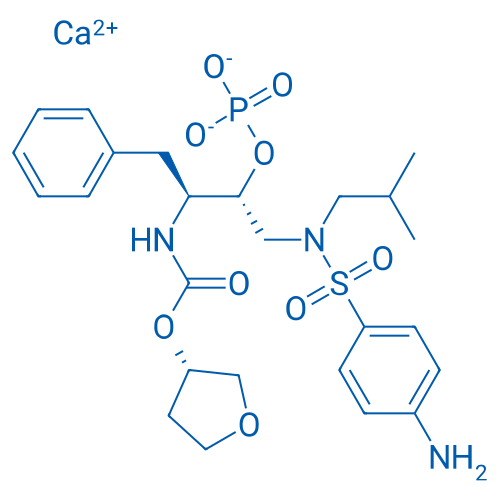
CAS No.: 226700-81-8
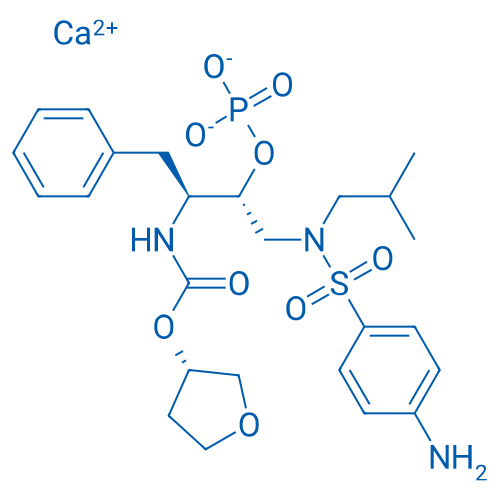
Fosamprenavir Calcium Salt/福沙那伟钙 Catalog No. CSN13578
Synonyms: GW433908G;Fosamprenavir (calcium salt);Fosamprenavir Calcium
Fosamprenavir Calcium the prodrug of amprenavir which is an HIV protease inhibitor with antiviral property.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03290131 Multiple Sclerosis ... more >> Spasticity, Muscle Collapse << Phase 3 Recruiting April 2019 United States, Arizona ... more >> Xenoscience Inc. Recruiting Phoenix, Arizona, United States, 85004 United States, California Neuro-Pain Medical Center Recruiting Fresno, California, United States, 93710 United States, Florida Meridien Research Recruiting Tampa, Florida, United States, 33634 Collapse << NCT03222349 Epilepsy Phase 2 Recruiting November 24, 2018 United States, Arizona ... more >> University of Arizona Recruiting Tucson, Arizona, United States, 85719 Contact: Sejal Jain United States, Florida NW FL Clinical Research Group, LLC Recruiting Gulf Breeze, Florida, United States, 32561 Contact: Weldon Mauney United States, New Jersey Children's St. Peters University Hospital Recruiting New Brunswick, New Jersey, United States, 08901 Contact: Carlos Lastra United States, New York Icahn School of Medicine at Mount Sinai Recruiting New York, New York, United States, 10029 Contact: Harriet Kang, MD 914-428-0529 Principal Investigator: Harriet Kang, MD Sub-Investigator: Patricia McGoldrick, NP Sub-Investigator: Steven Wolf, MD University of Rochester Recruiting Rochester, New York, United States, 14607 Contact: Inna Hughes United States, North Carolina Onsite Clinical Solutions LLC Recruiting Charlotte, North Carolina, United States, 28203 Contact: Robert Nahouraii United States, Pennsylvania Children's Hospital of Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Dennis Dlugos United States, Texas Dell Children's Medical Center Recruiting Austin, Texas, United States, 78723 Contact: David Clarke Austin Epilepsy Care Center Recruiting Austin, Texas, United States, 78758 Contact: Sami Aboumatar, MD 512-339-8831 sami@northaustinneuro.com United States, Virginia Virginia Commonwealth University Recruiting Richmond, Virginia, United States, 23298 Contact: Syndi Seinfeld Collapse << NCT03179891 Epilepsy Phase 2 Recruiting April 29, 2019 United States, Arizona ... more >> Arizona Health Sciences Center Recruiting Tucson, Arizona, United States, 85724-5023 Contact: David M. Labiner, MD 520-626-2006 United States, California Rancho Research Institute Recruiting Downey, California, United States, 90242 Contact: Hui Gong, MD 818-822-4916 hgong2@dhs.lacounty.gov United States, Connecticut Yale University School of Medicine-Comprehensive Epilepsy Center Recruiting New Haven, Connecticut, United States, 06520-8018 Contact: Kamil Detyniecki, MD 203-785-3865 kamil.detyniecki@yale.edu United States, Hawaii Hawaii Pacific Neuroscience Recruiting Honolulu, Hawaii, United States, 96817 Contact: Kore Liow, MD 808-261-4476 kliow@hawaiineuroscience.com United States, Maryland Mid-Atlantic Epilepsy and Sleep Center Recruiting Bethesda, Maryland, United States, 20817 Contact: Pavel Klein, M.B. 301-530-9744 kleinp@epilepsydc.com Contact: Diep Bui, M.D. United States, New Jersey Saint Peter's University Hospital Recruiting New Brunswick, New Jersey, United States, 08901 Contact: Carlos Lastra, MD 732-745-8600 clastra@saintpetersuh.com United States, New York University of Rochester Medical Center Recruiting Rochester, New York, United States, 14642 Contact: Trenton J. Tollefson, MD 507-273-4872 trenton_tollefson@urmc.rochester.edu United States, North Carolina Onsite Clinical Solutions LLC Recruiting Charlotte, North Carolina, United States, 28203 Contact: Robert Ataollah Nahouraii United States, Pennsylvania Hospital of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Michael A. Gelfand, MD, PhD 215-349-5166 michael.gelfand@uphs.upenn.edu United States, Texas Austin Epilepsy Care Center Recruiting Austin, Texas, United States, 78758 Contact: Sami Aboumatar, MD 512-339-8831 sami@northaustinneuro.com United States, Virginia Virginia Commonwealth University Medical Center Recruiting Richmond, Virginia, United States, 23219 Contact: Syndi A. Seinsefl, DO 804-828-0445 syndi.seinfeld@vcuhealth.org Collapse << - 更多
- 参考文献
- [1] Gruber VA, Rainey PM, et al. Interactions between buprenorphine and the protease inhibitors darunavir-ritonavir and fosamprenavir-ritonavir. Clin Infect Dis. 2012 Feb 1;54(3):414-23.
- [2] Falcoz C, Jenkins JM, et al. Pharmacokinetics of GW433908, a prodrug of amprenavir, in healthy male volunteers. J Clin Pharmacol. 2002 Aug;42(8):887-98.
