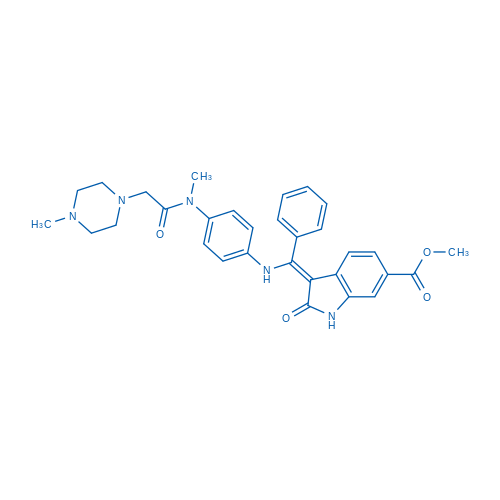
CAS No.: 656247-17-5
尼达尼布 Catalog No. CSN12341
Synonyms: BIBF 1120;Intedanib;尼达尼布
Nintedanib is a potent triple angiokinase inhibitor for VEGFR1/2/3, FGFR1/2/3 and PDGFRα/β with IC50 of 34 nM/13 nM/13 nM, 69 nM/37 nM/108 nM and 59 nM/65 nM.
纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 VEGFR3
IC50:13nMVEGFR2
IC50:13nMVEGFR1
IC50:34nM- 描述
- 作用机制
- 细胞研究
- Cell Data
- 更多
动物研究 剂量 Mice: 50 mg/kg[2] (p.o.), 3 mg/kg - 100 mg/kg[3] (p.o.); 5 mg/kg[4] (i.p.)
Rat: 25 mg/kg - 100 mg/kg[1] (p.o.)给药途径 p.o., i.p.
动物药代数据 Animal Mice[5] Rats[5] Monkeys[5] Dose 50 mg/kg 50 mg/kg (p.o.)
2 mg/kg (i.v.)40 mg/kg (p.o., cynomolgus monkey)
5 mg/kg (i.v., cynomolgus monkey)Administration p.o. p.o.
i.v.p.o.
i.v.MRT 5.19 h 3.25 h (i.v.) 3.82 h (i.v.) F 11.9% (p.o.) 13.2% (p.o.) T1/2 5.15 h 3.95 h (i.v.) 5.95 h (i.v.) AUC 2720 ng·h/ml 375 ng·h/ml (p.o.)
181 ng·h/ml (i.v.)2390 ng·h/ml (p.o.)
2260 ng·h/ml (i.v.)CL 202 ml/min/kg (i.v.) 37.5 ml/min/kg (i.v.) Cmax 547 ng/ml 105 ng/ml (p.o.)
124 ng/ml (i.v.)175 ng/ml (p.o.)
1300 ng/ml (i.v.)Protein bound (%) 92.9% (p.o.) Protein binding (free drug %) 0.972 98.5% (p.o.) Vss 41.2 L/kg (i.v.) 8.64 L/kg (i.v.) - 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT03361319 NSCLC, Recurrent ... more >> Adenocarcinoma of Lung Collapse << Phase 1 Phase 2 Not yet recruiting January 2022 - NCT02863055 Malignant Pleural Mesothelioma Phase 2 Recruiting October 2020 Belgium ... more >> UZ Antwerpen Recruiting Antwerpen, Belgium Contact: Jan Van Meerbeeck, prof UZ Gent Recruiting Gent, Belgium Contact: Veerle Surmont, Prof Italy Ospedale San Paolo Recruiting Milan, Italy Contact: Andrea Luciani, Dr United Kingdom Manchester University NHS Foundation Trust - UHSM-Wythenshawe Hospital Recruiting Wythenshawe, Manchester, United Kingdom, M23 9LT Principal Investigator: Paul Taylor Royal Marsden Hospital Recruiting Chelsea, United Kingdom Contact: Sanjay Popat, Dr Royal Marsden Hospital - Kingston Recruiting Kingston, United Kingdom Contact: Sanjay Popat, Dr Sheffield Teaching Hospitals NHS Foundation Trust - Weston Park Hospital Recruiting Sheffield, United Kingdom Contact: Robin Young, Dr NHS South Tyneside-South Tyneside District Hospital Recruiting South Shields, United Kingdom Contact: Rhona McMenemin, Dr Royal Marsden Hospital Recruiting Sutton, United Kingdom Contact: Sanjay Popat, Dr Collapse << NCT02597933 Scleroderma, Systemic Phase 3 Completed - - - 更多
- 参考文献
- [1] Hilberg F, Roth GJ, et al. BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. 2008 Jun 15;68(12):4774-82.
- [2] Roth GJ, Heckel A, et al. Design, synthesis, and evaluation of indolinones as triple angiokinase inhibitors and the discovery of a highly specific 6-methoxycarbonyl-substituted indolinone (BIBF 1120). J Med Chem. 2009 Jul 23;52(14):4466-80.
- [3] Wollin L, Maillet I, et al. Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosis. J Pharmacol Exp Ther. 2014 May;349(2):209-20.
- [4] Öztürk Akcora B, Storm G, et al. Tyrosine kinase inhibitor BIBF1120 ameliorates inflammation, angiogenesis and fibrosis in CCl4-induced liver fibrogenesis mouse model. Sci Rep. 2017 Mar 14;7:44545.
- [5] Nintedanib
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 656247-17-5 | 储存条件 |
|
|||||||||||||
| 分子式 | C31H33N5O4 | 运输 | 蓝冰 | |||||||||||||
| 分子量 | 539.62 | 别名 | BIBF 1120;Intedanib;尼达尼布 | |||||||||||||
| 溶解度 |
|
动物实验配方 |
|
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| A549 | 2/5 μM | Function Assay | 24 h | has a general EMT reversal effect | 26061747 |
| A549 | 0.01–5 μM | Function Assay | 24 h | induces SFTPD mRNA expression dose dependently | 25843005 |
| A549 | 0.01–5 μM | Function Assay | 72 h | enhances SP-D protein expression in a dose-dependent manner at concentrations of up to 5 μM | 25843005 |
| A549 | 5 μM | Function Assay | 0-1 h | increases AP-1 activation after 30 min | 25843005 |
| H1703 | - | Growth Inhibition Assay | - | IC50=0.05 μM | 23729403 |
| Hep3B | 0-20 μM | Cell Viability Assay | 48 h | decreases cell viability dose dependently | 24657398 |
| Hep3B | 0-20 μM | Apoptosis Assay | 48 h | induces cell apoptosis dose dependently | 24657398 |
| HepG2 | 0-20 μM | Cell Viability Assay | 48 h | decreases cell viability dose dependently | 24657398 |
| HepG2 | 0-20 μM | Apoptosis Assay | 48 h | induces cell apoptosis dose dependently | 24657398 |
| HuH7 | 0-20 μM | Cell Viability Assay | 48 h | decreases cell viability dose dependently | 24657398 |
| HuH7 | 0-20 μM | Apoptosis Assay | 48 h | induces cell apoptosis dose dependently | 24657398 |
| Mia-Paca2 | 2/5 μM | Function Assay | 24 h | has a general EMT reversal effect | 26061747 |
| PLC5 | 0-20 μM | Cell Viability Assay | 48 h | decreases cell viability dose dependently | 24657398 |
| PLC5 | 0-20 μM | Apoptosis Assay | 48 h | induces cell apoptosis dose dependently | 24657398 |
| SK-Hep1 | 0-20 μM | Cell Viability Assay | 48 h | decreases cell viability dose dependently | 24657398 |
| SK-Hep1 | 0-20 μM | Apoptosis Assay | 48 h | induces cell apoptosis dose dependently | 24657398 |
| SKOV3 | 5 µM | Function Assay | 24 h | induces a significant increase in the promoter activities of E-cad, CDH1, and CDH3 | 26061747 |
| T24 | 2/5 μM | Function Assay | 24 h | has a general EMT reversal effect | 26061747 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT03361319 | NSCLC, Recurrent ... more >> Adenocarcinoma of Lung Collapse << | Phase 1 Phase 2 | Not yet recruiting | January 2022 | - |
| NCT02863055 | Malignant Pleural Mesothelioma | Phase 2 | Recruiting | October 2020 | Belgium ... more >> UZ Antwerpen Recruiting Antwerpen, Belgium Contact: Jan Van Meerbeeck, prof UZ Gent Recruiting Gent, Belgium Contact: Veerle Surmont, Prof Italy Ospedale San Paolo Recruiting Milan, Italy Contact: Andrea Luciani, Dr United Kingdom Manchester University NHS Foundation Trust - UHSM-Wythenshawe Hospital Recruiting Wythenshawe, Manchester, United Kingdom, M23 9LT Principal Investigator: Paul Taylor Royal Marsden Hospital Recruiting Chelsea, United Kingdom Contact: Sanjay Popat, Dr Royal Marsden Hospital - Kingston Recruiting Kingston, United Kingdom Contact: Sanjay Popat, Dr Sheffield Teaching Hospitals NHS Foundation Trust - Weston Park Hospital Recruiting Sheffield, United Kingdom Contact: Robin Young, Dr NHS South Tyneside-South Tyneside District Hospital Recruiting South Shields, United Kingdom Contact: Rhona McMenemin, Dr Royal Marsden Hospital Recruiting Sutton, United Kingdom Contact: Sanjay Popat, Dr Collapse << |
| NCT02597933 | Scleroderma, Systemic | Phase 3 | Completed | - | - |
| NCT01770392 | - | - | Completed | - | - |
| NCT02607722 | - | - | Active, not recruiting | January 31, 2020 | Japan ... more >> Multiple Locations, Japan Collapse << |
| NCT01770392 | Healthy | Phase 1 | Completed | - | Germany ... more >> 1199.162.1 Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02856425 | Patients With Any Advanced Sol... more >>id Tumors Collapse << | Phase 1 | Suspended(Safety event) | July 2026 | France ... more >> Gustave Roussy Villejuif, Val De Marne, France, 94805 Collapse << |
| NCT03710824 | - | - | Not yet recruiting | January 13, 2021 | - |
