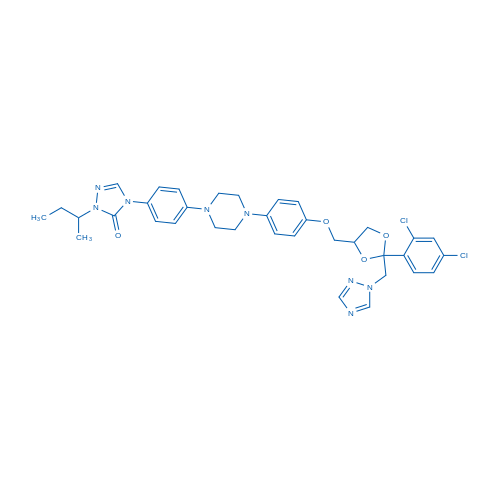
CAS No.: 84625-61-6
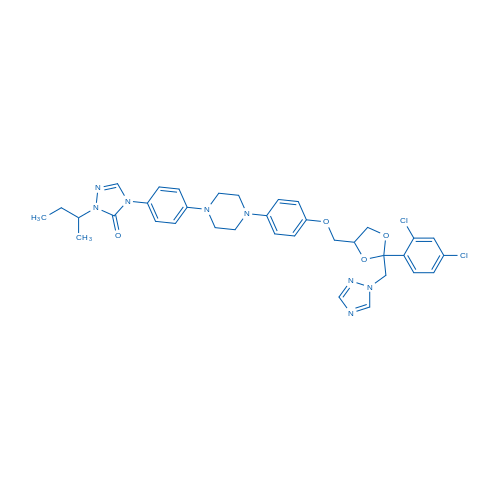
Itraconazole/伊曲康唑 Catalog No. CSN13003
Synonyms: R51211;Oriconazole;Orungal
Itraconazole is a relatively potent inhibitor of CYP3A4 with IC50 of 6.1 nM, used as a triazole antifungal agent.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 CYP3A4
IC50:6.1nM- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT00895453 - - Completed - - NCT01060462 - - Completed - - NCT02831972 Healthy Phase 1 Completed - United States, Arizona ... more >> Tempe, Arizona, United States, 85283 Collapse << - 更多
- 参考文献
- [1] Kim J, Tang JY, et al. Itraconazole, a commonly used antifungal that inhibits Hedgehog pathway activity and cancer growth. Cancer Cell. 2010 Apr 13;17(4):388-99.
- [2] Isoherranen N, Kunze KL, et al. Role of itraconazole metabolites in CYP3A4 inhibition. Drug Metab Dispos. 2004 Oct;32(10):1121-31.
- [3] Kim J, Tang JY, Gong R, Kim J, Lee JJ, Clemons KV, Chong CR, Chang KS, Fereshteh M, Gardner D, Reya T, Liu JO, Epstein EH, Stevens DA, Beachy PA. Itraconazole, a commonly used antifungal that inhibits Hedgehog pathway activity and cancer growth. Cancer Cell. 2010 Apr 13;17(4):388-99
- [4] Chong CR, Xu J, Lu J, Bhat S, Sullivan DJ Jr, Liu JO. Inhibition of angiogenesis by the antifungal drug itraconazole. ACS Chem Biol. 2007 Apr 24;2(4):263-70
- [5] Deng H, Huang L, Liao Z, Liu M, Li Q, Xu R. Itraconazole inhibits the Hedgehog signaling pathway thereby inducing autophagy-mediated apoptosis of colon cancer cells. Cell Death Dis. 2020 Jul 17;11(7):539
- [6] Caputo R. Itraconazole (Sporanox) in superficial and systemic fungal infections. Expert Rev Anti Infect Ther. 2003 Dec;1(4):531-42
- [7] Slain D, Rogers PD, Cleary JD, Chapman SW. Intravenous itraconazole. Ann Pharmacother. 2001 Jun;35(6):720-9
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 84625-61-6 | 储存条件 |
|
|||||||||||
| 分子式 | C35H38Cl2N8O4 | 运输 | 蓝冰 | |||||||||||
| 分子量 | 705.63 | 别名 | R51211;Oriconazole;Orungal;Sporanox;Itraconazolum;Itraconazol;Itrizole;伊曲康唑 | |||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| human hepatocytes | - | Function assay | - | Inhibition of CYP3A4 in human hepatocytes using testosterone as substrate by HPLC/MS/MS method, IC50=0.07 μM | 24948565 |
| human HTLA cells | - | Function assay | 20 mins | Inhibition of CX3CL1-stimulated CX3CR1 in human HTLA cells pre-incubated for 20 mins measured on day 4 by beta arrestin-recruitment mediated luciferase reporter gene assay, IC50=0.1 μM | 23437772 |
| human HUVEC cells | 2 μM | Function assay | 24 h | Inhibition of VEGFR2 glycosylation in human HUVEC cells at 2 uM after 24 hrs by Western blot analysis | 21936514 |
| human MRC5 cells | - | Cytotoxicity assay | - | Cytotoxicity against human MRC5 cells, CC50=49.33 μM | 20805398 |
| human PBMC | - | Cytotoxicity assay | 72 h | Cytotoxicity against human PBMC assessed as cell viability after 72 hrs by MTT assay, IC50=1.53 μM | 24485783 |
| LLC-PK1 epithelial cells | - | Function assay | - | Inhibition of P-glycoprotein, mouse L-mdr1a expressed in LLC-PK1 epithelial cells using calcein-AM polarisation assay, IC50=0.2 μM | 12699389 |
| Topp 3 cells | - | Function assay | - | Inhibition of human CYP51 expressed in Topp 3 cells by lanosterol demethylase assay, IC50=3.6 μM | 17194716 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT00895453 | - | - | Completed | - | - |
| NCT01060462 | - | - | Completed | - | - |
| NCT02831972 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States, 85283 Collapse << |
| NCT02653872 | Healthy Subjects | Phase 1 | Completed | - | United Kingdom ... more >> Research Site London, United Kingdom Collapse << |
| NCT01913379 | Healthy Subjects ... more >> Pharmacokinetics Drug-Drug Interaction Collapse << | Phase 1 | Completed | - | France ... more >> SGS Paris, France, 75015 Collapse << |
| NCT02342717 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1346.10.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02653872 | - | - | Completed | - | - |
| NCT02248259 | Healthy | Phase 1 | Completed | - | Korea, Republic of ... more >> 1289.23.8201 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of Collapse << |
| NCT01795937 | - | - | Completed | - | - |
| NCT02817100 | Clinical Trials, Phase I as To... more >>pic Collapse << | Phase 1 | Completed | - | Germany ... more >> CRS Clinical Research Services Berlin GmbH Berlin, Germany, 13353 Collapse << |
