Drive Province of the Western Cape, Cape Town, South Africa, 7500 Site 43 - Allergy Diagnostic & Clinical Research Unit, UCT Lung Institute, George Street Province of the Western Cape, Cape Town, South Africa, 7700 Site 34 - Synopsis Research, Room 8, First floor, Fountain Centre, Belmont Road Rondebosch, Cape Town, South Africa, 7700 Site 41 - Tiervlei Trial Centre, Karl Bremer Hospital, c/o Mike Pienaar Boulevard & Frans Conradie Avenue, Bellville Western Cape, Cape Town, South Africa, 7530 Site 45 - 343 Randles Road Sydenham, Durban, South Africa, 4091 Site 44 - Dr B van der Berg and Associates, 162 Pretoria Road, Rynfield Benoni, Gauteng, South Africa, 1511 Site 31 - MD Search, 1 Paul Smit Street Boksburg North, Gauteng, South Africa, 1459 Site 32 - EMMED Research, Emmed Research, 641 5th Avenue Eloffsdal, Gauteng, South Africa, 0084 Site 35 - I Engelbrecht Research (Pty) Ltd, 174 Cradock Avenue Lyttleton, Gauteng, South Africa, 0157 Site 36 - Midrand Medical Centre, Shop #1, Health Emporium, Cnr Church and Market Street Midrand, Gauteng, South Africa, 1685 Site 39 - Medicross Sophiatown, Cnr Edward and Millar Streets Sophiatown, Gauteng, South Africa, 2092 Site 38 - Perinatal HIV Research Unit, New Nurses Home, Chris Hani Baragwanath Academic Hospital, Chris Hani Road Soweto, Gauteng, South Africa, 1862 Site 37 - Excellentis Clinical Trial Consultants, Suite 201, York Building, 72 York Street, George, South Africa, 6529 Site 40 - Newtown Clinical Research Centre, Suite 3, Newgate Centre, 104 Jeppe Street Newtown, Johannesburg, South Africa, 2113 Site 46 - Helderberg Clinical Trials Centre, Suite 7G&H Arun Place Sir Lowry's Pass Road, Somerset West, South Africa, 7130 Thailand Site 53 - Clinical Trials Unit, Office for Research and Development, His Majesty the King's 80th Birthday December 2007 Building, 3rd floor, room 307, Faculty of Medicine Siriraj Hospital, Mahidol University, 2 Prannok Rd. Bangkoknoi, Bangkok, Thailand, 10700 Site 52 - 1. Faculty of Medicine, Chulalongkorn University Rama 4 Rd., Pathumwan, Bangkok, Thailand, 10300 Site 52 - 2. Queen Saovabha Memorial Institute Thai Red Cross Society, Rama 4 Rd., Pathumwan, Bangkok, Thailand, 10300 Site 51 - Faculty of Medicine, Srinakharinwirot University, HRH Princess, Sirindhorn Medical Center Ongkarak, Nakhorn, Nayok, Thailand, 26120 Collapse << |
| NCT01867021 | - | - | Completed | - | - |
| NCT03334396 | Atopic Dermatitis | Phase 3 | Recruiting | August 15, 2019 | - |
| NCT01991990 | Healthy Volunteer | Phase 4 | Completed | - | United Kingdom ... more >> Cambridge, United Kingdom, CB2 0QQ Collapse << |
| NCT00662103 | Breast Cancer ... more >> Cancer Survivor Fatigue Long-term Effects Secondary to Cancer Therapy in Adults Psychosocial Effects of Cancer and Its Treatment Collapse << | Not Applicable | Completed | - | United States, Oregon ... more >> Knight Cancer Institute at Oregon Health and Science University Portland, Oregon, United States, 97239-3098 Collapse << |
| NCT01991990 | - | - | Completed | - | - |
| NCT00098774 | Lymphoma | Phase 2 | Completed | - | - |
| NCT03696446 | Coronary Heart Disease | Not Applicable | Recruiting | December 31, 2020 | Canada, Ontario ... more >> University of Ottawa Heart Institute Recruiting Ottawa, Ontario, Canada, K1Y 4W7 Contact: Evyanne Wooding 613-696-7000 ext 17596 ewooding@ottawaheart.ca Collapse << |
| NCT02205762 | Langerhans Cell Histiocytosis | Phase 2 Phase 3 | Recruiting | July 2023 | United States, Arizona ... more >> Phoenix Children's Hospital Recruiting Phoenix, Arizona, United States, 85006 Contact: Michael Henry, MD mhenry@phoenixchildrens.com Principal Investigator: Michael Henry, MD United States, Arkansas Arkansas Children's Hospital Recruiting Little Rock, Arkansas, United States, 72202 Contact: Kimo Stine, MD stinekimoc@uams.edu Principal Investigator: Kimo Stine, MD United States, California Children's Hospital of Los Angeles Recruiting Los Angeles, California, United States, 90027 Contact: Rima Jubran, MD rjubran@chla.usc.edu Principal Investigator: Rima Jubran, MD Valley Children's Healthcare Not yet recruiting Madera, California, United States, 93636 Contact: David Samuel, MD DSamuel@valleychildrens.org Contact: Faisal Razzaqi, MD FRazzaqi@valleychildrens.org Principal Investigator: David Samuel, MD United States, Connecticut Connecticut Children's Medical Center Not yet recruiting Hartford, Connecticut, United States, 06106 Contact: Andrea Orsey, MD aorsey@connecticutchildrens.org Principal Investigator: Andrea Orsey, MD United States, District of Columbia Children's National Medical Center Recruiting Washington, District of Columbia, United States, 20010 Contact: Stephan Ladisch, MD sladisch@childrensnational.org Contact: Jay Greenberg, MD jagreenb@childrensnational.org United States, Maryland Johns Hopkins University Not yet recruiting Baltimore, Maryland, United States, 21287 Contact: Elias Zambidis, MD EZambid1@jhmi.edu Contact: Christine Pratilas, MD Cpratil1@jhmi.edu United States, Massachusetts Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Mary Huang, MD 617-726-2737 mshuang@partners.org Principal Investigator: Mary Huang, MD Dana Farber Cancer Institute Recruiting Boston, Massachusetts, United States, 02115 Contact: Barbara Degar, MD 617-632-6801 bdegar@partners.org Principal Investigator: Barbara Degar, MD United States, Minnesota Children's Minnesota Recruiting Minneapolis, Minnesota, United States, 55404 Contact: Michael Richards, MD Michael.richards@childrensmn.org Contact: Nathan Gossai, MD nathan.gossai@childrensmn.org United States, New Jersey Hackensack University Medical Center Recruiting Hackensack, New Jersey, United States, 07601 Contact: Steven Diamond, MD Steven.Diamond@hackensackmeridian.org Contact: Burton Appel, MD Burton.appel@hackensackmeridian.org United States, New York Cohen Children's Medical Center Recruiting New Hyde Park, New York, United States, 11040 Contact: Carolyn Fein Levy, MD clevy4@northwell.edu Contact: Jeffery Lipton, MD jlipton@northwell.edu Columbia University / Herbert Irving Cancer Center Recruiting New York, New York, United States, 10032 Contact: Maria Luis Sulis, MD mls95@cumc.columbia.edu Contact: Julia Glade Bender, MD jg589@cumc.columbia.edu Staten Island University Hospital Recruiting Staten Island, New York, United States, 10305 Contact: Sarah Vaiselbuh, MD 718-226-6435 svaiselbuh@nshs.edu Principal Investigator: Sarah Vaiselbuh`, MD SUNY Upstate Medical University Recruiting Syracuse, New York, United States, 13210 Contact: Melanie Comito, MD comitom@upstate.edu Contact: Andrea Dvorak, MD dvoraka@upstate.edu United States, Ohio Cincinnati Children's Hospital Medical Center Recruiting Cincinnati, Ohio, United States, 45229 Contact: Michael Jordan, MD 513-636-1773 Michael.jordan@cchmc.org Contact: Ashish Kumar, MD 513-803-1631 ashish.kumar@cchmc.org United States, South Carolina Medical Universtiy of South Carolina/Hollings Cancer Center Not yet recruiting Charleston, South Carolina, United States, 29425 Contact: Jacqueline Kraveka, DO 843-792-2957 kravekjm@musc.edu Principal Investigator: Jacqueline Kraveka, DO United States, Tennessee St. Jude Children's Research Hospital Recruiting Memphis, Tennessee, United States, 38105 Contact: Patrick Campbell, MD patrick.campbell@stjude.org Principal Investigator: Patrick Campbell, MD United States, Washington Providence Sacred Heart Children's Hospital Recruiting Spokane, Washington, United States, 99204 Contact: Stefanos Intzes, MD stefanos.intzes@providence.org Contact: Judy Felgenhauer, MD judy.felgenhauer@providence.org Collapse << |
| NCT00046228 | Myocardial Infarction | Phase 3 | Completed | - | - |
| NCT00297414 | - | - | Completed | - | - |
| NCT00441727 | Gastric Ulcer ... more >> Duodenal Ulcer Collapse << | Phase 3 | Completed | - | - |
| NCT00098774 | - | - | Completed | - | - |
| NCT00046228 | - | - | Completed | - | - |
| NCT03517995 | Bladder Cancer ... more >> Bladder Tumor Urothelial Carcinoma Collapse << | Phase 2 | Not yet recruiting | July 2022 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Not yet recruiting Tampa, Florida, United States, 33612 Contact: Kayoko Kennedy 813-745-1947 kayoko.kennedy@moffitt.org Contact: Nagi Kumar, Ph.D. 813-745-6885 nagi.kumar@moffitt.org James A. Haley Veteran's Administration Hospital Not yet recruiting Tampa, Florida, United States, 33612 Contact: Cesar Ercole, M.D. 813-910-4088 cesar.ercole1@va.gov Principal Investigator: Cesar Ercole, M.D. Collapse << |
| NCT00441727 | - | - | Completed | - | - |
| NCT00071331 | Congestive Heart Failure | Phase 3 | Completed | - | - |
| NCT03493802 | - | - | Recruiting | November 1, 2020 | United States, Minnesota ... more >> Mayo Clinic Recruiting Rochester, Minnesota, United States, 55905 Contact: Kathleen R Leistikow, ACRC 507-266-1316 leistikow.kathleen@mayo.edu Contact: Charles D Madsen, CCRP 507-266-9391 madsen.charles@mayo.edu Principal Investigator: Maria V Irazabal, M.D. Collapse << |
| NCT02127762 | Diabetes Mellitus ... more >> Type I Diabetes Type II Diabetes Diabetic Peripheral Neuropathy Chronic Pain Collapse << | Not Applicable | Completed | - | Canada, Ontario ... more >> The Ottawa Hospital Ottawa, Ontario, Canada, K1H 8L6 Collapse << |
| NCT00430898 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT00206154 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 3 | Completed | - | - |
| NCT00160667 | Neuralgia, Postherpetic | Phase 2 | Completed | - | - |
| NCT00479401 | Early Parkinson Disease (Early... more >> PD) Collapse << | Phase 3 | Completed | - | - |
| NCT01164501 | Diabetes Mellitus, Type 2 ... more >> Renal Insufficiency Collapse << | Phase 3 | Completed | - | - |
| NCT00144339 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01164501 | - | - | Completed | - | - |
| NCT00144339 | - | - | Completed | - | - |
| NCT01313676 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00378690 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT00479401 | - | - | Completed | - | - |
| NCT00601523 | Parkinson Disease | Phase 3 | Completed | - | - |
| NCT00407537 | Hypertension ... more >>Hypercholesterolemia Collapse << | Phase 4 | Completed | - | - |
| NCT01313676 | - | - | Completed | - | - |
| NCT00601523 | - | - | Completed | - | - |
| NCT01061567 | - | - | Completed | - | - |
| NCT00802178 | - | - | Completed | - | - |
| NCT01061567 | - | - | Completed | - | - |
| NCT00407537 | - | - | Completed | - | - |
| NCT00802178 | - | - | Completed | - | - |
| NCT00679627 | - | - | Terminated(Due to a pre-specif... more >>ied imbalance of deaths between treatment groups, the DSMB recommended early termination of the trial) Collapse << | - | - |
| NCT00679627 | Alzheimer's Disease | Phase 3 | Terminated(Due to a pre-specif... more >>ied imbalance of deaths between treatment groups, the DSMB recommended early termination of the trial) Collapse << | - | - |
| NCT00466167 | - | - | Completed | - | - |
| NCT00391872 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT00617669 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT00466167 | Parkinson Disease | Phase 3 | Completed | - | - |
| NCT00391872 | - | - | Completed | - | - |
| NCT00626548 | Prostate Cancer | Phase 3 | Terminated(early efficacy revi... more >>ew by the Independent Data Monitoring Committee indicated it was unlikely to meet its primary efficacy endpoints) Collapse << | - | - |
| NCT00577460 | - | - | Completed | - | - |
| NCT00617669 | - | - | Completed | - | - |
| NCT00626548 | - | - | Terminated(early efficacy revi... more >>ew by the Independent Data Monitoring Committee indicated it was unlikely to meet its primary efficacy endpoints) Collapse << | - | - |
| NCT00781820 | Onychomycosis | Phase 3 | Completed | - | - |
| NCT00577460 | Parkinson Disease | Phase 3 | Completed | - | - |
| NCT00643201 | Venous Thrombosis | Phase 3 | Completed | - | - |
| NCT01068678 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01536418 | - | - | Terminated | - | - |
| NCT00781391 | Stroke Atrial... more >> Fibrillation Embolism Collapse << | Phase 3 | Completed | - | - |
| NCT01470612 | Ulcerative Colitis | Phase 3 | Active, not recruiting | July 27, 2020 | - |
| NCT01068678 | - | - | Completed | - | - |
| NCT00809965 | Acute Coronary Syndrome ... more >> Myocardial Infarction Myocardial Ischemia Unstable Angina Collapse << | Phase 3 | Completed | - | - |
| NCT00809965 | - | - | Completed | - | - |
| NCT01536418 | Crohn's Disease | Phase 3 | Terminated | - | - |
| NCT01107886 | Type 2 Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02058160 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00781391 | - | - | Completed | - | - |
| NCT00643201 | - | - | Completed | - | - |
| NCT01609582 | Type 2 Diabetes ... more >> Cardiovascular Disease Collapse << | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01620762 | Rhinoconjunctivitis | Phase 3 | Completed | - | - |
| NCT01609582 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT03531554 | VLCAD Deficiency ... more >> Fatty Acid Oxidation Defects Collapse << | Not Applicable | Completed | - | Netherlands ... more >> Academic Medical Center Amsterdam, Noord-Holland, Netherlands, 1105 AZ Dept of Neuroscience/ Neuroimaging Center Groningen, Netherlands, 9700RB Collapse << |
| NCT01620762 | - | - | Completed | - | - |
| NCT02058160 | - | - | Completed | - | - |
| NCT01265654 | - | - | Completed | - | Czech Republic ... more >> Research Site Benesov u Prahy, Czech Republic Research Site Brno, Czech Republic Research Site Ceske Budejovice, Czech Republic Research Site Cesky Krumlov, Czech Republic Research Site Cheb, Czech Republic Research Site Chomutov, Czech Republic Research Site Havlickuv Brod, Czech Republic Research Site Hradec Kralove, Czech Republic Research Site Jablonec nad Nisou, Czech Republic Research Site Jicin, Czech Republic Research Site Jihlava, Czech Republic Research Site Karvina, Czech Republic Research Site Kladno, Czech Republic Research Site Kolin, Czech Republic Research Site Liberec, Czech Republic Research Site Most, Czech Republic Research Site Nachod, Czech Republic Research Site Novy Jicin, Czech Republic Research Site Olomouc, Czech Republic Research Site Opava, Czech Republic Research Site Ostrava - Fifejdy, Czech Republic Research Site Ostrava - Vitkovice, Czech Republic Research Site Ostrava, Czech Republic Research Site Pardubice, Czech Republic Research Site Pisek, Czech Republic Research Site Plzen, Czech Republic Research Site Praha, Czech Republic Research Site Prerov, Czech Republic Research Site Prostejov, Czech Republic Research Site Rakovnik, Czech Republic Research Site Sokolov, Czech Republic Research Site Strakonice, Czech Republic Research Site Sumperk, Czech Republic Research Site Tabor, Czech Republic Research Site Usti nad Labem, Czech Republic Research Site Zlin, Czech Republic Research Site Znojmo, Czech Republic Collapse << |
| NCT01107886 | - | - | Completed | - | - |
| NCT01259297 | - | - | Terminated(Terminated early in... more >> agreement with Health Authorities for feasibility reasons) Collapse << | - | - |
| NCT01259297 | Cardiovascular Events | Phase 3 | Terminated(Terminated early in... more >> agreement with Health Authorities for feasibility reasons) Collapse << | - | - |
| NCT01681992 | Measles Mumps... more >> Rubella Collapse << | Phase 3 | Completed | - | - |
| NCT01130246 | Acute Coronary Syndrome | Phase 3 | Terminated(Lack of efficacy) | - | - |
| NCT01748591 | - | - | Terminated(Terminated due to p... more >>roblems enrolling adequate number of patients.) Collapse << | - | Czech Republic ... more >> Nemocnice Rudolfa a Stefanie Benešov, Czech Republic Nemocnice Blansko Blansko, Czech Republic Nemocnice Břeclav Břeclav, Czech Republic Děčínská nemocnice Děčín, Czech Republic Nemocnice Havlíčkův Brod Havlíčkův Brod, Czech Republic Fakultní nemocnice Hradec Králové Hradec Králové, Czech Republic Nemocnice Jablonec Jablonec nad Nisou, Czech Republic Nemocnice Jihlava Jihlava, Czech Republic Privátní GE Jihlava, Czech Republic FN Olomouc Olomouc, Czech Republic Všeobecná fakutlní nemocnice Praha 2, Czech Republic Sanatorium Sv.Anny Praha 3, Czech Republic Privátní GE Praha 4 Praha 4, Czech Republic Thomayerova nemocnice Praha 4, Czech Republic FN Bulovka Praha 8, Czech Republic Nemocnice Příbram Příbram, Czech Republic Lužická nemocnice Rumburk, Czech Republic Nemocnice Teplice Teplice, Czech Republic Nemocnice Tábor Tábor, Czech Republic Nemocnice Znojmo Znojmo, Czech Republic Ústecká poliklinika Ústí nad Labem, Czech Republic Soukrmá ambulance Česká Lípa, Czech Republic Collapse << |
| NCT02065570 | Crohn's Disease | Phase 3 | Active, not recruiting | December 11, 2020 | - |
| NCT02770131 | - | - | Recruiting | June 24, 2021 | - |
| NCT01681992 | - | - | Completed | - | - |
| NCT01241591 | - | - | Completed | - | - |
| NCT01225562 | - | - | Completed | - | - |
| NCT01241591 | Psoriasis | Phase 3 | Completed | - | - |
| NCT02706873 | Rheumatoid Arthritis | Phase 3 | Active, not recruiting | March 28, 2021 | - |
| NCT01549964 | Glycemic Control | Phase 3 | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT03708185 | Healthy | Not Applicable | Recruiting | September 30, 2020 | United Kingdom ... more >> School of Sports and Health Sciences Recruiting Exeter, Devon, United Kingdom, EX4 4JA Contact: David J Childs 01392 722771 ext 2771 d.j.childs@exeter.ac.uk Collapse << |
| NCT01225562 | Myocardial Infarction ... more >> Cardiovascular Death Atherothrombosis Stroke Collapse << | Phase 3 | Completed | - | - |
| NCT02394028 | Crohn Disease | Phase 3 | Recruiting | February 19, 2021 | - |