| NCT02452463 | Radiation-Induced Pneumonitis ... more >> Stage IIA Non-Small Cell Lung Carcinoma Stage IIB Non-Small Cell Lung Carcinoma Stage IIIA Non-Small Cell Lung Cancer Stage IIIB Non-Small Cell Lung Cancer Stage IV Non-Small Cell Lung Cancer Collapse << | Phase 2 | Recruiting | January 7, 2020 | United States, New York ... more >> Roswell Park Cancer Institute Recruiting Buffalo, New York, United States, 14263 Contact: Grace Dy 716-845-3099 Grace.Dy@RoswellPark.org Principal Investigator: Grace Dy United States, West Virginia West Virginia University Recruiting Morgantown, West Virginia, United States, 26506 Contact: Carloa Ross 304-581-1158 cjross@hsc.wvu.edu Principal Investigator: Malcom Mattes, MD Collapse << |
| NCT03287947 | Appendix Cancer | Phase 2 | Recruiting | March 1, 2022 | United States, North Carolina ... more >> Levine Cancer Institute Recruiting Charlotte, North Carolina, United States, 28204 Contact: Nathan Thompson, RN 980-487-3241 Nathan.Thompson@carolinashealthcare.org Collapse << |
| NCT02399215 | Carcinoid Tumor ... more >> Metastatic Carcinoid Tumor Neuroendocrine Neoplasm Collapse << | Phase 2 | Active, not recruiting | August 1, 2019 | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 United States, Ohio Ohio State University Comprehensive Cancer Center Columbus, Ohio, United States, 43210 Collapse << |
| NCT03513484 | Acute Myeloid Leukemia With t(... more >>9;11)(p22.3;q23.3); MLLT3-KMT2A Fibroblast Growth Factor Basic Form Measurement FLT3 Internal Tandem Duplication Recurrent Adult Acute Myeloid Leukemia Refractory Acute Myeloid Leukemia Collapse << | Phase 1 | Recruiting | January 2021 | United States, Illinois ... more >> Northwestern University Recruiting Chicago, Illinois, United States, 60611 Contact: Jessica K. Altman 312-695-6180 j-altman@northwestern.edu Principal Investigator: Jessica K. Altman Collapse << |
| NCT02730416 | Endometrial Cancer | Phase 2 | Recruiting | July 2022 | Denmark ... more >> NSGO Recruiting Copenhagen, Sjaelland, Denmark, 2100 Contact: Mette Berensen, RN mette.berensen.02@regionh.dk Collapse << |
| NCT02531737 | Lung Neoplasms | Phase 2 | Recruiting | January 2019 | France ... more >> CH de Beauvais Recruiting Beauvais, France Contact: Jacky Crequet, MD 33.3 44 11 22 23 Jacky.crequit@wanadoo.fr Sub-Investigator: Jacky crequet, MD Ch de Beauvais Recruiting Beauvais, France Principal Investigator: Jacky CREQUIT, MD CHU Brest Recruiting Brest, France Contact: G. ROBINET, MD Principal Investigator: G ROBINET, MD Service de Pneumologie Recruiting Créteil, France Contact: Isabelle Monnet, MD 33.1 45 17 50 14 Isabelle.Monnet@chicreteil.fr Principal Investigator: Isabelle Monnet, MD Service de Pneumologie Recruiting Gap, France Contact: P Thomas, MD 33.4 92 40 61 26 pneumologie@ch-gap.fr Principal Investigator: P Thomas, MD CHU de Limoges Recruiting Limoges, France, 87000 Contact: A. Vergnenegre, MD alain.vergnenegre@unilim.fr Principal Investigator: B. Melloni, MD Principal Investigator: A. Vergnenegre, MD CH de Bretagne Sud Recruiting LOrient, France Contact: Regine LAMY, MD Principal Investigator: Regine LAMY, MD Centre Hospitalier F. Quesnay Recruiting Mantes La Jolie, France, 78200 Contact: JB. Auliac, MD 33 (0)1 34 97 40 70 j-b.auliac@ch-mantes-la-jolie.rss.fr Principal Investigator: JB. Auliac, MD AP-HM Recruiting Marseille, France Principal Investigator: Fabrice BARLESI, MD Institut Paoli-Calmettes Recruiting Marseille, France Contact: Anne MADROSZYK, MD Principal Investigator: Anne MADROSZYK, MD Instiut de Cancérologie Recruiting Saint Etienne, France Principal Investigator: Pierre FOURNEL, MD Service de Pneumologie Recruiting Villefranche, France Contact: L falchero, MD 33.4 74 09 27 23 lfalchero@ch-villefranche.fr Principal Investigator: L Falchero, MD Collapse << |
| NCT02665143 | Relapsed/Refractory Acute Myel... more >>oid Leukemia Collapse << | Phase 1 Phase 2 | Recruiting | December 2019 | United States, Connecticut ... more >> Yale University Recruiting New Haven, Connecticut, United States, 06520 Contact: Rebecca Lilian 203-737-8358 rebecca.lilian@yale.edu United States, Tennessee Vanderbilt Ingram Cancer Center Recruiting Nashville, Tennessee, United States, 37232 Contact: Channing V Dudley, RN, MSN 615-875-5503 channing.v.dudley@vanderbilt.edu Principal Investigator: Stephen Strickland, MD Collapse << |
| NCT03377023 | Non Small Cell Lung Cancer ... more >> Lung Cancer, Nonsmall Cell Non Small Cell Lung Cancer Metastatic Collapse << | Phase 1 Phase 2 | Recruiting | February 2021 | United States, Florida ... more >> H. Lee Moffitt Cancer Center and Research Institute Recruiting Tampa, Florida, United States, 33612 Contact: Germaine Gonzalez-Vazquez 813-745-6636 germaine.gonzalezvazquez@moffitt.org Contact: Jhanelle E. Gray, M.D. 813-745-6895 jhanelle.gray@moffitt.org Collapse << |
| NCT02149108 | Colorectal Neoplasms | Phase 3 | Completed | - | - |
| NCT01658462 | Breast Cancer | Phase 2 | Unknown | December 2016 | France ... more >> CHU Amiens- Hôpital Sud Amiens, France, 80 054 Hôpital Privé les Bonnettes Arras, France, 62000 Centre Pierre Curie Beuvry, France, 62660 CH Compiègne-Noyon Compiègne, France, 60200 Centre Léonard de Vinci Dechy, France, 59 187 Centre Oscar Lambret Lille, France, 59 020 Polyclinique de Limoges - site Chénieux Limoges, France, 87039 Institut Jean Godinot Reims, France, 51056 CMCO de la Côte d'Opale Saint Martin les Boulogne, France, 62280 Hôpital Bretonneau Tours, France, 37044 Nouvelle Clinique des Dentellières Valenciennes, France, 59300 Centre Alexis Vautrin Vandoeuvre Les Nancy, France, 54 500 Collapse << |
| NCT02149108 | - | - | Completed | - | - |
| NCT02572752 | - | - | Completed | - | - |
| NCT02572752 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT01907100 | Mesothelioma | Phase 3 | Completed | - | - |
| NCT02191865 | Hepatic Insufficiency | Phase 1 | Completed | - | Germany ... more >> 1199.200.49001 Boehringer Ingelheim Investigational Site Kiel, Germany Collapse << |
| NCT02619162 | Breast Cancer | Phase 1 | Unknown | April 2018 | Spain ... more >> Hospital Universitario de Fuenlabrada Recruiting Fuenlabrada, Madrid, Spain, 28942 Contact: Miguel Angel Quintela, PhD +34-91.732.80.00 ext 2930 mquintela@cnio.es Contact: Berta Nasarre +34-91.600.65.84 bnasarre@ext.cnio.es Principal Investigator: Miguel Quintela, PhD Sub-Investigator: Juan Victor Apala Sub-Investigator: Juan Antonio Guerra Sub-Investigator: Diego Malón Sub-Investigator: Laura Rodríguez Hospital de La Princesa Recruiting Madrid, Spain, 28006 Contact: Ramón Colomer +34.91.5202276 rcolomer@seom.org Contact: Anabel Ballestero +34.91.5202276 Principal Investigator: Ramón Colomer, PhD MD Anderson Cancer Center Withdrawn Madrid, Spain, 28033 Clínica Quirón Recruiting Madrid, Spain, 28223 Contact: Miguel Quintela, PhD 91.732.8000 ext 2930 mquintela@cnio.es Principal Investigator: Miguel Quintela, PhD Collapse << |
| NCT02191865 | - | - | Completed | - | - |
| NCT00806819 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | - |
| NCT00806819 | - | - | Completed | - | - |
