CAS No.: 164656-23-9
度他雄胺 Catalog No. CSN10879
Synonyms: GG 745;GI 198745;LS-173584
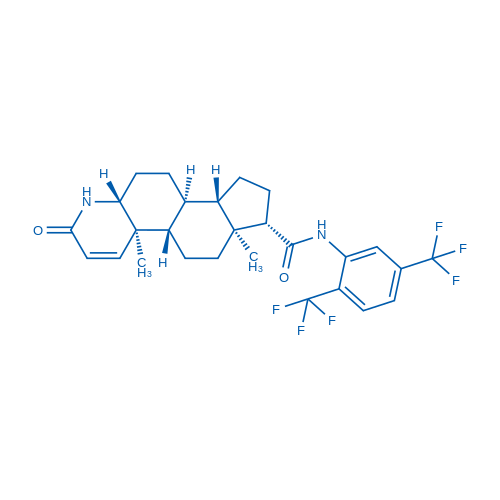
Dutasteride is a potent inhibitor of both 5 alpha-reductase isozymes. Dutasteride may possess off-target effects on the androgen receptor (AR) due to its structural similarity to DHT.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 作用机制
动物研究 剂量 Mice: 5 mg/kg, 12.5 mg/kg[4] (i.p., SID)
给药途径 i.p.
动物药代数据 Animal Rats[5] Dose 2.5 mg/kg (i.v.)
5 mg/kg (p.o.)Administration i.v.
p.o.AUCinf 1148 ± 81 μg·min/ml (i.v.) T1/2 1607 ± 196 min (i.v.) Tmax 60 min (p.o.) AUClast 275 ± 9 μg·min/ml (p.o.) CL 2.19 ± 0.16 ml/min/kg (i.v.) Cmax 289 ± 10 ng/ml (p.o.) Vss 4900 ± 326 ml/kg (i.v.) - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02147964 Gonadotropin Deficiency Phase 2 Not yet recruiting December 2026 United States, Washington ... more >> University of Washington Medical Center (Health Sciences) Not yet recruiting Seattle, Washington, United States, 98195 Contact: Iris Nielsen 206-221-5473 nielseni@uw.edu Contact: Kathy Winter 206-616-0484 klwinter@uw.edu Principal Investigator: Mara Roth, MD Sub-Investigator: John Amory, MD, MPH Sub-Investigator: Stephanie Page, MD, PhD Sub-Investigator: Bradley Anawalt, MD Collapse << NCT00298155 - - Completed - - NCT00298155 Cancer Prosta... more >>te Neoplasms Collapse << Phase 2 Completed - United States, Washington ... more >> Veterans' Administration Puget Sound Health Care System (VAPSHCS) Seattle, Washington, United States, 98108-1532 University of Washington Seattle, Washington, United States, 98195-6158 Collapse << - 更多
- 参考文献
- [1] Jun JEJ, Kinkade A, et al. 5α-Reductase Inhibitors for Treatment of Benign Prostatic Hyperplasia: A Systematic Review and Meta-Analysis. Can J Hosp Pharm. 2017;70(2):113-119.
- [2] Frye SV. Discovery and clinical development of dutasteride, a potent dual 5alpha-reductase inhibitor. Curr Top Med Chem. 2006;6(5):405-21.
- [3] Vitalis OO, Christopher CO. The Role of Dutasteride in Acute Prostatic Haematuria. 2017 Nov Vol.8 No.11.
- [4] Litim N, Bourque M, et al. The 5α-reductase inhibitor Dutasteride but not Finasteride protects dopamine neurons in the MPTP mouse model of Parkinson's disease. Neuropharmacology. 2015 Oct;97:86-94.
