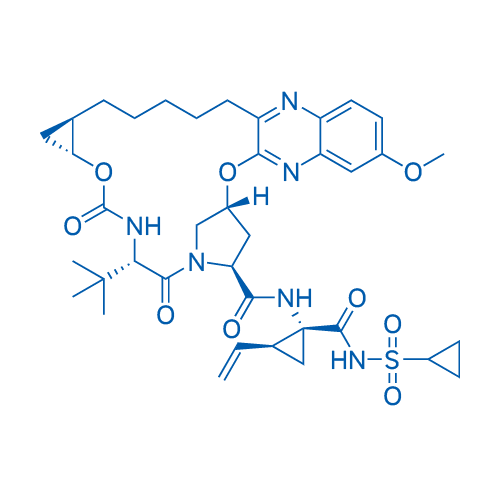
CAS No.: 1350514-68-9
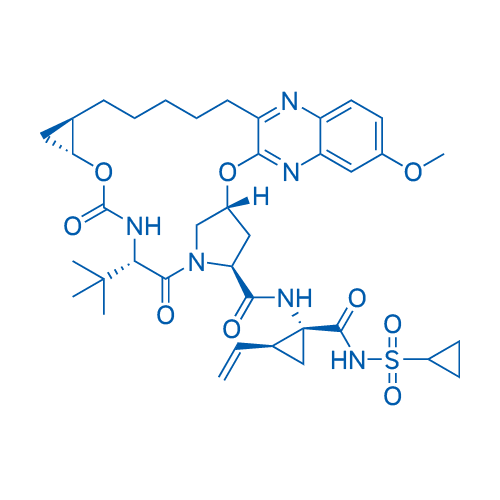
Grazoprevir/格拉瑞韦 Catalog No. CSN13898
Synonyms: MK-5172;格拉瑞韦
MK-5172 inhibits Hepatitis C virus NS3/4a protease with Ki of 0.01 nM (gt1b), 0.01 nM (gt1a), 0.08 nM (gt2a), 0.15 nM (gt2b), 0.90 nM (gt3a), respectively.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 HCV (gt1a)
Ki:0.01nMHCV (gt1b)
Ki:0.01nMHCV (gt2a)
Ki:0.08nMHCV (gt2b)
Ki:0.15nMHCV (gt3a)
Ki:0.90nM更多 
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03423641 - - Completed - - NCT02333292 - - Recruiting December 2020 Spain ... more >> Valme University Hospital Recruiting Seville, Andalusia, Spain, 41014 Contact: Karin I Neukam, Dr 0034955015871 karin.neukam@gmail.com Contact: Juan A Pineda, Dr 0034955015684 japineda@telefonica.net Collapse << NCT02057003 - - Recruiting December 2020 Spain ... more >> Valme University Hospital Recruiting Seville, Spain, 41014 Contact: Karin Neukam, PhD 0034955015799 karin.neukam@gmail.com Contact: Juan A Pineda, MD 0034955015684 japineda@telefonica.net Collapse << - 更多
- 参考文献
- [1] Summa V, Ludmerer SW, et al. MK-5172, a selective inhibitor of hepatitis C virus NS3/4a protease with broad activity across genotypes and resistant variants. Antimicrob Agents Chemother. 2012 Aug;56(8):4161-7.
- [2] Harper S, McCauley JA, et al. Discovery of MK-5172, a Macrocyclic Hepatitis C Virus NS3/4a Protease Inhibitor. ACS Med Chem Lett. 2012 Mar 2;3(4):332-6.