| NCT02308553 | Cutaneous Malignant Melanoma | Phase 1 Phase 2 | Recruiting | June 2018 | Germany ... more >> University Hospital Essen Recruiting Essen, NRW, Germany, 45147 Contact: Dirk Schadendorf, Prof. Dr. +49-201- 723 ext 4342 dirk.schadendorf@uk-essen.de Principal Investigator: Dirk Schadendorf, Prof. Dr. Elbeklinikum Buxtehude Recruiting Buxtehude, Germany, 21614 Contact: Peter Mohr, Dr. med. peter.mohr@elbekliniken.de Principal Investigator: Peter Mohr, Dr. med. SRH Wald-Klinikum Gera Not yet recruiting Gera, Germany, 07548 Contact: Martin Kaatz, PD Dr. martin.kaatz@wkg.srh.de Principal Investigator: Martin Kaatz, PD Dr. National Centre for Tumour Diseases (NCT) Recruiting Heidelberg, Germany, 69120 Contact: Jessica Hassel, Dr. med. jessica.hassel@med.uni-heidelberg.de Principal Investigator: Jessica Hassel, Dr. med. Universitätsklinikum des Saarlandes Not yet recruiting Homburg, Germany, 66421 Contact: Claudia Pföhler, PD Dr. claudia.pfoehler@uks.eu Principal Investigator: Claudia Pföhler, PD Dr. Klinikum der Stadt Ludwigshafen am Rhein gGmbH Recruiting Ludwigshafen, Germany, 67063 Contact: Edgar Dippel, Prof. Dr. dippele@klilu.de Principal Investigator: Edgar Dippel, Prof. Dr. Universitätsklinikum Schleswig-Holstein, Campus Lübeck Not yet recruiting Lübeck, Germany, 23538 Contact: Patrick Terheyden, PD Dr. patrick.terheyden@uksh.de Principal Investigator: Patrick Terheyden, PD Dr. University Hospital München Recruiting München, Germany, 80337 Contact: Carola Berking, Prof. Dr. carola.berking@med.uni-muenchen.de Principal Investigator: Carola Berking, Prof. Dr. Fachklinik Hornheide Recruiting Münster, Germany, 48157 Contact: Michael Fluck, Dr. med. michael.fluck@fachklinik-hornheide.de Principal Investigator: Michael Fluck, Dr. med. Collapse << |
| NCT02234596 | Esophagogastric Adenocarcinoma | Phase 2 | Active, not recruiting | September 2019 | United States, New Jersey ... more >> Memoral Sloan Kettering Cancer Center Basking Ridge, New Jersey, United States Memorial Sloan Kettering Monmouth Middletown, New Jersey, United States, 07748 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Commack, New York, United States, 11725 Memorial Sloan Kettering Westchester Harrison, New York, United States, 10604 Memorial Sloan Kettering Cancer Center 1275 York Avenue New York, New York, United States, 10065 Memorial Sloan Kettering at Mercy Medical Center Rockville Centre, New York, United States Collapse << |
| NCT02300298 | - | - | Completed | - | - |
| NCT02393755 | Colon Adenocarcinoma ... more >> Rectal Adenocarcinoma Recurrent Colon Carcinoma Recurrent Rectal Carcinoma Stage IVA Colon Cancer Stage IVA Rectal Cancer Stage IVB Colon Cancer Stage IVB Rectal Cancer Collapse << | Phase 2 | Active, not recruiting | January 1, 2019 | United States, California ... more >> City of Hope Comprehensive Cancer Center Duarte, California, United States, 91010 United States, New York Roswell Park Cancer Institute Buffalo, New York, United States, 14263 Collapse << |
| NCT01417156 | Pulmonary Fibrosis | Phase 2 | Completed | - | Japan ... more >> Boehringer Ingelheim Investigational Site Himeji, Hyogo, Japan Boehringer Ingelheim Investigational Site Sakai, Osaka, Japan Boehringer Ingelheim Investigational Site Seto, Aichi, Japan Boehringer Ingelheim Investigational Site Yokohama, Kanagawa, Japan Collapse << |
| NCT02866370 | Ovarian Clear Cell Carcinoma ... more >> Endometrial Clear Cell Carcinoma Collapse << | Phase 2 | Recruiting | March 2021 | United Kingdom ... more >> Beatson West of Scotland Cancer Centre Recruiting Glasgow, Lanarkshire, United Kingdom, G12 0YN Contact: Ros Glasspool Principal Investigator: Ros Glasspool Sub-Investigator: Iain McNeish Ninewells Hospital Recruiting Dundee, Tayside, United Kingdom Principal Investigator: Michelle Ferguson Clatterbridge Cancer Centre Recruiting Liverpool, Wirral, United Kingdom Principal Investigator: Rosemary Lord Belfast City Hospital (Northern Ireland Cancer Centre) Recruiting Belfast, United Kingdom Principal Investigator: Sarah McKenna Bristol Heamatology and Cancer Centre Recruiting Bristol, United Kingdom Principal Investigator: Axel Walther Velindre Hospital Recruiting Cardiff, United Kingdom Principal Investigator: Rachel Jones Kent & Canterbury Hospital Recruiting Kent, United Kingdom Principal Investigator: Justin Waters Queen Elizabeth Queen Mother Hospital Recruiting Kent, United Kingdom Principal Investigator: Justin Waters William Harvey Hospital Recruiting Kent, United Kingdom Principal Investigator: Justin Waters St James Hospital Recruiting Leeds, United Kingdom Principal Investigator: Geoff Hall Guy's Hosital Recruiting London, United Kingdom Principal Investigator: Ana Montes Royal Marsden Hospital Recruiting London, United Kingdom Principal Investigator: Susannah Bannerjee St Bartholomew's Hospital Not yet recruiting London, United Kingdom Principal Investigator: Melanie Powell University College London Hospital Not yet recruiting London, United Kingdom Principal Investigator: Jonathan Ledermann The Christie Hospital Recruiting Manchester, United Kingdom Principal Investigator: Andrew Clamp Great Western Hospital Recruiting Swindon, United Kingdom Principal Investigator: Omar Khan Musgrove Park Hospital Recruiting Taunton, United Kingdom Principal Investigator: Clare Barlow Collapse << |
| NCT00514683 | Pulmonary Fibrosis | Phase 2 | Completed | - | - |
| NCT02300298 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Japan ... more >> 1199.90.81001 Boehringer Ingelheim Investigational Site Chiba , Kashiwa, Japan 1199.90.81003 Boehringer Ingelheim Investigational Site Kanagawa, Yokohama, Japan 1199.90.81007 Boehringer Ingelheim Investigational Site Osaka, Osakasayama, Japan 1199.90.81006 Boehringer Ingelheim Investigational Site Osaka, Osaka, Japan 1199.90.81004 Boehringer Ingelheim Investigational Site Shizuoka, Sunto-gun, Japan 1199.90.81002 Boehringer Ingelheim Investigational Site Tokyo, Chuo, Japan Collapse << |
| NCT01417156 | - | - | Completed | - | - |
| NCT02579603 | - | - | Completed | - | - |
| NCT02667704 | Healthy | Phase 1 | Completed | - | Germany ... more >> Boehringer Ingelheim Investigational Site Biberach, Germany Collapse << |
| NCT02667704 | - | - | Completed | - | - |
| NCT00514683 | - | - | Completed | - | - |
| NCT01225887 | Endometrial Adenocarcinoma ... more >> Endometrial Clear Cell Adenocarcinoma Endometrial Mucinous Adenocarcinoma Endometrial Serous Adenocarcinoma Endometrial Squamous Cell Carcinoma Endometrial Transitional Cell Carcinoma Endometrial Undifferentiated Carcinoma Malignant Uterine Corpus Mixed Epithelial and Mesenchymal Neoplasm Recurrent Uterine Corpus Carcinoma Collapse << | Phase 2 | Completed | - | - |