- [5] Seo SW, Park JW, et al. In Vitro and In Vivo Assessment of Metabolic Drug Interaction Potential of Dutasteride with Ketoconazole. Pharmaceutics. 2019 Dec 11;11(12). pii: E673.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 164656-23-9 | 储存条件 |
|
|||||||||||||
| 分子式 | C27H30F6N2O2 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 528.53 | 别名 | GG 745;GI 198745;LS-173584;Brand name: Avodart;Avidart;Avolve;Duagen;Dutas;Dutagen;Duprost;度他雄胺 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02147964 | Gonadotropin Deficiency | Phase 2 | Not yet recruiting | December 2026 | United States, Washington ... more >> University of Washington Medical Center (Health Sciences) Not yet recruiting Seattle, Washington, United States, 98195 Contact: Iris Nielsen 206-221-5473 nielseni@uw.edu Contact: Kathy Winter 206-616-0484 klwinter@uw.edu Principal Investigator: Mara Roth, MD Sub-Investigator: John Amory, MD, MPH Sub-Investigator: Stephanie Page, MD, PhD Sub-Investigator: Bradley Anawalt, MD Collapse << |
| NCT00298155 | - | - | Completed | - | - |
| NCT00298155 | Cancer Prosta... more >>te Neoplasms Collapse << | Phase 2 | Completed | - | United States, Washington ... more >> Veterans' Administration Puget Sound Health Care System (VAPSHCS) Seattle, Washington, United States, 98108-1532 University of Washington Seattle, Washington, United States, 98195-6158 Collapse << |
| NCT00490555 | Healthy | Phase 2 Phase 3 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT00490555 | - | - | Completed | - | - |
| NCT01777269 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01830829 | Benign Prostatic Hyperplasia ... more >> Chronic Prostatitis Collapse << | Phase 3 | Terminated(Difficulty in enrol... more >>ling particpants) Collapse << | - | Canada, Ontario ... more >> Centre for Applied Urological Research Kingston, Ontario, Canada, K7L 3J7 Collapse << |
| NCT01777269 | - | - | Completed | - | - |
| NCT00883909 | - | - | Completed | - | - |
| NCT00609596 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, New York ... more >> GSK Investigational Site Buffalo, New York, United States, 14202 Collapse << |
| NCT01342367 | Prostate Cancer | Not Applicable | Recruiting | April 2019 | United States, Illinois ... more >> University of Chicago Recruiting Chicago, Illinois, United States, 60637 Contact: Fauzia Arif farif@radonc.uchicago.edu Collapse << |
| NCT00983554 | Obesity Insul... more >>in Resistance Collapse << | Not Applicable | Unknown | October 2009 | United States, California ... more >> University of California, San Diego San Diego, California, United States, 92103 VA San Diego Healthcare System San Diego, California, United States, 92161 Collapse << |
| NCT01351987 | - | - | Unknown | December 2013 | Taiwan ... more >> Taipei Medical University - WanFang Hospital Taipei, Taiwan Collapse << |
| NCT01547299 | Prostate Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Boston, Massachusetts, United States, 02215 United States, Washington Seattle, Washington, United States, 98195 Canada, British Columbia Vancouver, British Columbia, Canada, V5Z 1M9 Canada, Ontario Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT01484808 | - | - | Unknown | May 2012 | United States, Rhode Island ... more >> Division of Urology, Brown University Not yet recruiting Providence, Rhode Island, United States, 02903 Principal Investigator: Steven I Cohen, MD Collapse << |
| NCT01547299 | - | - | Completed | - | - |
| NCT00680680 | Urinary Retention ... more >> Benign Prostatic Hyperplasia Collapse << | Not Applicable | Completed | - | - |
| NCT01337258 | - | - | Completed | - | - |
| NCT00593593 | - | - | Completed | - | - |
| NCT00056407 | Neoplasms, Prostate | Phase 3 | Completed | - | - |
| NCT00470834 | Neoplasms, Prostate | Phase 4 | Completed | - | - |
| NCT00056407 | - | - | Completed | - | - |
| NCT01332435 | - | - | Completed | - | - |
| NCT01332435 | - | - | Completed | - | - |