- [3] Summa V, Ludmerer SW, McCauley JA, et al. MK-5172, a selective inhibitor of hepatitis C virus NS3/4a protease with broad activity across genotypes and resistant variants. Antimicrob Agents Chemother. 2012 Aug;56(8):4161-7
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1350514-68-9 | 储存条件 |
|
|||||||
| 分子式 | C38H50N6O9S | 运输 | 蓝冰 | |||||||
| 分子量 | 766.90 | 别名 | MK-5172;格拉瑞韦 | |||||||
| 溶解度 |
|
动物实验配方 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03423641 | - | - | Completed | - | - |
| NCT02333292 | - | - | Recruiting | December 2020 | Spain ... more >> Valme University Hospital Recruiting Seville, Andalusia, Spain, 41014 Contact: Karin I Neukam, Dr 0034955015871 karin.neukam@gmail.com Contact: Juan A Pineda, Dr 0034955015684 japineda@telefonica.net Collapse << |
| NCT02057003 | - | - | Recruiting | December 2020 | Spain ... more >> Valme University Hospital Recruiting Seville, Spain, 41014 Contact: Karin Neukam, PhD 0034955015799 karin.neukam@gmail.com Contact: Juan A Pineda, MD 0034955015684 japineda@telefonica.net Collapse << |
| NCT03236506 | Hepatitis C | Phase 2 | Recruiting | March 2020 | United Kingdom ... more >> University of Dundee Not yet recruiting Dundee, Tayside, United Kingdom, DD1 9SY NHS Tayside Recruiting Dundee, United Kingdom, DD1 9SY Contact: John Dillon, MD Collapse << |
| NCT02858050 | - | - | Completed | - | United States, New Jersey ... more >> Saint Michael's Medical Center Newark, New Jersey, United States, 07102 Collapse << |
| NCT02743897 | End Stage Renal Disease | Phase 1 Phase 2 | Recruiting | December 2020 | United States, Pennsylvania ... more >> Hospital of the University of Pennsylvania Recruiting Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT03093740 | Renal Failure Chronic ... more >> Hepatitis C Collapse << | Phase 4 | Not yet recruiting | January 31, 2021 | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Collapse << |
| NCT03249194 | Kidney Transplant ... more >> Hepatitis C Collapse << | Early Phase 1 | Active, not recruiting | June 30, 2022 | United States, Virginia ... more >> Virginia Commonwealth University Richmond, Virginia, United States, 23298 Collapse << |
| NCT02858050 | - | - | Completed | - | - |
| NCT03420300 | Hepatitis C | Phase 4 | Recruiting | September 2019 | Taiwan ... more >> National Taiwan University Hospital, Yun-Lin Branch Recruiting Douliu, Taiwan Contact: Yu-Ren Fang, MD Sub-Investigator: Chen-Hua Liu, MD, PhD China Medical University Hospital Recruiting Taichung, Taiwan Contact: Cheng-Yuan Peng, MD, PhD Taichung Veterans General Hospital Recruiting Taichung, Taiwan Contact: Sheng-Shun Yang, MD, PhD Far Eastern Memorial Hospital Recruiting Taipei county, Taiwan Contact: Cheng-Kuan Lin, MD National Taiwan University Hospital Recruiting Taipei, Taiwan Contact: Chen-Hua Liu, MD, PhD +886-223123456 ext 63572 jacque_liu@mail2000.com.tw Sub-Investigator: Jia-Horng Kao, MD, PhD Sub-Investigator: Pei-Jer Chen, MD, PhD Sub-Investigator: Chun-Jen Liu, MD, PhD Sub-Investigator: Ding-Shinn Chen, MD Sub-Investigator: Hung-Chih Yang, MD, PhD Taipei Medical University Recruiting Taipei, Taiwan Contact: Wei-Yu Kao, MD Sub-Investigator: Chun-Chao Chang, MD, PhD Collapse << |
| NCT03146741 | Heart Failure | Phase 1 Phase 2 | Active, not recruiting | December 2020 | United States, Pennsylvania ... more >> Hospital of the University of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 Collapse << |
| NCT03724149 | Lung Diseases | Phase 1 Phase 2 | Not yet recruiting | November 2021 | United States, Pennsylvania ... more >> Hospital of the University of Pennsylvania Not yet recruiting Philadelphia, Pennsylvania, United States, 19104 Principal Investigator: David Goldberg, MD, MSCE Principal Investigator: Peter Reese, MD, MSCE Collapse << |