- [3] The Triple Threat of HIV-1 Protease Inhibitors
- [4] Fosamprenavir : Clinical Pharmacokinetics and Drug Interactions of the Amprenavir Prodrug
- [5] Amprenavir or Fosamprenavir Plus Ritonavir in HIV Infection: Pharmacology, Efficacy and Tolerability Profile
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 226700-81-8 | 储存条件 |
|
|
| 分子式 | C25H34CaN3O9PS | 运输 | 蓝冰 | |
| 分子量 | 623.67 | 别名 | GW433908G;Fosamprenavir (calcium salt);Fosamprenavir Calcium;Lexiva;Amprenavir phosphate calcium;UNII-ID1GU2627N;GW-433908;GW-908;福沙那伟钙 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03290131 | Multiple Sclerosis ... more >> Spasticity, Muscle Collapse << | Phase 3 | Recruiting | April 2019 | United States, Arizona ... more >> Xenoscience Inc. Recruiting Phoenix, Arizona, United States, 85004 United States, California Neuro-Pain Medical Center Recruiting Fresno, California, United States, 93710 United States, Florida Meridien Research Recruiting Tampa, Florida, United States, 33634 Collapse << |
| NCT03222349 | Epilepsy | Phase 2 | Recruiting | November 24, 2018 | United States, Arizona ... more >> University of Arizona Recruiting Tucson, Arizona, United States, 85719 Contact: Sejal Jain United States, Florida NW FL Clinical Research Group, LLC Recruiting Gulf Breeze, Florida, United States, 32561 Contact: Weldon Mauney United States, New Jersey Children's St. Peters University Hospital Recruiting New Brunswick, New Jersey, United States, 08901 Contact: Carlos Lastra United States, New York Icahn School of Medicine at Mount Sinai Recruiting New York, New York, United States, 10029 Contact: Harriet Kang, MD 914-428-0529 Principal Investigator: Harriet Kang, MD Sub-Investigator: Patricia McGoldrick, NP Sub-Investigator: Steven Wolf, MD University of Rochester Recruiting Rochester, New York, United States, 14607 Contact: Inna Hughes United States, North Carolina Onsite Clinical Solutions LLC Recruiting Charlotte, North Carolina, United States, 28203 Contact: Robert Nahouraii United States, Pennsylvania Children's Hospital of Philadelphia Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Dennis Dlugos United States, Texas Dell Children's Medical Center Recruiting Austin, Texas, United States, 78723 Contact: David Clarke Austin Epilepsy Care Center Recruiting Austin, Texas, United States, 78758 Contact: Sami Aboumatar, MD 512-339-8831 sami@northaustinneuro.com United States, Virginia Virginia Commonwealth University Recruiting Richmond, Virginia, United States, 23298 Contact: Syndi Seinfeld Collapse << |
| NCT03179891 | Epilepsy | Phase 2 | Recruiting | April 29, 2019 | United States, Arizona ... more >> Arizona Health Sciences Center Recruiting Tucson, Arizona, United States, 85724-5023 Contact: David M. Labiner, MD 520-626-2006 United States, California Rancho Research Institute Recruiting Downey, California, United States, 90242 Contact: Hui Gong, MD 818-822-4916 hgong2@dhs.lacounty.gov United States, Connecticut Yale University School of Medicine-Comprehensive Epilepsy Center Recruiting New Haven, Connecticut, United States, 06520-8018 Contact: Kamil Detyniecki, MD 203-785-3865 kamil.detyniecki@yale.edu United States, Hawaii Hawaii Pacific Neuroscience Recruiting Honolulu, Hawaii, United States, 96817 Contact: Kore Liow, MD 808-261-4476 kliow@hawaiineuroscience.com United States, Maryland Mid-Atlantic Epilepsy and Sleep Center Recruiting Bethesda, Maryland, United States, 20817 Contact: Pavel Klein, M.B. 301-530-9744 kleinp@epilepsydc.com Contact: Diep Bui, M.D. United States, New Jersey Saint Peter's University Hospital Recruiting New Brunswick, New Jersey, United States, 08901 Contact: Carlos Lastra, MD 732-745-8600 clastra@saintpetersuh.com United States, New York University of Rochester Medical Center Recruiting Rochester, New York, United States, 14642 Contact: Trenton J. Tollefson, MD 507-273-4872 trenton_tollefson@urmc.rochester.edu United States, North Carolina Onsite Clinical Solutions LLC Recruiting Charlotte, North Carolina, United States, 28203 Contact: Robert Ataollah Nahouraii United States, Pennsylvania Hospital of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Michael A. Gelfand, MD, PhD 215-349-5166 michael.gelfand@uphs.upenn.edu United States, Texas Austin Epilepsy Care Center Recruiting Austin, Texas, United States, 78758 Contact: Sami Aboumatar, MD 512-339-8831 sami@northaustinneuro.com United States, Virginia Virginia Commonwealth University Medical Center Recruiting Richmond, Virginia, United States, 23219 Contact: Syndi A. Seinsefl, DO 804-828-0445 syndi.seinfeld@vcuhealth.org Collapse << |
| NCT03236207 | Cow Milk Allergy | Not Applicable | Recruiting | January 2019 | United States, California ... more >> Hoag Medical Group Recruiting Huntington Beach, California, United States, 92648 Contact: Rana Najjar 714-477-8080 Principal Investigator: Zabaneh, MD Stanford University Recruiting Mountain View, California, United States, 94040 Contact: Laurie Kost 650-724-0293 Principal Investigator: Sayantani Sindher, MD United States, Minnesota Clinical Research Institute Withdrawn Plymouth, Minnesota, United States, 55441 United States, Missouri Midwest Clinical Research Recruiting Saint Louis, Missouri, United States, 63141 Contact: Liz Poeppel, CCRC Contact liz@stlouisresearch.com Principal Investigator: Robert F Onder, MD United States, New Mexico Breathe America Recruiting Albuquerque, New Mexico, United States, 87109 Contact: Richard Wachs 856-417-6185 Principal Investigator: Richard Wachs, MD United States, New York Northwell Health System Recruiting Great Neck, New York, United States, 11021 Contact: Annica Bryson Contact abryson1@northwell.edu Principal Investigator: Punita Ponda, MD Icahn School of Medicine at Mount Sinai Recruiting New York, New York, United States, 10029 Contact: Christopher Knight Contact Christopher.knight1@mssm.edu Principal Investigator: Anna Nowak-Wegrzyn, MD United States, Oklahoma Allergy, Asthma and Clinical Research Center Recruiting Oklahoma City, Oklahoma, United States, 71320 Contact: Ricardo Pulido 405-286-9431 Principal Investigator: Martha Tarpay, MD United States, South Carolina Carolina ENT Recruiting Orangeburg, South Carolina, United States, 29118 Contact: Simone Ansley 803-378-1607 Principal Investigator: John Ansley, MD United States, Tennessee Memphis & Shelby County Pediatrics Recruiting Memphis, Tennessee, United States, 38116 Contact: Renita Talley renitact@hotmail.com Principal Investigator: James Carter, MD United States, Texas Houston Clinical Research Associates Recruiting Houston, Texas, United States, 77090 Contact: A Banker, MD abanker@hcratrials.com Principal Investigator: Siddiqi, MD Allergy Asthma Research Institute Recruiting Waco, Texas, United States, 76633 Contact: Dana Neilson dneilson@aactx.com Principal Investigator: Amar, MD Collapse << |
| NCT03319732 | Multiple Sclerosis ... more >> Spasticity, Muscle Collapse << | Phase 3 | Recruiting | May 2020 | United States, California ... more >> Neuro Pain Medical Center Recruiting Fresno, California, United States, 93710 Contact: Diana Sirico 908-809-1372 Collapse << |
| NCT03267108 | Pulmonary Fibrosis ... more >> Pulmonary Hypertension Collapse << | Phase 2 | Recruiting | December 2021 | United States, California ... more >> University of California Davis Health Recruiting Sacramento, California, United States, 95817 Contact: Macey Sockolov, CCRP 916-734-1554 mlsockolov@ucdavis.edu Principal Investigator: Roblee Allen, MD United States, Colorado UC Denver Anschutz Medical Center Recruiting Aurora, Colorado, United States, 80046 Contact: Colton Carter 303-724-7938 Contact: Cheryl Abbott, RN, CCRP 303-724-7466 Principal Investigator: Todd Bull, MD National Jewish Health Recruiting Denver, Colorado, United States, 80206 Contact: Kris Eliopoulos 303-270-2622 Principal Investigator: Amy Olson, MD United States, Florida University of Miami Recruiting Miami, Florida, United States, 33125 Contact: Emmanuelle Simonet, MA, CCRP 305-243-9383 Principal Investigator: Roger Alvarez, MD Central Florida Pulmonary Group Recruiting Orlando, Florida, United States, 33136 Contact: Sharon Foust, CCRC 407-841-1100 ext 135 sfoust@cfpulmonary.com Principal Investigator: Syed Mobin, MD United States, Georgia Piedmont Healthcare Recruiting Austell, Georgia, United States, 30106 Contact: Sheila Greene, MSA 770-745-1404 ext 2 sheila.greene@piedmont.org Principal Investigator: Amy Case, MD United States, Kentucky