| NCT01282879 | Graft vs Host Disease | Phase 4 | Terminated(In interim analysis... more >>, this study met the primary hypothesis.) Collapse << | - | Korea, Republic of ... more >> Chonnam National University Hwasun Hospital Hwasun, Jeollanam-do, Korea, Republic of Soonchunhyang University Bucheon Hospital Bucheon, Kyounggi-do, Korea, Republic of Gachon University Gil Hospital Incheon, Korea, Republic of Inha University Hospital Incheon, Korea, Republic of Inje University Pusan Paik Hospital Pusan, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of, 135-710 Chung-ang University Hospital Seoul, Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of Soonchunhyang University Seoul Hospital Seoul, Korea, Republic of Collapse << |
| NCT02370615 | Japanese Healthy Adult Males | Phase 1 | Completed | - | Japan ... more >> Osaka-shi, Osaka, Japan Collapse << |
| NCT00336531 | Neuroblastoma ... more >> Brain Tumor Retinoblastoma Wilms Tumor Mycoses Collapse << | Phase 4 | Completed | - | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of Collapse << |
| NCT02370615 | - | - | Completed | - | - |
| NCT02044393 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1334.10.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01814709 | Healthy Volunteer | Phase 1 | Completed | - | United States, Texas ... more >> Dallas, Texas, United States, 75247 Collapse << |
| NCT01795937 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1220.61.1 Boehringer Ingelheim Investigational Site Ingelheim, Germany Collapse << |
| NCT03579719 | Healthy Volunteers | Phase 1 | Recruiting | November 10, 2018 | Netherlands ... more >> Pra International Group B.V Recruiting Groningen, Netherlands, 9728 NZ Collapse << |
| NCT02044393 | - | - | Completed | - | - |
| NCT03774576 | Healthy Volunteers | Phase 1 | Not yet recruiting | March 2, 2019 | France ... more >> BIOTRIAL Not yet recruiting Rennes, France, 35042 Collapse << |
| NCT02456129 | Clinical Trial, Phase I | Phase 1 | Completed | - | United Kingdom ... more >> Leeds, West Yorkshire, United Kingdom, LS2 9LH Collapse << |
| NCT02838264 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT03614455 | Pharmacokinetics | Early Phase 1 | Completed | - | United States, Texas ... more >> Covance Clinical Research Unit, Inc. Dallas, Texas, United States, 75247 Collapse << |
| NCT02807909 | Thrombosis | Phase 1 | Completed | - | - |
| NCT01900028 | Solid Tumours | Phase 1 | Completed | - | Belgium ... more >> Research Site Gent, Belgium, 9000 Research Site Gent, Belgium Research Site Leuven, Belgium, 3000 Research Site Leuven, Belgium Research Site Liege, Belgium, 4000 Research Site Liege, Belgium Denmark Research site Herlev, Denmark, 2730 Research Site København, Denmark, 2100 Netherlands Research Site Amsterdam, Netherlands, 1081 HV Research Site Maastricht, Netherlands, 6229 HX Research Site Nijmegen, Netherlands, 6525 GA United Kingdom Research Site London, United Kingdom Research Site Newcastle, United Kingdom, NE7 7DN Research Site Sutton, United Kingdom, SM2 5PT Collapse << |
| NCT00695071 | Healthy | Phase 1 | Completed | - | United States, New Jersey ... more >> Advanced Biomedical Research Hackensack, New Jersey, United States, 07601 Collapse << |
| NCT02130661 | Alzheimer's Disease | Phase 1 | Withdrawn | January 2018 | - |
| NCT02621905 | Neutropenia | Phase 4 | Completed | - | Australia, New South Wales ... more >> St Vincent's Hospital Darlinghurst, New South Wales, Australia, 2010 Collapse << |
| NCT02230033 | Healthy | Phase 1 | Completed | - | United States, Arizona ... more >> Tempe, Arizona, United States Collapse << |
| NCT03515382 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Quotient Sciences Limited Ruddington, United Kingdom Collapse << |
| NCT03135106 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Clinical Pharmacology Unit Merksem, Belgium, 2170 Collapse << |
| NCT02199431 | Healthy | Phase 1 | Completed | - | United Kingdom ... more >> Covance Leeds, United Kingdom Collapse << |
| NCT03403439 | Healthy | Phase 1 | Completed | - | Germany ... more >> Humanpharmakologisches Zentrum Biberach Biberach, Germany, 88397 Collapse << |
| NCT02729467 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Overland Park, Kansas, United States Collapse << |
| NCT02706535 | Drug Interactions | Phase 1 | Completed | - | United States, Maryland ... more >> GSK Investigational Site Baltimore, Maryland, United States, 21225 Collapse << |
| NCT02440841 | Nocturia | Phase 1 | Completed | - | Germany ... more >> PAREXEL Early Phase Clinical Unit Berlin Berlin, Germany, 14050 Collapse << |