| NCT02895958 | Hepatitis C | Phase 4 | Recruiting | January 2019 | Ireland ... more >> Thompson Centre Recruiting Dublin, Ireland Contact: Suzanne Barror +35317164491 sbarror@gmail.com Collapse << |
| NCT02786537 | Chronic Hepatitis C | Phase 4 | Active, not recruiting | February 2022 | - |
| NCT03144440 | - | - | Active, not recruiting | January 1, 2020 | - |
| NCT03093415 | Hepatitis C S... more >>ubstance Use Disorders Substance Abuse, Intravenous Collapse << | Phase 4 | Enrolling by invitation | March 30, 2019 | United States, Oregon ... more >> Old Town Clinic Portland, Oregon, United States, 97214 Outside In Portland, Oregon, United States, 97214 Collapse << |
| NCT03221582 | Human Immunodeficiency Virus ... more >> Hepatitis C Collapse << | Phase 4 | Recruiting | December 1, 2019 | United States, Texas ... more >> Dallas VA Medical Center Recruiting Dallas, Texas, United States, 75216 Contact: Dindi Moore-Matthews, MS 214-857-1415 Dindi.Moore-Matthews@va.gov Contact: Ashley Liggion-Turk, BS 214-857-1606 Ashley.Liggion-Turk@va.gov Principal Investigator: Roger J Bedimo, MD Collapse << |
| NCT03127358 | Hepatitis C M... more >>edication Adherence Collapse << | Not Applicable | Recruiting | March 1, 2019 | United States, New York ... more >> Albert Einstein College of Medicine Division of Substance Abuse clinics Recruiting Bronx, New York, United States, 10461 Contact: Julia Arnsten, MD 718-944-3840 jarnsten@montefiore.org Principal Investigator: Julia Arnsten, MD Collapse << |
| NCT02781649 | - | - | Completed | - | - |
| NCT02781649 | End-Stage Renal Disease ... more >> Hepatitis C Collapse << | Phase 4 | Completed | - | United States, Maryland ... more >> Johns Hopkins Hospital Baltimore, Maryland, United States, 21205 Collapse << |
| NCT03407703 | - | - | Recruiting | December 31, 2019 | United States, California ... more >> University of California, San Francisco Enrolling by invitation San Francisco, California, United States, 94115 San Francisco VA Medical Center Recruiting San Francisco, California, United States, 94121 Contact: Phyllis C Tien, MD 415-221-4810 ext 22577 phyllis.tien@ucsf.edu Contact: Heather Freasier, MS 415-379-5518 heather.freasier@ucsf.edu Collapse << |
| NCT03105349 | HCV | Phase 4 | Withdrawn(No availability of i... more >>nvestigational medication.) Collapse << | February 2018 | Spain ... more >> Hospital Univ. La Paz Madrid, Madri, Spain, 28046 Hospital Univ. Gregorio Marañon Madrid, Spain, 28007 Hospital Infanta Leonor Madrid, Spain, 28031 Hospita 12 de octubre Madrid, Spain, 28041 Hospital Univ. La Paz Madrid, Spain, 28046 Collapse << |
| NCT02890719 | Liver Transplantation ... more >> Hepatitis C Collapse << | Phase 3 | Withdrawn(impossibility of sup... more >>plying the medication in research of the study by the pharmaceutical company that gave it to the trial) Collapse << | December 2017 | - |
| NCT02785666 | Hepatitis C H... more >>IV Collapse << | Phase 3 | Completed | - | Switzerland ... more >> Division of Infectious Diseases and Hospital Epidemiology, University Hospital Zurich, University of Zurich Zurich, Switzerland, 8091 Collapse << |
| NCT02897596 | Hepatitis C H... more >>IV Collapse << | Phase 3 | Active, not recruiting | October 2018 | Spain ... more >> Hospital Clínico y provincial de Barcelona Barcelona, Spain, 08036 Collapse << |
| NCT03365635 | Hepatitis C H... more >>emodialysis Nosocomial Infection Collapse << | Phase 4 | Not yet recruiting | April 2019 | United States, Pennsylvania ... more >> Penn Presbyterian Medical Center Philadelphia, Pennsylvania, United States, 19428 Collapse << |