| NCT02579603 | Idiopathic Pulmonary Fibrosis | Phase 4 | Completed | - | United States, Connecticut ... more >> Western CT Medical Group, P.C. Danbury, Connecticut, United States, 06810 United States, Louisiana Tulane University Hospital and Clinic New Orleans, Louisiana, United States, 70112 United States, Minnesota Minnesota Lung Center Minneapolis, Minnesota, United States, 55407 United States, Missouri The Lung Research Center, LLC Chesterfield, Missouri, United States, 63017 United States, South Carolina Lowcountry Lung and Crit Care Charleston, South Carolina, United States, 29406 United States, Tennessee Vanderbilt University Medical Center Nashville, Tennessee, United States, 37232-5735 Canada, British Columbia St. Paul's Hospital Vancouver, British Columbia, Canada, V6Z 1Y6 Canada, Manitoba Concordia Hospital Winnipeg, Manitoba, Canada, R2K 3S8 France HOP Avicenne Bobigny, France, 93009 HOP de la Cavale Blanche Brest, France, 29609 HOP Louis Pradel Bron cedex, France, 69677 HOP Calmette Lille, France, 59037 HOP Pasteur Nice, France, 06001 HOP Bichat Paris, France, 75018 HOP Pontchaillou Rennes, France, 35033 Germany Klinik Donaustauf Donaustauf, Germany, 93093 Ruhrlandklinik, Westdeutsches Lungenzentrum am Universitätsklinikum Essen gGmbH Essen, Germany, 45239 Thoraxklinik-Heidelberg gGmbH am Universitätsklinikum Heidelberg Heidelberg, Germany, 69126 Italy A.O.U. Policlinico Vittorio Emanuele Catania, Italy, 95124 Osp. S. Giuseppe Fatebenefratelli Milano, Italy, 20123 A.O.U. Senese Policlinico Santa Maria alle Scotte Siena, Italy, 53100 Netherlands Sint Antonius Ziekenhuis Nieuwegein, Netherlands, 3435 CM Erasmus Medisch Centrum Rotterdam, Netherlands, 3015 CE Collapse << |
| NCT02009579 | Uterine Cervical Neoplasms | Phase 2 | Recruiting | July 2020 | Belgium ... more >> CHU Saint-Pierre Active, not recruiting Bruxelles, Belgium Institut Jules Bordet Recruiting Bruxelles, Belgium Grand Hopital de Charleroi Active, not recruiting Charleroi, Belgium UZ Antwerpen Active, not recruiting Edegem, Belgium AZ Groeninge Recruiting Kortrijk, Belgium UZ Leuven Recruiting Leuven, Belgium, 3000 Principal Investigator: Ignace Vergote, MD CHR Citadelle Recruiting Liege, Belgium CHU de Liège Recruiting Liège, Belgium, 4000 Contact: Christine Gennigens, MD Clinique et maternite St. Elisabeth Recruiting Namur, Belgium Cliniques Universitaires mont godinne Active, not recruiting Yvoir, Belgium Germany Charité Med Uni Berlin Recruiting Berlin, Germany Contact: Radoslav Chekerov, MD Universitätsklinikum Carl Gustav Carus Dresden Recruiting Dresden, Germany Contact: Pauline Wimberger, MD Kliniken Essen Mitte Active, not recruiting Essen, Germany Georg-August University Göttingen Active, not recruiting Gottingen, Germany Medical University Greifswald Recruiting Greifswald, Germany Contact: Alexander Mustea, MD University Tübingen Recruiting Tubingen, Germany Contact: Andreas Hartkopf, MD Italy Centro Riferimento Oncologico Not yet recruiting Aviano, Italy Contact: Roberto Sorio, MD Spedali Civili Not yet recruiting Brescia, Italy Contact: Germana Tognon, MD Azienda Ospedaliera Cannizzaro Not yet recruiting Catania, Italy Contact: Paolo Scollo, MD National Cancer Institute Recruiting Milano, Italy Contact: Ketta Lorusso, MD Istituto Nazionale Tumori-Pascale Naples Active, not recruiting Naples, Italy Padova Istituti Oncologico Veneto Not yet recruiting Padova, Italy Contact: Maria Nicoletto, MD University Pisa Not yet recruiting Pisa, Italy Contact: Angiolo Gadducci, MD AUSL Reggio Emilia Not yet recruiting Reggio Emilia, Italy Contact: allesandra bologna, MD Poloclinico A Gemelli Not yet recruiting Rome, Italy Contact: Giovanni Scambia, MD Mauriziano -Torino Active, not recruiting Torino, Italy S. Anna Torino Recruiting Torino, Italy Contact: Paolo Zola, MD Spain Hospital Provincial Reina Sofia Recruiting Córdoba, Spain, 14004 Contact: Maria Jesús Rubio, MD H. Ramón y Cajal Recruiting Madrid, Spain, 28034 Contact: Eva Maria Guerra, MD Hospital Clinico San Carlos Recruiting Madrid, Spain, 28040 Contact: Antonio Casado, MD Hospital Universitario Morales Meseguer Recruiting Murcia, Spain, 30008 Contact: Helena Garcia, MD Hospital Son Llatzer Recruiting Palma Mallorca, Spain, 07198 Contact: Isabel Bover, MD Collapse << |
| NCT03062943 | Lymphangioleiomyomatosis | Phase 2 | Recruiting | October 2020 | Italy ... more >> Pneumology unit Recruiting Milan, Italy, 20123 Contact: Sergio A Harari, MD +39 02 55 40 4580 sergioalfonso.harari@multimedica.it Contact: Mara Cattaeno +39 02 55 40 4127 mara.cattaneo@multimedica.it Principal Investigator: Sergio A Harari, MD Sub-Investigator: Olga Torre, MD Sub-Investigator: Roberto Cassandro, MD Collapse << |
| NCT00706628 | Prostatic Neoplasms | Phase 2 | Completed | - | United Kingdom ... more >> 1239.3.4402 Boehringer Ingelheim Investigational Site Belfast, United Kingdom 1239.3.4406 Boehringer Ingelheim Investigational Site Bournemouth, United Kingdom 1239.3.4408 Boehringer Ingelheim Investigational Site Brighton, United Kingdom 1239.3.4409 Boehringer Ingelheim Investigational Site Cheltenham, United Kingdom 1239.3.4405 Boehringer Ingelheim Investigational Site Glasgow, United Kingdom 1239.3.4403 Boehringer Ingelheim Investigational Site Newcastle Upon Tyne, United Kingdom 1239.3.4411 Boehringer Ingelheim Investigational Site Southampton, United Kingdom 1239.3.4401 Boehringer Ingelheim Investigational Site Sutton, United Kingdom 1239.3.4410 Boehringer Ingelheim Investigational Site Truro, United Kingdom Collapse << |
| NCT00706628 | - | - | Completed | - | - |
| NCT00987935 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Korea, Republic of ... more >> 1199.39.82001 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1199.39.82002 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1199.39.82003 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1199.39.82004 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1199.39.82005 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of 1199.39.82006 Boehringer Ingelheim Investigational Site Seoul, Korea, Republic of Taiwan 1199.39.88606 Boehringer Ingelheim Investigational Site Changhua, Taiwan 1199.39.88609 Boehringer Ingelheim Investigational Site Kaohsiung, Taiwan 1199.39.88610 Boehringer Ingelheim Investigational Site Kaohsiung, Taiwan 1199.39.88605 Boehringer Ingelheim Investigational Site Taichung, Taiwan 1199.39.88608 Boehringer Ingelheim Investigational Site Tainan City, Taiwan 1199.39.88602 Boehringer Ingelheim Investigational Site Tainan, Taiwan 1199.39.88601 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1199.39.88603 Boehringer Ingelheim Investigational Site Taipei, Taiwan 1199.39.88604 Boehringer Ingelheim Investigational Site Taoyuan County, Taiwan 1199.39.88607 Boehringer Ingelheim Investigational Site Yunlin County, Taiwan Collapse << |
| NCT00710762 | - | - | Completed | - | - |
| NCT02299141 | Carcinoma, Non-Small-Cell Lung... more >> Non-Small Cell Lung Cancer Nonsmall Cell Lung Cancer Collapse << | Phase 1 | Recruiting | February 28, 2020 | United States, Missouri ... more >> Washington University School of Medicine Recruiting Saint Louis, Missouri, United States, 63110 Contact: Ramaswamy Govindan, M.D. 314-362-5654 rgovindan@wustl.edu Contact: Cindy Fogal 314-362-1518 clfogal@wustl.edu Principal Investigator: Ramaswamy Govindan, M.D. Sub-Investigator: Saiama Waqar, M.D. Sub-Investigator: Daniel Morgensztern, M.D. Sub-Investigator: Ticiana Leal, M.D. United States, Wisconsin University of Wisconsin Clinical Science Center Recruiting Madison, Wisconsin, United States, 53705 Contact: Ticiana Leal, M.D. 608-265-1700 tbleal@medicine.wisc.edu Principal Investigator: Ticiana Leal, M.D. Sub-Investigator: Toby C Campbell, M.D. Sub-Investigator: Anne Traynor, M.D. Sub-Investigator: Justine Yang Bruce, M.D. Sub-Investigator: Rosemary Sahagian Neider, N.P. Collapse << |