| NCT01549964 | - | - | Terminated(Due to concerns abo... more >>ut potential liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01674712 | Dyslipidemia | Phase 3 | Completed | - | - |
| NCT02403323 | Crohn Disease | Phase 3 | Recruiting | April 19, 2024 | - |
| NCT01604928 | Urinary Bladder, Overactive | Phase 2 | Completed | - | Belgium ... more >> Site: 12 Brussels, Belgium, 1090 Site: 13 Edegem, Belgium, 2650 Site: 11 Gent, Belgium, 9000 Site: 10 Leuven, Belgium, 3000 Czech Republic Site: 25 Melnik, Czech Republic, 276 01 Site: 22 Prague, Czech Republic, 128 08 Site: 24 Prague, Czech Republic, 140 59 Site: 20 Prague, Czech Republic, 180 81 Site: 21 Usti nad Labem, Czech Republic, 40001 Site: 23 Usti nad Labem, Czech Republic, 401 13 Germany Site: 32 Bad Ems, Germany, 56130 Site: 30 Emmendingen, Germany, 79312 Site: 35 Frankfurt, Germany, 60326 Site: 34 Hagenow, Germany, 19230 Site: 33 Hamburg, Germany, 20253 Site: 36 Koblenz, Germany, 56068 Site: 31 Trier, Germany, 54290 Spain Site: 42 Alzira-Valencia, Spain, 46600 Site: 40 Madrid, Spain, 28046 Site: 43 Miranda de Ebro, Spain, 09200 Sweden Site: 55 Gotenburg, Sweden, 413 45 Site: 53 Linkoping, Sweden, 582 24 Site: 50 Lund, Sweden, 221 85 Site: 51 Stockholm, Sweden, 141 86 Site: 52 Uppsala, Sweden, 751 85 United Kingdom Site: 64 Bimingham, United Kingdom, B15 2TG Site: 62 London, United Kingdom, N19 5LW Site: 63 London, United Kingdom, W1P 7PN Site: 60 Sheffield, United Kingdom, S10 2JF Site: 61 Swansea, United Kingdom, SA6 6NL Collapse << |
| NCT01730534 | Diabetes Mellitus, Non-Insulin... more >>-Dependent High Risk for Cardiovascular Event Collapse << | Phase 3 | Completed | - | - |
| NCT02234583 | Pain Associated With Fibromyal... more >>gia Collapse << | Phase 3 | Completed | - | - |
| NCT02157935 | COPD Patients | Phase 3 | Completed | - | - |
| NCT02207634 | Dyslipidemia | Phase 3 | Completed | - | - |
| NCT02207413 | - | - | Completed | - | - |
| NCT02207413 | Influenza | Phase 3 | Completed | - | - |
| NCT02207634 | - | - | Completed | - | - |
| NCT02345161 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01674712 | - | - | Completed | - | - |
| NCT02108262 | Acute Myocardial Infarction | Phase 2 | Completed | - | - |
| NCT02345161 | - | - | Completed | - | - |
| NCT02157935 | - | - | Completed | - | - |
| NCT01870778 | Acute Heart Failure | Phase 3 | Completed | - | - |
| NCT02676089 | Asthma | Phase 3 | Completed | - | - |
| NCT02065791 | Diabetes Mellitus, Type 2 ... more >> Diabetic Nephropathy Collapse << | Phase 3 | Active, not recruiting | June 28, 2019 | - |
| NCT02585934 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT02585934 | - | - | Completed | - | - |
| NCT02586909 | Alzheimer's Disease | Phase 3 | Terminated(Intepirdine did not... more >> meet its primary efficacy endpoints in lead-in study RVT-101-3001.) Collapse << | - | - |
| NCT02107196 | Irritable Bowel Syndrome With ... more >>Diarrhea Collapse << | Phase 3 | Completed | - | - |
| NCT02111564 | Heart Failure ... more >> Respiratory Insufficiency Stroke Acute Infectious Diseases Rheumatic Diseases Collapse << | Phase 3 | Completed | - | - |
| NCT02121613 | Benign Prostatic Hyperplasia (... more >>BPH) Collapse << | Phase 4 | Completed | - | - |
| NCT01870778 | - | - | Completed | - | - |
| NCT02415400 | Acute Coronary Syndromes | Phase 4 | Completed | - | - |
| NCT03325556 | Dementia-related Psychosis | Phase 3 | Recruiting | March 2020 | - |
| NCT03036280 | Alzheimer's Disease | Phase 3 | Recruiting | June 29, 2021 | - |
| NCT02174731 | Anemia | Phase 3 | Completed | - | - |
| NCT03611751 | Psoriasis | Phase 3 | Recruiting | July 8, 2020 | - |
| NCT02107404 | Prostate Cancer | Phase 2 | Completed | - | Czechia ... more >> Brno, Czechia Hradec Kralove, Czechia Jablonec nad Nisou, Czechia Jihlava, Czechia Mnisek pod Brdy, Czechia Novy Jicin, Czechia Olomouc, Czechia Plzen, Czechia Praha 10, Czechia Praha 4, Czechia Praha 5, Czechia Uherske Hradiste, Czechia Usti nad Labem, Czechia Collapse << |
| NCT00316095 | Hypertension ... more >>Dyslipidemias Collapse << | Phase 3 | Completed | - | - |
| NCT02107196 | - | - | Completed | - | - |
| NCT02704091 | Acute Diarrhoea | Phase 4 | Recruiting | June 2019 | - |
| NCT02554786 | Asthma | Phase 3 | Recruiting | June 24, 2019 | - |
| NCT01807520 | - | - | Completed | - | - |
| NCT01807520 | Moderate to Severe Nail Psoria... more >>sis Collapse << | Phase 3 | Completed | - | - |
| NCT02555878 | Neoplasms | Phase 3 | Completed | - | - |
| NCT01813422 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT01791361 | - | - | Completed | - | - |
| NCT01813422 | - | - | Completed | - | - |
| NCT01844856 | Complicated Intra-abdominal In... more >>fections Collapse << | Phase 3 | Completed | - | - |
| NCT01844856 | - | - | Completed | - | - |
| NCT01853384 | Venous Ulcer ... more >>Venous Stasis Ulcer Ulcer Collapse << | Phase 3 | Terminated(based on outcome of... more >> trial NCT01656889.) Collapse << | - | - |
| NCT01853384 | - | - | Terminated(based on outcome of... more >> trial NCT01656889.) Collapse << | - | - |
| NCT02615873 | Parkinson's Disease | Phase 3 | Recruiting | December 2019 | - |
| NCT02461589 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 2 | Completed | - | - |
| NCT01859988 | Atopic Dermatitis | Phase 2 | Completed | - | - |
| NCT01009593 | Hepatocellular Carcinoma Non-r... more >>esectable Hepatocellular Carcinoma Recurrent Carcinoma, Hepatocellular Liver Diseases Neoplasms by Histologic Type Digestive System Neoplasms Carcinoma Liver Neoplasms Neoplasms Neoplasms by Site Digestive System Diseases Adenocarcinoma Neoplasms, Glandular and Epithelial Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT01859988 | - | - | Completed | - | - |
| NCT01170065 | Pulmonary Fibrosis | Phase 2 | Completed | - | - |
| NCT01439360 | Influenza | Phase 3 | Completed | - | - |
| NCT01015287 | Acute Coronary Syndromes | Phase 3 | Completed | - | - |
| NCT01439360 | - | - | Completed | - | - |
| NCT01435616 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01015287 | - | - | Completed | - | - |
| NCT01018173 | Diabetes Mellitus Type 2 | Phase 3 | Completed | - | - |
| NCT01463397 | Neuropathic Pain | Phase 2 | Completed | - | - |
| NCT01028963 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | Australia, Queensland ... more >> Auchenflower, Queensland, Australia Kippa-Ring, Queensland, Australia Czech Republic Beroun, Czech Republic Hlucin, Czech Republic Neratovice, Czech Republic Novy Jicin, Czech Republic Ostrava, Czech Republic Pardubice, Czech Republic Prague, Czech Republic Prelouc, Czech Republic Slany, Czech Republic Uhersky Brod, Czech Republic Unicov, Czech Republic Usti nad Labem, Czech Republic Germany Basenheim, Germany Berlin, Germany Cologne, Germany Dresden, Germany Heidelberg, Germany Mannheim, Germany Nuremberg, Germany Saarlouis, Germany Hungary Balatonfuered, Hungary Budapest, Hungary Satoraljaujhely, Hungary Szikszo, Hungary New Zealand Christchurch, New Zealand Wellington, New Zealand Collapse << |
| NCT01447147 | Diabetic Nephropathy ... more >> Type 2 Diabetes Mellitus Collapse << | Phase 2 | Completed | - | - |
| NCT01036438 | - | - | Completed | - | - |
| NCT01435616 | - | - | Completed | - | - |
| NCT01036438 | Venous Leg Ulcers ... more >> Mixed Leg Ulcers Collapse << | Phase 4 | Completed | - | - |
| NCT01465997 | - | - | Completed | - | - |
| NCT01020383 | Unstable Angina ... more >> Non ST Segment Elevation Myocardial Infarction (NSTEMI) Stable Angina (Associated With High Risk PCI) Collapse << | Phase 2 | Completed | - | - |
| NCT01772147 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01634152 | Asthma | Phase 3 | Completed | - | - |
| NCT01465997 | Epilepsy Mono... more >>therapy Collapse << | Phase 3 | Completed | - | - |
| NCT01634152 | - | - | Completed | - | - |
| NCT01528254 | Type 2 Diabetes Mellitus | Phase 4 | Active, not recruiting | April 18, 2019 | - |
| NCT01131676 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01582451 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01772147 | - | - | Completed | - | - |
| NCT01145560 | - | - | Completed | - | - |
| NCT01076764 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT01582451 | - | - | Completed | - | - |
| NCT01149044 | Acute Coronary Syndrome ... more >> ST Elevation Myocardial Infarction Percutaneous Coronary Intervention Collapse << | Not Applicable | Completed | - | - |
| NCT01163253 | Psoriasis | Phase 3 | Terminated | - | - |
| NCT01908140 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 3 | Completed | - | - |
| NCT01318941 | - | - | Completed | - | - |
| NCT01145560 | Severe Sepsis ... more >> Septic Shock Collapse << | Phase 2 | Completed | - | - |
| NCT01131676 | - | - | Completed | - | - |
| NCT01616459 | - | - | Completed | - | - |
| NCT01616459 | Infections, Streptococcal | Phase 2 | Completed | - | - |
| NCT01163253 | - | - | Terminated | - | - |
| NCT01908140 | - | - | Completed | - | - |
| NCT01776424 | Prevention & Control | Phase 3 | Active, not recruiting | June 30, 2021 | - |
| NCT01187407 | - | - | Completed | - | - |
| NCT01955161 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT01217697 | - | - | - | - | - |
| NCT01955161 | - | - | Completed | - | - |
| NCT01969136 | Alzheimer's Disease ... more >> Dementia Collapse << | Phase 3 | Terminated(Study has been susp... more >>ended due to clinical hold.) Collapse << | January 2017 | - |
| NCT01776424 | - | - | Active, not recruiting | - | - |
| NCT01222117 | Acute Peripheral Arterial Occl... more >>usion Collapse << | Phase 2 | Completed | - | - |
| NCT01225380 | Chronic Hepatitis C Infection | Phase 2 | Completed | - | - |
| NCT01187407 | Major Depressive Disorder (MDD... more >>) Collapse << | Phase 3 | Completed | - | - |
| NCT01380730 | Hyperlipidemia | Phase 2 | Completed | - | - |
| NCT02004392 | Alzheimer's Disease ... more >> Dementia Collapse << | Phase 3 | Terminated(Study has been susp... more >>ended due to clinical hold.) Collapse << | August 2017 | - |
| NCT01235962 | Cancer | Phase 3 | Active, not recruiting | April 15, 2019 | - |
| NCT01380730 | - | - | Completed | - | - |
| NCT01222117 | - | - | Completed | - | - |
| NCT02006641 | - | - | Completed | - | - |
| NCT01244191 | Non Squamous, Non-small-cell L... more >>ung Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT01234311 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT01877915 | Heart Failure ... more >> Coronary Artery Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01744496 | Advanced Idiopathic Parkinson'... more >>s Disease Collapse << | Phase 4 | Completed | - | United States, Arizona ... more >> 2113 Gilbert, Arizona, United States United States, California 2120 Sunnyvale, California, United States United States, Florida 2109 Tampa, Florida, United States United States, Illinois 2107 Chicago, Illinois, United States United States, Kentucky 2102 Lexington, Kentucky, United States United States, North Carolina 2118 Advance, North Carolina, United States United States, Ohio 2117 Cincinnati, Ohio, United States United States, Oklahoma 2103 Tulsa, Oklahoma, United States United States, Virginia 2101 Roanoke, Virginia, United States United States, Wisconsin 2104 Milwaukee, Wisconsin, United States Germany 1204 Gera, Germany Poland 1607 Gdansk, Poland 1603 Krakow, Poland 1609 Olsztyn, Poland Slovakia 1804 Bratislava, Slovakia 1805 Dubnica Nad Vahom, Slovakia Collapse << |
| NCT02006641 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT01285609 | Lung Cancer - Non Small Cell S... more >>quamous Collapse << | Phase 3 | Completed | - | - |
| NCT01867125 | Asthma | Phase 3 | Completed | - | - |
| NCT01439880 | Hypercholesterolemia | Phase 2 | Completed | - | - |
| NCT01285609 | - | - | Completed | - | - |
| NCT02079246 | Alzheimer's Disease | Phase 3 | Completed | - | - |
| NCT01744496 | - | - | Completed | - | - |
| NCT02079246 | - | - | Completed | - | - |
| NCT01300013 | Heart Failure | Phase 2 | Completed | - | - |
| NCT01768559 | - | - | Completed | - | - |
| NCT01768559 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01287897 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT01790438 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01287897 | - | - | Completed | - | - |
| NCT02119286 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01193335 | 13-valent Pneumococcal Vaccine... more >> Premature Birth Immunization Safety Collapse << | Phase 4 | Completed | - | Poland ... more >> NZOZ "HIPOKRATES-II" Sp. z o.o. Krakow, Poland, 31-223 Hanna Czajka Indywidualna Praktyka Specjalistyczna Lekarska Krakow, Poland, 31-302 SP ZOZ Wojewodzki Specjalistyczny Szpital im. W. Bieganskiego w Lodzi Lodz, Poland, 91-347 Specjalistyczny ZOZ nad Matka i Dzieckiem Poznan, Poland, 61-825 Szpital im. Sw. Jadwigi Slaskiej, Oddzial Pediatryczny Trzebnica, Poland, 55-100 Samodzielny Publiczny Szpital Kliniczny nr 1 we Wroclawiu Wroclaw, Poland, 50-345 Spain Hospital Universitario de Santiago de Compostela Santiago de Compostela, A Coruña, Spain, 15706 Complexo Hospitalario Xeral Cies Vigo, Pontevedra, Spain, 36204 Complejo Hospitalario DE Torrecardenas Almeria, Spain, 04009 Hospital 12 de Octubre Madrid, Spain, 28041 Hospital Universitario de La Paz Madrid, Spain, 28046 Complejo Hospitalario de Navarra Pamplona, Spain, 31008 Collapse << |
| NCT02119286 | - | - | Completed | - | - |
| NCT01345318 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT02138916 | Moderate to Very Severe Chroni... more >>c Obstructive Pulmonary Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01193335 | - | - | Completed | - | - |
| NCT01790438 | - | - | Completed | - | - |
| NCT01335464 | Pulmonary Fibrosis | Phase 3 | Completed | - | - |
| NCT01335464 | - | - | Completed | - | - |
| NCT01345318 | - | - | Completed | - | - |
| NCT02146430 | Pain Associated With Fibromyal... more >>gia Collapse << | Phase 3 | Completed | - | - |
| NCT01975389 | Cardiovascular Disease | Phase 3 | Terminated(See Detailed Descri... more >>ption) Collapse << | - | - |
| NCT01975376 | Cardiovascular Disease | Phase 3 | Terminated(See Detailed Descri... more >>ption) Collapse << | - | - |
| NCT00746681 | Overactive Bladder | Phase 2 | Completed | - | Czech Republic ... more >> Pfizer Investigational Site Hradec Kralove, Czech Republic, 500 05 Pfizer Investigational Site Praha 1, Czech Republic, 110 00 Pfizer Investigational Site Praha 3, Czech Republic, 130 00 Pfizer Investigational Site Praha 4 - Krc, Czech Republic, 140 59 Pfizer Investigational Site Usti Nad Labem, Czech Republic, 401 13 Lithuania Pfizer Investigational Site Kaunas, Lithuania, LT-47144 Pfizer Investigational Site Kaunas, Lithuania, LT-50009 Pfizer Investigational Site Vilnius, Lithuania, LT-01118 Pfizer Investigational Site Vilnius, Lithuania, LT-08661 Norway Pfizer Investigational Site Haugesund, Norway, 5507 Pfizer Investigational Site Moelv, Norway, N-2390 Pfizer Investigational Site Trondheim, Norway, 7006 Slovakia Pfizer Investigational Site Bratislava, Slovakia, 901 01 Pfizer Investigational Site Kosice, Slovakia, 040 11 Pfizer Investigational Site Martin, Slovakia Pfizer Investigational Site Skalica, Slovakia, 909 82 Sweden Pfizer Investigational Site Lulea, Sweden, 97180 Pfizer Investigational Site Norrkoping, Sweden, 601 82 Pfizer Investigational Site Stockholm, Sweden, 141 86 United Kingdom Pfizer Investigational Site Dundee, Tayside, United Kingdom, DD1 9SY Pfizer Investigational Site Bristol, United Kingdom, BS10 5NB Pfizer Investigational Site Dundee, United Kingdom, DD1 9SY Pfizer Investigational Site Plymouth, United Kingdom, PL6 8DH Collapse << |
| NCT01358357 | - | - | Completed | - | - |
| NCT01975376 | - | - | Terminated(See Detailed Descri... more >>ption) Collapse << | - | - |
| NCT01369329 | Crohn's Disease ... more >> IBD Colitis Inflammatory Bowel Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01358357 | Bipolar I Disorder | Phase 3 | Completed | - | - |
| NCT01369329 | - | - | Completed | - | - |
| NCT01369355 | Crohn's Disease ... more >> Colitis IBD Inflammatory Bowel Disease Collapse << | Phase 3 | Active, not recruiting | October 21, 2019 | - |
| NCT01690299 | Psoriasis Pso... more >>riatic Arthritis Collapse << | Phase 3 | Completed | - | - |
| NCT01532648 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT01369355 | - | - | Active, not recruiting | - | - |
| NCT01379703 | - | - | Completed | - | - |
| NCT01202565 | - | - | Completed | - | - |
| NCT02547818 | Alzheimer's Disease | Phase 3 | Recruiting | November 2019 | - |
| NCT01975389 | - | - | Terminated(See Detailed Descri... more >>ption) Collapse << | - | - |
| NCT01690299 | - | - | Completed | - | - |
| NCT01379703 | - | - | Completed | - | - |
| NCT02224157 | Asthma | Phase 3 | Completed | - | - |
| NCT01072383 | Psoriasis Vulgaris | Phase 2 | Completed | - | Czech Republic ... more >> Dermatologic Clinic Ostrava, Czech Republic Dermatologic Clinic I Prague, Czech Republic Dermatologic Clinic Prague III Prague, Czech Republic Dermatologic Clinic Prague II Prague, Czech Republic Dermatologic Clinic Ústí nad Labem, Czech Republic Hungary Dermatologic Clinic Budapest, Hungary Dermatologic Clinic Debrecen, Hungary Dermatologic Clinic Miskolc, Hungary Dermatologic Clinic Szeged, Hungary Dermatologic Clinic Szikszo, Hungary Dermatologic Clinic Szolnok, Hungary Dermatologic Clinic Szombathely, Hungary Dermatologic Clinic Veszprém, Hungary Collapse << |
| NCT01202565 | - | - | Completed | - | - |
| NCT02236611 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00811941 | Alcohol Dependence | Phase 3 | Completed | - | - |
| NCT02236611 | - | - | Completed | - | - |
| NCT01447394 | Hepatitis C Virus (HCV) | Phase 3 | Withdrawn | August 2014 | - |
| NCT02281318 | Asthma | Phase 3 | Completed | - | - |