| NCT00316732 | - | - | Completed | - | United Kingdom ... more >> GSK Investigational Site Exeter, Devon, United Kingdom, EX2 5DW GSK Investigational Site Plymouth, Devon, United Kingdom, PL6 8DH GSK Investigational Site Colchester, Essex, United Kingdom, CO4 5JL GSK Investigational Site Stevenage, Hertfordshire, United Kingdom, SG2 4AB GSK Investigational Site Manchester, Lancashire, United Kingdom, M13 9WL GSK Investigational Site Manchester, Lancashire, United Kingdom, M8 5RB GSK Investigational Site Leicester, Leicestershire, United Kingdom, LE5 4PW GSK Investigational Site Leytonstone, London, United Kingdom, E11 1NR GSK Investigational Site Enfield, Middlesex, United Kingdom, EN2 8JL GSK Investigational Site Nottingham, Nottinghamshire, United Kingdom, NG5 1PB GSK Investigational Site Tauton, Somerset, United Kingdom, TA1 5DA GSK Investigational Site Sunderland, Tyne & Wear, United Kingdom, SR4 7TP GSK Investigational Site Sutton Coldfield, West Midlands, United Kingdom, B75 7RR GSK Investigational Site Birmingham, United Kingdom, B15 2TH GSK Investigational Site Bradford, United Kingdom, BD5 0NA GSK Investigational Site Bradford, United Kingdom, BD9 6RJ GSK Investigational Site Bristol, United Kingdom, BS2 8HW GSK Investigational Site East Kilbride, United Kingdom, G75 8RG GSK Investigational Site Hull, United Kingdom, HU16 5JD GSK Investigational Site Ilkeston, Derbyshire, United Kingdom, DE7 8LN GSK Investigational Site Leeds, United Kingdom, LS1 3EX GSK Investigational Site London, United Kingdom, EC1 7BE GSK Investigational Site London, United Kingdom, NW3 2QG GSK Investigational Site London, United Kingdom, W18 6BJ GSK Investigational Site Sheffield, United Kingdom, S10 2JF GSK Investigational Site Torquay, United Kingdom, TQ2 7AA GSK Investigational Site Wakefield, United Kingdom, WF1 4DG Collapse << |
| NCT00375765 | Benign Prostatic Hyperplasia ... more >> Prostate Cancer Collapse << | Phase 4 | Completed | - | Netherlands ... more >> GSK Investigational Site Nijmegen, Netherlands, 6525 GA GSK Investigational Site Nijmegen, Netherlands, 6532 SZ Collapse << |
| NCT00161421 | Contraception ... more >> Hypogonadism Collapse << | Phase 2 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT00470834 | - | - | Completed | - | - |
| NCT01594502 | - | - | Active, not recruiting | July 2021 | United States, Illinois ... more >> University of Illinois at Chicago Chicago, Illinois, United States, 60612 Collapse << |
| NCT00537654 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Indiana ... more >> GSK Investigational Site Evansville, Indiana, United States, 47714 United States, Texas GSK Investigational Site Austin, Texas, United States, 78752 Collapse << |
| NCT00734656 | Alcohol Related Disorders ... more >> Alcoholism Alcohol Abuse Collapse << | Not Applicable | Completed | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 Collapse << |
| NCT00734656 | - | - | Completed | - | - |
| NCT00953576 | Prostate Cancer | Phase 1 Phase 2 | Terminated(The study terminate... more >>d early due to concerns about drug toxicity.) Collapse << | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Collapse << |
| NCT02159690 | Prostate Cancer ... more >> Localized Prostate Cancer Collapse << | Phase 2 | Withdrawn(loss of funding) | - | United States, Maryland ... more >> Johns Hopkins Hospital Baltimore, Maryland, United States, 21231 Collapse << |
| NCT01193855 | Prostate Cancer | Phase 2 | Unknown | - | United Kingdom ... more >> University College of London Hospitals Recruiting London, England, United Kingdom, W1T 7NF Contact: Contact Person 44-207-380-9194 markemberton1@btinternet.com Collapse << |
| NCT01294592 | Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT01923090 | Healthy Volunteers | Phase 2 | Unknown | October 2013 | United Kingdom ... more >> Wellcome Trust Clinical Research Facility, Queen Elizabeth Hospital Recruiting Birmingham, West Midlands, United Kingdom, B15 2TT Contact: Jeremy w Tomlinson, MD PhD 44(0)121 4158715 J.W.Tomlinson@bham.ac.uk Principal Investigator: Jeremy W Tomlinson, MD PhD Collapse << |