| NCT03722173 | Healthy | Phase 1 | Recruiting | December 18, 2018 | Germany ... more >> Humanpharmakologisches Zentrum Biberach Recruiting Biberach, Germany, 88397 Contact: Thomas Gießmann +49 (7351) 5493829 thomas.giessmann@boehringer-ingelheim.com Collapse << |
| NCT00895453 | Vulvovaginal Candidiasis | Not Applicable | Completed | - | Austria ... more >> Vienna Medical School Vienna, Austria, 1090 Collapse << |
| NCT00791219 | Onychomycosis | Phase 2 | Completed | - | United States, Florida ... more >> Synergyst Research Altamonte Springs, Florida, United States, 32701 FXM Research Corp Miami, Florida, United States, 33175 United States, Idaho Northwest Clinical Trials Boise, Idaho, United States, 83704 United States, North Carolina PMG Research Salisbury, North Carolina, United States, 28144 United States, Oregon Oregon Medical Research Center, P.C Portland, Oregon, United States, 97223 United States, South Carolina Coastal Carolina Research Mt. Pleasant, South Carolina, United States, 29464 United States, Texas JS Studies College Station, Texas, United States, 77845 Endeavor Clinical Trials San Antonio, Texas, United States, 78229 Collapse << |
| NCT01344681 | Febrile Neutropenia ... more >> Hematological Diseases Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> Dong-A Medical Center Busan, Korea, Republic of Severance Hospital Seoul, Korea, Republic of, 120-752 Korea University Guro Hospital Seoul, Korea, Republic of, 152-703 ASAN Medical Center Seoul, Korea, Republic of Ewha Womans University Mokdong Hospital Seoul, Korea, Republic of Samsung Medical Center Seoul, Korea, Republic of Ajou University Medical Center Suwon, Korea, Republic of Collapse << |
| NCT02118909 | Healthy Volunteers ... more >> Non-smokers Collapse << | Phase 1 | Completed | - | United States, Arizona ... more >> Celerion Tempe, Arizona, United States, 67296 Collapse << |
| NCT01772563 | Neoplasms | Phase 1 | Active, not recruiting | April 15, 2019 | Hungary ... more >> PRA Hungary Ltd., Phase I. Clinical Pharmacology Unit Budapest, Hungary, 1077 National Institute of Oncology Budapest, Hungary, 1122 Collapse << |
| NCT02122692 | Healthy Volunteers | Phase 1 | Completed | - | France ... more >> FR801 Rennes, France, 35042 Collapse << |
| NCT00794703 | Fungemia Fung... more >>al Infections Collapse << | Phase 3 | Completed | - | China, Fujian ... more >> Fuzhou, Fujian, China China, Hubei Wuhan, Hubei, China China, Sichuan Chengdu, Sichuan, China China, Zhejiang Hangzhou, Zhejiang, China China Jiangsu, China Nanning, China Peking, China Shanghai, China Tianjin, China Xian, China Collapse << |
| NCT02824042 | Medical Oncology | Phase 1 | Active, not recruiting | June 28, 2019 | United States, California ... more >> UCLA-Santa Monica Medical Center Santa Monica, California, United States, 90404 United States, Michigan Henry Ford Health System Detroit, Michigan, United States, 48202 United States, Missouri Washington University School of Medicine Saint Louis, Missouri, United States, 63110 United States, Ohio University Hospitals Cleveland Medical Center Cleveland, Ohio, United States, 44106-2602 United States, Texas Mary Crowley Medical Research Center Dallas, Texas, United States, 75230 Australia, New South Wales Blacktown Cancer & Haematology Centre Blacktown, New South Wales, Australia, 2148 Australia, Victoria Epworth HealthCare Richmond, Victoria, Australia, 3122 Belgium CU Saint-Luc/UZ St-Luc Bruxelles - Brussel, Belgium, 1200 UZ Gent Gent, Belgium, 9000 France Hôpital Henri Mondor Creteil, France, 94010 Centre Georges Francois Leclerc Dijon Dijon, France, 21000 Hôpital de la Timone - Marseille Marseille, France, 13005 Netherlands Nederlands Kanker Instituut Amsterdam, Netherlands, 1066 CX Academisch Ziekenhuis Vrije Universiteit Amsterdam, Netherlands, 1081 HV Universitair Medisch Centrum St. Radboud Nijmegen, Netherlands, 6525 GA Spain Ciutat Sanitària i Universitaria de la Vall d'Hebron Barcelona, Spain, 08035 Fundacion Jimenez Diaz (Clinica de la Concepcion) Madrid, Spain, 28040 Hospital Virgen de la Victoria Málaga, Spain, 29010 Collapse << |
| NCT02883114 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02065739 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Merksem, Belgium Collapse << |