| NCT03381859 | Hepatitis C Virus Infection, R... more >>esponse to Therapy of Collapse << | Phase 4 | Not yet recruiting | June 1, 2020 | Egypt ... more >> Kasr Alainy Hospital Not yet recruiting Cairo, Almanial, Egypt Contact: Rabab Fouad, MD +201069920756 Collapse << |
| NCT03585101 | Hepatitis C | Phase 4 | Withdrawn(Funding no longer av... more >>ailable) Collapse << | April 2020 | United States, California ... more >> UCLA CARE Center Los Angeles, California, United States, 90025 Collapse << |
| NCT03037151 | Compensated Cirrhosis | Phase 4 | Recruiting | January 19, 2022 | Thailand ... more >> Faculty of Medicine, Chulalongkorn University Recruiting Bangkok, Thailand, 10330 Contact: Pisit Tangkijvanich, MD pisittkvn@yahoo.com HIV-NAT, Thai Red Cross AIDS Research Centre Recruiting Bangkok, Thailand, 10330 Contact: Anchalee Avihingsanon, MD, PhD 662 652 3040 anchaleea2009@gmail.com Collapse << |
| NCT03110055 | HCV, HCC | Not Applicable | Not yet recruiting | May 1, 2019 | - |
| NCT03111966 | - | - | Recruiting | November 30, 2017 | Spain ... more >> HepaC Recruiting Madrid, Spain, 28020 Contact: Marta Hernández Conde 0034679611532 marta.hernandez.conde@gmail.com Collapse << |
| NCT02886624 | Acute Hepatitis C ... more >> HIV Collapse << | Phase 2 | Active, not recruiting | December 2019 | France ... more >> CHU de Lyon Lyon, France, 69317 CHU de Nice Nice, France, 06200 Hôpital Saint-Antoine Paris, France, 75012 Hôpital La Pitié-Salpêtrière Paris, France, 75013 Hôpital Bichat Paris, France, 75018 Hôpital Tenon Paris, France, 75020 Collapse << |
| NCT03186365 | Hepatitis C, Chronic | Phase 3 | Recruiting | October 31, 2018 | Taiwan ... more >> Kaohsiung Medical University Hospital Recruiting Kaohsiung, Taiwan, 807 Contact: Ming-Lung Yu, MD., PhD. +88673121101 ext 7475 fish6069@gmail.com Collapse << |
| NCT02661126 | - | - | Completed | - | - |
| NCT03026023 | Cardiac Transplant Disorder ... more >> Hepatitis C Collapse << | Phase 4 | Not yet recruiting | June 30, 2021 | United States, Massachusetts ... more >> Masschusetts General Hospital Not yet recruiting Boston, Massachusetts, United States, 02114 Contact: Jenna L Gustafson, MS 617-724-3836 jlgustafson@partners.org Contact: Raymond T Chung, MS rchung@partners.org Sub-Investigator: Emily Bethea, MD Collapse << |
| NCT03145623 | - | - | Completed | - | France ... more >> University Hospital Caen, France, 14000 University Hospital Toulouse Toulouse, France, 31059 Collapse << |
| NCT02661126 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT02902120 | Hepatitis C R... more >>enal Insufficiency, Chronic Disorder of Transplanted Kidney Collapse << | Phase 4 | Recruiting | June 2019 | United States, Maryland ... more >> University of Maryland Medical Center Recruiting Baltimore, Maryland, United States, 21201 Contact: Jennifer Husson, MD 410-706-6973 Contact: Ilise Marrazzo, RN 4107062564 Collapse << |
| NCT03022006 | Chronic Hepatitis C | Phase 4 | Recruiting | - | Japan ... more >> Hokkaido University Recruiting Sapporo, Hokkaido, Japan, 060-8638 Contact: Goki Suda, Dr Collapse << |
| NCT02647632 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT02945150 | Renal Failure | Phase 4 | Recruiting | December 2020 | United States, Massachusetts ... more >> Massachusetts General Hospital Recruiting Boston, Massachusetts, United States, 02114 Contact: Donald Chute, BA LiverResearchGroup@partners.org Principal Investigator: Raymond T Chung, MD Sub-Investigator: Meghan E Sise, MD Collapse << |
| NCT03496233 | Chronic HCV Hepatitis | Phase 3 | Not yet recruiting | December 2018 | Italy ... more >> Policlinico Paolo Giaccone Not yet recruiting Palermo, Italy, 90127 Contact: Vincenza Calvaruso, MD, PHD vincenza.calvaruso@unipa.it Collapse << |