Kentuckiana Pulmonary Associates Recruiting Louisville, Kentucky, United States, 40202 Contact: Kimberly Robinson 502-587-8000 krobinson@kpadocs.com Principal Investigator: John McConnell, MD United States, Michigan University of Michigan Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Candace Flaherty, CCRP 734-936-8301 cflah@med.umich.edu Contact: Christi Getty 734-232-5844 cgetty@med.umich.edu Principal Investigator: Elizabeth Belloli, MD United States, Missouri The Lung Research Center, LLC Recruiting Chesterfield, Missouri, United States, 63017 Contact: Anna Shipp, RRT 314-682-3653 Anna.Shipp@stlukes-stl.com Contact: Kathleen McNulty, MHS 314-682-3653 Kathleen.McNulty@stlukes-stl.com Principal Investigator: Neil A Ettinger, MD, FACCP United States, Pennsylvania Temple University Recruiting Philadelphia, Pennsylvania, United States, 19140 Contact: Delores Fehrle 215-707-4260 delores.fehrle@tuhs.temple.edu Principal Investigator: Jeffrey Stewart, MD United States, South Carolina Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Jordyn DeMartino 843-792-8092 demartij@musc.edu Principal Investigator: Rahul Argula, MD United States, Tennessee Vanderbilt University Medical Center Recruiting Nashville, Tennessee, United States, 37232 Contact: James Del Greco, MSN, RN 615-343-7068 james.del.greco@vanderbilt.edu Principal Investigator: James Loyd, MD United States, Texas UT Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390 Contact: Oluwatosin Igenoza 214-645-6493 oluwatosin.igenoza@utsouthwestern.edu Principal Investigator: Sonja Bartolome, MD United States, Utah University of Utah Health Sciences Recruiting Salt Lake City, Utah, United States, 84108 Contact: Scott Sweeten 801-581-5811 Principal Investigator: MaryBeth Scholand, MD United States, Virginia Inova Heart and Vascular Institute Advanced Lung Disease Clinic Recruiting Falls Church, Virginia, United States, 22042 Contact: Serina Zorrilla, RN 703-776-6147 serina.zorrilla@inova.org Contact: Edwinia Battle, RN 703-776-3067 edwinia.battle@inova.org Principal Investigator: Christopher King, MD Pulmonary Associates of Richmond Recruiting Richmond, Virginia, United States, 23229 Contact: Betsy Daniel, CCRC 804-288-5945 bdaniel@paraccess.com Principal Investigator: Shilpa Johri, MD Collapse << |
| NCT03530553 | Weight Loss | Not Applicable | Recruiting | October 11, 2018 | United States, New Jersey ... more >> Penn Medicine Princeton Health Recruiting Plainsboro, New Jersey, United States, 08536 Contact: Missy Trammel 609-853-7220 mtrammel@princetonhcs.org Collapse << |
| NCT03478982 | Epilepsy | Phase 2 | Recruiting | May 2019 | United States, California ... more >> Havana Research Institute LLC. Recruiting Pasadena, California, United States, 91105 Contact grazy@havanaresearchinstitute.com Principal Investigator: John De Beixedon, MD United States, District of Columbia GW Medical Faculty Associates Recruiting Washington, District of Columbia, United States, 20037 Contact raly@mfa.gwu.edu Principal Investigator: Anumeha Sheth, MD, MD United States, Florida Advanced Pharma Cr, LLC Recruiting Miami, Florida, United States, 33147 Contact kblackburn@advancedpharmacr.com Principal Investigator: Kim Cruz, MD Research Institute of Orlando, LLC Recruiting Orlando, Florida, United States, 32806 Contact michele.nsor@gmail.com Principal Investigator: Ahmed Sadek, MD United States, Georgia Center for Rare Neurological Diseases Recruiting Norcross, Georgia, United States, 30093 Contact daniel@rareneuro.com Principal Investigator: Daniel Tarquinio, MD Clinical Research Institute Recruiting Stockbridge, Georgia, United States, 30281 Contact dwilliams@globaltrials.org Principal Investigator: Phillip Nowlin, MD United States, Hawaii Hawaii Pacific Neuroscience Recruiting Honolulu, Hawaii, United States, 96817 Contact: Director 808-261-4476 Principal Investigator: Kore Liow, M.D. United States, Louisiana Ochsner Health System Recruiting New Orleans, Louisiana, United States, 70121 Contact samantha.bright@ochsner.org Principal Investigator: Fawad Khan, MD United States, Maryland Mid-Atlantic Epilepsy And Sleep Center, LLC Recruiting Bethesda, Maryland, United States, 20817 Contact kleinp@epilepsydc.com Principal Investigator: Pavel Klein, MD United States, Nevada Impact Clinical Trials Las Vegas Recruiting Las Vegas, Nevada, United States, 89106 Contact fabiola@impactlasvegas.org Principal Investigator: Constance Brown, MD United States, New York SUNY Downstate Medical Center - Comprehensive Epilepsy Center Recruiting Brooklyn, New York, United States, 11203 Contact Reem.Deeb@downstate.edu Principal Investigator: Arthur Grant, MD NYU Comprehensive Epilepsy Center Recruiting New York, New York, United States, 10016 Contact Oluwatobi.clement@nyumc.org Principal Investigator: Blanca Vazquez, MD United States, North Carolina OnSite Clinical Solutions, LLC Recruiting Concord, North Carolina, United States, 28025 Contact TMeeks@onsiteclinical.com Principal Investigator: Michael Amiri, MD United States, Oregon Oregon Health & Science University - Brain Institute - Comprehensive Epilepsy Center Recruiting Portland, Oregon, United States, 97239 Contact dolan@ohsu.edu Principal Investigator: David Spencer, MD United States, Pennsylvania University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact Ashlie.Jefferson@uphs.upenn.edu Principal Investigator: Michael Gelfand, MD, PhD Thomas Jefferson University Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact Pranoti.Pradhan@jefferson.edu Principal Investigator: Michael Sperling, MD United States, Texas University of Texas Southwestern Medical Center - Neurology Clinic Recruiting Dallas, Texas, United States, 75390 Contact Giselle.Huet@UTSouthwestern.edu Principal Investigator: Hina Dave, MD UT Houston Recruiting Houston, Texas, United States, 77030 Contact Pamela.A.Weaver@uth.tmc.edu Principal Investigator: Omotola Hope, MD United States, Virginia Centra Medical Group Neurology Center Recruiting Lynchburg, Virginia, United States, 24502 Contact Joyce.White@Centrahealth.com Principal Investigator: Batool Kirmani, MD Collapse << |
| NCT03359525 | Tranexamic Acid Adverse Reacti... more >>on Collapse << | Phase 4 | Recruiting | December 31, 2019 | United States, New York ... more >> Albany Medical Center Recruiting Albany, New York, United States, 12208 Contact: Samik Banerjee, MBBS Contact: Ravneet Bhullar, MD bhullar@amc.edu Collapse << |
| NCT03581578 | - | - | Recruiting | June 2020 | United States, Arizona ... more >> Chandler Regional Medical Center Recruiting Chandler, Arizona, United States, 85224 Contact: Brian R Tiffany, MD Principal Investigator: Brian R Tiffany, MD United States, Connecticut Yale New Haven Hospital Recruiting New Haven, Connecticut, United States, 06519 Contact: Basmah Safdar, MD Principal Investigator: Basmah Safdar, MD United States, Kansas University of Kansas Medical Center Recruiting Kansas City, Kansas, United States, 66160 Contact: Chad M Cannon, MD Principal Investigator: Chad M Cannon, MD United States, Maryland University of Maryland Recruiting Baltimore, Maryland, United States, 21201 Contact: Robert Christenson, Ph.D. Principal Investigator: Robert Christenson, Ph.D. United States, Michigan Wayne State University/Detroit Receiving Hospital Recruiting Detroit, Michigan, United States, 48201 Contact: Elizabeth Dubey, MD Principal Investigator: Elizabeth Dubey, MD Henry Ford Health System Recruiting Detroit, Michigan, United States, 48202 Contact: Richard M Nowak, MD Principal Investigator: Richard M Nowak, MD Beaumont Recruiting Royal Oak, Michigan, United States, 48073 Contact: Carol Clark, MD Principal Investigator: Carol Clark, MD United States, Minnesota Hennepin County Medical Center/Minneapolis Medical Research Foundation Recruiting Minneapolis, Minnesota, United States, 55404 Contact: Fred Apple, PhD Principal Investigator: Fred Apple, PhD United States, New York Stony Brook University Hospital Recruiting Stony Brook, New York, United States, 11794 Contact: Adam J Singer, MD Principal Investigator: Adam J Singer, MD United States, Ohio University of Cincinnati Recruiting Cincinnati, Ohio, United States, 45267 Contact: Gregory Fermann, MD Principal Investigator: Gregory Fermann, MD United States, Oregon Oregon Health Science University Hospital Recruiting Portland, Oregon, United States, 97239 Contact: Bory Kea, MD Principal Investigator: Bory Kea, MD United States, Pennsylvania Thomas Jefferson