| NCT02608034 | Metastatic Melanoma, BRAF V600... more >> Mutation Positive Collapse << | Phase 1 | Active, not recruiting | January 8, 2019 | United States, Kansas ... more >> University of Kansas Cancer Center and Medical Pavilion Westwood, Kansas, United States, 66205 United States, Texas Mary Crowley Medical Research Center Dallas, Texas, United States, 75230 Israel Rambam Health Care Campus; Oncology Haifa, Israel, 3109601 Hadassah Ein Karem Hospital; Oncology Dept Jerusalem, Israel, 9112001 Tel Aviv Sourasky Medical Center Tel Aviv, Israel, 64239 Korea, Republic of Seoul National University Hospital Seoul, Korea, Republic of, 03080 Samsung Medical Center; Gastroenterology Seoul, Korea, Republic of, 135-710 Asan Medical Center; Division of Oncology Seoul, Korea, Republic of, 138-736 Severance Hospital - Yonsei Uni ; Obstetrics & Gynaecology Dept. Seoul, Korea, Republic of Russian Federation Republican Clinical Oncologic Dispensary of Republic Of Tatarstan Kazan, Russian Federation, 420029 FSBSI "N. N. Blokhin Russian Cancer Research Center" Moscow, Russian Federation, 115478 St. Petersburg Oncology Hospital St Petersburg, Russian Federation, 198255 Collapse << |
| NCT02401295 | Relapsed Multiple Myeloma | Phase 1 | Completed | - | United States, Iowa ... more >> University of Iowa Hospitals and Clinics Iowa City, Iowa, United States, 52242 Collapse << |
| NCT02272764 | Pharmacokinetics | Phase 1 | Completed | - | United States, Kansas ... more >> Alkermes Investigational Site Overland Park, Kansas, United States, 66212 Collapse << |
| NCT02755857 | Neutropenia | Phase 2 | Completed | - | Australia, New South Wales ... more >> St Vincent's Hospital Darlinghurst, New South Wales, Australia, 2010 Collapse << |
| NCT03121664 | Healthy | Phase 1 | Completed | - | Belgium ... more >> Pfizer Clinical Research Unit Brussels, Belgium, B-1070 Collapse << |
| NCT02513446 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1320.20.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT03209648 | Healthy Participants | Phase 1 | Completed | - | United Kingdom ... more >> Covance Clinical Research Unit Ltd. Leeds, United Kingdom, LS2 9LH Collapse << |
| NCT01160952 | Hematological Diseases ... more >> Allogeneic Stem Cell Transplantation Collapse << | Phase 2 | Unknown | March 2011 | China, Guangdong ... more >> Guangzhou General Hospital of Guangzhou Military Command Recruiting Guangzhou, Guangdong, China, 510010 Contact: Yonghua Li, MD 8613751880527 lyhood@163.com Contact: Xiaohui Zeng, Pharm D 8613560327666 gzlcyljd@163.com Principal Investigator: Yang Xiao, MD Collapse << |
| NCT02440009 | Allergic Bronchopulmonary Aspe... more >>rgillosis Collapse << | Phase 2 Phase 3 | Recruiting | December 2018 | India ... more >> Chest Clinic, PGIMER Recruiting Chandigarh, India, 160012 Contact: Ritesh Agarwal, MD, DM 0172-2756825 riteshpgi@gmail.com Contact: Ashutosh Aggarwal, MD, DM 0172-2756824 dr.anaggarwal@gmail.com Collapse << |
| NCT02895529 | Hematologic Neoplasms | Phase 4 | Terminated(Study is a commitme... more >>nt for license renew, we target on Apr19 submission, considering timeline, a cut off should be scheduled in Apr18.) Collapse << | - | China ... more >> Beijign, China Beijing, China Changchun, China Changzhou, China Fuzhou, China Guangzhou, China Guiyang, China Hefei, China Shanghai, China Suzhou, China Tianjin, China Zhengzhou, China Collapse << |
| NCT03103646 | Cytochrome P450 Interaction | Phase 1 | Recruiting | April 2019 | United States, Texas ... more >> Covance - Dallas Recruiting Dallas, Texas, United States, 75247 Contact: Email contact via H. Lundbeck A/S United Kingdom Covance Clinical Research Unit Ltd Recruiting Leeds, United Kingdom Contact: Email contact via H. Lundbeck A/S Collapse << |
| NCT01752023 | Non-small Cell Lung Cancer Met... more >>astatic Collapse << | Phase 2 | Terminated(Low Accrual) | - | United States, Maryland ... more >> Bayview Medical Center at Johns Hopkins Baltimore, Maryland, United States, 21224 Johns Hopkins University, SKCCC Baltimore, Maryland, United States, 21287 Collapse << |
| NCT01752023 | - | - | Terminated(Low Accrual) | - | - |
| NCT02402595 | Healthy | Phase 1 | Completed | - | United States, Kansas ... more >> Vince and Associates Clinical Research, Inc. Overland Park, Kansas, United States, 66212 Collapse << |
| NCT01259336 | Chronic Cavitary Pulmonary Asp... more >>ergillosis Collapse << | Phase 4 | Completed | - | India ... more >> PGIMER Chandigarh, Punjab, India, 160012 Collapse << |
