| NCT02612623 | Refractory Chronic Cough | Phase 2 | Completed | - | United States, California
... more >>
Afferent Investigative Site
Mission Viejo, California, United States
Afferent Investigative Site
San Jose, California, United States
United States, Colorado
Afferent Investigative Site
Colorado Springs, Colorado, United States
United States, Florida
Afferent Investigative Site
Largo, Florida, United States
United States, North Carolina
Afferent Investigative Site
Charlotte, North Carolina, United States
United States, Oklahoma
Afferent Investigative Site
Tulsa, Oklahoma, United States
United States, Texas
Afferent Investigative Site
Dallas, Texas, United States
Afferent Investigative Site
San Antonio, Texas, United States
United States, Washington
Afferent Investigative Site
Bellevue, Washington, United States
United States, Wisconsin
Afferent Investigative Site
Greenfield, Wisconsin, United States Collapse << |
| NCT01554579 | Osteoarthritis of the Knee | Phase 2 | Completed | - | - |
| NCT01554579 | - | - | Completed | - | - |
| NCT01569438 | - | - | Completed | - | - |
| NCT02612610 | Refractory Chronic Cough | Phase 2 | Completed | - | - |
| NCT01432730 | Chronic Cough | Phase 2 | Completed | - | United Kingdom
... more >> North West Lung Research Centre, Wythenshawe Hospital
Manchester, United Kingdom, M23 9LT Collapse << |
| NCT03449134 | Chronic Cough | Phase 3 | Recruiting | July 10, 2020 | - |
| NCT02612610 | - | - | Completed | - | - |
| NCT01569438 | Bladder Pain Syndrome | Phase 2 | Completed | - | - |
| NCT02502097 | Idiopathic Pulmonary Fibrosis
... more >>
Cough Collapse << | Phase 2 | Completed | - | - |
| NCT02349425 | Refractory Chronic Cough | Phase 2 | Completed | - | - |
| NCT03654326 | Endometriosis-related Pain | Phase 2 | Recruiting | October 22, 2019 | - |
| NCT03108924 | Renal Insufficiency | Phase 1 | Completed | - | United States, Florida
... more >>
Clinical Pharmacology of Miami ( Site 0001)
Hialeah, Florida, United States, 33014
Orlando Clinical Research Center ( Site 0002)
Orlando, Florida, United States, 32809 Collapse << |
| NCT03449147 | Chronic Cough | Phase 3 | Recruiting | January 8, 2021 | - |
| NCT03569033 | Acute Cough | Phase 2 | Recruiting | June 21, 2019 | United Kingdom
... more >> Hvivo Service Limited. Queen Mary BioEnterprises ( Site 0003)
Recruiting
London, United Kingdom, E1 2AX
Contact: Study Coordinator +442077561300 Collapse << |
| NCT02476890 | Refractory Chronic Cough | Phase 2 | Completed | - | - |
| NCT02790840 | Healthy | Phase 1 | Completed | - | United States, Arizona
... more >>
Celerion
Tempe, Arizona, United States, 85283 Collapse << |
| NCT03696108 | Chronic Cough | Phase 3 | Recruiting | October 6, 2020 | Japan
... more >> Oishi Clinic ( Site 3818)
Recruiting
Kasuya-gun, Fukuoka, Japan, 811-2310
Contact: Study Coordinator +81929381900
Nagata Hospital ( Site 3815)
Recruiting
Yanagawa, Fukuoka, Japan, 832-0059
Contact: Study Coordinator +81944723501
Japan Community Health care Organization Kanazawa Hospital ( Site 3817)
Recruiting
Kanazawa, Ishikawa, Japan, 920-8610
Contact: Study Coordinator +81762522200
Matsusaka City Hospital ( Site 3825)
Recruiting
Matsusaka, Mie, Japan, 515-8544
Contact: Study Coordinator +81598231515
Sugiura Clinic ( Site 3806)
Recruiting
Kawaguchi, Saitama, Japan, 332-0012
Contact: Study Coordinator +81482225015
National Hospital Organization Matsue Medical Center ( Site 3848)