| NCT03359746 | Hepatitis C | Phase 4 | Not yet recruiting | December 31, 2018 | - |
| NCT02823457 | Patient Adherence, Chronic Hep... more >>atitis C, Alcohol-related Disorders Collapse << | Not Applicable | Recruiting | March 2019 | United States, Mississippi ... more >> GV Sonny Montgomery VAMC Recruiting Jackson, Mississippi, United States, 39216 Contact: Mary Burton, MD Collapse << |
| NCT03379506 | HCV Infection | Phase 2 | Recruiting | June 17, 2020 | United States, California ... more >> University of California San Francisco ( Site 0020) Recruiting San Francisco, California, United States, 94158 Contact: Study Coordinator 415-476-1756 United States, Florida Florida Hospital ( Site 0006) Recruiting Orlando, Florida, United States, 32803 Contact: Study Coordinator 407-303-7756 United States, Georgia Children's Center for Advanced Pediatrics ( Site 0204) Recruiting Atlanta, Georgia, United States, 30329 Contact: Study Coordinator 404-785-0421 United States, Massachusetts Children's Hospital Boston ( Site 0009) Recruiting Boston, Massachusetts, United States, 02115 Contact: Study Coordinator 617-355-5837 United States, Ohio Cincinnati Childrens Hospital Medical Center ( Site 0003) Recruiting Cincinnati, Ohio, United States, 45229 Contact: Study Coordinator 513-636-7511 United States, Pennsylvania Children's Hospital of Pittsburgh ( Site 0024) Recruiting Pittsburgh, Pennsylvania, United States, 15224 Contact: Study Coordinator 412-692-6444 United States, Texas American Research Corporation ( Site 0200) Recruiting San Antonio, Texas, United States, 78215 Contact: Study Coordinator 210-918-8851 United States, Washington Children's Hospital and Medical Center, [Seattle, WA] ( Site 0017) Recruiting Seattle, Washington, United States, 98105 Contact: Study Coordinator 206-987-4636 Germany Medizinische Hochschule Hannover Kinderklinik K10 ( Site 0105) Recruiting Hannover, Germany, 30625 Contact: Study Coordinator +495115323208 Klinikum Starnberg ( Site 0107) Recruiting Starnberg, Germany, 82319 Contact: Study Coordinator +498151182330 Helios Klinikum Wuppertal GmbH ( Site 0104) Recruiting Wuppertal, Germany, 42283 Contact: Study Coordinator +492028963831 Poland WSOZ im.T.Browicza w Bydgoszczy ( Site 0800) Recruiting Bydgoszcz, Poland, 85-030 Contact: Study Coordinator +48602575396 Wojewodzki Specjalistyczny Szpital im. dr W. Bieganskiego w Lodzi ( Site 0810) Recruiting Lodz, Poland, 91-347 Contact: Study Coordinator +48601272443 MED-POLONIA Sp. z o.o. ( Site 0808) Recruiting Poznan, Poland, 60-693 Contact: Study Coordinator +48616561700 Sweden Karolinska Universitetssjukhuset Huddinge. ( Site 0062) Recruiting Stockholm, Sweden, 141 86 Contact: Study Coordinator +46858587321 Collapse << |
| NCT02940691 | Hepatitis C | Phase 4 | Active, not recruiting | February 2019 | Australia, New South Wales ... more >> Kirketon Road Centre Darlinghurst, New South Wales, Australia, 2010 St Vincent's Hospital Darlinghurst, New South Wales, Australia, 2010 The Langton Centre Darlinghurst, New South Wales, Australia, 2010 Nepean Hospital Kingswood, New South Wales, Australia, 2751 Drug and Alcohol Clinical Services (Hunter) Newcastle, New South Wales, Australia, 2300 Collapse << |
| NCT02601573 | - | - | Completed | - | - |
| NCT02601573 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT03222167 | Chronic Hepatitis C Genotype 1... more >>B Metabolic Syndrome Fibrosis, Liver Cirrhoses, Liver Collapse << | Phase 3 | Not yet recruiting | August 2019 | Kazakhstan ... more >> National Research Institute of Cardiology and Internal Medicine Not yet recruiting Almaty, Kazakhstan, 050000 Contact: Alexandre V. Nersesov, MD, Prof. +7 701 799 82 12 alexander.nersesov@gmail.com Contact: Almagul Jumabayeva, MD, PhD +7 701 512 23 26 almusa010@mail.ru Collapse << |
