| NCT01309542 | Major Depressive Disorder | Phase 3 | Completed | - | - |
| NCT01414920 | Diabetes Mellitus, Type 2 | Phase 2 | Completed | - | - |
| NCT00201981 | Keratoconjunctivitis Sicca | Phase 3 | Completed | - | - |
| NCT00848107 | Systemic Sclerosis | Phase 2 | Terminated(The TDE-DU-202 exte... more >>nsion study was discontinued after the randomized, placebo-controlled TDE-DU-201 study did not meet its primary efficacy objective.) Collapse << | - | - |
| NCT02909725 | Sedentary Lifestyle
... more >> Diabetes Mellitus, Gestational Collapse << | Not Applicable | Unknown | December 2016 | United States, Iowa
... more >>
Iowa State University
Recruiting
Ames, Iowa, United States, 50011
Contact: Caroline McKinney, BS 515-202-5569 clmck@iastate.edu
Contact: Christina Campbell, PhD 515-294-4260 ccampbel@iastate.edu
Principal Investigator: Christina Campbell, PhD Collapse << |
| NCT01104870 | Pulmonary Hypertension | Phase 2 | Completed | - | United States, Arizona
... more >>
St. Joseph's Hospital and Medical Center
Phoenix, Arizona, United States, 85013
University of Arizona
Tucson, Arizona, United States, 87524
United States, California
University of California Los Angeles Pulmonary Division
Los Angeles, California, United States, 90095
University of California Davis Medical Center
Sacramento, California, United States, 95817
United States, New York
University of Rochester Medical Center, Mary Parkes Center
Rochester, New York, United States, 14623
United States, Ohio
The Carl and Edyth Lindner Research Center at The Christ Hospital
Cincinnati, Ohio, United States, 45219
University of Cincinnati
Cincinnati, Ohio, United States, 45267
The Ohio State University Medical Center
Columbus, Ohio, United States, 43221
United States, Pennsylvania
University of Pittsburgh Medical Center
Pittsburgh, Pennsylvania, United States, 15213 Collapse << |
| NCT02160730 | Cushings Disease | Phase 2 | Terminated(NIH grant ended.) | - | United States, California
... more >>
Cedars-Sinai Medical Center
Los Angeles, California, United States, 90048 Collapse << |
| NCT00848107 | - | - | Terminated(The TDE-DU-202 exte... more >>nsion study was discontinued after the randomized, placebo-controlled TDE-DU-201 study did not meet its primary efficacy objective.) Collapse << | - | - |
| NCT00151398 | Alzheimer Disease | Phase 2 | Completed | - | - |
| NCT01546649 | Premenopausal Breast Cancer | Phase 3 | Completed | - | Japan
... more >>
Nagoya-shi, Aichi, Japan
Fukushima-shi, Fukushima, Japan
Maebashi-shi, Gunma, Japan
Ohta-shi, Gunma, Japan
Sapporo-shi, Hokkaido, Japan
Isehara-shi, Kanagawa, Japan
Yokohama-shi, Kanagawa, Japan
Kumamoto-shi, Kumamoto, Japan
Kyoto-shi, Kyoto, Japan
Nigata-shi, Nigata, Japan
Kurashiki-shi, Okayama, Japan
Osaka-shi, Osaka, Japan
Suita-shi, Osaka, Japan
Kita-adachi-gun, Saitama, Japan
Shinjuku-ku, Tokyo, Japan Collapse << |
| NCT01546649 | - | - | Completed | - | - |
| NCT00654927 | Multiple Sclerosis | Phase 3 | Completed | - | - |
| NCT00053417 | - | - | Completed | - | - |
| NCT00709371 | Obesity | Phase 2 | Completed | - | - |
| NCT01027949 | Pulmonary Arterial Hypertensio... more >>n Collapse << | Phase 3 | Active, not recruiting | December 2020 | - |
| NCT01104870 | - | - | Completed | - | - |
| NCT01235221 | Multiple Sclerosis | Phase 3 | Completed | - | Canada, Alberta
... more >>
Foothills Medical Center
Calgary, Alberta, Canada
Canada, British Columbia
University of British Columbia
Vancouver, British Columbia, Canada
Canada, New Brunswick
River Valley Health
Fredericton, New Brunswick, Canada
Canada, Nova Scotia
QEII Health Sciences Centre
Halifax, Nova Scotia, Canada
Canada, Ontario
Ottawa Hospital General Campus
Ottawa, Ontario, Canada Collapse << |
| NCT00654927 | - | - | Completed | - | - |
| NCT02393612 | Peripheral Arterial Disease | Phase 3 | Completed | - | - |
| NCT00053417 | Multiple Sclerosis | Phase 2 | Completed | - | - |
| NCT00526136 | Atrial Fibrillation | Phase 2 | Completed | - | - |
| NCT02547428 | Attention-Deficit Hyperactivit... more >>y Disorder Collapse << | Phase 2 | Completed | - | United States, California
... more >>
Newport Beach, California, United States
United States, Florida
Bradenton, Florida, United States
Maitland, Florida, United States
United States, Nevada
Las Vegas, Nevada, United States
United States, North Carolina
Durham, North Carolina, United States Collapse << |
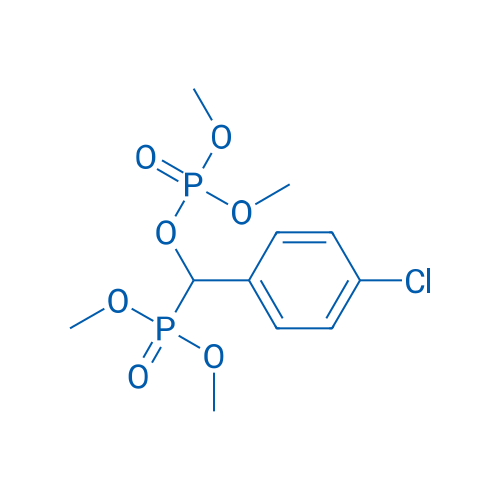

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