University Hospital Recruiting Philadelphia, Pennsylvania, United States, 19107 Contact: Anna Marie Chang, MD Principal Investigator: Anna Marie Chang, MD United States, South Carolina Medical University of South Carolina Recruiting Charleston, South Carolina, United States, 29425 Contact: Gary F Headden, MD Principal Investigator: Gary F Headden, MD United States, Texas UT Southwestern Medical Center Not yet recruiting Dallas, Texas, United States, 75390 Contact: Ava Pierce, MD Principal Investigator: Ava Pierce, MD Texas Tech University Health Sciences Center El Paso Recruiting El Paso, Texas, United States, 79905 Contact: Edward Michelson, MD Principal Investigator: Edward Michelson, MD Ben Taub General Hospital / Baylor College of Medicine/Harris Health System Recruiting Houston, Texas, United States, 77030 Contact: Zubaid Rafique, MD Principal Investigator: Zubaid Rafique, MD United States, Virginia INOVA Alexandria Hospital Recruiting Alexandria, Virginia, United States, 22304 Contact: Brian Krieger, MD Principal Investigator: Brian Krieger, MD Virginia Commonwealth University Medical Center Recruiting Richmond, Virginia, United States, 29298 Contact: Christopher J Hogan, MD Principal Investigator: Christopher J Hogan, MD United States, Wisconsin Medical College of Wisconsin and Froedtert Hospital Not yet recruiting Milwaukee, Wisconsin, United States, 53226 Contact: Tom P Aufderheide, MD Principal Investigator: Tom P Aufderheide, MD Collapse << |
| NCT03747939 | Arthritis, Psoriatic | Phase 4 | Not yet recruiting | March 31, 2021 | - |
| NCT03415126 | Cancer Malign... more >>ancy Neoplasia Neoplasm Neoplasm Metastasis Colon Cancer Colonic Neoplasms Colon Cancer Liver Metastasis Metastatic Cancer Metastatic Melanoma Metastatic Colon Cancer Metastatic Lung Cancer Non Small Cell Lung Cancer Metastatic Pancreatic Cancer Pancreas Cancer Pancreas Adenocarcinoma Pancreas Neoplasm Metastatic Nonsmall Cell Lung Cancer Metastatic Pancreatic Cancer Collapse << | Phase 1 | Recruiting | May 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Principal Investigator: Ryan Sullivan, MD United States, Michigan START - Grand Rapids Recruiting Grand Rapids, Michigan, United States, 49546 Principal Investigator: Nehal Lakhani, MD United States, Texas MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Principal Investigator: Filip Janku, M.D. South Texas Accelerated Research Therapeutics Recruiting San Antonio, Texas, United States, 78229 Principal Investigator: Drew Rasco, M.D. NEXT Oncology Recruiting San Antonio, Texas, United States, 78240 Principal Investigator: Anthony Tolcher, M.D Collapse << |
| NCT03544567 | Angiosarcoma of Skin | Phase 1 | Recruiting | December 2020 | United States, Texas ... more >> Texas Oncology Not yet recruiting Dallas, Texas, United States, 75251 Contact: Stephanie Cannon, BS, CCRC 972-490-2939 Stephanie.Cannon@USOncology.com Principal Investigator: Robert Mennel, MD MD Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Gilberto Botello 713-792-3789 Gbotello@mdanderson.org Principal Investigator: Vinod Ravi, MD United States, Washington University of Washington/Fred Hutchinson Cancer Center Recruiting Seattle, Washington, United States, 98109 Contact: Roxanne Moore 206-606-6425 romoore@seattlecca.org Principal Investigator: Michael J. Wagner, MD Hong Kong Prince of Wales Hospital, Shatin Recruiting Hong Kong, Hong Kong Contact (852) 3505 2119 enquiry@clo.cuhk.edu.hk Principal Investigator: Herbert Loong, MD Collapse << |
| NCT03567226 | Glaucoma | Not Applicable | Recruiting | December 31, 2018 | China, Zhejiang ... more >> The affiliated eye hospital of Wenzhou Medical University Recruiting Wenzhou, Zhejiang, China Contact: Yuanbo Liang Collapse << |
| NCT03712280 | Hepatic Cirrhosis ... more >> Hepatic Encephalopathy Collapse << | Phase 2 | Recruiting | July 30, 2019 | United States, California ... more >> Anaheim Clinical Trials, LLC Recruiting Anaheim, California, United States, 92801 Southern California Research Center Recruiting Coronado, California, United States, 928118 Collapse << |
| NCT03428360 | Epilepsy | Phase 3 | Recruiting | June 7, 2019 | United States, Hawaii ... more >> Hawaii Neuroscience Center Recruiting Honolulu, Hawaii, United States, 96187 Contact: Kore Liow, MD 808-261-4476 kliow@hawaiineuroscience.com Principal Investigator: Kore Liow, MD Sub-Investigator: Patricia Borman, MD Sub-Investigator: Allison Przekop, DO Sub-Investigator: Paul Adapon III United States, Idaho Consultants in Epilepsy and Neurology, PLLC Recruiting Boise, Idaho, United States, 83702 Contact: Robert T Wechsler, MD 208-275-8585 rtw@idahoepilepsy.com Principal Investigator: Robert T Wechsler, MD Sub-Investigator: Jessica Bishop, PA-C Sub-Investigator: Lisa Gravett, RN United States, New Jersey Clinical Research Center of New Jersey (CRCNJ) Recruiting Voorhees, New Jersey, United States, 08043 Contact: Mark Mintz, MD 856-346-0005 ext 1101 mmintz@cnnh.org Principal Investigator: Mark Mintz, MD Sub-Investigator: Roger M Kurlan, MD United States, North Carolina OnSite Clinical Solutions LLC Recruiting Charlotte, North Carolina, United States, 28203 Contact: Robert A Nahouraii, MD 800-785-3150 rnahouraiiMD@onsiteclinical.com Principal Investigator: Robert A Nahouraii, MD Sub-Investigator: Kurt R Washburn, MD Sub-Investigator: Kayla "Brooke" Withers Sub-Investigator: Syed Mehdi United States, Pennsylvania Hospital of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Contact: Michael A Gelfand, MD 215-349-5166 michael.gelfand@uphs.upenn.edu Principal Investigator: Michael A Gelfand, MD Sub-Investigator: Ashlie Jefferson Sub-Investigator: Meryl Lozano Sub-Investigator: Kelly Ca Sub-Investigator: Lee Jeon United States, Texas Austin Epilepsy Care Center Recruiting Austin, Texas, United States, 78758 Contact: Sami Aboumatar 515-339-8831 sami@austinepilepsy.com Collapse << |
| NCT03288766 | - | - | Recruiting | January 2019 | United States, North Carolina ... more >> Vidant Medical Center Recruiting Greenville, North Carolina, United States, 27834 Contact: Connie Rogers, ADN CRogers@vidanthealth.com Collapse << |
| NCT03713099 | Cancer of the Lung ... more >> Lung Cancer Neoplasms, Lung Carcinoma, Non-Small Cell Lung Non-Small Cell Lung Cancer Non-Small Cell Lung Carcinoma Collapse << | Not Applicable | Not yet recruiting | December 31, 2020 | United States, California ... more >> City of Hope Not yet recruiting Duarte, California, United States, 91010 Contact: Jae Kim Principal Investigator: Jae Kim United States, Minnesota Mayo Clinic Not yet recruiting Rochester, Minnesota, United States, 55905 Contact: Janani Reisenauer Principal Investigator: Janani Reisenauer United States, New York New York Presbyterian-Weill Cornell Medicine Not yet recruiting New York, New York, United States, 10065 Contact: Bradley Pua Principal Investigator: Bradley Pua United States, North Carolina FirstHealth Moore Regional Hospital Not yet recruiting Pinehurst, North Carolina, United States, 28374 Contact: Michael Prithcett Principal Investigator: Michael Pritchett United States, Ohio Cleveland Clinic Not yet recruiting Cleveland, Ohio, United States, 44195 Contact: Thomas Gildea Principal Investigator: Thomas Gildea Collapse << |
| NCT03656692 | Uveitis, Posterior ... more >> Uveitis, Intermediate Panuveitis Collapse << | Phase 4 | Recruiting | February 27, 2020 | United States, Arizona ... more >> Retinal Research Institute, LLC Recruiting Phoenix, Arizona, United States, 85014 Retina Center PC Recruiting Tucson, Arizona, United States, 85704 United States, Illinois University Retina and Macula Associates PC Recruiting Lemont, Illinois, United States, 60439 United States, Massachusetts Massachusetts Eye Research and Surgery Institution (MERSI) Recruiting Waltham, Massachusetts, United States, 02451 United States, New Jersey Metropolitan Eye Research and Surgery Institute Recruiting Palisades Park, New Jersey, United States, 07650 United States, Texas Valley Retina Institute, PA Recruiting McAllen, Texas, United States, 78503 Collapse << |
| NCT03548909 | - | - | Recruiting | August 2019 | United States, Alabama ... more >> Drug Research Analysis Corp Recruiting Montgomery, Alabama, United States, 36106 Contact: John Frazer 334-265-2700 Principal Investigator: Steven G O'Mara, MD Collapse << |
| NCT00935480 | HIV Infections | Phase 3 | Completed | - | France ... more >> Centre Hospitalier Intercommunal de Toulon La Seyne sur Mer, Service d'infectiologie Toulon, France, 83056 Centre Hospitalier Intercommunal de Toulon La Seyne sur mer Toulon, France, 83500 Collapse << |