| NCT00811941 | - | - | Completed | - | - |
| NCT02128932 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02281318 | - | - | Completed | - | - |
| NCT00537914 | Small for Gestational Age | Phase 4 | Active, not recruiting | June 30, 2021 | Belgium ... more >> Sandoz Investigative Site Edegem, Belgium Czechia Sandoz Investigative Site Hradec Králové, Czechia Sandoz Investigative Site Prague, Czechia Sandoz Investigational Site Ústí nad Labem, Czechia Georgia Sandoz Investigative Site Tiblisi, Georgia Germany Sandoz Investigative Site Munich, Germany Sandoz Investigative Site Sankt Augustin, Germany Hungary Sandoz Investigative Site Budapest, Hungary Sandoz Investigative Site Győr, Hungary Sandoz Investigative Site Miskolc, Hungary Sandoz Investigative Site Szeged, Hungary Poland Sandoz Investigative Site Bydgoszcz, Poland Sandoz Investigative Site Gdańsk, Poland Sandoz Investigative Site Katowice, Poland Sandoz Investigative Site Kielce, Poland Sandoz Investigative Site Kraków, Poland Sandoz Investigative Site Lódz, Poland Sandoz Investigative Site Poznań, Poland Sandoz Investigative Site Rzeszów, Poland Sandoz Investigative Site Szczencin, Poland Sandoz Investigative Site Warszawa, Poland Sandoz Investigative Site Wroclaw, Poland Sandoz Investigative Site Wrocław, Poland Sandoz Investigative Site Zabrze, Poland Romania Sandoz Investigative Site Bucharest, Romania Sandoz Investigative Site Cluj Napoca, Romania Sandoz Investigative Site Craiova, Romania Sandoz Investigative Site Iasi, Romania Sandoz Investigative Site Timisoara, Romania Collapse << |
| NCT02128932 | - | - | Completed | - | - |
| NCT02104674 | Asthma | Phase 3 | Completed | - | - |
| NCT01493271 | Hypertension, Glaucoma, Open-A... more >>ngle Collapse << | Phase 2 | Completed | - | United States, Georgia ... more >> Morrow, Georgia, United States, 30260 United States, North Carolina High Point, North Carolina, United States, 27262 United States, South Carolina Mt. Pleasant, South Carolina, United States, 29464 Bulgaria Sofia, Bulgaria, 1517 Czech Republic Hradec Kralove, Czech Republic, 500 05 Litomysi, Czech Republic, 570 14 Praha, Czech Republic, 100 34 Usti Nad Labem, Czech Republic, 401 13 Zlin, Czech Republic, 762 75 Hungary Budapest, Hungary, 1115 Budapest, Hungary, 1125 Esztergom, Hungary, 2500 Collapse << |
| NCT01985334 | COPD | Phase 4 | Completed | - | - |
| NCT01508286 | - | - | - | - | - |
| NCT02352363 | - | - | Completed | - | - |
| NCT02322762 | - | - | Active, not recruiting | June 30, 2019 | - |
| NCT02054130 | Asthma | Phase 2 | Completed | - | - |
| NCT02726386 | Parkinson's Disease | Phase 2 | Active, not recruiting | August 2021 | - |
| NCT01583218 | Venous Thromboembolism (VTE) | Phase 3 | Completed | - | - |
| NCT02352363 | Idiopathic Parkinson's Disease | Phase 3 | Completed | - | - |
| NCT02054130 | - | - | Completed | - | - |
| NCT02453386 | Idiopathic Parkinson's Disease | Phase 3 | Terminated(New Safety Informat... more >>ion) Collapse << | - | - |
| NCT01763866 | Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT00408954 | Prostatic Hyperplasia | Phase 2 | Completed | - | Czech Republic ... more >> Pfizer Investigational Site Brno, Czech Republic, 612 00 Pfizer Investigational Site Hradec Kralove, Czech Republic, 500 02 Pfizer Investigational Site Olomouc, Czech Republic, 775 20 Pfizer Investigational Site Usti nad Labem, Czech Republic, 401 13 Netherlands Pfizer Investigational Site Amsterdam, Netherlands, 1081 HV Pfizer Investigational Site Nijmegen, Netherlands, 6525 GA Slovakia Pfizer Investigational Site Bratislava, Slovakia, 833 05 Pfizer Investigational Site Kosice, Slovakia, 040 11 Pfizer Investigational Site Kosice, Slovakia, 041 90 Pfizer Investigational Site Martin, Slovakia, 036 59 Pfizer Investigational Site Trencin, Slovakia, 911 01 Collapse << |
| NCT02200614 | Prostate Cancer Non-Metastatic... more >> Castration-Resistant Collapse << | Phase 3 | Active, not recruiting | June 30, 2020 | - |
| NCT01788046 | Hyperparathyroidism, Secondary | Phase 3 | Completed | - | - |
| NCT01583218 | - | - | Completed | - | - |
| NCT02164513 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01785875 | - | - | Completed | - | - |
| NCT01763866 | - | - | Completed | - | - |
| NCT01788046 | - | - | Completed | - | - |
| NCT01785875 | Hyperparathyroidism, Secondary | Phase 3 | Completed | - | - |
| NCT02194699 | Uncontrolled Asthma | Phase 3 | Completed | - | - |
| NCT02187159 | Pain Associated With Fibromyal... more >>gia Collapse << | Phase 3 | Completed | - | - |
| NCT01694485 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT02164513 | - | - | Completed | - | - |
| NCT02194699 | - | - | Completed | - | - |
| NCT01696396 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT01697072 | Gastric Cancer | Phase 3 | Terminated(All Amgen sponsored... more >> AMG102 clinical studies were terminated following a pre-planned Data Monitoring Committee safety review of study 20070622.) Collapse << | - | - |
| NCT01708603 | Moderate to Severe Plaque Psor... more >>iasis Collapse << | Phase 3 | Terminated(Sponsor decision) | August 2015 | - |
| NCT01896232 | Secondary Hyperparathyroidism ... more >> Chronic Kidney Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02620020 | Low Back Pain | Phase 2 Phase 3 | Completed | - | - |
| NCT02452190 | Asthma | Phase 3 | Completed | - | - |
| NCT02107937 | Ovarian Neoplasms ... more >> Ovarian Epithelial Cancer Collapse << | Phase 2 | Recruiting | December 2023 | Czechia ... more >> Recruiting Brno, Czechia, 625 00 Recruiting Brno, Czechia, 656 53 Recruiting Hradec Králové, Czechia, 500 05 Withdrawn Jihlava, Czechia, 586 00 Recruiting Nový Jičín, Czechia, 741 01 Recruiting Olomouc, Czechia, 775 20 Recruiting Ostrava, Czechia, 708 52 Recruiting Plzeň, Czechia, 304 60 Recruiting Praha 5, Czechia, 150 06 Recruiting Praha, Czechia, 100 34 Recruiting Praha, Czechia, 128 08 Withdrawn Ústí nad Labem, Czechia, 400 01 Recruiting České Budějovice, Czechia, 370 01 Germany Withdrawn Dresden, Germany, 01465 Withdrawn Erlangen, Germany, 91054 Withdrawn Köln, Germany, 50931 Poland Not yet recruiting Bialystok, Poland, 15-276 Withdrawn Krakow, Poland, 31-501 Recruiting Lublin, Poland, 20-090 Withdrawn Poznan, Poland, 60-569 Collapse << |
| NCT02226120 | Chronic Heart Failure With Red... more >>uced Ejection Fraction Collapse << | Phase 3 | Completed | - | - |
| NCT02298192 | - | - | Completed | - | - |
| NCT02461589 | - | - | Completed | - | - |
| NCT01891864 | - | - | Completed | - | - |
| NCT02298192 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT02534935 | Meningococcal B Disease | Phase 2 | Active, not recruiting | March 2021 | Australia, Australian Capital ... more >>T The Canberra Hospital, Paediatric Research and Clinical Trials Canberra, Garran, Australian Capital T, Australia, 2605 Australia, New South Wales Australian Clinical Research Network Maroubra, New South Wales, Australia, 2035 Maroubra Medical Centre Maroubra, New South Wales, Australia, 2035 Australia, South Australia Women's And Children's Hospital North Adelaide, South Australia, Australia, 5006 Australia, Victoria Murdoch Children's Research Institute, Royal Children's Hospital Melbourne Parkville, Victoria, Australia, 3052 Australia, Western Australia Princess Margaret Hospital for Children Subiaco, Western Australia, Australia, 6008 Czechia Ordinace praktickeho lekare pro deti a dorost Jindrichuv Hradec, Czechia, 377 01 Samostatna Ordinace Praktickeho Lekare Pro Deti A Dorost Jindrichuv Hradec, Czechia, 377 01 Medicentrum 6 s.r.o Praha 6 - Vokovice, Czechia, 160 00 Prakticky Lekar Pro Deti A Mladez Tynec Nad Sazavou, Czechia, 257 41 Finland Espoo Vaccine Research Clinic Espoo, Finland, 02230 Helsinki South Vaccine Research Clinic Helsinki, Finland, 00100 Helsinki East Vaccine Research Clinic Helsinki, Finland, 00930 Jarvenpaa Vaccine Research Clinic Jarvenpaa, Finland, 04400 Pori Vaccine Research Clinic Pori, Finland, 28100 Tampere Vaccine Research Clinic Tampere, Finland, 33100 Turku Vaccine Research Clinic Turku, Finland, 20520 Poland NZOZ Vitamed Bydgoszcz, Poland, 85-079 Prywatny Gabinet Lekarski dr n. med. Jerzy Brzostek Debica, Poland, 39-200 Indywidualna Specjalistyczna Praktyka Lekarska Hanna Czajka Krakow, Poland, 31-302 Specjalistyczna Praktyka Lekarska GRAVITA Lodz, Poland, 91-347 "NZOZ Praktyka Lekarza Rodzinnego ""Eskulap"" sp. z o.o." Lublin, Poland, 20-044 Niepubliczny Zaklad Lecznictwa Ambulatoryjnego "Michalkowice"Jarosz i Partnerzy Spolka Lekarska Siemianowice Slaskie, Poland, 41-103 Szpital im. Sw. Jadwigi Slaskiej w Trzebnicy Oddzial Pediatryczny Trzebnica, Poland, 55-100 Samodzielny Publiczny Szpital Kliniczny nr 1 we Wroclawiu, Klinika Pediatrii i Chorob Infekcyjnych Wroclaw, Poland, 50-368 Collapse << |
| NCT02102204 | - | - | Completed | - | - |
| NCT02102204 | Hyperparathyroidism, Secondary | Phase 3 | Completed | - | - |
| NCT01891864 | Chronic Stable Plaque Psoriasi... more >>s Collapse << | Phase 3 | Completed | - | - |
| NCT01951326 | Crohn's Disease | Phase 3 | Active, not recruiting | April 2019 | - |
| NCT01949311 | Atopic Dermatitis | Phase 3 | Enrolling by invitation | December 2018 | - |
| NCT01896232 | - | - | Completed | - | - |
| NCT02534935 | - | - | Active, not recruiting | - | - |
| NCT01951586 | Non-Small Cell Lung Cancer | Phase 2 | Completed | - | - |
| NCT02346240 | Psoriasis Pla... more >>que Psoriasis Collapse << | Phase 3 | Active, not recruiting | December 11, 2018 | - |
| NCT01054885 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01201356 | Relapsing Forms of Multiple Sc... more >>lerosis Collapse << | Phase 3 | Completed | - | - |
| NCT01054885 | - | - | Completed | - | - |
| NCT01951586 | - | - | Completed | - | - |
| NCT00941798 | Asthma | Phase 2 | Completed | - | - |
| NCT00941798 | - | - | Completed | - | - |
| NCT00439725 | Venous Thromboembolism | Phase 3 | Completed | - | - |
| NCT00439725 | - | - | Completed | - | - |
| NCT02346240 | - | - | Active, not recruiting | - | - |
| NCT00439777 | Pulmonary Embolism | Phase 3 | Completed | - | - |
| NCT00439777 | - | - | Completed | - | - |
| NCT00440193 | Venous Thrombosis | Phase 3 | Completed | - | - |
| NCT00552058 | - | - | Completed | - | - |
| NCT00552058 | Crohn Disease | Phase 3 | Completed | - | - |
| NCT00445185 | Hepatitis B | Phase 3 | Completed | - | Belgium ... more >> O.L.Vrouwziekenhuis Aalst Aalst, Belgium, 9300 RHMS La Madeleine ATH Ath, Belgium, 7800 RHMS Clinique Louis Caty Baudour Baudour, Belgium, 7331 CHU Brugmann (site V Horta) Service de néphrologie Bruxelles, Belgium, 1020 ULB Hôpital Erasme Département de Néphrologie Bruxelles, Belgium, 1070 AZ -VUB Dienst Nefrologie Bruxelles, Belgium, 1090 Cliniques universitaires Saint Luc Bruxelles, Belgium, 1200 CHU Hôpital civil de Charleroi, Belgium, 6000 UZ Gent Gent, Belgium, 9000 CHU Tivoli La Louvière, Belgium, 7100 UZ Gasthuisberg Leuven Nierziekten Leuven, Belgium, 3000 CHU Andre VESALE Montigny Le Tilleul, Belgium, 6110 RHMS TournayService de néphrologie Tournai, Belgium, 7500 Czech Republic Hospital JihlavaVrchlického Jihlava, Czech Republic, 59586 33 Regional Hospital Liberec Liberec, Czech Republic, 46063 University Hospital with Outpatient Clinic Ostrava Ostrava, Czech Republic, 708 52 Dept. of Internal Medicine StrahovSermirska 5 Prague, Czech Republic, 169 00 Masaryk´s Hospital Socialni pece 3316/12A Usti Nad Labem, Czech Republic, 401 13 Hungary St. Rókus Hospital Budapest, Hungary, 1085 St. István Hospital Budapest, Hungary, 1096 Petz Aladár Teaching Hospital Győr, Hungary, 9023 Pest County Flór Ferenc Hospital Kistarcsa, Hungary, 2143 Vas and Szombathely County Markusovszky Hospital Szombathely, Hungary, 9700 Collapse << |
| NCT00696813 | - | - | Completed | - | - |
| NCT00457002 | Venous Thrombosis ... more >> Pulmonary Embolism Collapse << | Phase 3 | Completed | - | - |
| NCT00440193 | - | - | Completed | - | - |
| NCT00848549 | - | - | Completed | - | - |
| NCT00848549 | Epilepsy | Phase 3 | Completed | - | - |
| NCT01106677 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01106677 | - | - | Completed | - | - |
| NCT00580216 | Atrial Fibrillation | Phase 3 | Terminated(early discontinuati... more >>on based on strategic sponsor decision not driven by any safety concern) Collapse << | - | - |
| NCT00988156 | - | - | Completed | - | - |
| NCT00457392 | Carcinoma, Non-Small Cell Lung | Phase 3 | Completed | - | - |
| NCT00457002 | - | - | Completed | - | - |
| NCT00326976 | Myocardial Ischemia ... more >> Coronary Disease Myocardial Infarction Collapse << | Phase 2 | Completed | - | - |
| NCT00988156 | Partial Epilepsy in Children a... more >>nd Adolescents Collapse << | Phase 3 | Completed | - | - |
| NCT00480025 | Lung Cancer, Non-Small Cell | Phase 3 | Terminated(The study was termi... more >>nated following assessment of the lack of efficacy of the study product by the Independent Data monitoring Committee for the study.) Collapse << | - | - |
| NCT00457392 | - | - | Completed | - | - |
| NCT00894868 | Congestive Heart Failure ... more >> Type II Diabetes Mellitus Collapse << | Phase 4 | Completed | - | - |
| NCT00490139 | Neoplasms, Breast | Phase 3 | Active, not recruiting | July 1, 2021 | - |
| NCT00490139 | - | - | Active, not recruiting | - | - |
| NCT00501046 | - | - | Completed | - | - |
| NCT00500331 | - | - | Completed | - | - |
| NCT00505388 | - | - | Completed | - | - |
| NCT00633893 | Venous Thrombosis | Phase 3 | Completed | - | - |
| NCT00514683 | Pulmonary Fibrosis | Phase 2 | Completed | - | - |
| NCT00633893 | - | - | Completed | - | - |
| NCT00501046 | Chronic Kidney Disease | Phase 3 | Completed | - | - |
| NCT00500331 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00523952 | Dermatitis, Atopic | Phase 3 | Completed | - | Czech Republic ... more >> Brno, Czech Republic Hradec Kralove, Czech Republic Ostrava, Czech Republic Plzen, Czech Republic Prague, Czech Republic Tabor, Czech Republic Usti nad Labem, Czech Republic Hungary Budapest, Hungary Debrecen, Hungary Györ, Hungary Kecskemet, Hungary Miskolc, Hungary Pecs, Hungary Poland Bialystok, Poland Czestochowa, Poland Gdansk, Poland Katowice, Poland Krakow, Poland Lodz, Poland Lublin, Poland Poznan, Poland Sopot, Poland Szczecin, Poland Warsaw, Poland Wroclaw, Poland Collapse << |
| NCT00514683 | - | - | Completed | - | - |
| NCT00524433 | Acute Heart Failure ... more >> Acute Decompensation of Chronic Heart Failure New Onset of Heart Failure Collapse << | Phase 3 | Completed | - | - |
| NCT00510406 | Lower Urinary Tract Symptoms ... more >> Prostatic Hyperplasia Collapse << | Phase 2 | Completed | - | - |
| NCT00523991 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00554229 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT00542815 | Chronic Kidney Disease ... more >> Dialysis Hyperphosphatemia Collapse << | Phase 3 | Completed | - | - |
| NCT00542815 | - | - | Completed | - | - |
| NCT00554229 | - | - | Completed | - | - |
| NCT00600821 | Non-Small-Cell Lung Carcinoma ... more >> Adenocarcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT00523991 | - | - | Completed | - | - |
| NCT00604214 | Sepsis | Phase 3 | Completed | - | - |
| NCT01224106 | Alzheimer's Disease | Phase 3 | Active, not recruiting | July 22, 2020 | - |
| NCT01029886 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT00614939 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00606450 | Psoriasis | Phase 2 | Completed | - | Canada, Alberta ... more >> Division of Dermatology and Cutaneous Science Edmonton, Alberta, Canada Canada, British Columbia Division of Dermatology Vancouver, British Columbia, Canada Canada, New Brunswick Duronder C.P. Inc Moncton, New Brunswick, Canada, E1C 8X3 Canada, Nova Scotia Eastern Canada Cutaneous Research Associates Halifax, Nova Scotia, Canada, B3H 1Z4 Canada, Ontario Ultranova Skincare Barrie, Ontario, Canada, L4M 6L2 Dermatrials Research Hamilton, Ontario, Canada, L8N 1V6 The Lynde Center for Dermatology Markham, Ontario, Canada, L3P 7N8 North Bay Dermatology Centre North Bay, Ontario, Canada, P1B3Z7 K. Papp Clinical Research Waterloo, Ontario, Canada, L3P 7N8 Canada, Quebec Innovaderm Montreal, Quebec, Canada, H2K 4L5 Dr Yves Poulin Quebec City, Quebec, Canada, G1V 4X7 Czechia Department of Dermatology Brno, Czechia Department of Dermatology Hradec Kralove, Czechia Department of Dermatovererology Olomouc, Czechia Department of Dermatovererology Praha, Czechia Depart of Dermatology Usti nad Labem, Czechia Germany Celgene Clinical Site Ausburg, Germany Celgene Clinical Site Berlin, Germany Department of Dermatologie and Venerology Dresden, Germany Department of Dermatology and Venerology Frankfurt Main, Germany Celgene Clinical Site Hamburg, Germany Celgene Clinical Site Heidelberg, Germany Celgene Clinical Site Herborn, Germany Celgene Clinical Site Homburg, Germany Celgene Clinical Site Leipzig, Germany Celgene Clinical Site Mannheim, Germany Celgene Clinical Site Munster, Germany Celgene Clinical Site Salzwedel, Germany Celgene Clinical Site Schwerin, Germany Celgene Clinical Site Wiesbaden, Germany Celgene Clinical Site Wurzburg, Germany Collapse << |
| NCT00615199 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT00600821 | - | - | Completed | - | - |
| NCT00615199 | - | - | Completed | - | - |
| NCT00631371 | Renal Cell Carcinoma | Phase 3 | Completed | - | - |
| NCT00604214 | - | - | Completed | - | - |
| NCT01029886 | - | - | Completed | - | - |