| NCT01294592 | - | - | Completed | - | - |
| NCT00953576 | - | - | Terminated(The study terminate... more >>d early due to concerns about drug toxicity.) Collapse << | - | - |
| NCT01529086 | - | - | Completed | - | United States, Ohio ... more >> Cleveland Clinic Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01376284 | - | - | Completed | - | - |
| NCT00969072 | Benign Prostatic Hyperplasia ... more >> Prostatic Hyperplasia Collapse << | Phase 2 | Completed | - | - |
| NCT00690950 | Benign Prostatic Hyperplasia | Phase 4 | Unknown | - | United States, Pennsylvania ... more >> Urologic Consultants of Southeastern PA Bala Cynwyd, Pennsylvania, United States, 19004 Collapse << |
| NCT01942551 | Benign Prostate Hyperplasia | Phase 1 | Completed | - | Korea, Republic of ... more >> Chungnam National University Hospital Daejeon, Korea, Republic of, 301-721 Collapse << |
| NCT00421421 | Benign Prostatic Hyperplasia | Phase 4 | Terminated(Incomplete informat... more >>ion) Collapse << | - | United Kingdom ... more >> GSK Clinical Trials Call Center Barnet, United Kingdom, EN5 3DJ GSK Clinical Trials Call Center Bath, United Kingdom, BA1 1BX GSK Clinical Trials Call Centre Birmingham, United Kingdom, B15 2TH GSK Clinical Trials Call Center Bradford, United Kingdom, BD2 0NA GSK Clinical Trials Call Center Bristol, United Kingdom, BS2 8HW GSK Clinical Trials Call Center Chester, United Kingdom, CH2 1UL GSK Clinical Trials Call Center Colchester, United Kingdom, CO4 5JL GSK Clinical Trials Call Center Crewe, United Kingdom, CW1 4QJ GSK Clinical Trials Call Centre Derby, United Kingdom, DE22 3NE GSK Clinical Trials Call Center Edinburgh, United Kingdom, EH4 2XU GSK Clinical Trials Call Center Glasgow, United Kingdom, G51 4TF GSK Clinical Trials Call Center Hull, United Kingdom, HU16 5JQ GSK Clinical Trials Call Center Leeds, United Kingdom, LS9 7TF GSK Clinical Trials Call Center Leicester, United Kingdom, LE5 4PW GSK Clinical Trials Call Center London, United Kingdom, E11 1NR GSK Clinical Trials Call Center Newcastle Upon Tyne, United Kingdom, NE7 7DN GSK Clinical Trials Call Center Nottingham, United Kingdom, NG5 1PB GSK Clinical Trials Call Center Oldham, United Kingdom, OL1 2JH GSK Clinical Trials Call Center Ormskirk, United Kingdom, L39 2AZ GSK Clinical Trials Call Center Plymouth, United Kingdom, PL6 8DH GSK Clinical Trials Call Center Stevenage, United Kingdom, SG1 4AB GSK Clinical Trials Call Center Sunderland, United Kingdom, SR4 7TP GSK Clinical Trials Call Center Sutton Coldfield, United Kingdom, B75 7RR GSK Clinical Trials Call Center Torquay, United Kingdom, TQ2 7AA GSK Clinical Trials Call Center Wakefield, United Kingdom, WF1 4DG Collapse << |
| NCT00244309 | Prostate Cancer | Phase 3 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Taussig Cancer Institute, Case Comprehensive Cancer Center Cleveland, Ohio, United States, 44195 Collapse << |
| NCT02058368 | Prostatic Hyperplasia | Phase 3 | Completed | - | - |
| NCT00082043 | Premenstrual Syndrome ... more >> PMS Healthy Depression Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00403000 | Prostate Cancer | Phase 2 | Completed | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263-0001 Collapse << |
| NCT01473030 | - | - | Active, not recruiting | December 2018 | United States, Illinois ... more >> University of Illinois at Chicago Chicago, Illinois, United States, 60612 Collapse << |
| NCT00399165 | Contraception | Phase 1 Phase 2 | Completed | - | United States, Washington ... more >> University of Washington Seattle, Washington, United States, 98195 Collapse << |
| NCT01428869 | - | - | Completed | - | Canada, Ontario ... more >> University Health Network Toronto, Ontario, Canada, M5G 2M9 Collapse << |