| NCT00027339 | HIV Infections | Phase 2 | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States Stanford CRS Palo Alto, California, United States, 94305-5107 UC Davis Medical Center Sacramento, California, United States, 95814 Ucsd, Avrc Crs San Diego, California, United States, 92103 United States, Colorado University of Colorado Hospital CRS Aurora, Colorado, United States, 80262 United States, Florida Univ. of Miami AIDS CRS Miami, Florida, United States United States, Indiana Indiana Univ. School of Medicine, Wishard Memorial Indianapolis, Indiana, United States, 46202 Methodist Hosp. of Indiana Indianapolis, Indiana, United States, 46202 United States, New York Beth Israel Med. Ctr., ACTU New York, New York, United States, 10003 NY Univ. HIV/AIDS CRS New York, New York, United States, 10016 United States, North Carolina Unc Aids Crs Chapel Hill, North Carolina, United States, 275997215 Duke Univ. Med. Ctr. Adult CRS Durham, North Carolina, United States, 27710 United States, Tennessee Vanderbilt Therapeutics CRS Nashville, Tennessee, United States, 37203 Puerto Rico Puerto Rico-AIDS CRS San Juan, Puerto Rico, 00936-5067 Collapse << |
| NCT01585753 | - | - | Completed | - | Argentina ... more >> Hospital General de Agudos "Dr. José María Ramos Mejía" Buenos Aires, Argentina, C1221ADC Fundacion IDEAA Buenos Aires, Argentina, C1405CKC Hospital Italiano de Buenos Aires Buenos Aires, Argentina Australia, New South Wales St. Vincent's Hospital Sydney, New South Wales, Australia, 2010 Germany Johann Wolfgang Goethe-University Hospital, Medical HIVCENTER Frankfurt, Frankfurt am Main, Germany, 60590 Thailand Chulalongkorn University Hospital Bangkok, Thailand, 10330 Collapse << |
| NCT03559387 | Chemotherapy-induced Neutropen... more >>ia Collapse << | Phase 2 | Recruiting | December 31, 2019 | France ... more >> Hôpital Saint Louis - Center des Maladies du Sein Not yet recruiting Paris, France, 75475 Institut de cancérologie Jean Godinot Not yet recruiting Reims, France, 51100 Strasbourg Oncologie Libérale Not yet recruiting Strasbourg, France, 67000 CHU de Tours Not yet recruiting Tours, France, 37044 Netherlands Erasmus Medical Center Recruiting Rotterdam, Netherlands, 3008 Ikazia Ziekenhuis Recruiting Rotterdam, Netherlands, 3038 Maasstad Ziekenhuis Recruiting Rotterdam, Netherlands, 3079 Franciscus Gasthuis & Vlietland Recruiting Schiedam, Netherlands, 3118 Collapse << |
| NCT00224172 | - | - | Completed | - | United States, New York ... more >> New York Presbyterian Hospital-Weill Cornell Medical College New York, New York, United States, 10065 Collapse << |
| NCT01348308 | HIV-1 Infection ... more >> AIDS Collapse << | Phase 3 | Completed | - | France ... more >> Hôpital Henri Mondor Creteil, France, 94010 Collapse << |
| NCT00501696 | Multiple Sclerosis | Phase 3 | Completed | - | United States, California ... more >> MS Center UCSF, Parnassus Ave, suite #908 SAn Francisco, California, United States, 94117 Collapse << |
| NCT00002453 | HIV Infections | Not Applicable | Completed | - | United States, California ... more >> Tenofovir Coordinating Center Foster City, California, United States, 94404 Collapse << |
| NCT02486510 | HIV-1 Infection | Early Phase 1 | Terminated(Futility criteria) | - | Spain ... more >> Hospital Universitario Ramon Y Cajal Madrid, Spain, 28034 Hospital Universitario La Paz Madrid, Spain, 28046 Collapse << |
| NCT01232361 | - | - | Completed | - | United States, Alabama ... more >> Univ. of Alabama Birmingham NICHD CRS (5096) Birmingham, Alabama, United States, 35294 United States, California Miller Children's Hospital Long Beach (5093) Long Beach, California, United States, 90806 Usc La Nichd Crs (5048) Los Angeles, California, United States, 90033 UCLA-Los Angeles/Brazil AIDS Consortium (LABAC) CR (3601) Los Angeles, California, United States, 90095 Univ of California, San Diego (UCSD) (4601) San Diego, California, United States, 92103 United States, Colorado Childrens Hospital (U. Colorado, Denver) NICHD CRS (5052) Denver, Colorado, United States, 80218-1088 United States, District of Columbia Children's National Med. Ctr. Washington DC NICHD CRS (5015) Washington, District of Columbia, United States, 20010 United States, Florida South Florida CDC Ft Lauderdale NICHD CRS (5055) Ft Lauderdal, Florida, United States, 33316 Univ of Miami Pediatric/Perinatal HIV/AIDS (4201) Miami, Florida, United States, 33136 United States, Illinois Chicago Children's CRS (4001) Chicago, Illinois, United States, 60611 Rush University Cook County Hospital NICHD CRS (5083) Chicago, Illinois, United States, 60612 United States, Maryland Johns Hopkins University NICHD CRS (5092) Baltimore, Maryland, United States, 21287 United States, Massachusetts Boston Medical Center Ped. HIV Program NICHD CRS (5011) Boston, Massachusetts, United States, 02118 WNE Maternal Pediatric Adolescent AIDS CRS (7301) Worcester, Massachusetts, United States, 01605 United States, Michigan Wayne State University/Children's Hospital of Michigan NICHD CRS (5041) Detroit, Michigan, United States, 48201 United States, New York Jacobi Med. Ctr. Bronx NICHD CRS (5013) Bronx, New York, United States, 10461 New York University NY (5012) New York, New York, United States, 10016 Columbia IMPAACT Center (4101) New York, New York, United States, 10032 SUNY Stony Brook (5040) Stony Brook, New York, United States, 11794-8111 United States, Tennessee St. Jude Childrens Research Hospital, Memphis (6501) Memphis, Tennessee, United States, 38105-2794 United States, Texas Texas Children's Hosp. CRS (3801) Houston, Texas, United States, 77030 Puerto Rico San Juan City Hosp. PR NICHD CRS (5031) San Juan, Puerto Rico, 00927 Collapse << |
| NCT01908660 | - | - | Completed | - | Spain ... more >> Hospital Universitario de Valme Seville, Spain, 41014 Collapse << |
| NCT00837616 | - | - | Completed | - | - |
| NCT02105987 | - | - | Completed | - | - |
| NCT03311568 | Critical Illness ... more >> Septic Shock Collapse << | Not Applicable | Recruiting | October 2022 | Norway ... more >> St Olavs Hospital, Dept Gastrointestinal Surgery, Dept Thoracic Surgery, Dept Intensive Care Medicine Recruiting Trondheim, Norway Contact: Daniel Bergum, md phd daniel.bergum@stolav.no Collapse << |
| NCT00001007 | HIV Infections | Phase 1 | Completed | - | United States, Alabama ... more >> Univ of Alabama at Birmingham Birmingham, Alabama, United States, 35294 United States, California Stanford Univ School of Medicine Stanford, California, United States, 94305 United States, Maryland Johns Hopkins Hosp - Pediatric Baltimore, Maryland, United States, 212874933 Johns Hopkins Hosp Baltimore, Maryland, United States, 21287 United States, Massachusetts Boston Med Ctr Boston, Massachusetts, United States, 02118 United States, North Carolina Duke Univ Med Ctr Durham, North Carolina, United States, 277103499 Collapse << |
| NCT00837616 | Turner Syndrome ... more >> Hypogonadism Premature Ovarian Failure Collapse << | Phase 4 | Completed | - | United States, Florida ... more >> Nemours Children's Clinic Jacksonville, Florida, United States, 32207 United States, Pennsylvania Jefferson Medical College of Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 Chile University of Chile/Clinica las Condes Santiago, Chile Collapse << |
| NCT00583076 | Pouchitis | Phase 2 | Completed | - | United States, Ohio ... more >> Cleveland Clinic Cleveland, Ohio, United States, 44195 Collapse << |
| NCT01431781 | Post-ERCP Acute Pancreatitis | Not Applicable | Unknown | March 2013 | China, Fujian ... more >> Fujian Province Hospital Active, not recruiting Fuzhou, Fujian, China China, Guangdong Nanfang Hospital Active, not recruiting Guangzhou, Guangdong, China China, Heilongjiang The People' Hospital of Heilongjiang Province Active, not recruiting Haerbin, Heilongjiang, China China, Jiangsu Nanjing Drum Tower Hospital Active, not recruiting Nanjing, Jiangsu, China China, Jiangxi The First Affiliated Hospital of Nanchang University Active, not recruiting Nanchang, Jiangxi, China China, Shanxi Xijing Hospital Active, not recruiting Xi,an, Shanxi, China China, Xinjiang Wulumuqi General Hospital of Chinese PLA Active, not recruiting Wulumuqi, Xinjiang, China China, Zhejiang Hangzhou First People Hospital Active, not recruiting Hangzhou, Zhejiang, China China Beijing Friendship Hospital Active, not recruiting Beijing, China Xinan Hospital Active, not recruiting Chongqing, China Jiangsu Province of TCM Active, not recruiting Nanjing, China Changhai Hospital Recruiting Shanghai, China Principal Investigator: Shen Zh Li, Pro. Eastern Hepatobiliary Surgery Hospital Active, not recruiting Shanghai, China Shanghai First People Hospital Active, not recruiting Shanghai, China Tongji Hospital Active, not recruiting Wuhan, China Collapse << |