| NCT01313650 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT00980798 | Pain Osteoart... more >>hritis, Hip Osteoarthritis, Knee Collapse << | Phase 3 | Completed | - | Czech Republic ... more >> Klatovy, Czech Republic Olomouc, Czech Republic Pelhøimov, Czech Republic Praha 2, Czech Republic Praha 3, Czech Republic Roudnice Nad Labem, Czech Republic Romania Bucharest, Romania Bucuresti, Romania Cluj-Napoca, Romania Iasi, Romania Slovakia Bratislava, Slovakia Hlohovec, Slovakia Piestany, Slovakia United Kingdom London, United Kingdom Collapse << |
| NCT00614939 | - | - | Completed | - | - |
| NCT00980798 | - | - | Completed | - | - |
| NCT01313650 | - | - | Completed | - | - |
| NCT00631371 | - | - | Completed | - | - |
| NCT00637377 | Macular Degeneration | Phase 3 | Completed | - | - |
| NCT00642850 | Anemia | Phase 3 | Completed | - | Czech Republic ... more >> Brno, Czech Republic, 656 91 Cesky Krumlov, Czech Republic, 38127 Decin, Czech Republic, 405 99 Havirov, Czech Republic, 736 24 Hradec Kralove, Czech Republic, 500 05 Jihlava, Czech Republic, 586 33 Karlovy Vary, Czech Republic, 360 73 Kolin Iii, Czech Republic, 280 20 Liberec, Czech Republic, 460 63 Novy Jicin, Czech Republic, 741 11 Olomouc, Czech Republic, 775 20 Ostrava, Czech Republic, 708 52 Pisek, Czech Republic, 397 23 Praha 6, Czech Republic, 169 00 Praha, Czech Republic, 128 08 Praha, Czech Republic, 14000 Praha, Czech Republic, 150 30 Strakonice, Czech Republic, 38629 Sumperk, Czech Republic, 787 01 Tabor, Czech Republic, 390 03 Teplice, Czech Republic, 415 01 Trebic, Czech Republic, 674 01 Usti Nad Labem, Czech Republic, 401 13 Znojmo, Czech Republic, 76275 Collapse << |
| NCT00642850 | - | - | Completed | - | - |
| NCT00801437 | - | - | Completed | - | Slovakia ... more >> Pfizer Investigational Site Banovce n. Bebravou, Slovakia, 957 01 Pfizer Investigational Site Banska Bystrica, Slovakia, 975 17 Pfizer Investigational Site Bojnice, Slovakia, 972 01 Pfizer Investigational Site Bratislava, Slovakia, 811 05 Pfizer Investigational Site Bratislava, Slovakia, 820 07 Pfizer Investigational Site Bratislava, Slovakia, 833 01 Pfizer Investigational Site Bratislava, Slovakia, 851 04 Pfizer Investigational Site Brezno, Slovakia, 977 01 Pfizer Investigational Site Dunajska Streda, Slovakia Pfizer Investigational Site Galanta, Slovakia Pfizer Investigational Site Kezmarok, Slovakia, 060 01 Pfizer Investigational Site Kosice, Slovakia, 040 01 Pfizer Investigational Site Kosice, Slovakia, 04004 Pfizer Investigational Site Krompachy, Slovakia, 053 42 Pfizer Investigational Site Lipany, Slovakia, 080 01 Pfizer Investigational Site Lucenec, Slovakia, 984 01 Pfizer Investigational Site Martin, Slovakia, 036 01 Pfizer Investigational Site Myjava, Slovakia, 907 13 Pfizer Investigational Site Nitra, Slovakia Pfizer Investigational Site Nova Bana, Slovakia, 968 01 Pfizer Investigational Site Nove Mesto n. Vahom, Slovakia, 915 01 Pfizer Investigational Site Nove Zamky, Slovakia Pfizer Investigational Site Poprad, Slovakia, 058 01 Pfizer Investigational Site Presov, Slovakia, 080 01 Pfizer Investigational Site Rimavska Sobota, Slovakia, 979 01 Pfizer Investigational Site Roznava, Slovakia, 048 01 Pfizer Investigational Site Ruzomberok, Slovakia, 034 01 Pfizer Investigational Site Sabinov, Slovakia, 083 01 Pfizer Investigational Site Sala, Slovakia Pfizer Investigational Site Sliac, Slovakia, 962 31 Pfizer Investigational Site Snina, Slovakia, 06901 Pfizer Investigational Site Spisska Nova Ves, Slovakia, 052 01 Pfizer Investigational Site Stara Lubovna, Slovakia, 064 01 Pfizer Investigational Site Stara Tura, Slovakia, 916 01 Pfizer Investigational Site Topolcany, Slovakia, 955 01 Pfizer Investigational Site Ziar nad Hronom, Slovakia, 965 01 Pfizer Investigational Site Zilina, Slovakia, 010 01 Pfizer Investigational Site Zlate Moravce, Slovakia Pfizer Investigational Site Zvolen, Slovakia, 960 01 Collapse << |
| NCT00656890 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT00799903 | - | - | Completed | - | - |
| NCT00808067 | Atrial Fibrillation | Phase 3 | Completed | - | - |
| NCT00637377 | - | - | Completed | - | - |
| NCT00799903 | Atherosclerosis | Phase 3 | Completed | - | - |
| NCT00679588 | Venous Thromboembolism | Phase 3 | Completed | - | - |
| NCT00680186 | Thromboembolism | Phase 3 | Completed | - | - |
| NCT00657709 | Serogroup B Meningococcal Meni... more >>ngitis Collapse << | Phase 3 | Completed | - | - |
| NCT00676650 | Prostatic Neoplasms | Phase 3 | Terminated(Study A6181120 was ... more >>prematurely discontinued due to futility on 27 September 2010. No new or unexpected safety issues were identified.) Collapse << | - | - |
| NCT00680186 | - | - | Completed | - | - |
| NCT00676650 | - | - | Terminated(Study A6181120 was ... more >>prematurely discontinued due to futility on 27 September 2010. No new or unexpected safety issues were identified.) Collapse << | - | - |
| NCT00688688 | Urinary Bladder, Overactive | Phase 3 | Completed | - | - |
| NCT00657709 | - | - | Completed | - | - |
| NCT00683176 | Diabetic Macular Edema | Phase 2 | Completed | - | Bulgaria ... more >> Site Reference ID/Investigator# 54268 Sofia, Bulgaria, 1000 Site Reference ID/Investigator# 54291 Sofia, Bulgaria, 1233 Site Reference ID/Investigator# 54279 Sofia, Bulgaria, 1408 Site Reference ID/Investigator# 54273 Sofia, Bulgaria, 1517 Czech Republic Site Reference ID/Investigator# 54267 Brno, Czech Republic, 62500 Site Reference ID/Investigator# 54282 Ostrava-Vitkovice, Czech Republic, 703 84 Site Reference ID/Investigator# 54283 Prague, Czech Republic, 169 02 Site Reference ID/Investigator# 54270 Usti nad Labem, Czech Republic, 40113 Denmark Site Reference ID/Investigator# 54274 Glostrup, Denmark, 2600 Germany Site Reference ID/Investigator# 54263 Leipzig, Germany, 04103 Site Reference ID/Investigator# 54284 Muenster, Germany, 48145 Hungary Site Reference ID/Investigator# 54269 Budapest, Hungary, 1106 Site Reference ID/Investigator# 54277 Gyor, Hungary, H-9023 Site Reference ID/Investigator# 54275 Zalaegerszeg - Pozva, Hungary, 8900 Italy Site Reference ID/Investigator# 54272 Udine, Italy, 33100 Netherlands Site Reference ID/Investigator# 54293 Amsterdam, Netherlands, 1105 AZ Poland Site Reference ID/Investigator# 54265 Gdansk, Poland, 80-147 Site Reference ID/Investigator# 54266 Katowice, Poland, 40-760 Spain Site Reference ID/Investigator# 54280 Madrid, Spain, 28002 Site Reference ID/Investigator# 54264 Santiago de Compostela, Spain, 15705 United Kingdom Site Reference ID/Investigator# 54271 Liverpool, United Kingdom, L7 8XP Collapse << |
| NCT00688688 | - | - | Completed | - | - |
| NCT00689104 | Urinary Bladder, Overactive | Phase 3 | Completed | - | - |
| NCT00808067 | - | - | Completed | - | - |
| NCT00831441 | Acute Coronary Syndrome | Phase 3 | Terminated | - | - |
| NCT00699751 | Hormone Refractory Prostate Ca... more >>ncer Bone Metastases Collapse << | Phase 3 | Completed | - | - |
| NCT00878501 | Pain | Phase 2 | Terminated(The study was termi... more >>nated after the planned interim analysis.) Collapse << | - | - |
| NCT00630279 | Pancreatic Exocrine Insufficie... more >>ncy Due to Chronic Pancreatitis Collapse << | Phase 2 | Terminated(This trial disconti... more >>nued March 7,2009 due to high screen failure rate.) Collapse << | - | - |
| NCT00831441 | - | - | Terminated | - | - |
| NCT00699751 | - | - | Completed | - | - |
| NCT00689104 | - | - | Completed | - | - |
| NCT00878501 | - | - | Terminated(The study was termi... more >>nated after the planned interim analysis.) Collapse << | - | - |
| NCT01392300 | - | - | Completed | - | - |
| NCT00737568 | Hepatitis B | Phase 3 | Completed | - | - |
| NCT01392300 | Post-stroke Spasticity of the ... more >>Upper Limb Collapse << | Phase 3 | Completed | - | - |
| NCT01427270 | Low Back Pain | Phase 3 | Completed | - | - |
| NCT00925587 | Anemia Chroni... more >>c Kidney Disease Collapse << | Phase 3 | Completed | - | - |
| NCT01464307 | Post-stroke Spasticity of the ... more >>Lower Limb Collapse << | Phase 3 | Completed | - | - |
| NCT00737568 | - | - | Completed | - | - |
| NCT01336023 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT01337089 | - | - | Completed | - | - |
| NCT01337089 | Pain Advanced... more >> Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT00925587 | - | - | Completed | - | - |
| NCT00787319 | - | - | Completed | - | Czechia ... more >> Pfizer Investigational Site Brno, Czechia, 625 00 Pfizer Investigational Site Brno, Czechia, 62500 Pfizer Investigational Site Olomouc, Czechia, 775 20 Pfizer Investigational Site Olomouc, Czechia, 77520 Pfizer Investigational Site Ostrava - Poruba, Czechia, 70852 Pfizer Investigational Site Plzen, Czechia, 304 60 Pfizer Investigational Site Praha 2, Czechia, 128 08 Pfizer Investigational Site Praha 2, Czechia, 12808 Pfizer Investigational Site Praha 6, Czechia, 169 00 Pfizer Investigational Site Praha 6, Czechia, 169 02 Pfizer Investigational Site Usti nad Labem, Czechia, 40113 Collapse << |
| NCT01464307 | - | - | Completed | - | - |
| NCT00787319 | - | - | Completed | - | - |
| NCT01336023 | - | - | Completed | - | - |
| NCT01340027 | Urologic Diseases ... more >> Urinary Bladder Diseases Urological Manifestations Signs and Symptoms Urinary Bladder, Overactive Collapse << | Phase 2 | Completed | - | - |
| NCT00787202 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT00787202 | - | - | Completed | - | - |
| NCT00790907 | - | - | Completed | - | - |
| NCT01478607 | Painful Diabetic Peripheral Ne... more >>uropathy (PDPN) Collapse << | Phase 3 | Completed | - | - |
| NCT01340027 | - | - | Completed | - | - |
| NCT00790907 | Acute Coronary Syndrome | Phase 4 | Completed | - | - |
| NCT00806923 | Non-Squamous Non-Small Cell Lu... more >>ng Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT01481116 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT00799877 | - | - | Active, not recruiting | September 15, 2022 | - |
| NCT01361607 | Pain Advanced... more >> Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT01361607 | - | - | Completed | - | - |
| NCT01516541 | Cardiovascular Disease, Corona... more >>ry Heart Disease, Dyslipidemia, Peripheral Arterial Disease (PAD) Collapse << | Phase 3 | Completed | - | - |
| NCT01243177 | Epilepsy Mono... more >>therapy Collapse << | Phase 3 | Completed | - | - |
| NCT00800748 | Hepatitis C, Chronic | Phase 4 | Completed | - | Czechia ... more >> Beroun, Czechia Brno, Czechia, 62500 Ceska Lipa, Czechia Ceské Budejovice, Czechia, 370 87 Chomutov, Czechia Decin, Czechia, 0 Havirov, Czechia Havlickuv Brod, Czechia Hradec Kralove, Czechia, 500 12 Hradec Kralove, Czechia Jablonec/nisou, Czechia, 466 60 Jihlava, Czechia Karlovy Vary, Czechia Kolin, Czechia Liberec, Czechia Melnik, Czechia Most, Czechia Olomouc, Czechia Opava, Czechia Ostrava, Czechia, 708 52 Pardubice, Czechia Praha 2, Czechia, 128 08 Praha, Czechia, 00000 Praha, Czechia, 140 00 Praha, Czechia, 180 01 Praha, Czechia Prostejov, Czechia Usti Nad Labem, Czechia Slovakia Banska Bystrica, Slovakia, 957 17 Bratislava, Slovakia, 833 05 Bratislava, Slovakia, 851 07 Kosice, Slovakia, 040 01 Kosice, Slovakia, 04001 Martin, Slovakia, 036 59 Trencin, Slovakia, 911 07 Collapse << |
| NCT01481116 | - | - | Terminated(Due to potential co... more >>ncerns about liver safety (See Detailed Description)) Collapse << | - | - |
| NCT01243177 | - | - | Completed | - | - |
| NCT01000727 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT00847145 | - | - | Completed | - | - |
| NCT00847145 | Meningococcal Disease | Phase 3 | Completed | - | - |
| NCT00876421 | Overactive Bladder | Phase 2 | Completed | - | - |
| NCT01424228 | Constipation | Phase 4 | Completed | - | - |
| NCT01476475 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01476475 | - | - | Completed | - | - |
| NCT01627327 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 3 | Completed | - | - |
| NCT01717638 | Meningococcal Disease ... more >> Meningococcal Meningitis Collapse << | Phase 3 | Completed | - | Czech Republic ... more >> Ordinace praktickeho lekare pro deti a dorost Jaromer, Alšova 466, Czech Republic, 55101 Ordinace praktickeho lekare pro deti a dorost Jaromer, Dr. E.Beneše 191, Czech Republic, 55101 Ordinace praktickeho lekare pro deti a dorost Sezemice, Havlickova 168, Czech Republic, 53304 Ordinace praktickeho lekare pro deti a dorost Hronov, Hostovského 485, Czech Republic, 54931 Ordinace praktickeho lekare pro deti a dorost Ceska Skalice, Husovo namesti 36, Czech Republic, 55203 Ordinace praktickeho lekare pro deti a dorost Pardubice, L.Male 656, Czech Republic, 53012 Ordinace praktickeho lekare pro deti a dorost Hradec Králové, Manesova 646, Czech Republic, 50002 Ordinace praktickeho lekare pro deti a dorost Hronov, Palackeho 517, Czech Republic, 54931 Ordinace praktickeho lekare pro deti a dorost Hradec Králové, Pardubicka 752, Czech Republic, 50004 Ordinace praktickeho lekare pro deti a dorost Chlumec nad Cidlinou, Pernstynska 127/I, Czech Republic, 50351 Nemocnice Náchod Nachod, Purkynova 446, Czech Republic, 54701 Ordinace praktickeho lekare pro deti a dorost Jindrichuv Hradec, Ruských legií 352, Czech Republic, 37701 Ordinace praktickeho lekare pro deti a dorost Pardubice, Sladkovskeho 2617, Czech Republic, 53002 Ordinace praktickeho lekare pro deti a dorost Jindrichuv Hradec, Sídliste Vajgar 724/III, Czech Republic, 37701 Fakulta vojenskeho zdravotnictvi UO Hradec Kralove, Trebesska 1575, Czech Republic, 50001 Ordinace praktickeho lekare pro deti a dorost Holice, U kaplicky 1042, Czech Republic, 53401 Ordinace praktickeho lekare pro deti a dorost Jindrichuv Hradec, U nemocnice380/III, Czech Republic, 37701 Italy Universita di Firenze -Pediatria Florence, Italy, 50139 IRCCS Cà Granda Milan, Italy, 20122 Ospedale Maggiore della Carita Novara, Italy, 28100 Dip Pediatria AO Padova Padova, Italy, 35128 Spain Hospital Clinico Universitario de Santiago de Compostela Santiago de Compostela A Coruña, Spain, 15706 Hospital Universitario Dr. Peset Valencia, Spain, 46017 Centro Superior de Investigacion en Salud Publica/Clinica Universitaria San Vicente Martir Valencia, Spain, 46020/46001 Complexo Hospitalario Xeral Cies Vigo Pontevedra, Spain, 36204 United Kingdom Oxford Vaccine Group - Centre for Clinical Vaccinology and Tropical Medicine Churchill Hospital Oxford, Headington, United Kingdom, OX3 7LJ North Bristol NHS Trust Bristol, United Kingdom, BS1 3NU Royal Devon and Exeter NHS Foundation Trust Exeter, United Kingdom, EX2 5DW St Georges Hospital London, United Kingdom, SW17 0RE Collapse << |
| NCT01627327 | - | - | Completed | - | - |
| NCT01717638 | - | - | Completed | - | - |
| NCT01000727 | - | - | Completed | - | - |
| NCT00925600 | Cancer Catara... more >>ct Low Bone Mineral Density Osteopenia Osteoporosis Prostate Cancer Collapse << | Phase 3 | Completed | - | - |
| NCT01466088 | Alzheimer's Disease | Phase 2 | Completed | - | United States, Arizona ... more >> Banner Alzheimer Institute Phoenix, Arizona, United States, 85006 United States, Florida Meridien Research Brooksville, Florida, United States, 34601 MD Clinical Hallandale Beach, Florida, United States, 33009 Czech Republic Policlinic Chocen, Czech Republic BRAIN-SOULTHERAPY s.r.o. Kladno, Czech Republic Bialbi.s.r.o. Psychiatrické oddělení Litoměřice, Czech Republic Psychiatricka Ambulance Olomouc, Czech Republic Vojenska Nemocnice Psychiatricke oddeleni Olomouc, Czech Republic Clintrial Praha, Czech Republic PRAGTIS s.r.o. Praha, Czech Republic Psychosocialni centrum Prerov, Czech Republic Romania Crucea Alba - Dr. Oros si Asociatii - Societate Civila Medicala Oradea, Romania, 410163 Spitalul de Psihiatrie Sibiu Sibiu, Romania, 550012 Spitalul Clinic Judetean de Urgenta SIBIU Sibiu, Romania, 550166 Spitalul Clinic Judetean de Urgenta, Clinica Psihiatrie Timisoara, Romania, 300128 Spitalul Clinic Judetean de Urgenta, Clinica Neurologie 2 Timisoara, Romania, 300736 Slovakia Neurologicka Ambulancia, s.r.o. Banska Bystrica, Slovakia Univerzitna nemocnica Bratislava, Geronto-psychiatrické oddelenie Bratislava, Slovakia KONZILIUM s.r.o. Dubnica nad Váhom, Slovakia Neurologická ambulancia Krompachy, Slovakia Ukraine Dnipropetrovsk Regional Clinical Hospital Dnipropetrovsk, Ukraine, 49005 Donetsk National Medical University of M. Gorky Donetsk, Ukraine, 83003 Donetsk Regional Clinical Psychiatric Hospital Donetsk, Ukraine, 83008 Crimean Republican Institution Psychoneurological Dispensary Kerch, Ukraine, 98310 Central Clinical Hospital Ukrzaliznytsi Kharkiv, Ukraine, 61103 Department of Age Physiology and Pathology of Nervous System; Institute of Gerontology NAMS Kyiv, Ukraine, 04114 Lugansk Regional Clinical Psychoneurological Hospital Lugansk, Ukraine, 91045 Lviv National Medical University named after Galytskyy Lviv, Ukraine, 79010 Lviv Regional Clinical Psychiatric Hospital Lviv, Ukraine, 79021 Odessa Regional Psychoneurology Dispensary Odessa, Ukraine, 65014 Odessa Regional Psychiatric Hospital # 2 Oleksandrivka, Ukraine, 67513 Ukrainian Medical Stomatological Academy Poltava, Ukraine, 36006 Department #3 of the Kherson Regional Psychiatric Hospital Stepanovka, Ukraine, 73488 Collapse << |
| NCT01424228 | - | - | Completed | - | - |
| NCT00938730 | Atrial Fibrillation | Phase 2 | Completed | - | - |
| NCT01486940 | Diabetes Diab... more >>etes Mellitus, Type 1 Collapse << | Phase 3 | Completed | - | - |
| NCT00940095 | Aneurysmal Subarachnoid Hemorr... more >>hage Collapse << | Phase 3 | Terminated(Lack of efficacy da... more >>ta from the Phase 3 clinical study (AC-054-301; CONSCIOUS-2)) Collapse << | - | - |
| NCT00949286 | - | - | Completed | - | - |