| NCT00887458 | Prostate Cancer | Phase 2 | Completed | - | United States, Maryland ... more >> Johns Hopkins Hospital Baltimore, Maryland, United States, 21231 United States, Michigan University of Michigan Comprehensive Cancer Center Ann Arbor, Michigan, United States, 48109 Karmanos Cancer Center Detroit, Michigan, United States, 48201 United States, New York Memorial Sloan-Kettering Cancer Center New York, New York, United States, 10021 Collapse << |
| NCT03301181 | Healthy Volunteers: Asian, Non... more >>-Asian Collapse << | Phase 1 | Completed | - | United States, California ... more >> West Coast Clinical Trials (WCCT) Cypress, California, United States, 90630 Collapse << |
| NCT02699723 | Skin Basal Cell Carcinoma | Not Applicable | Not yet recruiting | November 2019 | United States, California ... more >> Stanford Cancer Institute Not yet recruiting Palo Alto, California, United States, 94304 Contact: Irene Bailey-Healy 408-892-7261 baileyhi@stanford.edu Principal Investigator: Jean Tang Collapse << |
| NCT02790671 | Pharmacokinetics | Phase 1 | Completed | - | United States, Texas ... more >> Worldwide Clinical Trials San Antonio, Texas, United States, 78217 Collapse << |
| NCT02054793 | Prostate Cancer ... more >> Castration-resistant Prostate Cancer Collapse << | Phase 1 Phase 2 | Withdrawn(This study was never... more >> able to open due to lack of funding from the pharmaceutical company.) Collapse << | July 2016 | United States, District of Col... more >>umbia Sibley Memorial Hospital Washington, District of Columbia, United States, 20016 United States, Maryland Johns Hopkins University Baltimore, Maryland, United States, 21231 United States, Michigan Karmanos Cancer Institute Detroit, Michigan, United States Collapse << |
| NCT01450683 | - | - | Terminated(Low accrual) | - | - |
| NCT01198236 | Invasive Fungal Infection | Phase 4 | Completed | - | China, Zhejiang ... more >> The First Hospital of Zhejiang Medical Colleage Zhejiang University Hangzhou, Zhejiang, China, 310003 Collapse << |
| NCT03513211 | Prostate Cancer | Phase 1 Phase 2 | Recruiting | March 30, 2021 | Australia, New South Wales ... more >> St Vincent's Hospital Recruiting Darlinghurst, New South Wales, Australia, 2010 Contact: Robert Kent 61 2 9355 5611 svhs.cancerresearch@svha.org.au Collapse << |
| NCT00887458 | - | - | Completed | - | - |
| NCT03161405 | Healthy Participants | Phase 1 | Completed | - | United States, Kansas ... more >> Takeda Investigational Site Lenexa, Kansas, United States, 66219 Collapse << |
| NCT01450683 | Prostate Cancer ... more >> Prostatic Neoplasms Castrate-resistant Prostate Cancer (CRPC) Androgen-insensitive Prostate Cancer Hormone-refractory Prostate Cancer Metastatic Disease Collapse << | Phase 2 | Terminated(Low accrual) | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT02131298 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer Investigational Site New Haven, Connecticut, United States, 06511 Collapse << |
| NCT02354261 | Basal Cell Carcinoma in Basal ... more >>Cell Nevus Syndrome Collapse << | Phase 2 | Active, not recruiting | June 2019 | United States, California ... more >> Dermatology Specialists, Inc Oceanside, California, United States, 92056 United States, Florida Ameriderm Research Ormond Beach, Florida, United States, 32174 United States, Michigan Grekin Skin Institute Warren, Michigan, United States, 48034 United States, New York Stony Brook University East Setauket, New York, United States, 11733 United States, Pennsylvania Penn State Hershey Medical Center Hershey, Pennsylvania, United States, 17033 Collapse << |
| NCT01967979 | Healthy Volunteer | Phase 1 | Completed | - | Netherlands ... more >> Zuidlaren, Netherlands, 9471 GP Collapse << |
| NCT03173170 | Healthy Participants | Phase 1 | Completed | - | United States, Kansas ... more >> Pharmaceutical Research Associates, Inc. Lenexa, Kansas, United States, 66219 Collapse << |
| NCT02836171 | Neoplasms | Phase 1 | Completed | - | China, Shanghai ... more >> Shanghai Xuhui Central Hospital Shanghai, Shanghai, China, 200030 Collapse << |
| NCT02735356 | Skin Basal Cell Carcinoma | Early Phase 1 | Active, not recruiting | January 31, 2019 | United States, California ... more >> Stanford Cancer Institute Palo Alto, California, United States, 94304 Collapse << |
| NCT00356915 | - | - | Completed | - | - |
| NCT02341482 | Healthy | Phase 1 | Completed | - | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 Collapse << |
| NCT00356915 | Onychomycosis | Phase 3 | Completed | - | - |