| NCT00884793 | HIV Infections | Not Applicable | Completed | - | United States, California ... more >> San Francisco General Hospital San Francisco, California, United States, 94110 San Francisco VA Medical Center (SFVAMC) San Francisco, California, United States, 94121 Collapse << |
| NCT02105987 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Completed | - | - |
| NCT00884793 | - | - | Completed | - | - |
| NCT01054638 | - | - | Completed | - | - |
| NCT00005129 | - | - | Active, not recruiting | November 2023 | United States, Louisiana ... more >> Tulane University New Orleans, Louisiana, United States, 70112 Collapse << |
| NCT01137981 | - | - | Recruiting | December 31, 2024 | United States, North Carolina ... more >> GSK Investigational Site Recruiting Wilmington, North Carolina, United States, 28405 Contact: US Clincal Trials Call Center 877-379-3718 Contact: EU Clinical Trials Call Center +44 (0) 20 8990 4466 Collapse << |
| NCT00901979 | Type 2 Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00908323 | - | - | Completed | - | United States, Alabama ... more >> Alabama Vaccine CRS Birmingham, Alabama, United States, 35294 United States, California Bridge HIV CRS San Francisco, California, United States, 94143 United States, Washington Seattle Vaccine and Prevention CRS Seattle, Washington, United States, 98109-1024 Collapse << |
| NCT00624195 | HIV Infections | Phase 2 Phase 3 | Completed | - | United States, California ... more >> HIV Neurobehavioral Research Center, University of California San Diego San Diego, California, United States, 92103 University of California, San Francisco San Francisco, California, United States, 94110 United States, Maryland Johns Hopkins University- School of Medicine Baltimore, Maryland, United States, 21287 United States, Missouri Washington University St. Louis, Missouri, United States, 63110 United States, New York Mount Sinai Medical Center New York, New York, United States, 10024 Collapse << |
| NCT00307502 | HIV Infections | Phase 1 | Completed | - | Spain ... more >> Germans Trias i Pujol Hospital Badalona, Barcelona, Spain, 08916 Hospital de Figueres Figueras, Barcelona, Spain, 17600 Fundació Hospital-Asil de Granollers Granollers, Barcelona, Spain, 08400 Hospital de Vic Vic, Barcelona, Spain, 08500 Hospital Universitari Sant Joan de Reus Reus, Tarragona, Spain, 43201 Hospital de la Santa Creu i Sant Pau Barcelona, Spain, 08025 Collapse << |
| NCT00624195 | - | - | Completed | - | - |
| NCT01450618 | - | - | Completed | - | - |
| NCT02061410 | Knee Osteoarthritis | Not Applicable | Completed | - | Brazil ... more >> Exercise Research Laboratory, School of Physical Education, Federal University of Rio Grande do Sul Porto Alegre, RS, Brazil Collapse << |
| NCT00000868 | HIV Infections ... more >> HIV Seronegativity Collapse << | Phase 1 | Completed | - | United States, Maryland ... more >> JHU AVEG Baltimore, Maryland, United States, 21205 Collapse << |
| NCT00028366 | HIV Infections | Not Applicable | Completed | - | United States, California ... more >> USC CRS Los Angeles, California, United States, 90033-1079 Ucsf Aids Crs San Francisco, California, United States, 94110 United States, Georgia The Ponce de Leon Ctr. CRS Atlanta, Georgia, United States, 30308 United States, Indiana Indiana Univ. School of Medicine, Infectious Disease Research Clinic Indianapolis, Indiana, United States, 462025250 Indiana Univ. School of Medicine, Wishard Memorial Indianapolis, Indiana, United States, 46202 Methodist Hosp. of Indiana Indianapolis, Indiana, United States, 46202 United States, New York HIV Prevention & Treatment CRS New York, New York, United States United States, Ohio Univ. of Cincinnati CRS Cincinnati, Ohio, United States, 452670405 Case CRS Cleveland, Ohio, United States, 44106 MetroHealth CRS Cleveland, Ohio, United States, 441091998 The Ohio State Univ. AIDS CRS Columbus, Ohio, United States, 432101228 United States, Tennessee Vanderbilt Therapeutics CRS Nashville, Tennessee, United States, 37203 United States, Washington University of Washington AIDS CRS Seattle, Washington, United States, 98104 Collapse << |
| NCT02513147 | HIV-1 | Phase 4 | Completed | - | Spain ... more >> Hospital Universitari Vall d'Hebron Barcelona, Spain, 08035 Collapse << |
| NCT00877591 | Opioid Dependency ... more >> HIV Infections Collapse << | Phase 1 | Completed | - | United States, California ... more >> San Francisco General Hospital San Francisco, California, United States, 94110 Collapse << |
| NCT00543530 | HIV Infections | Phase 3 | Completed | - | - |
| NCT00092053 | Postmenopausal Osteoporosis | Phase 3 | Completed | - | - |
| NCT00085943 | HIV Infection ... more >> Infection, Human Immunodeficiency Virus Collapse << | Phase 3 | Completed | - | - |
| NCT03130959 | Various Advanced Cancer | Phase 2 | Recruiting | June 15, 2021 | - |
| NCT00756730 | HIV Infections | Phase 4 | Completed | - | United States, Arizona ... more >> Spectrum Medical Group Phoenix, Arizona, United States, 85012 United States, California AIDS Healthcare Foundation Los Angeles, California, United States, 02319 United States, Florida Orlando Immunology Center Orlando, Florida, United States, 32803 United States, Massachusetts Community Research Initiative Boston, Massachusetts, United States, 02215 Community Research Initiative - West Springfield, Massachusetts, United States, 01107 United States, Minnesota Abbott Northwestern Infectious Disease and Travel Clinic Minneapolis, Minnesota, United States, 55404 United States, New York AIDS Community Health Center Rochester, New York, United States, 14804 United States, Pennsylvania Philadelphia Fight Philadelphia, Pennsylvania, United States, 19107 United States, Texas David M. Lee, M.D., P.A., a/b/a Uptown Physicians Group Dallas, Texas, United States, 75204 Nicholaos C. Bellos, MD, PA Dallas, Texas, United States, 75204 United States, Wisconsin Medical College of Wisconsin Milwaukee, Wisconsin, United States, 53226 Collapse << |
| NCT00009061 | HIV Infections | Phase 3 | Unknown | - | - |
| NCT00122603 | HIV Infections | Phase 2 | Completed | - | France ... more >> Service des Maladies infectieuses et tropicales Hopital Bichat Claude Bernard Paris, France, 75018 Collapse << |
| NCT03762291 | Multiple Myeloma | Phase 1 | Not yet recruiting | November 1, 2026 | - |
| NCT00242840 | Infection, Human Immunodeficie... more >>ncy Virus I HIV Infection Collapse << | Phase 3 | Completed | - | United States, Florida ... more >> GSK Investigational Site Fort Lauderdale, Florida, United States, 33308 GSK Investigational Site Orlando, Florida, United States, 32804 GSK Investigational Site Tampa, Florida, United States, 33614 United States, Georgia GSK Investigational Site Atlanta, Georgia, United States, 30308/30309 GSK Investigational Site Atlanta, Georgia, United States, 30308 GSK Investigational Site Augusta, Georgia, United States, 30912 Italy GSK Investigational Site Brescia, Lombardia, Italy, 25125 Collapse << |
| NCT01384682 | HIV | Phase 4 | Completed | - | - |
| NCT00756730 | - | - | Completed | - | - |
| NCT01054586 | - | - | Completed | - | - |