| NCT00050297 | Prostate Cancer | Phase 2 | Terminated | - | Belgium ... more >> University Clinic AZ Brussels, Belgium University Clinic UCL St. Luc Brussels, Belgium Czech Republic Private Urology Out-patient Ward Jablonec nad Nisou, Czech Republic Clinic of Urology, University Hospital Králové, Czech Republic Clinic of Urology, University Hospital Olomouc, Czech Republic Clinic of Urology, University Hospital Prague, Czech Republic France Hôpital Bichat, service de Urology Paris, France Germany Haingasse 22 Bad Homburg, Germany, 61348 Klinik und Poliklinik für Urologie, Universitätsklinikum Carl-Gustav-Carus Dresden, Germany Klinik u. Poliklinik für Urologie, Philipps Universität, Baldingerstraße Marburg, Germany Krummbogen 15 Marburg, Germany Poland Regional Hospital, Department of Urology Bydgoszcz, Poland MedSource Poland Gdansk, Poland Bielanski Hospital, Department of Urology Warszawa, Poland Oncology Center, Department of Urinary Tract Cancer Warszawa, Poland Medical University, Clinic of Urology Wroclaw, Poland Spain Complejo Hospitaliaro Juan Canalejo, Servicio de Urología Corunna, Spain Fundacion Hospital Alcorcón, Servicio Urología Madrid, Spain Hospital Universitario Principe de Asturias, Servicio de Urología, Ctra. Madrid, Spain United Kingdom Department of Urology, Bristol Royal Infirmary Bristol, United Kingdom Department of Urology, Gartnavel Hospital Glasgow, United Kingdom Department of Urology, Norfolk & Norwich Hospital Norwich, United Kingdom Department of Urology, Taunton & Somerset Hospital Taunton, United Kingdom Collapse << |
| NCT00925600 | - | - | Completed | - | - |
| NCT01517373 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00958776 | Cough Sore Th... more >>roat Nasal Congestion Headache Fever Seasonal Influenza Collapse << | Phase 3 | Terminated(This study was term... more >>inated for futility) Collapse << | - | - |
| NCT00404703 | Non-Small Cell Lung Cancer | Phase 2 | Terminated(Primary (safety) en... more >>dpoint reached) Collapse << | - | Australia ... more >> Tugun, Australia Wollongong, Australia Belgium Liege, Belgium Czech Republic Ostrava, Czech Republic Usti nad Labem, Czech Republic France Bobigny, France Marseille, France Nantes, France Hungary Szekesfehervar, Hungary Szombathely, Hungary Israel Kfar Saba, Israel Ramat Gan, Israel Poland Lublin, Poland Poznan, Poland Szczecin, Poland Warszawa, Poland Russian Federation Balashikha, Russian Federation Moscow, Russian Federation St. Petersburg, Russian Federation Spain Madrid, Spain Sevilla, Spain Taiwan Kueishan, Taiwan Taichung, Taiwan Taipei, Taiwan Taoyuan, Taiwan Collapse << |
| NCT00977080 | Chronic Kidney Disease ... more >> Secondary Hyperparathyroidism Hemodialysis Collapse << | Phase 4 | Completed | - | - |
| NCT00958776 | - | - | Terminated(This study was term... more >>inated for futility) Collapse << | - | - |
| NCT01421459 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00993473 | Type 1 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01538251 | Ulcerative Colitis | Phase 3 | Terminated(Evidence of very lo... more >>w probability to success. No safety issues.) Collapse << | - | - |
| NCT00977080 | - | - | Completed | - | - |
| NCT01517373 | - | - | Completed | - | - |
| NCT01147926 | Male Subjects With Chronic Con... more >>stipation Collapse << | Phase 3 | Completed | - | - |
| NCT00993473 | - | - | Completed | - | - |
| NCT01421459 | - | - | Completed | - | - |
| NCT00986154 | - | - | Completed | - | - |
| NCT00986154 | Venous Thromboembolism ... more >> Deep Vein Thrombosis (DVT) Pulmonary Embolism (PE) Thromboembolism Venous Thrombosis Collapse << | Phase 3 | Completed | - | - |
| NCT01147926 | - | - | Completed | - | - |
| NCT01719003 | Diabetes Mellitus, Type 2 ... more >> Hyperglycemia Collapse << | Phase 3 | Completed | - | - |
| NCT02540993 | Diabetic Kidney Disease | Phase 3 | Active, not recruiting | May 25, 2020 | - |
| NCT01719003 | - | - | Completed | - | - |
| NCT02558296 | Type 2 Diabetes Mellitus | Phase 3 | Active, not recruiting | April 2019 | - |
| NCT01970657 | - | - | Completed | - | - |
| NCT02738151 | - | - | Completed | - | - |
| NCT02738151 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01966419 | Acute Episode of Overt Hepatic... more >> Encephalopathy Hepatic Encephalopathy Collapse << | Phase 2 | Completed | - | - |
| NCT03402659 | Alzheimer Disease | Phase 2 | Recruiting | July 31, 2019 | - |
| NCT02545049 | Diabetic Kidney Disease | Phase 3 | Active, not recruiting | July 21, 2021 | - |
| NCT01972841 | Urinary Bladder Overactive ... more >> Urinary Bladder Diseases\Urologic Diseases Overactive Bladder Urgency Incontinence Collapse << | Phase 3 | Completed | - | - |
| NCT01976104 | Thrombocytopenia Associated Wi... more >>th Liver Disease Collapse << | Phase 3 | Completed | - | - |
| NCT02065349 | Post-Herpetic Neuralgia (PHN) ... more >> Neuropathic Pain Painful Diabetic Peripheral Neuropathy (PDPN) Collapse << | Phase 2 | Completed | - | Czechia ... more >> Site: CZ42003 Chocen, Czechia, 561 01 Site: CZ42004 Litomysl, Czechia, 517 14 Site: CZ42011 Olomouc, Czechia, 77900 Site: CZ42014 Praha 2, Czechia, 12000 Site: CZ42001 Rychnov nad Kneznou, Czechia, 516 01 Site: CZ42002 Slezska Ostrava, Czechia, 710 00 Germany Site: DE49003 Koeln, Germany, 50937 Site: DE49005 Neuss, Germany, 41460 Poland Site: PL48003 Bialystok, Poland, 15-950 Site: PL48004 Poznan, Poland, 60-773 Site: PL48001 Poznan, Poland, 61-655 Site: PL48002 Torun, Poland, 87-100 Site: PL48005 Warszawa, Poland, 00-465 United Kingdom Site: GB44001 Glasgow, Scotland, United Kingdom, G12OYN Site: GB44003 Ipswich, United Kingdom, IP45PD Site: GB44006 London, United Kingdom, SE17EH Site: GB44002 Manchester, United Kingdom, M320UT Collapse << |
| NCT01456052 | Ulcerative Colitis | Phase 2 | Completed | - | United States, Arkansas ... more >> Lexicon Investigational Site Little Rock, Arkansas, United States, 72205 United States, California Lexicon Investigational Site Anaheim, California, United States, 92801 Lexicon Investigational Site Santa Monica, California, United States, 90404 United States, New York Lexicon Investigational Site Great Neck, New York, United States, 11021 Lexicon Investigational Site New York, New York, United States, 10017 United States, Oklahoma Lexicon Investigational Site Tulsa, Oklahoma, United States, 74135 United States, Texas Lexicon Investigational Site Houston, Texas, United States, 77030 Lexicon Investigational Site San Antonio, Texas, United States, 78229 United States, Utah Lexicon Investigational Site Ogden, Utah, United States, 84405 Lexicon Investigational Site Sandy City, Utah, United States, 84094 United States, Washington Lexicon Investigational Site Seattle, Washington, United States, 98195 Lithuania Lexicon Investigational Site Kaunas, Lithuania, LT-50009 Lexicon Investigational Site Klaipeda, Lithuania, LT-92288 Lexicon Investigational Site Siauliai, Lithuania, LT-76231 Lexicon Investigational Site Vilnius, Lithuania, LT-03215 Lexicon Investigational Site Vilnius, Lithuania, LT-08661 Poland Lexicon Investigational Site Krakow, Poland, 31531 Lexicon Investigational Site Lodz, Poland, 90302 Lexicon Investigational Site Sopot, Poland, 81756 Lexicon Investigational Site Warszawa, Poland, 03580 Lexicon Investigational Site Wroclaw, Poland, 53025 Lexicon Investigational Site Wroclaw, Poland, 54144 Slovakia Lexicon Investigational Site Nitra, Slovakia, 94901 Lexicon Investigational Site Nove Mesto nad Vahom, Slovakia, 91501 Collapse << |
| NCT03202979 | Painful Diabetic Neuropathy | Phase 2 | Completed | - | Czechia ... more >> NEUROHK s.r.o. Chocen, Czechia, 565 01 Litnea s.r.o. Neurologicka ambulance Litomerice, Czechia, 412 01 Neurosanatio s.r.o. Litomysl, Czechia, 570 01 MP-neuro s.r.o. Poliklinika Modry pavilon Ostrava, Czechia, 710 00 Nemocnice Pardubickeho kraje a.s. Pardubicka nemocnice Neurologická klinika Pardubice, Czechia, 532 03 Diabetologicka ambulance Milan Kvapil s.r.o. Praha 4, Czechia, 149 00 Vestra Clinics s.r.o. Rychnov nad Knežnou, Czechia, 516 01 Hungary Budai Irgalmasrendi Korhaz Belgyógyászati Centrum Budapest, Hungary, 1027 Semmelweis Egyetem AOK I. sz. Belgyogyaszati Klinika Budapest, Hungary, 1083 Markhot Ferenc Oktatokorhaz es Rendelointezet Diabetesz Gondozo Eger, Hungary, 3300 Bekes Megyei Kozponti Korhaz Pandy Kalman Tagkorhaz I. sz. Belgyogyaszati Osztaly Gyula, Hungary, 5700 Bacs-Kiskun Megyei Korhaz II. sz. Belgyogyaszati Osztaly Kecskemet, Hungary, 6000 Szegedi Tudomanyegyetem Szent-Gyorgyi Albert Klinikai Kozpont I. Sz. Belgyogyaszati Klinika Szeged, Hungary, 6720 Poland Silmedic Sp. z o.o. Katowice, Poland, 40-282 Pro Familia Altera Sp. z o.o. Katowice, Poland, 40-648 NZOZ Neuromed M. i M. Nastaj Sp. P. Lublin, Poland, 20-064 RCMed Oddział Sochaczew Sochaczew, Poland, 96-500 Jeka Sławomir Niepubliczny Zakład Opieki Zdrowotnej "Nasz Lekarz" Praktyka Grupowa Lekarzy Rodzinnych z Przychodnia Specjalistyczna Torun, Poland, 87-100 NBR Polska Tomasz Klodawski Warszawa, Poland, 00-465 Medycyna Kliniczna Warszawa, Poland, 00-660 Collapse << |
| NCT02575833 | - | - | Completed | - | - |
| NCT01456052 | - | - | Completed | - | - |
| NCT01475461 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT01976104 | - | - | Completed | - | - |
| NCT01972841 | - | - | Completed | - | - |
| NCT01624259 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT01111383 | Cystic Fibrosis | Phase 3 | Completed | - | France ... more >> CHR Clemenceau Caen, France, 14 033 Hopital Arnaud de Villeneuve, Clinique des maladies respiratoires Montpellier, France, 34 295 Hopital Necker Paris, France, 75 015 Poland Specjalistyczny ZOZ nad Matka i Dzieckiem, Poradnia Leczenia Mukowiscydozy Gdansk, Poland, 80-308 I Oddzial Chorob Dzieciecych, Wojewodzki Specjalistyczny Szpital Dzieciecy Kielce, Poland, 25-381 Oddzial Kliniczny Interny Dzieciecej i Alergologii, Wojewodzki Szpital Specjalistyczny Lodz, Poland, 93-513 Dzieciecy Szpital Kliniczny Akademii Medycznej, Klinika Chorob Pluc I Reumatologii Lublin, Poland, 20-093 Klinika Pneumonologii, Alergologii Dzieciecej i Immunologii Klinicznej Szpital Kliniczny Uniwersytetu Medycznego w Poznaniu Poznan, Poland, 60-572 Klinika Pneumonologii i Mukowiscydozy, Instytut Gruzlicy i Chorob Pluc w Rabce Zdroj Rabka Zdroj, Poland, 34-700 Poradnia Mukowiscydozy Wojewodzkiej, Przychodni Specjalistycznej dla Dzieci, Szpitala Wojewodzkiego Nr 2 Rzeszow, Poland, 35-301 Klinika Pediatrii Instytut Matki I Dziecka Warszawa, Poland, 01-211 Ukraine Dnipropetrovsk City Children Clinical Hospital # 2 Dnipropetrovsk, Ukraine, 49101 Donetsk Regional Children Clinical Hospital Donetsk, Ukraine, 83052 Kriviy Rig City Clinical Hospital # 8 Kriviy Rig, Ukraine, 50047 Institute of Phthysiology and Pulmonology n.a., F.G.Yanovskiy of the Academy of Medical Science of Ukraine Kyiv, Ukraine, 03680 Institute of Pediatrics, Obstetrics and Gynecology of the Academy of Medical Science of Ukraine Kyiv, Ukraine, 04050 Lviv Regional Children Specialized Clinical Hospital Lviv, Ukraine, 79035 Odesa Regional Children Clinical Hospital Odesa, Ukraine, 65031 Simferopol Central District Clinical Hospital Simferopol, Ukraine, 95033 Zaporizhya Regional Clinical Children Hospital Zaporizhya, Ukraine Collapse << |
| NCT02575833 | Stable Angina | Phase 2 | Completed | - | - |
| NCT01624259 | - | - | Completed | - | - |
| NCT03105479 | Clostridium Difficile Infectio... more >>n Collapse << | Phase 2 Phase 3 | Terminated(Study suspended in ... more >>October 2017 and terminated April 17, 2018 after decision to discontinue the study drug development) Collapse << | - | United States, Idaho ... more >> Snake River Research, PLLC Idaho Falls, Idaho, United States, 83404 United States, Illinois University of Chicago, Dept. Of Medicine Chicago, Illinois, United States, 60637 United States, Louisiana Louisiana State University Health Sciences Center - Shreveport Shreveport, Louisiana, United States, 71103 United States, Ohio SUNY Upstate Medical University - Upstate Golisano Children's Hospital (GCH) - Pediatric Designated AIDS Center Syracuse, Ohio, United States, 13210 United States, Texas Texas Children's Hospital Feigin Cente Houston, Texas, United States, 77030 Belgium Universitair Ziekenhuis Brussel - Kinderziekenhuis Jette, Belgium, 1090 Canada Infection Prevention & Control, AGW5 Foothills Medical Center 1403 29th Street N.W. Calgary, Canada, T2N 2T9 Czechia FN Brno Brno, Czechia, 62500 Hungary Egyesített Szent István és Szent László Kórház - Rendelőintézet / Gyermekinfektológiai Osztály Budapest, Hungary, 1097 Pándy Kálmán Megyei Kórház Gyula, Hungary, 5700 Italy Ospedale Buzzi Milano, Italy, 20154 Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) - Ospedale Pediatrico Bambino Gesu Roma, Italy, 00165 Poland Wojewodzki Szpital Obserwacyjno-Zakazny im. Tadeusza Browicza Bydgoszcz, Poland, 85-030 Specjalistyczny Zespół Opieki Zdrowotnej nad Matką i Dzieckiem Poznan, Poland, 61-734 Gabinet Lekarski Bartosz Korczowski Rzeszow, Poland, 35-302 Klinika Gastroenterologii, Hepatologii, Zaburzeń Odżywiania i Pediatrii, Instytut "Pomnik - Centrum Zdrowia Dziecka" Warsaw, Poland, 04-730 Romania Institutul National De Boli Infectioase "Prof. Dr. Matei Bals", sectia IX pediatrie Bucharest, Romania, 21105 Spitalul Clinic de Boli Infectioase "Sfanta Parascheva" Iasi, Clinica de Boli Infectioase I, Iasi, Romania, 700116 Spain Hospital Sant Joan de Déu, Esplugues Barcelona, Spain, 8950 Hospital Universitario Infantil LA PAZ Madrid, Spain, 28046 Collapse << |
| NCT01475461 | - | - | Completed | - | - |
| NCT02227862 | Type 1 Diabetes | Phase 3 | Completed | - | - |
| NCT02629159 | Rheumatoid Arthritis | Phase 3 | Active, not recruiting | August 10, 2020 | - |
| NCT00090103 | Prostatic Hyperplasia | Phase 3 | Completed | - | - |
| NCT02683863 | Multiple Sclerosis | Phase 4 | Completed | - | United States, New York ... more >> Multiple Sclerosis Center of Northeastern New York Latham, New York, United States, 12110 Collapse << |
| NCT02528188 | Chronic Pain ... more >>Osteoarthritis, Hip Osteoarthritis, Knee Collapse << | Phase 3 | Active, not recruiting | February 2019 | - |
| NCT00216619 | Migraine | Phase 3 | Completed | - | - |
| NCT00048295 | Crohn's Disease | Phase 3 | Unknown | - | Belgium ... more >> Kortrijk, Belgium Roeselare, Belgium Czech Republic Hradic Kralove 2, Czech Republic Olomouc, Czech Republic Prague, Czech Republic Praha, Czech Republic Usti nad Orlici, Czech Republic Germany Minden, Germany Munster, Germany Italy Torino, Italy Poland Krakow, Poland Szczecin, Poland Warszawa, Poland Wroclaw, Poland Spain Barcelona, Spain Valencia, Spain Collapse << |
| NCT00224497 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT00280059 | - | - | Completed | - | - |
| NCT00280059 | Epilepsy, Partial | Phase 3 | Completed | - | - |
| NCT00090103 | - | - | Completed | - | - |
| NCT02709486 | Osteoarthritis, Hip ... more >> Osteoarthritis, Knee Collapse << | Phase 3 | Active, not recruiting | December 2018 | - |
| NCT03214367 | Type 1 Diabetes Mellitus | Phase 3 | Active, not recruiting | September 5, 2019 | - |
| NCT01494532 | Parkinson Disease | Phase 4 | Completed | - | - |
| NCT02674386 | Osteoarthritis | Phase 3 | Recruiting | August 12, 2019 | - |
| NCT00256178 | Hypercholesterolemia | Phase 3 | Completed | - | - |
| NCT00351611 | Epilepsies, Partial | Phase 4 | Recruiting | November 11, 2019 | - |
| NCT01064817 | Glaucoma | Phase 2 | Completed | - | Belgium ... more >> University Hospital Leuven Leuven, Belgium, B-3000 Czech Republic Oftalmologicka klinika Brno-Bohunice Brno, Czech Republic, 639 00 Facility Hospital Hradec Kralove Hradec Kralove, Czech Republic, 500 05 Palacky University Olomouc, Czech Republic, 775 20 Hospital Pardubice Pardubice, Czech Republic, 53203 Charles University Praha, Czech Republic Masaryak's Hospital Usti' Nad/Labem, Czech Republic, 40013 Netherlands UMC St. Radboud West Nijmegen, Netherlands, 6525 EX Eramus Medical Center Rotterdam, Netherlands, 3015 CE United Kingdom Cheltenham General Hospital Gloucestershire, United Kingdom, GL53 7AN St. Thomas London, United Kingdom Norfolk and Norwich University Hospital-NHS Trust Norwich, United Kingdom, NR4 7UY Oxford Eye Hosiptal Oxford, United Kingdom, OX3 9DU Royal Hallamshire Hospital Sheffield, United Kingdom, S10 2JF Collapse << |
| NCT03104400 | Psoriatic Arthritis | Phase 3 | Recruiting | September 6, 2023 | - |
| NCT01064817 | - | - | Completed | - | - |
| NCT00150813 | Epilepsy, Tonic-clonic | Phase 3 | Completed | - | Czechia ... more >> Beroun, Czechia Brno, Czechia Ceske Budejovice, Czechia Praha 1, Czechia Praha 5, Czechia Rychnov nad Kneznou, Czechia Hungary Budapest, Hungary Debrecen, Hungary Pécs, Hungary Szeged, Hungary Poland Białystok, Poland Gdańsk, Poland Katowice, Poland Lublin, Poland Szczecin, Poland Warszawa, Poland Łódź, Poland Sweden Göteborg, Sweden Helsingborg, Sweden Huddinge, Sweden Karlstad, Sweden Uppsala, Sweden Collapse << |
| NCT00368472 | Epilepsy | Phase 2 | Completed | - | - |
| NCT00175903 | Epilepsy | Phase 3 | Completed | - | - |
| NCT00318461 | Diabetes Diab... more >>etes Mellitus, Type 2 Collapse << | Phase 3 | Completed | - | - |
| NCT00150813 | - | - | Completed | - | - |
| NCT00381095 | Bone Neoplasms ... more >> Pain, Intractable Cancer Collapse << | Phase 4 | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00368472 | - | - | Completed | - | - |
| NCT00318461 | - | - | Completed | - | - |
| NCT00381095 | - | - | Terminated(See termination rea... more >>son in detailed description.) Collapse << | - | - |
| NCT00206310 | Heart Failure | Phase 3 | Completed | - | - |
| NCT00077597 | Anemia | Phase 3 | Completed | - | - |
| NCT03235050 | Diabetes Mellitus, Type 2 | Phase 2 | Active, not recruiting | March 9, 2020 | - |
| NCT01494532 | - | - | Completed | - | - |
| NCT00444925 | Overactive Bladder | Phase 3 | Completed | - | - |
| NCT00329602 | - | - | Completed | - | - |
| NCT00329602 | Restless Legs Syndrome | Phase 4 | Completed | - | - |