| NCT01787331 | Prostate Adenocarcinoma ... more >> Recurrent Prostate Carcinoma Stage I Prostate Adenocarcinoma AJCC v7 Stage II Prostate Adenocarcinoma AJCC v7 Stage III Prostate Adenocarcinoma AJCC v7 Collapse << | Phase 2 | Completed | - | United States, California ... more >> University of California, San Francisco San Francisco, California, United States, 94115 Collapse << |
| NCT01787331 | - | - | Completed | - | - |
| NCT02341482 | - | - | Completed | - | - |
| NCT00871728 | Onychomycosis, Toe | Phase 4 | Completed | - | - |
| NCT01823289 | - | - | Completed | - | - |
| NCT00769600 | Recurrent Non Small Cell Lung ... more >>Cancer Collapse << | Phase 2 | Terminated(Low accrual.) | - | United States, Maryland ... more >> Johns Hopkins Sidney Kimmel Comprehensive Cancer Center Baltimore, Maryland, United States, 21231 Singapore Singapore General Hospital Jalan Tan Tock Seng, Singapore, 308433 Collapse << |
| NCT02175407 | Pharmacokinetics ... more >> DDI (Drug-Drug Interaction) Healthy Subjects Collapse << | Phase 1 | Completed | - | Germany ... more >> PAREXEL International GmbH Berlin, Germany, 14050 Collapse << |
| NCT03479411 | Asthma | Phase 1 | Completed | - | United Kingdom ... more >> Medicines Evaluation Unit Manchester, United Kingdom Quotient Sciences Nottingham, United Kingdom Collapse << |
| NCT01823289 | Pulmonary Fungal Infection | Phase 4 | Completed | - | - |
| NCT01929876 | - | - | Completed | - | - |
| NCT01929876 | Healthy Participants | Phase 1 | Completed | - | United States, Texas ... more >> Dallas, Texas, United States, 75247 Collapse << |
| NCT00871728 | - | - | Completed | - | - |
| NCT02475746 | Healthy | Phase 1 | Withdrawn | August 2015 | United States, Connecticut ... more >> Pfizer New Haven Clinical Research Unit New Haven, Connecticut, United States, 06511 CRU New Haven New Haven, Connecticut, United States Collapse << |
| NCT02455856 | Healthy | Phase 1 | Completed | - | Germany ... more >> Berlin, Germany Collapse << |
| NCT03656081 | Chronic Pulmonary Aspergillosi... more >>s Collapse << | Phase 3 | Not yet recruiting | May 2023 | - |
| NCT00798135 | Breast Neoplasms ... more >> Neoplasm Metastasis Collapse << | Not Applicable | Completed | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT03081702 | Platinum-resistant Epithelial ... more >>Ovarian Cancer Collapse << | Phase 1 Phase 2 | Recruiting | October 1, 2022 | Canada, Ontario ... more >> Princess Margaret Cancer Centre Recruiting Toronto, Ontario, Canada, M5G 2M9 Contact: Stephanie Lheureux, M.D. 416-946-2818 Collapse << |
| NCT00798135 | - | - | Completed | - | - |
| NCT01108094 | Basal Cell Carcinoma (BCC) ... more >> Skin Cancer Collapse << | Phase 2 | Completed | - | United States, California ... more >> Stanford University School of Medicine Stanford, California, United States, 94305 Collapse << |
| NCT01021683 | - | - | Completed | - | - |
| NCT01108094 | - | - | Completed | - | - |
| NCT00588016 | Sinusitis | Phase 1 | Completed | - | United States, Minnesota ... more >> Mayo Clinic Rochester, Minnesota, United States, 55905 Collapse << |
| NCT01021683 | - | - | Completed | - | - |
| NCT03086226 | Mycetoma | Phase 2 | Recruiting | March 31, 2020 | Sudan ... more >> Mycetoma Research Centre Recruiting Khartoum, Sudan Contact: Ahmed H Fahal +249912346703 ahfahal@hotmail.com Contact: Rowa Alrahman +249914110128 roaalbasha@hotmail.com Collapse << |
| NCT01409018 | Pediatric, Cancer | Phase 1 | Completed | - | Korea, Republic of ... more >> Seoul National University Hospital Seoul, Chongno-gu, Korea, Republic of Collapse << |
| NCT02120677 | Basal Cell Carcinoma | Early Phase 1 | Recruiting | December 2018 | United States, Maryland ... more >> Johns Hopkins School of Medicine, Department of Dermatology Recruiting Baltimore, Maryland, United States, 21287 Contact: Nikki Tang, MD 410-502-7546 ctrep@jhmi.edu Contact: Bao Chau Ly, MS 410-502-7547 Baochau.Ly@jhmi.edu Collapse << |
| NCT02749513 | Esophageal Cancer | Early Phase 1 | Recruiting | - | United States, Texas ... more >> Dallas VA Medical Center Recruiting Dallas, Texas, United States, 75216 Contact: Sanjai Sabu 214-857-4237 sanjai.sabu@va.gov Collapse << |
| NCT03664115 | Lung Cancer | Phase 2 | Recruiting | December 2, 2020 | Egypt ... more >> oncology department Ain shams university Recruiting Cairo, Egypt, 11591 Contact: Amr sh Tawfik, MD Contact: Asmaa WH Mohamed, Master 01003538597 drasmaa_wahid@hotmail.com Collapse << |
| NCT03264079 | Healthy Volunteers | Phase 1 | Completed | - | United States, Ohio ... more >> Medpace Clinical Pharmacology Unit Cincinnati, Ohio, United States, 45227 Collapse << |