| NCT00274417 | Benign Prostatic Hyperplasia ... more >> Lower Urinary Tract Symptoms Collapse << | Not Applicable | Unknown | May 2009 | United States, Virginia ... more >> Urology of Virginia Norfolk, Virginia, United States, 23510 Urology of Virginia Virginia Beach, Virginia, United States, 23454 Collapse << |
| NCT01231607 | Androgenetic Alopecia | Phase 3 | Completed | - | - |
| NCT00752869 | Hypogonadism | Phase 4 | Completed | - | United States, Massachusetts ... more >> Men's Health Boston Boston, Massachusetts, United States, 002445 United States, Rhode Island The Miriam Hospital Providence, Rhode Island, United States, 02906 Collapse << |
| NCT02352311 | Benign Prostate Hyperplasia ... more >> Healthy Collapse << | Phase 1 | Unknown | June 2015 | Korea, Republic of ... more >> Chonbuk National University Hospital Recruiting Jeonju, Jeollabuk-do, Korea, Republic of, 561-712 Contact: Min-Gul Kim, M.D. +82-63-250-1487 mgkim@jbctc.org Collapse << |
| NCT01653925 | Prostatic Neoplasms ... more >> Low Grade Prostate Cancer Collapse << | Not Applicable | Active, not recruiting | December 2019 | Canada ... more >> Hotel-Dieu of Quebec Quebec, Canada, G1R 2J6 Institute of nutraceuticals and functional food of Laval University Quebec, Canada, G1V 0A6 Collapse << |
| NCT00431626 | - | - | Terminated(Low enrollment) | - | - |
| NCT00431626 | Benign Prostatic Hyperplasia | Phase 3 | Terminated(Low enrollment) | - | - |
| NCT00398281 | Prostate Cancer | Phase 3 | Completed | - | United States, Pennsylvania ... more >> Kimmel Cancer Center at Thomas Jefferson University - Philadelphia Philadelphia, Pennsylvania, United States, 19107-5541 Collapse << |
| NCT00398580 | Hypogonadism ... more >>Hypogonadism, Male Collapse << | Phase 2 | Completed | - | United States, California ... more >> GSK Investigational Site Torrance, California, United States, 90502 United States, Kentucky GSK Investigational Site Louisville, Kentucky, United States, 40202 United States, Maryland GSK Investigational Site Baltimore, Maryland, United States, 21287 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27710 United States, Texas GSK Investigational Site San Antonio, Texas, United States, 78229 United States, Washington GSK Investigational Site Seattle, Washington, United States, 98108 GSK Investigational Site Seattle, Washington, United States, 98195 Collapse << |
| NCT01231607 | - | - | Completed | - | - |
| NCT00558363 | Neoplasms, Prostate ... more >> Prostate Cancer After a Radical Treatment Collapse << | Phase 2 | Completed | - | - |
| NCT01495026 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT00062790 | Prostatic Hyperplasia | Phase 4 | Completed | - | United States, California ... more >> GSK Investigational Site Long Beach, California, United States, 90806 United States, Connecticut GSK Investigational Site New Britain, Connecticut, United States, 06052 GSK Investigational Site Trumbull, Connecticut, United States, 06611 United States, Georgia GSK Investigational Site Augusta, Georgia, United States, 30912 United States, Illinois GSK Investigational Site Chicago, Illinois, United States, 60612 GSK Investigational Site Niles, Illinois, United States, 60714 United States, Massachusetts GSK Investigational Site Boston, Massachusetts, United States, 02114-3139 United States, New Hampshire GSK Investigational Site Lebanon, New Hampshire, United States, 03756 United States, Oregon GSK Investigational Site Eugene, Oregon, United States, 97401-8122 United States, Texas GSK Investigational Site Dallas, Texas, United States, 75235 United States, Virginia GSK Investigational Site Richmond, Virginia, United States, 23249 Collapse << |