| NCT01225887 | - | - | Completed | - | - |
| NCT00710762 | Ovarian Neoplasms | Phase 2 | Completed | - | United Kingdom ... more >> 1199.9.4413 Boehringer Ingelheim Investigational Site Burton on Trent, United Kingdom 1199.9.4412 Boehringer Ingelheim Investigational Site Cambridge, United Kingdom 1199.9.4407 Boehringer Ingelheim Investigational Site Creigiau, Cardiff, United Kingdom 1199.9.4410 St James's University Hospital Leeds, United Kingdom 1199.9.4401 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.9.4404 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.9.4409 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.9.4411 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.9.4406 Boehringer Ingelheim Investigational Site Manchester, United Kingdom 1199.9.4402 Boehringer Ingelheim Investigational Site Northwood, United Kingdom 1199.9.4405 Boehringer Ingelheim Investigational Site Sutton, United Kingdom Collapse << |
| NCT00801294 | Colorectal Neoplasms | Phase 2 | Completed | - | France ... more >> 1239.2.3305A clinique Saint Jean Lyon, France 1239.2.3305B Cabinet Médical Lyon, France 1239.2.3301A Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301B Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301C Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301D Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301E Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301F Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301G Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301H Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301I Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301J Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3301K Hôpital Saint Antoine Paris Cedex 12, France 1239.2.3302A Hôpital Tenon Paris Cedex 20, France 1239.2.3302B Hôpital Tenon Paris Cedex 20, France 1239.2.3304A Hôpital Robert Debré Reims Cedex, France 1239.2.3304B Hôpital Robert Debré Reims Cedex, France 1239.2.3304C Hôpital Robert Debré Reims Cedex, France 1239.2.3303A Institut Gustave Roussy Villejuif Cedex, France 1239.2.3303B Institut Gustave Roussy Villejuif Cedex, France 1239.2.3303C Institut Gustave Roussy Villejuif Cedex, France 1239.2.3303D Institut Gustave Roussy Villejuif Cedex, France 1239.2.3303E Institut Gustave Roussy Villejuif Cedex, France 1239.2.3303F Institut Gustave Roussy Villejuif Cedex, France Collapse << |
| NCT02808247 | Sarcoma, Soft Tissue | Phase 2 | Recruiting | January 2022 | Belgium ... more >> Hopitaux Universitaires Bordet-Erasme - Institut Jules Bordet Recruiting Brussels, Belgium, 1000 Contact: Gil Thierry +32 25413188 thierry.gil@bordet.be Cliniques Universitaires Saint-Luc (121) Recruiting Brussels, Belgium Contact: Filomena Mazzeo Filomena.Mazzeo@uclouvain.be U.Z. Leuven - Campus Gasthuisberg (147) Recruiting Leuven, Belgium Contact: Patrick Schöffski patrick.schoffski@uzleuven.be France Institut Bergonie Recruiting Bordeaux, France, 33076 Contact: Antoine Italiano a.italiano@bordeaux.unicancer.fr Centre Leon Berard (227) Recruiting Lyon, France Contact: Jean-Yves Blay jean-yves.blay@lyon.unicancer.fr Gustave Roussy (225) Recruiting Villejuif, France Contact: Olivier Mir olivier.mir@gustaveroussy.fr Lithuania Vilnius University Hospital Santariskiu Santaros Clinics Klinikos (9453) Recruiting Vilnius, Lithuania, 08661 Netherlands The Netherlands Cancer Institute-Antoni Van Leeuwenhoekziekenhuis (301) Recruiting Amsterdam, Netherlands, 1066 Leiden University Medical Centre (310) Recruiting Leiden, Netherlands, 2300 Poland Maria Sklodowska-Curie Memorial Cancer Centre Recruiting Warsaw, Poland, 02 781 Contact: Piotr Rutkowski rutkowskip@coi.waw.pl Spain Institut Catala d'Oncologia - ICO Badalona - Hospital Germans Trias i Pujol (Institut Catala D'Oncologia) Recruiting Barcelona, Spain, 08916 Contact: Anna Estival estivalanna@gmail.com Hospital Universitario San Carlos (366) Recruiting Madrid, Spain, 28040 United Kingdom Royal Marsden Hospital - Chelsea, London (613) Recruiting London, United Kingdom, SW3 6JJ Collapse << |
| NCT02225405 | Stage IB Non-Small Cell Lung C... more >>arcinoma AJCC v7 Stage II Non-Small Cell Lung Cancer AJCC v7 Stage IIA Non-Small Cell Lung Carcinoma AJCC v7 Stage IIB Non-Small Cell Lung Carcinoma AJCC v7 Stage IIIA Non-Small Cell Lung Cancer AJCC v7 Collapse << | Phase 1 | Active, not recruiting | April 30, 2020 | United States, Texas ... more >> M D Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02568449 | Recurrent Pleural Malignant Me... more >>sothelioma Stage IV Pleural Mesothelioma Collapse << | Phase 2 | Recruiting | January 1, 2018 | United States, California ... more >> University of California Davis Comprehensive Cancer Center Not yet recruiting Sacramento, California, United States, 95817 Contact: David R. Gandara 916-734-8452 david.gandara@ucdmc.ucdavis.edu Principal Investigator: David R. Gandara United States, Michigan University of Michigan Comprehensive Cancer Center Recruiting Ann Arbor, Michigan, United States, 48109 Contact: Gregory P. Kalemkerian 734-615-4762 kalemker@umich.edu Principal Investigator: Gregory P. Kalemkerian, M.D. Wayne State University/Karmanos Cancer Institute Recruiting Detroit, Michigan, United States, 48201 Contact: Antoinette J. Wozniak 313-576-8752 wozniakt@karmanos.org Principal Investigator: Antoinette J. Wozniak, M.D. Sub-Investigator: Shirsh Gadgeel, M.D. Sub-Investigator: Ammar Sukari, M.D. United States, New York Memorial Sloan Kettering Cancer Center Not yet recruiting New York, New York, United States, 10065 Contact: Marjorie G. Zauderer, M.D. 646-888-4201 zauderem@mskcc.org Principal Investigator: Marjorie G. Zauderer, M.D. Collapse << |
| NCT01362361 | Colorectal Cancer | Phase 2 | Unknown | December 2013 | Germany ... more >> Universitätsklinikum Halle Recruiting Halle, Sachsen-Anhalt, Germany, 06120 Contact: Thomas Seufferlein, MD Principal Investigator: Thomas Seufferlein, MD Sub-Investigator: Thomas J. Ettrich, MD Schwerpunktpraxis für Hämatologie und Onkologie Bottrop und Dorsten Recruiting Bottrop, Germany Principal Investigator: Carla V. Hannig, MD Universitätsklinikum Greifswald -Klinik für Innere Medizin A Recruiting Greifswald, Germany Principal Investigator: Julia Mayerle, MD Gemeinschaftspraxis und Tagesklinik Onkologie und Gastroenterologie - Halle Recruiting Halle, Germany Principal Investigator: Stefanie Frank-Gleich, MD Klinikum Karlsruhe, Medizinische Klinik III Recruiting Karlsruhe, Germany Principal Investigator: Margarethe Schmier, MD Kliniken der Stadt Köln gGmbH - Krankenhaus Holweide Recruiting Köln, Germany Principal Investigator: Claudia Lang, MD Universitätsmedizin Mannheim - TTZ am Interdisziplinären Tumorzentrum Recruiting Mannheim, Germany Principal Investigator: Ralf Hofheinz, MD Joh. Wesling Klinikum Minden Recruiting Minden, Germany Principal Investigator: Hans-Joachim Tischler, MD Stauferklinikum Schwäbisch Gmünd - Mutlangen - Zentrum Innere Medizin Recruiting Mutlangen, Germany Principal Investigator: Holger Hebart, MD Klinikum der Universität München-Großhadern - Medizinische Klinik und Poliklink III Recruiting München, Germany Principal Investigator: Volker Heinemann, MD Leopoldina-Krankenhaus Schweinfurt - Medizinische Klinik 2 Recruiting Schweinfurt, Germany Principal Investigator: Stephan Kanzler, MD Universitätsklinikum Ulm - Klinik für Innere Medizin I Recruiting Ulm, Germany Principal Investigator: Götz von Wichert, MD Collapse << |