| NCT03723824 | Chronic Hepatitis c ... more >> Liver Transplant Infection Kidney Transplant Infection Collapse << | Phase 4 | Not yet recruiting | December 31, 2021 | - |
| NCT01353911 | - | - | Completed | - | - |
| NCT03111108 | Hepatitis C Virus (HCV) Infect... more >>ion Collapse << | Phase 4 | Completed | - | France ... more >> CHU Amiens-Picardie - Hopital Sud ( Site 0217) Amiens, France CHU Jean Minjoz ( Site 0213) Besancon, France CHU Henri Mondor ( Site 0206) Creteil, France CHU de Grenoble - Hopital Michallon ( Site 0208) Genoble, France CHU Dupuytren ( Site 0209) Limoges, France Hopital Saint Eloi ( Site 0207) Montpellier, France C.H.U. de Nice Hopital de l Archet 2 ( Site 0215) Nice, France Centre Hospitalier Regional du Orleans ( Site 0212) Orleans, France Hopital Beaujon ( Site 0201) Paris, France Hopital Cochin ( Site 0211) Paris, France Hopital Saint Antoine ( Site 0200) Paris, France CHU de Toulouse - Hopital Purpan ( Site 0216) Toulouse, France CHU de Nancy Hopital Brabois Adultes ( Site 0204) Vandoeuvre les Nancy, France Collapse << |
| NCT02332707 | - | - | Completed | - | - |
| NCT02332707 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT01353911 | Hepatitis C, Chronic | Phase 2 | Completed | - | - |
| NCT03706222 | - | - | Not yet recruiting | June 2019 | - |
| NCT03578640 | Hepatitis C, Chronic | Phase 3 | Not yet recruiting | February 28, 2019 | Saudi Arabia ... more >> King Fahad Medical City Not yet recruiting Riyadh, Saudi Arabia, 11525 Contact: Omar H Kasule, PhD +966 11 288 9999 ext 26913 okasule@kfmc.med.sa Contact: Isamme N AlFayyad, MA BioEthics +966 11 288 9999 ext 10848 ialfayyad@kfmc.med.sa Principal Investigator: Ahmad A AlEid, Consultant Sub-Investigator: Abdullah A AlKhathlan, Consultant Sub-Investigator: Areej N Al Balkhi, MBBS Sub-Investigator: Adel Al Qutub, Consultant Collapse << |
| NCT02613403 | - | - | Terminated | - | - |
| NCT03098121 | To Assess the Efficacy of Graz... more >>oprevir 100mg and Elbasvir 50mg by Determining the Proportion of Sustained Virological Response 12 Weeks After the End of Therapy Collapse << | Phase 4 | Not yet recruiting | March 31, 2018 | - |
| NCT02973503 | Chronic HCV Infection | Phase 3 | Recruiting | February 2018 | France ... more >> CHU Clermont-Ferrand Recruiting Clermont-Ferrand, France, 63003 Contact: Patrick LACARIN 04 73 75 11 95 placarin@chu-clermontferrand.fr Principal Investigator: Armand ABERGEL Collapse << |
| NCT03144635 | Hepatitis C Viral ... more >> Chronic Kidney Disease stage3 Collapse << | Phase 4 | Recruiting | March 31, 2020 | Japan ... more >> Kyushu University Hospital Recruiting Fukuoka, Japan, 812-8582 Contact: Eiichi Ogawa, MD, PhD +81-92-642-5909 eogawa@gim.med.kyushu-u.ac.jp Collapse << |
| NCT02613403 | Hepatitis Hep... more >>atitis C Digestive System Diseases Flaviviridae Infections Hepatitis, Viral, Human Liver Diseases RNA Virus Infections Virus Diseases Collapse << | Phase 2 | Terminated | - | - |
| NCT02203149 | Hepatitis C | Phase 2 Phase 3 | Completed | - | - |
| NCT02332720 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT02105454 | - | - | Completed | - | - |
| NCT02105467 | - | - | Completed | - | - |
| NCT02105454 | Hepatitis C Virus | Phase 2 | Completed | - | - |
| NCT02105467 | Chronic Hepatitis C Virus | Phase 3 | Completed | - | - |
