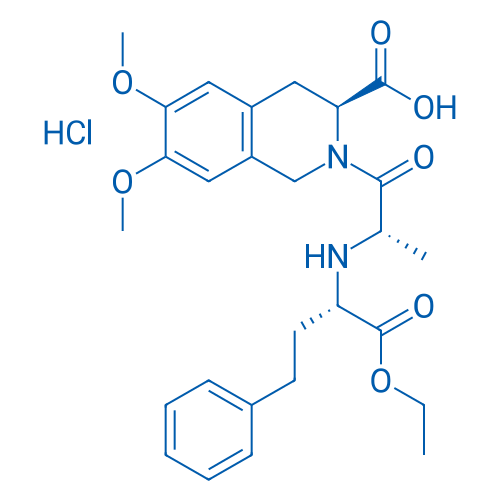
CAS No.: 82586-52-5
Moexipril HCl/盐酸莫西普利 Catalog No. CSN11458
Synonyms: RS-10085;Moexipril (hydrochloride);CI-925
Moexipril HCl is a potent orally active non-sulfhydryl angiotensin converting enzyme (ACE) inhibitor, which is used for the treatment of hypertension and congestive heart failure.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01867047 Adverse Effects of Angiotensin... more >>-converting-enzyme Inhibitors Hypotension Collapse << Not Applicable Terminated(Following an interi... more >>m analysis the study team felt that there was enough data that showed no significant difference.) Collapse << - United States, North Carolina ... more >> Duke University Durham, North Carolina, United States, 27710 Collapse << NCT01669434 - - Completed - - NCT01669434 Hypotension on Induction Phase 4 Completed - United States, Nebraska ... more >> University of Nebraska Medical Center Omaha, Nebraska, United States, 68198 Collapse << - 更多
- 参考文献
- [1] Ravati A, Junker V, et al. Enalapril and moexipril protect from free radical-induced neuronal damage in vitro and reduce ischemic brain injury in mice and rats. Eur J Pharmacol. 1999 May 28;373(1):21-33.
- [2] Ma YF, Stimpel M, et al. Impact of antihypertensive therapy on the skeleton: effects of moexipril and hydrochlorothiazide on osteopenia in spontaneously hypertensive ovariectomized rats. J Endocrinol. 1997 Sep;154(3):467-74.
- [3] LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012–. Moexipril. 2018 Feb 11
- [4] Chrysant SG, Chrysant GS. Pharmacological and clinical profile of moexipril: a concise review. J Clin Pharmacol. 2004 Aug;44(8):827-36
- [5] Edling O, Bao G, Feelisch M, Unger T, Gohlke P. Moexipril, a new angiotensin-converting enzyme (ACE) inhibitor: pharmacological characterization and comparison with enalapril. J Pharmacol Exp Ther. 1995 Nov;275(2):854-63
- [6] Koch B, Oparil S, Stimpel M. Co-administration of an ACE-inhibitor (moexipril) and hormonal replacement therapy in postmenopausal women. J Hum Hypertens. 1999 May;13(5):337-42
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 82586-52-5 | 储存条件 |
|
|
| 分子式 | C27H35ClN2O7 | 运输 | 蓝冰 | |
| 分子量 | 535.03 | 别名 | RS-10085;Moexipril (hydrochloride);CI-925;RS 10085-197;SPM-925;Moexipril. trade name Univasc |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