| NCT00008554 | HIV Infections | Phase 3 | Unknown | - | United States, California ... more >> East Bay Clinical Trial Ctr Concord, California, United States, 94520 Ocean View Internal Medicine Long Beach, California, United States, 90803 United States, Florida Florida ID Group Orlando, Florida, United States, 32801 Hillsborough County Health Dept Tampa, Florida, United States, 33602 Clinical Pharmacology Services Tampa, Florida, United States, 33617 United States, Illinois Veterans Affairs Med Ctr of North Chicago Chicago, Illinois, United States, 60064 United States, Kansas Univ of Kansas Med Ctr Kansas City, Kansas, United States, 661607415 United States, New Jersey Saint Michael's Med Ctr Newark, New Jersey, United States, 07102 UMDNJ - New Jersey Med School Newark, New Jersey, United States, 071032757 United States, New York Bronx Municipal Hosp Ctr/Jacobi Med Ctr Bronx, New York, United States, 10461 Addiction Research and Treatment Corp Brooklyn, New York, United States, 11201 Brookdale Univ Hosp and Med Ctr Brooklyn, New York, United States, 112123198 Gervais Frechette New York, New York, United States, 10011 Howard Grossman New York, New York, United States, 10011 Mount Sinai School of Medicine New York, New York, United States, 10029 Univ of Rochester Med Ctr Rochester, New York, United States, 14642 United States, North Carolina SMO-USA Inc Charlotte, North Carolina, United States, 28211 United States, Oregon Advanced Clinical Trials Inc Eugene, Oregon, United States, 97401 United States, Pennsylvania Thomas Jefferson Univ Philadelphia, Pennsylvania, United States, 19107 United States, Texas Univ of Texas Med Branch Galveston, Texas, United States, 775550835 MacGregor Med Association Houston, Texas, United States, 77054 Walter Gaman Irving, Texas, United States, 75038 Southwest Texas Methodist Hosp San Antonio, Texas, United States, 78229 Collapse << |
| NCT00043888 | HIV Infections | Phase 3 | Completed | - | United States, Arkansas ... more >> GSK Clinical Trials Call Center Little Rock, Arkansas, United States, 72205 United States, California GSK Clinical Trials Call Center Long Beach, California, United States, 90813 GSK Clinical Trials Call Center Newport Beach, California, United States, 92663 GSK Clinical Trials Call Center San Diego, California, United States, 92101 United States, Colorado GSK Clinical Trials Call Center Denver, Colorado, United States, 80220 United States, District of Columbia GSK Clinical Trials Call Center Washington, District of Columbia, United States, 20036 United States, Florida GSK Clinical Trials Call Center Altamonte Springs, Florida, United States, 32701 GSK Clinical Trials Call Center Fort Lauderdale, Florida, United States, 33308 GSK Clinical Trials Call Center Fort Lauderdale, Florida, United States, 33316 GSK Clinical Trials Call Center Fort Lauderdale, Florida, United States, 33334 United States, Georgia GSK Clinical Trials Call Center Atlanta, Georgia, United States, 30339 United States, North Carolina GSK Clinical Trials Call Center Greenville, North Carolina, United States, 27858 United States, Pennsylvania GSK Clinical Trials Call Center Philadelphia, Pennsylvania, United States, 19107 France GSK Clinical Trials Call Center Lyon, France, 69437 GSK Clinical Trials Call Center Paris, France, 75475 GSK Clinical Trials Call Center Paris, France, 75679 United Kingdom GSK Clinical Trials Call Center London, United Kingdom, SE5 9RS GSK Clinical Trials Call Center Manchester, United Kingdom, M8 6RL Collapse << |
| NCT00148785 | HIV Infections | Phase 4 | Completed | - | United States, Georgia ... more >> Grady Infectious Diseases Program Atlanta, Georgia, United States, 30308 Collapse << |
| NCT00025727 | HIV Infections | Phase 3 | Unknown | - | - |
| NCT01054586 | - | - | Completed | - | - |
| NCT01077635 | - | - | Completed | - | - |
| NCT00775125 | Hepatitis C T... more >>elaprevir HIV Collapse << | Phase 1 | Completed | - | - |
| NCT00727597 | Human Immunodeficiency Virus I... more >>nfections Collapse << | Phase 3 | Completed | - | United States, District of Col... more >>umbia Georgetown University Washington, District of Columbia, United States, 20007 Collapse << |
| NCT00614991 | Healthy | Not Applicable | Completed | - | - |
| NCT00614991 | - | - | Completed | - | - |
| NCT00727597 | - | - | Completed | - | - |
| NCT00802074 | Healthy | Not Applicable | Completed | - | United States, New Jersey ... more >> Garden State Infectious Disease Associates, PA Voorhees, New Jersey, United States, 08043 Collapse << |
| NCT00335270 | HIV | Phase 4 | Completed | - | United States, Florida ... more >> University of Miami School of Medicine Miami, Florida, United States, 33136 Collapse << |
| NCT00481182 | Healthy Subjects | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21287-5554 United States, New York GSK Investigational Site Buffalo, New York, United States, 14215-1199 Collapse << |
| NCT00802074 | - | - | Completed | - | - |
| NCT01140412 | Healthy | Phase 1 | Terminated(The trial premature... more >>ly terminated on November 1, 2010, due to healthy volunteer participants experiencing non-serious fosamprenavir-related skin rash.) Collapse << | - | Singapore ... more >> Pfizer Investigational Site Singapore, Singapore, 188770 Collapse << |
| NCT00040664 | HIV Infection | Phase 2 | Completed | - | - |
| NCT00144833 | HIV-1 | Phase 3 | Terminated(Incomplete data) | - | - |
| NCT00040664 | - | - | Completed | - | - |
| NCT00242216 | HIV Infections | Phase 4 | Completed | - | United States, Texas ... more >> Thomas Street Health Center Houston, Texas, United States, 77009 Collapse << |
| NCT01290211 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Investigational Site Bruxelles, Belgium, B-1070 Collapse << |
| NCT00071760 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 2 | Active, not recruiting | June 2021 | United States, Florida ... more >> GSK Investigational Site Jacksonville, Florida, United States, 32209 United States, North Carolina GSK Investigational Site Durham, North Carolina, United States, 27705 Argentina GSK Investigational Site Buenos Aires, Argentina, 1405 Mexico GSK Investigational Site Mexico, D.F., Mexico, 06720 GSK Investigational Site Mexico, Mexico, 6720 Portugal GSK Investigational Site Almada, Portugal, 2805-267 GSK Investigational Site Amadora, Portugal, 2700 GSK Investigational Site Lisboa, Portugal, 1649-035 Puerto Rico GSK Investigational Site San Juan, Puerto Rico, 00935 Russian Federation GSK Investigational Site Moscow, Russian Federation, 105275 GSK Investigational Site Moscow, Russian Federation, 129110 GSK Investigational Site St. Petersburg, Russian Federation, 196645 South Africa GSK Investigational Site Durban, KwaZulu- Natal, South Africa, 4013 GSK Investigational Site Parow Valley, Western Province, South Africa, 7505 GSK Investigational Site Soweto, South Africa, 2013 Collapse << |
| NCT00450580 | - | - | Completed | - | - |
| NCT00242216 | - | - | Completed | - | - |
| NCT00764465 | - | - | Completed | - | - |
| NCT00450580 | Infection, Human Immunodeficie... more >>ncy Virus I HIV-1 Infection Collapse << | Phase 3 | Completed | - | - |
| NCT00363142 | HIV Infection ... more >> Infection, Human Immunodeficiency Virus Collapse << | Phase 3 | Completed | - | - |
| NCT00764465 | Healthy | Phase 2 | Completed | - | United States, New Jersey ... more >> Garden State Infectious Disease Associates,PA Voorhees, New Jersey, United States, 08043 Collapse << |
| NCT00089583 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 2 | Completed | - | - |
| NCT00071760 | - | - | Active, not recruiting | - | - |
| NCT00363142 | - | - | Completed | - | - |
| NCT00089583 | - | - | Completed | - | - |
| NCT00296504 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 3 | Completed | - | United States, California ... more >> GSK Investigational Site Fountain Valley, California, United States, 92708 GSK Investigational Site San Francisco, California, United States, 94115-1931 United States, Colorado GSK Investigational Site Denver, Colorado, United States, 80262 United States, Florida GSK Investigational Site Fort Lauderdale, Florida, United States, 33145 GSK Investigational Site Orlando, Florida, United States, 32804 GSK Investigational Site Orlando, Florida, United States, 32806 GSK Investigational Site Sarasota, Florida, United States, 34239 GSK Investigational Site Tampa, Florida, United States, 33614 United States, New York GSK Investigational Site Manhasset, New York, United States, 11030 United States, Texas GSK Investigational Site Galveston, Texas, United States, 77555-1188 Brazil GSK Investigational Site Campinas, São Paulo, Brazil, 13083970 Chile GSK Investigational Site Santiago, Región Metro De Santiago, Chile France GSK Investigational Site Le Kremlin Bicêtre Cedex, France, 94275 GSK Investigational Site Marseille, France, 13005 GSK Investigational Site Paris Cedex 10, France, 75475 GSK Investigational Site Paris Cedex 20, France, 75970 GSK Investigational Site Vandoeuvre Les Nancy Cedex, France, 54511 GSK Investigational Site Villejuif Cedex, France, 94805 Italy GSK Investigational Site Genova, Liguria, Italy, 16128 Portugal GSK Investigational Site Coimbra, Portugal, 3000-075 Spain GSK Investigational Site Badajoz, Spain, 6080 GSK Investigational Site Barcelona, Spain, 08036 United Kingdom GSK Investigational Site London, United Kingdom, NW3 2QG GSK Investigational Site London, United Kingdom, SE1 7EH Collapse << |