| NCT02600325 | Acute Hepatitis C ... more >> Human Immunodeficiency Virus Hepatitis C Collapse << | Phase 3 | Active, not recruiting | July 2018 | Belgium ... more >> Institute of Tropical Medicine Antwerp (ITG) Antwerpen, Belgium Netherlands Erasmus Medical Center (EMC) Rotterdam, Zuid Holland, Netherlands, 3000 CA Onze Lieve Vrouwe Gasthuis (OLVG) Amsterdam, Netherlands Slotervaart Hospital Amsterdam, Netherlands Rijnstate Hospital Arnhem, Netherlands University Medical Center Groningen (UMCG) Groningen, Netherlands Maastricht University Medical Center (MUMC) Maastricht, Netherlands Radbout University Medical Center Nijmegen, Netherlands Utrecht Medical University Center (UMCU) Utrecht, Netherlands Collapse << |
| NCT01547312 | Chronic Hepatitis C | Phase 1 | Withdrawn | November 2012 | - |
| NCT02332720 | - | - | Completed | - | - |
| NCT01390428 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT02203149 | - | - | Completed | - | - |
| NCT01390428 | - | - | Completed | - | - |
| NCT03143998 | Hepatitis C | Phase 4 | Withdrawn(Business reasons) | January 12, 2019 | - |
| NCT02204475 | Hepatitis C | Phase 3 | Withdrawn | September 2016 | - |
| NCT02358044 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT01717326 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT01937975 | Chronic Hepatitis C ... more >> Renal Impairment Collapse << | Phase 1 | Completed | - | - |
| NCT02358044 | - | - | Completed | - | - |
| NCT02252016 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00998985 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT01716156 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT01717326 | - | - | Completed | - | - |
| NCT02252016 | - | - | Completed | - | - |
| NCT02732405 | Hepatitis C C... more >>ompensated Cirrhosis Collapse << | Phase 3 | Unknown | March 2017 | - |
| NCT00998985 | - | - | Completed | - | - |
| NCT01716156 | - | - | Completed | - | - |
| NCT01932762 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT01937975 | - | - | Completed | - | - |
| NCT01710501 | Chronic Hepatitis C (CHC) | Phase 2 | Completed | - | - |
| NCT01932762 | - | - | Completed | - | - |
| NCT01710501 | - | - | Completed | - | - |
| NCT01667081 | - | - | Enrolling by invitation | July 7, 2023 | - |
| NCT02092350 | Hepatitis C Virus | Phase 2 Phase 3 | Completed | - | - |
| NCT02092350 | - | - | Completed | - | - |
| NCT02105701 | Hepatitis C Infection | Phase 3 | Completed | - | - |
| NCT01440595 | Hepatitis C, Chronic | Phase 2 | Terminated(Preliminary results... more >> of MK-5172 PN003 (NCT01353911) suggested a possible dose relationship to elevated transaminase levels in treatment with grazoprevir.) Collapse << | - | - |
| NCT02105701 | - | - | Completed | - | - |
| NCT02115321 | Chronic Hepatitis C | Phase 2 Phase 3 | Completed | - | - |
| NCT02115321 | - | - | Completed | - | - |
| NCT01537900 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT02133131 | - | - | Completed | - | - |
| NCT01537900 | - | - | Completed | - | - |
| NCT01440595 | - | - | Terminated(Preliminary results... more >> of MK-5172 PN003 (NCT01353911) suggested a possible dose relationship to elevated transaminase levels in treatment with grazoprevir.) Collapse << | - | - |
| NCT02105662 | Chronic Hepatitis C | Phase 3 | Completed | - | - |
| NCT02133131 | Hepatitis C | Phase 2 | Completed | - | - |
| NCT02251990 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT03433326 | - | - | Not yet recruiting | December 31, 2018 | - |
| NCT02105662 | - | - | Completed | - | - |
| NCT02105688 | Chronic Hepatitis C | Phase 3 | Active, not recruiting | December 6, 2018 | - |
| NCT02251990 | - | - | Completed | - | - |
| NCT02105688 | - | - | Active, not recruiting | - | - |
| 靶点 | Description | IC50 |
|---|---|---|
| HCV (gt1a) | Ki:0.01nM | |
| HCV (gt1b) | Ki:0.01nM | |
| HCV (gt2a) | Ki:0.08nM | |
| HCV (gt2b) | Ki:0.15nM | |
| HCV (gt3a) | Ki:0.90nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