| NCT01706562 | - | - | Completed | - | - |
| NCT03498170 | Interaction | Phase 1 | Completed | - | United States, Florida ... more >> inVentiv Health Clinical Research Services LLC Miami, Florida, United States, 33136 Collapse << |
| NCT03398421 | Pulmonary Disease, Chronic Obs... more >>tructive Collapse << | Phase 1 | Completed | - | United States, Kansas ... more >> GSK Investigational Site Overland Park, Kansas, United States, 66211 Collapse << |
| NCT02259010 | Advanced Solid Tumors ... more >> Relapsed/Refractory Lymphoma Collapse << | Phase 1 | Unknown | April 2016 | United States, Missouri ... more >> St. Louis, Missouri, United States United States, Oklahoma Oklahoma City, Oklahoma, United States United States, Tennessee Germantown, Tennessee, United States United States, Texas Dallas, Texas, United States Collapse << |
| NCT02357836 | Non-small Cell Lung Cancer | Early Phase 1 | Recruiting | June 2019 | United States, Texas ... more >> UT Southwestern Medical Center Recruiting Dallas, Texas, United States, 75390 Contact: Jessica Saltarski, B.S., CCRP 214-648-7097 Jessica.Saltarski@utsouthwestern.edu Contact: David E Gerber, M.D. 214-648-4180 David.Gerber@UTSouthwestern.edu Collapse << |
| NCT03572049 | Fungal Infection | Phase 2 Phase 3 | Recruiting | December 31, 2019 | United States, Alabama ... more >> University of Alabama at Birmingham Recruiting Birmingham, Alabama, United States, 35233 Contact: Alisa Peinhardt, MAIS apeinhardt@uabmc.edu Contact: Rachel McMullen, BS rlmcmullen@uabmc.edu Principal Investigator: Peter G Pappas, MD Collapse << |
| NCT01936870 | - | - | Completed | - | - |
| NCT01936870 | - | - | Completed | - | - |
| NCT02329808 | - | - | Recruiting | December 2018 | Netherlands ... more >> Radboud University Medical Center Recruiting Nijmegen, Gelderland, Netherlands Contact: Roger Brüggemann, PharmD PhD roger.bruggemann@radboudumc.nl Contact: Lisa Martial, PharmD lisa.martial@radboudumc.nl Sub-Investigator: Maroeska Te Loo Sub-Investigator: Michiel Schreuder Sub-Investigator: Stefanie Henriet Collapse << |
| NCT02366884 | Neoplasms | Phase 2 | Enrolling by invitation | December 31, 2020 | United States, Maryland ... more >> Atavistic Chemotherapy Clinical Trial Bethesda, Maryland, United States, 20814 Mexico Dr. Frank Arguello Cancer Clinic San Jose del Cabo, Baja California Sur, Mexico Instituto de Ciencia y Medicina Genomica Torreon, Coahuila, Mexico Collapse << |
| NCT00673309 | Burns | Phase 2 Phase 3 | Completed | - | United States, Texas ... more >> University of Texas Medical Branch Galveston, Texas, United States, 77550 Collapse << |
| NCT01686607 | - | - | Active, not recruiting | January 2020 | United States, Maryland ... more >> Site US4 Baltimore, Maryland, United States, 21205 United States, Massachusetts Site US2 Boston, Massachusetts, United States, 02119 United States, Michigan Site US6 Ann Arbor, Michigan, United States, 48109 United States, North Carolina Site US3 Durham, North Carolina, United States, 27710 United States, Pennsylvania Site US5 Philadelphia, Pennsylvania, United States, 19104 Site US1 Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT00675714 | Burns | Phase 2 Phase 3 | Recruiting | September 2018 | United States, Texas ... more >> University of Texas Medical Branch Recruiting Galveston, Texas, United States, 77551 Contact: David N Herndon, MD 409-770-6731 dherndon@utmb.edu Contact: Cathy L Reed, BSN 409-770-6987 ca2reed@utmb.edu Principal Investigator: David N Herndon, MD Sub-Investigator: Oscar E Suman, PhD Sub-Investigator: Celeste FInnerty, PhD Collapse << |
| NCT01951430 | - | - | Recruiting | December 2018 | Italy ... more >> S.O.C. di Ematologia - Azienda Ospedaliera - SS. Antonio e Biagio e Cesare Arrigo Recruiting Alessandria, Italy Contact: Daniela Pietrasanta Principal Investigator: Daniela Pietrasanta, Dr. Sub-Investigator: Lorella De Paoli, Dr. Azienda Ospedaliera - Nuovo Ospedale 'Torrette' Recruiting Ancona, Italy Contact: Antonella Poloni Principal Investigator: Antonella Poloni, Dr. Sub-Investigator: Benedetta Costantini, Dr. UO Ematologia con trapianto-Università degli Studi di Bari Aldo Moro Recruiting Bari, Italy Contact: Giorgina Specchia Principal Investigator: Giorgina Specchia Divisione di Ematologia Ospedale A. Perrin Recruiting Brindisi, Italy Contact: Angela Melpignano Principal Investigator: Angela Melpignano Sub-Investigator: Gianluca Guaragna ASL N.8 - Ospedale "A. Businco" - Struttura Complessa di Ematologia e CTMO Recruiting Cagliari, Italy Contact: Anna Angela Di Tucci Principal Investigator: Anna Angela Di Tucci, Dr. Sub-Investigator: Graziella Pilo, Dr. Policlinico di Careggi Recruiting Firenze, Italy Contact: Valeria Santini Principal Investigator: Valeria Santini Sub-Investigator: Alessandro Sanna IRCCS_AOU San Martino-IST-Ematologia 1-Monoblocco 11°piano- lato ponente Recruiting Genova, Italy Contact: Marco Gobbi Principal Investigator: Marco Gobbi Sub-Investigator: Maurizio Miglino ASL Le/1 P.O. Vito Fazzi - U.O. di Ematologia ed UTIE Recruiting Lecce, Italy Contact: Nicola Di Renzo Principal Investigator: Nicola Di Renzo Sub-Investigator: Valentina Vezzoli Azienda Ospedaliera Universitaria - Università degli Studi di Napoli "Federico II" - Facoltà di Medicina e Chirurgia Recruiting Napoli, Italy Contact: Fabrizio Pane Principal Investigator: Fabrizio Pane Sub-Investigator: Giuseppe Cerciello S.C.D.U. Ematologia - DIMECS e Dipartimento Oncologico - Università del Piemonte Orientale Amedeo Avogadro Recruiting Novara, Italy Contact: Gianluca Gaidano Principal Investigator: Gianluca Gaidano Sub-Investigator: Monia Lunghi Università degli Studi di Padova - Ematologia ed Immunologia Clinica Recruiting Padova, Italy Contact: Renato Zambello Sub-Investigator: Gianni Binotto Principal Investigator: Renato Zambello U.O. di Ematologia con trapianto - Centro di Riferimento Regionale per le coagulopatie rare nel bambino e nell'adulto Dipart. Biomedico di Medicina Interna - A.U. Policlinico "Paolo Giaccone" Recruiting Palermo, Italy Contact: Maria Enza Mitra Principal Investigator: Maria Enza Mitra Sub-Investigator: Cinzia Maugeri Cattedra di Ematologia CTMO Università degli Studi di Parma Recruiting Parma, Italy Contact: Monica Crugnola Principal Investigator: Monica Crugnola Med. Int. ed Oncologia Medica IRCCS Policlinico S. Matteo Recruiting Pavia, Italy Contact: Rosangela Invernizzi Principal Investigator: Rosangela Invernizzi, Dr. Sub-Investigator: Federica Quaglia, Dr. Div. di Ematologia di Muraglia - CTMO Ospedale San Salvatore Recruiting Pesaro, Italy Contact: Giuseppe Visani Sub-Investigator: Sara Barulli Principal Investigator: Giuseppe Visani U.O. Ematologia Clinica - Azienda USL di Pescara Recruiting Pescara, Italy Contact: Paolo Di Bartolomeo Principal Investigator: Paolo Di Bartolomeo Unità Operativa Ematologia e Centro Trapianti - Dipartimento di Oncologia ed Ematologia - AUSL Ospedale di Piacenza Recruiting Piacenza, Italy Contact: Costanza Bosi Principal Investigator: Costanza Bosi, Dr. U.O. di Ematologia - Centro Oncologico Basilicata Recruiting Rionero in Vulture, Italy Contact: Pellegrino Musto Principal Investigator: Pellegrino Musto, Dr. Sub-Investigator: Oreste Villani, Dr. Complesso Ospedaliero S. Giovanni Addolorata Recruiting Roma, Italy Contact: Susanna Fennu Principal Investigator: Susanna Fennu Sub-Investigator: Stefania Cortese U.O.C. Ematologia - Ospedale S.Eugenio Recruiting Roma, Italy Contact: Pasquale Niscola Principal Investigator: Pasquale Niscola Sub-Investigator: Laura Scaramucci Università Cattolica del Sacro Cuore - Policlinico A. Gemelli Recruiting Roma, Italy Contact: Livio Pagano, Dr. Principal Investigator: Livio Pagano, Dr. Sub-Investigator: Maria Teresa Voso, Dr. Università degli Studi - Policlinico di Tor Vergata Not yet recruiting Roma, Italy Contact: Adriano Venditti Principal Investigator: Adriano Venditti, dr. Sub-Investigator: Luca Maurillo, Dr. Istituto di Ematologia - IRCCS Ospedale Casa Sollievo della Sofferenza Recruiting S. G. Rotondo, Italy Contact: Nicola Cascavilla Principal Investigator: Nicola Cascavilla Sub-Investigator: Grazia Sanpaolo, Dr. Dipartimento di Oncologia ed Ematologia S.C. Ematologia 2 A.O. Città della Salute e della Scienza di Torino San Giovanni Battista Recruiting Torino, Italy Contact: Bernardino Allione Sub-Investigator: Stefano D'Ardia Principal Investigator: Bernardino Allione Clinica Ematologica-Centro Trapianti e Terapie cellulari Azienda Ospedaliero-Universitaria, Udine Recruiting Udine, Italy Contact: Anna Candoni Principal Investigator: Anna Candoni Sub-Investigator: Erica Simeone Collapse << |
| NCT02770378 | Glioblastoma | Phase 1 | Active, not recruiting | March 2020 | Germany ... more >> University of Ulm School of Medicine Ulm, Baden-Württemberg, Germany, 89081 Collapse << |
| 靶点 | Description | IC50 |
|---|---|---|
| CYP3A4 | IC50:6.1nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