| NCT00537238 | Partial Seizures | Phase 3 | Completed | - | - |
| NCT00444925 | - | - | Completed | - | - |
| NCT00553267 | Hypertension | Phase 3 | Completed | - | - |
| NCT00553267 | - | - | Completed | - | - |
| NCT00537238 | - | - | Completed | - | - |
| NCT00126022 | Atrial Fibrillation ... more >> Atrial Flutter Collapse << | Phase 3 | Completed | - | - |
| NCT00095238 | Congestive Heart Failure | Phase 3 | Completed | - | - |
| NCT00095238 | - | - | Completed | - | - |
| NCT00472953 | Diabetes Diab... more >>etes Mellitus, Type 1 Diabetes Mellitus, Type 2 Chronic Obstructive Pulmonary Disease Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | Argentina ... more >> Novo Nordisk Investigational Site Buenos Aires, Argentina, B1636DSU Novo Nordisk Investigational Site Buenos Aires, Argentina, C1425AGC Novo Nordisk Investigational Site Ciudad de Buenos Aires, Argentina, 1280 India Novo Nordisk Investigational Site Chandigarh, Punjab, India, 160012 Novo Nordisk Investigational Site Vellore, Tamil Nadu, India, 632004 Novo Nordisk Investigational Site Kolkata, West Bengal, India, 700020 Novo Nordisk Investigational Site Hyderabad, India, 600034 Novo Nordisk Investigational Site Mumbai, India, 400 067 Novo Nordisk Investigational Site Pune, India, 411011 Romania Novo Nordisk Investigational Site Bucharest, Romania, 020475 Novo Nordisk Investigational Site Bucharest, Romania, 020992 Slovakia Novo Nordisk Investigational Site Kosice, Slovakia, 04190 Novo Nordisk Investigational Site Lubochna, Slovakia, 03491 Novo Nordisk Investigational Site Moldava nad Bodvou, Slovakia, 045 01 Novo Nordisk Investigational Site Zilina, Slovakia, 01001 Novo Nordisk Investigational Site Zilina, Slovakia, 01207 Taiwan Novo Nordisk Investigational Site Changhua, Taiwan, 500 Novo Nordisk Investigational Site Chiayi City, Taiwan, 600 Novo Nordisk Investigational Site Taipei, Taiwan, 231 Thailand Novo Nordisk Investigational Site Bangkok, Thailand, 10330 Novo Nordisk Investigational Site Bangkok, Thailand, 10700 Turkey Novo Nordisk Investigational Site Erzurum, Turkey, 25240 Novo Nordisk Investigational Site Gaziantep, Turkey, 27070 Novo Nordisk Investigational Site Istanbul, Turkey, 34400 Novo Nordisk Investigational Site Istanbul, Turkey, 34722 Novo Nordisk Investigational Site Istanbul, Turkey, 34890 Novo Nordisk Investigational Site Kocaeli, Turkey, 41380 Collapse << |
| NCT00508157 | - | - | Terminated(Slow Accrual) | - | - |
| NCT00153101 | Cardiovascular Diseases | Phase 4 | Completed | - | - |
| NCT00624052 | - | - | Completed | - | - |
| NCT00624052 | Hypertension | Phase 3 | Completed | - | - |
| NCT00523042 | Diabetes Diab... more >>etes Mellitus, Type 1 Diabetes Mellitus, Type 2 Asthma Collapse << | Phase 3 | Terminated(See termination rea... more >>son in detailed description) Collapse << | - | - |
| NCT00662649 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00251680 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00662649 | - | - | Completed | - | - |
| NCT03495102 | Diabetes Mellitus, Type 2 | Phase 3 | Active, not recruiting | October 7, 2019 | - |
| NCT00262600 | Atrial Fibrillation ... more >> Stroke Collapse << | Phase 3 | Completed | - | - |
| NCT00558259 | Venous Thromboembolism | Phase 3 | Completed | - | - |
| NCT00189813 | Irritable Bowel Syndrome | Phase 2 | Completed | - | - |
| NCT00508157 | Metabolic Syndrome ... more >> Schizophrenia Collapse << | Phase 4 | Terminated(Slow Accrual) | - | Belgium ... more >> Local Institution Brussels, Belgium, 1160 Czech Republic Local Institution Brno, Czech Republic, 602 00 Local Institution Brno, Czech Republic, 625 00 Local Institution Havirov, Czech Republic, 736 01 Local Institution Hradec Kralove, Czech Republic, 500 05 Local Institution Prague, Czech Republic, 190 00 Local Institution Praha 10, Czech Republic, 100 00 Local Institution Praha 6, Czech Republic, 160 00 Local Institution Prerov, Czech Republic, 750 02 Local Institution Roudnice Nad Labem, Czech Republic, 41301 France Local Institution Fains Veel, France, 55000 Local Institution Lille Cedex, France, 59037 Local Institution Limoges Cedex, France, 87025 Local Institution Montpellier, France, 34295 Local Institution Paris Cedex 14, France, 75674 Local Institution Paris, France, 75013 Local Institution Poitiers, France, 86000 Local Institution Strasbourg, France, 67000 Germany Local Institution Ellwangen, Germany, 73479 Local Institution Werneck, Germany, 97440 Greece Local Institution Chania-Crete, Greece, 73300 Local Institution Corfu, Greece, 49100 Hungary Local Institution Budapest, Hungary, 1125 Local Institution Gyula, Hungary, 5700 Spain Local Institution Oviedo, Asturias, Spain, 33011 Local Institution Barcelona, Spain, 08025 Local Institution Barcelona, Spain, 08036 Local Institution Barcelona, Spain, 08907 Switzerland Local Institution Wetzikon, Switzerland, 8620 Turkey Local Institution Izmir, Turkey, 35370 Collapse << |
| NCT00558259 | - | - | Completed | - | - |
| NCT00153101 | - | - | Completed | - | - |
| NCT00692003 | Epilepsy | Phase 3 | Terminated(Sponsor's decision) | - | - |
| NCT00693017 | - | - | Terminated(Sponsor's decision) | - | - |
| NCT00567346 | Allergic Rhinoconjunctivitis | Phase 2 Phase 3 | Completed | - | - |
| NCT00693017 | Epilepsy | Phase 3 | Terminated(Sponsor's decision) | - | - |
| NCT00575588 | Type 2 Diabetes | Phase 3 | Completed | - | - |
| NCT00262600 | - | - | Completed | - | - |
| NCT00692003 | - | - | Terminated(Sponsor's decision) | - | - |
| NCT00575588 | - | - | Completed | - | - |
| NCT00477295 | Epilepsy | Phase 3 | Completed | - | - |
| NCT00115908 | Hepatitis C, Chronic | Phase 2 | Completed | - | - |
| NCT00477295 | - | - | Completed | - | - |
| NCT00731692 | Primary Progressive Multiple S... more >>clerosis Collapse << | Phase 3 | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT00128531 | Prostate Cancer | Phase 3 | Completed | - | - |
| NCT03687827 | Diabetes Mellitus, Type 2 | Phase 4 | Recruiting | December 30, 2019 | - |
| NCT00731692 | - | - | Terminated(The extension study... more >> was terminated early after the results of the core study showed the study did not meet primary endpoint; confirmed disability progression) Collapse << | - | - |
| NCT00143455 | Small Cell Lung Carcinoma | Phase 3 | Completed | - | - |
| NCT00156741 | Bradycardia; Sick Sinus Syndro... more >>me, AV Block Collapse << | Phase 4 | Unknown | - | - |
| NCT00207740 | Asthma | Phase 2 | Completed | - | - |
| NCT00501059 | Moderate Risk of CVD | Phase 3 | Completed | - | - |
| NCT00207740 | - | - | Completed | - | - |
| NCT00209326 | Labor, Premature ... more >> Premature Birth Collapse << | Phase 2 | Completed | - | Belgium ... more >> ULB Erasme Hospital Gynecology, Route de Lennik 808 Brussels, Belgium, BE-1070 CHR Citadelle, Boulevard du 12iede Ligne 1 Liege, Belgium, BE-4000 AZ Heilig Hart Gynecology, Kliniekstraat 44 Tienen, Belgium, BE-3299 Czech Republic Gynekologicko-porodnicka klinika Fakultni nemocnice Brno, Obilni trh 11 Brno, Czech Republic, CZ-625 00 Gynekologicko-porodnicke oddeleni nemocnice Jihlava, Vrchlickeho 59 Jihlava, Czech Republic, CZ-58 501 Gynekologicka-porodnicka klinika Fakultni nemocnice, Capkovo namesti 1 Plzen, Czech Republic, CZ-307 08 Gynekologicko-porodnicka klinika 1. LF UK a VFN v Praze, Apolinarska 18 Praha 2, Czech Republic, CZ-128 51 Gynekologicko-porodnicka klinika Fakultni nemocnice Motol, V uvalu 84 Praha 5, Czech Republic, CZ-15006 Gynekologicko.porodnicke oddeleni, Socialni pece 12A Usti nad Labem, Czech Republic, CZ-400 01 Finland Kuopio University Hopital Kuopio, Finland, FI-70210 Tampere University Hospital Tampere, Finland, FI-33521 Turku University Hospital, Kiinamyllynkatu 4-6 Turku, Finland, FIN-20521 Lithuania Kaunas Medical University Clinics, Eiveniu 2 Kaunas, Lithuania, LT-50009 Vilnius University, Antakalnio 57 Vilnius, Lithuania, LT-10207 Poland Zaklad Patofizjologii Ciazy AM w Bialymstoku, M.C. Sklodowskiej 24a Bialystok, Poland, PO - 15-276 Romania Spitalul Clinic de Urgenta "Elias", Nr. 17, Str. Marasti, sector 1 Bucuresti, Romania, RO-011461 Spitalul Clinic Judetean Cluj-Napoca Ginecologie No. 1, Clinica Nr. 1 Ginecologie Nr. 3-5, Str. Clinicilor Cluj-Napoca, Romania, RO-3400 Collapse << |
| NCT00163358 | Asthma | Phase 3 | Completed | - | - |
| NCT00143455 | - | - | Completed | - | - |
| NCT00252837 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT00501059 | - | - | Completed | - | - |
| NCT00252005 | Deep-Vein Thrombosis | Phase 2 | Completed | - | - |
| NCT00254891 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Terminated(See Termination Rea... more >>son in Detailed Description) Collapse << | - | - |
| NCT00252876 | Type 2 Diabetes | Phase 3 | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT00240331 | Renal Failure | Phase 3 | Completed | - | - |
| NCT00256711 | Non-Small-Cell Lung Carcinoma | Phase 2 | Completed | - | - |
| NCT00362934 | Hyperlipidemia | Phase 3 | Completed | - | - |
| NCT00240331 | - | - | Completed | - | - |
| NCT00274469 | Metastatic Breast Cancer | Phase 2 | Completed | - | - |
| NCT00279045 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00255645 | - | - | Terminated(The development pro... more >>gram has been terminated) Collapse << | - | - |
| NCT00274469 | - | - | Completed | - | - |
| NCT00291330 | Thromboembolism | Phase 3 | Completed | - | - |
| NCT00291330 | - | - | Completed | - | - |
| NCT00280657 | Acne Vulgaris | Phase 3 | Completed | - | - |
| NCT00599339 | - | - | Completed | - | - |
| NCT00294437 | Prostate Cancer | Phase 3 | Terminated(underfunding) | - | - |
| NCT00510822 | Major Depression | Phase 2 | Completed | - | Austria ... more >> Landeskrankenhaus Klagenfurt, Abteilung für Psychiatrie und Psychotherapie Klagenfurt, Austria, A-9020 Gemeinnützige Salzburger Landeskliniken Betriebsgesellschaft mbH Salzburg, Austria, A-5020 Czech Republic Faculty Hospital Brno Brno, Czech Republic, 639 00 Hospital Ceske Budejovice Ceske Budejovice, Czech Republic, 370 87 Pardubice Regional Hospital Pardubice, Czech Republic, 532 03 1st Medical Faculty Prague Prague, Czech Republic, 120 00 Prague Psychiatric Centrum Praha, Czech Republic, 181 03 Masaryk Hospital Ústí nad Labem, Czech Republic, 401 13 Germany Charite - Center for Psychiatry and Psychotherapy Berlin, Germany, D-10117 LWL-Universitätsklinik Bochum Bochum, Germany, 44791 University Bonn, Center for Psychiatry and Psychotherapy Bonn, Germany, D-53105 Carl Gustav Carus University Dresden, Center for Psychiatry and Psychotherapy Dresden, Germany, D-01307 Georg-August-University Goettingen, Department of Psychiatry and Psychotherapy Goettingen, Germany, D-37075 Hospital Guenzburg, Center for Psychosomatic Medicine Guenzburg, Germany, D-89312 University Jena, Center for Psychiatry and Psychotherapy Jena, Germany, D-07743 Klinik für Psychiatrie und Psychotherapie der Universität zu Köln Köln, Germany, 50924 Fachklinik Katzenelnbogen Limburg an der Lahn (Katzenelnbogen), Germany, 56368 Otto-von-Guericke University Magdeburg, Department of Psychiatry, Psychotherapy and Psychosomatic Medicine Magdeburg, Germany, D-39120 Klinikum der Johannes Gutenberg-Universität Mainz Mainz, Germany, 55131 Zentralinstitut für Seelische Gesundheit, Klinik für Psychiatrie und Psychotherapie Mannheim, Germany, 68159 Center for Psychiatry and Psychotherapy, University of Muenster Muenster, Germany, D-48149 Ludwig-Maximilians University Munich Munich, Germany, D-80336 Max Planck Institute of Psychiatry Munich, Germany, D-80804 Bezirksklinikum Regensburg Regensburg, Germany, 93053 Ernst Moritz Arndt University of Greifswald, Center for Psychiatry and Psychotherapy Stralsund, Germany, D-18437 Collapse << |
| NCT00291941 | Hepatitis B | Phase 3 | Completed | - | Belgium ... more >> O.L.Vrouwziekenhuis Aalst Aalst, Belgium, 9300 RHMS La Madeleine ATH ATH, Belgium, 7800 RHMS Clinique Louis Caty Baudour Baudour, Belgium, 7331 AZ -VUB Dienst Nefrologie Bruxelles, Belgium, 1090 Cliniques universitaires Saint Luc Bruxelles, Belgium, 1200 CHU Brugmann (site V Horta) Service de néphrologie Bruxelles, Belgium, B-1020 ULB Hôpital Erasme Département de Néphrologie Bruxelles, Belgium CHU Hôpital civil de Charleroi, Belgium, 6000 UZ AntwerpenDienst nefrologie Edegem, Belgium, B-2650 UZ Gent Gent, Belgium, 9000 CHU Tivoli La Louvière, Belgium, 7100 UZ Gasthuisberg Leuven Nierziekten Leuven, Belgium, 3000 CHU Andre VESALE Montigny le tilleul, Belgium, 6110 RHMS TournayService de néphrologie Tournai, Belgium, 7500 Czech Republic Dept. of Heamodialysis Hospital JihlavaVrchlického Jihlava, Czech Republic, 59586 33 Regional Hospital Liberec Liberec, Czech Republic, 46063 Infection Diseases and AIDS Treatment ClinicUniversity Hospital with Outpatient Clinic Ostrava - Poruba, Czech Republic, 1790708 52 Dept. of Internal Medicine StrahovSermirska 5 Prague, Czech Republic, 169 00 Masaryk´s Hospital Socialni pece 3316/12A Usti nad Labem, Czech Republic, 401 13 Hungary St. Rókus Hospital Budapest, Hungary, 1085 St. István Hospital Budapest, Hungary, 1096 Petz Aladár Teaching Hospital Vasvári Győr, Hungary, H-9023 Pest County Flór Ferenc Hospital Kistarcsa, Hungary, 2143 Vas and Szombathely County Markusovszky Hospital Szombathely, Hungary, 9700 Collapse << |
| NCT00860262 | Hypertension | Phase 3 | Completed | - | - |
| NCT00329238 | Thromboembolism | Phase 3 | Completed | - | - |
| NCT00599339 | - | - | Completed | - | - |
| NCT00860262 | - | - | Completed | - | - |
| NCT00412984 | Atrial Fibrillation ... more >> Atrial Flutter Collapse << | Phase 3 | Completed | - | - |
| NCT00923091 | - | - | Completed | - | - |
| NCT00337090 | Overactive Bladder | Phase 2 | Completed | - | - |
| NCT00930059 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT00329238 | - | - | Completed | - | - |
| NCT00923091 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT00312975 | Carcinoma, Non-small-cell Lung | Phase 2 | Completed | - | - |
| NCT00824447 | Allergic Rhinoconjunctivitis | Phase 2 Phase 3 | Completed | - | - |
| NCT00350415 | Ulcerative Colitis | Phase 3 | Completed | - | - |
| NCT00358215 | Heart Failure ... more >> Anemia Cardiovascular Disease Ventricular Dysfunction Congestive Heart Failure Collapse << | Phase 3 | Completed | - | - |
| NCT00412984 | - | - | Completed | - | - |
| NCT00366834 | Vomiting Naus... more >>ea Nausea and Vomiting, Chemotherapy-Induced Collapse << | Phase 3 | Completed | - | - |
| NCT00388674 | - | - | Completed | - | - |
| NCT00358215 | - | - | Completed | - | - |
| NCT00394927 | - | - | Completed | - | - |
| NCT00395772 | Venous Thromboembolism | Phase 2 | Completed | - | - |
| NCT00496769 | Atrial Fibrillation | Phase 3 | Completed | - | - |
| NCT00598442 | - | - | Completed | - | - |
| NCT00400478 | Diffuse Large B-Cell Lymphoma ... more >>(DLBCL) Follicular NHL Grade 3b Collapse << | Phase 3 | Completed | - | - |
| NCT00598442 | Chronic Renal Failure ... more >> Chronic Kidney Disease Anemia Collapse << | Phase 3 | Completed | - | - |
| NCT01019421 | Alzheimer's Disease | Phase 2 | Completed | - | - |
| NCT00402428 | Chronic Hepatitis C | Phase 3 | Completed | - | - |
| NCT00409188 | Non-small Cell Lung Cancer | Phase 3 | Completed | - | - |
| NCT00388674 | - | - | Completed | - | - |
| NCT00416520 | Chronic Kidney Disease ... more >> Hyperphosphatemia Collapse << | Phase 3 | Completed | - | - |
| NCT00409188 | - | - | Completed | - | - |
| NCT00496769 | - | - | Completed | - | - |
| NCT01032629 | Diabetes Mellitus, Type 2 ... more >> Cardiovascular Diseases Risk Factors Collapse << | Phase 3 | Completed | - | - |
| NCT02603393 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 4 | Completed | - | - |
| NCT02038647 | Small Cell Lung Cancer (SCLC) | Phase 2 | Active, not recruiting | August 7, 2017 | - |
| NCT00416520 | - | - | Completed | - | - |
| NCT02041533 | - | - | Active, not recruiting | - | - |
| NCT02045862 | Urinary Bladder Overactive ... more >> Overactive Bladder Urgency Incontinence Urinary Bladder Diseases\Urologic Diseases Collapse << | Phase 3 | Completed | - | - |
| NCT01032629 | - | - | Completed | - | - |
| NCT02858453 | Interstitial Cystitis ... more >> Bladder Pain Syndrome Painful Bladder Syndrome Chronic Interstitial Cystitis Collapse << | Phase 3 | Active, not recruiting | February 2020 | - |
| NCT02041533 | Stage IV or Recurrent Non-Smal... more >>l Cell Lung Cancer Collapse << | Phase 3 | Active, not recruiting | October 1, 2018 | - |
| NCT00682786 | Rectal Carcinoma | Phase 2 | Completed | - | United States, Missouri ... more >> Washington University School of Medicine Saint Louis, Missouri, United States, 63110 Collapse << |
| NCT02064439 | Pulmonary Embolism ... more >> Thromboembolism Thrombosis Venous Thrombosis Venous Thromboembolism Collapse << | Phase 3 | Completed | - | - |
| NCT01762085 | Diabetic Ulcer of Plantar Aspe... more >>ct of Left Foot Diabetic Ulcer of Plantar Aspect of Right Foot Diabetic Polyneuropathy Collapse << | Not Applicable | Unknown | December 2016 | United States, Arizona ... more >> Barrett Foot & Ankle Institute Recruiting Phoenix, Arizona, United States, 85024 Contact: Steven L Barrett, DPM 480-478-0780 slbarrettpod@me.com Contact: John C Tassone, DPM (602) 938-6960 jctassone@cox.net Principal Investigator: Stephen L Barrett, DPM Richard P. Jacoby Recruiting Scottsdale, Arizona, United States, 85251 Contact: Richard P Jacoby, DPM 480-994-5977 jacobydpm@gmail.com Principal Investigator: Richard P Jacoby, DPM Southern Arizona Limb Salvage Alliance, Recruiting Tucson, Arizona, United States, 85724-5072 Contact: Manish Bharara, PhD 520-626-1349 manish.bharara@gmail.com Contact: David Armstrong, DPM (520) 626-1349 Principal Investigator: Manish Bharara, PhD United States, Indiana Andrew Rader, DPM Recruiting Jasper, Indiana, United States, 47546 Contact: Andrew P Rader, DPM 812-634-2778 pvppc@psci.net Principal Investigator: Andrew P Rader, DPM United States, Texas Damien Dauphinee Recruiting Denton, Texas, United States, 76210 Contact: Damien Dauphinee, DPM 940-300-3054 dauphinee@completefootandanklecare.com Principal Investigator: Damien Dauphinee, DPM Maria Buitrago, DPM Recruiting Houston, Texas, United States, 77092 Contact: Maria Buitrago, DPM 713-680-3668 mobui@hotmail.com Principal Investigator: Maria Buitrago, DPM Collapse << |