| NCT02184585 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT00368979 | Prostatic Hyperplasia | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Chiba, Japan, 263-0043 GSK Investigational Site Chiba, Japan, 266-0031 GSK Investigational Site Chiba, Japan, 272-0107 GSK Investigational Site Fukuoka, Japan, 802-0077 GSK Investigational Site Fukuoka, Japan, 810-0001 GSK Investigational Site Fukuoka, Japan, 830-0027 GSK Investigational Site Hyogo, Japan, 660-0052 GSK Investigational Site Kanagawa, Japan, 215-0021 GSK Investigational Site Kanagawa, Japan, 226-0025 GSK Investigational Site Kanagawa, Japan, 229-1103 GSK Investigational Site Kanagawa, Japan, 245-0015 GSK Investigational Site Kanagawa, Japan, 252-0804 GSK Investigational Site Kanagawa, Japan, 259-1132 GSK Investigational Site Kyoto, Japan, 604-8436 GSK Investigational Site Oita, Japan, 871-0012 GSK Investigational Site Oita, Japan, 874-0937 GSK Investigational Site Osaka, Japan, 542-0073 GSK Investigational Site Osaka, Japan, 562-0036 GSK Investigational Site Osaka, Japan, 584-0074 GSK Investigational Site Tokyo, Japan, 130-0026 GSK Investigational Site Tokyo, Japan, 131-0032 GSK Investigational Site Tokyo, Japan, 150-0002 GSK Investigational Site Tokyo, Japan, 152-0001 GSK Investigational Site Tokyo, Japan, 153-0051 GSK Investigational Site Tokyo, Japan, 183-0044 GSK Investigational Site Tokyo, Japan, 186-0011 Collapse << |
| NCT00368979 | - | - | Completed | - | - |
| NCT01393730 | Prostate Cancer | Phase 2 | Completed | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Washington Seattle Cancer Care Alliance Seattle, Washington, United States, 98109 University of Washington Medical Center Seattle, Washington, United States, 98195 Collapse << |
| NCT00558363 | - | - | Completed | - | - |
| NCT01393730 | - | - | Completed | - | - |
| NCT00090103 | Prostatic Hyperplasia | Phase 3 | Completed | - | - |
| NCT00527605 | - | - | Completed | - | - |
| NCT00527605 | Benign Prostatic Hyperplasia ... more >> Prostatic Hyperplasia Collapse << | Phase 3 | Completed | - | China, Guangdong ... more >> GSK Investigational Site Guangzhou, Guangdong, China, 510180 China, Hubei GSK Investigational Site Wuhan, Hubei, China, 430030 China, Jiangsu GSK Investigational Site Nanjing, Jiangsu, China, 210006 China, Zhejiang GSK Investigational Site Hangzhou, Zhejiang, China, 310003 China GSK Investigational Site Beijing, China, 100034 GSK Investigational Site Beijing, China, 100050 GSK Investigational Site Beijing, China, 100853 GSK Investigational Site Beijing, China GSK Investigational Site Shanghai, China, 200001 GSK Investigational Site Shanghai, China, 200025 GSK Investigational Site Shanghai, China, 200030 GSK Investigational Site Tianjin, China, 300211 Collapse << |
| NCT00866554 | Prostate Cancer ... more >> Erectile Dysfunction Lower Urinary Tract Symptoms Collapse << | Phase 2 | Unknown | December 2015 | Canada ... more >> CHUQ- Hotel-Dieu de Quebec Quebec, Canada, G1R 2J6 Collapse << |
| NCT01368003 | Adenocarcinoma of the Prostate | Phase 2 | Withdrawn(Loss of funding.) | - | United States, Massachusetts ... more >> Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02215 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 Collapse << |
| NCT00701779 | Benign Prostatic Hyperplasia | Phase 4 | Completed | - | United States, Indiana ... more >> Research Institute of Deaconess Clinic Evansville, Indiana, United States, 47713 Collapse << |
| NCT00701779 | - | - | Completed | - | - |
| NCT00090103 | - | - | Completed | - | - |
| NCT00194675 | Hypogonadism ... more >>Benign Prostatic Hyperplasia Collapse << | Phase 4 | Completed | - | United States, Washington ... more >> VA Puget Sound Health Care System Seattle, Washington, United States, 98108 Collapse << |
| NCT00939120 | - | - | Completed | - | - |
| NCT00194675 | - | - | Completed | - | - |
| NCT00939120 | Benign Prostatic Hyperplasia (... more >>BPH) Collapse << | Phase 4 | Completed | - | United States, Indiana ... more >> Deaconess Clinic Gateway Health Center Newburgh, Indiana, United States, 47630 Collapse << |
| NCT00400335 | Hypogonadism ... more >>Hypogonadism, Male Collapse << | Phase 1 | Completed | - | United States, Washington ... more >> GSK Investigational Site Tacoma, Washington, United States, 98418 Collapse << |