| NCT01948141 | Recurrent Non-small Cell Lung ... more >>Cancer Squamous Cell Lung Cancer Stage III Non-small Cell Lung Cancer Stage IV Non-small Cell Lung Cancer Collapse << | Phase 2 | Completed | - | United States, New York ... more >> Roswell Park Cancer Institute Buffalo, New York, United States, 14263 United States, Ohio University Hospitals Case Medical Center Cleveland, Ohio, United States, 44106 Collapse << |
| NCT00987935 | - | - | Completed | - | - |
| NCT00715403 | Neoplasms | Phase 1 | Completed | - | France ... more >> 1199.16.3306A Boehringer Ingelheim Investigational Site Bordeaux cedex, France 1199.16.3311A Boehringer Ingelheim Investigational Site Clichy Cedex, France 1199.16.3311B Boehringer Ingelheim Investigational Site Clichy Cedex, France 1199.16.3313A Boehringer Ingelheim Investigational Site Paris Cedex 10, France 1199.16.3313E Boehringer Ingelheim Investigational Site Paris Cedex 10, France 1199.16.3302A Boehringer Ingelheim Investigational Site Paris cedex 15, France 1199.16.3312A Boehringer Ingelheim Investigational Site Paris, France Germany 1199.16.49001 Boehringer Ingelheim Investigational Site Freiburg/Breisgau, Germany 1199.16.49004 Boehringer Ingelheim Investigational Site Grosshansdorf, Germany 1199.16.49008 Boehringer Ingelheim Investigational Site Tübingen, Germany 1199.16.49005 Boehringer Ingelheim Investigational Site Wiesbaden, Germany Collapse << |
| NCT01015118 | Ovarian Neoplasms ... more >> Peritoneal Neoplasms Collapse << | Phase 3 | Completed | - | - |
| NCT02598193 | - | - | Completed | - | - |
| NCT01015118 | - | - | Completed | - | - |
| NCT01948141 | - | - | Completed | - | - |
| NCT01004003 | Carcinoma, Hepatocellular | Phase 2 | Completed | - | Austria ... more >> 1199.37.43001 Boehringer Ingelheim Investigational Site Wien, Austria 1199.37.43002 Boehringer Ingelheim Investigational Site Wien, Austria France 1199.37.33001 Boehringer Ingelheim Investigational Site Paris, France 1199.37.33002 Boehringer Ingelheim Investigational Site Paris, France Germany 1199.37.49008 Boehringer Ingelheim Investigational Site Berlin, Germany 1199.37.49009 Boehringer Ingelheim Investigational Site Erlangen, Germany 1199.37.49002 Boehringer Ingelheim Investigational Site Freiburg, Germany 1199.37.49001 Boehringer Ingelheim Investigational Site Hannover, Germany 1199.37.49010 Boehringer Ingelheim Investigational Site Heidelberg, Germany 1199.37.49005 Boehringer Ingelheim Investigational Site Jena, Germany 1199.37.49004 Boehringer Ingelheim Investigational Site Magdeburg, Germany 1199.37.49003 Boehringer Ingelheim Investigational Site München, Germany 1199.37.49006 Boehringer Ingelheim Investigational Site Tübingen, Germany Hungary 1199.37.36001 Boehringer Ingelheim Investigational Site Debrecen, Hungary Netherlands 1199.37.31002 Boehringer Ingelheim Investigational Site Leiden, Netherlands 1199.37.31001 Boehringer Ingelheim Investigational Site Utrecht, Netherlands Poland 1199.37.48002 Boehringer Ingelheim Investigational Site Olsztyn, Poland 1199.37.48003 Boehringer Ingelheim Investigational Site Warsaw, Poland 1199.37.48001 Boehringer Ingelheim Investigational Site Warszawa, Poland Romania 1199.37.40002 Boehringer Ingelheim Investigational Site Bucharest, Romania 1199.37.40003 Boehringer Ingelheim Investigational Site Cluj-Napoca, Romania United Kingdom 1199.37.44001 Boehringer Ingelheim Investigational Site Edgbaston, Birmingham, United Kingdom 1199.37.44005 Boehringer Ingelheim Investigational Site Glasgow, United Kingdom 1199.37.44008 Boehringer Ingelheim Investigational Site Liverpool, United Kingdom 1199.37.44002 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.37.44003 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.37.44006 Boehringer Ingelheim Investigational Site Manchester, United Kingdom 1199.37.44004 Boehringer Ingelheim Investigational Site Nottingham, United Kingdom Collapse << |
| NCT00715403 | - | - | Completed | - | - |
| NCT00904839 | Colorectal Neoplasms | Phase 2 | Completed | - | - |
| NCT01004003 | - | - | Completed | - | - |
| NCT02598193 | Idiopathic Pulmonary Fibrosis | Phase 4 | Completed | - | - |
| NCT01251484 | Recurrent Glioblastoma | Phase 2 | Completed | - | Denmark ... more >> Rigshospitalet Copenhagen, Denmark, 2100 Collapse << |
| NCT00904839 | - | - | Completed | - | - |
| NCT00730821 | Neoplasms | Phase 1 | Completed | - | United States, Arizona ... more >> 1239.1.3 Boehringer Ingelheim Investigational Site Scottsdale, Arizona, United States United States, Florida 1239.1.1 Boehringer Ingelheim Investigational Site Tampa, Florida, United States United States, Michigan 1239.1.2 Boehringer Ingelheim Investigational Site Detroit, Michigan, United States United States, Nebraska 1239.1.4 Boehringer Ingelheim Investigational Site Omaha, Nebraska, United States Collapse << |
| NCT02902484 | Cancer of Pancreas | Phase 1 Phase 2 | Recruiting | September 2020 | United States, Texas ... more >> University of Texas Southwestern Medical Center Recruiting Dallas, Texas, United States, 75063 Contact: Silvia Pilarski, RN 214-648-5919 Silvia.Pilarski@UTSouthwestern.edu Principal Investigator: Muhammad Beg, MD Collapse << |
| NCT01484080 | Breast Cancer | Phase 1 Phase 2 | Completed | - | Spain ... more >> Hospital Universitari de Bellvitge Hospitalet de Llobregat, Barcelona, Spain, 08907 Hospital Universitario de Fuenlabrada Fuenlabrada, Madrid, Spain, 28942 MD Anderson Cancer Centre Madrid Madrid, Spain, 28033 Collapse << |
| NCT02182258 | Healthy | Phase 1 | Completed | - | - |
| NCT02182193 | Healthy | Phase 1 | Completed | - | - |
| NCT01136174 | - | - | Completed | - | - |
| NCT02182050 | Carcinoma, Non-Small-Cell Lung | Phase 2 | Completed | - | - |
| NCT01170065 | Pulmonary Fibrosis | Phase 2 | Completed | - | - |
| NCT00979576 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Terminated | - | Japan ... more >> 1199.28.003 Boehringer Ingelheim Investigational Site Chiba,Kashiwa, Japan 1199.28.002 Boehringer Ingelheim Investigational Site Miyakojima-ku, Osaka, Japan 1199.28.001 Boehringer Ingelheim Investigational Site Osaka-Sayama, Osaka, Japan Collapse << |