| NCT02398734 | Internal Carotid Artery Stenos... more >>is Collapse << | Not Applicable | Recruiting | January 2023 | Czechia ... more >> University Hospital Ostrava Recruiting Ostrava, Czech Republic, Czechia, 70852 Contact: David Skoloudik, MD,PhD,FESO 00420597375613 skoloudik@hotmail.com Contact: Martin Roubec, MD, PhD 00420597375630 martin.r@centrum.cz Principal Investigator: David Skoloudik, MD, PhD Collapse << |
| NCT02064439 | - | - | Completed | - | - |
| NCT02727894 | - | - | Completed | - | - |
| NCT01686204 | Intestinal Barrier Function ... more >> Chronic Inflammation Collapse << | Not Applicable | Completed | - | United States, Wisconsin ... more >> Department of Food Science, Babcock Hall Madison, Wisconsin, United States, 53706 Collapse << |
| NCT02045862 | - | - | Completed | - | - |
| NCT02023632 | Motor Activity | Not Applicable | Completed | - | Canada, British Columbia ... more >> Surrey Family YMCA Surrey, British Columbia, Canada, V3S 7S6 Langara YMCA Vancouver, British Columbia, Canada, V5Y 2Z5 Robert Lee YMCA Vancouver, British Columbia, Canada, V6Z 1Y2 Collapse << |
| NCT00682786 | - | - | Completed | - | - |
| NCT03704337 | Healthy | Not Applicable | Completed | - | United States, Texas ... more >> Exercise & Sport Nutrition Lab - Human Clinical Research Facility College Station, Texas, United States, 77845 Collapse << |
| NCT01813370 | - | - | Active, not recruiting | March 2019 | United States, Maryland ... more >> John Hopkins Medical Center Baltimore, Maryland, United States, 21287 United States, New York Memorial Sloan Kettering Cancer Center New York, New York, United States, 10065 United States, North Carolina Duke University Medical Center Durham, North Carolina, United States, 27701 United States, Pennsylvania Urological Associates of Lancaster Lancaster, Pennsylvania, United States, 17601 United States, South Carolina Carolina Urologic Research Center Myrtle Beach, South Carolina, United States, 29579 Canada, Manitoba Cancer Care Manitoba Winnipeg, Manitoba, Canada Lithuania Hospital of Lithuanian University Health Sciences Kauno Klinikos Kauno, Klinikos, Lithuania Klaipeda University Hospital Klaipeda, Lithuania, 92288 Collapse << |
| NCT00071279 | Pulmonary Embolism ... more >> Deep Vein Thrombosis Collapse << | Phase 3 | Completed | - | - |
| NCT00289978 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 3 | Completed | - | - |
| NCT00289978 | - | - | Completed | - | - |
| NCT03128021 | Major Depressive Disorder | Phase 4 | Recruiting | July 2022 | United States, Pennsylvania ... more >> University of Pittsburgh Recruiting Pittsburgh, Pennsylvania, United States, 15213 Contact: Dana Williams, MS 412-246-5924 barvdm@upmc.edu Contact: Rachel Berta, BA 412-246-5685 goodra@upmc.edu Sub-Investigator: Meryl A Butters, Ph.D. Sub-Investigator: Tae Kim, Ph.D. Sub-Investigator: Dana Tudorascu, Ph.D. Principal Investigator: Howard Aizenstein, M.D., Ph.D. Principal Investigator: Carmen Andreescu, M.D. Collapse << |
| NCT03244475 | Mild Traumatic Brain Injury (m... more >>TBI) Post-traumatic Stress Disorder Collapse << | Not Applicable | Recruiting | September 30, 2021 | United States, California ... more >> VA San Diego Healthcare System, San Diego, CA Recruiting San Diego, California, United States, 92161 Contact: Annemarie A Quinto, BA 858-822-2352 adangeles@ucsd.edu Contact: Ashley R Swan, BA (858) 822-6874 arobb@ucsd.edu Principal Investigator: Mingxiong Huang, PhD Collapse << |
| NCT03304613 | Chronic Spine Pain ... more >> Fibromyalgia Collapse << | Not Applicable | Recruiting | October 2022 | United States, Michigan ... more >> Back & Pain Center, University of Michigan Recruiting Ann Arbor, Michigan, United States, 48106 Contact: Joseph Long 734-763-5226 josephlo@med.umich.edu Collapse << |
| NCT01327703 | Exocrine Pancreatic Insufficie... more >>ncy Cystic Fibrosis Collapse << | Phase 4 | Completed | - | Germany ... more >> Klinikum-Bochum Bochum, Germany Universitätsklinikum Carl Gustav Carus Dresden, Germany Universitaetsklinikum Erlangen Erlangen, Germany Jena University Hospital, Universitaetsklinikum Jena Jena, Germany Klinikum der Universitat Munchen Medizinische Klinik-Innenstadt München, Germany University Children's Clinic Tubingen Tubingen, Germany Poland Specjalistyczny Zespół Opieki Zdrowotnej nad Matką i Dzieckiem Poradnia Leczenia Mukowiscydozy Gdansk, Poland Wojewodzki Specjalistityczny Szpital Dziect Im Sw Ludwika Krakow, Poland Dziecięcy Szpital Kliniczny im. Prof. Antoniego Gębali Lublin, Poland Szpital Kliniczny im Karola Jonschera Poznan, Poland NZOZ Sanatorium Cassia Villa Medica Rabka Zdrój, Poland NZOZ Podkarpacki Osrodek Pulmonologii i Alergologii Rzeszow, Poland Children's Health Memorial Institute Warszawa, Poland Collapse << |
| NCT00389519 | - | - | Terminated(A planned interim a... more >>nalysis was performed after approx. 240 subjects completed the trial. The study was stopped, as permitted by protocol, after the analysis.) Collapse << | - | - |
| NCT00389519 | Hypertension | Phase 3 | Terminated(A planned interim a... more >>nalysis was performed after approx. 240 subjects completed the trial. The study was stopped, as permitted by protocol, after the analysis.) Collapse << | - | - |
| NCT01375049 | - | - | Completed | - | - |
| NCT00216476 | Schizophrenia ... more >> Psychotic Disorders Collapse << | Phase 3 | Completed | - | - |
| NCT01641393 | Exocrine Pancreatic Insufficie... more >>ncy: Cystic Fibrosis Collapse << | Phase 3 | Completed | - | - |
| NCT01375049 | Cystic Fibrosis | Phase 2 | Completed | - | - |
| NCT01327703 | - | - | Completed | - | - |
| NCT00216476 | - | - | Completed | - | - |
| NCT00734162 | Hepatitis B Virus (HBV) | Phase 3 | Completed | - | United States, California ... more >> Children's Hospital & Research Center at Oakland Oakland, California, United States, 94609 United States, Indiana Riley Hospital for Children Indianapolis, Indiana, United States, 46202 United States, Washington Children's Hospital & Regional Medical Center, d/b/a Seattle Children's Research Institute Seattle, Washington, United States, 98105 Bulgaria Multiprofile Hospital for Active Treatment Sveti Georgi Plovdiv, Bulgaria, 4002 Clinic of Gastroenterology, Specialized Hospital for Active Treatment of Pediatric Diseases, Sofia Sofia, Bulgaria, 1606 France Hopital Femmes Meres Enfants Bron Cedex, France, 69677 Hôpital Claude Huriez Lille Cedex, France, 59037 Poland Samodzielny Publiczny Dzieciecy Szpital Kliniczny Akademii Medycznej w Bialymstoku Białystok, Poland, 15-274 Wojewodzki Specjalistyczny Szpital im Bieganskiego Bydgoszcz, Poland, 85-030 Wojewodzki Szpital Obserwacyjno-Zakazny im. T. Browicza Bydgoszcz, Poland, 85-030 Krakowski Szpital Specjalistyczny im. Jana Pawla II Kraków, Poland, 31-202 Samodzielny Publiczny Szpital Kliniczny im. Karola Johschera Poznan, Poland, 60-572 Specjalistyczny Zespol Opieki Zdrowotnej nad Matka i Dzieckiem Poznań, Poland, 61-734 Wojewodzki Szpital Zakazny Warszawa, Poland, 01-201 Samodzielny Publiczny Szpital Kliniczny Nr 1 Wrocław, Poland, 50-368 Romania Fundeni Clinical Institute Bucharest, Romania, 022328 Institute for Infectious Diseases Bucharest, Romania, 21105 Cluj Childrens Emergency Hospital Napaco, Romania, 400217 Spain Hosp Univ y Politecnico La Fe de Valencia Madrid, Spain, 28046 Hospital Universitario De Getafe Madrid, Spain, 46009 Turkey Ege Universitesi Tip Fakultesi Hastanesi Izmir, Turkey, 35100 Collapse << |
| NCT00475605 | - | - | Active, not recruiting | August 2022 | - |
| NCT00241527 | NSAIDs Upper ... more >>GI Symptoms Collapse << | Phase 3 | Completed | - | - |
| NCT01830855 | Meningococcal Vaccine | Phase 3 | Completed | - | - |
| NCT00808028 | Meningitis, Meningococcal | Phase 2 | Completed | - | Australia, Queensland ... more >> Queensland Paediatric Infectious Diseases (QPID) Laboratory Herston, Queensland, Australia, 4029 Australia, South Australia Department of Paediatrics, Women's & Children's Hospital North Adelaide, South Australia, Australia, 5006 Australia, Victoria Royal Children's Hospital Carlton, Victoria, Australia, 3053 Australia, Western Australia Children's Clinical Research Facility, Vaccine Trials Group (VTG), Subiaco, Western Australia, Australia, 6008 Poland ZOZ w Debicy, Poradnia Chorob Zakaznych Debica, Poland, 39-200 SP ZOZ Krakowski Szpital Specjalistyczny im. Jana Pawla II w Krakowie Krakow, Poland, 31-202 Wojewodzki Specjalistyczny Szpital Dzieciecy im. Sw. Ludwika w Krakowie, Krakow, Poland, 31-503 NZOZ Salmed Leczna, Poland, 21-010 Wojewodzki Specjalistyczny Szpital im. dr Wl. Bieganskiego w Lodzi Lodz, Poland, 91-347 Wojewodzki Specjalistyczny Szpital im. W. Bieganskiego Lodz, Poland, 91-347 SPZOZ w Lubartowie, Oddzial Pediatryczny, ul. Cicha 14 Lubartow, Poland, 21-100 Eskulap Sp. z o. o., ul. Weteranow 46 Lublin, Poland, 20-044 SP ZOZ, Poradnia Dziecieca w Grucie Melno, Poland, 86-330 NZOZ Pratyka Lekarza Rodzinnego Alina Grocka-Wlazlak Oborniki Slaskie, Poland, 55-120 Specjalistyczny ZOZ nad Matka i Dzieckiem w Poznaniu Oddzial Obserwacyjno-Zakazny A Poznan, Poland, 61-734 NZLA Michalkowice, Jarosz i Partnerzy Siemianowice Slaskie, Poland, 41-03 NZOZ Nasz Lekarz Praktyka Grupowa Lekarzy Rodzinnych z Przychodnia Torun, Poland, 87-100 Oddzial Dzieciecy Szpital im. Swietej Jadwigi Slaskiej Trzebnica, Poland, 55-100 Klinika Pediatrii i Chorob Infekcyjnych Wroclaw, Poland, 50-345 Samodzielny Publiczny Szpital Kliniczny Nr 1 Wroclaw, Poland, 50-345 Spain Hospital Clinico Universitario de Santiago de Compostela Santiago de Compostela, La Coruna, Spain, 15706 Hospital U. de Getafe Getafe, Madrid, Spain, 28905 Complexo Hospitalario Xeral-Cies de Vigo Vigo, Pontevedra, Spain, 36204 Clinica Virgen del Mar Almeria, Spain, 04120 Hospital General de Cataluna Barcelona, Spain, 08195 Hospital U. 12 de Octubre Madrid, Spain, 28041 Centro de Salud Nazaret Valencia, Spain, 46024 Collapse << |
| NCT00351910 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT00309608 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00734162 | - | - | Completed | - | - |
| NCT01830855 | - | - | Completed | - | - |
| NCT00241553 | NSAIDs Upper ... more >>GI Symptoms Collapse << | Phase 3 | Completed | - | - |
| NCT00532558 | Hypercholesterolemia | Phase 3 | Terminated(Overall profile of ... more >>the compound does not offer significant clinical advantage to patients over currently available lipid lowering agents) Collapse << | - | - |
| NCT00309608 | - | - | Completed | - | - |
| NCT00808028 | - | - | Completed | - | - |
| NCT00488631 | Colitis, Ulcerative | Phase 3 | Completed | - | - |
| NCT00487539 | Colitis, Ulcerative | Phase 2 Phase 3 | Completed | - | - |
| NCT00496470 | - | - | Completed | - | - |
| NCT00496470 | Chronic Obstructive Pulmonary ... more >>Disease, COPD Collapse << | Phase 4 | Completed | - | - |
| NCT01392378 | Pneumococcal Vaccines | Phase 4 | Completed | - | Poland ... more >> Gabinet Lekarski Debica, Poland, 39-200 Hanna Czajka Indywidualna Praktyka Lekarska Krakow, Poland, 31-302 NZOZ "Praktimed" sp. z o.o. Krakow, Poland, 31-422 NZOZ Salmed Leczna, Poland, 21-010 Specjalistyczna Praktyka Lekarska Gravita Lodz, Poland, 91-347 SP ZOZ Lubartow Lubartow, Poland, 21-100 NZOZ Praktyka Lekarza Rodzinnego Eskulap Lublin, Poland, 20-044 NZOZ Praktyka Lekarza Rodzinnego Alina Grocka-Wlazlak Oborniki Slaskie, Poland, 55-120 Specjalistyczny ZOZ nad Matka i Dzieckiem, Oddzial Obserwacyjno Zakazny A, Szpital Dzieciecy Poznan, Poland, 61-734 NZLA Michalkowice Jarosz i Partnerzy Siemianowice Slaskie, Poland, 41-103 NZOZ Nasz Lekarz Torun, Poland, 87-100 Szpital im. Sw. Jadwigi Slaskiej, Oddzia Pediatryczny Trzebnica, Poland, 55-100 DEN-MED Gabinet Lekarsko-Stomatologiczny Joanna i Jacek Witwiccy Warszawa, Poland, 02-127 Samodzielny Publiczny Szpital Kliniczny nr 1 we Wroclawiu Wroclaw, Poland, 50-345 Collapse << |
| NCT01392378 | - | - | Completed | - | - |
| NCT00487539 | - | - | Completed | - | - |
| NCT00885365 | Cystic Fibrosis | Phase 3 | Completed | - | - |
| NCT01404234 | Cystic Fibrosis ... more >> Pseudomonas Aeruginosa Collapse << | Phase 3 | Completed | - | - |
| NCT00488631 | - | - | Completed | - | - |
| NCT00621140 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT01404234 | - | - | Completed | - | - |
| NCT00621140 | - | - | Completed | - | - |
| NCT00789035 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00789035 | - | - | Completed | - | - |
| NCT00821977 | Diabetes Mellitus, Type 2 | Phase 2 Phase 3 | Completed | - | - |
| NCT00885365 | - | - | Completed | - | - |
| NCT02152371 | - | - | Completed | - | - |
| NCT02152371 | Diabetes Mellitus, Type 2 | Phase 3 | Completed | - | - |
| NCT00949975 | Chronic Obstructive Pulmonary ... more >>Disease Collapse << | Phase 2 | Completed | - | - |
| NCT00881530 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00949975 | - | - | Completed | - | - |
| NCT00968708 | Diabetes Mellitus, Type 2 ... more >> Acute Coronary Syndrome Collapse << | Phase 3 | Completed | - | - |
| NCT00881530 | - | - | Completed | - | - |
| NCT01023516 | Chronic Obstructive Pulmonary ... more >>Disease (COPD) Collapse << | Phase 2 | Completed | - | - |
| NCT02973100 | - | - | Completed | - | - |
| NCT01323270 | Meningococcal Vaccine ... more >> rLP2086 Repevax N Meningitidis Serogroup B Meningitis Collapse << | Phase 2 | Completed | - | Finland ... more >> Espoo Vaccine Research Clinic Espoo, Finland, 02100 Helsinki South Vaccine Research Clinic Helsinki, Finland, 00100 Ita-Helsinki Vaccine Research Clinic Helsinki, Finland, 00930 Järvenpää Vaccine Research Clinic Järvenpää, Finland, 04400 Kokkola Vaccine Research Clinic Kokkola, Finland, 67100 Lahti Vaccine Research Clinic Lahti, Finland, 15140 Oulu Vaccine Research Clinic Oulu, Finland, 90220 Pori Vaccine Research Clinic Pori, Finland, 28100 Seinäjoki Vaccine Research Clinic Seinäjoki, Finland, 60100 Tampereen Yliopisto University Of Tampere Tampere, Finland, 33014 Tampere Vaccine Research Clinic Tampere, Finland, 33100 Vaccine Research Center Tampere, Finland, FIN-33014 Turku Vaccine Research Clinic Turku, Finland, 20520 Vantaa Vaccine Research Clinic Vantaa, Finland, 01300 Germany Gemeinschaftspraxis Dr. med. Guido Hein, Dr. med. Rainer Lauf Bad Sobernheim, Germany, 55566 Gerhard Bleckmann Kinder- und Jugendarzt Baunatal, Germany, 34225 Kinderarzt-Praxis Bramsche, Germany, 49565 Gemeinschaftspraxis fuer Kinder- und Jugendmedizin Dres. Behre, Burgert, Gunkel Kehl, Germany, 77694 Dr. med. Sobhi Mahdi Kinderarzt und Jugendmedizin Luebeck, Germany, 23566 Universitaetsmedizin der Johannes Gutenberg-Universitaet Mainz Mainz, Germany, 55131 Dr. Michael Vomstein, Hermann Oesterle, Kinder- und Jugendaerzte Schwaebisch-Hall, Germany, 74523 Thomas Lenz & (Frau) Dr. med. Marin Eggers Vellmar, Germany, 34246 Dres.T. Schmitz, H. Knee, Ch, Wittermann Fachaerztliche Weilheim, Germany, 82362 Gemeinschaftspraxis fuer Kinder und Jugendliche Welzheim Welzheim, Germany, 73642 Poland Gabinet Lekarski Debica, Poland, 39-200 Krakowski Szpital Specjalistyczny, im. Jana Pawla II Krakow, Poland, 31-202 NZOZ Salmed Leczna, Poland, 21-010 Specjalistyczna Praktyka Lekarska Gravita Lodz, Poland, 91-347 Samodzielny Publiczny Zaklad Opieki Zdrowotnej, Oddzial Pediatryczny Lubartow, Poland, 21-100 NZOZ Praktyka Lekarza Rodzinnego Eskulap Lublin, Poland, 20-044 NZOZ Praktyka Lekarza Rodzinnego Alina Grocka-Wlazlak Oborniki Slaskie, Poland, 55-120 Specjalistyczny ZOZ nad Matka i Dzieckiem, Przychodnia Wieku Rozwojowego Poznan, Poland, 61-825 NZLA Michalkowice Jarosz i Partnerzy Siemianowice Slaskie, Poland, 41-103 NZOZ Nasz Lekarz Torun, Poland, 87-100 Szpital im. Sw. Jadwigi Slaskiej, Oddzial Pediatryczny Trzebnica, Poland, 55-100 Samodzielny Publiczny Szpital Kliniczny nr 1 we Wroclawiu Wroclaw, Poland, 50-345 Collapse << |
| NCT02973100 | Type 2 Diabetes | Phase 2 | Completed | - | - |
| NCT01023516 | - | - | Completed | - | - |
| NCT02597049 | Type 2 Diabetes Mellitus | Phase 3 | Completed | - | - |
| NCT01323270 | - | - | Completed | - | - |
| NCT00968708 | - | - | Completed | - | - |
| NCT02597049 | - | - | Completed | - | - |
| NCT01117350 | - | - | Completed | - | - |
| NCT01117350 | Diabetes Mellitus, Type 2 | Phase 4 | Completed | - | - |
| NCT01276509 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT01276509 | - | - | Completed | - | - |
| NCT01035749 | Influenza | Phase 2 | Completed | - | Estonia ... more >> GSK Investigational Site Tartu, Estonia, 50106 Slovakia GSK Investigational Site Cifer, Slovakia, 919 43 GSK Investigational Site Dolny Kubin, Slovakia, 026 01 GSK Investigational Site Dunajska Streda, Slovakia, 929 01 GSK Investigational Site Nova Dubnica, Slovakia, 018 51 GSK Investigational Site Nove Mesto nad Vahom, Slovakia, 915 01 GSK Investigational Site Puchov, Slovakia, 020 01 GSK Investigational Site Ruzomberok, Slovakia, 034 01 GSK Investigational Site Trencin, Slovakia, 911 01 Collapse << |
| NCT01298492 | Crohn's Disease | Phase 2 | Completed | - | - |
| NCT01277666 | - | - | Completed | - | - |
| NCT01035749 | - | - | Completed | - | - |