| NCT02839122 | Benign Prostate Hyperplasia | Phase 1 | Completed | - | - |
| NCT00780754 | Prostate Cancer | Phase 3 | Completed | - | Lithuania ... more >> Urology dep. of Kaunas University of Medicine Kaunas, Lithuania, 50009 Collapse << |
| NCT00363311 | Neoplasms, Prostate | Phase 4 | Completed | - | - |
| NCT00363311 | - | - | Completed | - | - |
| NCT00827814 | Benign Prostatic Hyperplasia | Phase 4 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT00303446 | Kennedy's Disease ... more >> Spinal and Bulbar Muscular Atrophy Collapse << | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT00673127 | Prostate Cancer | Phase 2 | Completed | - | United States, Maryland ... more >> Sidney Kimmel Comprehensive Cancer Center at John Hopkins University Baltimore, Maryland, United States United States, Massachusetts Beth Israel Deaconess Medical Center Boston, Massachusetts, United States, 02115 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02115 Massachusetts General Hospital Boston, Massachusetts, United States, 02214 United States, Oregon Oregon Health and Science University Portland, Oregon, United States United States, Texas MD Anderson Cancer Center Houston, Texas, United States Canada Sunnybrook and Women's College Health Sciences Center Toronto, Canada Collapse << |
| NCT00880672 | Benign Prostatic Hyperplasia | Phase 4 | Completed | - | - |
| NCT00668642 | Prostate Cancer | Phase 2 | Completed | - | United States, Illinois ... more >> Northwestern University Medical Center Chicago, Illinois, United States, 60611 University of Chicago Hospitals and Clinics Chicago, Illinois, United States, 60637 NorthShore University HealthSystem Evanston, Illinois, United States, 60201 Collapse << |
| NCT00303446 | - | - | Completed | - | - |
| NCT02213107 | Prostate Cancer | Phase 2 | Active, not recruiting | September 2020 | United States, New York ... more >> University of Rochester Rochester, New York, United States, 14642 Collapse << |
| NCT00553878 | Prostate Cancer | Phase 2 Phase 3 | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT00853697 | Prostate Cancer ... more >> Castration-resistant, Metastatic Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10065 Collapse << |
| NCT02058576 | Urologic Diseases | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Belfast, United Kingdom, BT9 6AD Collapse << |
| NCT01299571 | - | - | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Seoul, Korea, Republic of, 110-749 Collapse << |
| NCT00441116 | - | - | Completed | - | - |
| NCT00441116 | Alopecia | Phase 3 | Completed | - | - |
| NCT00673127 | - | - | Completed | - | - |
| NCT01577693 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Texas ... more >> GSK Investigational Site Austin, Texas, United States, 78744 Collapse << |
| NCT01299571 | - | - | Completed | - | - |
| NCT01929330 | Alopecia | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT00382356 | Benign Prostatic Hypertrophy | Not Applicable | Completed | - | - |
| NCT01673490 | Prostatic Hyperplasia | Phase 4 | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | Vietnam ... more >> GSK Investigational Site Ho Chi Minh, Vietnam Collapse << |
| NCT02052713 | Urologic Diseases | Phase 1 | Completed | - | United Kingdom ... more >> GSK Investigational Site Belfast, United Kingdom, BT9 6AD Collapse << |
| NCT01673490 | - | - | Terminated(The sole investigat... more >>ive site refused to accept the amended protocol and declined to continue the study. There was no safety signal nor any other reason.) Collapse << | - | - |