| NCT00977301 | HIV Infections | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud Universtity Nijmegen Medical Centre Nijmegen, Netherlands Collapse << |
| NCT01594983 | Non Familial Chylocmicronemia ... more >>Syndrome (Non-FCS) Collapse << | Phase 2 | Completed | - | - |
| NCT01387958 | Hepatitis C | Phase 2 | Terminated(Terminated early du... more >>e to futility) Collapse << | - | New Zealand ... more >> Novartis Investigative Site Auckland, New Zealand Novartis Investigative Site Christchurch, New Zealand Collapse << |
| NCT01146522 | Hyperlipoproteinemia | Phase 1 Phase 2 | Completed | - | Canada ... more >> Novartis Investigative Site Quebec, Canada Collapse << |
| NCT01209065 | Infections, Human Immunodefici... more >>ency Virus and Herpesviridae Collapse << | Phase 1 | Completed | - | United States, New York ... more >> GSK Investigational Site Buffalo, New York, United States, 14202 Collapse << |
| NCT01474434 | Coronary Artery Disease ... more >> Hypertriglyceridemia Collapse << | Phase 2 | Terminated(The study was termi... more >>nated based on interim analysis. See detailed description.) Collapse << | - | United States, California ... more >> Novartis Investigative Site Pasadena, California, United States, 91105 Collapse << |
| NCT01474434 | - | - | Terminated(The study was termi... more >>nated based on interim analysis. See detailed description.) Collapse << | - | - |
| NCT00240552 | Infection, Human Immunodeficie... more >>ncy Virus Collapse << | Phase 4 | Completed | - | Switzerland ... more >> GSK Investigational Site Aarau, Switzerland, 5001 GSK Investigational Site Basel, Switzerland, 4031 GSK Investigational Site Bern, Switzerland, 3010 GSK Investigational Site Bruderholz, Switzerland, 4101 GSK Investigational Site La Chaux-de-Fonds, Switzerland, CH 2301 GSK Investigational Site Lausanne, Switzerland, 1011 GSK Investigational Site Lugano, Switzerland, 6900 GSK Investigational Site Luzern, Switzerland, 6000 GSK Investigational Site St Gallen, Switzerland, 9007 GSK Investigational Site Zurich, Switzerland, 8008 GSK Investigational Site Zurich, Switzerland, 8038 GSK Investigational Site Zurich, Switzerland, 8091 Collapse << |
| NCT01010399 | Hypertriglyceridemia ... more >> HIV Infection Collapse << | Phase 4 | Completed | - | United States, California ... more >> Franco Felizarta, MD Bakersfield, California, United States, 93301 Collapse << |
| NCT01010399 | - | - | Completed | - | - |
| NCT00296504 | - | - | Completed | - | - |
| NCT01811472 | Non-alcoholic Fatty Liver Dise... more >>ase (NAFLD) Collapse << | Phase 2 | Completed | - | United States, Alabama ... more >> Novartis Investigative Site Mobile, Alabama, United States, 36608 United States, California Novartis Investigative Site San Diego, California, United States, 92114 United States, Florida Novartis Investigative Site Gainesville, Florida, United States, 32610-0277 Novartis Investigative Site Miami, Florida, United States, 33126 Novartis Investigative Site Tamarac, Florida, United States, 33319 United States, Hawaii Novartis Investigative Site Honolulu, Hawaii, United States, 96814 United States, Kentucky Novartis Investigative Site Louisville, Kentucky, United States, 40213 United States, Mississippi Novartis Investigative Site Tupelo, Mississippi, United States, 38801 United States, Texas Novartis Investigative Site Houston, Texas, United States, 77030 Novartis Investigative Site Plano, Texas, United States, 75093 United States, Virginia Novartis Investigative Site Richmond, Virginia, United States, 23298 Collapse << |
| NCT00094523 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 3 | Completed | - | - |
| NCT01811472 | - | - | Completed | - | - |
| NCT00817765 | HIV Infection ... more >> Fungal Infection Collapse << | Phase 1 | Completed | - | Netherlands ... more >> Radboud University Nijmegen Medical Centre Nijmegen, Gelderland, Netherlands Collapse << |
| NCT01589237 | Familial Chylomicronemia Syndr... more >>ome (FCS) (HLP Type I) Collapse << | Phase 3 | Terminated(Interim analysis su... more >>ggested that size of benefit anticipated from continued participation of patients in Part B no longer supported trial extension beyond Part A) Collapse << | - | United States, Washington ... more >> Novartis Investigative Site Seatlle, Washington, United States, 98104 Canada, Quebec Novartis Investigative Site Chicoutimi, Quebec, Canada, G7H 7P2 Novartis Investigative Site Ste-Foy, Quebec, Canada, G1V4M6 Canada Novartis Investigative Site Ouest-Montreal, Canada, H2W1R7 France Novartis Investigative Site Nantes, France, 44093 Novartis Investigative Site Paris Cedex 13, France, 75651 Germany Novartis Investigative Site Hamburg, Germany, 20246 Netherlands Novartis Investigative Site Meibergdreef 9, Netherlands, 1105 AZ South Africa Novartis Investigative Site Cape Town, South Africa, 7925 United Kingdom Novartis Investigative Site Manchester, United Kingdom, M13 9NT Collapse << |
| NCT01594957 | Hepatic Impairment | Phase 1 | Completed | - | United States, Florida ... more >> Novartis Investigative Site Miami, Florida, United States, 33136 Novartis Investigative Site Orlando, Florida, United States, 32809 Collapse << |
| NCT01514461 | Familial Chylomicronemia Syndr... more >>ome (FCS) Collapse << | Phase 3 | Completed | - | United States, Washington ... more >> Novartis Investigative Site Seatlle, Washington, United States, 98104 Canada, Quebec Novartis Investigative Site Chicoutimi, Quebec, Canada, G7H 7P2 Novartis Investigative Site Ste-Foy, Quebec, Canada, G1V4M6 Canada Novartis Investigative Site Ouest-Montreal, Canada, H2W1R7 France Novartis Investigative Site Bron, France, 69677 Novartis Investigative Site Nantes, France, 44093 Novartis Investigative Site Paris Cedex 13, France, 75651 Germany Novartis Investigative Site Hamburg, Germany, 20246 Novartis Investigative Site Köln, Germany, 50937 Netherlands Novartis Investigative Site Meibergdreef 9, Netherlands, 1105 AZ South Africa Novartis Investigative Site Cape Town, South Africa, 7925 Spain Novartis Investigative Site Malaga, Andalucia, Spain, 29010 Novartis Investigative Site Sevilla, Andalucia, Spain, 41013 United Kingdom Novartis Investigative Site Manchester, United Kingdom, M13 9NT Collapse << |
| NCT01589237 | - | - | Terminated(Interim analysis su... more >>ggested that size of benefit anticipated from continued participation of patients in Part B no longer supported trial extension beyond Part A) Collapse << | - | - |
| NCT01222611 | Chronic HIV Infection ... more >> HCV Coinfection Collapse << | Phase 4 | Completed | - | Spain ... more >> Hospital Príncipe de Asturias Alcalá de Henares, Madrid, Spain Hospital Gregorio Marañón Madrid, Spain, 28007 Hospital La Paz Madrid, Spain, 28046 Hospital Clínico San Carlos Madrid, Spain Hospital Doce de Octubre Madrid, Spain Hospital Ramón y Cajal Madrid, Spain Collapse << |
| NCT01514461 | - | - | Completed | - | - |
| NCT01558323 | Renal Impairment | Phase 1 | Completed | - | United States, Florida ... more >> Novartis Investigative Site Orlando, Florida, United States, 32809 United States, Tennessee Novartis Investigative Site Knoxville, Tennessee, United States, 37920 Collapse << |
| NCT00359788 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 4 | Completed | - | - |
| NCT00359788 | - | - | Completed | - | - |
| NCT00877929 | Hypertension | Phase 3 | Completed | - | - |
| NCT00909038 | - | - | Completed | - | - |
| NCT00886288 | - | - | Completed | - | - |
| NCT00877929 | - | - | Completed | - | - |
| NCT00886288 | - | - | Completed | - | - |
| NCT01491178 | - | - | Completed | - | - |
| NCT00197145 | Infection, Human Immunodeficie... more >>ncy Virus I Collapse << | Phase 3 | Terminated | - | - |
| NCT00909038 | - | - | Completed | - | - |
| NCT01721837 | - | - | Completed | - | - |
| NCT01491178 | - | - | Completed | - | - |
| NCT00197145 | - | - | Terminated | - | - |
| NCT02443116 | Nonalcoholic Steatohepatitis (... more >>NASH) Collapse << | Phase 2 | Recruiting | September 2019 | - |
| NCT01721837 | - | - | Completed | - | - |
| NCT02895100 | Ulcerative Colitis | Phase 2 | Terminated(Data Monitoring Com... more >>mittee discontinued the trial due to futility-based outcome) Collapse << | - | - |
| 靶点 | Description | IC50 |
|---|

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