| NCT01277666 | Crohn's Disease | Phase 3 | Completed | - | - |
| NCT01316939 | Crohn's Disease | Phase 3 | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT01316939 | - | - | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT01338870 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT01298492 | - | - | Completed | - | - |
| NCT01318993 | Crohn's Disease | Phase 3 | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT02105675 | Prostate Cancer ... more >> Prostate Cancer Metastatic Collapse << | Phase 2 | Completed | - | Czechia ... more >> Brno, Czechia Hradec Kralove, Czechia Jihlava, Czechia Liberec, Czechia Nova Ves pod Plesi, Czechia Olomouc, Czechia Ostrava, Czechia Praha 10, Czechia Praha 4, Czechia Praha 5, Czechia Praha 8, Czechia Usti nad Labem, Czechia Zlin, Czechia Collapse << |
| NCT02111577 | Metastatic Castrate Resistant ... more >>Prostate Cancer Collapse << | Phase 3 | Active, not recruiting | June 2020 | - |
| NCT02935608 | Lumbosacral Radiculopathy | Phase 2 | Completed | - | - |
| NCT02130362 | - | - | Active, not recruiting | February 27, 2028 | - |
| NCT01318993 | - | - | Terminated(This study was term... more >>inated due to the lack of efficacy of GSK1605786A in Crohn's disease based on the results of Study CCX114151.) Collapse << | - | - |
| NCT02670538 | Bipolar Disorder ... more >> Depression Collapse << | Phase 3 | Completed | - | - |
| NCT02766608 | Chronic Obstructive Pulmonary ... more >>Disorder Collapse << | Phase 3 | Completed | - | - |
| NCT01338870 | - | - | Completed | - | - |
| NCT02145468 | - | - | Completed | - | - |
| NCT02145468 | Acute Coronary Syndrome | Phase 3 | Completed | - | - |
| NCT02166476 | Urinary Tract Infection Compli... more >>cated Acute Pyelonephritis Collapse << | Phase 3 | Completed | - | - |
| NCT02207244 | Psoriasis | Phase 3 | Active, not recruiting | July 15, 2020 | - |
| NCT03011450 | Severe Hypertriglyceridemia | Phase 3 | Active, not recruiting | June 2019 | - |
| NCT02166476 | - | - | Completed | - | - |
| NCT03308968 | Migraine Prophylaxis | Phase 3 | Active, not recruiting | June 6, 2019 | - |
| NCT03052725 | Eosinophils, Asthma | Phase 3 | Completed | - | - |
| NCT02207244 | - | - | Active, not recruiting | - | - |
| NCT03066804 | Heart Failure With Preserved E... more >>jection Fraction Collapse << | Phase 3 | Recruiting | December 4, 2019 | - |
| NCT02260986 | Atopic Dermatitis | Phase 3 | Completed | - | - |
| NCT03381196 | Influenza A | Phase 3 | Recruiting | October 29, 2019 | - |
| NCT03185013 | Cervical Dysplasia ... more >> Cervical High Grade Squamous Intraepithelial Lesion HSIL Collapse << | Phase 3 | Recruiting | August 2020 | - |
| NCT02957617 | Neuropathic Pain From Lumbosac... more >>ral Radiculopathy Collapse << | Phase 2 | Active, not recruiting | August 5, 2019 | - |
| NCT02260986 | - | - | Completed | - | - |
| NCT02975349 | Relapsing-remitting Multiple S... more >>clerosis Collapse << | Phase 2 | Active, not recruiting | August 12, 2020 | - |
| NCT02974153 | Migraine Disorders | Phase 3 | Completed | - | - |
| NCT03478683 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Active, not recruiting | March 14, 2019 | - |
| NCT02414958 | Diabetes Mellitus, Type 1 | Phase 3 | Completed | - | - |
| NCT02929329 | Heart Failure | Phase 3 | Recruiting | January 27, 2021 | - |
| NCT03001817 | Severe Hypertriglyceridemia | Phase 3 | Active, not recruiting | June 2019 | - |
| NCT03315143 | Type 2 Diabetes Mellitus ... more >> Chronic Kidney Diseases Collapse << | Phase 3 | Recruiting | March 2022 | - |
| NCT02764229 | Colitis, Ulcerative | Phase 2 | Active, not recruiting | November 2018 | - |
| NCT03094195 | Post-herpetic Neuralgia | Phase 2 | Recruiting | August 28, 2020 | - |
| NCT02337608 | Ulcerative Colitis | Phase 2 | Completed | - | - |
| NCT02465515 | Diabetes Mellitus | Phase 4 | Completed | - | - |
| NCT02470468 | Stage IV Non-small Cell Lung C... more >>ancer Collapse << | Phase 1 Phase 2 | Unknown | November 2018 | Czechia ... more >> Brno, Czechia, 625 00 Hradec Kralove, Czechia, 500 05 Jindřichův Hradec, Czechia, 377 38 Kutna Hora, Czechia, 284 01 Nachod, Czechia, 547 69 Olomouc, Czechia, 775 20 Ostrava, Czechia, 708 52 Pardubice, Czechia, 530 03 Plzen, Czechia, 305 99 Praha, Czechia, 128 08 Praha, Czechia, 140 59 Praha, Czechia, 150 06 Pribram, Czechia, 261 95 Usti nad Labem, Czechia, 401 13 Zlin, Czechia, 762 75 Slovakia Kosice, Slovakia, 040 01 Piest'any, Slovakia, 921 01 Poprad, Slovakia, 058 01 Collapse << |
| NCT02942576 | Atrial Fibrillation | Phase 3 | Active, not recruiting | November 2018 | - |
| NCT03152552 | Diabetes Type 2 With Heart Fai... more >>lure Collapse << | Phase 2 | Completed | - | - |
| NCT02540954 | Macular Degeneration | Phase 3 | Recruiting | January 12, 2021 | - |
| NCT03496298 | Type 2 Diabetes Mellitus | Phase 3 | Recruiting | April 2021 | - |
| NCT02414958 | - | - | Completed | - | - |
| NCT02566993 | Small-cell Lung Cancer | Phase 3 | Active, not recruiting | - | - |
| NCT03107793 | Crohn Disease | Phase 3 | Recruiting | July 24, 2021 | - |
| NCT02581995 | Macular Edema | Phase 4 | Completed | - | - |
| NCT02607618 | - | - | Completed | - | - |
| NCT02581995 | - | - | Completed | - | - |
| NCT02607618 | Skin Structures and Soft Tissu... more >>e Infections Collapse << | Phase 3 | Completed | - | - |
| NCT03395184 | Crohn's Disease | Phase 2 | Recruiting | February 18, 2021 | - |
| NCT02340234 | Atopic Dermatitis | Phase 2 | Completed | - | - |
| NCT00796354 | Constipation | Phase 3 | Completed | - | Czech Republic ... more >> Gastroenterologie s.r.o. Hradec Králové, Czech Republic, 500 02 EGK.s.r.o, Sanatorium sv. Anny Praha, Czech Republic, 130 00 Orlickoustecká Nemocnice a.s. Ústi Nad Orlicí, Czech Republic, 562 18 France Hôpital Hotel Dieu Clermont-Ferrand, France, 63058 Hôpital Archet II Nice, France, 06200 Hôpital Charles Nicolles Rouen, France, 76031 Germany Martin-Luther-Krankenhaus-Betriebs-GmbH Berlin, Germany, 14193 Gastroenterologische Gemeinschaftspraxis Münster, Germany, 48159 Facharztpraxis für Innere Medizin Wiesbaden, Germany, 65185 Italy Policlinico S. Orsola Malpighi Bologna, Italy, 40138 Università degli Studi di Genova Genova, Italy, 6 - 16132 Università degli Studi di Palermo Palermo, Italy, 129 - 90127 Università La Sapienza Roma, Italy, 00162 Poland Slaskie Centrum Osteoporozy Katowice, Poland, 40-084 Centrum Onkologii ul.Rentgena 5 Warsaw, Poland, 02-781 Prosen SMO Warszawa, Poland, 01-231 Katedra i Klinika Gastroenterologii i Hepatologii Wroclaw, Poland, 50-556 Sweden Sodra Alvsborgs Hospital (SAS) Borås, Sweden, SE-176-65 Karolinska University Hospital Solna Stockholm, Sweden, SE-171-76 United Kingdom St Marks Hospital Harrow, United Kingdom, HA1 3UJ John Radcliffe Hospital Oxford, United Kingdom, OX3 9DU Collapse << |
| NCT02670915 | Diabetes Diab... more >>etes Mellitus, Type 1 Collapse << | Phase 3 | Completed | - | - |
| NCT03449134 | Chronic Cough | Phase 3 | Recruiting | July 10, 2020 | - |
| NCT03449147 | Chronic Cough | Phase 3 | Recruiting | January 8, 2021 | - |
| NCT02743806 | Colitis, Ulcerative ... more >> Crohn Disease Collapse << | Phase 4 | Active, not recruiting | February 15, 2022 | - |
| NCT02762500 | Colitis, Ulcerative | Phase 2 | Completed | - | - |
| NCT02752776 | Moderate to Severe Plaque Psor... more >>iasis Collapse << | Phase 4 | Completed | - | - |
| NCT03751657 | Diabetes Mellitus, Type 2 | Phase 2 | Recruiting | November 7, 2019 | - |
| NCT02767804 | Non-small Cell Lung Cancer | Phase 3 | Recruiting | April 2020 | - |
| NCT02365610 | Epilepsy Foca... more >>l Seizures Collapse << | Phase 2 | Completed | - | Czechia ... more >> Havířov, Czechia Hradec Králové, Czechia Praha, Czechia Rychnov nad Kněžnou, Czechia Hungary Budapest, Hungary Esztergom, Hungary Hodmezovasarhely, Hungary Kecskemét, Hungary Pecs, Hungary Szombathely, Hungary Italy Bologna, Italy Pavia, Italy Rome, Italy Poland Bydgoszcz, Poland Elbląg, Poland Golub-Dobrzyn, Poland Katowice, Poland Krakow, Poland Kraków, Poland Warszawa, Poland Wrocław, Poland Spain Barcelona, Spain Madrid, Spain United Kingdom Birmingham, United Kingdom Glasgow, United Kingdom Great Yarmouth, United Kingdom Stoke-on-Trent, United Kingdom Collapse << |
| NCT03560986 | Complex Regional Pain Syndrome... more >> (CRPS) Collapse << | Phase 3 | Recruiting | April 2021 | - |
| NCT00265135 | Carcinoma, Renal Cell | Phase 1 Phase 2 | Completed | - | Czech Republic ... more >> Brno, Czech Republic Hradec Nad Svitavou, Czech Republic France Caen, France Lyon Cedex 08, France Montpellier Cedex 5 N/A, France Montpellier, France Villejuif N/A, France Netherlands Groningen, Netherlands Nijmegen, Netherlands Rotterdam, Netherlands United Kingdom Birmingham, United Kingdom Leeds, United Kingdom Manchester, United Kingdom Plymouth, United Kingdom Collapse << |
| NCT03619213 | Heart Failure With Preserved E... more >>jection Fraction Collapse << | Phase 3 | Recruiting | June 22, 2021 | - |
| NCT02871635 | Crohn Disease | Phase 3 | Active, not recruiting | May 2, 2019 | - |
| NCT02891226 | Crohn's Disease | Phase 2 | Recruiting | November 28, 2020 | - |
| NCT03006770 | Critical Limb Ischemia (CLI) | Phase 3 | Recruiting | May 2020 | - |
| NCT03069014 | Mild to Moderate Alzheimer's D... more >>isease Collapse << | Phase 1 Phase 2 | Recruiting | October 31, 2019 | Austria ... more >> University Hospital Graz Recruiting Graz, Styria, Austria, 8034 Contact: Birgit Reinhart +43 316 385 833 96 birgit.reinhart@medunigraz.at Principal Investigator: Reinhold Schmidt, Prof. Dr. Sub-Investigator: Anita Lechner, Dr. med. Landeskrankenhaus Hall Recruiting Hall in Tirol, Tirol, Austria, 6060 Contact: Elisabeth Klingler +43 50504 88265 elisabeth.klingler@tirol-kliniken.at Principal Investigator: Douglas Imarhiagbe, OA Dr. med. Sub-Investigator: Cornelia Hessler-Piock, Dr. med. Czechia Charles University Recruiting Praha, Hlavni Mesto Praha, Czechia, 16000 Contact: Marcela Sedlackova +42 0 702 001 917 marcelasedlackova@seznam.cz Principal Investigator: Jakub Hort, Prof. MD Sub-Investigator: Jan Lazco, MD Neurology Clinic of Martin Urbanek Recruiting Brno, Jihomoravsky Kraj, Czechia, 60200 Contact: Jana Pacinkova 00420 737 157 670 j.pacinkova@gmail.com Principal Investigator: Katerina Sheardová, MD Sub-Investigator: Martin Urbanek, MD Vestra clinics s.r.o Recruiting Rychnov nad Kneznou, Kralovehadrecky Kraj, Czechia, 51601 Contact: Jitka Adamcova +42 0607 818683 jitka@vestraclinics.org Principal Investigator: Ladislav Pazdera, MD Sub-Investigator: Veselý Pazdera, MD NEUROHK s.r.o Recruiting Chocen, Pardubicky Kraj, Czechia, 56501 Contact: Jitka Prouzová +420 604 444 808 kucerjiri@gmail.com Principal Investigator: Martin Valis, MD Sub-Investigator: Petr Barton, MD Germany Nordwestkrankenhaus Sanderbusch Recruiting Sande, Friesland, Germany, 26452 Contact: Christina Eiben +49 4422 80-1434 c.eiben@sanderbusch.de Contact: Anne Stargardt a.stargardt@sanderbusch.de Principal Investigator: Pawel Kermer, Prof.Dr.med Sub-Investigator: Deeg Sebastian, Dr.med Zentrum für klinische Forschung Recruiting Bad Homburg, Hesse, Germany, 61348 Contact: Justine Orth +49 6172 1710784 justineorth@dr-schoell.de Contact: Rebecca Schwegler rebeccaschwegler@dr-schoell.de Principal Investigator: Irma Schöll, MD Sub-Investigator: Elvira Steidl, MD Studienzentrum Nordwest Recruiting Westerstede, Niedersachsen, Germany, 26655 Contact: Irene Reinisch +49 448852 1500 ireinisch@studienzentrum-nordwest.de Contact: Nicole Mildt nmildt@studienzentrum-nordwest.de Principal Investigator: Joachim Springub, Dr. med. Sub-Investigator: Wolfgang Schwarz, Dr. med. Universitätsklinik Magdeburg, Klinik für Neurologie Recruiting Magdeburg, Sachsen Anthal, Germany, 39120 Contact: Urte Schneider urte.schneider@med.ovgu.de Principal Investigator: Daniel Bittner, MD Sub-Investigator: Peter Körtvelyessy, MD Sächsisches Krankenhaus Arnsdorf Recruiting Arnsdorf, Sachsen, Germany, 01477 Contact: Ramona Rost +49 35200 26-2270 Ramona.Rost@skhar.sms.sachsen.de Principal Investigator: Peter Schönknecht, Prof. Dr. Sub-Investigator: Majdy Abu Bakr, OA Dr. Pharmakologisches Studienzentrum Chemnitz GmbH Recruiting Chemnitz, Sachsen, Germany Contact: Anne Merkel +49 3727 991006 merkel@mvz-mittweida.de Principal Investigator: Ralf Bodenschatz, Dipl.-med. Sub-Investigator: Gunter Schumann, Dr. med. Charité Universitätsmedizin Berlin Recruiting Berlin, Germany, 13125 Contact: Nadine Krüger +49 3045 540097 nadine.krueger@charite.de Principal Investigator: Oliver Peters, Doz. Dr. Sub-Investigator: Heidrun Mehling, Dr. med. Neurologie Sendlinger Strasse Studien- und Gedächtniszentrum München Recruiting München, Germany, 80331 Contact: Susanne Dombrowka susanne.dombrowka@web.de Contact: Barbara Wittek goodmeds@web.de Principal Investigator: Andrej Pauls, Dipl.Psy.MD Sub-Investigator: Susanne Adel, MD LMU München Klinik für Psychiatrie und Psychotherapie Recruiting München, Germany, 80336 Contact: Thomas Görlitz thomas.goerlitz@med.uni-muenchen.de Principal Investigator: Robert Perneczky, Prof. MD Sub-Investigator: Carolin Kurz, MD Spain Fundació ACE Not yet recruiting Barcelona, Catalonia, Spain, 08028 Contact: Mar Buendia +34 - 93 430 47 20 Principal Investigator: Mercé Boada Rovira, MD Fundación de Gestión Sanitaria del Hospital de la Santa Creu I Sant Pau, C Not yet recruiting Barcelona, Catalonia, Spain, 08036 Contact: Roser Ribosa +34 - 93 553 71 12 Principal Investigator: Rafael Blesa González, MD Hospital Clínic de Barcelona Not yet recruiting Barcelona, Catalonia, Spain, 08036 Contact: Guadalupe Fernández Principal Investigator: Raquel Sánchez del Valle, MD Hospital la Paz Not yet recruiting Madrid, Spain, 28046 Contact: Ana Frank García, Prof. MD. Contact: Vega Mauleón Principal Investigator: Ana Frank García, Prof. MD. Hospital Virgen del Rocío Not yet recruiting Sevilla, Spain, 41013 Contact: Antonio Cervera Barajas +34-955-01 43 57 Principal Investigator: Emilio Franco Macías, MD Sweden Karolinska University Not yet recruiting Stockholm, Stockholms Iän, Sweden, 14186 Contact: Malin Aspö +46 8 585 854 68 malin.aspo@karolinska.se Principal Investigator: Niels Andreasen, Prof.Dr. Collapse << |
| NCT01138098 | Hepatitis B | Phase 4 | Completed | - | Slovakia ... more >> GSK Investigational Site Banska Bystrica, Slovakia, 974 01 GSK Investigational Site Dolny Kubin, Slovakia, 026 01 GSK Investigational Site Dubnica Nad Vahom, Slovakia, 018 41 GSK Investigational Site Martin, Slovakia, 036 01 GSK Investigational Site Nitra, Slovakia, 949 01 GSK Investigational Site Nova Dubnica, Slovakia, 018 51 GSK Investigational Site Nove Zamky, Slovakia, 940 01 GSK Investigational Site Puchov, Slovakia, 020 01 GSK Investigational Site Roznava, Slovakia, 048 01 GSK Investigational Site Trebisov, Slovakia, 075 01 GSK Investigational Site Zlate Moravce, Slovakia, 953 01 Collapse << |
| NCT03334435 | Atopic Dermatitis | Phase 3 | Recruiting | August 27, 2021 | - |
| NCT01138098 | - | - | Completed | - | - |
| NCT03043872 | Small Cell Lung Carcinoma Exte... more >>nsive Disease Collapse << | Phase 3 | Recruiting | September 30, 2020 | - |
| NCT03535194 | Psoriasis | Phase 3 | Recruiting | April 9, 2020 | - |
| NCT03599622 | Granulomatous Colitis ... more >> Crohn's Disease Crohn's Enteritis Granulomatous Enteritis Collapse << | Phase 2 | Recruiting | September 28, 2021 | - |
| NCT03556202 | Psoriasis | Phase 3 | Recruiting | May 16, 2024 | - |
| NCT03551964 | Acute Myocardial Infarction ... more >> Cardiogenic Shock Collapse << | Phase 4 | Recruiting | June 2021 | Czechia ... more >> University Hospital Kralovske Vinohrady Recruiting Prague, Please Select, Czechia, 10034 Contact: Zuzana Motovska, MD PhD +420731573253 motovska.zuzana@gmail.com Sub-Investigator: Jiri Knot, MD PhD Sub-Investigator: Jaroslav Ulman, MD St. Anne's University Hospital Brno Active, not recruiting Brno, Czechia, 656 91 Department of Cardiology, University Hospital Brno-Bohunice Not yet recruiting Brno, Czechia Contact: Petr Kala, MD. PhD. Cardiology Department, Regional Hospital Active, not recruiting Ceske Budejovice, Czechia University Hospital Hradec Králové Active, not recruiting Hradec Králové, Czechia, 500 05 Cardiocenter, Regional Hospital Recruiting Karlovy Vary, Czechia Contact: Alexander Schee, MD Contact alexandr.schee@gmail.com Krajská nemocnice Liberec Recruiting Liberec, Czechia, 460 63 Contact: Jiri Karasek, MD +420 485 312 646 jirikarry@gmail.com Principal Investigator: Jiri Karasek University Hospital Olomouc Active, not recruiting Olomouc, Czechia, 77900 University Hospital Ostrava Recruiting Ostrava, Czechia, 70852 Contact: Jan Mrozek, MD +420 596 920 115 honzamrozek@email.cz Principal Investigator: Jan Mrozek, MD Department of Cardiology, Regional Hospital, Active, not recruiting Pardubice, Czechia University Hospital Pilsen Recruiting Pilsen, Czechia, 304 60 Contact: Milan Hromadka, MD PhD +00420377 103 3166 hromadka@fnplzen.cz Principal Investigator: Milan Hromadka, MD PhD General University Hospital in Prague Active, not recruiting Prague, Czechia, 12808 Institute of Clinical and Experimental Medicine Active, not recruiting Prague, Czechia, 14021 Na Homolce Hospital Active, not recruiting Prague, Czechia, 150 30 Cardiocenter, Hospital Podlesi Not yet recruiting Trinec, Czechia Contact: Libor Sknouril, MD. PhD. Contact libor.sknouril@nempodlesi.cz Regional Hospital T. Bati Active, not recruiting Zlin, Czechia, 762 75 Masaryk Hospital Active, not recruiting Ústí Nad Labem, Czechia, 40011 Collapse << |
| NCT03354429 | Acute Ischaemic Stroke ... more >> Transient Ischaemic Attack Collapse << | Phase 3 | Recruiting | December 16, 2019 | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