| NCT02014584 | Alopecia | Phase 3 | Completed | - | Chile ... more >> GSK Investigational Site Santiago, Región Metro De Santiago, Chile, 7580206 Hong Kong GSK Investigational Site Pokfulam, Hong Kong GSK Investigational Site Shatin, Hong Kong Korea, Republic of GSK Investigational Site Chungcheongnam-do, Korea, Republic of GSK Investigational Site Daejeon, Korea, Republic of, 301-721 GSK Investigational Site Gwangju, Korea, Republic of, 501-757 GSK Investigational Site Gyeonggi-do, Korea, Republic of, 463-707 GSK Investigational Site Incheon, Korea, Republic of, 400-711 GSK Investigational Site Jeonju-si, Jeollabuk-do, Korea, Republic of, 561-712 GSK Investigational Site Kangwon-Do, Korea, Republic of, 220-701 GSK Investigational Site Pusan, Korea, Republic of, 602-739 GSK Investigational Site Seoul, Korea, Republic of, 110-744 GSK Investigational Site Seoul, Korea, Republic of, 134-090 GSK Investigational Site Seoul, Korea, Republic of, 143-729 GSK Investigational Site Seoul, Korea, Republic of, 156-755 Singapore GSK Investigational Site Singapore, Singapore, 169608 Taiwan GSK Investigational Site Taipei, Taiwan, 106 GSK Investigational Site Taipei, Taiwan Collapse << |
| NCT01471678 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01758523 | Alcoholism Al... more >>cohol Abuse Alcohol Dependence Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 Collapse << |
| NCT01657851 | Prostatic Hyperplasia | Phase 1 | Completed | - | Russian Federation ... more >> GSK Investigational Site Reutov, Moscow Region, Russian Federation, 143964 Collapse << |
| NCT01831791 | Alopecia | Phase 3 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 812-0025 GSK Investigational Site Osaka, Japan, 530-0057 GSK Investigational Site Osaka, Japan, 532-0003 GSK Investigational Site Tokyo, Japan, 103-0028 GSK Investigational Site Tokyo, Japan, 160-0022 Collapse << |
| NCT00805701 | Prostate Cancer | Phase 4 | Completed | - | United States, Massachusetts ... more >> Bay State Clinical Trials, Inc. Watertown, Massachusetts, United States, 02472 Collapse << |
| NCT01831791 | - | - | Completed | - | - |
| NCT01254071 | Prostatic Hyperplasia | Phase 1 | Completed | - | Australia, New South Wales ... more >> GSK Investigational Site Randwick, New South Wales, Australia, 2031 Collapse << |
| NCT01262287 | Alcoholism Al... more >>cohol Abuse Alcohol Dependence Collapse << | Phase 4 | Completed | - | United States, Connecticut ... more >> University of Connecticut Health Center Farmington, Connecticut, United States, 06030 Collapse << |
| NCT00706966 | - | - | Completed | - | - |
| NCT00802321 | Alcohol Related Disorders ... more >> Alcoholism Alcohol Abuse Collapse << | Not Applicable | Completed | - | United States, Connecticut ... more >> Unversity of Connecticut Health Center Farmington, Connecticut, United States, 06030 Collapse << |
| NCT02509104 | Prostatic Hyperplasia | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT01004809 | - | - | Completed | - | Korea, Republic of ... more >> GSK Investigational Site Daejeon, Korea, Republic of, 301-721 Collapse << |
| NCT00985738 | - | - | Terminated(Low recruitment) | - | - |
| NCT02014584 | - | - | Completed | - | - |
| NCT00985738 | Prostate Cancer | Phase 2 | Terminated(Low recruitment) | - | United States, Colorado ... more >> University of Colorado Hospital Aurora, Colorado, United States, 80045 Collapse << |
| NCT01525914 | - | - | Completed | - | Canada, Ontario ... more >> Sunnybrook Health Sciences Centre Toronto, Ontario, Canada, M4N 3M5 Collapse << |
| NCT02578953 | Prostatic Hyperplasia | Phase 1 | Completed | - | Japan ... more >> GSK Investigational Site Fukuoka, Japan, 813-0017 Collapse << |
| NCT01262287 | - | - | Completed | - | - |
| NCT03240939 | Benign Prostatic Hyperplasia | Phase 1 | Completed | - | Korea, Republic of ... more >> Asan Medical Center Seoul, Korea, Republic of, 05505 Collapse << |
| NCT01957189 | Prostatic Hyperplasia | Phase 1 | Completed | - | China ... more >> GSK Investigational Site Shanghai, China, 200030 Collapse << |
| NCT00706966 | Nonmalignant Neoplasm ... more >> Prostate Cancer Collapse << | Not Applicable | Completed | - | United States, California ... more >> UCSF Helen Diller Family Comprehensive Cancer Center San Francisco, California, United States, 94115 Collapse << |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