| NCT01788982 | Medullary Thyroid Cancer (MTC)... more >> Differentiated Thyroid Cancer (DTC) Collapse << | Phase 2 | Active, not recruiting | December 2018 | Belgium ... more >> A.Z. St. Jan Brugge, Belgium, 8000 Hopitaux Universitaires Bordet-Erasme - Institut Jules Bordet Brussels, Belgium, 1000 Cliniques Universitaires Saint-Luc Brussels, Belgium, 1200 Universitair Ziekenhuis Antwerpen Edegem, Antwerpen, Belgium, 2650 Universitair Ziekenhuis Gent Gent, Belgium, 9000 U.Z. Leuven - Campus Gasthuisberg Leuven, Belgium, 3000 Denmark Odense University Hospital Odense, Denmark, 5000 France CHU d'Angers Angers, France, 49933 Institut Bergonie Bordeaux, France, 33076 Centre Regional Francois Baclesse Caen, France, 14076 Centre Georges-Francois-Leclerc Dijon, France, 21079 Centre Leon Berard Lyon, France, 69008 Assitance Publique - Hopitaux de Paris - Hopital Saint-Louis Paris, France, 75475 Assistance Publique - Hopitaux de Paris - La Pitié Salpétrière Paris, France, 75651 Centre Jean Godinot Reims, France, 51056 Institut Gustave Roussy Villejuif, France Germany Ludwig-Maximilians-Universitaet Muenchen - Klinikum der Universitaet Muenchen - Campus Grosshadern Munich, Germany, DE 81377 Universitaetsklinikum Wuerzburg Wuerzburg, Germany, DE 97080 Italy Fondazione IRCCS Istituto Nazionale dei Tumori Milano, Italy Azienda Ospedaliera Universitaria "Federico II" Napoli, Italy, 80131 Netherlands University Medical Center Groningen Groningen, Netherlands, 9713 Leiden University Medical Centre Leiden, Netherlands, 2300 Radboud University Medical Center Nijmegen Nijmegen, Netherlands, 6500 Poland Maria Sklodowska-Curie Memorial Cancer Centre Warsaw, Poland, PL 02 781 Spain Hospital General Vall D'Hebron Barcelona, Spain, ES 08035 United Kingdom Royal Marsden Hospital - Sutton, Surrey Sutton, Surrey, United Kingdom, SM2 5PT NHS Greater Glasgow and Clyde - Beatson West of Scotland Cancer Centre - Gartnavel General Hospital Glasgow, United Kingdom, G12 0YN Collapse << |
| NCT00979576 | - | - | Terminated | - | - |
| NCT01488344 | Acute Myeloid Leukemia | Phase 1 Phase 2 | Unknown | - | Germany ... more >> Universitätsklinikum Münster, Medizinische Klinik und Poliklinik A Münster, Germany, 48149 Collapse << |
| NCT03283007 | Lung-transplant Recipients | Phase 3 | Not yet recruiting | May 2022 | - |
| NCT03562416 | Idiopathic Pulmonary Fibrosis ... more >> Lung Transplant; Complications Collapse << | Phase 2 | Not yet recruiting | December 2021 | United States, Pennsylvania ... more >> Temple University Hospital Not yet recruiting Philadelphia, Pennsylvania, United States, 19140 Contact: Francine McGonagle, BSN,RN 215-707-2682 Francine.McGonagle@tuhs.temple.edu Contact: Shubhra Srivastava-Malhotra 215-707-0945 Shubhra.Srivastava-Malhotra@tuhs.temple.edu Principal Investigator: Jonathan A Galli, MD Sub-Investigator: Gerard J Criner, MD Sub-Investigator: Patrick Mulhall, MD Sub-Investigator: Kartik Shenoy, MD Sub-Investigator: James Brown, MD Sub-Investigator: Thomas Rogers, PhD Collapse << |
| NCT02182154 | Healthy | Phase 1 | Completed | - | - |
| NCT00805194 | Carcinoma, Non-Small-Cell Lung | Phase 3 | Completed | - | - |
| NCT01022853 | Neoplasms | Phase 1 | Completed | - | Italy ... more >> 1230.7.39002 Boehringer Ingelheim Investigational Site Ancona, Italy 1230.7.39001 Boehringer Ingelheim Investigational Site Milano, Italy Collapse << |
| NCT00805194 | - | - | Completed | - | - |
| NCT02182245 | Genital Neoplasms, Female | Phase 1 | Completed | - | - |
| NCT00876460 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Japan ... more >> 1199.29.002 Boehringer Ingelheim Investigational Site Fukuoka, Fukuoka, Japan 1199.29.001 Boehringer Ingelheim Investigational Site Osaka-Sayamashi, Osaka, Japan Collapse << |
| NCT02182063 | Prostatic Neoplasms | Phase 2 | Completed | - | - |
| NCT01024920 | Carcinoma, Renal Cell | Phase 2 | Active, not recruiting | February 15, 2019 | Hungary ... more >> University of Pecs Medical School, Dept. of Oncotherapy Pecs, Hungary, 7624 Poland Ziemia Lubelska Oncological Center, Lublin Lublin, Poland, 20-099 Onco.Cent. - Instit. of Maria Sklodowskiej-Curie Warszawa, Poland, 02-781 Romania Military Central Clinical Emergency Hospital Bucharest, Romania, 010825 Sf. Nectarie Oncology Center, Craiova Craiova, Romania, 200347 ONCOLAB SRL, Craiova Craiova, Romania, 200385 Ukraine Municipal Establishment Cherkasy Oncology Centre Cherkasy, Ukraine, 18009 Bukovynsk State Medical University Chernivtsi, Ukraine, 58013 City Clinical Hospital #4, Dnipropetrovsk State Medical Academy Dnipropetrovks, Ukraine, 49102 CI of LRC Lviv Onco.Reg.Treat.&Diag.Cent. Lviv, Ukraine, 79031 Uzhgorod National University, Oncology Centre Uzhgorod, Ukraine, 88000 United Kingdom Addenbrooke's Hospital Cambridge, United Kingdom, CB2 0QQ Beatson West of Scotland Cancer Centre Glasgow, United Kingdom, G12 0YN Surrey Cancer Research Institute Guildford, United Kingdom, GU2 7WG St James's University Hospital Leeds, United Kingdom, LS9 7TF Collapse << |
| NCT01380782 | Glioblastoma ... more >>Gliosarcoma Anaplastic Astrocytoma Anaplastic Oligodendroglioma Anaplastic Oligoastrocytoma Collapse << | Phase 2 | Completed | - | United States, Massachusetts ... more >> Massachusetts General Hospital Boston, Massachusetts, United States, 02114 Dana-Farber Cancer Institute Boston, Massachusetts, United States, 02215 United States, Ohio Cleveland Clinic Cleveland, Ohio, United States, 44195 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908-4324 Collapse << |
| NCT01022853 | - | - | Completed | - | - |
| NCT01335464 | Pulmonary Fibrosis | Phase 3 | Completed | - | - |
| NCT02835833 | Renal Cell Carcinoma ... more >> Colorectal Adenocarcinoma Non-squamous Non-small Cell Lung Cancer Platinum-refractory Ovarian Carcinoma Cervical Carcinoma Collapse << | Phase 1 | Completed | - | United States, Alabama ... more >> University of Alabama at Birmingham Comprehensive Cancer Center Birmingham, Alabama, United States, 35294 Collapse << |
| NCT01136174 | Idiopathic Pulmonary Fibrosis | Phase 2 | Completed | - | Japan ... more >> 1199.31.002 Boehringer Ingelheim Investigational Site Bunkyo-ku,Tokyo, Japan 1199.31.004 Boehringer Ingelheim Investigational Site Hamamatsu, Shizuoka, Japan 1199.31.008 Boehringer Ingelheim Investigational Site Himeji, Hyogo, Japan 1199.31.006 Boehringer Ingelheim Investigational Site Nagoya, Aichi, Japan 1199.31.007 Boehringer Ingelheim Investigational Site Sakai, Osaka, Japan 1199.31.005 Boehringer Ingelheim Investigational Site Seto, Aichi, Japan 1199.31.001 Boehringer Ingelheim Investigational Site Shimotsuke,Tochigi, Japan 1199.31.003 Boehringer Ingelheim Investigational Site Yokohama, Kanagawa, Japan Collapse << |
| NCT01380782 | - | - | Completed | - | - |
| NCT00998296 | Neoplasms | Phase 1 | Completed | - | France ... more >> 1239.14.3301A Boehringer Ingelheim Investigational Site Villejuif Cedex, France Collapse << |
| NCT00876460 | - | - | Completed | - | - |
| NCT01335477 | - | - | Completed | - | - |
| NCT01314105 | Ovarian Neoplasms ... more >> Peritoneal Neoplasms Collapse << | Phase 1 | Completed | - | Spain ... more >> 1199.119.34001 Boehringer Ingelheim Investigational Site Barcelona, Spain 1199.119.34002 Boehringer Ingelheim Investigational Site Barcelona, Spain 1199.119.34003 Boehringer Ingelheim Investigational Site L'Hospitalet de Llobregat, Spain Collapse << |
| NCT01441297 | Small Cell Lung Cancer ... more >> Small Cell Lung Cancer Recurrent Collapse << | Phase 2 | Completed | - | Korea, Republic of ... more >> National Cancer Center Goyang-si, Gyeonggi-do, Korea, Republic of, 410-769 Collapse << |
| NCT01335477 | Pulmonary Fibrosis | Phase 3 | Completed | - | - |
| NCT00998296 | - | - | Completed | - | - |
| NCT02182232 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | - |
| NCT01683682 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Germany ... more >> Krankenhaus Großhansdorf Großhansdorf, Germany, D-22927 Klinikum Kassel GmbH Kassel, Germany, D-34125 Collapse << |
| NCT01329549 | Ovarian Neoplasms | Phase 1 | Terminated | - | Japan ... more >> 1199.117.003 Boehringer Ingelheim Investigational Site Akashi, Hyogo, Japan 1199.117.002 Boehringer Ingelheim Investigational Site Chuo-ku,Tokyo, Japan 1199.117.001 Boehringer Ingelheim Investigational Site Hidaka, Saitama, Japan Collapse << |
| NCT01666600 | Glioblastoma Multiforme | Phase 1 Phase 2 | Terminated(Interim Analysis fo... more >>r Feasibility) Collapse << | - | Germany ... more >> University Hospital Heidelberg, Department of Neurooncology Heidelberg, Baden-Württemberg, Germany, 69120 University Hospital Heidelberg, Department of Pharmacology Heidelberg, Baden-Württemberg, Germany, 69120 Collapse << |
| NCT01314105 | - | - | Completed | - | - |
| NCT01346540 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Italy ... more >> 1199.82.39004 Boehringer Ingelheim Investigational Site Milano, Italy Netherlands 1199.82.3102 Boehringer Ingelheim Investigational Site Maastricht, Netherlands Spain 1199.82.3401 Boehringer Ingelheim Investigational Site Barcelona, Spain 1199.82.3406 Boehringer Ingelheim Investigational Site Madrid, Spain 1199.82.3410 Boehringer Ingelheim Investigational Site Málaga, Spain United Kingdom 1199.82.4401 Boehringer Ingelheim Investigational Site London, United Kingdom 1199.82.4402 Boehringer Ingelheim Investigational Site Manchester, United Kingdom Collapse << |
| NCT02558387 | Recurrent or Metastatic Saliva... more >>ry Gland Cancer of the Head and Neck Collapse << | Phase 2 | Unknown | January 2017 | Korea, Republic of ... more >> Samsung Medical Center Seoul, Korea, Republic of, 135-710 Collapse << |
| NCT01329549 | - | - | Terminated | - | - |
| NCT01335464 | - | - | Completed | - | - |
| NCT01684111 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | Germany ... more >> Klinikum Kassel GmbH Kassel, Hessen, Germany, 34125 LungenClinic Großhansdorf GmbH Großhansdorf, Germany, 22927 Thoraxklinik Universitätsklinikum Heidelberg Heidelberg, Germany, D-69126 Klinikum der Universität München München, Germany, D-80336 Collapse << |
| NCT02182141 | Multiple Myeloma | Phase 1 | Completed | - | - |
| NCT01610869 | Ovarian Cancer ... more >> Fallopian Tube Cancer Collapse << | Phase 2 | Active, not recruiting | January 2019 | United Kingdom ... more >> Kent Oncology Centre Maidstone, Kent, United Kingdom Beatson West of Scotland Cancer Centre Glasgow, Scotland, United Kingdom Velindre Hospital Cardiff, Wales, United Kingdom Royal United Hospital Bath, United Kingdom Addenbrookes Hospital Cambridge, United Kingdom Royal Derby Hospital Derby, United Kingdom Royal Surrey County Hospital Guildford, United Kingdom St James's University Hospital Leeds, United Kingdom Clatterbridge Centre for Oncology Liverpool, United Kingdom Mount Vernon Hospital London, United Kingdom Royal Marsden Hospital London, United Kingdom St Bartholomew's Hospital London, United Kingdom University College London Hospital (UCLH) London, United Kingdom Christie Hospital Manchester, United Kingdom Churchill Hospital Oxford, United Kingdom Wexham Park Hospital Slough, United Kingdom Collapse << |
| NCT02496585 | Lung Cancer L... more >>ung Metastases Collapse << | Phase 2 | Recruiting | July 2019 | United States, Massachusetts ... more >> Brigham and Women's Hospital Recruiting Boston, Massachusetts, United States, 02115 Contact: Raymond Mak, MD 617-632-3591 United States, New Jersey Memorial Sloan Kettering Cancer Center at Basking Ridge Recruiting Basking Ridge, New Jersey, United States, 07920 Contact: Andreas Rimner, MD 212-639-6025 Memorial Sloan Kettering Monmouth Recruiting Middletown, New Jersey, United States, 07748 Contact: Andreas Rimner, MD 212-639-6025 Memorial Sloan Kettering Bergen Recruiting Montvale, New Jersey, United States, 07645 Contact: Andreas Rimner, MD 212-639-6025 United States, New York Memorial Sloan Kettering Cancer Center @ Suffolk Recruiting Commack, New York, United States, 11725 Contact: Andreas Rimner, MD 212-639-6025 Memorial Sloan Kettering Westchester Recruiting Harrison, New York, United States, 10604 Contact: Andreas Rimner, MD 212-639-6025 Memorial Sloan Kettering Cancer Center Recruiting New York, New York, United States, 10065 Contact: Andreas Rimner, MD 212-639-6025 Contact: Jamie Chaft, MD 646-888-4545 Principal Investigator: Andreas Rimner, MD Memorial Sloan Kettering at Mercy Medical Center Recruiting Rockville Centre, New York, United States Contact: Andreas Rimner, MD 212-639-6025 Contact: Jamie Chaft, MD 646-888-4545 Principal Investigator: Andreas Rimner, MD United States, Texas Md Anderson Cancer Center Recruiting Houston, Texas, United States, 77030 Contact: Daniel Gomez, MD 713-563-8446 Principal Investigator: Daniel Gomez, MD Collapse << |
| NCT01346540 | - | - | Completed | - | - |
| NCT01619085 | Idiopathic Pulmonary Fibrosis | Phase 3 | Active, not recruiting | March 4, 2019 | - |
| NCT02182219 | Prostatic Neoplasms | Phase 1 | Completed | - | - |
| NCT02182102 | Carcinoma, Non-Small-Cell Lung | Phase 1 | Completed | - | - |
| NCT01951846 | Malignant Solid Tumour | Phase 1 | Completed | - | Germany ... more >> Klinik für Tumorbiologie, Abteilung Internistische Onkologie Freiburg, Germany, 79106 Collapse << |
| NCT02152059 | Small Cell Lung Cancer ... more >> Platinum-sensitive Collapse << | Phase 2 | Withdrawn(contract issues) | - | Canada, Alberta ... more >> Cross Cancer Institute Edmonton, Alberta, Canada, T6G 1Z2 Collapse << |
| NCT02278978 | Urothelial Carcinoma | Phase 2 | Unknown | March 2016 | Taiwan ... more >> Chia-Chi Lin Taipei, Taiwan Collapse << |
| NCT02182206 | Tumors | Phase 1 | Completed | - | - |
| NCT01349296 | Solid Tumors | Phase 1 | Completed | - | Germany ... more >> University Hospital of Cologne Cologne, NRW, Germany, 50937 Collapse << |
| NCT01485874 | Ovarian Cancer | Phase 1 | Terminated(Funding withdrawn d... more >>ue to drug unavailability) Collapse << | - | United States, Indiana ... more >> Indiana University Melvin and Bren Simon Cancer Center Indianapolis, Indiana, United States, 46202 Collapse << |
| NCT01669798 | Ovarian Cancer ... more >> Fallopian Tube Cancer Peritoneal Cancer Collapse << | Phase 2 | Completed | - | United States, North Carolina ... more >> Duke Cancer Institute Durham, North Carolina, United States, 27710 United States, Virginia University of Virginia Charlottesville, Virginia, United States, 22908 Virginia Oncology Associates Norfolk, Virginia, United States, 23502 Collapse << |
| NCT02182128 | Tumors | Phase 1 | Completed | - | - |
| NCT01619085 | - | - | Active, not recruiting | - | - |
| NCT02389764 | Breast Cancer | Phase 2 | Terminated(Slow Accrual) | - | United States, Texas ... more >> University of Texas MD Anderson Cancer Center Houston, Texas, United States, 77030 Collapse << |
| NCT02182076 | Healthy | Phase 1 | Completed | - | - |
| NCT01669798 | - | - | Completed | - | - |
| 靶点 | Description | IC50 |
|---|---|---|
| VEGFR3 | IC50:13nM | |
| VEGFR2 | IC50:13nM | |
| VEGFR1 | IC50:34nM |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