Recruiting
Matsue, Shimane, Japan, 690-8556
Contact: Study Coordinator +81852216131
Kiheibashi Otolaryngology ( Site 3828)
Recruiting
Kodaira, Tokyo, Japan, 187-0044
Contact: Study Coordinator +81423323387
Tochigi Takao Clinic ( Site 3833)
Recruiting
Kagoshima, Japan, 890-0073
Contact: Study Coordinator +81998138111
Nagano Red Cross Hospital ( Site 3859)
Recruiting
Nagano, Japan, 380-8582
Contact: Study Coordinator +81262264131
Saiseikai Niigata Daini Hospital ( Site 3831)
Recruiting
Niigata, Japan, 950-1104
Contact: Study Coordinator +81252336161
Oita Red Cross Hospital ( Site 3837)
Recruiting
Oita, Japan, 870-0033
Contact: Study Coordinator +81975326181
Yamagata Clinic ( Site 3813)
Recruiting
Oita, Japan, 870-0921
Contact: Study Coordinator +81975562456
Chibana Clinic ( Site 3809)
Recruiting
Okinawa, Japan, 904-2143
Contact: Study Coordinator +81989391301
Ito ENT Clinic ( Site 3816)
Recruiting
Shizuoka, Japan, 420-0803
Contact: Study Coordinator +81542651000 Collapse << |
| NCT02492776 | Healthy | Phase 1 | Completed | - | United States, Arizona
... more >>
Celerion
Tempe, Arizona, United States, 85283 Collapse << |
| NCT01993329 | Asthma | Phase 1 | Completed | - | United Kingdom
... more >> Medicines Evaluation Unit
Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT02477709 | Idiopathic Pulmonary Fibrosis | Phase 2 | Completed | - | United States, North Carolina
... more >>
American Health Research
Charlotte, North Carolina, United States, 28207 Collapse << |
| NCT02476890 | - | - | Completed | - | - |
| NCT02477709 | - | - | Completed | - | - |
| NCT02229877 | Healthy Volunteers | Phase 1 | Completed | - | United States, Arizona
... more >>
Celerion
Tempe, Arizona, United States, 85283 Collapse << |
| NCT02397460 | Refractory Chronic Cough | Phase 2 | Completed | - | United Kingdom
... more >> The Medicines Evaluation Unit
Manchester, United Kingdom, M23 9QZ Collapse << |
| NCT03482713 | Chronic Cough | Phase 2 | Completed | - | Japan
... more >> Nagoya City University Hospital ( Site 3328)
Nagoya, Aichi, Japan, 467-8602
Idaimae Minamiyojo Int Clinic ( Site 3321)
Sapporo, Hokkaido, Japan, 064-0804
Tsumura Cardiovascular-internal-medicine Clinic ( Site 3314)
Kakogawa, Hyogo, Japan, 675-0101
Hitachi, Ltd. Hitachinaka General Hospital ( Site 3301)
Hitachinaka, Ibaraki, Japan, 312-0057
Saiseikai Kanazawa Hospital ( Site 3337)
Kanazawa, Ishikawa, Japan, 920-0353
Komatsu Municipal Hospital ( Site 3308)
Komatsu, Ishikawa, Japan, 923-8560
Kamei Internal Medicine and Respiratory Clinic ( Site 3309)
Takamatsu, Kagawa, Japan, 761-8073
Yokohama City Minato Red Cross Hospital ( Site 3306)
Yokohama, Kanagawa, Japan, 231-8682
Matsusaka City Hospital ( Site 3325)
Matsusaka, Mie, Japan, 515-8544
Nagaoka Red Cross Hospital ( Site 3307)
Nagaoka, Niigata, Japan, 940-2085
Kawaguchi Respiratory Clinic ( Site 3304)
Higashiosaka, Osaka, Japan, 577-0843
Fukushima Medical University Hospital ( Site 3338)
Fukushima, Japan, 960-1295
Doujin Memorial Medical Foundation, Meiwa Hospital ( Site 3310)
Tokyo, Japan, 101-0041
Nihonbashi Medical & Allergy Clinic ( Site 3334)
Tokyo, Japan, 103-0022
Fukuwa Clinic ( Site 3311)
Tokyo, Japan, 103-0027
Showa University Hospital ( Site 3331)
Tokyo, Japan, 142-8666 Collapse << |
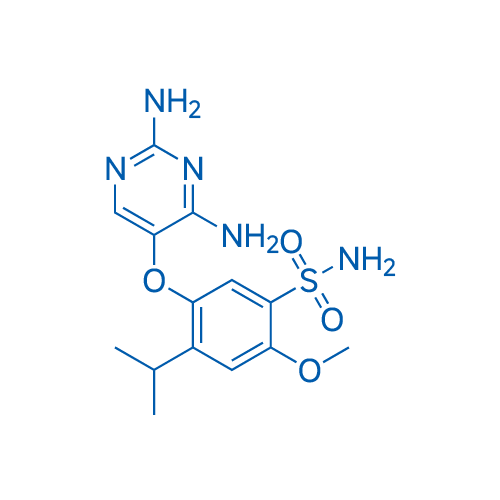
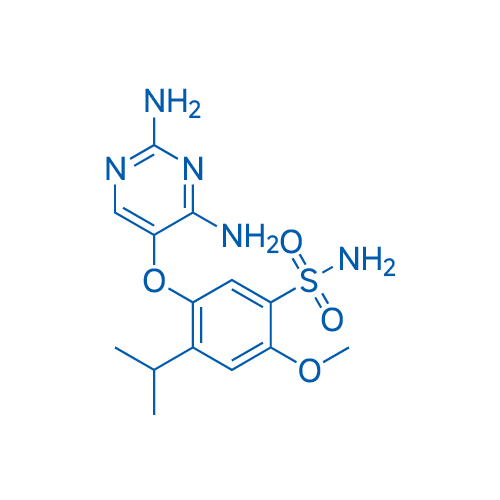

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
